Abstract
Three levels of nasal cavity sections (anterior, middle, and most posterior) are routinely examined as protocol required tissues in our 26-week carcinogenicity studies involving Tg.rasH2 mice. Exudative inflammation of the nasal cavity was noted in the most posterior section of both males and females that were administered vehicle and/or test article via oral gavage, particularly when the vehicle and/or test article had irritant properties, was in the form of a salt, had a low pH, and/or was viscous. The exudative inflammatory lesion was characterized by the presence of eosinophilic proteinaceous fluid, fibrin, mucin, sloughed cells, and degenerate neutrophils within the nasal cavities. In lesions of increased severity, there was often degeneration, necrosis, and erosion of the underlying mucosa. Often, there was hyperplasia as well as squamous metaplasia of the mucosa. Retrospective analysis of our data, involving thirty-two 26-week Tg.rasH2 carcinogenicity studies, revealed that despite the presence of these exudative inflammatory changes with degeneration, necrosis, and mucosal hyperplasia, progression to tumor formation in the nasal cavities was rare and the incidence of nasal tumors was comparable in animals with or without exudative inflammatory lesions.
Keywords
Introduction
The 2-year rodent carcinogenicity assays involving conventional rats and mice have been conducted for over 3 decades. As an alternative to the 2-year mouse carcinogenicity bioassays, the 26-week short-term carcinogenicity bioassays were approved using genetically modified mouse strains including Tg.rasH2. 1 The Tg.rasH2 model, which can be used for both genotoxic and nongenotoxic compounds, has gained popularity and its use has increased over the years. Currently, more than 75% of all mouse carcinogenicity studies are being conducted in the Tg.rasH2 mice. 2 The Tg.rasH2 model predicts neoplastic findings relevant to human cancer risk assessment and produces fewer nonbiologically significant neoplastic outcomes and is thus preferable to a 2-year mouse study. 3
There are no published reports on exudative inflammatory lesions and their association with mucosal hyperplasia and/or tumors in the nasal cavities of Tg.rasH2 mice in noninhalation gavage studies. However, there have been many papers published, mainly in rats, that have shown exudative inflammatory lesions commonly in the most posterior sections and concurrent erosion, necrosis, hyperplasia, squamous metaplasia in (noninhalation) oral gavage studies, most likely caused by gastric reflux. 4 -9 The occurrence of neoplasia in these gavage rat studies is very rare. 10 These authors 4 -10 identified several causes for the inflammatory lesions in nasal cavities with (noninhalation) gavage studies, which included irritant properties, low pH, and/or viscosity of the vehicle and/or the test article. These authors also discussed the complexity of differentiating these nasal cavity lesions induced by gastric reflux in gavage studies, which may mask or exaggerate compound-related toxicity. As described in this article, exudative inflammation in the nasal cavities of Tg.rasH2 mice was commonly present in gavage studies, when the vehicle and/or test article were in the form of a salt, had a low pH, had irritant properties, and/or was viscous. Accompanying the exudative inflammatory changes were degeneration, necrosis, erosion of underlying mucosa as well as hyperplasia and squamous metaplasia of the respiratory mucosa. These lesions were more commonly seen in the most posterior section of the nasal cavity. Despite these changes, the incidence of carcinomas and hemangiosarcomas in the nasal cavities from animals that had exudative inflammation was low and comparable to animals that did not have exudative inflammatory changes. Thus, these exudative inflammatory lesions do not predispose Tg.rasH2 mice to the development of nasal cavity tumors.
Materials and Methods
Animals
CByB6F1-Tg(HRAS)2Jic (+/− hemizygous c-Ha-ras) mice, obtained from Taconic Biosciences (Germantown, New York), were used in all studies. The knock-in Tg element (human prototype c-Ha-ras gene with its own promoter/enhancer) is injected into C57BL/6 × BALB/c F2 zygotes, which were crossed back to C57BL/6J forming C57BL/6JJic-Tg(HRAS)2Jic. The CByB6F1-Tg(HRAS)2Jic (+/− hemizygous c-Ha-ras) is the offspring from a cross of the C57BL/6JJic-Tg(HRAS)2Jic hemizygous male mice with the BALB/cByJJic female mice. Each mouse was genotyped by Taconic Biosciences to verify the presence of the transgene before being placed on the study. Animals were randomized by body weight into groups using a computer program (Provantis [Instem] Version 8.2.0.8). On the first day of treatment, animals were 7 to 9 weeks of age and weighed at least 17 or 15 g (males and females, respectively). Individual body weights for each dose group of each sex were within ± 20% of the mean at the start of the study.
Housing and Environmental Conditions
Housing and environmental conditions were similar in all studies. Animals were multiple housed during acclimation and single housed in polycarbonate cages following randomization, with hardwood bedding chips in environmentally controlled rooms. Animals were verified to be free of illness prior to being placed on a study. All animals had ad libitum access to water and powdered feed (Harlan TEKLAD Global Diet, Madison, Wisconsin). The environmental conditions at the animal facility, including the type of feed, bedding, cleaning detergents, the lighting cycle, humidity, or temperature ranges, were the same for all studies conducted at BioReliance.
Regulatory Requirements
The numbers of animals, procedures, and experimental design for each study were reviewed and approved by the BioReliance Institutional Animal Care and Use Committee. All procedures followed the specifications recommended in The Guide for the Care and Use of Laboratory Animals and were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care–accredited facility. All procedures involving, but not limited to, quarantine and acclimation, randomization, application of unique identification system, housing, provision of food and water, administration of test article, recording of clinical signs, necropsy, and tissue processing were followed in strict accordance with the good laboratory practice regulations, standard operating procedures, and protocol for each study.
