Abstract
Azo dyes are used widely as color additives in food, drugs, and cosmetics; hence, there is an increasing concern about their safety and possible health hazards. In the present study, we chose 4 azo dyes tartrazine, Sunset Yellow, amaranth, and Allura red and evaluated their developmental toxicity on zebrafish embryos. At concentration levels of 5 to 50 mM, we found that azo dyes can induce hatching difficulty and developmental abnormalities such as cardiac edema, decreased heart rate, yolk sac edema, and spinal defects including spinal curvature and tail distortion. Exposure to 100 mM of each azo dye was completely embryolethal. The median lethal concentration (LC50), median effective concentration (EC50), and teratogenic index (TI) were calculated for each azo dye at 72 hours postfertilization. For tartrazine, the LC50 was 47.10 mM and EC50 value was at 42.66 mM with TI ratio of 1.10. For Sunset Yellow, the LC50 was 38.93 mM and EC50 value was at 29.81 mM with TI ratio of 1.31. For amaranth, the LC50 was 39.86 mM and EC50 value was at 31.94 mM with TI ratio of 1.25. For Allura red, the LC50 was 47.42 mM and EC50 value was 40.05 mM with TI ratio of 1.18. This study reports the developmental toxicity of azo dyes in zebrafish embryos at concentrations higher than the expected human exposures from consuming food and drugs containing azo dyes.
Introduction
Azo dyes are organic compounds with the functional group R−N=N−R′, in which R and R′ are usually aryl. The azo bond and different, mainly aromatic, side groups make azo compounds stable and tend to produce vivid colors. Owing to the simple synthetic procedures involved, the great structural diversity, high molar extinction coefficient, and medium-to-high fastness properties both in light and in wetness, azo dyes are widely used in artificial coloring. 1,2
In 1881, artificial coloring was first authorized in food use. 3 By 1900, artificial coloring of food was common. Currently, azo dyes account for approximately 50% of all organic dyes used globally as color additives in food, drugs, and cosmetics. 4 For many years, the toxicity of azo dyes was reported to be rather low for classification of dangerous substances. However, not all of the azo dyes are harmless, and some can cause a variety of chronic effects. Reactive Brilliant Red can inhibit the function of human serum albumin and cause a conformational change or even precipitation at a concentration of 0.35 mM. 5 Sudan Red dyes, containing azo and hydroxyl groups, have been shown to result in toxic effects such as membrane blocking and membrane expansion at a concentration of 0.8 mM. 6 Furthermore, several azo dyes have been found to be mutagenic or carcinogenic in laboratory rodents at a dose level of 200 to 1,000 mg/kg body weight. 7
Since the 20th century, the governments in different countries have enforced the regulatory oversight for azo dyes used as color additives, and large numbers of toxicity assessments have been performed on them. 8 Nevertheless, their potential impact on human health is still difficult to determine, especially in pregnant women and children. 9 Traditional toxicity studies of azo dyes have been conducted on mammalian models, mainly rats and mice, which generally focus on immunological, serological, biochemical, and histopathological features. 10 -12 However, developmental toxicity testing is seldom carried out.
Zebrafish (
In the present study, we attempted to evaluate the developmental toxicity of azo dyes on zebrafish embryos as a model system. We chose 4 azo dyes that are commonly used as color additives: tartrazine, Sunset Yellow, amaranth, and Allura red. The developmental features and teratogenic potential were critically analyzed in zebrafish embryos from the blastula stage (2 hours postfertilization [hpf]) until 72 hpf.
Materials and Methods
Chemicals and Preparation of Embryo Medium and Exposure Media
The 4 azo dyes, tartrazine, Sunset Yellow, amaranth, and Allura red were obtained from Sigma-Aldrich (St Louis, Missouri). The embryo E3 medium (5 mM NaCl, 0.17 mM KCl, 0.33 mM CaCl2, and 0.33 mM MgSO4) was prepared as the protocol described in the Zebrafish Book (2007). 13,17 As these dyes are water-soluble, all the exposure media were prepared in E3 medium just before use. The Chemical Abstracts Service number, FD&C number, purity, solubility, and test concentrations for each azo dye are shown in Table 1.
CAS Number, FD&C Number, Purity, Solubility, and Test Concentrations of the 4 Azo Dyes.

Zebrafish Maintenance and Embryo Collection
Wide-type zebrafish (Tubingen line) were obtained from Model Animal Research Center of Nanjing University. They were kept in fish tanks at 28.5°C and pH of 7.0 followed the Organization for Economic Cooperation and Development (OECD) Test Guidelines (2013). The light–dark cycle was 14:10 hours. 15 Adult male and female fish (2:1) were placed in a spawning tank, and spawning was stimulated by the onset of light. The next day, fertilized embryos were found at the bottom of spawning tank and then collected into Petri dishes containing E3 medium for washing thoroughly. 18 All the studies on zebrafish were approved by the Institutional Animal Care and Use Committee in Nanjing Medical University.
Experimental Design
The embryos were randomly distributed in 24-well plates at a density of 20 embryos/well, containing 1 mL of exposure media per well with the various test concentrations of each azo dye (n = 20/concentration) and exposed from the blastula stage (2 hpf) until 72 hpf. The test concentrations were selected following the OECD Test Guidelines (2013) and methods of Joshi and Katti. 19 All experiments were done in triplicate, and each assay was repeated 3 times.
Lethality and Developmental Toxicity
Embryolethality was monitored daily during the whole experiment. Dead embryos were removed, and survival rate was reported every day until 72 hpf. Hatching rate was calculated by the rate of successful hatching embryos divided by the total number of embryos at each concentration and recorded from 2 to 72 hpf.
At 72 hpf, morphological defects, including cardiac edema, yolk sac edema, spinal defects (including spinal curvature and tail distortion), and heartbeats per minute were assessed using a dissecting microscope (SMZ800N; Nikon, Tokyo, Japan). The incidences of cardiac edema, yolk sac edema, spinal defects, and decreased heartbeats were calculated as the number of abnormal embryos divided by the number of surviving embryos at each concentration. Each treatment was performed in triplicate.
