Abstract
Introduction
Also one of the world’s oldest medicinal plants, its use first described in Divine Husbandman’s Materia Medica (the earliest known Chinese pharmacopoeia).
8
Dried leaves of
The most predominant iminosugar in
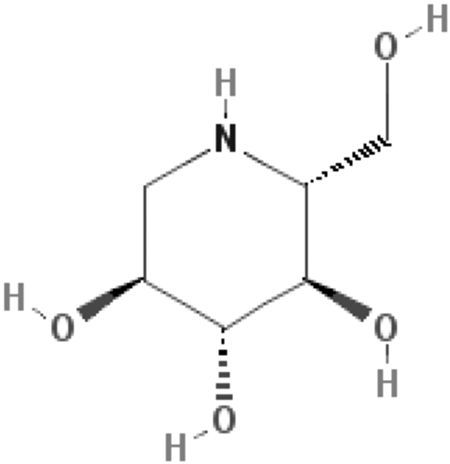
Chemical Structure of 1-deoxyjirimycin (DNJ)
Reducose 5% (formerly IminoNorm 5%), produced by Phynova Group Limited, is an iminosugar-rich aqueous extract of
Although available toxicological data suggest that
Material and Methods
Test Article
The test article was Reducose 5%, a commercial aqueous extract of the leaves of
Reducose 5% (batch number IM150522), a water-soluble, light brown powder with a characteristic odor and slightly sour taste, containing 4.65% DNJ was provided to Toxi-Coop Zrt (Budapest, Hungary) along with specifications, a certificate of analysis, and MSDS by the sponsor of the studies (Phynova Group Limited, Oxford, United Kingdom).
Twenty Eight–Day Repeated Dose Oral Toxicity Study in Rats
This 28-day repeated dose oral toxicity study was conducted to characterize the toxicity profile of Reducose 5%, the test article. The study was conducted in compliance with GLP and internationally accepted guidelines Organisation for Economic Co-operation and Development (OECD) 407
32
and
Test article doses were prepared daily as a solution by dissolving Reducose 5% in distilled water (vehicle) to achieve concentrations of 100, 200, and 400 mg/mL (approximately 5, 10 and 20 mg/mL DNJ) in order to provide a constant dosing volume of 10 mL/kg bw. Stability data conducted by the manufacturer have indicated that DNJ is stable in an aqueous (water) solution for up to 3 months. Doses were prepared by careful weight measurement and administered within 4 hours of preparation. The negative control groups received the same volume of the distilled water vehicle only.
Specific pathogen-free male and female Hsd.Han Wistar rats (Toxi-Coop) were housed individually with a 12-hour light–dark cycle at 22°C ± 3°C and 30% to 70% relative humidity in type II polypropylene/polycarbonate cages with Lignocel certified laboratory wood bedding (J. Rettenmaier & Sohne GmbH+Co.KG; D-73494 Rosenberg). Animals received ssniff SM R/M-Z+H complete diet for rats and mice (ssniff Spezialdiäten GmbH, Soest, Germany) and potable tap water ad libitum.
At the start of the experimental period, animals were 37 to 40 days old and weighed 131 to 162 g (males) and 103 to 121 g (females). Eighty rats were stratified by weight and randomly assigned to 4 groups of 10 rats/sex/group. The test article was administered by gavage in doses of 0 (vehicle-control), 1,000, 2,000, and 4,000 mg/kg bw/d (approximately 50, 100 and 200 mg/kg bw/d of DNJ) for 28 days (males) or 29 days (females).
Because the available literature on
Animals were observed twice daily for morbidity and mortality, and general cage-side observations for clinical signs were made twice during the acclimation period and once daily after administration of the test article. Detailed clinical observations were conducted on the day prior to the first test article administration and once weekly during the experimental period. A functional observational battery was performed during the final week to assess parameters such as general physical condition and behavior, sensory reactions to various stimuli, grip strength, and motor activity using a modified Irwin test. 35 Individual body weights were recorded twice during the acclimation period, on the first experimental day prior to treatment, twice weekly during weeks 1 to 4, and immediately prior to sacrifice. Food intake was determined and food efficiency calculated once weekly (body weight gain/food consumption; calculated using SPSS PC+ statistical software). Ophthalmological examination (using Humapent 5 mg/mL eye drops; TEVA Pharmaceutical Works Private Ltd. Co, Gödöllő, Hungary) was carried out on all animals prior to the experimental period and prior to the study termination in control and high-dose group animals.
