Abstract
The reproductive toxicity potential of Ammonium Salt of Perfluorinated Hexanoic Acid (PFHxA Ammonium Salt) in pregnant Crl: CD1(ICR) mice was investigated. Twenty females/group were administered the test substance or vehicle once daily from gestation day 6 through 18. Phase 1 doses: 0, 100, 350, and 500 mg/kg/d; phase 2: 0, 7, 35, and 175 mg/kg/d. Parameters evaluated include mortality, viability, body weights, clinical signs, abortions, premature deliveries, pregnancy and fertility, litter observations, maternal behavior, and sexual maturity in the F1 generation. The level of PFHxA Ammonium Salt was measured in the liver of F0 and F1 mice. At doses of 350 and 500 mg/kg/d maternal mortalities, excess salivation and changes in body weight gains occurred. Pup body weights were reduced on postpartum day (PPD) 0 in all the dosage groups, but persisted only in the 350 and 500 mg/kg/d groups. Additional effects at 300 and 500 mg/kg/d included stillbirths, reductions in viability indices, and delays in physical development. Levels of PFHxA Ammonium Salt in the livers of the 100 mg/kg/d dams were all below the lower limit of quantization (0.02 µg/mL); in the 350 mg/kg/d group, 3 of the 8 samples had quantifiable analytical results. In phase 2 no PFHxA Ammonium Salt was found in the liver. Adverse effects occurred only in the 175 mg/kg/d group and consisted of increased stillborn pups, pups dying on PPD 1, and reduced pup weights on PPD 1. Based on these data, the maternal and reproductive no observable adverse effect level of PFHxA Ammonium Salt is 100 mg/kg/d.
Introduction
Perfluorohexanoic acid (PFHxA) is a highly stable, ultimate degradant of the class of “C6” surfactant chemicals which is currently being developed to be used as a durable water and oil repellent for textiles, carpets, nonwoven, and paper applications. In the environment, any activity of PFHxA will undoubtedly be due to the dissociated PFHxA ion. Since the PFHxA salt readily dissociates in water to yield the PFHxA ion, it is a good vehicle to test the toxicity of “C6” chemicals without introducing a confounding factor of acid toxicity at higher concentrations that are not environmentally relevant. Further, in some applications, the PFHxA Ammonium Salt itself may find application as an industrial surfactant.
The developmental toxicity of perfluoroalkyl acids (PFAAs) has been extensively studied. 1,2 The mouse has been used to evaluate the developmental toxicity of other PFAAs including perfluorooctanic acid (PFOA). Unlike the rat, sex differences in exposure levels in the mouse do not appear to exist. 3 Although only female mice were evaluated in this study, the mouse appears to be an appropriate and sensitive model for evaluation of PFHxA. Developmental toxicity observed in rodents showed a lack of teratological findings (structural anomalies) from PFHxA. Effects on pup survival and postnatal growth have been consistently observed in both rats and mice in a dose-dependent pattern. 4 The general toxicity and toxicokinetics of PFHxA were investigated in a 90-day repeat-dose toxicity and metabolism studies in rats. 5,6 The toxicity and toxicokinetics of other PFHxA, including sodium perfluorohexanoate and ammonium perfluorohexanoate, have also been studied. 7,8
The purpose of this research, conducted in 2 phases, was to test for toxic effects resulting from PFHxA Ammonium Salt treatment of Crl: CD1(ICR) pregnant female mice and evaluate the development of the embryo and fetus consequent to exposure of the dam from implantation to closure of the hard palate and during lactation. This study was designed to evaluate the International Conference on Harmonisation Harmonised Tripartite Guideline stages C through F of the reproductive process and detect effects on gestation, parturition, lactation, and maternal behavior in female mice and on the development of the offspring of the treated female mice. Because manifestations of effects induced during this period may be delayed in the offspring, observations were continued through sexual maturity of the F1-generation mice.
These studies were conducted in compliance with the Good Laboratory Practice (GLP) regulations of the US Environmental Protection Agency, 9 the Japanese Ministry of Agriculture, Forestry and Fisheries, 10 and the Organization for Economic Cooperation and Development (OECD). 11 The pharmacokinetic analysis and analytical portion of the study were conducted in accordance with the appropriate Food and Drug Administration and OECD Principles of GLP (ENV/MC/CHEM(98)17). 11
The data presented here are from research conducted in 2 phases. The first phase used doses of the test substance that did not allow determination of a clear no observable adverse effect level (NOAEL). The second phase was conducted at lower doses to determine a clear NOAEL.
Materials and Methods
Animals and Husbandry
The methodology was comparable for each phase with the exception of the dosage levels. Eighty Crl: CD1(ICR; Charles River Laboratories, Inc, St Constant Canada or Kingston, New York) mice were used in each phase. Mice were approximately 61 to 63 days of age and weighed 24 to 33 g at arrival to the Testing Facility. They were bred with male mice of the same strain and source that were maintained as a breeding colony at the Testing Facility. Female F0-generation mice were given temporary numbers at receipt and given permanent identification numbers when assigned to the study on the basis of body weights obtained on the first day of gestation (day 0). Female mice were permanently identified using a tail tattoo. Pups were not individually identified during the lactation period; all parameters were evaluated in terms of the litter. At weaning, F1-generation mice were identified by tail tattoo.
All husbandry, cage sizes, and housing conditions were in compliance with the Guide for the Care and Use of Laboratory Animals. 12 The study rooms were maintained under conditions of positive airflow relative to a hallway and independently supplied with a minimum of 10 changes per hour of 100% fresh air that had been passed through 99.97% high-efficiency particulate absorption filters. Room temperature and humidity were monitored constantly throughout the studies. Room temperature was targeted at 64°F to 79°F (18°C-26°C); relative humidity was targeted at 30% to 70%.
F0-generation mice were individually housed in stainless steel, wire-bottomed cages, except during the cohabitation and postpartum periods. During cohabitation, each pair of male and female mice was housed in the male mouse’s cage. Each dam and delivered litter were housed in a common nesting box during the postpartum period. After weaning (postpartum day [PPD] 20), F1-generation mice were housed in nesting boxes. Mice were pair housed until at least PPD 27, after which they were individually housed. An automatically controlled 12-hour light–12-hour dark fluorescent light cycle was maintained. Each dark period began at 1900 hours (±30 minutes).
Cages were changed approximately every other week. Bedding was changed as often as necessary to keep the mice dry and clean. Mice were given ad libitum access to Certified Rodent Diet #5002 (PMI Nutrition International, Inc, St Louis, Missouri) in individual feeders.
Local water that had been processed by passage through a reverse osmosis membrane (RO water) was available to the mice ad libitum from individual water bottles attached to the cages. Chlorine was added to the processed water as a bacteriostat. Bed-o’cobs bedding (The Andersons Industrial Products Group, Maumee, Ohio) was used as the nesting material. Each lot was certified for contaminates by the Testing Facility.
Test Substance
PFHxA Ammonium Salt, a granular white solid, is also known as C-1500N and Ammonium Salt of Perfluorinated Hexanoic Acid. The test substance was supplied as a 50% aqueous solution in RO deionized water. The aqueous solution was a colorless liquid. Purity was 93.4%. Solutions of the test substance were prepared once weekly at the Testing Facility and stirred continuously for at least 24 hours prior to dosage administration and stored at room temperature. The vehicle (RO water) was available from a continuous source at the Testing Facility and maintained at room temperature.
The study samples analyzed were within the acceptance criteria of ±10% of their mean nominal concentrations. For homogeneity, the relative standard deviation for the formulation for the grand mean of the average value for the top, middle, and bottom formulations for each group was ±5%. Homogeneity results showed that the formulation technique used produced homogenous preparations. Stability was demonstrated in a separate study for 10 days at room temperature from 7 to 70 mg/mL.
Mating and Treatment
After acclimation of approximately 1 week, 100 virgin female mice were cohabitated with 100 breeder male mice, 1 male mouse per female mouse. The cohabitation period consisted of a maximum of 5 days. Female mice with a copulatory plug observed in situ were considered to be at gestation day (DG) 0 and assigned to individual housing.
F0-generation female mice were administered the test substance and/or vehicle once daily via oral gavage from DG 6 through DG 18. Dosages were adjusted daily for body weight changes and given at approximately the same time each day. Dams in the process of delivering pups were not adminstered the test substance or vehicle in order to preclude possible disruption to maternal behavior and/or cannibalization of the pups. F1-generation pups were not directly administered the test substance and/or vehicle but may have been possibly exposed to the test substance and/or vehicle during maternal gestation (in utero exposure) or via maternal milk during the lactation period. The treatment regimen for each of the 2 phases is summarized in Table 1.
Treatment Regimen for Phase 1 and Phase 2.

F0-generation mice were observed for viability at least twice each day of the study and for clinical observations and general appearance once weekly during acclimation and on DG 0. The mice were also examined for clinical observations, abortions, premature deliveries, and deaths prior to dosage administration and between 1 and 2 hours after dosage administration and once daily during the postdosage period. Body weights were recorded once weekly during the acclimation period, on DG 0, and daily during the dosage and postdosage periods.
F0-generation mice were evaluated for adverse clinical signs observed during parturition, duration of gestation, litter sizes (all pups delivered) and pup viability at birth, fertility index (percentage of matings that result in pregnancies), gestation index (percentage of pregnancies that result in birth of live litters), number of offspring per litter (live and dead pups), number of implantation sites, general condition of dam and litter during the postpartum period, viability indices (percentage of pups born that survive 4 and 7 days), and lactation index (percentage of pups born that survive 20 days). Maternal behavior was evaluated on PPDs 0, 4, 7, 14, and 20.
Day 0 of lactation (postpartum) was defined as the day of birth and was also the first day on which all pups in a litter were individually weighed (pup body weights were recorded after all pups in a litter were delivered and groomed by the dam). Each litter was evaluated for viability and general appearance at least twice daily. The pups in each litter were counted once daily. Clinical observations were recorded once daily during the preweaning period. Pup body weights were recorded on PPDs 0 (birth), 4, 7, 14, and 20. During the preweaning period, pups were evaluated for eye opening, beginning on PPD 10.
F1-generation mice postweaning were observed for viability daily. These mice were also examined for clinical observations and general appearance once daily. Body weights were recorded weekly. Female mice were evaluated for the age of vaginal patency, beginning on PPD 20. Male mice were evaluated for the age of preputial separation, beginning on PPD 26.
Gross Necropsy
All mice were necropsied and gross lesions were retained in neutral-buffered 10% formalin for possible future evaluation. Unless specifically cited subsequently, all other tissues were discarded. F0-generation mice were killed by carbon dioxide asphyxiation. Pups were killed by an intraperitoneal injection of sodium pentobarbital (pups ≤14 days of age) or by carbon dioxide asphyxiation (pups ≥15 days of age).
At weaning of the F1-generation litters, 5 livers per group from the F0-generation mice were excised, weighed, and frozen on dry ice. Livers were maintained frozen (≤70°C) for analysis. Mice that did not deliver a litter were killed on DG 23 and examined for gross lesions. The number and distribution of implantation sites were recorded after staining with 10% ammonium sulfide 9 to confirm the absence of implantation sites.
Dams with no surviving pups were killed after the last pup was found dead or missing, presumed cannibalized. A gross necropsy of the thoracic, abdominal, and pelvic viscera was performed and implantation sites were recorded after staining with 10% ammonium sulfide. 13
Mice that died before scheduled termination were examined for the cause of death as soon as possible after the observation was made. The mice were examined for gross lesions. The lungs, trachea, and esophagus were perfused and saved in neutral-buffered 10% formalin for possible future evaluation. The heart, kidneys, stomach, and spleen were retained in neutral-buffered 10% formalin for possible histological evaluation. Gravid uterine weights were recorded. Pregnancy status and uterine contents of female mice were recorded. Conceptuses in utero were examined to the extent possible, using the same methods described for term fetuses/pups. The livers were excised, weighed, and frozen on dry ice. Livers were maintained frozen (≤−70ºC) until shipped for analysis to Charles River Preclinical Services Montreal.
Pups that died before initial examination of the litter for pup viability were evaluated for vital status at birth. The lungs were removed and immersed in water. Pups with lungs that sank were considered stillborn; pups with lungs that floated were considered liveborn and to have died shortly after birth.
Pups found dead were examined for gross lesions and for the cause of death as soon as possible. All pups found dead on PPD 1 to 3 were preserved in Bouin solution for possible future evaluation; all pups found dead on PPD 4 to 20 were preserved in neutral-buffered 10% formalin.
