Abstract
The immune system of the rat undergoes substantial functional and morphological development during the postnatal period. Some aspects of this development are genetically predetermined, while other aspects depend on environmental influences. Detailed information on postnatal development is important in the interpretation of histopathologic findings in juvenile toxicology and pubertal assay studies, as well as other studies conducted in juvenile rats. Studies were conducted to provide detailed characterization of histologic features of the major functional compartments of immune system organs in male and female Sprague-Dawley rats at weekly intervals from the day of birth through postnatal day (PND) 42. Maturation of the individual immune system organs occurred across a range of ages, with histologic maturation of T-cell-related compartments typically occurring prior to maturation of B-cell-related compartments. The sequence of histologic maturation was bone marrow and thymus on PND 14, mesenteric lymph node on PND 21, Peyer’s patches and bronchus-associated lymphoid tissue on PND 28, mandibular lymph node, nasopharynx-associated lymphoid tissue, and diffuse mucosal mononuclear cell population of small intestine on PND 35, and spleen on PND 42. An estimation of functional maturation can be made based on the morphological indications of maturity of each compartment of immune system organs, but histologic indications of maturity do not confirm functional immunocompetence.
Keywords
Introduction
Knowledge of the histogenesis of immune system organs of the rat is of particular importance in the interpretation of histopathological observations in juvenile toxicology and developmental immunotoxicology (DIT) studies, but could be of importance in other studies conducted in animals of an age where the immune system organs are not fully developed.
Interest in juvenile toxicology is based on recognition that responses to xenobiotics of juvenile humans and animals may be quite different from responses of adults (Burns-Naas et al. 2008; Holsapple 2003). In addition, xenobiotic-induced cellular or molecular modifications during gestational, neonatal, or juvenile periods may alter responses to chemical influences later in life (Chang and Bellward 1996; Kikkawa, Fujita, and Sindhu 1994; Singh et al. 2013). Interpretation of histologic changes in juvenile toxicology studies is particularly complex. In addition to the direct, indirect, nutrition-, and stress-related histologic alterations that are encountered in non-juvenile toxicology studies and in juvenile toxicology studies, the pathologist must be concerned with xenobiotic-associated alterations in postnatal organ development. As an additional complicating factor, postnatal development of the immune system is partially driven by environmental factors, as opposed to the genetically determined histologic development of most other organ systems. Challenges in histopathologic interpretation in juvenile toxicology studies are most apparent when dealing with histologic observations in unscheduled death animals, where age- and sex-matched concurrent control animals typically are not available for comparison.
Concern with regard to developmental immunotoxicity (DIT) is based on the premise that exposure to xenobiotics early in life may result in alterations in immune responsiveness that are not manifested until later in life (for review, see Burns-Naas et al. 2008). Effects on the developing immune system have been documented for a number of chemicals (Bunn et al. 2001b; Gehrs and Smialowicz 1999; Miller et al. 1998; Mustafa et al. 2009; Weinstein et al. 2008). This concern resulted in a symposium (Holsapple 2002) with participants from government, academia, and industry that considered many aspects of DIT. Among other conclusions, symposium attendees decided (1) DIT studies would be most effectively conducted in rats rather than mice, due to the large toxicology-related database in rats; (2) DIT investigations could probably be added to existing developmental and reproductive toxicology (DART) protocols such as 2-generation studies; and (3) histopathologic evaluation of tissue specimens would probably serve as one end point for DIT studies. It was acknowledged that gaps exist in the information available on immune system development in rats. A desire to fill some of the histology information gaps was a major incentive for conducting the present studies.
Published information on histologic features of the full extent of the immune system organs is generally sparse, though there is an excellent review of morphology and immunohistochemical (IHC) staining properties of thymus and spleen of rats at 1, 2, 3, 4, 9, 19, 23, 46, and 57 weeks of age (Kodama et al. 2012). Selected observations from our preliminary study on the histogenesis of immune system organs in the rat were included in other publications (Burns-Naas et al. 2008) and was the subject of a satellite symposium of the 2009 annual meeting of the American College of Veterinary Pathologists (Weinstock et al. 2010). These prior publications contain late gestational and widely spaced postnatal time points but do not have a systematic evaluation of all major immune system organs at closely spaced intervals. The present report addresses the histologic features of an extended selection of immune system organs at weekly intervals, spanning the ages typically encountered in juvenile toxicology studies or ages that would be anticipated in animals collected at termination of multigenerational DART studies.
Materials and Methods
For the primary study, juvenile Sprague-Dawley Crl:CD rats were necropsied at postnatal day (PND) 0 (day of birth), 7, 14, 21, 28, 35, and 42. Thirty males and 30 females were examined on PND 0, and 10 rats/sex/PND interval were examined at PND 7, 14, 21, 28, 35, and 42. Multiple litters were represented in each PND interval group, as shown in Table 1. The rat pups for this study were collected during a single time period that extended approximately 8 weeks. All animals were housed in stock animal rooms at WIL Research in Ashland, Ohio.
Summary of compartment maturation in immune system organs of the juvenile rat based on histomorphology.
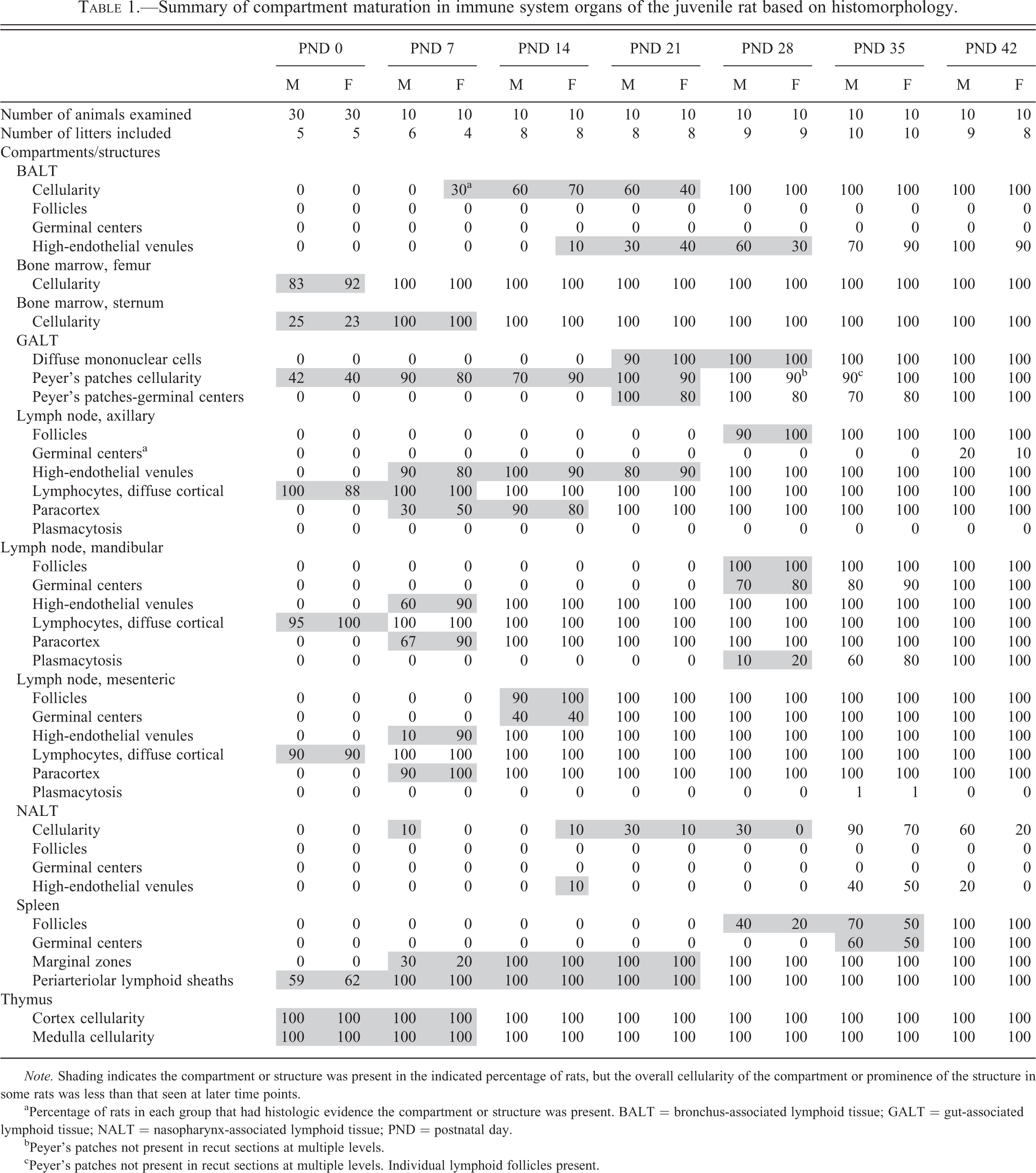
Note. Shading indicates the compartment or structure was present in the indicated percentage of rats, but the overall cellularity of the compartment or prominence of the structure in some rats was less than that seen at later time points.
aPercentage of rats in each group that had histologic evidence the compartment or structure was present. BALT = bronchus-associated lymphoid tissue; GALT = gut-associated lymphoid tissue; NALT = nasopharynx-associated lymphoid tissue; PND = postnatal day.
bPeyer’s patches not present in recut sections at multiple levels.
cPeyer’s patches not present in recut sections at multiple levels. Individual lymphoid follicles present.
In addition, this report includes observations in liver specimens of the preliminary study, which were collected at gestation day (GD) 15 and GD 20 as well as PND 0, 1, 2, 3, 10, 21, and 42. These animals were also housed in stock animal rooms at WIL Research in Ashland, Ohio, and tissue specimens were collected during a different time period.
Necropsy and General Histology
Juvenile rats were euthanized via intraperitoneal injection of sodium pentobarbital (PND 0, 7, and 14) or carbon dioxide asphyxiation (PND 21 through PND 42), and the following tissues were collected and fixed in neutral buffered formalin: thymus, spleen, axillary lymph node, mandibular lymph node, mesenteric lymph node, small intestine, lung, sternum, femur, and posterior aspect of nasal cavity. The small intestine was processed in toto, using the “Swiss roll” technique (Moolenbeek and Ruitenberg 1981). Tissue specimens were processed by routine histology techniques for paraffin embedding, sectioned at approximately 5 μm, and stained with hematoxylin and eosin (H&E). In order to demonstrate additional histologic features necessary for immune system characterization, selected sections were subjected to IHC staining for CD3 (T cells), immunohistochemistry marker for B cells (CD45RA; B cells), or Ki67 (proliferation marker to aid in identification of germinal centers), using routine staining procedures with diaminobenzidine as the chromagen.