After an overnight fast (approximately 16 hours) following final administration of the test article, blood samples were collected from the retro–orbital venous plexus under Isoflurane CP anesthesia (CP-Pharma Hadnelsgesellschaft GmbH, Ostlandring, Burgdorf, Germany), after which the animals were euthanized by exsanguination. Blood samples were analyzed for hematologic, blood coagulation, and clinical chemistry parameters. Gross pathological examinations were conducted on male animals on day 28 and on female animals on day 29, and absolute and relative organ weights were determined on all animals. Full histopathological examinations were conducted on preserved organs (as specified by OECD guidelines) of all animals of the control and high-dose groups.
Statistical Analyses
Statistical analyses were conducted using SPSS PC+ software (version 4; SPSS, Inc, Chicago, Illinois). Bartlett homogeneity of variance test was used to assess heterogeneity of variance between groups. One-way analysis of variance (ANOVA) was performed if no significant heterogeneity was detected, and Duncan multiple range test was used to assess the significance of intergroup differences if a positive ANOVA result was obtained. Where significant heterogeneity was detected by Bartlett test, the Kolmogorov-Smirnov test was performed to examine normally distributed data. Kruskal-Wallis nonparametric 1-way ANOVA, followed by the Mann-Whitney
Results
No mortality was observed in any group throughout the study. Daily and weekly detailed clinical and functional observations of the animals did not reveal any toxicologically relevant abnormalities. Slight salivation occurred in 3 female rats in the 4,000 mg/kg bw/d group appearing shortly after administration and ceasing approximately half an hour thereafter. Scarring and alopecia on the neck were noted for a single male in the 2,000 mg/kg bw/d group from day 18 until the end of the observational period.
No test item–related changes were noted in body weight (see Supplemental Figures 1a and 1b), body weight gain, daily food consumption, or feed efficiency in male or female rats at any dose level. A statistically significant decrease was noted in mean body weight gain in female animals at 4,000 mg/kg bw/d compared to controls between days 18 and 21. Mean daily food consumption was slightly but significantly lower compared to controls in female animals in the 4,000 mg/kg bw/d group during the first week of the study, and a slightly higher feed efficiency was noted in the same animals during week 2. Additionally, a lower feed efficiency was noted in female and male animals in the 4,000 mg/kg bw/d group during weeks 3 and 4, respectively.
No alterations were noted in ophthalmoscopic examinations. Slight, statistically significant changes were noted in various hematology and clinical chemistry parameters in both sexes compared to control animals (see Tables 1 and 2). Among these changes was a significant increase in neutrophils (NEU) in all treated females. In females of the 2,000 and 4,000 mg/kg bw/d groups, a significant increase in activated partial thromboplastin time (APTT) and a significant decrease in lymphocytes (LYMs) were also noted. Significant increases in alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were noted in male animals in the 4,000 mg/kg bw/d group and appeared to be dose dependent.
