Abstract
A safety evaluation was performed for EpiCor, a product produced by a proprietary fermentation process using Saccharomyces cerevisiae. Studies included the following assays: bacterial reverse mutation, mouse lymphoma cell mutagenicity, mitogenicity assay in human peripheral lymphocytes, and a cytochrome P450 ([CYP] CYP1A2 and CYP3A4) induction assessment as well as 14-day acute, 90-day subchronic, and 1-year chronic oral toxicity studies in rats. No evidence of genotoxicity or mitogenicity was seen in any of the in vitro or in vivo studies. The CYP assessment showed no interactions or inductions. No toxic clinical symptoms or histopathological lesions were observed in the acute, subchronic, or chronic oral toxicity studies in the rat. Results of the studies performed indicate that EpiCor does not possess genotoxic activity and has a low order of toxicity that is well tolerated when administered orally. The no observable adverse effect level (NOAEL) was 1500 mg/kg body weight (bw)/d for the 90-day study and 800 mg/kg bw/d for the 1 year study, for the highest doses tested.
Keywords
Introduction
EpiCor is a fermentate powder produced using a nongenetically engineered strain of Saccharomyces cerevisiae that undergoes anaerobic fermentation of a proprietary medium under conditions of stress. The wet fermentate is dehydrated and milled into a powder and contains inactive whole yeast cells including cell wall components, components from the medium, together with the fermentative byproducts and stress-induced metabolites. The food grade dried fermentate is intended for use as an ingredient in single- and multi-ingredient dietary supplements and as an ingredient in food to contribute nutritional benefits or technical functions of enhancing flavor and antioxidant properties.
Saccharomyces cerevisiae, also known as “brewer’s yeast” or “baker’s yeast,” is a species of budding yeast used for millennia by humans as a tool for producing food and beverage products. Baker’s yeast extract itself is regarded as “generally recognized as safe” (GRAS) as a direct food additive (21 CFR §184.1983). While EpiCor is produced utilizing S. cerevisiae fermentation, the production involves both a unique substrate and a stress process; hence, out of diligence, the ingredient, which is intended for human consumption, was placed through a battery of safety studies. The objective of the study was to determine the broad safety profile of EpiCor using accepted toxicology models. For the composition and characterization of EpiCor, supplemental Tables 1 to 5 are provided. EpiCor contains 13.7% phenolics, based on liquid chromatography/mass spectrometry (LC/MS) structural analysis for specific phenolics. The primary phenolic monomers were determined to be catechin and epicatechin, in addition to various phenolic polymers. A ribosomal DNA (rDNA) fungal identification sequencing kit using analysis software (MicorSeq D2 LSU; Applied Biosystems, Foster City, California) determined the test article to be a 100% genetic match to the species S. cerevisiae.
Summary of Hematology Findings in Chronic 1-Year Study a
Abbreviations: RBC, red blood cells; WBC, white blood cells; Hb, hemoglobin; Hct, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; PLT, platelet; Ly, lymphocytes; Ne, neutrophils; Eo, Eosinophils; Mo, Monocytes; Ba, Basophils; Pt, prothrombin time.
a Data represent the mean and the standard deviation
b P < 0.05.
EpiCor has been shown to have a high antioxidant content and immunomodulatory properties; it has a significant peroxyl radical scavenging capacity in vitro based on the oxygen radical absorbance capacity (ORAC) assay, an ability to enter and protect live human cells from oxidative damage from hydrogen peroxide, to activate human natural killer cells and B cells in vitro, and to have an immunomodulatory effect on polymorphonuclear cells. 1,2 In human studies, the ingredient has been shown to increase salivary IgA and hematocrit levels and is associated with support of the immune system with regard to seasonal allergies and the incidence and duration of colds and flu. 3 –5
Because these studies suggest an effect on the immune system, a lymphocyte proliferation assay was performed to evaluate its ability to function as a mitogen in terms of direct activation of immune cells. To evaluate mutagenic effects, a bacterial reverse mutation assay and in vitro mammalian cell gene mutation test in mouse lymphoma L5178Y cells were performed. In order to further determine the safety of EpiCor, the following short-term and repeat exposure, acute, subchronic, and chronic, oral toxicity studies were performed in the rat. Finally, because of the potential and growing concern for food–drug interactions with regard to natural products, 6 –8 a preliminary in vitro cytochrome P450 (CYP) induction study was performed using immortalized human hepatocytes. Cultured human hepatocytes provide needed insight in evaluating substances or new molecular entities as inducers of CYP enzymes. The results of these experiments are reported herein.
Materials and Methods
Test Article
The test article is EpiCor powder (Embria Health Sciences, Ankeny, Iowa). All assays and animal studies were conducted in accordance with good laboratory practice (GLP) regulations. The animal studies were conducted in a GLP laboratory certified by the Hungarian Ministry of Health as well as Hungarian Ministry of Agriculture and Regional Development. The lymphocyte proliferation assay and the CYP1A2 and CYP3A4 inducer assay in Fa2N-4 cells were performed in the United States and conducted in accordance with the US Food and Drug Administration (FDA) compliance guidelines.
Ames Test (Bacterial Reverse Mutation Assay)
The test substance, EpiCor, was evaluated in a bacterial reverse mutation assay in accordance with the Organisation for Economic Co-operation and Development (OECD) 471 guidelines employing both Salmonella typhimurium strains TA97a, TA98, TA100, and TA1535 (obtained from Bruce Ames, PhD, Berkeley, California) along with Escherichia coli strain WP2 urvA (pKM101+; National Collection of Industrial and Marine Bacteria, Aberdeen, UK) in the presence and absence of an exogenous metabolic activation system, at concentrations of 5, 10, 50, 100, 500, 1000, 2500, and 5000 μg per plate with S. typhimurium strains TA98 and TA100 and 1, 5, 10, 50, 100, 500, 1000, and 2500 μg per plate for S. typhimurium strains TA97a and TA1535 as well as E. coli strain WP2 uvrA (328). In the confirmatory preincubation assay, concentrations of 10, 50, 100, 500, and 1000 μg per plate were assessed in all strains. Sterile water served as the negative control. The criterion for classifying a substance as mutagenic was an increase in the mean number of revertants that was at least 2 times greater than the mean in the negative control (or 3 times greater for strain TA1535). 9
Mammalian Cell Gene Mutation Study in Mouse Lymphoma L5178Y Cells
The mammalian cell gene mutation test was conducted in accordance with the OECD 476 guidelines. The mouse lymphoma L5178Y cell line was obtained from the European Collection of Cell Cultures, Health Protection Agency (Wiltshire, UK) and determined free of mycoplasma (Mycoplasma Laboratory, Copenhagen, Denmark). Methyl methanesulphonate and cyclophosphamide (Sigma Chemical, St Louis, Missouri) were used as the positive controls at a final concentration of 5 and 0.2 µg/mL, respectively, for a 3-hour treatment (and 0.1 µg/mL for a 24-hour treatment). Estimation was determined by percentage of relative total growth (RTG%), cell survival or cytotoxicity (cloning efficiency [CE]), and mutant frequencies (MFs). Distribution of colony forming units over the wells is assumed to follow Poisson distribution.
