Abstract
Lubricating oil base stocks (LOBs) are substances used in the manufacture of finished lubricants and greases. They are produced from residue remaining after atmospheric distillation of crude oil that is subsequently fractionated by vacuum distillation and additional refining steps. Initial LOB streams that have been produced by vacuum distillation but not further refined may contain polycyclic aromatic compounds (PACs) and may present carcinogenic hazards. In modern refineries, LOBs are further refined by multistep processes including solvent extraction and/or hydrogen treatment to reduce the levels of PACs and other undesirable constituents. Thus, mildly (insufficiently) refined LOBs are potentially more hazardous than more severely (sufficiently) refined LOBs. This article discusses the evaluation of LOBs using statistical models based on content of PACs; these models indicate that insufficiently refined LOBs (potentially carcinogenic LOBs) can also produce systemic and developmental effects with repeated dermal exposure. Experimental data were also obtained in ten 13-week dermal studies in rats, eight 4-week dermal studies in rabbits, and seven dermal developmental toxicity studies with sufficiently refined LOBs (noncarcinogenic and commonly marketed) in which no observed adverse effect levels for systemic toxicity and developmental toxicity were 1000 to 2000 mg/kg/d with dermal exposures, typically the highest dose tested. Results in both oral and inhalation developmental toxicity studies were similar. This absence of toxicologically relevant findings was consistent with lower PAC content of sufficiently refined LOBs. Based on data on reproductive organs with repeated dosing and parameters in developmental toxicity studies, sufficiently refined LOBs are likely to have little, if any, effect on reproductive parameters.
Introduction
Lubricating oil base stocks (LOBs, petroleum mineral oils) constitute 1 of 13 categories of petroleum substances that were sponsored by the Petroleum High Production Volume Testing Group (PHPVTG) in response to the High Production Volume (HPV) Challenge Program 1 of the United States Environmental Protection Agency (US EPA). These categories encompassed approximately 400 petroleum substances. This article reports previously unpublished data on the toxicological hazards of repeated exposures to LOBs.
Lubricating oil base stocks, derived from petroleum crude oil, include refinery streams with a range of potential toxicity depending on the extent of their refining. “Insufficiently refined” LOBs are intermediate refinery streams that contain polycyclic aromatic compounds (PACs) at levels that are known to present carcinogenic hazards. “Sufficiently refined” LOBs have much lower levels of PACs, are not considered carcinogenic, and are the starting liquid in the formulation of a wide array of lubricants. Finally, more intensively refined LOBs (white oils [WOs], also known as “highly refined” LOBs) may also be used in food-contact applications, pharmaceuticals, laxatives, body lotions, baby oils, cosmetics, or direct food additives.
The initial starting material for manufacture of LOBs is the residuum from the atmospheric distillation of crude oil. This complex material has constituents that boil principally above ∼650°F (343°C), including the higher boiling aromatic constituents that are present in crude oil. The atmospheric residuum is subsequently distilled under vacuum to separate these constituents further by boiling point into various distillate fractions and a residuum from vacuum distillation. These products of vacuum distillation are further refined to yield a set of related substances (base oils, WOs, waxes, aromatic extracts, and asphalt) that can be considered as related, not separate, entities.
The LOB fractions obtained directly from vacuum distillation are considered to be insufficiently refined and can contain a number of undesirable components that can negatively affect the performance of the LOB and must be removed. The undesirable components include heterocyclic aromatics and PACs. Polycyclic aromatic compounds are predominantly found in the distillate streams although, depending on the processing conditions, they can also be found in the overhead and residual streams. Most commonly, the PACs are removed by solvent extraction that selectively extracts compounds with 2 or more aromatic rings. In essence, PACs are transferred from the insufficiently refined LOBs to an aromatic extract, leaving sufficiently refined LOBs with low levels of PACs. The LOBs can also receive hydrogen treatment that is primarily used to reduce sulfur levels and can also reduce the levels of PACs under the appropriate processing conditions, usually by ring opening and saturation. Additional processing for finishing LOBs can also include “de-waxing” to remove long chain paraffins (waxes). The resulting “sufficiently refined” LOBs can be used to manufacture commercial products, primarily lubricants. Figure 1 shows a diagram of common refining steps and information from the current production of 2 sufficiently refined LOBs, including the extraction of PACs into a distillate aromatic extract (DAE).
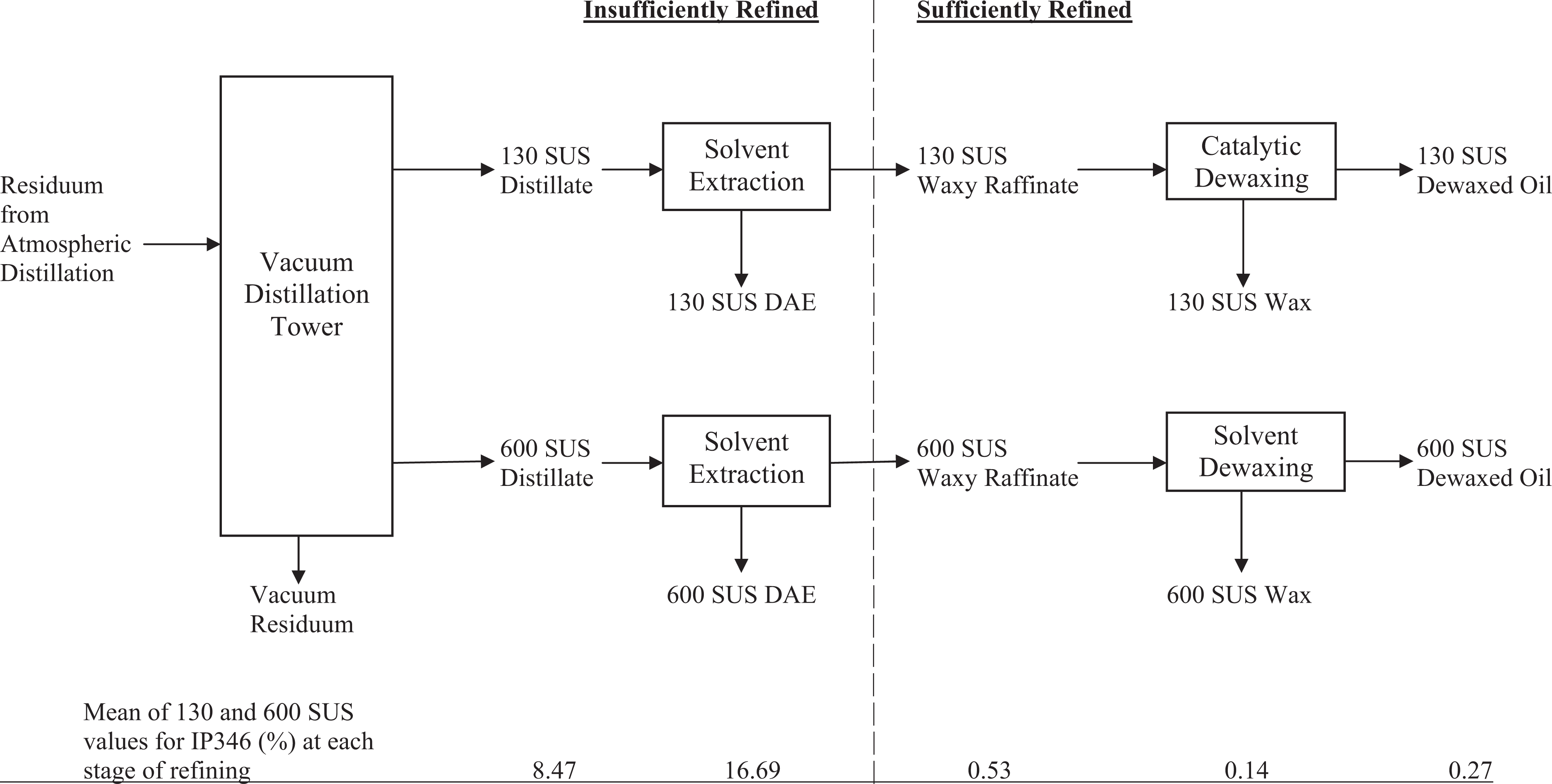
Schematic of refining process and mean IP346 values on 2 currently produced vacuum distillates and the refinery streams derived from them. IP indicates Institute of Petroleum.
In certain cases, the sufficiently refined LOBs receive additional treatment either by more vigorous hydrogenation or by acid oxidation/extraction to further reduce or eliminate aromatic constituents to produce WOs (highly refined LOBs), which meet the regulatory requirements for food contact and/or pharmaceutical applications. The Appendix contains information on the relevance of other groups of LOBs, namely, whether LOBs are distillate or residual and also whether the LOBs were derived from paraffinic or naphthenic crude oils.
As with other refinery streams, the definitions of individual Chemical Abstracts Services (CAS) numbers of LOBs are written in general terms due to the complex and variable nature of these substances. The definitions typically refer to the final process step a refinery stream has undergone rather than to chemical composition. Therefore, the severity of the various refining steps used to produce a LOB may not be apparent from the CAS number alone. As a result, knowledge of refining history or results of at least 1 screening test are commonly used to assess whether the LOBs have been sufficiently refined before their use in finished lubricants. Those LOBs that have not been assessed by testing or do not have well-defined refining histories typically are considered to have been insufficiently refined and are classified accordingly. This approach has been reviewed elsewhere. 2 -11 One short-term screening test involves measurement of the total amount of material extractable in dimethyl sulfoxide (DMSO) using Institute of Petroleum (IP) 346 12 as an index of PAC content. Oils with levels >3.0 weight% of DMSO-extractable material are considered to be potentially carcinogenic. In a second short-term assay, the optimized Ames test, a mutagenicity index (MI) is calculated as the initial linear slope of the dose–response curve in which dose is µL of a DMSO extract of the test substance per cultured plate of bacteria and response is the number of revertant colonies on the respective plates. 2,13 A value of “1” for the MI is commonly used as a cutoff for distillate streams, that is, samples for which MI ≥1 are considered potentially carcinogenic. 14
Although information was available on many aspects of health effects with LOBs, data on laboratory studies of subchronic and developmental toxicity were relatively limited. 15,16 This article provides such information, starting with potential effects of insufficiently refined LOBs. These LOBs were known to be carcinogenic when applied dermally. In addition, statistical models have been developed to predict the outcome of systemic and developmental toxicity tests from compositional information of many refinery streams. 17 -19 In essence, these models showed that the potential for systemic and developmental toxicity was associated with the aromatic ring class (ARC) profile of the types and levels of PACs. Given the known potential toxicity and available models to estimate that toxicity, additional studies on subchronic and developmental toxicity were not performed with insufficiently refined LOBs. Instead, the potential for systemic and developmental toxicity as predicted using these models for the representative insufficiently refined LOBs in Figure 1 and Table 1 is presented here. Note that both of the insufficiently refined LOBs in Table 1 had values of IP346 and “Total ARC weight %” that were well above 3% and MIs well above 1.0; thus, those samples can be considered potentially carcinogenic. (Method II, which was also used to derive the weight percent of DMSO-extractable material [“Total ARC weight %” in Table 1] is similar to the IP346 method.)
ARC Profile, IP346 Value, and MI for 2 Insufficiently Refined Distillate LOBs (130 SUS and 600 SUS) and Petroleum Streams Derived From Them.
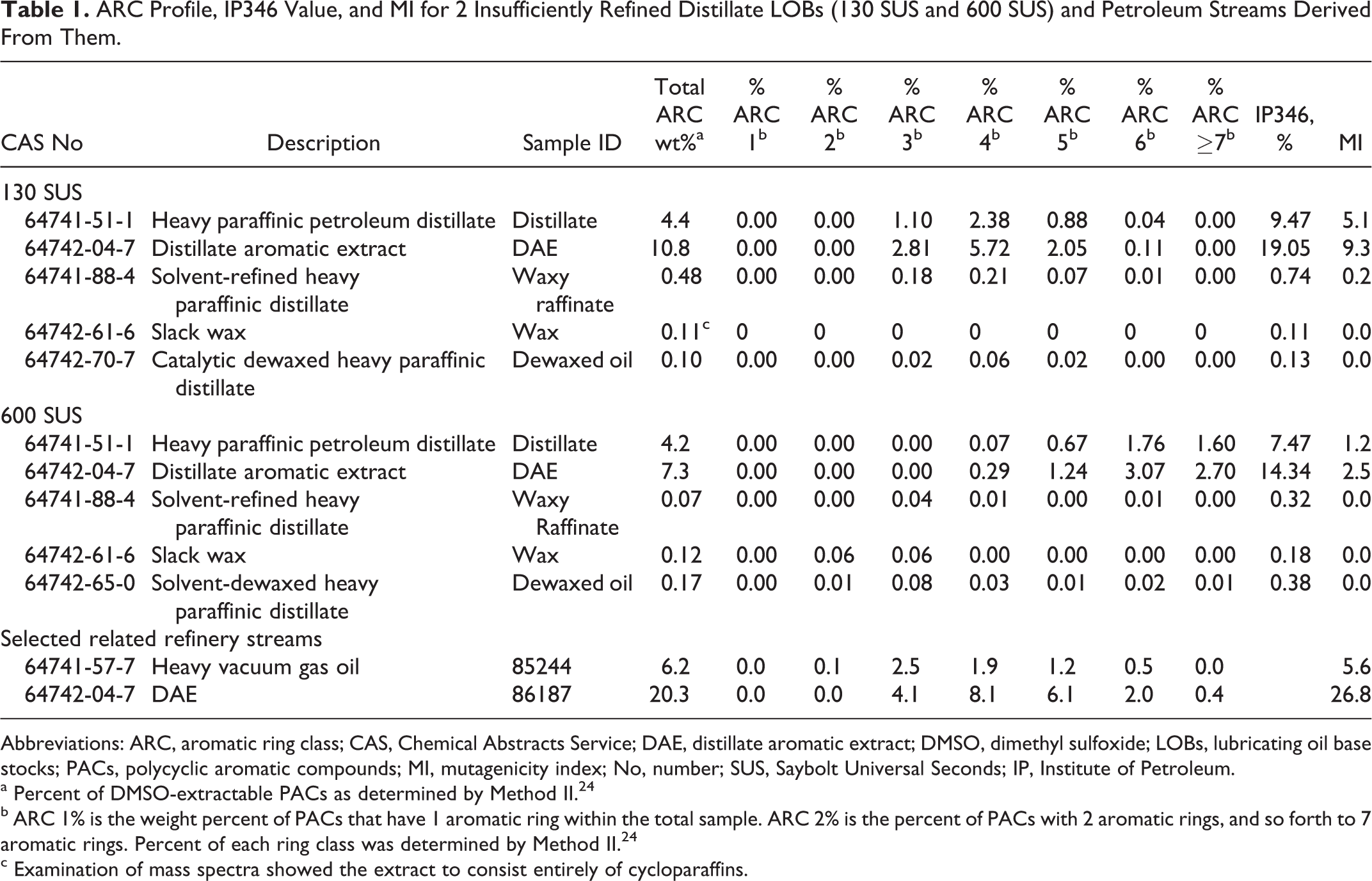
Abbreviations: ARC, aromatic ring class; CAS, Chemical Abstracts Service; DAE, distillate aromatic extract; DMSO, dimethyl sulfoxide; LOBs, lubricating oil base stocks; PACs, polycyclic aromatic compounds; MI, mutagenicity index; No, number; SUS, Saybolt Universal Seconds; IP, Institute of Petroleum.
a Percent of DMSO-extractable PACs as determined by Method II. 24
b ARC 1% is the weight percent of PACs that have 1 aromatic ring within the total sample. ARC 2% is the percent of PACs with 2 aromatic rings, and so forth to 7 aromatic rings. Percent of each ring class was determined by Method II. 24
c Examination of mass spectra showed the extract to consist entirely of cycloparaffins.
