Abstract
Objectives:
This study compared different cytotoxicity test models for evaluating resin-based composites (RBCs) and assessed the biocompatibility of standard and bulk-fill RBCs.
Methods:
A standard (spectrum TPH) and a bulk-fill (smart dentin replacement (SDR)) RBC were selected. Disc-shaped specimens (7 mm diameter) of 2 and 4 mm thickness were polymerized for 20 s with a LED curing light of 700 mW/cm2 irradiance. The specimens (n = 5) were subjected to micro-hardness testing and three cytotoxicity test models (direct contact, indirect contact and extract tests) with the established L-929 cell line. Hardness ratios of top and bottom surfaces of specimens were computed to assess the effectiveness of cure. For the direct and indirect contact tests, the cells were stained and zones of inhibition were analyzed after material contact for 24 h. For the extract test, cells were exposed to extracts for 24 h, and cell viability was measured. Data was analyzed using analysis of variance/Scheffe’s post hoc test and Pearson’s correlation (p < 0.05).
Results:
The lowest mean hardness ratio and highest cytotoxicity were observed for TPH at 4 mm. At 4-mm thickness, SDR was found to be biocompatible with all three models. Correlations between hardness ratio and cell viability ranged from r = 0.89–0.96 for the various tests. A significant correlation (r = 0.97) was also observed between the three test models.
Conclusion:
Our data indicated consistency between direct contact, indirect contact and extract test models for cytotoxicity testing of RBCs. Bulk placement and curing at 4 mm for the bulk-fill RBC evaluated did not result in undue cytotoxicity.
Introduction
Amalgam has been used to restore posterior teeth for more than 150 years. Its popularity as a dental restorative material has, however, been on the decline due to environmental concerns and fear of mercury toxicity. With continued technological improvements, resin-based composites (RBCs), which are tooth-coloured composite resins, now have comparable strengths to amalgam and can adhere to tooth structure, reducing the need for unnecessary tooth structure removal for restoration retention. 1 Being tooth-coloured and available in different shades, RBCs are also highly aesthetic and can be used to restore both anterior and posterior teeth. Nonetheless, RBCs shrink during polymerization and the shrinkage stress generated can lead to poor marginal seal, marginal staining, secondary caries, enamel micro-cracks as well as post-operative sensitivity. 2 Moreover, standard light-polymerized RBCs have limited depths of cure of up to 2 mm, entailing the need for incremental placement and adaptation of materials in cavities to ensure optimal material properties. 3 Besides additional clinical time and effort, disadvantages of the increment filling technique include voids or contamination between material layers, bond failure between layers and placement difficulty in small cavities. Innovative bulk-fill RBCs were recently introduced with the aim of simplifying and expediting the restorative procedures. Through the use of novel proprietary resins, modulators, unique fillers and filler control, manufacturers of bulk-fill RBCs claim curing depths of up to 4 mm coupled with low polymerization shrinkage. 4
Dental biomaterials used in the mouth should ideally be harmless to both hard and soft oral tissues. They should also be free of toxic, leachable or diffusible substances that can produce adverse local effects or systemic responses. At curing depths of 4 mm, curing light penetration may be compromised, leading to reduced monomer to polymer conversion, leaching of unreacted monomers and biocompatibility issues. Unreacted components from incompletely polymerized RBCs have been shown to cause irritation of local tissues and pulpal inflammation. 5,6 Unreacted monomers including bisphenol-A, triethylene glycol dimethacrylate, urethane dimethacrylate (UDMA) and hydroxyethyl methacrylate have also been found to have cytotoxic and mutagenic effects on cells both in vitro 7,8 and in vivo. 9 To date, few studies have evaluated the cytotoxicity of bulk-fill RBCs. 10 As the number of varying bulk-fill products continue to increase, the toxicity of bulk-fill RBCs has become a growing concern. There is, therefore, a need to evaluate the cytotoxicity of these bulk-fill RBCs using reliable and clinically relevant in vitro test models.
Several in vitro test models have been proposed by the International Standards Organization (ISO) 10993 guidelines for evaluating the cytotoxicity of dental biomaterials. 11,12 These include the direct contact test model where the biomaterial contacts the cell layer; the indirect contact test where there is a barrier between the biomaterial and cell layer and the extract test where the cells are exposed to the eluate of the biomaterial. The purpose of having three different test models is to provide for a more rigorous evaluation of the biomaterial’s cytotoxicity. Due to resource and technical constraints, most cytotoxicity studies on RBCs do not conduct all three test models in tandem. An ideal in vitro test model should mimic closely the in vivo physiological situation. All three models are, however, relevant to RBCs depending on the specific clinical scenario. The aims of this study were to compare the different ISO 10993 cytotoxicity test models for evaluating RBCs and to assess the effectiveness of cure and biocompatibility of standard and bulk-fill RBCs. The correlation between effectiveness of cure and biocompatibility was also explored. It was hypothesized (null hypothesis) that no significant difference exits between the three cytotoxicity test models as well as the standard and bulk-fill RBCs with regards to effectiveness of cure and biocompatibility.
