Abstract
The Food and Drug Administration Animal Rule requires evaluation of cardiovascular and central nervous system (CNS) effects of new therapeutics. To characterize an adult and juvenile mouse model, neurobehavioral and cardiovascular effects and pathology of a single sublethal but toxic, 8 mg/kg, oral dose of potassium cyanide (KCN) for up to 41 days postdosing were investigated. This study describes the short- and long-term sensory, motor, cognitive, and behavioral changes associated with oral dosing of a sublethal but toxic dose of KCN utilizing functional observation battery and Tier II CNS testing in adult and juvenile mice of both sexes. Selected tissues (histopathology) were evaluated for changes associated with KCN exposure with special attention to brain regions. Telemetry (adult mice only) was used to evaluate cardiovascular and temperature changes. Neurobehavioral capacity, sensorimotor responsivity or spontaneous locomotor activity, and rectal temperature were significantly reduced in adult and juvenile mice at 30 minutes post-8 mg/kg KCN dose. Immediate effects of cyanide included bradycardia, adverse electrocardiogram arrhythmic events, hypotension, and hypothermia with recovery by approximately 1 hour for blood pressure and heart rate effects and by 2 hours for body temperature. Lesions consistent with hypoxia, such as mild acute tubular necrosis in the kidneys corticomedullary junction, were the only histopathological findings and occurred at a very low incidence. The mouse KCN intoxication model indicates rapid and completely reversible effects in adult and juvenile mice following a single oral 8 mg/kg dose. Neurobehavioral and cardiovascular measurements can be used in this animal model as a trigger for treatment.
Introduction
Cyanide (CN) is a rapidly lethal compound due to its inhibition of electron transport by binding to the iron of mitochondrial cytochrome C oxidase. 1 Initial effects of CN are on organ systems with the highest demand for energy, such as the central nervous, cardiovascular, and pulmonary systems. 2 A secondary effect of CN intoxication involves a hypotensive state with bradycardia, not solely explained as a sequelae of hypoxia.
Concerns of increased exposure to CN have increased in recent years due to its increased usage in industry, increased exposure from burning plastic products in fires, 3 -5 and homeland security assessments of the potential for CN usage as an intentional and malicious contaminant of food or water. 6
Animal models of CN toxicity are needed to assess the efficacy of medical countermeasures in preclinical development. Little information is available concerning characterization of CN intoxication in juvenile animal models. McMahon and Birnbaum reported age differences in rhodanese activity, a key enzyme in degradation of CN. 7 The aim of this study was to characterize the neurobehavioral and cardiovascular effects of CN in adult and juvenile mice. The Agency for Toxic Substances and Disease Registry Toxicological Profile for CN indicated that “no data were available for CN-induced neurotoxicity in humans following intermediate-duration exposures by any route, but a number of animal studies are available, none of which, however, systematically evaluated neurotoxicity using a neurobehavioral test battery.” 8(p17) This study was conducted at a single sublethal but toxic oral dose of potassium cyanide (KCN), 8 mg/kg body weight, 9 in order to systematically evaluate acute exposure neurotoxicity.
Materials and Methods
Animals
Adult CD-1 mice, 70 to 84 days of age at dosing, and juvenile CD-1 mice, 21 to 25 days of age at dosing, were obtained from Charles River Laboratories (Portage, Michigan site for all juvenile mice and some adult mice and Kingston, New York and Raleigh, North Carolina site for remaining adult mice). Both sexes of this strain of mouse achieved an adult size (minimum of 26 g) sufficient for implantation of telemetry probes. Mice were dosed as juveniles (approximately 21-25 days old) or as adults (approximately 12-14 weeks old). For juvenile mice, CD-1 dams with approximately 10 nursing pups were received at 10 days of age. Juvenile mice were weaned at 21 to 23 days of age and used at an age of approximately 21 to 25 days on study. All mice were quarantined for at least 10 days prior to use. Animal experiments adhered to the eighth edition of the Guide for the Care and Use of Laboratory Animals 10 and were conducted under a protocol approved by the Battelle Institutional Animal Care and Use Committee.
Chemicals
Potassium cyanide (CAS 151-50-8, purity of 99.8%) and tetrabutylammonium sulfate were obtained from Sigma-Aldrich, St Louis, Missouri.
Experimental Design
Dose volumes were based on individual body weights recorded within 1 day of dosing and were approximately 10 mL/kg body weight for adult mice and 12 mL/kg for juvenile mice. Dosing solution concentrations were adjusted to deliver the target doses.
Functional Observational Battery
The FOB is a series of observational tests designed to identify areas of neurobehavioral defect and can be used to select appropriate Tier II tests for additional testing. The FOB testing followed the procedure described by Moser et al 11 except it was modified for mice as detailed in the methods section. An observer recorded each rodent’s posture, presence of tremors, convulsive activity, and eyelid closure in the home cage. The rodent was then removed from the cage, and the observer evaluated the ease of removal and handling. The observer also performed a detailed observation regarding the general condition/appearance, salivation, piloerection, fur appearance, vocalization, respiration, lacrimation, and palpebral reflex responsivity. The rodent was then placed in an open-field observation chamber for approximately 2 minutes to assess locomotion, number of rearings, posture, tremors, convulsive activity, arousal, and gait. At the end of 2 minutes, the number of fecal pellets and urine pools on the flooring was recorded. Stereotypy, bizarre behavior, and vocalizations were also noted. Reflex testing followed the open-field test and consisted of recording each rodent’s responses to the approach of a blunt object, touch response of a blunt object to the rump, startle response, tail pinch, paw placement on a grid, and grip strength. Finally, a rectal temperature was taken.
Tier II Neurobehavioral Analyses
Tier II tests included quantitation of spontaneous locomotor activity (MotorMonitor; Kinder Scientific, Poway, California), equilibrium (Rotamex-5 Rotarod/Accelarod; Columbus Instruments, Columbus, Ohio), and anxiety (Elevated Plus Maze; Kinder Scientific). These tests followed the FOB (Tier I) tests; predose, 0.5 and 24 hours postdose; and 6, 13, 27, and 41 days postdose. A separate set of 10 male and 10 female animals were tested at each time point to minimize animal perturbation that could compromise the testing results. Separate cohorts also allowed for necropsy and histopathology of the same animals at each time point in order to compare neurobehavioral effects and histopathological findings at the same point in time.
MotorMonitor testing followed the procedure described by Loggi et al. 12 The MotorMonitor system (Kinder Scientific) consists of 4 separate open-field enclosures containing aligned photo beams to measure both horizontal and vertical planes. Over the course of 10 minutes, the MotorMonitor system measured total basic movements, ambulations, fine motor movements, rearings, distance/total time/rest/entries/pokes into the SmartFrame periphery (outer 50% of the apparatus grid), and distance/total time/rest/entries/pokes into the SmartFrame center (inner 50% of the apparatus grid).
