Abstract
Reproductive toxicity of isobornyl acetate (IA), a widely used fragrance ingredient, was investigated in a 1-generation reproduction study in which 25 Crl: CD (Sprague-Dawley) rats/sex/group were gavaged with dosages of 0 (corn oil vehicle), 30, 100, or 300 mg/kg/d during premating, mating, gestation, and lactation. After weaning, 25 F1 generation pups/sex/dosage group were randomly selected for evaluation until sexual maturity. The following parameters were evaluated in P generation males and females: viability, clinical signs, body weights, feed consumption, mating and fertility, organ weights, gross and microscopic observations, sperm assessments (motility and concentration), natural delivery and litter observations, and ovarian follicle counts. In F1 generation pups, viability, body weights, sexual maturation, anogenital distance (days 1 and 22 postpartum), nipple eruption (day 12 postpartum), and gross necropsy observations were recorded. Isobornyl acetate did not adversely affect any of the investigated parameters. Based on the results of this investigation, the no observable adverse effect level (NOAEL) for toxicity of IA is considered to be 300 mg/kg/d. Increased incidences of excess salivation occurred in P generation male and female rats at 100 and/or 300 mg/kg/d throughout the dosage period, and low incidences of urine-stained abdominal fur were seen in females at 300 mg/kg/d during the gestation period. These clinical signs were not considered as adverse effects of IA administration. Thus, the NOAEL for reproductive toxicity in the P generation rats and the NOAEL for viability and growth of the F1 generation offspring is considered to be ≥300 mg/kg/d.
Introduction
Isobornyl acetate (IA; C12H20O2) is an aliphatic ester with an odor of pine needles and soluble in fixed oils and mineral oils. The molecular structure is presented in Figure 1. It is a fragrance ingredient widely used in toiletries, soaps, and antiseptics and as a flavoring agent. Its worldwide volume of use is greater than 1,000 metric tons/y. 1 The toxicology of this compound has been extensively documented in a variety of animal species, 2 and the Flavor and Extract Manufacturers Association has classified IA as generally recognized as safe when used as a flavor ingredient. 3
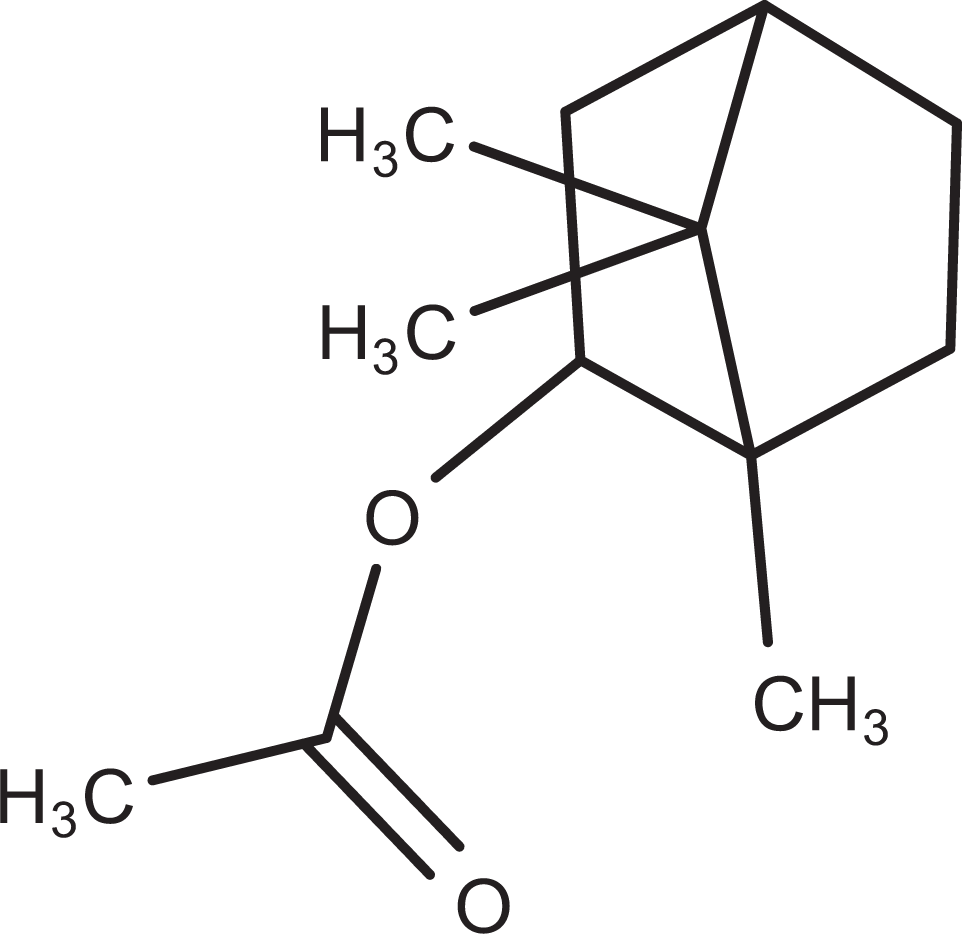
Structural formula of isobornyl acetate.
The purpose of the present study was to provide information regarding the effects of IA on gonadal function, estrous cycles, mating behavior, conception, parturition, lactation, and the growth and development of offspring up to day 60 postpartum. The study was also designed to provide information about the possibility of potential teratogenesis.
Dosages were selected on the basis of a dosage range reproduction study in which 8 rats/sex/dosage group were gavaged with 0 (vehicle), 5, 15, 45, or 135 mg/kg/d IA in a corn oil vehicle. 4 Dosing occurred 14 days prior to cohabitation, through cohabitation, gestation, and postpartum day (PPD) 5 or the day of euthanasia. There were no treatment-related effects in male rats. In female rats, maternal body weight gains were reduced in the 45 and 135 mg/kg/d dosage groups during gestation, but all pregnant rats delivered a litter and there were no effects on natural delivery or litter observations. There were no effects on pup body weights.
Based on these data, a high dose of 300 mg/kg/d was proposed for the final study to ensure that sufficient toxicity would be elicited. Middle and low dosages were selected in 3-fold increments.
