Abstract
Nicotinamide riboside (NR) is a naturally occurring form of vitamin B3 shown to preferentially elevate the nicotinamide adenine dinucleotide (NAD+) metabolome compared to other vitamin B3 forms (nicotinic acid and nicotinamide). Although daily requirements of vitamin B3 are typically met through the diet, recent studies have shown that additional supplementation with NR may be an effective method to counter the age-related decline in NAD+ levels as NR bypasses the rate-limiting step in NAD+ biosynthesis. Furthermore, pharmaceutical applications of NR for age-related disorders have been proposed. In this study, the safety of a high-purity, nature-identical, synthetic NR (NR-E), manufactured under the guidelines of good manufacturing practices for dietary supplements (21 CFR 111) as well as for drugs (21 CFR 210), was investigated in a 90-day oral toxicity study in Sprague Dawley rats at 300, 500, and 1,200 mg/kg/d. There were no mortality or clinical observations attributable to the test substance at any dose. A small but statistically significant decrease in body weight was observed at day 92 in the 1,200 mg/kg/d NR-treated male rats only. In contrast to a previously published safety assessment using a different synthetic NR (NIAGEN), whose no-observed-adverse-effect-level (NOAEL) was reported to be 300 mg/kg/d, there were no adverse changes in clinical pathology parameters and no notable macroscopic or microscopic findings or treatment-related effects at similar doses. In the current study, the NOAEL for systemic toxicity of NR-E in Sprague-Dawley rats was conservatively determined to be 500 mg/kg/d for males (solely based on body weight) and 1,200 mg/kg/d for females.
Introduction
Nicotinamide riboside (NR) is a naturally occurring pyridine nucleoside member of the vitamin B3 family, shown to efficiently induce biosynthesis of nicotinamide adenine dinucleotide (NAD+), an essential cofactor found in all living cells involved in enzyme-catalyzed reduction–oxidation (redox) reactions. 1 -6 NAD+ plays a canonical role as a coenzyme catalyzing oxidative phosphorylation and redox reactions in fundamental metabolic processes such as glycolysis, fatty acid β-oxidation, and the tricarboxylic acid cycle. 7 -9 NAD+ also serves as a substrate for NAD+-dependent enzymes including poly-adenosine diphosphate-ribose polymerases, sirtuins (SIRTs), and CD38 and CD157 ectoenzymes that mediate critical cellular processes. 10 -14 For instance, NAD+-dependent activation of SIRTs regulates important physiological aspects such as circadian rhythm, glucose, and fat metabolism, and is implicated in controlling the processes of normal aging. 13,15 Levels of NAD+ have been shown to decline with age in a range of human tissues, including skin, 16 blood, 17 and brain. 18 Furthermore, perturbations to the NAD+ metabolome have been observed in a number of preclinical disease models. 19 -21 Therefore, augmentation of NAD+ with various forms of vitamin B3 is emerging as a potential strategy to alleviate a wide range of age-associated conditions. 22 -27
In rodents, it has been shown that oral administration of NR effectively elevates hepatic NAD+ levels, with a favorable pharmacokinetic profile relative to other forms of vitamin B3 (nicotinamide and nicotinic acid). 2 In this previous study, both NR and nicotinamide effectively elevated NAD+ levels; however, NR increased markers of NAD+ utility, such as adenosine diphosphate ribose and nicotinamide, to a greater extent. As both of these metabolites are by-products of NAD+ consumption, these findings indicate that NR is effectively being utilized by NAD+ consuming enzymes with a comparatively higher turnover rate. Subsequent human clinical studies have demonstrated the safety and tolerability of NR at doses up to 2,000 mg/d for 12 weeks and confirmed the efficacy of NR for inducing NAD+ biosynthesis. 1,3,4,26,28,29 More recently, dozens of clinical trials have been initiated to better understand the safety and therapeutic potential of increasing NAD+ levels using NR (See Clinicaltrials.gov). 22 This has motivated the development of NR-E: a nature identical, synthetic NR that is manufactured under the guidelines of good manufacturing practices (GMP) 21 CFR 111/210 that would be suitable for both dietary supplementation and therapeutic applications.
Previous toxicological assessment by Conze et al with a synthetic NR (branded as NIAGEN) established that NR possesses a similar safety profile to that of another vitamin B3, nicotinamide. 30 This prior safety study included a 90-day repeat dose toxicity study, in which Sprague Dawley (SD) rats were administered NIAGEN orally at the dose levels 300, 1000, and 3,000 mg/kg/d; the tolerable upper intake level (UL) in humans for these doses corresponds to 180, 600, and 1,800 mg/d, assuming an average human weight of 60 kg and a 100-fold safety factor. The no-observed-adverse-effect-level (NOAEL) was determined to be the lowest dose of 300 mg/kg/d, and the lowest-observed adverse-effect-level was determined to be 1,000 mg/kg/d. Adverse effects observed included treatment-related adverse effects in liver, kidney, testes, epididymides, and ovaries, along with increases in clinical parameters related to hepatocyte damage (alanine aminotransferase [ALT], alkaline phosphatase [ALP], γ-glutamyl transferase [GGT]), corresponding liver weight increase, centrilobular hepatocellular hypertrophy, and single cell necrosis. Notably, the adverse effects observed at the highest dosage level of 3,000 mg/kg/d were also observed with a similar magnitude in a separate cohort receiving an equimolar dose of nicotinamide at 1,260 mg/kg/d as a positive control. Target organs of toxicity were similar as well: liver, kidney, ovary, and testes. Based on the NOAEL of 300 mg/kg/d, the UL in humans was determined to be 180 mg/d.
The purpose of the present study was to evaluate the safety profile of NR-E: a nature-identical, synthetic form of NR that was produced using a proprietary manufacturing process developed by Elysium Health, under the guidelines of GMP 21 CFR 111/210. We conducted a 90-day subchronic toxicity study of NR-E, administered orally at doses of 300, 500, and 1,200 mg/kg/d, to male and female SD rats, followed by a 28-day recovery period. This study expands on previous assessment of NR (NIAGEN) by Conze et al, as it includes a smaller dosage range, in addition to a 28-day recovery arm. The inclusion of a recovery arm enables a more sensitive characterization of findings among the different dosage levels.
Materials and Methods
Test Substance
A synthetic, nature-identical nicotinamide riboside chloride (CAS #23111-00-4) designated as NR-E was manufactured using a proprietary process under the guidelines of GMP 21 CFR 111/210 by Elysium Health (New York). The specification for the NR-E used in this 90-day study was set to GMP 21 CFR 111. The crystalline product was verified by high-performance liquid chromatography with mass spectroscopy detection using a positive-mode electrospray ionization indicating a >97% purity.
Study Design
Nicotinamide riboside chloride was tested in a Good Laboratory Practices (GLP)-compliant 90-day oral toxicity study for potential toxicity in CD IGS Sprague-Dawley (CrL: SD) rats. The study was performed at Product Safety Labs (PSL; Dayton, New Jersey) in accordance with (1) Organization for Economic Co-operation and Development (OECD) Guidelines for the Testing of Chemicals, Section 4 Health Effects (Part 408): Repeated Dose 90-day Oral Toxicity Study in Rodents; (2) US Food and Drug Administration (FDA) Redbook (2000): Chapter IV.C.4.a Sub-Chronic Toxicity Studies with Rodents; and (3) standard operating procedures at PSL. This study utilized both FDA and OECD guidance principles which take into consideration animal welfare concerns, with the minimal number of animals used to make accurate inferences about the sample population. The study is in accordance with the requirements set forth by the 21 CFR 58: US FDA GLP Standards, 1987, and OECD Principles of GLP, 1997.
