Abstract
Gas oils, used to manufacture diesel fuel and residential heating oil, are complex hydrocarbon substances with carbon numbers of C9-C30 and boiling ranges of approximately 150°C to 450°C. Target organ (liver enlargement, reduced thymus weights, and reductions in hematological parameters) and developmental (reduced fetal viability, increased resorption frequency, and reduced fetal weights) effects are associated with aromatic constituents present in some gas oils. Two types of gas oils were tested for repeated-dose and developmental toxicity following repeated dermal administration. A blend of commercial diesel fuels containing 26% aromatics, primarily single-ring compounds, did not cause either target organ or developmental effects at levels up to 600 mg/kg/d. “Cracked” gas oils containing higher levels of aromatic constituents were also tested. Because of limited sample availability, 2 cracked gas oil samples were tested, one for systemic effects and the other for developmental toxicity. The sample tested in the repeated-dose toxicity study (81% aromatics including approximately 10% 3-ring compounds) produced increased liver weights, reduced thymus weights, and reductions in hematological parameters. The overall no observed adverse effect level (NOAEL) was 100 mg/kg/d. The sample tested for developmental toxicity (65% aromatics including approximately 5% 3-ring compounds) resulted in significant reductions in fetal survival, significant increases in resorption frequency, and significant reductions in fetal weights with an overall NOAEL of 100 mg/kg/d. In summary, gas oils may or may not cause target organ and/or developmental effects depending on the levels and types of aromatic constituents that they contain.
Keywords
Introduction
The US Environmental Protection Agency (EPA) announced a voluntary chemical data collection effort in 1998 called the High Production Volume (HPV) Challenge Program. 1 The HPV chemicals are those produced or imported to the United States in aggregate quantities of at least 1 million pounds per year. Approximately 400 petroleum substances, sponsored in the EPA’s Challenge Program by companies belonging to the Petroleum HPV Testing Group (PHPVTG), were organized into 13 categories to facilitate data sharing and to avoid redundant testing. The categories include crude oil, gases, gasoline, kerosene/jet fuel, gas oils, heavy fuel oils, lubricating oils, waxes, aromatic extracts, asphalts, grease thickeners, petroleum coke, and wastes. This article reports an investigation into the toxicological hazards of gas oils, a category of substances that includes diesel fuel, residential heating oils, and the refinery streams that are used to manufacture these types of end products.
Gas oils and their blending streams are complex petroleum-derived substances with complex and variable compositions, best described as substances of unknown or variable composition (UVCBs). 2 The constituents are hydrocarbons (normal paraffins, iso [branched] paraffins, naphthenes [cycloparaffins], olefins, and aromatics) with carbon numbers ranging from approximately C9 to C30 and distilling within a range of approximately 150°C to 450°C. The gas oil blending streams are produced either by atmospheric or vacuum distillation (straight-run gas oils) or by secondary processing of these or other petroleum-derived substances. The secondary processing often includes “cracking,” a generic term for processes that convert higher to lower molecular weight material, producing streams that may have higher aromatic and/or olefinic content than those produced by atmospheric distillation. The compositions of the different refinery streams vary depending on the feedstocks and the processing steps utilized; but ultimately the gas oil streams are blended to make end products such as diesel fuel and heating oil. Although there are no compositional specifications for the individual gas oil blending streams, the specifications for diesel fuel and heating oil include limits on boiling range (the 90% boiling point [T90] is less than 338°C) and total sulfur content (15 ppm) that effectively limit the types of aromatic constituents that may be present in the end products to those with 1 to 3 aromatic rings. 3
From a toxicological perspective, there are some generic hazards of gas oils including the potential for chemical pneumonitis if aspirated and the potential to cause severe skin irritation following prolonged and repeated dermal contact. There are also some additional toxicological properties that are related to the specific compositions of the individual substances. In particular, aromatic constituents are associated with systemic and developmental toxicity, and gas oils containing aromatics with 3 or more rings may also be mutagenic and carcinogenic. Accordingly, the potential for toxicity of the substances in the gas oil category is related to both boiling range and aromatic content.
In the initial studies to provide base toxicological data, blended diesel and home heating fuels with differing aromatic levels were not toxic in acute oral and dermal studies and did not produce eye irritation or allergic contact sensitization but did produce moderate to severe skin irritation, particularly when applied repeatedly at high doses. 3,4 Repeated dermal application studies in rabbits provided evidence of severe dermal irritation, but no clear evidence of systemic toxicity. 4 Results of dermal carcinogenesis studies showed that middle distillate streams (ie, the precursors for kerosene and gas oils) could produce skin tumors when repeatedly applied to mouse skin, 5 and these observations led to further investigations to better understand the mechanisms. Initial studies showed that the carcinogenic activity of these streams was not directly related to aromatic content. 5 –7 It was later shown that gas oils that contained aromatic molecules with 3 or more rings were mutagenic when tested in optimized Salmonella assays and could initiate skin tumors. 8 In contrast, the aliphatic constituents of gas oils were not mutagenic- or carcinogenic-initiating agents, but some had tumor-promoting properties. 9 –11 It was shown that the aliphatic gas oils could also produce skin cancer in mice in studies involving repeated dermal application, but the tumors seemed to have been due to the tumor-promoting properties of the aliphatic constituents and a consequence of repeated dermal irritation. 10,11
The potential for the more highly high-boiling petroleum substances (ie, with final boiling points >650°F/344°C) to produce target organ and/or developmental toxicity was first studied systematically by Feuston et al, who reported that target organ and developmental effects of dermal exposure to these high-boiling petroleum substances were correlated with the presence of polycyclic aromatic compounds (PACs; note 1) with 3 to 7 aromatic rings. 12 These data and a subsequent evaluation that included additional, previously unpublished data established that the outcomes associated with these aromatic constituents included increased liver weights, reduced thymus weights, alterations in serum chemistry and hematology parameters, increased resorption frequency, and reduced fetal body weight. The aliphatic constituents (normal paraffins, iso [branched] paraffins, and cycloparaffins) of these substances are dermal irritants as described previously but do not contribute substantially to the other toxicological properties of gas oils. Published information on 4 gas oil streams (light catalytically cracked gas oil [LCCO], light coker gas oil [LCGO], heavy atmospheric gas oil [HAGO], and vacuum tower overhead [VTO]) with differing levels and types of aromatic constituents identified a lowest effect level (LOEL) for target organ effects of 125 mg/kg/d based on the increased liver weight. The LOEL in the developmental toxicity studies was 500 mg/kg/d based on the increased frequencies of resorption.
