Abstract
Petroleum (commonly called crude oil) is a complex substance primarily composed of hydrocarbon constituents. Based on the results of previous toxicological studies as well as occupational experience, the principal acute toxicological hazards are those associated with exposure by inhalation to volatile hydrocarbon constituents and hydrogen sulfide, and chronic hazards are associated with inhalation exposure to benzene and dermal exposure to polycyclic aromatic compounds. The current assessment was an attempt to characterize the potential for repeated dose and/or developmental effects of crude oils following dermal exposures and to generalize the conclusions across a broad range of crude oils from different sources. Statistical models were used to predict the potential for repeated dose and developmental toxicity from compositional information. The model predictions indicated that the empirical data from previously tested crude oils approximated a “worst case” situation, and that the data from previously tested crude oils could be used as a reasonable basis for characterizing the repeated dose and developmental toxicological hazards of crude oils in general.
Introduction
The United States Environmental Protection Agency (US EPA) announced a voluntary chemical data collection effort in 1998 called the High Production Volume (HPV) Challenge Program. 1 The HPV chemicals are those produced or imported into the United States in aggregate quantities of at least 1 million pounds per year. Approximately 400 petroleum substances were sponsored in the EPA’s Challenge Program by companies belonging to the Petroleum HPV Testing Group (PHPVTG). The various substances were organized into 13 categories to facilitate data sharing and to avoid redundant testing. These categories include crude oil (petroleum), gases, gasoline, kerosene/jet fuel, gas oils, heavy fuel oils, lubricating oils, waxes, aromatic extracts, asphalts, grease thickeners, petroleum coke, and wastes. This article reports an investigation into the toxicological hazards of petroleum (commonly called crude oil), a complex substance of variable composition identified by CAS number 8002-05-9 and defined as a complex combination of hydrocarbons. It consists predominantly of aliphatic, alicyclic, and aromatic hydrocarbons. It may also contain small amounts of nitrogen, oxygen, and sulfur compounds. This category encompasses light, medium, and heavy petroleums as well as the oils extracted from tar sands. Hydrocarbonaceous materials requiring major chemical changes for their recovery or conversion to petroleum refinery feedstocks such as crude shale oils, upgraded shale oils, and liquid coal fuels are not included in this definition.
It is difficult to precisely define the toxicological hazards of any specific crude oil, because oils from different sources are compositionally distinct and because the toxicological hazards are ultimately the consequence of the constituents that they contain. In acute situations, the hazards of crude oils are largely related to central nervous system effects resulting from overexposure to hydrocarbon vapors or other volatile constituents such as hydrogen sulfide or other sulfur-containing molecules such as mercaptans. 2,3 The hazards associated with prolonged and repeated dermal exposures to crude oils are mostly due to those associated with polycyclic aromatic compounds (PACs) including both polycyclic aromatic hydrocarbons and other aromatic molecules in which 1 or more of the carbons is replaced by a sulfur, oxygen, or nitrogen atom. In short, crude oil contains a number of hazardous constituents but as the levels of these hazardous constituents vary between crude oils, the overall hazard characteristics of crude oils also vary in accordance with their individual compositions.
Historically, the principal concern about repeated exposures to crude oils during production or as a consequence of accidental discharge has been its potential to cause dermal cancer. Although the human evidence per se was judged as “inadequate” by International Agency for Research on Cancer, 4 crude oils do contain PAC, and have been shown to produce squamous cell carcinomas when tested in dermal administration assays in C3H mice. 5–7 However, there have also been investigations into the potential for mutagenic potential, 8,9 as well as systemic effects 10,11 and developmental toxicity 12,13 in mammalian species. It has been reported that for “high-boiling” petroleum-derived substances (ie, those with final boiling points ≥650°F [343°C]), the potential for systemic and developmental effects is associated with the levels of 3- to 7-ring PACs. 14 This association was further elaborated as part of the work done by the PHPVTG, 15 which resulted in a series of empirical models with which the potential for systemic and developmental effects could be predicted from compositional information. 15–17
To develop these models, compositional data, that is, polycyclic aromatic profiles, were compared to 4 indicators of target organ effects (increased liver-to-body weight ratios, reduced thymus weight, reduced platelet count, and reduced red blood cell count) and 3 indicators of developmental effects (live fetus count, fetal weight, and percent resorptions). The effects data were taken from approximately 19 studies of high-boiling petroleum substances including gas oils, heavy fuel oils, lubricant base oils, aromatic extracts, and crude oils. For the repeated dose studies, each dose group response was the mean response of all the animals in the dose group in the respective study. For the developmental toxicity studies, the dose group response was the unweighted mean of the means of all the litters in the dose group in the respective study.
A statistical model of the dose–response curve was developed independently for each of the 7 toxicological end points. The models were based on least-squares linear regression methods with the toxicological end point (eg, fetal body weight) as the dependent or predicted variable, with the relevant toxicological study design variables (eg, control group response, litter size, and sex) and the test material weight percentage of each of the aromatic ring fractions as the independent (or predicting) variables.
The models were developed to be as simple as possible but still adequately describing the data. The final model was selected based on the overall model multiple correlation coefficient (r) and the error mean square (EMS). These measures were selected because, among their other characteristics, the r value is a measure of the closeness of the observed and model predicted values, while the EMS is related to the width of the confidence interval of the predicted value. The final models showed a good correlation between the observed and predicted data, specifically, a mean r of 0.94 and a minimum r of 0.91.