Mitogenicity Study Using Human Peripheral Blood Lymphocytes
The lymphocyte proliferation assay was performed in a US FDA compliant laboratory and conducted in accordance with GLP, using a standard staining kit (Sigma-Aldrich, St Louis, Missouri). Freshly purified human peripheral blood lymphocytes were suspended in a diluent allowing for permanent incorporation of the lipophilic fluorescent dye PKH26 into the lymphocyte membranes. The reaction was stopped by addition of serum and by extensive washing. The lymphocytes were resuspended in appropriate culture medium at a density of 1 million per milliliter, and triplicate cultures were established in round-bottom tissue culture 96-well plates. Untreated cells served as the negative control and the known mitogen phytohemagglutinin (Sigma-Aldrich; 20µμg/mL) served as the positive control to induce cell division. Sets of triplicate wells were treated with serial dilutions of EpiCor prepared as follows: an amount of 0.5 g of EpiCor was added to 5 mL phosphate-buffered saline and incubated for 1 hour at room temperature. Solids were removed by centrifugation. The supernatant was diluted in cell culture medium by mixing 1 part extract to 9 parts cell culture medium and incubated for 1 hour at 37°C, after which any precipitate was removed by filtration. After treatment, the lymphocyte cultures were incubated at 37°C in 5% CO2 for 5 days. The lymphocyte cultures were then washed and the fluorescence intensity was evaluated by flow cytometry (FacsCalibur, Becton-Dickinson, San Jose, California), allowing for assessment of the relative distribution of cells in parent versus daughter populations for each culture. Cells that underwent mitosis displayed reduced membrane fluorescence as a result of the membrane staining being distributed between daughter cells. Data from each identical triplicate set of culture conditions were averaged and the mean and standard deviations were calculated.
Evaluation as CYP1A2 and CYP3A4 Inducer in Fa2N-4 Cells
This study evaluated the induction of human CYP1A2 and CYP34A in immortalized human hepatocytes (Fa2N-4 cells; XenoTech, Lenexa, Kansas). 10,11 Immortalized human Fa2N-4 hepatocytes were obtained from MultiCell Technologies (Lincoln, Rhode Island). The test article, EpiCor, was solubilized in dimethyl sulfoxide (DMSO) at 33.7 mg/mL. Fa2N-4 cells were plated on collagen-coated plates and maintained at 37°C in an atmosphere of 95% air/5% CO2 and humidity of 95%. Following a 3-day adaptation period, the cells were treated with 8 concentrations of EpiCor (33.70-0.01685 μg/mL) for 3 consecutive days to determine the level of toxicity. Lactate dehydrogenase (LDH) release was measured at 24 hours after each treatment using the Cytotoxicity Detection Kit (Roche Diagnostics, Indianapolis, Indiana). Cells were examined microscopically for signs of toxicity. The highest nontoxic concentration of the test article was serially diluted from 1.0× to 0.5× to 0.25× and used to determine enzyme induction; the Fa2N-4 cells were treated with 33.70, 16.85, and 8.43 μg/mL of EpiCor for 3 consecutive days. The CYP1A2 and CYP3A4 messenger RNA (mRNA) levels and enzymatic activity were determined 24 hours after the last treatment. The CYP mRNA levels were determined using a branched DNA (bDNA) assay and normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) mRNA. 12 The bDNA probes were manufactured by XenoTech. Reagents were purchased from Genospectra (Fremont, California). Fa2N-4 cells treated with omeprazole or rifampin served as positive controls for CYP1A2 and CYP3A4 induction, respectively. The CYP marker–substrate reactions of phenacetin O-dealkylation (CYP1A2) and midazolam 1′-hydroxylation (CYP3A4) were measured using LC/MS-MS after a 24-hour treatment of the cells with EpiCor, DMSO (negative control), or omeprazole or rifampin (positive controls).
Animal Studies
Each animal toxicology study utilized specific-pathogen-free (SPF) Sprague-Dawley rats (Charles River Laboratories, Budapest, Hungary) and was approved by the Animal Experimentation Committee of the laboratory. The studies were performed in accordance and conformity with the following protocols: acute (with 14 day postobservation), OECD 423; subchronic (90-day), OECD 408; and chronic (1 year), OECDs 408 and 452.
Test Material
EpiCor powder was weighed and suspended in 1% methylcellulose containing distilled water not earlier than 30 minutes before administration. Concentration and homogeneity of the test article were checked by gravimetry.
Acute Oral Toxicity Study With 14-Day Posttreatment Observation Period (Limit Test)
Twenty Sprague-Dawley rats (10 males and 10 females), aged 6 to 7 weeks, weighing 151.0 to 163.1 g (females) and 201.2 to 210 g (males) were used for the study. Housing conditions were maintained at a temperature of 22°C ± 3°C, relative humidity of 30% to 70%, air exchange of 15 times per hour, and a 12-hour light–dark cycle. Rats were given free access to tap water and a standardized rat and mouse diet S8106-SO11 SM R/M-Z+H (Ssniff Spezialdiäten, Soest, Germany). Animals were acclimatized and observed for 5 days prior to treatment. A single dose of 2000 mg/kg body weight (bw) was administered orally by gavage. Clinical observation of the rats was carried out once before exposure, then after the treatment for 6 hours continuously, and during the subsequent period in which animals were checked twice daily (at least once daily on weekends) for mortality, general state, external appearance, behavior, and clinical symptoms. Body weight was measured on days 1, 2, 8, and 15. Euthanasia was performed on day 15 using intravenous administration of 0.1 mL/100 g bw of T61 (Intervet International, Budapest, Hungary), which contains 0.2 g embutramide, 0.005 g tetracaine hydrochloride, and 0.05 g mebezonium iodide per mL.