Materials and Methods
Materials
Isobornyl acetate, CAS# 125-12-2, is a clear, colorless liquid (lot number J145) supplied by Takasago International Corp, Rockleigh, New Jersey. Corn oil (Charkit Chemical Corp, South Norwalk, Connecticut) was the vehicle and administered to control group rats. Formulations (suspensions) were prepared at least once weekly at the testing facility and a 10-day stability of IA was established. Both bulk substances and prepared formulations were stored at room temperature and protected from light. Prepared formulations were stirred continuously prior to and during dosage administration.
Animals
Crl: CD(SD) male and female rats (Charles River Laboratories, Inc, Portage, Michigan) were used in the study. After a short acclimation period, the P generation rats were randomly assigned to 4 groups using a computer-generated (weight-ordered) randomization procedure based on body weights. At study assignment, female rats weighed 212 to 253 g, while the male rats weighed 133 to 159 g. Each rat was housed individually in stainless steel, wire-bottomed cages, except during cohabitation when each pair of male and female rats were housed in the male rat’s cage. Beginning no later than gestation day 20 (GD 20), P generation females were individually housed in nesting boxes until they either naturally delivered litters or were euthanized on GD 25. Each dam and delivered litter was housed in a common nesting box during the postpartum period.
All rats were given ad libitum access to Certified Rodent Diet #5002 meal (PMI Nutrition International, Inc, St Louis, Missouri) in individual feeders. Water was available to the rats ad libitum from an automatic watering access system and/or individual water bottles attached to the cages. Chlorine was added to the reverse osmosis membrane (RO water) processed water as a bacteriostat. Bed-o’cobs bedding (The Andersons Industrial Products Group, Maumee, Ohio) was used as the nesting material. Chewable Nylabones (BioServe, Frenchtown, NJ) were supplied to all rats during the course of the study.
All cage sizes and housing conditions were in compliance with the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources 1996). 5 The study room was independently supplied with at least 10 changes/h of 100% fresh air passed through 99.97% high-efficiency particulate air (HEPA) filters. Room temperature and humidity were monitored constantly throughout the study. The room temperature was targeted at 66°F to 77°F (19°C-25°C) and relative humidity was targeted at 30% to 70%. An automatically controlled 12-hour light and 12-hour dark fluorescent light cycle was maintained. Each dark period began at 1900 hours.
Methods
The requirements of the Organization for Economic Cooperation and Development Guidelines for Testing of Chemicals No. 415 6 were used as the basis for study design and conducted according to Good Laboratory Practices (GLP) regulations. 7 One hundred P generation male and 100 P generation female rats were assigned to 4 dosage groups, 25 rats/sex/dosage group, for treatment with the vehicle or test substance. Suspensions of the test substance, IA, or the vehicle, corn oil, were administered via gavage to the P generation male rats once daily beginning 84 days before the cohabitation period, through the cohabitation period (maximum of 14 days), and continuing until the day before euthanasia; and to the P generation female rats once daily beginning 14 days before the cohabitation period, through the cohabitation period (maximum of 14 days), and continuing through the day of euthanasia (through GD 25 for rats that did not deliver or lactation day [LD] 22 for rats that delivered a litter). Dosage levels of IA were 0 (vehicle), 30, 100, or 300 mg/kg/d. The dosage volume was 4 mL/kg, which was adjusted weekly according to individual body weights. The presence of spermatozoa and/or a copulatory plug was designated as GD 0.
The following parameters were evaluated in P generation males and females: viability, clinical observations, body weights, food consumption, mating and fertility, organ weights, and gross and histopathological observations of any lesions. In P generation male rats, sperm assessments (motility and concentration) were also evaluated, while in P generation female rats, estrous cycling, natural delivery, and litter observations, as well as ovarian follicle counts, were evaluated. In F1 generation pups, viability, body weights, gross observations, sexual maturation, anogenital distance on PPDs 1 and 22, and nipple eruption on PPD 12 were evaluated.
Data generated during the course of this study were recorded either by hand or using the Argus Automated Data Collection and Management System, the Vivarium Temperature and Relative Humidity Monitoring System, and the Hamilton Thorne IVOS. All data were tabulated, summarized, and/or statistically analyzed using the Argus Automated Data Collection and Management System, the Vivarium Temperature and Relative Humidity Monitoring System, Microsoft Excel (part of Microsoft Office 97/2000/2003/XP), Quattro Pro 8 and The SAS System (version 6.12). Clinical observations and other proportional data were analyzed using the variance test for homogeneity of the binomial distribution. 8 Continuous data were analyzed using the Bartlett test of homogeneity of variances 9 and analysis of variance (ANOVA). 10 Dunnett test 11 was used to identify statistical significance of individual groups. If the ANOVA was not appropriate, the Kruskal-Wallis test 12 or Dunn test 13 was used to identify the statistical significance of individual groups. If there were >75% ties, Fisher exact test 14 was used. Group means and standard deviations of primordial follicles were calculated and compared across groups using the Kruskal-Wallis nonparametric ANOVA test. If a significant result occurred (P < 0.05), the Wilcoxon (Mann-Whitney U) test 15 was used for pairwise group comparison to controls.
Results
Mean IA concentrations for all dose formulations were within the acceptable limits (≤15% difference from nominal concentration). Homogeneity was acceptable (≤5% relative standard deviation [RSD]) for all dose formulations. Dosage formulations were within acceptable stability limits (±10% of initial mean measured concentration) after 14 days.
P Generation Male Rats
No mortality related to the test substance occurred during the study. One male rat in the vehicle control group was found dead on study day (SD) 42 following an apparent gavage accident and 1 male rat in the 100 mg/kg/d group was euthanized on SD 107 with adverse clinical signs (scab, ulceration, localized alopecia, abrasion on the neck and red discharge from the neck; in addition, localized alopecia on the limbs and underside were also observed; and the rat was apprehensive when or at handling) all unrelated to administration of the test substance.
The only clinical sign attributed to treatment with IA included slight and moderate increase in salivation at 300 mg/kg/d. Among males, slight increase in salivation was observed as early as SD 27 and occurred in 22 of 25 males in the 300 mg/kg/d dosage group. Moderate increase in salivation occurred in 6 of 25 males in the same dosage group. Among females, slight increase in salivation was observed as early as SD 2 and persisted into the lactation period; the clinical sign was more pronounced during the pre-cohabitation and gestation periods.