Summary of Hematological Findings.a
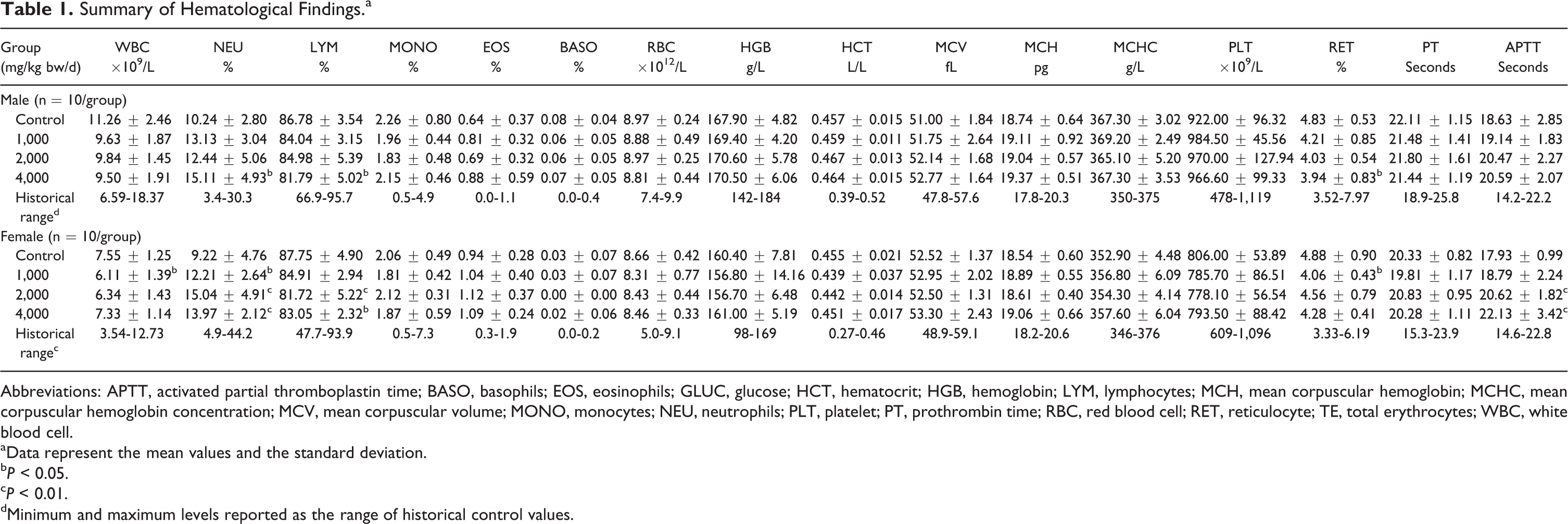
Abbreviations: APTT, activated partial thromboplastin time; BASO, basophils; EOS, eosinophils; GLUC, glucose; HCT, hematocrit; HGB, hemoglobin; LYM, lymphocytes; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; MONO, monocytes; NEU, neutrophils; PLT, platelet; PT, prothrombin time; RBC, red blood cell; RET, reticulocyte; TE, total erythrocytes; WBC, white blood cell.
aData represent the mean values and the standard deviation.
