Abstract
Toxicity is one of the major reasons for failure in drug development. Zebrafish, as an ideal vertebrate model, could also be used to evaluate drug toxicity. In this study, we aimed to show the predictability and highlight novel findings of toxicity in zebrafish model. Seven anticancer compounds, including triptolide (TP), gambogic acid (GA), mycophenolic acid (MPA), curcumin, auranofin, thalidomide, and taxol, were assessed in zebrafish for their toxicity. Three compounds (GA, TP, and taxol) showed highest acute lethality, with 50% lethal concentration ≈ 1 μmol/L. Missing tails, severe pericardial edema, and enlarged yolk sacs were observed in MPA-treated embryos. The development of pectoral fins was severely disturbed in thalidomide-, GA-, and TP-treated embryos. Bradycardia was observed in MPA- and thalidomide-treated groups. Our findings suggested that the zebrafish are a good model for toxicity assessment of anticancer compounds.
Introduction
As a vital concern and a major challenge for modern drug discovery, toxicity is the leading cause of drug development failure. In the past 40 years, 2.9% of approved drugs were delisted from the market because of a variety of serious adverse drug reactions, even death, from 1980s to the turn of the century. About 20% to 50% of drug development failures can be attributed to toxicity and clinical safety, 1 so drug toxicity evaluation is becoming critical for drug development.
Chemotherapy has played a major role in cancer treatment. Although anticancer drugs have successfully prolonged or even saved the patients’ lives, more and more clinical adverse reactions of anticancer drugs have been reported, such as cardiovascular toxicity, 2,3 neurotoxicity, 4 and toxicity in other organ systems, 5 which highlight the need for advanced testing that enables successful translation of preclinical toxicity assessment to the clinical setting with better accuracy and sensitivity. 6
Zebrafish share physiological, morphological, and histological similarities with mammals and have been recognized as valuable models for evaluating drug candidates for toxicity and safety liabilities. 7 The gene homology between zebrafish and humans is as high as 85%. In signal transduction pathways and at the protein level, the functional domains are highly conserved or structurally identical. 8 The zebrafish have its advantages compared to the traditional in vivo model like mouse in that multiple organs can be observed; pharmacodynamic, pharmacokinetic, and metabolite activity can be evaluated; low cost; short cycle; less amounts of test compound; and high throughput. Zebrafish model system has been successfully used in studying developmental toxicity, teratogenicity, cardiovascular toxicity, liver toxicity, behavioral toxicity, kidney toxicity, and a series of evaluation assays. 9
Seven small molecules, triptolide (TP), gambogic acid (GA), mycophenolic acid (MPA), curcumin, auranofin (AF), thalidomide, and taxol, in different stages of the drug developmental pipeline, were chosen for this study. Triptolide, a key biologically active component isolated from the Chinese medicinal herb Tripterygium wilfordii Hook F, has anti-inflammatory, immunosuppressive, and anticancer activities. 10 Despite its broad antitumor effects, the clinical use of TP is restricted due to its poor water solubility and some toxic effects. 11 Its prodrug PG490-88 and F60008 have been approved in phase I clinical trial for the treatment of prostate cancer and advanced solid tumors, respectively, in the United States. 12 Gambogic acid has strong anticancer activity by targeting diverse molecular targets but possess acceptable toxicity. 13 It has been approved by the Chinese Food and Drug Administration (FDA) for clinical trials in patients with cancer. 14 Mycophenolic acid is a known inosine monophosphate dehydrogenase (IMPDH) inhibitor and was used as an immunosuppressant in the last decades. It was recently shown to have anticancer activity through antiangiogenic and antifibrotic effects but the molecular basis warrants further clinical investigations. 15 Curcumin, an active component of the spice turmeric (Curcuma longa), possess biological activities that include chemopreventive and therapeutic properties against many tumors both in vitro and in vivo and in clinical trials. 16 Auranofinwas used in the clinic as an antirheumatoid arthritis medicine in the past years 17 due to its remarkable anti-inflammatory and analgesic effects. In recent years, it was reported that AF has inhibitory effects on both cancer cell lines and murine tumor models, 18,19 which led to clinical trials for the treatment of chronic lymphocytic leukemia and recurrent epithelial ovarian cancers. 20 Thalidomide was prescribed for use as a sedative and for the treatment of morning sickness before it caused a worldwide epidemic of multiple birth defects. 21 In May 2006, the FDA approved its use with dexamethasone for the treatment of patients with newly diagnosed multiple myeloma. 22 However, its use is limited due to the potent teratogenic activity. 23 Taxol is a known microtubule-targeting agent and is clinically used in the treatment of breast cancer, advanced ovarian cancer, nonsmall cell lung cancer, and advanced forms of Kaposi sarcoma. 24 All these 7 compounds have anticancer activity through different molecular mechanisms. We postulated that due to different chemical structures and drug targets, they should have different toxicity pattern/profile. Although there are studies about their activities and toxicities, there is no report to compare their toxicities simultaneously. In this study, the acute toxicities, developmental toxicities, and cardiovascular toxicities of these 7 anticancer drugs were assessed in zebrafish. The zebrafish model was found to be an alternative or even better predictive toxicity model compared to conventional system in drug safety assessment.