Calculation of Median Lethal Concentration, Median Effective Concentration, and Teratogenic Index
At 72 hpf, median lethal concentration (LC50) and median effective concentration (EC50) were calculated using linear regression. The teratogenic index (TI), a ratio of LC50 and EC50, was calculated to estimate the teratogenic potential of each azo dye. 20
Statistical Analysis
The data were expressed as mean ± standard error of the mean and analyzed using one-way analysis of variance followed by Dunnett multiple comparison tests (GraphPad Prism 5.0). Correlation coefficient
Results
Lethality
The azo dyes caused embryo lethality in zebrafish in a dose-dependent manner (Figure 1). Low concentrations (5-20 mM) of azo dyes caused minimal lethality during the exposure window, and the survival rate was coincident of more than 75%. In the 50-mM exposure groups, tartrazine, Sunset Yellow, amaranth, and Allura red caused lethality of 44.2%, 51.7%, 56.7%, and 46.7% at 72 hpf, respectively. There were few survivors in the 100-mM exposure groups of azo dyes.
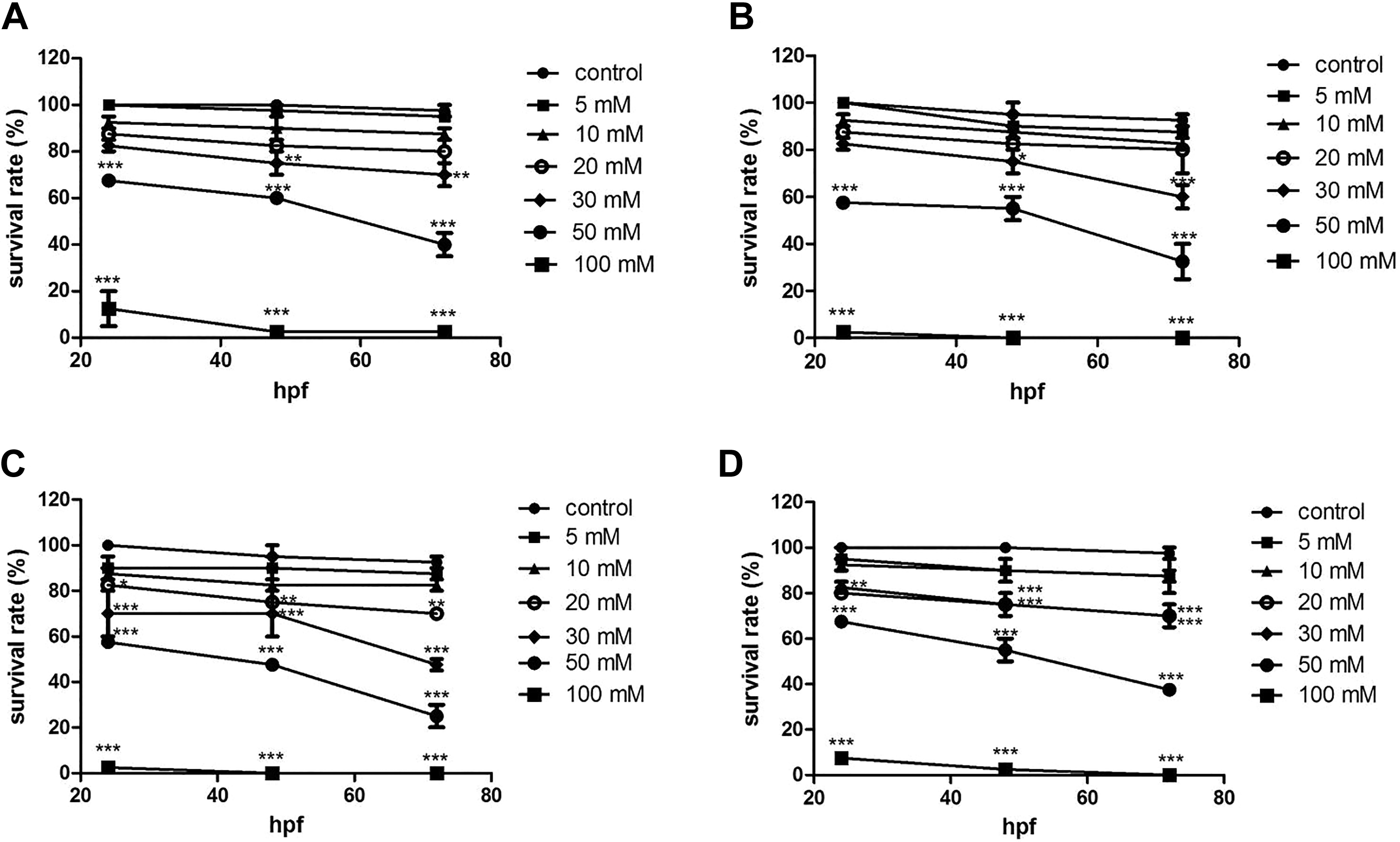
Survival rate of zebrafish embryos exposed to 4 azo dyes at different concentrations from 24 to 72 hours postfertilization (hpf). (A) tartrazine; (B) Sunset Yellow; (C) amaranth; and (D) Allura red. (***) indicates statistical significance
Developmental Toxicity
The azo dyes caused dose-dependent hatching difficulty and developmental abnormalities in zebrafish embryos, including cardiac edema, decreased heart rate, yolk sac edema, and spinal defects including spinal curvature and tail distortion (Figure 2).
