Abstract
HFPO-Amidol (CAS # 75888-49-2) is a new hexafluoropropylene oxide (HFPO)-based intermediate developed as an alternative to longer chain perfluorinated compounds. The repeated-dose toxicity of this material was evaluated in an Organization for Economic Cooperation and Development 422-compliant, 28-day oral exposure study with a concurrent reproductive/developmental toxicity screening test. Wistar rats received doses of 0, 30, 300, or 1000 mg/kg/d by oral gavage. Statistically significant changes in body weight gain of 1000 mg/kg/d females during the postcoitum period were possibly related to treatment but were considered not adverse, given the slight nature of the changes. The lower food consumption of 300 mg/kg/d females during the postcoitum and lactation period was not considered treatment related given the absence of a time- and dose-related trend and because food intake was generally similar to control levels after allowance for body weights. Statistically significant changes in motor activity (total movements and total ambulations) were noted in 1000 mg/kg/d main male and female rats. The changes observed in female rats were considered not treatment related in the absence of a dose–response trend. The higher motor activity of high-dose males was primarily apparent within the first 10 minutes of the 60-minute measurement period and was suggestive of temporary hyperreactivity to a new environment/stimulus. This increased peak motor activity remained present although at an apparent lower magnitude when measured 13 days after withdrawal of treatment. Because the possible toxicological relevance of the temporarily increased motor activity observed in 1000 mg/kg/d males could not be excluded, these changes were considered possibly adverse in nature. No treatment-related or toxicologically relevant effects were noted on the other parental, reproductive, and developmental parameters investigated in this study. The parental systemic no observed adverse effect level (NOAEL) for this study is 300 mg/kg/d (based on increased motor activity in males), while the reproductive and developmental NOAEL is 1000 mg/kg/d.
Introduction
Polymers of hexafluoropropylene oxide (HFPO) are known for their outstanding thermal and chemical resistance and are used widely in commerce today as greases, oils, surfactants, and lubricants. 1 HFPO-Amidol (CAS # 75888-49-2; poly[oxy[trifluoro(trifluoromethyl)-1,2-ethanediyl]], α-(heptafluoromethylethyl)-ω-[1,2,2,2-tetrafluoro-1-[[(2-hydroxyethyl)amino]carbonyl]ethoxy]-; Figure 1) is a new HFPO-based intermediate with a functionalized amidol end group. The chemical formula of this material is CF3CF2CF2O(CF(CF3)CF2O)nCF(CF3)C(O)NHCH2CH2OH. As synthesized, HFPO-Amidol is a complex mixture where n ranges from 2 to 14, with an average of 6. This chemistry was developed as an alternative to longer chain perfluorinated compounds, which are being scrutinized for possible environmental and human health effects. Some proposed applications of HFPO-Amidol include use as a monomer for polymeric synthesis, a film additive, and a surfactant.
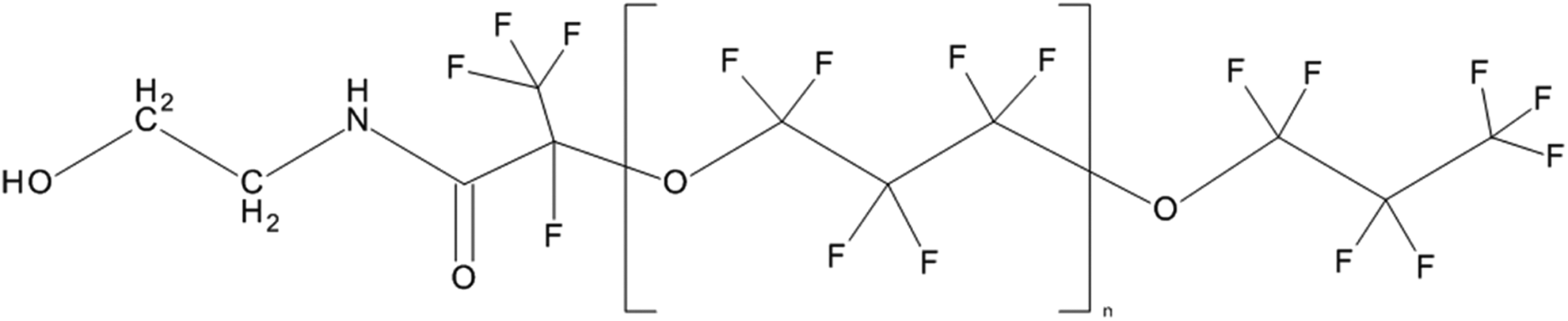
Structure of HFPO-Amidol (CASRN 75888-49-2). The repeated region (n) is repeated 6 times on average and can range from 2 to 14 repeats.
The purpose of this study was to evaluate the potential toxic effects of HFPO-Amidol when orally administered to Wistar Han rats for a minimum of 28 days followed by a 14-day recovery period and to evaluate the potential of HFPO-Amidol to affect reproductive end points such as gonadal function, mating behavior, conception, parturition, and early postnatal development. The oral route was selected as one possible route of exposure during use of the test substance. This work was conducted as part of a materials stewardship program and to generate GLP- and guideline-compliant test data to support its potential use as an industrial intermediate and surfactant.
Materials and Methods
Regulatory Compliance
The study procedures described in this publication were in compliance with Organization for Economic Cooperation and Development (OECD) Test Guideline 421, 2 OECD Test Guideline 422, 3 USEPA OPPTS 870.3650, 4 and 870.3550. 5 The study was conducted in compliance with OECD Good Laboratory Practice guidelines. 6
Test Article and Vehicle
The test substance, HFPO-Amidol (CASRN 75888-49-2; Lot No. N.B.# 156794-047-2/L-18535), was synthesized by 3M. Purity was 99.5% and the test sample was described as a colorless to slightly hazy liquid. The test material contained approximately 1% of the 5 repeating unit moiety (5-mer) and less than 0.02% of the 4 repeating moiety (4-mer). The number average molecular weight (Mw) for the test material was 1350, while the weight average Mw was 1404. The test substance was stored at room temperature in the dark and was considered stable under these conditions.
The test substance was formulated for dosing as a suspension in propylene glycol (Merck, Darmstadt, Germany), specific gravity 1.036, based on trial formulations performed at the test facility. Formulations (w/w) were prepared daily within 6 hours prior to dosing, homogenized to a visually acceptable level, and stored at ambient temperature. Adjustment was made for specific gravity of the vehicle (1.036) and test substance (1.8). Chemical analysis of dose preparations using gas chromatography was conducted on a single occasion during the treatment phase, according to a validated method. Samples of formulations were analyzed for homogeneity (highest and lowest concentration) and accuracy of preparation (all concentrations). Stability in vehicle over 6 hours at room temperature under normal laboratory light conditions was also determined (highest and lowest concentration). The accuracy of preparation was considered acceptable if the mean measured concentrations were 85% to 115% of the target concentration. Homogeneity was demonstrated if the coefficient of variation was ≤10%. Formulations were considered stable if the relative difference before and after storage was maximally 10%.
Study design, Animals, and Animal Husbandry
The study was conducted using outbred, SPF-quality Crl:WI(Han) rats obtained from Charles River Deutschland, Sulzfeld, Germany. This species and strain of rat is recognized as appropriate for general and reproductive toxicity studies and has been shown to be susceptible to the effects of reproductive toxicants. The study laboratory has general and reproduction/developmental historical data for this species from the same strain and source (see Online Supplemental Information).
Nulliparous and nonpregnant females and experimentally naive animals were used for the study. A total of 50 male and 70 female animals were acclimatized at least 5 days prior to start of treatment. A health inspection was performed prior to commencement of treatment to ensure that all animals were in a good state of health. Study animals were approximately 11 weeks old at the start of treatment. Study animals were allocated to treatment groups as shown in Table 1 by computer-generated random algorithm according to body weight (not stratified). All animals (including reserve animals) were weighed and arranged per sex from low to high body weight based on the total number of animals required for the study. Animals were randomized across the groups starting with the animal with the lowest body weight. The maximum allowed variation in individual body weights at randomization was predefined as ±20% of the mean for each sex. Reserve animals were not allocated since the ±20% criterion was reached.
Group Assignment of Rats Treated With HFPO-Amidol.
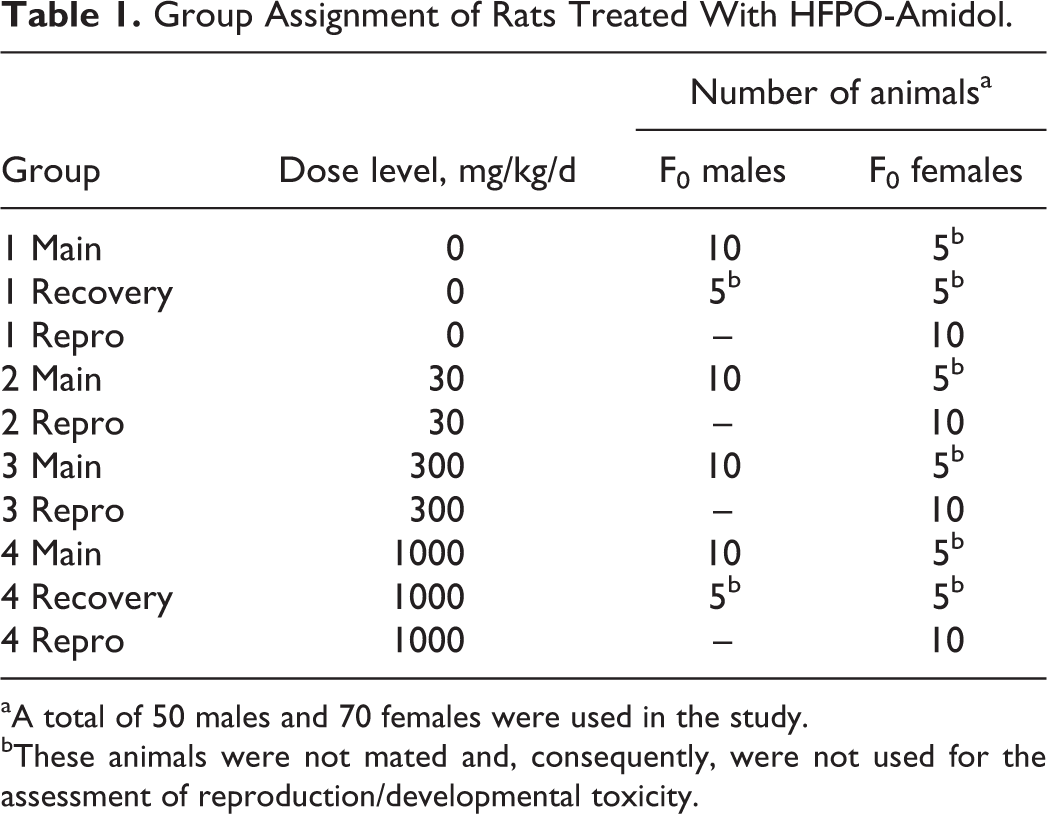
aA total of 50 males and 70 females were used in the study.
bThese animals were not mated and, consequently, were not used for the assessment of reproduction/developmental toxicity.
Animals were housed in Makrolon plastic cages (Type III and IV, height 18 cm; Technilab-BMI B.V., Someren, The Netherlands) with bedding. Mating pairs were housed by pair during cohabitation, and pregnant/nursing females were housed singly. Otherwise animals were housed by sex, 5/cage throughout the study. Study animals were housed in a controlled environment in which optimal conditions were considered to be 15 air changes per hour, a temperature of 21°C ± 3°C (actual range 18.0°C-21.6°C), relative humidity of 40% to 70% (actual range 25%-93%), and 12 hours of artificial light and 12 hours darkness per day. There were 2 occasions when the relative humidity exceeded 70% and 1 instance when the relative humidity was below 40%. Temporary fluctuations from the light–dark cycle (maximum of one hour) occurred during performance of papillary reflex tests in the animal room. These fluctuations in relative humidity and light–dark cycle were considered not to have affected the study integrity based on laboratory historical data. Animals had ad libitum access to tap water and to pelleted rodent diet (SM R/M-Z from SSNIFF Spezialdiäten GmbH, Soest, Germany) except during the period of fasting prior to clinical pathology blood collection. Animals were maintained in accordance with the Ethical Committee of WIL Research Europe BV as required by the Dutch Act on Animal Experimentation (February 1997).
Dose Justification and Administration
Dose levels were based on the results of a range-finding study. Relevant dosages were evaluated in a 10-day dosage range study in which 500 or 1000 mg/kg of HFPO-Amidol in propylene glycol was intubated daily into female rats. HFPO-Amidol produced no adverse effects on mortality, clinical signs, body weight, food consumption, gross findings, or organ weight at either dose level; therefore, dose levels of 0 (vehicle), 30, 300, and 1000 mg/kg/d were chosen for the main study.
The test article was suspended in propylene glycol and stirred continuously during dosing. All doses were administered via oral gavage at a dose volume of 5 mL/kg. Actual dose volumes were calculated according to the latest body weight. Doses were administered once daily for 7 days per week. Males were exposed for 30 days, that is, 2 weeks prior to mating, during mating, and up to the day prior to scheduled necropsy. Females in the main and recovery groups also were exposed for 30 days. At the end of the 30-day dose period, males and females in the recovery groups were held without treatment for 2 weeks. Females in the reproductive groups were exposed for 42 to 53 days, that is, for 2 weeks prior to mating, during mating, during postcoitum, and during at least 4 days of lactation (up to the day prior to scheduled necropsy).
Clinical Observations, Body Weight, and Food Consumption
Detailed clinical observations were made for all animals at least immediately after dosing. Observations were performed outside the home cage in a standard arena once prior to the start of treatment and at weekly intervals during the treatment phase. The time of onset, grade, and duration of any observed sign were recorded. Observations included evaluation of the skin, fur, eyes, ears, nose, oral cavity, thorax, abdomen, external genitalia, limbs and feet, respiratory and circulatory effects, autonomic effects such as salivation, and nervous systems effects including tremors, convulsions, reactivity to handling, and abnormal behavior.