Although the term “sufficiently refined” originated in relation to carcinogenicity, on a practical basis, it can be applied to both systemic and developmental effects. In other words, it is hypothesized that exposure to a sufficiently refined LOB would not result in significant systemic or developmental toxicity. Inherent in this approach is the premise (primarily based on Feuston et al 20 ) that PACs in the oil are responsible for both target organ effects and developmental effects and that the levels of biologically active PACs are so low in sufficiently refined LOBs that the hazards of systemic and/or developmental toxicity are minimal. This premise was investigated in several studies reported here on developmental toxicity and repeated-dose exposures.
These studies were focused primarily on dermal exposures to sufficiently refined LOBs since exposure via the skin is expected to be the main type of contact that would occur during normal use of these products. Additional justification for the use of dermal dosing is provided in the Appendix. A few studies reported here were performed with oral or inhalation exposure, and those routes are discussed further.
In addition, information by which it was possible to assess the potential for reproductive toxicity of LOBs was derived from these and additional studies and is discussed here. Finally, results from acute toxicity tests on LOBs, compiled from company records, are summarized here to complete the profile of information which is required to fulfill HPV human health obligations for this category of substances. Data on acute oral and dermal toxicity of other paraffinic and naphthenic base oils have been reported previously. 15,21 All LOBs in the acute studies discussed herein were sufficiently refined except for American Petroleum Institute (API) 83-12.
Materials and Methods
Acute Toxicity Studies With Oral, Dermal, and Inhalation Exposures
Oral and dermal toxicity tests were performed on 4 LOBs, namely, API 83-12, S-141, S-142, and S-150. In the acute oral tests, rats were observed daily after a single oral gavage. Body weights were measured periodically and animals were necropsied at terminal sacrifice. Acute dermal toxicity was evaluated in New Zealand white rabbits. One dose was given for 24 hours under an occlusive dressing. Animals were observed daily for 2 weeks with periodic measurement of body weight and necropsy at terminal sacrifice. Additional details of the studies are available. 22
Acute inhalation studies were conducted on 7 LOBs. For the first LOB (API 83-12), groups of rats (5/sex) were exposed for 4 hours to aerosolized LOB at concentrations of 1.0, 1.5, 2.4, 3.5, or 5.0 mg/L. Clinical observations were made daily during the following 2 weeks, and body weights were recorded weekly. Surviving animals were necropsied on day 14. This initial study was followed by studies of 3 LOBs (87-099, 87-101, and 87-102) in which groups of Sprague-Dawley rats (5/sex) were exposed for 4 hours for each aerosolized sample. Sham-exposed controls were provided. End points included daily clinical observation, weekly body weights, and necropsy at terminal sacrifice. Finally, studies were conducted for 3 sufficiently refined LOBs (S-141, S-142, and S-461) in which groups of Sprague-Dawley rats (10/sex) were exposed for 4 hours to 3 aerosol concentrations of each LOB. Half of each group (5/sex) was sacrificed on the day after exposure; the remaining half was observed for 2 weeks before sacrifice. End points included daily clinical observations, periodic body weights, necropsy, weights of liver, kidney, and lung (wet and dry), and histopathology of nose, lung, liver, kidney, and thoracic lymph nodes.
Modeled Predictions for Subchronic and Developmental Toxicity Studies With Dermal Exposures to Insufficiently Refined LOBs
Polycyclic aromatic compounds in the DMSO extracts of 2 insufficiently refined LOBs and their related petroleum streams were assessed by Mobil method II as described in more detail subsequently. Both starting samples were heavy paraffinic petroleum distillates from current production; the viscosity of 1 sample was 130 Saybolt Universal Seconds (SUS) and the other was 600 SUS. Additional samples of petroleum streams were taken at each stage of subsequent refining as the insufficiently refined LOBs were solvent extracted to remove aromatic constituents, and the resulting waxy raffinate (a solvent-refined heavy paraffinic distillate) was treated to remove long-chain paraffinic constituents to yield a final dewaxed LOB (Figure 1).
Detailed descriptions of the Mobil method II can be found elsewhere. 4,23,24 Briefly, each sample was initially extracted with DMSO, further purified, and analyzed by gas chromatography/mass spectrometry (GC/MS). The GC/MS chromatograms of the extracts were integrated in the slice mode. Data were reported by ring number of the PACs (1-ring, 2-ring, etc) and given as fractions of the starting material.
The resulting ARC profiles were then used in statistical models to predict the dose associated with a 10% difference from controls in characteristic target organ effects associated with repeated exposure to a range of PAC-containing refinery streams, namely, increased liver weights, decreased thymus weights, decreased platelet counts, and decreased hemoglobin (Hb) concentration. The models were also used to predict 10% differences in developmental parameters, including reductions in fetal body weight, reductions in percent offspring live-born, and increased percentage of resorptions. The predictions were based on a series of statistically developed empirical models described elsewhere, 17 and the end points were identified in an extensive analysis of data from several studies on petroleum streams as the most sensitive end points. 18,19 The predicted dose was identified as the predicted dose–response at 10% (PDR10). The lowest PDR10 in each study for each sample was identified as the “sample PDR10” for that study.
Subchronic Studies With Dermal Exposures to Sufficiently Refined LOBs
Subchronic (13 weeks) dermal studies in Sprague-Dawley rats were performed over several years on 10 sufficiently refined LOBs. Descriptions of these LOBs are given in Table 2. The designs of these studies were essentially similar although there were differences as described subsequently. The LOBs were applied to the clipped backs of the rats, treated sites were left uncovered, and animals wore Elizabethan collars to minimize ingestion of the LOBs. Each study included a sham-treated control group. Dosing was done 5 days/week. Clinical signs were evaluated daily and body weight was measured weekly. Evaluated end points included clinical chemistry (∼18 parameters), hematology (white blood cell [WBC] number, WBC differentials, hematocrit (Hct), Hb, red blood cell [RBC] number, and associated parameters), and organ weights (adrenals, gonads, heart, kidneys, liver, spleen, and thymus). Statistical analyses included analysis of variance (ANOVA), Dunnett or Duncan test, and chi-square test for all studies except those with WOLCCD and WOULSDF in which Student t test was used. A summary of other differences in the conduct of these studies in rats is given in Table 3.
Information on Samples of LOBs Used in Subchronic Dermal Toxicity Tests.
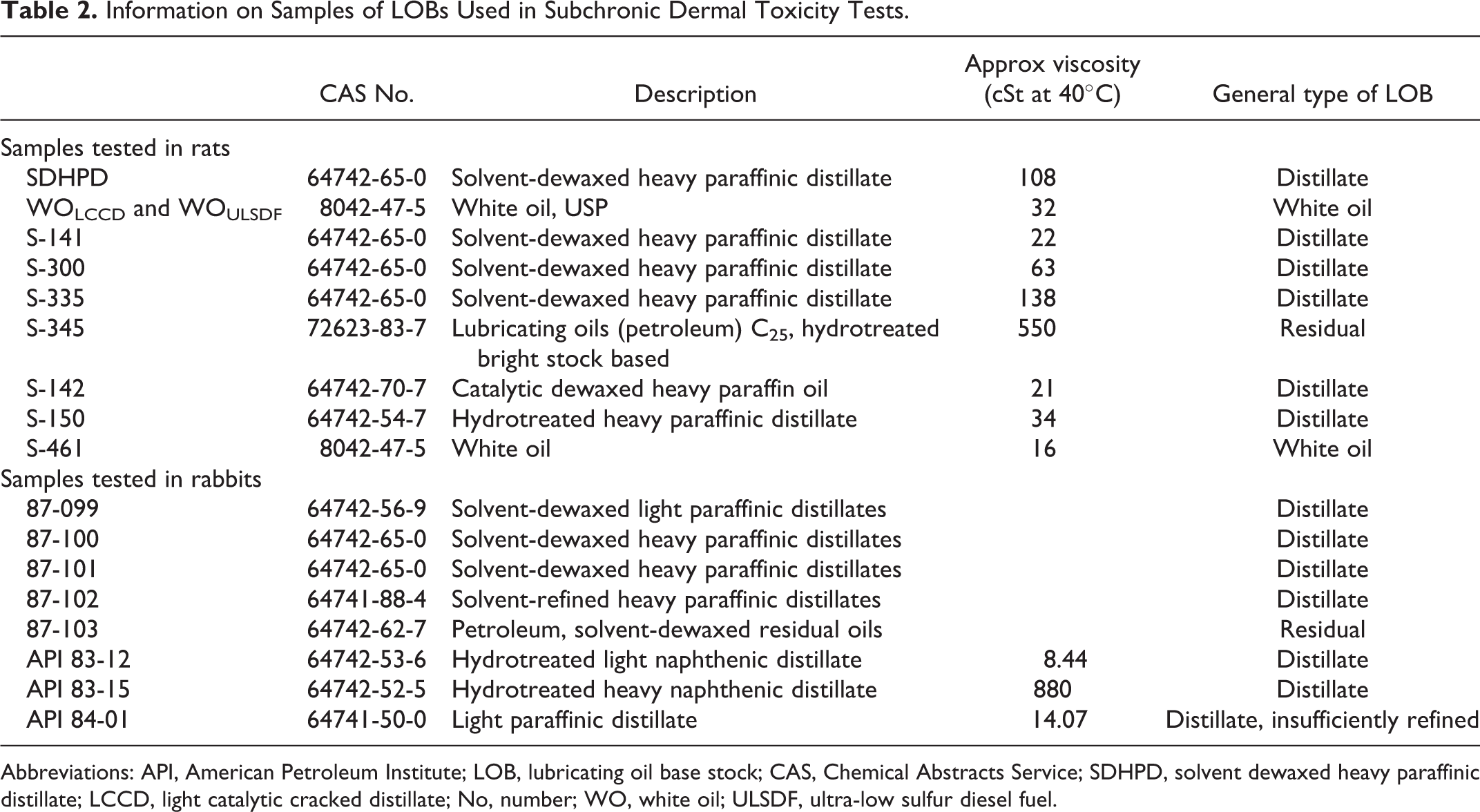
Abbreviations: API, American Petroleum Institute; LOB, lubricating oil base stock; CAS, Chemical Abstracts Service; SDHPD, solvent dewaxed heavy paraffinic distillate; LCCD, light catalytic cracked distillate; No, number; WO, white oil; ULSDF, ultra-low sulfur diesel fuel.