Absolute (g/d) and relative (g/kg/d) food consumption, as well as body weights and body weight gains, were unaffected by IA dosages as high as 300 mg/kg/d. Mean body weight on SD 112 (termination of treatment) was 103%, 104%, and 103% of vehicle controls in the 30, 100, and 300 mg/kg/d groups, respectively (Table 1).
Terminal Body Weights and Organ Weights—P Generation Male Rats.a
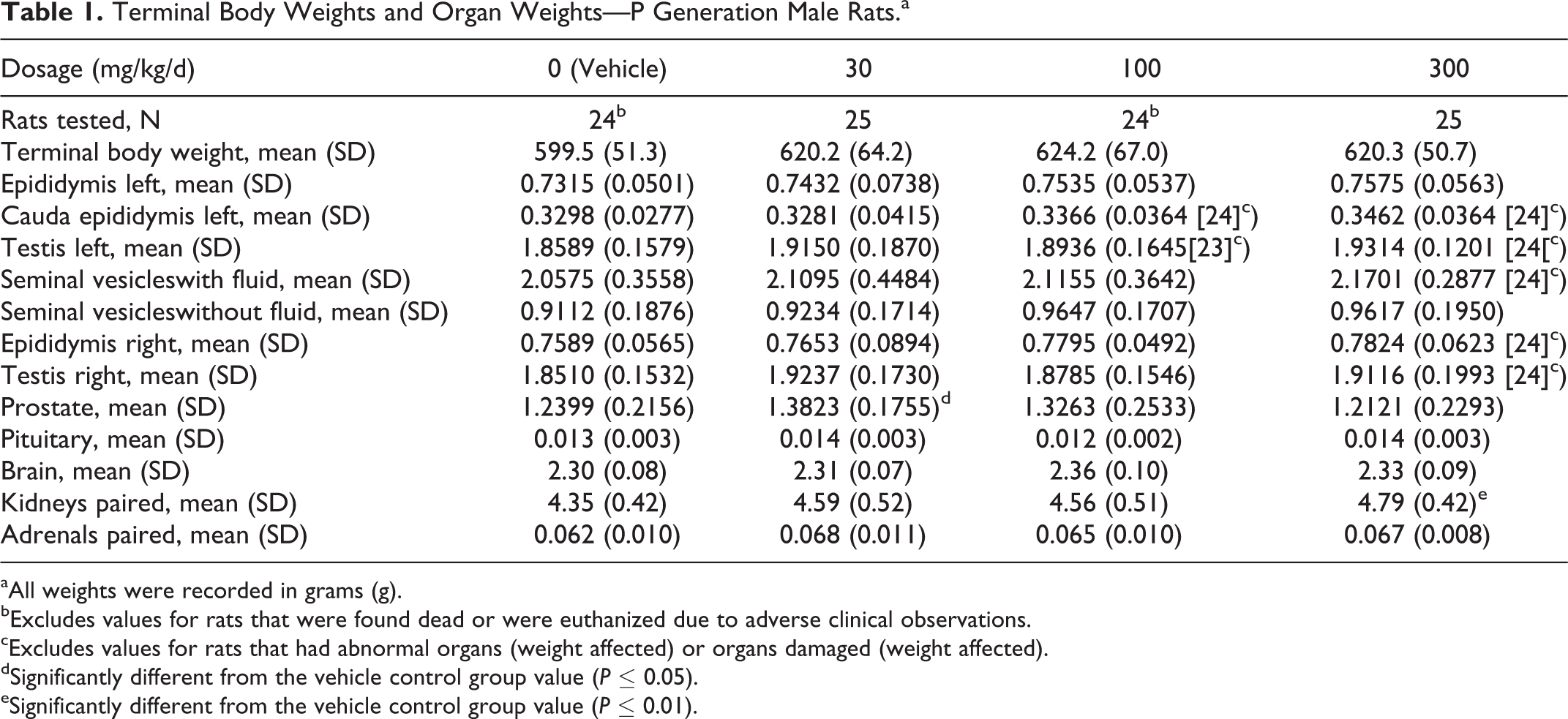
aAll weights were recorded in grams (g).
bExcludes values for rats that were found dead or were euthanized due to adverse clinical observations.
cExcludes values for rats that had abnormal organs (weight affected) or organs damaged (weight affected).
dSignificantly different from the vehicle control group value (P ≤ 0.05).
eSignificantly different from the vehicle control group value (P ≤ 0.01).
All mating and fertility parameters (numbers of days in cohabitation, number rats that mated, the fertility index [number of pregnancies per number of rats that mated], rats with confirmed mating dates during the first or second weeks of cohabitation and number of pregnancies per number of rats in cohabitation) were unaffected by IA dosages as high as 300 mg/kg/d. All values were comparable among the 4 dosage groups and did not significantly differ from the vehicle control group.
There were no test substance–related necropsy observations. All gross lesions were considered unrelated to treatment because (1) the incidences were not dosage dependent, (2) the findings occurred in only 1 rat in any dosage group, and/or (3) the finding occurred only in rats that were euthanized early. Gross lesions among male rats observed included, enlarged mandibular lymph nodes, an edematous thymus, a perforation in the esophagus, the presence of a white substance in the thoracic cavity, a dark red appearance to the lungs, slight dilation of the pelvis in both kidneys, a dark red appearance to the liver, an enlarged left testis, small testes or epididymides, and a small left cauda epididymis.
The epididymides, caudal epididymis, testes, seminal vesicles (with and without fluid), prostate, pituitary, brain, paired kidneys, and paired adrenals were weighed (Table 1), and the ratios of these organ weights to terminal body weight were assessed. A statistically significant increase (P ≤ 0.05) in the absolute weight of the prostate at 30 mg/kg/d and a statistically significant increase (P ≤ 0.01) in the absolute weight of the paired kidneys at 300 mg/kg/d were noted but were not attributed to IA because (1) the increase was not dosage dependent, (2) there was no change in the relative weight of the affected organs, (3) the kidney weights were not affected in the P generation female rats, and (4) there were no microscopic correlates that could account for the increased organ weight.
All evaluated sperm parameters were unaffected by IA dosages as high as 300 mg/kg/d (Table 2). Values for number and percentage of motile sperm, number of nonmotile sperm, total sperm count from the vas deferens, and cauda epididymal sperm count and density were comparable among the 4 dosage groups and did not significantly differ from the vehicle control group values.