b
c
dMinimum and maximum levels reported as the range of historical control values.
Summary of Clinical Chemistry Findings.a
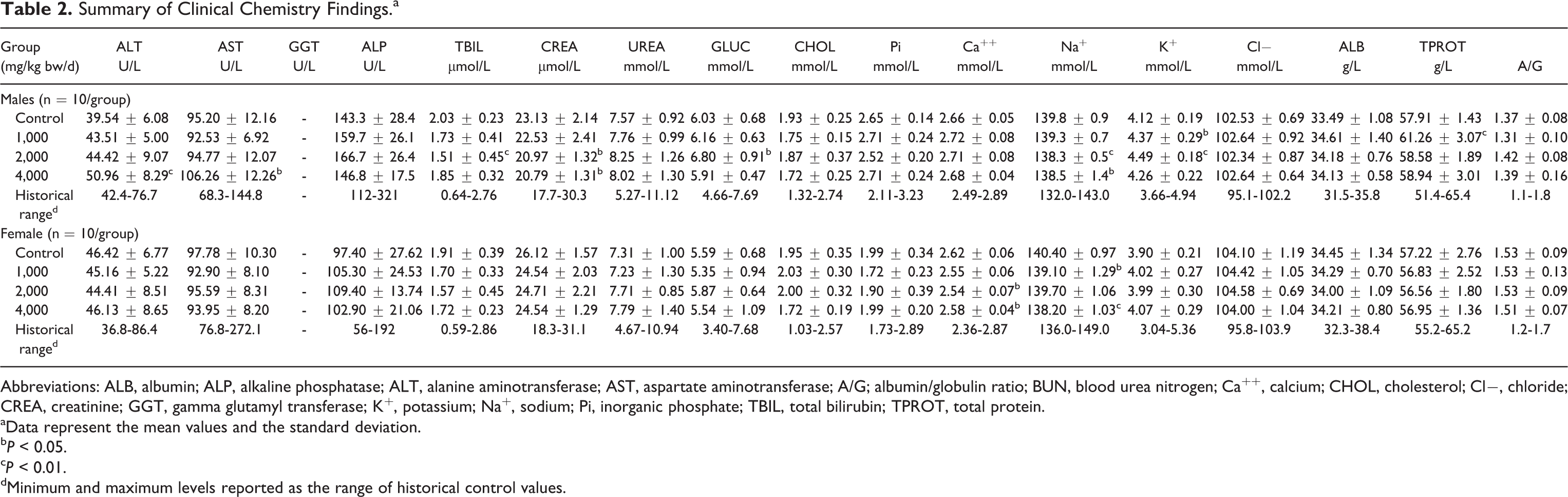
Abbreviations: ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; A/G; albumin/globulin ratio; BUN, blood urea nitrogen; Ca++, calcium; CHOL, cholesterol; Cl−, chloride; CREA, creatinine; GGT, gamma glutamyl transferase; K+, potassium; Na+, sodium; Pi, inorganic phosphate; TBIL, total bilirubin; TPROT, total protein.