The microscopic examination of each tissue was based on the principles outlined for enhanced histopathology of immune system organs (Elmore 2006a, 2006b, 2006c, 2006d, 2006e, 2012). The following histologic features were subjectively scored for each organ: Bone marrow (sternum and femur)—overall cellularity; Thymus—cortical cellularity and medullary cellularity; Axillary, mandibular, and mesenteric lymph nodes—cortical lymphocyte population, follicles, germinal centers within follicles, paracortex, high-endothelial venules (HEVs), and plasmacytosis; Small intestine—Peyer’s patches (PPs), germinal centers within PPs, and diffuse mucosal mononuclear cell population. The diffuse mucosal lymphocyte population included lymphocytes within the superficial epithelium as well as lymphocytes within the lamina propria of villi; Lung—presence of bronchus-associated lymphoid tissue (BALT), follicles, germinal centers, and HEVs; Nasopharynx—presence of nasopharynx-associated lymphoid tissue (NALT), follicles, germinal centers, and HEVs; and Spleen—follicles, germinal centers within follicles, periarteriolar lymphoid sheaths (PALS), and marginal zones (MZs).
Animals used in these studies were maintained in an Association for Assessment and Accreditation of Laboratory Animal Care–accredited animal facility under appropriate animal welfare guidelines (U.S. Department of Agriculture’s Animal Welfare Act [9 CFR Parts 1, 2, and 3]) and the Guide for the Care and Use of Laboratory Animals, Eighth Edition (Institute for Laboratory Animal Research 2010). The study protocols were reviewed and approved by the facility’s Institutional Animal Care and Use Committee (IACUC), and all procedures complied with facility standard operating procedures.
Results
A summary of postnatal development of the various immune system organ compartments is presented in Table 1, which presents the percentage of rats that exhibited the individual compartment or structure at the various PND intervals. Highlighting of data entries indicates compartments or structures were less developed histologically than the comparable compartments or structures at later time points. A narrative summary of observations in individual tissues follows.
Thymus
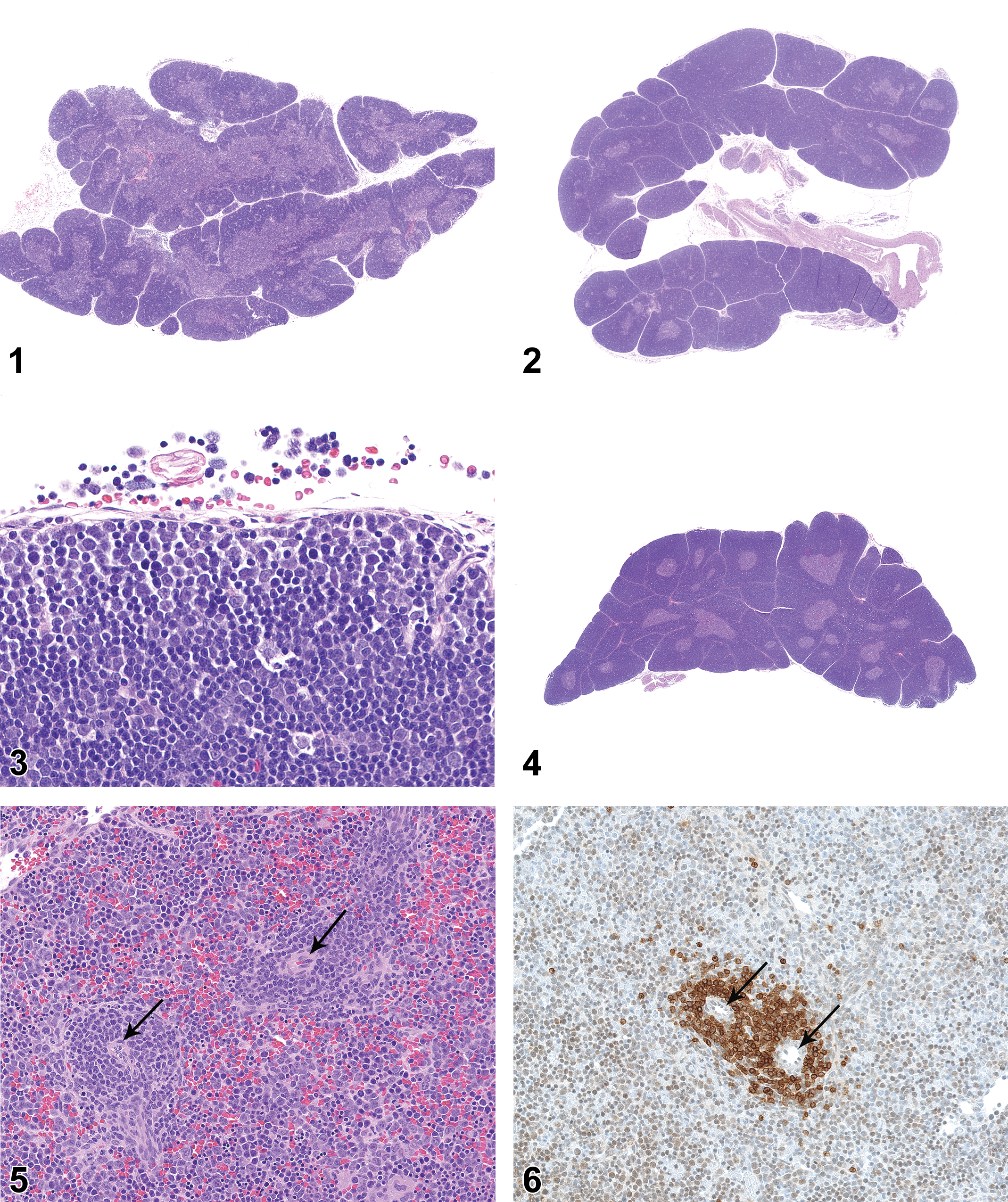
On PND 0 (day of birth), the bilobed thymus was moderately developed and had obvious corticomedullary distinction, though the cortex was not as thick (lower corticomedullary ratio) and had somewhat lower cellular population density than the cortex of young adult rats (Figure 1). By PND 7, the cortical cellularity approached that of the adult organ (Figure 2), and the rate of apoptosis was equivalent to that typically seen in young adult rats. The superficial cortex at PND 7 commonly contained a population of large mononuclear cells (Figure 3) that were microscopically distinctive from the small, densely stained mature lymphocytes that characterize the thymic cortex. From PND 14 onward, the microscopic features of the thymus were typical of the adult organ (Figure 4), with the only difference being a progressive increase in overall size of the organ.
Thymus of newborn rat (postnatal day 0). Corticomedullary definition is apparent, but cortex is thinner than fully developed thymus. Hematoxylin and eosin, original objective 2.5×.
Spleen
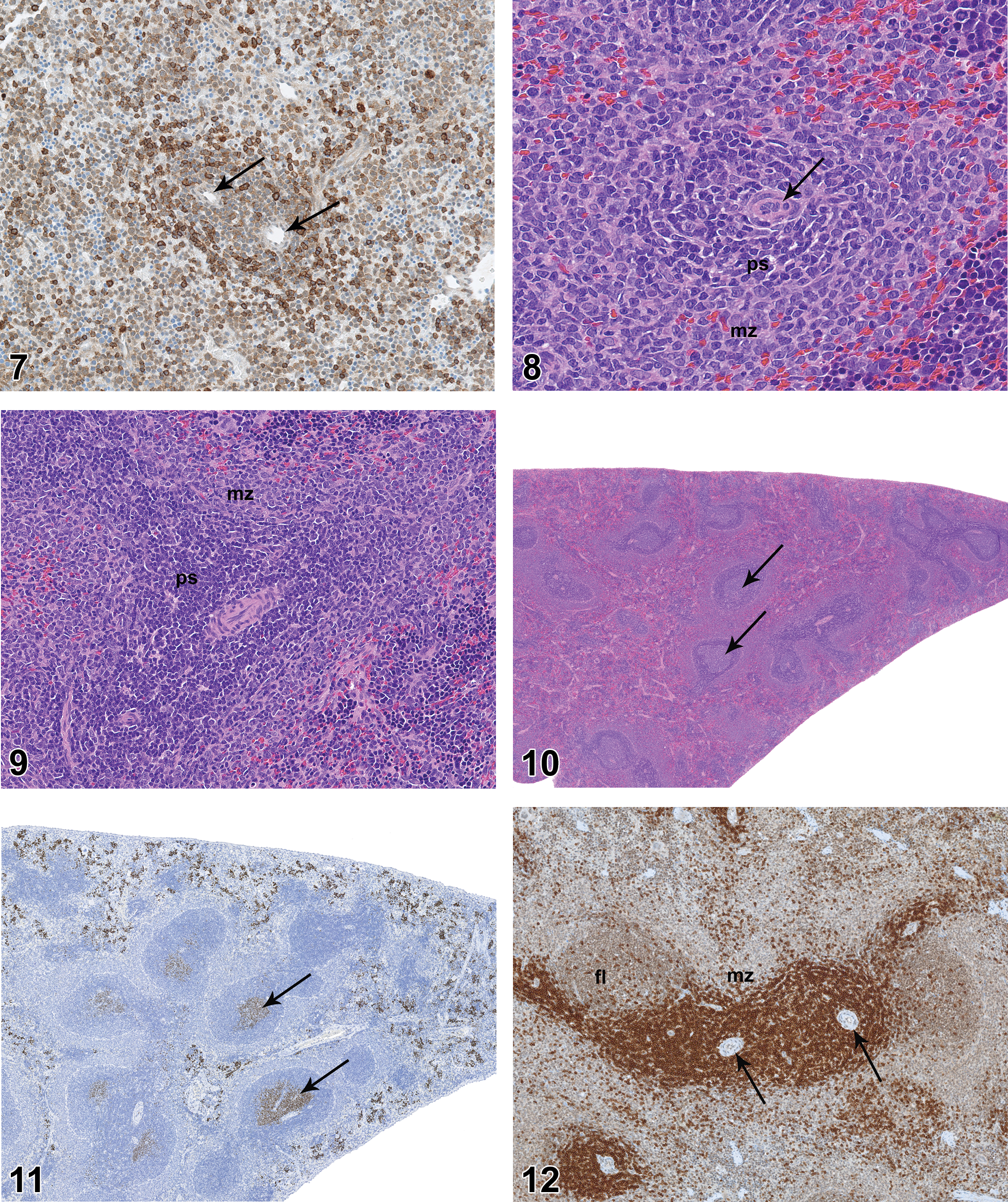
On PND 0, the parenchyma of the spleen consisted of a uniform sheet of cells with recognizable arterioles (Figure 5). The only suggestion of compartmental structure was periarteriolar aggregation of CD3-positive mononuclear cells (Figure 6). In some animals, there was a slight concentration of CD45RA-positive cells around the periphery of the periarteriolar aggregates (Figure 7). There were no lymphoid follicles or germinal centers at PND 0. By PND 7, the spleen of some rats had features of young adult spleen, with distinct PALS composed of a central zone of small, densely stained lymphoid cells and an indistinctly bounded MZ composed of larger mononuclear cells (Figure 8). On PND 14, both the PALS and the MZs were broader, approaching adult proportions (Figure 9). There was little change at PND 21 other than an increase in the overall size of the organ and internal structures. On PND 28, the PALS and MZs were fully developed. Incipient follicles were apparent in a few animals, but mature follicles with germinal centers were not present. On PND 35, there were well-formed follicles with germinal centers in more than half of the animals. On PND 42, the spleen had prominent PALS, well-developed MZs, and numerous follicles with germinal centers in all animals (Figure 10). On PND 42, Ki67 IHC staining revealed substantial proliferative activity in germinal centers of lymphoid follicles (Figure 11), and CD3 and CD45RA staining revealed prominent T- and B-cell differentiation (Figures 12 and 13, respectively).
