Abstract
A survey was undertaken to evaluate juvenile animal studies conducted for drug applications reviewed by the Center for Drug Evaluation and Research between 2009 and 2014. Some conclusions about the nonclinical pediatric safety assessment based on studies performed in support of central nervous system–active compounds are presented here. A total of 44 completed studies from 32 New Drug Applications submitted to the Divisions of Psychiatry and Neurology Products were evaluated. Data on animal species and age range used, endpoints evaluated, and outcomes included in labeling were analyzed. Of the drugs evaluated, all but one had studies conducted in rats. In some cases, a second study in a nonrodent species (dog) was also conducted. Indices of growth and development and standard general toxicity parameters were included in all of the studies. Expanded neurohistopathology evaluations, bone mineral density measurements, and reproductive and neurobehavioral functional assessments were also generally carried out. A variety of neurological and neurobehavioral tests were employed. In the majority of rat studies, the potential for long-term cognitive impairment was evaluated using a complex water maze. Juvenile animal studies provided safety information considered relevant to drug use in children and that was included in labeling for 78% of the applications surveyed. The most commonly reported findings in labeling were for neurobehavioral effects, including changes in locomotor activity, auditory startle habituation, and learning and memory. Of the studies described in labeling with neurobehavioral effects, 54% found these effects to be persistent and to provide evidence of developmental neurotoxicity.
Introduction
The juvenile animal study (JAS) has become a key component of the nonclinical safety assessment of drugs being developed for use in children. 1,2 The need for these studies became apparent as the promulgation of regulations requiring that certain new and marketed drugs be studied in pediatric patients focused increased attention on practices for evaluating drug safety in this population. It was recognized that safety data from clinical studies in adults supported by nonclinical studies in adult animals provided an insufficient basis for predicting adverse drug effects in pediatric patients. This applied in particular to effects on developmental processes specific to pediatric age-groups, some of which (eg, developmental neurotoxicity [DNT]) would be very difficult to detect in clinical trials or during postmarketing surveillance. Furthermore, the standard battery of nonclinical reproductive and developmental toxicity studies does not adequately address postnatal development, since postnatal exposure is limited to that which occurs via the milk during lactation, the outcome can be confounded by in utero exposure, and some endpoints of interest may not be examined. A role for studies “involving direct treatment of offspring, at ages to be specified,” as noted in the ICH S5(R2) nonclinical guideline when it was first issued in 1993, emerged as a means to address this gap in the safety evaluation. 3,4 Such specialized studies have been requested by regulatory agencies with increasing frequency since the mid-1990s. Recommendations for nonclinical testing strategies that employ a JAS in order to better assess the potential for drug toxicity in pediatric patients have been the subject of numerous literature articles as well as regulatory guidance documents. 5 -9 A new ICH (S11) guideline intended to provide guidance on the nonclinical safety studies needed to support a pediatric drug development program is currently in the draft stage. 10
The United States Food and Drug Administration (FDA)/Center for Drug Evaluation and Research (CDER) guidance on nonclinical studies performed to detect potential adverse effects of drug use in pediatric populations describes a case-by-case approach that relies upon selection of an appropriate, scientifically justified study design. 7 A number of issues are addressed, notably the identification of appropriate animal models and outcome measures. In designing nonclinical toxicity studies to support pediatric drug safety, consideration of the role of developmental status in the pediatric patient’s response to a drug is paramount, since a distinguishing feature of the pediatric organism is its dynamic state. In order to adequately assess risk in the pediatric population, the potential for postnatal maturational processes to affect or be affected by drug actions must be taken into account. Thus, the selection of appropriate animal models that will allow evaluation of adverse drug effects during developmental stages comparable to those during which drug exposure will occur in humans is a key aspect of study design. 11 Although the general term juvenile has been applied to these studies, dosing of animals may occur during the developmental equivalents of the human neonatal, infancy, childhood, and/or adolescent periods; and observations are made in adults to assess delayed or long-term effects.
Another critical aspect of JAS design involves the selection of outcome measures that will adequately reveal adverse effects resulting from exposure during the relevant developmental periods. Given the developmental focus of the JAS, toxicity endpoints that reflect changes in organ systems undergoing significant postnatal development have received the most attention. The JAS can provide data on the potential for effects that have longer term impact and cannot easily be evaluated in clinical trials or during postmarketing surveillance. According to the FDA/CDER guidance, the toxicological endpoints to be monitored in a JAS should include assessments of reproductive and neurobehavioral function in addition to standard general toxicity parameters. For DNT evaluations, well-established methods should be used to monitor important central nervous system (CNS) functions, including assessments of reflex ontogeny, sensorimotor function, locomotor activity, reactivity, and learning and memory. 12 Experience gained with juvenile animal studies may make it possible to draw some valuable conclusions about which endpoints and methods are best suited for identifying postnatal developmental toxicity or other age-related risk. With this in mind, we have surveyed the juvenile animal studies conducted for drug applications reviewed in the Divisions of Psychiatry and Neurology Products between 2009 and 2014. This study was presented in part at the Society of Toxicologic Pathology 36th Annual meeting; June 24-29, Montreal, Quebec, Canada (https://www.toxpath.org/abstracts.asp; 2017 Poster Abstracts, P56).