On PPD 20, all pups not selected for continued evaluation were killed by carbon dioxide asphyxiation and examined for gross lesions. Necropsy of the pups included a single cut at the suture of the frontal and parietal bones of the skull, and the cross-sectioned brain was examined for hydrocephaly.
One male mouse that died before scheduled termination was examined for the cause of death as soon as possible after the observation was made. The mouse was examined for gross lesions. The heart, kidneys, lungs, stomach, and spleen were retained in neutral-buffered 10% formalin for possible histological evaluation. The liver was excised, weighed, and frozen on dry ice.
Five F1-generation mice per sex per group (total 40 mice) in each phase were killed on PPD 41 for sample collection for determination of body burden. Blood samples (0.5-1.0 mL) and livers were collected from these mice. Blood samples were collected via the vena cava after killing. The blood samples were transferred into uncoated (red top) tubes and spun in a refrigerated (4°C) centrifuge for 10 minutes at 3500 rpm. The resulting serum was transferred into appropriately labeled polypropylene tubes. All samples were frozen on dry ice as soon as possible and maintained frozen (≤70°C) for the analysis. The remaining mice were killed by carbon dioxide asphyxiation on PPD 41. A gross necropsy of the thoracic, abdominal, and pelvic viscera was performed.
Statistical Analyses
Averages and percentages were calculated. Litter values were used where appropriate. Clinical observations and other proportional data were analyzed using the variance test for homogeneity of the binomial distribution. 14
Continuous data, such as body weights, organ weights, percentage of litter reaching a developmental landmark, and percentage of mortality per litter, were analyzed as described under the parametric heading of the schematic. Bartlett test of homogeneity of variances 15 was used to estimate the probability that the dosage groups had different variances. A nonsignificant result (P > 0.001) indicated that an assumption of homogeneity of variance was not inappropriate, and the data were compared using the analysis of variance. 16 If that test was significant (P ≤ 0.05), the groups given the test substance were compared with the control group using Dunnett test. 17 If Bartlett test was significant (P > 0.001), the analysis of variance test was inappropriate and the data were analyzed as described under the nonparametric heading of the schematic. When 75% or fewer of the scores were tied, the Kruskal-Wallis test 18 was used to analyze the data, and in the event of a significant result (P ≤ 0.05), Dunn method of multiple comparisons 19 was used to compare the groups given the test substance with the control group. When more than 75% of the scores were tied, Fisher exact test 20 was used to compare the proportion of ties in the dosage group.
Variables with graded count scores, such as litter size, were analyzed using the nonparametric procedures described previously. The pup weights for all weigh days were covaried against litter size at birth. 21
Results
Mortality, Clinical, and Necropsy Observations
Phase 1
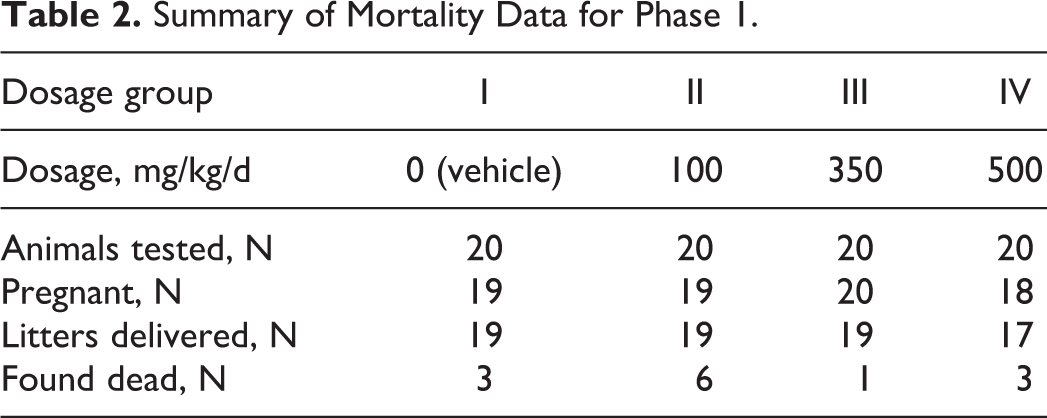
A total of 3, 6, 1, and 3 F0-generation female mice were found dead in the 0, 100, 350, and 500 mg/kg/d dosage groups, respectively (Table 2). Single deaths that occurred in the mid-dosage and high-dosage groups during the gestation period appear to have been related to administration of the test substance based on the timing of the deaths (DGs 8 and 13). No other mortality related to PFHxA Ammonium Salt occurred. The deaths in the control and low dosage group all occurred between PPDs 13 and 16. These deaths, which were within the historical range for this Testing Facility, and possibly 2 of the 3 deaths that occurred in the highest dosage group appeared to be due to the stress of nursing, which is known to occur in mice. 22 –26 Additionally, a total of 1, 0, 2, and 6 mice in the 0, 100, 350, and 500 mg/kg/d dosage groups, respectively, were killed due to no surviving pups.
Summary of Mortality Data for Phase 1.
Phase 2
No mortality related to PFHxA Ammonium Salt occurred. All mice survived until scheduled killing, with the exception of 1 mouse in the 7 mg/kg/d dosage group that was killed on DG 17 when it delivered its litter; and 1 mouse in the 35 mg/kg/d dosage group that was killed on PPD 2 due to no surviving pups.
All clinical observations during the gestation and lactation periods were considered unrelated to the test substance because: (1) the incidences were not dosage dependent and (2) the observations occurred in only 1 mouse in a group. These clinical observations included a red perivaginal substance and urine-stained abdominal fur.
There were no test substance-related necropsy observations. All necropsy observations were considered unrelated to the test substance because (1) the incidences were not dosage dependent or (2) the observations occurred in only 1 mouse. These necropsy observations included numerous clear cysts in the liver (one 7 mg/kg/d dosage group mouse), clear fluid-filled cyst in the capsule of the kidney (one 35 mg/kg/d dosage group mouse), thick walls of the uterus (one 7 mg/kg/d dosage group mouse), and clear fluid-filled cysts in the uterus (one 35 mg/kg/d dosage group mouse).
Body Weights and Body Weight Gains
Phase 1
Body weights and body weight gains during the gestation period and body weights during the lactation period were unaffected by dosages of the test substance as high as 500 mg/kg/d. All values were comparable among the 4 dosage groups and did not differ significantly (Figure 1).
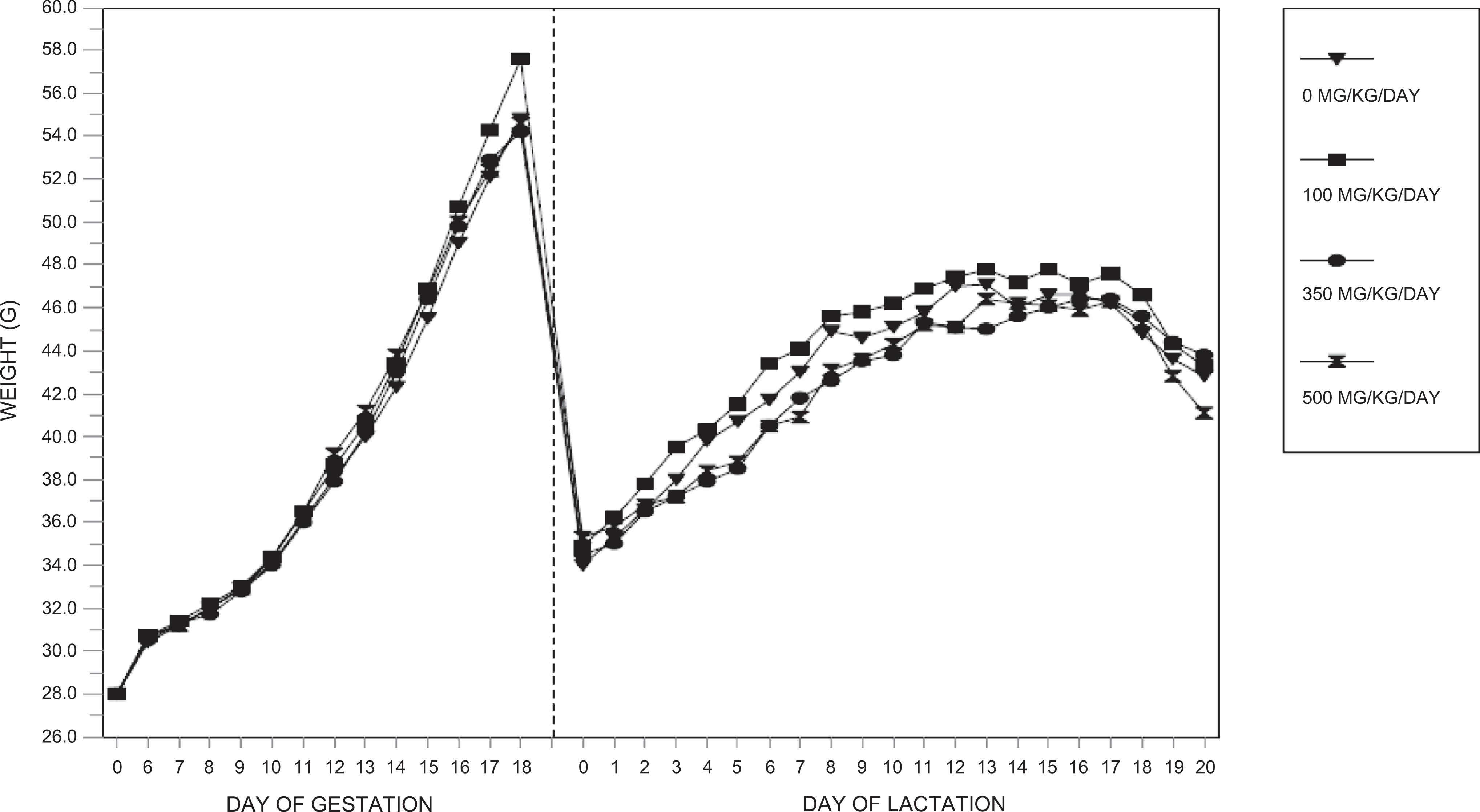
Maternal body weights (phase 1)—F0-generation female mice.
Body weight gains during lactation were significantly reduced (P ≤ 0.05 to P ≤ 0.01) for PPDs 0 to 4 in the 350 and 500 mg/kg/d dosage groups compared to the control group value. Although no additional significant differences occurred among the groups for body weight gain during lactation, the average gain during the entire lactation period was reduced in the 500 mg/kg/d dosage group compared to the control group value. Body weight gains from PPDs 0 to 20 were 97.7%, 110.3%, and 64.4% of the control group value.
Phase 2
Body weights and body weight gains during the gestation and lactation periods were unaffected by dosages of the test substance as high as 175 mg/kg/d. All values were comparable among the 4 dosage groups and did not differ significantly (Figure 2).