Materials and Methods
Zebrafish Handling
Wild-type zebrafish (Danio rerio; Tubingen line) were obtained from Model Animal Research Center of Nanjing University. Adult zebrafish below 1-year-old were housed in a light- and temperature-controlled aquaculture facility with a standard 14:10-hour light–dark photoperiod with a pH of 7 ± 0.2 and fed with live brine shrimp once daily and dry shrimp flakes twice a day. 25 Embryos were produced by pairwise mating using fish hatch box. Embryos were maintained at 28.5°C in E3 embryo medium (containing antifungal solvent [0.01% methylene blue] and salts [60 × E3 embryo medium 1 L: 17.2 g NaCl, 0.76 g KCl, 4.9 g MgSO4•7H2O, 2.9 g CaCl2•2H2O]). 26 Media was diluted in double distilled water (ddH2O; 4 mL 60 × E3 embryo medium in ddH2O to make up of 250 mL 1 × E3 embryo medium with 0.75 mL 0.01% methylene blue). Normally developed embryos were dechorionated using forceps before drug treatment. All zebrafish studies were approved by the institutional animal care and use committee at Nanjing University of Technology.
Drugs
The tested drugs thioridazine (Chemical Abstracts Service Registry [CAS] no. 130-61-0, purity ≥ 99%), thalidomide (CAS no.50-35-1, purity ≥ 98%), taxol (CAS no. 33069-62-4, purity ≥ 98%), TP (CAS no. 38748-32-2, purity ≥ 98%), GA (CAS no. 2752-65-0, purity ≥ 98%), MPA (CAS no. 24280-93-1, purity ≥ 99%), curcumin (CAS no.458-37-7, purity ≥ 98%), and AF (CAS no. 34031-32-8, purity ≥ 99%) were purchased from Sigma-Aldrich (Sigma-Aldrich, St. Louis, USA).
Drug Delivery by Soaking
Stock solutions (100 mmol/L) for each compound were prepared in 100% dimethyl sulfoxide (DMSO; Sigma). Series of dilutions for each compound were made in DMSO before adding to the embryo medium by 1:1000 (v/v). Zebrafish treated with 0.1% DMSO were used as a vehicle control in all assays to confirm that the vehicle solvent did not have an adverse effect on the zebrafish. To protect compounds from light-induced decomposition, experiments were carried out at a constant temperature (28.5°C) in the dark. After treatment, zebrafish were subjected to visual observation and image acquisition under a dissecting stereomicroscope (Nikon SMZ745T; Nikon, Japan).
Acute Toxicity Assay
The morphogenesis and function of the primary organ systems of zebrafish embryos was completed by 72 hours postfertilization (hpf) 27 ; hatching from the chorion was also completed by this time point. Thus, zebrafish embryos at 72 hpf were selected for the acute toxicity assay. Drug concentrations, designed on the basis of preexperiment data, used in the acute toxicity tests are summarized in Table 1. Zebrafish embryos at 72 hpf were assayed in 24-well plate, 20 larvae/well for each concentration, for 24 hours. The observation of zebrafish was made directly in the 24-well plate using an inverted dissecting microscope. During the acute toxicity experiment, fish were not fed and dead fish were removed from the well. The number of dead zebrafish in each concentration solution within 24 hours was recorded and the survival rate (%) was calculated. GraphPad Prism 5 (log[inhibitor] vs normalized response-variable slope nonlinear model) was used to calculate 50% lethal concentration (LC50) values for each drug. GraphPad Prism 5 was purchased from GraphPad Prism 5.0 (GraphPad Software, San Diego, CA, USA).
The Concentration and LC50 Value of Each Drug in Acute Toxicity Assay and LD50 in Mice from Related References.
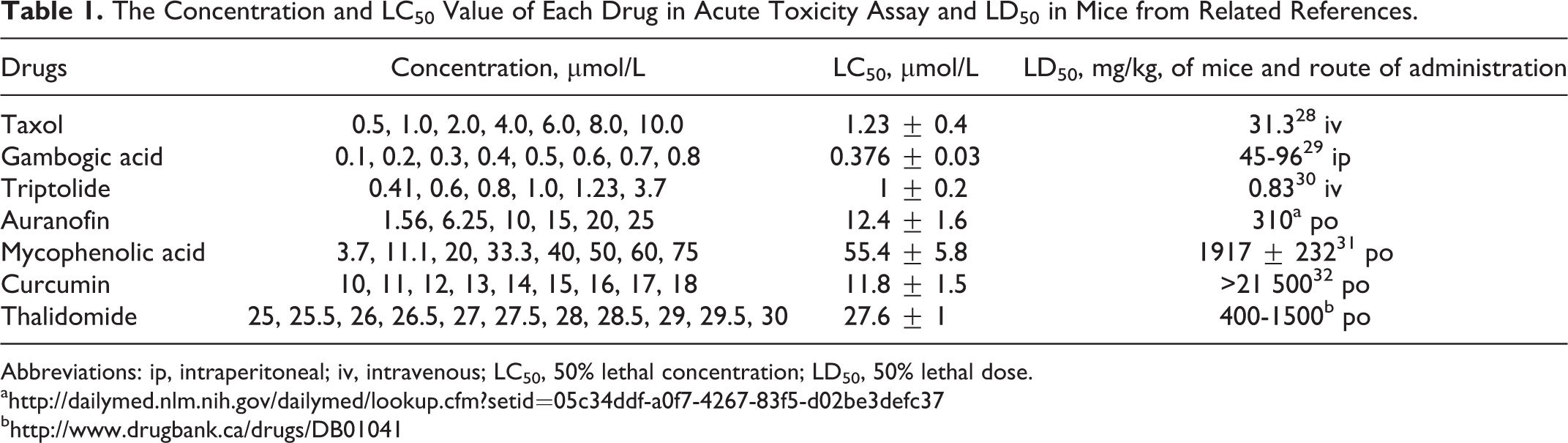
Abbreviations: ip, intraperitoneal; iv, intravenous; LC50, 50% lethal concentration; LD50, 50% lethal dose.