Retrospective Analysis
The database was derived from Tg.rasH2 mice assigned to 32 studies conducted at our facility, following the same study design. Animals in all of these studies were dosed by gavage. The first of these studies was completed in 2004 and the last study was completed in 2015. Each study included 25 mice/sex/dose groups. There were 4 dose groups in each study, designated as vehicle, low dose, mid dose, and high dose. In addition, there was a positive control group in each study which was administered urethane intraperitoneally. Of these 32 studies, there were 16 studies in which the vehicle and/or test article was in the form of a salt, had irritant properties, had low pH, and/or was viscous. In all of these studies, there was presence of exudative inflammation of the nasal cavities. In the remaining 16 studies, the vehicle and/or test article lacked these characteristics and did not show exudative inflammatory lesions. Upon death or moribund sacrifice of an animal and at the termination of the study, all animals were humanely sacrificed by carbon dioxide overdose (when applicable) and subjected to complete necropsy. Per our standard operating procedures, 3 sections of the nasal cavity (anterior, middle, and most posterior) were trimmed and examined for each animal. Protocol required tissues were saved in 10% neutral-buffered formalin, processed routinely, stained with hematoxylin and eosin (H and E), and evaluated microscopically. Each study was peer reviewed and tumor data statistics was performed on each study based on a modified version of the Food and Drug Administration (FDA) draft Guidance for Industry. 11
Statistical Methods
The Cochran-Armitage trend test, 12,13 a 2-sided test, was used to detect any dose-related increasing or decreasing trends among the proportions of incidence of exudative inflammation, mucosal hyperplasia, and nasal cavity carcinomas and hemangiosarcomas, as presented in Tables 1 and 2 (16 studies with nasal cavity exudative inflammatory lesions) and Tables 3 and 4 (16 studies without nasal cavity exudative inflammatory lesions).
Incidence of Mucosal Hyperplasia, Carcinomas, and Hemangiosarcomas in Males in 16 Studies Where Nasal Cavity Exudative Inflammatory Lesions Were Noted.
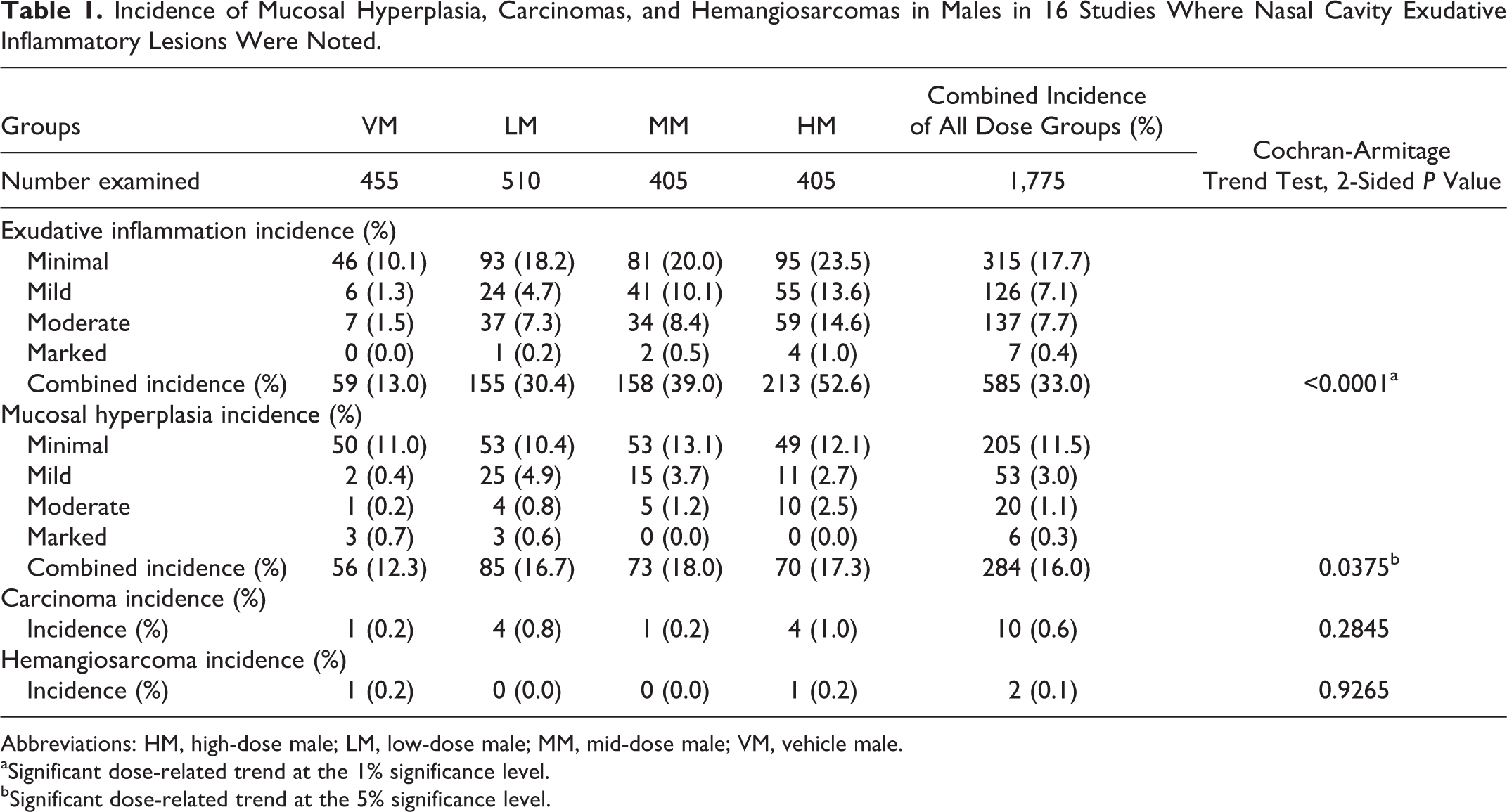
Abbreviations: HM, high-dose male; LM, low-dose male; MM, mid-dose male; VM, vehicle male.
aSignificant dose-related trend at the 1% significance level.
bSignificant dose-related trend at the 5% significance level.