Materials and methods
Preparation of RBC specimens
The experimental design is illustrated in Figure 1. A standard (spectrum TPH) and a bulk-fill (smart dentin replacement (SDR)) RBC were selected for the study. Both RBCs were from the same manufacturer (Dentsply Caulk, Milford, Delaware, USA). The technical profiles of the materials are shown in Table 1. The RBCs were placed in customized black polyvinyl moulds with cylindrical cavities of 7-mm diameter and depths of either 2 or 4 mm (Figure 2). The moulds were filled in a single increment and excess materials were extruded by compression between two 1-mm thick glass slides. The specimens were subsequently cured for 20 s through the glass slides using the BlueShot LED curing light (Shofu, Kyoto, Japan) with an irradiance of 700 mW/cm2 and an exit window of 8-mm diameter. The consistency of the curing light intensity was verified using an LED radiometer (Kerr Corporation, Middleton, Wisconsin, USA) at regular intervals. Five specimens (n = 5) were made for each material-depth combination and test model. 13
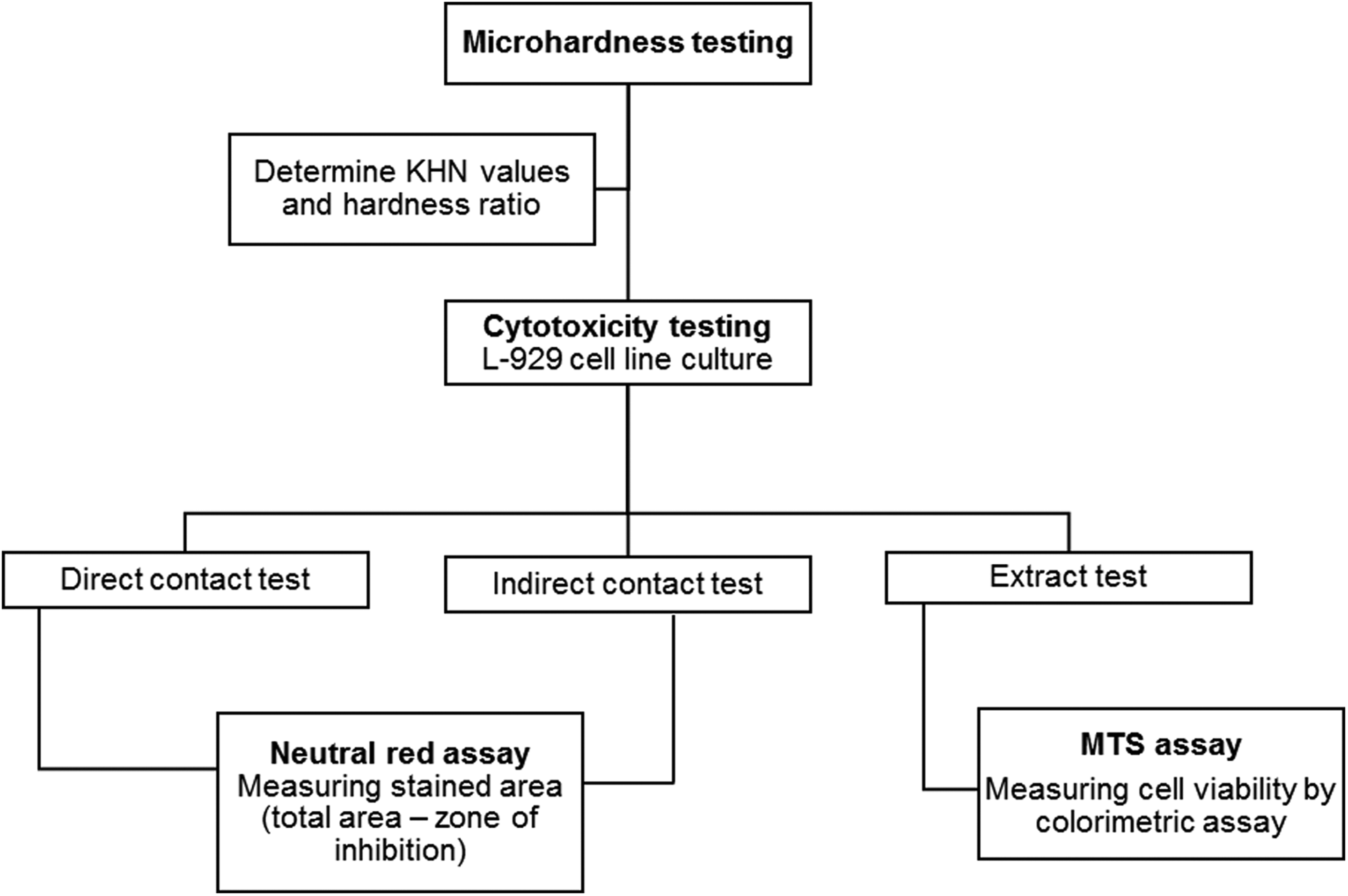
Overview of the experiment.
Technical profiles of the RBCs.

