Abstract
The fate of dermally applied [14C]d-limonene was evaluated in humans and Long-Evans rats. In rats, 5 mg/kg body weight of [14C]d-limonene applied dermally to the shaved back under occlusion, resulted in the absorption of approximately 12% of the dose. The absorbed d-limonene was completely metabolized and excreted rapidly, primarily from the urine (80%) with a small fraction (20%) excreted in the feces. There was no long-term retention of the test material in body tissues. In humans, following dermal application of 12 mg of [14C]d-limonene in ethanol (1 mL) to the back under nonocclusive conditions (for 1 h after application to allow the material to dry, thereafter under occlusion), only 0.16% of the dose was absorbed and the radioactivity was recovered from the urine. Radioactivity in human feces was below the limit of detection. These results indicate that under conditions of simulated use of fragrances and cosmetics, d-limonene has a low potential for dermal absorption and tissue accumulation, and the d-limonene that is absorbed is rapidly excreted in the urine. Based upon these findings and the knowledge that d-limonene possesses a low-systemic toxicity profile, it is reasonable to conclude that dermal exposure to d-limonene from fragrance and cosmetic applications is highly unlikely to result in any clinically significant human toxicity.
Keywords
Introduction
d-Limonene (1-methyl-4(1-methylethnyl)cyclohexene; CAS-RN 5989-27-5) is a naturally occurring cyclic monoterpene found in numerous fruits and oils, especially citrus oils. It is listed in the US Food and Drug Administration (FDA) Generally Recognized as Safe List and is approved for use by the FDA as a food additive and flavor ingredient in the baking and confectionery industries. As a fragrance ingredient, it has a long-established history of use in cosmetic skin lotions and creams, soaps and detergents, and in perfumes and colognes. 1 The worldwide yearly use volume of d-limonene in fragrances was reported to be between 100 and 1000 metric tons. 2 The upper 97.5 percentile human systemic exposure from dermal fragrance use in cosmetic products was calculated to be 0.034 mg/kg body weight (bw)/d and the maximum skin level in fine fragrances (hydroalcoholics) was reported to be 2.6%. Although the primary route of human exposure to d-limonene from use as a fragrance and cosmetic material occurs via the skin, there is a relative paucity of dermal exposure studies, most likely because of its documented lack of toxicity.
The oral and inhalation toxicity of d-limonene has been well characterized in numerous animal models, and it has been shown to have a relatively low-systemic toxicity profile. An acceptable maximum daily intake of 1.5 mg/kg of bw for humans was established by the Joint FAO/WHO Expert Committee. In 1993, based upon the opinion that d-limonene did not represent a health hazard, the acceptable daily intake was withdrawn. 3
Only the male rat has shown exposure-related kidney tumors that have been attributed to the well-characterized non-DNA reactive mechanism of action mediated by the increased level of the male rat-specific protein, α2µ-globulin. 4 The rat metabolite of d-limonene, limonene-1,2-oxide, has been specifically associated with the male rat kidney α2µ-globulin toxicity. 5,6 This finding has also been confirmed by Dietrich and Swenberg 7 who found no evidence of d-limonene-related nephrotoxicity in NCI-black-Reiter rats, the only strain of male rat known not to synthesize α2µ-globulin, concluding that the presence of this protein was causal to the development of d-limonene-induced renal toxicity. Nephrotoxicity was also not observed in other species of animals, including mice, dogs, and guinea pigs which do not synthesize α2µ-globulin. 8
Most of the dermal exposure studies conducted with d-limonene have been conducted in vitro. Meyer and Meyer
9
reported that d-limonene was absorbed through mouse abdominal skin, but that its skin absorption both with and without occlusion was low (6% in 72 hours). In shaved mouse skin studies, the dermal absorption of [
3
H] d/
The present study was conducted to determine the fate of [14C]d-limonene after dermal application in male Long-Evans rats and after dermal application of a 1.2% solution in humans, a level which simulates human fragrance and cosmetic use levels.