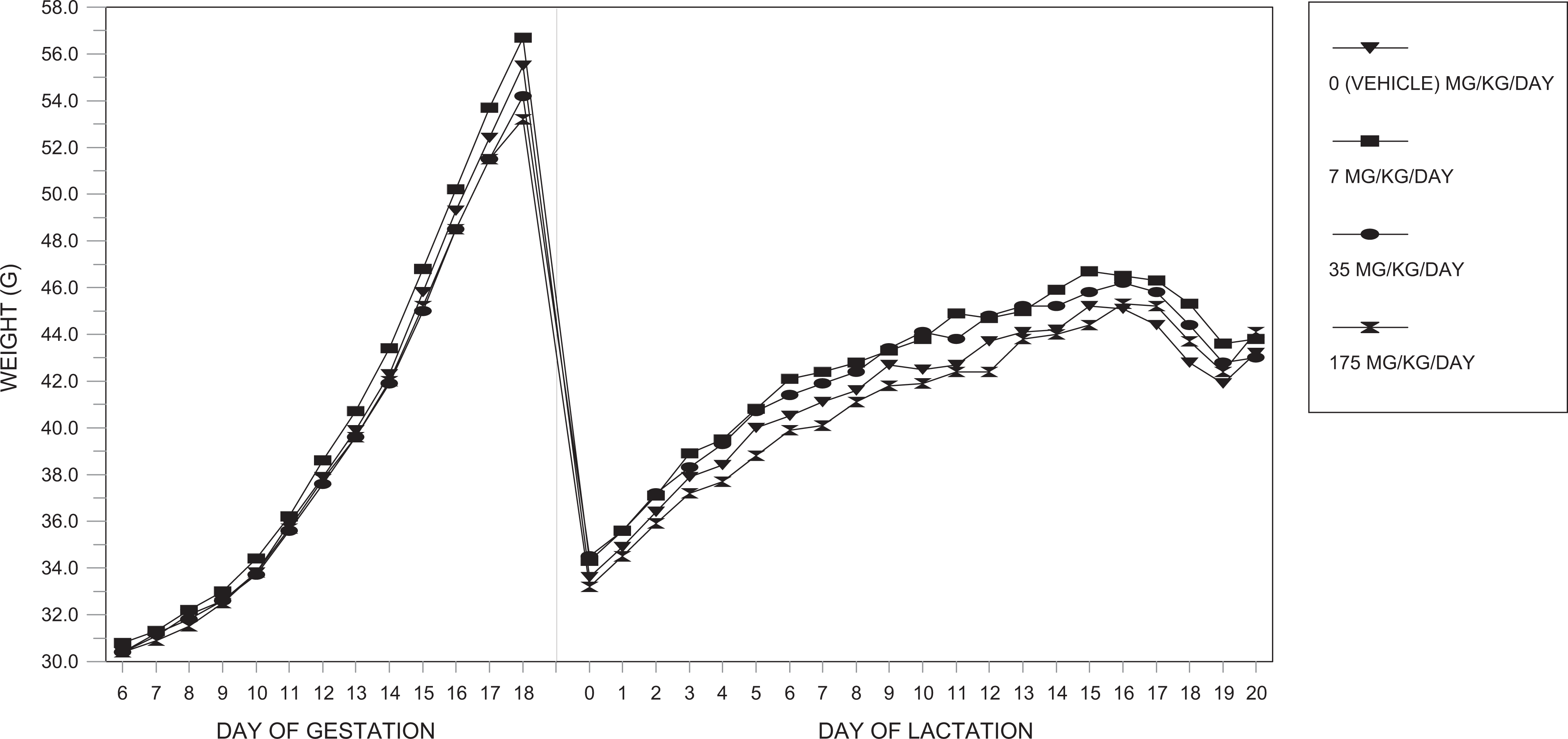
Maternal body weights (phase 2)—F0-generation female mice.
Pregnancy, Fertility, and Litter Observations
Phase 1
Pregnancy occurred in 19, 19, 20, and 18 of the 20 mated female mice in the 0 (vehicle), 100, 350, and 500 mg/kg/d dosage groups, respectively (Table 3). All pregnant dams delivered litters, with the exception of 1 mouse in each of the groups III and IV that died during gestation. All mated mice were pregnant and all that survived delivered a litter.
F0-Generation Female Mice: Summary of Natural Delivery Observations.
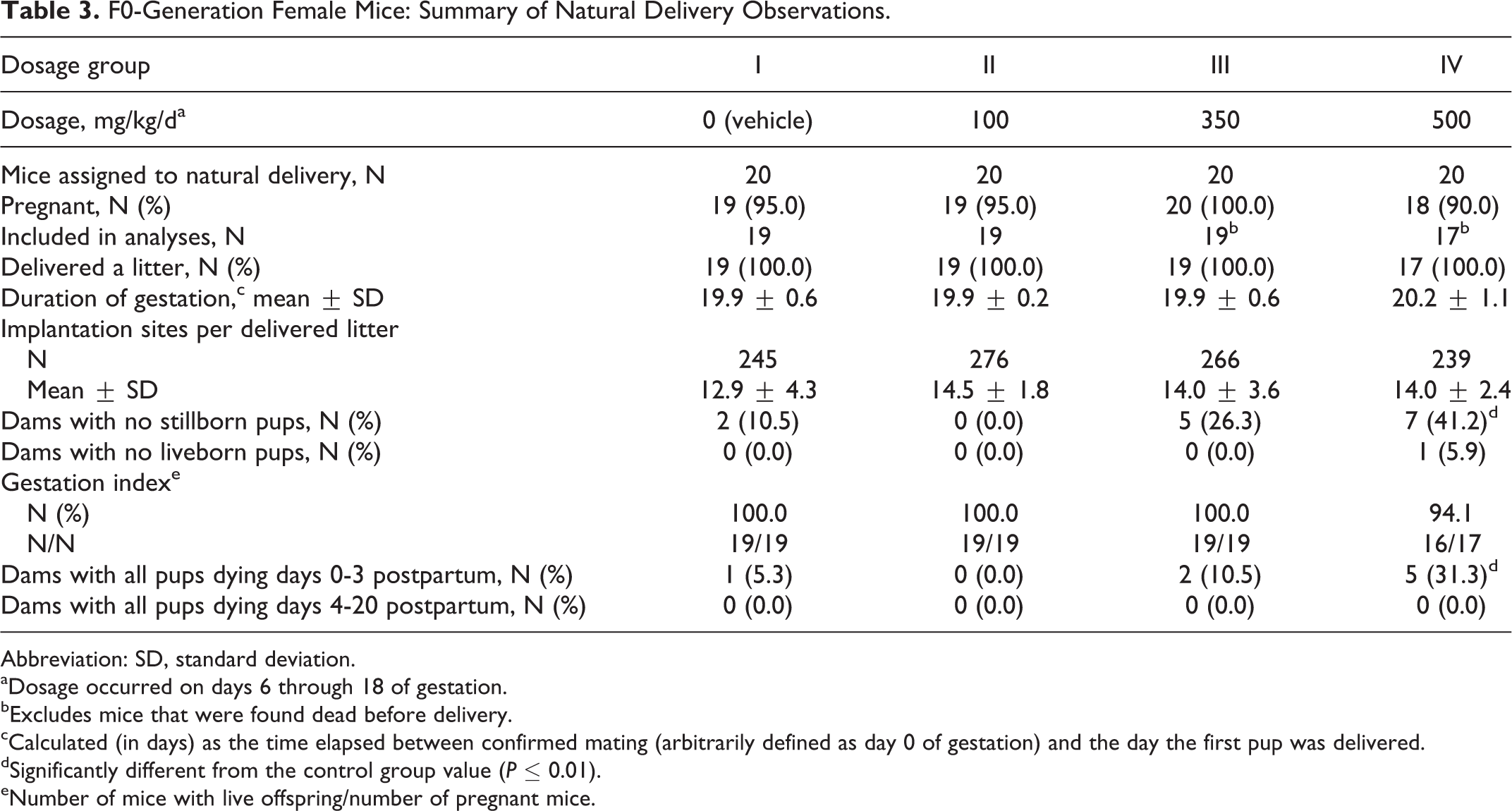
Abbreviation: SD, standard deviation.
aDosage occurred on days 6 through 18 of gestation.
bExcludes mice that were found dead before delivery.
cCalculated (in days) as the time elapsed between confirmed mating (arbitrarily defined as day 0 of gestation) and the day the first pup was delivered.
dSignificantly different from the control group value (P ≤ 0.01).
eNumber of mice with live offspring/number of pregnant mice.
The number of pups dying on PPDs 1 to 4 in the 350 and 500 mg/kg/d dosage groups was significantly increased (P ≤ 0.01) compared to the control group (Table 4). The number of pups dying on PPD 0 was increased in the 300 mg/kg/d dosage group and significantly increased (P ≤ 0.01) in the 500 mg/kg/d dosage group compared to the control group.
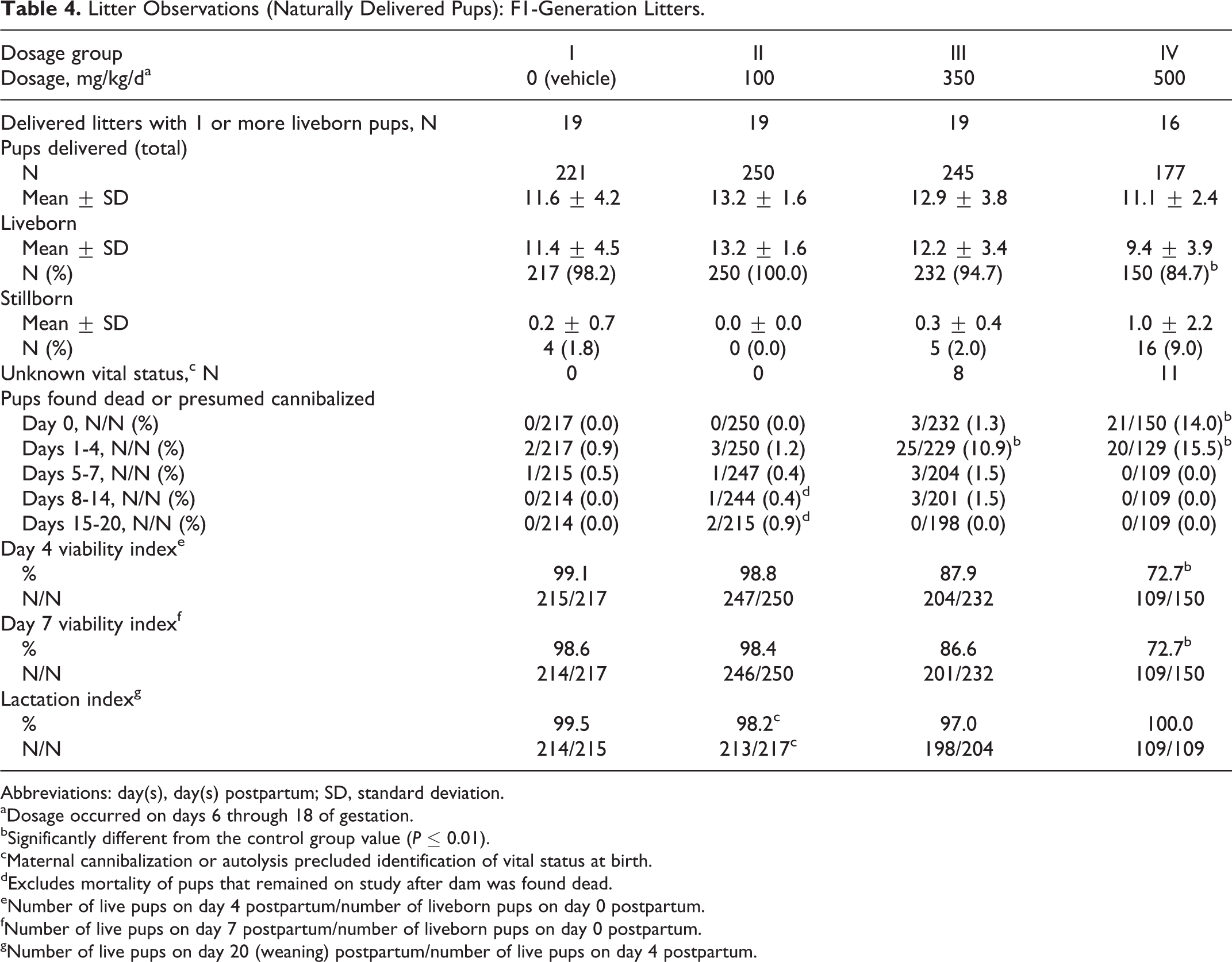
Litter Observations (Naturally Delivered Pups): F1-Generation Litters.
Abbreviations: day(s), day(s) postpartum; SD, standard deviation.
aDosage occurred on days 6 through 18 of gestation.
bSignificantly different from the control group value (P ≤ 0.01).
cMaternal cannibalization or autolysis precluded identification of vital status at birth.
dExcludes mortality of pups that remained on study after dam was found dead.
eNumber of live pups on day 4 postpartum/number of liveborn pups on day 0 postpartum.
fNumber of live pups on day 7 postpartum/number of liveborn pups on day 0 postpartum.
gNumber of live pups on day 20 (weaning) postpartum/number of live pups on day 4 postpartum.
The number of mice with stillborn pups was increased in the highest dosage group, and the number of mice with all pups dying on PPDs 0 to 3 was significantly increased (P ≤ 0.01) in the 500 mg/kg/d dosage group compared to the control group values. The following additional effects occurred in the 500 mg/kg/d dosage group: the average litter size was reduced at birth and throughout the lactation period with a significant reduction (P ≤ 0.05) on PPD 4; and the PPD 4 and 7 viability indices were significantly reduced (P ≤ 0.01) compared to the control group value (Table 4). The day 7 viability index was significantly reduced (P ≤ 0.05) in the 350 mg/kg/d dosage group. The average number of surviving pups per litter was significantly reduced (P ≤ 0.01) in the 500 mg/kg/d dosage group for PPDs 4, 7, 14, and 20 compared to the control group values (Table 5).