The PHPVTG collected and summarized the available published information on crude oils along with unpublished information from industry sources. 18 The PHPVTG also selected 46 samples of different crude oils from those in commerce in 2004. 19 The necessary compositional information for these samples was obtained, and predictions of toxicity were made and compared to the previously existing data. The lowest levels associated with these predicted results were similar to the results of a previously tested crude oil, 11,13 indicating that the potential hazards of crude oils were generally consistent with those previously identified and that the potential for target organ effects and developmental toxicity of any crude oil could be characterized using information on the types and levels of PACs that it contains.
Materials and Methods
Samples
From a group of 170 crude oils, which were being processed in US refineries in 2004, 46 samples of uniquely identified crude oils were randomly chosen. 19 One way in which crude oils are differentiated within the petroleum industry is by the use of specific gravity to separate them into “light” (ie, crudes with specific gravities ≤0.860) and “heavy” (ie, crudes with specific gravities ≥0.887). Of the 46 crude oils assessed, 12 were heavy, 19 were intermediate, and 15 were light. (The underlying data are given in Table 1.) These 46 samples were pentane deasphalted (modified ASTM D6560) to remove very high-molecular-weight, nonaromatic constituents to allow the remaining materials to be evaluated for PAC content as described subsequently. Chromatographic standards were obtained from Sigma-Aldrich (St. Louis, Missouri).
Relative Density Information for 46 Crude Oils.
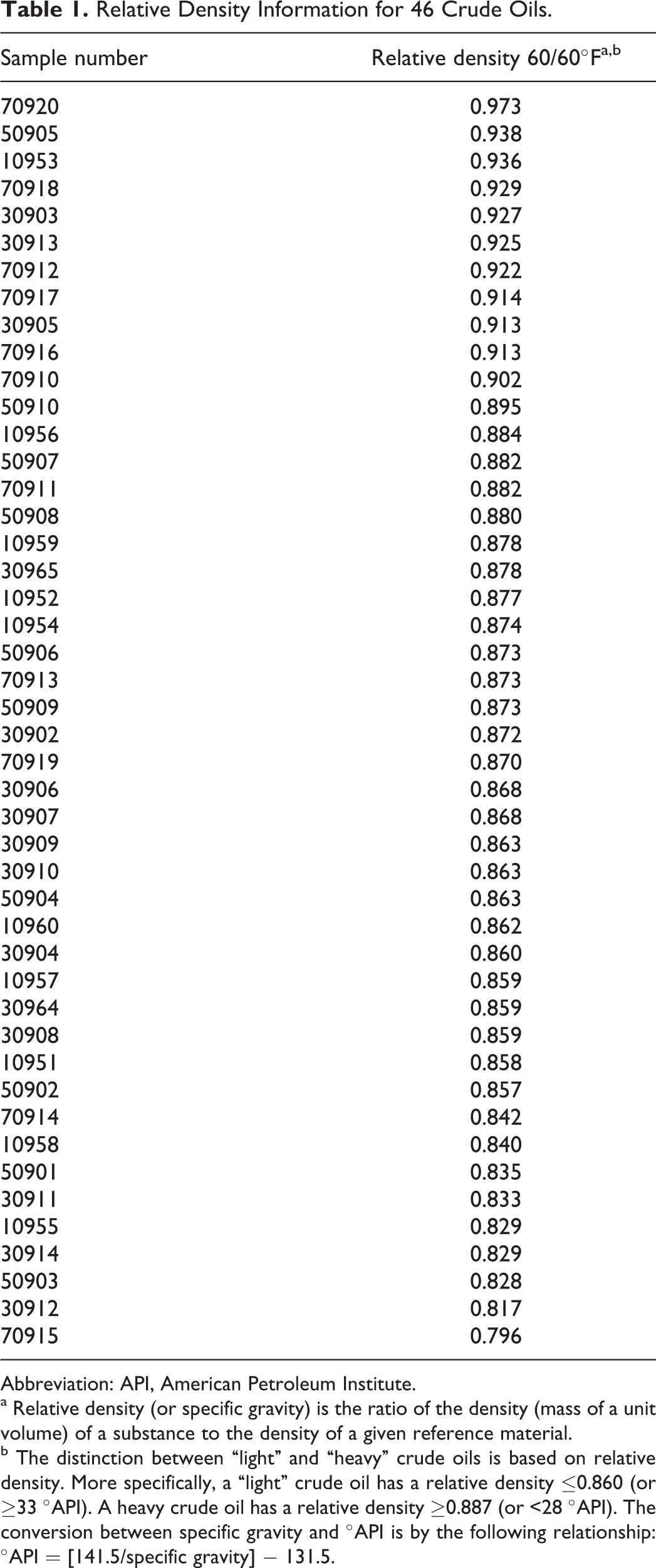
Abbreviation: API, American Petroleum Institute.
a Relative density (or specific gravity) is the ratio of the density (mass of a unit volume) of a substance to the density of a given reference material.
b The distinction between “light” and “heavy” crude oils is based on relative density. More specifically, a “light” crude oil has a relative density ≤0.860 (or ≥33 °API). A heavy crude oil has a relative density ≥0.887 (or <28 °API). The conversion between specific gravity and °API is by the following relationship: °API = [141.5/specific gravity] − 131.5.
The PAC Analysis
The 46 crude oil samples were evaluated for PAC content based on the method described by Roy et al 20,21 (for a more complete description, see Gray et al 22 ). Briefly, a 4-g sample of each deasphalted crude oil was weighed in a 60-mL separatory funnel and dissolved in 10 mL of cyclohexane. The sample was then extracted with 2 × 10 mL dimethyl sulfoxide (DMSO). The DMSO extracts were combined in a 125-mL separatory funnel, diluted 2:1 with 40 mL of 4% aqueous sodium chloride, and extracted with 20 and 10 mL of cyclohexane. The cyclohexane fractions were combined in a 60-mL separatory funnel, washed with 2 × 5 mL of distilled water, and filtered through anhydrous sodium sulfate into a 50-mL flask. The cyclohexane was evaporated to near dryness at 40°C followed by further evaporation at 80°C (15-20 Torr) for 0.5 hour. Once equilibrated to room temperature, the flask was weighed and the residue dissolved in cyclohexane (∼50 mg/mL) and stored at 4°C pending analysis.