Subchronic (90-Day) Oral Toxicity Study in Rats
One hundred sixty SPF Sprague-Dawley rats (80 females and 80 males), 4 to 5 weeks of age at arrival, weighing 101.3 to 124.5 g (females) and 101.4 to 124.9 g (males), were used in the study and 2 of the same sex were housed in a cage. Housing conditions and diet were as described in the acute toxicity study. Animals were observed for at least 5 days prior to initiation of treatment. EpiCor was prepared as described once daily by gavage over 90 consecutive days at doses of 30, 200, and 1500 mg/kg bw. The dosages were based on proposed human use levels: the oral dose of 500 mg once daily (approximately 7 mg/kg, calculated as 70 kg bw in an adult). The highest dose 1500 mg/kg corresponds to about 200-fold of the 7 mg/kg daily dose if consumed by an adult, in addition to a previous acute toxicity study and dose range finding preliminary experiment. The lower dose of 30.0 mg/kg is still more than 4-fold the adult dose. The intermediate dose is approximately the geometrical mean of the high and the lower dose. The applied volume was adjusted weekly in accordance with the changes in the animals’ body weight. Doses were adjusted for weight on a weekly basis, as well as food and water, the latter of which was performed in the microbiology department.
Animals were checked for mortality, general state, external appearance, behavior, and clinical signs twice daily at the beginning and end of the workday on weekdays and once on weekend days until the morning of the 91st day. They were weighed on the first day of treatment (day 1) and weekly thereafter. Food consumption was measured weekly and the daily average food consumption was calculated. Water consumption was measured once a week for 24 hours, and microbiological testing was performed. Direct opthalmological examination was performed on the first 5 males and first 5 females of each dose group prior to the treatment period and all high-dose and control animals once during week 11 of treatment by a veterinary ophthalmologist using a Welch Allyn Pan Optic Ophthalmoscope (Jungingen, Germany). Sensory reactivity to auditory, visual, and proprioceptive stimuli, assessment of grip strength, and motor activity was conducted on all animals during week 11, according to routine methods. 13
After the last treatment, rats were fasted overnight then anesthetized and blood samples were examined for hematological and clinical chemistry parameters. Urinalysis was carried out once during week 12 on 10 males and 10 females. Necropsy occurred on the 91st day after a 16-hour fast. Organ weights were recorded and histopathological examinations were performed.
Chronic (1-Year) Oral Toxicity Study
One hundred sixty (80 females and 80 males) 4- to 5-week-old SPF Sprague-Dawley rats, weighing 101.0 to 112.8 g (females) and 126.0 to 149.3 g (males), were housed 1 per cage and observed for at least 5 days prior to initiation of treatment. Dosages applied in this study were selected on the basis of results obtained in previous acute toxicity and 90-day subchronic studies, where it was well tolerated in daily doses up to 1500 mg/kg for 90 days. The safety factor of 100 to 1 for food additives used in applying animal experimentation data to man was taken into consideration, that is, a food additive for use by man will not be granted a tolerance that will exceed 1/100th of the maximum amount demonstrated to be without harm to experimental animals. The highest dose chosen, 800 mg/kg, corresponds to 100-fold of the approximate 7.1 mg/kg daily dose, for a planned supplement oral dose of 500 mg once daily, if consumed by an adult or 32-fold of the 25 mg/kg daily dose when calculated for a child's body weight (ie, 20 kg). The lower dose of 20 mg/kg is still 2.8-fold of the adult's dose. The intermediate dose is 2.8-fold of the 7.1 mg/kg daily dose if consumed by an adult. Hence, application of the test article was by gavage at doses of 20, 200, and 800 mg/kg bw once daily over 364 consecutive days.
EpiCor was prepared as previously described. Animals were weighed upon arrival to the laboratory, on the day of randomization, on day 1 (first treatment), once weekly during the treatment period, and on the day of necropsy. Food consumption was measured weekly and the daily average food consumption was calculated. The applied volume was adjusted weekly in accordance with the changes in the animals’ body weight. Water consumption was measured once a week for 24 hours and microbiologically tested.
Animals were checked for mortality, general state, external appearance, behavior, and clinical signs twice daily at the beginning and end of the workday on weekdays and once on weekend days until the morning of the final day of the study. Should any grossly visible or palpable tumor appear, time of onset, location, dimensions, appearance, and progression, are recorded. Direct ophthalmologic examination was conducted on all animals prior to the treatment period and on all high-dose and control animals at week 51 by a veterinary ophthalmologist using a Welch Allyn Pan Optic Ophthalmoscope (Jungingen, Germany). Sensory reactivity to auditory, visual, and proprioceptive stimuli, assessment of grip strength, and motor activity were conducted on all animals during weeks 15, 27, 40, and 52 according to methods described by Irwin. 13 Blood samples were obtained twice during the study at weeks 14 and 33 from 10 male and 10 female rats per group and 1 day after the last treatment before necropsy. Rats were fasted overnight, anesthetized, and blood samples were examined for hematological and clinical chemistry parameters. Urinalysis was carried out during weeks 13, 32, and 51 on 10 males and 10 females. Because live bacteria were seen in the sediment of 2 rats from the 800 mg/kg group during week 32, sediment analysis was repeated on week 36, and sediments from the 2 rats were sent to an outside laboratory for parasitology and bacteriology examinations. Necropsy occurred on day 365 after fasting for 16 hours. Organ weights were recorded and histopathological examinations were performed.
Statistical Analysis
Depending on the study, statistical evaluations were performed by Statistica Version 5.5 (StatSoft, Edition 99, Tulsa, Oklahoma). Calculations included mean values, standard deviations, comparison of variances, 1-way analysis of variance (ANOVA), Tukey test, and/or Kruskal-Wallis nonparametric 1-way ANOVA.
Results
Ames Test (Bacterial Reverse Mutation Assay)
In the bacterial reverse mutation assay, the test material was not observed to be toxic at any concentration level in the confirmatory preincubation assay for any of the strains tested. EpiCor was concluded to be nonmutagenic in this assay.
Mammalian Cell Gene Mutation Test in Mouse Lymphoma L5178Y Cells
For the preliminary toxicity test, the %RTG from the highest concentration of the test material (5000 µg/mL) was 63.25 and 19.86 over the negative control, in the presence and absence of metabolic activation, respectively, for the 3-hour exposure. The %RTG was 22.57 for the 24-hour exposure. In the definitive mutation assay, at the highest concentration tested, the reduction in the %RTG of the cells was in the range of 22.01% to 66.59% for the experiments in the presence and absence of metabolic activation of the first trial and was in the range of 21.61% to 61.11% for the negative control in the presence and absence of metabolic activation in the confirmatory experiments. Mutation frequencies were not found to be increased compared to negative control in the presence and absence of metabolic activation under the tested concentrations. The positive controls produced statistically significant increases in the MF. EpiCor was not observed to cause gene mutation in this test system.
Mitogenicity Study Using Human Peripheral Blood Lymphocytes
No differences in cell division levels between untreated cells and cells treated with serial dilutions of the test material were observed. In contrast, the lymphocytes treated with the positive control (phytohemagglutinin), showed substantial proliferation, reflected in a significant loss of membrane fluorescence. EpiCor was not considered to be mitogenic under the conditions of this assay.