Bis-GMA: bisphenol A glycidyl methacrylate adduct; Bis-EMA: ethoxylated bisphenol A dimethacrylate; TEGDMA: triethylene glycoldimethacrylate; UDMA: urethane dimethacrylate
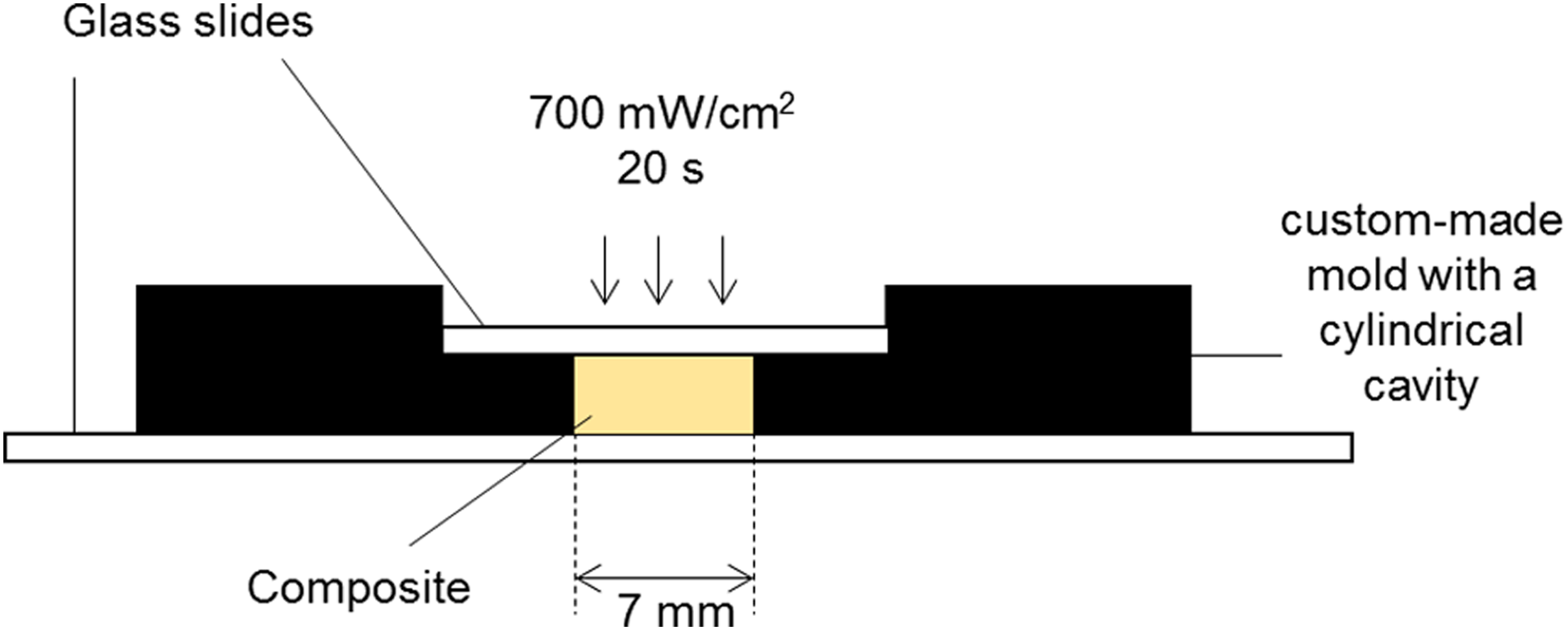
Schematic of the set-up used to fabricate the RBC specimens. RBC: resin-based composite.
Micro-hardness testing
Micro-hardness testing was performed at the top (closer to the curing light) and bottom surfaces of each specimen using a digital micro-hardness tester (FM7; Future-Tech Co., Tokyo, Japan). A 10 gf load force was applied through the indenter at the centre and 1 mm from border of each specimen surface with a dwell time of 15 s. The Knoop hardness number (KHN) corresponding to each indentation was computed by measuring the dimensions of the indentations using the below formula:

Where F is the test load in kgf and d is the longer, diagonal length of an indentation in millimetres.
The hardness ratio of the bottom surface to the top surface for each specimen was subsequently calculated. Effective cure at the bottom surface was set at 80% of the top surface hardness, equivalent to a hardness ratio of 0.8 as advocated by previous studies. 14,15 The specimens were subjected to cytotoxicity evaluation immediately after micro-hardness testing (Figure 3).
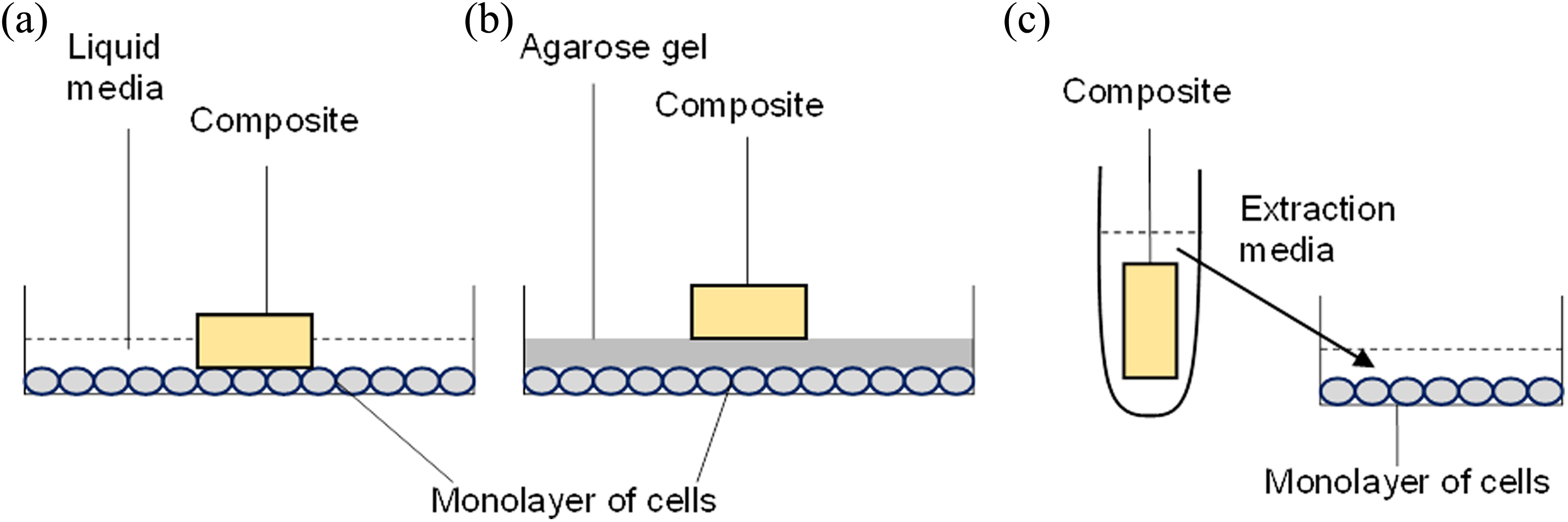
Experimental setup for (a) direct contact test, (b) indirect contact test and (c) extract test.
