Abstract
7-Acetyl-1,1,3,4,4,6-hexamethyl-1,2,3,4-tetrahydronaphthalene (AHTN ) and 1,3,4,6,7,8-hexahydro-4,6,6,7,8,8-hexamethylcyclopenta-gamma-2-benzopyran (HHCB) are polycyclic musks widely used as fragrance ingredients in consumer products. Because their metabolic fate following systemic exposure is not fully characterized, disposition and excretion of 14C-AHTN- and 14C-HHCB-derived radioactivity were studied in Sprague-Dawley rats and domestic pigs following a single intravenous dose. Rats administered with AHTN or HHCB excreted 21% or 28% of the radioactivity in urine and 67% or 61% in feces, respectively, within 7 days. In pigs administered AHTN or HHCB, 86% or 74% of the dose was excreted in the urine, and 12% or 15% in feces, respectively, during the 14-day collection period. Radioactivity in the whole blood and plasma of both species and tissues of rats declined steadily until the end of the study (28 days) for both the materials. Radioactivity in rat adipose tissue reached peak at 2 hours after dosing, decreasing steadily thereafter. Radioactivity in pig blood declined rapidly from 70 ng equivalents/g at 10 minutes to 1 ng equivalent/g or less by 28 days after administration of either AHTN or HHCB. Radioactivity in pig skin and adipose tissue decreased to below the limit of detection by 28 days for both the materials. Thin-layer chromatography showed multiple radioactive components in both species’ urine after administration of either material. Components found in the urine of the 2 species were qualitatively similar but quantitatively different. Both AHTN and HHCB were completely metabolized and excreted. No unchanged parent compound was detected in rat or pig urine.
Keywords
Introduction
7-Acetyl-1,1,3,4,4,6-hexamethyl-1,2,3,4-tetrahydronaphthalene (AHTN) and 1,3,4,6,7,8-hexahydro-4,6,6,7,8,8-hexamethylcyclopenta-gamma-2-benzopyran (HHCB) are polycyclic musks (Figure 1) used as fragrance materials. The chemical stability of polycyclic musk compounds has led to their extensive use in a variety of cosmetic products including body lotion, perfumes and colognes, shampoo, bath products, shower gel, toilet soap, and hair spray. Based upon the upper 97.5 percentile level of these materials in fragranced consumer products and the average amount of these products applied daily (g), human systemic exposure to AHTN and HHCB is estimated to be 18.6 mg/d (0.31 mg/kg/d) and 25.8 mg/d (0.43 mg/kg/d), respectively. 1 Since these products are primarily applied to skin or clothes, exposure is expected to be mostly percutaneous.
Chemical structures of AHTN and HHCB. AHTN indicates 7-acetyl-1,1,3,4,4,6-hexamethyl-1,2,3,4-tetrahydronaphthalene; HHCB, 1,3,4,6,7,8-hexahydro-4,6,6,7,8,8-hexamethyl-cyclopenta-gamma-2-benzopyran.
Chemical stability coupled with high lipophilicity explains the tendency of these materials to bioconcentrate. Chemical stability has also resulted in their detection in water supply and discharge flows of municipal clarification units and in fish. 2,3 They have also been detected in human adipose tissue. Rimkus and Wolf 4,5 reported that human adipose tissue contains AHTN and HHCB at levels of 8-33 and 28-189 ng/g fat, respectively. In another study, Muller et al 6 reported similar concentrations of AHTN and HHCB (1-23 and 12-171 ng/g fat, respectively) in human adipose tissue. High lipophilicity also explains the detection of polycyclic musks in breast milk. In 107 human breast milk samples obtained from nursing mothers, AHTN and HHCB were detected in 88 samples at mean levels of 16 ng/g fat and 80 ng/g fat, respectively. 7 These concentrations are similar to those reported by Rimkus and Wolf. 4 The presence of AHTN and HHCB in breast milk and adipose tissue suggests some dermal absorption.
Numerous toxicological studies have been conducted on these materials, demonstrating no genotoxic potential, 8,9 no adverse effects in repeat-dose toxicity testing, 10 –15 adverse effects on development only at doses that were toxic to dams, 16 and estrogenic activity at a level too low to affect humans or wildlife species 17 or in another study, no estrogenic potency at all. 18 Green discoloration of liver, mesenteric lymph nodes, and lachrymal glands was observed in a 13-week dietary repeat-dose test with AHTN, but only at higher doses. 19 The nature of this green discoloration has not been established, and although due to the administration of the materials, it was not considered to be toxicologically significant.
In previous studies, 20 dermal application of 14C-labeled AHTN and HHCB for 6 hours to rats resulted in the systemic absorption of 19% and 14% of the applied dose, respectively. Most of the absorbed dose was excreted in the feces (14.5% AHTN and 11.6% HHCB), with only 2.1% (AHTN) and 1.3% (HHCB) of the dose excreted in the urine. A large concentration of the material was also found in the intestines of rats and their contents during early periods after application. Studies conducted in 3 human volunteers 20 showed no significant excretion of radiolabel in urine or feces collected 120 hours after dermal application of either 14C-AHTN or 14C-HHCB. This suggests minimal percutaneous absorption of these materials by human skin.
To better understand the disposition of systemically available AHTN and HHCB as would occur following percutaneous absorption, a distribution and excretion study following intravenous (iv) administration was conducted in female rats and male domestic pigs. Excretion of the radioactivity was measured in urine and feces over several days following a single iv dose of 14C-AHTN or 14C-HHCB. In addition, the levels of 14C equivalents of AHTN and HHCB were determined in plasma, whole blood, adipose tissue, liver, and kidney of rats and in pig skin and underlying adipose tissue. In the rat, plasma and adipose tissue were analyzed for the presence of parent compound. In both the rat and the pig, urine was analyzed for the presence of parent compound and metabolites.
Materials and Methods
Materials
Ring-labeled 14C-AHTN (28.5 mCi/mmol; radiochemical purity >98.4%) and 14C-HHCB (36.7 mCi/mmol; radiochemical purity >98.7%) were obtained from Wizard Laboratories Inc (West Sacramento, California). The reported radiochemical purities were confirmed in house by thin-layer chromatography (TLC) using 3 different solvent systems. Nonradiolabeled AHTN (>99% purity) was obtained from Givaudan-Roure SA, Geneva, Switzerland); nonradiolabeled HHCB (98% purity) was obtained from International Flavors and Fragrances, Union Beach, New Jersey.
Animals
Female Sprague-Dawley rats (Crl: CDBR; age 10-11 weeks, weighing 213-230 g) were obtained from Charles River, UK Ltd, Margate Kent, United Kingdom. Animals were housed in stainless steel cages with suspended mesh floors. They were maintained in rooms with a 12-hour light/dark cycle at (21 ± 2°C) with a relative humidity of 40% to 60%. Food and water were provided ad libitum.