Materials and Methods
Rat Study
At the time the rat study was conducted, all applicable laws regarding animal care and use were followed. (+) [propene-1-14C]-Limonene CAS# 5989-27-5, 96.8% pure, with a specific activity of 50.7 µCi/mg and hexadecane standard was supplied by Amersham International (Amersham, UK). (+)-Limonene was purchased from Aldrich Chemical Co, Ltd (Gillingham, UK); Special Scintillator MI 31 from Packard Instrument Company Limited (Reading, UK); Potisorb I and Optisorb S scintillator System from Fision plc (Loughborough, UK); sample excisers (Amersham International plc); Colworth Stomacher 400 homogenizer (A.J. Seward, London, UK); Automatic Sample Oxidizer (Model 306 Mk3 Tricard) Packard Instrument Company Limited; Philips Automatic Liquid Scintillation Analyser (Phillips NV, Eindhoven, Holland); VG 7070E mass spectrometer (VG Analytical Ltd, Manchester, UK); and prelayered silica gel plates were obtained from Merck (Darmstadt, Germany).
Twelve adult male Long-Evans rats with a nominal body weight of 200 g (6 weeks old) were obtained from Charles River (Margate, Kent, UK). A single dose of 14C-limonene in ethanol (about 1 mg/mL) was applied at a nominal dose of 5 mg/kg (10-20 μCi/mg) over a 9 cm2 shaved area of the back. The treated area was covered with aluminum foil and held in place with Sleek® waterproof adhesive dressing for 3 hours (in those animals sacrificed at this time) or for 6 hours in the other animals. Upon removal, the treated area was washed with cotton wool swabs moistened with ethanol, and the dressing, foil, and swabs were retained for radioactivity measurement. Urine was collected at intervals of 0 to 3/6, 6 to 24, 24 to 48, and 48 to 72 hours. Feces were collected at 24, 48, and 72 hours. Air from the cage environment was collected in traps containing ethanolamine: 2-ethoxyethanol (1:3, v/v) from the rats sacrificed at 24, 48, and 72 hours. Groups of rats were sacrificed by exsanguination under halothane anesthesia at 3, 6, 12, 24, 48, and 72 hours after application of the test material. Samples of whole blood, plasma, liver, kidney, muscle, fat, testes, heart, eyes, brain, lungs, adrenals, bone marrow, lymph nodes, pancreas, spleen, thymus, thyroid, gastrointestinal tract with contents, treated skin, and untreated skin were collected. All the samples were stored together with the carcasses at −15°C.
Feces were mixed with a water–methanol mixture (1:1) and homogenized. Urine and plasma samples and expired air trap contents, cage washings, and other liquid samples were mixed with Special Scintillator MI 31. Samples of homogenized tissues and whole blood were mixed with dry cellulose powder and burned in oxygen. The combustion products were absorbed into Potisorb I and mixed with Optisorb S scintillator System. Liquid scintillation analysis of reference standards generally exceeded 97%, and therefore, the results were not corrected for efficiency. The mode of radioactivity counting was preset at 4 minutes or 106 counts for samples containing higher levels of radioactivity and at 10 minutes or 104 counts for samples containing low levels of radioactivity. Radioactivity in amounts less than twice background levels was considered to be below the limit of accurate detection. The maximum statistical counting error at this limit was approximately 10% of the net count rate. Mass spectroscopy was used to verify the identity of the radioactive standard. Thin layer chromatography (TLC) was carried out on 0.25-mm prelayered silica gel plates.
Human Study
The human study was subjected to review and approval by the Medical Ethics Committee of Huntingdon Health Authority and the Department of Health and Social Security for the United Kingdom.
(+) [propene-1-14C]-Limonene, 96.8% pure, with a specific activity of 50.7 µCi/mg and hexadecane standard were supplied by Amersham International. (+)-Limonene was purchased from Aldrich Chemical Co, Ltd; Special Scintillator MI 31 and Automatic Sample Oxidizer (Model 306 Mk2 Tricarb) from Packard Instrument Company Limited; Potisorb I and Optisorb S scintillator System from Fision plc; sample excisers (Amersham International pl); Philips Automatic Liquid Scintillation Analyser (Phillips NV); VG 7070E mass spectrometer (VG Analytical Ltd); Colworth Stomacher 400 (A.J. Seward); β-glucuronidase (Helix pomatia, type H1; Sigma Chemical Co Ltd, St. Louis, MO, USA); Berthold TLC Linear Analyzer (Models LB2832 or LB2842; Laboratory Impex Ltd, Twickenham, UK); and 0.25-mm prelayered silica gel plates (Merck).