Summary of Dosing Procedures and Selected Measured End Points in 13-Week Dermal Studies in Sprague-Dawley rats.a
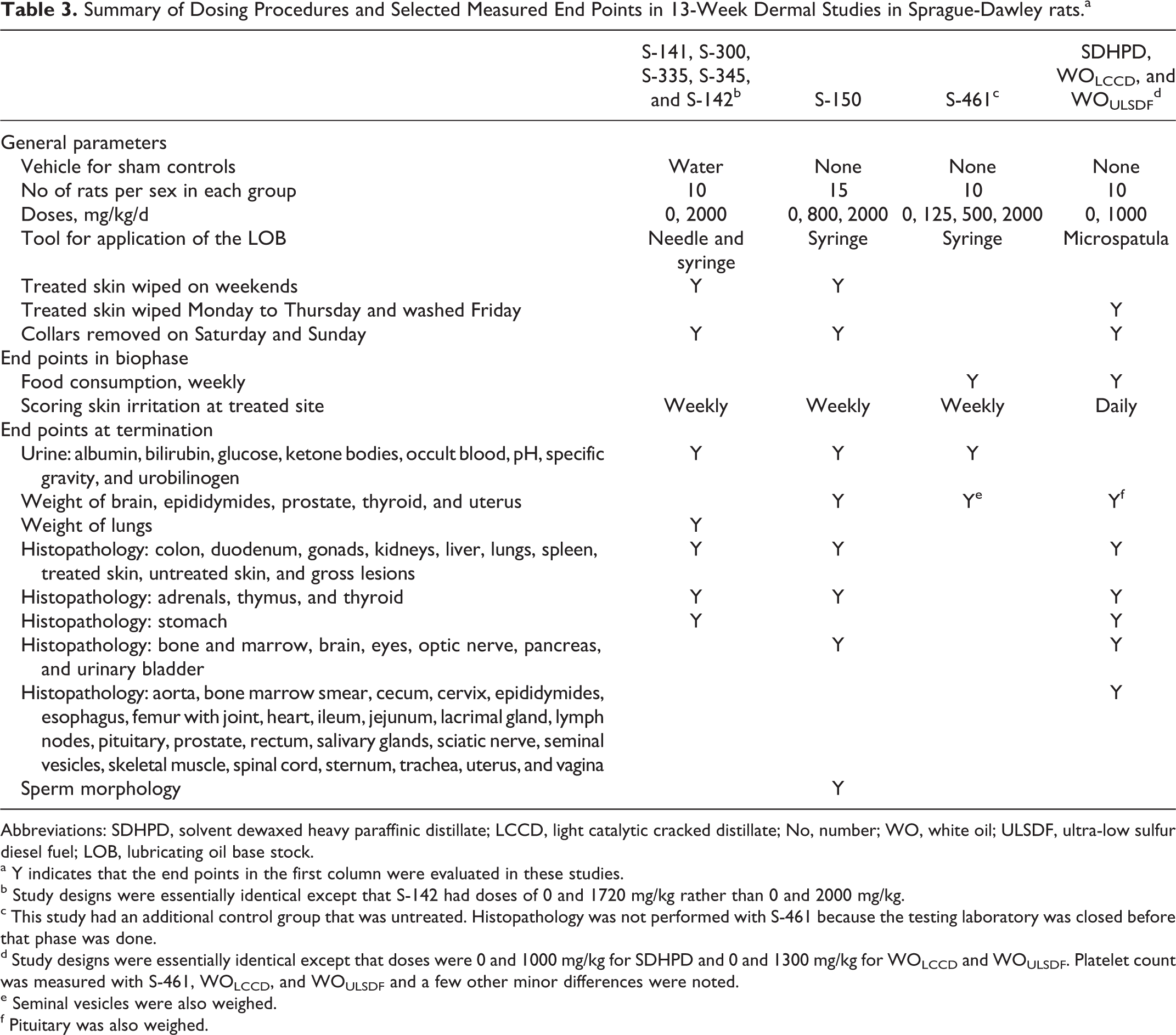
Abbreviations: SDHPD, solvent dewaxed heavy paraffinic distillate; LCCD, light catalytic cracked distillate; No, number; WO, white oil; ULSDF, ultra-low sulfur diesel fuel; LOB, lubricating oil base stock.
a Y indicates that the end points in the first column were evaluated in these studies.
b Study designs were essentially identical except that S-142 had doses of 0 and 1720 mg/kg rather than 0 and 2000 mg/kg.
c This study had an additional control group that was untreated. Histopathology was not performed with S-461 because the testing laboratory was closed before that phase was done.
d Study designs were essentially identical except that doses were 0 and 1000 mg/kg for SDHPD and 0 and 1300 mg/kg for WOLCCD and WOULSDF. Platelet count was measured with S-461, WOLCCD, and WOULSDF and a few other minor differences were noted.
e Seminal vesicles were also weighed.
f Pituitary was also weighed.
The most recent of these studies was performed with a solvent dewaxed heavy paraffinic distillate (SDHPD, CAS No 64742-65-0) that had been refined by solvent extraction to remove the PAC constituents. No DMSO-extractable material was found in the analysis of this sample by Method II. The study was conducted in accordance with the Office of Chemical Safety and Pollution Prevention (OCSPP) Guideline 870.3250 and Organization for Economic Cooperation and Development (OECD) Guideline 411. Solvent dewaxed heavy paraffinic distillate was applied undiluted at a dose of 1.14 mL/kg (1000 mg/kg spread over ∼10% of body surface). The application areas were gently washed on Fridays after a 6-hour exposure. The washing was done with a mild soap solution to remove any residual test material, followed by a rinse with deionized water and drying. Starting on study day 35 and occurring on each dosing day thereafter, each animal was wiped gently with a dry paper towel to remove residual test substance after a 6-hour exposure period. The treated skin was scored following the method of Draize. 25
Two additional studies in rats were not designed specifically to address the toxicity of LOBs but did yield useful data. These 13-week studies were conducted on light catalytic cracked distillates (LCCDs) and ultra-low sulfur diesel fuel (ULSDF). Although these test substances were not LOBs, each test substance was diluted in a LOB (WO) for the purpose of reducing dermal irritation that might confound the assessment of systemic toxicity. Both a vehicle control group (receiving only WO at 1300 mg/kg/d) and a sham-dosed control group (no WO) were included in each study. Vehicle controls were compared to the sham-dosed controls. The results represent 2 identical studies in which groups dosed dermally with the same WO were compared to respective sham-dosed controls. The studies were conducted in accordance with OCSPP Guideline 870.3250 and OECD Guidelines 411 and 474. The same sample of US Pharmacopoeia WO was used as the vehicle in both the studies (CAS No 8042-47-5). No DMSO-extractable material was seen in this sample by analysis with Method II. Both sham-dosed and vehicle control groups were subjected to the same procedures except that no WO was applied to the sham controls. Further details on the conduct of the studies are given in Table 3. For clarity, the data for the WO in each study are shown using the name of the respective test substance in that study, that is WOLCCD and WOULSDF.
Seven other 13-week studies were performed in rats over several years. Each of these LOBs was considered to be sufficiently refined based on process history or appropriate testing (eg, optimized Ames or skin-painting tests). Performed earlier than the study on SDHPD, these studies were essentially comparable to the one on SDHPD but did not include all of the same specific end points, particularly in regard to histopathological examination of organs. Further details are given in Table 3.
In addition to the studies performed with rats, 5 LOBs (samples 87-099 through 87-103 in Table 2) were tested via dermal application in groups of 10 New Zealand white rabbits (5/sex) at doses of 0 (sham-dosed controls) and 1000 mg/kg/d on 5 days/week for 4 weeks. Finally, 4-week studies were performed using New Zealand White rabbits with 3 LOBs (API 83-12, 83-15, and 84-01 in Table 2). In each study, groups of animals (5/sex) were treated dermally 3 times per week with doses of 0, 200, 1000, or 2000 mg/kg. Additional details on these studies are provided in the Appendix.
Developmental Toxicity Studies With Dermal Exposures to Sufficiently Refined LOBs
A developmental toxicity study using Sprague-Dawley rats was conducted with SDHPD in accordance with OCSPP 870.3700 and OECD Guideline 414. Groups of 25 pregnant females were dosed with 0 (sham-treated controls) or 1000 mg/kg/d daily from gestational day (GD) 0 through 19. As with the subchronic studies, all animals wore Elizabethan collars. Among the end points evaluated at necropsy of the dams on GD 20, adrenals and thymus glands were weighed. The uteri and ovaries from all dams were excised and weighed, the number of both corpora lutea and implantation sites was recorded, and the uterine contents examined. Uteri with no apparent evidence of implantation were opened and placed in 10% ammonium sulfide solution for detection of early implantation loss. 26 Each viable fetus was examined externally, individually sexed, and euthanized. The crown-rump length was determined for each fetus. The internal examinations of the viable fetuses followed the methods of Stuckhardt and Poppe 27 and included fresh dissection to assess the heart and major blood vessels. The sex of each fetus was confirmed by internal examination. Fetal kidneys were examined and graded for renal papillae development. 28 Heads from approximately one-half of the fetuses in each litter were placed in Bouin fixative for subsequent soft-tissue examination by the Wilson sectioning technique. 29 The heads from the remaining one-half of the fetuses were examined by mid-coronal slice. All carcasses were eviscerated and fixed in 100% ethyl alcohol. Following fixation, each fetus was macerated in potassium hydroxide and stained with Alizarin Red S and Alcian Blue. 30 Fetuses were then examined for skeletal malformations and developmental variations. External, visceral, and skeletal findings were recorded as either malformations or variations. Statistical analyses included ANOVA, Dunnett test, 2-sample t test, or Kruskal-Wallis nonparametric ANOVA, as appropriate.
As with the subchronic dermal studies, 2 developmental toxicity studies were performed on LCCD and ULSDF. Again these test substances were diluted with the same WO to reduce dermal irritation, and both a vehicle control group (receiving only WO at 1300 mg/kg/d) and a sham-dosed control group (no WO) were included in each study. The sample of WO was the same as that used in the related subchronic studies. The methods of dosing, necropsy, fetal evaluations, and statistical analyses were the same as with SDHPD with the addition of maternal liver weight. The results represent 2 identical developmental toxicity studies with female Sprague-Dawley rats dosed dermally with the same WO and compared to the respective sham-dosed controls.
Similar developmental toxicity studies on a sufficiently refined LOB (S-141) and a white mineral oil (S-461) were also performed. Although generally comparable to the study on SDHPD, doses in the study on S-461 were 0 and 2000 mg/kg/d on GD 6 through 19, group size was 20 females, and gravid uterine weight was not measured. Clinical chemistry was assessed in dams at necropsy (∼21 parameters). The methods used for evaluation of fetuses also differed. Half of the fetuses were preserved in Bouin fixative and examined for alterations in soft tissues by use of a modified Wilson technique. The other half of the fetuses were stained, cleared, and examined for skeletal changes. The study design with S-141 was the same as that for S-461 except that dosing was on GD 0 through 19, group size was 15 dams, and gravid uterine weight was measured. This study also had additional groups of 10 females that received 125 or 500 mg/kg/d. Statistical analyses included ANOVA and Fisher exact test or Dunnett test, as appropriate.
Developmental Toxicity Study With Inhalation or Oral Exposures to Sufficiently Refined LOBs
A white mineral oil (S-461) was tested by inhalation in a developmental toxicity study. A group of 20 pregnant female rats was exposed by inhalation to 1000 mg/m3 for 6 hours/d. Animals were housed individually and exposed whole body. The aerosol was generated with a Laskin nebulizer; large particles were removed before the aerosol entered the 400-liter inhalation chamber. The aerosol concentration was monitored gravimetrically and particle size was measured by cascade impaction. End points were the same as in the dermal developmental toxicity study with S-461. A sham-exposed group of 20 females was included.
In an oral study with a similar design, S-461 was also tested . The LOB was administered orally to a group of 21 pregnant female rats at a dose of 5000 mg/kg/d on GD 6-19. Animals were dosed using a 3 cm3 syringe fitted with a 16-gauge stainless steel gavage needle. End points were the same as in the dermal developmental toxicity study with S-461 and a sham-dosed group of 20 females was included.
Reproductive Toxicity With Dermal Exposures to Sufficiently Refined LOBs
No 2-generation reproductive toxicity studies were identified for LOBs. However, for purposes of the HPV Program, guidance from the US EPA indicates that the requirements to assess the potential for reproductive toxicity can be satisfied by data from (1) a developmental toxicity study and (2) a 90-day repeated-dose study in which the potential for effects on reproductive organs was assessed. 31 A summary of the relevant data from the present studies is given in the Results section.
In addition, a reproductive study was conducted with a white mineral oil (S-461) using a study design similar to OECD Guideline 415 (One-Generation Reproduction Toxicity Study). Differences from the guidelines included the use of 2000 mg/kg rather than the limit dose of 1000 mg/kg and administration of doses 5 times per week during much of the study rather than 7 times per week. Dermal doses applied to groups of Sprague-Dawley rats (20/sex) were 0 (untreated controls), 0 (sham-exposed controls), 125, 500, and 2000 mg/kg/d. The test substance was applied to the shaved backs of both males and females, and the sites were left uncovered. Elizabethan collars were used. Dosing began approximately 10 weeks before mating and continued during mating. Dosing of females was daily during gestation and then 5 days/week during a 3-week postpartum period. Dams were sacrificed on day 21 of lactation. Dosing of males was 5 days/week throughout, but half of the males were sacrificed after mating while the remainder was sacrificed within 2 weeks of the last sacrifice of pups (ie, during postpartum weeks 5 and 6).
Maternal body weights were measured weekly during premating and at intervals during gestation and lactation. Maternal food consumption was measured at intervals during premating and gestation. Females that did not deliver were sacrificed on GD 25 and necropsied. Females that delivered were sacrificed on postpartum day 21 and necropsied. At necropsy, ovaries and uteri were examined grossly, weighed, and preserved. The number of implantations and any remarkable findings were recorded. In addition, the estrus cycle was followed 5 days/week in 5 females in the untreated controls, sham-exposed controls, and rats dosed with 2000 mg/kg for 2 weeks prior to mating and during mating until breeding activity began. All offspring were observed individually during the postpartum period until sacrifice for body weight, behavior, and appearance. All viable neonates were examined as early as possible for sex and external anomalies. Litters of sufficient size were culled to 8 pups on postpartum day 4 (4/sex if possible). The number of open eyelids for each pup was recorded on postpartum day 10 and continued until both eyelids were open. All pups were tested for surface righting reflex on postpartum day 14. Pups were weaned on postpartum days 21 and then sacrificed and necropsied with gross observations on postpartum day 28. Data from the gestation and postpartum phases were analyzed with ANOVA followed by group comparisons using Fisher exact test or Dunnett test.
Results
Acute Toxicity Studies With Oral, Dermal, and Inhalation Exposures
Results of the acute toxicity studies are summarized in Table 4. Minimal systemic effects were seen in most of the oral and dermal studies. In the acute inhalation studies, no significant treatment-related toxicity was seen with S-141, S-150, or S-461. Therefore, the lethal concentration 50 (LC50) values are shown as greater than the highest concentration that was tested for each sample. No significant effects were seen in clinical signs, body weight, or observations at necropsy with acute inhalation exposures to 87-101 or 87-102. With 87-099, reddened skin after exposure, loss of body weight between days 0 and 7 with gain in body weight from days 7 to 14, and discolored lungs in a few animals at necropsy were seen. This sample also has the largest mass median aerodynamic diameter (MMAD) and the widest range of aerosol size (geometric standard deviation, GSD), possibly resulting in greater deposition of the aerosol on the fur.
Summary of Data on Acute Toxicity of LOBs in Mammals.
