Abstract
With the increasing use of mycophenolic acid (MPA) in solid organ transplantation, some clinical studies indicate that it is also a human teratogen. However, it is unknown by which mechanism MPA acts as a teratogen. Mycophenolic acid was a selective blocker of de novo purine synthesis, and its immunosuppressive effect is mediated by the inhibition of inosine monophosphate dehydrogenase, which could be a target for MPA-induced toxicity as well. The aim of our study was to examine the direct influence of MPA exposure on zebrafish (
Introduction
Mycophenolic acid (MPA) is a metabolized product and an active element of mycophenolate mofetil (MMF), which has been approved for the prevention of acute graft rejection in kidney, heart, and liver transplantation in 1995. 1,2 Mycophenolic acid is a known potent uncompetitive inhibitor of inosine monophosphate dehydrogenase (IMPDH), the rate-limiting enzyme for the de novo synthesis of guanosinenucleotides, 3,4 which plays crucial roles in cell proliferation and other cellular functions including DNA replication, RNA and protein synthesis, and cellular signaling. 5 Consequently, MPA can block T- and B-lymphocyte proliferation and clone expansion and prevent the generation of cytotoxic T cells and other effector T cells. Through depletion of guanosine nucleotides, MPA can suppress glycosylation and the expression of several adhesion molecules, thereby decreasing the recruitment of lymphocytes and monocytes into the sites of inflammation and graft rejection. 4 With the increasing use of MPA in solid organ transplantation, the most common adverse effects of MPA had been reported, including gastrointestinal disturbances and myelosuppression. 6,7 However, the use of MPA during pregnancy poses significant risk for patients and their unborn offsprings, 8 –11 so the importance of drug-related developmental toxicity became evident.
Zebrafish (
According to our previous research, MPA induced significant growth defects during the development of zebrafish embryo, including tail defects and severe pericardial edema. However, it was unknown by which mechanism MPA acted as a teratogen. In this study, we focused on the developmental toxicity caused by MPA. We identified the target
Materials and Methods
Zebrafish Maintenance
Wild-type zebrafish (Tübingen line) were obtained from Model Animal Research Center of Nanjing University. They were kept at 28.5°C as described. 16 The light–dark cycle was 14:10 hours. Wild-type fish were mated, and spawning was stimulated by the onset of light. Embryos were collected and placed at 28.5°C in Petri dishes containing embryo medium (0.2 g/L of Instant Ocean Salt in distilled water with 0.01% methylene blue). The zebrafish studies were approved by the Institutional Animal Care and Use Committee at Nanjing Tech University. Embryos and larvae were staged according to Kimmel et al. 17 The age of the embryos and larvae is indicated as hours postfertilization (hpf).
Substances
Mycophenolic acid (CAS No. 50-35-1), dimethyl sulfoxide (DMSO; CAS No. 67-68-5), and guanosine (CAS No. 118-00-3) were purchased from Sigma-Aldrich (St Louis, Missouri). Stock solutions were prepared by dissolving the pure chemicals in DMSO and then diluted to the desired concentrations in embryo medium. Final DMSO concentrations were 0.1% in the treatment solution. Dimethyl sulfoxide 0.1% was used as vehicle control during the assays.
Drug Treatment and Larvae Observation
Zebrafish embryos were distributed into 24-well plates with 20 embryos per well and 1 mL embryo medium. Survival and morphological defects were assessed using a dissecting microscope (SMZ745T; Nikon, Japan). Zebrafish embryos with any morphological defect were representative for the teratogenicity of MPA. For assessing both the lethal toxicity and the teratogenic toxicity, zebrafish embryos at 2 hpf were treated with various concentrations (0.5-50 µmol/L)of MPA for 70 hours. The number of dead embryos and embryos with any of the developmental defects were calculated. Image-Pro Plus 6.0 (Media Cybernetics, Rockville, Maryland) was used to define the tail curvature angle and the area of pericardial edema. GraphPad Prism 5.0 (San Diego, California) was used to calculate the 25% lethal concentration value (LC25), 50% lethal concentration value (LC50), 50% teratogenic concentration (EC50), and the concentration with no observed adverse effect level (NOAEL), respectively. Each experiment was done in triplicate and was repeated 3 times.
