Abstract
Rabbits are frequently used in studies assessing the toxicity of ophthalmic drugs; however, the postnatal histological changes that occur in the rabbit eye have not been fully described. To characterize postnatal ocular development in white rabbits, a histological investigation of the eyes and eyelids was sequentially performed between postnatal days (PNDs) 1 and 42. The eyes opened during PNDs10 to 12. Significant changes prior to eyelid opening included the proliferation of uveal and optic nerve cells, regression of the lenticular vasculature, and thinning of the retina with a decreasing number of retinal cells. After eyelid opening, several significant changes occurred in the anterior segment, including thickening of the cornea and the development of lacrimation-related tissues in the eyelid and conjunctiva. Additionally, the differentiation of retinal layer-derived cells and optic nerve thickening occurred. The lens size continued to increase throughout the postnatal period. The histological structure of the eyes and eyelids was nearly mature by PNDs28 to 42. This study characterizes the postnatal changes in the histological features of the eyes in juvenile white rabbits, providing fundamental knowledge on the appropriate design of histological studies of the eyes in juvenile rabbits, particularly ophthalmic drug evaluations.
Keywords
Introduction
Rabbits are commonly used experimental animals for the preclinical safety assessment of ophthalmic drugs. 1 –3 Their conjunctival sac is relatively large, and drug substances can thus be easily applied to the eyes in appropriate volumes. Rabbit eyes are also larger than those of mice and rats, facilitating the evaluation of ocular irritation potentials. Evaluation of the safety of drug administration on immature ocular tissues has been performed in several studies using juvenile rabbits. These have included juvenile rabbit toxicity studies examining the effects of topical ocular instillation of chloroquine or amiodarone, 4 or the intravitreal administration of bevacizumab. 5,6 A knowledge of comparative postnatal morphological characteristics is important to inform the choice of the age of animals used in juvenile toxicity studies for the assessment of risk in pediatrics. In addition, the study pathologists engaged in juvenile toxicity studies need to understand the normal histological development of the eye, the identification of time-sensitive points in development, and the effects on tissue differentiation and development exerted by the administration of pharmaceutical drugs. Several reports have described the postnatal development of the rabbit cornea, 7,8 ciliary body, 9 and retina. 10 More recently, the histological features of postnatal ocular development in New Zealand white rabbits between gestational day 13 to postnatal day (PND) 14 was investigated. 11 However, despite the inclusion of figures illustrating ocular histological development in that article, there is still insufficient information available to fully inform the histopathological examination of ocular toxicity studies in juvenile rabbits. Currently, the histological characteristics of postnatal ocular development are widely dispersed in these literature, meaning that there is limited information available to support the design of, and histopathological examination of ocular toxicity studies in juvenile rabbits. Therefore, there is a need to acquire further information regarding postnatal ocular morphological development, including sequential developmental changes, and to present images to illustrate this development.
In the past 2 decades, the regulatory guidance regarding pediatric drug use has been revised, along with the guidance describing the necessity of toxicological evaluation of these drugs using juvenile animals. In 2000, regulatory guidance has been issued by the Food and Drug Administration (FDA) for assessing the safety of drugs that may need to be administered to children. This guidance refers to the requirement for juvenile animal studies, and the pharmaceutical industry should consider the necessity of conducting toxicity studies using juvenile animals on a case-by-case basis according to the concerns regarding drug-induced postnatal developmental toxicity. 12 Guidance documents regarding conducting nonclinical toxicity studies using juvenile animals have been issued by the FDA, 13 the European Medicines Agency, 14 and the Ministry of Health, Labour, and Welfare in Japan 15 in 2006, 2008, and 2010, respectively. In these guidelines, juvenile toxicity studies are usually required as part of a pediatric safety assessment for any new drug application unless a waiver has been granted. 16 –18 When juvenile toxicity studies are designed, it is necessary to use information regarding the timing of maturation in that species, including organ/tissue development, in order to provide a rationale for the timing of dosing. In addition, knowledge of the normal histology of immature organs/tissues and the histological characteristics at each age is required for the study pathologists. Considering the current regulatory requirements, and the extent of scientific information now available, it is necessary to conduct further histological studies of postnatal ocular development in rabbits in order to elucidate the normal histological features at each age and thus support nonclinical toxicity studies. The purpose of this study was to characterize the histological features of postnatal ocular development in juvenile Japanese white rabbits, in order to inform the design and performance of juvenile ocular toxicity studies using rabbits.
Materials and Methods
Animals
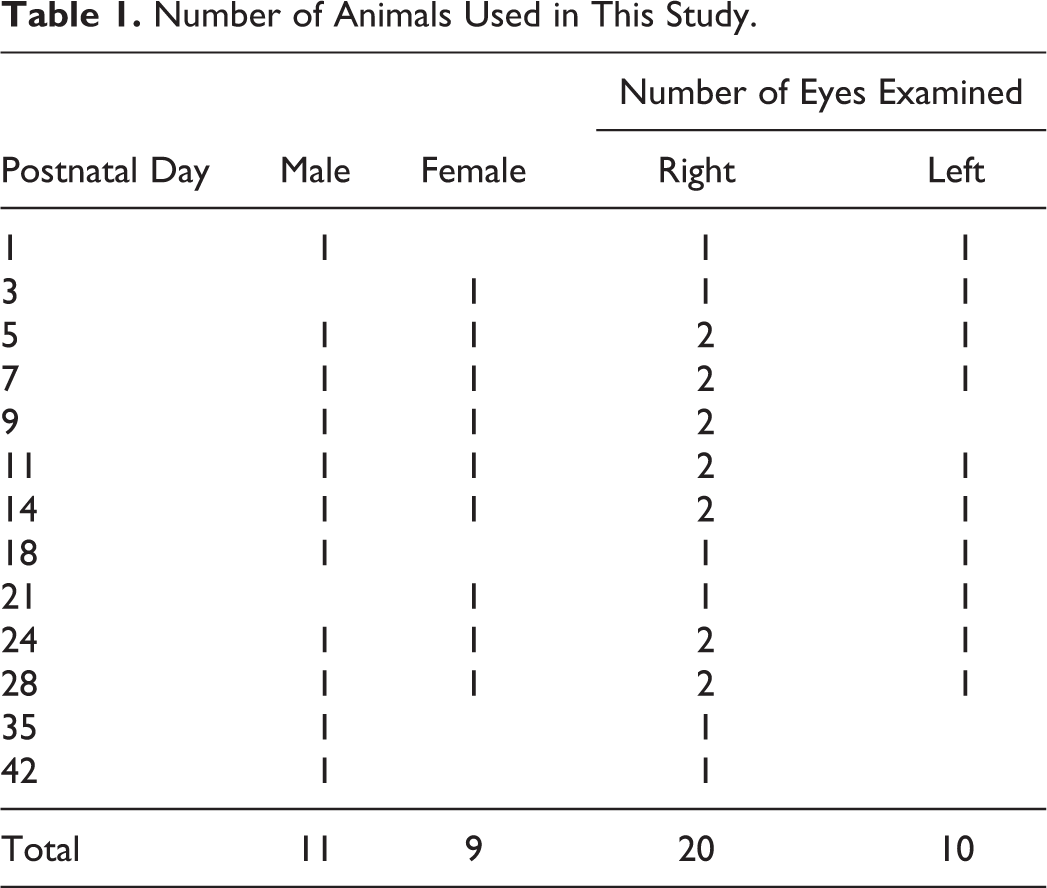
Eleven male and 9 female Japanese white rabbit (Kbl: JW) neonates/kits were used in this study (Table 1). The dams of the experimental animals were specific pathogen-free Kbl: JW. The neonates were either purchased from Kitayama Labes Co, Ltd (Nagano, Japan) or were delivered at the animal facility of Senju Pharmaceutical Co, Ltd by a pregnant female purchased from Kitayama Labes Co, Ltd. The neonates/kits were housed in plastic cages with their lactating dams, at 22°C ± 3°C and 55% ± 10% relative humidity with a 12/12 hour light/dark cycle in conventional animal rooms. Cage-side observation to monitor the health of the animals was performed every day. The dams were fed a commercial diet (Lab R stock; Nosan Corporation, Tokyo, Japan) once daily, and all animals were supplied with tap water ad libitum. A wooden box (Kitayama Labes Co, Ltd) was placed in each cage as a suitable location for suckling. Treatment and care of the animals were conducted according to standard procedures and the housing conditions in the facility were fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. All procedures were conducted in accordance with the guidelines for animal experimentation at Senju Pharmaceutical Co, Ltd, and the protocol was reviewed by the Institutional Animal Care and Use Committee.
Number of Animals Used in This Study.
Experimental Procedures
Clinically normal rabbit neonates and kits were randomly selected, and each euthanized in turn at PND1, 3, 5, 7, 9, 11, 14, 18, 21, 24, 28, 35, or 42. Before killing, rabbit neonates and kits were evaluated as whether their eyelids opened through gross observation. On the last day, they were separated from their dams, anesthetized using sevoflurane (Mylan Inc, Canonsburg, Pennsylvania) as an inhalational agent, and were subsequently euthanized by exsanguination through the abdominal aorta.
The number of eyes examined in each age is shown in Table 1. After euthanasia, the eyes and eyelids were removed by enucleation and were immersed in Davidson fixative (33% ethanol [vol/vol], 22% formalin [vol/vol], and 11% glacial acetic acid [vol/vol]). The caudal aspects of the eyes were cut to ensure rapid penetration of fixative. The next day, the fixative was replaced with 10% neutral-buffered formalin (FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan). From PND11 (immediately after eyelid opening), the eyelids were separated from the eye and were immersed in the 10% neutral-buffered formalin fixative. Following fixation, the eyes and eyelids were processed and embedded in paraffin, and sagittal sections of approximately 3 µm thickness were obtained at the level of the optic nerve head (Figure 1). These sections were stained with hematoxylin and eosin (H&E). The sections of eyes obtained at PND1, 3, 5, 7, 9, 11, 14, 18, 21, 24, and 28 were also stained with periodic acid-Schiff (PAS) staining. The H&E and PAS-stained preparations were observed using a light microscope. In addition, terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) was also performed to detect apoptosis in retinal cells. Briefly, the paraffin-embedded sagittal sections of the eyes at PND1, 3, 5, 7, 9, 11, 14, 18, 21, 24, and 28 were prepared and then stained using the Apoptosis in situ Detection Kit Wako (FUJIFILM Wako Pure Chemical Corporation) according to the manufacturer’s instructions. Thereafter, the sections were counterstained with hematoxylin to identify the retinal morphology. Terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling-positive cells were identified under light microscopy.
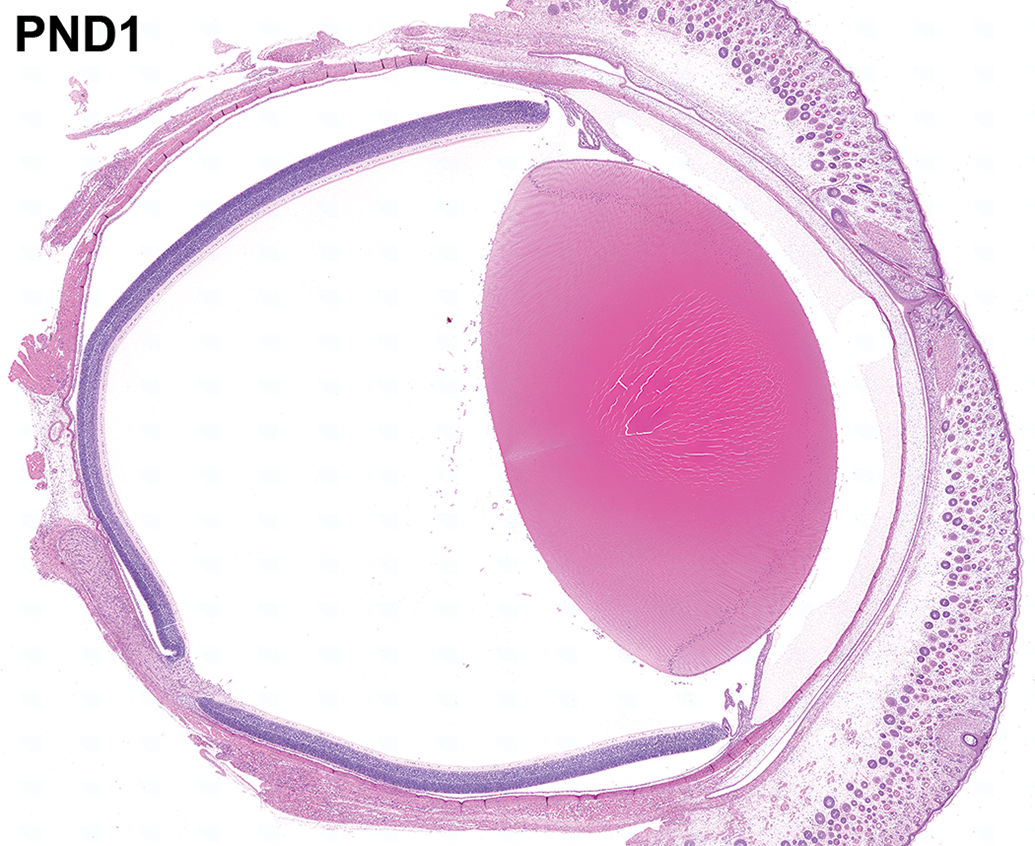
The entire eye and eyelid at postnatal day 1. A sagittal section of the ocular globe with the optic nerve at postnatal day 1. The anterior surface of the eye is completely covered by the fused eyelid. Hematoxylin and eosin staining.