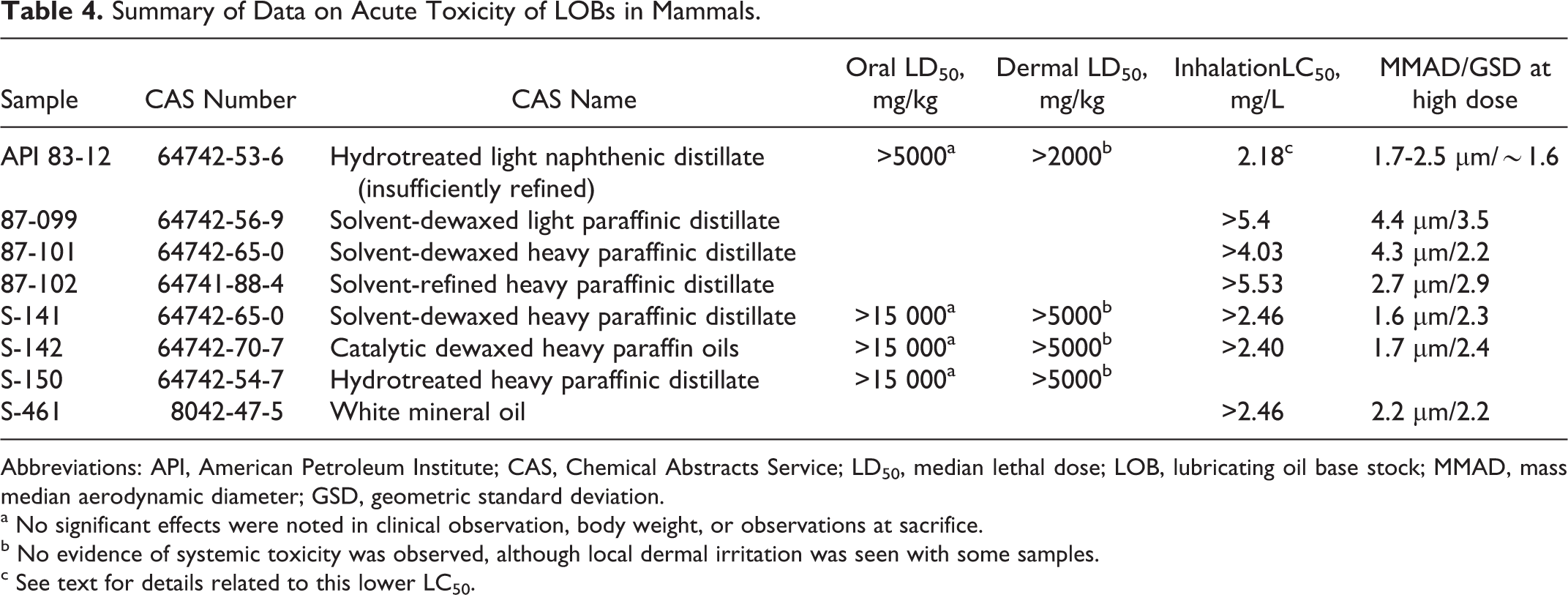
Abbreviations: API, American Petroleum Institute; CAS, Chemical Abstracts Service; LD50, median lethal dose; LOB, lubricating oil base stock; MMAD, mass median aerodynamic diameter; GSD, geometric standard deviation.
a No significant effects were noted in clinical observation, body weight, or observations at sacrifice.
b No evidence of systemic toxicity was observed, although local dermal irritation was seen with some samples.
c See text for details related to this lower LC50.
The LC50 in the acute inhalation study with API 83-12 was low (2.18 mg/L) when compared to the other LOB samples tested. The MMAD of the aerosolized LOB ranged from 1.7 to 2.5 µm among the 5 groups; the GSDs were ∼1.6. Body weight differences did not show a consistent dose-related pattern. Dose-related decreased activity, wet inguinal area, eyes partially closed, wet coat, loose stool, and oily coat were seen during exposure. Similar signs, poor condition, respiratory distress, and some deaths occurred during the first week after exposure. Most survivors appeared normal by the second week. Dark red lungs were described for some animals at necropsy; the incidence ranged from 2 of 10 animals at 1.0 mg/L to 10 of 10 animals at 3.5 mg/L. Histologically, affected animals exhibited diffuse pulmonary congestion and perivascular edema that were mostly moderate or marked in degree. Less consistently spotty alveolar edema was also seen. There was widespread damage to alveolar walls resulting in fibronecrotic debris resembling hyaline membranes in more marked cases and extravasation of RBCs and polymorphonuclear leukocytes. Necrosis and inflammation were seen in the walls of small blood vessels and there was spotty epithelial necrosis in small bronchioles, but the most severe damage seemed to be centroacinar. The larger airways were relatively unaffected. None of the surviving animals exhibited these acute changes. However, most of the surviving animals exposed to 2.4 or 1.0 mg/L and above exhibited chronic inflammatory changes that were not seen in the controls and only occasionally in animals exposed at the 1.5 mg/L level, and then to a lesser degree of severity.
Modeled Predictions for Subchronic and Developmental Toxicity Studies With Dermal Exposures to Insufficiently Refined LOBs
The goal of obtaining MI, IP346, and analyses of PACs for the 2 insufficiently refined LOBs and their subsequent refining streams (Figure 1) was to demonstrate the removal of PACs during refining and the concomitant reduction in toxicity as the final sufficiently refined LOBs were prepared. These samples were identified by the viscosity of starting insufficiently refined LOB (130 and 600 SUS). Reductions in MI and IP346 are apparent as shown in Table 1. In addition, means of the 2 IP346 values are shown at each refining step in Figure 1. Since solvent extraction of the raw distillate removes many of the aromatic constituents and concentrates them in a DAE, the IP346 values increase in the DAE compared to the initial distillate. The oils that remain after removal of the aromatics (the raffinates) are sufficiently refined based on IP346 and MI, and both the waxes and the dewaxed oils derived from the raffinates are sufficiently refined (Table 1 and Figure 1). These data provide a useful illustration of the efficacy of modern refining practices in the production of LOBs.
As stated previously, further testing of the systemic or developmental toxicity of insufficiently refined LOBs was not considered appropriate since they are already known to be carcinogenic and are used only under controlled conditions. Instead of testing, the potential for systemic and developmental toxicity was predicted using the data from the PAC profiles shown in Table 1. The PDR10s for each sensitive end point with subchronic dermal exposures were calculated and are shown in Table 5. The sample PDR10 values for the insufficiently refined LOBs were 91 and 77 mg/kg/d for the 130 and 600 SUS samples, respectively. As expected, the corresponding DAEs had lower PDR10s (31 and 45 mg/kg/d). After the PACs had been extracted into the DAEs, no toxicity was predicted for the raffinates, waxes, and final dewaxed LOBs; all PDR10s were >2000 mg/kg/d. The PDR10 values calculated for developmental effects are shown in Table 6. Sample PDR10 values were 550 and 30 mg/kg/d for the initial 130 and 600 SUS distillates, respectively. The corresponding DAEs again had lower sample PDR10 values (159 and 18 mg/kg/d) and all subsequent, sufficiently refined samples had PDR10 values >2000 mg/kg/d. In summary, the results of the modeling exercise indicate that repeated dermal exposure to insufficiently refined LOBs can result in target organ and developmental effects, while sufficiently refined LOBs are not expected to have significant effects on these end points.
Repeated-Dose PDR10s (mg/kg/d) Predicted for Dermal Dosing With 2 Insufficiently Refined LOBs and Petroleum Streams Derived From Them.
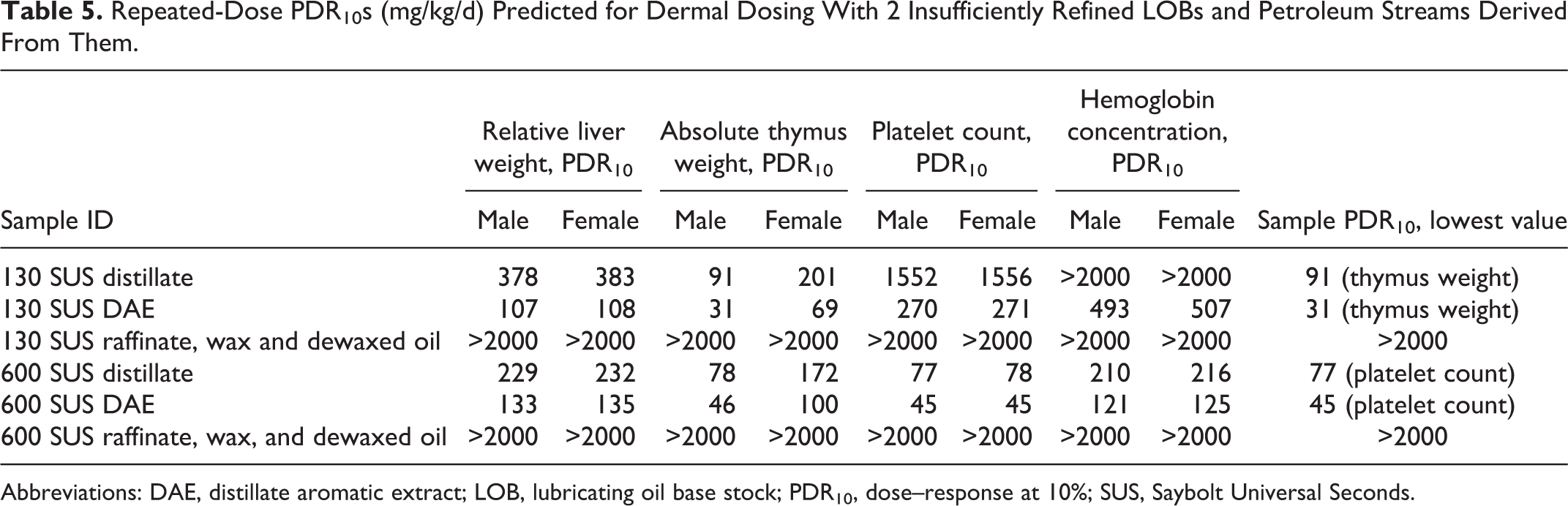
Abbreviations: DAE, distillate aromatic extract; LOB, lubricating oil base stock; PDR10, dose–response at 10%; SUS, Saybolt Universal Seconds.
Developmental PDR10s (mg/kg/d) Predicted for Dermal Dosing With 2 Insufficiently Refined LOBs and Petroleum Streams Derived From Them.
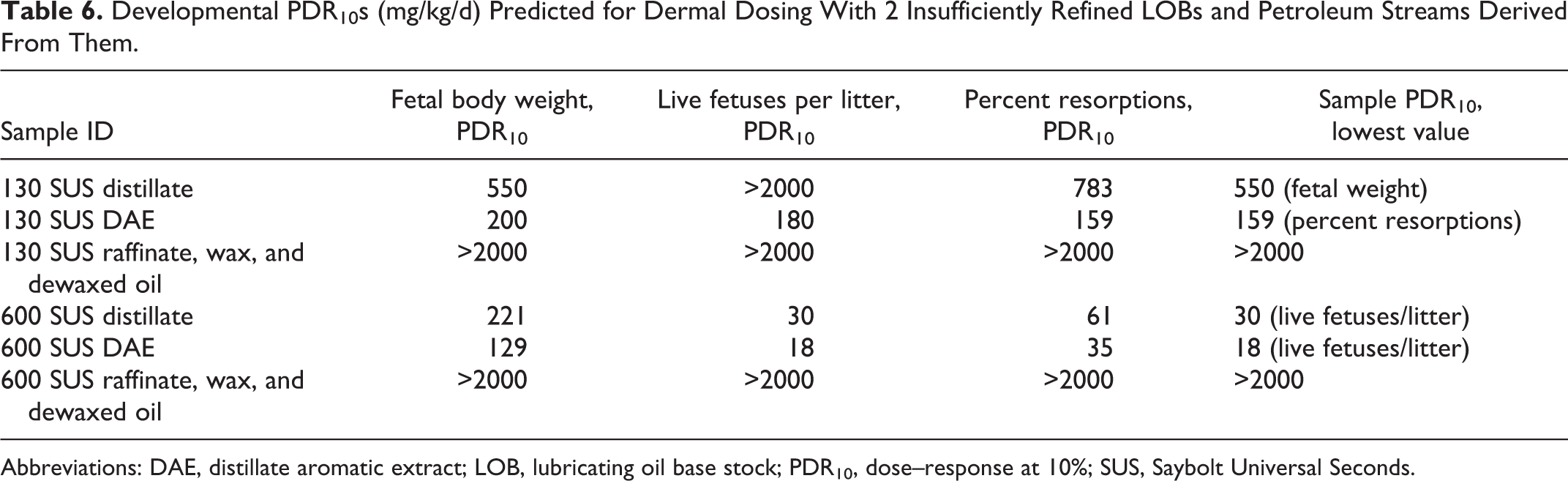
Abbreviations: DAE, distillate aromatic extract; LOB, lubricating oil base stock; PDR10, dose–response at 10%; SUS, Saybolt Universal Seconds.
Subchronic Studies With Dermal Exposures to Sufficiently Refined LOBs
In the study on SDHPD, no test substance-related clinical or dermal observations were noted. Treated males exhibited significantly lower final body weights than those of controls (Table 7), but weights among females were unaffected. No notable findings were seen in the gross examinations at necropsy, but treated males had statistically significantly lower absolute weights of liver and spleen (Table 7). These lower organ weights may have been related to lower terminal body weights as neither difference was significant when organ weight was expressed as a percentage of body weight. In females, both absolute and relative liver weights were significantly higher in treated animals. Histopathology revealed minimal epithelial hyperplasia in treated skin, but no treatment-related systemic findings were noted.
Final Body Weight and Other Selected End Points (Mean ± SD) in 13-Week Dermal Studies in Rats With Sufficiently Refined LOBs.