Developmental Toxicity Assay
Zebrafish embryos develop rapidly during the first 3 days. By 72 hpf, morphogenesis and function of the primary organ systems of zebrafish embryos are completed, 27 so the time frame from 2 to 72 hpf was used for the developmental assay. Dechorionated zebrafish embryos were exposed to a test solution at 2 hpf. Viability and morphological defects were assessed on 24, 48, and 72 hpf. Ten embryos were selected for each treatment condition. As the treatment time in developmental and cardiovascular toxicity assays was earlier than that in acute toxicity assay, the tolerant drug concentrations was lower than that in acute toxicity assay. We choose 1/10 LC50, 1/5 LC50, and 1/2 LC50 for the developmental and cardiovascular assay. Drug concentrations used in the developmental toxicity tests are summarized in Table 2. In preliminary experiments, the potential time points for the assessment of compound-induced dysmorphology and general toxicity parameters were evaluated and 72 hpf was found to be the optimum developmental stage for evaluation.
Drug Concentrations Tested for Assessing Developmental Toxicity and Cardiovascular Toxicity.
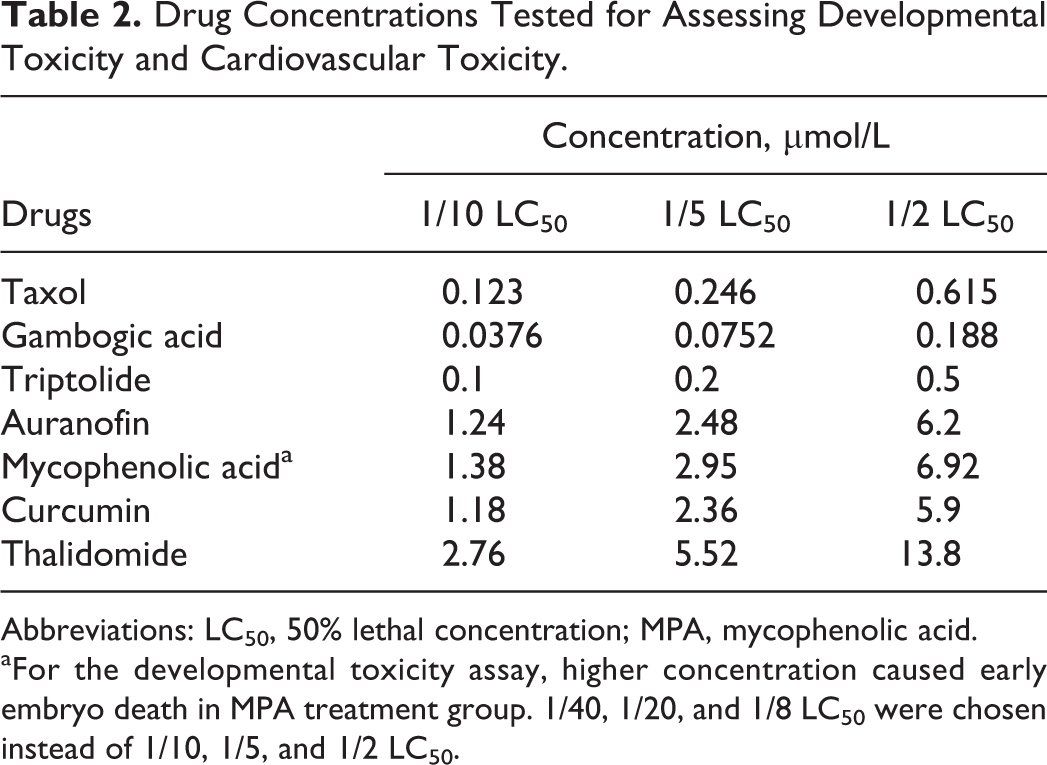
Abbreviations: LC50, 50% lethal concentration; MPA, mycophenolic acid.
aFor the developmental toxicity assay, higher concentration caused early embryo death in MPA treatment group. 1/40, 1/20, and 1/8 LC50 were chosen instead of 1/10, 1/5, and 1/2 LC50.
Cardiovascular Toxicity Assay
The zebrafish heart begins to beat by 22 hpf and the cardiovascular system is fully functional and exhibits a complex repertoire of ion channels and metabolic processes by 48 hpf
7
; thus, dechorionated zebrafish embryos at 48 hpf were selected as treatment initiation point. Drug concentrations used in the cardiovascular toxicity tests are summarized in Table 2. At the end of treatment, 10 zebrafish of each group were used for visual observation and image acquisition under a dissecting stereomicroscope. Six specific phenotypic end points were used in cardiovascular toxicity assessment as described: heart rate, heart rhythm, pericardial edema, circulation, hemorrhage, and thrombosis. The heart rate and rhythm were assessed at 52 hpf and pericardial edema, circulation, hemorrhage, and thrombosis were observed at 72 hpf. Thioridazine had known effects on heart rate at 5 µmol/L
33
and thus served as a positive control for heart rate test. A normalized heart rate was calculated using the following formula: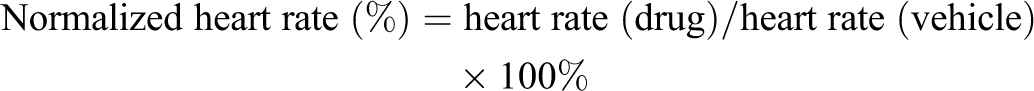
Statistical Analysis
All experiments were repeated at least twice after the experimental conditions were optimized. The LC50 was calculated with GraphPad Prism 5. Heart rate was expressed as mean ± standard error of the mean using GraphPad Prism 5 and heart rate differences were analyzed using 1-way analysis of variance followed by the Dunnett multiple comparison tests.