For the RESCUE study, 20 zebrafish embryos per well were treated with 33.3 μmol/L of guanosine and 8.0 μmol/L of MPA at 2 hpf. The evaluation of rescue phenotype was determined at 72 hpf. Each assay was done in triplicate and was repeated 3 times.
Reverse Transcription and Quantitative Real-Time Polymerase Chain Reaction Analysis
Total RNA from 10 embryos per well of 8.0 μmol/L of MPA-treated or control groups was isolated with TRIzol (Invitrogen, Cergy-Pontoise, France) and reverse transcribed by Moloney murine leukemia virus reverse transcriptase (Invitrogen) using oligo (dT) primers (Invitrogen). Real-time quantitative polymerase chain reaction (qPCR) was performed in an ABI 7900HT FastReal-Time PCR system (Applied Biosystems, Foster City, California) using the standard EvaGreen assay detection protocol. Briefly, all reactions were performed in a total volume of 30 μL containing 10 μL template, 1× PCR buffer, 0.2 mmol/L each deoxynucleotide nucleoside triphosphate, 2.5 mmol/L MgCl2, 0.75 U Taq polymerase, 0.5× EvaGreen (Biotium, Hayward, California), and 20 nmol/L fluorescein (Bio-Rad, Hercules, California). The expressions of β-actin under various treatment conditions during different time points were checked first to confirm that β-actin expressions were not changed by chemical treatment. The qPCR results were analyzed using β-actin as an internal standard, and fold of change was calculated relative to the untreated control using the ΔΔ
The Primers Used for Zebrafish Embryos.
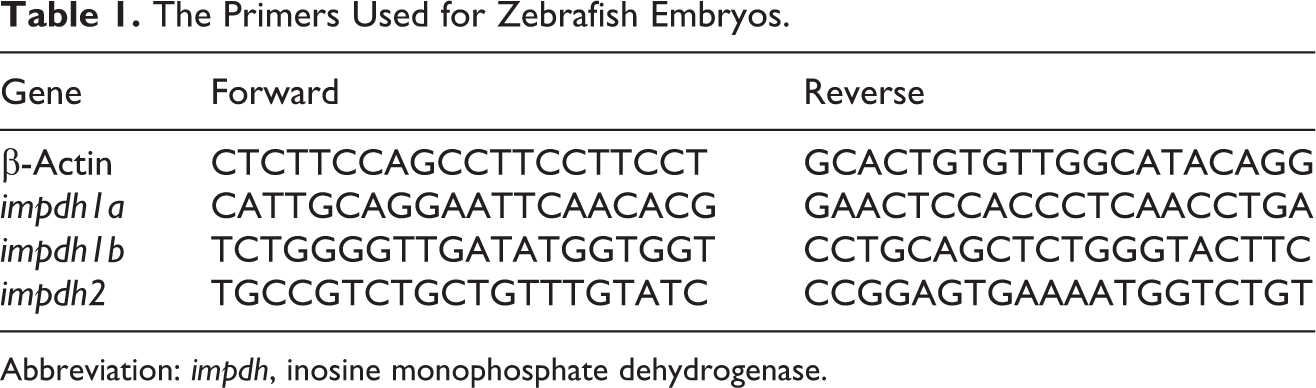
Abbreviation:
Statistical Analysis
All statistical analyses were expressed as mean ± standard error of the mean using GraphPad Prism 5.0. The decrease/increase in gene expression as determined by qPCR was analyzed using 2-way analysis of variance followed by Bonferroni posttests. Paired
Results
Lethality and Teratogenicity of MPA in Zebrafish Embryos
Mycophenolic acid treatment caused teratogenicity and lethal toxicity in zebrafish embryos in a dose-dependent manner (Figure 1). In the zebrafish model, compounds with a teratogenic index (LC25/NOAEL ratio) greater than or equal to 10 are classified as teratogens. 19 According to our assessment, the LC25, LC50, EC50, and NOAEL values were calculated as 8.0, 12, 6.9, and 0.5 μmol/L, respectively (Table 2). The LC25/NOAEL ratio was 16, which was greater than 10, so MPA could be classified as a teratogen.
