Abstract
A 3-mo-old, female Boer goat was presented because of respiratory difficulties. Tachypnea and inspiratory dyspnea were noted during physical examination. Thoracic radiographs were unremarkable; however, upper airway and nasal passage radiographs revealed a soft tissue mass within the nasal passages. The patient underwent cardiorespiratory arrest and did not respond to resuscitation efforts during endoscopy. A large, pedunculated, semi-firm mass originated from the soft palate and obstructed 90% of the nasopharynx on autopsy. Histologically, the mass was composed of primitive cells that multifocally formed tubules and glomeruloid structures intermingled with areas of fusiform and blastemal cells. The neoplastic cells were positive for cytokeratin (tubular and glomeruloid cells), vimentin (fusiform population and blastemal cells), and Wilms tumor 1 protein (glomeruloid structures) on immunohistochemistry, consistent with a triphasic nephroblastoma. To our knowledge, nasopharyngeal nephroblastoma has not been reported previously in any species.
Nephroblastomas (Wilms tumors) are one of the most common pediatric renal tumors.5,11,15,16 Retained embryonic metanephric blastema cells, known as nephrogenic rest cells in the neonate, undergo neoplastic transformation that results in failure to either differentiate or apoptose.2,7,11 Humans and dogs are the only species with well-characterized lesions and locations for nephroblastomas. However, other species that have been diagnosed with nephroblastomas include bovine fetuses, swine, cats, fish, chickens, a rabbit, an adult sheep, and a pair of hedgehogs.3,4,6,10,13,21
In humans, nephroblastomas typically affect children under the age of 6, have no sex predilection, are unilateral, will appear as a palpable abdominal mass, and are the most common malignant renal tumor in children.5,9,15 Additional clinical signs are relatively uncommon, but can include abdominal pain, abdominal distension, and constipation most commonly, with hypertension, hematuria, loss of appetite, weight loss, or fever occurring less frequently.5,15
In dogs, these tumors usually appear as primary renal tumors or as a primary ectopic nephroblastoma within the thoracolumbar spinal cord.2–4,13 Canine spinal cord nephroblastomas generally affect younger dogs, and are typically intradural extramedullary masses with clinical signs of thoracolumbar myelopathy secondary to spinal cord compression.2,7,8,20 The rarity of this tumor makes elucidating the pathogenesis and best treatment option challenging.
A 3-mo-old, female Boer goat was presented to the Texas A&M Large Animal Hospital (College Station, TX) with a 3-wk history of respiratory difficulty and nasal discharge. The goat had inspiratory dyspnea, tachypnea, and bilateral ocular and nasal discharge on physical examination. Thoracic radiographs were unremarkable; however, a soft tissue mass was evident on radiographs of the upper airway and nasal sinuses. The patient underwent cardiorespiratory arrest during endoscopic examination.
Postmortem examination revealed a 4.4 × 2.5 × 1.5 cm, smooth, tan-to-white, semi-firm, pedunculated mass that originated from the soft palate and obstructed 90% of the nasopharynx (Fig. 1). The mass was presumed to be a large nasopharyngeal polyp after gross examination. Airway obstruction by the mass during the endoscopic examination was presumed to be the cause of death. Samples were processed for routine histopathology, and slides were stained with hematoxylin and eosin. Additionally, serial sections from the neoplasm were stained with the following antibodies: pancytokeratin (AE1/AE3; monoclonal mouse antibody; Biocare Medical), vimentin (monoclonal mouse antibody, clone V9; Agilent/Dako), and Wilms tumor 1 protein (WT1; mouse monoclonal, clone 6F-H1; Cell Marque).