Results
50% Lethal Concentration Acute Toxicity Assay
Zebrafish lethality curves are presented in Figure 1. Based on the zebrafish lethality curves of the acute toxicity assay, LC50 of each drug was calculated and the results are indicated in Table 1.
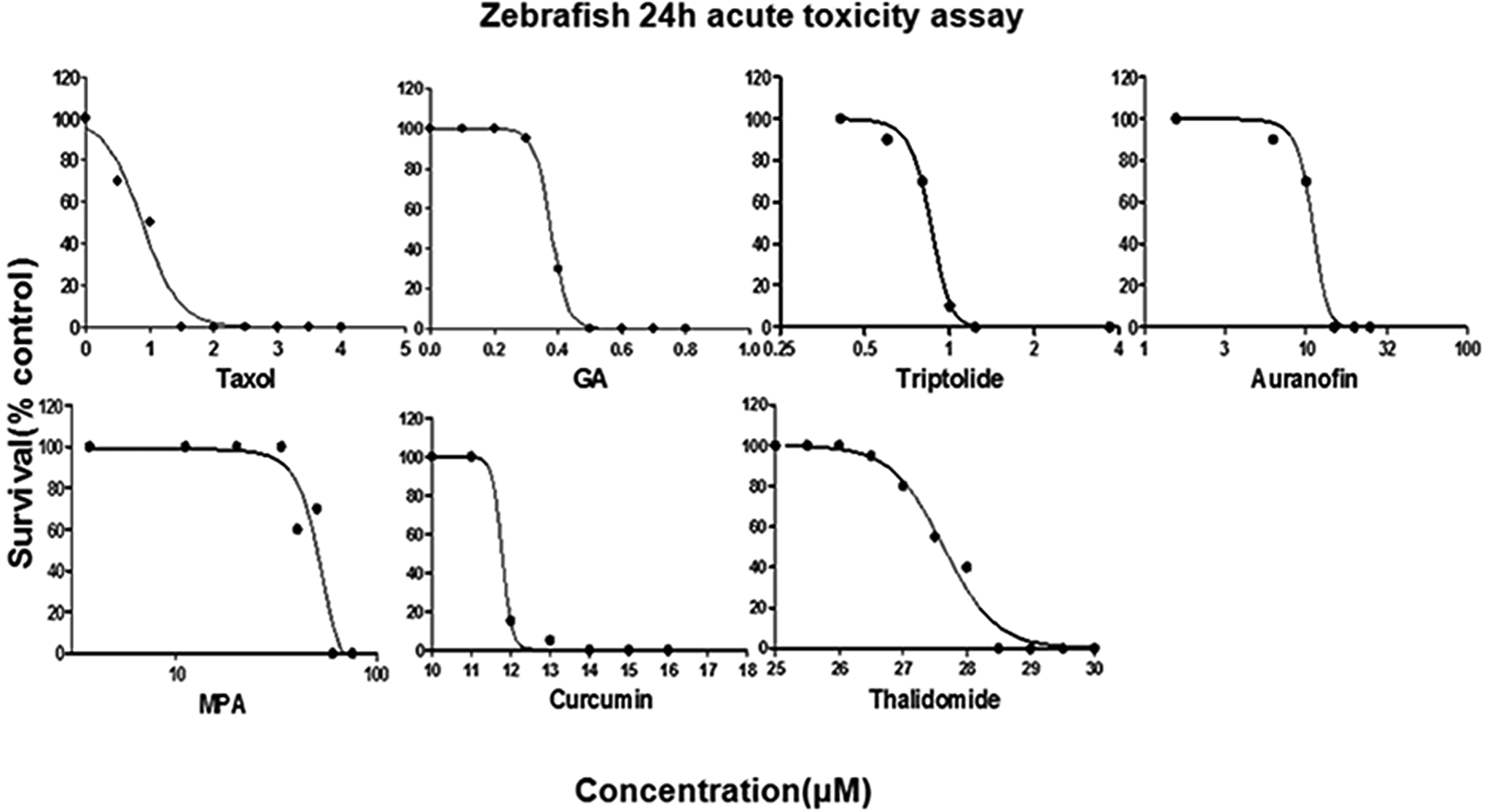
Drug-induced dose-dependent zebrafish survival rate at 96 hours postfertilization (hpf).
Phenotypic Changes in Developing Zebrafish Embryos/Larvae
For embryos/larvae in the exposure set, observations were made at 24, 48, and 72 hpf. Morphological abnormalities included edema of the periphery and/or abdomen, hemorrhage (a pool of blood clustered in an area outside the normal circulation), abnormal body shape (scoliosis, tail bent, and tail detachment), large yolk sac, pigmentation, motility reduction, and absence of pectoral fins (summarized in Table 3). For the developmental toxicity assay, higher drug concentrations caused early embryo death in MPA-treated group. Doses of 1/40, 1/20, and 1/8 LC50 were used. Pericardial edema was observed in TP-, AF-, MPA-, and thalidomide-treated groups. Hemorrhage was evident in the AF-treated groups. The GA-, TP-, AF-, MPA-, and thalidomide-treated embryos displayed abnormal body shape. Motility was decreased in the TP-, AF-, and MPA-treated groups. Pigmentation was reduced in taxol-, GA-, and TP-treated groups. The TP- and MPA-treated groups displayed axis shortening. Individual drugs induced morphological abnormalities at higher concentrations and had specific toxicity patterns. The AF-treated group presented with abnormal body shape and severe pericardial edema (Figure 2B). The TP-treated group exhibited pericardial edema, abnormal body shape, axis shortening, and enlarged yolk sac (Figure 2C). An interesting phenomenon was found in the MPA-treated group in that the zebrafish had a defect in axis formation, severe pericardial edema, and enlarged yolk sac (Figure 2D), suggesting that MPA caused severe growth arrest in zebrafish embryo. It was immediately apparent that in thalidomide-, GA-, and TP-treated embryos, development of pectoral fins was disturbed at certain concentrations whereas other general aspects of development were not affected (Figure 3).