The dosage levels 300, 500, and 1,200 mg/kg/d were selected based on a previous 90-day toxicity study performed and on the doses previously evaluated in human clinical studies; these range from 100 to 2,000 mg/d. 1,3 –5,26,28,30 Using a 100-fold safety factor and assuming a 60 kg human, the dosages assessed here corresponded to a human upper limit (UL) daily dose of 180, 300, and 720 mg/d, respectively. Twenty animals (10 males and 10 females) were randomized to each treatment arm including placebo; 10 animals (5 males and 5 females) were randomized to each recovery arm (Table 1).
Experimental Design of a 90-Day Toxicity Study of NR-E in Rats With a 28-Day Recovery Period.
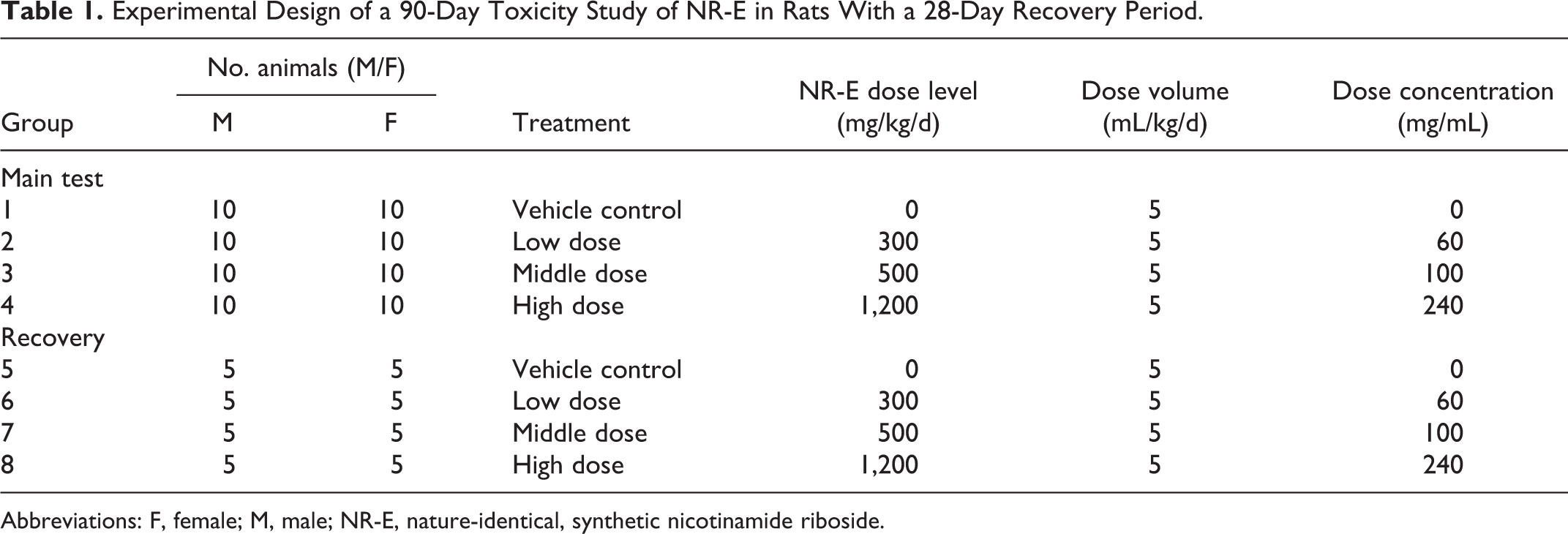
Abbreviations: F, female; M, male; NR-E, nature-identical, synthetic nicotinamide riboside.
Animals
One hundred and thirty healthy Sprague-Dawley rats (Charles River Laboratories, Raleigh, North Carolina) at approximately 7 weeks of age were acclimated to the laboratory environment for 7 consecutive days. Animals were then examined for general well-being and weighed prior to dose administration. One hundred and twenty animals (60 males and 60 females) within this cohort lacking clinical observations indicative of disease or injury and having an adequate body weight range within ± 20% of the mean body weight were selected. Animals were randomized and stratified to test groups by body weight, weighing 222 to 293 grams (males) and 168 to 209 grams (females) at approximately 8 weeks of age.
Animals were housed individually in suspended stainless-steel cages confirming to the size recommendations in the latest Guide for the Care and Use of Laboratory Animals 31 with a change of litter changing at minimum 3 times per week. Housing was regulated with a 12-hour light/dark cycle and was kept clean and vermin free in an environmentally controlled room with temperature and humidity maintained at 19 °C to 25 °C and 33% to 60%, respectively.
2016 Certified Envigo Teklad Global Rodent Diet (Envigo Teklad, Inc) was stored in a dedicated temperature and humidity monitored feed storage site and available ad libitum during acclimation and throughout the study, except when animals were fasted prior to clinical sample collections and terminal sacrifice. All animals were provided with filtered tap water ad libitum from individual bottles attached to the cages or from an automatic watering access system. A sentinel rodent health surveillance program monitored using test animals housed in the same room evaluated animals for the absence of viruses near the end of the in-life portion of the study and showed no viral contaminants.
Dose Formulation and Administration
Fresh dosage formulations containing 60, 100, and 240 mg/mL concentrations of NR were prepared daily by mixing the neat test substance weight to volume (wt/vol) in distilled water. Formulations were maintained on a stir plate during administration and used within 2 hours of preparation. Individual doses were calculated based on the most recent weekly body weights and were adjusted each week to maintain the targeted dose level for all rats (ie, mg/kg/d). All doses, including the vehicle control group, were administered volumetrically at 5 mL/kg.
Animals were dosed via oral gavage once daily for a consecutive period of at least 90 days, with terminal necropsy for main group animals scheduled on day 94 for males and day 95 for females. Recovery animals remained on the study without dose administration for a period of at least 28 days, with terminal necropsy for the recovery animals scheduled on day 123 for both sexes.
In-Life Parameters (Ophthalmological Examination, Clinical Observation, Body Weight, Feed Consumption, and Feed Efficiency)
During the acclimation period, the eyes of all rats being considered for study were examined by focal illumination, indirect ophthalmoscopy and, when indicated, slit-lamp microscopy. Mydriatic eye drops were administered prior to ophthalmoscopy, and the eyes were examined in subdued light. Subdued light was maintained in the animal room for approximately 2 to 3 hours following the examination. These procedures were repeated on all test animals prior to termination (groups 1-4 main test animals) and at the end of the recovery period (groups 5-8 recovery animals) to further investigate changes observed during the main test.
All animals were observed at least twice daily for viability. Cage-side observations of all animals were performed daily during the study, and all findings were recorded. Prior to the first treatment with the test substance on day 1, and weekly thereafter, a detailed clinical observation was conducted while handling the animal, generally occurring on days that the animals were weighed, and food consumption measurements were taken. Potential signs noted included, but were not limited to changes in skin, fur, eyes, and mucous membranes, occurrence of secretions and excretions and autonomic activity (eg, lacrimation, piloerection, pupil size, unusual respiratory pattern). Likewise, changes in gait, posture, and response to handling, as well as the presence of clonic or tonic movements, stereotypes (eg, excessive grooming, repetitive circling), or bizarre behavior (eg, self-mutilation, walking backwards) were also recorded. The date and clock time of all observations and/or mortality checks were recorded.
Individual body weights were recorded at least 2 times during acclimation. All animals were weighed on day 1 (prior to study start) and weekly thereafter (intervals of 7 days). The animals were also weighed prior to sacrifice in order to calculate organ-to-body weight ratios. Body weight gain was calculated for selected intervals and for the study overall. Individual food consumption was measured and recorded to coincide with body weight measurements. Food efficiency was also calculated and reported.