Rotamex-5 RotaRod/Accelarod testing followed the procedure described by Gerald and Gupta. 13 The Rotamex-5 Rotarod/Accelarod apparatus (Columbus Instruments) was used to assess sensorimotor integrity and equilibrium by measuring the running duration at a fixed speed (20 revolutions/min) on the elevated rotating rod.
Elevated Plus testing followed the procedure described by Belzung and Griebel. 14 The Elevated Plus apparatus (Kinder Scientific) was used to quantitate subject anxiety. In this test, an animal was placed at the choice point of a “plus symbol” shaped apparatus that is elevated approximately 36 inches from the floor. It is the natural tendency of normal rodents to remain primarily in the 2 dark and enclosed areas (closed arms) of the Elevated Plus Maze as opposed to in the 2 more illuminated and open areas (open arms).
Cardiovascular Evaluation
Adult mice (approximately 12-14 weeks old at time of dosing) were surgically implanted with telemetry units (model No. HD-X11) by Data Sciences Incorporated, St Paul, Minnesota to continuously measure systemic blood pressure (systolic, diastolic, and mean), heart rate, ECG, and body temperature in adult mice for approximately 24 hours predose and for approximately 24 hours prior to and after each time point (0.5 and 24 hours and 6, 13, 27, and 41 days postdose). The predose baseline blood pressure, heart rate, and body temperature values were determined by taking a 2-hour average between 4:00 and 6:00
Electrocardiogram Evaluation
Electrocardiogram waveforms were evaluated in adult mice (approximately 12-14 weeks old at time of dosing) for rhythm and morphology by a veterinarian cardiologist. Time points evaluated for ECG analysis were the day prior to KCN dosing and at approximately 10 minutes prior to dosing and 1, 3, 5, 30, and 60 minutes and 4 and 24 hours following KCN dosing. Electrocardiogram waveform tracings were also evaluated prior to dosing on days 6, 13, 27, and 41 postdose at approximately 8:00 to 10:00
Histopathology
Selected tissue samples were fixed in formalin, embedded in paraffin and sectioned between 4 and 6 μ, and sections stained with hematoxylin and eosin for microscopic analysis. Tissues weighed and examined microscopically by a board-certified veterinary pathologist included heart, brain, liver, kidney, thyroid gland (postfix weight), and cauda epididymis (males). Microscopic analysis of the brain was performed on 5 distinct brain sections that included samples of the olfactory bulbs, cerebral cortex and midbrain (2 sections), cerebellum, and brain stem (2 sections).
Statistical Evaluation
Functional observation battery and CNS Tier II test end points were compared between KCN-treated and vehicle-treated groups at each collection time using statistical methods for continuous, categorical, or binary response variables (P ≤ 0.05). Statistical approaches were different depending on the type of variables being analyzed. Male and female mice were combined for these analyses.
Among the FOB variables, lines crossed, rearings, urine pools, and fecal pellets were characterized as count variables, rectal temperature was characterized as a continuous variable, vocalization and spontaneous vocalizations were characterized as yes/no variables, and the rest of the FOB variables were characterized as scoring variables, grading with qualitative scales. Among the CNS Tier II data, all entries variables and rearing for motor monitor were characterized as count variables, and the rest of the Tier II variables were regarded as continuous variables.
For scoring variables, a mean response model was used to predict the likelihood of observing a particular pattern of responses among the possible response categories, as a function of dose group. 15 A chi-square test was used to test the significant differences between KCN and vehicle groups.
For yes/no variables, a logistic regression model was used with dose group as a fixed effect. A chi-square test was used to test significant differences between KCN- and vehicle-treated groups.
For variables classified as having continuous responses, a 1-way fixed effects analysis of variance (ANOVA) model was fitted to the observed responses. 16 The model included dose group as a fixed effect and assumed the residual error (ie, difference in observed and model-predicted values) followed a normal distribution with zero mean and a constant variance. Chi-square tests were used to test significant differences between KCN- and vehicle-treated groups.
For variables recorded as count data, square root transformation (
The percentages of responses within each category and each dose group were tabulated for the scoring variable and yes/no variables. Means and standard errors of means of the continuous responses were calculated for each dose group. All data were analyzed using version 12 (Release 12.1) of STATA.
Systemic blood pressure (systolic, diastolic, and mean), heart rate, and body temperature data collected by telemetry were divided into consecutive time intervals of specific length and averaged. On the day of KCN dosing, telemetry data were averaged every 1 minute for the first 30 minutes postdose, every 5 minutes from 30 to 120 minutes postdose, every 30 minutes from 120 to 360 minutes postdose, and hourly from 6 to 24 hours postdose. On 6, 13, 27, and 41 days postdose, only a single 1-hour average was analyzed during the approximate time as the 1-hour postdose time period on the day of dosing. Data collected within these time intervals were averaged and “baseline adjusted” by subtracting the animal’s corresponding predosing baseline average. The baseline-adjusted postdose group averages were analyzed using a repeated measures mixed effect ANOVA model that included dose level, time after dosing, and the interaction of time after dosing, with dose level as fixed effects and animal as a random effect. The ANOVA model tested for significant differences between CN-treated and control groups baseline-adjusted averages at each time interval following dosing. Male and female mice were combined for these analyses.
Results
The dose level was selected for this study based on toxicity studies conducted in this laboratory. 9 Based on these toxicity studies, the median lethal doses (LD50s) for orally administered KCN in mice were determined to be 10.9 mg/kg ± 0.5 mg/kg and 11.8 mg/kg ± 0.6 mg/kg KCN in juvenile and adult male mice, respectively, and 9.9 mg/kg ± 0.4 mg/kg and 11.0 mg/kg ± 0.7 mg/kg in juvenile and adult female mice, respectively. The oral LD50s were similar to those reported for rats and mice in previous studies. 8 A single oral dose that would be toxic, but <1% lethal (8 mg KCN/kg body weight), was chosen based on the results of the previous study. 9
Neurobehavioral Assessment
Oral dosing of adult or juvenile mice with 8 mg/kg KCN resulted in statistically significant reductions in a number of FOB measures of neurobehavioral capacity, generally involving reduced sensorimotor responsivity, including spontaneous locomotor activity (Tables 1 –4). Data from male and female mice were combined for analyses of each age-group of mice.
Incidence of Abnormal Findings Using the Home Cage FOB for Adult and Juvenile Mice.a

Abbreviations: FOB, functional observation battery; KCN, potassium cyanide.
aEach mouse received a single oral dose of 8 mg/kg KCN in water or the vehicle control (water) on day 1. Home cage FOB tests were conducted on 10 male and 10 female adult mice or 9 male and 10 female juvenile mice at various times postdosing. Juvenile animal 907/male died early on day 1 of the study resulting in lower n value (9) for juvenile male mice. Abnormal findings and the number of findings (in parenthesis) are indicated by an entry of Adult (adult mice) or Juv (juvenile mice) in the cell. Data from male and female mice were combined for each age group. There were no abnormal findings in the home cage on day 14 (13 days postdose), so the data are not shown.
bA significant difference from the control group.