Incidence of Mucosal Hyperplasia, Carcinomas, and Hemangiosarcomas in Females in 16 Studies Where Nasal Cavity Exudative Inflammatory Lesions Were Noted.
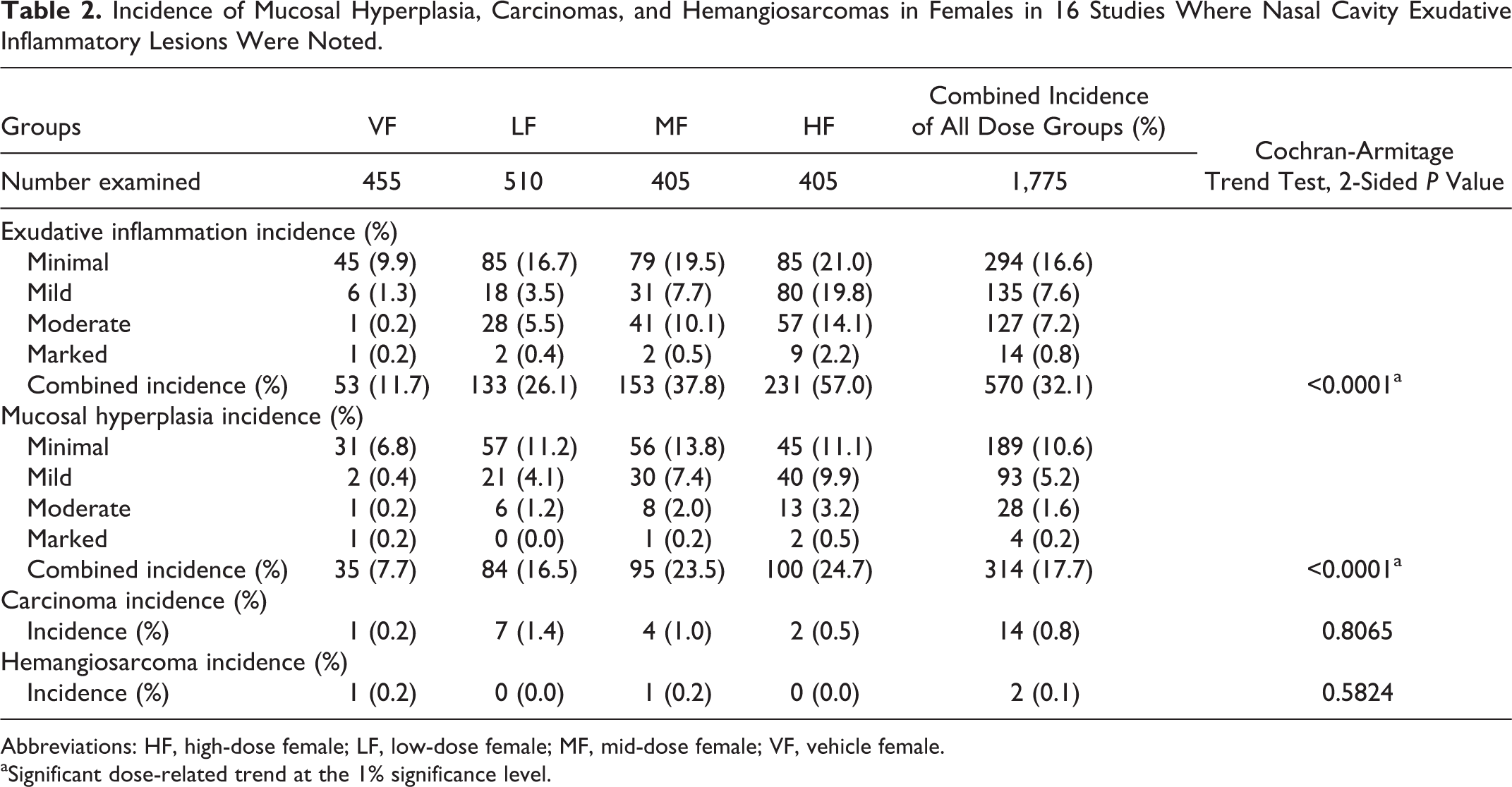
Abbreviations: HF, high-dose female; LF, low-dose female; MF, mid-dose female; VF, vehicle female.
aSignificant dose-related trend at the 1% significance level.
Incidence of Mucosal Hyperplasia, Carcinomas, and Hemangiosarcomas in Males in 16 Studies Where Nasal Cavity Exudative Inflammatory Lesions Were Not Noted.