Morphometric analyses of the thickness of the retinal layers in the histological preparations were conducted. Morphometric analysis was performed by capturing the images of the visual streak region of the retina in each eye. All images were captured with a ×20 objective using the BX51 light microscope, which was equipped with a DP71 color charge coupled device camera and the color imaging software cellSens Standard version 1.9 (Olympus Corporation, Tokyo, Japan). Image analysis was achieved using 24-bit color tagged image file format (1360 × 1024 pixels) images acquired using Olympus cellSens Standard version 1.9, which were analyzed with the freely available software program ImageJ Fiji version 1.52n. 19,20 Thickness was measured at the center of the visual streak. Border settings for the measuring range in each layer were as follows: nerve fiber layer (NFL), between the inner limiting membrane and the inner side of the innermost ganglion cell body; ganglion cell layer (GCL), from the inner side of the innermost ganglion cell body to the outer side of the outermost ganglion cell body; inner plexiform layer (IPL), between the outer side of the outermost ganglion cell body and the inner edge of the nuclei in the inner nuclear layers (INL); INL, between the inner and outer edges of the nuclei; outer plexiform layer (OPL), from the outer edge of the nuclei in the INL to the inner edge of the nuclei in the outer nuclear layer (ONL); ONL, between the inner and outer edges of the nuclei; photoreceptor layer (PRL), from the outer edge of the nuclei in the ONL to the end of the retinal outer segment.
Results
Eyelid
On gross observation, the upper and lower eyelids of all newborn neonates were fused at the palpebral fissure, which could be clearly identified at birth. The surface of the eye was completely covered by this fused eyelid. The fused region of the eyelid consisted of epidermal tissue, which was composed of stratified cuboidal/columnar epithelium. This was present on the center of the fused eyelid and was responsible for partitioning the dermis of the upper and lower eyelids. The dermis of the eyelid was composed of relatively scant connective tissue. Immature Meibomian glands composed of several small acini were found on the margins of the upper and lower eyelids. A few mitotic figures were observed on the basal aspect of the acini. The orbicularis oculi muscle encircled the eyelid, but its muscle fibers were still immature (Figure 2A). The epidermis of the palpebral fissure became keratinized with age, forming a stratified squamous epithelium on the margin of the eyelids. At PND9, the appearance of the palpebral fissure indicated that it was soon likely to be separated by the keratinization of epidermis at the eyelid margins. The Meibomian glands showed an increase in the number and length of acini with age (Figure 2B).
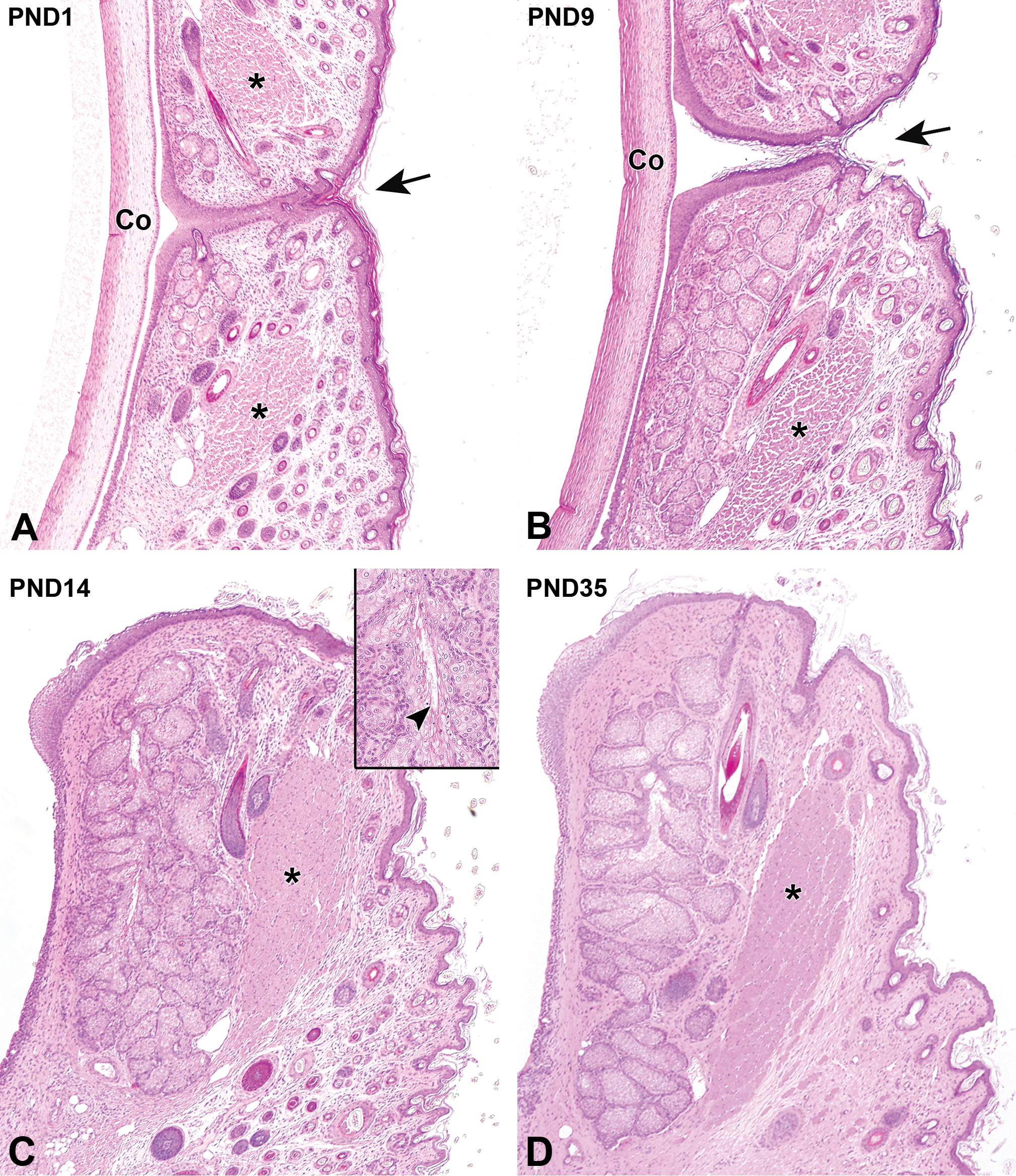
Postnatal histological development of the eyelid in white rabbits. A, At postnatal day (PND) 1, the palpebral fissure (arrow) is closed. The immature orbicularis oculi muscle (asterisks) and the short Meibomian gland can be observed at the eyelid margin. B, At PND9, the palpebral fissure (arrow) will soon be separated by the keratinization of the epidermis at the eyelid margin. The orbicularis oculi muscle (asterisk) is still immature. The Meibomian gland increases in length and develops an increasing number of acini. C, At PND14, the orbicularis oculi muscle fibers continue to develop (asterisk). Note further increases in the length of the Meibomian gland and the number of acini with initiation of secretion (inset arrowhead). Dermal connective tissue increases in density. D, At PND35, the hair follicles have decreased in number, and each component of the eyelid is histologically mature. Original objective ×4. Hematoxylin and eosin staining. Co indicates cornea.
The eyes of all kits except one opened at PND11 by the separation of the upper and lower eyelids. In the remaining kit, the right and left eyes opened at PND10 and PND12, respectively. The upper and lower eyelids were separated at the keratinization site, and the margin of the eyelid epidermis was covered by keratinized stratified squamous epithelium. At PND14, the density of the dermal connective tissue of the eyelid increased, and the orbicularis oculi muscle fibers subsequently developed. The Meibomian gland acini continued to increase in length. Secretions from the Meibomian gland acini could be detected from PND14. The orbicularis oculi muscle fibers continued to develop from this time point. The number of hair follicles in the dermis increased from birth until PND14 (Figure 2C), after which the number began to decrease. Development of the dermis and the Meibomian gland was completed at PND35, at which point the eyelid appeared to be mature on histological examination (Figure 2D, Table 2).
Summary of Postnatal Histological Development of Anterior Segments of the Eye and the Eyelid in White Rabbits Ranging From Postnatal Day (PND) 1 to 42.
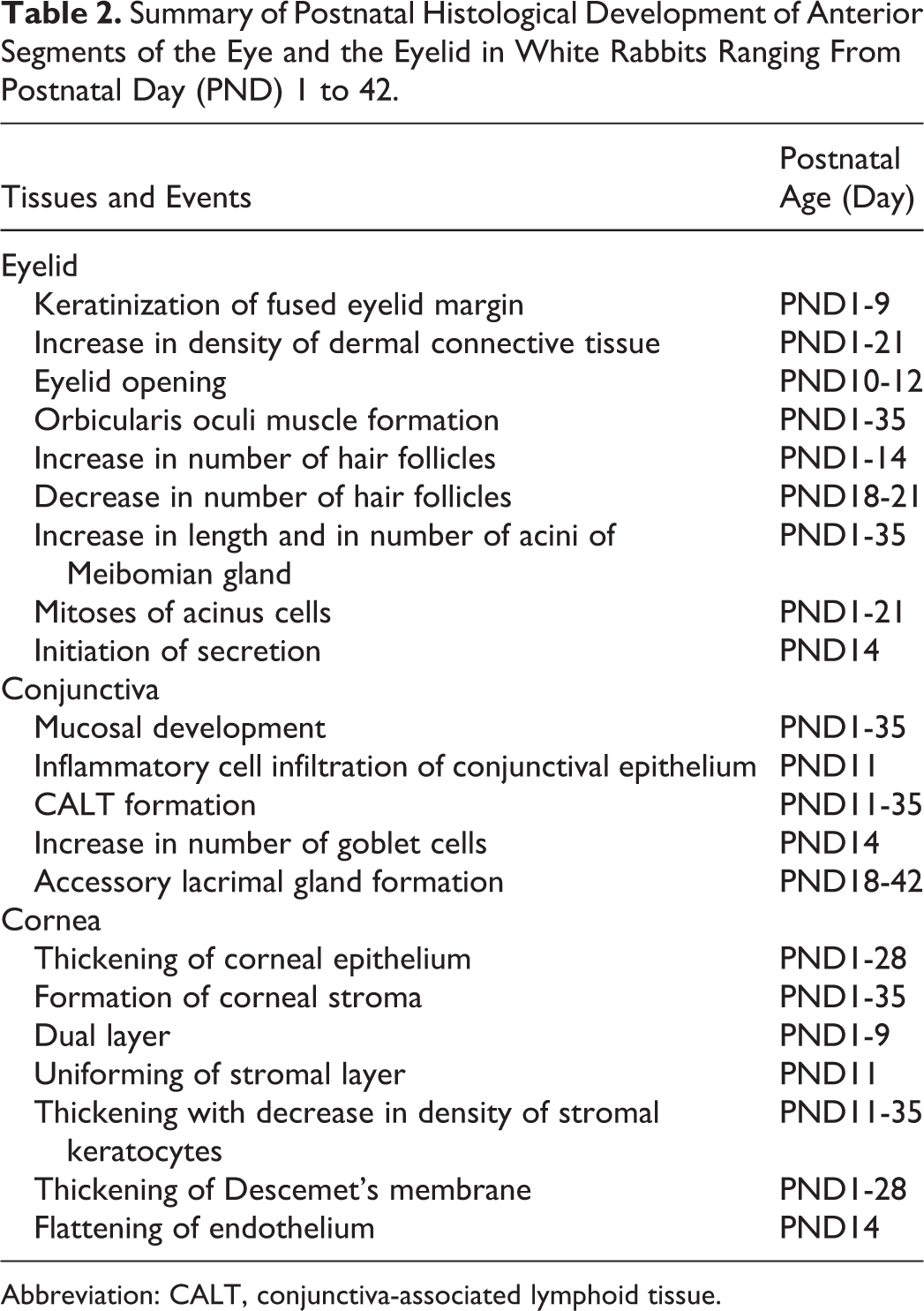
Abbreviation: CALT, conjunctiva-associated lymphoid tissue.