Main and recovery group animals were weighed on the first day of exposure and weekly thereafter. Mated females in the reproductive groups were weighed on postcoitum days (PCDs) 0, 4, 7, 11, 14, 17, and 20 and lactation days (LDs) 1 and 4.
Food consumption was measured by cage for 2 cages per sex per group, excluding the second cage of main group males in each dose group to provide the same number of observations for male and female test animals. Food consumption was measured weekly for the main and recovery groups except during cohabitation. For the reproductive groups, food consumption was measured weekly prior to cohabitation, on PCDs 0, 4, 7, 14, 17 and 20, and LDs 1 and 4.
Functional Observation Tests
Functional observation tests were performed during week 4 of treatment. All tests were conducted before blood sampling, within 3 hours after dosing, and after completing clinical observations. Tests were performed on each individual animal of the first 5 main males/group, all main females/group, and all recovery animals. Hearing ability, pupillary reflex, static righting reflex, and grip strength were evaluated as normal/present or abnormal/absent. Locomotor activity was evaluated for 1 hour (12 five-minute measurement intervals for each animal) under normal laboratory light conditions using a computerized monitoring system (Kinder Scientific LLC, Poway, California). The system included a high-density frame with 22 infrared beams (7 beams on the x-axis and 15 beams on the y-axis in a 60.33 × 26.7 × 2.11 cm metal frame. A polycarbonate cage (Ancare Corp, Bellmore, New York; 48.3 × 26.7 × 20.3 cm) was placed within the frame. Because a statistically significant effect on motor activity was noted for males at 1000 mg/kg/d, an additional motor activity evaluation was conducted for all recovery group males during the last week of the recovery period.
Hematology and Clinical Chemistry
Blood samples were collected from the first 5 main males/group, all main females/group, and all recovery animals at the end of the treatment period. In addition, blood samples were collected from the recovery animals at the end of the posttreatment recovery period. The animals were fasted overnight (for a maximum of 20 hours) before blood sampling, with water provided. Blood samples were collected under anesthesia using an isoflurane (Abbott BV, Hoofddorp, the Netherlands) in nitrous oxide/oxygen (Air Products, Amsterdam, the Netherlands) combination immediately prior to scheduled postmortem examination. Blood samples were drawn from the retro-orbital sinus and collected into tubes (Greiner Bio-One, Bad Haller, Austria) prepared with EDTA for hematological parameters (0.5 mL), with citrate for clotting tests (0.45 mL), and Li-heparin-treated tubes for clinical biochemistry parameters (0.5 mL). An additional 0.25 mL blood sample was collected into untreated tubes for determination of bile acids.
Hematology parameters were obtained using an ADVIA 120 automated hematology analyzer (Siemens Medical Solution Diagnostics, Breda, the Netherlands). Parameters evaluated included red blood cell count, reticulocyte count, red blood cell distribution width, hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, platelet count, white blood cell count, and differential white blood cell count.
Clotting potential was determined using an STA Compact coagulation analyzer (Roche Diagnostics). Parameters measured were prothrombin time and activated partial thromboplastin time.
Clinical biochemistry parameters were evaluated using an Olympus AU400 chemistry analyzer (Beckman Coulter Nederland BV, Woerden, the Netherlands). Parameters evaluated included alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, total protein, albumin, total bilirubin, urea, creatinine, glucose, cholesterol, inorganic phosphorus, sodium, potassium, chloride, calcium, and bile acids.
Necropsy
For necropsy of parental animals, the first 5 main group males, all main group females, and all recovery animals were deprived of food overnight prior to planned necropsy, but water was provided. Animals surviving to scheduled necropsy were anaesthetized using an isoflurane (Abbott BV) in nitrous oxide/oxygen (Air Products) combination and subsequently exsanguinated. Main males and females were necropsied after 30 days of dose administration. Recovery males and females were necropsied after the recovery period of at least 14 days, which was at least 14 days after the scheduled kill of the respective main sex. Repro females that delivered were necropsied on LDs 5 to 6. Repro females that failed to deliver were necropsied on PCDs 26 to 27.
All animals were subjected to macroscopic examination of the cranial, thoracic, and abdominal tissues and organs, with special attention being paid to the reproductive organs. Descriptions of all macroscopic abnormalities were recorded. The numbers of former implantation sites and corpora lutea were recorded for all paired Repro females.
Weights of the following organs were recorded for the first 5 main males/group, all main females/group, and all recovery animals: adrenal glands, brain, epididymides, heart, kidneys, liver, ovaries, spleen, testes, thymus, and uterus (including cervix). Weights of the following organs were recorded for the same groups after fixation for at least 24 hours: prostate, seminal vesicles including coagulating glands, and thyroid including parathyroid. Weights of the epididymides and testes were recorded for all remaining males.
Samples of the following tissues and organs were collected from the first 5 main males/group, all main females/group, and all recovery animals and fixed in 10% buffered formalin (neutral phosphate buffered 4% formaldehyde solution; Klinipath, Duiven, the Netherlands): adrenal glands, aorta, brain (cerebellum, mid-brain, and cortex), cecum, cervix, clitoral gland, colon, coagulation gland, duodenum, esophagus, female mammary gland area, femur including joint, heart, ileum, jejunum, kidneys, lacrimal gland, larynx, liver, lung (infused with formalin), lymph nodes (mandibular and mesenteric), nasopharynx, ovaries, pancreas, Peyer patches (if detectable), pituitary gland, preputial gland, prostate gland, rectum, salivary glands (mandibular and sublingual), sciatic nerve, seminal vesicles, skeletal muscle, skin, spinal cord (cervical, midthoracic, and lumbar), spleen, sternum with bone marrow, stomach, thymus, thyroid (including parathyroid if detectable), tongue, trachea, urinary bladder, uterus, vagina, and all detected gross lesions. The following tissues and organs were collected from all remaining animals, including females that fail to deliver: cervix, clitoral gland, coagulation gland, ovaries, preputial gland, prostate gland, seminal vesicles, uterus, vagina, and all detected gross lesions. Epididymides, eyes with optic nerve, and testes were first fixed in modified Davidson solution for at least 24 hours and then transferred to formalin.
Histopathological Examinations
The OECD-specified organs and tissues collected at necropsy were processed, embedded, and sectioned at a thickness of 2 to 4 μm and stained with hematoxylin and eosin (Klinipath) for histopathological examination. The aorta, lacrimal gland, larynx, esophagus, nasopharynx, pancreas, mandibular and sublingual salivary glands, skin, and tongue were not evaluated by the pathologist because no signs of toxicity were noted at macroscopic examination and because they are not OECD-specified organs and tissues.
Additional slides of the testes were prepared to examine staging of spermatogenesis in the following animals: the first 5 main control group males, the first 5 males at 1000 mg/kg/d, and the male at 30 mg/kg/d whose cohoused female did not become pregnant. The testes were processed, sectioned at 3 to 4 μm, and stained with periodic acid–Schiff/hematoxylin (Klinipath).
The following slides were examined by a pathologist: the preserved organs and tissues of the first 5 main males and all main females of the control and 1000 mg/kg/d groups; the additional slides of the testes for staging of spermatogenesis; all gross lesions of all animals; and the reproductive organs of females that failed to deliver (cervix, clitoral gland, ovaries, uterus, and vagina) and males that failed to sire (coagulation gland, epididymides, preputial gland, prostate gland, seminal vesicles, and testis).
Reproductive Toxicity Assessment
Reproductive group females were dosed daily with HFPO-Amidol for 14 consecutive days prior to mating, during the mating period (2 weeks), throughout gestation and through lactation (LD 4). During the mating period, each female was cohabited with a male of the same treatment group until evidence of mating (copulatory plug, sperm in the lavage sample, and/or determination of estrus stage) was obtained. The day on which mating was confirmed was designated PCD 0. Once mating occurred, the males and females were separated. At the end of the 2-week cohabitation period, females with no confirmed evidence of mating were returned to individual cages until study termination.
Pregnant females were allowed to give birth undisturbed. The day parturition was complete, and pups were nursing was designated as LD 1. The first litter check occurred on LD 1. On LD 1, all pups were sexed and body weights and descriptions of all external abnormalities were recorded. The stomach was examined for the presence of milk. Also on LD 1, pups were randomized per litter and individually identified by tattoo marking. The offspring were individually examined daily for abnormal behavior and appearance. Dead and abnormal pups were recorded daily, and, if possible, defects or cause of death were evaluated. On LD 4, sex was determined, and body weight was recorded for all surviving offspring. On LD 5, all surviving dams and offspring were weighed and sacrificed. Paired females which failed to deliver were weighed and sacrificed on PCD 26 to 27.
All offspring surviving to LD 5 were euthanized by decapitation and discarded. Procedures followed for euthanasia, necropsy, and gross examination of females in the reproductive groups were consistent with those described previously for the subacute main and recovery groups. Additionally, these females were examined macroscopically for the presence and number of uterine implantation sites and ovarian corpora lutea. For females which failed to deliver, the reproductive organs were collected, processed, and examined microscopically in the same manner as for the main group females.
Data Collection
Observations and measurements in the study were recorded electronically using the following programs: REES Centron Environmental Monitoring system version SQL 2.0 (REES Scientific, Trenton, New Jersey) was used to record environmental conditions. TOXDATA version 8.0 (NOTOX B.V.,’s-Hertogenbosch, the Netherlands) was used to record mortality, clinical signs, body weights, food consumption, reproduction parameters, pup observations, and organ weights. Advia 120 version V.3.1.8.0.MS (Siemens Medical Solution Diagnostics) was used to collect hematology data. Sta Compact version 1.06.06 (Stago Instruments, Gennevilliers, France) was used for clotting data. Olympus AU 400 version 8.2A (Beckman Coulter Nederland BV) was used to collect clinical chemistry data. Motor Monitor version 08356-14 (Kinder Scientific LLC) was used to record motor activity. Pathdata version 6.2D (Pathology Data Systems, Basel, Switzerland) was used for histopathology data. Empower version 5.00 (Waters, Milford, Massachusetts) was used for analytical chemistry data.
Statistical Analysis
For variables assumed to follow a normal distribution, the Dunnett test 7 based on a pooled covariance estimate was applied for comparison of the treated groups and the control groups for each sex. The Steel test 8 was applied if the data could not be assumed to follow a normal distribution. The Fisher Exact test 9 was applied to frequency data. The following additional methods of statistical analysis were used. Motor activity data were subjected to the Kruskal-Wallis nonparametric analysis of variance test 10 to determine intergroup differences, followed by the Wilcoxon test, 11 to compare the treated groups to the control group. All statistical tests were 2 sided, and in all cases, P < 0.05 was accepted as the lowest level of significance.
Results
Verification of Test Article Concentration, Homogeneity, and Stability
The concentrations analyzed in all HFPO-Amidol formulations were in agreement with target concentrations (ie, mean accuracies between 85% and 115%). No HFPO-Amidol was detected in the vehicle (control) formulation. The assayed HFPO-Amidol formulations were homogeneous (ie, coefficient of variation ≤10%). Formulations across the entire range were stable when stored at room temperature under normal laboratory light conditions for at least 6 hours.
Mortality and Clinical Observations
No mortality occurred during the study period. No clinical signs of toxicity were noted during the observation period. Incidental findings included alopecia, scabs, salivation, and opacity of the right eye. These findings occurred within the range of background incidence expected for rats of this age and strains that are housed and treated under the conditions in this study (data not shown). At the incidence observed, these findings were not considered to be of toxicological relevance.
Body Weight
No toxicologically relevant changes in body weights and body weight gain were noted (Figures 2 and 3, Tables 2 and 3; see Online Supplemental Information for historical control data). The statistically significant lower mean body weights of males and Repro females at 1000 mg/kg/d on day 1 of treatment/premating were not treatment related since this finding occurred prior to the first dose.