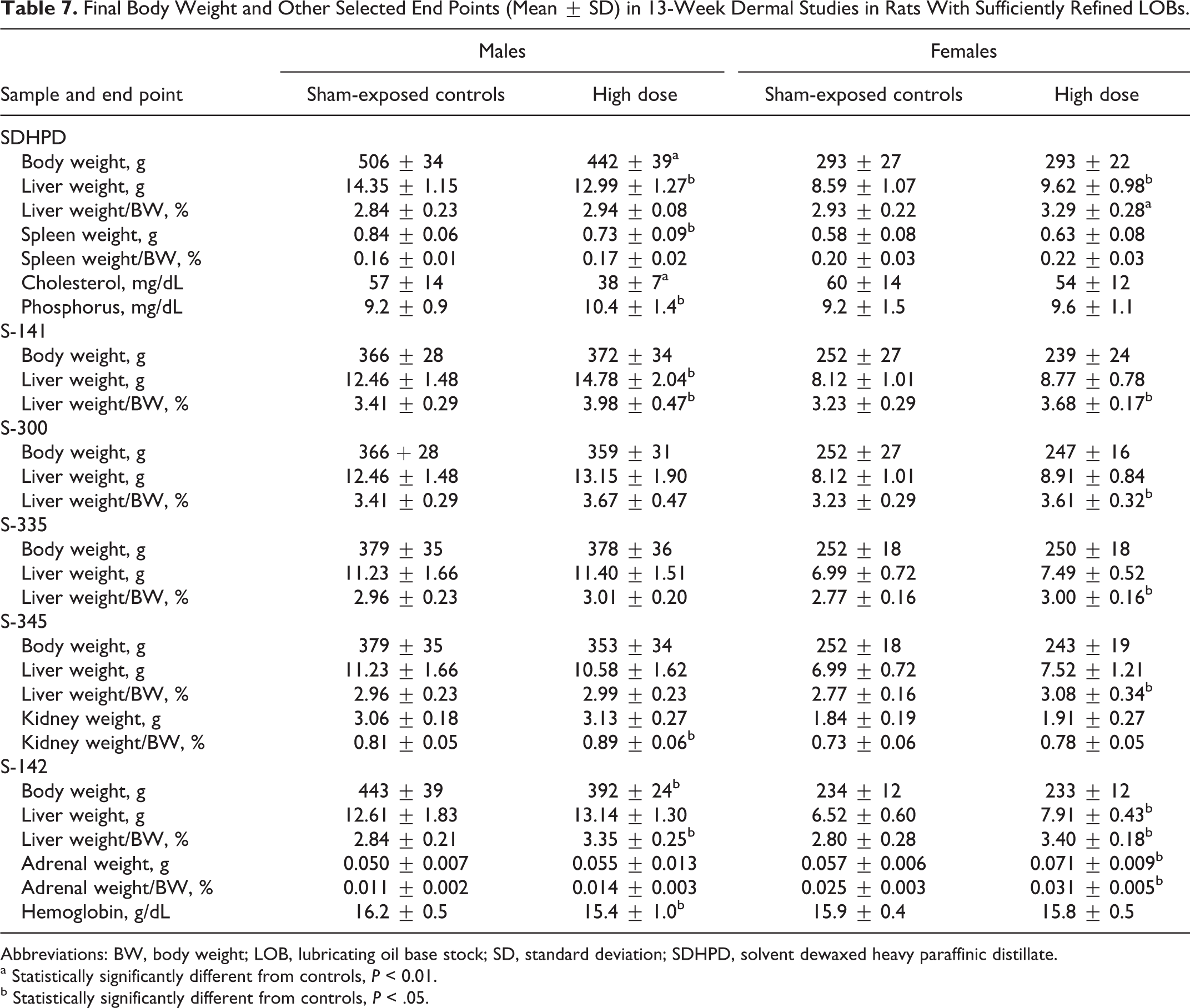
Abbreviations: BW, body weight; LOB, lubricating oil base stock; SD, standard deviation; SDHPD, solvent dewaxed heavy paraffinic distillate.
a Statistically significantly different from controls, P < 0.01.
b Statistically significantly different from controls, P < .05.
Statistically significant findings in the hematology and clinical chemistry evaluations were noted in treated males, namely, higher alkaline phosphatase and activated partial thromboplastin time and lower WBC count, platelet count, and absolute monocyte count. Data are not shown because, although these differences were statistically significant, they were small and within the range of historical controls. Accordingly, none of these was considered to have been toxicologically important. A test substance-related lower mean cholesterol value was noted in treated males when compared to controls (33.3% lower), with a smaller, nonsignificant reduction present in females (10.0% lower; Table 7). Although this finding in males was outside the range of historical controls, it was considered to have been toxicologically irrelevant due to the direction and small magnitude of change in both sexes. A higher mean serum phosphorus value was noted in treated males. Although the mean value was 13% higher than the mean for controls and was outside the range of historical controls, a similar difference was not observed in the females. Therefore, it may have been secondary to the effect on body weight in males. Overall, both the no observed adverse effect level (NOAEL) and lowest observed effect level (LOEL) were judged to be 1000 mg/kg/d based primarily on differences in body weight and organ weights.
With the 2 parallel studies on WO (WOLCCD and WOULSDF), no differences between the sham-dosed and the vehicle controls were seen in clinical signs, body weights, or changes in body weight. In both studies, food consumption was periodically higher in females in the vehicle controls than that in sham controls. Sporadic statistically significant differences were noted among the end points for hematology, clinical chemistry, and organ weights, particularly with WOLCCD (Table 8). These differences were not consistent except for the higher number of circulating neutrophils in both sexes receiving WO and a tendency for higher relative liver weight in females that received the WO. No differences were seen histologically between the sham and vehicle controls. Overall, no toxicologically significant effects were observed with the WO.
Selected End Points (Mean ± SD) for Sham and Vehicle (White oil) Control Groups in 13-Week Dermal Studies With LCCD and ULSDF.
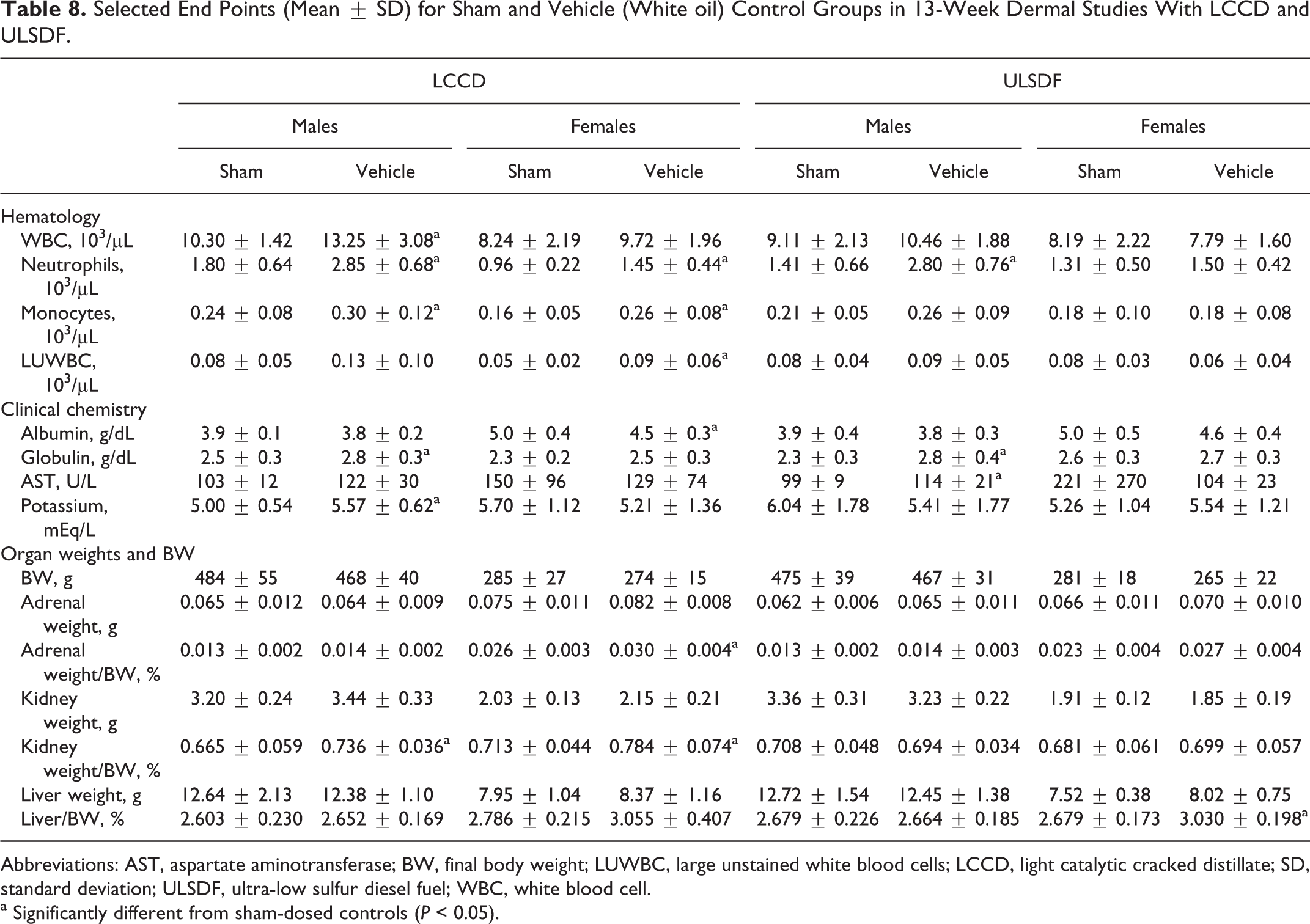
Abbreviations: AST, aspartate aminotransferase; BW, final body weight; LUWBC, large unstained white blood cells; LCCD, light catalytic cracked distillate; SD, standard deviation; ULSDF, ultra-low sulfur diesel fuel; WBC, white blood cell.
a Significantly different from sham-dosed controls (P < 0.05).
In the study with S-141, liver weight was higher in treated males, and relative liver weight was higher in both sexes of treated animals (Table 7), but no histological effects were observed in the liver. Albumin was slightly higher (6%) in treated males than in controls, and urea was increased (18%), but the differences were small and values for both groups were within the normal range. Therefore, these differences are judged not to be toxic responses to S-141. No other significant differences were noted. The NOAEL for systemic effects was judged to be 2000 mg/kg/d, and a LOEL of 2000 mg/kg was assigned based on absolute and relative liver weights.
With S-300, S-335, and S-345, relative liver weights were higher in treated females than in controls with each LOB (Table 7), but no histological effects were observed. With S-345, relative kidney weight was higher in treated males, primarily reflecting a decrease in body weight (Table 7). Sporadic differences from the controls were seen among serum chemistry parameters, but values in treated groups were within normal range, and the differences were not judged to be a response to treatment. No other significant differences were noted. Therefore, NOAELs for systemic effects with S-300, S-335, and S-345 were judged to be 2000 mg/kg/d, and LOELs were 2000 mg/kg based on relative liver weight in females.
In the study with S-142, body weights were significantly lower in treated males, but the differences were thought to result from unusually high body weights in the control group. Weights of the liver and adrenals were higher in treated females (Table 7), but no significant histological changes were observed except for occasional small aggregates of cells with foamy appearing cytoplasm (vacuolar degeneration) in a small portion of the livers of treated females. The increase in liver weight might have been an adaptive response. Hematocrit and Hb were significantly lower in treated males, but the difference was not judged to be toxicologically significant since the values were within the normal range. The NOAEL for systemic effects was judged to be 1720 mg/kg/d, the high dose. This dose was also the LOEL due to increased liver weight in females.
With S-150, body weights were significantly lower in treated males at both 800 and 2000 mg/kg (Table 9). No dose response was apparent, and the lower body weights were judged not to be toxicologically significant. (Lowered body weights in some of these dermal studies are addressed further in the Discussion section.) Liver weights were higher in females than those in controls by 14% at 2000 mg/kg, and liver weight relative to body weight was increased in both sexes. However, histological changes were minimal, and the higher weights were considered as an adaptive response. Thymus weight was lower in males at 2000 mg/kg, and adrenal weight was higher in females at both doses. No histological changes were observed in either organ, and the differences in weight were not judged to represent an adverse effect. Values for the following serum chemistry parameters in males at 2000 mg/kg were statistically lower than control values: albumin (8% lower), cholesterol (13%), triglycerides (28%), and calcium (5%). Alanine aminotransferase was 39% higher. No similar differences were seen in females. These differences were slight, and no abnormalities were seen microscopically; therefore the differences were not considered to be toxicologically significant. The NOAEL for systemic effects was judged to be 2000 mg/kg/d, and a LOEL of 800 mg/kg was set based on lower body weights in treated males.
Final BW and Selected Organ Weights (Mean ± SD) in Rats Treated Dermally With S-150 for 13 Weeks.