Conjunctiva
The conjunctival epithelium was composed of 2 to 3 layers of stratified oval cells. The submucosal connective tissue was filled with a relatively small amount of collagen fibers, and contained several blood vessels (Figure 3A). Up to PND9 (before eyelid opening), the conjunctival epithelium was relatively thin, and the histological development was not apparent. In the lamina propria, the density of the submucosal collagen fibers increased and the muscularis mucosa gradually developed with age (Figure 3B). The presence of goblet cells in the conjunctival epithelium was particularly rare before the opening of the eyelids. After the eyelids opened, the conjunctival epithelium increased in thickness. Observations at this stage revealed that it was now composed of 2 to 3 layers of stratified cuboidal/columnar epithelium, and a drastic increase in the number of goblet cells was seen. At PND11, a small lymphocytic nodule, termed conjunctiva-associated lymphoid tissue (CALT), was observed on the dermis of the conjunctiva (Figure 3C). The infiltration of heterophils transiently increased in the conjunctival epithelium (Figure 3D). At PND18, the conjunctival epithelium was considerably thickened, and the lamina propria continued to develop (Figure 3E). In the palpebral conjunctiva, the accessory lacrimal gland gradually formed from PND18 through the infolding of the conjunctival epithelium. At PND35, CALT could be clearly identified (Figure 3F). The formation of the accessory lacrimal gland in the palpebral conjunctiva was completed at PND42, by which time the conjunctiva was histologically mature (Table 2).
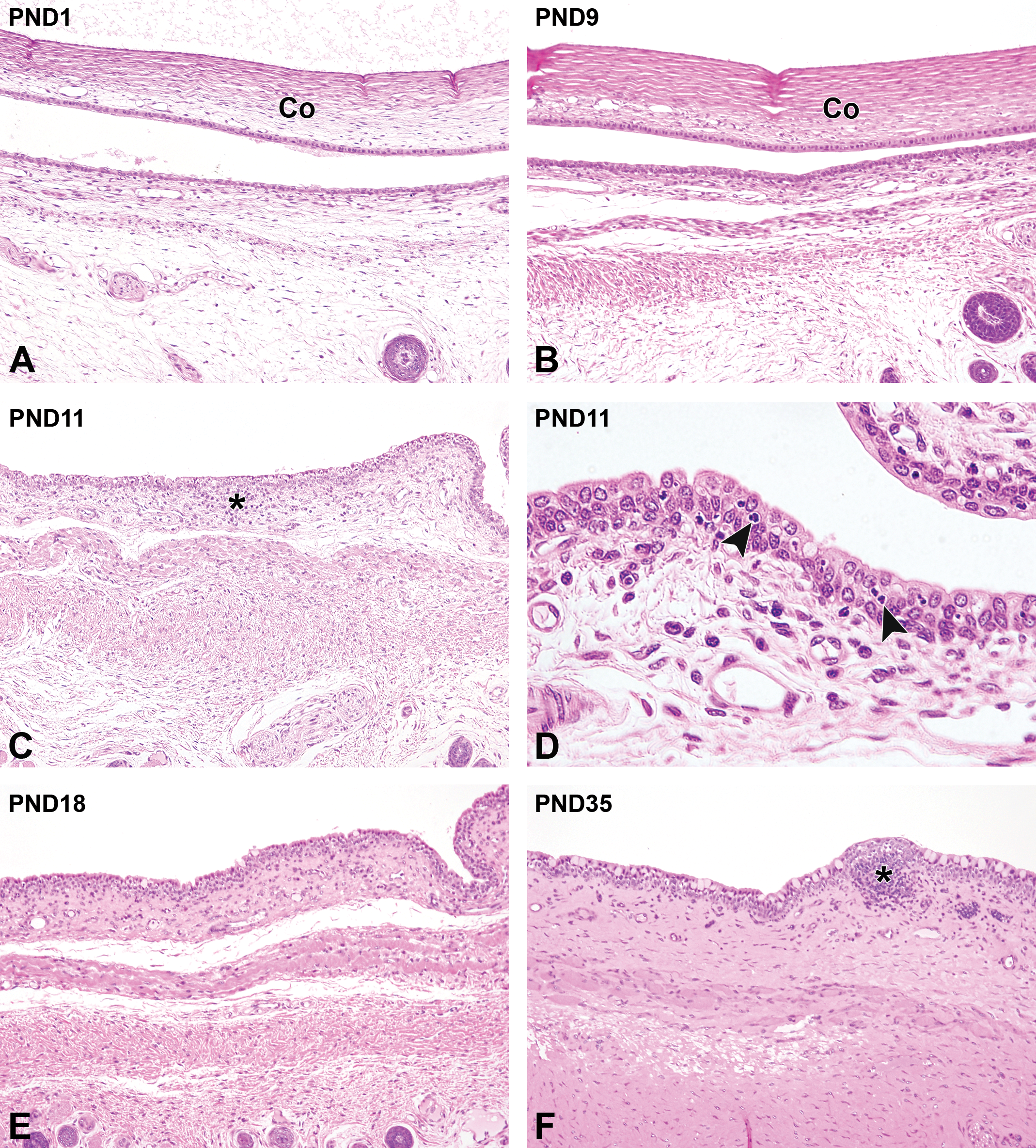
Postnatal histological development of the conjunctiva in white rabbits. A, At postnatal day (PND) 1, the conjunctiva is composed of tiny stratified cuboidal epithelial cells with an immature lamina propria. B, At PND9, the conjunctival epithelium increases in thickness. The connective tissue and muscularis mucosa in the lamina propria are gradually developing at this stage. C, At PND11, an accumulation of lymphocytes appears in the lamina propria, and a small lymphocytic nodule, termed conjunctiva-associated lymphoid tissue (CALT) forms (asterisk). D, At PND11, an infiltration of heterophils (arrowheads) is transiently observed in the conjunctival epithelium. E, At PND18, further development of the lamina propria is observed. F, At PND35, The histological development of the lamina propria is completed. Note the clear appearance of the CALT (asterisk). A-C, E, and F, Original objective ×10; D: original objective ×40. Hematoxylin and eosin staining. Co indicates cornea.
Cornea
Corneal epithelium
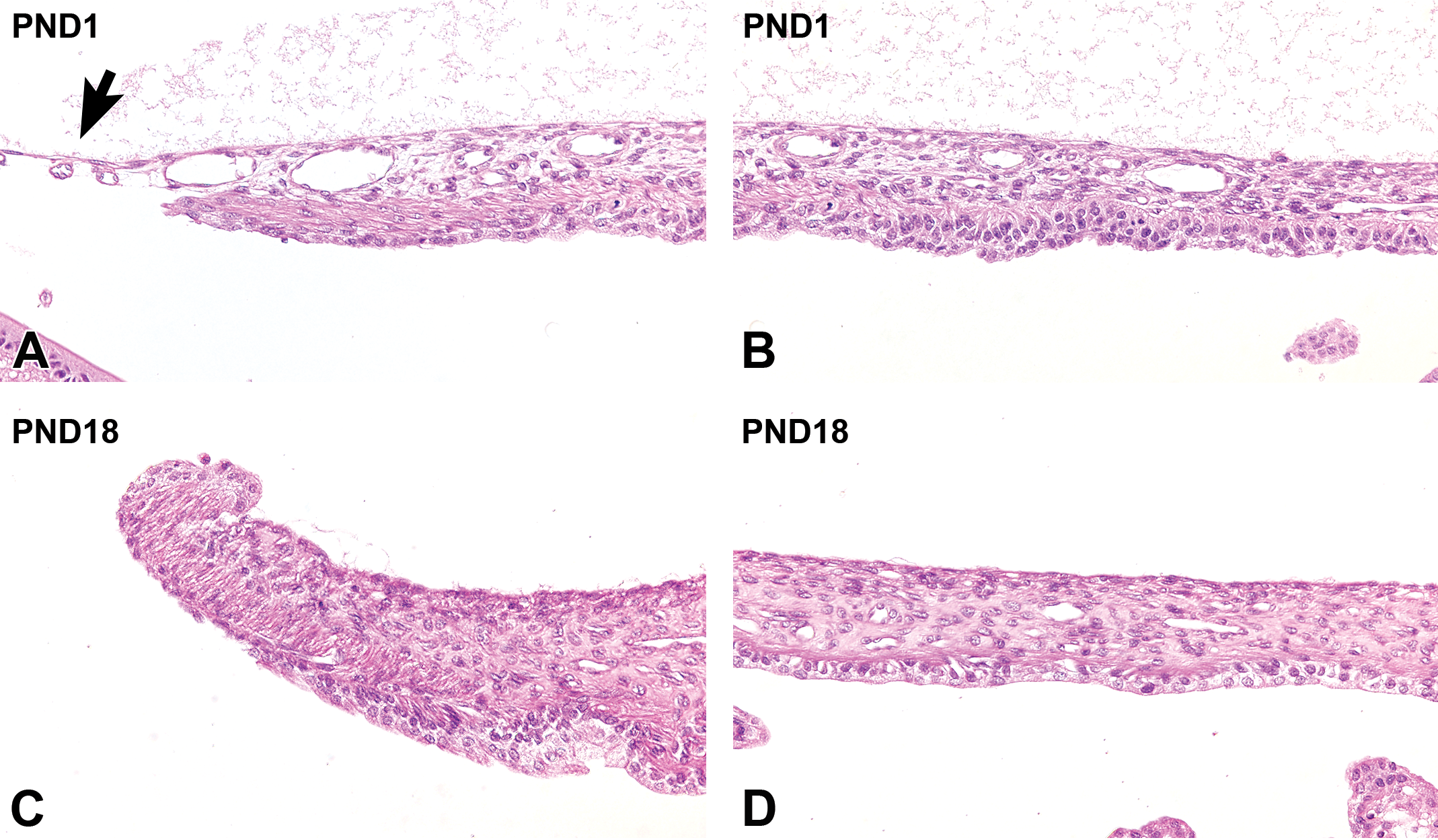
At PND1, the corneal epithelium completely covered the entire surface of the cornea and was composed of cuboidal basal cells with 2 to 3 layers of flattened cells at the epithelial surface. The basement membrane was still not clearly defined (Figure 4A). From PND3 to PND9 (before eyelid opening), the basal cells gradually formed from cuboidal to columnar cells, and the number of cell layers increased through the formation of a second layer called the wing-cell layer. The basement membrane was still not evident (Figure 4B). At PND14 (after eyelid opening), the corneal epithelium significantly thickened by transforming to a nonkeratinized squamous epithelium composed of 4 to 5 layers. The basal cells and wing cells appeared to be more crowded than they were before eyelid opening. The columnar basal cells continued to increase in height, and the wing cells became more cuboidal, causing the corneal epithelium to thicken. Periodic acid-Schiff-positive material appeared in the basal portion of the cytoplasm (Figure 4C). At PND28, the corneal epithelium was composed of 5 to 6 layers and was histologically mature (Figure 4D, Table 2).