Two healthy male participants aged 26 and 29 years and weighing 86.1 and 70 kg were kept under medical supervision for 12 hours before dosing and during the experimental period. Following a 12-hour fast, 12 mg of 14C-limonene in ethanol (50 μCi/mL) was applied to a 100-cm2 area of intact unshaven skin on the back. The material was allowed to dry for 1 hour, and the treated areas were occluded with light gauze dressing held in place with Sleek waterproof adhesive tape. Six hours after application, the dressing was removed, washed with cotton wool swabs moistened in ethanol, and the dressing and swabs were retained for radioactivity measurement. A 2 × 5 cm2 treated area was stripped with 5 successive applications of adhesive tape, and each tape strip was retained separately for radioactivity measurement. Blood samples were collected prior to test material application and at 0.5, 1, 2, 3, 4, 6, 8, 10, 12, 16, 24, 36, 48, 72, 96, and 120 hours after application. Urine was collected at intervals of 0 to 3, 3 to 6, 6 to 12, 12 to 24, 24 to 48, 48 to 72, 72 to 96, and 96 to 120 hours after application of the test material. Feces were collected at 24-hour increments between 0 and 120 hours after dose application. Urine and feces were stored at −20°C until analyzed. Feces were mixed with a water–methanol mixture (1:1) and homogenized in a Colworth Stomacher 400 or extracted with methanol. Samples of urine, plasma, contents of expired air traps, cage washings, extracts of feces, and other liquid samples were mixed with Special Scintillator MI 31. Samples of homogenized tissues and whole blood were mixed with dry cellulose powder and burned in oxygen using an Automatic Sample Oxidizer. The combustion products were absorbed into Optisorb I and mixed with Optisorb S scintillator System. Recoveries of radioactivity from carbon-14 standards for sample oxidizers burned in the oxidizer generally exceeded 97%, and therefore, the results were not corrected for efficiency. Radioactivity was measured in a liquid scintillation counter. Upon evaporation, the radioactivity was extracted from the residue with methanol by trituration and centrifugation, and the supernatant was separated. The methanol extraction was repeated 3 times, and the supernatant extracts were combined and concentrated under nitrogen at 37°C. The extracted residual solid was suspended in water and radioactivity was measured in both methanol extracts and aqueous residues. There was no appreciable loss of radioactivity by these procedures, and most of the radioactivity was obtained in the methanol concentrates.
The TLC was carried out on 0.25-mm prelayered silica gel plates with dichloromethane–methanol–acetic acid (90:10:1, v/v) and n-butanol–water–acetic acid (4:1:1, v/v). Radioactive components on the plates were detected with a Berthold TLC Linear Analyzer. The reference materials, perillyl alcohol, perillic acid, and p-menth-1-ene-8,9-diol, were obtained from Aldrich Chemical Co Ltd. The compounds were located by exposure to ultraviolet light or by spraying with aqueous potassium permanganate solution. Urine extracts were incubated for 18 hours at 37°C in 0.1 mol/L acetate buffer at pH 5 with β-glucuronidase. Enzyme-treated urine was evaporated to near dryness under a stream of nitrogen at 37°C and the residue was reconstituted in methanol for TLC.
Results
Rat Study
Following a 6-hour occluded dermal application of [14C]d-limonene, the total mean recovery of applied radioactivity was 78.4% in 72 hours (Table 1). The largest percentage of radioactivity was recovered from skin area washing (47.9%) and air traps (17.5%). Urinary 72-hour excretion accounted for 11% of radioactivity with most (9%) of the excretion occurring within 24 hours (4% within 0-6 hours and 5% within 6-24 hours). Fecal excretion accounted for 2.6% of the dose with half of this radioactivity excreted in the first 24 hours (Figure 1). Recovery of the applied radioactivity from treated skin tissue was 0.2%. A total of 1% of the applied radioactive dose was recovered from untreated skin, tissues, and rat carcasses. The unaccounted (21%) for radioactivity was most likely volatilized and/or evaporated during application. Based upon these recoveries, approximately 12% of the applied dose was absorbed through the skin.
Mean Recovery of Radioactivity After Single-Rat Dermal Application of 13C-Limonene.