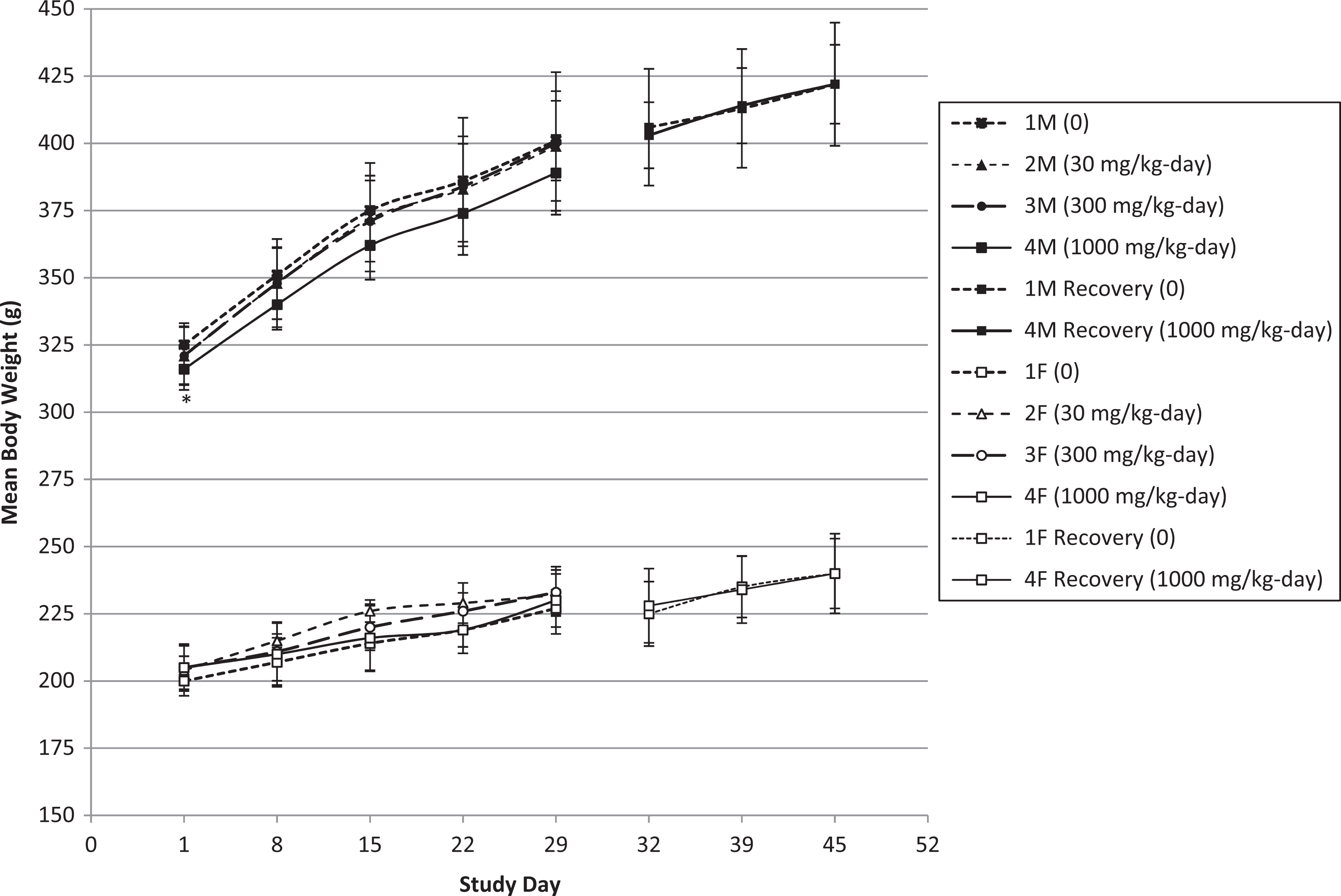
Mean body weights for male (M) and female (F) rats during the 28-day oral exposure period for HFPO-amidol and the 14-day recovery period. Error bars represent ±1 standard deviation. *1000 mg/kg/d-treated male group significantly decreased relative to the control group (Dunnett test based on pooled variance P < 0.05).
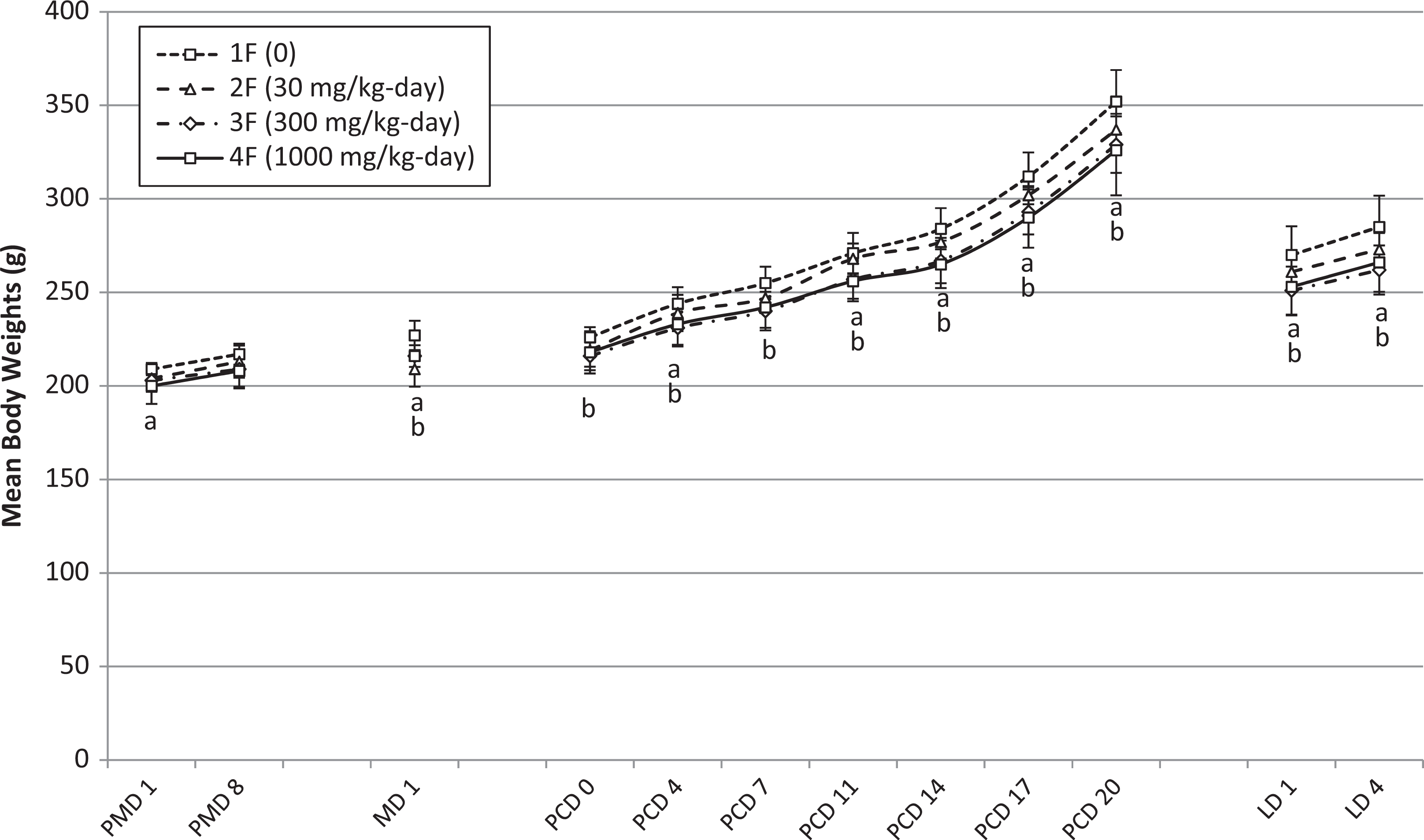
Mean body weights for reproductive group female rats during the premating, mating, postcoitum, and lactation oral exposure periods for HFPO-amidol. Error bars represent ± 1 standard deviation. One female treated with 1000 mg/kg/d successfully mated during mating week 2 (MD 8 body weight = 245 g). All other 1000 mg/kg/d-treated females mated during mating week 1. A, 1000 mg/kg/d-treated group significantly decreased relative to the control group (Dunnett test based on pooled variance, P < 0.05 or P < 0.01). B, 300 mg/kg/d-treated group significantly decreased relative to the control group (Dunnett test based on pooled variance, P < 0.05 or P < 0.01). PMD indicates premating day; MD, mating day; PCD, postcoitum day; LD, lactation day.
Body Weights (g) for Male and Female Rats in the Main and Recovery Groups.a
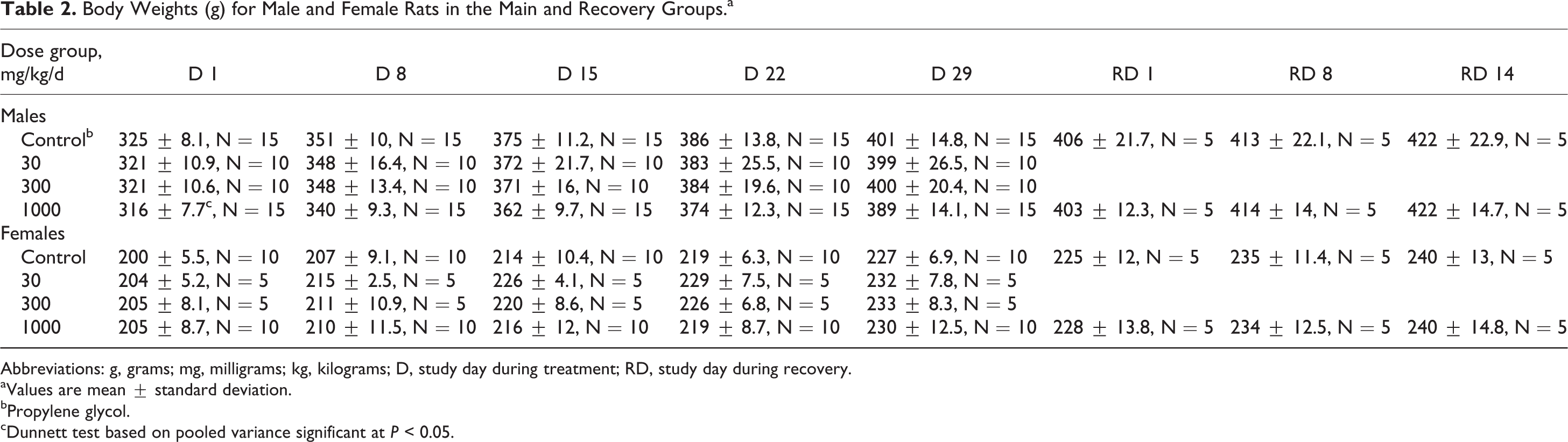
Abbreviations: g, grams; mg, milligrams; kg, kilograms; D, study day during treatment; RD, study day during recovery.
aValues are mean ± standard deviation.
bPropylene glycol.
cDunnett test based on pooled variance significant at P < 0.05.
Body Weights (g) for Reproductive Group Female Rats During Premating, Mating, Postcoitum, and Lactation.a
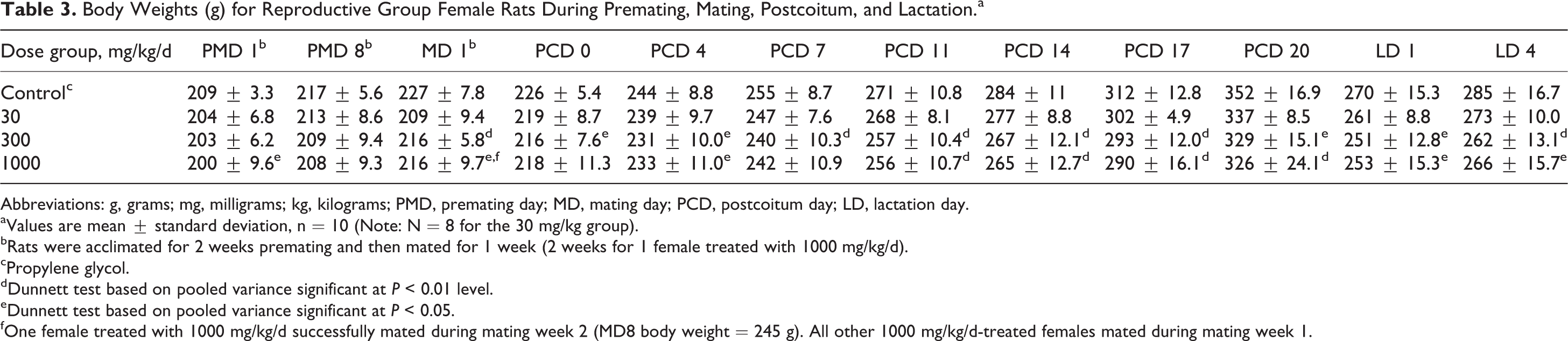
Abbreviations: g, grams; mg, milligrams; kg, kilograms; PMD, premating day; MD, mating day; PCD, postcoitum day; LD, lactation day.
aValues are mean ± standard deviation, n = 10 (Note: N = 8 for the 30 mg/kg group).
bRats were acclimated for 2 weeks premating and then mated for 1 week (2 weeks for 1 female treated with 1000 mg/kg/d).
cPropylene glycol.
dDunnett test based on pooled variance significant at P < 0.01 level.
eDunnett test based on pooled variance significant at P < 0.05.
fOne female treated with 1000 mg/kg/d successfully mated during mating week 2 (MD8 body weight = 245 g). All other 1000 mg/kg/d-treated females mated during mating week 1.
Reproductive group females showed statistically significant lower body weights during the mating, postcoitum, and lactation phases at 300 and 1000 mg/kg/d and a statistically significantly lower body weight gain at 1000 mg/kg/d during the postcoitum period on PCD 17 (33% gain over PCD 0 weight vs control gain of 38% over PCD 0 weight). Body weight gain of these females remained slightly lower on PCD 20 but not statistically different to control levels (Table 4
Body Weight Gains (%) for Male and Female Rats in the Main and Recovery Groups.a
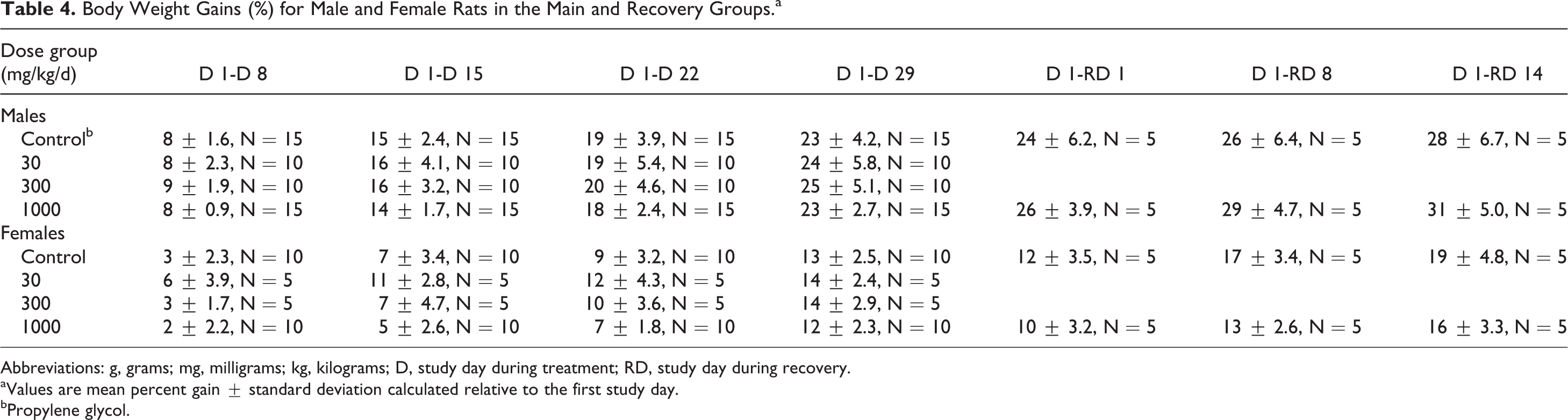
Abbreviations: g, grams; mg, milligrams; kg, kilograms; D, study day during treatment; RD, study day during recovery.
aValues are mean percent gain ± standard deviation calculated relative to the first study day.
bPropylene glycol.