Methods
To understand the value and regulatory impact of juvenile animal studies in pediatric drug development, the FDA/CDER/OND Nonclinical Safety Testing for Pediatric Drugs Subcommittee retrospectively examined proprietary JAS data submitted to CDER under various Investigational New Drug (IND) applications and/or New Drug Applications (NDAs) between 2009 and 2014. The IND and NDA applications with juvenile animal studies were identified from the CDER Document Archiving, Reporting, and Regulatory Tracking System using the “communication content search” option and content search criterion set as “juvenile.” The identified juvenile animal studies were indexed and further analyzed in detail by members of the Nonclinical Safety Testing for Pediatric Drugs Subcommittee and the information/data obtained was collated and archived in a database. Preliminary results of the analysis of juvenile animal studies submitted to CDER in support of pediatric indications were presented in part at the Society of Toxicology 55th Annual Meeting, March 13-17, 2016; New Orleans, LA (https://www.toxicology.org/pubs/amp/amPubs.asp. The Toxicologist Late-Breaking Supplement; P378. Accessed October 5, 2018).
Using the above database, a total of 120 applications were identified for products supporting CNS-active compounds from the Division of Psychiatry Products (DPP) and Division of Neurology Products (DNP). Of these, 32 applications that reached the NDA phase of drug development were selected for further evaluation and 44 completed studies conducted for these applications were examined. The original study reports and Agency reviews were used to collect information regarding animal species and age, endpoints assessed (including bone and reproductive parameters, neurohistopathology examinations, and type of learning and memory task used), and findings described in the labeling.
Results
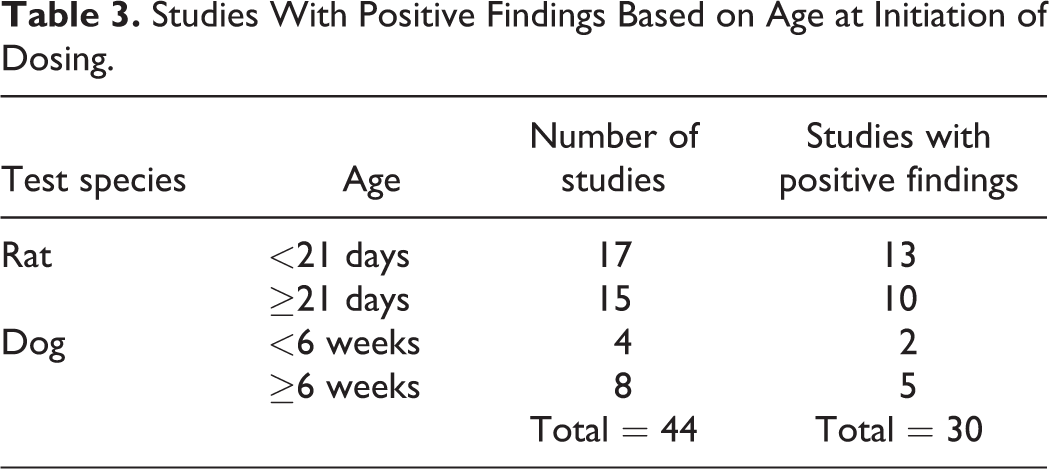
The majority of the applications were for drugs indicated for use in the treatment of epilepsy and attention-deficit hyperactivity disorder (ADHD), which are typically pediatric in onset, although adults are also affected. Other indications included psychiatric (depression, schizophrenia) and sleep disorders that occur primarily in older children and adults (Table 1). The indicated pediatric use was generally reflected in the age at initiation of dosing in the JAS. Of the total number of applications evaluated (32), all but 1 had a JAS conducted in rats (1 application had 2 rat studies), 1 had only a dog study, and 11 had studies in 2 species, rat and dog (Table 2). The age of the animals at initiation of dosing in these studies ranged from postnatal day (PND) 4 to PND 28 in rats and from PND 4 to 4 months in dogs (Table 3). Dosing continued through sexual maturation in all rat studies (duration of dosing 6-13 weeks) and in all but 1 dog study (duration of dosing 4-10 months).
Number of Applications by Indication.