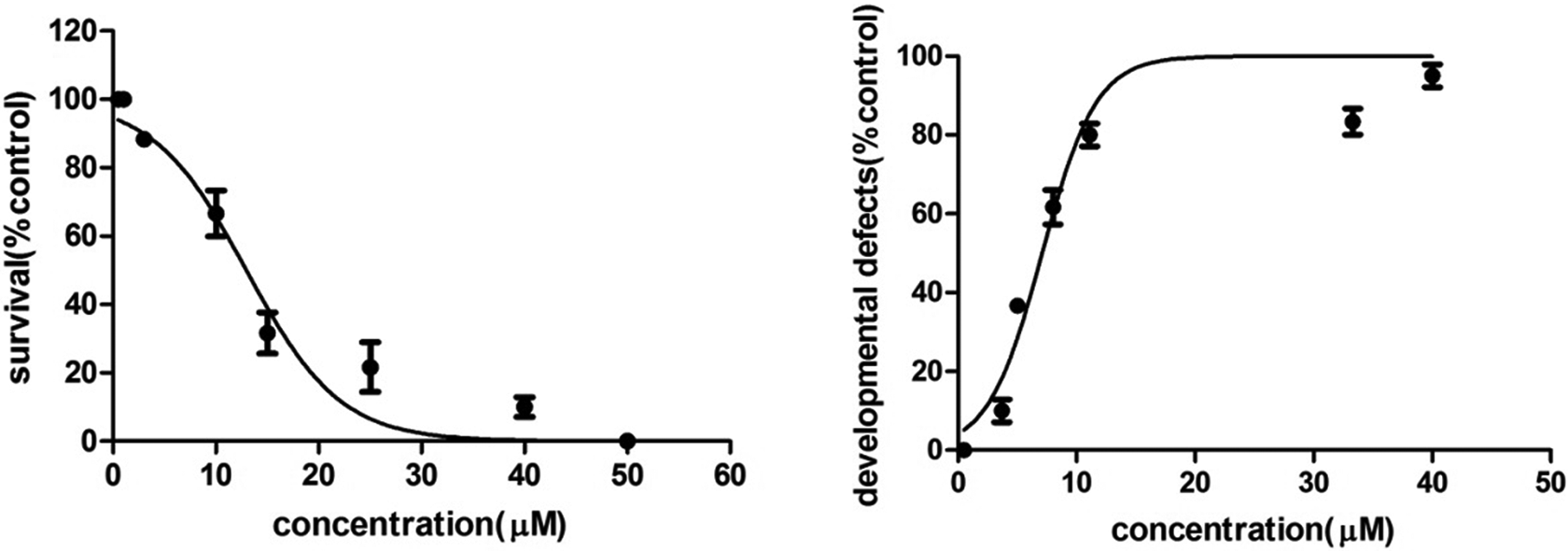
Mycophenolic acid (MPA) induced dose-dependent lethality and developmental defects at 72 hours postfertilization (hpf) in zebrafish.
LC25, LC50, EC50, and NOAEL Values of MPA.

Abbreviations: EC50, 50% teratogenic concentration; LC25, 25% lethal concentration; LC50, 50% lethal concentration; MPA, mycophenolic acid; NOAEL, no observed adverse effect level.