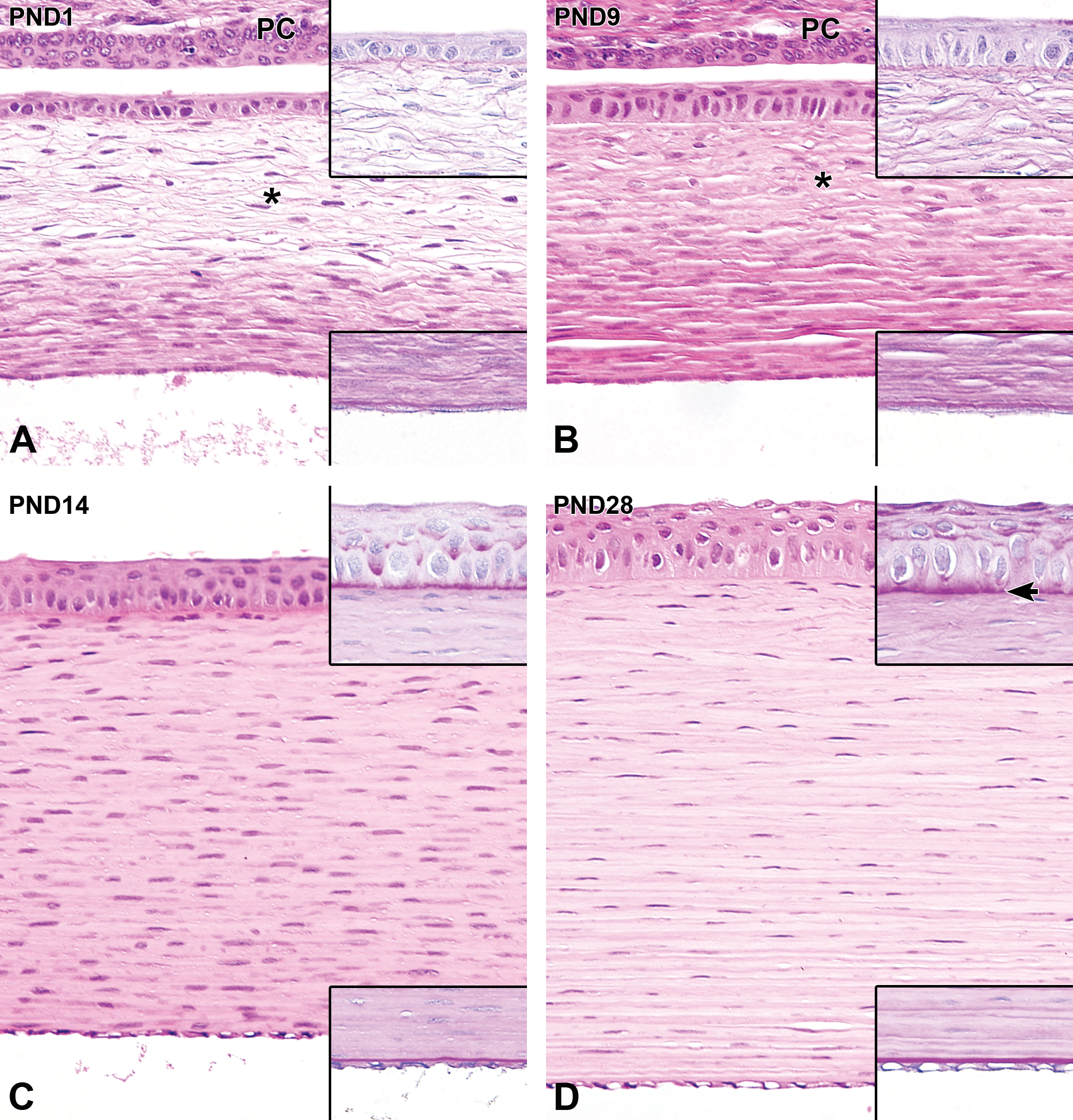
Postnatal histological development of the cornea in white rabbits. A, At postnatal day (PND) 1, the corneal epithelium is a 2- to 3-cell layer that consists of stratified cuboidal epithelium. The basement membrane is not apparent (upper inset). The corneal stroma is composed of an anterior (surface side) fibroareolar layer (asterisk) and a posterior (inner side) lamellated fibrous layer before eyelid opening. The corneal endothelium is composed of a single layer of cuboidal epithelium. Descemet membrane is extremely thin (lower inset). B, At PND 9, the corneal epithelium is approximately 4 cells thick. The basement membrane is still not apparent (upper inset). The corneal stroma has an increased number of extracellular lamellated fibers. Descemet's membrane is thicker than at PND1 (lower inset). C, After eyelid opening, at PND14, the corneal stroma drastically increases in thickness due to the expansion of the posterior lamellated fibrous layer across the whole stroma, while the anterior fibroareolar layer disappears. The corneal epithelium increases in thickness, along with the appearance of PAS-positive materials in the basal portion of the cytoplasm. The basement membrane is still not apparent (upper inset). The corneal endothelium is flattened. Descemet's membrane is more apparent (lower inset). D, At PND28, a further increase in epithelial thickness occurs. The basement membrane gradually becomes evident, along with an increasing amount of PAS-positive materials in the cytoplasm (upper inset arrow). The corneal stroma thickens further. The stromal keratocytes decrease in density and the nuclei become spindle-shaped. Descemet's membrane (lower inset) continues to increase in thickness. Original objective ×20. Hematoxylin and eosin staining; Insets: PAS, periodic-acid Schiff staining. PC indicates palpebral conjunctiva.
Corneal stroma
At PND1, the corneal stroma consisted of 2 layers: an anterior (surface side) fibroareolar layer and a posterior (inner side) lamellated fibrous layer. The anterior fibroareolar layer was comprised of relatively scant stromal connective tissue with linearly arranged stromal keratocytes. The posterior lamellated fibrous layer was comprised of relatively dense lamellated collagen fibers with highly dense stromal keratocytes. At the limbus, the anterior fibroareolar layer and the posterior lamellated fibrous layer appeared to transition into the bulbar conjunctival submucosa and the sclera, respectively. Both layers contained stromal keratocytes with oval nuclei (Figure 4A). This arrangement of the corneal stroma continued until the opening of the eyelid. From PND3 to PND9 (before eyelid opening), the extracellular lamellated collagen matrix increased in density in both the anterior fibroareolar and posterior lamellated fibrous layers. Thickening of the corneal stroma was not observed (Figure 4B). At PND14 (after eyelid opening), the corneal stroma drastically thickened and was composed of the lamellated fibrous layer, with disappearance of the anterior fibroareolar layer. After eyelid opening, a further increase in stromal thickness with a decreasing density of stromal keratocytes was observed with age (Figure 4C). The corneal stroma was histologically mature at PND28, by which time the formation of stromal connective tissue had been completed and the stromal keratocytes had decreased in density (Figure 4D, Table 2).
Corneal endothelium and Descemet's membrane
The corneal endothelium was initially composed of a single layer of cuboidal cells, and Descemet's membrane was extremely thin (Figure 4A). From PND3 to PND9 (before eyelid opening), no discernible histological change in the morphology of endothelial cells was observed, whereas Descemet's membrane gradually thickened (Figure 4B). At PND14 (after eyelid opening), the corneal endothelium was significantly flattened (Figure 4C). Descemet's membrane continued to thicken even after the opening of the eyelids (Figure 4D, Table 2).
Iridocorneal Angle
At PND1, the iridocorneal angle was already apparent between the corneoscleral junction and the iris (Figure 5A). The iridocorneal angle contained the trabecular meshwork composed of spindle-shaped cells with oval nuclei. The intercellular spaces could already be observed in the trabecular meshwork. Half of the angle recess was covered by a single layer of cuboidal corneal endothelial cells and the scattered termination of Descemet's membrane (Figure 5B). At PND7, the iridocorneal angle extended into the anterior ciliary body (Figure 5C). In the PAS-stained section, PAS-positive extracellular materials occasionally appeared in the intercellular spaces of the trabecular meshwork. The scattered termination of Descemet's membrane continued to remain evident in the angle recess (Figure 5D). At PND18, the iridocorneal angle was located on the roots of the ciliary processes (Figure 5E). The amount of extracellular material in the intercellular spaces of the trabecular meshwork increased. The termination of Descemet's membrane blended into the structure of the trabecular meshwork (Figure 5F). At this age, the iridocorneal angle appeared to be histologically mature (Table 3).
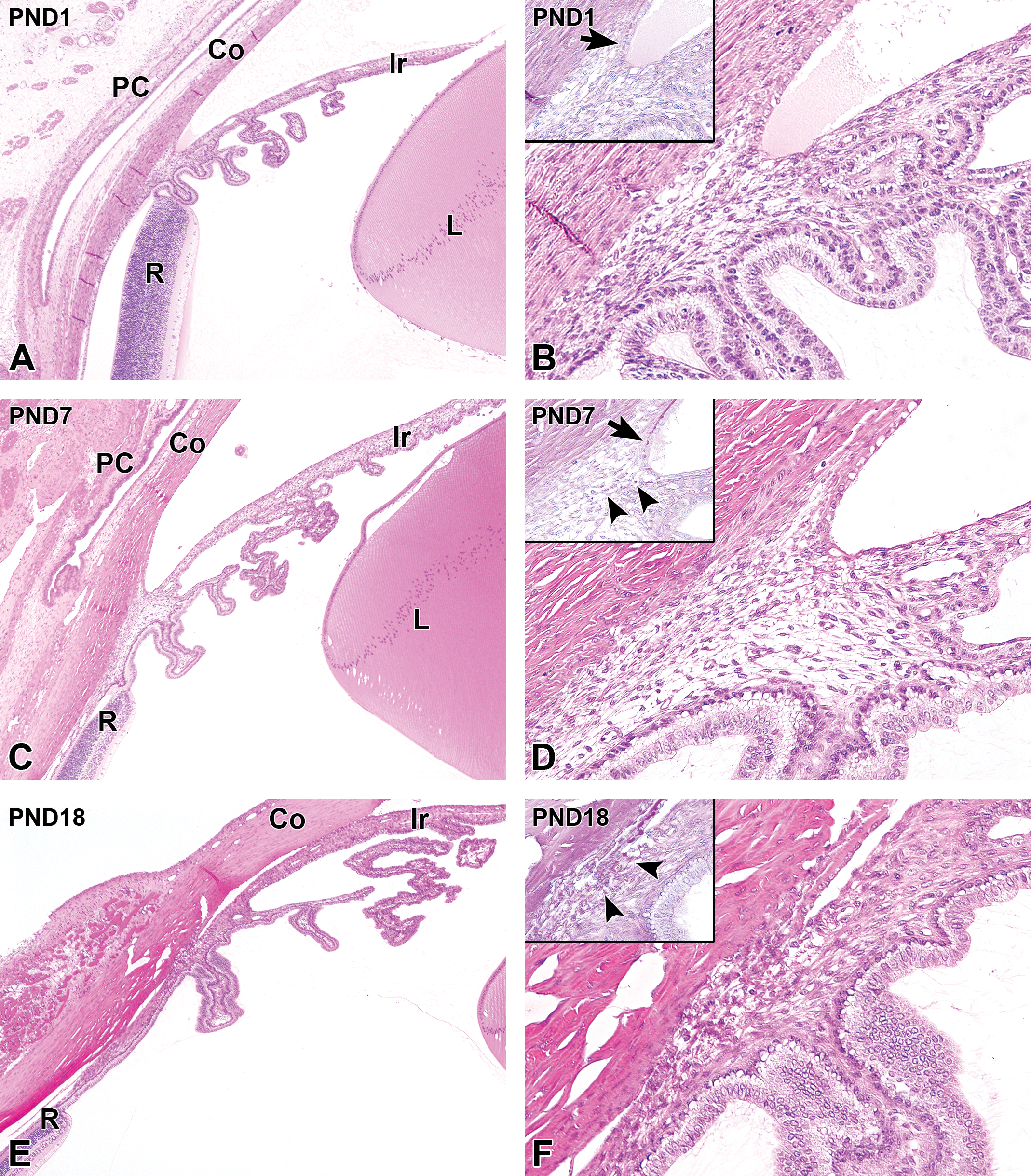
Postnatal histological development of the iridocorneal angle and the ciliary body in white rabbits. A, The iridocorneal angle and the ciliary body at postnatal day (PND) 1. The iridocorneal angle is evident between the corneoscleral junction and the iris. The ciliary processes are short and located adjacent to the ora serrata retinae. The posterior ciliary body is not evident. B, Higher magnification of (A). Half of the angle recess is covered by the corneal endothelium and the scattered Descemet's membrane termination (inset arrow). The ciliary processes are covered by a 2-cell layer of ciliary epithelium, and vascular structures are apparent in the relatively scant stromal connective tissue. C, At PND7, the iridocorneal angle extends into the anterior ciliary body. The ciliary processes are elongated and show complex branching. The posterior ciliary body forms between the ciliary processes and the ora serrata retinae. D, Higher magnification of (C). The scattered Descemet's membrane termination remains evident in the angle recess (inset arrow). The ciliary epithelium becomes eosinophilic. PAS-positive extracellular materials appear in the intercellular spaces of the trabecular meshwork (inset arrowheads). E, At PND18, the iridocorneal angle is located on the roots of the ciliary processes. The ciliary processes and posterior ciliary body undergo further branching and elongation, respectively. F, Higher magnification of (E). Further increases in the PAS-positive extracellular materials are observed in the trabecular meshwork (inset arrowheads). Descemet's membrane termination blends into the structure of the trabecular meshwork. The ciliary epithelium becomes more eosinophilic. Note an increase in the density of the stromal connective tissue of the ciliary processes. A, C, and E, Original objective ×4; B, D, and F: original objective ×20. Hematoxylin and eosin staining; Insets: PAS, periodic-acid Schiff staining. Co indicates cornea; Ir, iris; L, lens; PC, palpebral conjunctiva; R, retina; PAS, periodic acid-Schiff.