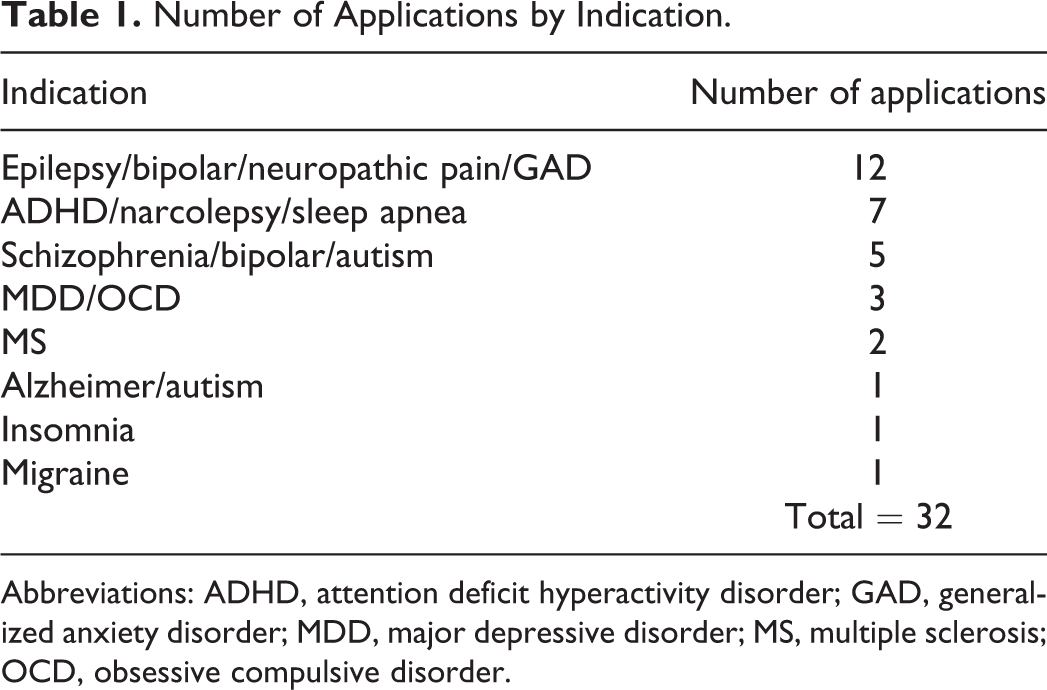
Abbreviations: ADHD, attention deficit hyperactivity disorder; GAD, generalized anxiety disorder; MDD, major depressive disorder; MS, multiple sclerosis; OCD, obsessive compulsive disorder.
Number of Applications by Juvenile Animal Species Used.

Studies With Positive Findings Based on Age at Initiation of Dosing.
Juvenile animal study findings were described in the labeling (eg, under Section 8: Use in Specific Populations, Subsection 8.4: Pediatric Use) for most (78%) of the applications. Table 4 shows selected study endpoints that detected what were considered important juvenile animal toxicity signals in the studies surveyed and which were described in labeling. For example, of the total number of studies that evaluated neurohistopathology and reproductive system structure or function (44), 7% and 16% showed an effect that was included in the labeling, respectively. Of the total studies that conducted neurological or neurobehavioral functional evaluations (43), 53% had findings for these endpoints described in the label. It should be recognized that endpoint categories may be interrelated, and alternative classifications are possible. This is particularly true for endpoints reflecting effects on growth, when the specificity of an effect is unclear. For example, sexual maturation is included under developmental landmarks rather than the reproductive system endpoints category, while bone length is combined with bone mineral density in a bone development category. And there may be overlap among endpoints, such as between evidence of neurotoxicity detected through use of clinical observations, neurohistopathology, and various neurobehavioral testing procedures. Multiple endpoints were generally affected in individual studies in which juvenile animal toxicity was observed.
Toxicity Signals Described in the Label for Selected Endpoints.