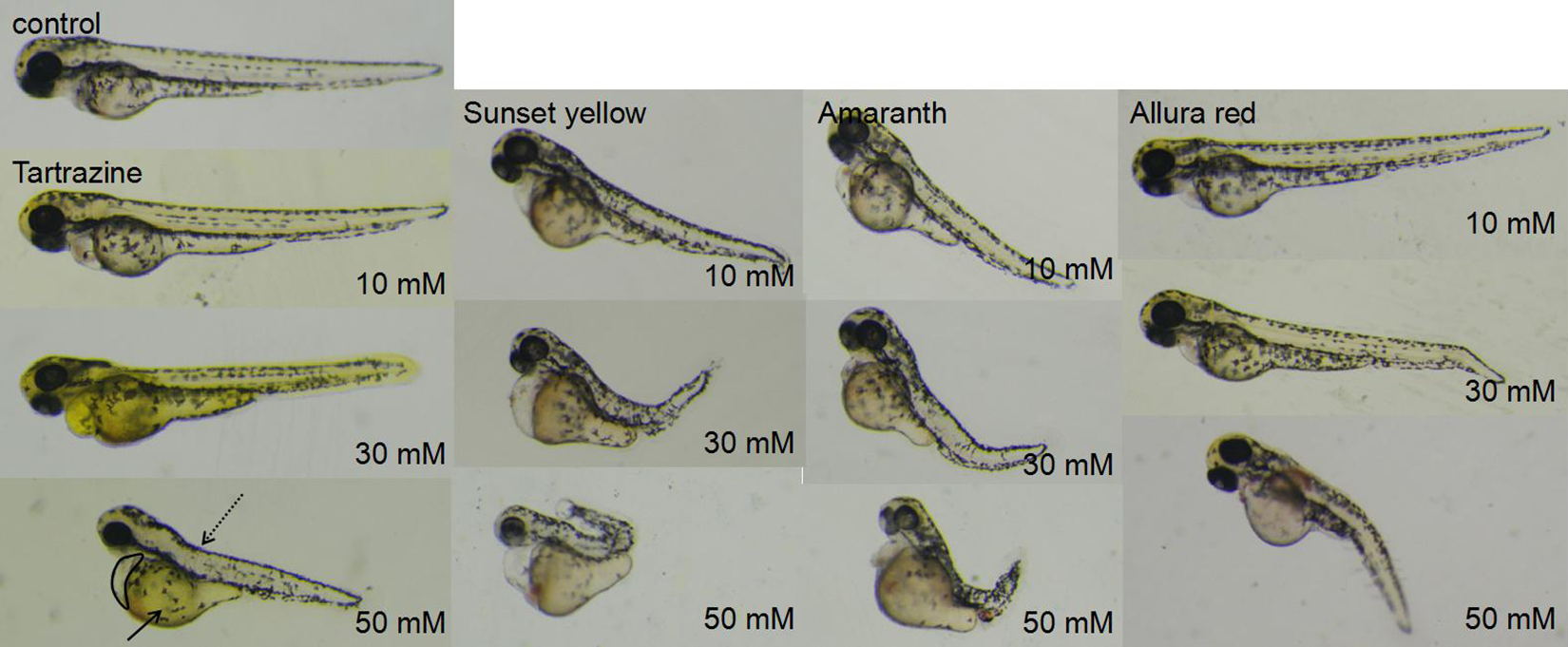
Developmental abnormalities in zebrafish embryos exposed to 4 azo dyes at 72 hours postfertilization (hpf), including cardiac edema (black circle), yolk sac edema (black arrow), and spinal defects including spinal curvature and tail distortion (black dotted arrow).
Hatching Rate
The hatching rate declined gradually at concentrations of 10 to 50 mM (Figure 3). In the 50-mM exposure groups, the hatching rates of tartrazine, Sunset Yellow, amaranth, and Allura red at 72 hpf were 27.5%, 28.3%, 15.0%, and 29.2%, respectively. When dosed at 100 mM, there were no hatching embryos due to lethality.
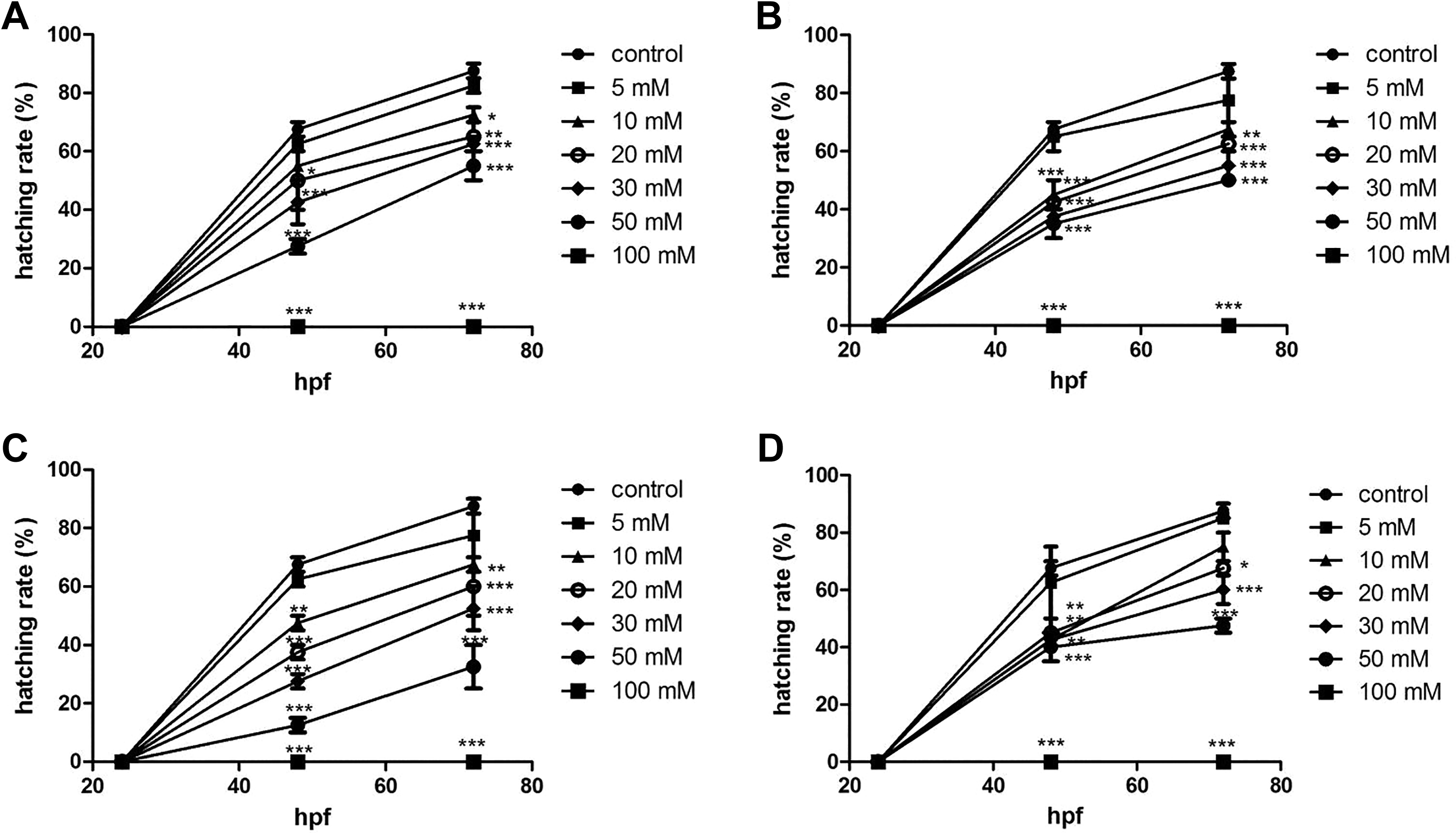
Hatching rate of zebrafish embryos exposed to 4 azo dyes at different concentrations from 24 to 72 hours postfertilization (hpf). (A) tartrazine; (B) Sunset Yellow; (C) amaranth; (D) Allura red. (***) indicates statistical significance
Cardiac Edema and Heart Rate
Cardiac edema was found at concentrations of 10 to 50 mM (Figure 4). In the 50-mM exposure groups of tartrazine, Sunset Yellow, amaranth, and Allura red, the rates of cardiac edema at 72 hpf were 58.3%, 72.1%, 61.7%, and 47.5%, respectively.