Sperm Motility, Count and Density—Summary of P Generation Male Rats.
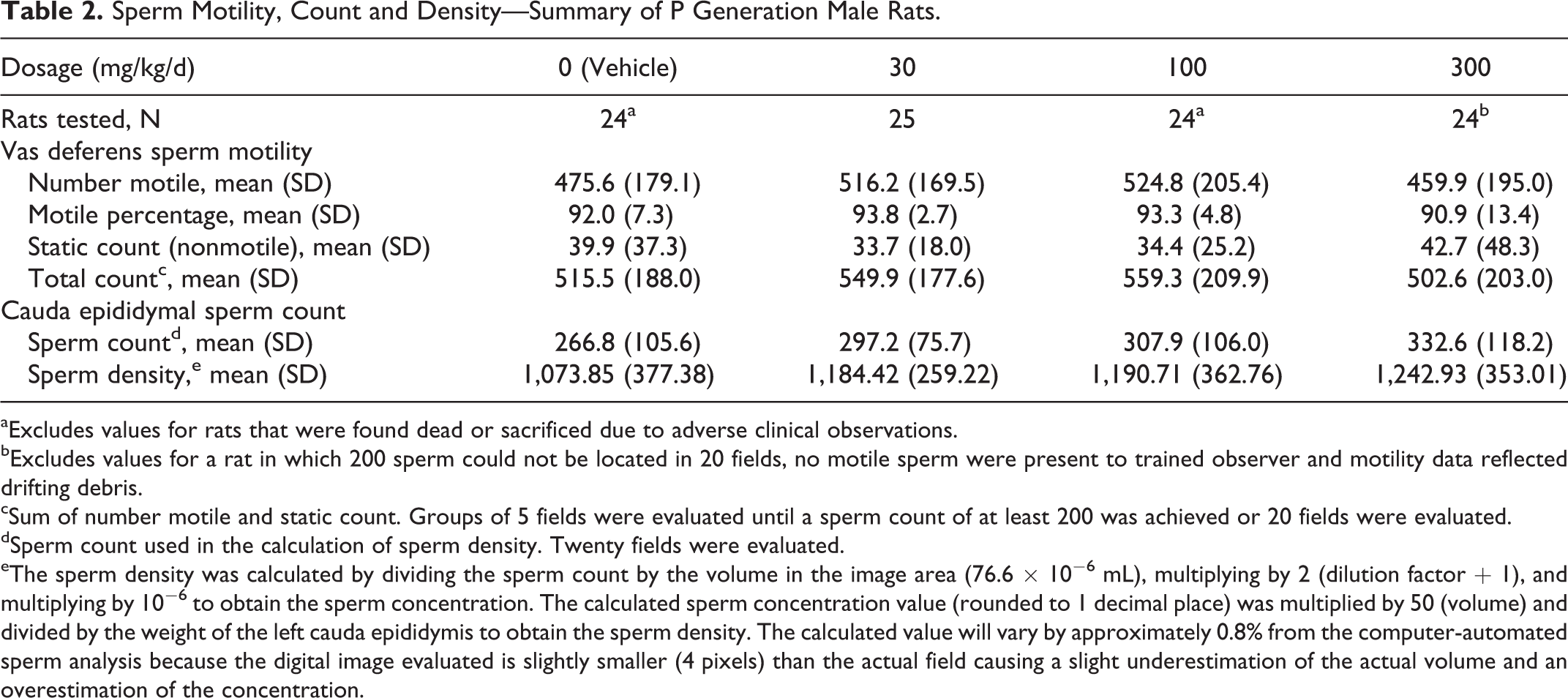
aExcludes values for rats that were found dead or sacrificed due to adverse clinical observations.
bExcludes values for a rat in which 200 sperm could not be located in 20 fields, no motile sperm were present to trained observer and motility data reflected drifting debris.
cSum of number motile and static count. Groups of 5 fields were evaluated until a sperm count of at least 200 was achieved or 20 fields were evaluated.
dSperm count used in the calculation of sperm density. Twenty fields were evaluated.
eThe sperm density was calculated by dividing the sperm count by the volume in the image area (76.6 × 10−6 mL), multiplying by 2 (dilution factor + 1), and multiplying by 10−6 to obtain the sperm concentration. The calculated sperm concentration value (rounded to 1 decimal place) was multiplied by 50 (volume) and divided by the weight of the left cauda epididymis to obtain the sperm density. The calculated value will vary by approximately 0.8% from the computer-automated sperm analysis because the digital image evaluated is slightly smaller (4 pixels) than the actual field causing a slight underestimation of the actual volume and an overestimation of the concentration.
P Generation Female Rats
No mortality related to the test substance occurred during this study. One female rat in the 100 mg/kg/d dosage group and 1 in the 300 mg/kg/d dosage group were euthanized during the gestation period due to adverse clinical observations (vocalization and an apparent tail injury in the mid-dose rat and signs of dystocia, including delivery of 9 live pups, and the presence of 5 dead fetuses in utero in the high-dose rat). These findings were not considered to be related to the test substance because they occurred only in single animals and were dissimilar in nature.
Similar to male rats, the number of P generation female rats with excess salivation (slight and/or moderate) was significantly increased (P ≤ 0.01) at 300 mg/kg/d, in comparison to the vehicle control group values. Slight excess salivation was observed as early as SD 2 and persisted into the lactation period, although it was more pronounced during pre-cohabitation and gestation and was also accompanied by a low incidence of transient urine-stained abdominal fur (P ≤ 0.01; N = 4 out of 25) during the gestation period. At 100 mg/kg/d, excess salivation was significantly increased (P ≤ 0.01) only during gestation, in comparison to the vehicle control group value. All other clinical observations during the pre-cohabitation, gestation, and lactation periods were considered unrelated to IA.