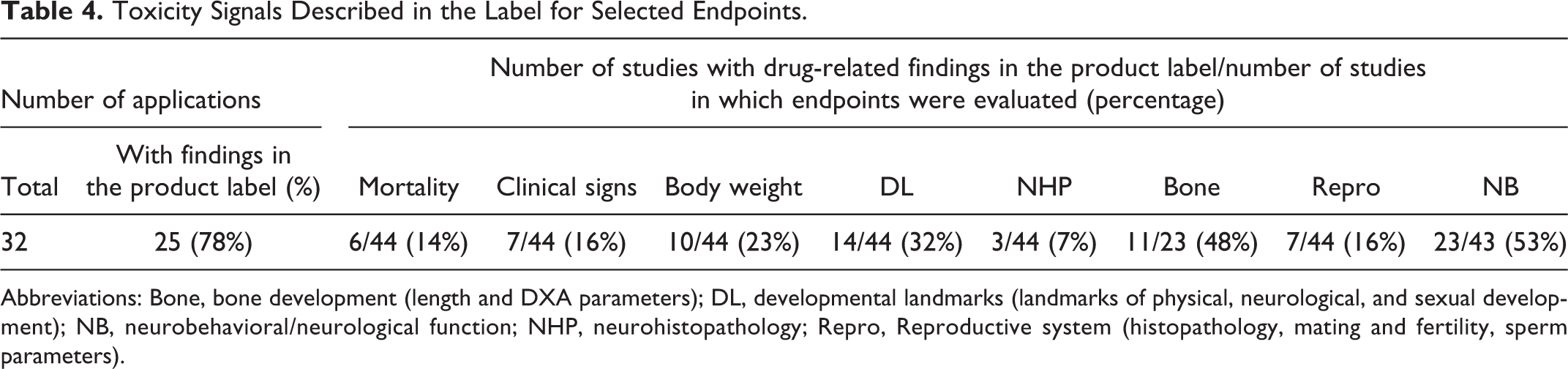
Abbreviations: Bone, bone development (length and DXA parameters); DL, developmental landmarks (landmarks of physical, neurological, and sexual development); NB, neurobehavioral/neurological function; NHP, neurohistopathology; Repro, Reproductive system (histopathology, mating and fertility, sperm parameters).
All studies in both rats and dogs examined the standard general toxicity parameters of mortality, body weight, clinical signs, clinical pathology, organ weights, and gross and microscopic pathology. Toxicity findings for these endpoints were generally described in labeling because juvenile animals appeared to be either uniquely susceptible or more sensitive compared to adults. Mortality was considered an age-related outcome and reported in the label in 6 of the 44 studies. Body weight reductions considered evidence of developmental toxicity, generally due to the persistence of deficits after cessation of dosing, were seen in 10 of 44 studies (8/32 rat, 2/12 dog). Clinical observations that were considered important juvenile toxicity signals were characterized as evidence of acute or long-term neurotoxicity and included convulsions observed during dosing and spontaneous convulsions observed in animals after the nondosing recovery period. These were described in labeling for 7 of the studies (2 rat and 5 dog). Clinical chemistry findings indicating unique muscle toxicity and increased sensitivity to urinary tract and hepatic toxicity compared to adults were observed in 3 of 44 studies (2 rat and 1 dog). A selective reduction in brain weight was found in 3 rat studies. Anatomical pathology examinations revealed drug-related toxicity in 11 of the 44 studies (9 rat, 2 dog). Affected organ systems included muscle, urinary tract, liver, male and female reproductive organs, and brain. Because of the considerable and critical postnatal development that takes place in the reproductive system and CNS, possible effects of drug exposure on these organ systems have been more thoroughly evaluated in juvenile animal studies, as described below.
Growth endpoints included, in addition to standard body weight measurements, landmarks of physical, neurological, and sexual development. Although these endpoints have been shown to be highly correlated with body weight, effects were more likely to be included in labeling if they appeared to be selective. Developmental delays indicating age-related toxicity were described in labeling in 32% (12/32 rat, 2/12 dog) of the studies examined.
More than half of the studies (25) included an expanded neurohistopathology evaluation based on recommendations by Bolon et al, 13,14 while the remainder (19) used what was considered a routine CNS histopathology examination. Of the studies that performed routine evaluations, none detected drug-related lesions. In studies that conducted expanded evaluations, 3 (2 rat studies and 1 dog study) reported CNS histopathology (Figure 1). Protocols for these 3 studies also included multiple discrete dosing periods and sacrifice times. Of note, both rat studies in which neurohistopathological abnormalities were observed also detected effects on neurobehavioral endpoints, while CNS function was not assessed in the dog study.