Body Weight Gains (%) for Reproductive Group Female Rats.a
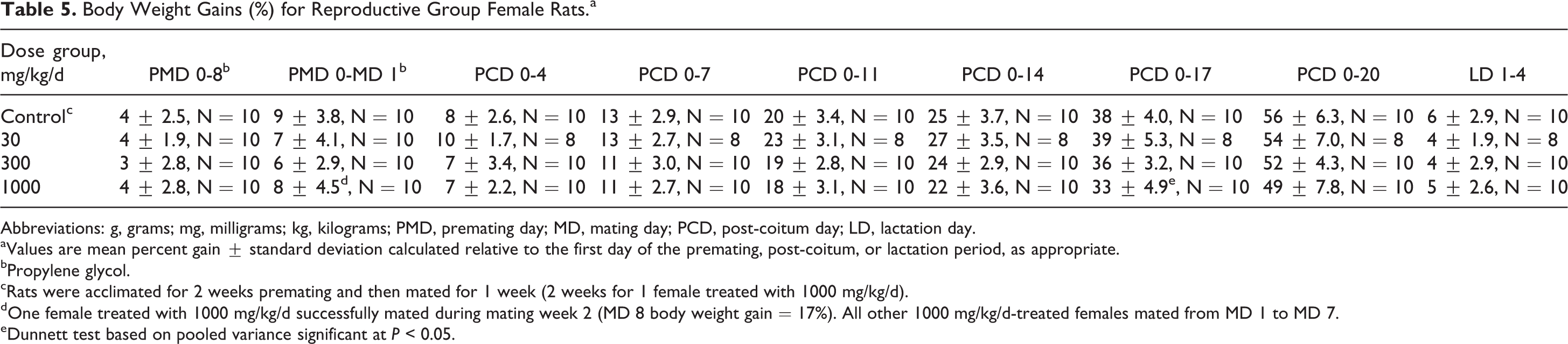
Abbreviations: g, grams; mg, milligrams; kg, kilograms; PMD, premating day; MD, mating day; PCD, post-coitum day; LD, lactation day.
aValues are mean percent gain ± standard deviation calculated relative to the first day of the premating, post-coitum, or lactation period, as appropriate.
bPropylene glycol.
cRats were acclimated for 2 weeks premating and then mated for 1 week (2 weeks for 1 female treated with 1000 mg/kg/d).
dOne female treated with 1000 mg/kg/d successfully mated during mating week 2 (MD 8 body weight gain = 17%). All other 1000 mg/kg/d-treated females mated from MD 1 to MD 7.
eDunnett test based on pooled variance significant at P < 0.05.
Food Consumption
No treatment-related changes in food consumption before or after allowance for body weight were noted. During the postcoitum and lactation phases, reproductive group females at 300 and 1000 mg/kg/d had lower food intake which was statistically significant for most measurement intervals (Table 6). No toxicological relevance was ascribed to these changes because food intake was essentially similar to control levels after allowance for body weight (except for the significantly decreased relative food consumption of reproductive group females at 1000 mg/kg/d for intervals PCD 7-11 and PCD 11-14; Table 7). These changes in food consumption before or after allowance for body weight occurred in the absence of a clear dose-related trend. In addition, the differences were inconsistently seen over time (eg, on PCD 17-20, there was no statistically significant difference in food intake between control and 300 and 1000 mg/kg/d groups, respectively). As such, these changes in food intake were considered to be unrelated to treatment with the test substance (see also Online Supplemental Information for historical control data).
Food Consumption (g/animal/d) Summary for Reproductive Group Female Rats.a
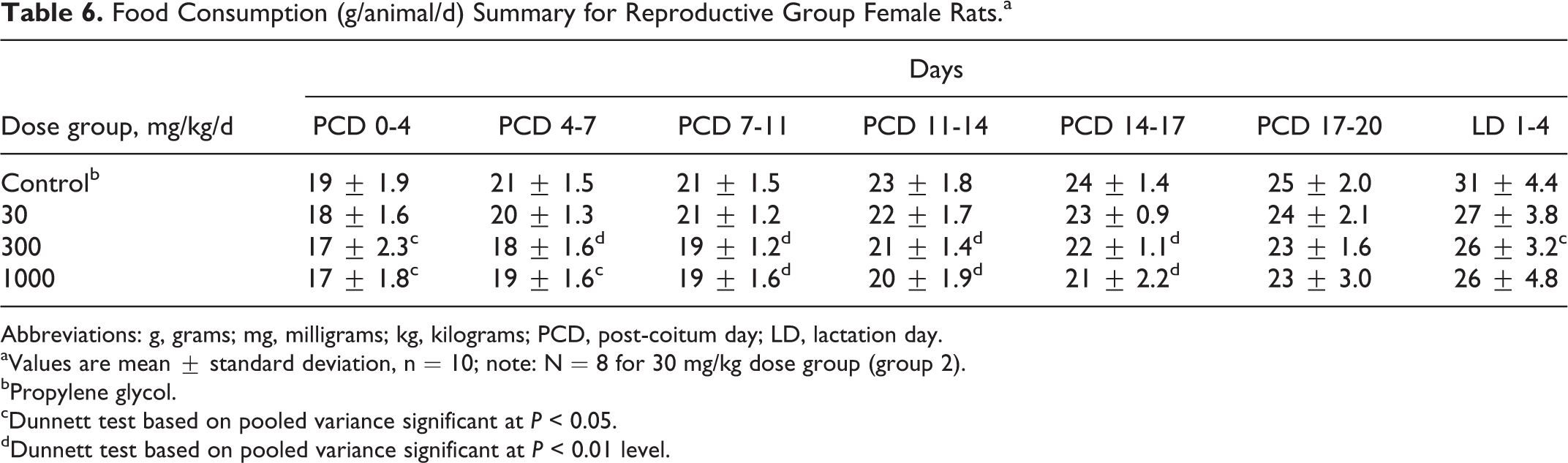
Abbreviations: g, grams; mg, milligrams; kg, kilograms; PCD, post-coitum day; LD, lactation day.
aValues are mean ± standard deviation, n = 10; note: N = 8 for 30 mg/kg dose group (group 2).
bPropylene glycol.
cDunnett test based on pooled variance significant at P < 0.05.
dDunnett test based on pooled variance significant at P < 0.01 level.
Relative Food Consumption (g/kg bw/d) summary for Reproductive Group Female Rats.a
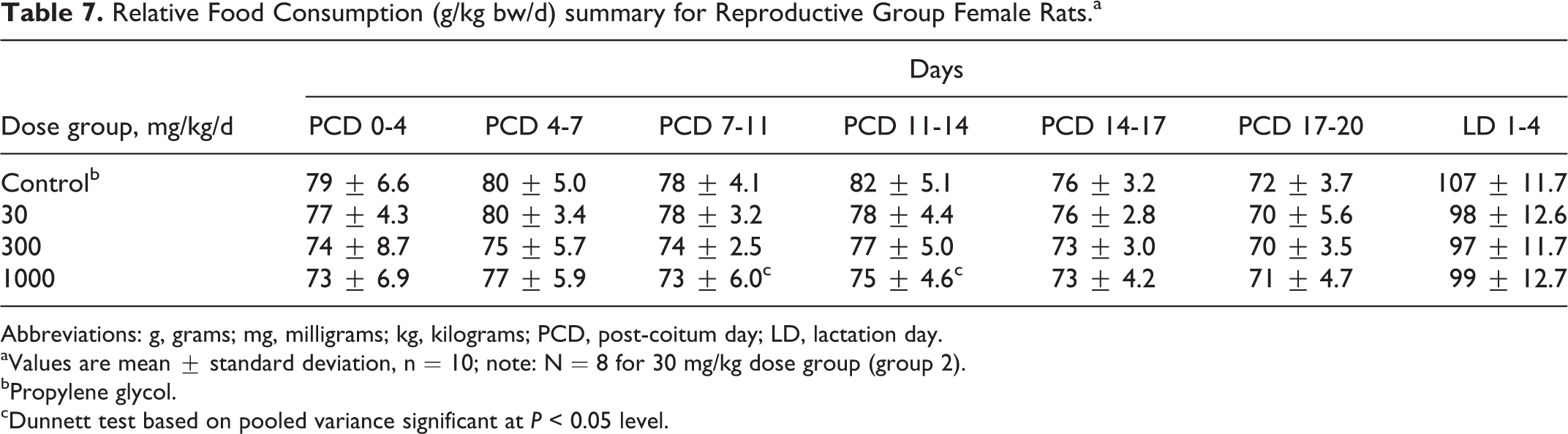
Abbreviations: g, grams; mg, milligrams; kg, kilograms; PCD, post-coitum day; LD, lactation day.
aValues are mean ± standard deviation, n = 10; note: N = 8 for 30 mg/kg dose group (group 2).
bPropylene glycol.
cDunnett test based on pooled variance significant at P < 0.05 level.
Functional Observations and Locomotor Activity
Hearing ability, pupillary reflex, static righting reflex, and grip strength were normal in all animals (data not shown). No treatment-related changes were noted in locomotor activity of animals at 30 and 300 mg/kg/d. Any statistically significant increases observed at these dose levels occurred in the absence of a dose-related trend and were intermittently noted during the 60-minute measurement interval (Table 8; see Online Supplemental Information for historical control data). Statistically significant increases in the total locomotor activity over the 60-minute interval (mean ambulatory and total motor activity counts) of males at 1000 mg/kg/d were observed at the end of the treatment and recovery periods (Table 8 and Figures 4 –7). Based on individual 5-minute intervals, a statistically significant increase was noted at the end of treatment for total movements during the first 5-minute interval and for ambulations during the first and third 5-minute interval. The possible toxicological relevance of these results could not be excluded. However, this increase in motor activity was transient in nature, and data showed that habituation occurred, that is, mean activity gradually decreased to values similar to control levels within approximately 15 to 20 minutes. This difference in initial exploratory activity was suggestive of a temporary hyperreactivity to a new environment/stimulus.
Summary of Interval and Total Mean Movements and Ambulations for Male and Female Rats Dosed With HFPO-Amidol for a Minimum of 28 Days and Male Rats After a 14-Day Recovery Period.
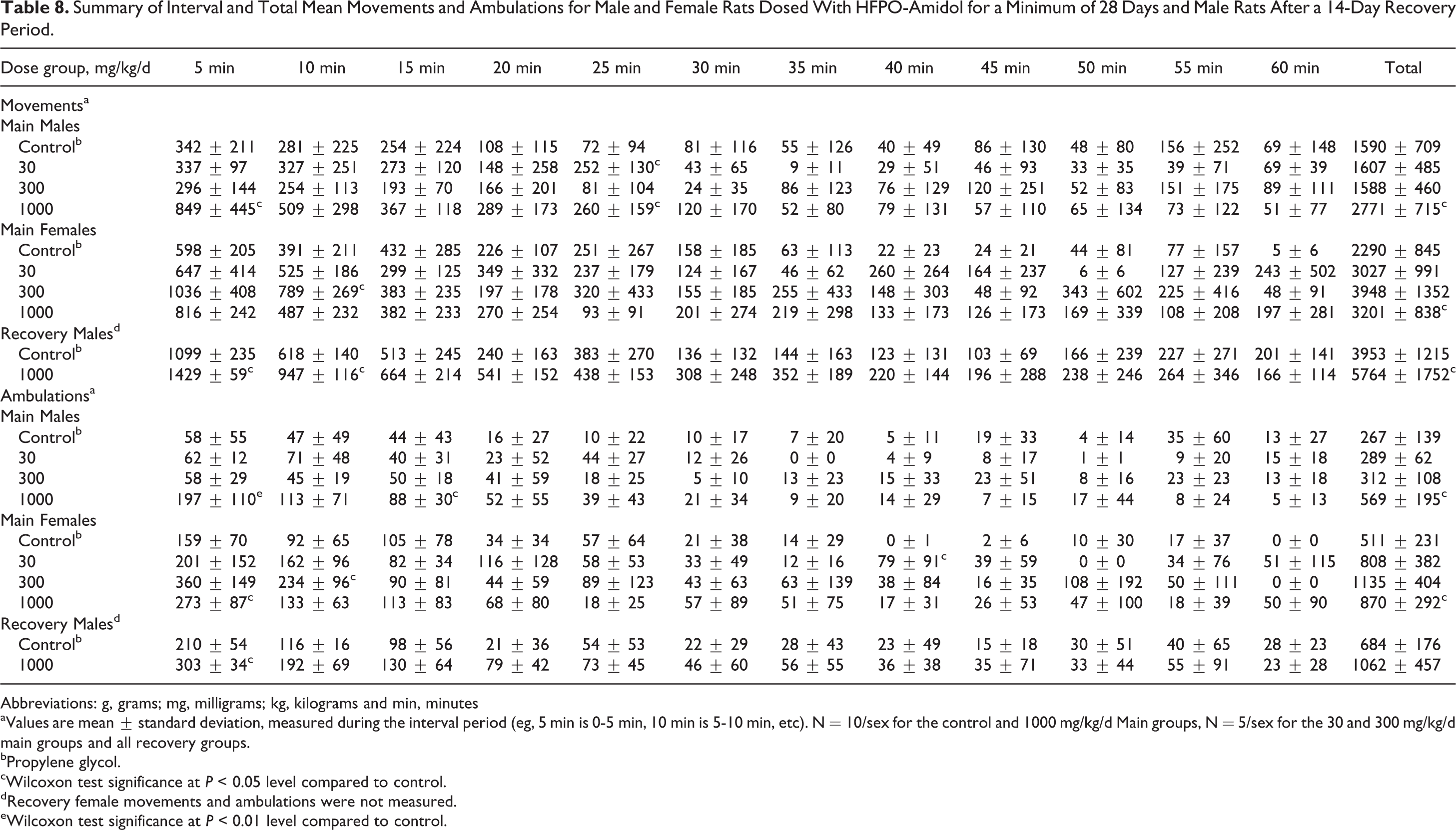
Abbreviations: g, grams; mg, milligrams; kg, kilograms and min, minutes
aValues are mean ± standard deviation, measured during the interval period (eg, 5 min is 0-5 min, 10 min is 5-10 min, etc). N = 10/sex for the control and 1000 mg/kg/d Main groups, N = 5/sex for the 30 and 300 mg/kg/d main groups and all recovery groups.
bPropylene glycol.
cWilcoxon test significance at P < 0.05 level compared to control.
dRecovery female movements and ambulations were not measured.
eWilcoxon test significance at P < 0.01 level compared to control.