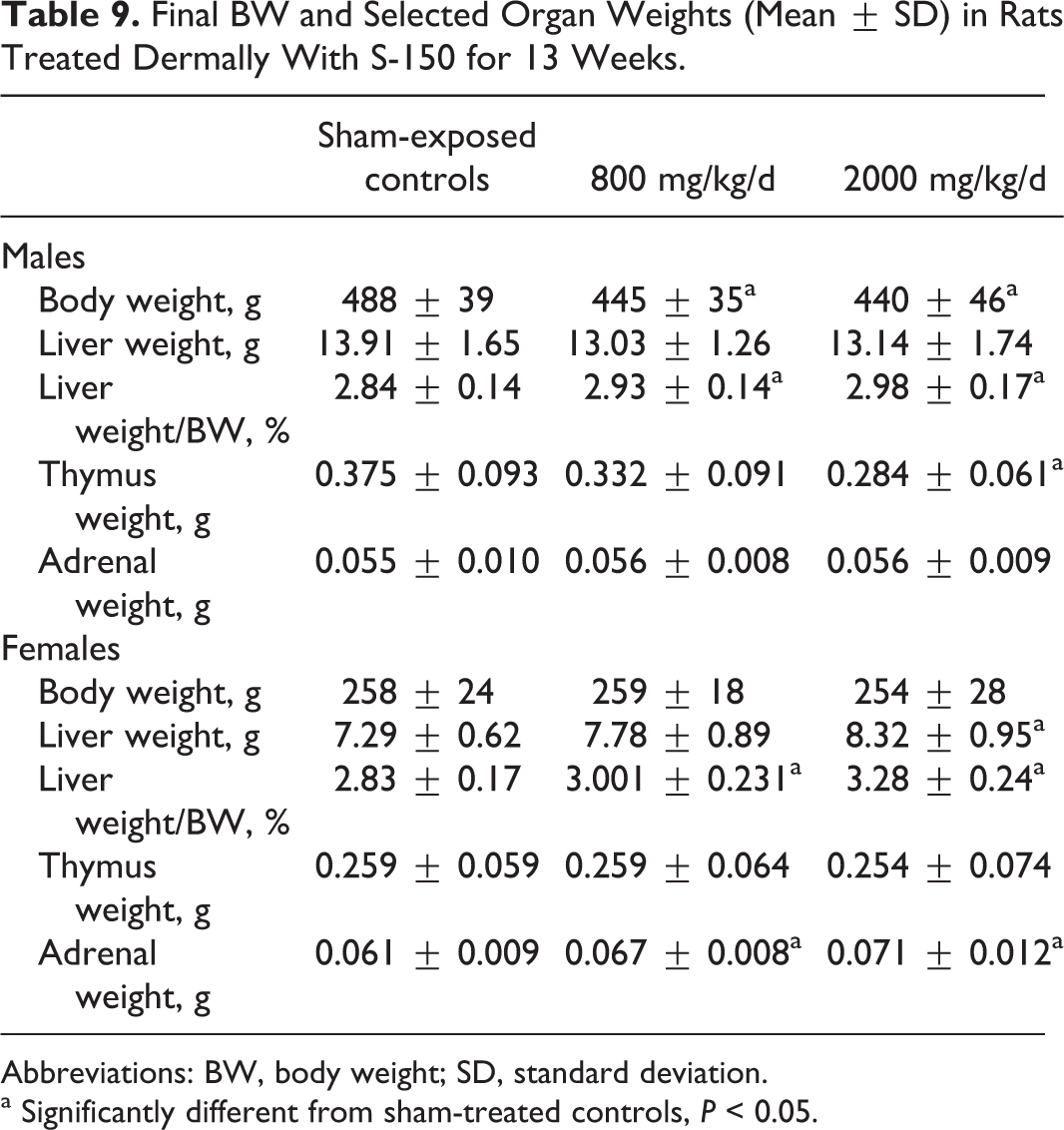
Abbreviations: BW, body weight; SD, standard deviation.
a Significantly different from sham-treated controls, P < 0.05.
In the study with S-461, body weights at the higher doses of S-461 tended to be lower than those of controls during the biophase, and final body weights with 2000 mg/kg at sacrifice were significantly lower in both sexes (Table 10). No significant treatment-related effects were seen in absolute organ weights in either sex. Relative weights of liver, brain, kidneys, and adrenals, expressed as a percent of final body weight, were greater at the high dose than those in sham-treated controls (Table 10). Given the number of relative organ weights that were higher in treated animals (including relative brain weight), the lack of treatment-related effects on absolute organ weights, and the lower body weights in treated animals, the differences in relative organ weights do not appear to be toxicologically significant. No differences were seen in hematology, urinalysis, and serum chemistry except for the following at a dose of 2000 mg/kg: albumin (11% lower than sham-treated controls), albumin/globulin ratio (18% lower), and alanine aminotransferase (33% higher) in males and glucose (11% lower) and triglycerides (29% lower) in females. A few smaller corresponding differences were seen at 500 mg/kg; however, the only values among these groups that were outside the range of historical controls were albumin levels in males. This 11% difference was considered marginal. The value of this study is limited since a histopathological evaluation was not conducted (see Table 3).
Body Weight and Weights of Selected Organs (Mean ± SD) in 13-Week Dermal Study With S-461.
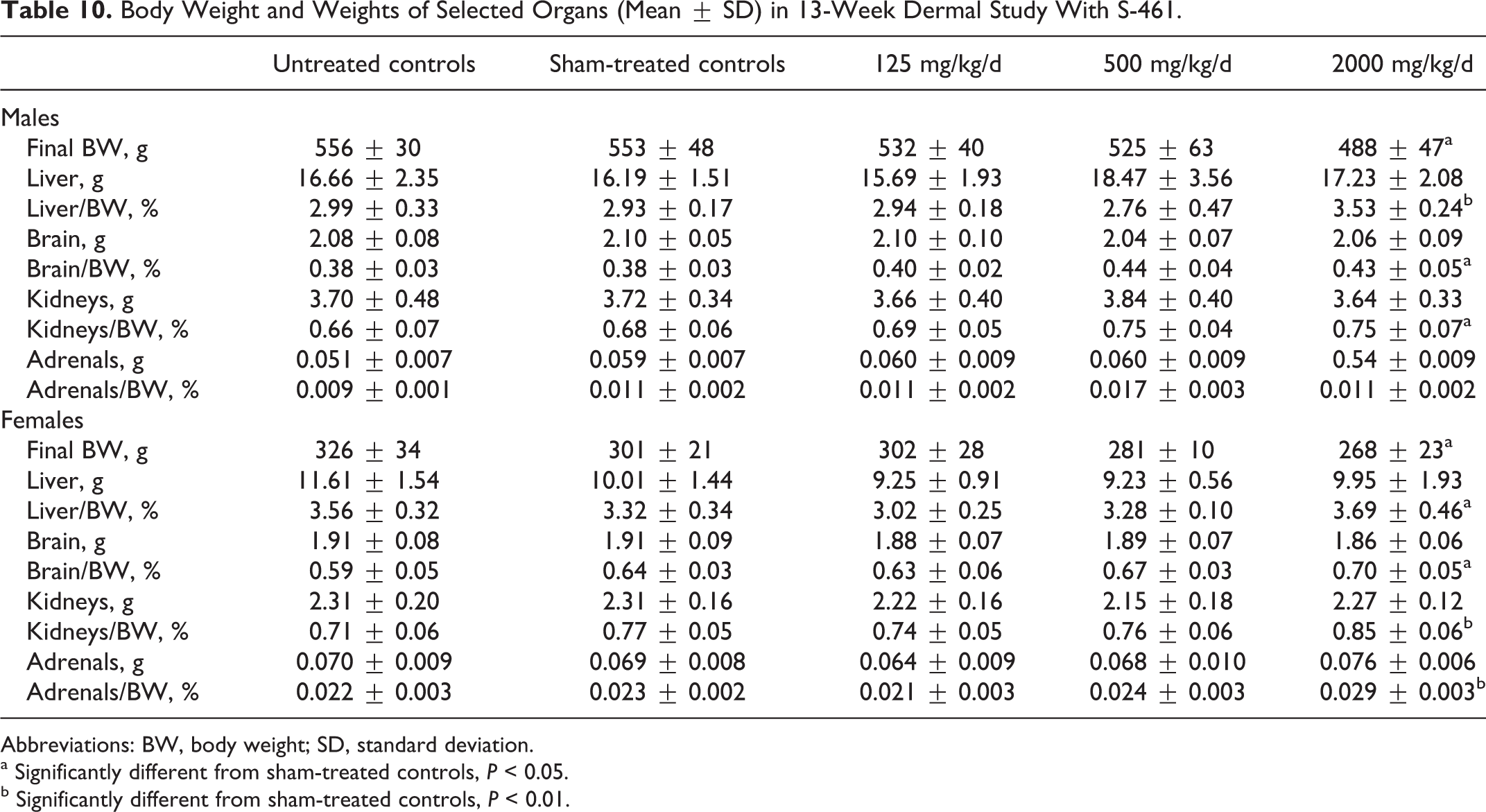
Abbreviations: BW, body weight; SD, standard deviation.
a Significantly different from sham-treated controls, P < 0.05.
b Significantly different from sham-treated controls, P < 0.01.
Overall, limited evidence of systemic effects was seen in 13-week dermal studies with these 8 sufficiently refined LOBs. More specifically, there were differences in organ weights in some studies (eg, with S-150), but no toxicologically relevant histological effects were reported. An analysis of factors that might have contributed to lower body weights in some of the studies led to the conclusion that the lower weights were not adverse. Thus, these weight differences could be used to set LOELs for treatment-related differences but not for adverse systemic effects related to the test substance. Similarly, the sporadic differences in some of the PAC-sensitive end points were considered to reflect small variations that can occur among groups that are not related to the test substance. This conclusion is consistent with the findings in the 2 parallel studies on the same WO (WOLCCD and WOULSDF). Given this logic and the absence of clear adverse systemic effects, the overall NOAELs for these subchronic studies in rats were the highest doses tested (1000 to 2000 mg/kg/d).
In the 4-week studies in rabbits on 5 LOBs identified as 87-099, 87-100, 87-101, 87-102, and 87-103, dermal irritation was noted in most cases, and sporadic changes occurred that were not judged to be adverse. The NOAEL for systemic effects was 1000 mg/kg/d in each study. Results of the studies with API 83-12, API 83-15, and API 84-01 were complicated by dermal irritation as described in more detail in the Appendix. The general interpretation of these 3 studies was that most of the differences observed in the treated rabbits occurred in the presence of significant dermal irritation and probably were secondary to that irritation and/or the stress of dosing. Therefore, these studies were not considered to demonstrate significant adverse systemic effects from the LOBs.
Developmental Toxicity Studies With Dermal Exposures to Sufficiently Refined LOBs
All dams survived to scheduled termination in the developmental toxicity study with SDHPD, and no effects were seen in clinical observations, treated skin, body weight gain, or terminal body weights (Table 11). No notable effects were reported from the gross necropsy. No treatment-related histological observations were noted in dams other than minimal, multifocal mononuclear infiltrate in the superficial dermis of the treated rats. Treatment-related effects in the dams were limited to statistically significant higher mean weights of adrenal glands and nonstatistically significant lower mean thymus weights (Table 11). The differences in weight were not considered adverse because they were small (both <13%) and other signs consistent with maternal toxicity were not observed. Intrauterine growth and survival in the treated group were also unaffected (Table 11). No treatment-related adverse effects on fetal morphology were observed, as measured by external, visceral, and skeletal malformations or developmental variations (Table 12). A low incidence of reduced ossification was noted, but the results were comparable between the control and treated groups. Based on the lack of observed maternal or developmental toxicity, the NOAEL for maternal and developmental toxicity was 1000 mg/kg/d.
Summary of Maternal Body Weight, Maternal Organ Weights, and Results From Examination of Uterine Contents (Mean ± SD) in Developmental Toxicity Study With Dermal Application of SDHPD.
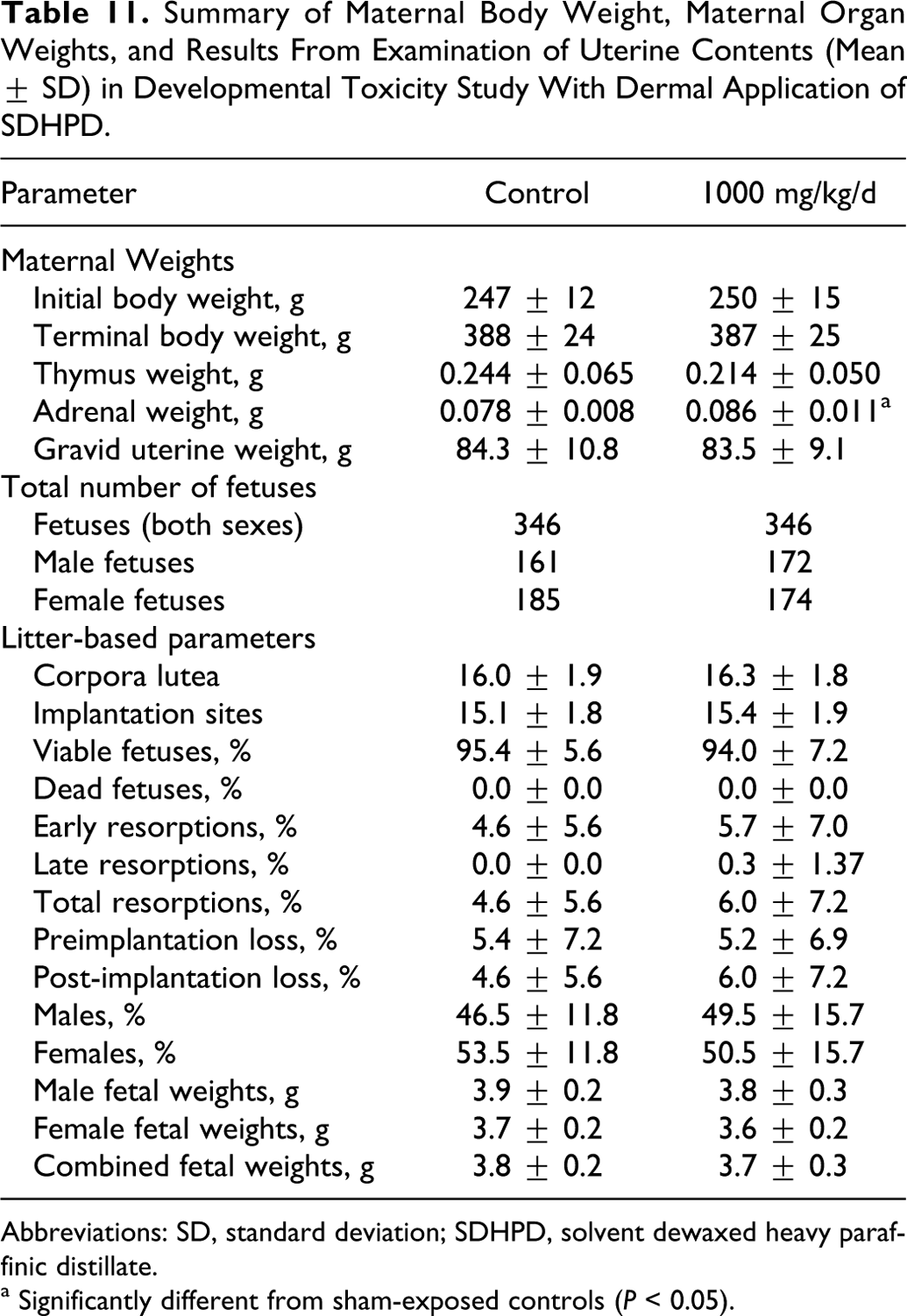
Abbreviations: SD, standard deviation; SDHPD, solvent dewaxed heavy paraffinic distillate.
a Significantly different from sham-exposed controls (P < 0.05).