Spleen of newborn rat (postnatal day 0) showing CD45RA-positive cells throughout splenic parenchyma, with slight concentration of CD45RA-positive cells around arterioles (arrows), suggesting incipient marginal zone. CD45RA immunohistochemical stain, original objective 20×.
Spleen of rat on postnatal day 42 showing brown-stained CD45RA-positive cells in follicles. Note sparse CD45RA staining in periarteriolar lymphoid sheath (ps). CD45RA immunohistochemical stain, original objective 5×.
Splenic extramedullary hematopoiesis (EMH), herein defined as erythropoiesis, myelopoiesis, and lymphopoiesis, was marked from PND 0 through PND 14, after which EMH gradually subsided to a moderate level at PND 42. Ki67 staining at PND 42 revealed substantial EMH persisted in the interfollicular pulp (Figure 11), but the level of EMH was reduced from that seen at PND 14.
Lymph Nodes (Axillary, Mandibular, and Mesenteric)
On PND 0, the lymph nodes were poorly developed, typically consisting of stromal elements and vascular structures with a minimal to mild diffuse cortical lymphocytic population (Figure 14), but no follicles, germinal centers, paracortex, HEVs, or plasmacytosis. The mesenteric lymph nodes were slightly more developed than the mandibular or axillary lymph nodes, and sometimes had ill-defined cortical clusters of lymphocytes that were consistent with incipient primary follicles.
On PND 7, none of the lymph nodes were considered to be fully developed, though the mesenteric lymph nodes of a few rats had lymphocytic clusters that suggested incipient follicle development. On PND 7, the axillary, mandibular, and mesenteric lymph nodes commonly had paracortex with HEVs (Figures 15–17). Overall, cortical cellularity was prominent in the mesenteric and mandibular lymph nodes, where it was judged to be fully developed, but the cortical lymphocyte population was somewhat less developed in the axillary lymph nodes. Germinal centers and plasmacytosis were not noted in any of the lymph nodes at PND 7.
On PND 14, all 3 lymph nodes were incompletely developed relative to later time points. The mandibular and mesenteric lymph nodes had fully developed diffuse lymphocytic populations and paracortical regions with HEVs. The diffuse cortical lymphocyte population of axillary lymph nodes was present in all rats but was judged to be somewhat underdeveloped relative to later time points. The paracortex and HEVs of axillary lymph nodes were also incompletely developed relative to later time points. Follicles were present in the mesenteric lymph nodes of nearly all rats, and small germinal centers were present in 4 of the 10 males and 4 of the 10 females. Neither follicles nor germinal centers were present in axillary or mandibular lymph nodes. Plasmacytosis was not present in any of the lymph nodes.
On PND 21, the mesenteric lymph nodes were fully developed, but the mandibular and axillary lymph nodes remained incompletely developed relative to later time points. Follicles with germinal centers were present in mesenteric lymph nodes of all rats (Figure 18). Follicles and germinal centers were not present in axillary or mandibular lymph nodes, and plasmacytosis was not noted in any of the lymph nodes. The diffuse cortical lymphocyte population of all 3 lymph nodes was fully developed, and paracortex with HEVs was fully developed in mandibular and mesenteric lymph nodes. Paracortex of axillary lymph nodes was fully developed in all rats, but the population of HEVs in axillary lymph nodes was slightly less developed than that seen at later time points.
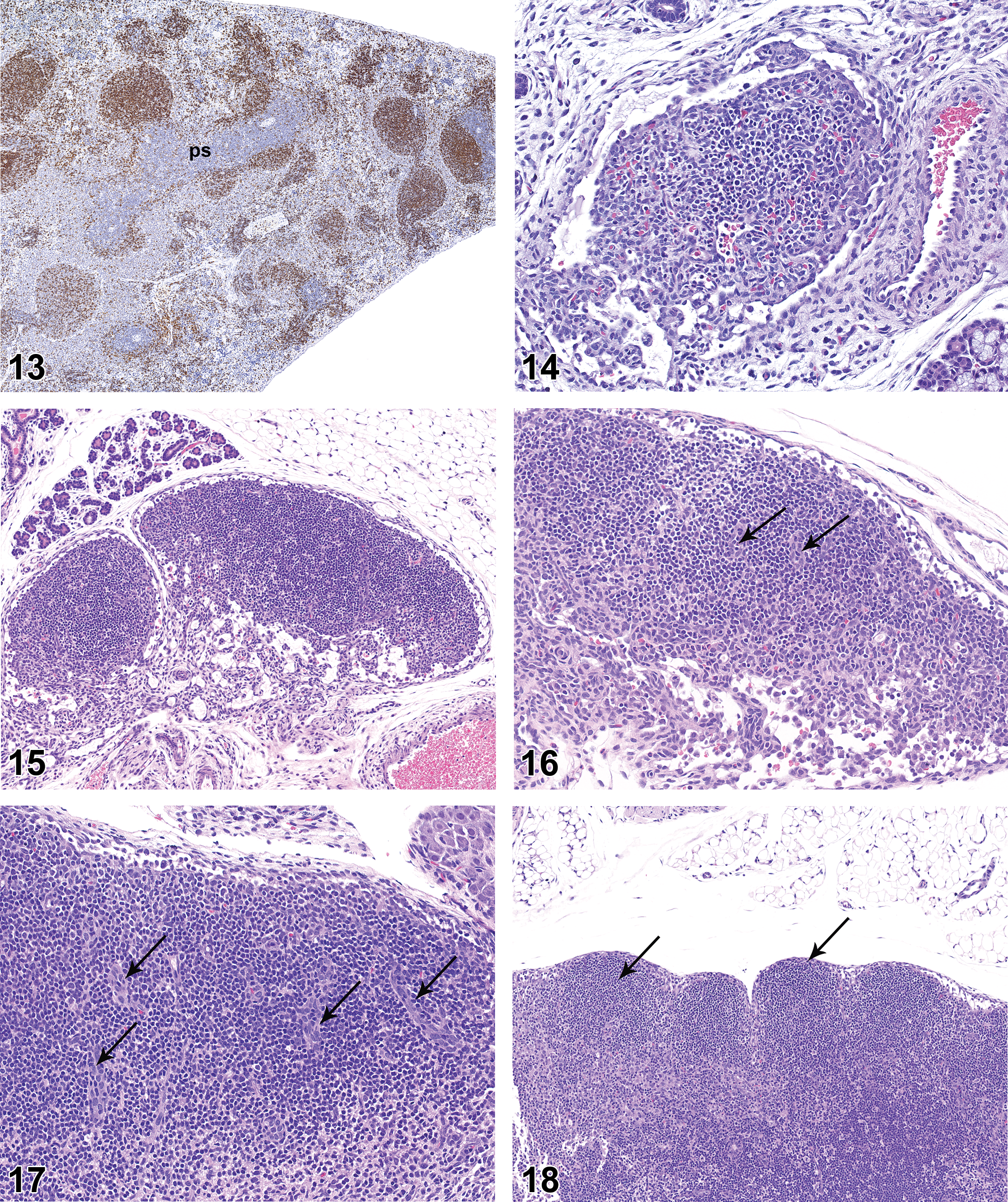
On PND 28, the axillary and mandibular lymph nodes remained incompletely developed relative to later time points. All mandibular lymph nodes had follicles and the majority had germinal centers (Figure 19), but the overall histologic features of the mandibular lymph nodes suggested the lymph nodes were underdeveloped relative to later time points. Follicles with mature germinal centers were present in all mesenteric lymph nodes (Figure 20). Follicles were present in axillary lymph nodes of nearly all rats but were underdeveloped as compared to later time points. Germinal centers were not present in axillary lymph nodes. Plasmacytosis was present in mandibular lymph nodes of 1 of the 10 males and 2 of the 10 females but was not present in axillary or mesenteric lymph nodes.
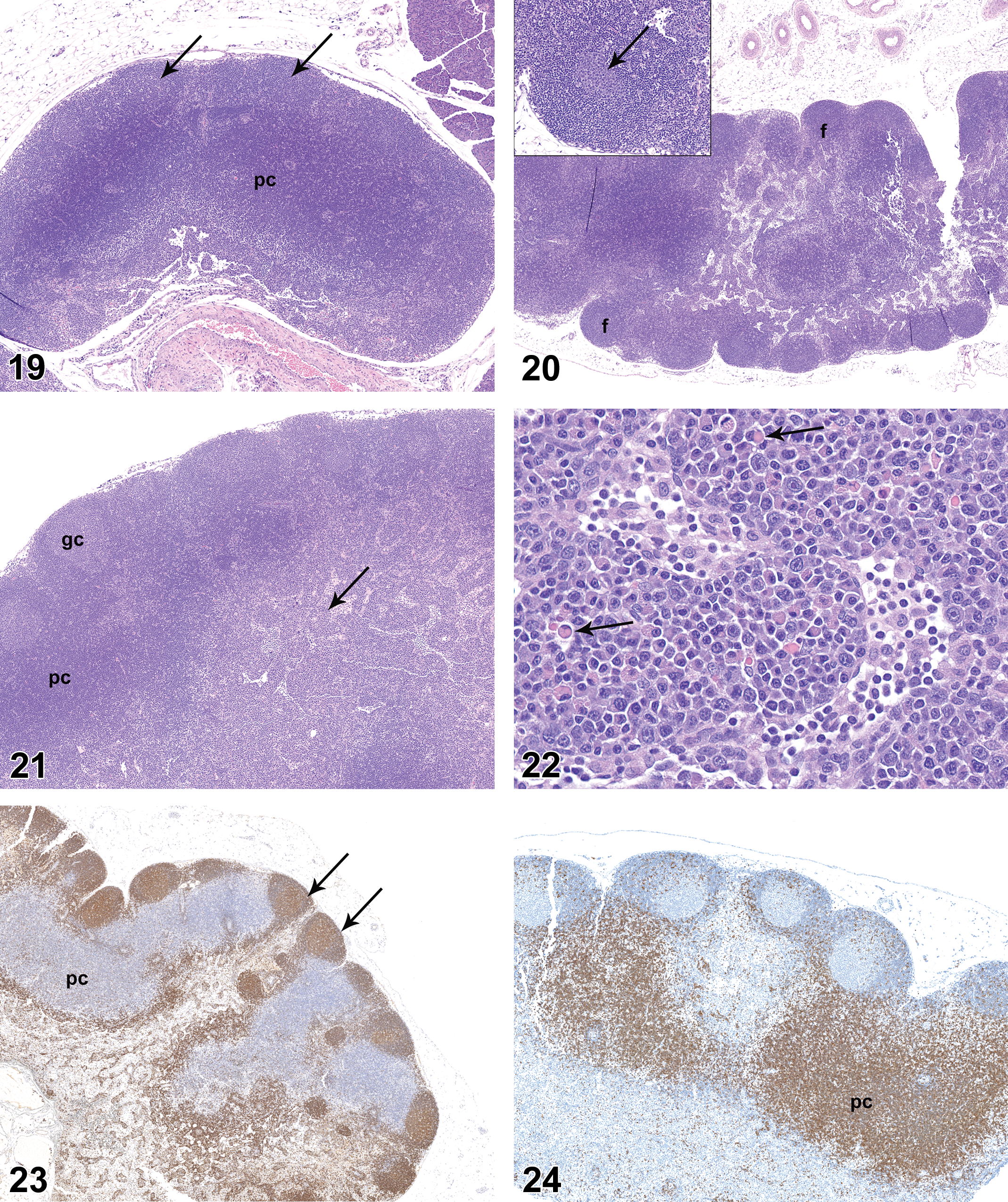
Mandibular lymph node from rat on postnatal day 28. Note dense cortical cellularity, multiple follicles (arrows), and highly cellular paracortex (pc). Hematoxylin and eosin, original objective 5×.