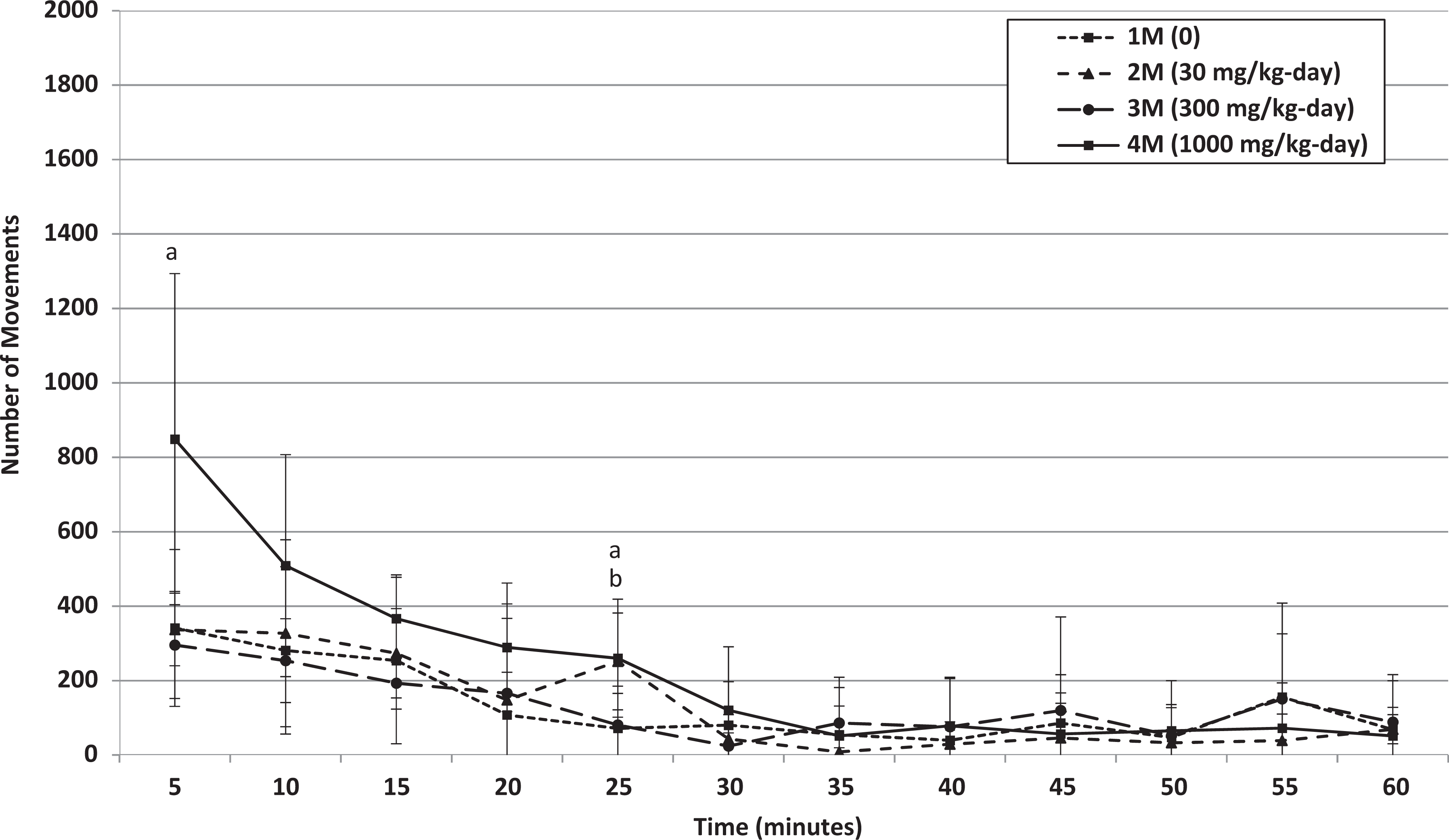
Week 4: Mean movements of male rats that received 0, 30, 300, or 1000 mg/kg/d HFPO-Amidol via daily oral gavage for 28 days. Error bars represent ±1 standard deviation. The measurement intervals are designated by the end time (eg, 5 minutes is the measurement from 0 to 5 minutes, etc). A, 1000 mg/kg/d-treated group significantly increased relative to the control group (Wilcoxon test, P < 0.05). B, 300 mg/kg/d-treated group significantly increased relative to the control group (Wilcoxon test, P < 0.05).
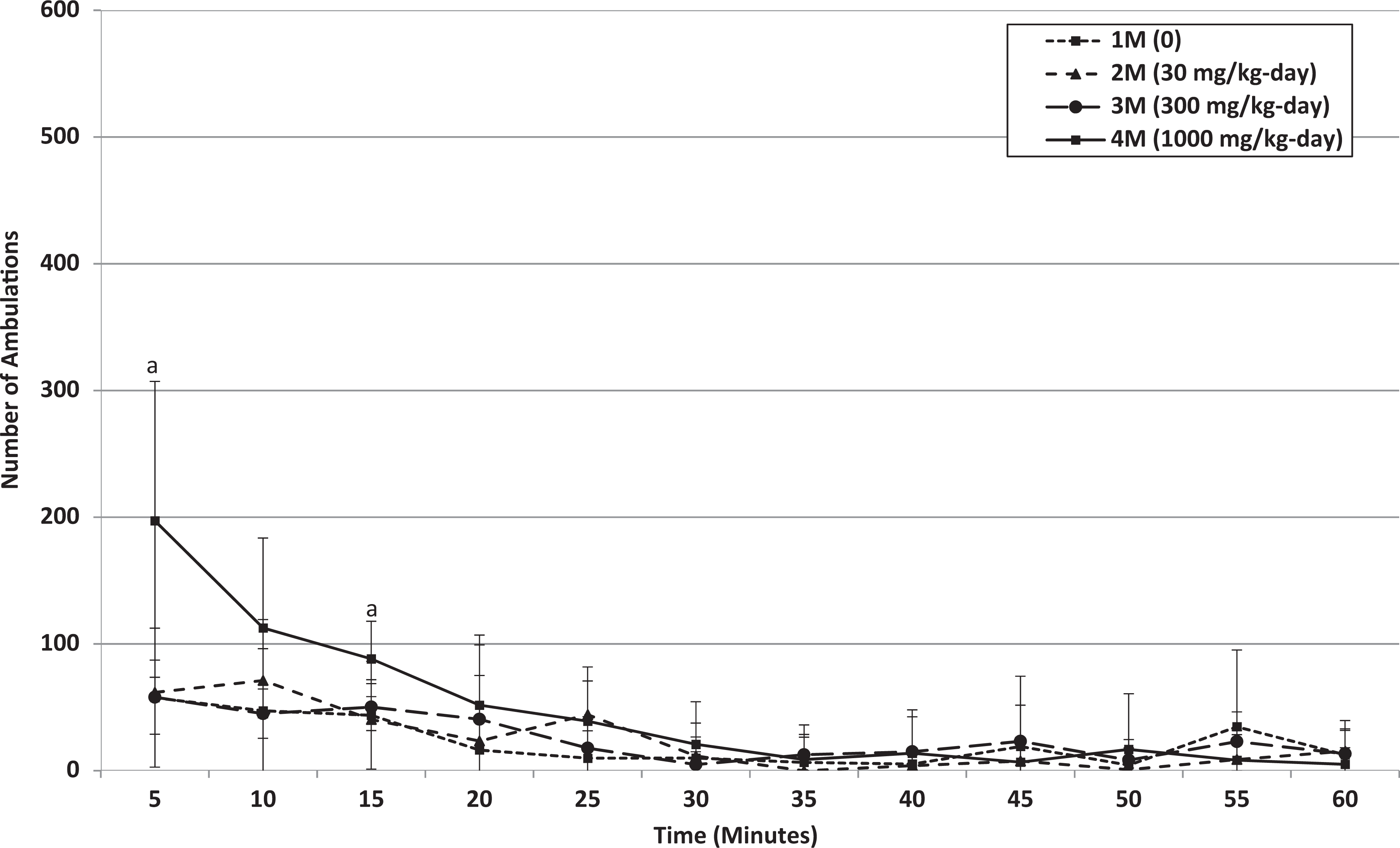
Week 4: Mean ambulations of male rats that received 0, 30, 300, or 1000 mg/kg/d HFPO-Amidol via daily oral gavage for 28 days. Error bars represent ±1 standard deviation. The measurement intervals are designated by the end time (eg, 5 minutes is the measurement from 0 to 5 minutes, etc). A, 1000 mg/kg/d-treated group significantly increased relative to the control group (Wilcoxon test, P < 0.05 or P < 0.01).
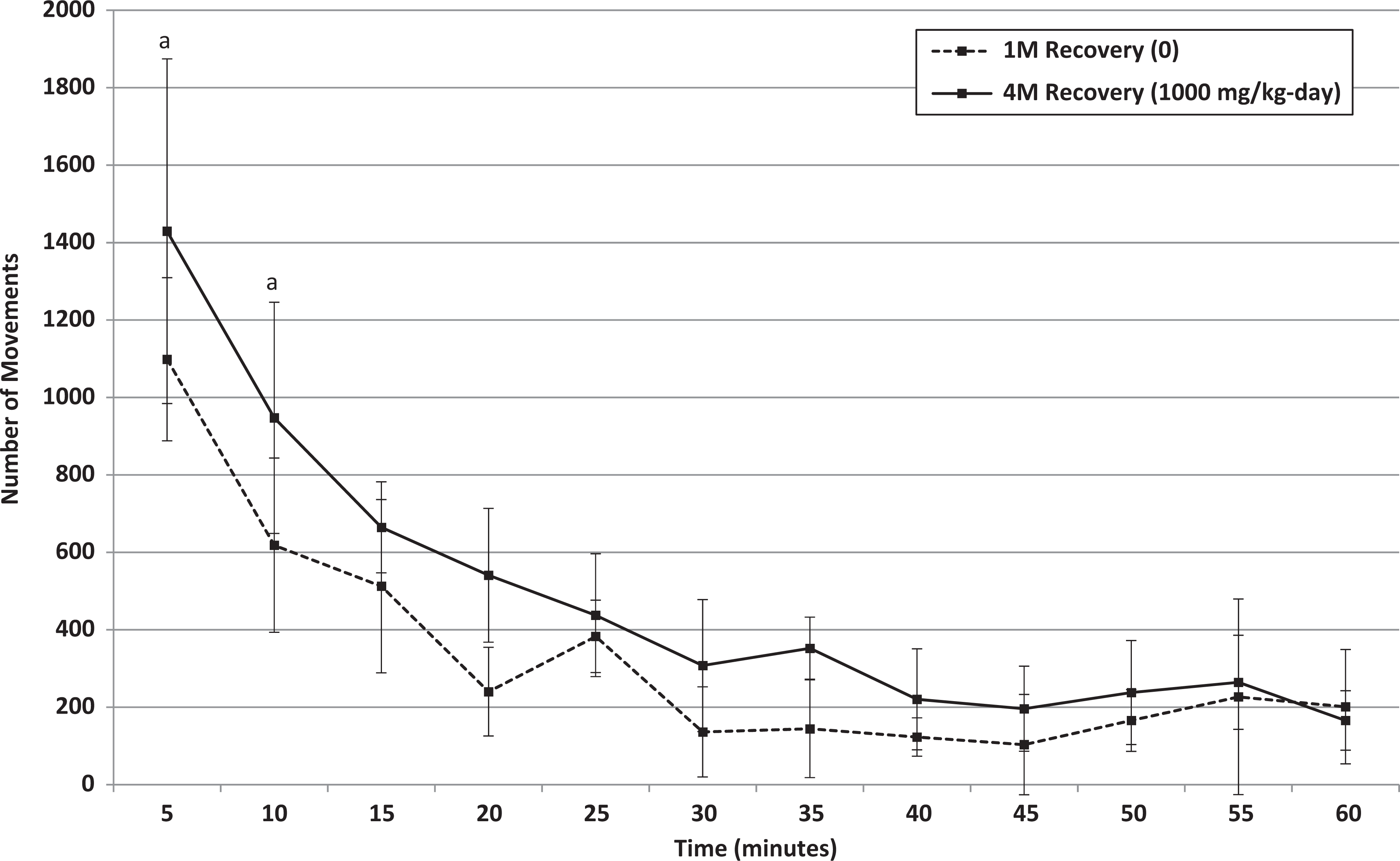
Recovery: Mean movements of male rats that received 0 or 1000 mg/kg/d HFPO-Amidol via daily oral gavage for 28 days. Error bars represent ±1 standard deviation. The measurement intervals are designated by the end time (eg, 5 minutes is the measurement from 0 to 5 min, etc). A, 1000 mg/kg/d-treated group significantly increased relative to the control group (Wilcoxon test, P < 0.05).
