Abstract
The reproductive toxicity potential of the resin monomer triethylene glycol dimethacrylate (TEGDMA; Chemical Abstracts Service Registry Number 109-16-0) was investigated in male and female Crl:CD1(ICR) mice, 4 dosage groups, 25 mice/sex/group. Formulations of TEGDMA (0, 0.01, 0.1, or 1.0 mg/kg/d) in reverse osmosis-processed deionized water were intubated once daily beginning 28 days before cohabitation and continuing through mating (males) or through gestation day 17 (females). The following parameters were evaluated: viability, clinical signs, body weights, estrous cyclicity, necropsy observations, organ weights, sperm concentration/motility/morphology, cesarean-sectioning and litter observations, and histopathological evaluation of select tissues. No deaths or clinical signs related to TEGDMA occurred. No significant changes in male and female body weights and body weight gains were recorded for any of the administered dosages of TEGMDA. All mating and fertility parameters and all litter and fetal data were considered to be unaffected by dosages of TEGMDA as high as 1 mg/kg/d. Gross or histopathologic tissue changes attributable to the test article were not observed. Reproductive and developmental no observed adverse effect levels (NOAELs) for TEGMDA were 1.0 mg/kg/d, the highest dose tested. Comparison of conservatively estimated TEGDMA exposures from dental treatments to the NOAEL of 1.0 mg/kg/d identified in this study indicates margins of exposure of at least 120- to 3000-fold depending on the exposure scenario. The results of this study support the continued safe use of TEGDMA in polymeric dental products applied according to the manufacturers’ instructions.
Introduction
The use of polymeric composite dental materials has increased dramatically since their introduction in the 1960s, as a result of their aesthetic qualities, improved handling and wear characteristics, and perceived health concerns associated with use of mercury amalgam. 1 In 2010, approximately 65% of dental restorations made in the United States used polymeric composite restorative materials. 1
Polymeric composite dental restorative materials are designed to replace biological tissue both in appearance and in function. The development of these materials is challenging. The resulting product must have a stable shelf life of several years plus the ability to react rapidly in the clinical setting to form a cross-linked polymer with high modulus, high hardness, and high glass transition temperature. 2 At the same time, the material must match the thermal expansion of the tooth, minimize extractable components and moisture uptake, be chemically inert, have minimal potential for shrinkage and shrinkage stress, and present an aesthetic appearance.
To date, methacrylate resin formulations represent the most successful approach to meeting these complex design criteria, and composites of this type currently dominate the commercial market. 2 The resin phase of these formulations consists of dimethacrylate monomers that convert from viscous liquid form to a cross-linked polymer network when exposed to light. Typically, bisphenol A glycidyl methacrylate, ethoxylated bisphenol A dimethacrylate, and/or urethane dimethacrylate are used as base monomers to produce dental materials with the essential qualities of adequate mechanical strength, rapid polymerization, and low shrinkage. 2 Because these base resins are highly viscous, a diluent resin is added to reduce the overall viscosity of the composite formulation, enabling higher filler loading and increased monomer conversion. 2 Triethylene glycol dimethacrylate (TEGDMA; Chemical Abstracts Service Registry Number [CASRN] 109-16-0; Figure 1), a low-viscosity reactive resin, is the most commonly used diluent for this purpose.
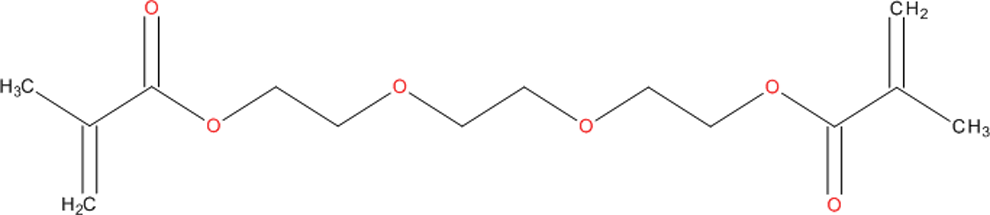
Structure of TEGDMA. TEGDMA indicates triethylene glycol dimethacrylate.
The potential for leaching of residual (unpolymerized) TEGDMA monomer from various types of polymerized composite dental materials has been recognized at least since the 1990s. 3 –8 The rate and extent of leaching, and thus potential exposure, depend on multiple factors, including product composition, amount applied, extent of cure, and cross-linking density. Numerous published studies have quantified TEGDMA release into organic and aqueous solvents under laboratory conditions. A recent meta-analysis 9 of data from 17 published studies calculated sample surface area-based geometric mean 24-hour and total cumulative release rates (figures rounded) of 0.19 and 0.32 nmol/mm 2 , respectively, for TEGDMA extraction in organic solvents. For aqueous solvents, the 24-hour and total cumulative release rates (figures rounded) were 0.04 and 0.15 nmol/mm2, respectively. When expressed on a sample volume basis, the geometric mean 24-hour and total cumulative release rates (figures rounded) were 2.51 and 5.67 nmol/mm3, respectively, for organic solvents and 0.21 and 3.15 nmol/mm3 for aqueous solvents.
In the clinical setting, TEGDMA may leach from composite dental materials into salivary fluids resulting in contact with the oral mucosa and gastrointestinal tract. Alternatively, some TEGDMA may leach into dentin and diffuse toward the pulp. 3 Oral exposure studies of 14 C-radiolabeled TEGDMA in guinea pigs and mice indicate that it is rapidly absorbed from the gastrointestinal tract. 10,11 Triethylene glycol dimethacrylate is widely distributed throughout the body following oral, intravenous, or intradermal administration. 10,11 Clearance is essentially complete within 24 hours of oral or intradermal dosing and within 48 hours of intravenous administration. 10 –12 In guinea pigs, approximately 60% of the 14 C-radiolabel is excreted as CO2 via the lungs and approximately 15% to 20% is excreted in the urine. 10 –12
Published in vivo data on the health effects of TEGDMA are limited to 2 studies, 13,14 reporting that TEGDMA produced adverse effects on the fertility and reproductive systems of male and female mice. In a study of male reproductive toxicity, Darmani and Al-Hiyasat 13 reported that TEGDMA, administered intragastrically in an alcoholic water solution to male mice for 28 days at 25 or 100 mcg/kg/d (0.025 or 0.100 mg/kg/d, respectively), caused statistically significant reductions in body weights and testis weights and reductions in the absolute weights of seminal vesicles and preputial glands. Testicular and epididymal sperm counts were also significantly reduced. Unexposed female mice mated to males exposed to 25 or 100 mcg/kg/d had significantly reduced pregnancy rate and an increase in the ratio of total resorptions to total implantation sites. Unexposed females mated to males exposed to 100 mcg/kg/d had a significantly increased number of resorptions. In a study of female reproductive toxicity by the same authors, 14 female mice exposed to 100 mcg/kg/d TEGDMA by intragastric gavage for 28 days prior to mating with unexposed males showed a significant reduction in pregnancy rate and significant increases in the total number of resorptions. Female mice exposed to 25 or 100 mcg/kg/d by intragastric gavage for 28 days prior to mating with unexposed males had significantly reduced body weight and uterus weights. A clear dose-response trend was not evident for the majority of the responses observed following treatment of males or females.