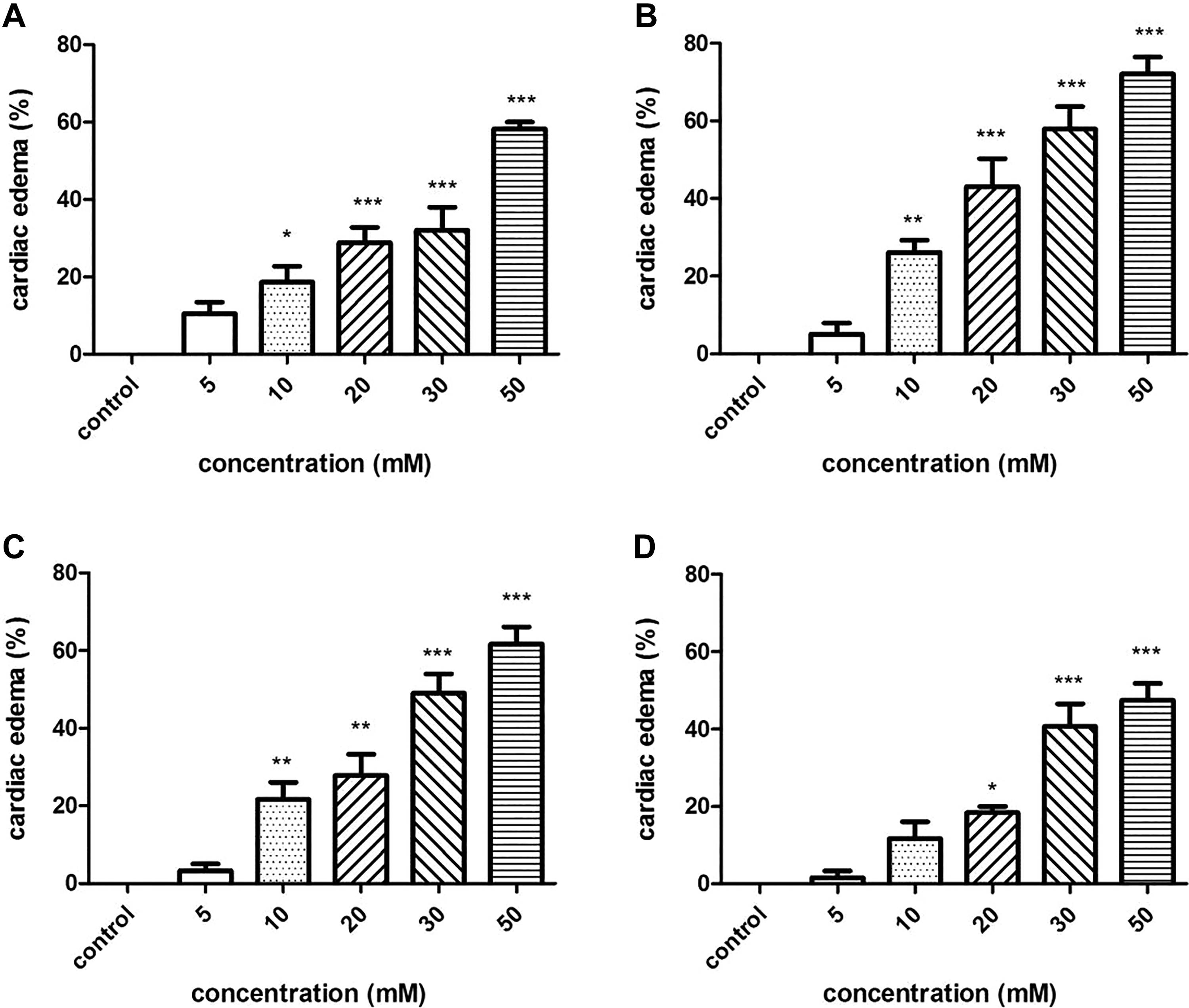
Cardiac edema in zebrafish embryos exposed to 4 azo dyes at different concentrations at 72 hours postfertilization (hpf). (A) tartrazine; (B) Sunset Yellow; (C) amaranth; (D) Allura red. (***) indicates statistical significance
At 72 hpf, the heart rate (beats per minute) of exposed embryos decreased at concentrations of 20 to 50 mM (Figure 5). In the 50-mM exposure groups of tartrazine, Sunset Yellow, amaranth, and Allura red, the heart beats per minute at 72 hpf were 90.7 ± 3.84, 65.3 ± 2.91, 82.3 ± 4.33, and 87.0 ± 7.00, respectively.