Clinical Pathology (Hematology, Clinical Chemistry, and Urinalysis)
Clinical pathology was performed on all main terminal sacrifice animals toward the end of the in-life dosing phase and on all recovery animals toward the end of the recovery period of study. The day before collection of samples for the clinical pathology evaluation, the animals were placed in metabolism cages and fasted (after 3
Hematological parameters included erythrocyte count, hemoglobin concentration, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, red cell distribution width, absolute reticulocyte count, platelet count, total white blood cell, absolute neutrophil (ANEU), absolute lymphocyte (ALYM), absolute monocyte (AMON), absolute eosinophil (AEOS), absolute basophil, absolute large unstained cell, and differential leukocyte count. Mean corpuscular hemoglobin concentration was calculated. In addition, separate blood smears, stained with new methylene blue or “Wright-Giemsa” stain, were prepared from each animal undergoing hematological evaluation and were examined, if required, to substantiate or clarify the results of hematology findings.
Clinical chemistry parameters measured included serum aspartate aminotransferase (AST), serum ALT, sorbitol dehydrogenase, ALP, total bilirubin, urea nitrogen, blood creatinine, total cholesterol (CHOL), triglycerides (TRIG), fasting glucose, total serum protein, albumin, globulin, calcium, inorganic phosphorus, sodium, potassium, and chloride (CL).
Urinalysis parameters included quality, pH, ketone, color, glucose, bilirubin, clarity, specific gravity, blood, volume, protein, urobilinogen, and microscopic urine sediment examination.
Pathology (Terminal Necropsy, Tissue Collection, and Histopathological Examination)
At termination, all surviving animals were euthanized by exsanguination from the abdominal aorta under isoflurane anesthesia. All animals in the study were subjected to a full necropsy, which included examination of the external surface of the body, all orifices, and the thoracic, abdominal, and cranial cavities and their contents. The following tissues were weighed wet as soon as possible after dissection to avoid drying: adrenals (combined), kidneys (combined), spleen, brain, liver, thymus, epididymides (combined), ovaries with oviducts (combined), uterus, heart, and testes (combined). Over 40 organs and tissues were collected and preserved in 10% neutral buffered formalin (inclusive of the STP standard list and following OECD guidelines). Additionally, epididymides, eyes with optic nerve, and testes were preserved in modified Davidson’s fixative and then retained in ethanol.
Histological examination was performed on the preserved organs and tissues of the main test animals from both the control and high-dose groups. The preserved/fixed tissues were trimmed, processed, embedded in paraffin, sectioned with a microtome, placed on glass microscope slides, stained with hematoxylin and eosin, and examined by light microscopy. Slide preparation and histological assessment, by a board-certified veterinary pathologist, was performed at Histo-Scientific Research Laboratories.
Statistical Analysis
For the in-life and organ weight data, mean and standard deviations were calculated for all quantitative data. If warranted by sufficient group sizes, data within groups were evaluated for homogeneity (HO) of variances and normality by Bartlett test. 32 Where Bartlett test indicated homogeneous variances, treated and control groups were compared using a one-way analysis of variance (ANOVA). When one-way ANOVA was significant, a comparison of the treated groups to control by Dunnett test 33,34 for multiple comparisons was performed. Where variances were considered significantly different by Bartlett test, groups were compared using a nonparametric method (Kruskal-Wallis nonparametric ANOVA). 35 When nonparametric ANOVA was significant, comparison of treated groups to control was performed using Dunn test. 36 Statistical analysis was performed on all quantitative data for in-life and organ weight parameters using Provantis version 9, Tables and Statistics, Instem LSS (Staffordshire, UK).
Clinical pathology data were initially evaluated by Levene test 37 for HO and Shapiro-Wilk test 38 for normality. If the preliminary test was not significant, a nonparametric one-way ANOVA followed with Dunn test was performed, while in the case of significance, a parametric one-way ANOVA followed by Dunnett test. Transforms of the data were used to achieve normality and variance HO. The order of transforms attempted were log, square-root, and rank order. If the log and square root transforms failed, the rank order was used. Other statistical methods were used when appropriate. Significance was judged at a probability value of P <0.05. Mean and standard deviations were calculated for all quantitative data. Male and female rats were evaluated separately.
Results
Experimental Design
Animals were randomly assigned to each treatment group (10 animals/sex/group for main test and 5 animals/sex/group for recovery) before undergoing a 90-day dosing period. Groups 1 and 5 received a vehicle control of distilled water, groups 2 and 6 received 300 mg/kg/d NR-E, groups 3 and 7 received 500 mg/kg/d NR-E, and groups 4 and 8 received 1,200 mg/kg/d NR-E. Test substances were administered via oral gavage. Once the dosing period was complete, all main test animals (groups 1-4) underwent terminal necropsy, whereas the recovery animals (groups 5-8) were maintained for an additional 28-day observational period following cessation of test substance administration. An overview of the experimental design including NR-E nominal dose levels and concentrations is presented in Table 1.
Survival, Clinical Observations, Ophthalmological Examination
There was no mortality in the study, and no clinical observations were attributable to the test substance administration at any dosage level. An ophthalmology evaluation of all animals prior to study initiation and again near experimental completion on day 88 (for main test animals) or day 116 (for recovery animals) did not reveal ocular abnormalities attributable to exposure of test substance. Additionally, there were no test substance-related microscopic ocular findings.
Body Weights and Feed Consumption
All rats gained weight over the course of the study; however, treatment-related decreases in mean weekly body weights and daily body weight gain were observed in the high dose-treated (1,200 mg/kg/d, group 4) male rats (Figure 1). These findings only reached statistical significance (P < 0.05) on day 92 for body weights (approximately 13% reduction) when compared to control animals. Body weights were not significantly different at any other time point (even for group 8) or during the recovery period. Additionally, treatment-related decreases in food efficiency and food consumption were observed in the high dose-treated male rats, yet these findings did not reach statistical significance (Figure 2). Conversely, in the low dose-treated (300 mg/kg/d, groups 2 and 6) and middle dose-treated (500 mg/kg/d, groups 3 and 7) male rats, there were no treatment-related changes in mean weekly body weights, daily body weight gain, food consumption, or food efficiency (Figures 1 and 2). The observed decrease (P < 0.01) in mean daily food consumption in group 6 males on days 15 to 22 was considered to be incidental and not toxicological relevant. For female rats, there were no changes in mean weekly body weights, daily body weight gain, daily food consumption, or food efficiency that were attributed to the test substances (Figures 1 and 2).