Incidence of Abnormal Findings Using the Open Field FOB for Adult and Juvenile Mice.a
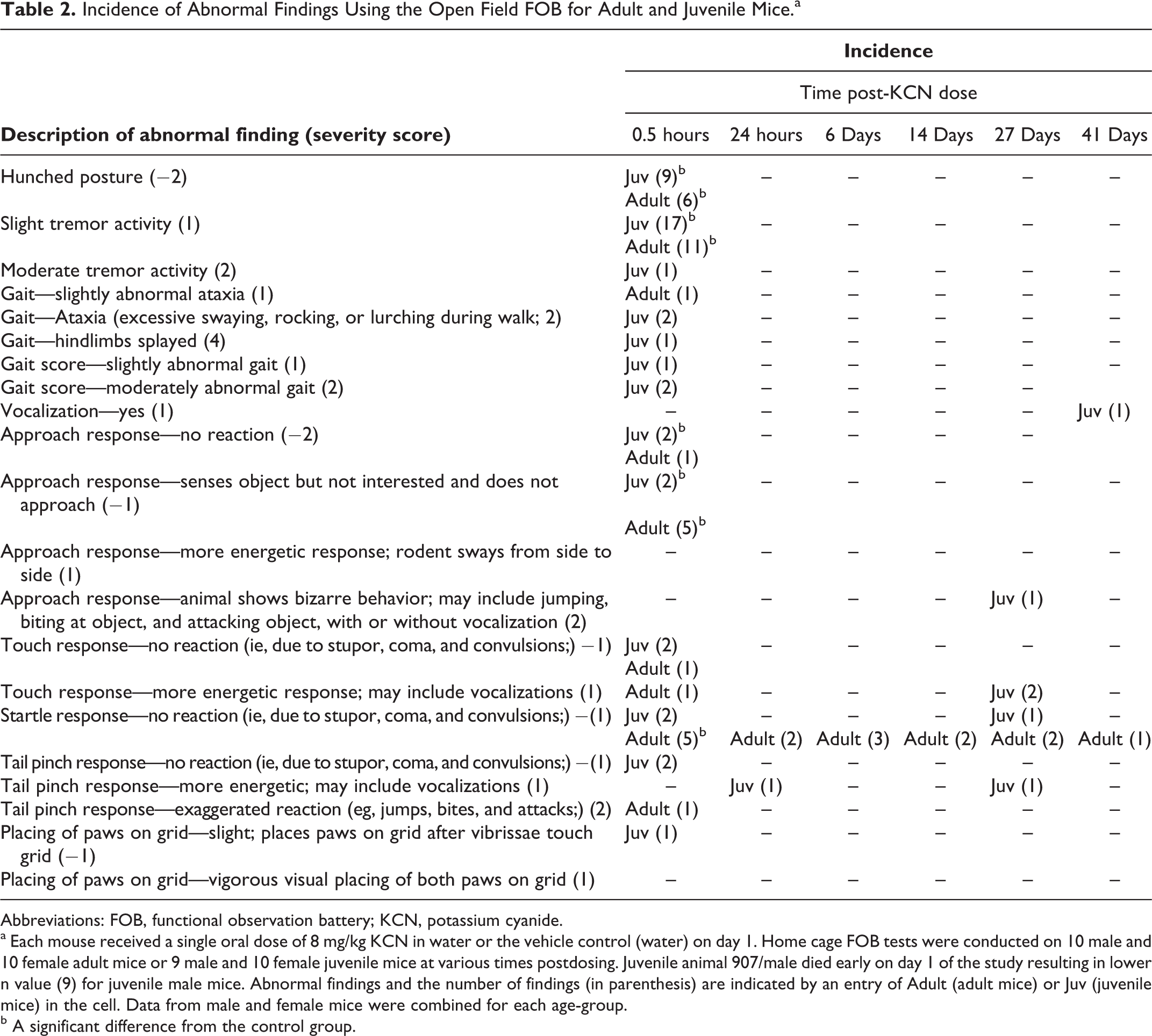
Abbreviations: FOB, functional observation battery; KCN, potassium cyanide.
aEach mouse received a single oral dose of 8 mg/kg KCN in water or the vehicle control (water) on day 1. Home cage FOB tests were conducted on 10 male and 10 female adult mice or 9 male and 10 female juvenile mice at various times postdosing. Juvenile animal 907/male died early on day 1 of the study resulting in lower n value (9) for juvenile male mice. Abnormal findings and the number of findings (in parenthesis) are indicated by an entry of Adult (adult mice) or Juv (juvenile mice) in the cell. Data from male and female mice were combined for each age-group.
bA significant difference from the control group.
Adult Mice—Summary of the FOB Data (Open Field Testing at 8 mg/kg KCN; n = 10 Male and 10 Female Mice).
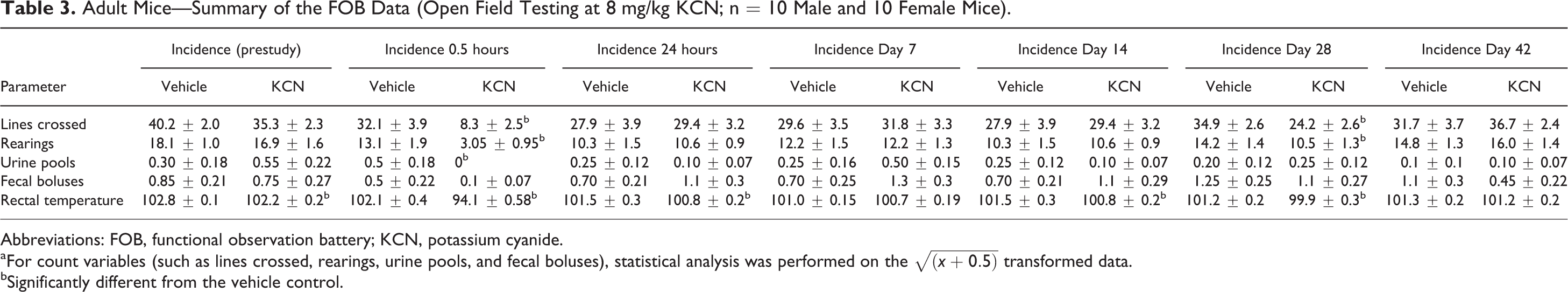
Abbreviations: FOB, functional observation battery; KCN, potassium cyanide.
aFor count variables (such as lines crossed, rearings, urine pools, and fecal boluses), statistical analysis was performed on the
bSignificantly different from the vehicle control.
Juvenile Mice—Summary of the FOB Data (Open Field Testing at 8 mg/kg KCN; n = 10 Male and 10 Female Mice).