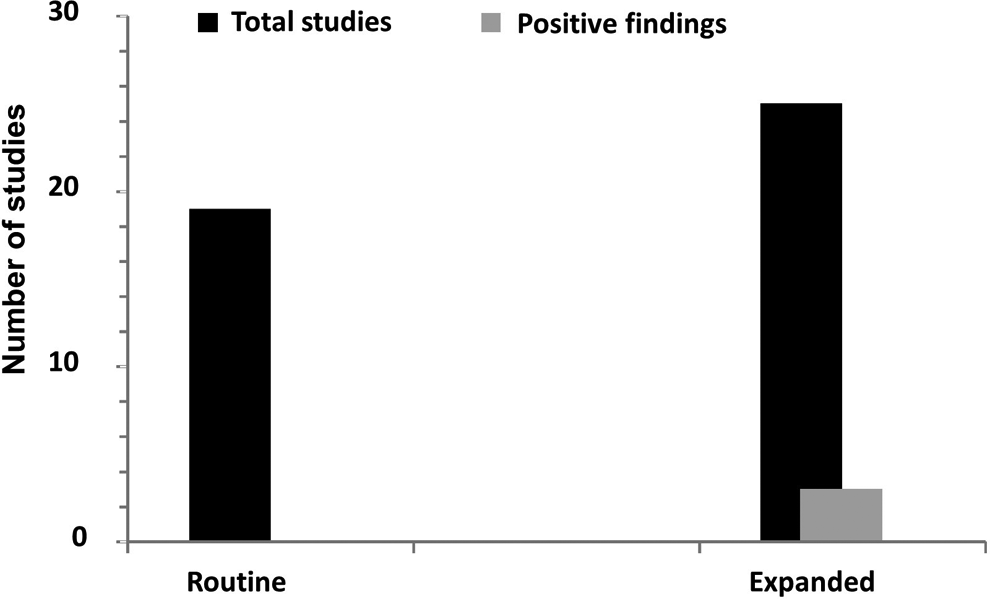
Number of studies that evaluated brain histopathology (black) and studies with positive findings (grey). Routine examination represents the evaluation of 3 brain sections and expanded examination represents the evaluation of at least 7 brain sections.
Because drug exposure occurs during a dynamic period of bone formation and growth in pediatric patients, bone development has also been a specific focus of JASs. There were 23 studies with specific bone development endpoints (other than histopathology): 14 studies in which both bone length and densitometry (dual-energy X-ray absorptiometry [DXA]) parameters were evaluated and 9 studies that included only bone length (Table 5). Of the 6 dog studies that included bone evaluations, all measured both length and density. Of the 17 rat studies, 8 evaluated both length and density, while 9 examined length only. In these combined studies, 11 (48%) reported positive findings for bone length (3), bone density (4), or both (4). Of the 8 studies that showed an effect on bone density, length was also affected in 4 (1 dog and 3 rat), while a selective effect on densitometry parameters was seen in 4 (2 dog and 2 rat).
Bone Densitometry and/or Length Measurements in Juvenile Rat and Dog Studies.
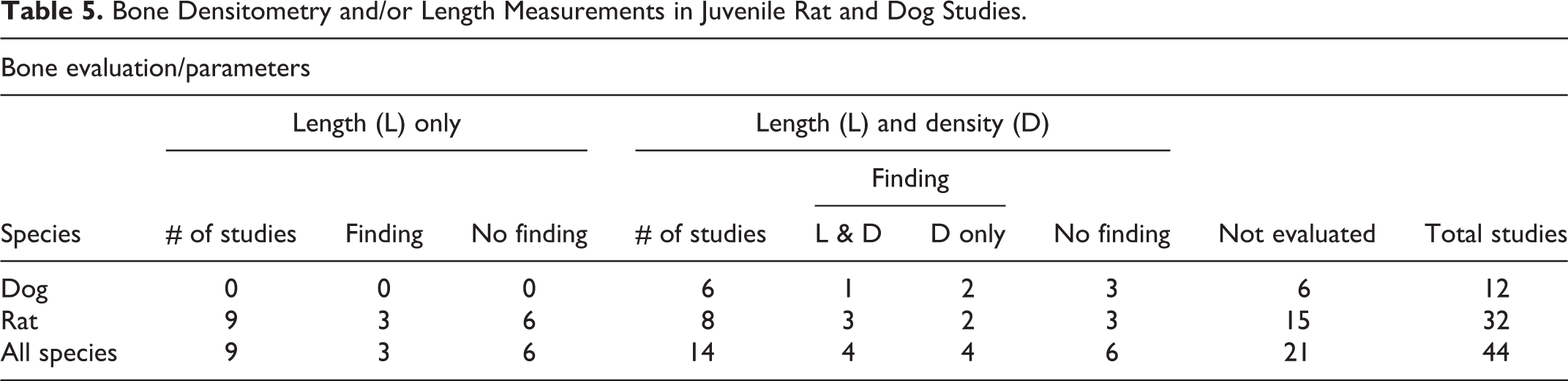
Mating and pregnancy outcome measures were evaluated in all rat studies (32), reproductive organ histopathology evaluation was conducted in all studies in both rats and dogs (44), and sperm analysis was conducted in 16 studies from both rats and dogs. A total of 7 studies (5 rat, 2 dog) conducted for 6 applications showed effects on one or more reproductive system endpoints (other than landmarks of sexual maturation which were included under growth, above): 2 of 32 studies showed an effect on mating and pregnancy outcomes, 5 of 44 studies showed an effect on reproductive organ histopathology (3 rat and 2 dog), and 3 of 16 (2 rat and 1 dog) showed an effect on sperm parameters (Figure 2). Two rat studies exhibited effects on more than 1 reproductive endpoint (testicular histopathology and sperm parameters, testicular histopathology and fertility), and effects on both testicular histopathology and sperm parameters were seen in a single dog study. Testicular histopathology and sperm parameter effects were produced in both the rat and the dog study conducted for 1 drug application. No adult animal reproductive toxicity was seen for 2 of the 6 drugs that produced reproductive effects in juvenile animal studies.