In the current study, samples of gas oils were tested in repeated dermal application studies to assess the potential for repeated-dose and developmental toxicity. A specific objective was to further characterize the relationship between toxicity and the levels of specific types of aromatic constituents. One sample, a blend of commercial diesel fuels (ultralow sulfur diesel [ULSD]), was selected in part because of its wide use in commerce, but also because although diesel fuels contain approximately 20% aromatics, these are almost entirely molecules with 1 or 2 rings. This sample provided a comparison to other, previously tested gas oils that contained higher levels of total aromatics with higher fractions of molecules containing more than 2 aromatic rings. Based on the previous data, ULSD was not expected to produce systemic or developmental toxicity. Two samples of LCCOs containing higher levels of molecules with more than 2 aromatic rings were also tested, one for repeated-dose toxicity and the other for developmental toxicity. It was expected that, because the LCCOs contained higher levels of aromatic constituents with ≥3 rings than ULSD, they would produce more profound effects in the repeated-dose and developmental toxicity studies than the ULSD. It was anticipated that the data from the diesel fuel and LCCO studies could be used to define the boundary conditions by which to assess the potential hazards of the other substances in the gas oil category. It was also expected that the additional toxicological information in combination with compositional information could be used to further refine models that could be used to predict the outcomes of repeated-dose and developmental toxicity tests of petroleum substances with final boiling points >650°F/344°C. 13 –15
Materials and Methods
Materials
Ultralow sulfur diesel oil (Chemical Abstract Services [CAS] registry number 68334-30-5) was a blend of 7 diesel fuels obtained from commercial vendors. It contained approximately 26.4% total aromatics (21.8% monoaromatics and 4.6% aromatics with more than 1 ring) by supercritical fluid chromatography (ASTM D5186) with the remaining constituents (approximately 73%) being aliphatics including n-, iso-, and cycloparaffins. The boiling range was approximately 170°C to 325°C. The dimethyl sulfoxide (DMSO)-extractable material included 2.2% 2-ring compounds and 0.6% 3-ring compounds using the methodology developed by Roy et al 16 (summarized recently by Gray et al 17 ). No DMSO-extractable aromatics with more than 3 rings were detected.
The 2 other test samples were LCCOs (CAS number 64741-59-9). The sample used in the systemic toxicity test had a boiling range of approximately 189°C to 350°C and contained 80.9% total aromatics (35.3% monoaromatics and 45.7% aromatics with more than 1 ring) by supercritical fluid chromatography (ASTM D5186). The DMSO-extractable material included 19.5% 2-ring compounds and 9.8% 3-ring compounds. The sample used in the developmental toxicity test had a boiling range of 184°C to 335°C and contained 65% total aromatics (28.9% monoaromatics and 36.1% aromatics with more than 1 ring) by supercritical fluid chromatography (ASTM D5186). The DMSO-extractable material included 16.7% 2-ring compounds and 4.8% 3-ring compounds. Based on the results of the DMSO extraction procedure, neither of the LCCO samples contained aromatic molecules with more than 3 rings.
The gas oil test samples were suspended in a US Pharmacopeia grade mineral oil (Spectrum Chemical Manufacturing Company, New Brunswick, New Jersey), a type of lubricant base oil that is severely refined to reduce the concentrations of aromatics to very low levels. There was 0.0% DMSO-extractable material in the vehicle.
Methods
Guidelines
The repeated-dose studies followed the guidelines for Organization for Economic Cooperation and Development (OECD) 411 and were also compliant with the US EPA guidelines (870.3250). The developmental toxicity studies followed the guidelines for OECD 414 (Prenatal Developmental Toxicity Study) and were also compliant with the corresponding EPA guidelines (Prenatal Developmental Toxicity Test Guidelines, OCSPP 870.3700).
Animal husbandry
For the repeated-dose tests, Sprague-Dawley rats [CrL: CD (SD)] were obtained from Charles River Laboratories, Inc (Raleigh, North Carolina) and were 45 days of age at the time of receipt. For the developmental toxicity tests, sexually mature virgin female Sprague-Dawley rats [Crl: CD (SD)], approximately 79 days old at receipt, were obtained from Charles River Laboratories (Portage, Michigan). The animals were examined for good health, weighed, and uniquely identified by ear tag. They were then held for 13 days during which they were acclimated to Elizabethan-style collars. The rats were individually housed in stainless steel wire mesh cages with temperatures maintained at 22°C ± 3°C and humidity at 50% ± 20%, and fluorescent lighting on a 12-hour on/off schedule. The rats were given ad libitum access to food (Certified Rodent LabDiet 5002; PMI Nutrition International, LLC, St. Louis, Missouri) and water. Other details of animal husbandry were in accordance with the guidelines of the National Research Council. 18
On the day prior to first treatment, the dermal surface of the back and sides of each rat was clipped to remove the hair and to allow the dose to be applied to an area of approximately 10% of the body surface. The test material was applied in dermal doses of 1.5 mL/kg. After 6 hours, the application areas were patted with paper towels to remove any remaining liquid. In the repeated-dose study, after each week (5 consecutive days of dermal application), residual test substance was removed using warm water and a mild soap solution. The animals all wore Elizabethan-style collars on dosing days of the study to prevent test material ingestion. The sham control animals treated in the same way as other animals on test but were not exposed to test material or vehicle. In the repeated-dose study, the rats were treated 5 days/week for 90 days. In the developmental toxicity study, the rats were treated daily from gestational day (GD) 0 to GD 19 and then euthanized on GD 20.