Interpretation of these study results was limited by several factors related to study design and execution, including the use of small group sizes for this type of study (10 animal/sex/dose), lack of information on preparation and delivery of dosing solutions, and choice of statistical methods for analysis of the results. Consequently, uncertainty regarding the results reported previously and the absence of a full reproductive and developmental toxicity study in mice prompted the initiation of the present study. Reproductive studies are usually conducted primarily in rats but due to the frequent use of the mouse in the published literature, verification of results in this species was considered essential.
Materials and Methods
Regulatory Compliance
A modified 1-generation reproductive toxicity study was designed and conducted following recommendations in Organization for Economic Cooperation and Development (OECD) Test Guideline 415. 15 In a 1-generation study, the test material is administered to male and female animals for a period of time covering sperm development in the males and at least 1 or 2 ovulations in the females through mating, gestation, lactation, and growth of the next generation. The study conformed to Good Laboratory Practice (GLP) regulations promulgated by the US Food and Drug Administration 16 and OECD. 17
Materials
The test substance was TEGDMA; CASRN 109-16-0, Lot # 082806, a clear, colorless liquid. The test substance was provided by the 3M ESPE Dental Products Division, St Paul, Minnesota and stored at 2°C to 8°C and protected from light.
The control article (vehicle) for the TEGDMA formulations was reverse osmosis-processed deionized water. Throughout the study all formulations (solutions) were prepared weekly at the testing facility and were stored in refrigerator (2°C-8°C) and protected from light. Prepared formulations were stirred continuously during sample collection and dosage administration. Each batch was analyzed for concentration.
Animals
Male and female Crl:CD1(ICR) mice (Charles River Laboratories, Inc, Raleigh, North Carolina and St Constant, Quebec, Canada) were used in the study because (1) they are a mammalian species accepted for use in toxicity studies; (2) historical data and experience exist at the testing facility; and (3) they are generally comparable (outbred strains) to the mice used in 2 existing studies of this compound. 13,14
Mice, approximately 50 days of age, arrived at the testing facility in 4 shipments and were assigned to individual housing on the basis of computer-generated random units to 4 replicates to accommodate the testing facility scheduling. After acclimation (14 days), male mice were selected for study on the basis of physical appearance and body weights. Female mice were selected after acclimation for 12 days, based on an estrous cycle evaluation, appearance, and body weights. The mice were then assigned to groups based on computer-generated (weight ordered) randomization procedures. The weight variation in mice used in this study did not exceed ±20% of the mean body weight of each sex. Male mice weighed 27.8 to 31.2 g and female mice weighed 19.4 to 29.8 g at study assignment.
Study rooms were maintained under conditions of positive airflow relative to a hallway and independently supplied with a minimum of 10 changes per hour of 100% fresh air that had been passed through 99.97% high-efficiency particulate absorption filters. Room temperature and relative humidity were targeted at 64°F to 79°F (18°C-26°C) and 30% to 70%, respectively. An automatically controlled 12-hour light–12-hour dark fluorescent light cycle was maintained. Mice were individually housed in stainless steel, wire-bottomed cages, except during cohabitation period when each pair of male and female mice was housed in the male’s cage. All cage sizes and housing conditions were in compliance with the Guide for the Care and Use of Laboratory Animals. 18
Mice were given ad libitum access to Certified Rodent Diet #5002 (PMI Nutrition International, Inc, St Louis, Missouri) in individual feeders. Water was available via an automatic watering access system to which chlorine was added as a bacteriostat.
Methods
Dosage Justification and Administration
The test substance, TEGDMA, is a leached component of approved and marketed dental composites; therefore, the oral (gavage) route was selected for testing because (1) it is the primary route of human patient exposure; (2) exact dosage can be accurately administered; and (3) 2 published studies 13,14 reported that oral dosages of 25 and 100 mcg/kg (0.025 and 0.100 mg/kg, respectively) TEGDMA had caused adverse fertility and reproductive effects in male and female mice.
Relevant dosages were also investigated in a 14-day dosage-range finding study in which 0.0375, 0.075, 0.5, or 5.0 mg/kg of TEGDMA were intubated daily into male and female mice. Triethylene glycol dimethacrylate produced no adverse effects at any dose level; therefore, dosages of 0 (vehicle), 0.01, 0.1, and 1.0 mg/kg/d of TEGDMA were selected for the reproductive study based on the consideration of TEGDMA concentrations observed in dental composite leaching studies and default safety factors used in risk assessment.
In the present study, the treatment period for male mice began 28 days before cohabitation (maximum 14 days) and continued through the day before euthanasia at completion of the cohabitation period. Treatment of female mice also began 28 days before cohabitation but continued through gestation day 17 (GD 17). Dosages were adjusted daily for body weight changes and intubated at approximately the same time each day. Specific information on the dosing regimen is summarized in Table 1.