Number of juvenile rat and dog studies that evaluated reproductive system effects (mating/pregnancy outcome, histopathological evaluation, sperm parameters). Black columns represent the total number of studies that evaluated one or more of these endpoints, and grey columns represent the number of studies with positive findings.
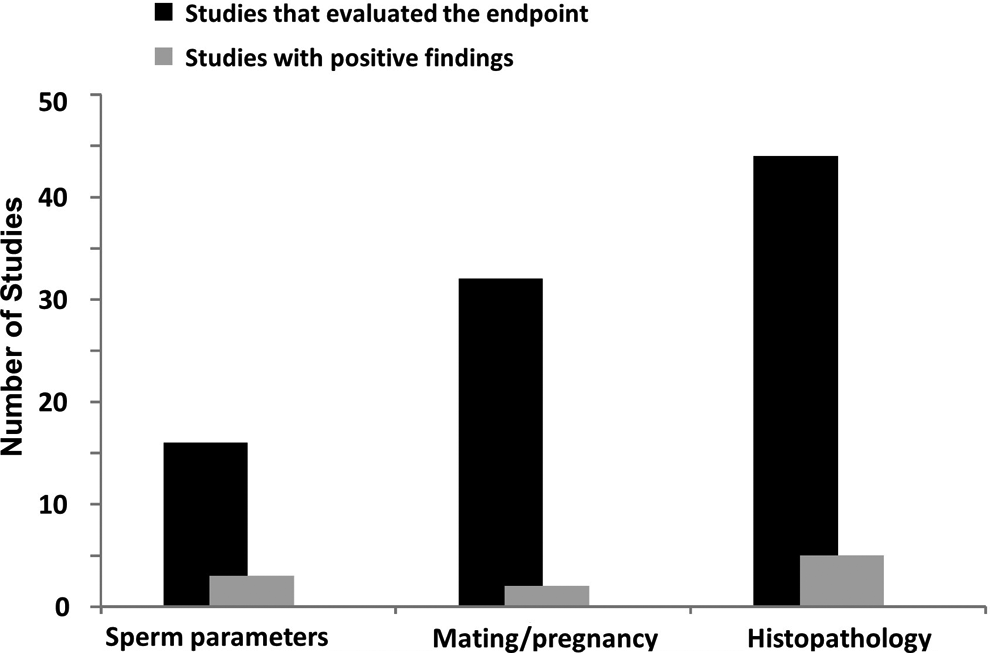
Neurobehavioral/CNS functional evaluations performed in these studies differed by species, consisting primarily of a neurological examination or functional observational battery (FOB) in dogs and more specific and quantitative evaluations, such as measurements of locomotor activity, auditory startle habituation, and learning and memory in rats. Effects on one or more of these endpoints were considered evidence of neurotoxicity in 53% (23/43) of the total number of studies in which CNS function was evaluated and in 69% (22/32) of the rat studies. All 32 rat studies performed one or more tests of learning and memory and most measured locomotor activity and auditory startle habituation using automated systems; drug-related effects on these measures were described in labeling for 47% (15/32), 32% (10/31), and 31% (9/29) of the studies in which they were performed, respectively (Figure 3). A signal of toxicity observed in the less frequently conducted rat FOB was included in labeling only once (1/12). All but one of the dog studies performed a neurological examination and/or a canine FOB; these detected evidence of neurotoxicity that had not been noted in clinical observations in 1 of 11 studies. Of the studies described in labeling with neurobehavioral effects, 54% found effects that persisted after discontinuation of drug administration, indicating long-term changes in CNS function.
Neurobehavioral tests conducted in juvenile rat studies (black) and number with positive findings (grey). AS indicates acoustic startle; FOB, functional observational battery; LM, learning and memory; MA, motor activity.