Repeated-dose toxicity studies
Four days prior to the initiation of dosing, the rats were weighed and examined. The rats that were judged to be in good health were assigned to study groups using a computerized randomization procedure based on the body weight stratification in a block design as shown in Table 1. Note that the doses were based on the preliminary range finding studies that were conducted to assess the potential for dermal irritation. The highest doses selected were expected to produce some degree of dermal irritation but not levels that were anticipated to cause excessive discomfort to the animals or to otherwise compromise the technical objectives of the studies.
Study Design for the Repeated-Dose Toxicity Tests of Gas Oil Samples.
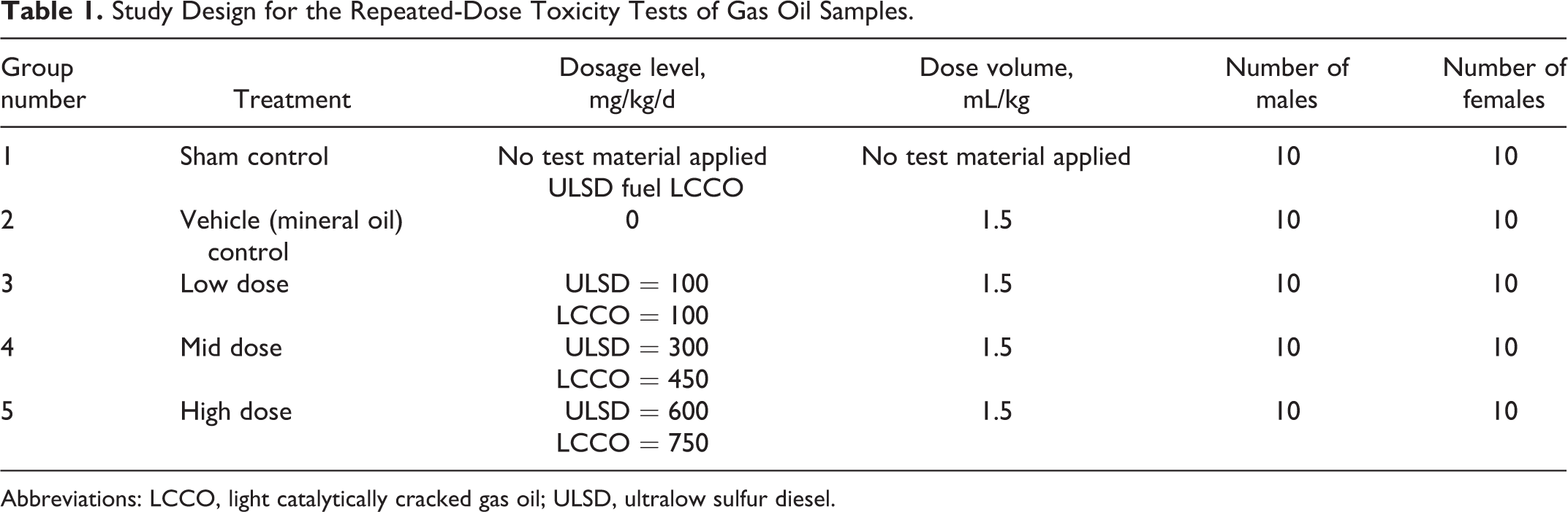
Abbreviations: LCCO, light catalytically cracked gas oil; ULSD, ultralow sulfur diesel.
All animals were checked twice daily for general condition. Detailed physical examinations, body weight measurements, and food consumption measurements were all done on a weekly basis. The sites of dose application were examined for dermal effects that were scored following the method of Draize et al. 19
Samples for clinical pathology (hematology, coagulation, and serum chemistry) were taken from all surviving animals. The animals were fasted overnight prior to blood collection. The animals were euthanized by inhalation of isoflurane, and the blood samples were taken from the vena cava as part of the gross necropsy. Parameters evaluated for hematology and coagulation included total leukocyte (WBC) count, erythrocyte (RBC) count, hemoglobin (HGB), hematocrit (HCT), mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, platelet count, prothrombin time, activated partial thromboplastin time, reticulocyte count (percentage, absolute, and differential) WBC count (percentage and absolute) neutrophil, lymphocyte, monocyte, eosinophil, basophil, large unstained cell, red cell distribution width, HGB distribution width, platelet estimate (if based on a manual differential assessment), and RBC morphology. The serum chemistry measurements included albumin, total protein, globulin (by calculation), albumin–globulin (A/G) ratio, total bilirubin, urea nitrogen, creatinine, alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase, γ-glutamyltransferase, glucose, total cholesterol, calcium, chloride, phosphorus, potassium, sodium, triglycerides, and sorbitol dehydrogenase.