The Developmental Defects Caused by MPA in Zebrafish Embryos
In the developmental toxicity assay, we found MPA induced significant morphological phenotypes, most prominently tail curvature and pericardial edema, in a dose-dependent manner (Figure 2A). Compared with the embryos in the control group, 8.0 µmol/L of MPA-treated embryos showed the severe ventral curvature with the average angle at almost 50° (Figure 2B), as well as the heart edema, which was 2-fold larger than that in the control embryos (Figure 2C).
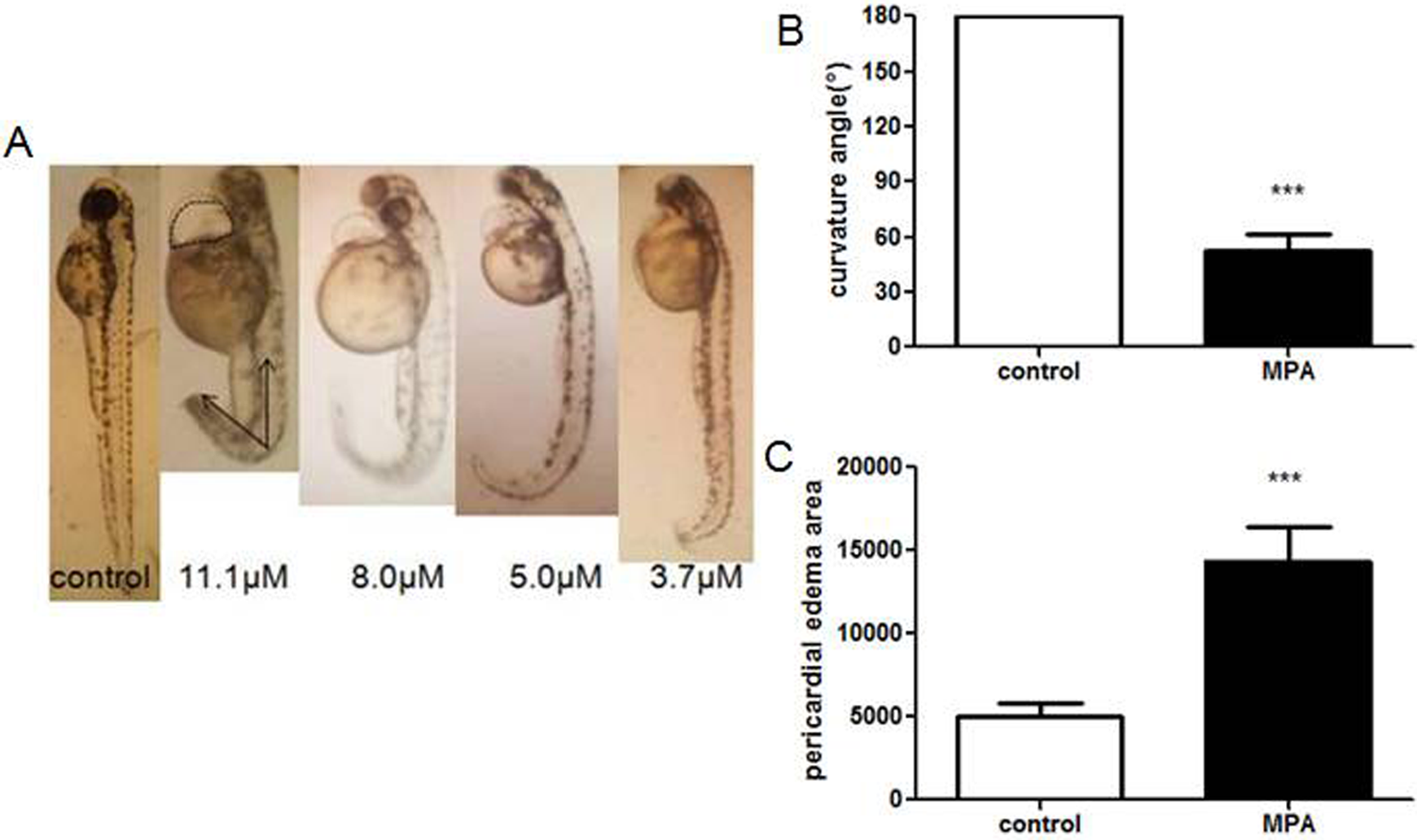
Zebrafish embryos were treated with mycophenolic acid (MPA) at 2 hours postfertilization (hpf) and observed at 72 hpf. Tail curvature angles are indicated by arrows and circled area was pericardial edema (A). Curvature angle (B) and the area of pericardial edema (C) in the 8.0 µmol/L of MPA-treated embryos were calculated with GraphPad Prism 5.0. ***Statistical significance
The Expression of impdh Genes Altered by MPA
As MPA is a known inhibitor of IMPDH, we studied
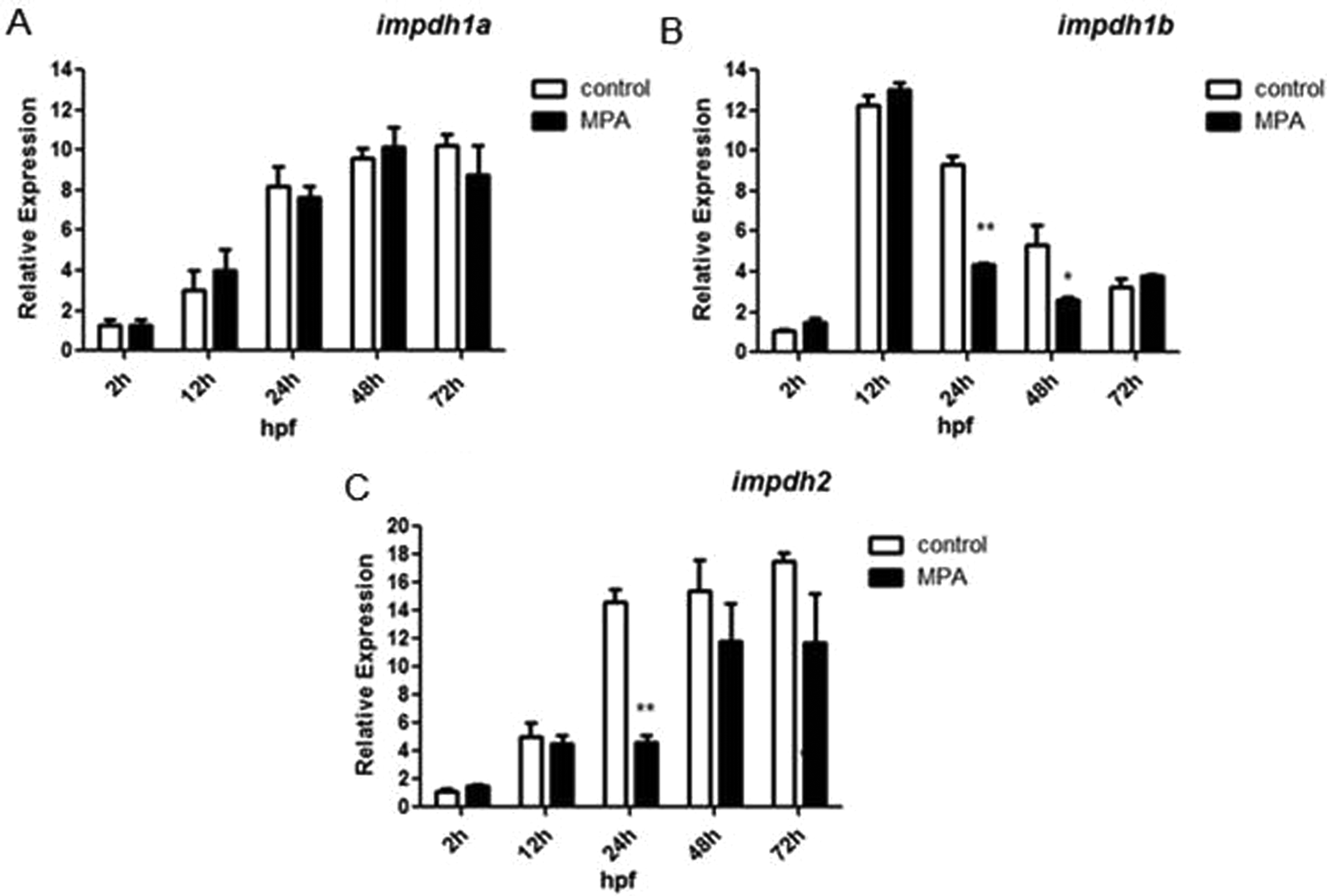