Cell culture
L-929 mouse fibroblasts were purchased from the American Type Culture Collection (ATCC, Manassas, Virginia, USA) and cultured in complete medium comprised of high glucose Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% foetal bovine serum (Biowest SAS, Nuaillé, France), 100 µg/mL streptomycin and 100 IU/mL penicillin (Biowest SAS). Culture of L-929 cells was performed within an incubator (ESCO, Singapore) set at 37°C, in a humidified atmosphere of 95% air and 5% CO2.
Direct contact test
The cells were counted using a haemocytometer and seeded at a density of 1.25 × 106 cells/5 mL in a 60-mm tissue culture dish. The cells were then incubated for 24 h for cell attachment. The specimens were surface swabbed with 70% ethanol and allowed to dry completely for at least 15 min inside the biosafety cabinet with the lights turned off. Each specimen was placed directly on top of the cell monolayer, with the bottom surface in contact with the cells, at the centre of the dish (Figure 3(a)). A 5 mL of culture medium was then added. The dishes were further incubated at 37°C, in a humidified atmosphere of 95% air and 5% CO2 for 24 h. This simulates the clinical situation where the bottom or least cured surface is in close approximation with underlying tissues.
Indirect contact test
For the indirect contact test (Figure 3(b)), the liquid medium was replaced by 1% agarose-complete medium (Sigma-Aldrich, St Louis, Missouri, USA). After gelation of the agarose, the composite specimen was placed in the centre of the plate on top of the agarose layer with the bottom surface in close proximity to the underlying cells. The dishes were further incubated at 37°C, in a humidified atmosphere of 95% air and 5% CO2 for 24 h. This simulates the clinical situation where there is a dentine barrier between the bottom or least cured surface and the underlying tissues.
Neutral red staining
Neutral red is a weak cationic dye that diffuses intracellularly and stored in lysosomes. Cytotoxicity of dental materials will result in the loss of membrane integrity, reducing retention of the stain and give a decolourized appearance relative to healthy cells. The procedures performed in this experiment were according that described by Borenfreund and Puerner. 16 Neutral red stock solution was prepared by mixing 5.5 mg of neutral red powder in 1.66 mL of distilled water. Neutral red working solution was prepared on the day of staining by mixing neutral red working solution with DMEM in a ratio of 1:80. The dishes were taken out from the incubator and the culture media and RBC specimens were then removed. A 2 mL of neutral red working solution was added to the dishes and incubated for 3 h at 37°C in a dark place. After 3 h, the neutral red working solution was separated and 10% formalin was added to fix the cells for 30 min at 37°C. After 30 min, formalin was removed and the dishes were washed twice with phosphate-buffered saline. The plates were left to dry overnight in the incubator and stored in a dark location.