After sacrifice, organs taken for weight and/or histological examination included adrenals, aorta, bone with marrow (sternum and femur with joint), bone marrow smear (sternum), brain (3 sections), cervix, epididymides, eyes with optic nerve, gastrointestinal tract (esophagus, stomach, duodenum, jejunum, ileum, cecum, colon, and rectum), heart, kidneys, lacrimal gland, liver (sections of 2 lobes), lungs (including bronchi), lymph nodes (axillary, mandibular, and mesenteric), ovaries with oviducts, pancreas, peripheral nerve (sciatic), pituitary, prostate, salivary glands, seminal vesicles, skeletal muscle, skin (with mammary gland), skin (treated and untreated skin from areas of dose application), spinal cord (cervical, thoracic, and lumbar), spleen, testes, thymus, thyroid (with parathyroid), trachea, urinary bladder, uterus, vagina, and gross lesions. Weights were taken for the following organs: adrenals, brain, epididymides, heart, kidneys, liver, ovaries with oviducts, pituitary, prostate, spleen, testes, thymus, thyroid with parathyroid, and uterus.
Developmental toxicity studies
At the end of the acclimation period, all female rats judged to be in good health were weighed and then cohoused with untreated, sexually mature male rats. Once successful mating was confirmed by the presence of a vaginal copulatory plug or the presence of sperm in the vaginal lavage, the mated females were assigned to treatment groups by a WTDMS computer program that randomized the rats based on stratification of the GD 0 body weights in a block design. Each study consisted of 5 groups, 2 controls (sham and vehicle) and 3 treatment groups (100, 300, and 600 mg/kg/d in the ULSD study and 100, 450, and 750 mg/kg/d in the LCCO study). The doses were chosen based on the preliminary 14-day skin irritation studies that were used to determine the maximum levels of the test substances that could be repeatedly applied without causing excessive skin irritation. Each group contained 25 mated females.
All animals were checked twice daily, and all observations were recorded. The application sites of all animals were evaluated on a daily basis, prior to dose administration for signs of dermal irritation and graded for dermal effects following the scoring system of Draize et al. 19 Body weights were taken on GD 0, 3, 6, 9, 12, 15, 18, and 20. Food consumption was recorded on the same days that the body weights were measured.
Rats were euthanized by carbon dioxide inhalation and subjected to a gross examination, which included the cranial, thoracic, abdominal, and pelvic cavities. Liver, brain, and thymus were weighed. Tissues taken for histological evaluation included sections of treated and untreated skin, liver, brain, thymus, and any gross lesions. The uterus and ovaries were then exposed and excised, and the numbers of corpora lutea were recorded. Each uterus was weighed and then opened, and the number and location of all fetuses, early and late resorptions, and the total number of implantation sites were recorded. Uteri with no macroscopic evidence of implantation were opened and subsequently placed in 10% ammonium sulfide solution for detection of early implantation loss. 20
Each viable fetus was examined externally, individually weighed, sexed, and then, after euthanasia, was examined for developmental effects. The detailed external examination included an assessment of the eyes, palate, and external orifices. To the extent possible, nonviable fetuses were examined to assess weight, crown-rump length, and sex. The internal examinations of the viable fetuses followed the methods of Stuckhardt and Poppe 21 and included fresh dissection to assess the heart and major blood vessels. The sex of each fetus was confirmed by internal examination. Fetal kidneys were examined and graded for renal papillae development. 22 Heads from approximately one-half of the fetuses in each litter were placed in Bouin fixative for subsequent soft tissue examination by the Wilson sectioning technique. 23 The heads from the remaining one-half of the fetuses were examined by mid-coronal slice. All carcasses were eviscerated and fixed in 100% ethyl alcohol.
Following fixation, each fetus was macerated in potassium hydroxide and stained with Alizarin Red S 24 and Alcian Blue. 25 Fetuses were then examined for skeletal malformations and developmental variations. External, visceral, and skeletal findings were recorded as either malformations or variations.
Statistical analysis
Parameters including body weight, body weight change, clinical pathology data (except γ-glutamyl transpeptidase [GGT] values), and food consumption data from the repeated-dose study as well as mean maternal body weights, organ weights, gravid uterine weights, numbers of corpora lutea, implantation sites, viable fetuses, and fetal body weights (separately by sex and combined) from the developmental toxicity study were evaluated using a parametric analysis of variance (ANOVA) to determine intergroup differences. 26 If the ANOVA revealed significant (P < 0.05) intergroup variance, the Dunnett test 27 or a 2-sample t test 26 was used, as appropriate, to compare the test substance-treated groups to the vehicle control group. The vehicle control group was evaluated using the Student t test 28 and compared to the sham control group. Mean litter proportions (percentage per litter) of prenatal data (viable and nonviable fetuses, early and late resorptions, total resorptions, pre- and postimplantation loss, and fetal sex distribution), GGT values, total malformations and developmental variations (external, visceral, skeletal, and combined), and each particular external, visceral and/or skeletal malformation, or variation were evaluated using the Kruskal-Wallis nonparametric ANOVA test to determine intergroup differences. 29 If the Levine test revealed significant (P < 0.05) intergroup differences in means, the Dunn Test 30 was used to compare the test substance-treated groups to the vehicle control group and the vehicle control group to the sham control group.
Results
The 90-Day Repeated Dermal Toxicity Study of ULSD Fuel
All the rats survived to scheduled termination with little evidence of treatment-related effects during the study. There were no effects on body weight or body weight gain (initial and terminal body weights given in Table 2) and no test substance-related clinical observations (data not shown). There was some evidence of dermal irritation, and the incidence of the findings increased with dose, but even in the highest dose group, the observations were largely limited to slight erythema, slight edema, and desquamation. The mild dermal effects were surprising as the highest dose had been selected with the expectation that relatively severe dermal irritation would be produced.