aData represent the mean values and the standard deviation.
b
c
dMinimum and maximum levels reported as the range of historical control values.
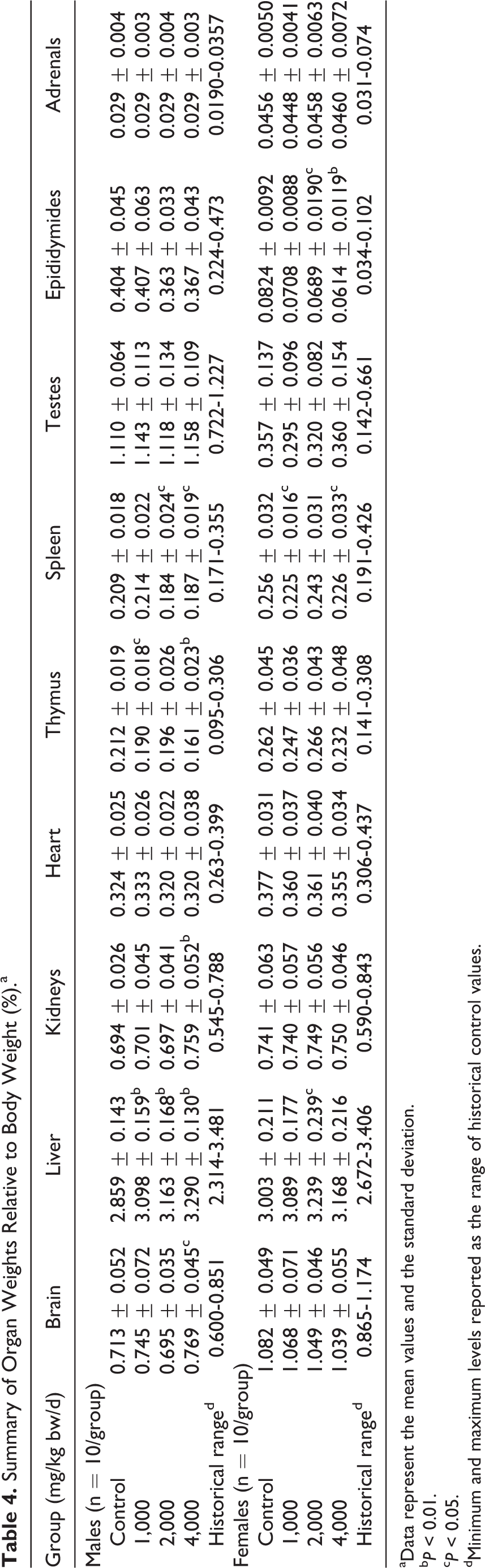
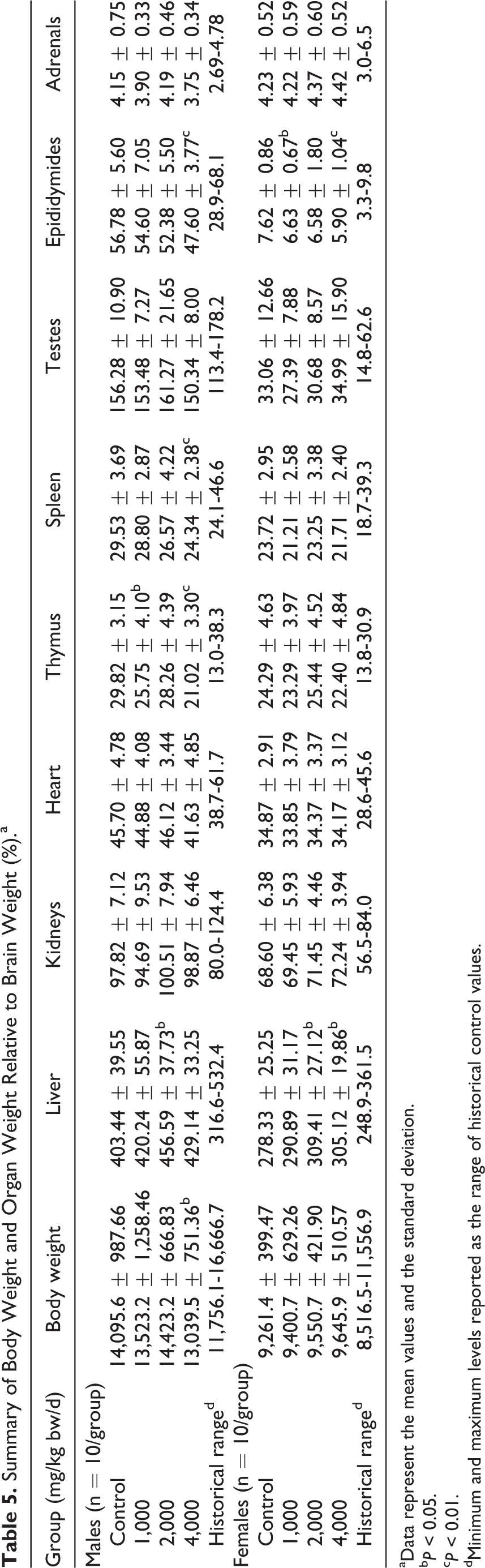
Similarly, slight, statistically significant differences compared to controls were found in various absolute and relative organ weights in both sexes (see Tables 3–5). A few of these changes appeared possibly dose dependent: a decrease in spleen (absolute and relative to body weight) and epididymides (absolute) weights in males of the 2,000 and 4,000 mg/kg bw/d groups, a decrease in epididymides weights (relative to brain weights) in the 4,000 mg/kg bw/d group, and an increase in liver weights (relative to body weight) in all treated males. Additionally, although a dose dependency was not noted, thymus weights (relative to body weight and brain weight) were significantly lower in males of the 1,000 and 4,000 mg/kg bw/d groups, and a significant decrease in thymus weights (absolute) in the 4,000 mg/kg bw/d group was observed.