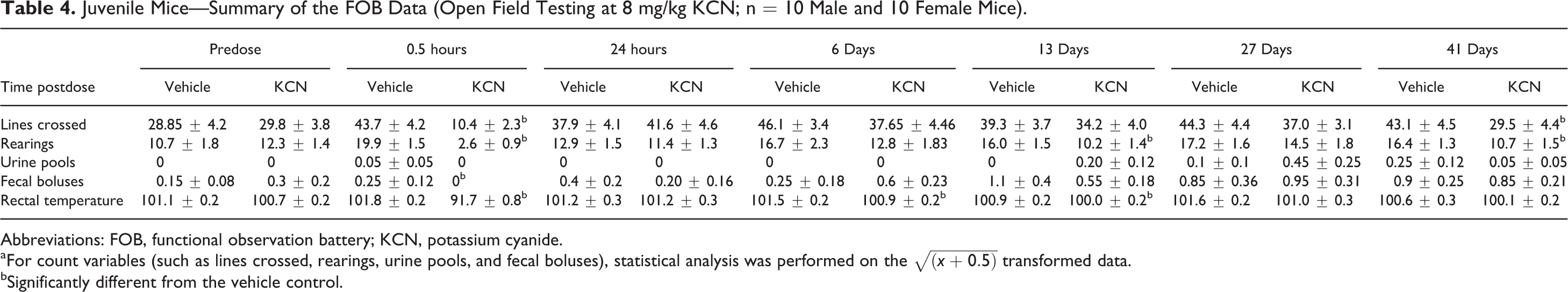
Abbreviations: FOB, functional observation battery; KCN, potassium cyanide.
aFor count variables (such as lines crossed, rearings, urine pools, and fecal boluses), statistical analysis was performed on the
bSignificantly different from the vehicle control.
Tier II neurobehavioral tests similarly quantified statistically significant reductions in a large number of measures of spontaneous locomotor activity compared to vehicle control mice of the same age in both adult and juvenile mice at approximately 30 minutes postdose using the Kinder Scientific Motor Monitor apparatus (Table 5). Data from male and female mice were combined for analyses of each age-group of mice. There were no statistically significant differences between dosed and control groups on the Elevated Plus Maze test that suggested treatment-related increased or decreased levels of anxiety following KCN dosing. At approximately 30 minutes postdose, there was no significant difference between the KCN and the control groups on the Rotamex-5 Rotarod indicating a lack of KCN-related changes in equilibrium and neuromuscular integrity (Table 6).
Motor Monitor Open Field Test Results at 0.5 Hours Postdose.
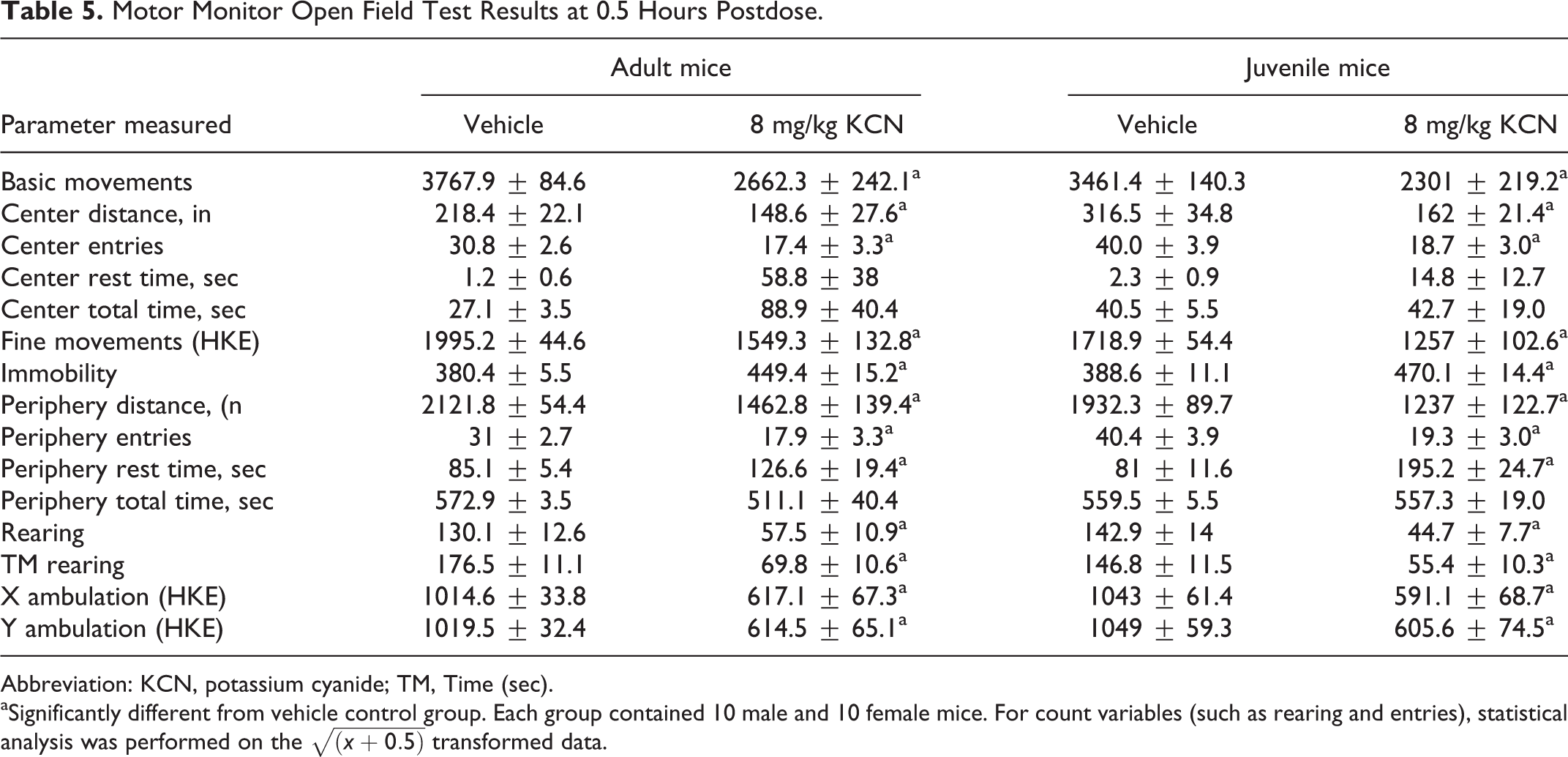
Abbreviation: KCN, potassium cyanide; TM, Time (sec).