Litter Viability (Naturally Delivered Pups): F1-Generation Litters.

Abbreviations: day, day of postpartum; SD, standard deviation; [ ], number of values averaged.
aDosage occurred on days 6 through 18 of gestation.
bAverage number of live pups per litter, including litters with no surviving pups.
cIncludes liveborn pups and pups that died before weighing on day 0 postpartum.
dExcludes litters with mortality of pups that remained on study after dam was found dead.
eSignificantly different from the control group value (P ≤ 0.01).
fExcludes values for litters that had no surviving pups.
gExcludes values for litters that had no surviving pups.
hSignificantly different from the control group value (P ≤ 0.05).
iExcludes litters with mortality of pups that remained on study after dam was found dead.
jWith pup body weights per litter covaried with litter size per litter, the analyses were not significant.
Pup body weights in all treated groups were generally lower in the treated groups compared to the control group values. Pup body weights were significantly reduced (P ≤ 0.05 to P ≤ 0.01) on PPD 0 in the 100 mg/kg/d and higher dosage groups compared to the control group value (Table 5). The variance in litter size among the groups also had an effect on the pup body weights. 27 When pup body weights were covaried against the litter size on PPD 0, the statistically significant differences among the groups for pup body weights were no longer present. This result indicated that the litter size rather than the test substance was affecting the pup weight differences between the groups. The litter size was largest in the 100 mg/kg/d dosage group and therefore, the reduced pup weight that occurred in this group was considered related to the litter size. The litter sizes in the 350 and 500 mg/kg/d dosage groups were affected by increased pup deaths that occurred in these dosage groups. Therefore, an effect on pup body weight was related to the test substance in these 2 dosage groups.
Statistically significantly reduced pup body weights persisted in the 350 mg/kg/d dosage group through PPD 7 and in the 500 mg/kg/d dosage group through PPD 4. On PPD 20, average pup weights per litter were 89%, 80%, and 88% of the control group value. The lack of dosage dependency can be attributed to the differences in litter size among the groups.
All other natural delivery and litter observations were unaffected by dosages of the test substance as high as 500 mg/kg/d. Values for the numbers of dams delivering litters, the duration of gestation, averages for implantation sites per delivered litter, the gestation index (number of dams with 1 or more liveborn pups/number of pregnant mice), total litter sizes, lactation index, and percentage of male pups per number of pups sexed per litter were comparable among the 4 dosage groups and did not significantly differ.
Phase 2
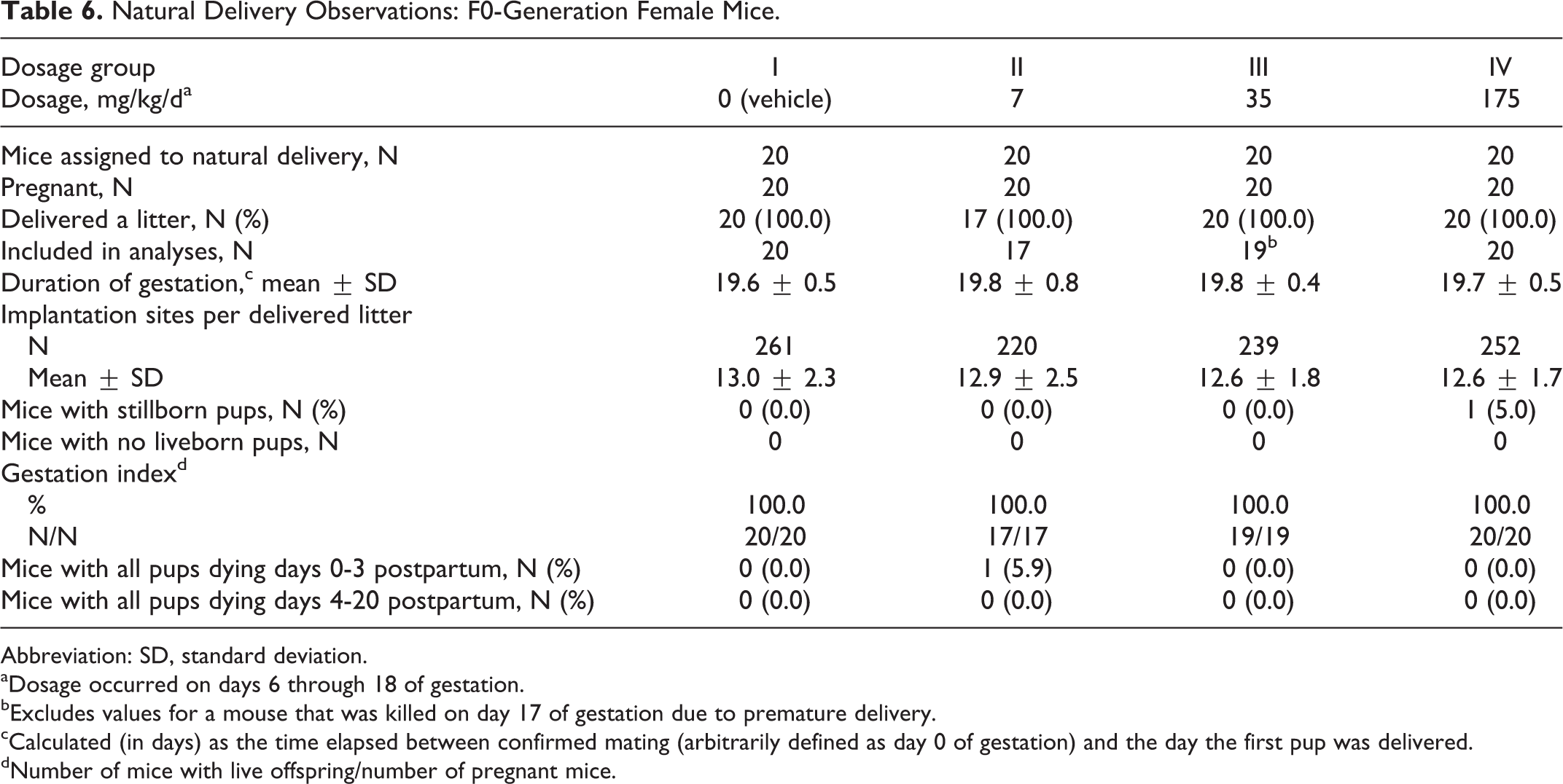
Pregnancy occurred in all the 20 mated female mice in the 0 (vehicle), 7, 35, and 175 mg/kg/d dosage groups, respectively. However, 20, 17, 20, and 20 mice, respectively, delivered litters (Table 6).
Natural Delivery Observations: F0-Generation Female Mice.
Abbreviation: SD, standard deviation.
aDosage occurred on days 6 through 18 of gestation.
bExcludes values for a mouse that was killed on day 17 of gestation due to premature delivery.
cCalculated (in days) as the time elapsed between confirmed mating (arbitrarily defined as day 0 of gestation) and the day the first pup was delivered.
dNumber of mice with live offspring/number of pregnant mice.
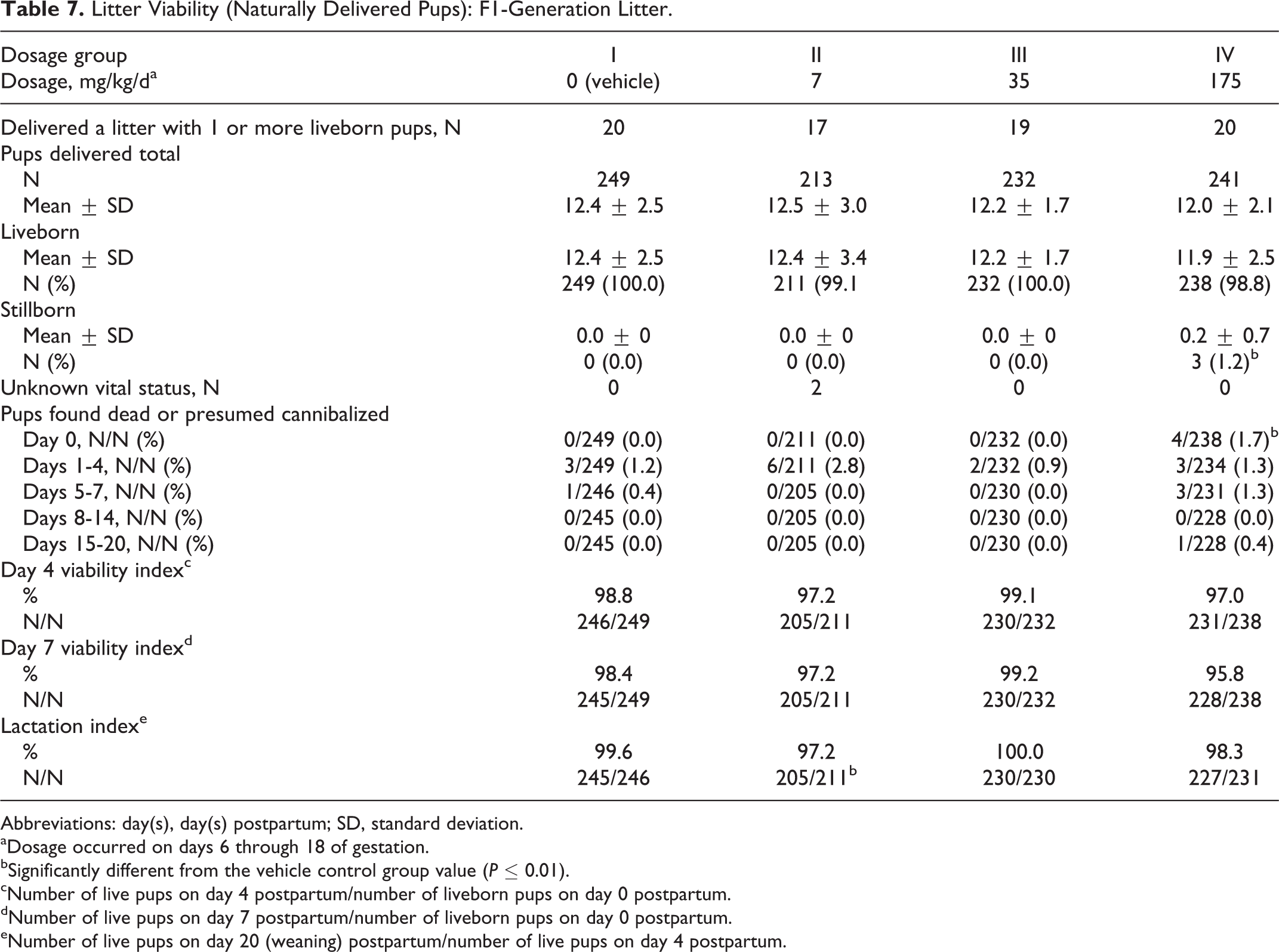
The number of stillborn pups and pups dying on day 1 postpartum were significantly increased (P ≤ 0.05), and the average pup weight per litter was significantly reduced on day 1 postpartum in the 175 mg/kg/d dosage group compared to the 0 (vehicle) mg/kg/d dosage group values (Tables 7 and 8).
Litter Viability (Naturally Delivered Pups): F1-Generation Litter.
Abbreviations: day(s), day(s) postpartum; SD, standard deviation.
aDosage occurred on days 6 through 18 of gestation.
bSignificantly different from the vehicle control group value (P ≤ 0.01).
cNumber of live pups on day 4 postpartum/number of liveborn pups on day 0 postpartum.
dNumber of live pups on day 7 postpartum/number of liveborn pups on day 0 postpartum.
eNumber of live pups on day 20 (weaning) postpartum/number of live pups on day 4 postpartum.