Summary of Organ weights (g).a
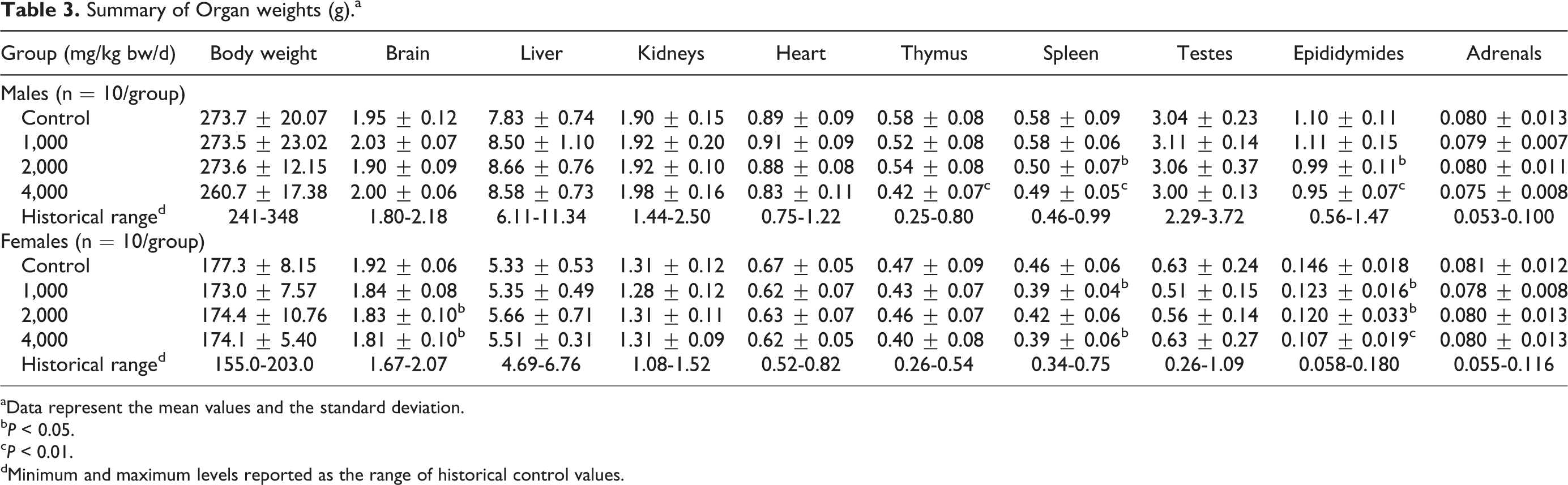
aData represent the mean values and the standard deviation.
b
c
dMinimum and maximum levels reported as the range of historical control values.
Summary of Organ Weights Relative to Body Weight (%).a
aData represent the mean values and the standard deviation.
b
c
dMinimum and maximum levels reported as the range of historical control values.
Summary of Body Weight and Organ Weight Relative to Brain Weight (%).a
aData represent the mean values and the standard deviation.
b
c
dMinimum and maximum levels reported as the range of historical control values.
In all treated female animals, ovarian weights (absolute) were significantly and dose dependently decreased, and a significant decrease in ovarian weights (relative to body weight) was observed in the 2,000 and 4,000 mg/kg bw/d groups. A significant decrease in ovarian weights (relative to brain weight) in the 1,000 and 4,000 mg/kg bw/d groups was also observed. Brain weights (absolute) were also significantly decreased in the 2,000 and 4,000 mg/kg bw/d groups. Lastly, although dose dependency was not clear, liver weights (relative to brain weight) were significantly higher in females of the 2,000 and 4,000 mg/kg bw/d groups and spleen weights (absolute and relative to body weight) were significantly decreased in females of the 1,000 and 4,000 mg/kg bw/d groups.
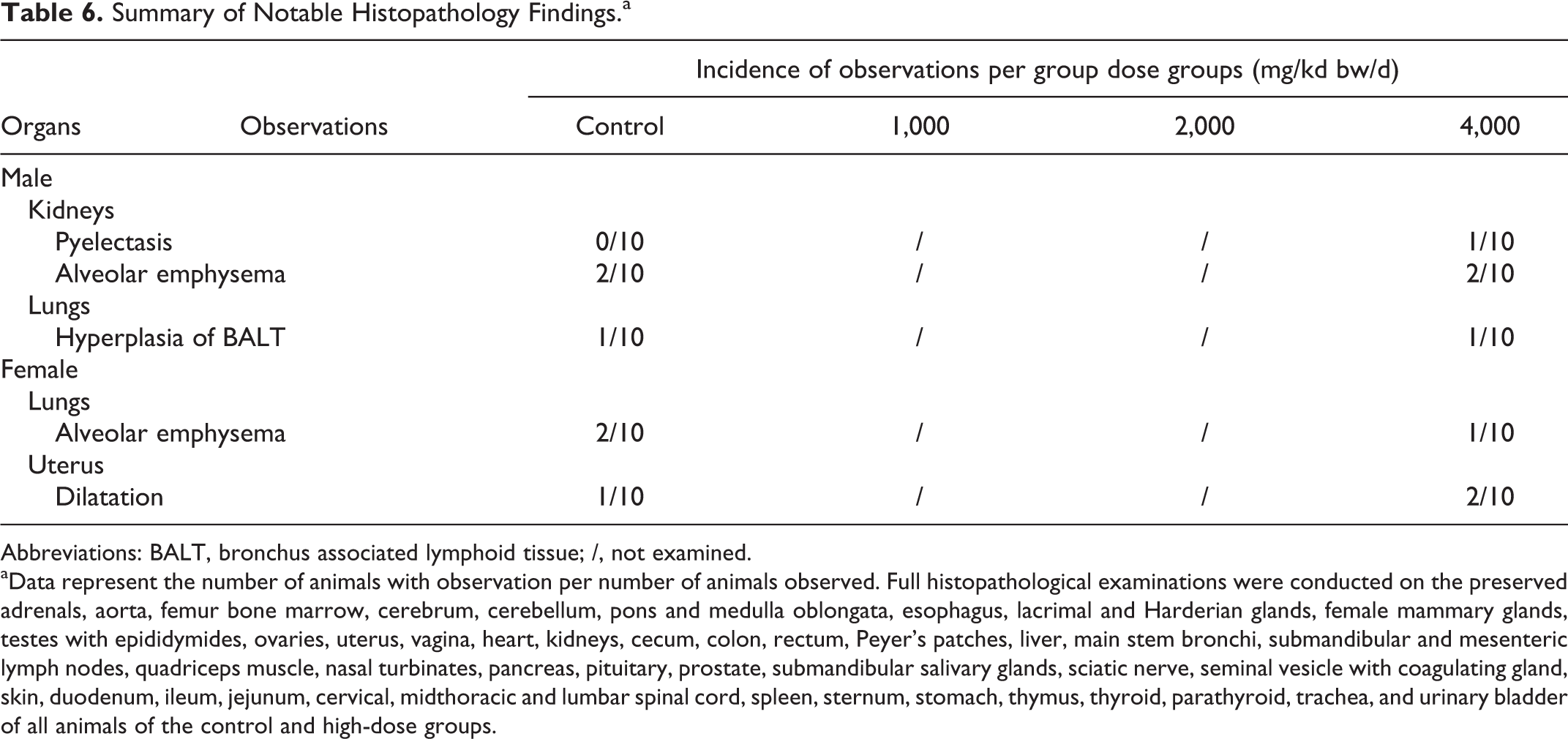