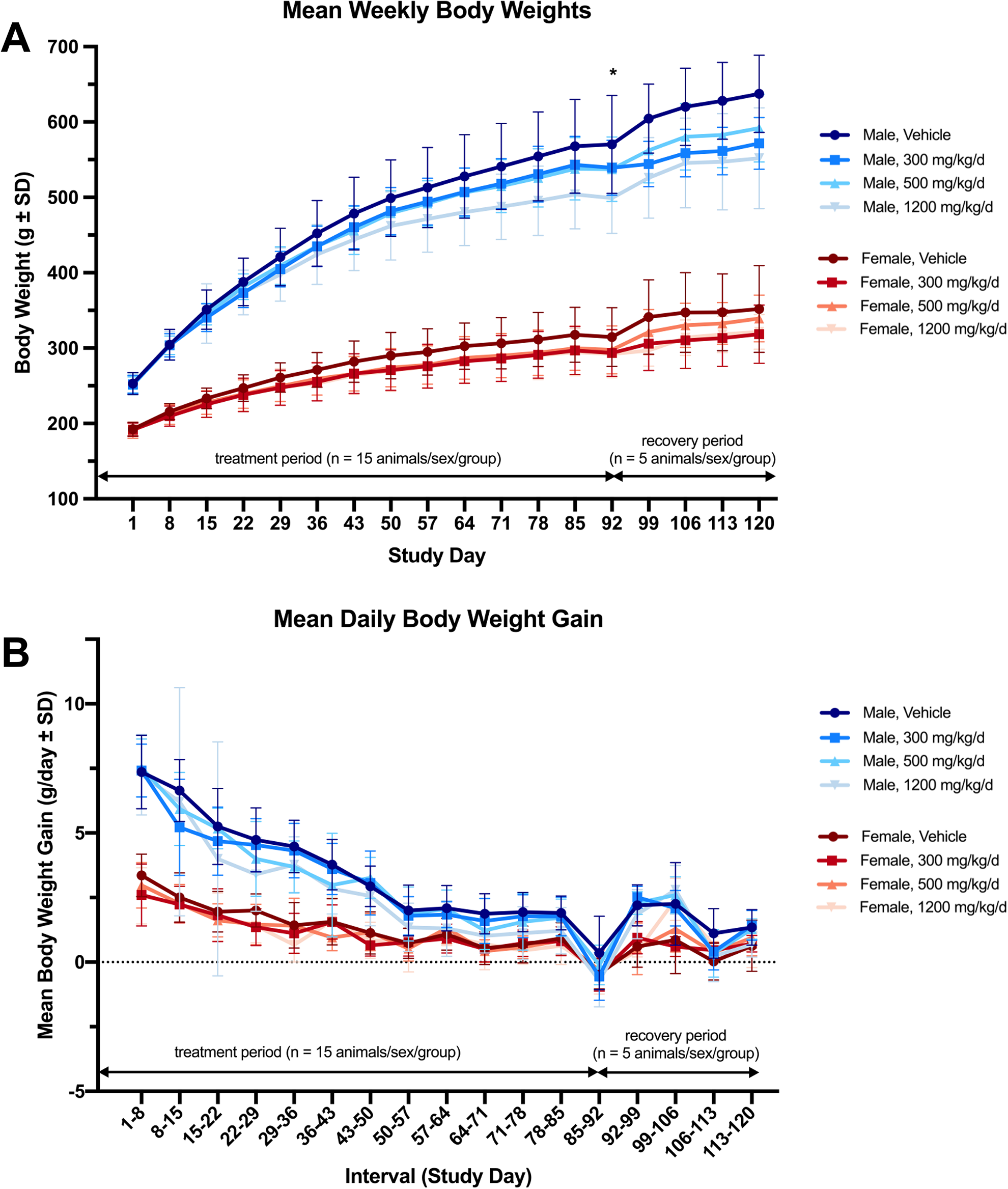
Mean weekly body weight and daily body weight gain of male and female rats orally administered vehicle control or NR-E at 300, 500, or 1,200 mg/kg/d for 90 days (n = 15 animals/sex/group; this includes recovery group) followed by a 28-day recovery (n = 5 animals/sex/group). A, Mean weekly body weights in the high dose-treated male rats trended toward a nonsignificant decrease throughout the study, which became statistically significant only on day 92. This finding was not statistically significant at any time point, including during the recovery. B, Mean daily body weight gain of all animals was comparable with the control throughout the study, although a slight but nonsignificant decrease was observed in the high dose-treated male rats (group 4) but not in other test substance-treated groups. (*P < 0.01). NR-E indicates nature-identical, synthetic nicotinamide riboside.
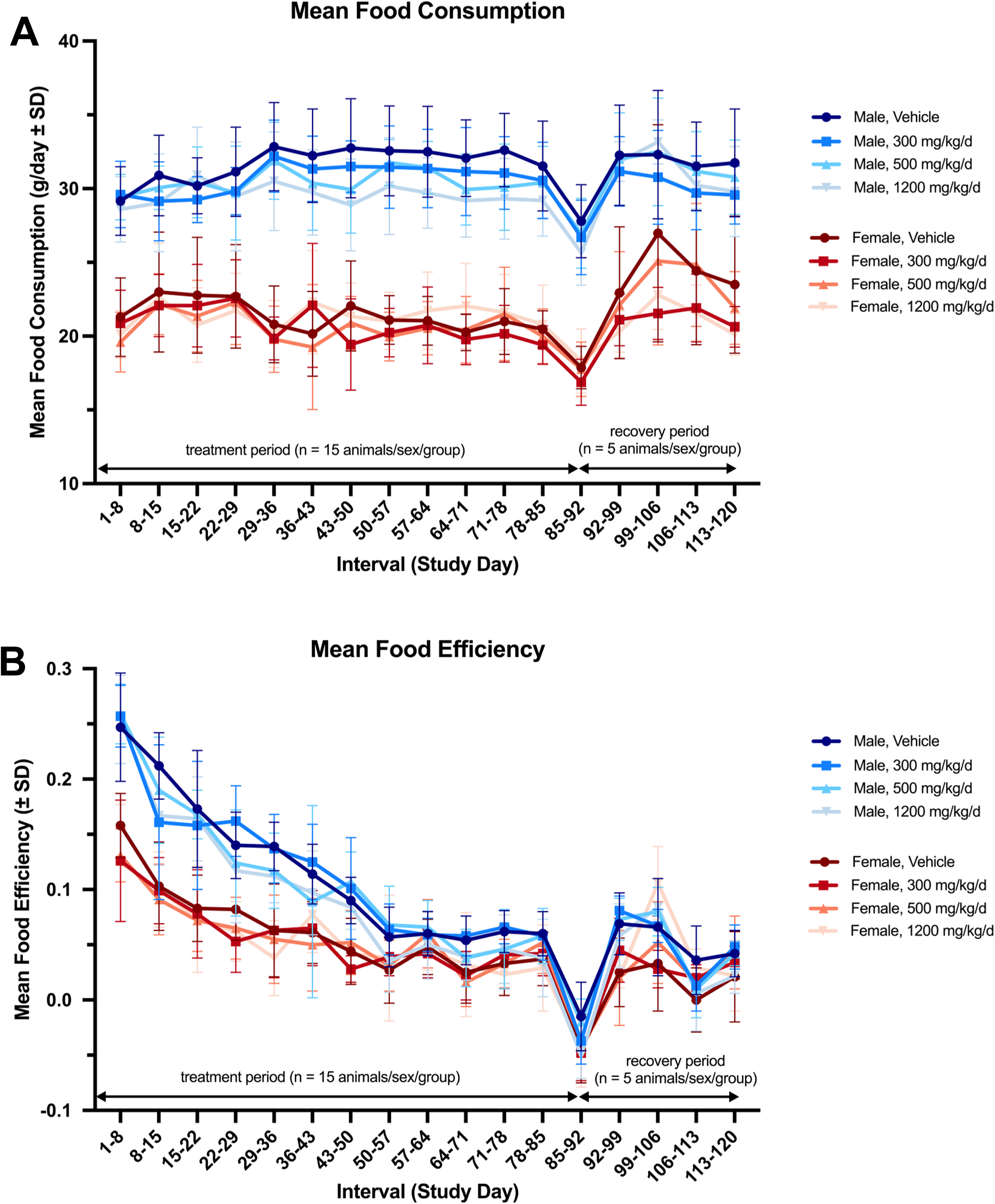
Mean daily food consumption (A) and food efficiency (B) for male and female rats administered 300, 500, and 1,200 mg/kg/d NR-E were generally comparable with the control group throughout the study; however, slight nonsignificant decreases in mean daily food consumption and food efficiency were noted in the high dose-treated males (group 4), but not in other test substance-treated groups. Treatment period n = 15 animals/sex/group (this includes the recovery group). NR-E indicates nature-identical, synthetic nicotinamide riboside.
Interestingly, all animals in groups 1 to 4 (including control animals) experienced a slight decrease in body weight at the final time point on day 92, which corresponded to a decrease in food consumption in all animals that was not significant. This was believed to be attributed to the overnight fasting in advance of blood collection for clinical pathology on day 88. Despite the slight decrease of the same magnitude observed across all animals of each sex, including control animals, this was unaccompanied by any relevant clinical signs and therefore was not considered toxicologically relevant.