A total of 52 rats were administered a single iv dose, (via the tail vein) of ring-labeled 14C-AHTN or 14C-HHCB at the dose of 2 mg/kg (based on previous studies). The vehicle was ethanol–Emulphor EL 620–isotonic saline (1:1:7, v/v/v). This dose, in excess of typical human systemic exposure from fragrance and cosmetic use, was chosen to match the previously studied doses used to investigate the transfer of these materials across the placenta and into milk of rats during and after pregnancy. 21,22 The rats were maintained in metabolism cages that allowed exhaled air to be drawn through 2 traps that contained 2-ethoxyethanol/ethoxyamine. Contents of the traps were collected at 24-hour intervals up to 48 hours. Urine and feces were collected at 24-hour intervals from those animals maintained for 7 days. Rats were euthanized in groups of 4 at 5, 15, and 30 minutes, at 1, 2, 4, 6, 12, 24, and 48 hours, and at 7, 14, and 28 days. Immediately prior to being euthanized, the blood samples were obtained by cardiac puncture. Blood and plasma were retained for analysis,at −20°C. At necropsy, the samples of adipose tissue and the liver and kidneys were taken for analysis. Carcasses were also retained for analysis.
Two male domestic pigs, Sus scrofa, of large white hybrid strain (age 8-12 weeks, body weight 33 kg) were obtained from R. Beedles, Shadymoor, Dorrington, Shropshire, United Kingdom and acclimatized for 12 days before dosing. They were housed in a pen of metal construction with wood shavings as bedding material. Prior to dosing and during collection of excreta, the pigs were maintained in stainless steel metabolism cages to facilitate separate collection of urine and feces. Food was provided twice daily, and water was available ad libitum.
One pig was used for each test material. A single intravenous dose of 14C-AHTN or 14C-HHCB (0.1 mg/kg) was administered via ear vein using a vehicle of ethanol–Emulphor EL 620–isotonic saline (1:1:7, v/v/v). The dose level of 0.1 mg/kg was chosen to mimic the systemic exposure to humans from an eau de toilette product (0.12 mg/kg exposure to AHTN for a consumer weighing 60 kg 23 ). Urine was collected from the metabolism cages after 6 hours, after 24 hours, and thereafter daily for up to 14 days. Feces were collected at 24-hour intervals over the same period. Blood samples were obtained from the jugular vein at predose, 10, 20, 40 minutes, at 1, 2, 4, 8, 12, 24 hours, and at 2, 3, 5, 7, 14, 21, and 28 days, respectively. Adipose tissue and skin biopsies (8 mm diameter) were taken at 9 and 16 days after administration of the dose under sedation and local anesthesia. Samples of skin and underlying adipose tissue (about 25 g) were collected from the carcass at day 28 when the animal was euthanized.
Measurement of Radioactivity
Liquid samples (urine, cage washings, plasma, cage air traps, and fecal extracts) were mixed with MI-31 scintillation cocktail (Canberra Packard Instrument Co Ltd, Pengbourne, Berkshire, United Kingdom) for measurement of radioactivity. Fecal residues and liver samples were combusted in oxygen using an automatic sample oxidizer (model 307 Tri-Car, Canberra Packard Instrument Co Ltd). Products of combustion were absorbed into Optisorb 1 and mixed with Optisorb S scintillator (Fisons Chemicals, Loughborough, United Kingdom) for measurement of radioactivity. Adipose tissue and kidney samples were solubilized with NCS-II tissue solubilizer at 55°C for 18 hours and then counted for radiolabel content. Radioactivity in amounts less than twice the background levels was considered to be below the limit of accurate determination.
Determination of Parent Compound in Rat Plasma and Adipose Tissue
Aliquots of plasma from the rats of each time point were pooled, extracted twice with acetonitrile, centrifuged, and the supernatant was concentrated by rotary evaporation. The residue was dissolved in 1.5 mL of acetonitrile and subjected to high-performance liquid chromatography (HPLC). Aliquots of adipose tissue were also pooled and homogenized by time point. The homogenates were extracted with acetonitrile (2×), n-hexane (2×), and water (1×). The extracts were pooled, concentrated by rotary evaporation, and analyzed as for plasma.
Aliquots of the resulting acetonitrile solution (100-250 μL for plasma and 50 μL adipose tissue) were applied to a reverse phase Spherisorb ODS1 column (25 cm × 4.6 mm) that was coupled to a SP 8800 HPLC system (Thermoseparation Products, St. Albans, United Kingdom). The mobile phase (acetonitrile–water 60:40) was maintained at a flow rate of 1 mL/min. Fractions were collected every minute for 30 minutes and subjected to radiochemical analyses. The HHCB eluted at 13 minutes and AHTN at 12 to 13 minutes. Radioactive peaks considered to be HHCB metabolites eluted between 4 and 9 minutes, and AHTN metabolites eluted between 2 and 4 minutes.
Characterization of Urinary Radioactivity by TLC
Urine from pigs was pooled to prepare collections representing 6 to 24 hours (AHTN), and 0 to 24 hours (HHCB). Urine from rats, collected between 0 and 24 hours was also pooled to prepare a representative sample for each compound. Aliquots of these urine samples were analyzed by TLC directly or after incubation with β-glucuronidase (1000 units/mL), or after incubation with aryl sulfatase (10 units). Enzyme incubations occurred at 37°C for 18 hours at pH 5 and allowed for determination of phase II conjugates. Normal phase TLC was carried out on prelayered, glass-backed Kieselgel 60 F254 plates (Merck Ltd, Poole, United Kingdom), with a layer thickness of 0.26 mm. The urine samples were applied as 2-cm wide bands. The developing systems were chloroform that demonstrated the absence of parent compound in the urine. The other solvent system was chloroform–methanol–ammonia (80:20:1 by volume). Preliminary studies using a variety of solvent systems demonstrated that this system gave the best separation of urinary components presumed to be metabolites. Images of the developed plates were obtained and quantified using a Fujix BAS 2000 Autoradiographic Imaging System (Fuji Photofilm Co, Japan). An associated personal computer employing PC-BAS 1000 software (Raytek Scientific Ltd, Sheffield, United Kingdom) was used for generation of linear-scaled radiochromatograms. Following development of the TLC plate, chromatographic correspondence was assessed by visual inspection of the plate under ultraviolet light and its associated radiochromatograms. Regions of radioactivity considered to be metabolites are referred to as H1-H13, each of which relates to Rf (distance migrated relative to the application site)
Results
Excretion of Radioactivity
In rats, total recovery of radioactivity at 7 days was 94% ± 1.7% for AHTN and 92% ± 1.9% for HHCB. The mean recovery of dose in feces was 67% ± 2.8% and 67% ± 4.5% for AHTN and HHCB, respectively. In urine, these recoveries were 21% ± 2.5% and 28% ± 2.5% for AHTN and HHCB, respectively (Figure 2). Small concentrations of radioactivity were present in kidney, liver, and carcass. No radioactivity was detected in air traps. For both the compounds, most of the urinary excretion occurred in the first 24 hours, whereas most of the fecal excretion occurred between 24 and 72 hours.