Evaluation as a CYP1A2 and CYP3A4 Inducer in Fa2N-4 Cells
Three days of exposure of Fa2N-4 cells to the test article did not induce CYP1A2 or CYP3A4 mRNA or enzymatic activity. The test material did not interfere with the induction of CYP1A2 and CYP3A4 by omeprazole and rifampin, respectively (Figures 1 and 2).
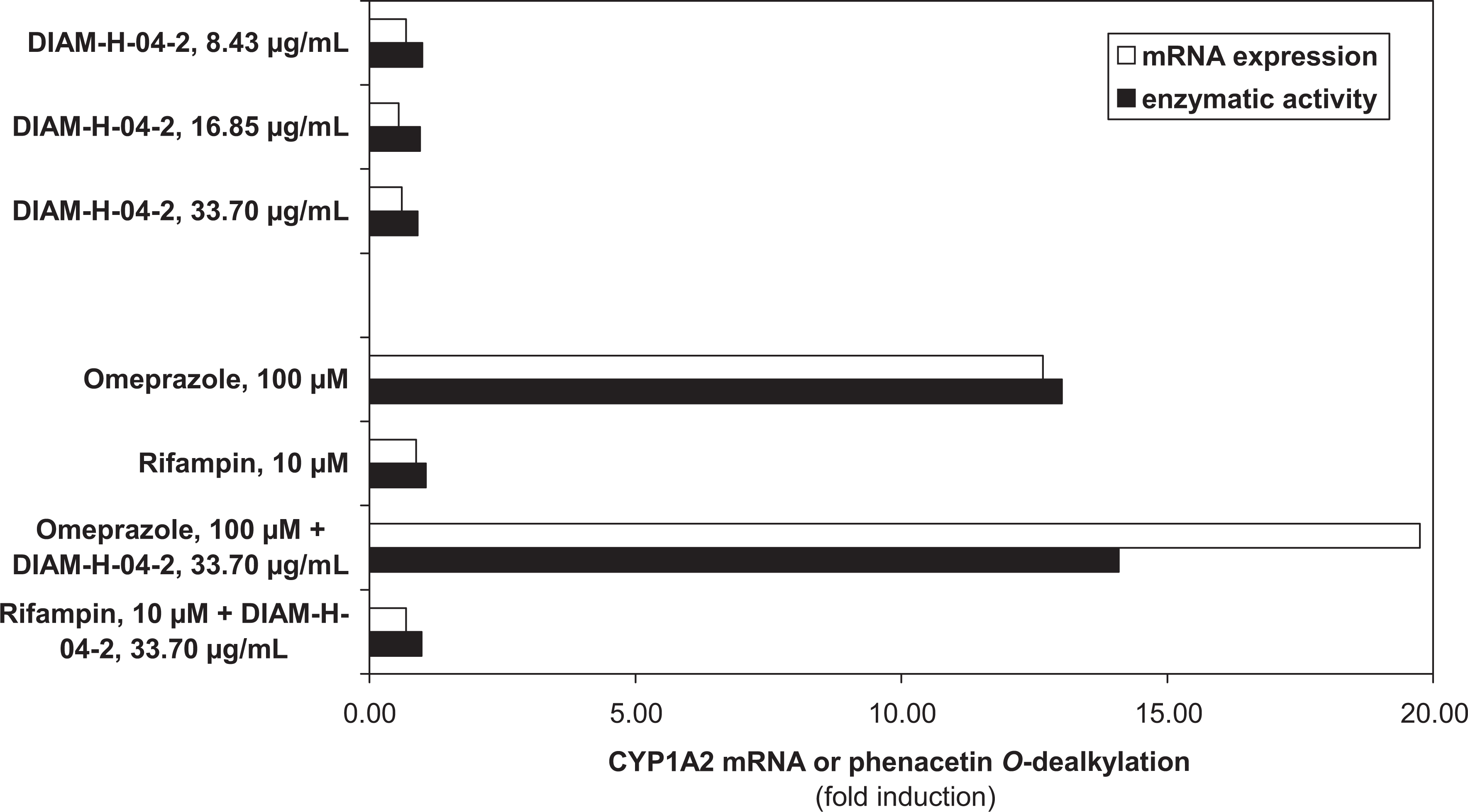
The effect of EpiCor (DIAM-H-04-2) on CYP1A2 messenger RNA expression and phenacetin O-dealkylase activity in Fa2N-4 cells.
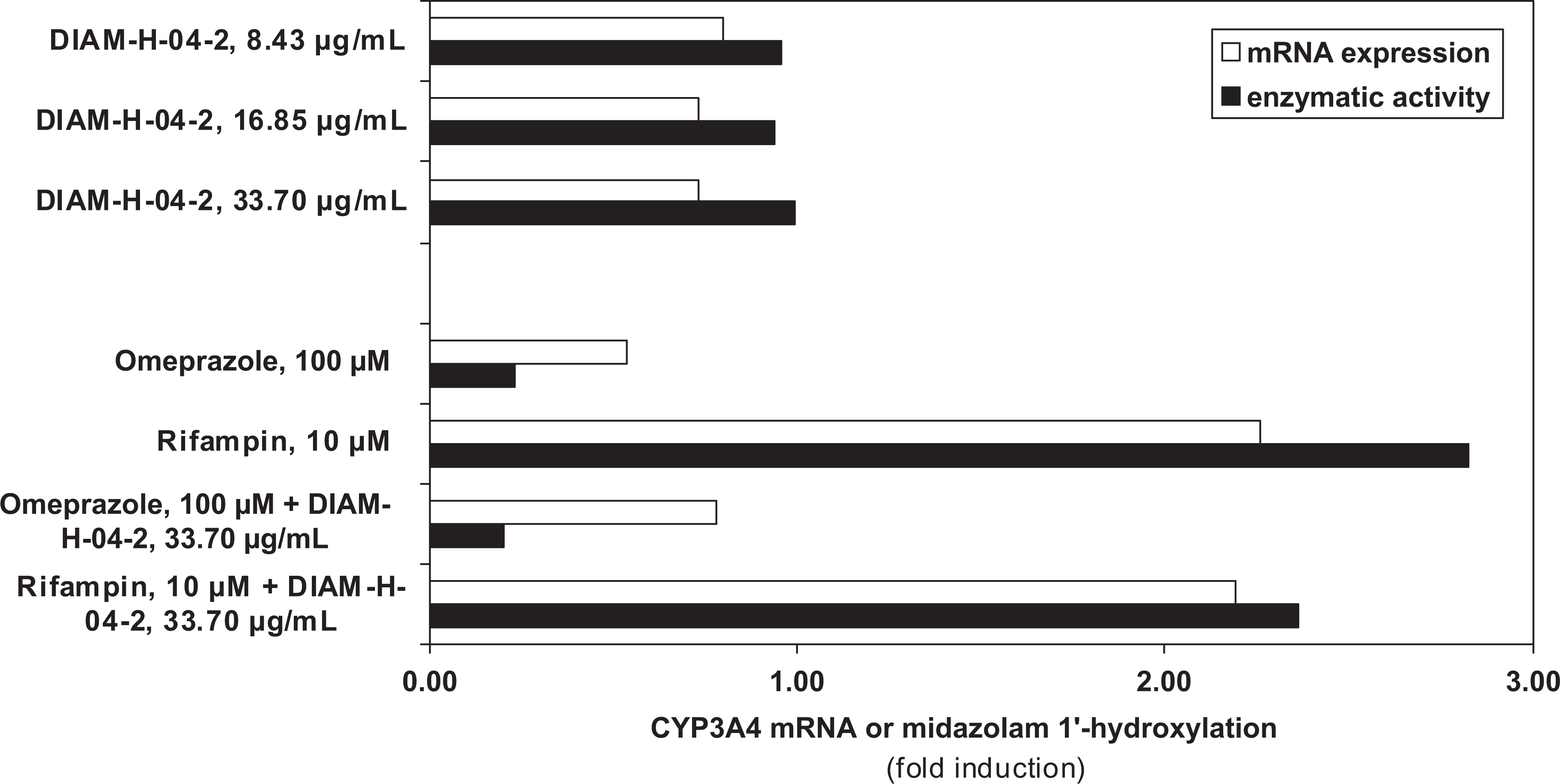
The effect of EpiCor (DIAM-H-04-2) on CYP3A4 messenger RNA expression and midazolam 1′-hydroxylase activity in Fa2N-4 cells.