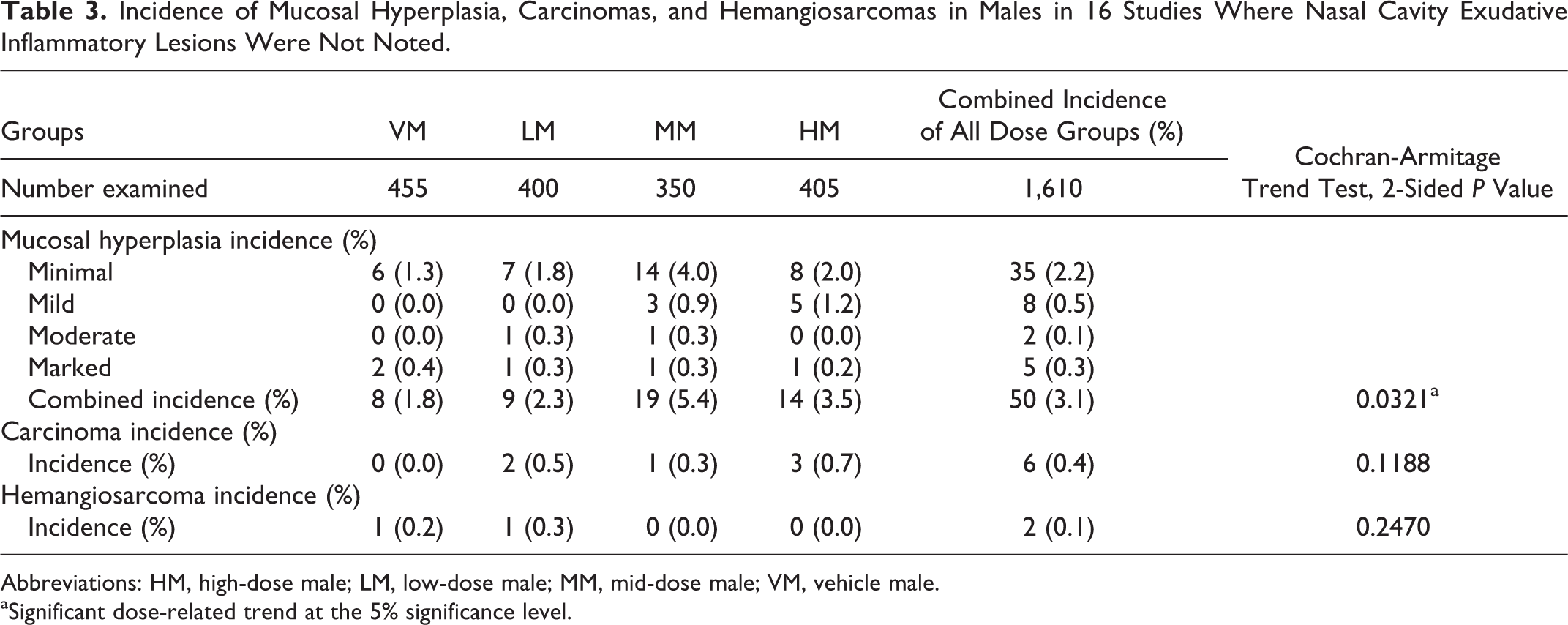
Abbreviations: HM, high-dose male; LM, low-dose male; MM, mid-dose male; VM, vehicle male.
aSignificant dose-related trend at the 5% significance level.
Incidence of Mucosal Hyperplasia, Carcinomas, and Hemangiosarcomas in Females in 16 Studies Where Nasal Cavity Exudative Inflammatory Lesions Were Not Noted.

Abbreviations: HF, high-dose female; LF, low-dose female; MF, mid-dose female; VF, vehicle female.
aSignificant dose-related trend at the 1% significance level.
Results
Tables 1 and 2 show the number of animals in 16 studies that had exudative inflammatory lesions, mucosal hyperplasia, and carcinomas and hemangiosarcomas in vehicle males (VMs), low-dose males (LMs), mid-dose males (MMs), and high-dose males (HMs) and vehicle females (VFs), low-dose females (LFs), mid-dose females (MFs), and high-dose females (HFs), respectively. Presented in these tables are the incidence and percentage of incidence for exudative inflammation and mucosal hyperplasia separated by degree of severity ranging from minimal to marked. These tables also show the incidence and percentages of carcinomas and hemangiosarcomas in the nasal cavities in each sex/dose group.
Tables 3 and 4 show the number of animals in the remaining 16 studies that did not have exudative inflammatory lesions but had mucosal hyperplasia, carcinomas, and hemangiosarcomas in VMs, LMs, MMs, and HMs and VFs, LFs, MFs, and HFs, respectively. Presented in these tables are the incidence and percentage of incidence for mucosal hyperplasia separated by degree of severity ranging from minimal to marked. These tables also show the incidence and percentages of carcinomas and hemangiosarcomas in the nasal cavity in each sex/dose group.
The differences in the number of animals belonging to low and mid doses of males and females, with or without exudative inflammation, are due to the variations in the number of animals assigned to each study per the protocol.
Figure 1 shows a normal nasal cavity from the middle section of a control mouse, whereas Figures 2, 3 (middle section), and 4 (posterior section) show typical exudative inflammatory lesions in the nasal cavities with mild to marked severity, respectively, in test article–treated mice. Figure 5 shows moderate mucosal hyperplasia in the middle section of a test article–treated mouse. Figure 6 shows a typical nasal cavity carcinoma in the most posterior section of a control mouse. Generally, in these carcinomas there was an adenomatous as well as squamous component with typical features of malignancy, such as pleomorphism, anisokaryosis, high degree of mitosis, and usually invasion of nasal turbinate bones. The exudative inflammatory lesions were generally characterized by the presence of eosinophilic proteinaceous fluid within the nasal turbinates that contained fibrin, mucin, sloughed respiratory mucosal, and olfactory cells along with inflammatory cells. Usually, degenerate neutrophils predominated in the inflammatory exudate. With increased severity of inflammation, there were often degenerative and necrotic changes involving the mucosa along with erosion. These exudative inflammatory lesions were most often present in the ventral areas of the most posterior section (third section) of the nasal cavity. The inflammation extended to the anterior and middle portions of the nasal cavity sections, when the the degree of severity of inflammation was moderate to marked.