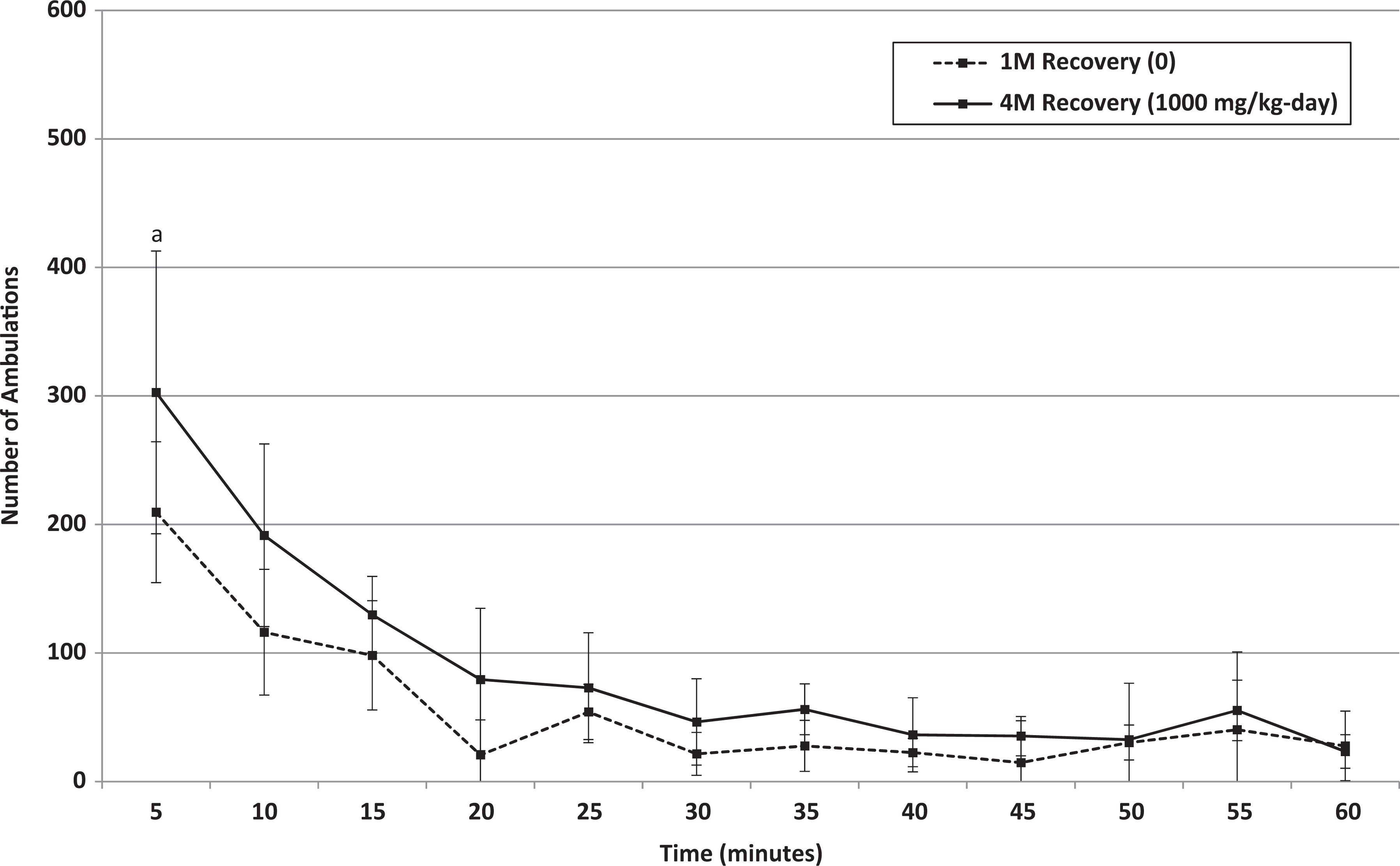
Recovery: Mean ambulations of male rats that received 0 or 1000 mg/kg/d HFPO-Amidol via daily oral gavage for 28 days. Error bars represent ±1 standard deviation. The measurement intervals are designated by the end time (eg, 5 minutes is the measurement from 0 to 5 minutes, etc). A, 1000 mg/kg/d-treated group significantly increased relative to the control group (Wilcoxon test, P < 0.05).
A statistically significant higher mean total movement counts for 1000 mg/kg/d males was also observed at the end of the recovery period. Based on individual 5-minute intervals, statistically significant increases at the end of the recovery period were noted for males at 1000 mg/kg/d for total movements during the first and second 5-minute intervals and for ambulations during the first 5-minute interval only. The difference in mean counts recorded between control and high-dose males during the first 5-minute interval was lower at the end of the recovery period compared to the end of the treatment period (by a factor of approximately 1.5 vs approximately 3 at the end of recovery and treatment, respectively). This may indicate that partial recovery occurred.
The statistically significant increase in total locomotor activity over the 60-minute interval (both total movements and ambulations and also for ambulations during the first 5-minute interval) of females at 1000 mg/kg/d at the end of the treatment period was not considered treatment related because no dose-related trend was noted (ie, total means over the 60-minute interval at 1000 mg/kg/d were similar to those at 30 mg/kg/d, Figures 8 and 9) and because the habituation profile appeared similar between treated and control females. For this reason, female motor activity was not retested in the recovery phase.
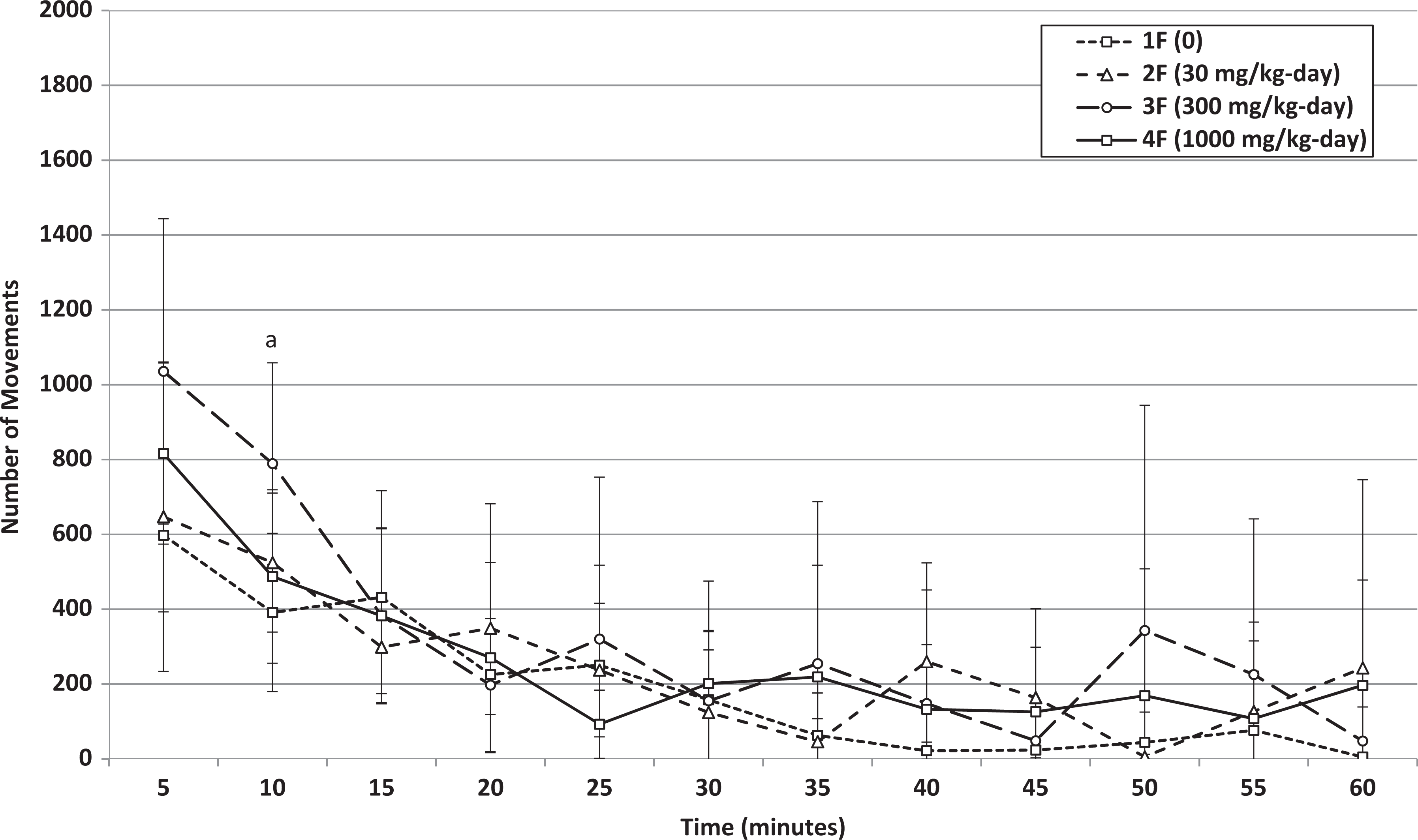
Week 4: Mean movements of female rats that received 0, 30, 300, or 1000 mg/kg-day HFPO-Amidol via daily oral gavage for 28 days. Error bars represent ±1 standard deviation. The measurement intervals are designated by the end time (eg, 5 minutes is the measurement from 0 to 5 min, etc). A 300 mg/kg/d-treated group significantly increased relative to the control group (Wilcoxon test, P < 0.05).
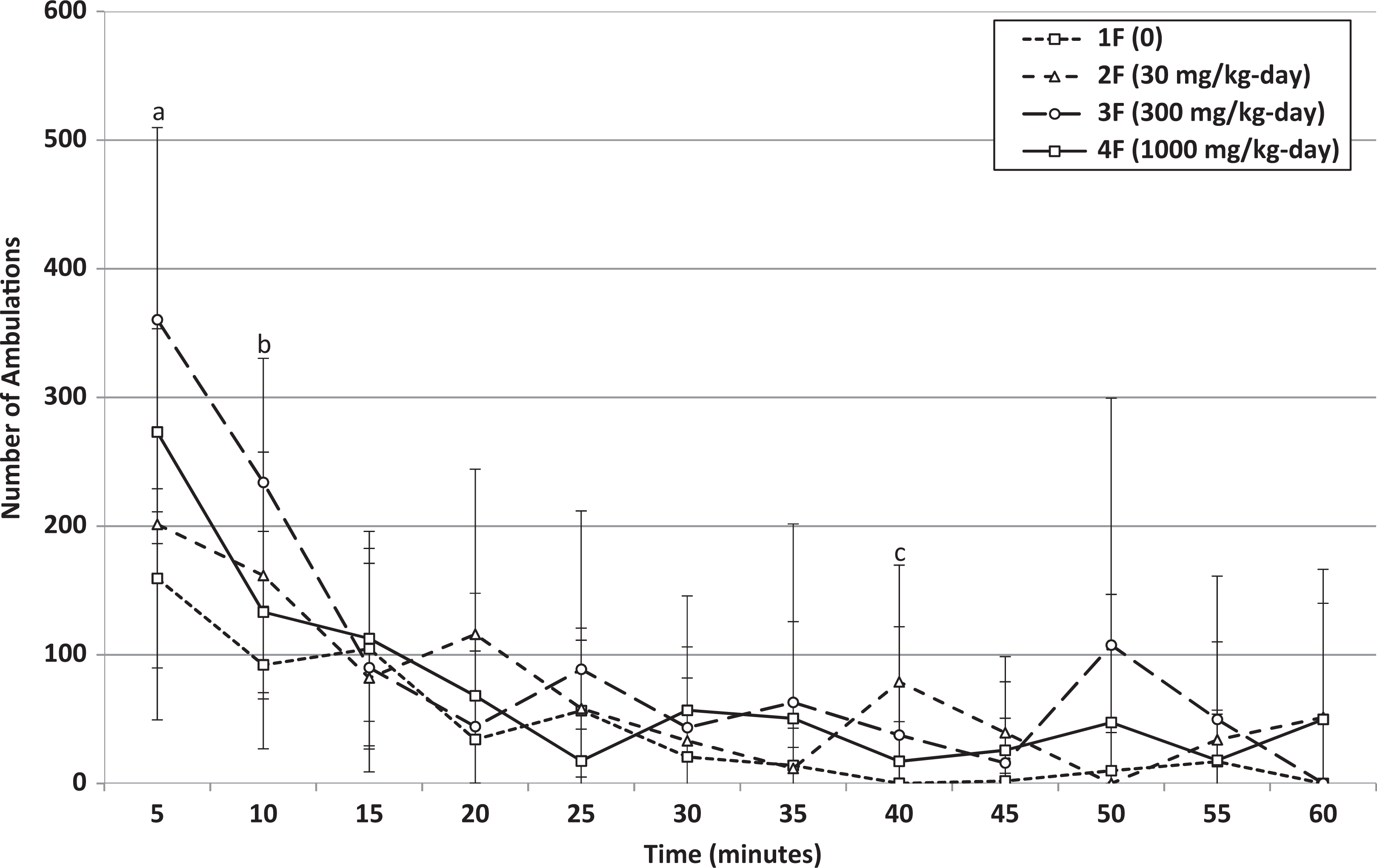
Week 4: Mean ambulations of female rats that received 0, 30, 300, or 1000 mg/kg-day HFPO-Amidol via daily oral gavage for 28 days. Error bars represent ±1 standard deviation. The measurement intervals are designated by the end time (eg, 5 minutes is the measurement from 0 to 5 minutes, etc). A, 1000 mg/kg/d-treated group significantly increased relative to the control group (Wilcoxon test, P < 0.05). B, 300 mg/kg/d-treated group significantly increased relative to the control group (Wilcoxon test, P < 0.05). C, 30 mg/kg/d-treated group significantly increased relative to the control group (Wilcoxon test, P < 0.05).