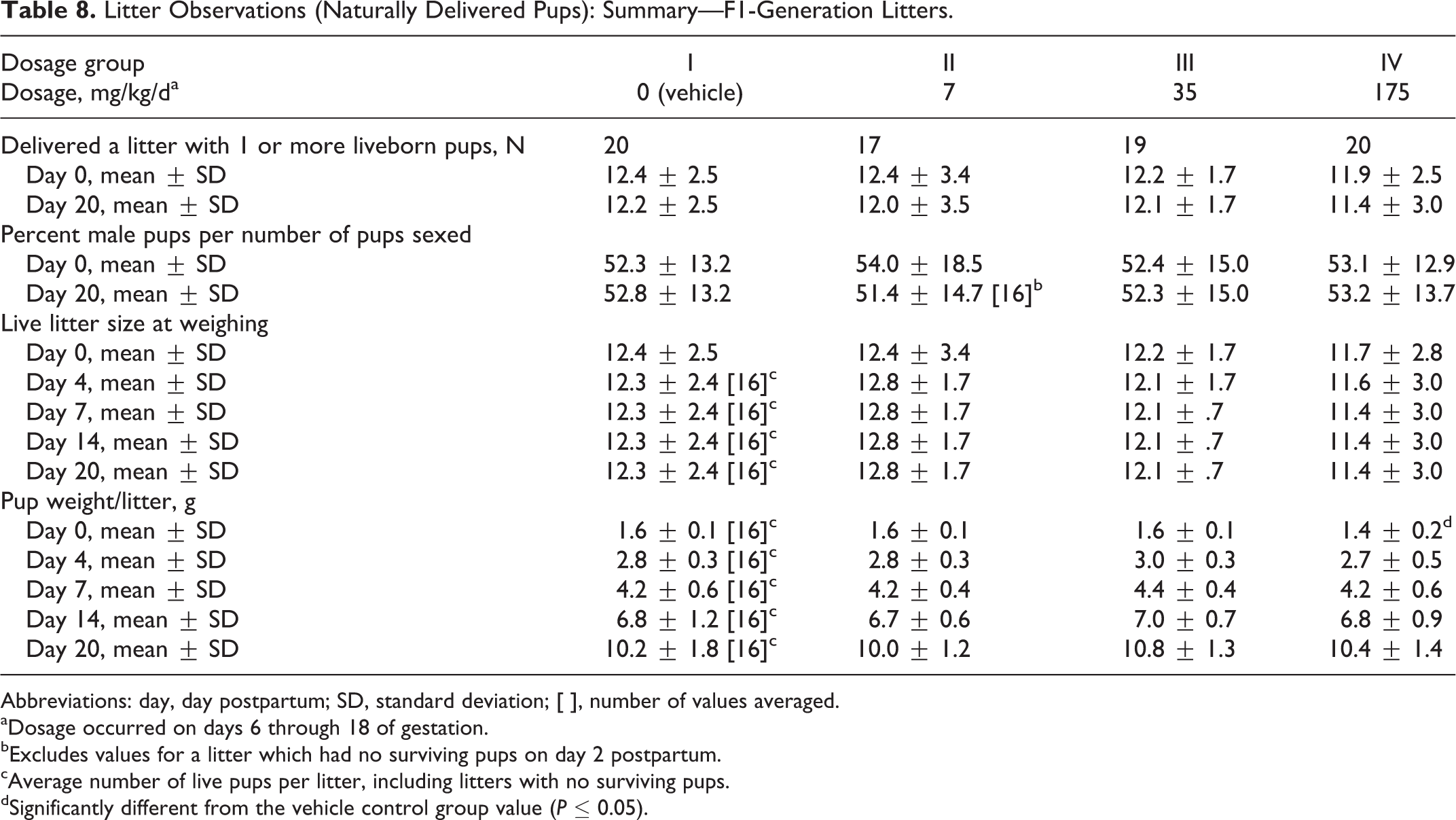
Litter Observations (Naturally Delivered Pups): Summary—F1-Generation Litters.
Abbreviations: day, day postpartum; SD, standard deviation; [ ], number of values averaged.
aDosage occurred on days 6 through 18 of gestation.
bExcludes values for a litter which had no surviving pups on day 2 postpartum.
cAverage number of live pups per litter, including litters with no surviving pups.
dSignificantly different from the vehicle control group value (P ≤ 0.05).
All other natural delivery and litter observations were unaffected by dosages of the test substance as high as 175 mg/kg/d (Table 8). Values for the numbers of dams delivering litters, the duration of gestation, averages for implantation sites per delivered litter, the gestation index (number of dams with 1 or more liveborn pups/number of pregnant mice), the numbers of dams with stillborn pups and of dams with all pups dying, litter sizes, viability index, surviving pups per litter, percentage of male pups per number of pups sexed per litter, live litter size at weighing, and pup weight per litter were comparable among the 4 dosage groups and did not significantly differ (Table 8). A significant reduction (P ≤ 0.01) in the lactation index in the 7 mg/kg/d dosage group was not considered related to the test substance because it was not dosage dependent.
Terminal Body Weights
Phase 1
Terminal body weights were comparable among the 4 dosage groups (Table 9). The absolute weights of the liver and the ratio of the liver weight to the terminal body weight did not differ significantly among the groups.
Terminal Body Weights, Liver Weights, and Ratios (%) of Liver Weight to Terminal Body Weight: F0-Generation Female Mice.a
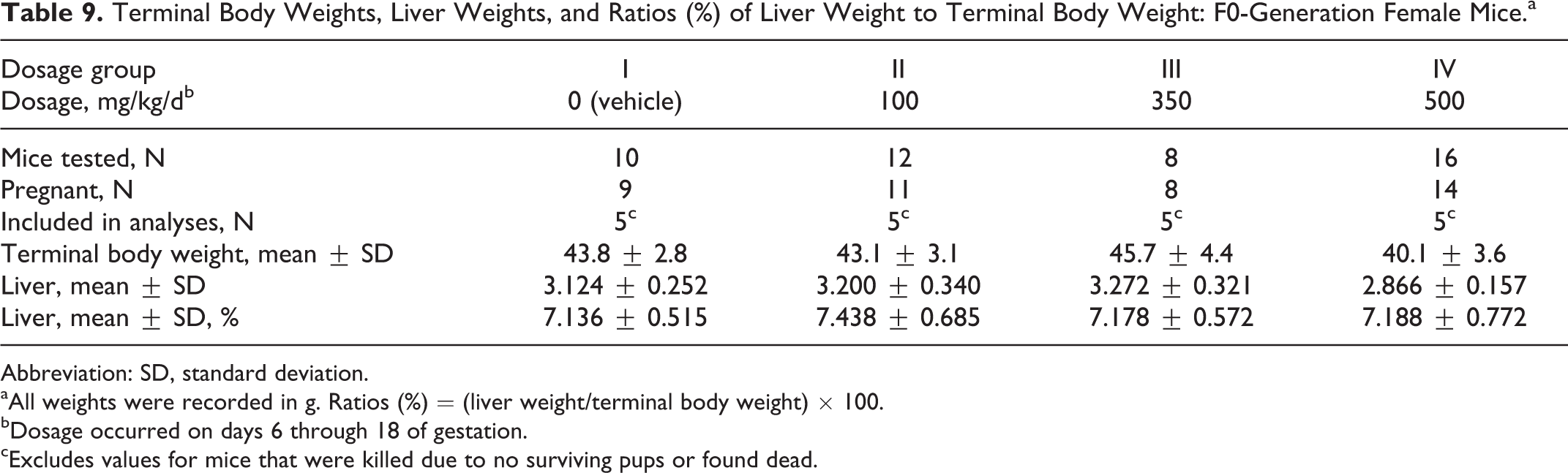
Abbreviation: SD, standard deviation.
aAll weights were recorded in g. Ratios (%) = (liver weight/terminal body weight) × 100.
bDosage occurred on days 6 through 18 of gestation.
cExcludes values for mice that were killed due to no surviving pups or found dead.
Phase 2
Terminal body weights were comparable among the 4 dosage groups (Table 10). The absolute weights of the liver and the ratio of the liver weight to the terminal body weight did not differ significantly among the groups.
Terminal Body Weights, Liver Weights, and Ratios (%) of Liver Weight to Terminal Body Weight: F0-Generation Female Mice.a
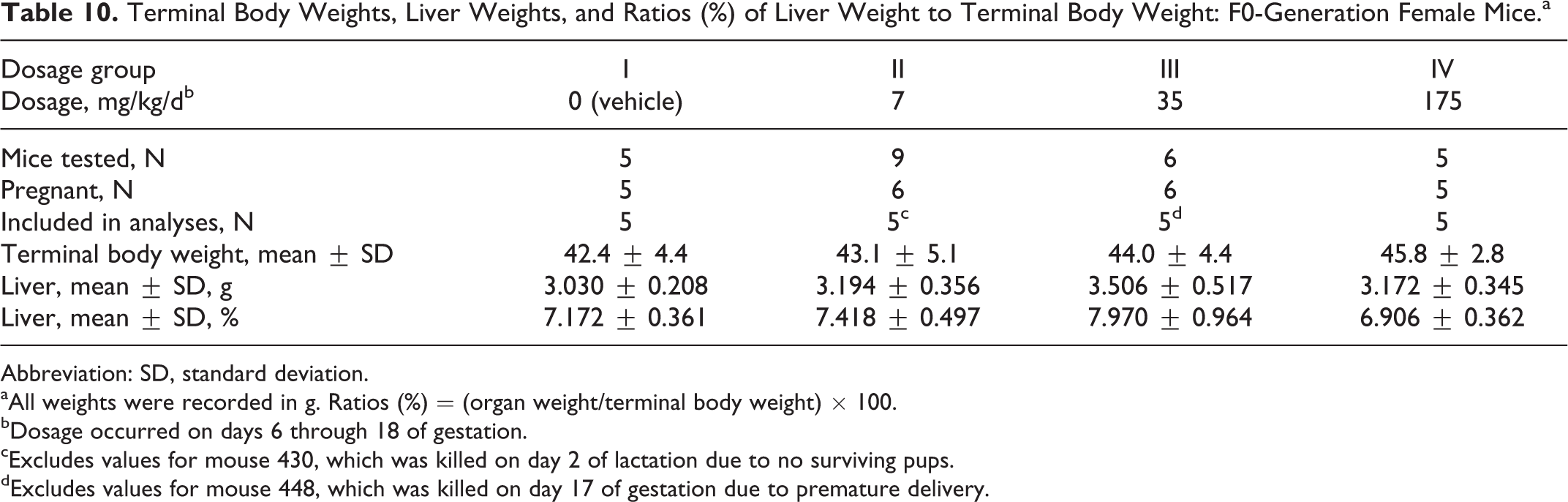
Abbreviation: SD, standard deviation.
aAll weights were recorded in g. Ratios (%) = (organ weight/terminal body weight) × 100.
bDosage occurred on days 6 through 18 of gestation.
cExcludes values for mouse 430, which was killed on day 2 of lactation due to no surviving pups.
dExcludes values for mouse 448, which was killed on day 17 of gestation due to premature delivery.
Necropsy Observations
Phase 1
Tan areas in the liver occurred in 1 mouse in the 350 mg/kg/d dosage group and 5 (P ≤ 0.01) of the 20 mice in the 500 mg/kg/d dosage group.
There were no other test substance-related necropsy observations. One mouse each in the 0 and 100 mg/kg/d dosage group had a bent sternum proximal to the xiphoid process, and 1 mouse in the 500 mg/kg/d dosage group had intestines that were distended with gas.
Phase 2
There were no test substance-related necropsy observations. Necropsy observations included numerous clear cysts in the liver (one 7 mg/kg/d dosage group mouse), clear fluid-filled cyst in the capsule of the kidney (one 35 mg/kg/d dosage group mouse), thick walls of the uterus (one 7 mg/kg/d dosage group mouse), and clear fluid-filled cysts in the uterus (one 35 mg/kg/d dosage group mouse).
Levels of PFHxA in Liver Homogenates
Phase 1
Results of the analyses of liver homogenates from 10 F0-generation control group mice were all below the lower limit of quantitation with the exception of 1 mouse for which a detectable level was found. Based on the quantity found (just above the lower limit of quantitation) and the lack of any adverse clinical observations in this mouse, this result appeared to indicate cross contamination of the sample during collection and/or processing. This mouse was found dead on PPD 17, and the liver was taken at approximately the same time as the livers from other early deaths in treated groups were taken.
All blood samples were below the limit of quantitation. In the 100 mg/kg/d dosage group, all liver homogenates analyzed were below the lower limit of quantitation. In the 350 mg/kg/d dosage group, 3 of the 8 samples had analytical results that were quantifiable. The highest level (87.5 μg/mL) occurred in a mouse that was found dead on DG 13. The other mice had much lower levels of PFHxA, but it is interesting that both of these mice were killed early after their litters had died off. These were the only mice in this group that lost their litters.
In the 500 mg/kg/d dosage group, 5 of the 16 samples had analytical results that were quantifiable. The highest level (98.4 μg/mL) occurred in a mouse that was found dead on DG 6. The other mice had much lower levels of PFHxA but each of these mice had litters that died early. Two samples that were below the lower limit of quantitation were from mice that lost their litters.