Morphological Abnormalities Induced by Several Anticancer Drugs and in Development Toxicity.
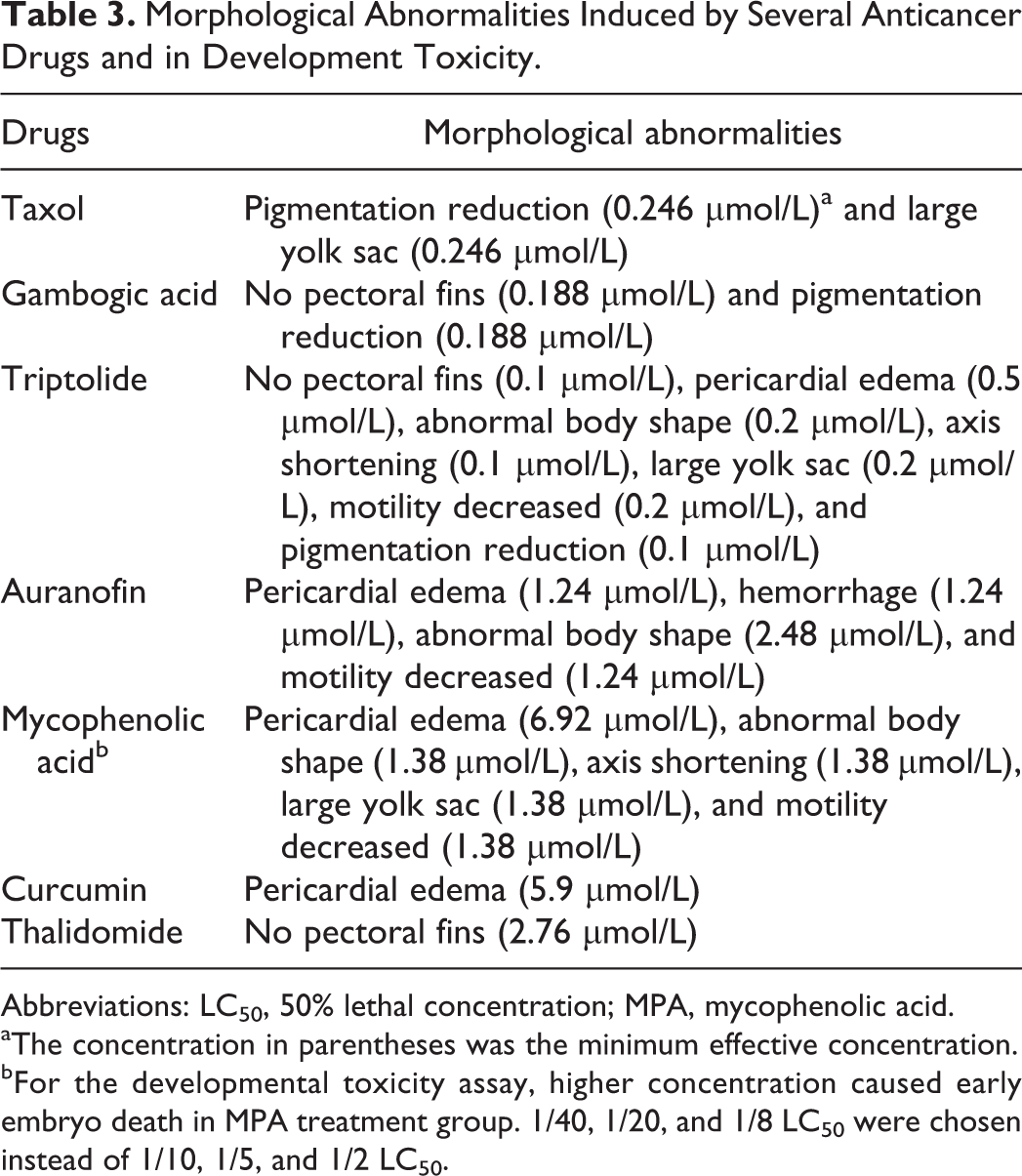
Abbreviations: LC50, 50% lethal concentration; MPA, mycophenolic acid.
aThe concentration in parentheses was the minimum effective concentration.
bFor the developmental toxicity assay, higher concentration caused early embryo death in MPA treatment group. 1/40, 1/20, and 1/8 LC50 were chosen instead of 1/10, 1/5, and 1/2 LC50.