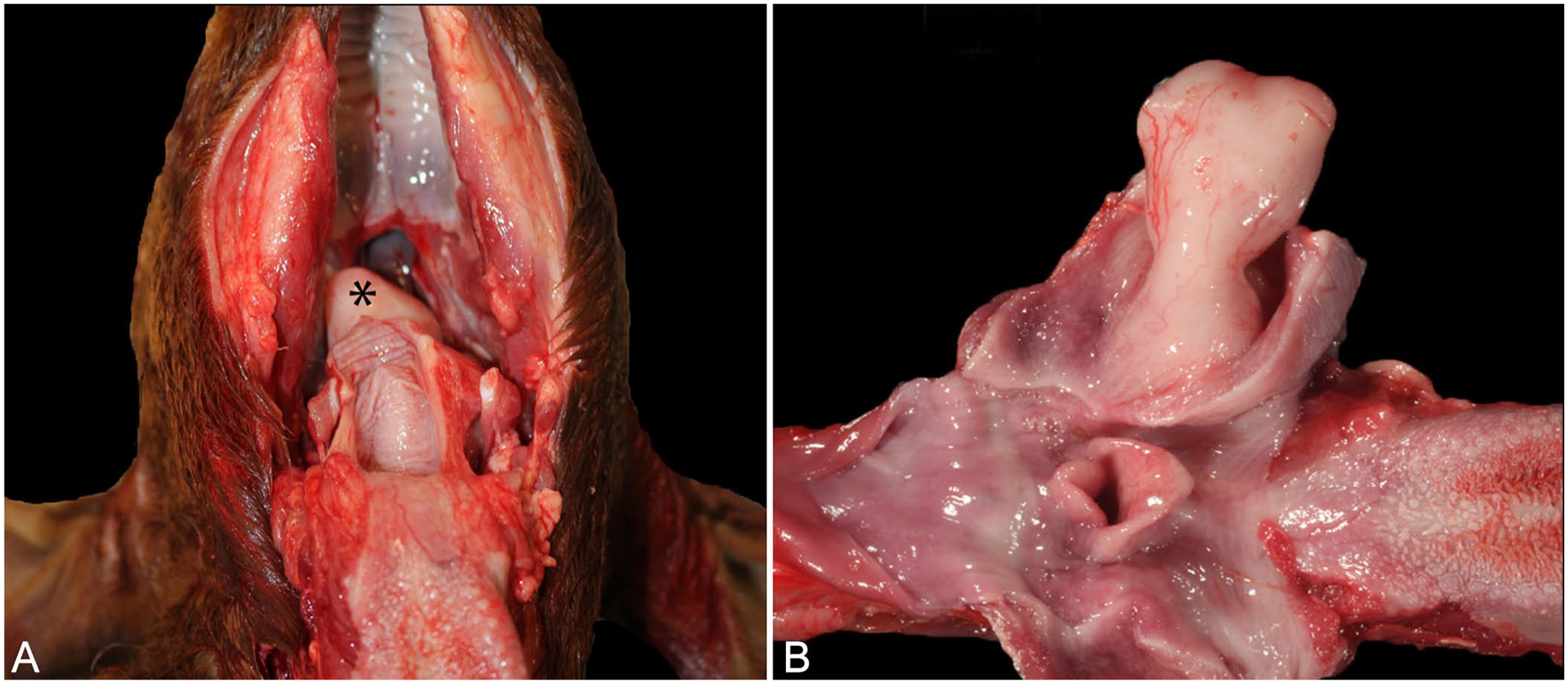
Nephroblastoma in the nasopharynx of a Boer goat. The nasopharyngeal mass occluded ~90% of the airway.
Histologically, the mass was characterized by proliferations of 3 distinct cell populations (Fig. 2). One of the populations was composed of epithelial cells multifocally forming tubules that were arranged infrequently into glomeruloid structures (Fig. 2A). These cells had indistinct cell borders, moderate amounts of eosinophilic cytoplasm, and round-to-oval nuclei with finely stippled chromatin and inconspicuous nucleoli. The second population was composed of fusiform mesenchymal cells loosely arranged in streams (embryonal mesenchyme; Fig. 2B). These cells had indistinct cell borders, moderate amounts of eosinophilic fibrillar cytoplasm, and oval-to-elongated nuclei with finely stippled chromatin and inconspicuous nucleoli. Although not a prominent feature in this neoplasm, a third population consisted of few small basophilic blastemal cells with round nuclei and coarsely stippled chromatin arranged in nests (Fig. 2C). On immunohistochemistry (IHC), the epithelial cells were positive for pancytokeratin (Fig. 2D), the glomeruloid structures were positive for both pancytokeratin (Fig. 2D) and WT1 protein IHC (Fig. 2E), and the mesenchymal and blastemal cells were positive for vimentin (Fig. 2F). The mass was diagnosed as a nephroblastoma.