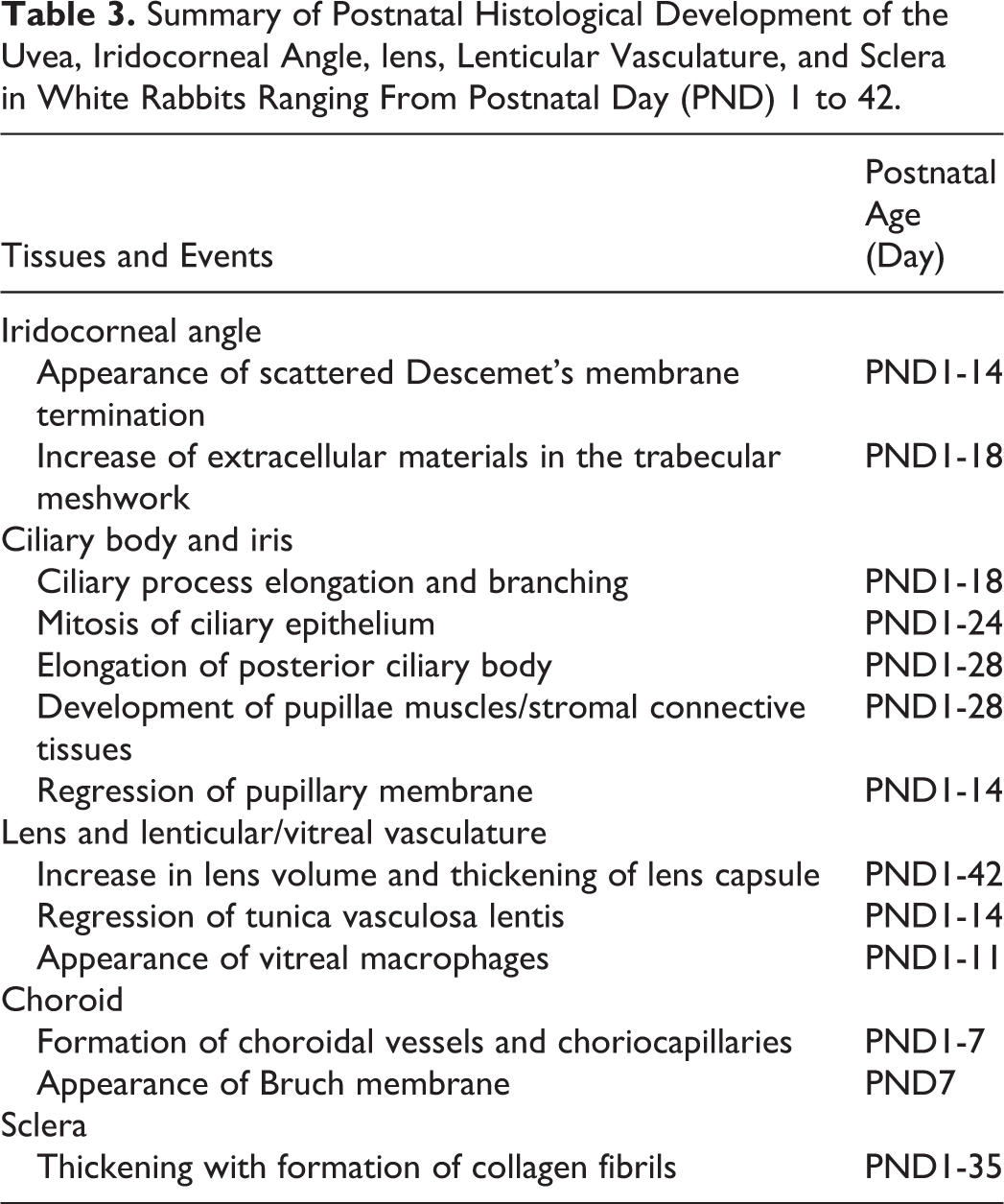
Summary of Postnatal Histological Development of the Uvea, Iridocorneal Angle, lens, Lenticular Vasculature, and Sclera in White Rabbits Ranging From Postnatal Day (PND) 1 to 42.
Ciliary Body
At PND1, the ciliary body was composed of short ciliary processes. The posterior ciliary body was not evident, meaning that the ciliary processes were adjacent to the edge of the retina, which is termed the ora serrata retinae (Figure 5A). The ciliary processes contained blood vessels in the stroma, and were covered by a 2-cell layer of ciliary body epithelium, which was composed of a single layer of cuboidal basal cells with a single layer of columnar surface cells. The basal cells had eosinophilic cytoplasm, and mitotic figures were occasionally found. The surface cells appeared to have round nuclei and pale eosinophilic cytoplasm (Figure 5B). The ciliary processes increased in length and developed complex branches with age, and the posterior ciliary body gradually elongated from birth (Figure 5C). The cytoplasm of the ciliary epithelium also became more eosinophilic with age, especially in the basal cells (Figure 5D). At PND18, the branching of the ciliary processes became more complex, and the posterior ciliary body appeared to elongate further (Figure 5E). The ciliary epithelium also became more eosinophilic, and the stromal connective tissue of the ciliary processes increased in density (Figure 5F). Mitotic figures were observed in the basal cell layer of the ciliary epithelium up to PND24. Histological maturation of the ciliary body was observed at PND28, at which point the formation of the posterior ciliary body was completed (Table 3).
Iris
At PND1, the pupillary membrane remained evident at the margin of the iris. The iridal stroma was hypercellular, and the density of the stromal connective tissue was relatively low. The sphincter and dilator pupillae muscles were still immature. The dilator pupillae muscle was aligned adjacent to the basal epithelium of the posterior iris, in which mitotic figures were occasionally observed, but its smooth muscle fibers were still not apparent (Figure 6A and B). The iridal stromal connective tissue increased in density with age, and the sphincter pupillae and dilator pupillae muscle fibers gradually became evident. The remnant of the pupillary membrane gradually regressed, and by PND18, it had diminished completely. At this stage, the connective tissue in the iridal stroma was abundant, and the sphincter pupillae and dilator pupillae muscle fibers were evident (Figure 6C and D). At PND28, the histological development of the iris was completed by the maturation of the sphincter and dilator pupillae muscles and the completion of the development of iridal stromal connective tissue (Table 3).
Postnatal histological development of the pupillary portion (A, C) and the ciliary portion (B, D) of the iris in white rabbits. A, The pupillary portion of the iris at postnatal day (PND) 1. The pupillary membrane (arrow) remains evident in the pupil. The sphincter pupillae muscle is still immature. B, In the ciliary portion at PND1, the iridal stroma is hypercellular. The dilator pupillae muscle fibers are still not apparent. C, The pupillary portion of the iris at PND18. The pupillary membrane has disappeared completely. D, In the ciliary portion at PND18, the sphincter pupillae muscle has smooth muscle fibers. Note the increased amount of iridal stromal connective tissue. Original objective ×20. Hematoxylin and eosin staining.
Lens and Lenticular/Vitreal Vasculature
At PND1, the lenticular vascular structure, termed the tunica vasculosa lentis, persisted in the vitreous and was seen circumscribing the lens (Figure 7A and B). The vitreal artery was not observed at PND1. Vitreal macrophages were occasionally found around the blood vessels at the posterior side of the lens (Figure 7C). Up to PND11 (immediately after eyelid opening), these vitreal macrophages could be temporarily found around the vasculature. The lens enlarged and developed an increasing number of lens fibers, and the lens capsule gradually thickened with age. The lenticular vasculature regressed gradually (Figure 7D and E). At PND14, the lenticular vasculature disappeared completely. Enlargement of the lens continued throughout all ages in this study (Table 3).
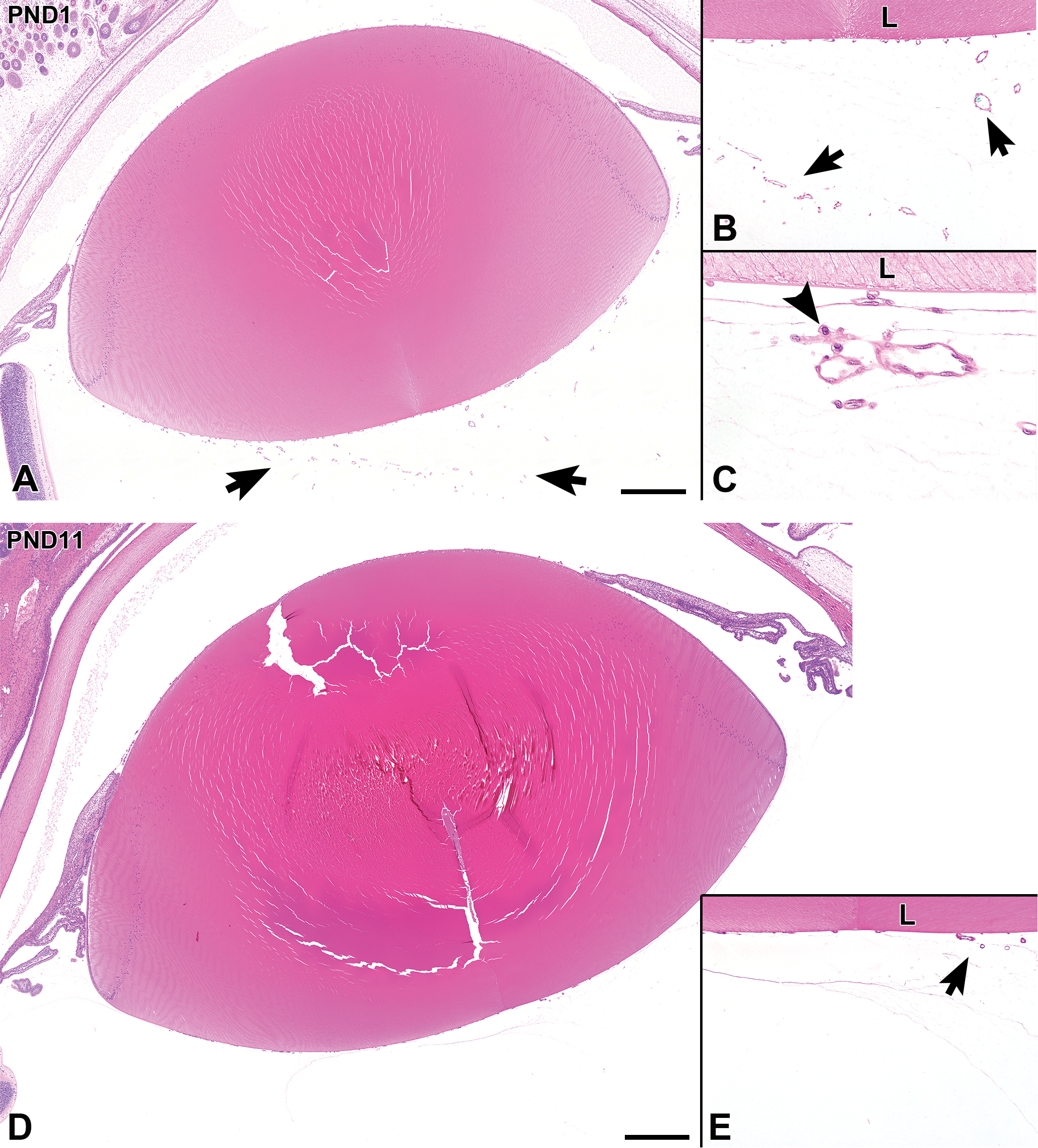
Postnatal histological development of the lens and surrounding lenticular vasculature in white rabbits. A, At postnatal day (PND) 1, the lens is relatively small. The lenticular vascular structure, termed the tunica vasculosa lentis, is found in the vitreous (arrows). B and C, Higher magnifications of (A). A vitreal macrophage (arrowhead) is observed in the area around the vasculature. D, At PND11, the lens is enlarged in comparison to its size at PND1. E, Higher magnification of (D). The lenticular vasculature appears to have regressed (arrow). A and D, Scale bar = 500 µm; B and E original objective ×10; C original objective ×40. Hematoxylin and eosin staining. L indicates lens.