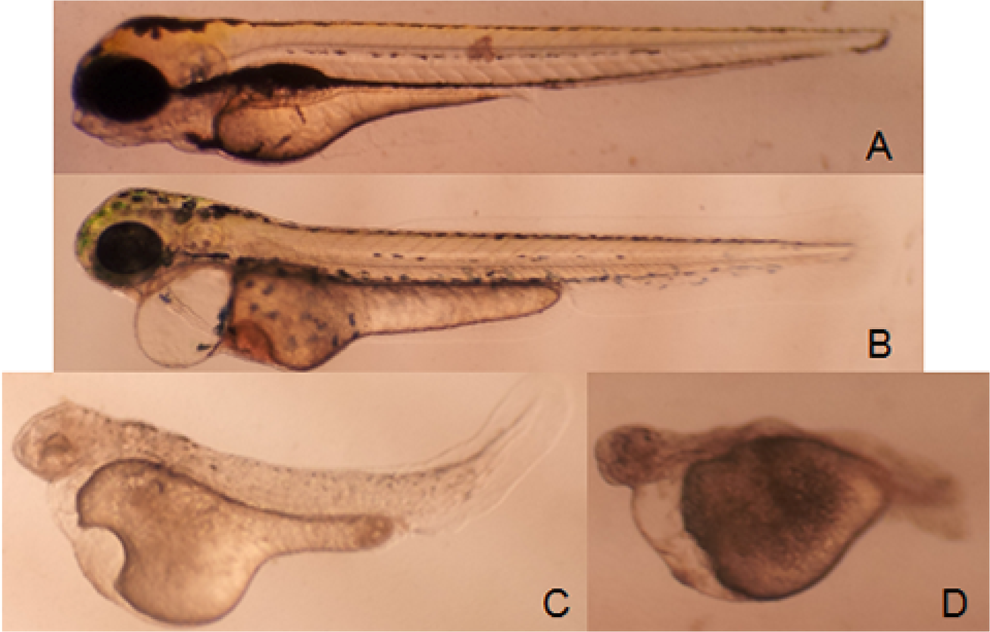
Zebrafish treated with compounds at 2 hpf and observed at 72 hpf. Control (A); 1.24 μmol/L auranofin (B); 0.5 μmol/L triptolide (C); and 6.92 μmol/L mycophenolic acid (MPA; D). hpf indicates hours postfertilization.
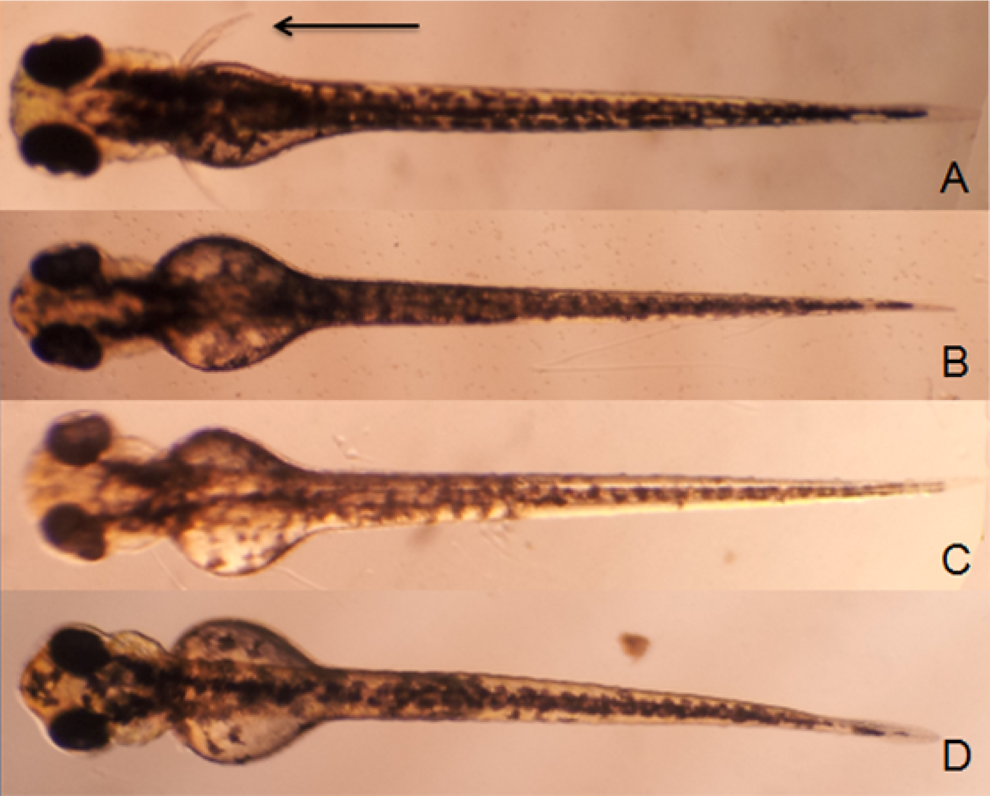
Zebrafish treated with compounds at 2 hpf and observed at 72 hpf. Pectoral fins are indicated by arrowheads. Control (A); 2.76 μmol/L thalidomide (B); 0.188 μmol/L gambogic acid (C); and 0.1 μmol/L triptolide (D). hpf indicates hours postfertilization.
Heart Rate and Heart Rhythm
Zebrafish atrial and ventricular rates were recorded at 4 hours posttreatment and analyzed for statistical significance (Figure 4). Taxol did not induce any observable adverse effect on the cardiovascular system. Significant bradycardia was observed in zebrafish treated at 1/2 LC50 by MPA and thalidomide as well as thioridazine at concentration of 5 µmol/L.