Dosage Regimen for Male and Female Mice Intubated With TEGDMA.a,b
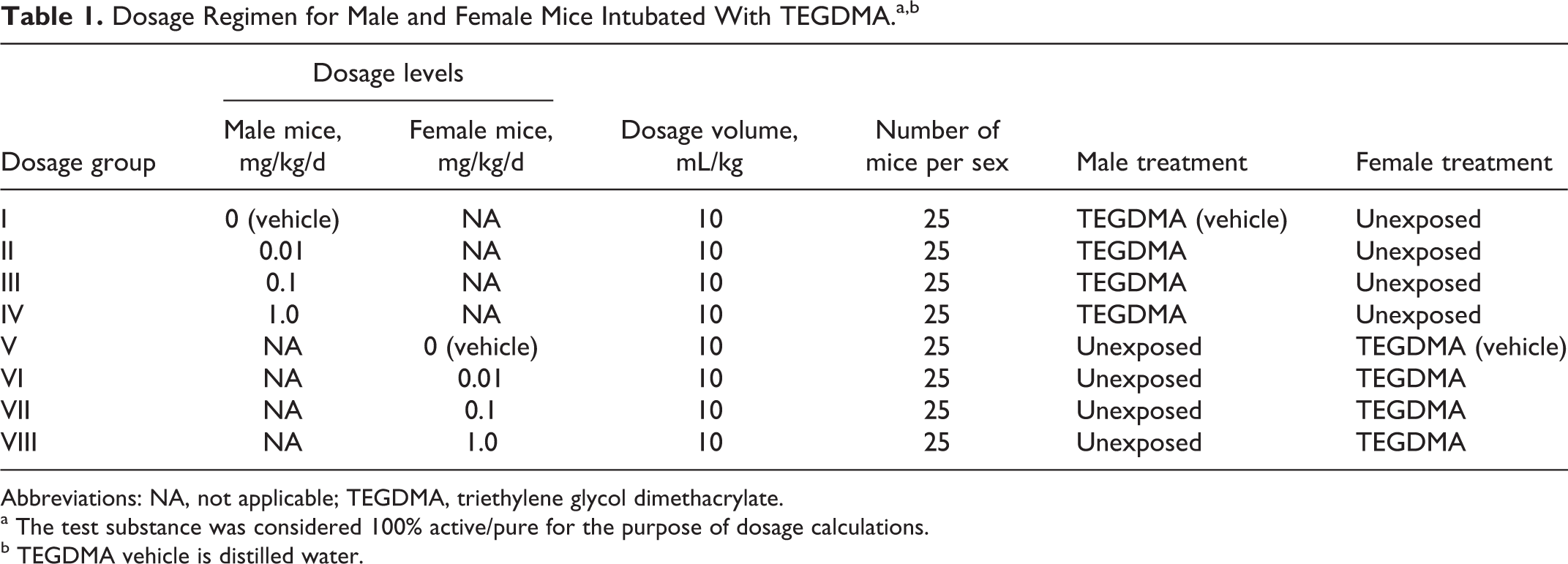
Abbreviations: NA, not applicable; TEGDMA, triethylene glycol dimethacrylate.
a The test substance was considered 100% active/pure for the purpose of dosage calculations.
b TEGDMA vehicle is distilled water.
Animals were observed twice daily for viability and examined for abnormal clinical signs, abortions, or premature deliveries before dosage administration and approximately 2 hours later. Body weights of treated mice were recorded weekly prior to the start of the study and daily during the dosage period and at euthanasia. Non-TEGDMA-exposed male mice were weighed weekly; non-TEGDMA-exposed females were weighed weekly during the acclimation and precohabitation periods and on GDs 0, 6, 9, 12, 15, and 18. Estrous cycling was evaluated by examination of vaginal cytology for 9 days before initiation of treatment, for 27 days beginning with the day after the first treatment, and then until a copulatory plug was observed in situ during the cohabitation period. In all cases, exposed mice were mated with nonexposed mice. Female mice with a copulatory plug in situ were considered to be at GD 0 and assigned to individual housing, as were female mice not mated within the 14-day cohabitation period.
All mice were euthanized by inhalation of carbon dioxide. Male mice were subjected to a gross necropsy of the thoracic, abdominal, and pelvic viscera, and the following reproductive organs were weighed: right and left testis, left epididymis (whole and cauda), right epididymis, and seminal vesicles (with coagulating gland and with and without fluid). With the exception of the pituitary, which was weighed after fixation, brain, kidneys, and liver were also weighed prior to fixation in all treated and vehicle control male mice. Sperm concentration and motility were evaluated using computer-assisted sperm analysis for all male mice treated with vehicle or a test article. Motility and sperm concentration (sperm/g tissue weight) were also evaluated via the Hamilton Thorne integrated visual optical system (IVOS) by collection of samples from the left cauda epididymis, while the remaining portion of the left cauda was used to manually evaluate sperm morphology. Brain, epididymides, kidneys, liver, pituitary, prostate, and seminal vesicles (with coagulating gland) were retained in 10% neutral-buffered formalin (NBF) for histological examination. Testes were initially fixed in Bouin solution and then transferred to 10% NBF.
Female mice were cesarean sectioned, and uteri of nonpregnant mice were examined while pressed between glass plates to confirm the absence of implantation sites. Uteri from pregnant mice were excised and examined for number and distribution of implantations, live and dead fetuses, and resorptions. Numbers of corpora lutea on each ovary were also recorded. Brain, kidneys, liver, ovaries, pituitary (fixed weight), and uterus were weighed and retained in 10% NBF.
Fetuses were removed from the uterus, weighed, and examined for gender and gross external alterations. Live fetuses were then euthanized by an intraperitoneal injection of pentobarbital before undergoing further examination. Approximately half of the fetuses in each litter were fixed in Bouin solution for possible future soft-tissue evaluation; the remaining fetuses were retained in alcohol for possible future skeletal evaluation.
Histopathological examination was performed on tissues from 10 randomly selected exposed male mice from groups I and IV and 10 randomly selected exposed female mice from groups V and VIII. The tissues were routinely processed, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. All gross lesions were also examined. Testes were fixed in Bouin fixative prior to being transferred to formalin. This procedure assures good fixation of the testes to allow for an adequate evaluation of all stages of sperm development and good visualization of the tubules in the testes. Periodic acid Schiff and hematoxylin staining was used for evaluation of the male reproductive organs.
Data generated during the course of study were recorded either by hand or by using the Argus Automated Data Collection and Management System, the Vivarium Temperature and Relative Humidity Monitoring System, and the Hamilton Thorne IVOS. Data were tabulated, summarized, and/or statistically analyzed using the above-mentioned systems in conjunction with Microsoft Excel (Microsoft Office 97/2000/XP) and/or the SAS System (version 6.12). Clinical observation and other proportional data were analyzed using the variance test for homogeneity of the binomial distribution. 19 Continuous data were analyzed using the Bartlett test of homogeneity of variances 20 and the analysis of variance. 21 The Dunnett test 22 was used to identify statistical significance of individual groups. If the analysis of variance was not appropriate, the Kruskal-Wallis test 23 or Dunn method of multiple comparisons 24 was used to identify the statistical significance of the individual groups. If there were greater than 75% ties, the Fisher exact test 25 was used to analyze the data.