Clinical Pathology: Hematology, Coagulation, Clinical Chemistry, and Urinalysis
There were no relevant test substance-related changes in hematology parameters for male and female rats of the main test groups on day 88 or the recovery groups on day 121 (Table 2). Mean corpuscular (cell) hemoglobin concentration was increased (P < 0.05) in group 4 males and groups 2 to 4 females but without an accompanying effect in other erythrocyte parameters and the absence of any other clinical or pathological correlates, this decrease was interpreted to have no toxicological relevance. White blood cell, ALYM, and AMON counts were decreased (P < 0.05) in group 8 males. However, similar changes were not observed in group 4 at the end of the treatment period and thus were considered to be incidental and independent from test substance administration. In group 3 females, increases in ANEU count and AMON count were statistically significant (P < 0.05) but observed in a nondose dependent manner—these were within PSL historical data base range and interpreted to be within expected biological variation and not toxicologically relevant.
Summary of Statistically Significant Main and Recovery Mean Hematology and Coagulation Values.a
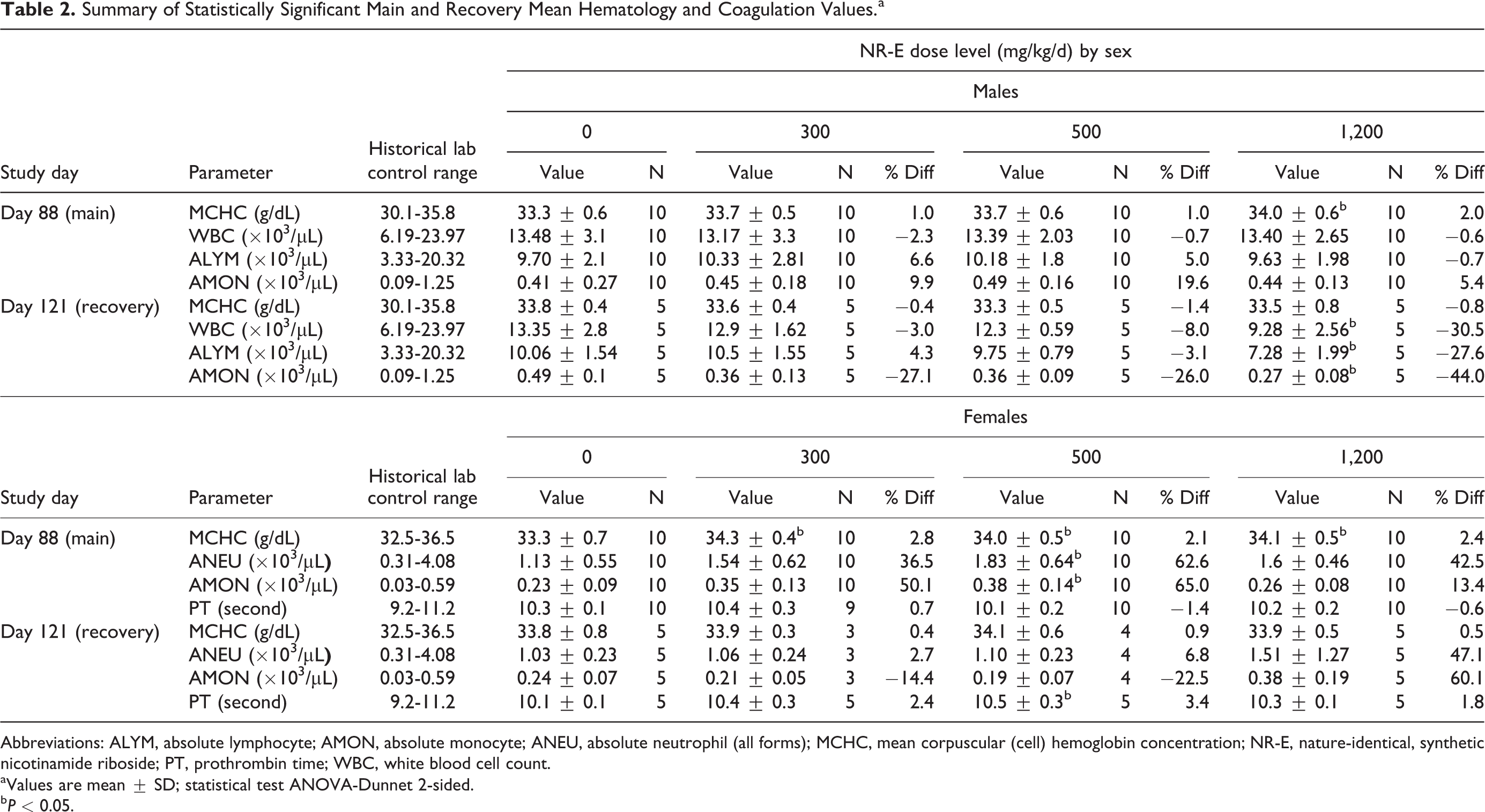
Abbreviations: ALYM, absolute lymphocyte; AMON, absolute monocyte; ANEU, absolute neutrophil (all forms); MCHC, mean corpuscular (cell) hemoglobin concentration; NR-E, nature-identical, synthetic nicotinamide riboside; PT, prothrombin time; WBC, white blood cell count.
a Values are mean ± SD; statistical test ANOVA-Dunnet 2-sided.
b P < 0.05.
There were no test substance-related changes in coagulation parameters for male and female rats of the main test groups on days 94/95 or the recovery groups on day 123 (Tables 2 and S1). A nondose dependent PT increase (P < 0.05) was observed in group 7 females. However, similar change was not observed at the end of the treatment period and thus was considered to be incidental and independent from test substance administration.
Small decreases in TRIGs (P < 0.05) were observed in groups 2, 4, 6, and 8 males. Sodium was decreased (P < 0.05) in groups 2, 3, and 4 males and group 4 females (Tables 3 and S2). Additionally, CL was decreased (P < 0.05) in group 4 males and females. Both changes in TRIGs and electrolytes were within PSL historical control range, had no histopathological or clinical observation correlated, and were interpreted to be nonadverse. A statistically significant decrease in CHOL was observed (P < 0.05) in groups 2 and 8 males, which was observed in a nondose dependent manner or time-dependent manner, within PSL historical data base range, and interpreted to be within expected biological variation and not toxicologically relevant. It should be noted that nicotinic acid has been shown to lower both TRIGs and CHOL in humans (although nicotinamide and NR not expected to have this effect) 39 and therefore this observation could be a real event but not of toxicological consequence.