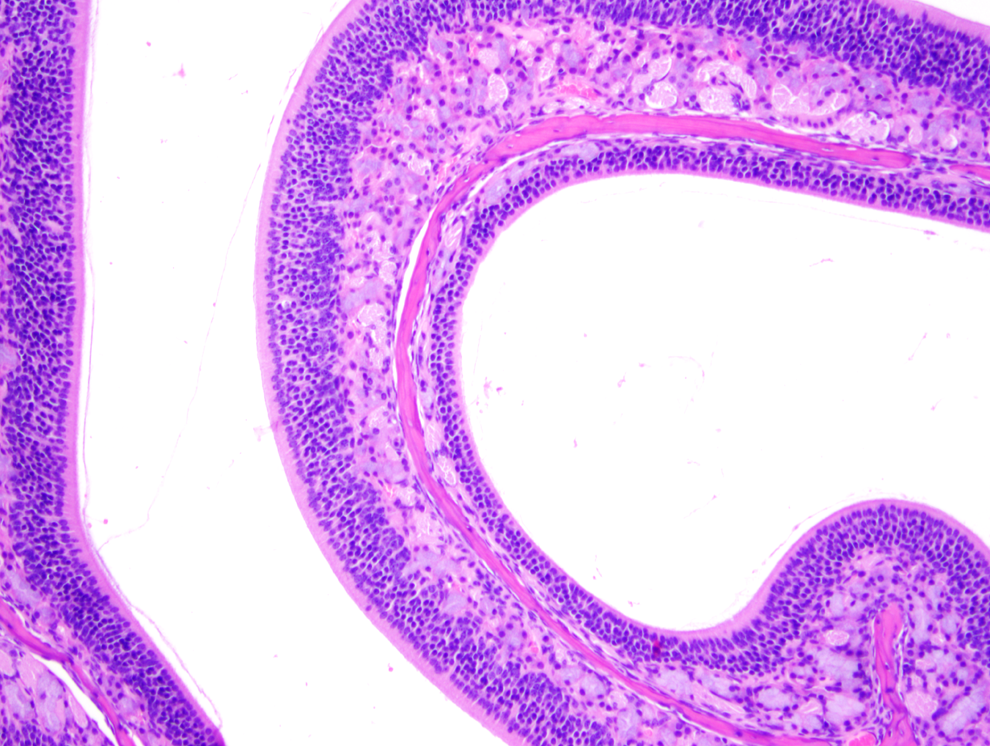
Normal nasal cavity (middle section) in a control mouse (H&E, ×100).
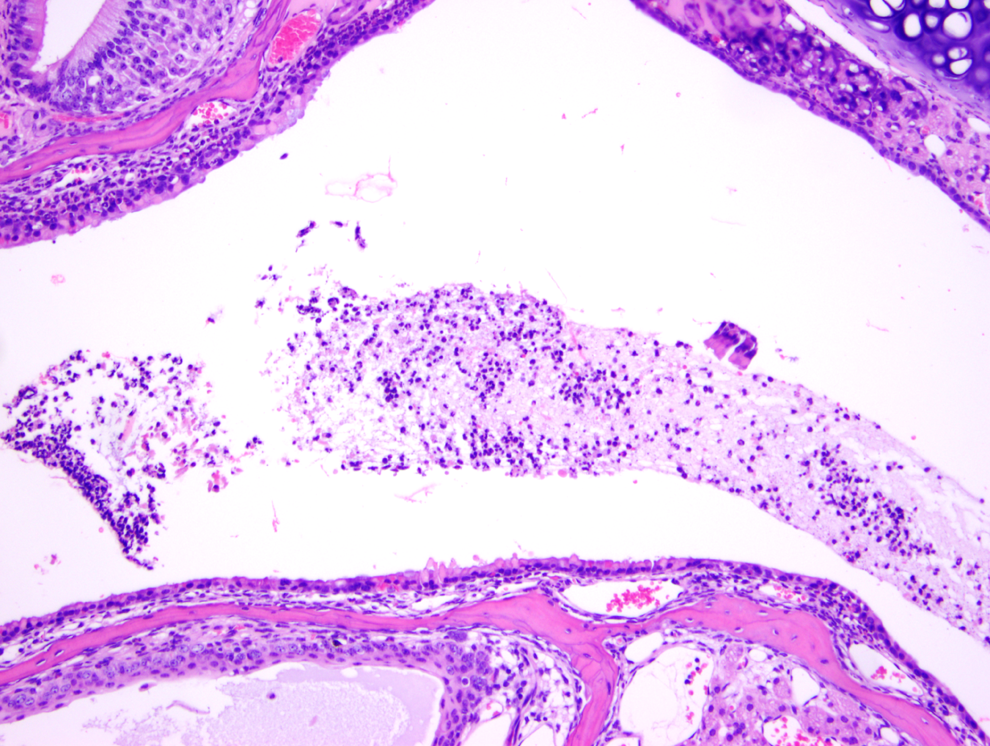
Exudative inflammation of mild degree, nasal cavity, in a test article–treated mouse (H&E, ×100). Mild infiltration of middle section with degenerate neutrophils.