The extracts were analyzed by gas chromatography with mass selective detection (GC/MSD). The gas chromatograph was equipped with a 30m × 0.25 mm × 0.25 μm Zebron-5HT capillary column (Phenomenex Inc, Torrance, California). The oven temperature was held at 70°C for 0.5 minutes and then programmed to 300°C at a rate of 5°C/min and held at the final temperature for 35 minutes. A split mode of injection (50:1) was used. Other general conditions of analysis were as follows: carrier gas—helium; 30 cm/sec; detector and injection port temperatures—300°C. The GC/MSD chromatograms of the extracts were integrated in the slice mode. Standards of naphthalene (Aldrich; 99+% purity), phenanthrene (Aldrich; 98% purity), 1,2-benzanthracene (Aldrich; 99% purity), benzo[a]pyrene (Sigma; ∼ 98% purity), benzo[ghi]perylene (Aldrich; 98% purity), and coronene (Aldrich; sublimed, 98% purity) standards were used to define the boundaries of retention times for PACs containing 2 through 7 rings. Data were reported by ring number as the weight percent of the starting material, and the results are referred to as the aromatic ring content (ARC) profile.
For comparative purposes, the literature information was taken from studies of systemic and developmental toxicity in which crude oils were administered to rats by dermal application. 11,13 The test materials (identified in the previous publications as crude I and crude II) had differing compositions in which the principal differentiating characteristics were the polycyclic aromatic constituents. More specifically, crude I contained approximately 50% saturated (aliphatic) and 50% aromatic constituents of which 35% was composed of 1- and 2-ring aromatics with the remaining 15% being aromatics of 3 or more rings. Crude II contained approximately 37% saturated constituents and 63% aromatics of which 42% was <3 rings and 21% was aromatics containing 3 or more rings.
Predictions of Systemic and Developmental Toxicity
The compositional information was used to predict the dose response for systemic and developmental effects for each of the 46 crude oils. Specifically, the predicted systemic end points are increases in liver-to-body weight ratios and decreases in thymus weights, platelet counts, and hematocrit. The predicted developmental end points are reduction in fetal body weights and offspring live-born and increased resorptions. The predictions were based on a series of statistically developed empirical models. 15–17 Each sample was characterized by the dose associated with a 10% change in the control group response, identified as the “predicted dose response at 10% (PDR10).” The PDR10 is similar in concept to the benchmark dose (BMD10). 23 However, there are several important differences; (1) unlike the BMD10 that is derived by fitting a model to data from a single study, the PDR is calculated by fitting a model derived from a series of toxicity studies covering a range of materials; (2) the PDR10 is based on 1 model, whereas there are several competing models for the BMD calculation making the BMD dependent on the specific model selected 24 ; and (3) the PDR10 is based on a large amount of data taken from many studies, whereas each BMD calculation is specific for a single study and is usually based on 3 to 5 data points.
Biological response data for crude oils were taken from the literature sources. 11,13 The BMD10 values were estimated for the 4 systemic and 3 developmental end points listed previously. The values were calculated using the linear model from the EPA BMD application. 25
The compositional information was then used to predict the potential for systemic and developmental effects for each of the crude oil samples using empirical models.
Results
As indicated in the Materials and Methods section, 46 samples of crude oils from various sources were obtained and evaluated for PAC content. As shown in Table 2, the total condensed ring aromatic contents of these oils (as measured by the DMSO extraction procedure 26 ) ranged from approximately 1.9% to 5.8%. For most of the oils, the 2-ring aromatic molecules were the most abundant aromatic species present, but many of the oils had appreciable concentrations of molecules with 3 or more aromatic rings.