Absolute (g/d) and relative (g/kg/d) food consumption, body weights, and body weight gains during the pre-cohabitation, gestation, and lactation periods were unaffected by IA dosages as high as 300 mg/kg/d (see Figure 2 for body weight graph). All values were comparable among the 4 dosage groups. The statistically significant increase (P ≤ 0.05) in absolute and relative food consumption that occurred in the 100 and 300 mg/kg/d dosage groups on LDs 8 to 11 were considered unrelated to the test substance because the increases were transient and not dosage dependent (Table 3). Statistically significant increase (P ≤ 0.05) in mean body weight at 100 mg/kg/d on SD 8, statistically significant increases (P ≤ 0.05 or P ≤ 0.01) in body weight gains at 100 and 300 mg/kg/d on SDs 5 to 8 and a statistically significant increase (P ≤ 0.01) in body weight gains at 300 mg/kg/d for the gestation period (GDs 1-22) also were considered unrelated to the test substance because the increases were not dosage dependent and/or reflected an overall reduction in body weight gains or net loss in body weight in the corresponding vehicle control group.
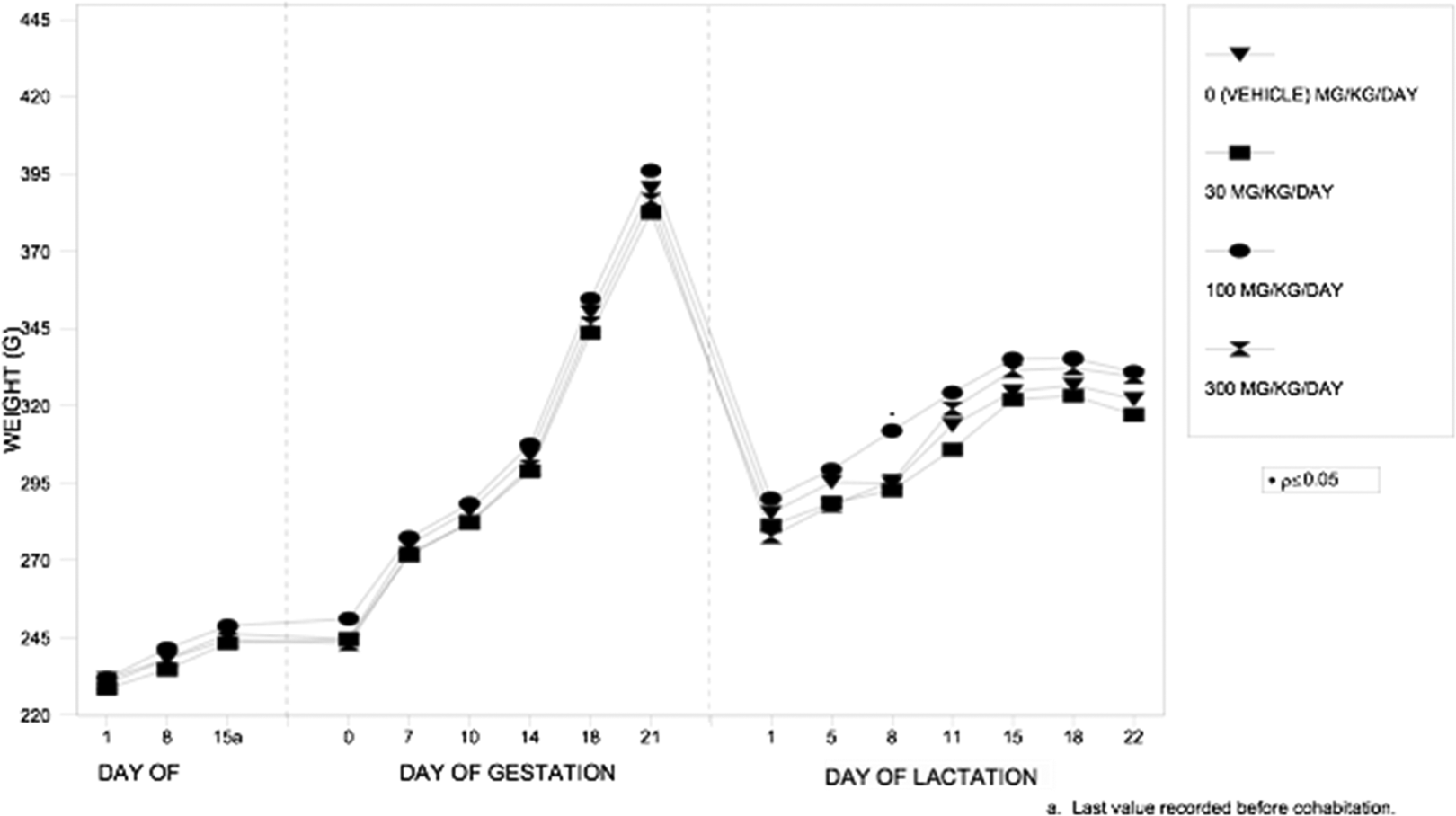
P generation female body weights administered isobornyl acetate via gavage at doses of 0, 30, 100, or 300 mg/kg/d.
Maternal Absolute Feed Consumption Values (g/d)—Lactation—Summary for P Generation Female Rats.
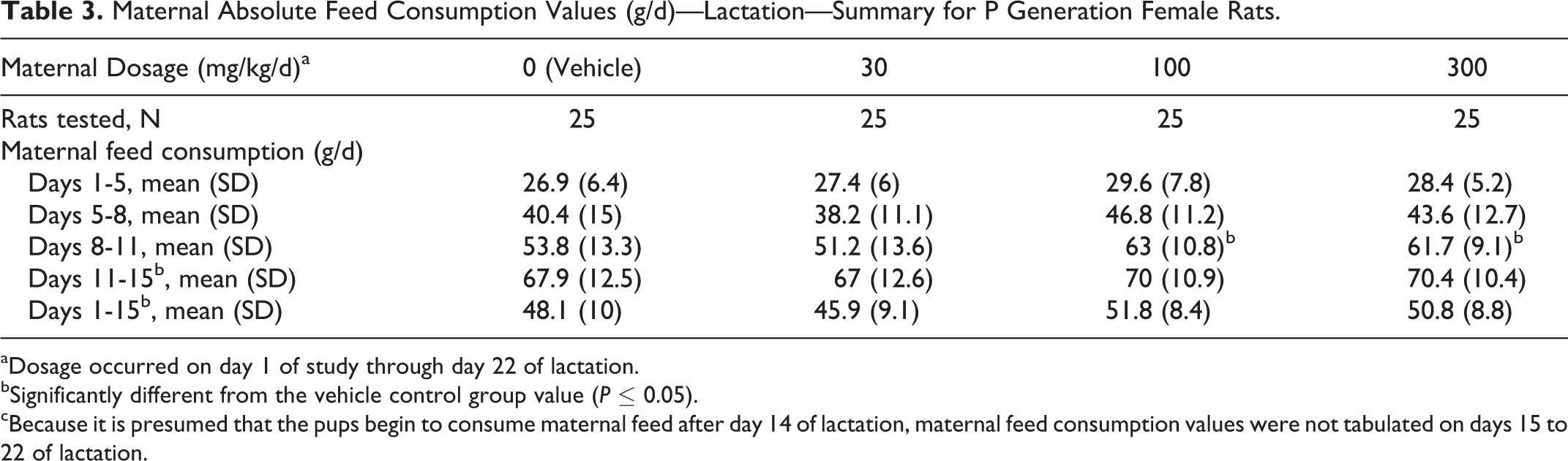
aDosage occurred on day 1 of study through day 22 of lactation.