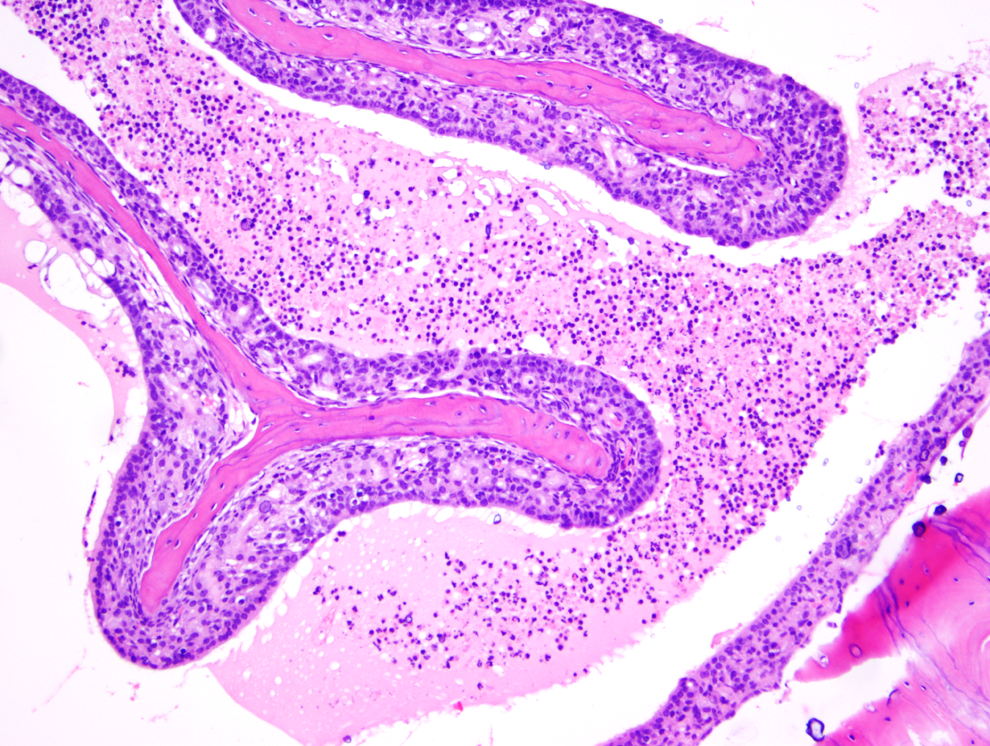
Exudative inflammation of moderate degree, nasal cavity, in a test article–treated mouse (H&E, ×100). Considerable number of neutrophils, eosinophilic fluid, attenuation, and degeneration of mucosa in the middle section.
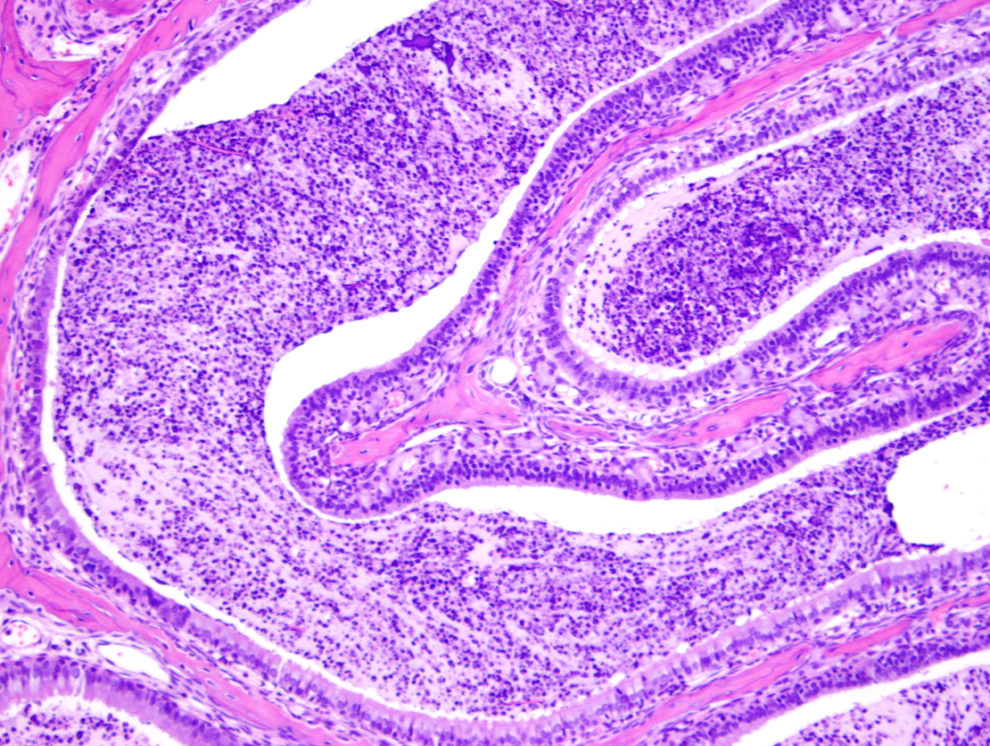
Exudative inflammation of marked degree, nasal cavity, in a test article–treated mouse (H&E, ×100). The posterior section shows extensive accumulation and degenerate neutrophils, fibrin, and sloughed cells.
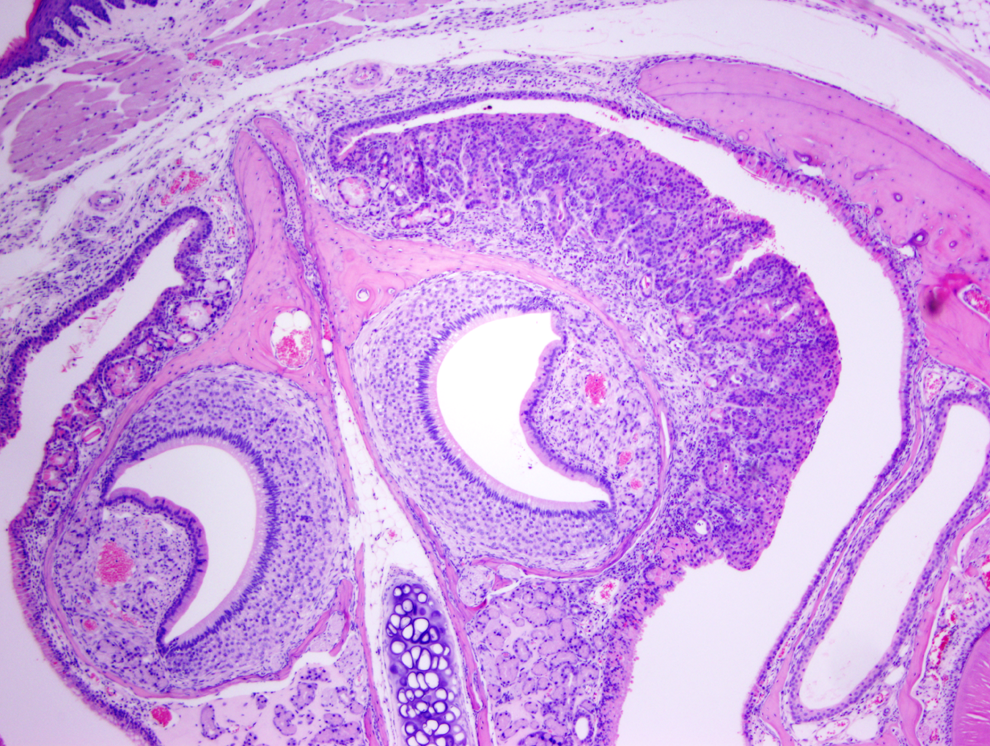
Mucosal hyperplasia, moderate degree, nasal cavity, middle section in a test article–treated mouse (H&E, ×40).