Real-time quantitative polymerase chain reaction (qPCR) analysis of
RESCUE Study of External Guanine for MPA-Exposed Embryos
Our study suggested that the expression of
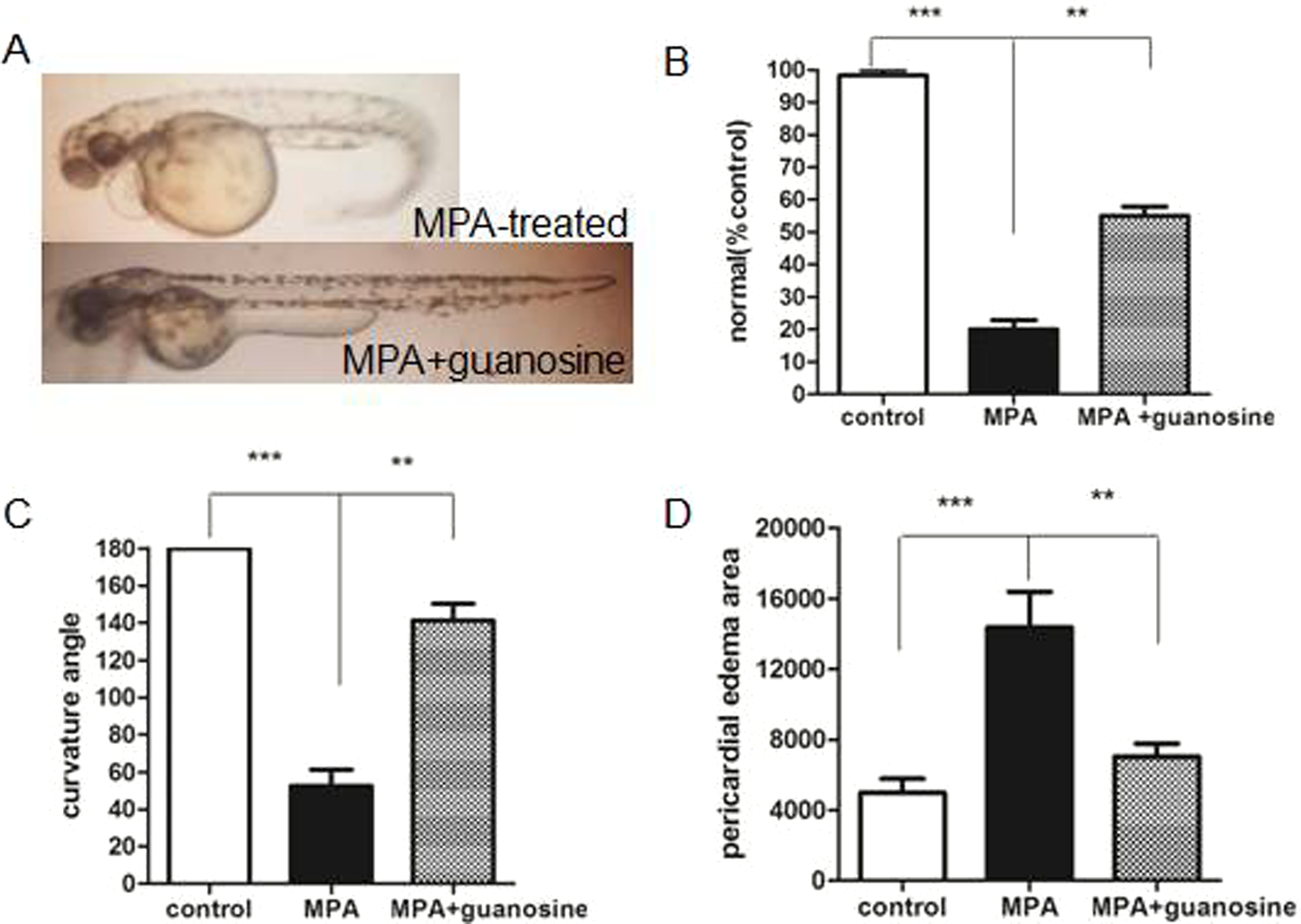
RESCUE study of guanosine supply on the development defects of mycophenolic acid (MPA)-treated embryos. (A) 8.0 µmol/L of MPA-treated embryos recovered at 72 hours postfertilization (hpf) after 33.3 µmol/L of guanosine supply. (B) Curvature angle and (C) the area of pericardial edema were calculated