Aromatic Ring Content (ARC) Profiles for 46 Crude Oils.a,b
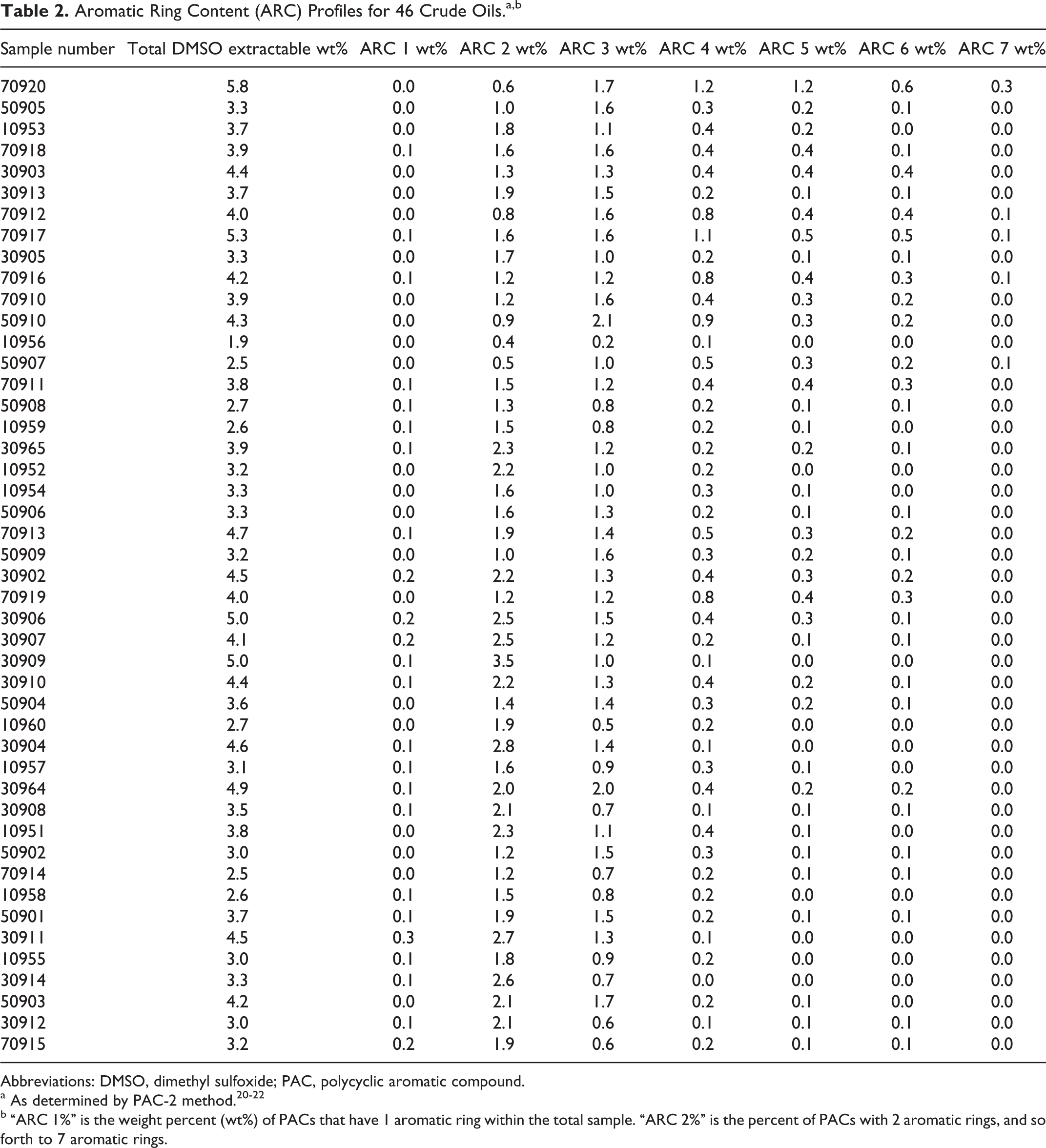
Abbreviations: DMSO, dimethyl sulfoxide; PAC, polycyclic aromatic compound.
b “ARC 1%” is the weight percent (wt%) of PACs that have 1 aromatic ring within the total sample. “ARC 2%” is the percent of PACs with 2 aromatic rings, and so forth to 7 aromatic rings.
Based on the compositional data, the PDR10 values (doses of dermally applied crude oil samples that were associated with 10% changes in systemic and developmental effects) for all 46 crude oil samples were estimated and plotted (Figure 1A and B). (All of the predicted values are listed in Tables 3 and 4.) Literature data from the 2 studies in which the systemic and developmental toxicity of crude oils administered dermally was assessed were also included for comparative purposes. 11,13 (The literature data are represented in the figures by a star [*] and a diamond [♦].) The systemic effects reported for these samples included increased relative liver weights and reductions in thymus weights, platelet counts, and red blood cell counts. The BMD10s for target organ effects of crude I were increased relative liver weights = 249 mg/kg/d (M) and 107 mg/kg/d (F); reduced thymus weights = 468 mg/kg/d (M) and 211 mg/kg/d (F); reduced platelet counts = 581 mg/kg/d (M) and 1900 mg/kg/d (F); and reduced red blood cell counts = 895 mg/kg/d (M) and 1900 mg/kg/d (F). The BMD10s for target organ effects of crude II were increased relative liver weights = 818 mg/kg/d (M) and 783 mg/kg/d (F); reduced thymus weights = 502 mg/kg/d (M) and 656 mg/kg/d (F), reduced platelet counts = 1039 mg/kg/d (M) and 2000 mg/kg/d (F); and reduced red blood cell counts = 1838 mg/kg/d (M) and 2000 mg/kg/d (F).