On PND 35, the mandibular and axillary lymph nodes were considered to be fully developed. Mandibular lymph nodes of all rats had numerous follicles and many follicles had prominent germinal centers (Figure 21). Germinal centers were not present in axillary lymph nodes. Plasmacytosis was present in the majority of mandibular lymph nodes (Figure 22) and was present to a minor degree in the mesenteric lymph nodes of 1 male and 1 female. Plasmacytosis was not noted in axillary lymph nodes.
On PND 42, all lymph nodes were considered to be histologically mature, but there were differences in histologic features of the various lymph nodes. Follicles and germinal centers of mandibular lymph nodes were less pronounced than those seen in the mesenteric lymph nodes. Accumulations of mature plasma cells (plasmacytosis) were present in the medulla of all mandibular lymph nodes, but plasmacytosis was not a prominent feature of mesenteric or axillary lymph nodes. Germinal centers were present in axillary lymph nodes of only 2 males and 2 females. Areas of B- and T-cell concentration in lymph nodes were readily demonstrated by CD45RA and CD3 IHC staining, respectively (Figures 23 and 24), and proliferative cells in germinal centers were clearly visible on Ki67 IHC staining.
In summary, the axillary, mandibular, and mesenteric lymph nodes on PND 0 had a diffuse cortical lymphocyte population with no additional distinctive lymphoid compartments. With time there was progressive development of additional lymphoid compartments, beginning with paracortex and HEVs and progressing to the formation of follicles with germinal centers. Mesenteric lymph nodes acquired the basic features of “adult morphology” (cortical follicles, germinal centers, and paracortex) earlier than either axillary or mandibular lymph nodes. Axillary lymph nodes on PND 42 rarely had well-developed germinal centers, though primary follicles were commonly observed. Mandibular lymph nodes developed a prominent plasma cell population that typically was not present in axillary or mesenteric lymph nodes.
Gut-associated Lymphoid Tissue (GALT)
The presence of GALT of the small intestine was scored as 2 separate elements, namely, mucosal lymphoid follicles (PP) and diffuse mucosal mononuclear cell population, the latter representing intraepithelial lymphocytes (IEL) and the mononuclear cell population in the lamina propria of villi.
On PND 0, the small intestinal mucosa had small cellular aggregates that were consistent with developing PPs (Figure 25), but the mucosa was essentially devoid of diffuse mononuclear cell population (Figure 26). On PND 7, the developing PPs were more pronounced and had distinct follicles, but germinal centers were not present (Figure 27). PPs became progressively more cellular on PND 14 and PND 21. On PND 21, the majority of PPs had germinal centers (Figures 28 and 29), though the germinal centers were not as developed as those seen at later time points. On PND 21, the lamina propria at the base of a few villi contained aggregations of lymphocytes that were consistent with developing lymphocyte-filled villi (Figure 30). On PND 28, the PPs were fully developed histologically, with prominent follicles and distinct germinal centers. On PND 28, the small intestinal mucosa also had individual lymphoid follicles (ILFs; Figure 31), distinct lymphocyte-filled villi (Figure 32), and cryptopatches (CPs; Figure 33), the latter being randomly distributed clusters of lymphoid cells in the basilar lamina propria (Pabst et al. 2005). IHC staining revealed the expected B- and T-cell areas in PPs (Figures 34 and 35), with Ki67-positive cells in germinal centers.
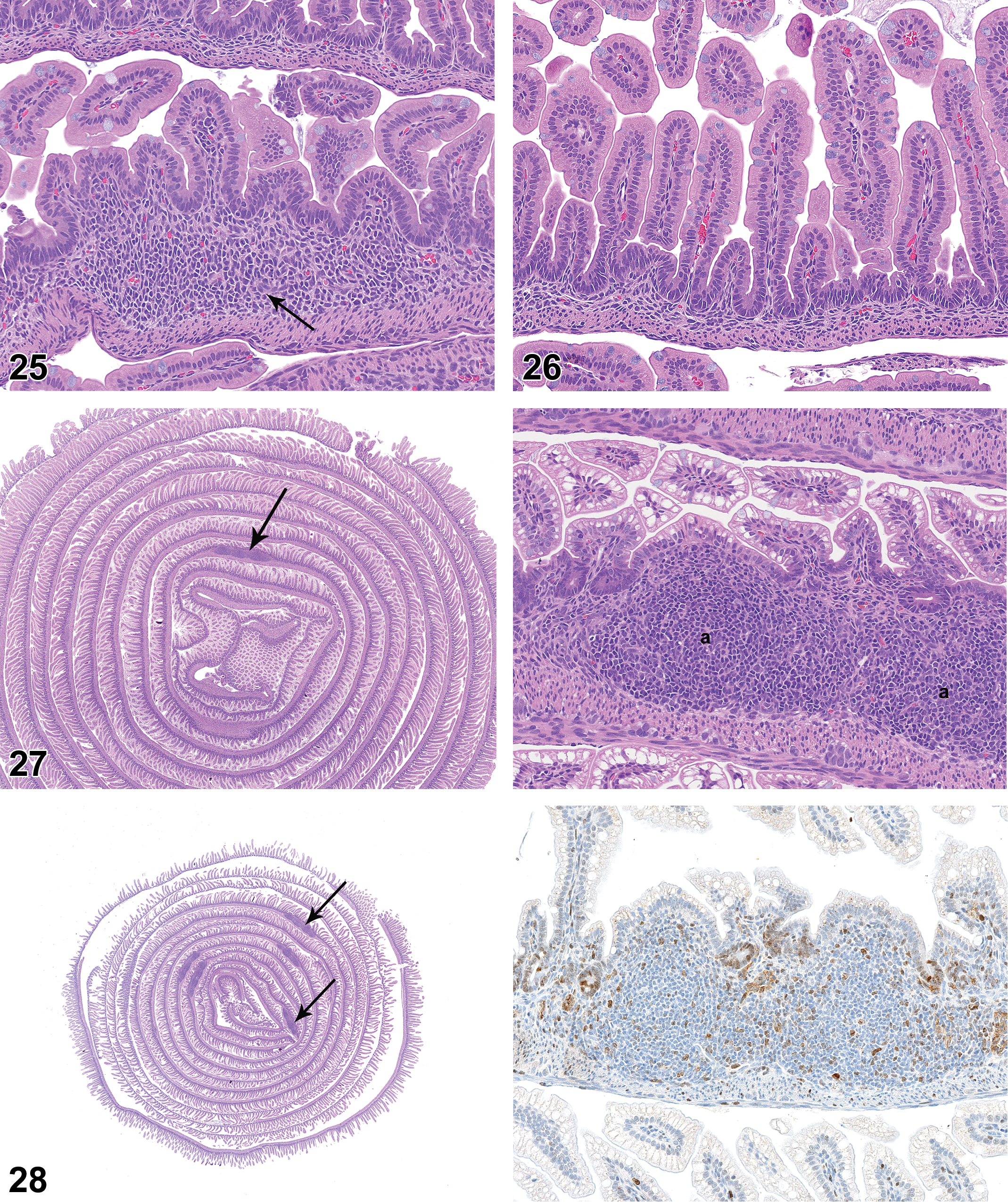
Small intestine of newborn rat (postnatal day 0). Note rudimentary Peyer’s patch (arrow). Hematoxylin and eosin, original objective 20×.
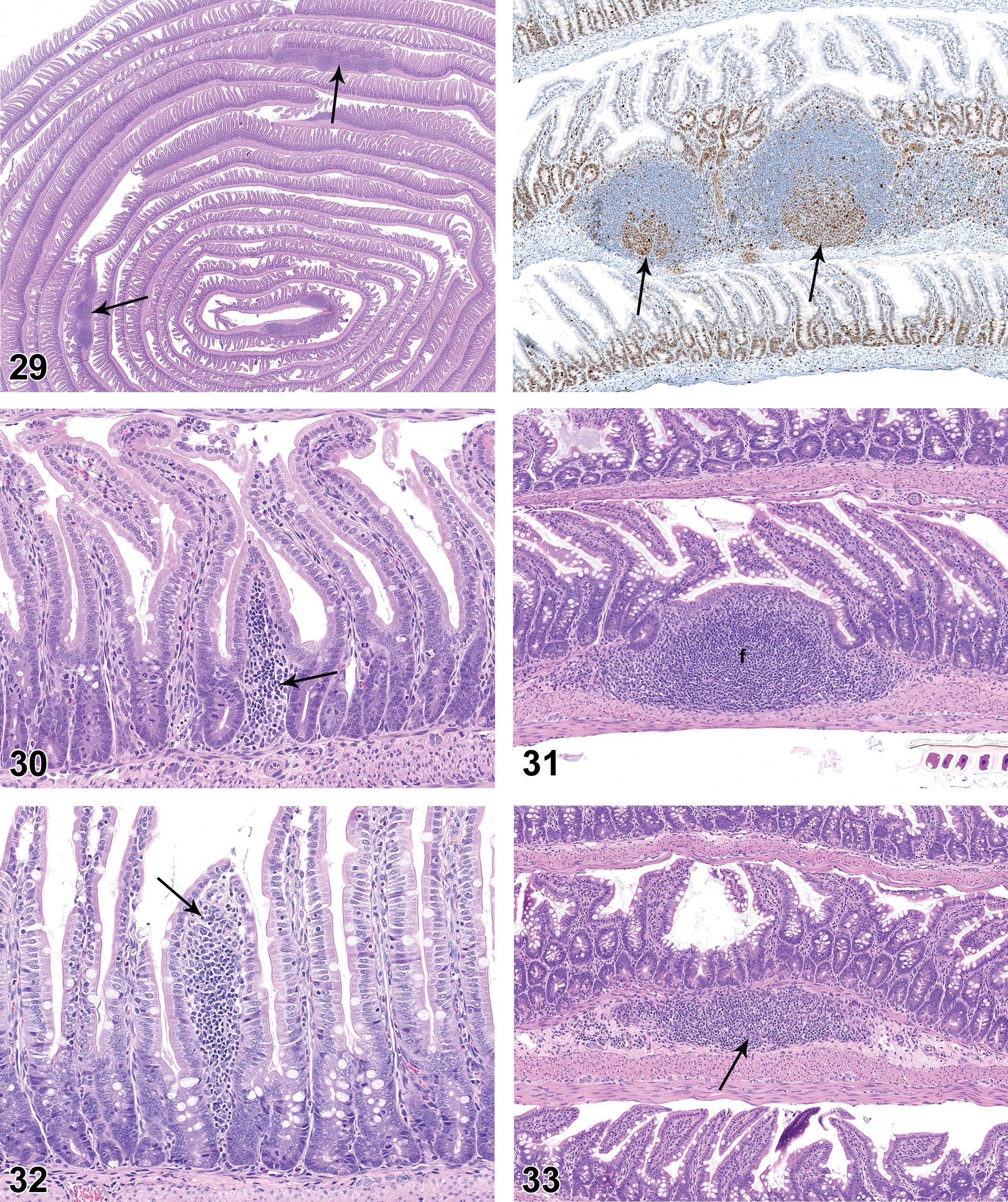
Left: Swiss roll preparation of small intestine from rat on postnatal day 21. Note multiple prominent Peyer’s patches (arrows). Hematoxylin and eosin, original objective 12.5×. Right: Positively stained proliferative cells in germinal centers (arrows). Ki67 immunohistochemical staining, DAB chromagen, original objective 10×).