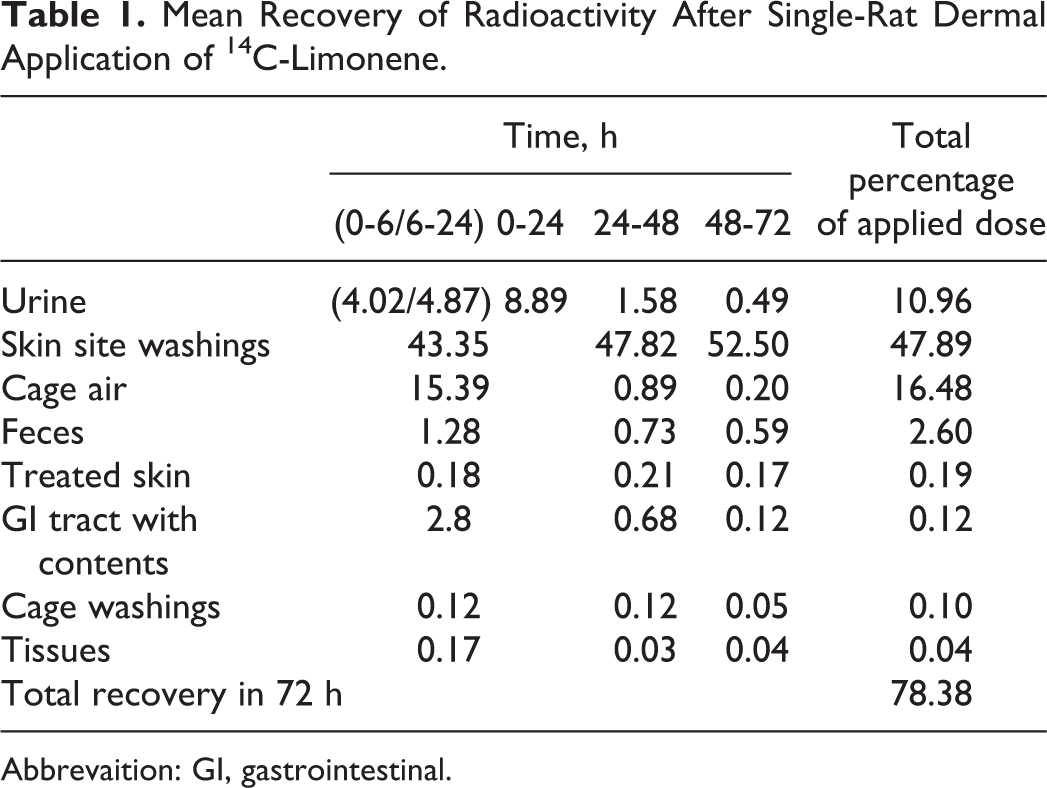
Abbrevaition: GI, gastrointestinal.
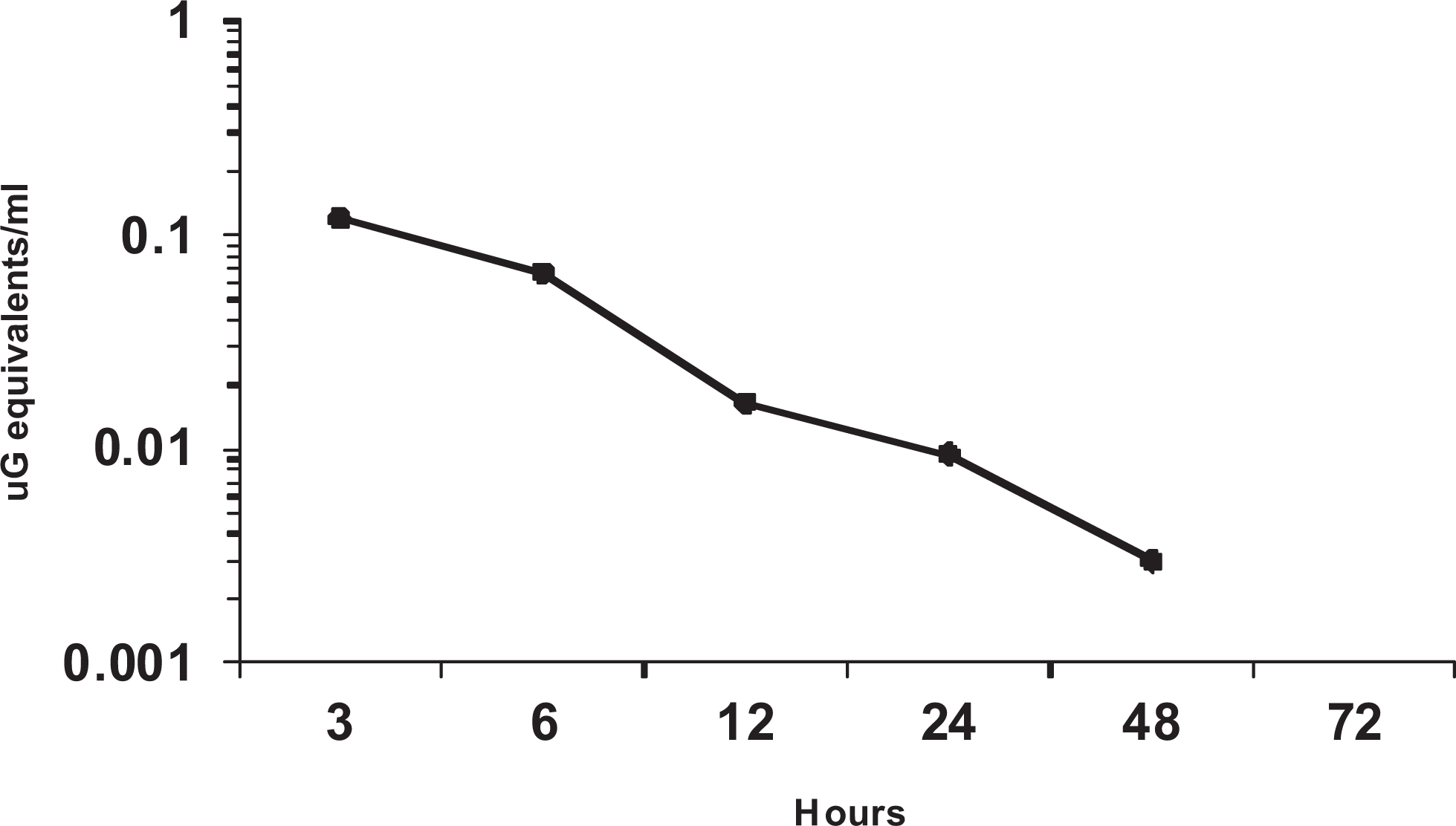
Mean plasma radioactivity in rats after occluded application of [13C]d-limonene.
Maximum radioactivity (0.12 μg equivalent/mL; <0.2% dose) in the plasma was seen in the 3-hour collection sample (Figure 1), and a rapid decline was observed thereafter with the radioactivity declining sharply to 15% of the peak value by 12 hours and 2.5% of the peak value by 48 hours. No radioactivity in plasma was detected at the 72-hour collection time.
In all tissues except the gastrointestinal (GI) tract/contents, the maximum radioactivity was observed in the first sample collected at 3 hours after dose application (Figure 2). At this time, the highest radioactivity was found in the GI tract (2.35 µg/g), kidneys (1.1 µg equivalent/g), liver (0.86 µg equivalent/g), and adipose tissue (0.5 µg equivalent/g). The radioactivity declined sharply in all the tissues after 12 hours. At 72 hours, radioactivity could only be detected in kidney, liver, adipose tissue, and GI tract and its contents. The slowest rate of decline was seen in the GI tract.