Phase 2
All liver samples from F1-generation pups euthanized on PPD 42 were below the limit of quantitation.
F1-Generation Preweaning Clinical Observations
Phase 1
No clinical observations in the F1-generation pups were attributed to dosages of the test substance as high as 500 mg/kg/d because (1) the incidences were not dosage dependent; (2) the observation occurred in only 1 to 3 litters; and/or (3) the observation occurred only in the vehicle control group. These clinical observations included scab, dehydration, tip of tail red or missing, not nursing, not nesting, and ungroomed coat.
The average day that 50% of the pups had open eyes was significantly longer (P ≤ 0.01) in the 350 mg/kg/d dosage group compared to the control group value (Table 11). The lack of a significant difference in the 500 mg/kg/d dosage group may be related to the higher number of early pups deaths and reduced litter size in this group. The percentage of pups per litter with open eyes was significantly reduced (P ≤ 0.05 to P ≤ 0.01) in the 350 and 500 mg/kg/d dosage groups on PPD 14 compared to the control group value.
Eye Opening by Litter: F1-Generation Litters.
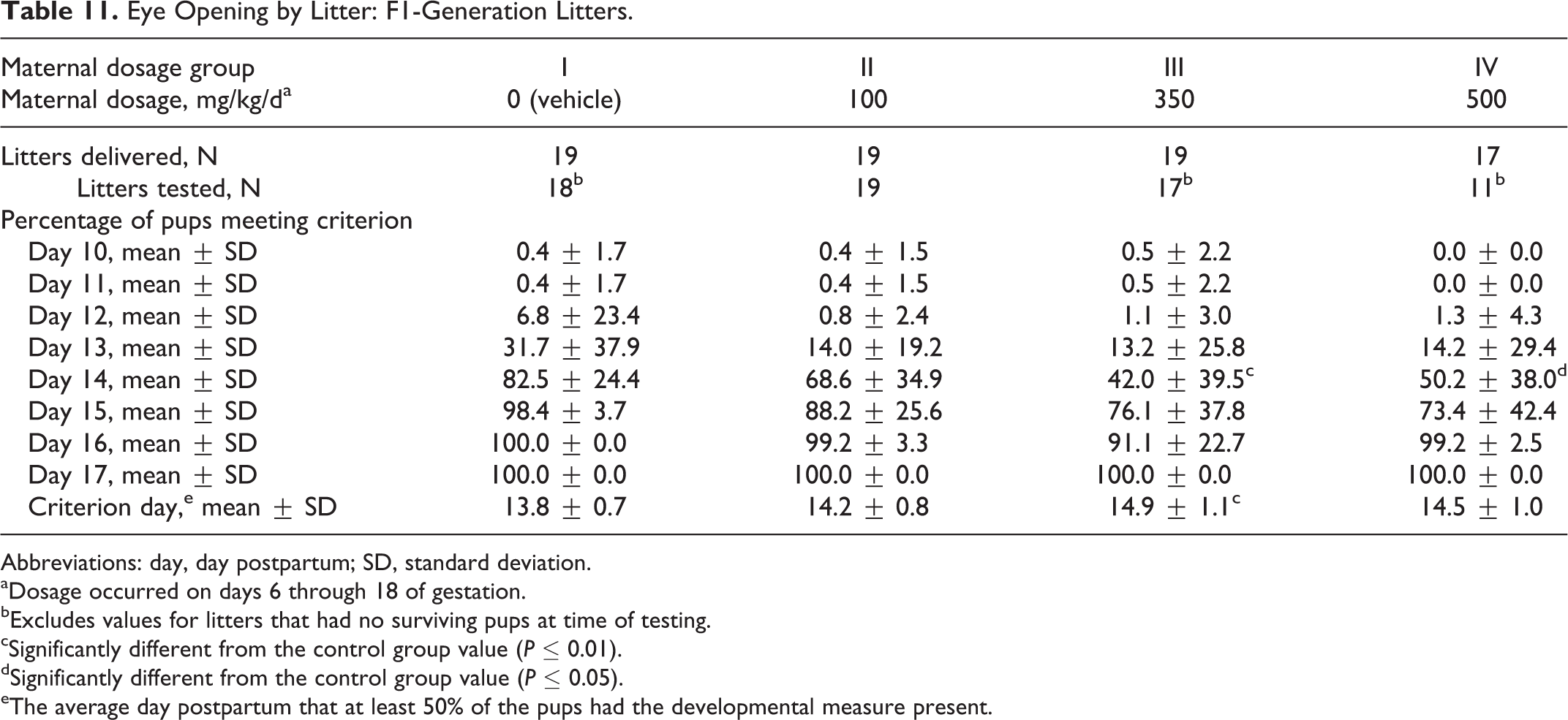
Abbreviations: day, day postpartum; SD, standard deviation.
aDosage occurred on days 6 through 18 of gestation.
bExcludes values for litters that had no surviving pups at time of testing.
cSignificantly different from the control group value (P ≤ 0.01).
dSignificantly different from the control group value (P ≤ 0.05).
eThe average day postpartum that at least 50% of the pups had the developmental measure present.
An increased number of pups in the 500 mg/kg/d dosage group that were found dead or stillborn in the 500 mg/kg/d dosage group had no milk in the stomach. All pups that survived to PPD 20 and were not continued on study appeared normal at necropsy.
Phase 2
Two litters in the 175 mg/kg/d dosage group had a pup each with corneal opacity and 1 pup each with microphthalmia. One litter in this dosage group also had a pup with a lenticular opacity. No other clinical observations in the F1-generation pups were attributed to dosages of the test substance as high as 175 mg/kg/d.
No other clinical observations in the F1-generation pups were attributed to dosages of the test substance as high as 175 mg/kg/d because (1) the incidences were not dosage dependent; (2) the observation occurred in only 1 to 3 litters; and/or (3) the observation occurred only in the vehicle control group.
These clinical observations included dehydration cold to touch, tip of tail red, pale body, not nesting, a head laceration, scab on the lower midline or head, and a mass on the back. The day of eye opening did not differ among the groups (Table 12).
Eye Opening by Litter: F1-Generation Litters.
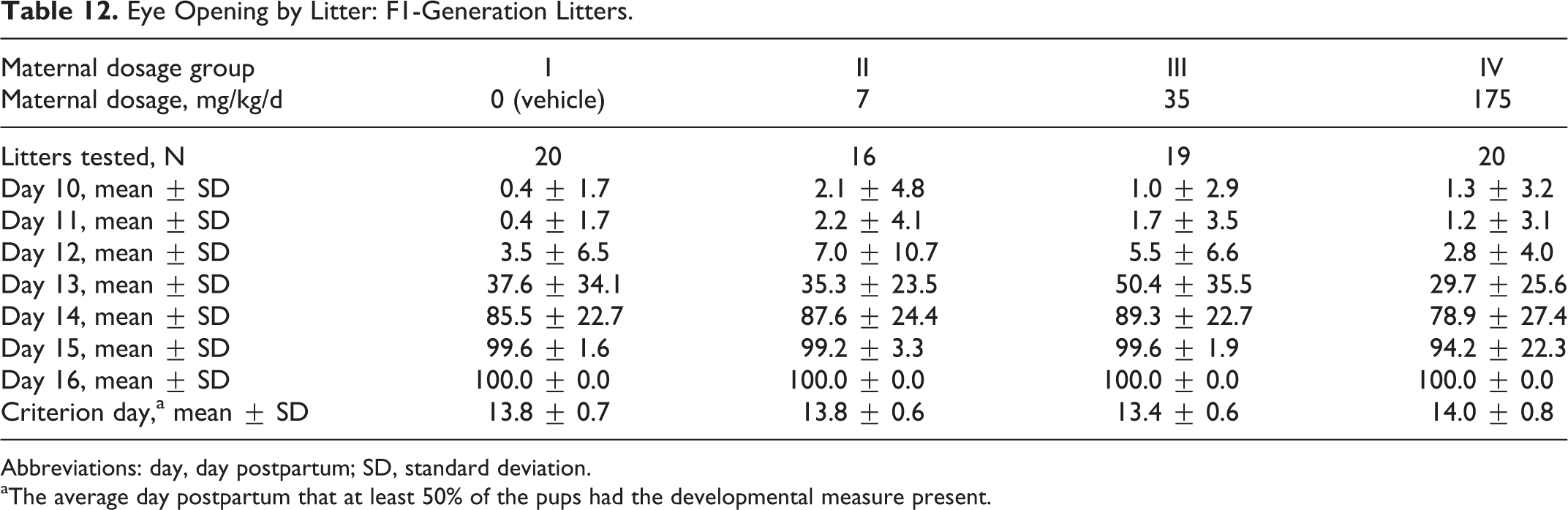
Abbreviations: day, day postpartum; SD, standard deviation.
aThe average day postpartum that at least 50% of the pups had the developmental measure present.
No necropsy observations in the F1-generation pups were attributed to dosages of the test substance as high as 175 mg/kg/d. There was only a single finding (gas-filled intestine) in a 7-mg/kg/d dosage group mouse for mice found dead, and all other mice appeared normal for those mice killed on day 20 postpartum.
F1-Generation Postweaning
Phase 1
One male mouse in the 350 mg/kg/d maternal dosage group was found dead on PPD 23. There were no clinical signs noted during the postweaning period. This mouse had the lowest body weight in its group. At necropsy, all tissues appeared normal for a moderate degree of autolysis. This mouse apparently did not thrive postweaning. All other F1-generation male and female mice survived to scheduled killing.
All clinical observations in the F1-generation male and female mice were considered unrelated to maternal administration of the test substance because (1) the incidences were not dosage dependent; (2) the observation occurred in only 1 mouse; and/or (3) the observation is common in this species and strain. These clinical observations were limited to common findings in the tail including constricted, bent, missing, or purple.
All F1-generation male mice appeared normal at necropsy. One F1-generation female mouse in the 100 mg/kg/d dosage group had a small left kidney. No other necropsy observations occurred in these mice.
Phase 2
All F1-generation male and female mice survived to scheduled killing. All clinical observations in the F1-generation male and female mice were considered unrelated to maternal administration of the test substance because (1) the incidences were not dosage dependent; (2) the observation occurred in only 1 mouse; and/or (3) the observation is common in this species and strain. These clinical observations included lacrimation, ptosis, scab on the tail, and a lenticular opacity.
Necropsy observations in the F1-generation female and male mice occurred in 1 mouse each in the 35 and 175 mg/kg/d dosage groups, respectively. One male mouse had a clear fluid-filled cyst in the liver and 1 female mouse had a dark flat red mass in the mesentery.
F1-Generation Body Weights and Body Weight Gains
Phase 1
Body weights and body weight gains of the F1-generation male and female mice were unaffected by maternal dosages of the test substance as high as 500 mg/kg/d (Figures 3 and 4).
Body weights (phase 1)—F1-generation male mice.
Body weights (phase 1)—F1-generation female mice.
Phase 2
Body weights and body weight gains of the F1-generation male and female mice were unaffected by maternal dosages of the test substance as high as 175 mg/kg/d (Figures 5 and 6). No significant differences occurred among the groups during the postweaning period (PPD 21-41).
Body weights (phase 2)—F1-generation male mice.

Body weights (phase 2)—F2-generation female mice.
F1-Generation Sexual Maturity
Phase 1
Sexual maturation was unaffected by maternal dosages of the test substance as high as 500 mg/kg/d (Table 13). The average day on which preputial separation or vaginal patency occurred was comparable among the 4 dosage groups.
Sexual Maturation: F1-Generation Mice.