Small intestine with Peyer’s patch from rat on postnatal day 28 showing prominent B-cell follicles (arrow). CD45RA immunohistochemical staining, DAB chromagen. original objective 5×.
Development of the diffuse intestinal mononuclear cell population lagged behind the development of PPs. A diffuse mucosal mononuclear cell population was not present on PND 0 to 14. As noted previously, clusters of mononuclear cells were noted within a few villi at PND 21, but these structures appeared to represent developing lymphocyte-filled villi rather than the diffuse mucosal mononuclear cell population. The diffuse mucosal mononuclear cell population that characterizes the small intestine of young adult rats was not present until PND 35 (Figure 36), which contrasts with the “adult” status of PPs observed a week earlier at PND 28.
BALT
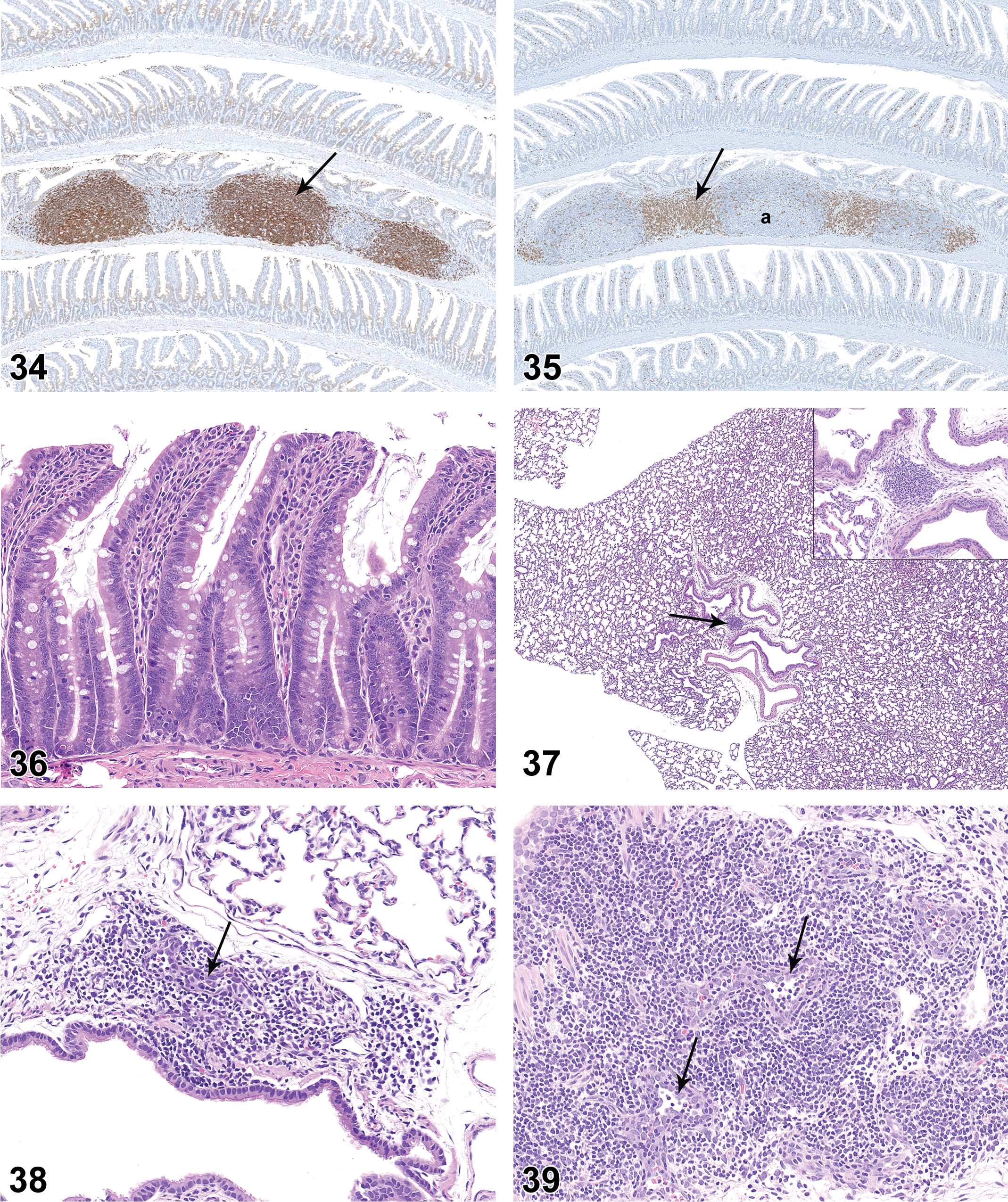
BALT was not present at PND 0. On PND 7, peribronchiolar cellular aggregates consistent with incipient BALT were noted in 3 of the 10 females (Figure 37). An increased number of rats had histologically discernible BALT on PND 14 and 21, and BALT was present in all rats from PND28 through PND 42. BALT had distinct HEV as early as PND 14, and by PND 21 HEVs were observed within BALT in a moderate number of rats (Figure 38). HEVs remained clearly visible as BALT cellularity increased through PND 35 and 42 (Figures 39 and 40). BALT was present in all animals on PND 42 and had prominent lymphoid cell populations that contained variably prominent HEVs. There was substantial variation in the prominence of BALT in individual juvenile rats at the various PND intervals. Follicles and germinal centers were not a feature of BALT through PND42.
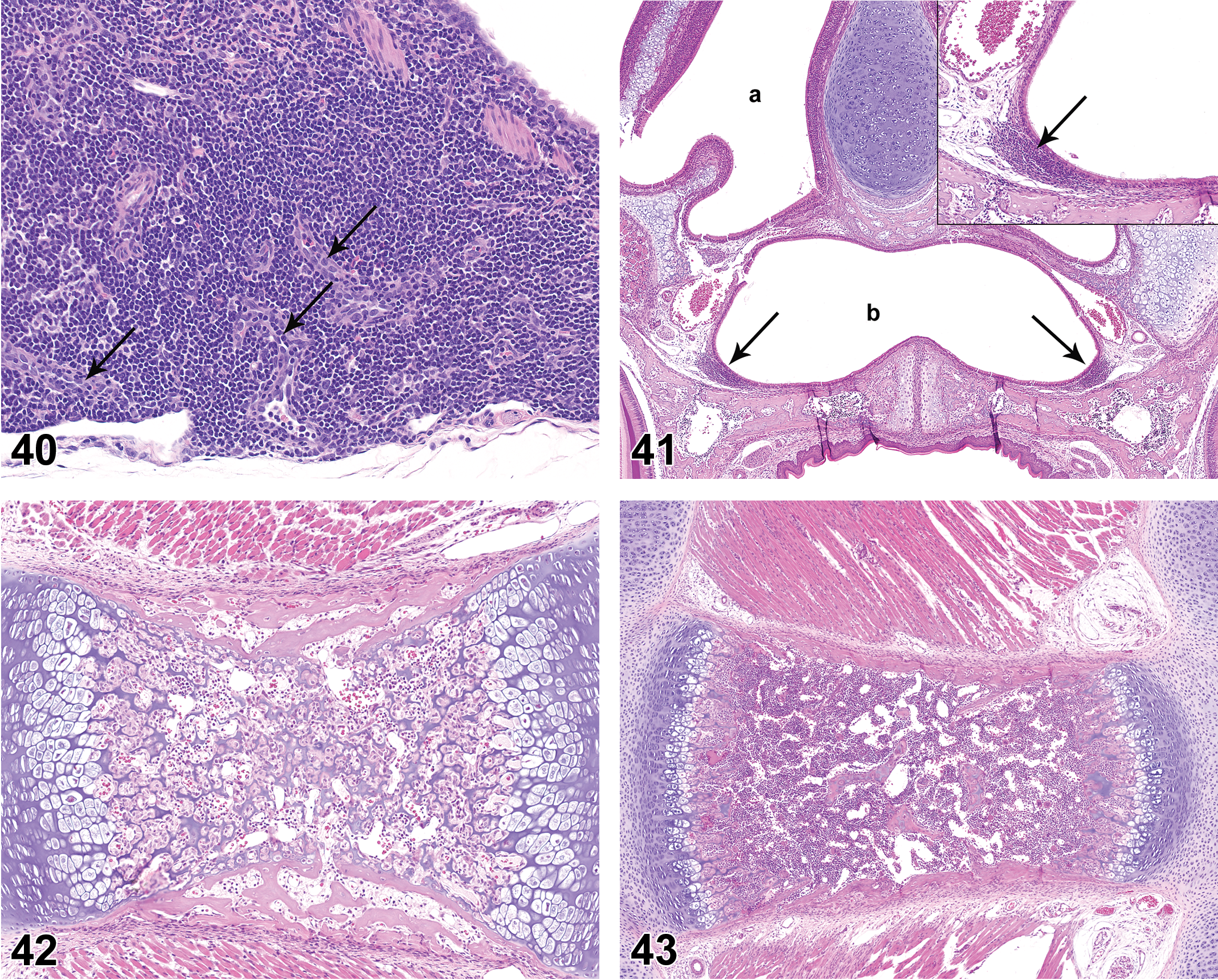
Lung of rat on postnatal day 42. Note highly cellular bronchus-associated lymphoid tissue with high-endothelial venules (arrows). Hematoxylin and eosin, original objective 20×.
NALTs
NALT was not present at PND 0. Nasopharyngeal lymphoid aggregates were sporadically present at PND 7 to 42 (Figure 41). HEVs were variably present within the lymphoid aggregates, but neither follicles nor germinal centers were present in NALT at any time point.