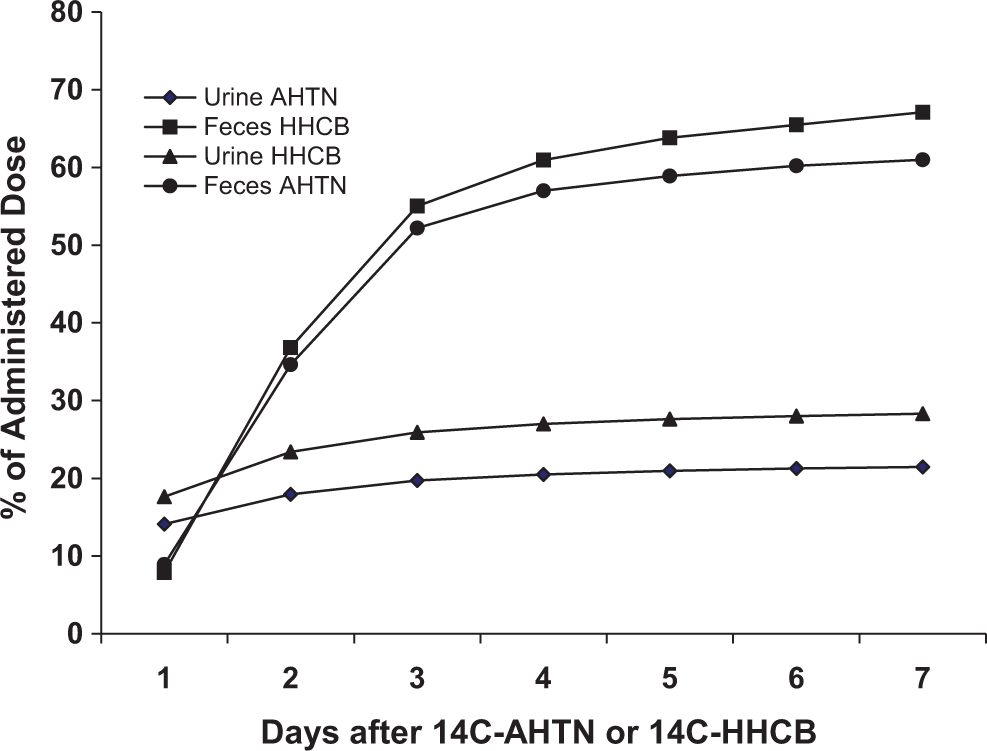
Cumulative excretion of radioactivity from 14C-AHTN and 14C-HHCB in urine and feces of rats following an intravenous injection of 2 mg/kg. Results are mean percentage excretion of radioactivity in specimens collected from 4 rats. Fecal excretion of the radioactivity was significantly greater than urinary excretion. 14C-AHTN indicates 7-acetyl-1,1,3,4,4,6-hexamethyl-1,2,3,4-tetrahydronaphthalene; 14C-HHCB, 1,3,4,6,7,8-hexahydro-4,6,6,7,8,8-hexamethyl-cyclopenta-gamma-2-benzopyran.
In pigs, the major route of elimination of both the compounds over 14 days was in the urine, 86.2% and 73.5% for AHTN and HHCB, respectively. Excretion in the feces was 12.2% for ATHN and 14.6% for HHCB (Figure 3). For both the compounds, most of the radioactivity in urine was excreted in the first 24 hours and in the feces between 24 and 48 hours.
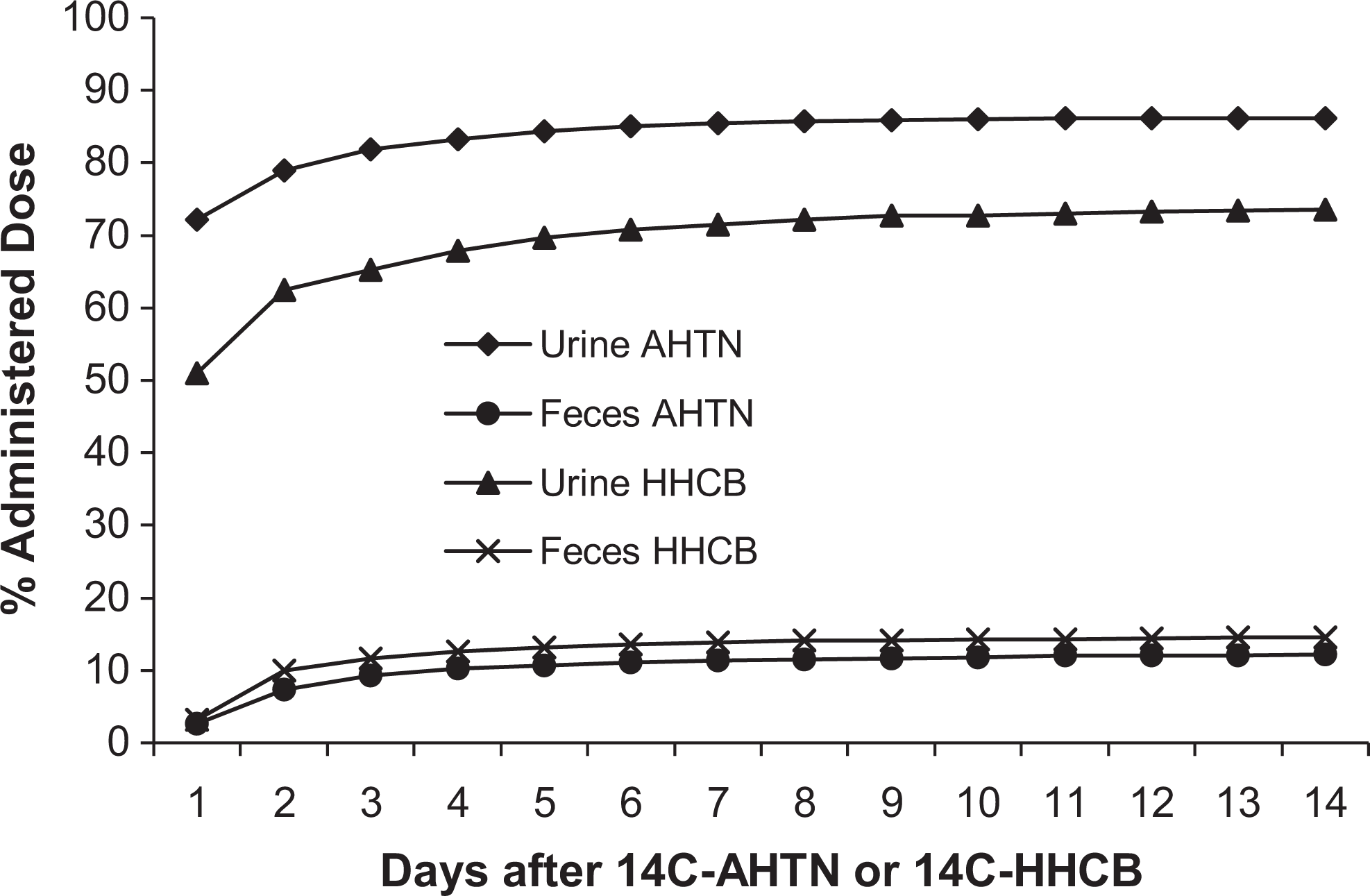
Cumulative excretion of 14C-AHTN or 14C-HHCB in urine and feces of the pig following intravenous injection of 0.1 mg/kg. Urinary excretion of the radioactivity was significantly greater than fecal excretion.14C-AHTN indicates 7-acetyl-1,1,3,4,4,6-hexamethyl-1,2,3,4-tetrahydronaphthalene; 14C-HHCB, 1,3,4,6,7,8-hexahydro-4,6,6,7,8,8-hexamethyl-cyclopenta-gamma-2-benzopyran.