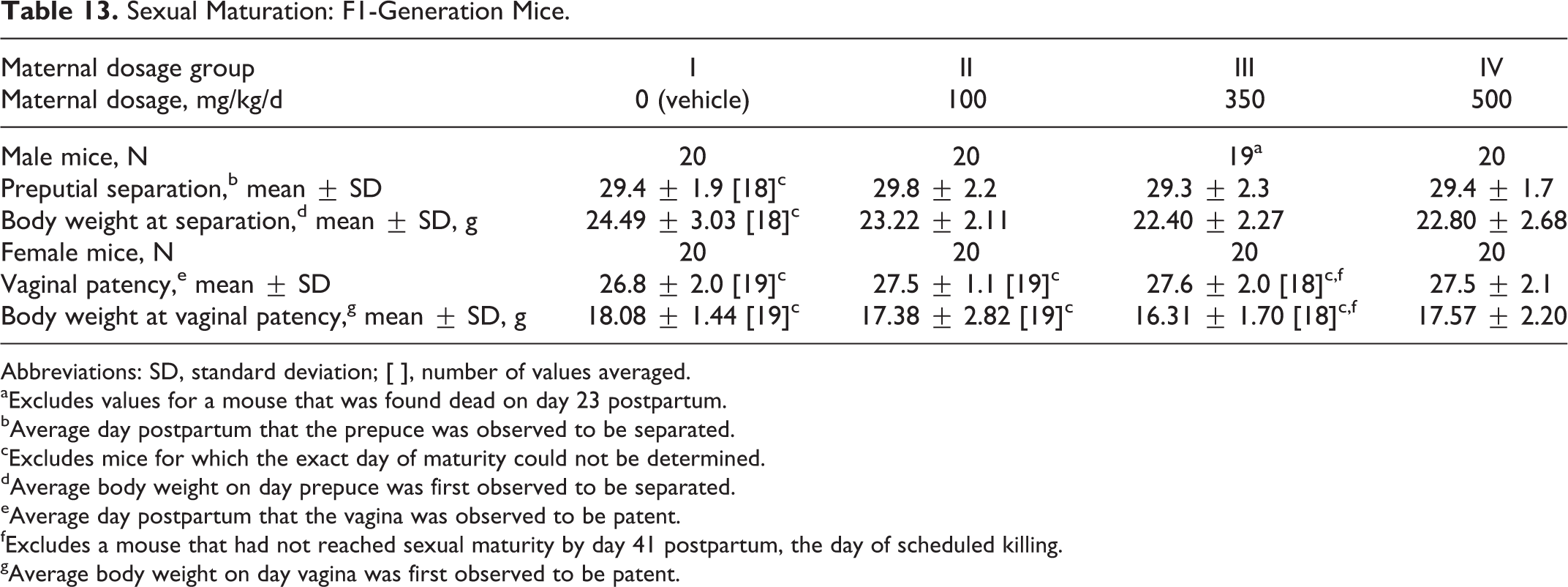
Abbreviations: SD, standard deviation; [ ], number of values averaged.
aExcludes values for a mouse that was found dead on day 23 postpartum.
bAverage day postpartum that the prepuce was observed to be separated.
cExcludes mice for which the exact day of maturity could not be determined.
dAverage body weight on day prepuce was first observed to be separated.
eAverage day postpartum that the vagina was observed to be patent.
fExcludes a mouse that had not reached sexual maturity by day 41 postpartum, the day of scheduled killing.
gAverage body weight on day vagina was first observed to be patent.
Phase 2
Sexual maturation was unaffected by maternal dosages of the test substance as high as 175 mg/kg/d (Table 14). The average day on which preputial separation or vaginal patency occurred was comparable among the 4 dosage groups.
Sexual Maturation: F1-Generation Mice.
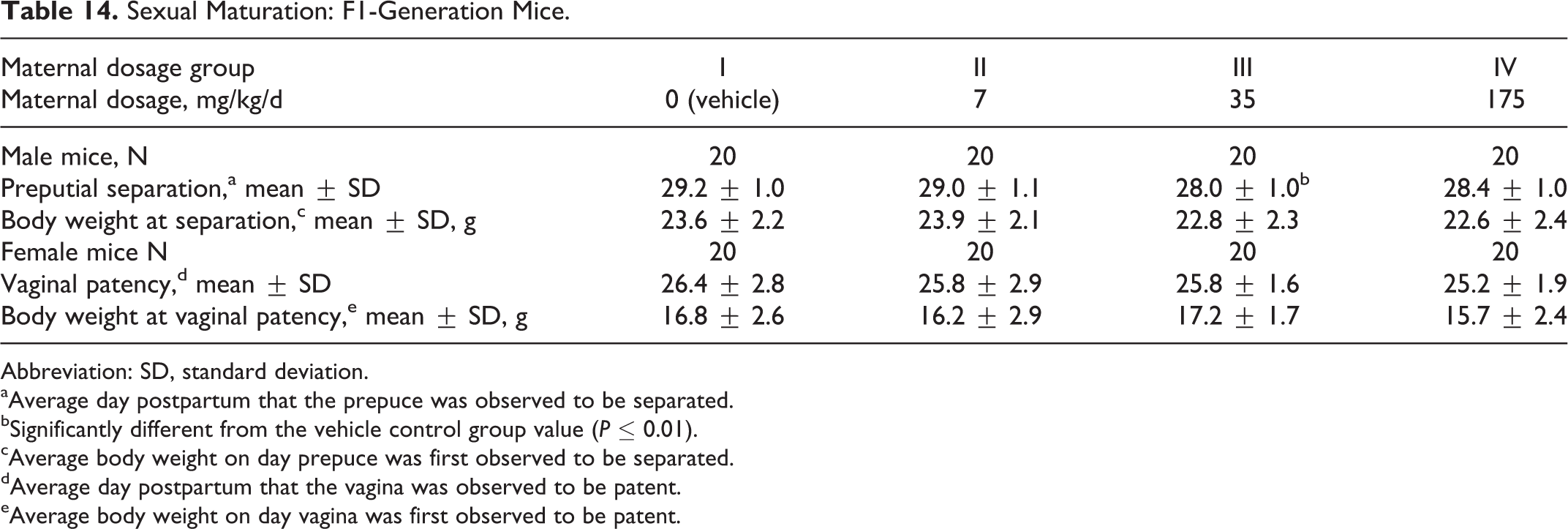
Abbreviation: SD, standard deviation.
aAverage day postpartum that the prepuce was observed to be separated.
bSignificantly different from the vehicle control group value (P ≤ 0.01).
cAverage body weight on day prepuce was first observed to be separated.
dAverage day postpartum that the vagina was observed to be patent.
eAverage body weight on day vagina was first observed to be patent.
F1-Generation Terminal Body Weights
Phase 1
Terminal body weights in the F1-generation male mice were comparable among the 4 groups (Table 15). The ratio of the liver weight to the terminal body weight was significantly reduced (P ≤ 0.05) in the 500 mg/kg/d dosage group compared to the control group value. This was considered related to PFHxA Ammonium Salt.
Terminal Body Weights, Liver Weights, and Ratios (%) of Liver Weight to Terminal Body Weight: F1-Generation Male Mice.a
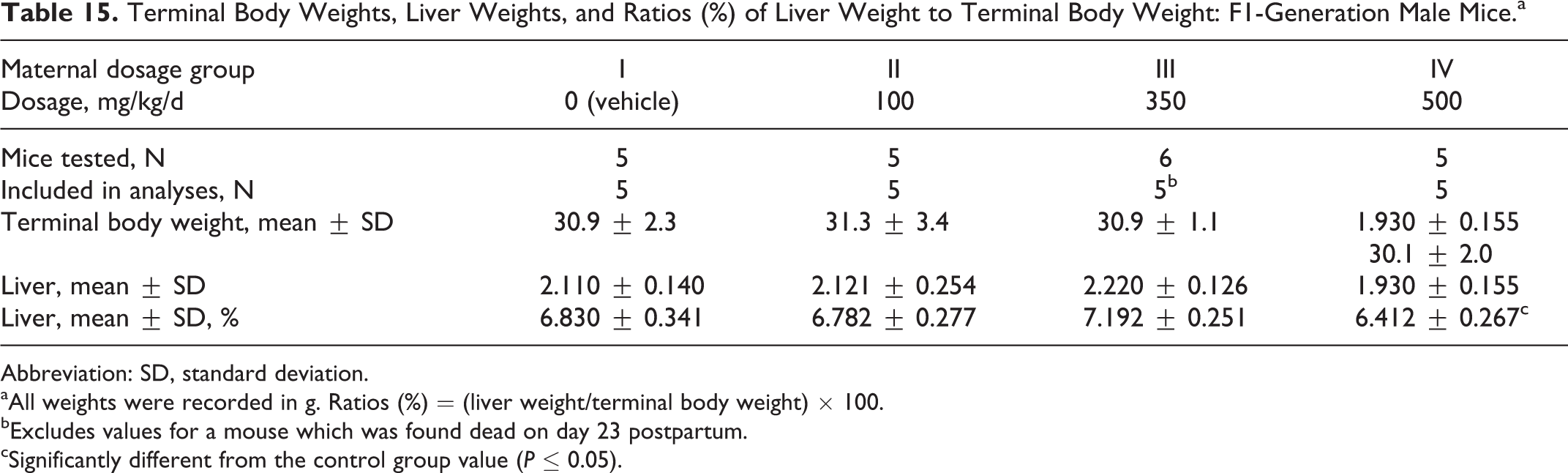
Abbreviation: SD, standard deviation.
aAll weights were recorded in g. Ratios (%) = (liver weight/terminal body weight) × 100.
bExcludes values for a mouse which was found dead on day 23 postpartum.
cSignificantly different from the control group value (P ≤ 0.05).
Terminal body weights in the F1-generation female mice were significantly reduced (P ≤ 0.05) in the 350 mg/kg/d dosage group compared to the control group value. Maternal dosages of the test substance as high as 500 mg/kg/d did not affect the liver weights or the ratio of liver weights to the terminal body weight (Table 16).
Terminal Body Weights, Liver Weights, and Ratios (%) of Liver Weight to Terminal Body Weight: F1-Generation Female Mice.a
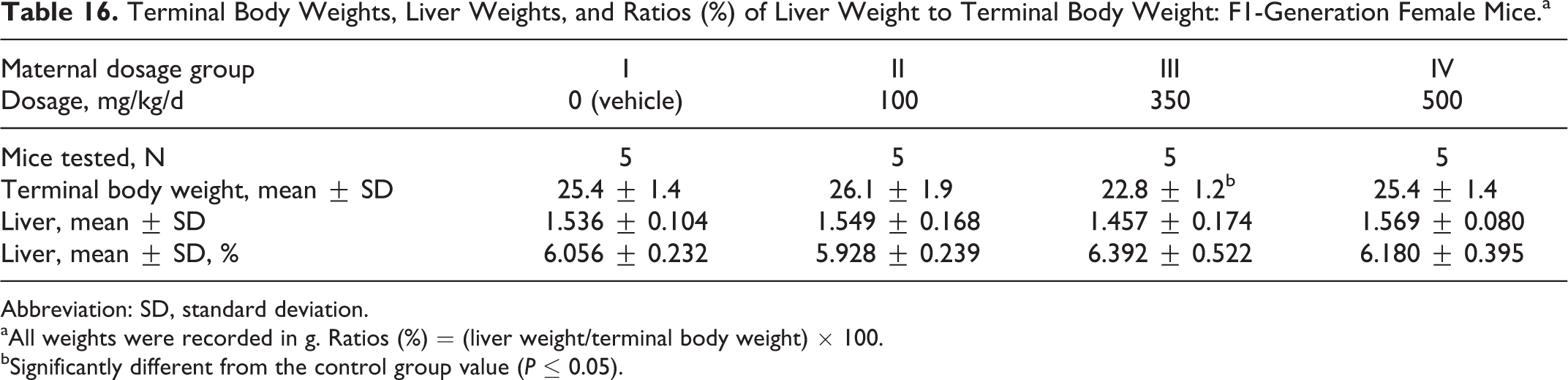
Abbreviation: SD, standard deviation.
aAll weights were recorded in g. Ratios (%) = (liver weight/terminal body weight) × 100.
bSignificantly different from the control group value (P ≤ 0.05).
Phase 2
Terminal body weights were comparable among the 4 groups (Table 17). Maternal dosages of the test substance as high as 175 mg/kg/d did not affect the liver weights or the ratio of liver weights to the terminal body weight (Table 18) for either sex. There were no significant differences among the groups.