Summary of Statistically Significant Main and Recovery Clinical Chemistry Values.a
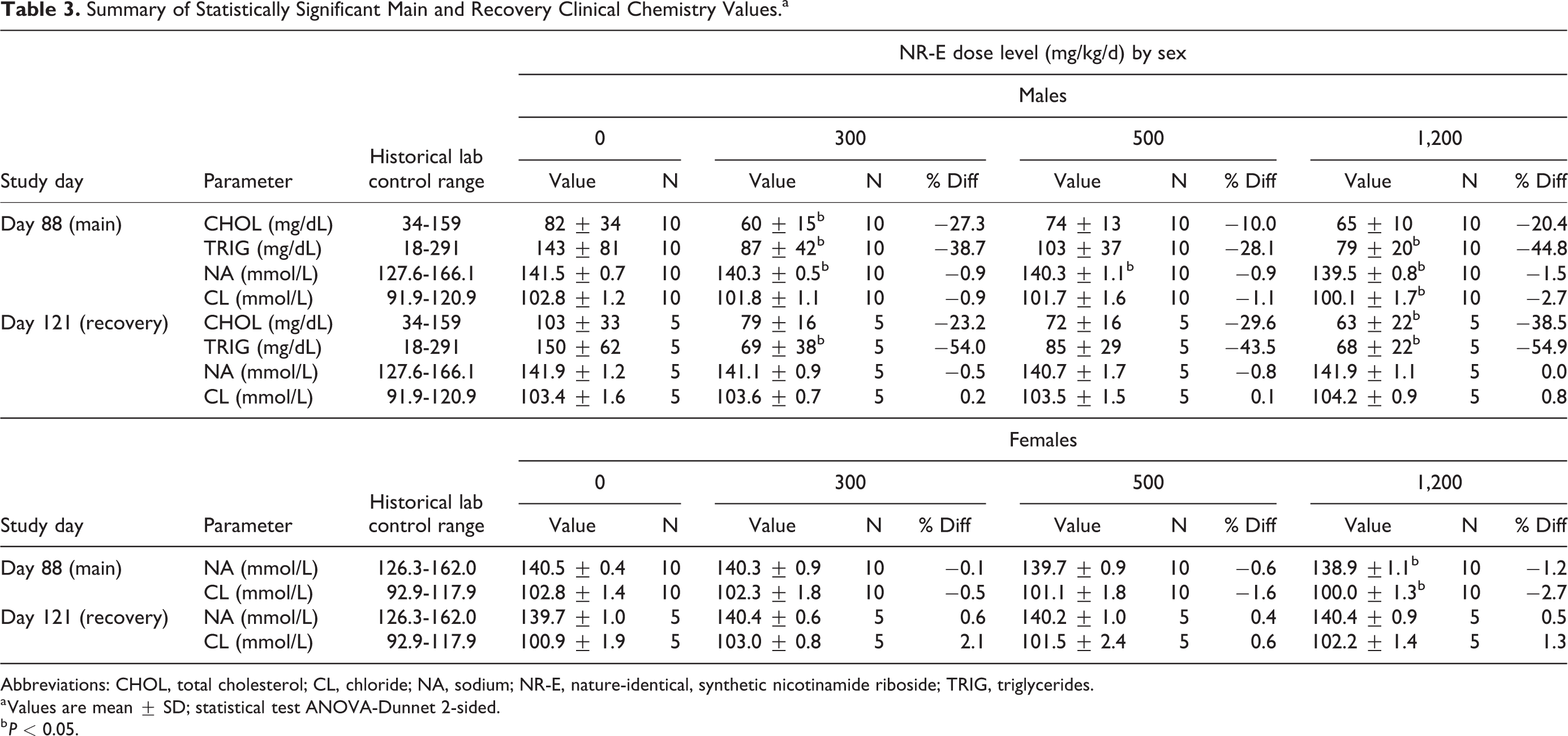
Abbreviations: CHOL, total cholesterol; CL, chloride; NA, sodium; NR-E, nature-identical, synthetic nicotinamide riboside; TRIG, triglycerides.
a Values are mean ± SD; statistical test ANOVA-Dunnet 2-sided.
b P < 0.05.
There were no test substance-related changes in urinalysis parameters on days 88 and 121 for male and female rats (data not shown). In group 6 males, urine volume decrease was statistically significant (P < 0.05); this was observed in a nondose dependent manner or time-dependent manner, within the lab historical data base range, and interpreted to be within expected biological variation and not toxicologically relevant.
Necropsy and Pathology
There was no mortality observed during the study, and there were no macroscopic or microscopic findings attributable to the test substance administration. Mean absolute organ weights of NR-E-treated animals were comparable to those of control animals throughout the study (Table 4).
Summary of Mean Terminal and Recovery Necropsy Body Weights and Organ Weights.a
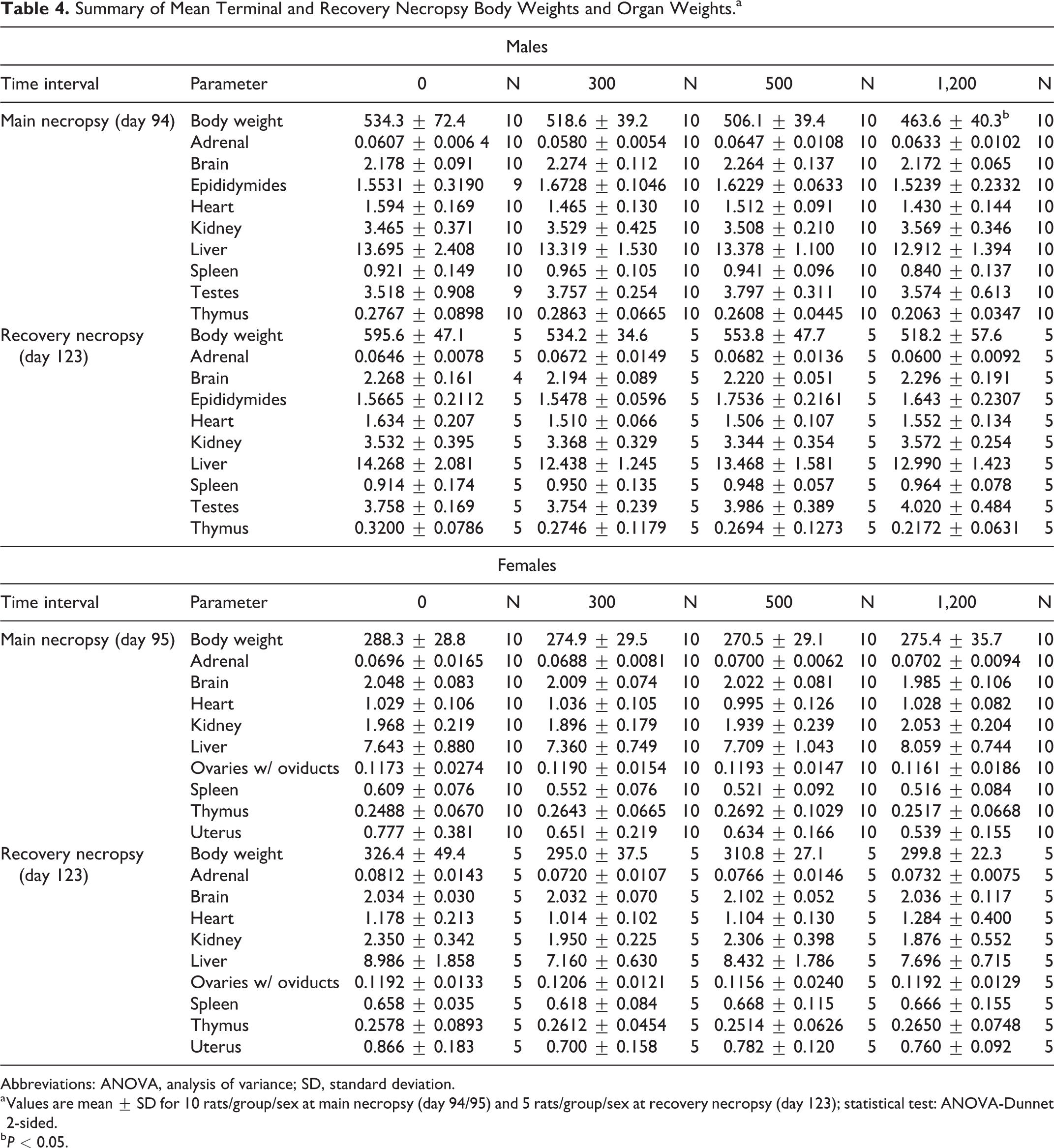
Abbreviations: ANOVA, analysis of variance; SD, standard deviation.
a Values are mean ± SD for 10 rats/group/sex at main necropsy (day 94/95) and 5 rats/group/sex at recovery necropsy (day 123); statistical test: ANOVA-Dunnet 2-sided.
b P < 0.05.
Statistically significant increases (P < 0.05-0.001) in mean adrenal, brain, kidneys, and liver-to-body weights were observed in the group 4 males, which were attributed to lower terminal body weight values (Table 5). In the recovery cohort, statistically significant increases (P < 0.05-0.01) in mean heart, kidneys, and testes-to-body weights were observed in the group 8 males and were also attributed to lower terminal body weight values and not directly attributable to a test substance administration effect. There was a statistically significant decrease (P < 0.05) in heart-to-brain weights in the group 2 males. However, as this was not dose-dependent and without associated histopathological findings, this was considered to be incidental and not toxicologically relevant (Table 6). In female rats, a statistically significant increase (P < 0.01) in liver-to-body weights was observed in the group 4 animals. As correlating changes in absolute liver weight, hepatic microscopic findings, or serum chemistry findings were not observed, this was interpreted to have little toxicological significance and to be nonadverse.