Bone Marrow
At PND 0, the bone marrow had a distinct hematopoietic cell population, though the marrow was very hypocellular in comparison to later time points (Figure 42). At PND 0, the femoral marrow was more frequently populated than the sternal marrow. At PND 7, the sternal marrow cavity remained somewhat underdeveloped relative to later time points (Figure 43), but the femoral marrow was fully developed. By PND 14, the sternal and femoral marrow cavities were filled with hematopoietic cells, essentially consistent with young adult rat histomorphology (Figure 44).
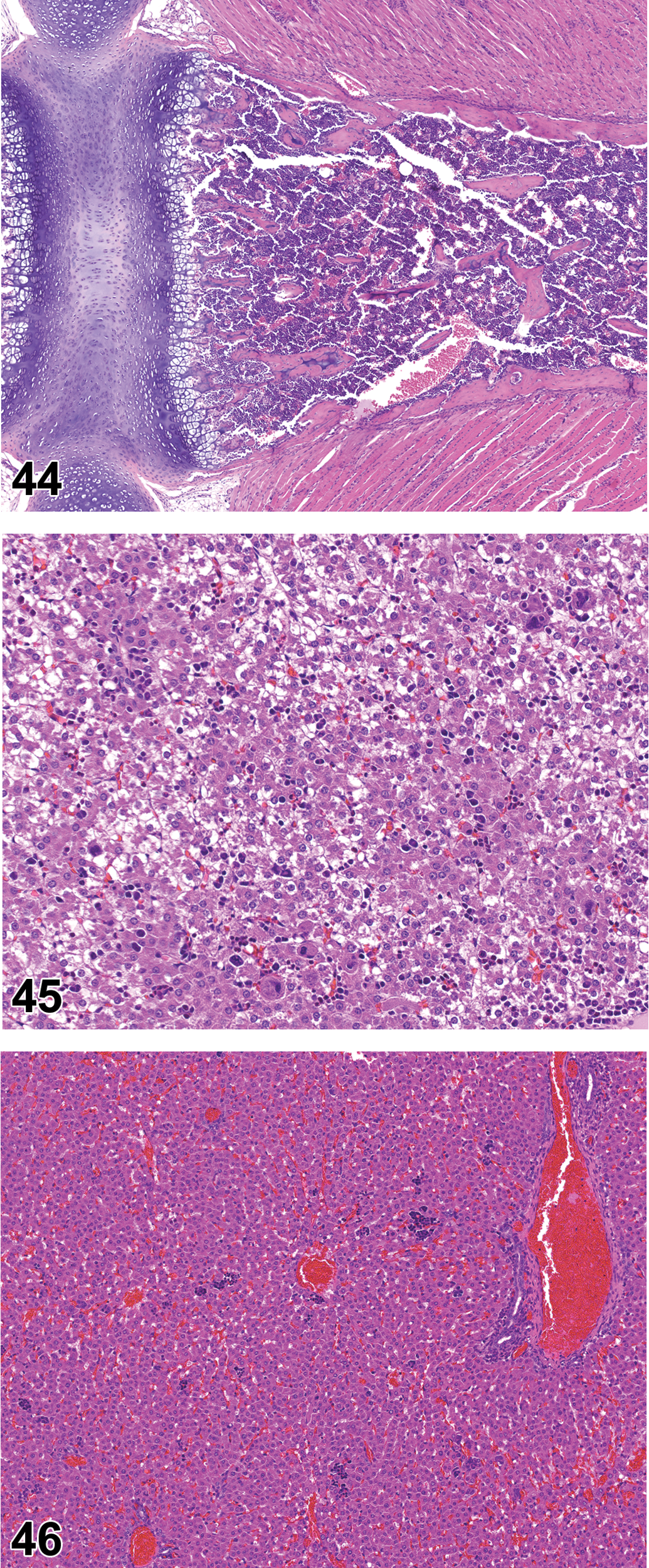
Sternum of rat on postnatal day 14. Note dense cellularity of marrow cavity, similar to that seen in young adult rats. Hematoxylin and eosin, original objective 5×.
Liver
At PND 0 through PND 3, the liver had a moderate diffuse hematopoietic cell population (Figure 45), but by PND 10 the hepatic hematopoietic cell population consisted of scattered clusters of cells (Figure 46). A gradual decline in hepatic hematopoiesis continued through PND 42, though widely dispersed clusters of hematopoietic cells were present in the liver of many rats on PND 42.
Discussion
Thymus
At GD 15 of our preliminary study, the thymus was near the anterior aspect of the pleural cavity and was composed of a uniform sheet of cells with no organoid structures. The thymus had distinct corticomedullary relationships at GD 20 but lacked the dense population of cortical lymphocytes that is present in young adult rats. The postnatal study indicated that the thymus reaches a histomorphological stage of maturity in advance of many other lymphoid organs, essentially in parallel with the maturing bone marrow. On PND 0, the thymic cortex was thinner and less cellular than that seen in young adult rats, though all cell populations appeared to be present and corticomedullary relationships were distinct. In the earlier stages of development (i.e., prior to PND 21), the thymus sometimes exhibited a less densely cellular peripheral zone that had a substantial population of large mononuclear cells, consistent with the “large lymphocyte population” in the thymic MZ described in the thymus embryology literature (Harland 1940) and the population of lymphoblasts and/or large lymphocytes described in cell proliferation analysis of the developing thymus (Aboussaouira, Moustafa, and Idelman 1988). The thymus rapidly matured to its final adult morphology with highly cellular cortex and less densely cellular medulla, and by PND 14 exhibited adult morphology, with highly cellular cortex and less densely cellular medulla. The only subsequent change was an increase in overall size of the organ.
Observations in the present studies generally correlated with published information. In a detailed morphometric study of IHC-stained thymus and spleen of rats at 1, 2, 3, 4, 9, 19, 23, 46, and 57 weeks of age (Kodama et al. 2012), the thymic T-cell population increased markedly from 3 to 4 weeks of age, in advance of the spleen, which correlates with the increase in size of the thymus seen in the present study. In the reported study, both the thymus and the spleen exhibited mature T- and B-cell populations by 9 weeks of age, which correlates with histologic observations in the present study. It should be emphasized that the reported study was based on time points that did not include the PND 35 and PND 42 time points of the present study.
Spleen
The present studies in rats suggested the postnatal histomorphological development of the immune system components of the spleen is similar to those observed in mice (Kaufman and Bard 1999; Landreth 1993; Paige et al. 1981). At GD 15, the spleen consisted of a uniform sheet of unidentified mononuclear cells with a superficial layer of plump mesothelial cells. There were no defining internal histologic features such as PALS, follicles, germinal centers, or MZs. At GD 20, the spleen was easily recognized histologically but lacked follicles, PALS, or other internal structures associated with the lymphoid system. At PND 0, there were clusters of large mononuclear cells centered on splenic arterioles, consistent with an early manifestation of PALS, though the individual cell morphology was not typical of the small mature lymphocytes that characterize the PALS of adult rats. The majority of the periarteriolar cellular clusters consisted of a uniform population of large mononuclear cells, but in a few animals the periarteriolar cellular aggregation had a central (periarteriolar) population of densely basophilic small mononuclear cells that were consistent with mature lymphocytes. IHC staining suggested the periarteriolar cell clusters had an internal concentration of CD3+ cells and a less pronounced peripheral concentration of CD45RA+ cells, similar to the pattern seen with the PALS and MZs of the mature spleen. On PND 7, there was expansion of the periarteriolar cellular sheath, which was typically composed of large mononuclear cells with a minor but distinct central population of mature lymphocytes. At PND 14, the PALS was more pronounced in comparison to PND 7, and there was an increased population of centrally located mature lymphocytes. The histologic features between PND 0 and PND 14 suggested the early presence and subsequent expansion of a periarteriolar cell population that eventually constituted the MZ, with subsequent manifestation of the centrally placed population of mature T lymphocytes.
In the present studies, the histologic features of the spleen were slow to develop postnatally, in comparison to the thymus and bone marrow, and did not exhibit fully mature histomorphology until PND 42. The T-cell component (PALS) of the spleen exhibited an adult level of maturity before the B-cell component (follicles and germinal centers), though a minor population of CD45RA+ cells appeared in the periarteriolar region very early in development. It should be stressed that the fully mature histomorphology of the spleen in the present studies was based on a “normal” level of antigenic stimulation in conventional rats in the relatively low-pathogen environment of a conventional laboratory animal colony. Quite possibly earlier maturation of secondary follicles with germinal centers would be observed if the animals received deliberate antigenic stimulation.
Observations in the present studies generally correlated with published observations. In a morphometric study of IHC-stained spleens of rats (Kodama et al. 2012), the area and cellularity of the white pulp increased postnatally until 9 weeks of age and remained stable thereafter. Lymphoid follicles were not apparent until 3 weeks of age and were clearly visible at 4 weeks of age. T-cell populations in PALS and MZs increased with age up to 9 weeks and remained stable thereafter, but continued to increase with age in lymphoid follicles. B-cell populations increased with age in all areas, with the most pronounced populations contained in lymphoid follicles.
Lymph Nodes
Lymph nodes develop relatively late in fetal life and persist throughout adulthood (Losco and Harlemen 1992), and their appearance at any given time is directly related to the type and amount of antigenic stimulation (Tizard 1987). In the neonate, lymph nodes have a distinct reticular framework with a small number of lymphocytes in the cortex (Arno 1980). IHC studies of popliteal lymph nodes of immature rats revealed T cells and interdigitating cells were present from birth (van Rees et al. 1985). B cells were present on the second day of life, and the first primary follicles appeared at day 18 (van Rees et al. 1985). When young rats of various ages were sacrificed 5 days following immunization by injections of trinitrophenyl (TNP)–keyhole limpet hemocyanin (KLH) in the hind footpads, popliteal lymph nodes had primary follicles at 19 days of age and secondary follicles with germinal centers at 23 days of age (van Rees, Dijkstra, and van Rooijen 1986). A similar study of responses to thymus-independent type-1 (TI-type 1) and TI type-2 antigens (TNP-lipopolysaccharide [LPS] and TNP-Ficoll, respectively) revealed a specific antibody response to NTP-LPS at 19 days after birth, but antibody response to TNP-Ficoll was low even at 6 weeks after birth (van Rees, Dijkstra, and van Rooijen 1987). The latter observations highlight the potential disconnection between morphologic evidence of maturity in immune system organs versus full functional maturity.
Observations in the current studies indicated that postnatal development of lymph nodes occurred somewhat later than the thymus and bone marrow, and there were differences in postnatal developmental schedule of enteric versus somatic lymph nodes. Development of the mesenteric lymph node started before development of the mandibular and axillary lymph nodes, and the mesenteric lymph node reached “adult histomorphology” prior to the mandibular and axillary lymph nodes.