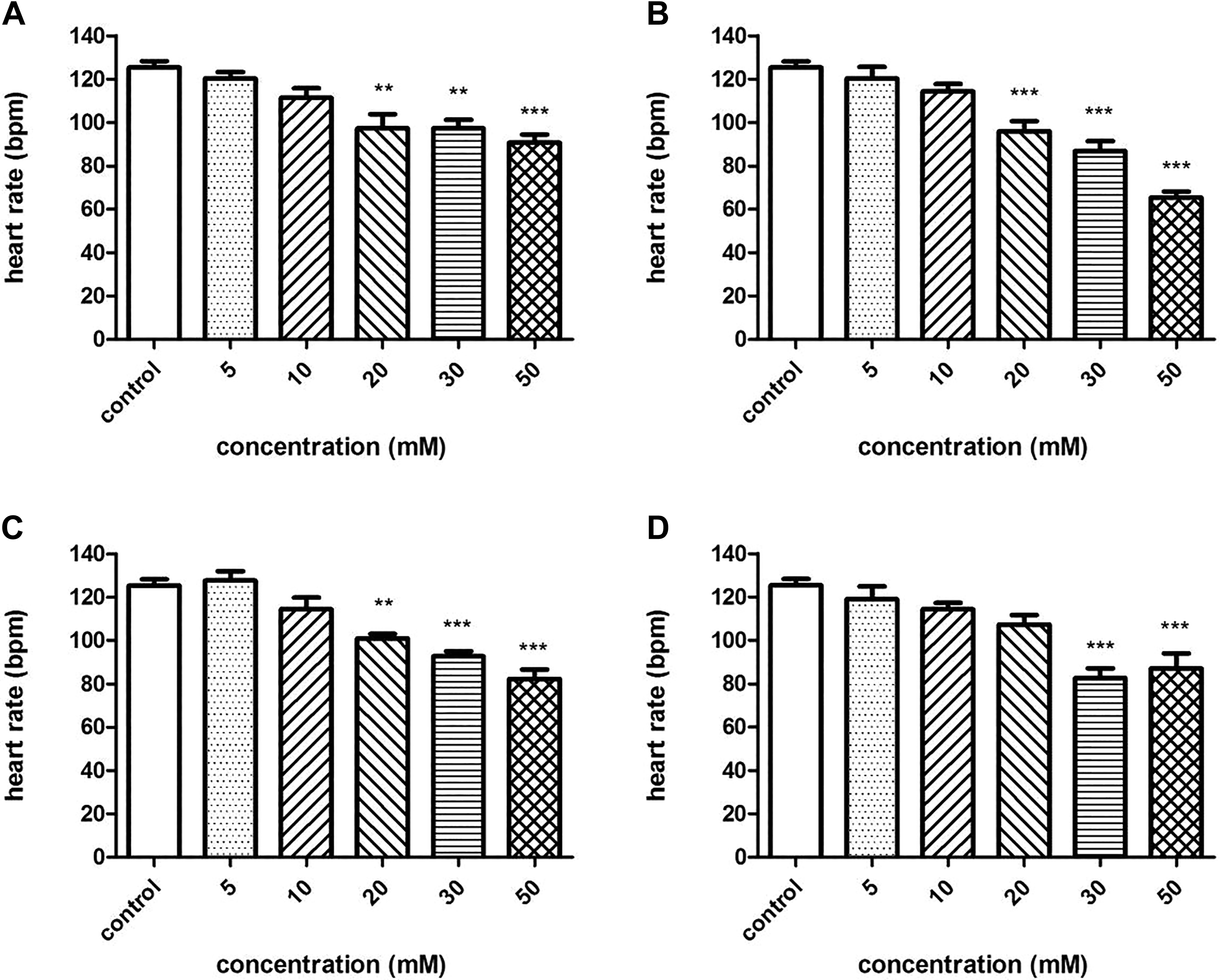
Heartbeats per minute in zebrafish embryos exposed to four azo dyes at different concentrations at 72 hours postfertilization (hpf). (A) tartrazine; (B) Sunset Yellow; (C) amaranth; (D) Allura red. bpm indicates beats per minute. (***) indicates statistical significance
Yolk Sac Edema
Yolk sac edema was observed at concentrations of 10 to 50 mM (Figure 6). In the 50-mM exposure groups of tartrazine, Sunset Yellow, amaranth, and Allura red, the rates of yolk sac edema at 72 hpf were 42.5%, 60.8%, 56.7%, and 35.8%, respectively.
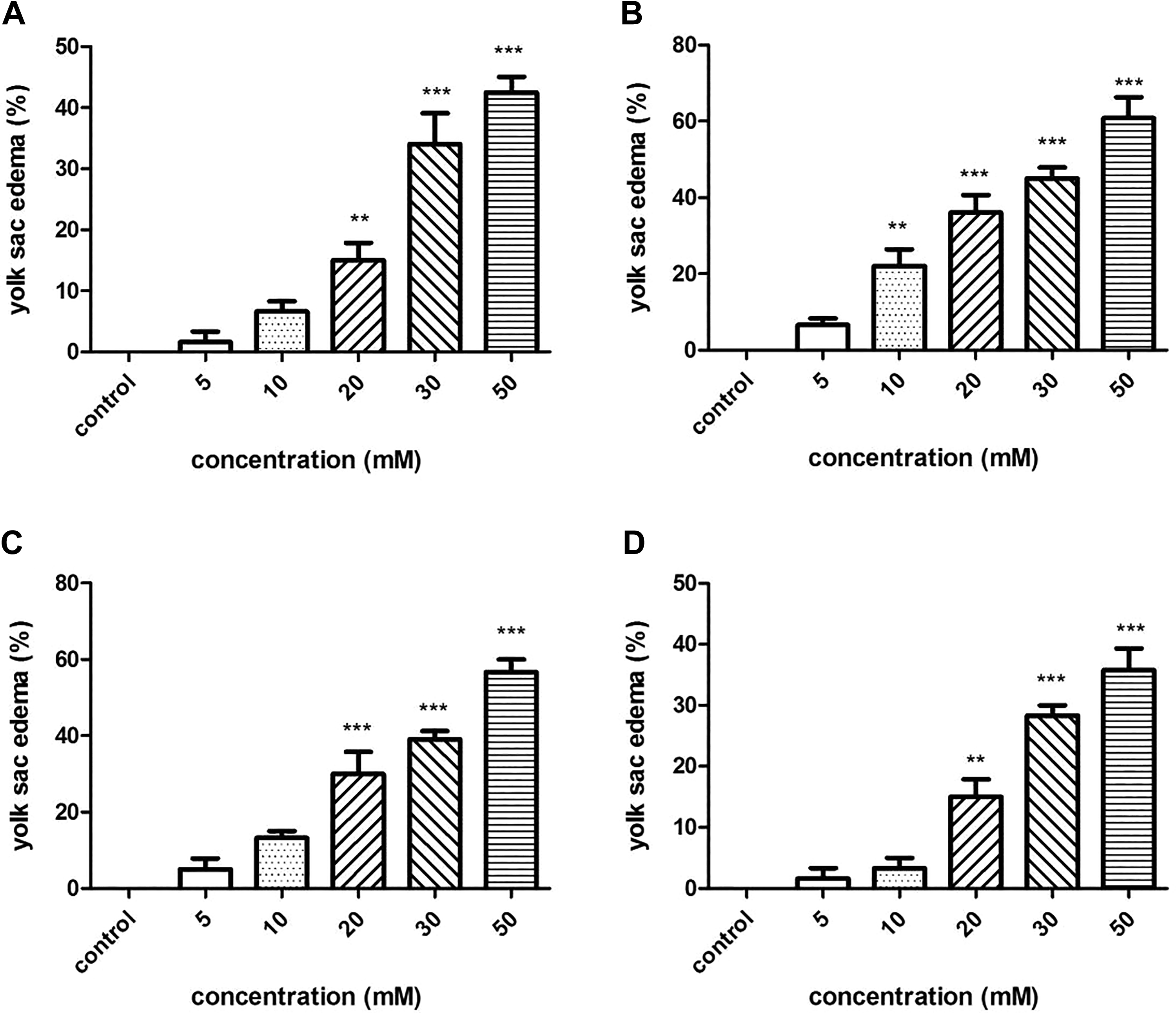
Yolk sac edema in zebrafish embryos exposed to four azo dyes at different concentrations at 72 hours postfertilization (hpf). (A) tartrazine; (B) Sunset Yellow; (C) amaranth; (D) Allura red. (***) indicates statistical significance
Spinal Defects
The spinal defects including spinal curvature and tail distortion were observed at concentrations of 10 to 50 mM (Figure 7). In the 50-mM exposure groups of tartrazine, Sunset Yellow, amaranth, and Allura red, the rates of spinal defects at 72 hpf were 34.4%, 58.3%, 40.9%, and 37.6%, respectively.