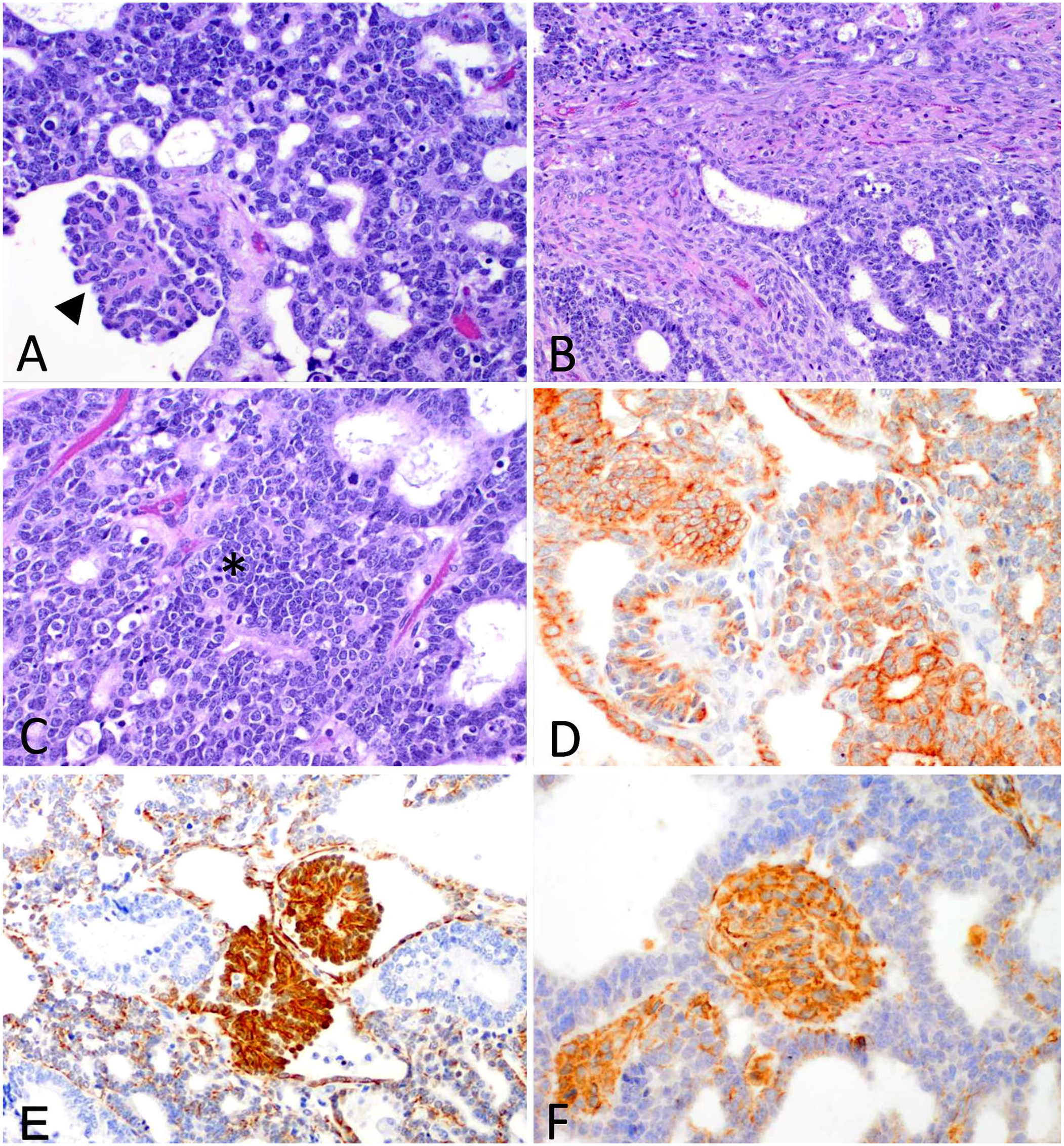
Nephroblastoma in the nasopharynx of a Boer goat; 3 distinct neoplastic cell populations were present.