Choroid
At PND1, the choroid consisted of immature choroidal vessels and randomly arranged spindle cells that had oval nuclei. Bruch membrane and choriocapillaries were not evident at PND1. A small number of mitotic figures were found, predominantly on the inner aspect of the choroid (Figure 8A). During the first week after birth, the choroidal vessels and choriocapillary layer gradually formed. At PND7, Bruch membrane became apparent adjacent to the retinal pigment epithelium (RPE), and at this point the choroid was histologically mature (Figure 8B, Table 3).
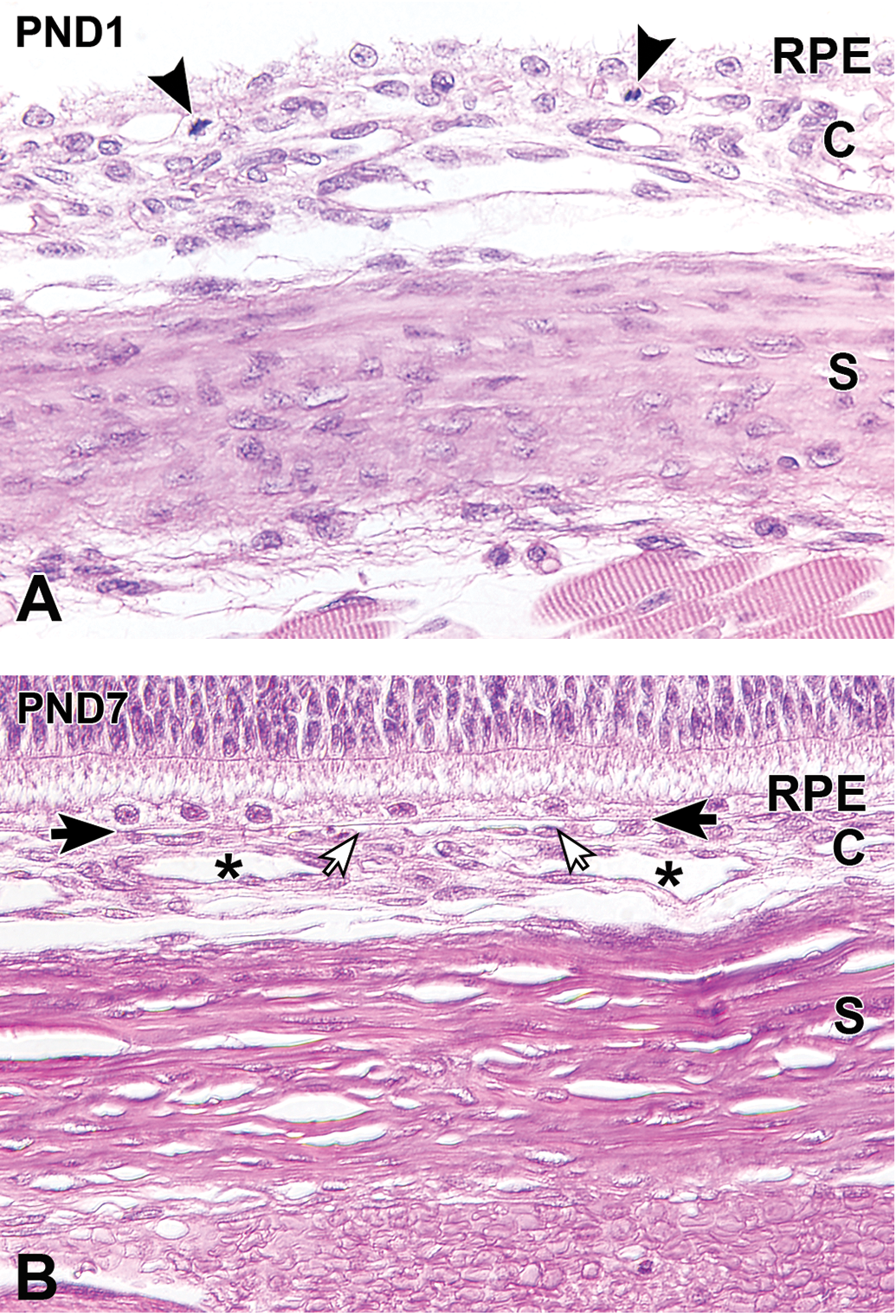
Postnatal histological development of the choroid and the sclera in white rabbits. A, At postnatal day (PND) 1, the choroid is composed of immature choroidal vessels and spindle cells that have oval nuclei. The sclera is hypercellular, and the scleral cells are spindle-shaped/elongated and have oval nuclei. Mitotic figures are found predominantly on the inner side of the choroid (arrowheads). B, At PND7, the choroidal blood vessels (asterisks) are evident, and the choriocapillary layer becomes apparent (open arrows). Bruch membrane (solid arrows) appears adjacent to the retinal pigment epithelium. The sclera increases in thickness as the extracellular collagen fibrils develop a lamellar arrangement. Original objective ×40. Hematoxylin and eosin staining. C indicates choroid; RPE, retinal pigment epithelium; S, sclera.
Sclera
The sclera at PND1 consisted of spindle-shaped or elongated cells that had oval to elongated nuclei, while extracellular collagen fibrils were not apparent at this stage (Figure 8A). Over time, the sclera gradually thickened, developing an increasing amount of extracellular collagen matrix with a lamellar arrangement of fibrils. The scleral cell nuclei appeared to elongate gradually as the amount of lamellated collagen fibrils increased and the cellular density decreased (Figure 8B, Table 3).
Retina
The retina was composed of a NFL, a GCL, an IPL, thickened INL, ONL, and a PRL. The OPL between the INL and ONL was extremely thin at PND1. The total thickness of the retina tended to decrease with age, mainly due to a reduction in the thickness of the INL and ONL. Immediately after eyelid opening at PND11, further histological changes of the retinal layers were observed (Figure 9). At PND35, the structure of the retinal layers was morphologically mature.
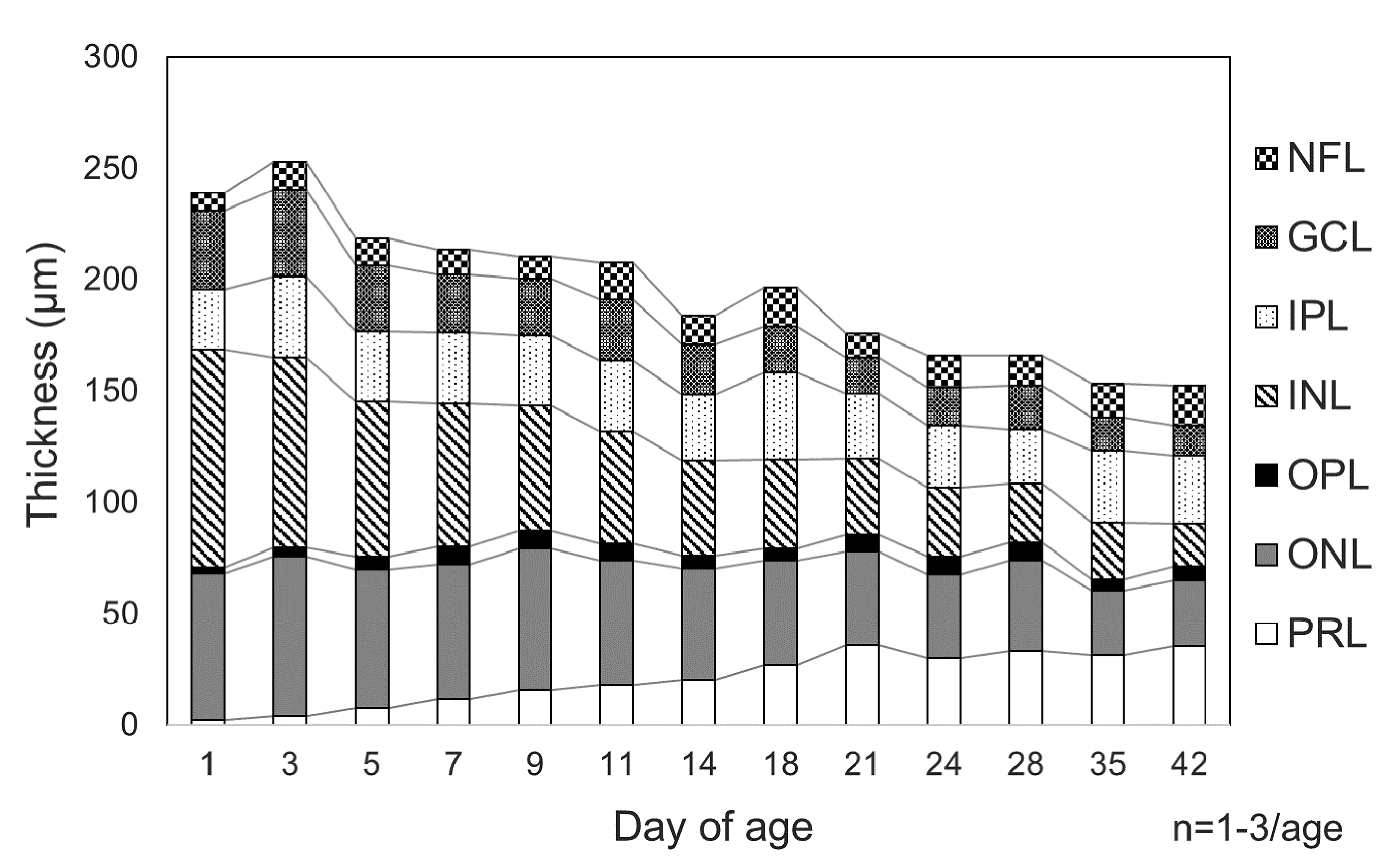
Postnatal changes in the thickness of the retinal layers seen in white rabbit eyes fixed in Davidson fixative. After birth, the retina tends to decrease in thickness with age, predominantly due to thinning of the inner and outer nuclear layers. The retinal photoreceptor layer increases in thickness up to postnatal day 21. GCL indicates retinal ganglion cell layer; IPL, inner plexiform layer; INL, inner nuclear layer; NFL, retinal nerve fiber layer; ONL, outer nuclear layer; OPL, outer plexiform layer; PRL, retinal photoreceptor layer.
Nerve fiber layer and ganglion cell layer
At PND1, the GCL was composed of retinal ganglion cell bodies that had round nuclei with scant cytoplasm, in which Nissl material was not observed. The visual streak, which was composed of densely aligned retinal ganglion cell bodies, had already formed on the lower side of the optic nerve head. The nerve fibers in the NFL at PND1 were still immature (Figure 10A). Up to PND5, a few pyknotic nuclei were observed in the GCL. At PND5, retinal ganglion cell bodies were plumper than those seen at PND1, and the nerve fibers could be clearly identified in the NFL (Figure 10B). At PND14, Nissl material appeared in the retinal ganglion cell bodies (Figure 10C). At PND21, the inner processes of Müller cells extended to the innermost boundary of the retina (Figure 10D), and was clearly evident in the H&E-stained section until PND35. At PND35, the GCL and NFL had a similar morphology to that seen in adults (Figure 10E, Table 4).