with GraphPad Prism 5.0 for different treatment groups. Data are presented as means ± standard error of the mean (SEM). Significance was considered when
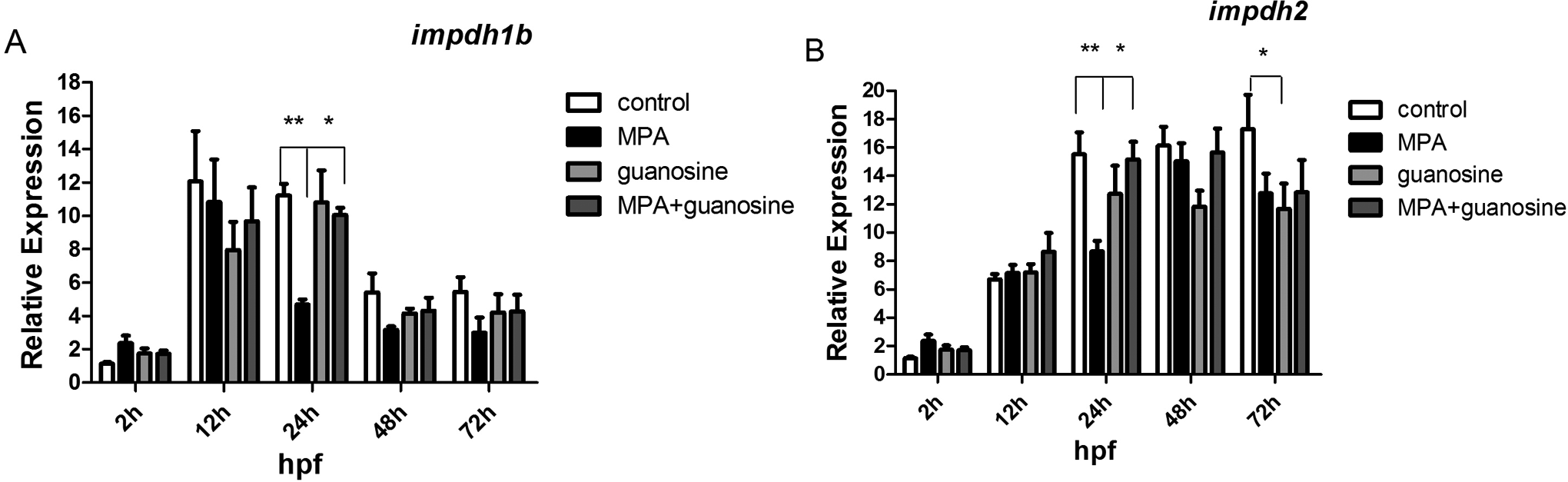
The relative expression levels of mycophenolic acid (MPA) target genes after external guanosine supply (33.3 μmol/L). (A)
In summary, our results demonstrated at morphological level and molecular level that the additional guanosine supply can partially rescue zebrafish embryos from developmental defects caused by MPA treatment.