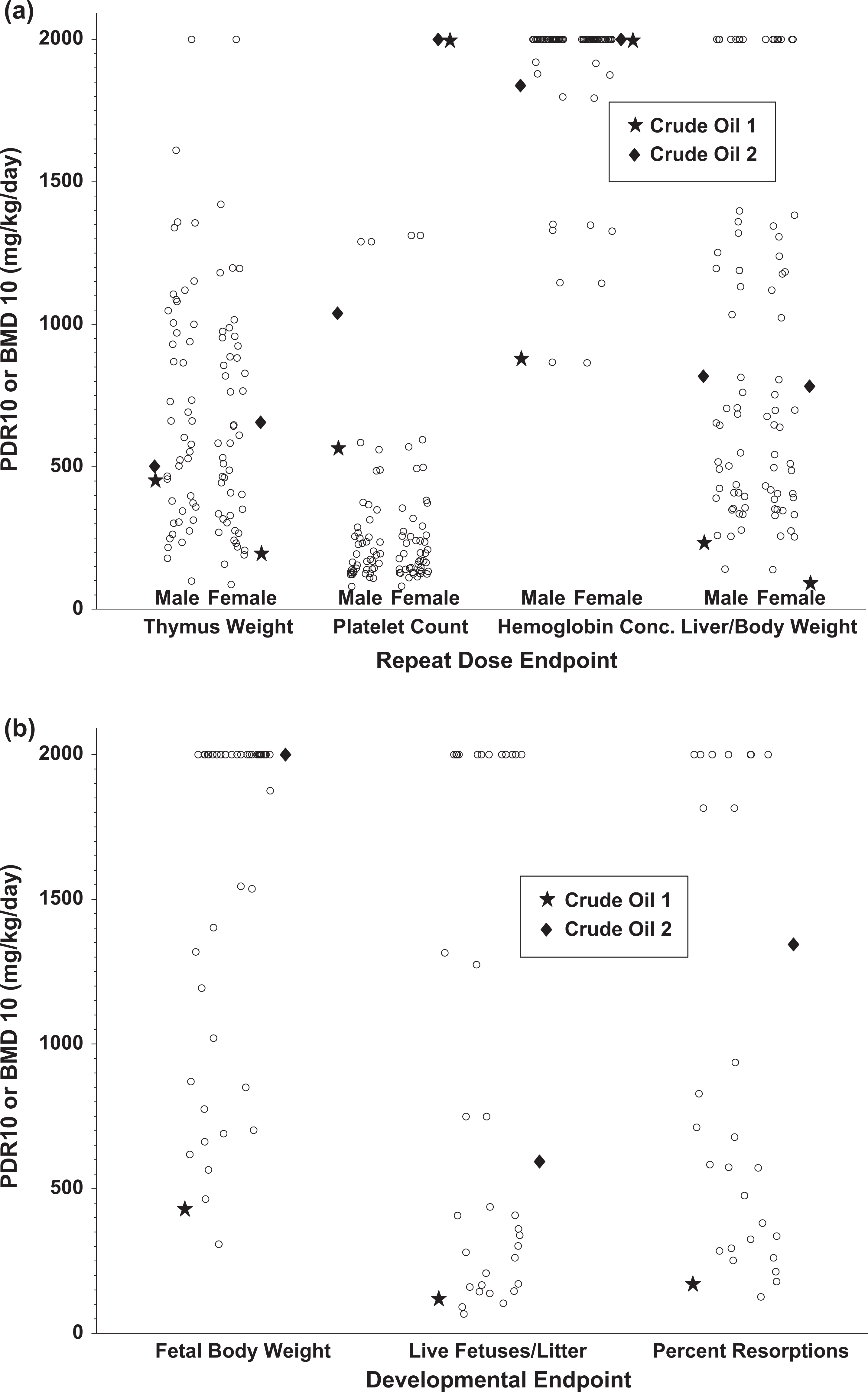
(a) Summary information on predicted dose for 10% change (PDR10) predictions for indicators of systemic effects (reduced thymus weights, reduced platelet counts, reduced hemoglobin concentrations, and increased liver-to-body weight ratios) for 46 crude oils as compared to the bench mark dose for a 10% change (BMD10) values for those same end points for 2 crude oils (crude oil 1* and crude oil 2♦). (b) Summary information on predicted dose for a 10% change (PDR10) predictions for indicators of developmental effects (reduced fetal body weight, reduced fetal survival, increased resorptions) for 46 crude oils as compared to the bench mark dose for a 10% change (BMD10) values for those same end points for 2 crude oils (crude oil 1* and crude oil 2 ♦).
Doses (mg/kg/d) of Crude Oil Predicted to Cause 10% Reductions in Thymus Weights, Platelet Count, and Hemoglobin Content and 10% Increases in Relative Liver Weights.a,b
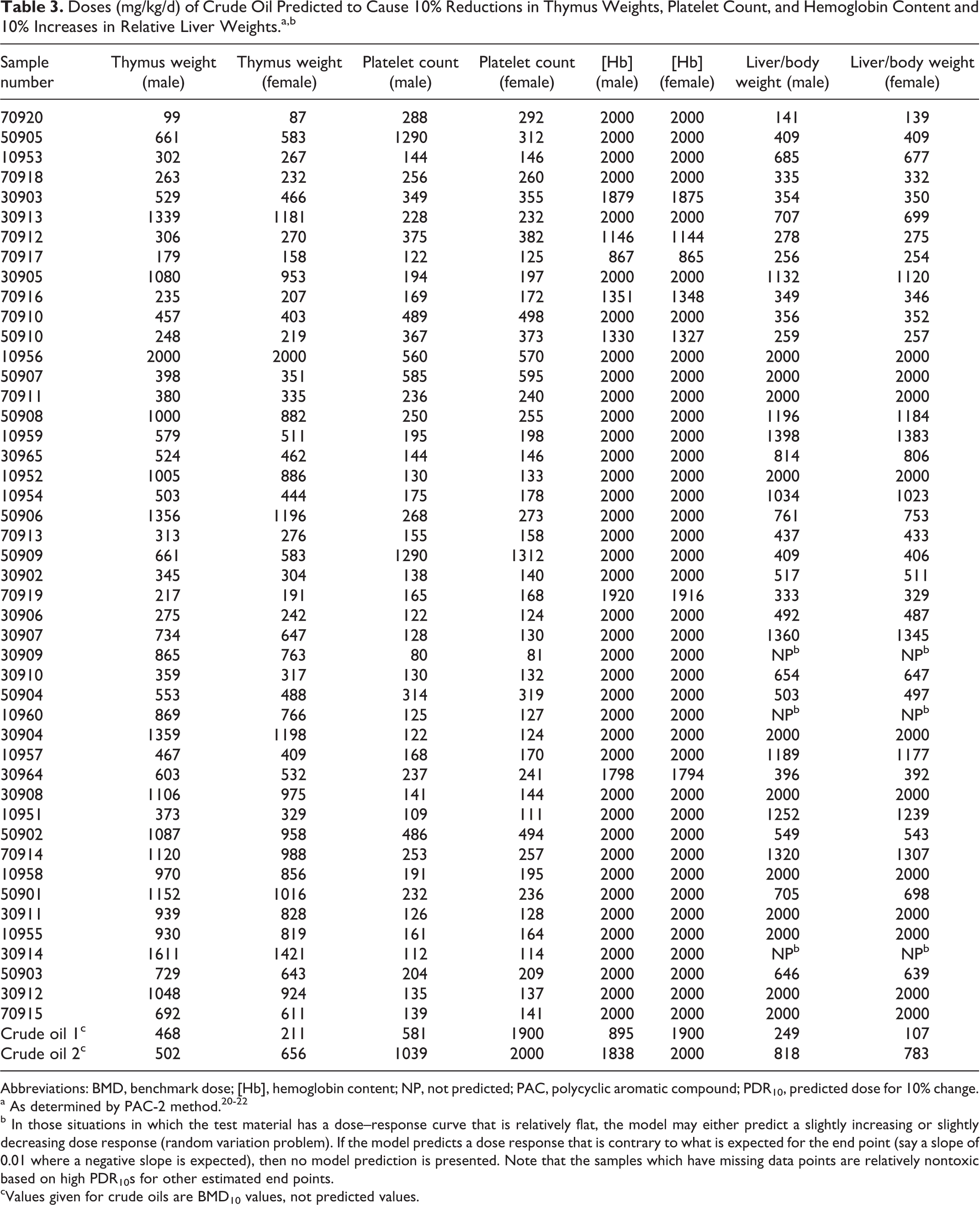
Abbreviations: BMD, benchmark dose; [Hb], hemoglobin content; NP, not predicted; PAC, polycyclic aromatic compound; PDR10, predicted dose for 10% change.