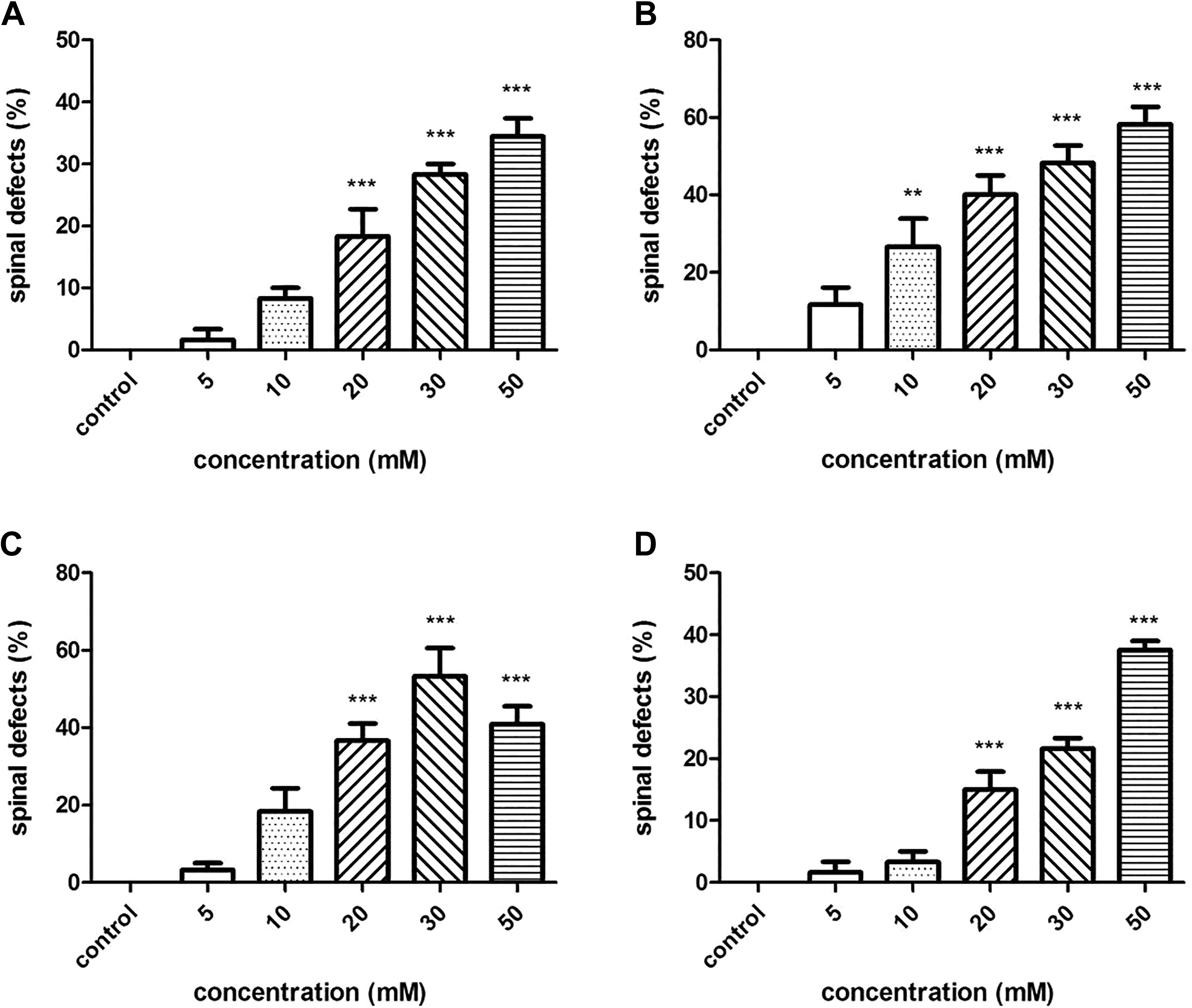
Spinal defects (spinal curvature and tail distortion) in zebrafish embryos exposed to 4 azo dyes at different concentrations at 72 hours postfertilization (hpf). (A) tartrazine; (B) Sunset Yellow; (C) amaranth; (D) Allura red. (***) indicates statistical significance
Median Lethal Concentration, EC50, and TI Value
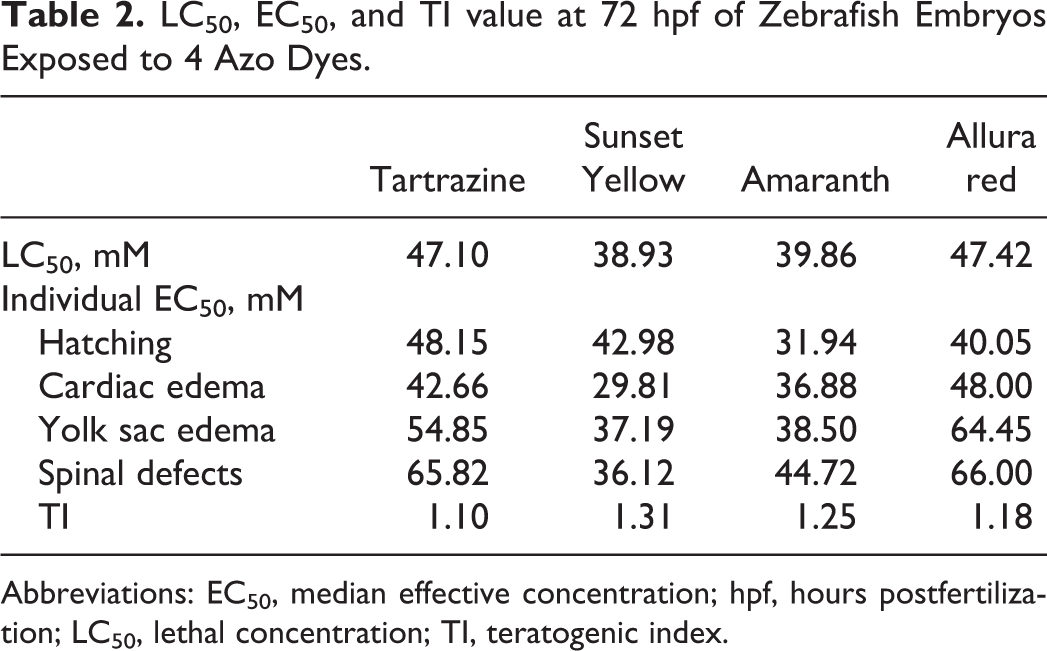
As shown on the mortality curve of experimental embryos in azo dye–exposed groups (Figure 8), LC50s of tartrazine, Sunset Yellow, amaranth, and Allura red at 72 hpf were 47.10, 38.93, 39.86, and 47.42 mM, respectively. Individual EC50s at 72 hpf were calculated for hatching rate, cardiac edema, yolk sac edema, and spinal defects, and the TI value based on the EC50 value of the most sensitive effect were shown for each azo dye in Table 2.