Results
Analytical
Concentrations and purity of the bulk TEGDMA and dosing solutions were analyzed by high-performance liquid chromatography and/or ultraviolet methodology. Chromatograms from bulk test articles at the beginning and end of the study did not differ. Weekly concentration analyses for TEGDMA formulations, used for administration, were all within ±10% of targeted concentrations except for one 0.01 mg/kg sample with a −16% mean bias. Homogeneity, based on triplicate samples from the top, middle, and bottom of a preparation, was within the acceptable range of ≤5% relative standard deviation. Stability of TEGDMA formulations was confirmed for 10 days of refrigeration at 5°C.
Treated Male Mice
No deaths or clinical signs occurred that were related to TEGDMA. One male mouse in the 0.01 mg/kg/d TEGDMA dosage group was euthanized due to an intubation error. Body weights, body weight gains, and terminal body weights were not affected by dosages of TEGDMA as high as 1.0 mg/kg/d. All values were comparable among the dosage groups and did not differ significantly (Figure 2).
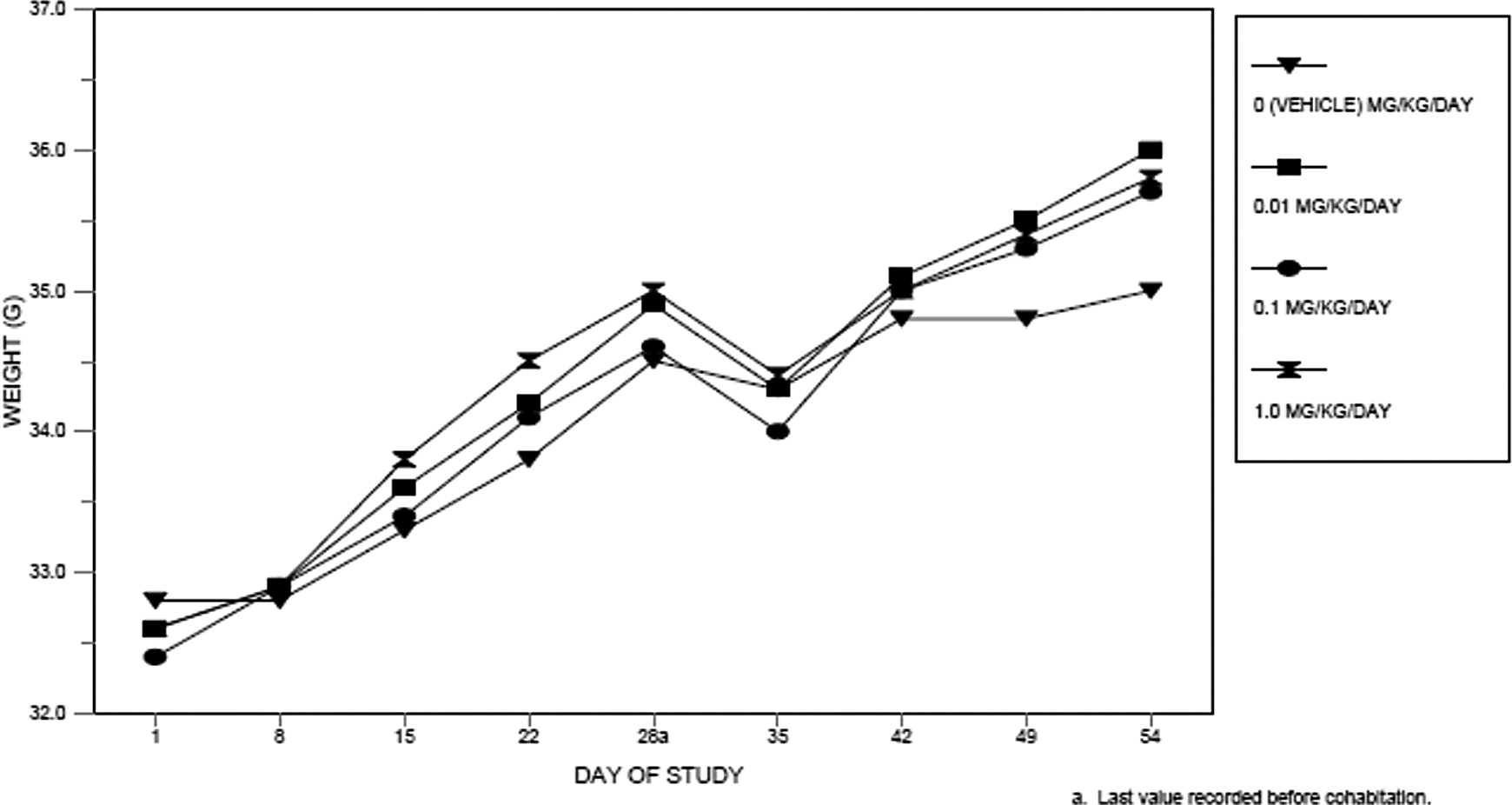
Body weights of male mice.
All mating and fertility parameters (days in cohabitation, mice that mated, the fertility index [pregnancies per mated mice], mice with confirmed mating dates during the first week of cohabitation, and number of pregnancies per number of mice in cohabitation) were comparable among control and treatment groups (Table 2). The test facility’s historical range for pregnancy rate is 80% to 100%. Litter averages for implantations, corpora lutea, litter size, live fetuses, dead fetuses, early and late resorptions, fetal weights, sex ratio, and percentage of dead and resorbed fetuses per litter were unaffected by paternal dosages as high as 1 mg/kg/d of TEGDMA (cesarean data from the unexposed female mice are not shown).