Discussion
Both animal and human studies revealed that MPA is a teratogen. Echardt group has reported the growth and development defects of rat embryos using whole embryo culture with MPA, and the effects were predominantly localized in the body regions. 21 Additionally, teratology studies of MPA in rabbits also showed fetal resorptions and malformations. 22 A prospective study of the European Network of Teratology Information Services has confirmed that mycophenolate is a human teratogen; there is a high incidence of major malformations (26%) after the first trimester exposure to mycophenolate. 23 The American National Transplantation Pregnancy Registry gathered pregnancy outcomes in female patients after organ transplantation and intake of MPA during gestation. Within this series of cases, 49% of all examined pregnancy outcomes were spontaneous abortions. Birth defects such as microtia and other deformities were also observed. 24 Using the zebrafish embryos, we also found MPA induced significant developmental defects, including tail defects and severe pericardial edema. The teratogenic index (LC25/NOAEL ratio) of MPA was greater than 10, a value representative of a teratogen in zebrafish model. Our results showed the same teratogenic toxicity of MPA as in other reports. Thus, it is concluded that teratogenic toxicity in the zebrafish model has a good correlation with other mammalian models.
Mycophenolic acid caused morphologic toxicity in zebrafish embryo in a dose-dependent manner. As IMPDH was the well-known target of MPA, we conducted qPCR assay to evaluate the expression change of
Mycophenolic acid is a potent, selective, and reversible inhibitor of IMPDH, which catalyzes the oxidation of inosine-5′-monophosphate to xanthosine monophosphate (XMP), a precursor of GMP. Mycophenolic acid exposure to human IMPDH results in cessation of cellular division limited to those cells that do not have the salvage pathway for guanine synthesis.
25
Interestingly, there are few evidence describing developmental toxicity dependent on this pathway. Here, we demonstrated that the impact of MPA on zebrafish was reversible by the addition of guanosine, the substrate of the salvage pathway of guanosine triphosphate (GTP) synthesis.
20
This result may confirm that MPA affects zebrafish development by guanosine nucleotide depletion due to the direct inhibition of
However, external guanosine addition could not totally rescue the development defect phenotype caused by MPA. We speculated there might be other events related to MPA effects on zebrafish. Our further goal will be discovering the remaining molecular targets of MPA in order to provide more mechanistic information useful for the risk assessment of its clinical use.
Although zebrafish has been recognized as a good animal model for the study of “predictive toxicology” (http://www.fda.gov/forconsumers/consumerupdates/ucm343940.htm), the equivalent concentration conversion between zebrafish and human is still lacking. Mycophenolic acid is available in 2 branded products: MMF (CellCept; Genentech, San Francisco, California) and enteric-coated MPA (Myfortic; Novartis, East Hanover, New Jersey). The clinic dose of MMF and enteric-coated MPA for humans is 1 to 2 g/d and 1.44 g/d, respectively.
26
After oral exposure of 1 g MMF, the mean maximum plasma MPA concentration (
Footnotes
Author Contributions
Ling-Ling Jiang and Ming-Fang He contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Mei-Hui Liu and Jian-Ying Li contributed to acquisition and analysis. Zhi-Heng He critically revised the manuscript. Huan Li contributed to the guidance of real-time PCR experiments. Ning Shen contributed to the zebrafish care, breeding, and embryo culture. Ping Wei contributed reagents.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key R & D Special Funds of Jiangsu Province (BE2015696) and New Teacher Fund from Chinese Ministry Education (20123221120005).