EpiCor was found to be nontoxic to Fa2N-4 cells at any of the concentrations examined in this assay.
Acute Oral Toxicity Study With 14-Day Posttreatment Observation Period in the Rat
All 20 rats survived the single oral administration of 2000 mg/kg day of the test material and through the 14-day observation period. There was no difference in body weights compared to the control throughout the study period. Body weight of the animals corresponded to their species and age throughout the study. No body weight loss was observed. Food consumption was similar to the control. No clinical toxicological symptoms were seen. No gross pathological changes were found in the organs.
Subchronic (90-Day) Oral Toxicity Study in Rats
No deaths occurred and no treatment related changes in general state, external appearance, or behavior were observed in 160 Sprague-Dawley rats, in the 30, 200, and 1500 mg/kg male and female groups. Furless areas without signs of inflammation were noted in 3 rats (1 from the 200 mg/kg males group and 2 from the 1500 mg/kg female group). However, these symptoms have been observed sporadically in other studies and are not considered treatment related. There were no significant differences in body weight or body weight gain among groups treated with EpiCor when compared to the control group at any time point during the experimental period. All EpiCor treated male and female groups consumed similar amounts of food and water compared to the corresponding control groups throughout the study. No ophthalmologic changes were observed in the control or 1500 mg/kg groups. No treatment related differences in the visual (finger approach) or auditory (startle) reactivity, pain perception (tall pinch), grip strength, or motor activity was observed at examination of male and female rats during week 11 of treatment.
No compound related effects were observed in red blood cells (RBCs), white blood cells (WBCs), hemoglobin (Hb), hematocrit (Hct), and platelet count values. No alterations in blood coagulation were seen via prothrombin time measurements. No clinical chemistry data showed any treatment related toxic alterations at the end of the 90-day lasting treatment. Individual values and group mean values were within the physiological ranges. Dose-related slight decease of total cholesterol level was measured in the male rats that reached the statistically significant level in the high-dose group (group 4 M) only. Total cholesterol levels of the female group were similar to that of the controls.
There were no differences in the volume, specific gravity, or pH of the discharged urine among control and test vehicle treated groups, nor for glucose, blood, or protein.
Gross necropsy examination revealed no compound-related lesions. The few sporadically developed pathological changes were determined unrelated to treatment. Internal examination showed that subcutaneous tissue, regional lymph nodes, fatty tissue, skeletal muscles, joints, and bone system were normal in all animals. No differences in the organ weights or organ weights related to body weights were noted, and no treatment related histopathological findings were observed.
The test material was well tolerated in daily doses up to 1500 mg/kg bw/d for the duration of the 90-day study. Based on these results, the no observable adverse effect level (NOAEL) for EpiCor is considered to be 1500 mg/kg bw/d.
Chronic (1-Year) Oral Toxicity Study in Rats
No spontaneous deaths occurred in any of the 160 animals during the 1-year study, in any group. One male and one female rat, both from 800 mg/kg dose groups, were found in tumor-provoked moribund states (the male had a mass on the skin of the left foreleg, and the female had 3 nodules in the inguinal region) and were consequently sacrificed. One female rat was accidentally over-anaesthetized during scheduled blood sampling at week 33. On gross examination, males and females from both control and treatment groups displayed varying degrees of insignificant fur loss without visible changes of epidermis or signs of injury or inflammation. Two female rats (200 and 800 mg/kg group) displayed moderate ataxy resulting from pituitary adenomas. An ulcerated area on the right hind leg of 1 female rat in the 20 mg/kg was histopathologically described as an inflamed lipoma.
The development of the animals during the experimental period corresponded appropriately to their species and age; body weight gains in treated and control groups of both genders were similar throughout the study. Statistically significant body weight loss at week 39 in the 200 mg/kg male group was attributed to low within-group differences, and statistically significant weight gain at week 44 in the male 20 mg/kg group and at weeks 10, 14, 29, and 52 in the female 800 mg/kg group were considered to be of no biological significance. Prolonged body weight losses occurred in 3 rats from different groups; 2 were attributed to pituitary adenomas (200 and 800 mg/kg females) and 1 to diabetes (control male). Food consumption was similar between all treatment groups and control groups.
Statistically significant decreased water consumption occurred in the 800 mg/kg group. The male 800 mg/kg group displayed a statistically significant decrease in water consumption on 24 nonconsecutive individual weeks. The female 800 mg/kg group displayed statistically significant decreased water consumption only during the first 10 weeks of the study. No dose-dependent correlation could be established between decreased water consumption and any findings of clinical or histological relevance related to the test substance.
No ophthalmologic changes were observed prior to treatment or at week 51 in the control or the treatment groups, except in the 800 mg/kg group, where 1 male kept its head posture in right rotation from week 35 until the end of the study, determined related to handling during blood sampling, resulting in corneal injury, and 1 male was found with congestion of conjunctival blood vessels noted on the left eyelid, considered incidental. Otherwise, no compound-related differences in visual (finger approach) or auditory (startle) reactivity, pain perception (tail pinch), grip strength, or motor activity were noted.