Results of Examination of Fetuses From Dams Treated Dermally With SDHPD.
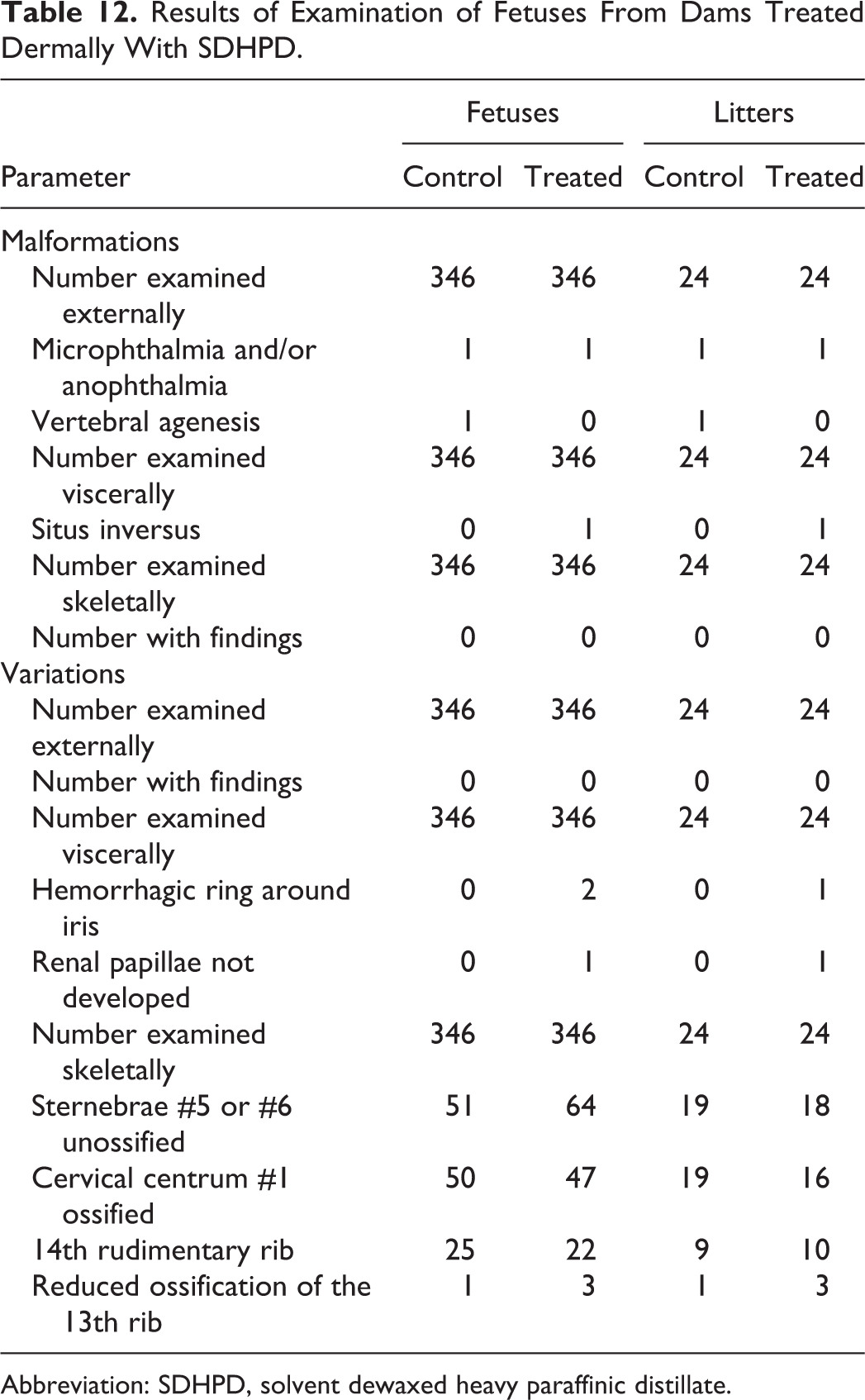
Abbreviation: SDHPD, solvent dewaxed heavy paraffinic distillate.
In the 2 parallel studies on the same sample of WO, clinical signs and dermal observations in dams treated with WOLCCD and WOULSDF were similar to the respective sham-dosed groups. Mean maternal body weights, body weight gains, net body weight, net body weight gain, and gravid uterine weight in females treated with WOLCCD were similar to the sham-dosed group. Mean body weights with WOULSDF were also generally similar to sham controls, but significantly (P < 0.01) lower mean body weight gains were noted with WOULSDF compared to the sham controls during GD 9-12 and 15-18. As a result, mean total body weight gain with WOULSDF was significantly (P < 0.01) lower than its sham controls for GD 0-20. In addition, mean body weight with WOULSDF was significantly (P < 0.05) lower (4.1%) than the sham controls on GD 18. The differences in mean body weights and mean body weight gains between WOULSDF and its sham controls were sporadic and/or slight in magnitude, and net body weights were similar between these 2 groups. Therefore, the differences in body weight parameters were attributed to biological variability. Food consumption tended to be sporadically high in the groups treated with either WOLCCD or WOULSDF compared to the respective sham controls, but there was no corresponding effect on body weights. The differences were attributed to biological variability. Macroscopic findings and organ weights in dams were not affected by treatment except for significantly (P < 0.05) lower mean thymus weight with WOULSDF compared to sham-treated controls (0.213 ± 0.061 vs 0.251 ± 0.055 g, respectively). Intrauterine growth and survival of fetuses with WOLCCD and WOULSDF were similar to the sham controls except that mean fetal sex ratios with WOLCCD were significantly (P < 0.05) different from the sham controls. However, the values were within the range of historical controls, and the differences were attributed to biological variability. No differences were noted in fetal external malformations, visceral variations or malformations, and skeletal variations or malformations with WOLCCD or WOULSDF compared to the respective sham-dosed controls. In short, the WO did not produce significant maternal or developmental toxicity in either of the parallel studies.
In the 2 remaining developmental toxicity studies, no treatment-related changes were reported in maternal or fetal end points, including maternal reproductive performance and survival or development of fetuses. The NOAELs for maternal and fetal effect from S-461 and S-141 were 2000 mg/kg/d.
Developmental Toxicity Study With Inhalation or Oral Exposures to Sufficiently Refined LOBs
Perianal staining was observed in all females treated orally and appeared to result from a clear oily anal discharge within a few hours of dosing. Otherwise, no adverse effects were seen in the inhalation and oral studies with S-461. The inhalation no observed adverse effect concentration (NOAEC) was 1000 mg/m3, and the oral NOAEL was 5000 mg/kg/d.
Reproductive Toxicity Assessments With Dermal Exposures to Sufficiently Refined LOBs
No significant treatment-related differences were seen in weights of reproductive organs with dermal doses of 2000 mg/kg in the 13-week dermal studies in rats (Table 13). In addition, testes were evaluated microscopically in controls and treated animals in the first 6 studies as shown in Table 13, and ovaries were examined microscopically following exposures to S-142. No treatment-related effects were seen microscopically in the examined organs.
Mean Final Body Weight and Weights of Reproductive Organs in 13-Week Dermal Studies With S-461 and Related LOBs.
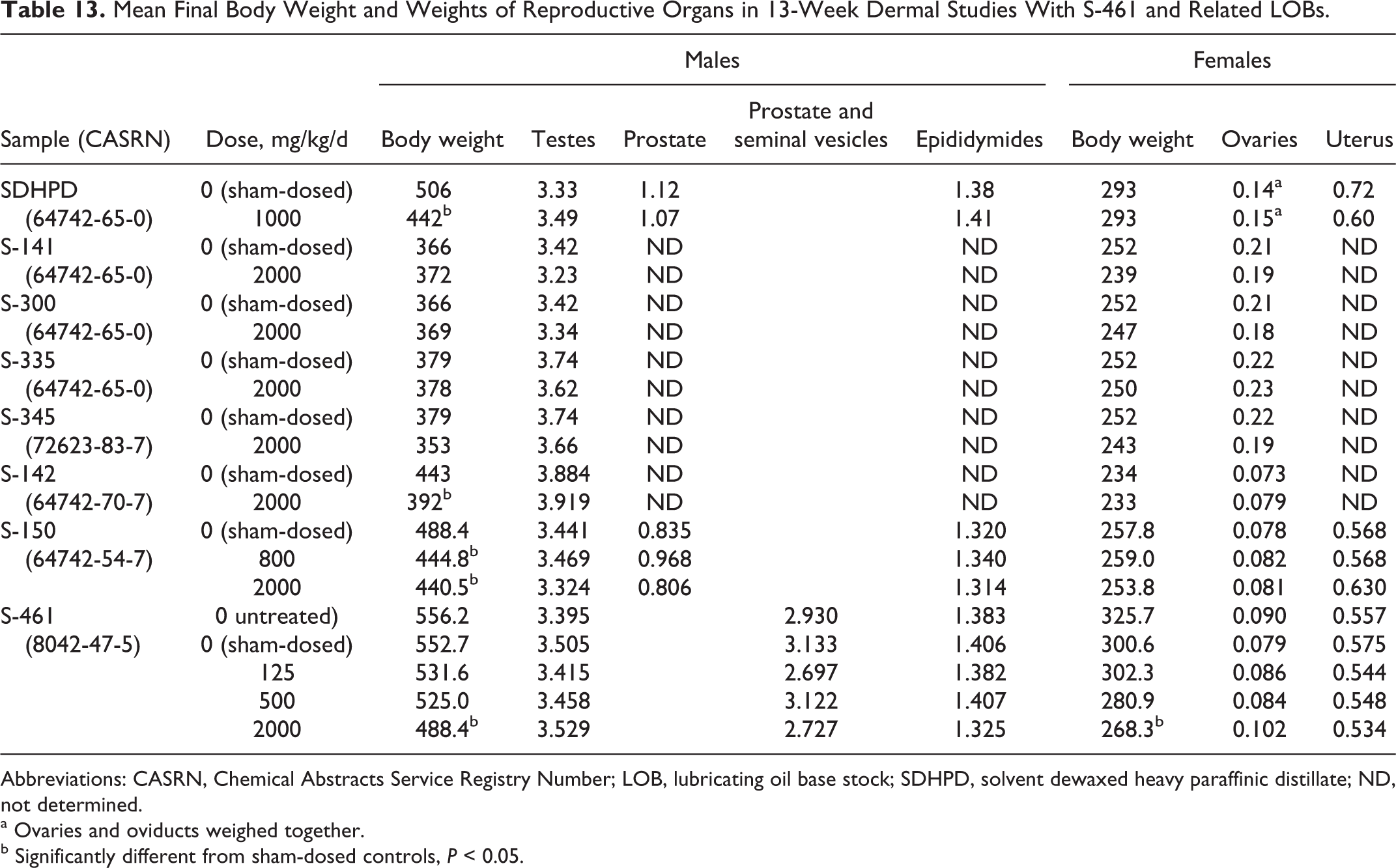
Abbreviations: CASRN, Chemical Abstracts Service Registry Number; LOB, lubricating oil base stock; SDHPD, solvent dewaxed heavy paraffinic distillate; ND, not determined.
a Ovaries and oviducts weighed together.
b Significantly different from sham-dosed controls, P < 0.05.
In the reproductive toxicity study on S-461, estrus cycle was not affected by treatment in limited subgroups of animals examined for this end point (5 females/group). The fertility index among these subgroups was 100% in the sham-exposed controls and 2000 mg/kg/d groups and 80% in the untreated controls due to 1 female with an abnormal estrus cycle. During gestation, erythema, scabs, and flaking were observed on the skin of nearly all animals treated with S-461. Similar findings were reported during lactation. Body weight gain during the gestation and postpartum periods appeared normal. Mean body weights of females in the 2000 mg/kg/d group were significantly lower than those of the untreated controls during the first half of gestation but were similar to mean weights for the sham-exposed controls (data not shown). Since the gain in body weight was not different in the treated groups, no treatment-related effect was apparent. No effects of treatment with S-461 were noted at necropsies of dams. No effects were seen in dams on the percentage of pregnant females, duration of gestation, or number of implantation sites per dam. No adverse effects were noted among the litters for Liveborn Index, Day 4 Survival Index, or Day 21 Survival Index. Mean pup weight was not affected by treatment during postpartum days 0 to 28. Eyelid dysjunction and surface righting reflex were not affected by treatment. Observations of offspring at birth and at necropsy were not affected by treatment. In short, dermal application of S-461 at doses up to 2000 mg/kg/d beginning 10 weeks before mating did not have any adverse effects on reproductive performance of female rats or on the in utero and postnatal survival or development of offspring. The apparent NOAEL was 2000 mg/kg/d, but confidence in that value is limited since histopathological evaluations of parental reproductive organs were not completed.
Discussion
Acute Toxicity Studies With Oral, Dermal, and Inhalation Exposures
Toxicity in the acute studies reported here was consistently low for LOBs administered by dermal, oral, or inhalation routes for both sufficiently and insufficiently refined LOBs. Nonlethal effects in the acute studies were minimal and, if present, generally consisted of dermal irritation with dermal dosing, loose stool with oral dosing, or no observed changes with inhalation. One exception was sample API 83-12, a carcinogenic LOB, that had a relatively low LC50 (2.18 mg/L) and caused pronounced pulmonary effects in an acute inhalation study. By comparison to the other test samples, API 83-12 has a low viscosity (53 SUS at 100°F (38°C), or about 8.3 cSt at 100°F). Such low viscosity (< ∼15 cSt at 100°F) can be the reason for an LC50 below 5 mg/L as well as the effects observed in the lung. 32
Repeated Dermal Exposures to Insufficiently Refined LOBs
Sample PDR10 values for the 2 insufficiently refined LOBs were 31 and 91 mg/kg for 13-week dermal studies in rats. These results were compared to 2 other refinery streams that could potentially be used as surrogates for insufficiently refined LOBs. Heavy vacuum gas oil (HVGO) was chosen because it is derived from the vacuum distillation tower similarly to streams that are further refined to produce sufficiently refined LOBs and also because this sample had PAC content similar to insufficiently refined LOBs (Table 1). A DAE was the second potential surrogate. The PAC content of the DAEs as shown in Table 1 was higher than their respective insufficiently refined LOBs. Polycyclic aromatic compound data were also available on an additional DAE (sample 86187 in Table 1) that was tested experimentally in animals and serves as a “worst-case” surrogate for insufficiently refined LOBs. This information on the aromatic constituents of the HVGO and DAE was previously published 20 and was provided for use by the company that conducted these studies.