Cytotoxicity was analysed by measuring the zone of inhibition (ZOI) surrounding the specimen, which was the unstained area including the area of the RBC specimen as a percentage of the area of the entire plate. The percentage of stained area was then calculated, which corresponds to cell viability. The RBC specimens were placed back onto the plates in their original positions and images of each plate were captured with a digital camera (Figure 4). The ZOI was then calculated using ImageJ software (National Institute of Health, Bethesda, Maryland, USA).
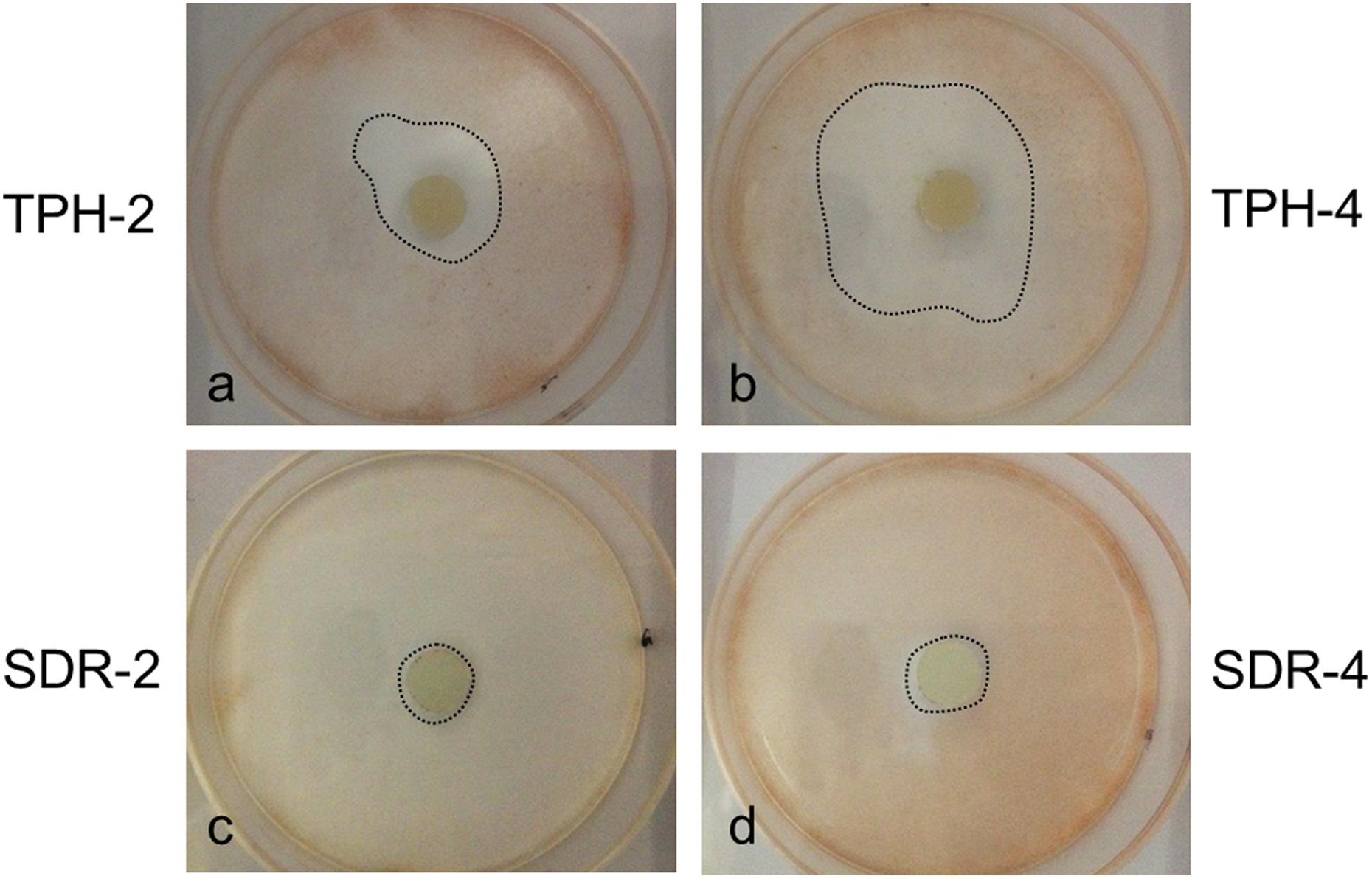
Representative images of direct contact test: (a) TPH-2, (b) TPH-4, (c) SDR-2 and (d) SDR-4. Zone of inhibition indicated by black dotted line. SDR: smart dentin replacement.
Extract test
The extracts were prepared in accordance with ISO 10993-5 (2009) and 10993-12 (2012) guidelines, using complete culture medium as the extraction medium. The recommended surface to volume ratio was fixed at 3 cm2/mL. The RBC specimens were totally immersed in the extraction medium and incubated at 37°C for 24 h under static condition (Figure 3(c)). L-929 mouse fibroblasts were seeded at a density of 2 × 104 cells per well in 96-well flat-bottomed plates. The cells were allowed to attach for 15–18 h, prior to exposure to the RBC extracts. The plating medium was removed and 100 µL of the extracts was added into each well for further incubation at 37°C for 24 h. Following incubation, cell morphological changes were observed by phase contrast microscope (DMI3000B; Leica, Wetzlar, Germany). Cell viability was assessed by MTS (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium) assay (CellTiter 96® AQueous One Solution Cell Proliferation Assay; G3582; Promega, Madison, Wisconsin, USA), following the manufacturer’s protocol. Briefly, 20 µL of MTS reagent was added to each well. The cells were incubated at 37°C for 2 h, and the absorbance readings were taken at 490 nM and 650 nM (reference) using Infinite 2000 plate reader (Tecan, Männedorf, Switzerland). Untreated cultures without any material served as negative control. Percentage cell viability was calculated by normalization of the absorbance readings against that of the negative control (set as 100%). Images of the cells were taken using inverted fluorescence phase contrast microscope (IX70; Olympus, Tokyo, Japan) and connected digital camera (DP70; Olympus).
Data analysis
The means and standard deviations were computed and data was statistically analysed with StatView (SAS Institute Inc., Cary, North Carolina, USA) using one-way analysis of variance and Scheffe’s post hoc test at a significance level of 0.05. Pearson’s correlation was also performed to assess the relationship between hardness ratio and cell viability as well as cell viability between the three test models at a significance level of 0.05.
Results
Mean KHN of the top/bottom surfaces and the mean hardness ratios are shown in Tables 2 and 3, respectively. Table 3 also reflects the percentage stained area and cell viability for the direct contact, indirect contact and extract test models. Results of statistical analysis are indicated in Table 4.
Mean KHN of RBCs.a

KHN: Knoop’s hardness number; RBC: resin-based composite; SDR: smart dentin replacement.
aSamples were prepared at 2 mm (TPH-2, SDR-2) and 4 mm (TPH-4 and SDR-4) depths (thickness).
Mean hardness ratio, percentage stained area and cell viability.