Distribution of Radioactivity in Plasma and Tissues
Figures 4 and 5 illustrate the uptake and decline of 14C-AHTN- and 14C-HHCB-derived radioactivity in plasma, whole blood, adipose tissue, kidney, and liver of rats. Following intravenous administration to rats, peak plasma concentrations of radioactivity occurred at 5 minutes and were 2.6 ± 0.2 and 2.6 ± 0.1 μg equivalents/g for AHTN and HHCB, respectively. In whole blood, concentrations of radioactivity were lower than in plasma but followed the same pattern. For both blood and plasma, concentrations of radioactivity were very low by day 28, between 0.0005 and 0.09 μg equivalents/g for both the materials. Peak liver and kidney levels of radioactivity also occurred at 5 minutes and were 9.6 ± 0.8 and 4.6 ± 0.5 μg equivalents/g for 14C-AHTN and 8.8 ± 0.4 and 4.6 ± 0.1 μg equivalents/g for 14C-HHCB. By day 28, the radioactivity levels in tissues of both the species for both the compounds had declined to very small levels (about 0.03 μg equivalents/g). Radioactivity in the adipose tissue did not reach peak concentrations until 2 hours after dosing and were 5.1 ± 1.5 μg equivalents/g for 14C-AHTN and 6.6 ± 1.9 μg equivalents/g for 14C-HHCB. These concentrations remained relatively stable until about 24 hours, at which time they began to decline to concentrations of 0.01 to 0.03 μg equivalents/g at the end of the study.
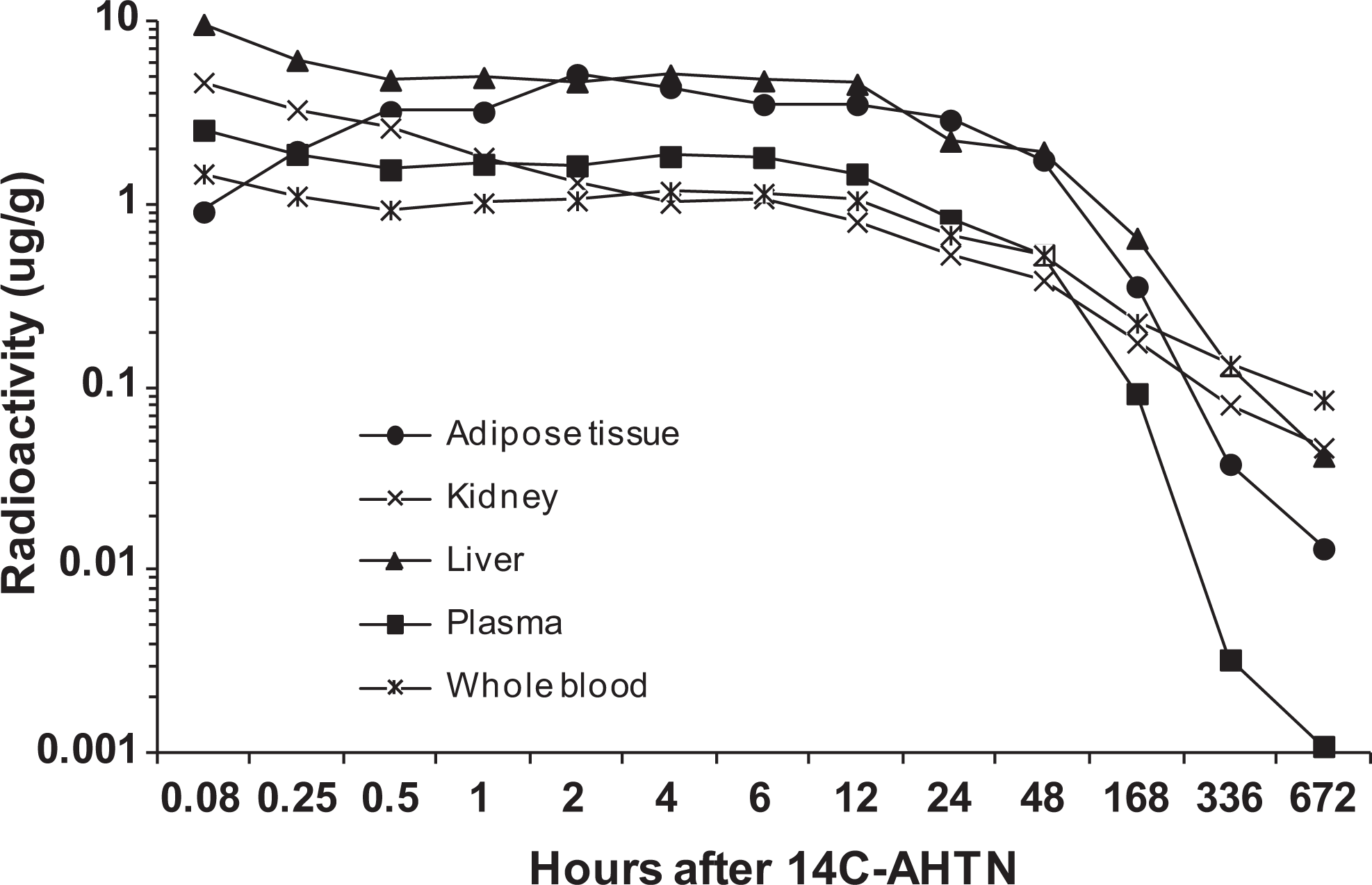
Distribution of radioactivity in rat tissues following intravenous injection of 14C-AHTN. Groups of 4 rats were killed at different time periods and radioactivity in the tissues was measured. Results are expressed as mean at each time point.14C-AHTN indicates 7-acetyl-1,1,3,4,4,6-hexamethyl-1,2,3,4-tetrahydronaphthalene.