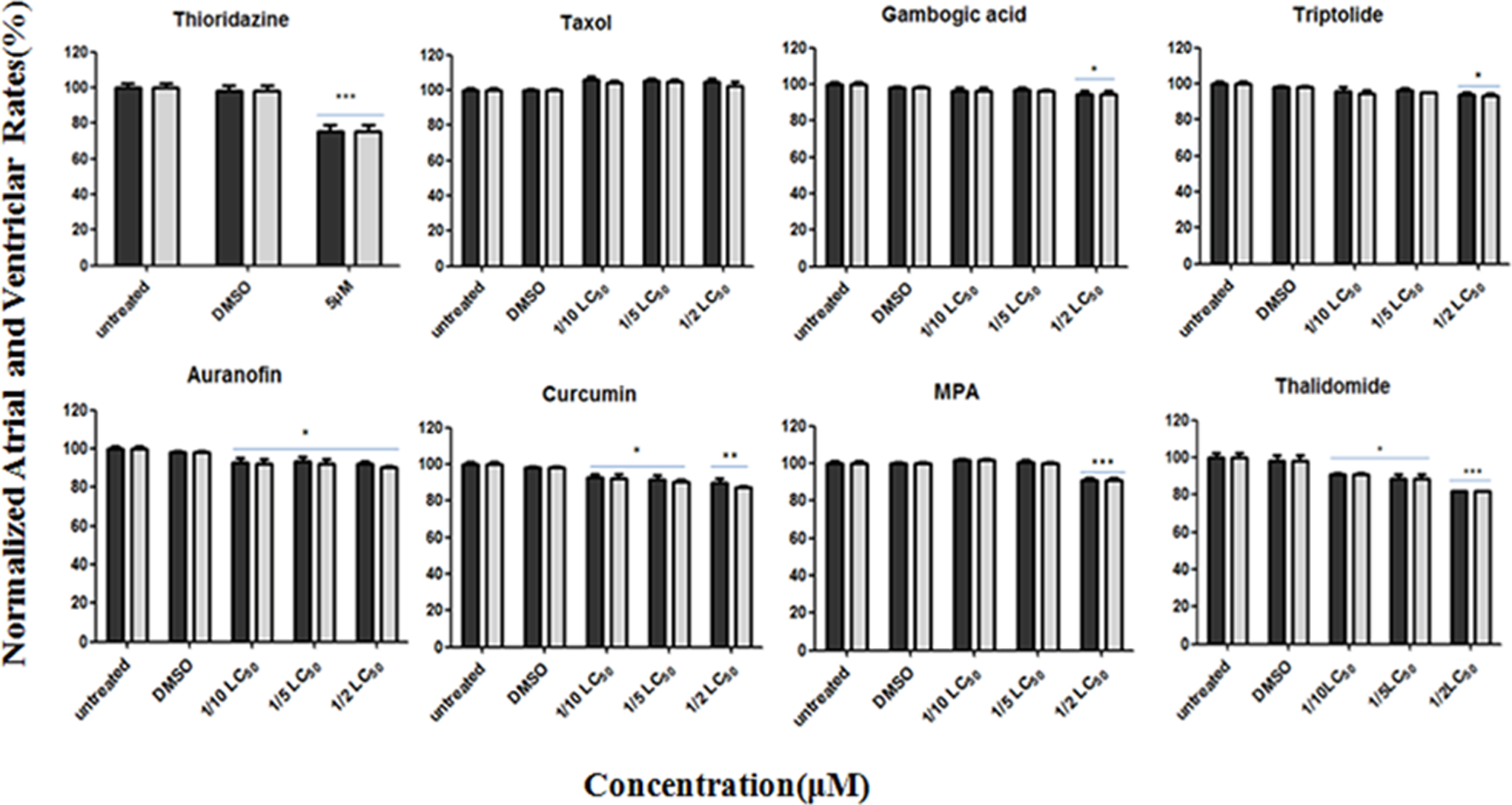
Zebrafish heart rate changes versus vehicle control (0.1% dimethyl sulfoxide [DMSO]) or untreated zebrafish after 4 hours of drug treatment. Dark represents the atrial rate, and grey represents the ventricular rate. Each point represents mean ± standard error of the mean (SEM; n =10) from a representative experiment. *P < .05, **P < .01, and ***P < .001 in 1-way analysis of variance (ANOVA) followed by the Dunnett multiple comparison test.
Pericardial Edema, Circulation, Hemorrhage, and Thrombosis of Cardiovascular Toxicity Assay
After 24-hour drug treatment, 10 zebrafish per group were used to observe cardiovascular toxicity-associated morphology including pericardial edema, circulation, hemorrhage, and thrombosis, which was summarized in Table 4. Pericardial edema was observed in TP-, AF-, and MPA-treated groups. The AF- and MPA-treated groups caused hemorrhage. Curcumin, GA, and taxol did not cause any morphological abnormality. The AF caused slow blood flow in circulation system. No thrombosis was found from any treatment.
Morphological Abnormalities Induced by Several Anticancer Drugs in Cardiovascular Toxicity.

Discussion
Most zebrafish assays rely on aqueous exposure and zebrafish larvae absorb small molecules diluted in the surrounding water through their skin, gut, and gills. 9 Compound uptake is dictated by the physicochemical properties of each compound, and a false-negative result could occur in aqueous exposure experiments owing to low compound absorption. 34 In our study, all the tested compounds were hydrophobic (with positive log P value), which suggests that the drugs can penetrate the embryo freely.