Comparison of materials/cavity depths for the various tests.a
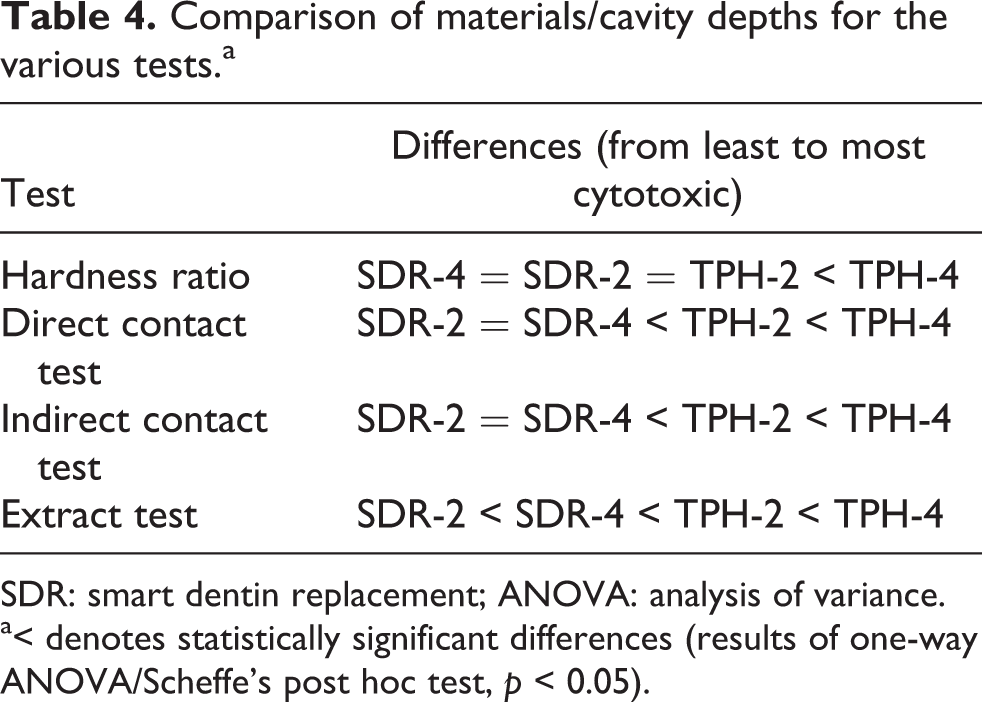
SDR: smart dentin replacement; ANOVA: analysis of variance.
a< denotes statistically significant differences (results of one-way ANOVA/Scheffe’s post hoc test, p < 0.05).
Micro-hardness testing
Hardness ratio derived from micro-hardness testing is a surrogate for effectiveness of RBC cure at 2- and 4-mm depths. Although TPH-2 and TPH-4 had similar KHN values at the top surfaces, KHN values at the bottom surfaces differed significantly. Hardness ratios for TPH-2 and TPH-4 were thus significantly different. The hardness ratio of TPH-4 was also significantly lower than SDR-2 and SDR-4. No significant difference in top/bottom KHN as well as hardness ratios was observed between SDR-2 and SDR-4. Hardness ratios of SDR-2/SDR-4 and TPH-2 were also not significantly different.
Direct contact test
Percentage stained area (which corresponds to cell viability) of TPH-4 was significantly lower (more cytotoxic) than TPH-2, SDR-2 and SDR-4. The percentage stained area of TPH-2 was, in turn, significantly lower than of SDR-2 and SDR-4 (Figure 5). No significant difference in percentage stained area was observed between SDR at 2- and 4-mm depths. Ranking of materials from least (left) to most (right) cytotoxic was as follows: SDR-2 = SDR-4 < TPH-2 < TPH-4 (Table 4).
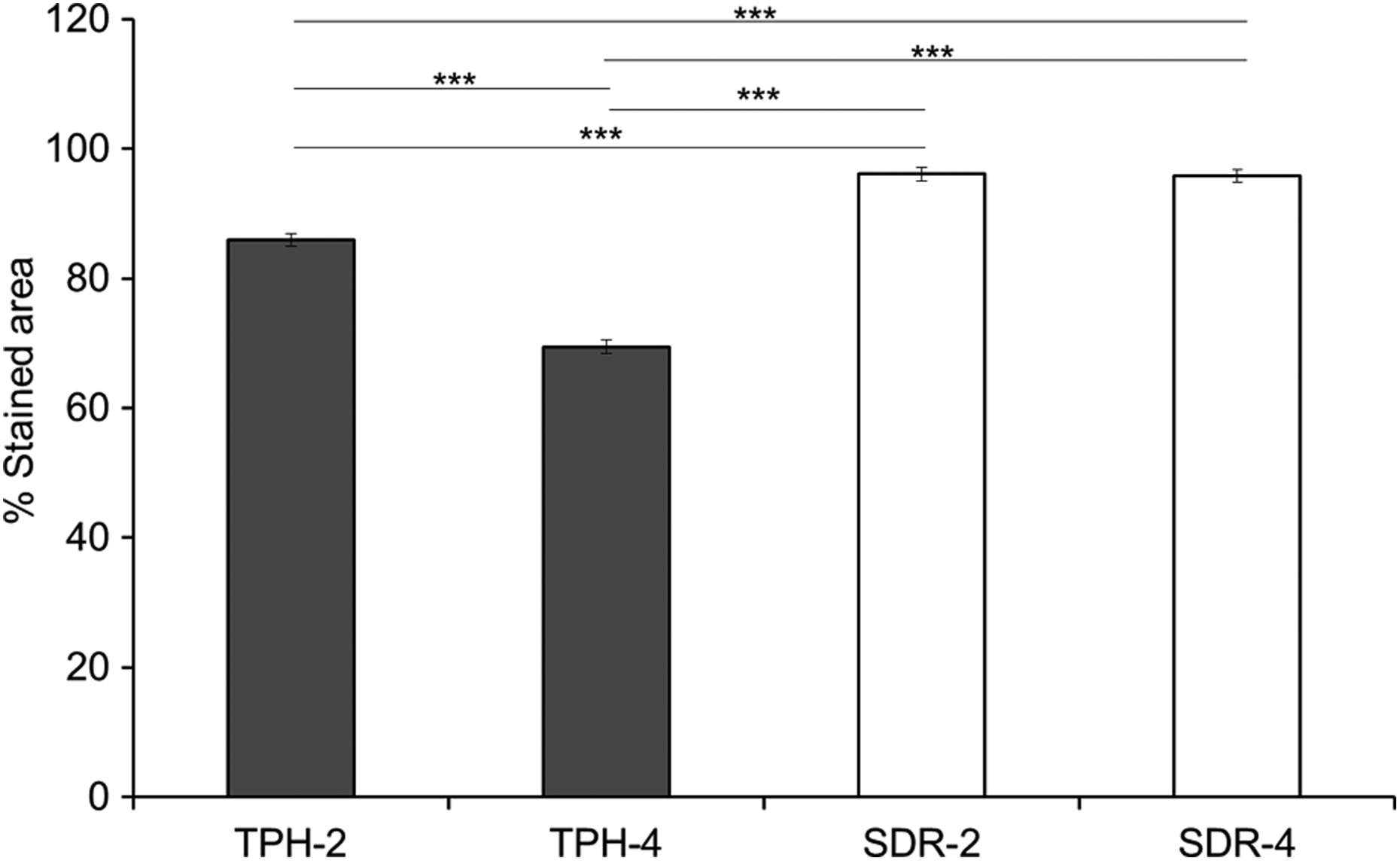
Percentage stained area of the various RBC-depth combinations for the direct contact test. Results of one-way ANOVA/Scheffe’s post hoc test. ***p < 0.001: statistically significant differences. RBC: resin-based composite; ANOVA: analysis of variance.