Summary of Mean Terminal and Recovery Necropsy Organ-to-Body Weight Ratios.a
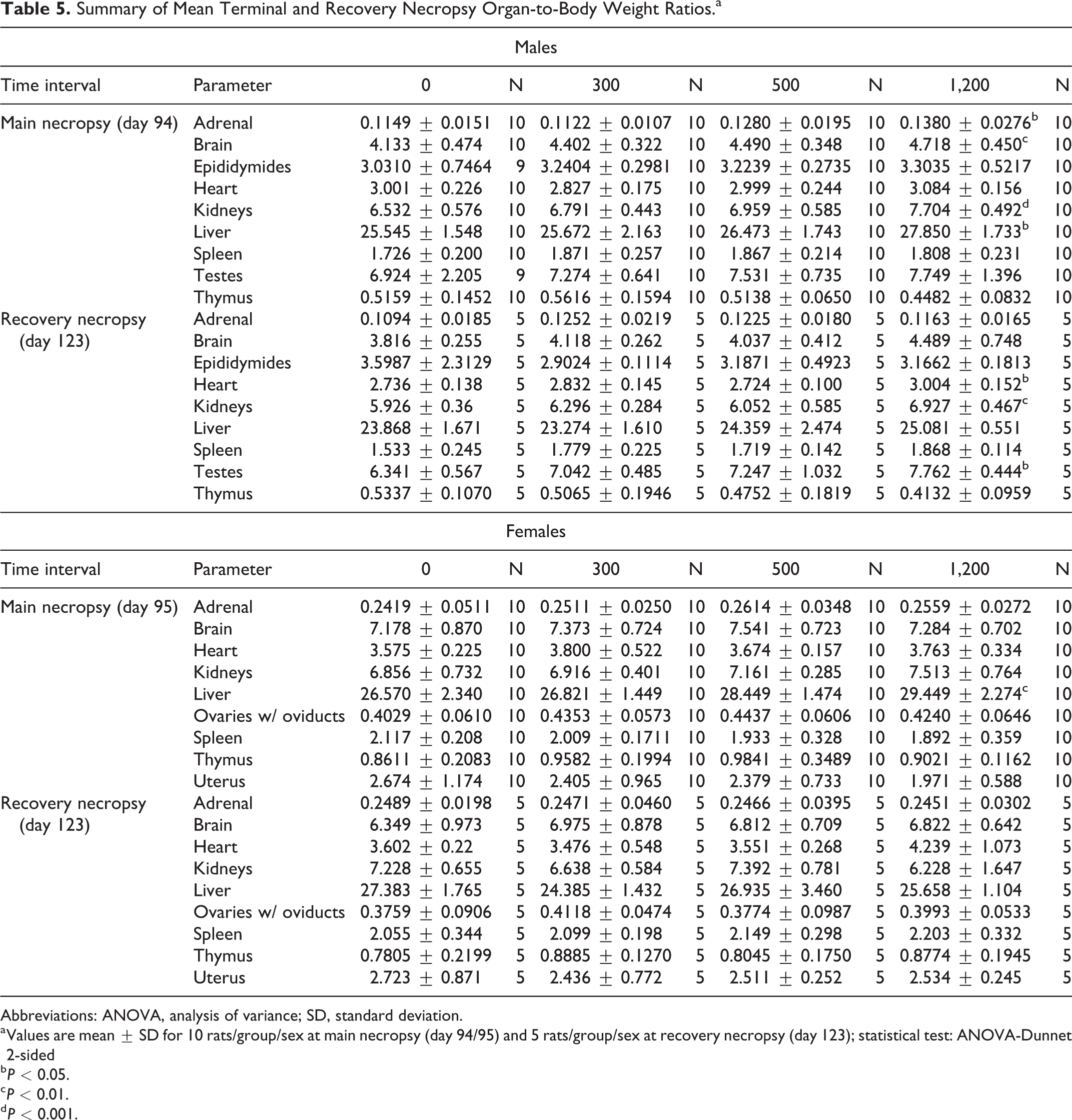
Abbreviations: ANOVA, analysis of variance; SD, standard deviation.
a Values are mean ± SD for 10 rats/group/sex at main necropsy (day 94/95) and 5 rats/group/sex at recovery necropsy (day 123); statistical test: ANOVA-Dunnet 2-sided
b P < 0.05.
c P < 0.01.
d P < 0.001.
Summary of Mean Terminal and Recovery Organ-to-Brain Ratios.a
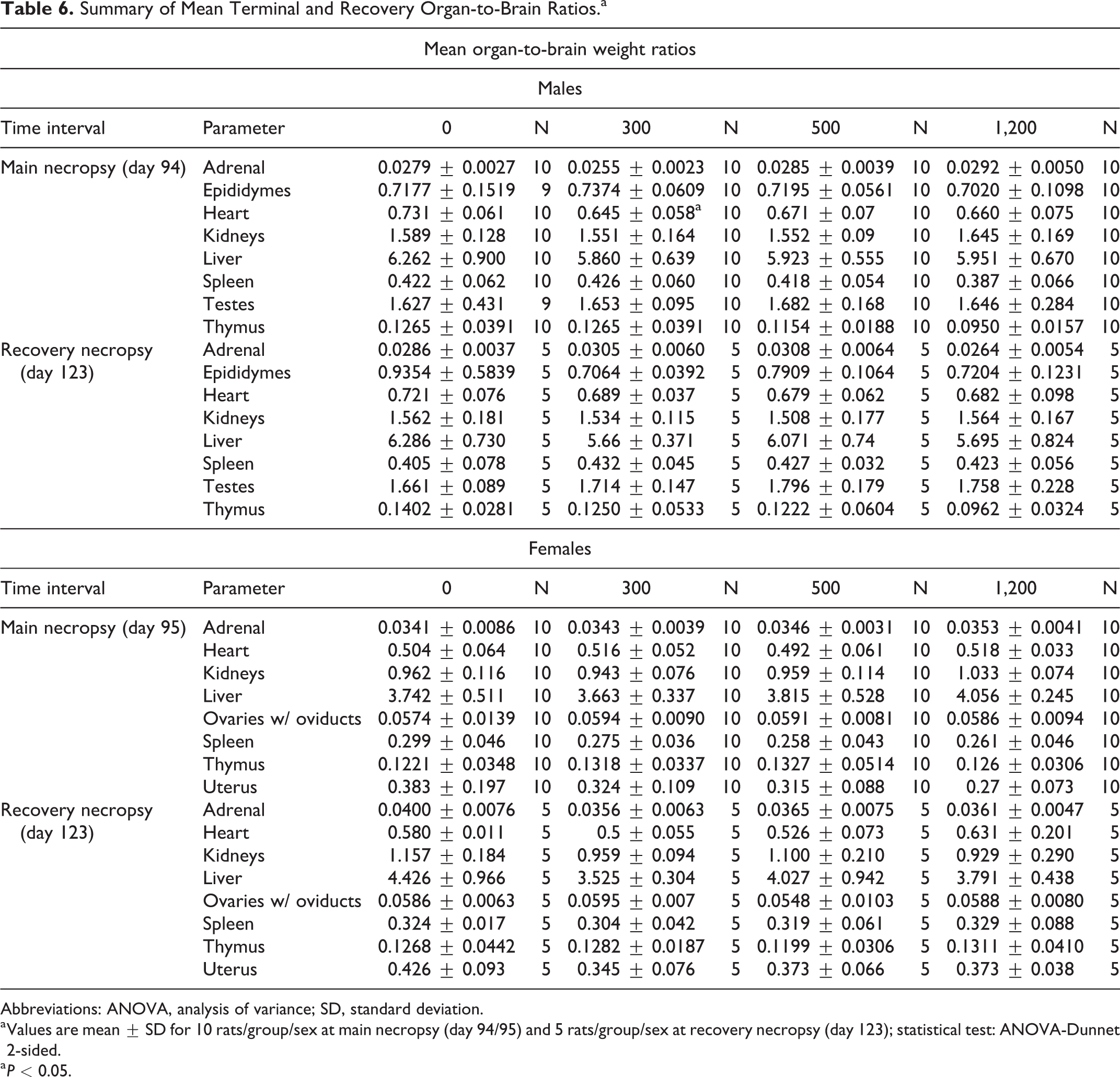
Abbreviations: ANOVA, analysis of variance; SD, standard deviation.
a Values are mean ± SD for 10 rats/group/sex at main necropsy (day 94/95) and 5 rats/group/sex at recovery necropsy (day 123); statistical test: ANOVA-Dunnet 2-sided.
a P < 0.05.