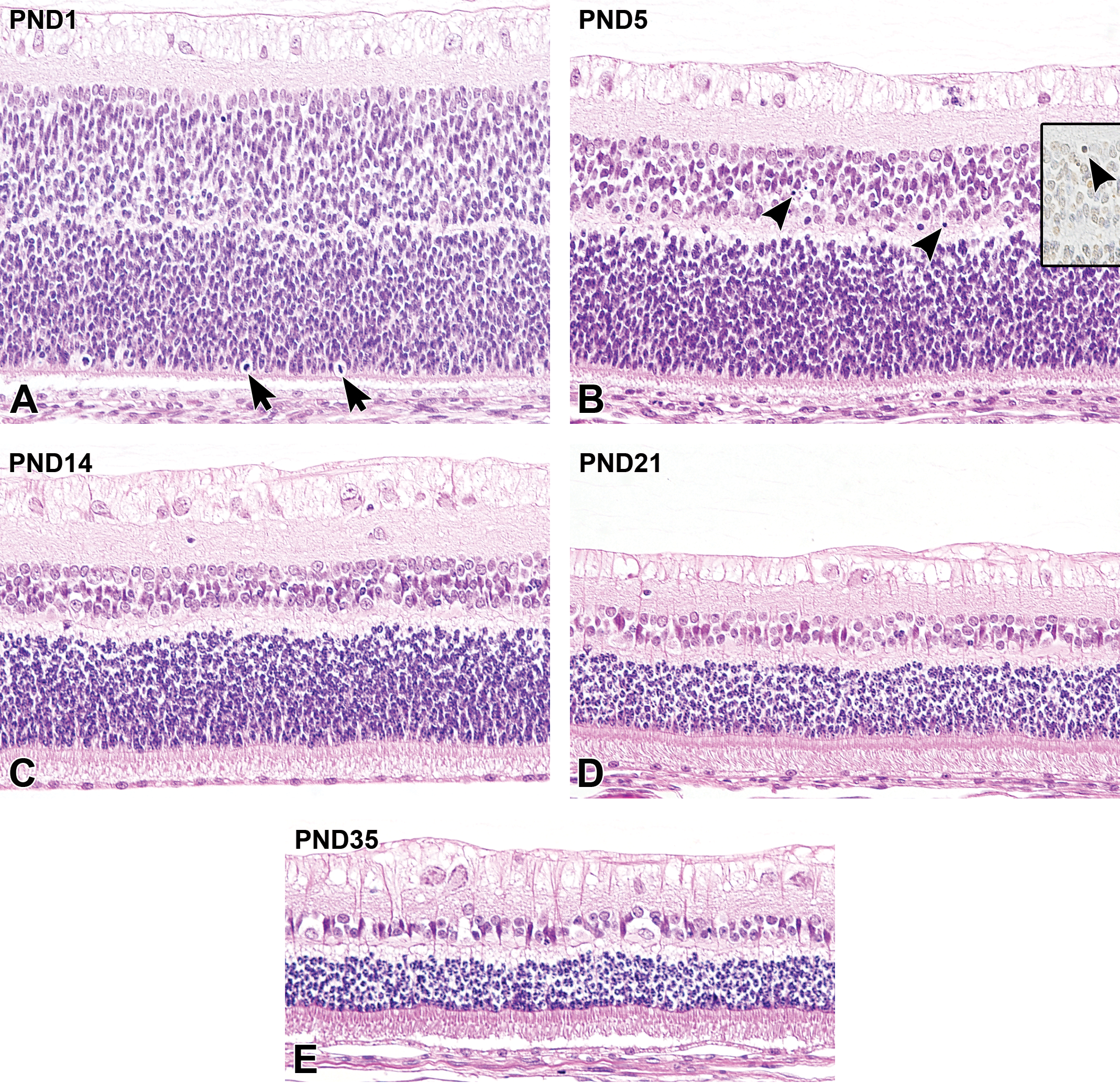
Postnatal histological development of the retina in white rabbits. A, At postnatal day (PND) 1, the retinal ganglion cells have scant cytoplasm. The retinal nerve fibers in the nerve fiber layer (NFL) are still immature. The inner nuclear layer (INL) and the outer nuclear layer (ONL) are composed of cells that have oval nuclei, and are separated by the tiny outer plexiform layer (OPL). Note mitotic figures in the peripheral region of the ONL (arrows). The photoreceptor layer (PRL) is extremely thin. The retinal pigment epithelium (RPE) is cuboidal and the cells themselves are relatively plump. B, At PND5, the retinal nerve fibers have developed and are evident in the NFL. The INL decreases in thickness and pyknotic nuclei appear (arrowheads). These pyknotic nuclei are positive on TUNEL staining (inset). The INL and ONL are clearly separated by the OPL. The PRL continues to elongate. C, At PND14, the retinal ganglion cell bodies are plump with the clear appearance of Nissl materials and nerve fibers. A further decrease in INL thickness occurs with cellular morphological differentiation. Note that the decreases in ONL thickness and cell number occur alongside increases in OPL thickness, the elongation of cones and rods in the photoreceptor layer (PRL), and the appearance of the outer limiting membrane (OLM). The RPE appears to be flattened. D, At PND21, the INL and ONL continue to decrease in thickness. The inner processes of Müller cells extend to the innermost boundary of the retina. In the PRL, further elongation of cones and rods is observed with the appearance of the photoreceptor inner and outer segments. Flattening of the RPE is completed. E, At PND35, the decreases in INL and ONL cell numbers and the formation of ganglion cell bodies/nerve fibers are complete, and the retinal structure is histologically mature. Original objective ×20. Hematoxylin and eosin staining; B inset: terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL).
Summary of Postnatal Histological Development of the Retina and Optic Nerve in White Rabbits Ranging From Postnatal Day (PND) 1 to 42.
Abbreviations: GCL, retinal ganglion cell layer; INL, inner nuclear layer; NFL, nerve fiber layer; ONL, outer nuclear layer; OPL, outer plexiform layer; RPE, retinal pigment epithelium.
Inner plexiform layer, inner nuclear layer, and outer plexiform layer
At PND1, the INL was composed of oval to elongated nuclei, and morphological differences between Müller, bipolar, horizontal, and amacrine cells could not be determined. The OPL was extremely thin at PND1 (Figure 10A). At PND5, the INL was thinner and contained a lower number of cells than at PND1, and the transient appearance of TUNEL-positive pyknotic nuclei was observed. In addition, morphological differentiation between different types of INL cells could be detected. On the inner side of the INL, occasional enlarged and rounded nuclei were observed at PND5. In the intermediate portion of the INL, spindle-shaped, deeply basophilic nuclei, which were recognized as Müller cells, were vertically arranged (Figure 10B). The OPL increased in thickness up to PND9. At PND14 (after eyelid opening), the INL further decreased in thickness and the number of cells again declined, but the pyknotic nuclei were no longer evident. Several cells on the outer side of the INL were enlarged and had round nuclei. In addition, most of the cells located adjacent to the IPL appeared to have round nuclei, and could be recognized as amacrine cells in H&E-stained sections (Figure 10C). At PND21, further morphological differentiation and development of the INL cells was observed, and the number of cells again decreased. The inner processes of Müller cells were apparent in the IPL and extended to the GCL (Figure 10D). At PND35, the decrease in the number of INL cells and the decrease in thickness of the INL were completed. Furthermore, the INL cells including Müller cells and horizontal cells could be distinguished by the shape of their nuclei in the H&E-stained sections (Figure 10E, Table 4).
Outer nuclear layer, PRL, and RPE
The ONL at PND1 was composed of cells that had round to oval nuclei, and the morphological differences between cone and rod nuclei could not be distinguished. Mitotic figures were frequently observed in the periphery of the ONL. The PRL at PND1 was extremely thin, and the inner and outer segments (IS, OS) of cones and rods could not be recognized. The RPE at PND1 was composed of a single layer of round to oval cells with clear cytoplasm (Figure 10A). At PND5, the PRL had increased in thickness compared to PND1 but the IS and OS of cones and rods were still not evident (Figure 10B). Mitotic figures in the ONL were only evident in the peripheral region. At PND14 (after eyelid opening), the ONL decreased in thickness, the number of cells in the ONL decreased and the PRL became significantly thickened. During the first 2 weeks after birth, the RPE gradually flattened (Figure 10C), and at PND21, each cell became cuboidal and exhibited a similar morphology to that seen in adults. Elongation of the PRL was completed, and the IS and OS were clearly distinguishable at PND21. The outer limiting membrane became evident between the ONL and PRL at PND18 to PND21. In the ONL, the decrease in thickness and in the number of cells continued after PND21, and the cell nuclei appeared to become rounded (Figure 10D). At PND35, the decrease in the number of ONL cells and in the thickness of the ONL was completed, and most of the cell nuclei displayed a concentric arrangement of chromatin at the periphery. At this stage, the retinal layers from the ONL to the RPE were histologically mature (Figure 10E, Table 4).
Optic Nerve
The optic nerve bundle was tiny and hypercellular at PND1, and the surrounding meninges were relatively thin (Figure 11A). The glial cells appeared to have round to oval nuclei and were randomly arranged in the optic nerve (Figure 11B). Mitotic figures were occasionally observed in the glial cells in the optic nerve bundle and surrounding meninges up to PND9. The optic nerve bundle and surrounding meninges gradually thickened with age (Figure 11C). At PND9, the glial cells were gradually decreasing in density and were linearly arranged. The blood vessels became apparent in the optic nerve at this stage (Figure 11D). At PND35, the thickening of the optic nerve bundle was complete (Figure 11E), and the glial cells had established their linear arrangement (Figure 11F). Blood vessels were clearly observed, and the optic nerve was histologically mature (Table 4).
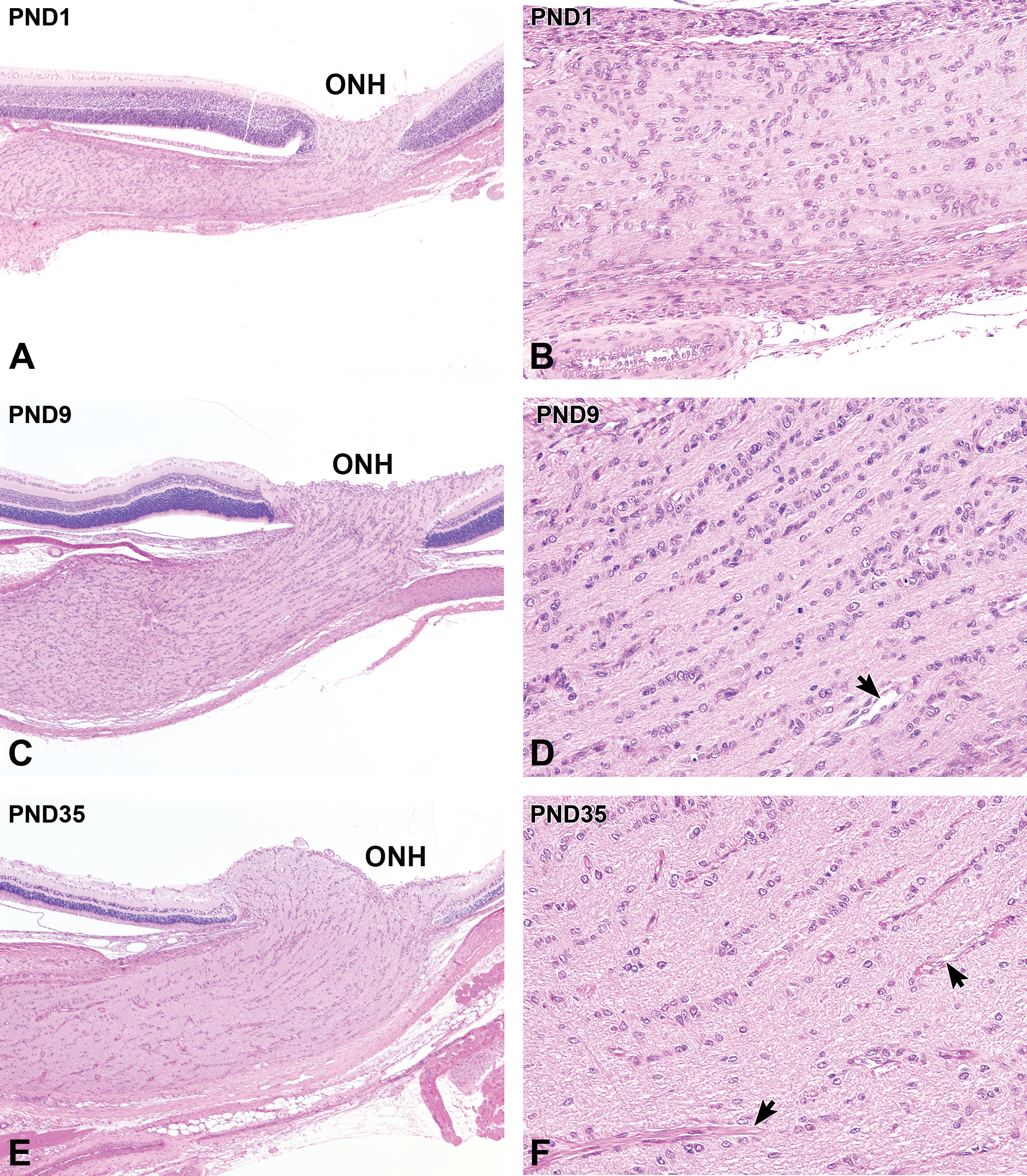
Postnatal histological development of the optic nerve in white rabbits. A, At postnatal day (PND) 1, the optic nerve bundle appears to be thin and hypercellular. B, Higher magnification of (A). The glial cells are randomly arranged, and the vascular structure in the optic nerve is not evident. C, At PND9, the optic nerve bundle and surrounding meninges increase in thickness. D, Higher magnification of (C). The glial cells in the optic nerve decrease in density and is gradually linearly arranged. Blood vessels appear in the optic nerve (arrow). E, At PND35, further thickening of the optic nerve bundle occurs. F, Higher magnification of (E). Note that the glial cells appear to be in linear arrangements in the optic nerve. The blood vessels in the optic nerve have sheaths (arrows). A, C, and E, Original objective ×4; B, D, and F, Original objective ×20. Hematoxylin and eosin staining; ONH indicates optic nerve head.