Hematology and Clinical Chemistry
Results for hematological measurements are summarized in Table 9. No toxicologically relevant changes occurred in hematological parameters of treated rats. Any statistically significant changes at the end of treatment were considered to be of no toxicological relevance, as they occurred in the absence of a treatment-related distribution. In male rats, these changes included lower prothrombin time at 30 mg/kg/d, lower white blood cell count at 300 mg/kg/d, and higher relative neutrophil count in males at 1000 mg/kg/d. The statistically significant lower white blood cell count in males at 1000 mg/kg/d at the end of the recovery phase was absent at the end of the treatment phase. No statistically significant changes were observed in the hematological parameters of female rats.
Hematology Summary.
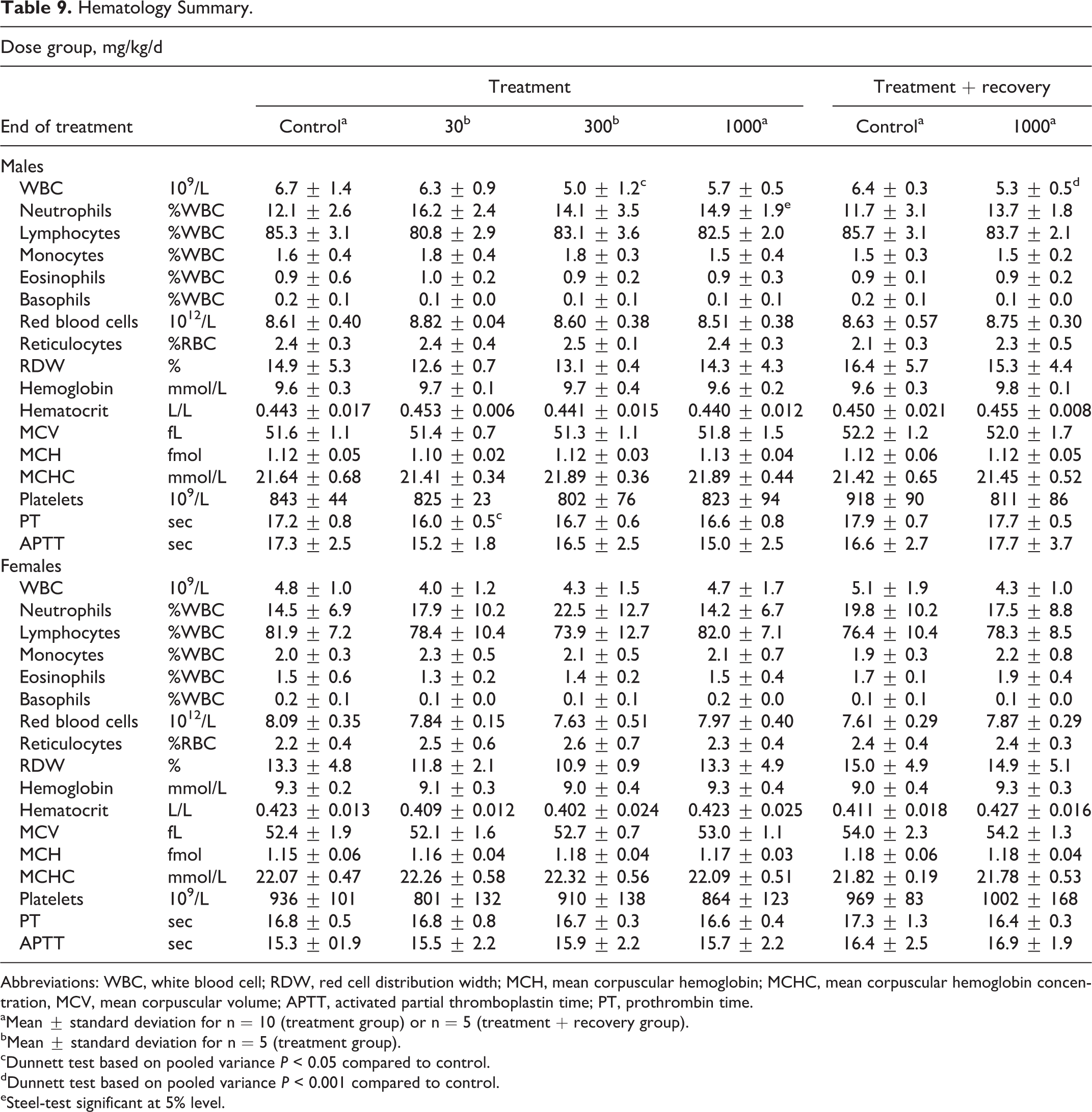
Abbreviations: WBC, white blood cell; RDW, red cell distribution width; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration, MCV, mean corpuscular volume; APTT, activated partial thromboplastin time; PT, prothrombin time.
aMean ± standard deviation for n = 10 (treatment group) or n = 5 (treatment + recovery group).
bMean ± standard deviation for n = 5 (treatment group).
cDunnett test based on pooled variance P < 0.05 compared to control.
dDunnett test based on pooled variance P < 0.001 compared to control.
eSteel-test significant at 5% level.
Results for selected clinical biochemistry measurements are summarized in Table 10. No toxicologically relevant changes occurred in clinical biochemistry parameters of treated rats. Any statistically significant changes at the end of treatment were considered to be of no toxicological relevance, as they occurred in the absence of a treatment-related distribution. These changes included lower total bilirubin and potassium levels in males at 1000 mg/kg/d, lower calcium levels in males at 300 mg/kg/d, and higher glucose and lower inorganic phosphate levels in females at 300 mg/kg/d. The statistically significant higher bile acid level in males at 1000 mg/kg/d at the end of the recovery phase was absent at the end of the treatment phase.
Clinical Biochemistry.
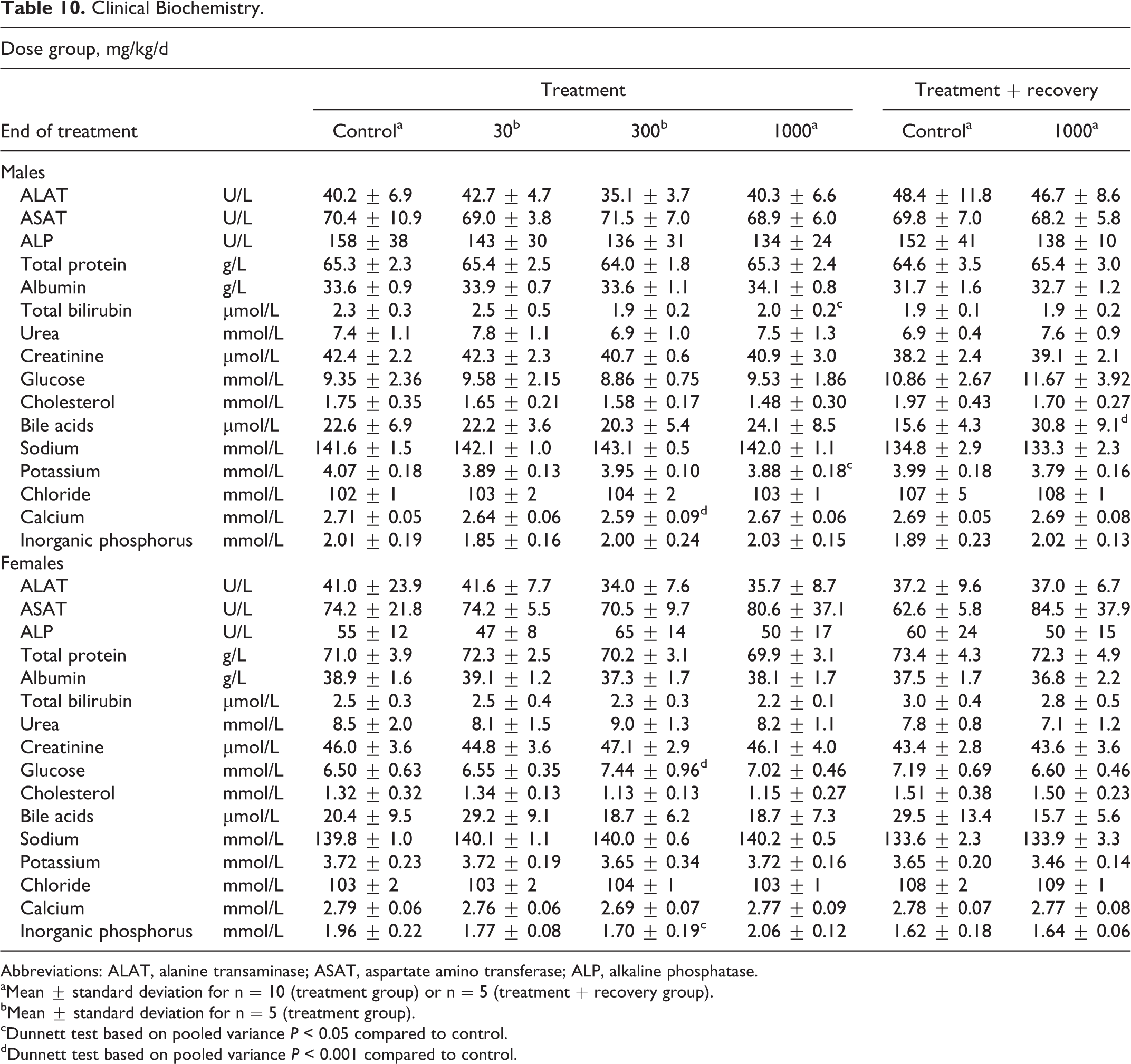
Abbreviations: ALAT, alanine transaminase; ASAT, aspartate amino transferase; ALP, alkaline phosphatase.
aMean ± standard deviation for n = 10 (treatment group) or n = 5 (treatment + recovery group).
bMean ± standard deviation for n = 5 (treatment group).
cDunnett test based on pooled variance P < 0.05 compared to control.
dDunnett test based on pooled variance P < 0.001 compared to control.
Necropsy
Necropsy did not reveal any toxicologically relevant alterations. Incidental findings among control and/or treated animals at the end of the treatment and/or recovery period included greenish foci on the kidneys, a yellowish nodule on the epididymides, reddish foci on the thymus, alopecia, irregular surface of the stomach, cloudy eyes, eye damaged during blood collection (single male at 1000 mg/kg/d), fluid in the uterus, red–brown foci on the clitoral glands, tan or greenish discoloration of the clitoral glands, black–brown focus on the adrenal glands, enlarged mandibular lymph nodes, and renal pelvic dilation. The incidence of these findings was considered to be within the range of findings that may be encountered among rats of this age and strain and did not show a dose-related incidence trend (data not shown). These necropsy findings were therefore considered to be of no toxicological relevance.
Organ weight
No toxicologically relevant changes were noted in organ weights and organ to body weight ratios. The statistically significant higher thyroid weight and thyroid to body weight ratio at the end of treatment in males at 1000 mg/kg/d (Table 11) was slight in nature (ie, remained within the range considered normal for rats of this age and strain (see Online Supplemental Information for historical control data) and had no histopathological correlate. The statistically significant higher liver, adrenal, and spleen weights and higher adrenal to body weight ratio in females at 300 mg/kg/d also remained within the range considered normal for rats of this age and strain (see Online Supplemental Information for historical control data) and occurred in the absence of a dose-related trend. Therefore, no toxicological relevance was ascribed to these changes.