b In those situations in which the test material has a dose–response curve that is relatively flat, the model may either predict a slightly increasing or slightly decreasing dose response (random variation problem). If the model predicts a dose response that is contrary to what is expected for the end point (say a slope of 0.01 where a negative slope is expected), then no model prediction is presented. Note that the samples which have missing data points are relatively nontoxic based on high PDR10s for other estimated end points.
cValues given for crude oils are BMD10 values, not predicted values.
Doses (mg/kg/d) of Crude Oil Predicted to Cause 10% Reductions in Fetal Body Weights and Live Fetuses/Litter and a 10% increase in Percent Resorptions.a
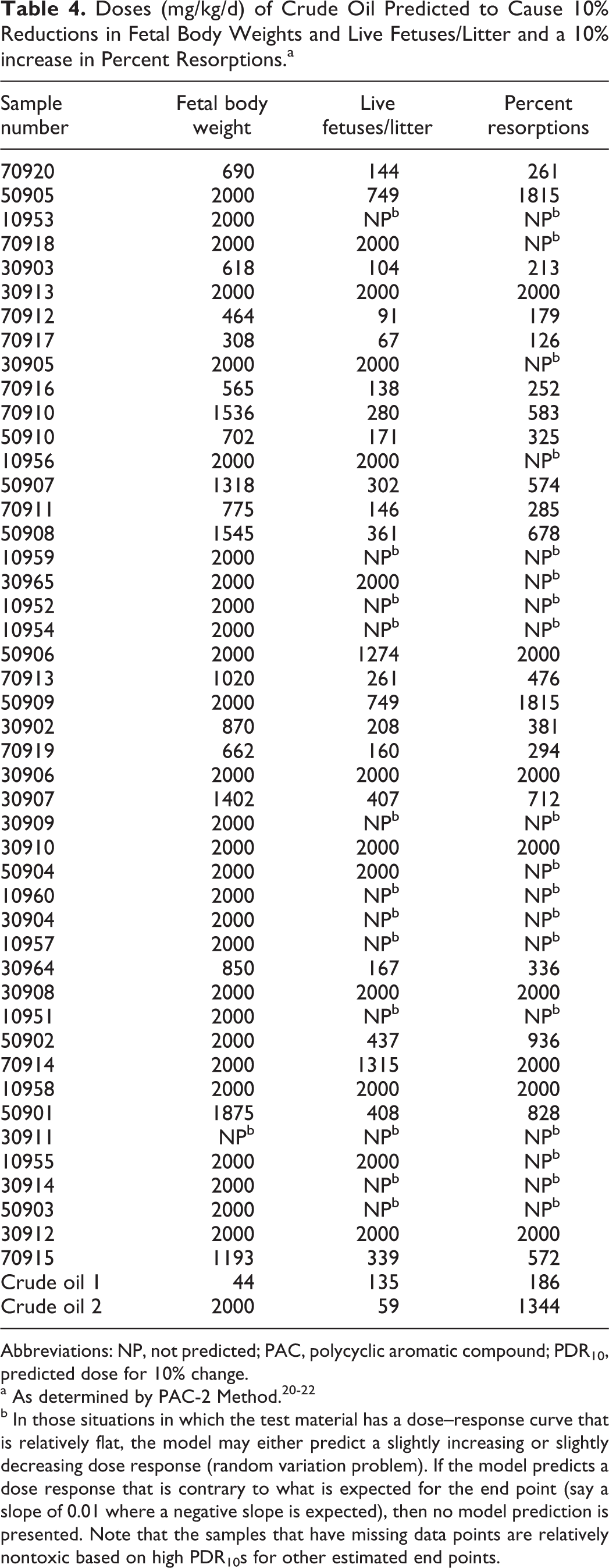
Abbreviations: NP, not predicted; PAC, polycyclic aromatic compound; PDR10, predicted dose for 10% change.
b In those situations in which the test material has a dose–response curve that is relatively flat, the model may either predict a slightly increasing or slightly decreasing dose response (random variation problem). If the model predicts a dose response that is contrary to what is expected for the end point (say a slope of 0.01 where a negative slope is expected), then no model prediction is presented. Note that the samples that have missing data points are relatively nontoxic based on high PDR10s for other estimated end points.