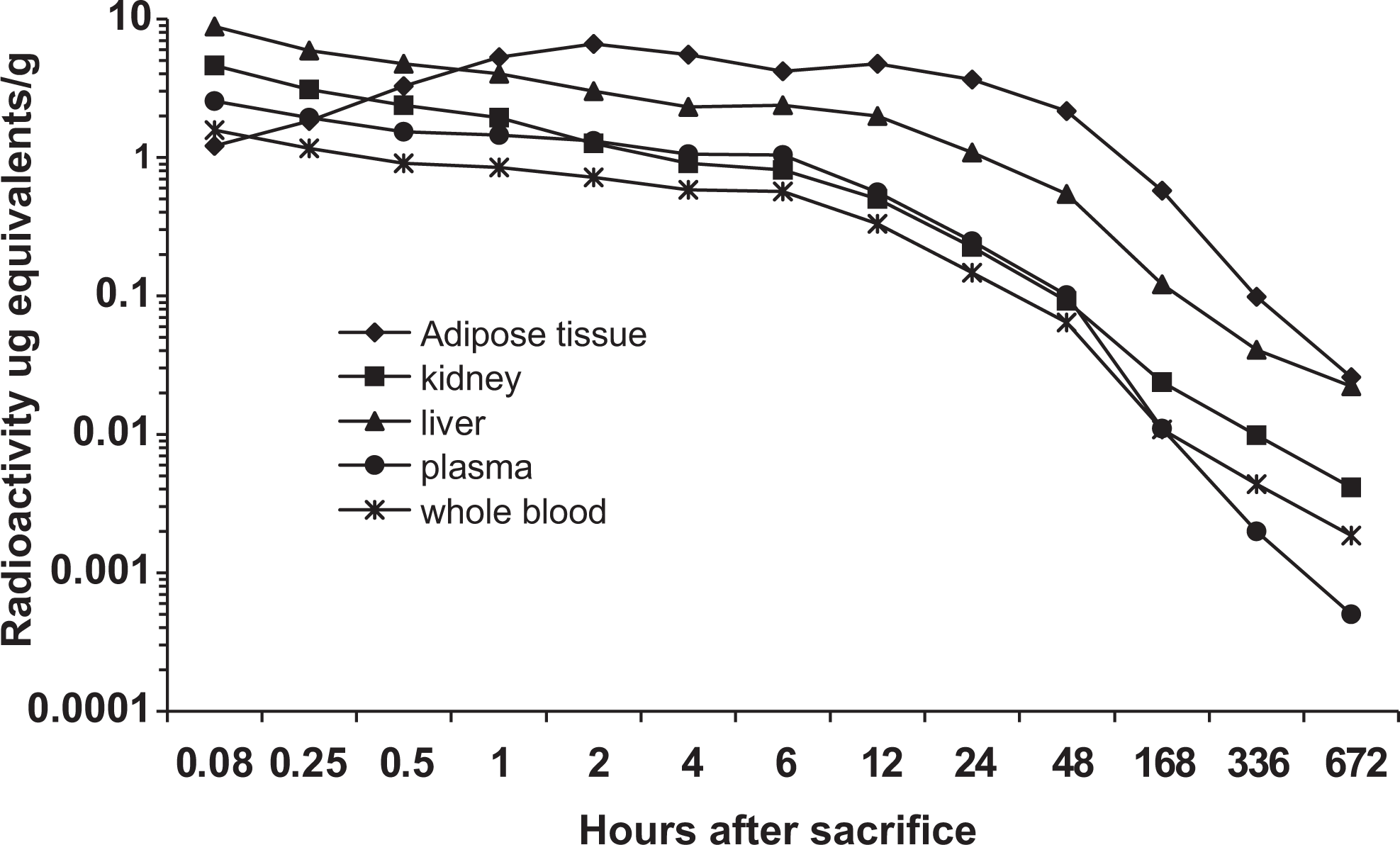
Distribution of radioactivity in rat tissues following intravenous injection of 14C-HHCB. Groups of 4 rats were sacrificed at different time periods and radioactivity in the tissues was measured. Results are expressed as mean at each time point. 14C-HHCB indicates 1,3,4,6,7,8-hexahydro-4,6,6,7,8,8-hexamethyl-cyclopenta-gamma-2-benzopyran.
The HPLC analysis of the plasma radioactivity in rat at 2 hours, 1 day, and 7 days revealed that only a small fraction was associated with parent AHTN or HHCB. Most of the radiolabel eluted at 4 to 9 minutes (HHCB) and 2 to 4 minutes (AHTN), which was well separated from the parent compound that eluted at 13 minutes (HHCB) and 12 to 13 minutes (AHTN). In adipose tissue of rats obtained at 2 hours, 1 day, and 7 days, most, but not all, of the radiolabel eluted at 12 to 13 minutes, the same time as parent 14C-AHTN or 14C-HHCB. Table 1 shows the levels (ug/g) of AHTN and HHCB found in the plasma and adipose tissue of rats at 2 hours, 1 day, and 7 days. The ratio of AHTN or HHCB found in adipose tissue compared to plasma is also given, and it indicates that plasma levels of AHTN and HHCB decreased by orders of magnitude from 2 hours to 1 day after dosing, while in adipose, the levels were much more stable during the same time frame. At all time points, the amount of AHTN and HHCB found in adipose tissue was higher than the amount found in plasma.
The Effect of Time on the Mean Concentrations (µg/g) of AHTN and HHCB in Adipose Tissue and Plasma and on the Adipose Tissue to Plasma Ratio of Female Rats Following a Single Intravenous Dose of 14C-AHTN or 14C-HHCB (2 mg/kg).a

Abbreviations: ND: below the limits of accurate detection; 14C-AHTN, 7-acetyl-1,1,3,4,4,6-hexamethyl-1,2,3,4-tetrahydronaphthalene; 14C-HHCB, 1,3,4,6,7,8-hexahydro-4,6,6,7,8,8-hexamethyl-cyclopenta-gamma-2-benzopyran.
a Results are expressed as ug/g; n = 4 for each material at each time point.
b Ratio is the proportion of material found in adipose tissue compared to plasma.