aSignificantly different from vehicle control group. Each group contained 10 male and 10 female mice. For count variables (such as rearing and entries), statistical analysis was performed on the
Summary for Tier II Rota Rod Data (sec).a
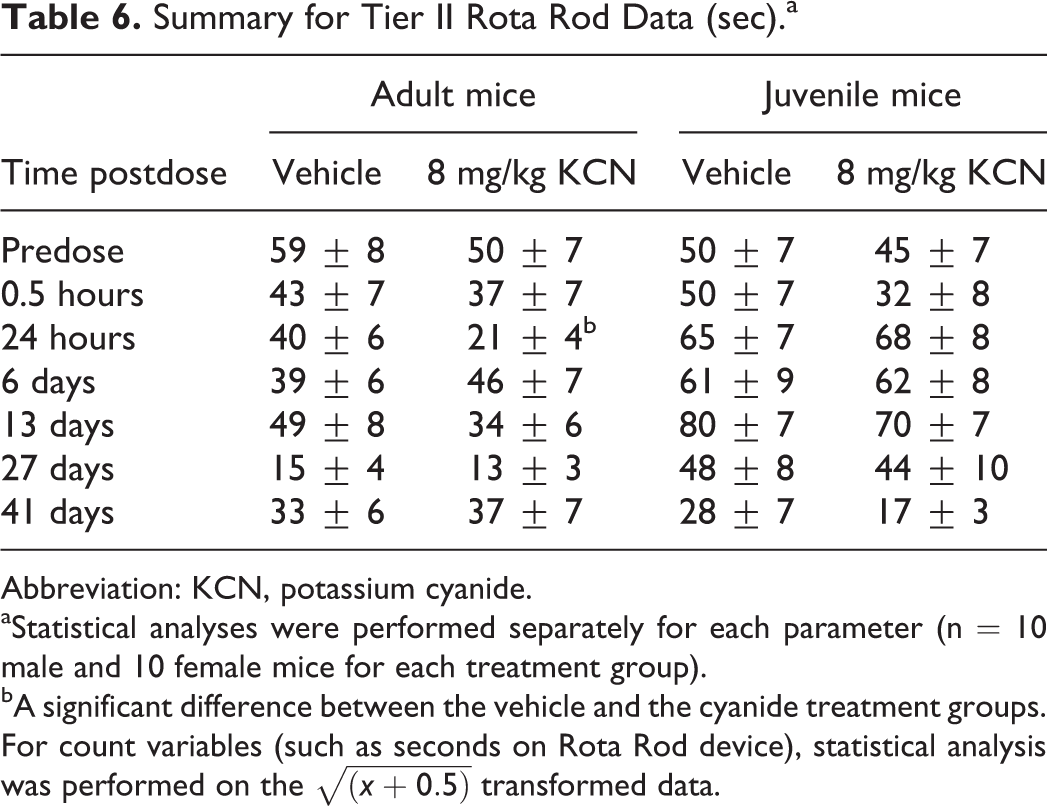
Abbreviation: KCN, potassium cyanide.
aStatistical analyses were performed separately for each parameter (n = 10 male and 10 female mice for each treatment group).
bA significant difference between the vehicle and the cyanide treatment groups. For count variables (such as seconds on Rota Rod device), statistical analysis was performed on the
A reduction in rectal temperature was observed for both adult and juvenile mice at 0.5 hours postdosing. A large reduction in mean rectal temperature was recorded in juvenile mice at approximately 0.5 hours postdosing with 8 mg/kg KCN; tremors were also observed at this time point.
Recovery from KCN-induced deficits, observed at 30 minutes postdose, appeared to resolve rapidly and nearly completely, in both adult and juvenile mice, as demonstrated by a lack of effect in FOB (Tables 1-4) and Tier II testing (Tables 6 and 7) at approximately 24 hours postdose. One exception to this was the statistically significant decrease in “time to leave the rotating rod” in the Rotamex-5 Rotarod testing for KCN-dosed adult mice (Table 6). These data would suggest a residual effect of the KCN exposure on equilibrium and/or neuromuscular integrity that was not identified at approximately 30 minutes postdose or in subsequent Rotarod tests on days 6, 13, 27, or 41 postdose. Functional observation battery and Tier II tests identified no neurobehavioral deficits or minimal deficits that were not considered to be of toxicological significance from day 6 to 41 postdose.
Motor Monitor Open Field Test Results at 24 Hours Postdose.
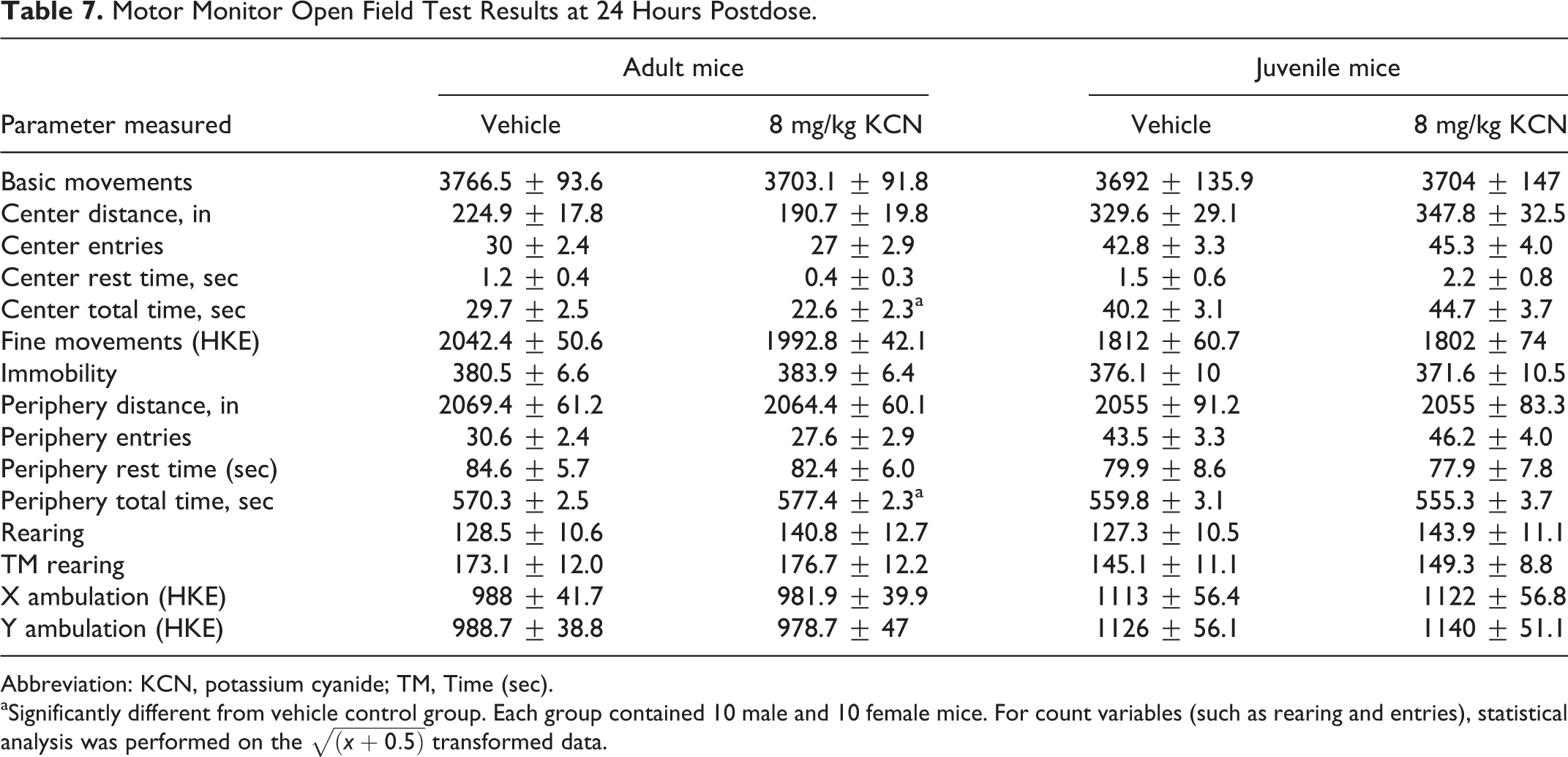
Abbreviation: KCN, potassium cyanide; TM, Time (sec).