Mating and Fertility: TEGDMA-Treated Male Mice With Unexposed Female Mice.a
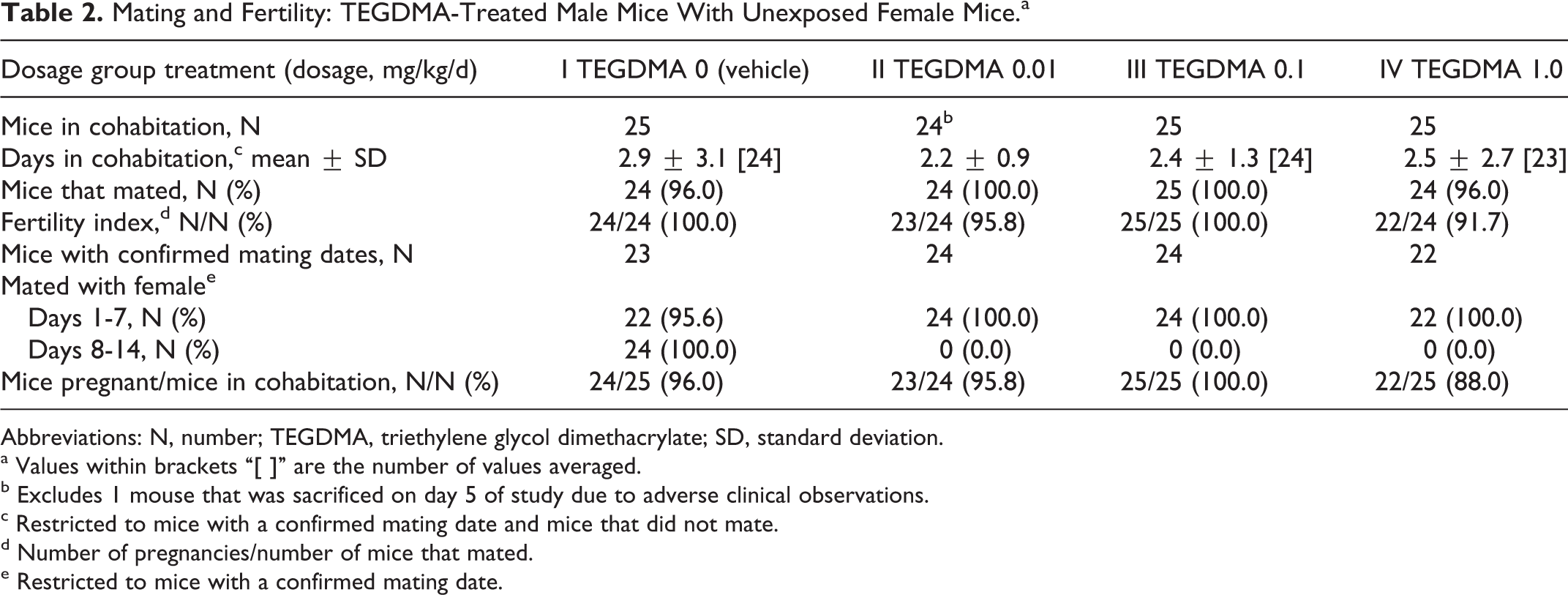
Abbreviations: N, number; TEGDMA, triethylene glycol dimethacrylate; SD, standard deviation.
a Values within brackets “[ ]” are the number of values averaged.
b Excludes 1 mouse that was sacrificed on day 5 of study due to adverse clinical observations.
c Restricted to mice with a confirmed mating date and mice that did not mate.
d Number of pregnancies/number of mice that mated.
e Restricted to mice with a confirmed mating date.
There were no gross morphological tissue changes at necropsy on study day 54 related to either test article. Absolute weights of the pituitary, brain, liver, kidneys, epididymides, caudal epididymis, testes, seminal vesicles (with and without fluid), and prostate, and the ratios of these organ weights to brain weight were unaffected by the administered dosages of TEGDMA. Only the ratio of the brain weight to the terminal body weight was significantly reduced in all 3 TEGDMA dosage groups compared to the control group value (Table 3). These reductions were not considered related to the test article because absolute terminal body and brain weights did not differ among the groups, and the reductions were not dosage dependent.
Ratios (%) of Organ Weight to Terminal Body Weight in Treated Male Mice.a,b
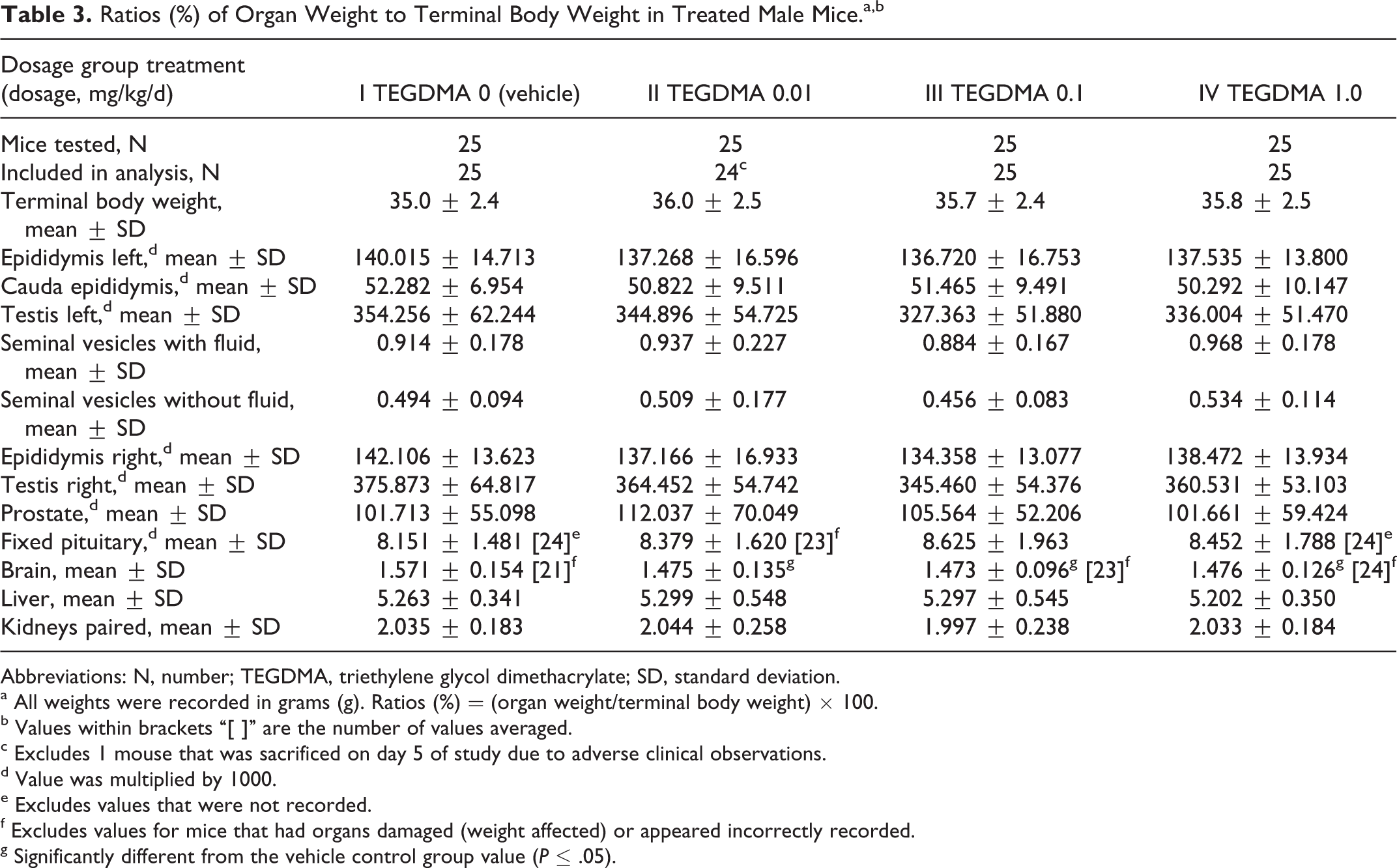
Abbreviations: N, number; TEGDMA, triethylene glycol dimethacrylate; SD, standard deviation.