Indirect contact test
Findings of the indirect contact test were similar to the direct contact test. TPH-4 had significantly lower percentage stained area than all the other material-depth combinations (i.e. TPH-2, SDR-2 and SDR-4). TPH-2, likewise, was more cytotoxic than SDR-2 and SDR-4 (Figure 6). Ranking of materials from least (left) to most (right) cytotoxic was as follows: SDR-2 = SDR-4 < TPH-2 < TPH-4 (Table 4).

Percentage stained area of the various RBC-depth combinations for the indirect contact test. Results of one-way ANOVA/Scheffe’s post hoc test. ***p < 0.001: statistically significant differences. RBC: resin-based composite; ANOVA: analysis of variance.
Extract test
Significant difference in cell viability was observed between all material-depth combinations (Figure 6). TPH-4 had significantly lower cell viability than all TPH-2, SDR-2 and SDR-4 while TPH-2 was more cytotoxic than SDR-2 and SDR-4. Unlike the direct and indirect contact models, significant difference in cell viability was observed between SDR-2 and SDR-4 (Figure 7). SDR-2 was significantly less cytotoxic than SDR-4. Ranking of materials from least (left) to most (right) cytotoxic was as follows: SDR-2 < SDR-4 < TPH-2 < TPH-4 (Table 4). This corresponded to our morphological observation where cells treated with extracts of TPH showed decrease in cell number and cells appeared rounded and retracted. On the other hand, cells treated with extracts of SDR remained spindle shaped and spread out, similar as the untreated negative control (Figure 8).

Percentage cell viability of the various RBC-depth combinations for the extract test. Results of one-way ANOVA/Scheffe’s post hoc test. **p < 0.01 and ***p < 0.001: statistically significant differences. RBC: resin-based composite; ANOVA: analysis of variance.

Representative images of L-929 fibroblasts after 24 h exposure to the various extracts at 37°C: (a) TPH-2, (b) TPH-4, (c) SDR-2, (d) SDR-4 and (e) negative control. Note the reduced cell number and retracted cellular morphology of L-929 when treated with extracts of TPH. SDR: smart dentin replacement.
Correlation analysis
Results of Pearson’s correlation are showed in Tables 5 and 6. Correlations between hardness ratio and cell viability ranged from r = 0.89–0.96 for the various tests. All correlations were very strong and highly significant. When data from all three tests were pooled, the correlation coefficient between hardness ratio and cell viability was weaker (r = 0.60) but still highly significantly. Very strong and significant correlation (r = 0.97 and above) was also observed between the three test models. Correlation between direct and indirect contact test was 0.98 while that between the extract test and the two contact tests were both 0.97.
Correlation between hardness ratio and cell viability (p value).

aStatistically significant correlation.
Correlation of the cell viability between test models (p value).

aStatistically significant correlation.
Discussion
Choice of cell line
L-929 cells were selected, as they are easily available and have efficient in vitro growth due their homogenous morphology and growth characteristics. Use of such established cell lines, however, only serves as a screening assessment of the cytotoxicity of dental materials. More clinically relevant cell lines such as human dental pulp stem cells and human embryonic stem cells could be considered in future work to reaffirm the cytotoxic properties of test materials as animal cell lines lack human relevance and also present issues of metabolic and genetic abnormalities.