Upon gross examination, pyelectasis (dilatation of the renal pelvis) was seen unilaterally in 1 male in the 4,000 mg/kg bw/d group. An elevated percentage of NEU and decreased percentage of LYMs were detected in this animal; however, no correlating clinical or histopathological findings were discovered. In female animals, hydrometra (dilated and fluid-filled uterine horns) was detected in all groups including control upon macroscopic and microscopic evaluations (see Table 6). Mild alveolar emphysema and mild hyperplasia of bronchus-associated lymphoid tissue (BALT) were noted in both control and treated animals with similar incidence. No other histopathological findings were observed in the examined organs and tissues.
Summary of Notable Histopathology Findings.a
Abbreviations: BALT, bronchus associated lymphoid tissue; /, not examined.
aData represent the number of animals with observation per number of animals observed. Full histopathological examinations were conducted on the preserved adrenals, aorta, femur bone marrow, cerebrum, cerebellum, pons and medulla oblongata, esophagus, lacrimal and Harderian glands, female mammary glands, testes with epididymides, ovaries, uterus, vagina, heart, kidneys, cecum, colon, rectum, Peyer’s patches, liver, main stem bronchi, submandibular and mesenteric lymph nodes, quadriceps muscle, nasal turbinates, pancreas, pituitary, prostate, submandibular salivary glands, sciatic nerve, seminal vesicle with coagulating gland, skin, duodenum, ileum, jejunum, cervical, midthoracic and lumbar spinal cord, spleen, sternum, stomach, thymus, thyroid, parathyroid, trachea, and urinary bladder of all animals of the control and high-dose groups.