aSignificantly different from vehicle control group. Each group contained 10 male and 10 female mice. For count variables (such as rearing and entries), statistical analysis was performed on the
Cardiovascular Assessment, Body Temperature, and ECG Waveform
Telemetry measurements were conducted only in adult mice due to size constraints on implanting telemetry probes. Data from male and female mice were combined for analyses. Following an initial rapid increase and decline, the telemetry-monitored control mice demonstrated an elevation in systolic, diastolic, and mean arterial blood pressure and heart rate for approximately 30 minutes postdose, followed by a return to baseline levels (Figures 1 and 2). The maximum increases from baseline, after vehicle dosing, were approximately 25 mm Hg mean arterial pressure and 128 bpm for heart rate at approximately 1 minute postdose. Since the control vehicle was sterile water, the elevation in these parameters was considered an effect of dose administration and/or dosing procedures and not vehicle related.
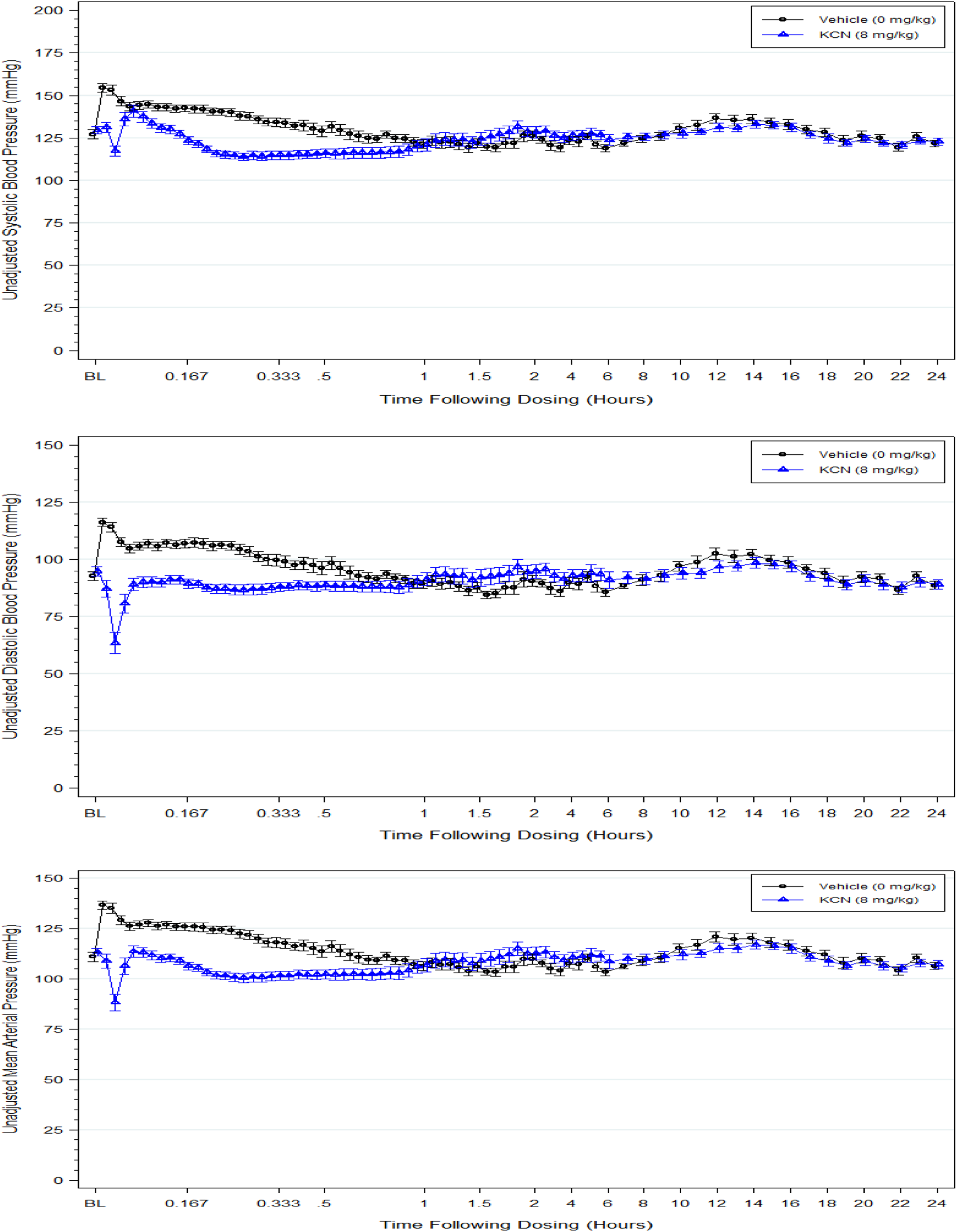
Systolic, diastolic, and mean arterial blood pressure (mm Hg). The data are the mean of 10 male and 10 female 8 mg/kg KCN-treated adult mice (triangle symbol) or 9 male and 9 female vehicle-treated mice (circle symbol) from predose (baseline) to 24-hour postdose.