Summarized Results of Terminal Body Weights and Weights of Target Organs Following Repeated Dermal Application of Ultralow Sulfur Diesel Fuel.a,b
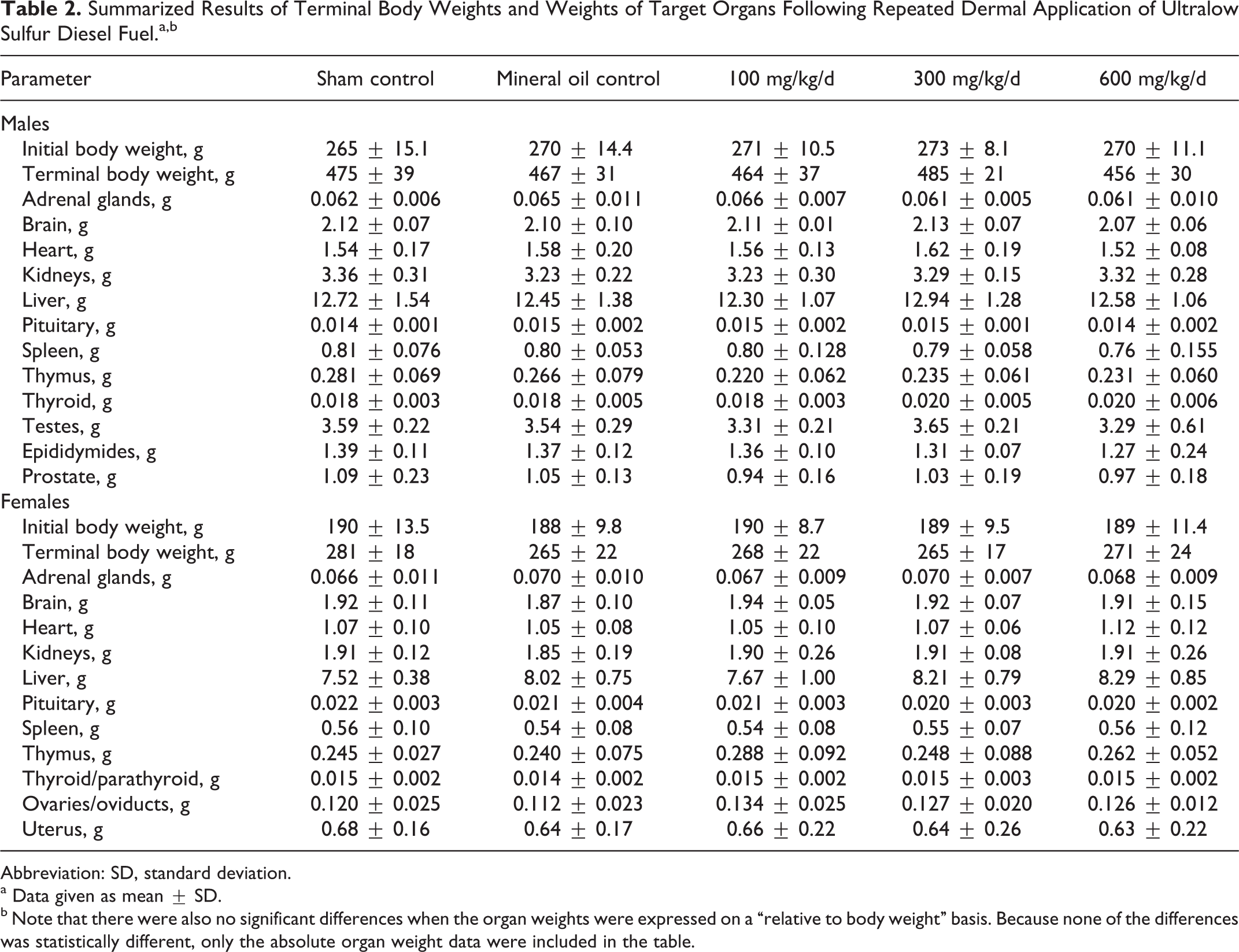
Abbreviation: SD, standard deviation.
a Data given as mean ± SD.
b Note that there were also no significant differences when the organ weights were expressed on a “relative to body weight” basis. Because none of the differences was statistically different, only the absolute organ weight data were included in the table.