The pharmacokinetic behavior of 14C-AHTN and 14C-HHCB in the plasma of the pig was nearly identical (Figure 6). Maximum concentration occurred at the earliest time point (10 minutes) and was 108 ng equivalents/g. In whole blood at 10 minutes, the concentration was lower at 70 ng equivalents/g. By 2 hours, the concentrations had declined to 26 to 34 ng equivalents/g for plasma and 17 to 21 ng equivalents/g for blood. These levels continued to decline steadily, with time to almost nondetectable levels by day 28. Corresponding concentrations in skin and adipose tissue followed a similar pattern, with maximum concentrations at the earliest sample decreasing to very low levels by day 28. Radioactivity in the earliest sample of skin, biopsied 9 days after dosing, was 1.08 and 3.8 ng equivalents/g for 14C-AHTN and 14C-HHCB, respectively. In pig adipose tissue, these concentrations were 7.6 and 30.5 ng equivalents/g at 9 days for AHTN and HHCB, respectively. By day 28, the radioactivity in both skin and adipose tissue decreased by at least a factor of 10.
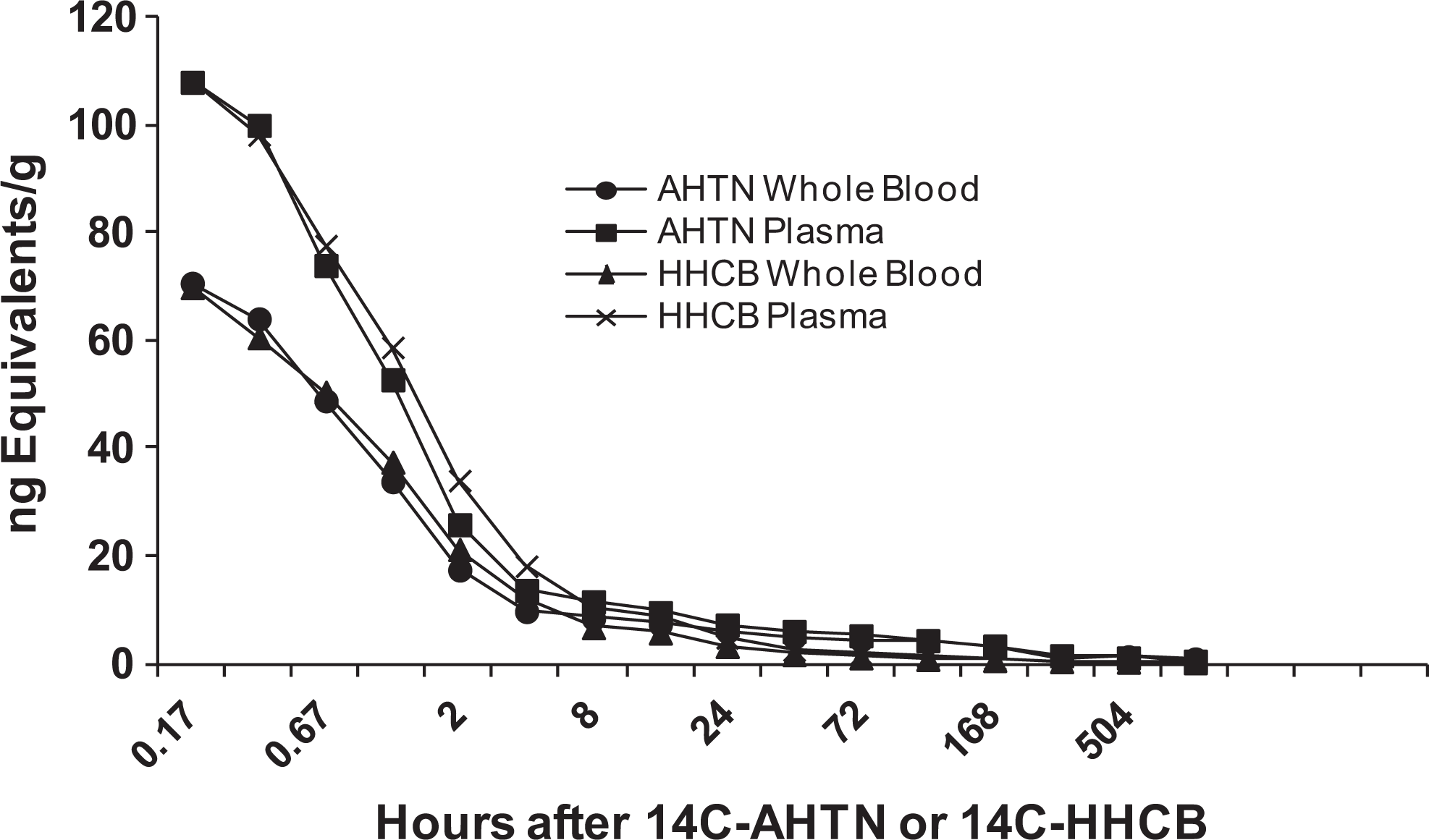
Concentrations of radioactivity in whole blood and plasma of the pig following intravenous injection of 14C-AHTN or 14C-HHCB at 0.1 mg/kg. Sharp decline in radioactivity was noticed in the first 4 hours after administration. 14C-AHTN indicates 7-Acetyl-1,1,3,4,4,6-hexamethyl-1,2,3,4-tetrahydronaphthalene; 14C-HHCB, 1,3,4,6,7,8-hexahydro-4,6,6,7,8,8-hexamethyl-cyclopenta-gamma-2-benzopyran.
Metabolite Profile
Thin-layer chromatography using chloroform as the solvent system separated parent AHTN and HHCB from radiolabeled components present in rat and pig urine. This system revealed that no parent for either compound was excreted in the urine of either species. When 0 to 24 hours pooled urine obtained from rats treated with 14C-ATHN was subjected to TLC analysis using chloroform–methanol–ammonia (80:20:1) as the solvent system, 8 discrete radiolabeled components were obtained (Table 2). These represented 0.17% to 3.4% of the administered dose. Enzyme treatment (β-glucuronidase or aryl sulfatase) produced minimal, if any, changes in the TLC profile. The TLC analysis of pig urine (6-24 hours; the 0-6 hours sample was lost) revealed 9 radiolabeled components (Table 2), representing 2% to 19% of the dose. β-Glucuronidase treatment resulted in the appearance of 3 new components (H3, H12, and H13), each representing <2% of the dose.