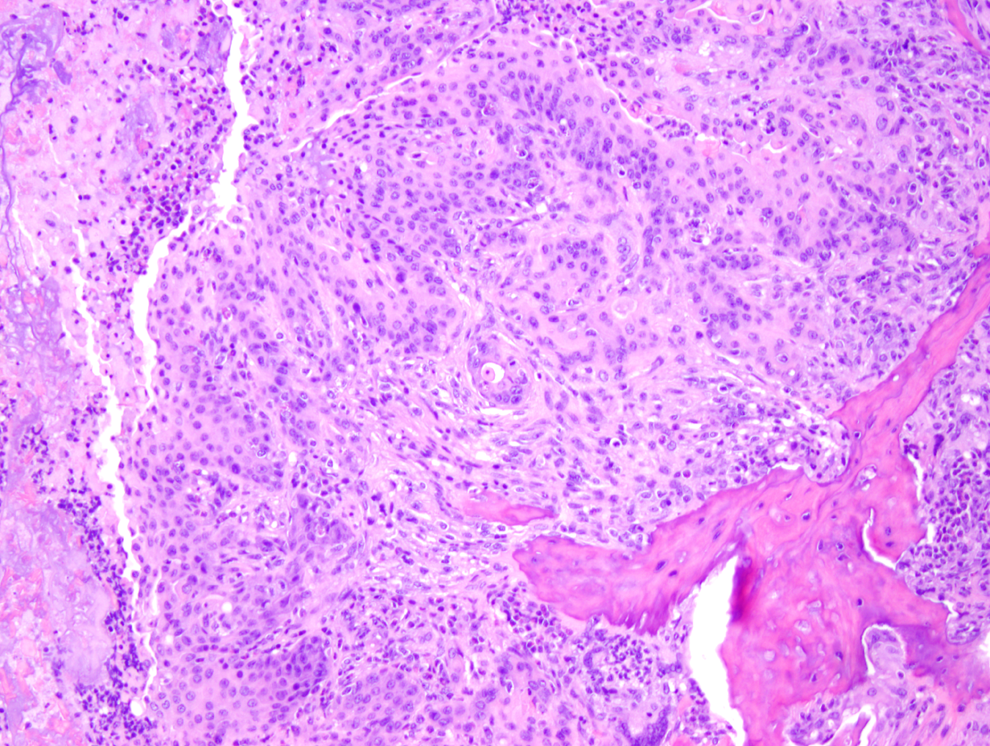
Carcinoma in the nasal cavity, posterior section, in a control mouse (H&E, ×100).
Comparison of Lesions in Male and Female Mice With or Without Exudative Inflammatory Lesions
In the male and female mice with exudative inflammatory lesions (16 studies), there was a dose-related statistically significant increase in the incidence of exudative inflammatory lesions as well as mucosal hyperplasia (Tables 1 and 2). In general, the vehicle-treated animals of both sexes had more minimal inflammatory and hyperplastic changes, whereas the test article–treated mice had more mild to marked inflammation as well as hyperplastic changes, indicating that the test article often exacerbated the lesions. Along with inflammation, the degree of erosion, necrosis, and attenuation was often more severe in the test article–treated groups of both sexes than in the vehicle groups of both sexes. The incidence of both inflammatory and hyperplastic lesions was always higher in the test article groups than in the vehicle groups of both sexes (Tables 1 and 2). In the male and female mice without the exudative inflammatory lesions (16 studies), there was also a dose-related statistically significant increase in the incidence and severity of mucosal hyperplasia. It is important to note that in the male and female mice without the exudative inflammatory lesions, the combined incidence of mucosal hyperplasia was much lower than that in the males and females with exudative inflammatory lesions for all degrees of severity. In males with exudative inflammatory lesions, the combined incidence of mucosal hyperplasia was 284/1,775, whereas the combined incidence of mucosal hyperplasia in males without the exudative inflammatory lesions was 50/1,610. Similarly, in females with exudative inflammatory lesions, the combined incidence of mucosal hyperplasia was 314/1,775, whereas in females without exudative inflammatory lesions, the combined incidence of mucosal hyperplasia was 59/1,610. When the incidence of carcinomas as well as hemangiosarcomas was compared between male and female mice with or without exudative inflammatory lesions, there were no statistically significant differences.