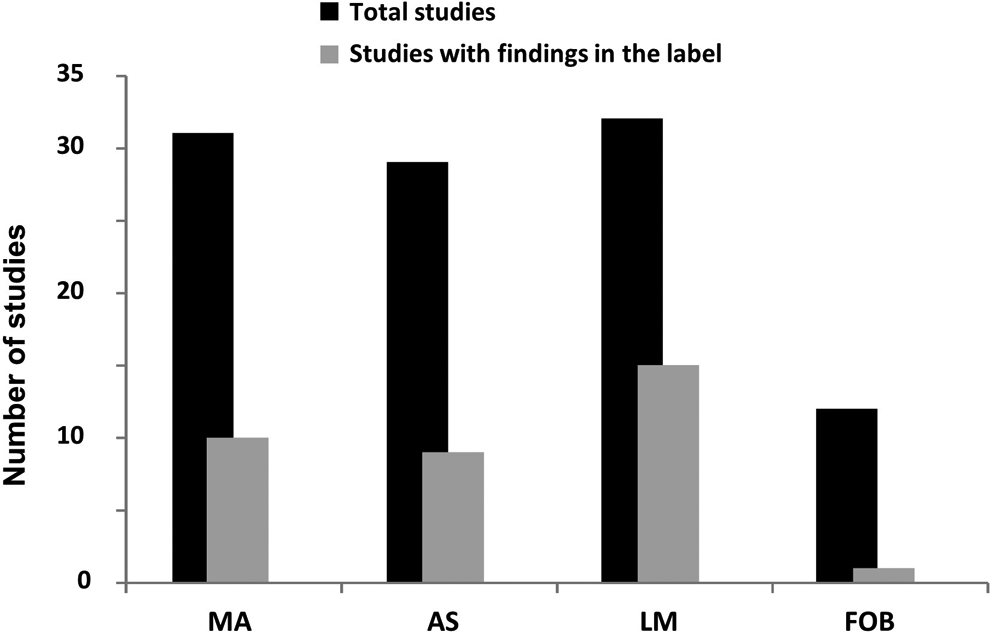
Different learning and memory tests, ranging in complexity and difficulty from simple 2-choice discrimination tasks and passive avoidance to multiple-T or Morris water mazes, were found in the submissions that included rat studies (Figure 4). The most frequently used learning and memory test was the Morris water maze (11), followed by the Biel (8) and Cincinnati water mazes (8). Positive findings, defined as some form of deficit, were reported in 64% of the studies in which the Morris water maze was used and in 63% of the studies in which the Biel maze was used. Fewer studies using the Cincinnati water maze indicated adverse effects, but in the majority of these studies insufficient numbers of animals per group (<15/sex) were used. The simple mazes had the lowest rate of positive findings (17%). Of the 2 studies that utilized the passive avoidance test, one was reported with positive findings.
Learning and memory tests used in juvenile rat studies (black) and number of studies with positive outcome (grey). Positive outcome is defined as an effect on any aspect of the test to indicate an adverse effect such as an increase in the number of errors or increase in the latency to reach the target. Note: more than one test was conducted in some studies.
Discussion
Recognition that product labels lacked information on the safety and efficacy of drug use in children provided much of the impetus for pediatric drug legislation. 15 The majority of juvenile animal studies conducted to support the safety of drugs for pediatric indications in DNP and DPP had a regulatory impact as reflected in labeling. Among the various parameters assessed in juvenile animal studies submitted to these divisions, toxicity signals considered to represent effects relevant to the safety of pediatric drug use were seen for endpoint categories that included mortality, neurohistopathology, growth and development (including reflex ontogeny and sexual maturation), bone composition, and neurobehavioral and reproductive function. Toxicity signals occurring at clinically relevant drug exposures were most frequently detected for growth and neurobehavioral endpoints. Clinical correlates have been observed for effects on growth, bone composition, and neurohistopathology seen in juvenile animal studies.
Neurobehavioral endpoints that have proven sensitive in detecting effects on juvenile CNS development included measures of locomotor activity, auditory startle habituation, and learning and memory. Effects on cognitive function, considered the most clinically meaningful neurobehavioral measure, 16 were most often detected using complex learning and memory tests such as the Morris, Biel, or Cincinnati water mazes. Both transient and long-term neurobehavioral changes could be observed by testing during and after cessation of dosing. Neurobehavioral effects were seen most often in studies in which dosing began in the preweaning period. For those studies with CNS histopathology findings, which were relatively few, expanded neurohistopathology techniques were used to detect and more fully characterize the effects.
Effects observed in developing animals can be compared with those seen in mature animals to determine whether they represent toxicities to which pediatric patients may be uniquely or more susceptible. However, some endpoints (eg, developmental milestones) would not be applicable to adults, and other effects, such as on neurobehavioral and reproductive endpoints, have been shown to be highly age-specific and subject to developmental critical periods. 17 -20 The developing nervous and reproductive systems have target processes not seen adults, so extrapolations across age may not be valid for these endpoints. 11,21 -23 For example, reproductive toxicity observed in the current survey of juvenile toxicity studies, manifested as changes in mating and fertility, sperm parameters, or reproductive organ histopathology, was not always seen in adults or did not always correspond to the adult effects in terms of type, severity, or reversibility. Childhood and adolescence are also critical periods for bone mineralization; and various insults, including drug exposures, occurring during these periods have been associated with long-lasting adverse effects on bone health. 24 Such juvenile to adult comparisons should be specific for type of effect, rather than general target organ, and include analysis of exposure and metabolism data.