On blood examination, no compound-related effects were observed in hematology, blood coagulation, or blood chemistry at weeks 14, 33, and at the end of the study. Statistically significant alterations in hematology and blood chemistry can be found in Tables 1 and 2. Results were considered nondose-dependant or were within historical control ranges and/or were considered not of clinical significance; the results were unrelated to other findings.
Summary of Chemistry Findings in Chronic Study a
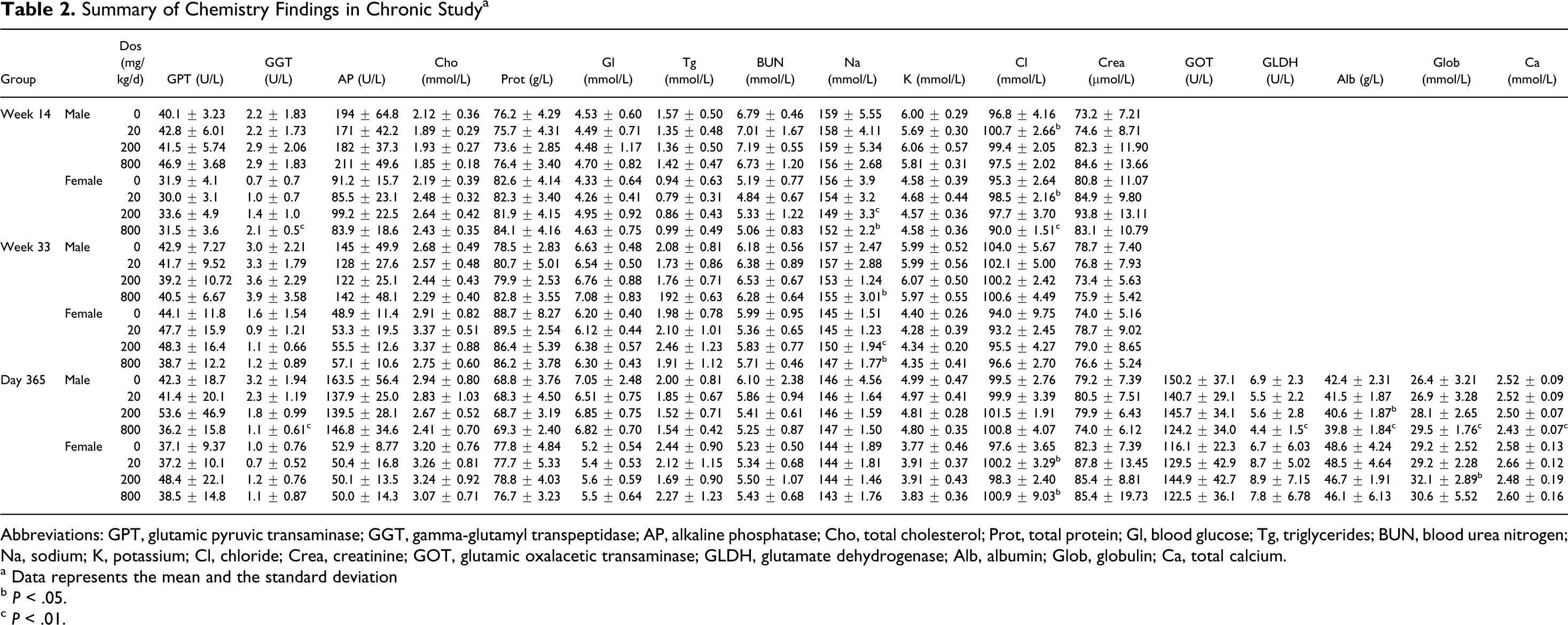
Abbreviations: GPT, glutamic pyruvic transaminase; GGT, gamma-glutamyl transpeptidase; AP, alkaline phosphatase; Cho, total cholesterol; Prot, total protein; Gl, blood glucose; Tg, triglycerides; BUN, blood urea nitrogen; Na, sodium; K, potassium; Cl, chloride; Crea, creatinine; GOT, glutamic oxalacetic transaminase; GLDH, glutamate dehydrogenase; Alb, albumin; Glob, globulin; Ca, total calcium.
a Data represents the mean and the standard deviation
b P < .05.
c P < .01.
On urinalysis during weeks 13, 32, and 51, no treatment-related differences were noted. Several sporadic statistically significant differences were noted in the urine samples of several groups. A low amount of total protein was present at a low incidence in the 20 and 800 mg/kg groups on week 32, and protein discharge was present in all dose groups at week 51. During week 51, the pH values of the 20 and 200 mg/kg females were lower compared to the control group. Low incidence of blood, crystals, and epithelial cells were noted, but were not consistent at any time point in the study. Live bacteria were noted in the urinary sediment from 2 symptom-free male rats from the 800 mg/kg group in week 32. The sediment analysis was repeated 1 month later, where live bacteria were again noted. These bacteria were identified as nonpathogenic bacteria (Enterobacter spp., Klebsiella spp., and Citrobacter freundii) generally present in the intestinal and urinary tract of rats. As the viable bacteria were facultative pathogens and the animals were free of clinical symptoms, the animals were left in the study. The sediments analyzed in week 51 were clear of viable bacteria.
Internal macroscopic examinations found no treatment-related pathological changes in subcutaneous tissues, regional lymph nodes, fatty tissues, skeletal muscles, joints, or bones in any animal. Sporadically developed pathological changes that were determined to be unrelated to treatment are summarized in Table 3 and include 1- and 2-sided testicular atrophy in low incidence, fawn-colored, and nutmeg-patterned livers in males and females of both control and treatment groups (with higher incidence in the control groups), adrenal gland cystic degeneration in female control and treated groups, age-related thymus atrophy in control and treated groups, occurrence of hydrometria, tumor-like changes in the pituitary and skin in low incidence, and tumor-like tissue changes in mammary glands of 15% to 20% of the 200 and 800 mg/kg female treatment groups. The mammary tumor rates were not significant and fell within the historical control incidences of 6.3% to 32%. Table 4 shows histopathological findings that are commonly considered incidental and spontaneous in nature. 14 Only benign tumors were found in the animals, which were confirmed histopathologically. Findings were not statistically significant and included 2 and 3 mammary gland adenoma in the 200 and 800 mg/kg female groups, respectively, a single mammary gland fibroma in each of the same female groups and pituitary adenomas in both treatment and control groups, along with 7 nondose treatment-related benign skin lesions. While the incidence of pituitary adenomas appeared dose-dependent in nature, the occurrences were not statistically significant, and were within historical control ranges (internal data).No treatment-related differences in organ weights or in differences in the bodyweight– to–organ weight ratios were found and no treatment-related histopathological changes were seen.