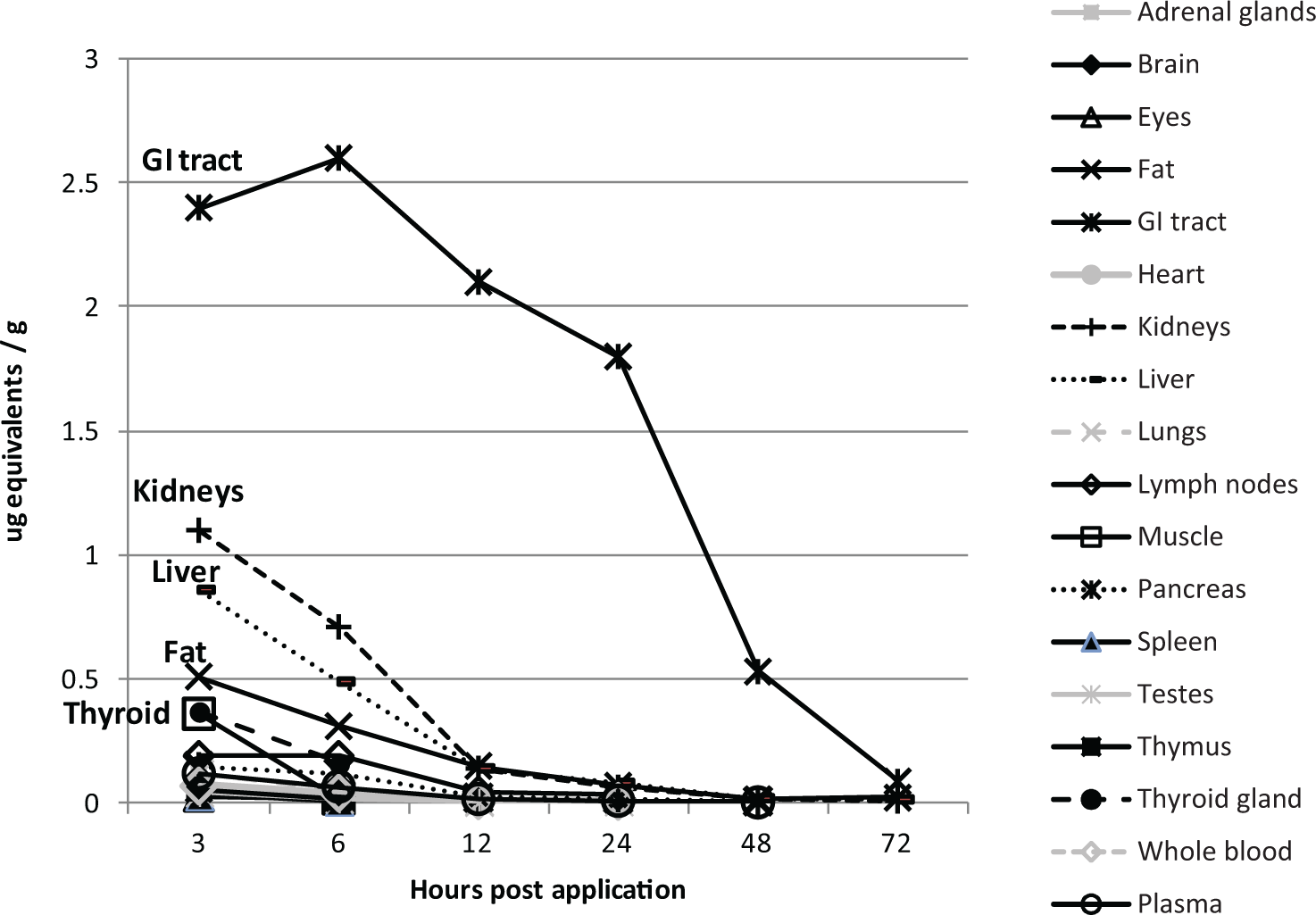
Rat tissue radioactivity after dermal 14C-limonene application. (Data calculated assuming muscle, whole blood, fat, plasma, and bone marrow represent 45%, 7%, 7%, 4%, and 0.35% body weight, respectively.)
The TLC of 0 to 6-hour collected rat urine using 2 different solvent systems showed the presence of a polar component that was not identified. Treatment of urine with β-glucuronidase hydrolyzed this component into 3 additional components. The major component cochromatographed with p-menth-1-ene-8,9-diol (42% of sample radioactivity) and the 2 minor components were associated with the metabolites perillyl alcohol (5% of sample radioactivity) and perillic acid (8% of sample radioactivity).
Human Study
The data from the 2 participantss showed almost identical results (Table 2). Six hours after dermal application of an ethanolic solution of 14C-limonene, an average of 0.36% of the dose was recovered from gauze dressing and 0.06% from swabs. Skin strippings yielded only 0.01% to 0.03% of the applied dose. The mean recovery from urine during the 120-hour study period was 0.16% of the dose. Radioactivity in feces was below the limit of quantification. The total mean recovery of radioactivity after the 120-hour study was only 0.6%. Based on the low total mean recovery, it is concluded that the applied [14C]d-limonene either evaporated during the drying time before occlusion or from the dressing after removal.