Growth and neurobehavioral endpoints have previously been shown to be sensitive indices of developmental toxicity; for example, in a survey of 69 EPA DNT studies of chemicals, the most common findings at the lowest observed adverse effect level (LOAEL) were changes in body weight, brain morphometrics, motor activity, brain weight, and auditory startle. 25 Although learning and memory measures were less frequent neurobehavioral endpoints identified as LOAELs in the studies reviewed by EPA, it has been pointed out that this was likely due to the choice of testing paradigms, for which the EPA DNT guideline allows considerable latitude. 26
The chronic pediatric indications for which the majority of juvenile animal studies were conducted were epilepsy and ADHD. Although the preponderance of neurobehavioral findings in these studies may reflect the intended mechanism of action of drugs submitted to these divisions, there is no reason to assume that such effects would be limited to drugs with CNS indications. Evaluation of the toxic potential of a pharmaceutical cannot be based solely on its therapeutic target; any drug that reaches the CNS should be considered potentially neurotoxic. 27 Neurotoxicity can also occur as the result of indirect effects, such as through endocrine disruption. 28
Appropriate neurobehavioral endpoints appear well suited for the detection of DNT in juvenile animal studies, and our findings generally support recent recommendations regarding regulatory neurobehavioral testing. 12,16,26 The innate sensitivity and prolonged postnatal development of the CNS make it particularly susceptible to environmental influences during childhood, so a thorough evaluaton of possible long-term effects of drug exposure on brain development is essential for an adequate pediatric safety assessment. 29 And, since many potent neurotoxic agents produce no detectable morphological changes, functional measures may provide the only evidence of DNT. 30 As concluded in a previous survey, neurobehavioral assessments make a valuable and unique contribution to the detection and characterization of toxicity resulting from exposure during vulnerable periods of CNS development. 31 Furthermore, the observation of alterations or deficits in well-validated neurobehavioral tests such as auditory startle or the Morris water maze implies clinical significance. 32,33
Notable in our results is the high proportion of studies in which impairment of cognitive function was observed. This was primarily seen in rat studies that assessed navigational learning and memory using either labyrinthine (Cincinnati and Biel) or open-field (Morris) water mazes, tests that have been recommended for regulatory use based on characteristics such as sensitivity, construct/theoretical validity, practicality, and interpretational/predictive value. 12,33 Cognitive impairment is among the most common neurotoxic effects associated with developmental exposure to environmental agents and drugs in epidemiological studies. 34 However, the clinical detection of cognitive deficits resulting from developmental exposures is fraught with difficulties; for example, clinical evaluation of the developmental effects of pediatric drug exposure may be confounded by an underlying neurological disorder. 35 Therefore, the inclusion of sensitive measures of cognitive impairment in nonclinical DNT assessments is considered critically important for providing information regarding potential human risk for functions that are central to quality of life. 16,26,34
Although neurobehavioral impairment occurred in both juvenile rat studies in which neurohistopathology was seen (no functional assessments were performed in the dog study), in most cases neurobehavioral changes were not accompanied by neurohistopathological observations, even when expanded examinations were performed. In the EPA DNT survey, qualitative neuropathology was never the basis for an LOAEL, suggesting that functional measures were more sensitive, although the question was raised whether enhancements of neuropathology methods could improve the sensitivity of these evaluations. 36 The conduct of neurohistopathology examinations at a single time point at the end of the dosing period, as is the usual practice, is not optimal or sufficient for detecting effects that may occur at any time over an extended dosing period. Given the importance of sacrifice time for adequate neuropathology evaluations, more frequent assessments during the course of treatment in a JAS may be needed. 37 Clearly, however, neurobehavioral and neurohistopathological evaluations are complementary and both contribute to an adequate assessment and characterization of possible DNT.
Footnotes
Authors’ Note
This article reflects the views of the authors and should not be construed to represent FDA’s views or policies.
Acknowledgments
The authors would like to acknowledge the contributions of the FDA/CDER/PTCC Nonclinical Safety Testing for Pediatric Drugs Subcommittee to data collection and compilation.
Author Contributions
All authors (J.E.F., A.R., and I.E.) made equal contributions to conception and design; data acquisition, analysis, and interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