Summary of Gross Pathological Findings in Chronic Study
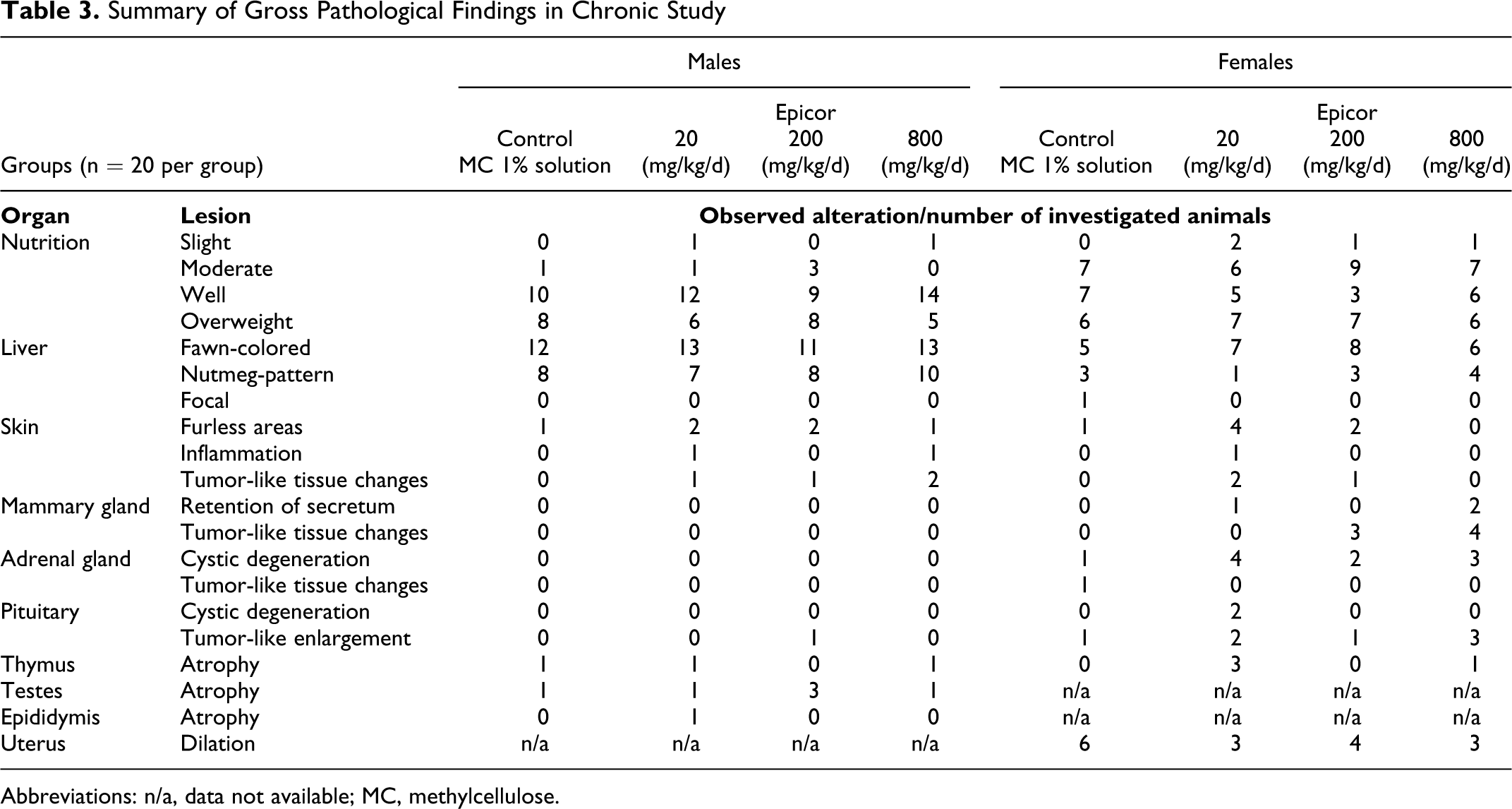
Abbreviations: n/a, data not available; MC, methylcellulose.
Summary of Histopathological Findings in Chronic 1-Year Study
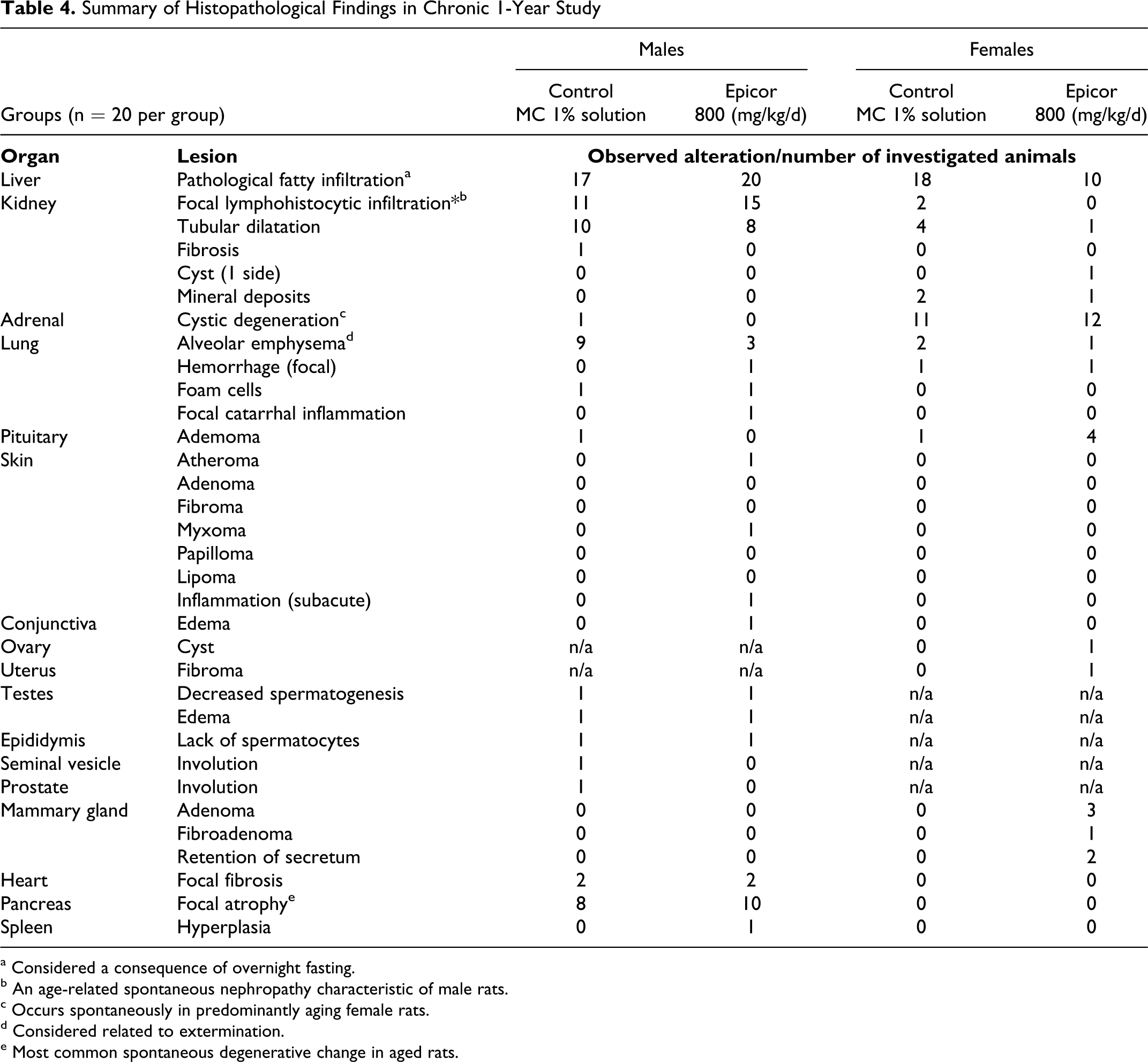
a Considered a consequence of overnight fasting.