Lymph nodes at PND 0 consisted primarily of stromal and vascular elements. Early postnatal development started with an accumulation of diffuse cortical lymphocytes and proceeded to formation of paracortical regions with HEVs. Formation of primary lymphoid follicles followed, with development of secondary lymphoid follicles bearing germinal centers as the final step in maturation of the histologic features. There was variation in the histomorphological maturation of lymph nodes in individual rats, but it was not clear whether this represented innate variability or differences in immune system stimulation of individual animals.
In addition to differences in postnatal development, the 3 types of lymph nodes had consistent differences in histomorphology. Mandibular lymph nodes had more pronounced populations of plasma cells in medullary cords than did mesenteric or axillary lymph nodes. This did not appear to be simply a matter of advanced maturity of the mandibular lymph nodes, as the histologic features of mesenteric lymph nodes suggested they started to develop earlier and reached morphological maturity earlier than the other lymph nodes. Pronounced plasmacytosis of mandibular lymph nodes is a common observation in young rats, but the underlying immunobiology is not apparent. Axillary lymph nodes rarely had germinal centers at PND 42, a feature that is commonly shared by young adult rats at the age of termination of typical 28-day toxicity studies (approximately PND 77). The chain of ILFs that constitute the elongated mesenteric lymph nodes commonly exhibited different stages of histologic maturity in different regions of the lymph node, presumably reflecting different levels of immunologic stimulation from various drainage areas.
GALT
The mucosa-associated lymphoid tissue (MALT) found in many internal organs is commonly known by the invested tissue, for example, GALT for gut-associated, BALT for bronchus-associated, NALT for nasopharynx-associated, LALT for larynx-associated lymphoid tissue, CALT for conjunctiva-associated lymphoid tissue, DALT for salivary duct-associated lymphoid tissue (Cesta 2006a), and GENALT for genital-associated lymphoid tissue (Lehner et al. 1995). The MALT constitutes an important line of defense against invasion of pathogens and, in the case of GALT, serves a role in development of immunological tolerance to ingested antigens. The GALT, along with the lymphocytic population of the biliary epithelium, also has the critical function of producing IgA, which is necessary for resistance to potentially pathogenic intestinal microflora (Belkaid 2013; Jung, Hugot, and Barreau 2010). In many species, but not all, there is a reciprocal relationship between GALT and intestinal microflora, with microflora being necessary for the maturation of GALT (O’Hara and Shanahan 2006).
PPs consist of B-cell follicles with intervening T-cell areas that also contain macrophages and dendritic cells. The lymphoid follicle is covered by a single layer of altered intestinal epithelial cells (follicle-associated epithelium [FAE]) that form a dome over the PPs. The FAE differs from adjacent villus epithelium in several ways that give the FAE intimate contact with luminal contents, immediate accessibility to antigens, and close contact with the immune system (Jung, Hugot, and Barreau 2010). An important morphologic feature of the FAE is the presence of specialized M (microfold) cells and associated basolateral compartments that contain immunocytes. M cells transport luminal antigens and bacteria toward the associated immune cells that activate or inhibit the immune response, leading to either tolerance or systemic immune response (Jung, Hugot, and Barreau 2010).
PPs are the best-known subset of GALT, but the diffuse IEL population probably has a greater number of lymphocytes (Bruder et al. 1999). The IEL has a substantial population of memory T cells in addition to a preponderant population of IgA-producing cells (Cheroutre 2005; Hayday et al. 2001). The IEL population also serves as a site of T-cell maturation following thymic involution (Abo 2001).
The large intestine has prominent lymphoid aggregates that differ from PPs in location (submucosal rather than the mucosal), vascular supply, and immunological reactions (Crouse et al. 1989). Herein, the term PPs refers only to the small intestinal structures.
In addition to PPs and the diffuse mucosal lymphocytic population, the mucosa of the small intestine has ILFs and smaller lymphoid aggregates known as CPs and lymphocyte-filled villi (Hamada et al. 2002; Moghaddami, Cummins, and Mayrhofer 1998). Studies on genetically modified mice suggest that PP, ILF, and CP represent 3 distinct types of organized small intestinal lymphoid tissues in the mouse (Hamada et al. 2002). CPs are phenotypically indistinguishable from lymphoid tissue inducer (LTi) cells of the fetus, suggesting that CP cells are the adult counterpart of fetal LTi cells (Lugering and Kucharzik 2006). It is likely that ILFs are derived from CPs under inflammatory conditions (Lugering and Kucharzik 2006).
Though the various components of MALT are grouped for purposes of general discussion, there are differences in histogenesis and immunobiology of MALT in different locations. As a result of these differences, postnatal development of MALT cannot be presented as a single unified process.
Information regarding embryogenesis and postnatal development of PPs in rats is less extensive than for mice, but the basic features have been described (Chen et al. 1995; Lyscom and Brueton 1983; Sminia, Janse, and Plesch 1983). Shifts in the cell populations of PPs are known to take place as the organs mature during the early postnatal period. A histological and IHC study (Chen et al. 1995) of PPs formation in rats revealed surface-IgM (sIgM)-bearing cells (B cells) scattered throughout the PPs at PND 1. On PND 3, sIgM-bearing cells accumulated to form primary follicles that were associated with domed elevations in the surface epithelial contour (ibid.). OX-2-positive cells (follicular dendritic cells) were present in the center of primary follicles by PND 5 (ibid.). Germinal centers first appeared within lymphoid follicles on PND 18, and by PND 21 most follicles contained a germinal center (ibid.). On PND 5, each PP contained 6 to 8 lymphoid follicles, with the lymphoid follicles subsequently increasing in size and number. The mean number of follicles/PPs reached the adult level (11.1 follicles/PPs) on PND 21 and remained at that level for the next 15 weeks, though individual follicles continued to enlarge during the observation period (ibid.). In another study (Lyscom and Brueton 1983) based on fluorescence microscopy of labeled cells extracted from the small intestine of neonatal and juvenile rats, the relative distribution of PPs lymphocyte subtypes at 4 weeks after birth was similar to that of mature rats, but the histomorphology of the tissue was not considered well differentiated until a week later. Observations in these published studies were generally consistent with observations in our study, though in our study the PPs were considered to be fully developed at PND 28 (4 weeks after birth).
While the development of PPs in humans, mice, and rats appears to be similar, and is consistent with PPs as secondary lymphoid organs that undergo full development in response to antigenic stimulation (Chen et al. 1995), the same cannot be said for all species. For example, PPs of sheep have a number of anatomical and functional characteristics that suggest they are primary rather than secondary lymphoid organs in that species and are fully developed at the time of birth (Reynolds and Morris 1983).
PPs and the diffuse intestinal lymphocyte population have different postnatal development sequences and somewhat different immunobiological features, but share responsibility for production of IgA (Beagley and Elson 1992), which is a critical component of defense against enteric pathogens (Endt et al. 2010; Hooper and Macpherson 2010). In human neonates and infants, the gastrointestinal immune system is functionally immature, and full production of gut IgA is not present until 2 years of age (Savilahti 1972). Studies in rats have shown the IEL population increases gradually from birth and reaches a mature level at 6 weeks of age (Lyscom and Brueton 1983). There was a transient increase in IEL in postnatal week 1 in rats, which was suspected to represent a population of maternal lymphocytes from the colostrum (milk lymphocytes; Lyscom and Brueton 1983). This maternal lymphocyte population is known to accumulate in the colostrum of humans (Diaz-Jouanen and Williams 1974; Richie et al. 1982) and have been shown to cross the mucosal barrier of the intestine of rats (Seelig and Billingham 1981).
PPs developed in advance of the diffuse IEL population in the present studies. Primitive lymphoid structures were observed in the small intestine at GD 20 and were invariably present at PND 0. Lymphoid follicles of PPs expanded in size through the period PND 7 to 21 and reached the adult level by PND 28. Germinal centers first appeared in PPs at PND 21 and reached adult proportions by PND 28. The diffuse IEL population started to appear at PND 21 but was not subjectively judged to be at the adult level of population density until PND 35. Observations in the current study were generally consistent with published information. One exception was the appearance of germinal centers, which were reported as present in most PP follicles by PND 21 (Chen et al. 1995). In our study, the “adult level” of germinal centers was not present until PND 28, while the number of germinal centers at PND 21 was judged to be immature. This minor difference may be due to different observational criteria or may reflect differences in the level of antigenic stimulation from the intestinal contents.
Observations in the present study were consistent with postnatal maturation of the intestinal immune system structures of rats in response to antigenic stimulation, presumably from intestinal contents; thus, the intestinal immune system structures would be classified as secondary lymphoid organs. Mesenteric lymph nodes typically had a minor diffuse cortical lymphocyte population at PND 0, and by PND 7 had immature follicles and paracortex with HEVs. At PND 21, the mesenteric lymph nodes were judged to be fully mature, with secondary lymphoid follicles containing germinal centers. PPs were discernible at PND 0 but did not reach fully mature histomorphology until PND 28. The diffuse IEL population of the small intestine started to appear at PND 21 but was not developed to adult level until PND 35.
Some caveats are in order regarding interpretation and extrapolation of the observations on GALT as presented herein. The present study was not designed to detect the total number of PP, as only 1 histologic section was taken from each Swiss roll preparation of small intestine. Observations and discussion related to PP of the small intestine should not be extrapolated to the submucosal lymphoid follicles of the large intestine, as these structures are anatomically and functionally different from PP of the small intestine (Crouse et al. 1989). References to the presence and number of PP in specific segments of small intestine in various species are obviously influenced by the defining boundaries of those intestinal segments. The ileum of humans and dogs is distinguished by antimesenteric as well as mesenteric vascular supply, and the ileum is a substantial component of the small intestine. In humans, approximately 46% of PPs are concentrated in the distal 25 cm of the ileum (Van Kruiningen et al. 2002). By contrast, in Wistar rats, the ileum is defined as the region of small intestine reached by the ileocecal ligament, thus the ileum as defined is only 1 to 2 cm long (Kuper et al. 2007) and is commonly devoid of PPs. These precise anatomical distinctions are largely academic except when study protocols for nonclinical toxicology studies have a generic, multi-species requirement for examination of “ileum with PP.” Ileum with PPs is easily collected in dogs and nonhuman primates, but is largely impossible to collect in rats. A more practical approach is to list PP separately in study protocols, thus avoiding the confines of intestinal anatomy and allowing necropsy personnel freedom to collect grossly visible PP regardless of their location in the small intestine. In addition to the influence that environmental variables have on immune system morphology and cellular constituents in rats, stain-related variation has been demonstrated (Bruder et al. 1999). PPs were smaller in Fischer 344 rats than in Wistar rats, and interindividual variability in IEL composition was greater in random-bred Wistar rats than in inbred Fischer 344 and Lewis rats (Bruder et al. 1999).
NALT
NALT and PP have similar immunological characteristics and the same types of resident immunocompetent cells, and antigen exposure to either NALT or PP can stimulate antigen-specific immunological responses. However, NALT-targeted immunization generally induces immunity in the respiratory and reproductive tissues, while PP-associated immunization generates protective immunity in the gastrointestinal tract (Belkaid 2013; Yuki and Kiyono 2003).