bSignificantly different from the vehicle control group value (P ≤ 0.05).
cBecause it is presumed that the pups begin to consume maternal feed after day 14 of lactation, maternal feed consumption values were not tabulated on days 15 to 22 of lactation.
All mating and fertility parameters (numbers of days in cohabitation, rats that mated, the fertility index [number of pregnancies per number of rats that mated], rats with confirmed mating dates during the first or second week of cohabitation and number of pregnancies/rats in cohabitation) were unaffected by IA dosages as high as 300 mg/kg/d. All values were comparable among the 4 dosage groups and did not significantly differ from the vehicle control group.
Pregnancy occurred in 23, 22, 24, and 22 of the 25 mated P generation female rats in the 0 (vehicle), 30, 100, and 300 mg/kg/d dosage groups, respectively. All pregnant dams delivered litters. Natural delivery and lactation parameters, as well as litter observations, were unaffected by IA dosages as high as 300 mg/kg/d (Table 4).
Pregnancy Results and Litter Data Through Lactation Period.a
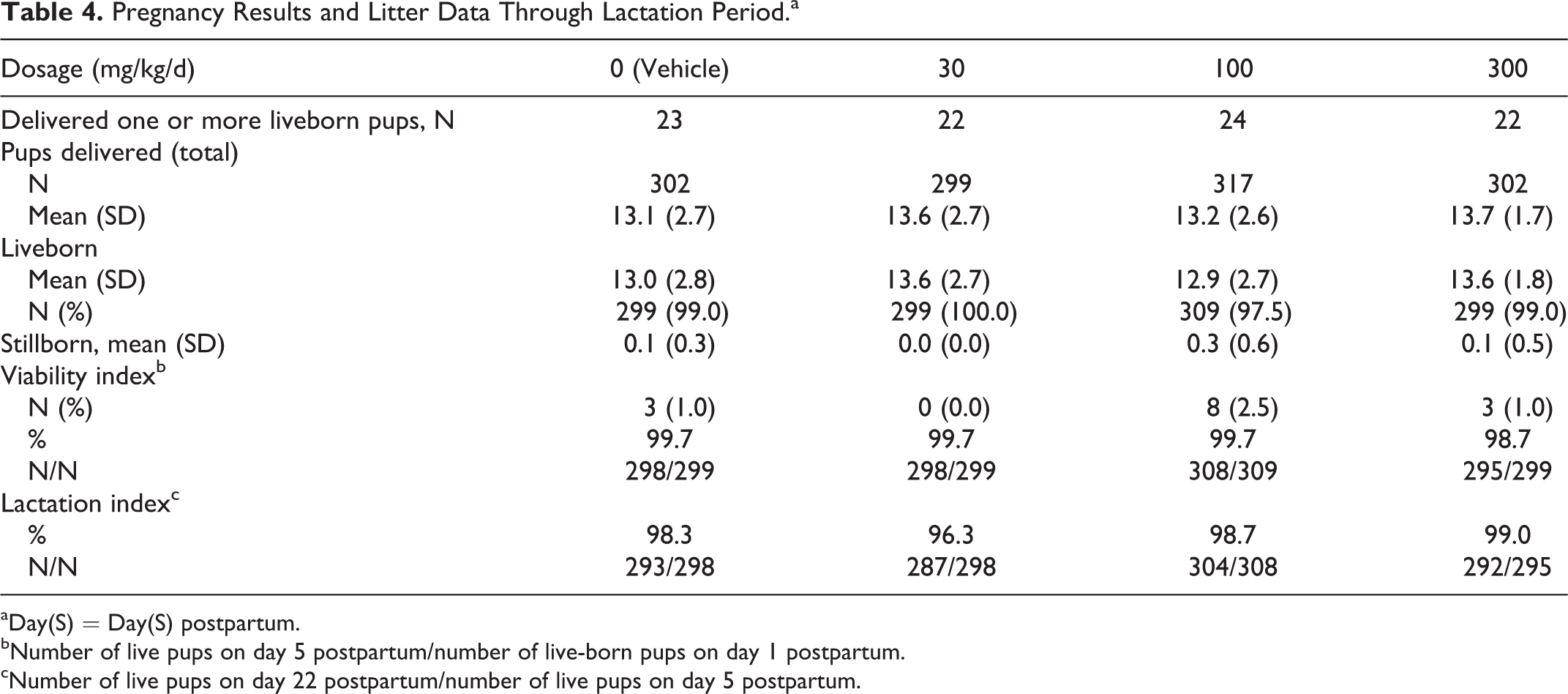
aDay(S) = Day(S) postpartum.
bNumber of live pups on day 5 postpartum/number of live-born pups on day 1 postpartum.
cNumber of live pups on day 22 postpartum/number of live pups on day 5 postpartum.
Terminal body weights were comparable among the 4 dosage groups and did not significantly differ from the vehicle control group value. There were no test substance–related necropsy observations. The weights of the left and right ovaries and the uterus with cervix and the ratios of these organ weights to terminal body weight were unaffected by IA dosages as high as 300 mg/kg/d. In addition, there were no apparent effects on the weights of the nonreproductive organs that were evaluated (ie, pituitary, brain, liver, paired kidneys, and paired adrenals) at scheduled euthanasia (Table 5).