Discussion
No toxicologically relevant abnormalities were observed in the clinical and functional observational examinations conducted throughout the study. The salivation in the three 4,000 mg/kg bw/d females was not considered toxicologically relevant due to its transient nature (occurring at least 1 day and up to 16 days, depending on the animal) and may have also been due to large doses of the test article. The scarring and alopecia findings in the single male in the 2,000 mg/kg bw/d group were also not considered toxicologically relevant because they are known to be common dermal changes in experimental rats. 36
The changes noted in body weight gain, daily food consumption, and feed efficiency were not considered to be of toxicological relevance due to their transient nature, the small degree of change, and because they had no significant influence on overall body weight or in body weight gain. In hematological examinations, changes in APTT were primarily due to a relatively low mean control value (with respect to historical control values) and were not related to any histopathological or clinical abnormalities. The changes noted in NEU and LYM were not dose dependent and remained well within historical controls. The significant increases in ALT and AST in male animals in the 4,000 mg/kg bw/d group were slight in magnitude, fell well within the historical control ranges, and lacked related histopathological changes. All of the remaining statistically significant changes in hematology and clinical chemistry parameters remained within historical controls and did not correlate with any clinical or histopathological findings and, therefore, were not considered to be of toxicological relevance.
Although there was some indication of dose dependency in certain organ weight changes, they were slight in magnitude, the values remained within historical control ranges, and there were no correlating clinical or histopathological findings. The degree of increase in the renal pelvic diameter noted in 1 high-dose male animal was slight and lacked medullar or cortical atrophy, inflammatory infiltrates, hemorrhage, hemosiderin, and degenerative or fibrotic lesions; therefore, it was considered an individual disorder without toxicological significance. The presence of hydrometra was not dose dependent and is considered a common neurohormonal phenomenon in connection with the proestrus phase of the female sexual cycle, a frequent observation in experimental animals. 37,38 The mild alveolar emphysema and mild hyperplasia of BALT were considered a consequence of exsanguination and a physiological immunomorphological phenomenon, respectively. 39 –42
All of the significant differences noted above were slight in magnitude and remained well within the laboratory’s historical control ranges for the species of animal used. No clinical or histopathological correlates were found with respect to hematological and clinical chemistry findings or organ weight changes; therefore, these findings were not considered to be toxicologically relevant.
Conclusions
In the present study, Reducose 5%, a commercial aqueous extract of
The safety of
Footnotes
Acknowledgments
The authors thank the following individuals for their contributions to the work: participating investigators István Buda, Ildikó Hermann, Beatrix Szilágyi Sümeginé, Ruth Weimer, Klára Fritz Kovácsné, Viktória Balogh, Ibolya Bogdán, Monika Csatári, Irén Somogyi Háriné, Brigitta Horváth, Isvánné Horváth, János Stáhl, Ágota Jó Schüllerné, Ferenc Szabó, Edit Szám, Viktória Matina, and Zsuzsanna Vuleta for the performance of experimental tasks, quality assurance, and/or collection of data; and Jared Brodin for administrative support in preparation of the manuscript.
Authors Contributions
Tennille K. Marx contributed to acquisition, analysis or interpretation of data, drafted the manuscript, and critically revised the manuscript for important intellectual content. Róbert Glávits contributed to acquisition, analysis or interpretation of data, and critically revised the manuscript for important intellectual content. John R. Endres substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation of data, and critically revised the manuscript for important intellectual content. Philip A. Palmer contributed to acquisition, analysis, or interpretation of data, drafted the manuscript, and critically revised the manuscript for important intellectual content. Amy E. Clewell contributed to acquisition, analysis, or interpretation of data and critically revised the manuscript for important intellectual content. Timothy S. Murbach contributed to acquisition, analysis, or interpretation of data and critically revised the manuscript for important intellectual content. Gábor Hirka substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation of data, and critically revised the manuscript for important intellectual content. Ilona Pasics substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation of data, and critically revised the manuscript for important intellectual content. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AIBMR Life Sciences, Inc. was contracted by the study sponsor, as an independent third party, to determine appropriate study protocols and dose selections, place the study, approve the study plan, monitor the toxicological study described above, as well as to analyze and interpret the resulting data and prepare the manuscript. Toxi-Coop Zrt was contracted by AIBMR to develop the study plan and conduct, analyze and interpret, and report the results of the toxicological study described above.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for the research was provided by Phynova Group Limited.