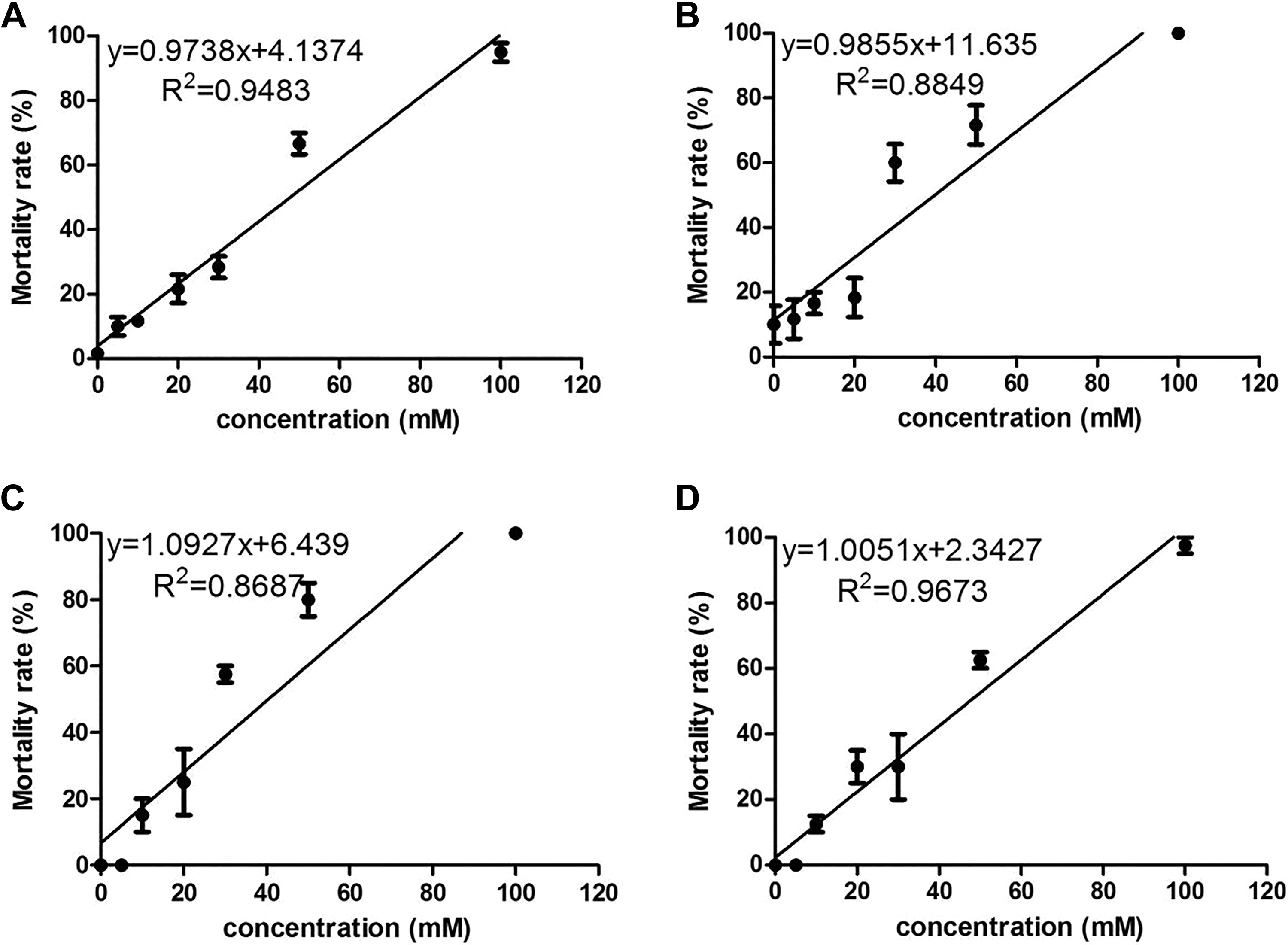
Mortality rate of zebrafish embryos exposed to four azo dyes at different concentrations at 72 hours postfertilization (hpf). (A) tartrazine; (B) Sunset Yellow; (C) amaranth; (D) Allura red.
LC50, EC50, and TI value at 72 hpf of Zebrafish Embryos Exposed to 4 Azo Dyes.
Abbreviations: EC50, median effective concentration; hpf, hours postfertilization; LC50, lethal concentration; TI, teratogenic index.