Proportions of Radioactive Components in Urine After Intravenous Doses of 14C-AHTN to Female Rats (2 mg/kg) and a Male Pig (0.1 mg/kg).a
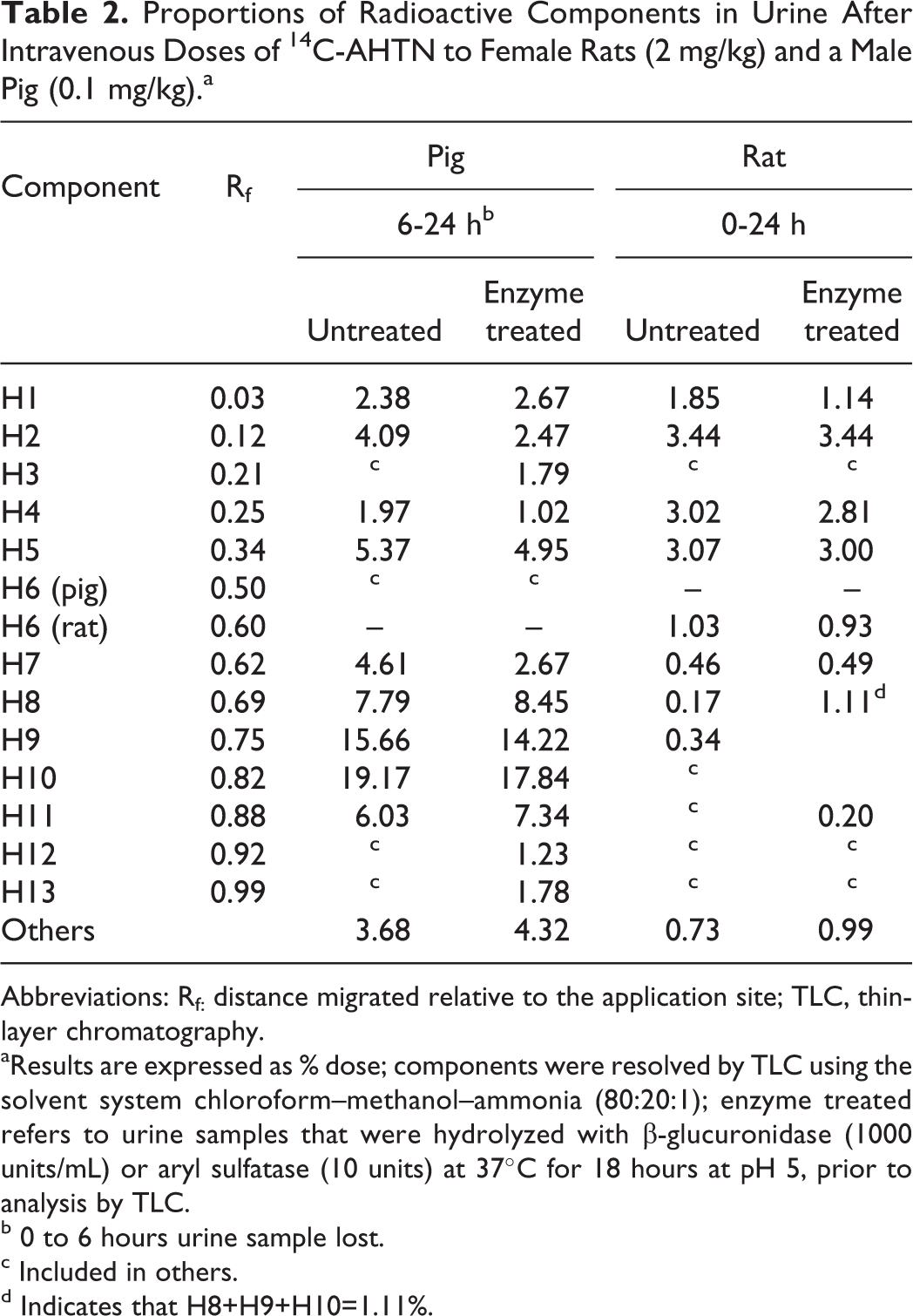
Abbreviations: Rf: distance migrated relative to the application site; TLC, thin-layer chromatography.
aResults are expressed as % dose; components were resolved by TLC using the solvent system chloroform–methanol–ammonia (80:20:1); enzyme treated refers to urine samples that were hydrolyzed with β-glucuronidase (1000 units/mL) or aryl sulfatase (10 units) at 37°C for 18 hours at pH 5, prior to analysis by TLC.
b 0 to 6 hours urine sample lost.
c Included in others.
d Indicates that H8+H9+H10=1.11%.
Nine discrete radioactive components were found in rat urine (0-24 hours) after 14C-HHCB administration (Table 3). These represented 0.24% to 3.9% of the dose. Enzyme treatment (β-glucuronidase) reduced component H1, with the appearance of 2 new components, H10 and H14. The TLC of pig urine showed 12 radioactive components (Table 3). The 6 major metabolites were components H1, H2, H8, H10, H11, and H12, which accounted for 36%, 1%, 1%, 2.7%, 1.3%, and 5.6% of the dose, respectively. The major changes after β-glucuronidase incubation were a 90% reduction in H1, and 3- to 4-fold increases in H9, H10, H11, and H12.
Proportions of Radioactive Components in Urine After Intravenous Doses of 14C-HHCB to Female Rats (2 mg/kg) and a Male Pig (0.1 mg/kg).a
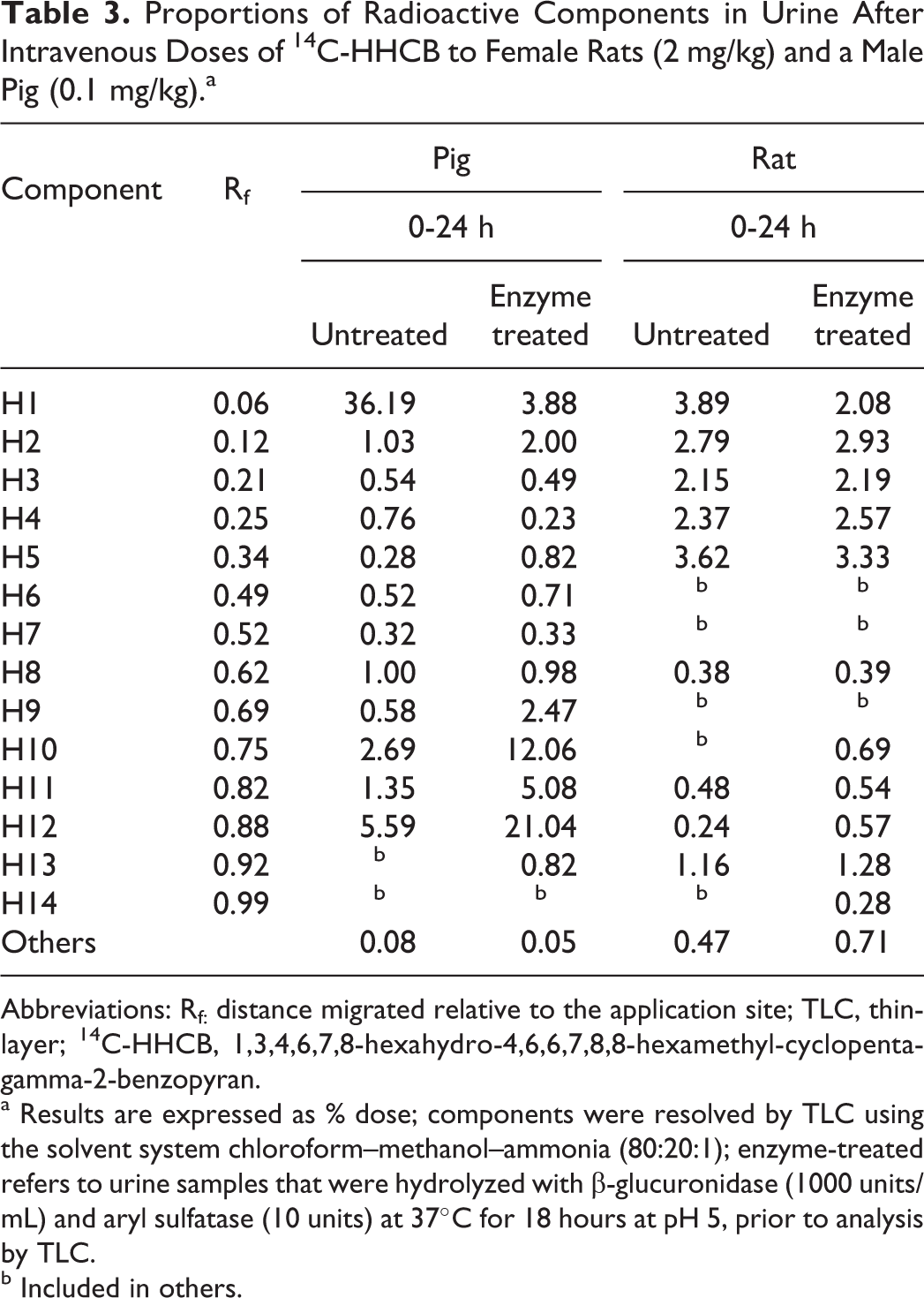
Abbreviations: Rf: distance migrated relative to the application site; TLC, thin-layer; 14C-HHCB, 1,3,4,6,7,8-hexahydro-4,6,6,7,8,8-hexamethyl-cyclopenta-gamma-2-benzopyran.