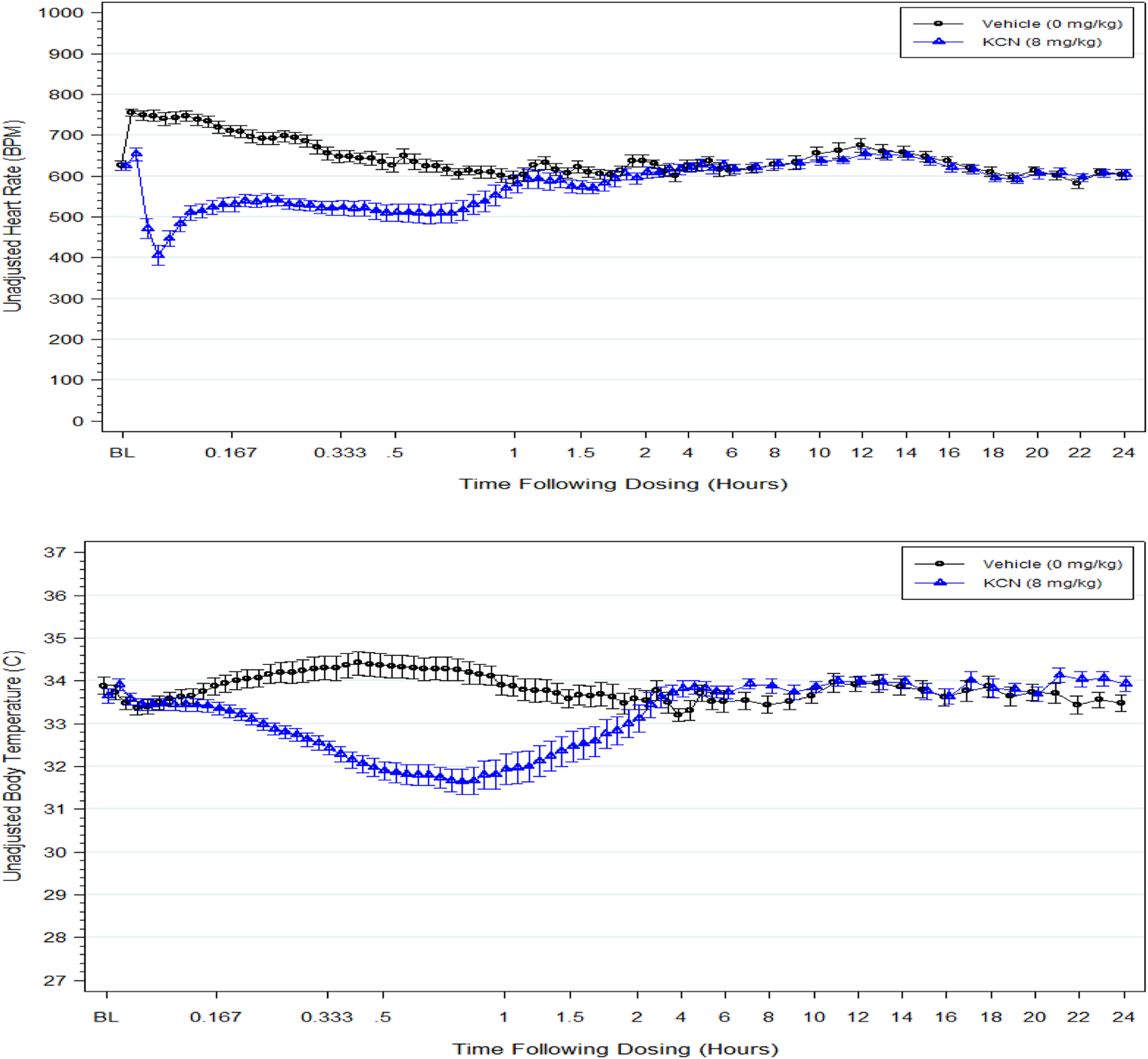
Heart rate and body temperature. The data are the mean of 10 male and 10 female 8 mg/kg KCN-treated mice (triangle symbol) or 9 male and 9 female vehicle-treated adult mice (circle symbol) from predose (baseline) to 24-hour postdose.
An 8 mg/kg KCN dose elicited a rapid decline in systolic, diastolic, and mean arterial blood pressure and heart rate in the first 2 minutes postdose (Figures 1 and 2). All blood pressure parameters were statistically significantly reduced within the first minute postdose and then rebounded but remained significantly lower than the pressures in vehicle control animals until approximately 35 minutes postdose. An exception to this was the systolic blood pressure that rebounded for a short period at approximately 5 minutes postdose to a pressure similar to that of the vehicle control; subsequently, the systolic blood pressure decreased again and remained less than that of the control group up to 35 minutes postdose. Heart rate was reduced from a baseline value of approximately 625 bpm to a minimum of approximately 218 bpm within the first 3 minutes of KCN dosing followed by a slight increase that did not recover to control group heart rates until approximately 100 minutes postdose. The difference between the heart rates of KCN-treated and vehicle-treated mice was statistically significant for 40 minutes postdose. Following recovery (approximately 1 hour postdose), there were no significant differences in blood pressure parameters or heart rate for the remainder of the study up to 41 days postdosing.
A slight increase in body temperature was observed over the first hour postdose in the control group mice returning to baseline temperature within 1 hour postdose. A statistically significant decline in body temperature occurred in KCN-treated mice at approximately 13 minutes postdose and reached a maximum decline of approximately −2°C by 45 minutes postdose. Body temperature remained statistically lower than that of the control group through 100 minutes postdose and did not recover to levels observed in the control group until approximately 2 hours postdose. The KCN-treated group had a small (maximum of 0.9°C), but statistically significant, elevation in temperature compared to the control group at approximately 4 to 4.5, 7 to 8, and 21 to 24 hours postdose. No significant differences were found in body temperature between groups on 6, 13, 27, or 41 days postdose.
Electrocardiogram Evaluation
Electrocardiogram waveforms were evaluated in 10 male and 10 female adult mice. Two of 20 mice, both females, in the control group experienced altered ECG signals. One mouse experienced atrioventricular (AV) block 1 minute after being dosed with the control and another had preexisting tachycardia evident 24 hours prior to control dosing. This same animal also experienced alternans 41days postdose. These events appear to be preexisting conditions in the mice and not related to the dosing event or the control article. In the 8 mg/kg KCN-dosed group, 4 (2 males and 2 females) of 20 adult mice experienced ECG events within the first 5 minutes that ranged from bradycardia in 2 animals (1 male and 1 female), a left ventricular depolarization in 1 animal (male), and a large Ta wave in another (male). Effects were also seen at the 30-minute postdose time point which included PR prolongation in 1 female mouse experiencing earlier issues, alternans in another female mouse, and Ta prolongation and enhancement in 1 female mouse. The bradycardia and left ventricular depolarization as well as the PR and Ta prolongation and enhancement appear to be effects of KCN, which occurred in a small percentage of exposed animals and are most likely due to effects on the sinoatrial (SA) node due either to altered autonomic traffic or an effect of CN on ion channels specific for phase 4 depolarization of the SA node.
Gross Necropsy and Histopathology
Only 1 male juvenile mouse died following a single oral 8 mg/kg KCN dose; death occurred on the day of dosing. No gross or microscopic lesions were evident in this animal. Within adult or juvenile age-groups, of either sex, there were no notable differences in terminal body weights or organ weights between animals administered 8 mg/kg KCN versus control at any time point, up to 41 days postdose, aside from minor changes considered to be spurious based on their presence in only 1 sex or at only 1 time point and a lack of correlation with any microscopic lesions. Gross lesions were observed in 4 mice as follows: mandibular lymph node enlargement in 1 control adult male mouse (corresponding to lymphoid hyperplasia), a small left kidney in control adult female mouse (corresponding to atrophy), jejunal diverticulum in 1 KCN-dosed adult female mouse, and a skin crust in 1 control juvenile female mouse (corresponding to inflammation). All of these lesions are deemed spontaneous incidental findings.