None of the hematological parameters was affected by the treatment (Table 3). The only statistically significant finding in the serum chemistry measurements (data not shown) was a significant increase in the A/G ratio in the high-dose males. This finding was not considered to be toxicologically important because it was similar to the untreated control values and within normal historical control values. The A/G ratio results were:
Results of Hematological Investigations of Rats Treated for 90 Days by Dermal Application With Ultralow Sulfur Diesel Fuel.a
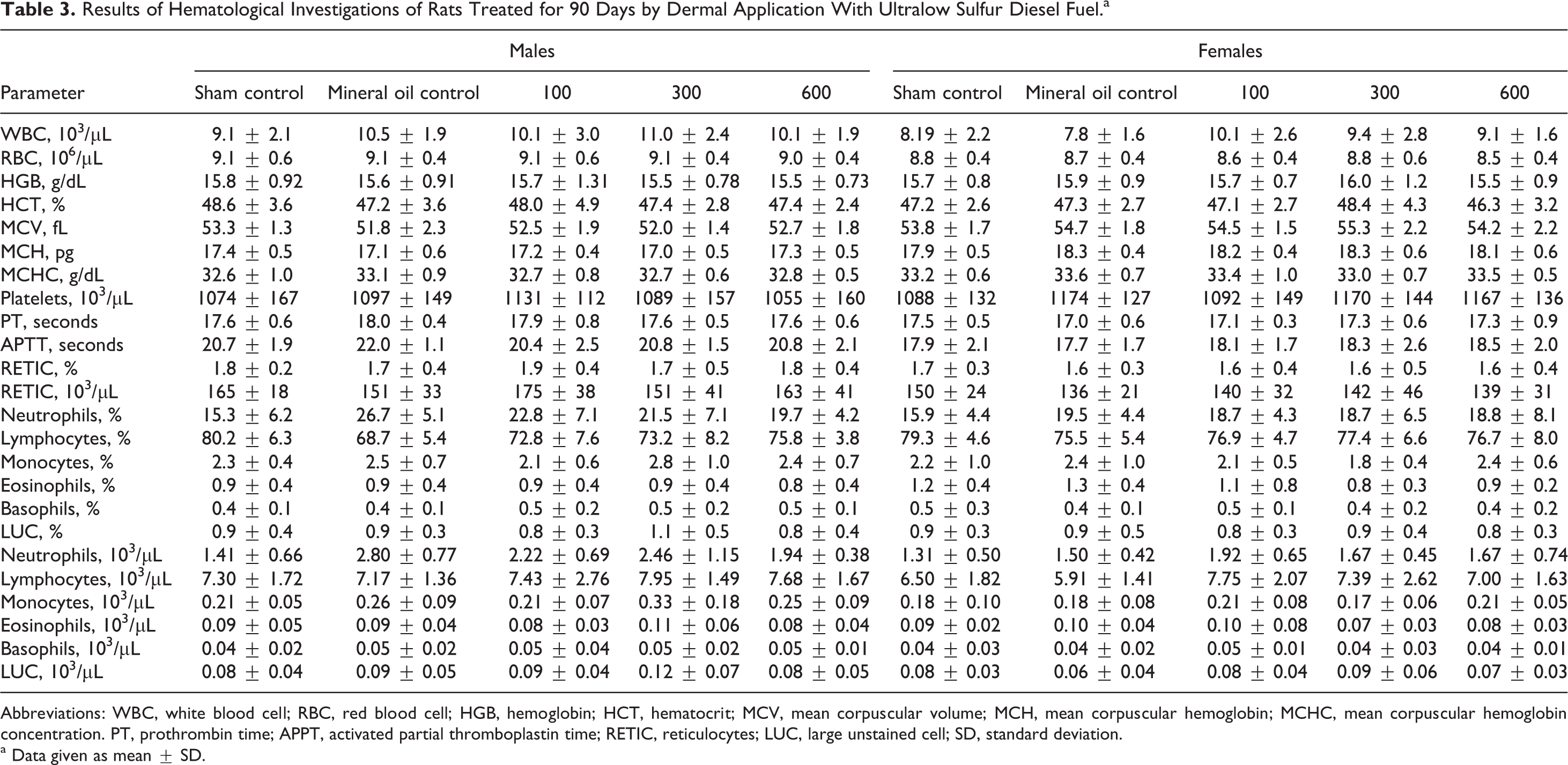
Abbreviations: WBC, white blood cell; RBC, red blood cell; HGB, hemoglobin; HCT, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration. PT, prothrombin time; APPT, activated partial thromboplastin time; RETIC, reticulocytes; LUC, large unstained cell; SD, standard deviation.
a Data given as mean ± SD.
Untreated control = 1.66 ± 0.16; mineral oil control = 1.39 ± 0.22; 100 mg/kg/d group = 1.49 ± 0.15; 300 mg/kg/d group = 1.48 ± 0.14; 600 mg/kg/d = 1.63 ± 0.19 (P < 0.05 by comparison to the mineral oil control).
There were no treatment-related gross observations, no differences in weights (absolute or relative to body or brain weight) of target organs (Table 2), and no pathological findings other than observations of dermal changes at the test material application sites were reported.
Prenatal Developmental Toxicity Study of ULSD Fuel
All of the rats survived to scheduled termination without evidence of test substance-related clinical findings. There were no dermal observations of note, and there were no differences in weight gain during the gestational period (Table 4). There were no gross findings indicative of treatment-related effects, and weights of the target organs were similar across the treatment groups (Table 5).
Gestational Body Weights of Rats Treated by Dermal Application With Ultralow Sulfur Diesel Fuel.a
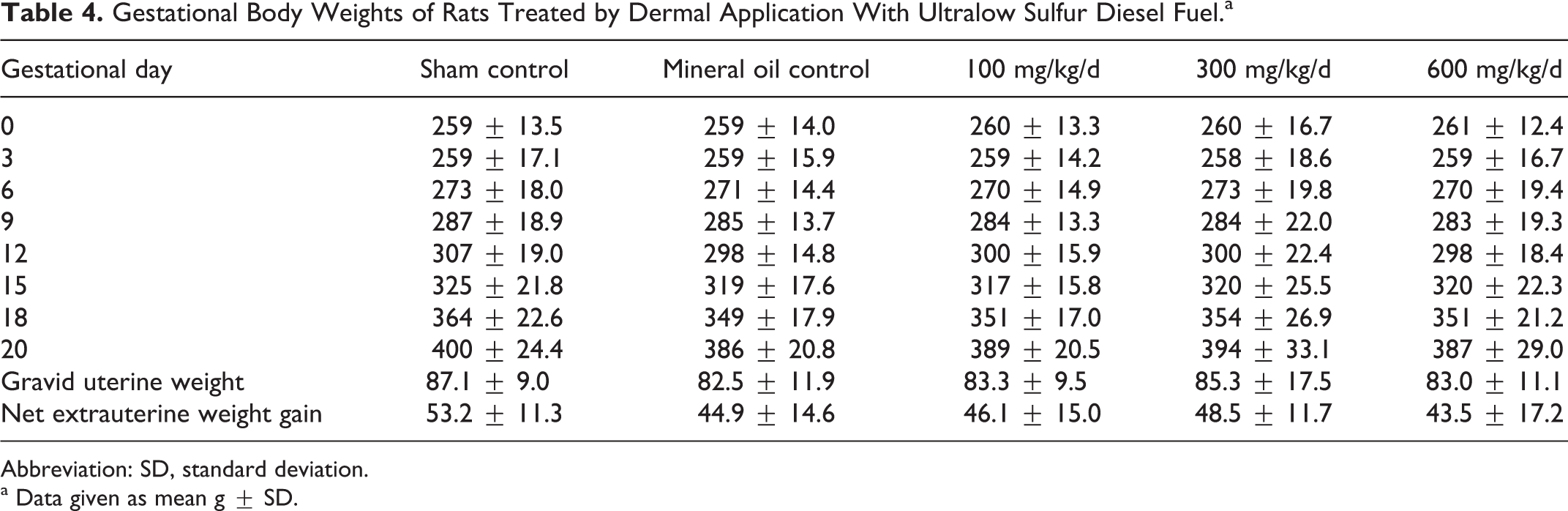
Abbreviation: SD, standard deviation.