Recovery of Radioactivity After Single-Human Dermal Application of 14C-Limonene. a
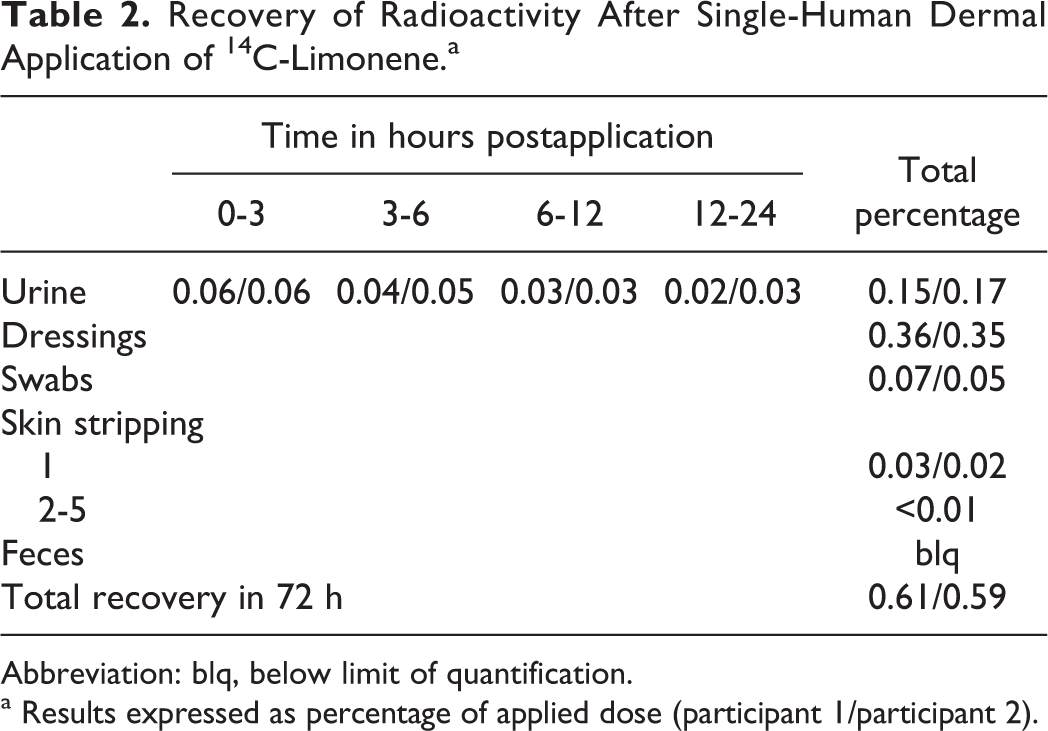
Abbreviation: blq, below limit of quantification.
a Results expressed as percentage of applied dose (participant 1/participant 2).
Excretion of radioactivity in human urine occurred only in the first 24 hours after application and accounted for 0.16% of the applied dose. No radioactivity was detected in human feces. Whole blood and plasma samples collected throughout the study contained no radioactivity above the limit of quantification which was 0.004 μg equivalent/mL in plasma and 0.01 μg equivalent/mL in whole blood.
Samples of the 0 to 6-hour urine contained the highest concentrations of radioactivity. The major peak remained at the origin of these TLC plates indicating a polar and probably conjugated component. The 0 to 6-hour urinary collection contained about 4% of the dose. No radioactivity was detected elsewhere on the plate, and treatment with β-glucuronidase did not yield any hydrolyzed product. Whether these results are indicative of a distinct lack of metabolite production is not known.
Discussion
This study demonstrates an extremely low human skin permeability of d-limonene, less than 1%, under open, or nonocclusive, conditions (for first hour after application) and a much greater dermal permeability (approximately 12%) in rats under occlusive conditions. The appreciable differences in absorbed dose between the 2 species are most likely a reflection of (1) the different conditions of application (open vs occlusive), (2) the differences in skin species permeability, and (3) the differences in applied concentrations.
The condition of dermal application, that is, occlusive versus nonocclusive, plays a distinct role in skin absorptive properties. As soon as the skin structure changes, as happens under occluded conditions, the dermal absorption increases. Numerous authors have documented dramatic increase in permeability of dermal absorption with increasing hydration states. 13 –15 The documented absorption differences are also consistent with the knowledge that volatile chemicals evaporate quickly, and given that human skin is normally a warm area, the evaporation rate of volatile chemicals when applied to a warm area is further increased. 16 Limonene has a high vapor pressure (1.45 mm Hg at 25°C), indicating it is volatile.