Discussion
NR is a form of vitamin B3 and precursor to NAD+. Results of this 90-day toxicity study demonstrated that oral administration of NR-E at dosages up to 500 mg/kg/d for male rats and 1,200 mg/kg/d for female rats was not associated with adverse events, as determined by the most conservative interpretation of the following toxicology end points: clinical observations, ophthalmologic examination, growth, feed consumption and efficiency, hematology, coagulation, clinical chemistry, urinalysis, organ weights, and histopathological findings. The UL in humans can be derived from the NOAELs of 500 and 1,200 mg/kg/d for males and females, respectively, by applying a standard 100-fold safety factor (10X for human variability and 10X for extrapolation from animals to humans), while assuming the average human weight of 60 kg. This thereby corresponds to an UL in humans of 300 mg/d for males and 720 mg/d for females.
No test substance-related clinical observations, ophthalmological examinations, hematology, coagulation, urinalysis, or macro- and microscopic histopathology findings were observed in males or females. Test substance-related decreases in mean weekly body weights, body weight gain, food consumption, and food efficiency were observed only in the high-dose 1,200 mg/kg/d males; however, statistical significance was only reached in body weights on day 92 (approximately 13% reduction). Body weights were not significantly different at any other time or during the recovery period; however, CHOL and TRIGs remained lower throughout the recovery phase at the highest dose. Despite the absence of additional corroborating evidence of toxicity in this study, the observed decrease in body weight and body weight gain was conservatively considered adverse in the high-dose 1,200 mg/kg/d males. Notably, there were no changes in these parameters, and in weekly body weights, observed in the female rats, suggesting a sex-specific effect that resulted in different NOAELs for males and females.
While the observed decrease in body weight and food consumption was the primary determinants of the NOAEL in male rats, it should be noted that decreased body weight did not correspond with any other clinical observations which would indicate concern of potential animal welfare. Furthermore, changes in body weight or composition have not been observed in any published human clinical trials with repeat-dose NR administered at dosages up to 2,000 mg/d. 1,4,5,26,28 Notably, in a double-blind, placebo-controlled clinical study using a different synthetic NR (branded as NIAGEN) by Conze et al, overweight but otherwise healthy adults were administered 1,000 mg/d of NIAGEN for 8 weeks and did not experience a significant change in weight. 26 In another placebo-controlled clinical study by Dollerup et al, 2,000 mg/d of NIAGEN for 12 weeks to obese but otherwise healthy men did not result in change of body composition. 28
The previously established NOAEL for NIAGEN, based on a 90-day toxicity study by Conze et al, was determined to be 300 mg/kg/d, 30 which corresponds to a human UL of 180 mg/d. The Conze study established the first UL for NR (180 mg/d), which is lower than the accepted UL for nicotinamide 40,41 and demonstrated that NR and nicotinamide have similar toxicological profiles. In this present study, the observed NOAEL of NR-E was found to be higher: 500 mg/kg/d for males and 1,200 mg/kg/d for females, corresponding to a human UL of 300 mg/d and 720 mg/d, respectively. While it must be acknowledged that a recovery period was not included in Conze’s study design and animals were dosed at a wider dosage range (300, 1,000, and 3,000 mg/kg/d), treatment-related adverse effects in the liver, kidney, testes, epididymis, and ovaries were observed that were not present in this current study. These included observations associated with hepatic toxicity (including increases in ALT, ALP, and GGT) that were accompanied by an increase in liver weight, centrilobular hepatocellular hypertrophy, and single cell necrosis. In contrast, no significant changes in ALT or AST or microscopic hepatic findings were observed in any groups in either sex in the present study. Conze et al also reported significant test substance-related increases in TRIGs in the 1,000 and 3,000 mg/kg/d females; however, the present study showed slight decreases in TRIGs in males and no change in females. Importantly, Conze et al observed that NIAGEN significantly elevated ALT levels and increased TRIG levels in females at the 1,000 mg/kg/d dose. 30 In contrast, the current study shows no hepatic toxicity or increase in TRIGs at 1,200 mg/kg/d of NR-E (Table 3). Thus, the treatment-related adverse events observed by Conze et al that were not observed in the present study could be attributed to differences in the manufacturing process and impurity profile of NIAGEN in comparison to the 21 CFR 111/210 NR-E used in this study.
In the present study, a sex-dependent difference in NOAEL was observed; a finding that was attributed primarily to the test-substance related decreases in mean weekly body weights and daily body weight gain observed in the high dose-treated male animals receiving 1,200 mg/kg/d. Significant dose-dependent decreases in body weights were similarly observed in the Conze study in male rats. 30 While the differences between NAD+ metabolism and utilization of NR between sexes have not been fully understood, studies have suggested potential sex-related differences exist in rodents and humans in regard to the NAD+ metabolome and utilization of NAD+. 17,42 -44 Notably, in the present study, a lack of treatment-related changes in body weight or weight gain in female rats resulted in a sex-specific effect and differing NOAELs between males and females. Further research is warranted to explore potential sex differences.
In summary, a conservative interpretation of the results of this present subchronic toxicity study suggests that oral administration of NR-E at levels up to 500 mg/kg/d in male and 1,200 mg/kg/d in female Sprague-Dawley rats for 90 days did not produce any adverse events under the conditions of the present study. Given the large safety factor between the determined NOAELs and the corresponding human UL, it is concluded from a critical evaluation of the present safety assessment and of previous toxicological information, these results are supportive of a UL of 300 mg/d for males and 720 mg/d for females.
Supplemental Material
Supplemental_Tables - Safety Assessment of High-Purity, Synthetic Nicotinamide Riboside (NR-E) in a 90-Day Repeated Dose Oral Toxicity Study, With a 28-Day Recovery Arm
Supplemental_Tables for Safety Assessment of High-Purity, Synthetic Nicotinamide Riboside (NR-E) in a 90-Day Repeated Dose Oral Toxicity Study, With a 28-Day Recovery Arm by Anca G. Marinescu, Jayson Chen, Holly E. Holmes, Leonard Guarente, Odete Mendes, Mark Morris and Ryan W. Dellinger in International Journal of Toxicology
Footnotes
Author Contribution
Marinescu contributed to analysis and interpretation, drafted manuscript, and critically revised the manuscript; Chen contributed to conception and design; contributed to acquisition, analysis, and interpretation; and critically revised the manuscript; Holmes contributed to interpretation, drafted the manuscript, and critically revised the manuscript. Mendes contributed to conception and design; contributed to acquisition, analysis, and interpretation; and critically revised the manuscript. Guarente contributed to conception and design and critically revised the manuscript. Morris contributed to conception and design and critically revised the manuscript. Dellinger contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that Marinescu, Holmes, Guarente, Morris and Dellinger recieve salary support and stock options from Elysium Health, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