Terminal Body Weights, Liver Weights, and Ratios (%) of Liver Weight to Terminal Body Weight: F1-Generation Male Mice.a
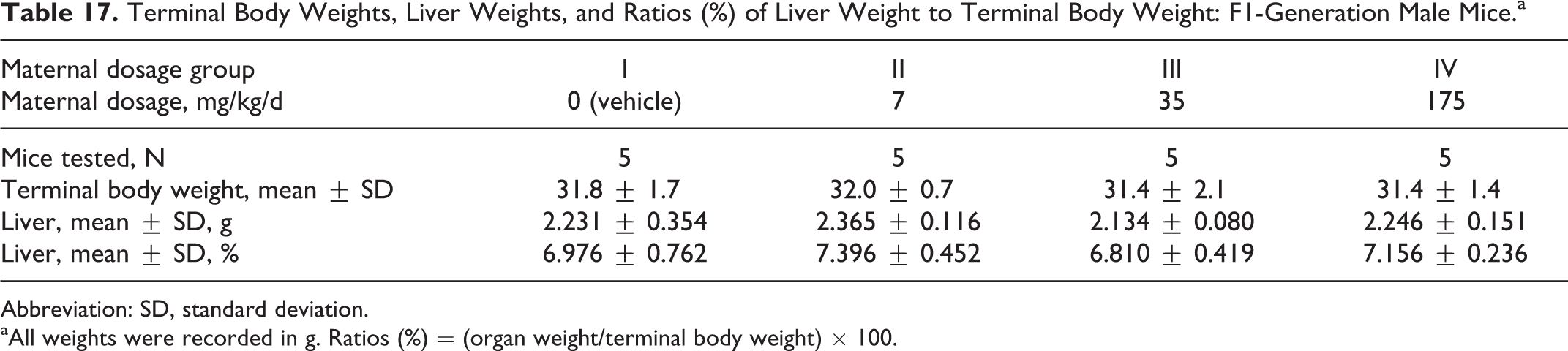
Abbreviation: SD, standard deviation.
aAll weights were recorded in g. Ratios (%) = (organ weight/terminal body weight) × 100.
Terminal Body Weights, Liver Weights, and Ratios (%) of Liver Weight to Terminal Body Weight: F-Generation Female Mice.a

Abbreviation: SD, standard deviation.
aAll weights were recorded in g. Ratios (%) = (organ weight/terminal body weight) × 100.
Levels of PFHxA in Blood and Liver Homogenates in F1 Generation
No detectable level of PFHxA was found in the blood or liver homogenate from any F1-generation male or female pup.
Discussion and Conclusion
Over the 2 phases of study conduct, F0-generation female mice were administered PFHxA and/or vehicle once daily from DG 6 through DG 18 at dosages of 0, 7, 35, 100, 175, 350, or 500 mg/kg/d. Dosages as high as 100 mg/kg/d produced no maternal or developmental toxicities. Maternally toxic doses of 175 mg/kg/d and higher produced increased number of stillborn pups and pups dying day 1 along with a reduction in pup weights on PPD 1, 2 litters with pups with corneal opacity, and delays in physical development.
The developmental toxicity of PFAAs and their derivatives was reviewed by Lau 28,29 in 2004 and again in 2007. Lau noted that developmental toxicity from PFAA exposure in rodents during gestation included embryo fetal death, teratological findings (malformations and variations), and reductions in fetal weight and these toxicities appeared only in the presence of maternal toxicity. Neonatal mortality as observed with PFAA was also observed with PFOS, again in a dose-dependent pattern. Cross-fostering of PFOS mouse pups did not improve the survival. 28 The mouse offspring exposed to PFOS at doses as low as 15 mg/kg/d did not survive 24 hours.
The mechanism of action of PFAAs toxicity is still being explored with reports of activation of peroxisome proliferator-activated receptors α demonstrated with PFOA and PFOS 29 and interference with the normal physiology function of pulmonary surfactants being 2 potential mechanisms. 30
Results of this study support what has been generally observed for other PFAAs in that developmental toxicity has generally only been seen in the presence of maternal toxicity. Similar to other PFAAs, the developmental toxicity observed has been related to pup survival and growth and not malformations, indicating that late gestational exposure to the test substance may be producing the toxicity observed. The dose of PFHxA that resulted in an effect on mouse pups was approximately 5 times the dose of PFOS that produced similar effects.
The PFHxA doses (175 and 350 mg/kg) in this study were chosen to approximate the area under the curves (AUCs) estimated to have resulted from doses of PFOA (5 and 10 mg/kg), at which severe developmental toxicity was previously indicated in CD-1 mice. 31 In studies of CD-1 mice, the developmental toxicity induced by exposure to PFOA throughout gestation included dose-related full litter resorptions, reduced postnatal survival, delayed eye opening, growth deficits, and sex-specific alterations in pubertal maturation. 31 Exposure to the C-4 chemical perfluorobutyric acid (PFBA) during pregnancy in the mouse did not recapitulate the profound adverse developmental effects previously reported with the C-8 chemicals PFOA and PFOS (neonatal morbidity and mortality, and postnatal growth deficits), although some subtler effects were still detectable. The milder responses of PFBA are likely accounted for by the rapid elimination of the chemical as well as its lower potency compared with the PFAAs with longer carbon chains. Similar to PFBA exposure, the absolute weights of the liver and the ratio of the liver weight to the terminal body weight did not differ significantly among all groups, and sexual maturation was also unaffected by maternal dosages PFHxA.
There are two 90-day repeated oral studies in rats of PFHxA and related salts. These studies included some reproductive end points. The NOAEL following 90 days of dosing with PFHxA was 50 mg/kg/d in male rats, based on liver weight increases and hepatocellular hypertrophy. 6 The NOAEL following 90 days of dosing with sodium perfluorohexanoate (NaPFHx) was 20 mg/kg/d, based on nasal lesions in male rats. 7 Both studies demonstrated higher liver weights, minimal hepatocellular hypertrophy, and slightly higher hepatic peroxisomal β-oxidation activity. The maternal and reproductive/developmental NOAELs for these studies was 100 mg/kg/d, based on reduced F1 pup weights. 7 This NOAEL was consistent with this study in mice.
Butenhoff and his colleagues conducted a comprehensive 2 generation reproductive toxicity study on PFOA with Sprague-Dawley rats and reported little toxicity, small postnatal weight gain deficits, slight delays of sexual maturation, and postweaning mortality (likely related to immaturity) were noted only in the F1-generation animals of the highest dose group (30 mg/kg). 32 The PFOA was less developmentally toxic than PFOS probably due, in part, to the efficient renal elimination of PFOA in the female rat, with a half-life estimate of 3 to 4 hours, compared to that of 6 to 8 days in the males. 33 –36 In contrast, no significant differences were seen between serum PFOA concentrations of male and female mice.
The toxicity of another C6 chemical, perfluorohexanesulfonic acid (PFHxS), has also been studied. Mean serum elimination half-life values in PFHxS were about 30 days between male and female mice. 37 The PFHxS reproductive and developmental toxicity study demonstrated, at 3 and 10 mg/kg/d, increased liver to body weight and liver to brain weight ratios, centrilobular hepatocellular hypertrophy, hyperplasia of thyroid follicular cells, and decreased hematocrit but no effects on reproduction or developmental toxicity. 38 However, it should be noted that because of the selectivity of the end points in this reproductive/developmental screening study, no definitive evidence of reproductive or developmental effects could be excluded.
Cassone and colleagues reported that in chick eggs effects of PFHxS and PFHxA exposure (maximum dose = 38 000 and 9700 ng/g egg, respectively) on embryonic death, developmental end points, tissue accumulation, messenger RNA expression in liver and cerebral cortex, and plasma thyroid hormone occurred. 39 Pipping success was reduced (37%) and tarsus length and embryo mass were reduced at the highest dose of PFHxS (38 000 ng/g). However, no effects were observed for PFHxA. 39 Free T4 levels in the plasma of developing chicken embryos exposed to PFHxS and PFHxA. PFHxS significantly decreased plasma-free T4 levels in a dose-dependent manner, whereas PFHxA had no effect on circulating thyroid hormone levels.
The depleted free T4 status observed in PFHxS-exposed embryos is in agreement with several of the altered end points presented in their study (ie, reduced pipping success, embryo growth, and increased deiodinase expression). Thyroid hormone-dependent neurodevelopmental pathways were affected in developing chicken embryos exposed to PFHxS. One thyroid hormone-independent mode of PFHxS action identified was gap junction intercellular communication (GJIC) via nephroblastoma-overexpressed gene (nov) downregulation. 40 The PFHxS was found to inhibit GJIC in a dose-dependent fashion, and this inhibition occurred rapidly and was reversible. 41 Upham and colleagues reported the effect of PFFAs on GJIC in WB-rat liver epithelial cells. The chain length of the PFFAs of 7 to 10 inhibited GJIC in a dose–response fashion, whereas carbon lengths of 2 to 5, 16, and 18 did not appreciably inhibit GJIC. Unfortunately, PFHxA was not specifically tested, but we expect that PFHxA would not inhibit GJIC. 42
Martin and colleagues compared bioconcentration factors (BCFs) for perfluoroalkyl carboxylates and sulfonates with the number of CF2 moieties. The PFHxS, which has 6 CF2 moieties, had a carcass BCF of about 10, whereas perfluoroheptanoic acid, which also has 6 perfluoroalkyl carbons, could not be detected in most tissues, despite a higher exposure concentration. 43
Some reports estimated that the BCF for PFHxA, which has 5 CF2 moieties, was about 0.1. 44,45 There is a 100 times difference in BCF between PFHxS and PFHxA. It appears that hydrophobicity of PFHxA is low and it hardly interacts with/permeates through the cell membrane. Therefore, this also suggests that PFHxA does not inhibit GJIC.
In this study, the body burden of PFHxA in F1 mice after weaning at postnatal day 41 was measured with detectable amount of the chemical found in the liver. Analysis of data from another study showed that the pharmacokinetics of PFHxA was characterized in female mice when administered once by oral gavage at dose levels of 35, 175, and 350 mg/kg (Table 19). The PFHxA was rapidly absorbed (Cmax reached within 30 minutes) and in general was not quantifiable at 24 hours after dose administration. The terminal elimination half-life of PFHxA ranged from 0.889 to 1.24 hours and was dose independent. The increase in Cmax (from 96.6 to 454 µg/mL) was lower than proportional, whereas the increase in AUC(0-inf) (from 178 to 1893 h µg/mL) was proportional to the increase in the dose from 35 to 350 mg/kg. In a mouse multiple-dose study,13 daily doses of orally administered PFHx Ammonium Salt was followed by a single oral administration of [ 14 C]-PFHxA Ammonium Salt. Excretion was rapid, with mean recoveries of over 90% of the administered dose (mean values >95% of the ultimately recovered material) at 24 hours after dosing, irrespective of gender 8 ; thus, by PD 22, most of the PFHxA Ammonium Salt would eliminated, even if one assumes a preferential accumulation of PFHxA in the liver (like PFOA). This most likely has to do with the rapid elimination of PFHxA as with PFBA.
Pharmacokinetic Exposure Parameters of PFHxA in Female Crl: CD1(ICR) Mice Serum Following Oral Gavage of PFHxA Ammonium Salt.

Abbreviations: AUC, area under the curve; PFHxA, Perfluorinated Hexanoic Acid.
Kudo and colleagues reported that the potency of the induction of hepatomegaly, peroxisomal β-oxidation, and microsomal 1-acylglycerophoshocholine(1-acyl-GPC) acyltransferase were compared among PFAAs with 6 to 9 carbon chain length in the liver of male and female mice. 46
The longer the perfluoroalkyl chain, the more PFAA accumulates in the liver of both male and female mice. The accumulated PFAAs induced hepatomegaly, peroxisomal β-oxidation, and microsomal 1-acyl-GPC acyltransferase, with little PFHxA accumulation in the liver.
Log octanol–water partition coefficient (Po/w) of PFBA, PFHxA, and PFOA using liquid chromatography–tandem mass spectrometry is 1.43, 2.51, and 3.60 (in our data). The half-life of PFBA would be faster than PFHxA. Mean terminal serum PFBA elimination half-lives were 1 to 10 hours in the rat, about 10 hours in the mouse, 40 hours in monkey, and approximately 3 days in humans. The elimination half-life of PFHxA in rats ranged between 0.4 and 9.8 hours, in mouse about 1 hour, in monkeys 14-47 hours, and in humans it is 14-49 days. It seems there was no difference in the elimination half-lives of PFBA and PFHxA. A comparison of elimination rates of PFHxA among various species (including mouse and humans) has been made by Russell and colleagues and Chang and colleagues. 47,48 Unlike PFHxS, perfluorooctanesulfonic acid (PFOS), and PFOA and like PFBA, PFHxA is rapidly eliminated, which may contribute to higher NOAELS for developmental and reproductive parameters in mammalian species.
On the basis of the data from this study, the maternal NOAEL for PFHxA Ammonium Salt is 100 mg/kg/d. The NOAEL in the F1 generation is also 100 mg/kg/d. None of the effects observed in the pups preweaning at any dose level persisted into the postweaning period.
Footnotes
Authors’ Note
The research for this article was conducted entirely at Charles River Laboratories under the sponsorship of Daikin Industries Ltd.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