a All weights were recorded in grams (g). Ratios (%) = (organ weight/terminal body weight) × 100.
b Values within brackets “[ ]” are the number of values averaged.
c Excludes 1 mouse that was sacrificed on day 5 of study due to adverse clinical observations.
d Value was multiplied by 1000.
e Excludes values that were not recorded.
f Excludes values for mice that had organs damaged (weight affected) or appeared incorrectly recorded.
g Significantly different from the vehicle control group value (P ≤ .05).
There were no statistically significant differences in sperm parameters, including sperm morphology (not shown), between the control group and treatment groups intubated with dosages as high as 1 mg/kg/d of TEGDMA (Table 4). Test article-associated histopathological changes were not observed in any organs mentioned above from 10 randomly chosen male mice in the TEGDMA high-dose group.
Sperm Motility, Count, and Density in Treated Male Mice.
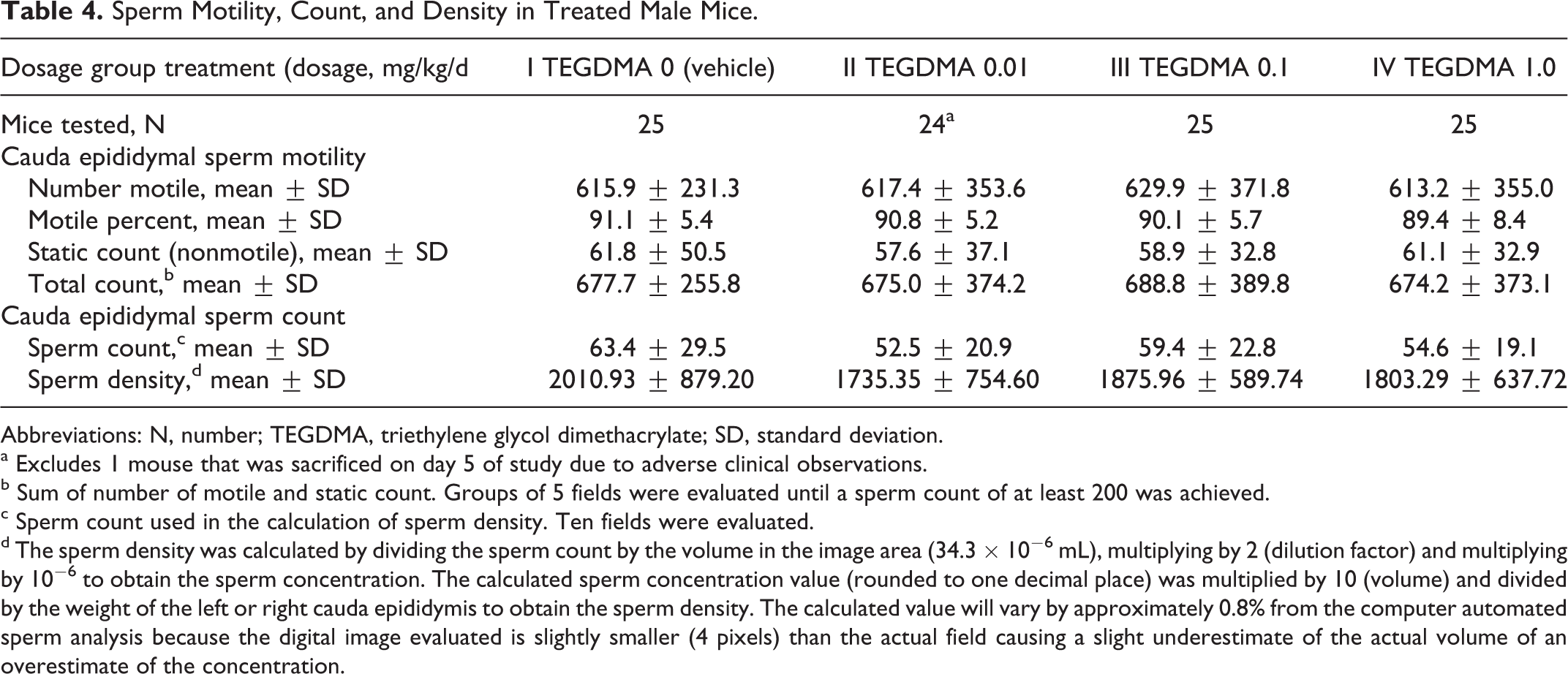
Abbreviations: N, number; TEGDMA, triethylene glycol dimethacrylate; SD, standard deviation.
a Excludes 1 mouse that was sacrificed on day 5 of study due to adverse clinical observations.
b Sum of number of motile and static count. Groups of 5 fields were evaluated until a sperm count of at least 200 was achieved.
c Sperm count used in the calculation of sperm density. Ten fields were evaluated.
d The sperm density was calculated by dividing the sperm count by the volume in the image area (34.3 × 10−6 mL), multiplying by 2 (dilution factor) and multiplying by 10−6 to obtain the sperm concentration. The calculated sperm concentration value (rounded to one decimal place) was multiplied by 10 (volume) and divided by the weight of the left or right cauda epididymis to obtain the sperm density. The calculated value will vary by approximately 0.8% from the computer automated sperm analysis because the digital image evaluated is slightly smaller (4 pixels) than the actual field causing a slight underestimate of the actual volume of an overestimate of the concentration.