Absolute and Relative Organ Weights of Male and Female Rats Following 28 Days of Daily Oral Exposure to HFPO-Amidol.a
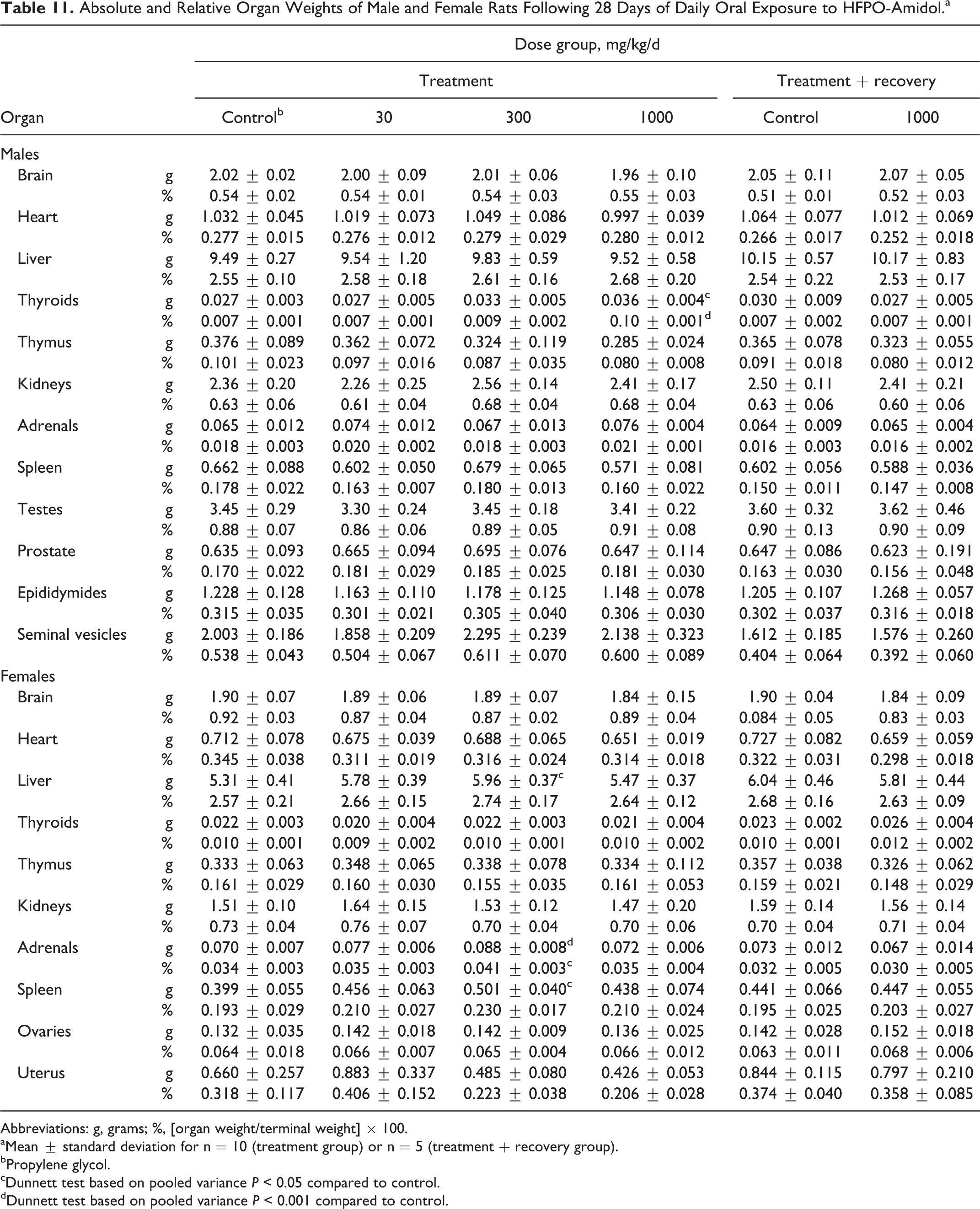
Abbreviations: g, grams; %, [organ weight/terminal weight] × 100.
aMean ± standard deviation for n = 10 (treatment group) or n = 5 (treatment + recovery group).
bPropylene glycol.
cDunnett test based on pooled variance P < 0.05 compared to control.
dDunnett test based on pooled variance P < 0.001 compared to control.
Histopathology
There were no treatment-related microscopic findings. All microscopic findings recorded were considered to be within the normal range of background pathology encountered in Wistar Han rats of this age and strain (data not shown). No abnormalities were seen in the reproductive organs of 2 nonpregnant females at 30 mg/kg/d or the paired male rats that could account for their infertility. The assessment of the integrity of the spermatogenetic cycle did not provide any evidence of treatment-related impaired spermatogenesis.
Reproductive Toxicity Assessment
No statistically significant or toxicologically relevant effects on reproductive parameters were noted. Mating, fertility, and conception indices, precoital time, and number of corpora lutea and implantation sites were unaffected by treatment (Table 12). For 1 female at 30 mg/kg/d, there was no evidence of mating, and this female along with another female of the same dose group did not become pregnant. This resulted in slightly lower mating, fertility, and conception indices at this dose level. Given that these lower indices occurred in the absence of a dose-related trend, this response was not considered to be treatment related.
Reproduction Observations for Rats Treated With HFPO-Amidol.a

aMean ± standard deviation.
bPropylene glycol.
cExcludes corpora lutea counts for non-pregnant females.
dMating index = (number of females mated/number of females paired) × 100.
eFertility index = (number of pregnant females/number of females paired) × 100.
fConception index = (number of pregnant females/number of females mated) × 100.
No toxicologically relevant effects on parturition or early postnatal pup development were observed (Table 13; see also Online Supplemental Information for historical control data). The gestation index was 100% for all groups. All animals displayed normal parturition and maternal care. The mean number of live pups per litter on LD 1 was statistically significantly lower at 30 mg/kg/d. This difference was considered to be not toxicologically relevant, since there was no dose-related trend and the mean remained well within the range considered normal for rats of this age and strain (see Online Supplemental Information for historical control data). There were no statistically significant differences in the number of dead pups on LD 1, postnatal loss, viability index, sex ratio, or body weights.
Parturition and Litter Observations for Rats Treated With HFPO-Amidol.a
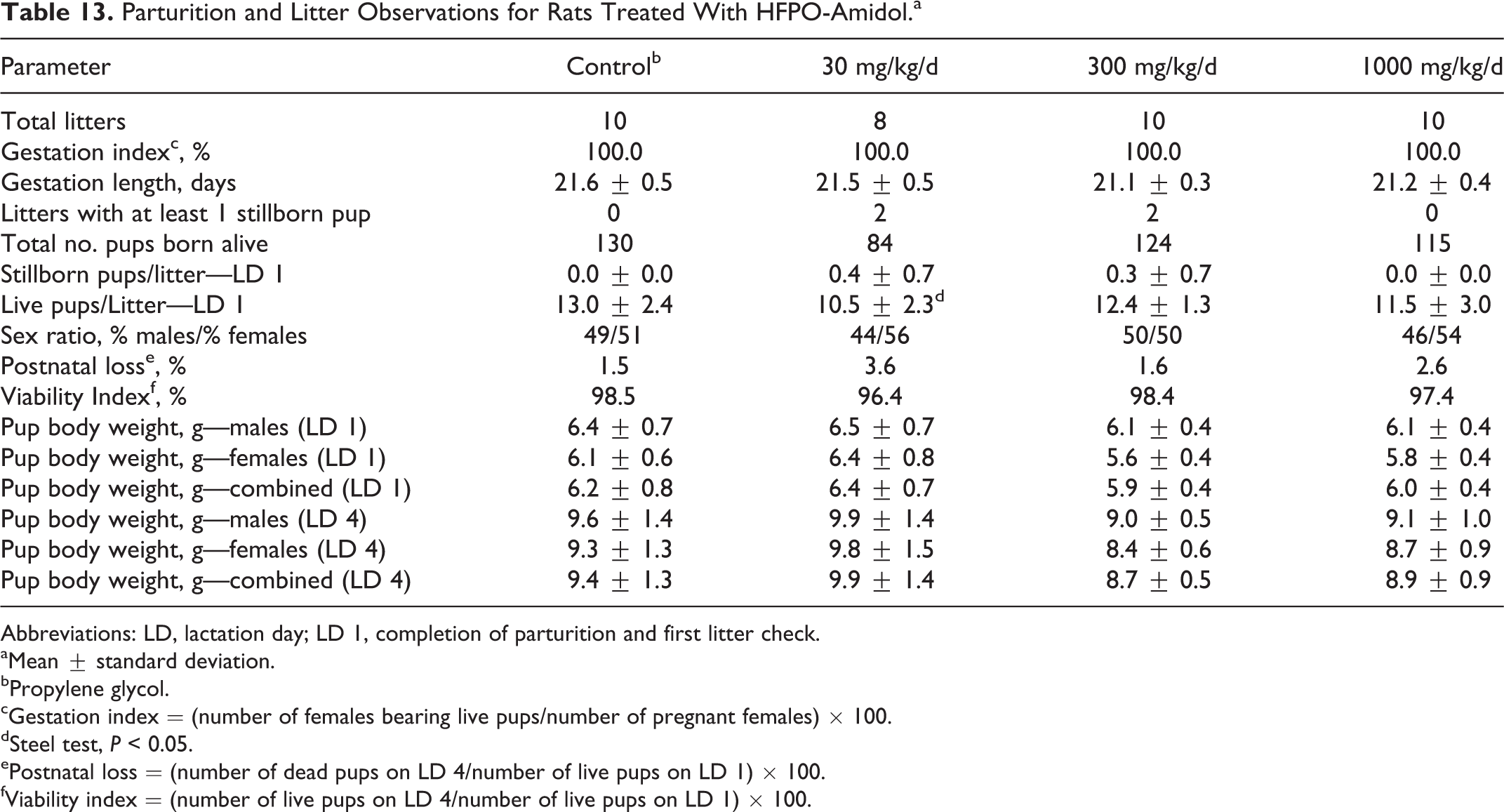
Abbreviations: LD, lactation day; LD 1, completion of parturition and first litter check.
aMean ± standard deviation.
bPropylene glycol.
cGestation index = (number of females bearing live pups/number of pregnant females) × 100.
dSteel test, P < 0.05.
ePostnatal loss = (number of dead pups on LD 4/number of live pups on LD 1) × 100.
fViability index = (number of live pups on LD 4/number of live pups on LD 1) × 100.
Two pups of the control group, 6 pups at 30 mg/kg/d, 5 pups at 300 mg/kg/d, and 3 pups at 1000 mg/kg/d were found dead or missing between LD 1 and LD 5. Pups missing were most likely cannibalized. Incidental macroscopic findings of pups that were found dead included absence of milk in the stomach, a missing tail tip, and a stage of beginning autolysis. The mortality incidence and the nature and incidence of macroscopic findings did not show a dose-related trend and were considered to be within the normal range for pups of this age.
Clinical signs noted in pups (both surviving pups and pups found dead) included absence of milk in the stomach, scabs, a wound on the hind leg, and a missing tail tip. The nature and incidence of these clinical signs were considered to be within the normal range for pups of this age.
Discussion
HFPO-Amidol was developed as an alternative to longer chain perfluorinated compounds. The purpose of this study was to provide information on the repeated-dose toxicity of HFPO-Amidol in male Wistar Han rats exposed for 2 weeks prior to mating, during mating, and up to termination (for 30 days) and in female Wistar Han rats exposed for 2 weeks prior to mating, during mating, during postcoitum, and at least 4 days of lactation (for 42-53 days). The study design incorporated evaluation of reproductive and developmental end points including gonadal function, mating behavior, conception, and parturition to provide screening information on possible effects of HFPO-Amidol on male and female reproductive performance.
Exposure to HFPO-Amidol had no treatment-related effect on mortality, clinical observations, food consumption, most functional parameters (hearing ability, pupillary reflex, static righting reflex, and grip strength), organ weights, gross findings, or histopathology in parental generation male or female Wistar Han rats at doses up to 1000 mg/kg/d. No treatment-related effects were noted on reproductive or developmental toxicity parameters under the conditions of this study.
The statistically significant changes in body weight gain of 1000 mg/kg/d females during the postcoitum period may be related to treatment, but since these changes were slight in nature, occurred in the absence of a clear dose-related trend, and remained within 10% of the control means throughout the treatment period, they were considered not to be toxicologically relevant or adverse. Body weight changes of 300 mg/kg/d females during the mating, postcoitum, and lactation phases were considered not treatment related, since body weight gain of these females in each of these phases remained similar to control levels.
The lower food consumption of reproductive group females at 300 and 1000 mg/kg/d during the postcoitum and lactation period was not considered adverse and to be unrelated to treatment with the test substance, since these variations occurred in the absence of a clear dose-related trend, were inconsistently seen over time, and were generally absent after allowance for body weights.
Statistically significant increases in total movements and ambulations were observed in males and females dosed with 1000 mg/kg/d. The response in 1000 mg/kg/d females was not considered treatment related because any statistically significant changes in both total counts and counts per interval did not show a dose-related trend. Nor were concurrent clinical signs, changes in other functional observation tests, or changes in the histopathology of the nervous system observed in the 1000 mg/kg/d females.
The statistically significant increase in motor activity in 1000 mg/kg/d males was present within the first 10 to 15 minutes of the 60-minute measurement period and was suggestive of a temporary hyperreactivity to a new environment/stimulus. At the end of the recovery phase of the study, a statistically significant increase in motor activity (both ambulations and total movements) remained present in these males during the first 5 to 10 minutes. The difference in mean counts between control and high dose males was lower at the end of the recovery period compared to the end of the treatment period (by a factor of approximately 1.5 vs approximately 3 at the end of recovery and treatment, respectively). This may indicate that partial recovery occurred. The toxicological relevance of these transient changes in motor activity is unclear. Clinical signs did not show any evidence of increased/persistent locomotor activity during treatment, and other functional observation tests and histopathological examination of neuronal tissues did not show any abnormalities. However, because the temporarily increased motor activity in 1000 mg/kg/d males essentially persisted in the recovery phase, the possibility that the observation represents an adverse neurological effect could not be fully excluded.
The parental systemic NOAEL in this study is 300 mg/kg/d, based on statistically significant increases in the number of total movements and total ambulations in males at 1000 mg/kg/d. No indications of reproductive or developmental toxicity were observed under the conditions of this study. The reproductive and developmental NOAELs are thus 1000 mg/kg/d, the highest dose tested.
Footnotes
Author Contribution
Moilanen, L contributed to conception and design, analysis, and interpretation and drafted the manuscript. Bagley, B contributed to design and interpretation and drafted the manuscript; Hakes, C contributed to acquisition, drafted the manuscript, and critically revised the manuscript. Hope, E contributed to interpretation, drafted the manuscript, and critically the revised manuscript; Reynolds, J contributed to interpretation and drafted the manuscript. Van Otterdijk, F contributed to design, acquisition, analysis, and interpretation and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy
Authors’ Note
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Authors Lori H. Moilanen, Daniel C. Hakes, Bradford D. Bagley, Esther F. Hope, and Jill E. Reynolds are current or former employees of 3M, a manufacturer of HFPO-Amidol. Francois van Otterdijk is an employee of WIL Research Europe B.V., the laboratory contracted by 3M to perform the study described in this publication.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All aspects of the study described in this publication were funded by 3M.