a Results are expressed as % dose; components were resolved by TLC using the solvent system chloroform–methanol–ammonia (80:20:1); enzyme-treated refers to urine samples that were hydrolyzed with β-glucuronidase (1000 units/mL) and aryl sulfatase (10 units) at 37°C for 18 hours at pH 5, prior to analysis by TLC.
b Included in others.
Discussion
Following iv administration, both the compounds were readily excreted by the rat and pig, although by different routes. Within 72 hours of dosing, 70% to 90% of the administered radioactivity had been excreted. Little, if any, unchanged AHTN or HHCB was excreted in the urine. In rats, radioactivity excreted in feces was 2- to 3-fold greater than that excreted in the urine for both 14C-HHCB and 14C-AHTN. This result is in agreement with the findings of Ford et al, 20 following dermal exposure of rats to radiolabeled AHTN and HHCB, where the materials were primarily excreted in the feces by the rat with much lower amounts in the urine. In the pig, excretion was predominantly via the urine. Fecal excretion was 5- to 7-fold lower than urinary excretion for both 14C-HHCB and 14C-AHTN. In the rat, the predominance of radioactivity in the feces indicates biliary excretion of the test material, or more likely, conjugated metabolites of the test material. The results of these studies are in agreement with those reported by Ford et al, 20 who found that after dermal administration of AHTN or HHCB, the absorbed material was found in the intestinal contents as well as the feces. No bile was collected from rats in the present study for the identification of metabolites, but as excretion of the test material was primarily in feces, the most likely source of radiolabel in the feces is expected to be via bile.
The interspecies difference in the route of elimination of systemically available HHCB and AHTN glucuronide (via the bile in rats and in the urine in pigs) most likely occurs as a result of the lower molecular weight threshold for biliary excretion in rats (325 Da) as compared to most other species. 24 The molecular weight of glucuronide conjugates of hydroxylated HHCB and AHTN would be in the range of 450, which exceeds this molecular weight threshold. Several examples appear in the literature that demonstrate these differences between rats and pigs. Biliary excretion of methylene disalicylic acid (molecular weight 288) in the rat was 54%, but there was no biliary excretion in the pig. 25 Excretion of stilbestrol glucuronide (molecular weight 445) was 95% in rat bile, but there was no excretion in pig bile. 25 Excretion of phenolphthalein glucuronide (molecular weight 495) with rat bile was 54% but was essentially zero in pig bile. 25 The pig appears to behave more similar to humans than rats with respect to secretion of xenobiotics into the bile. Excretion of chemicals in human bile is extremely low when compared with rats. Excretion of morphine glucuronide (molecular weight 461) was reported to be 63% in rat bile but was only 1% to 7% in human bile. 25 Excretion of chloramphenicol glucuronide (molecular weight 499) was 80% in rat bile and only 3% in human bile. 26 Thus, it is expected that if these compounds were absorbed percutaneously by humans, their metabolites would be excreted in the urine.
It is possible that the rat and the pig could form different metabolites that would explain this difference in preferred route of excretion. However, the TLC profiles of urine metabolites (Tables 2 and 3) indicate that while there were quantitative differences in the concentration of metabolites between rat and pig, metabolites present in rat urine and pig urine had similar retention times suggesting that the pig and the rat produce primarily the same metabolites. At least for HHCB, the change in the TLC profile after incubation with β-glucuronidase suggests that the predominant metabolites in pig urine are glucuronides. For AHTN, the loss of the 0 to 6 hours time point urine sample precludes such a conclusion.
Both AHTN and HHCB have large octanol/water partition coefficients (K ow = 6) and are highly lipophilic. This lipophilicity explains the distribution of parent AHTN and HHCB to adipose tissue during the first 2 hours after intravenous administration. These parent compounds maintained a steady state concentration in adipose tissue until about 48 hours, after which the concentrations declined. This decline can be explained by the continued hepatic conversion of parent molecule to less lipophilic metabolites. Because adipose tissue levels of ATHN and HHCB did not decline until after 48 hours, repeated daily dosing may lead to accumulation over time in adipose tissue. Accumulation would be dependent on the dose, dose route, and dosing frequency.
In summary, the results of this study demonstrate that the major route of excretion for AHTN and HHCB is different in rats and in domestic pig. Excretion was primarily via feces in rats and via urine in the pig. The evidence suggests both AHTN and HHCB were completely metabolized in both the species of animals and no unchanged compound was detected in urine. Only a slight distribution of radioactivity occurred in tissues, but the levels did not begin to decrease until 24 hours after administration. Since both pig and humans are “poor biliary excretors” as compared to rats, it is assumed that in humans, the excretion of any systemically available AHTN and HHCB will be similar to the pig, which is primarily via urine.
Footnotes
Declaration of Conflicting Interests
Two of the authors are employees of the Research Institute for Fragrance Materials, an independent research institute supported by the manufacturers of fragrances and consumer products containing fragrances.
Funding
This research was supported by the Research Institute for Fragrance Materials, an independent research institute that is funded by the manufacturers of fragrances and consumer products containing fragrances.