Treated Female Mice
No deaths or clinical signs related to TEGDMA were observed. One female mouse in the 1.0 mg/kg/d TEGDMA dosage group was euthanized on GD 9 due to an early (mistimed) pregnancy, and 1 female mouse in the vehicle control group was euthanized on GD 9 due to an intubation error. All other female mice survived to scheduled sacrifice on GD 18.
Body weights and body weight gains during the precohabitation and gestation periods, as well as terminal body weights, were unaffected by dosages as high as 1 mg/kg/d TEGDMA. All values were comparable among the dosage groups and did not differ significantly (Figure 3).
Body weights of female mice.
Estrous cycling and all mating and fertility parameters (days in cohabitation, mice that mated, the fertility index [pregnancies per mated mice], mice with confirmed mating dates during the first week of cohabitation, and number of pregnancies per number of mice in cohabitation) were unaffected by the maternal administration of TEGDMA (Table 5). Cesarean-sectioning and litter parameters also were devoid of adverse effects and were unaffected by any of the maternal dosages of TEGDMA (Tables 6 and 7).
Estrous Cycling, Mating, and Fertility of Treated Female Mice Mated With Unexposed Male Mice.a

Abbreviations: N, number; TEGDMA, triethylene glycol dimethacrylate; SD, standard deviation.
a Values within brackets “[ ]” are the number of values averaged.
b Restricted to mice with a confirmed mating date and mice that did not mate.
c Number of pregnancies/number of mice that mated.
d Restricted to mice with a confirmed mating date.
Cesarean-Sectioning Observations in Treated Female Mice.

Abbreviations: N, number; TEGDMA, triethylene glycol dimethacrylate; SD, standard deviation.
a Includes values for 4 mice that did not have a confirmed mating date.
b Includes values for 4 mice that delivered a litter on day 18 of gestation.
c Excludes values for a mouse that was sacrificed on day 9 of gestation due to mistimed pregnancy.
Litter Observations (Cesarean-Delivered Fetuses) in Treated Female Mice.a

Abbreviations: N, number; TEGDMA, triethylene glycol dimethacrylate; SD, standard deviation.
a Values within brackets “[ ]” are the number of values averaged.
b Includes values for 4 mice that did not have a confirmed mating date.
c Includes values for 4 mice that delivered a litter on day 18 of gestation.
d Excludes values for a mouse that was sacrificed on day 9 of gestation due to mistimed pregnancy.
e Excludes litters from a mouse that did not have a confirmed mating date.
f Total of male and female body weights.
There were no gross morphological tissue changes at necropsy related to TEGDMA. Absolute weights of the pituitary, brain, liver, kidneys, ovaries, and nongravid uterus with cervix, and the ratios of these organ weights to terminal body weight and brain weight were unaffected by the administered dosages of TEGDMA (Table 8). Test article-associated histopathological changes also were not observed in any of the tissues listed earlier, which were examined microscopically from 10 randomly chosen female mice in the TEGDMA high-dose group.
Ratios (%) of Organ Weight to Terminal Body Weight for Treated Female Mice.a,b
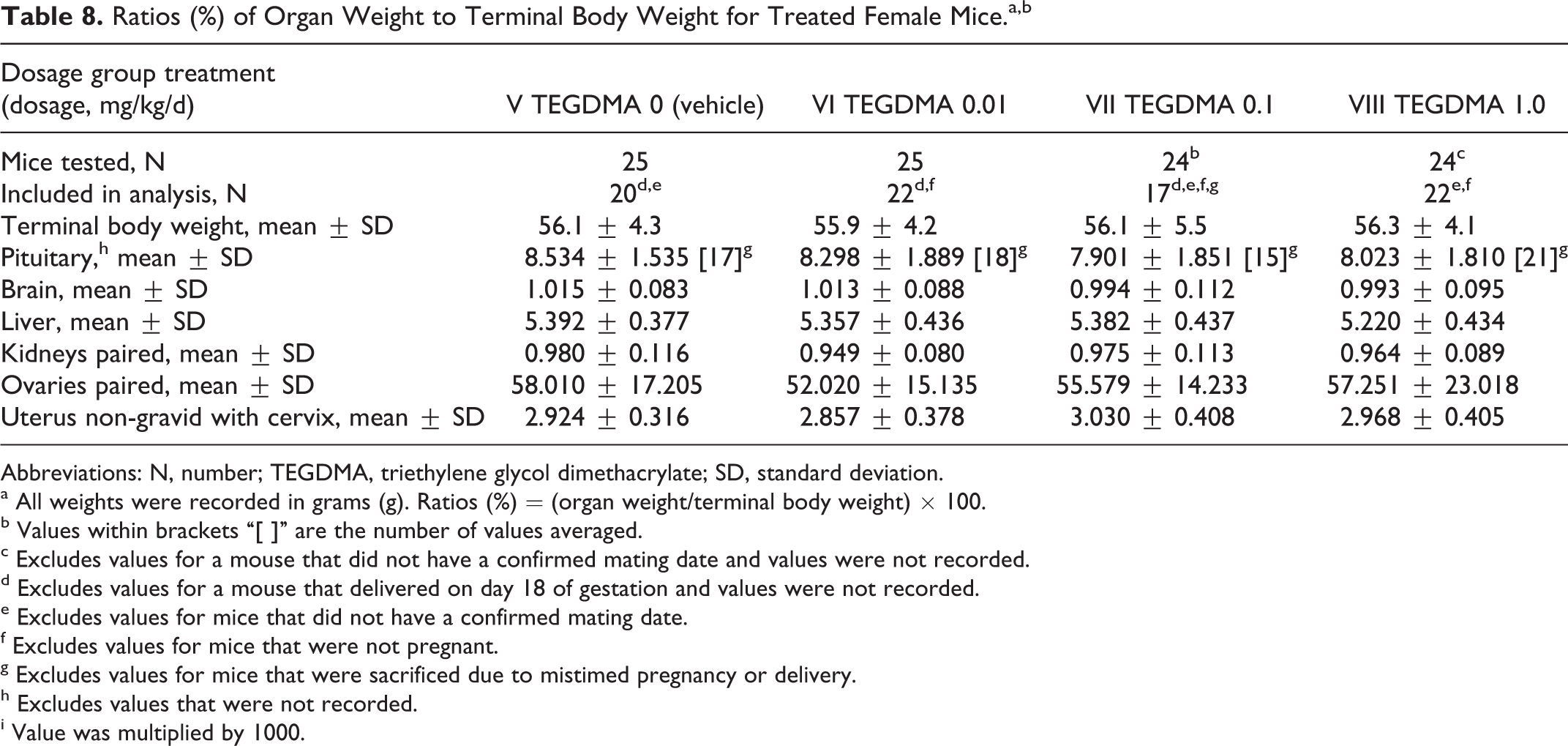
Abbreviations: N, number; TEGDMA, triethylene glycol dimethacrylate; SD, standard deviation.