Discussion
In this study, we investigated the postnatal histomorphological development of the eyes in Japanese white rabbits from immediately after birth to 6 weeks of age. The majority of the eyes opened at PND11. Prior to eyelid opening, proliferation of cells in the ciliary body, choroid, and optic nerve was observed. Regression of the lenticular/vitreal vasculature and a decrease in the number of retinal cells, especially in the INL, was also observed. After eyelid opening, drastic changes in the anterior segment of the eyes were observed, including the thickening of the cornea and the appearance of lacrimation-related tissues in the eyelid and conjunctiva. Conjunctiva-associated lymphoid tissue is a constitutive mucosa-associated lymphoid tissue (MALT) that underlies the conjunctiva in some mammalian species including rabbits. 21,22 In this study, the CALT developed as early as PND11, just after eyelid opening, as found in a previous study. 21 This finding suggests that CALT is part of the MALT system that develops postnatally after antigenic exposure. In addition, a transient increase in heterophil infiltration was noted in the conjunctival epithelium at PND11. These results suggest that eyelid opening is a key event in the formation of the immune system of the ocular surface. In the posterior segment, the differentiation of retinal layer-derived cells, such as Müller cells, bipolar cells, and photoreceptor cells, was apparent after eyelid opening. Optic nerve thickening occurred concurrently with the maturation of retinal ganglion cells. An increase in the size of the lens continued throughout the postnatal period. Furthermore, we observed the histological maturation of the eyes and eyelids in rabbits at PND28 to 42. The postnatal development of ocular dimensions has been investigated in commonly used laboratory animals, including rabbits. 23 –25 In rabbits, optical parameters, such as eyeball diameter, corneal thickness, and vitreous volume, continue to increase with age. Morphometric analyses of lens dimensions were not conducted in this study, but the size appeared to increase with age on microscopic observation of the H&E-stained sections. Histological investigations of postnatal development in rabbit eyes have also been conducted for the corneal stroma, 7 the corneal endothelium, 26 –28 ciliary body, 9 the retina, 10,29 –31 and the CALT. 21 The results of this study regarding these tissues are similar to the findings of these previous investigations. Furthermore, a recent study examined pre- and postnatal ocular histological development in New Zealand white rabbits. 11 Ocular histology during postnatal development in rabbits has not yet been sufficiently illustrated in the literature to fully inform histopathological examinations in nonclinical toxicology studies. In this study, we describe additional histological features of postnatal development of rabbit eyes and eyelids from the period immediately after birth to 6 weeks of age. In addition, the results of this study suggest that postnatal ocular development in Japanese white rabbits is similar to that in New Zealand white rabbits.
Ophthalmic drugs are sometimes used in children. 32 When safety evaluations of ophthalmic drugs are conducted, rabbits are generally used in nonclinical toxicology studies, 1 –3 including for intravitreal injection drugs. 33,34 There are several reports of juvenile rabbits being used for toxicological evaluations for topical ocular drugs or intravitreal injection agents. 4 –6,35 When conducting juvenile toxicity studies, it is important to distinguish the changes due to test agent administration from the normal histology of immature organs/tissues. In ocular instillation studies, the eye should be focused on the toxicological evaluation since it is directly exposed to the test articles. Knowledge of postnatal histologic development of the eyes is required for toxicologic pathologists in juvenile toxicity studies since it is necessary to distinguish whether the histological characteristics observed are drug-induced, spontaneous, or features of normal underdeveloped structures. Drastic histological changes in the cornea and other ocular surface tissues in rabbits occur immediately after the opening of the eyelids. Postnatal corneal development is associated with eyelid opening. 36 Light stimulation has been shown to affect mitosis in the corneal epithelium. 37 These studies suggest that changes in ocular surface conditions, such as exposure to light and/or the atmosphere, may affect the postnatal development of the cornea. The relationship between corneal and conjunctival development and environmental conditions remains unknown; however, the results of this study suggest that eyelid opening induces changes in the ocular surface environment and stimulates corneal and conjunctival development. In this study, after the opening of the eyelids, we noted increases in the corneal/conjunctival cell stratification and the number of conjunctival goblet cells, as well as initiation of CALT growth. Alternatively, changing the ocular surface environment through the ocular instillation of ophthalmic drugs may potentially affect postnatal development of the ocular surface. In recent years, various kinds of ophthalmic drug delivery systems have been developed. 38 The rabbit eye is an appropriate size for applying various ophthalmic drugs and conducting ophthalmological examinations.
From a regulatory standpoint, the International Conference on Harmonization M3 (R2) guidance emphasizes that rodents should be used in juvenile toxicity studies. 39 However, the eyes of mice and rats are relatively small and thus the application of drugs, particularly intravitreal administration, is difficult to perform. Furthermore, conducting ophthalmological examinations without inducing confounding lesions is also difficult due to the small globes. The eyes in nonhuman primates have a fovea and are morphologically similar to human eyes, indicating that nonhuman primates are the most suitable experimental animals to use in ocular toxicity studies to support the extrapolation of risks to humans. However, it is not always possible to obtain the requisite number of juvenile nonhuman primates to perform juvenile toxicity studies, since the reproductive efficiency of monkeys is lower than that of rodents and rabbits. Due to these reasons, rabbits are considered suitable for the nonclinical safety assessment of ophthalmic drugs. Regarding animal welfare, the use of rabbits in research is generally widely accepted and preferred over the use of dogs, a species commonly used as companion animals, and nonhuman primates, a group of animals with a greater phylogenetic relatedness to humans. This study and previous reports regarding postnatal ocular development in rabbits support the performance of juvenile toxicity studies using rabbits.
The anatomical differences between humans and laboratory animals are crucial to consider when nonclinical juvenile toxicity studies are conducted, and when the results of these toxicity studies are interpreted and the changes in laboratory animals are extrapolated to humans. In humans, the opening of the eyes occurs 20 minutes after birth, 40 whereas the eyes of laboratory rats and rabbits are known to open at PND9 to 11. 10,41 In humans, the histological development of the eyes is mostly completed during the gestational period. 42 This study showed that in rabbits, the conjunctiva, cornea, and retina continued to develop after birth, and regression of the lenticular vessels occurred during the first postnatal 2 weeks. In humans, however, events such as development of the eyelid components, flattening of the corneal endothelium, regression of the pupillary membrane and hyaloid vessels, development of the ciliary process, and formation of the retinal layers occur during the gestational period and are almost or fully complete at birth. 42,43 Therefore, for the design of nonclinical studies, it is important to recognize that the eye of the rabbit at PND9 to 11 would be developmentally equivalent to the eye of a human infant immediately after birth. In humans, ocular morphological development is completed at 2 years of age. 42 The postnatal morphological changes in the human eye occur in the iris, the optic nerve, and the foveal/macular region of the retina. 42,44 Results of sequential observations using spectral domain-optical coherence tomography support postnatal retinal development in human infants. 45,46 Human neonates primarily see shades of gray, and color discrimination begins to acquire at 3 months of age, but the cones on the fovea remain immature even at 15 months postpartum. 42 In the cornea in humans, the corneal endothelial cell density continues to decrease even after birth, 47 though these morphological changes are minimal. Taken together, these facts suggest that while topical ocular application studies using juvenile rabbits can be useful for the evaluation of ocular toxicity, including the effects of drugs on postnatal ocular histological development, the anatomical differences between humans and rabbits might affect the relevance of the risk assessments generated from these studies. In the cornea, for example, Bowman layer is rudimentary in rabbits, whereas in humans and nonhuman primates it is well developed. 2 In the posterior segment of the rabbit eye, the retinal vasculature has a merangiotic pattern, in which the vessels are confined to a broad horizontal band, whereas humans and nonhuman primates have holangiotic retina, in which the vessels extend from the optic nerve head to the periphery of the retina. 48 The retina in rabbits also lacks a fovea centralis. Monkeys, however, have a fovea centralis and its development continues even after birth. 49 These facts suggest that the use of juvenile monkeys rather than rabbits would provide more relevant data to support extrapolation of the risks associated with pediatric ophthalmic drugs. However, prenatal foveal development in monkeys is faster than that in humans, and thus the fovea is relatively mature at birth. 49 This may lead to an underestimation of the adverse effects of drugs on vision. In monkeys, the number of offspring is smaller than that in rabbits, meaning that it is hard to obtain a substantial number of juvenile animals. Considering all of these factors, the choice of the most suitable animal species for juvenile toxicity studies is still a matter of debate.
A limitation of this study is that we only examined 1 to 3 eyes per day from PND1 to PND42. Also, the histological examination of each eye was performed without considering the sex of the rabbit. No apparent histological differences between males and females were found in this study; however, the differences in postnatal ocular development between males and females were not specifically investigated. In rabbits, the testes develop until 6 months of age, suggesting that sexual maturation of male rabbits continues even after PND42. 50 The relationship between ocular development and sexual maturation was not investigated in this study. As well as sexual maturation continuing beyond 6 weeks of age, several tissues such as the lens continue to grow in size throughout life. Furthermore, the formation of CALT might dependent on the housing conditions of the animals. For a precise investigation into the continued postnatal development of the eyes and surrounding tissues, taking into account sex differences, it would be necessary to perform histological examination of a larger number of eyes in males and females throughout their whole life.
The New Zealand white rabbit is one of the main breeds employed as experimental animals. 51 The Japanese white rabbit, employed in this study, is also used experimentally including in ocular safety evaluations. 3,52 The growth in eyeball axial length, eyeball weight, and eyeball volume; lens thickness, lens weight, and lens volume; and vitreous weight and vitreous volume are similar between New Zealand and Japanese white rabbits. 24 No apparent histological differences in the eyes from PND1 to 14 were noted between this study and previous studies on the New Zealand white rabbit. 11 In addition, the timing of CALT appearance in this study was at PND11, the same as that in the New Zealand white rabbit. 21 However, the histological differences in postnatal ocular development between the New Zealand and Japanese white rabbits have not been fully investigated. Both New Zealand and Japanese white rabbits are albinos, and therefore the investigation of postnatal ocular development in pigmented breeds such as Dutch-belted rabbits will also contribute to ocular safety research since the human eye contains melanin. Functional analyses were not conducted in this study. Further examination of postnatal ocular development in rabbits will be needed in order to strengthen the rationale for the use of rabbits in juvenile ocular toxicity studies.
In summary, this study characterized the histological development of the eye in Japanese white rabbits from immediately after birth until 6 weeks of age. These results provide information to support histological evaluations in juvenile toxicity studies in rabbits, to inform age-matched comparisons of the normal histology of the rabbit eye with that of humans and other animals, to assist the design and planning of juvenile toxicity studies, and furthermore, to facilitate the extrapolation of data regarding the safety of drug use in children.
Footnotes
Authors’ Note
The authors contributed to the conception and design, data acquisition, analysis, interpretation, and drafting of the manuscript (Y.Y.); and to critically revising the manuscript (M.K., H.S.). All authors gave final approval and agreed to be accountable for all aspects of this study in ensuring that questions relating to the accuracy or integrity of any part of this work are appropriately investigated and resolved.
Acknowledgments
The authors thank Mr Yoshihiro Takei and the staff of Ina Research Inc for all their help in preparing the specimens for histological examination. Their dedication and excellent efforts made these investigations possible. The authors also thank Dr Akio Kawamura from Senju Pharmaceutical Co, Ltd for all their valuable comments to aid the production of this manuscript. The authors would like to thank Editage (![]() ) for English language editing.
) for English language editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