The observed different patterns of dermal absorption of d-limonene for human and rat skin are consistent with other documented studies. Hotchkiss 11 showed that rat skin is significantly more permeable to fragrance material chemicals than human skin. Schafer and Schafer 10 reported rapid absorption of dermally administered limonene in mice.
Finally, different concentrations used for humans versus rats may also have had an impact on the rate and quantity of dermal absorption. At least in vivo, the transdermal permeation rates of certain drugs have been shown to be significantly altered by enhancers such as d-limonene, with concentrations of 2% to 5% d-limonene having no effect, and 20% d-limonene increasing permeation. 17 However, other in vitro studies reported no appreciable differences in transdermal penetration of certain drugs when d-limonene was used between 1% and 10% concentrations. 18
The reaffirmation of a lack of significant human skin penetration of d-limonene is also interesting in light of the use of d-limonene as a penetration enhancer of various chemicals. 19,20 However, the percutaneous enhancing properties of limonene depend on its interaction with compounds such as propylene glycol and in most cases, penetration enhancement has been documented at levels (usually greater than 5%) that are much higher than those used in fragrance and cosmetic uses (maximum level 3.5%). 21 –23
Even with the observed differences in dermal absorption, both species showed rapid absorption and elimination primarily, via the urine, without any significant long-term systemic retention. In the rat, peak blood and tissue levels were noted in the first sample collected at 3 hours with maximum elimination of the radioactivity being observed in the first 24-hour sample. These results are similar to studies reported by Igimi et al, 24 where oral administration of [14C]d-limonene in rats produced peak blood levels at 2 hours and peak tissue levels within 2 hours after administration. No significant radioactivity was reported in blood and tissues after 48 hours, and almost 60% of the orally administered [14C]d-limonene was excreted in urine and 5% was excreted in feces. The urine was also found to be the major excretory pathway by Kodama et al 25 who reported that after oral administration of [14C]d-limonene to male rabbits, 72% of the dose was excreted in urine and 7% in feces during the first 72 hours after application. Oral exposure studies in various animal species have also shown urinary excretion to be the primary route of elimination: 82% in rats, 82% in guinea pigs, 96% in hamsters, 87% in rabbits, and 77% in dogs during 48 to 72 hours after [14C]d-limonene administration. 26 In other oral human excretion studies, the radioactivity in urine accounted for 83% in 1 participant and 55% in the other participant 48 hours after dosing. 26
No significant metabolites were identified in the human study, but the major metabolite in rat urine was a polar component, which after treatment with β-glucuronidase hydrolyzed into p-menth-1-ene-8,9-diol and 2 minor components including perillyl alcohol and perillic acid. Crowell et al
26
identified perillic acid and dihydroperillic acid as the major circulating metabolites in rats. Numerous other metabolites have been identified in rat urine after oral administration of [14C]d-limonene. They have included perillic acid, p-menth-1-ene-8,9-diol, perillic acid-8,9-diol, and 8-hydroxy-p-menth-1-en-9-yl-β-
Although urine is the major route of excretion of d-limonene, the rat study showed that 2.6% of the dermally applied radioactivity was excreted in feces. This finding is similar to the results of Igimi et al 23 who recovered 25% of the 14C-limonene radioactivity from rat bile and 5% from feces. Fecal excretion ranged from 2% in guinea pigs to 9% in rats and dogs. 26 These results indicate some portion of radioactivity finding its way into the enterohepatic circulation.
Conclusions
In summary, the results of this study reaffirm the distinct differences in human and rat absorption of dermally applied d-limonene related both to skin specificity and application differences and the distinct similarities in rapid elimination through the urine, without any significant systemic retention. Based upon this knowledge and the well-documented knowledge of the lack of toxicity associated with d-limonene in humans and animals via oral and inhalation exposure, it can be concluded that human dermal exposure to d-limonene in fragrance/cosmetic uses is highly unlikely to result in any systemic toxicity due to its insignificant human dermal absorption, lack of systemic retention, and rapid elimination.
Footnotes
Declaration of Conflicting Interests
Anne Marie Api and Gretchen Ritacco are employees of the Research Institute for Fragrance Materials, an independent research institute supported by the manufacturers of fragrances and consumer products containing fragrances.
Funding
This research was supported by the Research Institute for Fragrance Materials, an independent research institute supported by the manufacturers of fragrances and consumer products containing fragrances.