Terminal Body Weights and Organ Weights—P Generation Female Rats.a
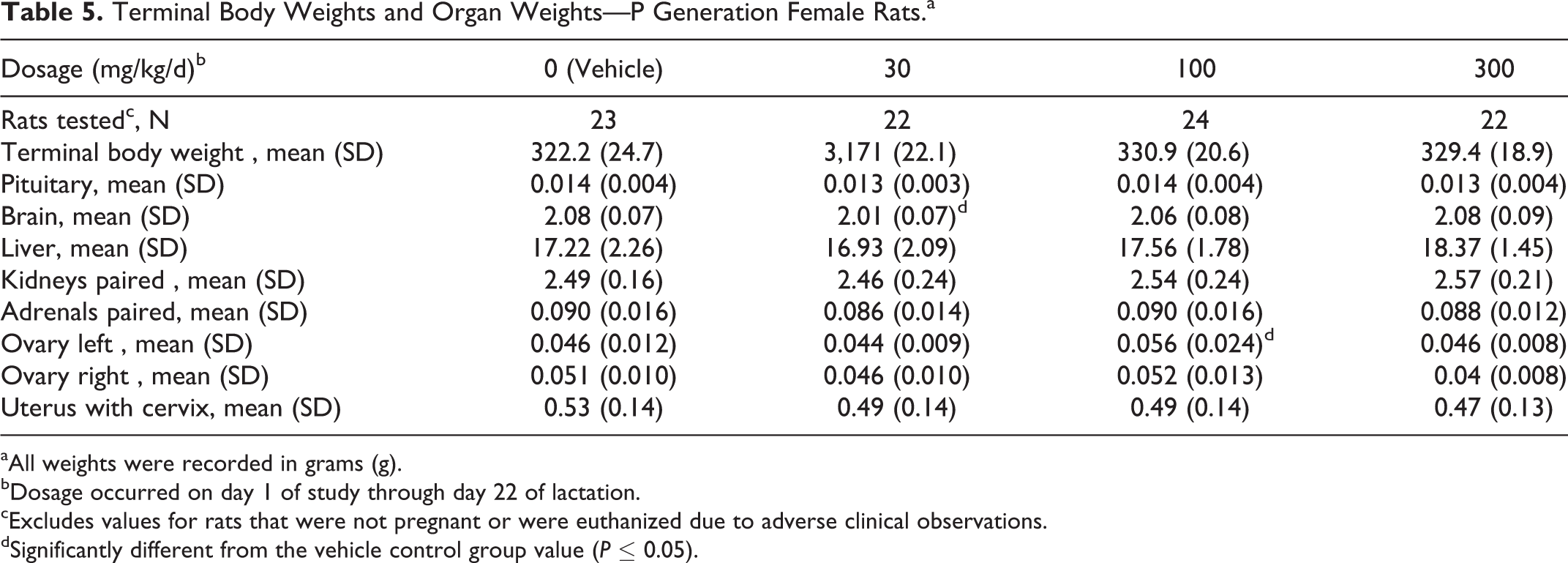
aAll weights were recorded in grams (g).
bDosage occurred on day 1 of study through day 22 of lactation.
cExcludes values for rats that were not pregnant or were euthanized due to adverse clinical observations.
dSignificantly different from the vehicle control group value (P ≤ 0.05).
No test substance–related histopathology was noted. The microscopic findings observed were considered incidental, of a nature commonly observed in this strain and age of rats, and/or were of similar incidence and severity in control and treated rats and were considered unrelated to IA administration. There were no statistically significant differences in the number of primordial ovarian follicles in left, right, or both ovaries combined, in P generation female rats administered 300 mg/kg/d IA when compared to vehicle control rats. Corpora lutea were present in all female rats evaluated.
F1 Generation Pups
At birth and during the lactation period, there were no treatment-related clinical signs observed in the F1 generation pups following the treatment of the P generation rats with IA at dosages as high as 300 mg/kg/d. All transient and persistent clinical observations, including missing digits on the left or right hind paw and a necrotic appearance to the right hind paw, were considered unrelated to IA because the incidences were not dosage dependent and/or the number of litters affected did not differ significantly from vehicle control group values.
Anogenital distance on PPD 1 or 22 in F1 generation male and female pups was not affected by treatment of the P generation rats with IA at any dosage level tested. Nipple eruption did not occur in any F1 generation male pup at any dosage level tested. All F1 generation female pups had nipples present on PPD 12 (Table 6).
Anogenital Distance Measurement and Nipple Eruption—F1 Generation Pups.

On postpartum day 1, an ocular micrometer (µm) was used, and it was arbitrarily determined that 10 units were equivalent to 1 mm, and the values were reported as whole µm units. On postpartum day 22, values were recorded in millimeters to tenths precision using a vernier caliper.
aExcludes values for litters that appeared incorrectly recorded or were incorrectly sexed.
There were no test substance–related gross lesions observed in F1 generation pups that survived to scheduled necropsy on PPD 22 (not selected for continued evaluation). One F1 generation pup in the 300 mg/kg/d dosage group had complete situs inversus; all other pups appeared normal.
No deaths related to IA occurred in the F1 generation rats during the postweaning period (1 F1 generation male rat in the 100 mg/kg/d dosage group was euthanized on PPD 63 because of a broken palate). All other F1 generation rats selected for continued evaluation during the 36- to 42-day postweaning period survived until scheduled euthanasia. Treatment of P generation rats with IA dosages as high as 300 mg/kg/d produced no significant changes in absolute and relative food consumption, body weights, and body weight gains for F1 generation male and female rats during the postweaning period when compared to vehicle controls.
Sexual maturation in F1 generation rats was unaffected by treatment of P generation rats with IA dosages as high as 300 mg/kg/d. The average day on which preputial separation was observed in F1 generation male rats (46-47 days) and vaginal opening was observed in F1 generation female rats (34-35 days) was comparable among the dosage groups. In addition, the average body weight on the day sexual maturation was achieved in F1 generation male and female rats was comparable among the dosage groups and did not differ significantly from vehicle control group values (Table 7).
Sexual Maturation of Pups—F1 Generation.