Discussion
There are no published reports on exudative inflammatory lesions and their association with mucosal hyperplasia and tumors in the nasal cavities of Tg.rasH2 mice. However, there have been many papers published, mainly in rats, that have shown exudative inflammatory lesions commonly in the most posterior sections and concurrent erosion, necrosis, hyperplasia, squamous metaplasia in (noninhalation) oral gavage studies, most likely caused by gastric reflux. 4 -9 The occurrence of neoplasia in these gavage rat studies is very rare. 10 These authors identified several causes for the inflammatory lesions in nasal cavities within gavage studies, which included irritant properties, low pH, and/or viscosity of the vehicle and/or the test article. These authors also discussed the complexity of differentiating these nasal cavity lesions induced by reflux in gavage studies, which may mask or exaggerate the compound-related toxicity. Based on the evaluation of the studies that we have performed, we feel that gastric reflux is the most likely cause of these lesions rather than the direct toxic effect of the test article. The lesions in our studies were also seen in the most posterior section of the nasal cavity section. The Tg.rasH2 mice are generally much smaller than the conventional 2-year mice as well as rats and their small esophageal size may play a role in causing rapid and repeated gastric reflux as these mice are dosed on a daily basis in these gavage studies for a period of 26 weeks. Besides, whenever we have seen the exudative inflammatory lesions as well as hyperplastic changes in the nasal cavities of these mice with vehicle and/or test article with irritant properties, low pH, or viscosity, we have not concurrently seen systemic changes such as gastrointestinal hyperplasia, necrosis, erosion, ulceration, or inflammation, which makes us believe that these lesions in the nasal cavity are due to repeated gastric reflux of transient duration on a daily basis.
Retrospective analysis of our data showed that in 16 studies, when the vehicle and/or test article administered via oral gavage was in the form of a salt, had irritant properties, had low pH, and/or was viscous, caused local irritation of the nasal cavity, most likely due to reflux leading to exudative inflammatory lesions in the nasal cavity. These exudative inflammatory lesions were characterized by the accumulation of eosinophilic edematous proteinaceous fluid, fibrin, mucin, sloughed epithelial cells, and infiltration of mostly degenerate neutrophils. Accompanying changes included degeneration, necrosis, and erosion of respiratory epithelium. Due to the decreased amount and lower severity of degeneration, necrosis, and erosion in minimal to mild exudative inflammatory lesions, and their more common presence with increasing severity in the moderate to marked exudative inflammatory changes, we think that degeneration, necrosis, and erosion are secondary to inflammation at higher degrees of severity rather than leading to secondary inflammation.
In most of the 16 studies with exudative inflammatory lesions in the nasal cavity, the nasal cavity was considered to be the target organ. In some studies, it was considered to be a target organ because of the lack of exudative inflammatory lesions in the vehicle groups of both sexes, but not in the test article–treated groups, or the exudative inflammatory lesions were present in the vehicle groups of both sexes and were exacerbated by the test article with increased incidence and/or degree of severity. In the later 2 instances, the vehicle was either less or nonirritating, less viscous, was not in the salt form, and/or did not have a low pH. However, upon addition of the test article in the vehicle, the test article formulations became an irritant, viscous, salty, and/or acidic. In very few studies, the nasal cavity was not considered to be the target organ despite the presence of exudative inflammatory lesions, as their incidence and degrees of severity were comparable between the vehicle and test article–treated groups.
In the remaining 16 studies, in which exudative inflammatory lesions were not noted, the overall incidence of mucosal hyperplasia for all degrees of severity was much lower than that in studies in which exudative inflammation was noted. This also shows that the mucosal hyperplasia can take place independently in the absence of concurrent exudative inflammatory lesions. This mucosal hyperplasia noted in nasal cavities without concurrent exudative inflammatory lesions may simply have been a background or incidental lesion. Nevertheless, its incidence and severity was very low compared to the studies with concurrent exudative inflammatory lesions.
This retrospective analysis was performed to determine if these exudative inflammatory lesions and hyperplastic changes in mucosa led to an increase in the incidence of nasal cavity carcinomas and hemangiosarcomas when compared with mice that lack these exudative inflammatory lesions. Nasal cavity hemangiosarcomas were included in this analysis, even though they were not considered to be caused by the irritating inflammation. We analyzed the hemangiosarcomas due to the fact that these mice are predisposed to hemangiosarcomas and because nasal cavity hemangiosarcomas are the second most common tumor of the nasal cavity after carcinoma. 14 The results of our analysis clearly demonstrate that these exudative inflammatory changes in both sexes of mice can cause mucosal hyperplasia with much higher incidence and degree of severity than in mice without the exudative inflammatory lesions. We believe that these exudative inflammatory changes were secondary to either the vehicle and/or test article formulations as both were irritant, were in the form of salt, were acidic in nature, and/or were viscous. We also believe that the inflammatory changes in the nasal cavity were because of gastric reflux. Despite the hyperplastic changes, there was no increase in the nasal cavity carcinomas and hemangiosarcomas in either sex when compared with the carcinomas and hemangiosarcomas without the accompanying exudative inflammatory lesions. The incidence of these carcinomas and hemangiosarcomas was comparable in mice with or without exudative inflammatory lesions and there were no statistically significant differences. In none of the individual 32 studies was there any statistically significant increase in the incidence of nasal cavity carcinomas or hemangiosarcomas based on tumor data statistics performed on individual studies.
Footnotes
Authors’ Contributions
Madhav G. Paranjpe contributed to conception, design and interpretation and drafted the manuscript. Jessica L. Belich contributed to design and analysis and drafted the manuscript. Dayauna R. Richardson contributed to design and analysis and critically revised the manuscript. T. Vidmar contributed to design and analysis and critically revised the manuscript. Peter C. Mann contributed to conception, design, and analysis and critically revised the manuscript. Marie E. McKeon contributed to conception and analysis and critically revised the manuscript. Reem H. Elbekai contributed to design and analysis and critically revised the manuscript. C. Brown contributed to design and analysis and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