a Data given as mean g ± SD.
Summary of Maternal Organ Weights Following Dermal Treatment With Ultralow Sulfur Diesel Fuel on Days 0 to 20 of Gestation.a
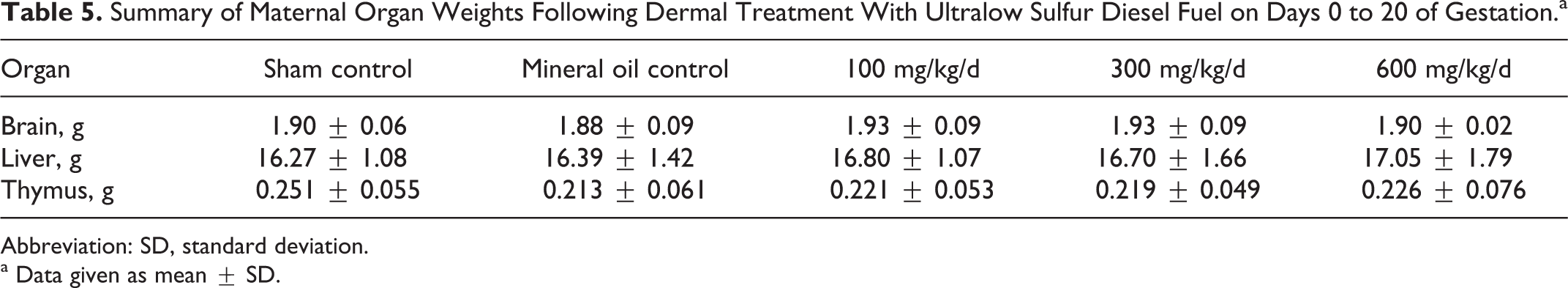
Abbreviation: SD, standard deviation.
a Data given as mean ± SD.
The uterine examinations did not reveal any treatment-related differences in the number of gravid dams, numbers of corpora lutea, implantation sites, viable fetuses, early or late resorptions, or fetal weights (Table 6). There were few malformations (Table 7) or developmental variants (data not shown) observed in this study, and no evidence that these variants were treatment related.
Results of Uterine Examinations Following Dermal Treatment of Dams by Ultralow Sulfur Diesel Fuel.
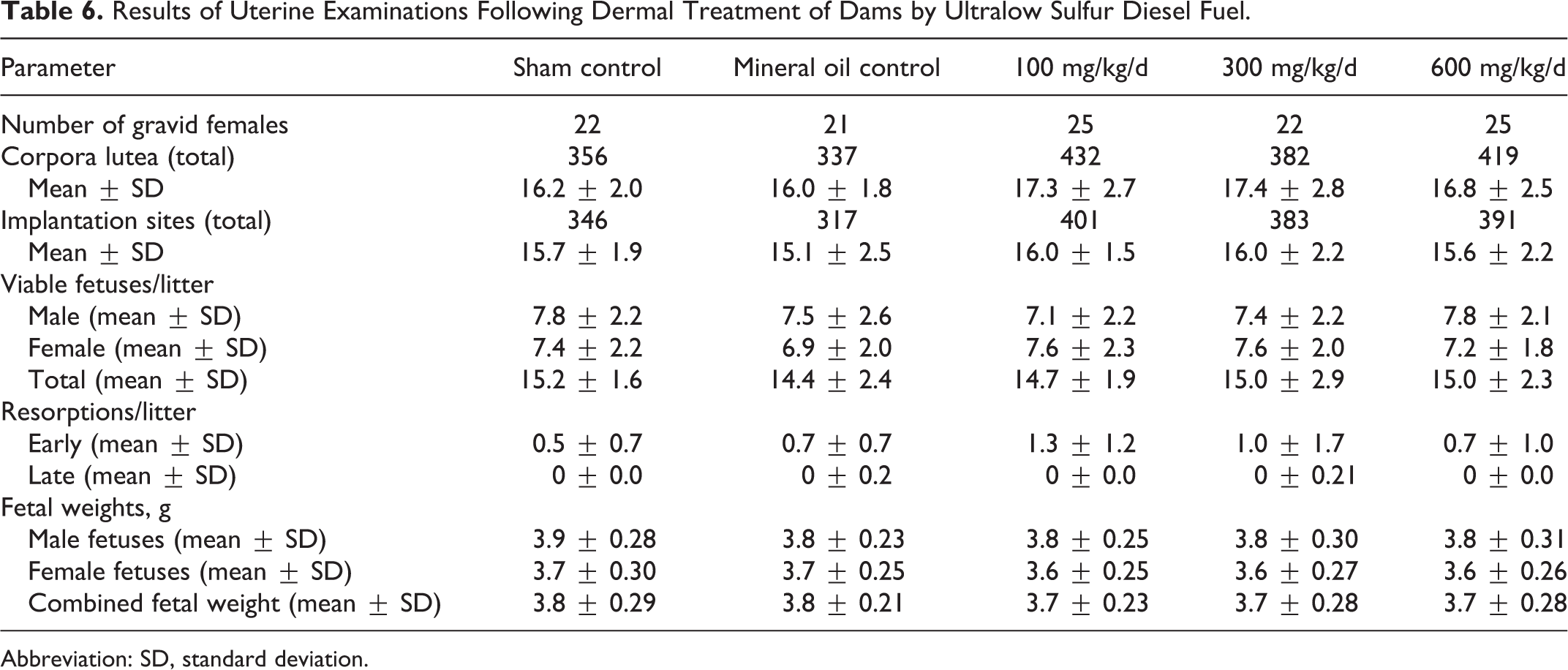
Abbreviation: SD, standard deviation.