Micro-hardness testing
To reduce inter-operator errors, micro-hardness testing was conducted by only one trained operator. Based on a hardness ratio of 0.8 indicating effective cure, TPH-2 exhibited adequate cure but not TPH-4. Therefore, the manufacturer’s recommended maximum depth of cure of 3 mm should be adhered to for TPH. SDR was, however, successfully cured at both 2- and 4-mm depths with hardness ratios of 0.82 and 0.88, respectively. SDR is the first bulk-fill RBC material introduced to the dental profession. It was designed to serve as a base material to replace dentin and is capped with any universal RBC. In addition to 4-mm thick incremental placements, manufacturer had also claimed a 60% reduction in shrinkage stress when compared to other RBCs. The latter is achieved with a proprietary ‘polymerization modulator’. SDR’s increased depth of cure can be attributed in part to the greater translucency and lower filler content of SDR (57% (TPH) versus 45% (SDR) by volume). Better curing light transmittance through the material is achieved resulting in better cure. 17 In our study, hardness ratio of SDR at 2 mm was found to be marginally lower than that at 4 mm. This contrasted with other studies reporting higher hardness ratios for SDR at 2 mm when compared to 4 mm. 18,19 The variance can be attributed to the lower KHN values obtained at the top surface of SDR-2 observed. Difference in hardness ratio between SDR-2 and SDR-4 was, however, statistically insignificant.
Comparison between materials
For all test models, the utmost cytotoxicity was observed with TPH-4. This corresponded to the relatively poor cure of TPH at 4 mm and anticipated quantity of unreacted monomers. Despite similar effectiveness of cure, cytotoxicity of TPH at 2 mm was significantly higher than that of SDR at both 2 and 4 mm. The forementioned can be attributed to differences in RBC composition. While both RBCs contain Bis-EMA and TEDGMA, SDR contains modified UDMA in place of Bis-GMA. Flowable RBCs with their higher BisGMA content has been associated with greater cytotoxicity when compared to their standard counterparts. 20 The better biocompatibility of SDR can also be attributed to its effective cure (even at 4 mm) and reduced quantity of unreacted monomers. In addition to SDR’s greater translucency and lower filler content, the polymerization modulator embedded within the SDR matrix is also purported to interact with the camphorquinone photoinitiator to increase the effectiveness of light cure and penetration. 21 As significant differences in effectiveness of cure and biocompatibility was observed between TPH and SDR especially at 4-mm depth, the secondary null hypothesis was rejected. Our findings supported manufacturer’s claim of bulk placement and curing without undue cytotoxicity for SDR.
Comparison between test models
The rank order for cytotoxicity was consistent between the three test models. In general, SDR displayed better biocompatibility than TPH. For all three test models, TPH-4 was significantly more cytotoxic than TPH-2 which in turn was significantly more cytotoxic than SDR-2 and SDR-4. Although no difference in biocompatibility was observed between SDR-2 and SDR-4 for the direct and indirect contact tests, a significant difference in cell viability was observed for the extract test. For the extract test, SDR-2 was found to be more biocompatible than SDR-4 which was anticipated in view of the better curing light penetration and monomer conversion. The number of studies comparing the different ISO test models for the assessment of cytotoxicity of RBCs is scarce. Cao and co-workers reported that the extract test was the least sensitive and least correlated amongst the various cytotoxicity test models. 13 In a related study, it was concluded that the L-929 direct contact test was the most reliable when compared to tooth slice organ culture. The extract test was again reported to be the least sensitive amongst the three test models. 22 Our findings contradicted these earlier works as the extract test appeared to be more sensitive than the two contact tests. The variance can be attributed to dissimilarities in RBC materials evaluated and experimental design. A single RBC polymerized using two halogen-based and two LED curing lights served as the basis of comparison for the earlier work. More studies using a broad spectrum of RBCs of varying chemistry and cured with differing light sources are necessary before a definitive conclusion can be drawn on the sensitivity of the various test models and their clinical relevance.
Although the extract test followed the ISO recommended parameters, further optimization of test parameters including duration of extraction, surface area and volume of extraction media 23 are required to produce sufficient concentration of leached components to interact with oral tissues and simulate in vivo conditions. As a difference was observed between the two contact tests and the extract test model, the primary null hypothesis was rejected. Both the direct and indirect contact tests showed similar trends in terms of ranking and statistical analysis of cytotoxicity. The percentage stained area with the indirect contact test was consistently higher than that of the direct contact test. The presence of an agar barrier hence reduced leaching of cytotoxic substances that caused cell death. This mimicked a dentine barrier in vivo, reducing the contact of cytotoxic substances with pulpal cells. Whether the use of an agar barrier effectively simulates dentine has yet to be determined.
The strong and significant correlation between hardness ratio and cell viability regardless of cytotoxicity test model reinforced the importance of ensuring adequate RBC cure. Tests relating to RBC cure including micro-hardness testing and degree of conversion, however, cannot accurately reflect a dental material’s biocompatibility as the novel proprietary resins and modulators introduced to achieve bulk light curing may be cytotoxic. Cytotoxicity testing therefore remains an important screening assessment for all new RBCs. It is, however, not widely investigated due to the need for special technical skills, equipment and facilities. As the tests are also resource and manpower intensive, seldom are all three ISO test models carried out in research evaluating RBCs. Based on our study, the extract test may be recommended if only one test model is feasible, due to its higher sensitivity. Further research based on more clinically relevant cell types as well as testing models and methods is warranted to validate the cytotoxicity of RBCs.
Conclusion
The results of direct contact, indirect contact and extract test models for cytotoxicity testing of RBCs were found to be consistent. The extract test, however, appeared to be more sensitive than the two contact tests. Strong and significant correlations were observed between effectiveness of cure (hardness ratio) and cytotoxicity. Manufacturer’s recommended depths of cure must be adhered to ensure adequate light curing and biocompatibility of RBCs. Findings also supported the manufacturer’s claim of bulk placement and curing at 4 mm without undue cytotoxicity for SDR.
Footnotes
Acknowledgement
The authors would like to thank Dentsply Singapore for their material support.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by start-up grant (R221000067133), Faculty of Dentistry, National University of Singapore, Singapore.