In the acute toxicity assay, tested compounds showed large variation in their acute toxicity. The GA has the lowest LC50 value (0.376 μmol/L) while MPA has the highest LC50 value (55.4 μmol/L). Three compounds, GA, TP, and taxol, showed high acute lethality with LC50 ≈ 1μmol/L. The 50% inhibitory concentration (IC50) of GA, TP, and taxol is about 200, 35 20, 36 and 250 nmol/L, 37 respectively, in several human cancer cell lines in 48 hours cytotoxicity assay. The AF, curcumin, and thalidomide showed moderate acute toxicity compared with GA and MPA. The IC50 of AF, curcumin, and thalidomide is about 1, 17 10, 38 and 25 μmol/L, 39 respectively, in several human cancer cell lines in 48 hours cytotoxicity assay. From the LC50 of zebrafish assay and the IC50 of cytotoxicity assay, we found that the acute toxicity of these compounds in zebrafish was comparable to the cytotoxicity in human cancer cell lines.
Acute toxicity of zebrafish also showed good correlation with other mammalian models. In this experiment, LC50 values of drugs in zebrafish were significantly correlated with those in rodents (mice) reported in the literature (Table 1). We found 3 compounds, GA, TP, and taxol, showed high acute lethality with LC50 ≈ 1 μmol/L in zebrafish assay. At the same time, these 3 compounds also showed high acute lethality with 50% lethal dose (LD50) lower than 100 mg/kg in mice. The other 4 compounds showed moderate acute toxicity in mice assay and in zebrafish assay. The inconsistency between the LC50 in zebrafish assay and LD50 in mice assay for each compound might be due to the species difference as well as the differences in drug delivery and drug metabolism. Although the 2 values may not be able to translate to each other directly, they reflected the same general toxicity trend. We concluded that it is feasible to use zebrafish model to rapidly assess compound toxicity, and the acute toxicity in zebrafish model is comparable to that in rodent model.
In the developmental toxicity assay, we found MPA induced a significant growth arrest in embryo development. Echardt group reported the same embryotoxicity of MPA when the rat whole embryos were cultured in vitro with MPA. 40 The MPA caused zebrafish embryo axis and tail defects and severe pericardial edema, similar to that treated with retinoids. 34 The MPA was a known potent uncompetitive inhibitor of IMPDH, the rate-limiting enzyme in the de novo synthesis of guanosine nucleotides. 15 The Lin group 41 reported that targeting IMPDH by MPA or morphorlinos in zebrafish resulted in suppressed embryo angiogenesis without significant impact on the gross morphology. Our study first found that MPA could cause severe embryo developmental defect. The discrepancy of the zebrafish phenotype after MPA treatment between Lin study and ours may be the different initial treatment time and drug concentration. Our research also gave clues that IMPDH may play an important role in the axis formation in vertebrate development besides angiogenesis or unknown target of MPA exists. Further study will be initiated to discover the link between IMPDH and axis formation in the zebrafish model.
Another interesting phenomenon was that we found severe pericardial edema in AF-treated embryos. Auranofin is the first metal phosphine complex introduced into clinical practice for chrysotherapy, the treatment of rheumatoid arthritis with gold-based drug. In particular, it was found that AF, acting as a potent inhibitor of thioredoxin (Trx) reductase (TrxR), causes an alteration in the redox state of the cell leading to an increased production of hydrogen peroxide and oxidation of the components of the Trx system. 17 The TrxR plays an important role in protecting cardiac tissue from oxidative stress. Auranofin was showed to increase the degree of postischemic apoptosis in rat ischemia–reperfusion assay. 42 Our research repeated the cardiotoxicity of AF in the zebrafish model and offered other evidence that zebrafish is a good model to assess the cardiovascular toxicity of small molecules. Our further study will focus on the role of TrxR in AF-induced zebrafish cardiotoxicity.
We found pectoral fins defects in thalidomide-, GA- and TP-treated embryos. It was immediately apparent in thalidomide-treated embryos at 2.8 μmol/L or higher concentrations that development of pectoral fins was disturbed whereas other aspects of development were not generally affected, which indicated that thalidomide targets analogous structures in human and zebrafish. Furthermore, the molecular targets of thalidomide that affect limb formation were highly conserved in human and zebrafish. 21 Our study found for the first time that GA and TP could also cause embryonic teratogenicity in zebrafish model with the same phenotype as that caused by thalidomide. It will be extremely important to investigate the limb teratogenic mechanism of the 2 compounds for their safe use in the clinic. Thalidomide showed high selectivity in the disturbance of pectoral fin formation. But GA and TP caused other developmental defects when dosed at higher concentration (Table 3).
In our cardiovascular toxicity study, thalidomide caused obvious bradycardia at concentrations ≥2.76 µmol/L. Our findings provide the same results with a clinical trial of thalidomide (ClinicalTrials.gov Identifier: NCT00231140) in which bradycardia was the most common adverse event of thalidomide treatment in patients with amyotrophic lateral sclerosis (ALS). This toxicity discouraged further clinical trials and compassionate use of thalidomide in ALS or other clinical applications. 43
Zebrafish embryogenesis progresses through a standard embryological developmental sequence of events choreographed by numerous signaling pathways. Perturbation of these molecular pathways leads to distinct and predictable defects in the embryos. In our study, small molecules with different targets and potency caused unique toxicity profile in zebrafish. The zebrafish model can not only provide toxicological responses similar to those by mammals but also provide novel findings of toxicities that yet to be discovered by conventional model system. In conclusion, zebrafish model has become a valuable tool for assessing and predicting drug toxicity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Jiangsu Provincial Natural Science Fund (Grant BK2011796), New Teacher Fund from Chinese Ministry of Education (Grant 20123221120005), and National Natural Science Foundation of China (Grants 81172947 and 81073129).