The BMD10 for developmental effects of crude I were 445 mg/kg/d for reduced fetal body weight, 135 mg/kg/d for reductions in numbers of live fetuses per litter, and 186 mg/kg/d for increases in percentage resorptions. For crude II, the corresponding BMD10 were 2000 mg/kg/d for reduced fetal body weight, 594 mg/kg/d for reductions in numbers of live fetuses per litter, and 1344 mg/kg/d for increases in percentage resorptions.
As shown in Table 5, the lowest PDR10 values for thymus weights, platelet counts, and liver-to-body weight ratios were in the 80 to 141 mg/kg/d range. The lowest PDR10 value for hematocrit reduction was approximately 865 mg/kg/d. Note that the lowest predicted doses for reduced thymus weight and increased liver-to-body weight ratios were associated with sample 70920, whereas the sample associated with the lowest dose predicted to reduce platelet count and hematocrit was 70917. As indicated in Table 1, these are the 2 samples with the highest content of DMSO extractable material of the 46 crude oils tested. However, as shown by Nicolich et al, 15 the effects of the PAC-containing oils are related not only to the total amount of DMSO extractable material but also to the ring distributions.
Minimum Predicted Dose for 10% Change (PDR10) Values for Indicators of Systemic Effects (mg/kg/d) Based on the Predicted Results From 46 Crude Oil Samples.

The estimated PDR10 values for developmental parameters are shown in Table 6. The sample associated with the lowest dose reducing maternal thymus weight (36 mg/kg/d) was 70920 (the same sample predicted to reduce thymus weights in male and nonpregnant female rats as indicated in the previous paragraph). The sample associated with the lowest doses causing reductions in fetal body weight (308 mg/kg/d), offspring survival (64 mg/kg/d), and increased resorptions (126 mg/kg/d) was 70917.
Minimum Predicted Dose for 10% Change (PDR10) Values for Indicators of Developmental Toxicity and Maternal Effects (mg/kg/d) Based on the Predicted Results From 46 Crude Oil Samples.

Discussion
Crude petroleum oils are complex substances of variable composition. The toxicological hazards of these oils are related to the levels of certain specific constituents that they contain. In those situations in which the exposures are to relatively high levels over short periods of time, the most hazardous constituents are volatile components including hydrogen sulfide, other volatile sulfur-containing compounds, and volatile hydrocarbon constituents. These volatile components can cause acute central nervous system effects that may have serious consequences, particularly under conditions of overexposure. 2,3 However, there are other crude oil constituents including benzene and PACs, which are carcinogenic and may present chronic hazards under conditions involving repeated exposures at lower levels. Historically, these hazards have been managed through efforts to identify and then limit exposure to particularly toxic constituents.
Another historical practice of the petroleum industry has been to test materials that are “representative” of certain types of substances in order to use the results to characterize the potential hazards of similar substances, a pragmatic approach necessitated by the complex nature of many petroleum-derived substances. Another early decision was to focus on dermal cancer as the toxicological end point of greatest concern. The decision to focus on dermal cancer was also a pragmatic choice but does not address whether crude oils might produce other toxic effects, and, if so whether control measures that minimize the potential cancer risks are also sufficient to mitigate these other hazards. The HPV Challenge afforded the opportunity to address the “noncancer” toxicological end points.
The specific overall objective of the PHPVTG was to characterize the potential for crude oil to cause repeated dose and developmental effects. As indicated previously, the potential for crude oil to cause nonacute effects in animals is associated with the levels of PACs that the oils contain. Because of the large number of crude oils, testing all possible substances is not practical. Rather, a series of empirical statistical models 15 was developed by which it was possible to predict the likelihood of systemic 17 and developmental 16 toxicity based on ARC profiles.
Two crude oils that have been tested for repeated dose and developmental toxicity by dermal administration were available for comparison. 11,13 Systemic effects included reduced thymus weight, platelet count, and hematocrit and increased liver-to-body weight ratios. Based on BMD10 analysis, the most sensitive parameters were increases in relative liver weights and decreases in thymus weights for which the BMD10 values ranged from 62 to 236 mg/kg/d. In the developmental toxicity studies, there was evidence of reduced fetal weight and increased frequency of resorption with the lowest BMD10 value of 122 mg/kg/d. When the BMD10 values from the experimental data from these 2 crude oils are compared to the predicted outcomes, it is apparent that there is reasonable agreement between the outcomes of studies of the more toxic of the crude oils and the lowest predicted levels for all of the outcome parameters except reduction in platelet counts for all the 46 crude oils that were examined in the predictive models (Figure 1A and B).
The differences between the predicted and the measured effects on platelet counts are difficult to rationalize, but since the predicted outcomes are below the measured outcomes, the predictive models appear to be conservative, that is, the models predict that dermal treatment with crude oils will result in significant reductions in platelet counts at lower doses than those found experimentally to cause such reductions. Accordingly, the models seem more likely to overestimate than to underestimate the potential for crude oils to reduce platelet counts. Aside from platelet counts, it is apparent that the predicted values are in reasonable agreement with the empirical evidence and that as the data obtained in the study of the more toxic of the crude oils are similar to the lowest estimated PDR10 values for the 46 crude oils for which estimates were made, the published data on crude oil provide a reasonable worst-case basis for purposes of characterizing the toxicological hazards of crude oils in general.