Experimentally, this HVGO had a NOAEL of 125 mg/kg in the original report of a 13-week dermal toxicity study, but a NOAEL of 30 mg/kg (next lower dose) was subsequently suggested based on lower Hct at 125 mg/kg. 33 The sample PDR10 for this HVGO was 118 mg/kg. These doses with HVGO were near the PDR10s found with the insufficiently refined LOBs. The DAE was also tested in a 13-week study at doses of 0, 30, 125, 500, and 1250 mg/kg/d. 20,34 In this study, the NOAEL was <30 mg/kg/d; the benchmark dose at 10% response (BMD10) for this study was calculated to be 15 mg/kg/d; and the sample PDR10 for the DAE was estimated as 58 mg/kg/d. 35 As might be expected, these values for the DAE were lower than those for HVGO and the PDR10 values for the 2 insufficiently refined LOBs.
Sample PDR10s for developmental toxicity end points with the 2 insufficiently refined LOBs were 550 and 30 mg/kg/d. Results from dermal developmental studies in rats with HVGO and DAE followed a pattern similar to that with the 13-week studies. The NOAEL for maternal and fetal effects of HVGO was 125 mg/kg/d,
33
close to its sample PDR10 of 157 mg/kg. The NOAEL with DAE was
Thus, as a worst-case assessment, it would be reasonable to assume that the systemic or developmental toxicity of insufficiently refined LOBs would be similar to that of DAEs. However, variability in PAC content and toxicity has been seen in other studies on PAC-containing refinery streams. 19 If a particular untested LOB is designated as insufficiently refined but is suspected to have low PACs, an assessment of that LOB can be made on a case-by-case basis and requires that ARC data be available to provide justification for a claim that the LOB is sufficiently refined.
Systemic Toxicity With Repeated Exposures to Sufficiently Refined LOBs
It was hypothesized that the DMSO-extractable aromatics represented the toxic constituents in insufficiently refined LOBs and their removal would result in sufficiently refined LOBs that would be nontoxic. The present results support that hypothesis. Repeated dermal application of sufficiently refined LOBs to rats (13-week exposures) and rabbits (3-4 week exposures) consistently resulted in NOAELs for systemic toxicity in the range of 1000 to 2000 mg/kg/d, typically the highest doses tested. There were no consistent pathological changes. Liver weights tended to be slightly increased in several studies, most often in only 1 gender of the treated animals. However, as no histopathological changes were seen in the livers, the effects on liver weight were considered to be evidence of systemic exposure but not toxicity. These results were consistent with 2-week dermal exposures. 15 Some treatment-related differences (eg, lower body weight gain and lower weights of heart, testes, and ovaries) were noted in the dermal studies with rabbits reported here; however many of these were considered secondary to dermal irritation. The effect on the testes in particular might have been secondary to dermal irritation. 37
Although dermal exposures are expected to be the most common route of exposure with sufficiently refined LOBs, inhalation exposures also occur, particularly in industrial settings. The number of publications on inhalation toxicity studies specifically on LOBs is limited, but several studies have been performed on metal removal fluids (MRFs) that are formulated using sufficiently refined LOBs together with performance additives. Restricting this discussion to LOBs and not MRFs, the only consistent effects in 3 separate 4-week inhalation studies were increased lung weight and concentration-related microscopic accumulation of foamy alveolar macrophages in lungs. 38 Although 1000 mg/m3 could justifiably be considered the NOAEL for these 3 studies due to the non-adverse nature of accumulation of alveolar macrophages in the lungs without other accompanying effects, a NOAEL of 210 mg/m3 was chosen because of increased lung weight. Supporting data were obtained from14-day inhalation studies with 2 highly refined base oils in which rats (both sexes) were exposed to aerosol concentrations of approximately 50, 500, and 1500 mg/m3 for 6 hours/d, and no observable effect levels were >50 mg/m3. 35,39 Further discussion of these and related inhalation studies is available. 40
Nonaccidental oral exposures to most sufficiently refined LOBs are not expected, but ingestion is one of the intended routes of exposure with WOs in foods, medicines, and cosmetics. Numerous studies involving ingestion of WOs have been previously performed. In subchronic studies, observed increases in weights of liver, lymph nodes, and spleen at the highest doses were associated with deposition of the lipophilic oil and minimal pathological changes in Fischer 344 rats. 41,42 Other rodent strains and species do not display this sensitivity to ingested mineral oil. 43,44 Extensive follow-up testing has demonstrated a unique sensitivity of F344 rats to mineral oil after ingestion, resulting in significant oil uptake, deposition, and a characteristic histological response. 44 -49 The general interpretation of these findings is that the granulomatous lesions experimentally induced by WO feeding, particularly in the liver of F344 rats, are exaggerated immunological responses peculiar to this strain of rats and that the changes induced by WOs in human are incidental and inconsequential. 50 Little difference is expected after ingestion of WOs or sufficiently refined LOBs due to low PAC content in both types of oils.
One finding in the present subchronic studies that deserved particular attention was that body weights of treated males were significantly lower than sham-treated controls in 4 of the 8 studies; mean weights ranged from 87.3% to 90.2% of control weights in these 4 studies. In contrast, body weights of females were affected in only 1 of the 8 studies. These lower body weights were not associated with pathological changes or, in most cases, differences in organ weights. No association was seen between the lower weights and other experimental variables as discussed in the Appendix. Given that and the fact that lower body weights were not reported in publications of repeated exposures of rats to sufficiently refined LOBs administered orally or by inhalation, the differences seen with dermal administration were considered to be related to the mode of dosing rather than to inherent toxicity of the LOBs. Since the lower body weights might be linked to the experimental model and did not appear to be associated with significant systemic effects, these differences in body weights are not considered as adverse, as suggested by European Centre for Ecotoxicology and Toxicology of Chemicals (ECETOC). 51 In other words, the lower body weights could be used to set a LOEL for treatment-related differences, but not a lowest observed adverse effect level for adverse systemic effects related to the test substance. Accordingly, the NOAEL for systemic toxicity following repeated dosing was the highest tested dose (1000-2000 mg/kg/d).
Sporadic differences in other end points were seen between treated and controls animals in the subchronic dermal studies in rats. These are also discussed further in the Appendix.
Developmental Toxicity With Exposures to Sufficiently Refined LOBs
Here, 5 developmental toxicity studies with dermal dosing of a sufficiently refined LOB or WOs in rats are reported. In each case, no adverse effects were seen in either maternal or fetal end points. Although weight of the adrenals was higher in dams treated with SDHPD, this difference was consistent with enlargement of the adrenal glands due to chronic stress. 52,53 Given this possible reason for the difference, the small amount of difference from controls, and the lack of signs of maternal toxicity, the difference in weight was not considered adverse. Therefore, the NOAEL in each study was the highest dose that was tested (1000 or 2000 mg/kg/d). In addition, the NOAEC in an inhalation study with a highly refined LOB (S-461, a WO) was 1000 mg/m3. In an oral developmental toxicity study using the same WO, the NOAEL was 5000 mg/kg/d. This NOAEL was supported by a second, more limited, study. These NOAEL and NOAEC values can apply to those LOBs that are sufficiently refined and have low PAC content.
Additional information supporting the lack of developmental toxicity by LOBs was obtained from an article by Schreiner et al 54 on a study based on OECD Test Guideline 421. Treatment with 1 mL/kg/d (∼900 mg/kg/d) of mineral oil did not significantly affect any of the measured end points. Further supporting data are obtained from a reproductive/developmental toxicity study in Sprague Dawley rats. 55 A white mineral oil (food/drug grade highly refined, CAS No 8012-95-1) was used as a vehicle control. Although there were no untreated animals for comparison to the WO group, the results were considered to be within normal limits. Consequently, these studies provide supporting evidence that WOs do not produce developmental effects.
Reproductive Toxicity Assessments With Dermal Exposures to LOBs
The results from the developmental toxicity studies on sufficiently refined LOBs presented here provide data to address the developmental toxicity of these LOBs in the assessment of possible reproductive toxicity. No significant effects were seen in these studies. In addition, the weights of reproductive organs were not affected in several subchronic dermal studies with sufficiently refined LOBs (Table 13). Testes were examined microscopically in 6 of these studies and ovaries in 1 study. No histopathological abnormalities were noted. Thus, the available data from both developmental and repeated-dose studies provide the information necessary to satisfy the HPV requirements for reproductive toxicity assessments and indicate that reproductive toxicity with sufficiently refined LOBs is expected to be minimal.
Even for insufficiently refined LOBs, reproductive toxicity does not appear to be a sensitive end point of toxicity compared to developmental and repeat-dose toxicity. In a recent study, high-boiling petroleum substances, including many with a high PAC content, have demonstrated low potential to cause male or female reproductive toxicity relative to developmental toxicity and systemic toxicity (in repeat-dose toxicity studies). 56
Further evidence of the low potential of insufficiently refined LOBs to cause reproductive toxicity is obtained from screening-level fertility studies of clarified slurry oil (CSO, CAS No 64741-62-4, also known as catalytically cracked clarified oil, syntower bottoms, and carbon black oil). This refinery stream contains such high levels of PACs that it is believed to be “worst case” by comparison to all other refinery streams. When CSO samples are tested in developmental toxicity studies, they typically produce severe developmental effects (resorptions, reduced fetal body weight, and cleft palate) at levels below 5 mg/kg/d. 57,58 In contrast, in screening studies to assess the potential for CSO to affect male and female reproductive parameters, 59 there were no effects at levels up to 250 mg/kg/d, the highest dose tested. These studies add additional weight to the view that further tests of the potential for unrefined LOBs to cause reproductive effects are not justified, because the critical effects, carcinogenicity and developmental toxicity, which would be used to assess the adequacy of current hazard control practices, have already been characterized.
Conclusions
Because LOBs are a family of substances with variable composition, the amount and profile of PACs can differ among various samples. As a result, mammalian toxicity with repeated exposures, mutagenicity, and carcinogenicity have been shown to vary with the PACs in the samples. The new data presented in this article are related to the refining history and PAC content of each sample and add to the body of information indicating low toxicity with sufficiently refined LOBs and potential toxicity with insufficiently refined LOBs. The main conclusions were as follows:
As with many other refinery streams with constituents boiling principally above 343°C17–20, the effects of LOBs on sensitive noncarcinogenic end points in subchronic and developmental toxicity studies are related to PAC content of the LOBs.
Insufficiently refined LOBs can produce target organ effects and can also be developmentally toxic with repeated dermal exposure.
The toxicity of insufficiently refined LOBs is associated with the aromatic constituents, which are removed during the production of sufficiently refined LOBs.
Additional testing of insufficiently refined LOBs was not considered justified since they are already recognized as potentially hazardous (carcinogenic and systemically toxic). The doses of an untested sample that could result in observable systemic or developmental effects can be modeled using the PAC profile of that sample or estimated by read-across from similar LOBs or refinery streams.
Experimental NOAELs for systemic toxicity and developmental toxicity with repeated dermal exposures to sufficiently refined LOBs were 1000 to 2000 mg/kg/d and typically were the highest doses tested. Although local effects can occur with repeated dermal or inhalation exposures, those effects seem more related to the physical presence of the oil rather than to inherent toxicity. The absence of toxicologically relevant findings was consistent with the lower PAC content of sufficiently refined LOBs.
Sufficiently refined LOBs as currently marketed would not be considered hazardous, although testing or a thorough knowledge of refining history is needed to verify this designation. The data on sufficiently refined LOBs presented here can be used for read-across to untested sufficiently refined LOBs.
Lower body weights were observed in some 13-week studies in rats with dermal dosing. These differences appeared to be related to the experimental model and are not adverse.
Sufficiently refined LOBs are likely to have little, if any, effects on reproductive parameters.
It is unlikely that insufficiently refined LOBs would be reproductively toxic, or at least not at dose levels which would be likely to produce developmental toxicity.
Tested LOBs had low acute toxicity by dermal, oral, and inhalation routes. Nonlethal effects in the acute studies were minimal.
Footnotes
Appendix
Acknowledgments
The authors would like to thank Chris Sexsmith for quality assurance support. WIL Research Laboratories performed the 90-day and developmental toxicity studies on SDHPD; Dr Patrick Crittenden and Dr Adam Kuhl were the respective study directors. Dr Mark Nicolich was instrumental in the development of the PAC models and calculated the PDR10s provided here. Chris D’Aleo was the study director on the acute oral and dermal studies. Dr George Cruzan was the study director for the subchronic studies on S-141, S-142, S-150, S-300, S-335, and S-345. Dr Maureen Feuston was the study director for the subchronic, developmental (oral and dermal), and reproduction studies with S-461 as well as the developmental toxicity study on S-141. Dr Angela Burke was the study director on the subchronic studies on LCCD and ULSDF. Dr Vincent Piccirillo was the study director on the subchronic studies with API 83-12, API 83-15, and API 84-01. Jeffrey Charlap was the study director on the developmental toxicity studies with LCCD and ULSDF. Gary Trimmer was the study director on the subchronic studies with 87-099 through 87-103. Fred Whitman was the study director on acute inhalation studies with 87-099, 87-101, and 87-102.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Potential conflicts of interest exist. The authors of this paper are or were employed by companies that manufacture petroleum products or are contractors working on behalf of the petroleum industry HPV program.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This project was sponsored and funded by the Petroleum HPV Testing Group (PHPVTG), an unincorporated group of manufacturers affiliated by contractual obligation to fund a voluntary data disclosure and toxicity testing program on certain petroleum-related chemical substances in response to EPA’s HPV Challenge Program. The American Petroleum Institute (API) manages the PHPVTG’s activities.