No microscopic lesions were definitively attributed to KCN exposure in this study. However, 3 adult mice had microscopic lesions with uncertain, and potential, relationship with KCN exposure. Two mice (1 male and 1 female) euthanized 24 hours post-KCN exposure had minimal to mild acute tubular necrosis in the kidneys, most notably near the corticomedullary junction. This lesion can be caused by hypoxia. Another female mouse, euthanized 30 minutes post-KCN exposure, had spongiosis (vacuolization of the neuropil) in the deep cerebral cortex in the most rostral brain section that was processed. While many CD-1 mice had similar spongiosis in the hippocampus as a background lesion unrelated to KCN exposure, this mouse was the only 1 found to have it in the cerebral cortex.
Discussion
Cyanide is a potent, systemic chemical asphyxiant. It interferes with the normal availability of oxygen and cellular utilization of available oxygen, particularly affecting those organ systems most sensitive to low oxygen levels: the central nervous system (CNS), the cardiovascular system, and the pulmonary system. 2,8,17 Significant effects occur in the brain following acute, high concentration, and CN exposure. 8 Central nervous system effects may include convulsions, coma, and death due to the toxicity at centers controlling respiration. Humans, surviving lethal doses of CN, may have delayed recovery displaying increased incidences of severe Parkinsion-like symptoms or milder symptoms such as tremor and headache. Extensive degenerative changes in the CNS have been produced experimentally in the brain of dogs and rats exposed to high levels of CN by inhalation. 8
A well-characterized model of oral KCN intoxication is needed to test new therapeutics against KCN intoxication. The purpose of this study was to characterize short- and long-term cardiovascular and CNS toxicity of a single sublethal oral dose of KCN in adult and juvenile mice using a neurobehavioral test battery and telemetry.
Neurobehavioral and cardiovascular end points were measured at a sublethal, but toxic, dose of KCN in mice, 8.0 mg/kg. The neurobehavioral effects observed in the present study suggest that 8 mg/kg KCN induced systemic consequences on critical bodily functions that led to statistically significant reductions in a number of measures of neurobehavioral capacity, generally involving reduced sensorimotor responsivity, including spontaneous locomotor activity in both adult and juvenile mice. The effects are also consistent with those observed in rodents during general malaise and are generally consistent with effects of nonlethal CN exposures reported in humans. 2,8,17
Neurobehavioral effects measured using FOB and Tier II test methods occurred rapidly, within approximately 30 minutes postdose, followed by a rapid recovery. These acute neurobehavioral changes included reductions in ambulatory movements, fine motor movements, rearing and rearing time, and entries into various portions of the open field, as well as increased immobility and resting. There were no statistically significant differences in anxiety (elevated maze test) or equilibrium and neuromuscular integrity (rotarod test) postdose.
Although a statistical comparison of FOB data in adult versus juvenile mice was not conducted, there appeared to be an increased incidence of hunched posture and slight tremor in juvenile versus adult mice in both home cage and open-field tests at 0.5 hours postdose. This is consistent with previous oral LD50 studies in adult and juvenile mice 9 and the lower rhodanese activity, a key enzyme in degradation of CN, reported in younger mice. 7 The KCN-induced changes in spontaneous locomotor activity (Table 5) and equilibrium and neuromuscular integrity (Table 6), observed at 0.5 hours postdose, were similar for both adult and juvenile mice.
At approximately 24 hours post-KCN dosing, there were virtually no neurobehavioral differences compared to a vehicle control group, except for a statistically significant decrease in the time on the rotating rod for adult, but not juvenile, mice, suggesting a residual effect of KCN exposure on equilibrium and/or neuromuscular integrity that was not identified at 0.5 hours postdose or in subsequent rotarod tests at 6, 13, 27, or 41 days postdose. From 6 to 41 days postdose, no significant neurobehavioral deficits were identified.
Heart rate and systolic, diastolic, and mean arterial pressure were reduced immediately after dosing adult mice with 8 mg/kg of KCN. Due to their small size, telemetry could not be conducted in juvenile mice. In addition, body temperature decreased 10 minutes after KCN dosing. The KCN-induced reduction in heart rate is likely due to activation of the parasympathetic system 18 and/or direct effects on the SA node due to the lack of oxygen. 19 The reduction in heart rate would lower cardiac output and reduce mean arterial pressure. In addition, KCN may directly affect contractility and vascular tone. 20 Hypothermia occurred after the effects on heart rate and blood pressure and remained lowered after blood pressure and heart rate had recovered. The change in body temperature may stem from systemic inhibition of metabolism, reduced blood flow due to hypotension, or as a result of lack of mobility in the KCN-exposed mice. The nonlethal dose of 8 mg/kg KCN was sufficient to cause immediate effects relating to bradycardia, hypotension, and hypothermia, with recovery of cardiovascular effects approximately 1 hour and body temperature approximately 2 hours postdose. There were no chronic effects of 8 mg/kg KCN on any of these parameters. These studies were generally consistent with effects of nonlethal CN exposures reported in humans. 2,8,17
In conclusion, the results presented in this article provide thorough descriptions of adult and juvenile mouse models of KCN intoxication that can be used to screen or conduct preclinical efficacy studies of potential countermeasures. The model description indicates that the effects of KCN are rapid and, at a dose of 8 mg/kg, completely reversible in surviving mice. This is a toxic dose and is close to the LD50 dose of 10.4 or 11.0 mg/kg for juvenile and adult mice, respectively. Based on the rapid toxicity observed, treatment would have to be initiated quickly following exposures for optimal results. Neurobehavioral and cardiovascular end points can clearly be used to monitor toxicity of CN real time to use as a trigger for treatment in the efficacy studies.
Footnotes
Author Contribution
Michael A. Hawk contributed to design, acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Glenn D. Ritchie contributed to design, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Kim A. Henderson contributed to design, acquisition, and analysis and drafted the manuscript. Katherine A. B. Knostman contributed to design, acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Brian M. Roche contributed to design, analysis, and interpretation and critically revised the manuscript. Zhenxu J. Ma contributed to analysis and drafted the manuscript. Claire M. Matthews contributed to design, analysis, and interpretation and drafted the manuscript. Carol L. Sabourin contributed to conception and critically revised the manuscript. Edward J. Wakayama contributed to conception and design and critically revised the manuscript. Patrick J. Sabourin contributed to conception and design, analysis and interpretation; drafted the manuscript; and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has been funded in whole or in part with Federal funds from Biomedical Advanced Research and Development Authority, Department of Health and Human Services, under HHSO1002011000005I/Task Order HHSO10033006T.