Histologically, nephroblastoma (also known as Wilms tumor and embryonal nephroma) is a triphasic tumor composed of 3 discrete cell populations including epithelial, stromal, and blastemal cells.3,4,6,11,13,17 It is possible to have monophasic or biphasic variants, depending on the amount of differentiation present, which can make the diagnosis challenging.4,8,9,13,16,21 The epithelial component can contain primitive tubules with rosette-like structures to somewhat-differentiated tubules and glomeruli.3,8,16 These primitive glomeruli are considered highly diagnostic.4,13 The stromal component may vary from dense, undifferentiated mesenchymal cells to loose, myxomatous areas.4,13,16 Occasionally, cartilaginous, muscular, or osseous metaplasia of the stromal component is present.3,4,6,10,13 Finally, the blastemal component is the least differentiated with small, round, basophilic cells in sheets or serpiginous aggregates that have overlapping nuclei and frequent mitoses.3,16 A classic pattern is the proliferation of blastemal cells that are surrounded by tubules, and glomeruli that are embedded in mesenchymal stroma. 13 This pattern was not observed in our case. Blastemal patterns are not predictive of prognosis. 16
The primary gross differential diagnosis for nephroblastoma is lymphoma. 13 Traditionally, histologic differential diagnoses for nephroblastoma depend on the monophasic, biphasic, or triphasic pattern of the tumor. Differentials listed here are specific to renal, not ectopic, origin. Differentials for a monophasic blastemal tumor include any other undifferentiated tumor such as a neuroblastoma, primitive neuroectodermal tumor, desmoplastic small round cell tumor, or synovial sarcoma. 16 IHC is critical to ensure proper diagnosis.2,6,16 Histologic features and IHC are needed to differentiate a pure epithelial tumor from a metanephric adenoma and hyperplastic perilobar nephrogenic rests. 15 Differentials for a monophasic stromal tumor include a clear cell sarcoma and mesoblastic nephroma. 16 In our case, differentials for the anatomic location of the tumor included squamous cell carcinoma, nasopharyngeal carcinoma, alimentary papilloma, and an adenomatous polyp. 1
It has been shown on IHC for canine nephroblastomas that tubules and glomeruli will stain positive for cytokeratin and negative for vimentin, whereas the blastemal and stromal components will stain positively for vimentin and negative for cytokeratin.6,19 WT1 is a protein product of the WT1 tumor suppressor gene that is normally expressed during renal development and overexpressed in children with Wilms tumor.6,9,21 WT1 is strongly immunoreactive to blastema cell nuclei within the glomeruloid structures in canine nephroblastomas. The WT1 protein orchestrates the transition from epithelial to mesenchymal tissue during nephrogenesis.6,11,20,21 This immunohistochemical stain is often used in dogs to differentiate between a nephroblastoma and primitive neuroectodermal tumor.2,4,7,8
The reports of nephroblastoma in ruminants include bovine fetuses and a single adult sheep. In the bovine fetuses, the stromal component could be as differentiated as the epithelial component. The reported cases were confined to a renal or extrarenal primary tumor.6,10,13 Furthermore, in ruminants, the tumor could be composed predominantly of the stromal component.4,13
There is a single case report of an 8-y-old boy with a primary renal nephroblastoma with multiple metastases including the nasopharynx. An initial nasopharyngeal biopsy had sheets of undifferentiated tumor cells. Primitive or abortive glomeruli and tubules were only histologically present within the left kidney. 8
The National Wilms Tumor Study Group staging criteria for humans has been applied to several canine cases.3,12,14,18 In humans, staging uses tumor location and metastatic distribution, and includes histologic features to guide surgical and/or chemotherapeutic protocols.3,9 This staging protocol could not be applied to our case, given that the animal was presented for postmortem examination and the primary tumor was not associated with the kidney.
We found no evidence of a primary tumor within the kidneys or other organs. Therefore, the nasopharyngeal nephroblastoma was considered primary, and not a metastatic site. To our knowledge, primary nasopharyngeal nephroblastoma has not been reported previously.
Footnotes
Acknowledgements
We thank the histology and IHC laboratory personnel at Texas A&M University and the University of Georgia Veterinary Diagnostic laboratories for their technical assistance with this case.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