b An age-related spontaneous nephropathy characteristic of male rats.
c Occurs spontaneously in predominantly aging female rats.
d Considered related to extermination.
e Most common spontaneous degenerative change in aged rats.
Discussion
EpiCor is a nutrient rich, high-metabolite product of fermentation that has been shown in studies to have significant antioxidant capacity, with desirable immune modulatory and anti-inflammatory effects in healthy adults based on randomized clinical trials. 3 –5,15,16
Some natural products that are considered antioxidant in nature such as quercetin have displayed mutagenic activity in vitro and are thought to have some pro-oxidant activity. 17 However, the test material showed no signs of mutagenicity in the Ames test or the mammalian cell gene mutation study.
The ingredient also did not induce or interfere with induction of 2 major CYP enzymes, CYP1A2 and CYP3A4, involved in drug metabolism in vitro. The test material did not interfere with the induction of CYP1A2 and CYP3A4 by omeprazole and rifampin (Figures 1 and 2). CYP1A2 is a member of the P450 oxidase system involved in metabolism of xenobiotics. It catalyzes numerous reactions involved in drug metabolism. Based on the in vitro evidence reported, EpiCor is not a substrate, inducer, or inhibitor of CYP1A2, as is seen with such methylxanthine stimulants as caffeine or theophylline or weak food inducers such as broccoli, Brussels sprouts, or barbecued meats. Food–drug interaction is a growing area of study gaining attention given the increased number of consumers simultaneously taking both natural products and pharmaceutical medications. 18 Since the ingredient did not induce or interfere with induction of the P450 enzyme, CYP3A4, this is of particular significance as it has a role in the metabolism of approximately 50% of all small molecule drugs prescribed today, 19 as this enzyme can lead to adverse drug–drug interactions when concomitant drug therapies require metabolic clearance by the same enzyme. 20 Certain natural products and organisms have been shown to be mitogenic, 21 –24 resulting in direct or indirect stimulation of cells causing them to divide and enter a highly activated state, thus raise a concern as to safety and/or immune overreactivity due. EpiCor was not found to be directly mitogenic in the lymphocyte proliferation assay. Given the lack of statistically significant differences and the magnitude of the changes in any of the assays seen as too small, the test material is considered to have no potential impact in terms of potential genotoxicity.
The dose applied in the acute study corresponds to 285-fold of the daily suggested dose of 500 mg by a human adult of 70 kg. EpiCor was well tolerated in the acute study with a 14-day observation follow-up after a single oral dose of 2000 mg/kg bw/d.
Sporadic dilatation of the lumen of the uterus with fluid is termed hydrometra and was observed in various groups in the subacute study, connected to the ovarian hormonal cycle and is a common finding in the Sprague-Dawley rat. Because no other pathological disturbances were seen in the affected females, and the total number of hydrometra bearing animals was not higher than in similar studies (15%-26%), the incidence of hydrometra seen in this study is considered to be of no significance.
The non-neoplastic histopathological findings observed in the 1-year oral toxicity study was spontaneous or incidental and typical of those encountered in control Sprague-Dawley rats. The low incidence of benign tumors in the female rats was not statistically significant and falls within the historical control range for this rat strain (6.3%-32% for mammary-gland adenomas and 26.0%-92.86% for pituitary adenomas). These benign neoplastic changes are well recognized to occur spontaneously in Sprague-Dawley rats. 14,25 Due to the lack of statistical significance and incidences falling within the historical control group ranges, the benign tumors in the treatment group were considered unrelated to treatment with the test vehicle. Based on the results of the 90-day study with histopathologies, the NOAEL for EpiCor is considered to be 1500 mg/kg bw/d.
In the chronic 1-year study, no macroscopic or microscopic evaluations, serum chemistry, hematological, urinary parameters, or histology results were determined to be of clinical significance. Overall, the test article was well tolerated in daily oral doses of up to 800 mg/kg, except for a statistically significant decrease in water consumption over nonconsecutive weeks in the highest dose group without clinical relevance. The observed fawn-colored and nutmeg-patterned livers in males and females of both control and treatment groups were considered to be unrelated to treatment, since they occurred in both the groups, with a higher incidence seen in the controls. Although there were some statistically significant hematological findings, results were not dose-dependent or were within historical control ranges and therefore not considered clinically significant. Hence, given the overall results in the 1-year study, it is felt reasonable that the NOAEL of EpiCor is 800 mg/kg bw/d.
The data obtained from the studies presented are significant in determining the safety of EpiCor when used in food and dietary supplements for human consumption based on the experimental models used for safety assessment. A human controlled clinical trial with an acute single dose of EpiCor showed a benefit to the immune system without adverse effects, 26 confirming previous observations that chronic exposure to the ingredient provides some protection against infectious agents. 27 Favorable outcomes associated with human controlled clinical trials continue to show benefits to immune function without adverse effects. 2,26 These benefits suggest that EpiCor may find increased exposure as an ingredient in food and/or dietary supplements, making it much more essential that experimental studies assessing safety, such as reported by the authors, continue to be conducted. No significant changes were seen suggestive of an effect on immune function, for example, hematology, in the in vivo studies. Further, specific immune markers were not measured in the rat studies. In future investigations of the efficacy of EpiCor in humans, safety panels should be included to gather further data on its safety.
Footnotes
Acknowledgments
The authors thank Drs Lijie Fu, Kristen Totaro, Maciej Czerwinski, and S. Shivaram, and (the late) Istvan Financsek for their contributions. Embria Health Sciences, LLC, provided the test article.
The author(s) declared no conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Embria Health Sciences, LLC, provided financial support.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