Discussion
With wide use as color additives in food, drugs, and cosmetics, the safety and potential postconsumption health hazards associated with azo dyes is an increasing concern. Among the azo dyes, tartrazine is the most common food colorant today. It was found to be nephrotoxic and hepatotoxic in rodents at a dose of 500 mg/kg body weight and has negative effects on fertility, reproductive performance at a dose level of 200 to 500 mg/kg body weight in mice and rats. 22,23 Sunset Yellow also is widely used in artificial coloring of food and drugs. It was reported to cause allergies and side effects in children by affecting the immune system followed a diet of 0.3 to 0.8 mg/kg body weight for 6 weeks and can induce hyperactivity and intolerance in individuals sensitive to salicylates. 24,25 Amaranth was a popular food coloring until 1976. It has been banned in many countries after numerous studies found that it can induce cancer, birth defects, and early fetal death at 200 or 1,000 mg/kg body weight in laboratory animals. 12,26 Allura red is one of the Food and Drug–approved color additives. It has been found to induce allergic reactions in individuals with preexisting moderate asthma, and its degradation products can cause bladder cancer in animals when orally given the dosage of 7.0 mg/kg body weight. 25,27 Therefore, the potential toxicity of azo dyes deserves more attention.
In the present study, we used zebrafish embryos to evaluate the toxicity of azo dyes by critically assessing the lethality, developmental defects, and teratogenic potential. The range of concentrations tested in zebrafish embryos was 0 to 100 mM (0-53.4 mg). It was reported that the typical use level of color additives in solid foods was 25 to 200 mg/kg and 10 to 300 mg/L in beverages, 27 and the maximum of acceptable daily intake (ADI) of the 4 azo dyes is 7.5 mg/kg body weight. 28 As the oral absorption of azo dyes is 2% to 5% in humans, their mean amount reaching the blood for an adult weighting 70 kg (7% of body weight, as well as 5 L of blood) would be approximately 2.1 to 5.25 μg/mL. 29 -31 Therefore, the zebrafish-exposed concentrations (0-53.4 mg) of azo dyes used in our study were higher than the expected human exposures at the ADI.
Our observations found that, a concentration of 5 mM (2.67 mg) was neither lethal to embryos nor induced noticeable morphological defects, the rates of which were low to 10%. At concentrations of 10 to 50 mM (5.34-26.7 mg), azo dyes can induce hatching difficulty and developmental abnormalities, such as cardiac edema, decreased heart rates, yolk sac edema, and spinal defects, including spinal curvature and tail distortion. At 100 mM (53.4 mg), there were few survivors in all exposed groups. These developmental toxicity values in zebrafish embryos proved that exceeding the permissible ADI for azo dyes may pose health risks, and teratogenicity and embryotoxicity may be of a concern for pregnant women.
Development can be extremely sensitive to exogenous substances. In zebrafish, hatching is commonly used for assessing the effects of chemicals on early development as an end point for successful completion of embryogenesis, leading to the release of free-swimming larvae. 32 Alteration in hatching is the result of biochemical and physical mechanisms. The activity of Hatching Enzyme 1 (HE1), an enzyme which is released by hatching gland cells (HGCs), plays an important role in the hatching process. It was reported that perturbing HGCs secretion could consequently affect HE1 choriolysin activity and then induce hatching failure. 33,34 Another explanation for hatching difficulty is neuromuscular deficits resulting in a weakened spontaneous muscular movement. 33 The decreased hatching rate has been observed in zebrafish embryos exposed to pharmaceuticals (including acetaminophen and auranofin), nanomaterials and nanoparticles, organic pesticides, and metal ions. 19,35,36
The heart is one of the first organs observed during zebrafish development. Heart rate is an important target for developmental toxicity in zebrafish, as an indicator of cardiac function. It was reported that zebrafish could actively regulate heartbeat and cardiac output in response to changes in the environment using both the parasympathetic and the sympathetic branches of the autonomic nervous system. 36 Furthermore, the decreased embryonic heartbeat would lead to a heart failure with the continuous exposure of azo dyes until 120 hpf. 37 Cardiac edema is a general response to toxicity owing to the interference in the water permeability barrier, and it has been reported to occur in zebrafish embryos exposed to different toxicants, such as organic pollutants and heavy metals. 13 The existence of a water permeability barrier surrounding the heart maintains the osmotic balance against the inward diffusion of water. 38 Alternatively, the edema may have occurred due to an impairment of renal excretion with a decrease of blood circulation and cardiac output 39 The cardiac edema and decreased embryonic heart rate observed in our study demonstrated that azo dyes may induce an impairment of cardiac function.
Likewise, yolk sac edema is also a commonly observed pathology in zebrafish developmental toxicity screens. The yolk sac environment is highly lipophilic and has low water and solute permeability. The embryo relies on a water barrier in the yolk sac to maintain the osmotic gradient compared to the surrounding aqueous environment. 40 Owing to their water solubility, azo dyes may impair the maintenance of the osmotic gradient, resulting in excessive water uptake into the embryo and subsequent edema formation.
In addition, spinal defects including spinal curvature and tail distortion were observed in exposed embryos. The neuronal and muscular development in zebrafish embryo is influenced by acetylcholine (Ach), acetylcholinesterase (AchE), and Ach receptor levels. Color additives containing azo dyes have been reported to influence the expression of nicotinic acetylcholine receptors, thereby affecting learning and memory behavior in resultant newborn rats. 41 Further, neuroinflammatory signals within cerebrospinal fluid may also be involved in the impairment of spinal nerve function caused by azo dyes. 42
In order to determine the teratogenic potential of the four azo dyes, we calculated the LC50, EC50, and TI values. As proposed by Weigt and colleagues, the TI is a useful parameter that allows comparisons of teratogenicity between different chemicals. 20 A TI value >1 indicates that the substance is considered to be teratogenic; when TI value is ≤1, the substance produces mainly embryolethality. 43 At 72 hpf, LC50s of tartrazine, Sunset Yellow, amaranth, and Allura red were 47.10 mM, 38.93 mM, 39.86 mM, and 47.42 mM, and the most sensitive EC50s of developmental malformation were 42.66 mM, 29.81 mM, 31.94 mM, and 40.05 mM, respectively. The TIs for tartrazine, Sunset Yellow, amaranth, and Allura red were 1.10, 1.31, 1.25, and 1.18, respectively. Therefore, the teratogenic potential of azo dyes has been confirmed, with Sunset Yellow and amaranth being the most teratogenic of these four azo dyes.
In conclusion, the present study reports the developmental toxicity potential of azo dyes in zebrafish embryos, although at concentrations higher than the expected human exposure after consumption of food and drugs containing azo dyes.
Footnotes
Author Contribution
L. L. Jiang contributed to conception and design and drafted manuscript; K. Li contributed to acquisition and analysis and critically revised manuscript; D. L. Yan and L. Ma contributed to analysis and critically revised manuscript; M. F. Yang and L.Z. contributed to interpretation and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Science and Technology Development Fund of Nanjing Medical University (2016NJMU050).