In rodents, NALT is found on both sides of the nasopharyngeal duct and is considered analogous to Waldeyer’s ring (composed of palatine tonsil, lingual tonsil, and nasopharyngeal tonsil) in humans (Kuper et al. 1990, 1992). Nasopharynx-associated lymphoid tissue consists of FAE, HEVs, and T- and B-cell areas that are similar to PP. Antigen-presenting cells, including dendritic cells and macrophages, and antigen-sampling M cells are present in the FAE of NALT (Hameleers et al. 1989; Porgador et al. 1998; Spit et al. 1989).
Despite similarities in structure and function, there are significant differences in the organogenesis of PP versus NALT. The HEV of PP express mucosal vascular addressin cell-adhesion molecule (MAdCAM-1; Csencsits, Jutila, and Pascual 1999) and NALT-associated HEVs express peripheral-node addressin (PNAd; Kiyono and Fukuyama 2004), thus different populations of naive B cells are sequestered in PP versus NALT. Embryological and postnatal development of PP and BALT are also different. Precursors to PP were observed in mice as early as GD 15.5 (Adachi et al. 1997), and dome-shaped PPs were observed in later stages of embryogenesis in mice (Hashi et al. 2001). By contrast, NALT was not observed during embryogenesis or in newborn mice (Fukuyama et al. 2002). In this study, PNAd+ HEVs with associated lymphocytes were first detected in nasal tissue of mice at PND 7, and complete bell-shaped NALT was not observed until 5 to 8 weeks after birth (Fukuyama et al. 2002). These observations in mice suggest prenatal initiation of lymphoid organogenesis for PPs and postnatal organogenesis for NALT, with the latter being dependent on environmental stimulation (Kiyono and Fukuyama 2004). Sparse published observations on rats suggest NALT development in rats is similar to that seen in mice (Hameleers et al. 1989).
In the present study, the nasopharyngeal region of rats at PND 0 and PND 7 was devoid of lymphoid elements. Mucosal lymphocytic clusters in the nasopharyngeal region were first noted in 1 male at PND 14 and were sporadically present at remaining time points. The cellular density of the NALT lymphoid population was subjectively judged to be at adult level at PND 35, but it was noted that NALT occurrence in PND 42 males was lower than NALT occurrence in PND 35 males. These observations suggest the presence of NALT is highly variable, presumably influenced by environmental stimulation. Neither follicles nor germinal centers were noted in NALT through the time period of the study.
BALT
BALT is a constitutive mucosal lymphoid tissue that lies adjacent to major airways in some mammalian species, including rats and rabbits, but not in humans and mice (Randall 2010). Microscopically similar tissue, known as inducible BALT (iBALT), is considered to be an ectopic (or tertiary) lymphoid tissue that may be found throughout chronically inflamed lungs of humans and mice. Both BALT and iBALT acquire antigens from the airways and initiate local immune responses in the lungs. Formation of iBALT in the lungs of mice has been shown to be dependent on T-cell production of interleukin 17 (IL-17), which promotes the lymphotoxin-α-independent expression of the chemokine CXCL13 (Randall 2010; Rangel-Moreno et al. 2011). CXCL13 is the ligand for CXCR5, which promotes homing of LTi cells to sites of future lymph node development (Rangel-Moreno et al. 2011). These observations suggest inflammation-related IL-17 produced by T cells is involved in the formation of iBALT via signaling pathways that are generally similar to those involved in the embryogenesis of lymph nodes.
Observations in the present study support classification of BALT as a constitutively expressed lymphoid tissue in rats, that is, secondary lymphoid tissue. BALT was present in the lungs of all rats at PND35 and PND42, though it varied in cellularity and overall prominence in individual animals.
Bone Marrow and Liver
In mice, hematopoietic stem cells (HSCs) migrate from the aorta-gonad-metanephros (AGM) region to the fetal liver by GD 10 (Cumano et al. 2001; Cumano and Godin 2001). In the fetal liver, the HSCs become progressively more differentiated/restricted stem cells and progenitor cells, which are defined by their ability to form colonies in vitro in response to specific cytokine stimulation. The fetal liver continues to be the primary hematopoietic organ throughout gestation, with emergence and rapid expansion of lineage-restricted progenitor cells for all types of leukocytes (Landreth 2002). However, this period of rapid expansion of progenitor cells is not accompanied by equivalent expansion of cells exhibiting the mature immunocompetent phenotype (Godin et al. 1999). Mature lymphocytes are not found in the developing liver until GD 18 in the mouse, after initiation of hematopoiesis in the embryonic bone marrow (Kincade 1981).
Observations in the present studies were consistent with a similar sequence of events in rats. The liver at GD 15 consisted of a sheet of hematopoietic elements intermixed with immature hepatocytes, with no indication of the lobular pattern that characterizes the liver of adult rats. At GD 20, the hematopoietic activity of the liver had subsided somewhat, but remained pronounced. During the period from GD 20 to PND 7, there was a rapid relocation of hematopoiesis from the liver to the bone marrow, resulting in substantial bone marrow cellularity and greatly reduced hepatic hematopoiesis at PND 7. Bone marrow cellularity was subjectively judged to be at adult level at PND 14.
Relationship of Immune System Function and Morphology
Current understanding of immune system development has come largely from experiments in mice. Less is known about the development of the immune system in rats (Holsapple, West, and Landreth 2003); however, the general sequence and timing of immune system development in both rats and humans appears to parallel studies in mice where data are available (Landreth 2002). Phenotypic analysis of rats on PND 10 indicated the absence of lymphocytic subsets, and histological analysis of the spleen revealed no germinal centers in either untreated rats or rats immunized with sheep erythrocytes (SRBC; Ladics et al. 2000). No anti-SRBC antibodies were detected in rats on PND 10 (ibid.). Phenotypic analysis of spleens from rats on PND 21 revealed B-cell populations that were comparable to adult rats, but overall T-cell population and T-cell subsets were reduced in comparison to adult rats (Ladics et al. 2000). Similar results were seen when KLH was used (Bunn, Ladics, et al. 2001). These published observations based on immunized rats contrast to some degree with morphological observations in the nonimmunized rats of the present studies, which suggest the T-cell component of the spleen develops before the mature B-cell component. In the present study, the spleen did not consistently demonstrate follicles and germinal centers until PND 35, and those structures were not judged to be at an adult level of morphological development until PND 42. The combination of published data and our present observations suggests the spleen is functionally mature and capable of responding to administered T-dependent antigens at approximately PND 21 but, in the protected environment of a typical toxicology laboratory, the lower level of antigenic stimulation results in a delay in the appearance of mature follicles and germinal centers.
It should be stressed that histologic indications of organ maturity, even those based on IHC identification of cell populations, do not necessarily imply functional maturity. Nevertheless, histopathologic evaluation of immune system organs is a useful scanning technique for detection of immunomodulation. Investigators must realize that histopathologic evaluation has limitations in the detection of xenobiotic-associated immunomodulation, particularly in the determination of pathogenesis of immune system alterations. Determination of immunopathologic mechanisms is typically based on a battery of analyses, which may or may not include histopathological evaluation. Similarly, histopathologic observations on immune system organs may be correlated with immunophenotyping data derived from flow cytometric analysis, but some level of discordance between the analyses is commonly encountered.
Information presented herein may serve as a general guideline for histologic evaluation of immune system organs in strains of rat other than the Sprague-Dawley, but detailed study of immune system organs from the various rat strains would be necessary to make specific comparisons. Several strain-related differences in immune system structure and responses have been reported (Bruder et al. 1999; Bunn, Ladics, et al. 2001; Bunn et al. 2001a), and undoubtedly additional differences remain unreported.
The reader is referred to published reviews for details of normal histologic features of immune system organs in the adult laboratory rat and for specific procedures used for histological detection of xenobiotic-associated changes: thymus (Elmore 2006e, 2012; Pearse 2006a, 2006b), spleen (Cesta 2006b; Elmore 2006d, 2012; Suttie 2006), lymph nodes (Elmore 2006c, 2006f, 2012; Willard-Mack 2006), MALT (Cesta 2006a; Elmore 2006a, 2012), and bone marrow (Elmore 2006b, 2012; Travlos 2006). The reader is referred to published reviews for details of liver involvement in immune responses (Parker and Picut 2005, 2012) as well as further information on immunobiology of MALT (Jung, Hugot, and Barreau 2010; Kuper et al. 2013).
Summary
Results of the present studies indicate the immune system organs of the rat mature at different rates in the postnatal period. Maturation of the primary immune system organs, that is, bone marrow and thymus, occurs prior to maturation of the secondary immune system organs. Bone marrow cellularity is at adult level by PND 14, and the thymus exhibits adult histomorphology by PND 14. Secondary immune system organs that have substantial exposure to environmental stimulants, for example, mesenteric lymph nodes and GALT, assume adult morphology early in postnatal life, while immune system organs in more protected locations (e.g., spleen and axillary lymph nodes) are slower to develop the histologic features of adult maturity. Though these slowly developing secondary immune system organs have histologic features of immaturity, as compared to the histologic features typically encountered in young adult rats, it should be noted the histologic features of immaturity may be related to a lower level of antigenic stimulation rather than differences in genetically programmed maturation. The secondary immune system organs that were classified as immature in the present study typically had all components of mature immune system organs except follicular germinal centers. Presence of germinal centers and plasmacytosis in lymph nodes were considered to be the result of antigenic stimulation and B-cell maturation, respectively, thus both would be directly affected by the level of stimulation of the lymph nodes.
Complex questions arise in classification and interpretation of histologic changes in juvenile rats that are unscheduled deaths, as study protocols typically do not provide age- and sex-matched control animals for comparison with unscheduled decedents. In these cases, histologic changes in immune system organs of decedent rats may be (a) normal for the age and state of development, (b) an indication of hindered organ development due to xenobiotic influences, (c) a manifestation of stress-related effects, or (d) some combination of these influences. In these situations, the pathologist must depend on personal knowledge and experience to distinguish between direct or indirect xenobiotic-related effects versus normal postnatal development, the latter possibly influenced by xenobiotic exposure. If extensive involvement in studies involving juvenile animals is anticipated, consideration should be given to accumulating a library of histologic specimens at various postnatal periods for comparison with tissues from decedent rats. This is an ideal use for scanned digital slides. Optionally, and perhaps ideally, study protocols for toxicology studies that involve juvenile animals would include additional naive control animals that could be euthanized concurrently with decedents, thus providing a biological control that would aid in differentiating normal postnatal organ development from xenobiotic influence.
Footnotes
Acknowledgments
The authors wish to thank Michelle Hackman and the DART team at WIL Research in Ashland, Ohio; Toni Carpenter and the necropsy team at WIL Research in Ashland, Ohio; and Sirena L. Hudgins, Tara McIver, and the histology team at WIL Research in Hillsborough, North Carolina.
Author Contribution
Authors contributed to conception or design (GP); data acquisition, analysis, or interpretation (GP, CP, JT, CS); drafting the manuscript (GP, CS); and critically revising the manuscript (GP, CP, JT). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was fully funded by WIL Research.