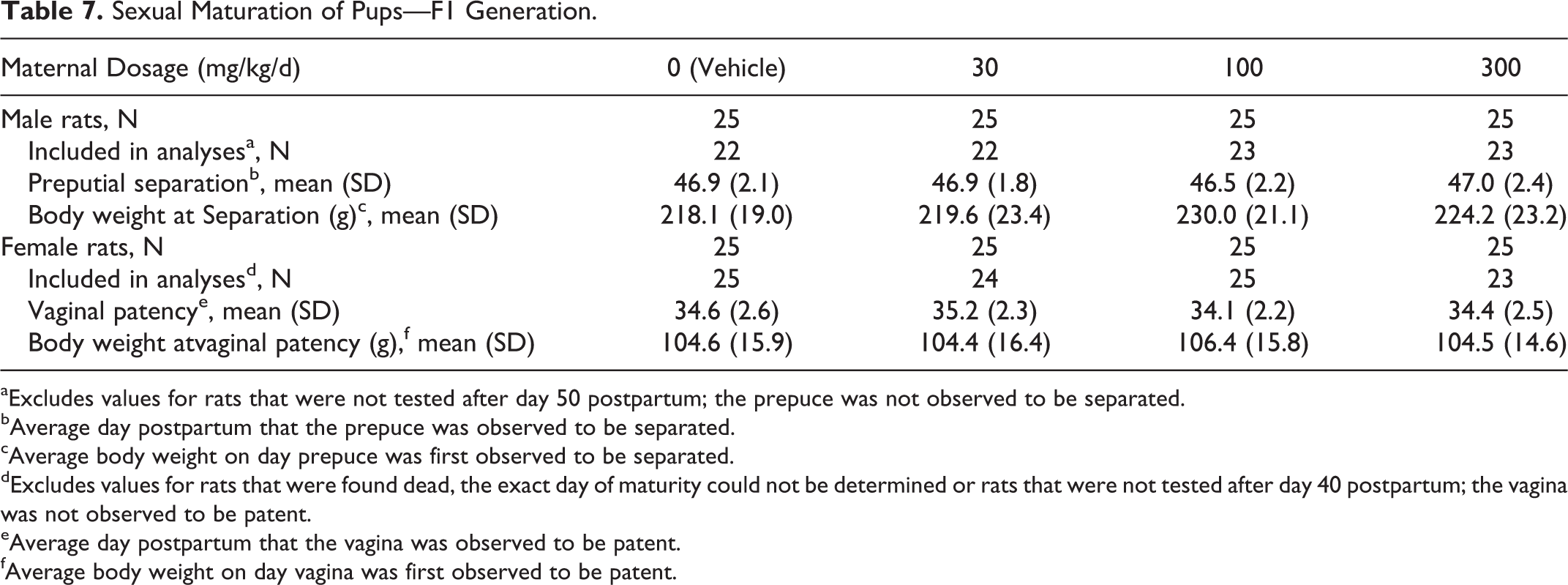
aExcludes values for rats that were not tested after day 50 postpartum; the prepuce was not observed to be separated.
bAverage day postpartum that the prepuce was observed to be separated.
cAverage body weight on day prepuce was first observed to be separated.
dExcludes values for rats that were found dead, the exact day of maturity could not be determined or rats that were not tested after day 40 postpartum; the vagina was not observed to be patent.
eAverage day postpartum that the vagina was observed to be patent.
fAverage body weight on day vagina was first observed to be patent.
There were no test substance–related necropsy findings or apparent effects of treatment from P generation rats with IA on the reproductive (for males, prostate, seminal vesicles, and testes were examined; for females, uterus with cervix and ovaries were examined) and nonreproductive tissue weights (brain, paired kidneys, paired adrenals) of the F1 generation male or female rats at any dosage level. A statistically significant reduction (P ≤ 0.05) in the relative (percentage of terminal body weight) weight of the paired adrenal glands occurred in F1 generation male rats at 100 mg/kg/d, in comparison to the vehicle control value. This reduction was considered unrelated to treatment of P generation rats with IA because there was no statistically significant change in the absolute weight of this organ; the reduction did not occur in the F1 generation female rats; and the increase was not dosage dependent.
Discussion
The present study was conducted to confirm the unpublished findings of the reproduction range finding study 3 and to provide additional information on the effects of IA on gonadal function and the growth and development of offspring up to 60 days postpartum. Based on the results of this study, the no observable adverse effect level (NOAEL) for toxicity of IA is considered to be 300 mg/kg/d.
Increased incidences of excess salivation occurred in P generation male and female rats at 100 and/or 300 mg/kg/d throughout the dosage period, and low incidences of urine-stained abdominal fur occurred in females at 300 mg/kg/d during the gestation period; these effects were considered dosing factors, not adverse effects of the test substance.
The IA did not affect body weight (including terminal body weights), body weight gains, or food consumption values in the P generation rats and did not cause any mortality or increase the incidence of gross or histologic pathology findings at any dosage level. However, significantly increased absolute kidney weights were observed in male P generation rats at 300 mg/kg/d, an effect that was also described only in male rats in an early 13-week study, 16 using gavaged doses of 270 mg/kg/d of IA. These doses produced a decrease in renal concentrating ability, an increase in water intake, and exfoliation and vacuolation of renal tubular cells. Signs of nephrotoxicity were also noted with daily doses of 90 mg/kg but not with 15 mg/kg/d. Histological examination of all kidneys from control and high-dose rats in the present study revealed no histopathological changes and relative organ weights for the kidneys when compared to vehicle control rats; therefore, the mean increase in absolute kidney weights in the high-dose male rats was not considered to be an adverse effect caused by IA.
The reproductive NOAEL in the P generation rats and the NOAEL for viability and growth of the F1 generation offspring is ≥300 mg/kg/d. There were no apparent effects on estrous cycling, mating, fertility, reproductive organ weights, natural delivery parameters in the P generation, and growth and development (including anogenital distance, nipple eruption, or sexual maturation) in the F1 generation rats at the highest dosage level tested (300 mg/kg/d). These NOAELs indicate that a safety margin of nearly 2,000 exists between the calculated maximum daily human exposure (covering dermal, oral, and inhalation routes) of 0.1523 mg/kg/d 17 to IA from dermally applied fragranced cosmetic products and the possibility of an adverse maternal or fetal exposure. 18
Footnotes
Authors’ note
The study was conducted at Charles River Laboratories Preclinical Services, Horsham, Pennsylvania, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: funded by the Research Institute for Fragrance Materials, Inc, Woodcliff Lake, New Jersey, USA.