In a larger sense, data provided to the EPA under the HPV program indicate that crude oils are minimally acutely toxic by oral and dermal routes of administration, do not cause eye or skin irritation, and do not produce allergic contact dermatitis. 18 These data also indicate that crude oils contain PAC and produce skin cancer when tested in mice. Systemic effects associated with dermal application of crude oils include reduction in thymus weights and red blood cell parameters and liver enlargement. Developmental effects include reduced fetal survival and reduced fetal weights. The doses producing these effects have been estimated from compositional information, and the lowest doses are consistent with previous crude oil data.
To meet the expectations of the HPV program, the industry provided toxicological data that encompassed acute, repeated dose, mutagenic, and developmental toxicity end points as well as reproductive toxicity. Previous data along with the information reviewed earlier covers the majority of these end points; the one not specifically addressed is reproductive toxicity. For EPA’s HPV Challenge Program, reproductive toxicity can be addressed in several different ways, among them by a combination of developmental toxicity data and evidence from repeated-dose tests showing that reproductive organs are not affected. 27 Examination of the data from the repeated dose studies of crude oils 11,13 indicates that, in fact, there were no significant pathological changes in reproductive organ tissues, providing evidence that reproductive organs were not affected by crude oil exposure at levels that produced other evidence of systemic toxicity.
There are 2 reports of the effects of orally administered crude oil on reproductive parameters in rats. In the first of these, crude oil was administered as Tween 80 solutions to male rats at levels of 200, 400, and 800 mg/kg/d. The animals were treated daily for 7 days, and then sacrificed for gross and pathologic examination. It was reported that testicular weights were significantly reduced in the 800 mg/kg/d group and epididymal sperm counts were significantly reduced in the 400 and 800 mg/kg/d groups. Some pathological effects in the testes were also reported, but the doses given in the table did not correspond to those in the text so the interpretation is somewhat difficult. 28 In a second study, Sprague-Dawley rats were given crude oil by oral administration at doses of 0.1, 0.2, or 0.4 mL crude oil. The rats were treated every other day for 4 weeks. The doses used corresponded to approximately 566, 1132, or 2264 mg/kg. 29 The authors reported significant reductions in epididymal sperm count in all dose groups, but relative testicular weights were unaffected. A critical evaluation of these studies is difficult as the methods were not described in detail; some important outcome data including information on the health of the treated animals were not provided, and there were some inconsistencies between the tabulated information and the text. It may also be important that the epididymal sperm counts in the controls were reported as approximately 135 × 106/mL in the first study and approximately 21 × 106 in the second, although it is not clear whether these data are comparable. Nevertheless, if these data are taken at face value, they raise the possibility that oral administration of crude oil at levels greater than 200 mg/kg/d can result in reductions in epididymal sperm count.
It should be noted that there are data from reproductive toxicity studies of a heavy oil with a high aromatic content derived from a catalytic cracking process and described most commonly as “clarified slurry oil.” 30,31 This type of substance contains the highest levels of PACs of any of the petroleum-derived substances. In the studies of clarified slurry oil, there were no effects on fertility following dermal administration of “clarified slurry oil” at doses of 0.1, 1, 10, 50, or 250 mg/kg/d, the highest level tested. 30 These data suggest that the PAC constituents in high-boiling petroleum-derived substances are unlikely to affect fertility. In contrast, in developmental toxicity studies of clarified slurry oil dermal treatment at levels between 1 and 250 mg/kg/d resulted in both maternal and fetal effects of which the most profound were significant increases in resorption and significantly reduced maternal and fetal body weights. 31 Developmental delays were also observed. These studies provide evidence that developmental toxicity is much more sensitive to the types of PACs found in high-boiling petroleum substances than fertility and reproductive organ effects.
In consideration of the that mentioned earlier, further assessment of the reproductive effects of crude oil would not seem to be justified, in part because the data already available indicate that if there are effects on fertility and/or reproductive organs, these effects are unlikely to occur at exposure levels below those causing developmental effects. This being the case, it is not clear what practical value would be provided by further assessment of reproductive parameters, particularly as there are already management measures to control exposure to crude oil due to its potential carcinogenic effects.
To facilitate appropriate classification and labeling of petroleum substances, including crude oil, IPIECA (the global oil and gas industry association for environmental and social issues) has developed guidance endorsed by the UN Subcommittee of Experts on globally harmonized system (GHS). 32,33 The first version of the document, called Guidance on the application of GHS criteria to petroleum substances, identifies the constituents of concern for crude oil as hydrogen sulfide, benzene, and PACs. It guides the user through the GHS criteria to meet national and international standards for classification and labeling. From there, appropriate risk management actions can be designed to minimize human health risks from potential exposure to crude oil.
In conclusion, the lowest predicted effect levels for systemic and developmental effects of 46 crude oils are similar to those obtained in toxicological studies. From this it is evident that of the crude oil samples already tested, crude I is a reasonable worst case for purposes of assessing the toxicological hazards of crude oils for these end points. Other available information indicates that the acute toxic hazards are most likely associated with the potential for exposure to volatile constituents, particularly hydrogen sulfide. The chronic hazard of greatest concern is most likely the potential for carcinogenic effects. Management practices to limit exposures to crude oil in order to protect from these hazards would also seem to afford a sufficient measure of protection from other toxicological hazards assessed as part of the HPV program.
Footnotes
Acknowledgments
The authors would like to thank Chris Sexsmith for quality assurance support and Lynn Bennett for assistance in manuscript preparation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors of this article are employees of the American Petroleum Institute or its member companies or are consultants paid by the PHPVTG.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was sponsored and funded by the Petroleum HPVTG, an unincorporated group of manufacturers affiliated by contractual obligation to fund a voluntary data disclosure and toxicity testing program on certain petroleum-related chemical substances in response to EPA’s HPV Challenge Program. The American Petroleum Institute (API) manages the PHPVTG’s activities.