a All weights were recorded in grams (g). Ratios (%) = (organ weight/terminal body weight) × 100.
b Values within brackets “[ ]” are the number of values averaged.
c Excludes values for a mouse that did not have a confirmed mating date and values were not recorded.
d Excludes values for a mouse that delivered on day 18 of gestation and values were not recorded.
e Excludes values for mice that did not have a confirmed mating date.
f Excludes values for mice that were not pregnant.
g Excludes values for mice that were sacrificed due to mistimed pregnancy or delivery.
h Excludes values that were not recorded.
i Value was multiplied by 1000.
Dosages of TEGDMA as high as 1.0 mg/kg/d administered to female mice prior to and during gestation did not cause any fetal gross external alterations. The only statistically significant finding was the occurrence of 4 fetuses from 2 litters in the 1.0 mg/kg/d TEGDMA dosage group in which exencephaly and open eye lids occurred; 2 of these fetuses also had a protruding tongue. These alterations were considered unrelated to TEGDMA because litter incidence (2 litters), the more relevant parameter, 26 was not significantly increased, and the anomaly (exencephaly with open eye lids and protruding tongue) is common in this strain of mouse (based on the historical control data of the testing facility). The absence of a relationship to the test article is further supported by the nondosage-dependent alteration in 2 fetuses in a litter from an unexposed female mouse mated by a low-dosage group (0.01 mg/kg/d TEGDMA) male mouse; 2 other littermates of these exencephalic fetuses had a domed head, depressed eye bulges, and a medial cleft palate or depressed eye bulges and a medially cleft palate and snout.
Discussion
The increased use of composite resins in dentistry and the observed leaching of components from dental resins have prompted questions about the safety of these dental products. The present reproductive and developmental toxicity study was conducted to provide GLP- and guideline-compliant basic animal reproductive information, which has been lacking, on the widely used resin component TEGDMA. The study was conducted in mice, rather than rats, in order to verify some of the data recently published by Darmani and Al-Hiyasat. 13,14
The results from the present GLP-compliant and guideline-driven mouse study indicate that TEGDMA does not produce any remarkable untoward effects in adult male or female Crl:CD1(ICR) mice, or in the F1 offspring, when administered in dosages as described in the study. Triethylene glycol dimethacrylate did not produce any decrease in pregnancy rate, increase in ovarian weights, increase in resorptions, decrease in sperm counts, or testicular weights, even at dosages that were 10× higher than reportedly administered by Darmani and Al-Hiyasat. 13,14 Routine microscopic examination of reproductive organs from high-dose male and female mice failed to find any histological changes that might indicate any untoward male or female reproductive effects. Based on these data, it was concluded that the reproductive and the developmental no observed adverse effect level (NOAEL) was 1.0 mg/kg/d, the highest dosage tested in this study.
The results of this study are in contrast to the previous studies in mice 13,14 that report TEGDMA-induced impairment of reproduction and fertility in male and female mice at doses as low as 25 mcg/kg/d (0.025 mg/kg/d). A direct comparison of study methodology and results between this study and the earlier studies is difficult because the published descriptions of these studies do not fully report methodological details and data. Nevertheless, it is important to note that there are numerous departures in the published studies from the standard approaches used for conducting reproductive and developmental toxicology studies for risk assessment and regulatory purposes. For example, the number of animals treated per group is smaller than recommended for reproductive toxicity studies. Neither publication reports measured concentration, homogeneity, stability, or administered volume of the dosing solutions. The method used to randomize animals to treatment groups was not reported, and no information was presented on initial body weights as an alternative. Confirmation of mating was not reported. In the evaluation of effects on male reproductive toxicity, males were sacrificed shortly after the mating period, which may have confounded measurement of sperm counts in the epididymis. Data for pregnancy rate are presented for all mated females, without reference to the results for individual, test article-treated breeding males. Finally, it is not clear that appropriate statistical methods were used for all end points. Taken together, these departures from standard methodology significantly limit the use of the Darmani and Al-Hiyasat data for human health risk assessment or regulatory purposes.
The results of the current study address the above-mentioned concerns and provide a basis for screening level risk assessment calculations. The amount of TEGDMA bioavailable from a clinical restoration depends on a number of factors including the surface area or volume of the dental restoration and time elapsed since treatment. The estimated typical surface area for a variety of restorations 9 ranges from 12 to 117 mm2, while estimated typical volume ranges from 7 to 271 mm3. Extraction studies routinely conducted for chemical characterization of polymeric dental devices indicate that bioavailability of residual unpolymerized TEGDMA is greatest within the first 24 hours after curing of the product and then rapidly diminishes with time (data not shown). Assuming 2 restorations per visit, a large restoration surface area value of 117 mm2, an upper confidence interval 9 (CI) peak 24-hour elution rate of 2.88E-10 mol/mm2, and a default body weight of 58 kg for an adult woman, 27 the resulting exposure would be approximately 3.3E-4 mg/kg/d. Comparison to the NOAEL of 1.0 mg/kg/d from the current study, the highest dose tested, indicates a margin of exposure of at least 3000-fold for this scenario using conservative risk assessment assumptions. Alternatively, assuming 2 restorations in 1 visit, a large restoration volume of 271 mm3, an upper CI peak 24-hour elution rate of 2.93E-9 mol/mm2,9 and a default body weight of 58 kg for an adult woman, the resulting exposure would be approximately 7.8E-3 mg/kg/d. The corresponding margin of exposure for this volume-based scenario is greater than 120.
The results of this study support the continued safe use of TEGDMA in polymeric dental products applied according to the manufacturers’ instructions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Lori H. Moilanen and Janell K. Dahms are employees of 3M, a manufacturer of dental products that contain the dental resin monomer analyzed in this study. Alan M. Hoberman is an employee of the laboratory contracted to perform the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study described in this publication was sponsored by 3M.