Summary of the Results of the Examination of Fetuses From Dams Treated With Ultralow Sulfur Diesel Fuel by Dermal Administration.
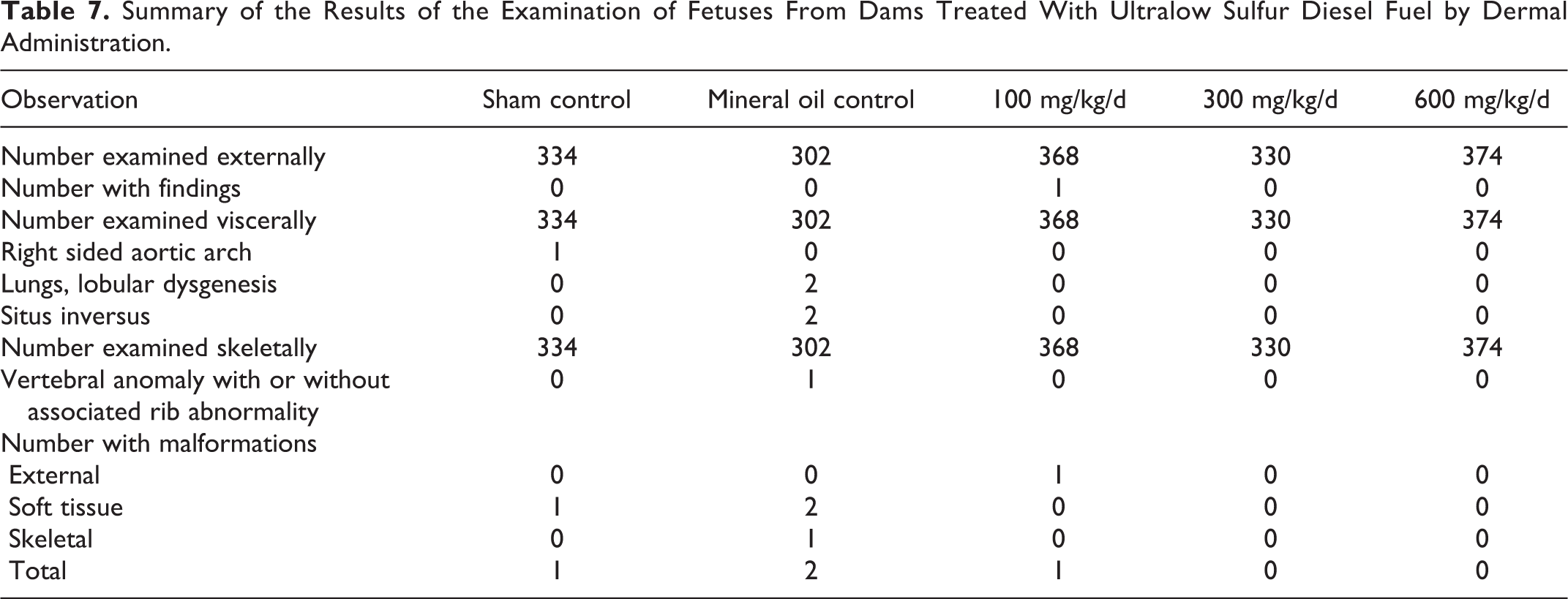
The 90-Day Repeated Dermal Toxicity Study of LCCO
Two rats died prior to scheduled termination; a male rat from the 750 mg/kg/d group was sacrificed in extremis due to a broken leg on study day 50 and a male rat from the 450 mg/kg/d group was found dead on study day 69. Neither of these deaths was considered to have been treatment related. There were no test material-related clinical observations during the study, and the dermal observations were limited to slight erythema, which was not considered to be treatment related as similar observations were made in control and treatment groups. Body weight gains were lower, and, by scheduled termination, the body weights of both males and females in the high-dose group were significantly below mineral oil control values (Table 8). The hematological investigations revealed statistically significant reductions in red blood cell counts, HGB content, and HCT (Table 9). When differences were found they were often significant in the 750 mg/kg/d groups, but in some cases, significant differences were also apparent at the 450 mg/kg/d treatment levels. Although statistical differences were found in other parameters, particular note was taken of the reductions in eosinophil counts that were significantly reduced (absolute and percentage) in both male and female rats in the 450 and 750 mg/kg/d groups. At 100 mg/kg/d, the absolute eosinophil count but not the percentage value was statistically significantly reduced in males compared to vehicle controls but not in females. No other effects were noted in any other hematology parameters for this dose group.
Summarized Results of Terminal Body Weights and Weights of Target Organs Following Repeated Dermal Application of Light Catalytically Cracked Gas Oil.a
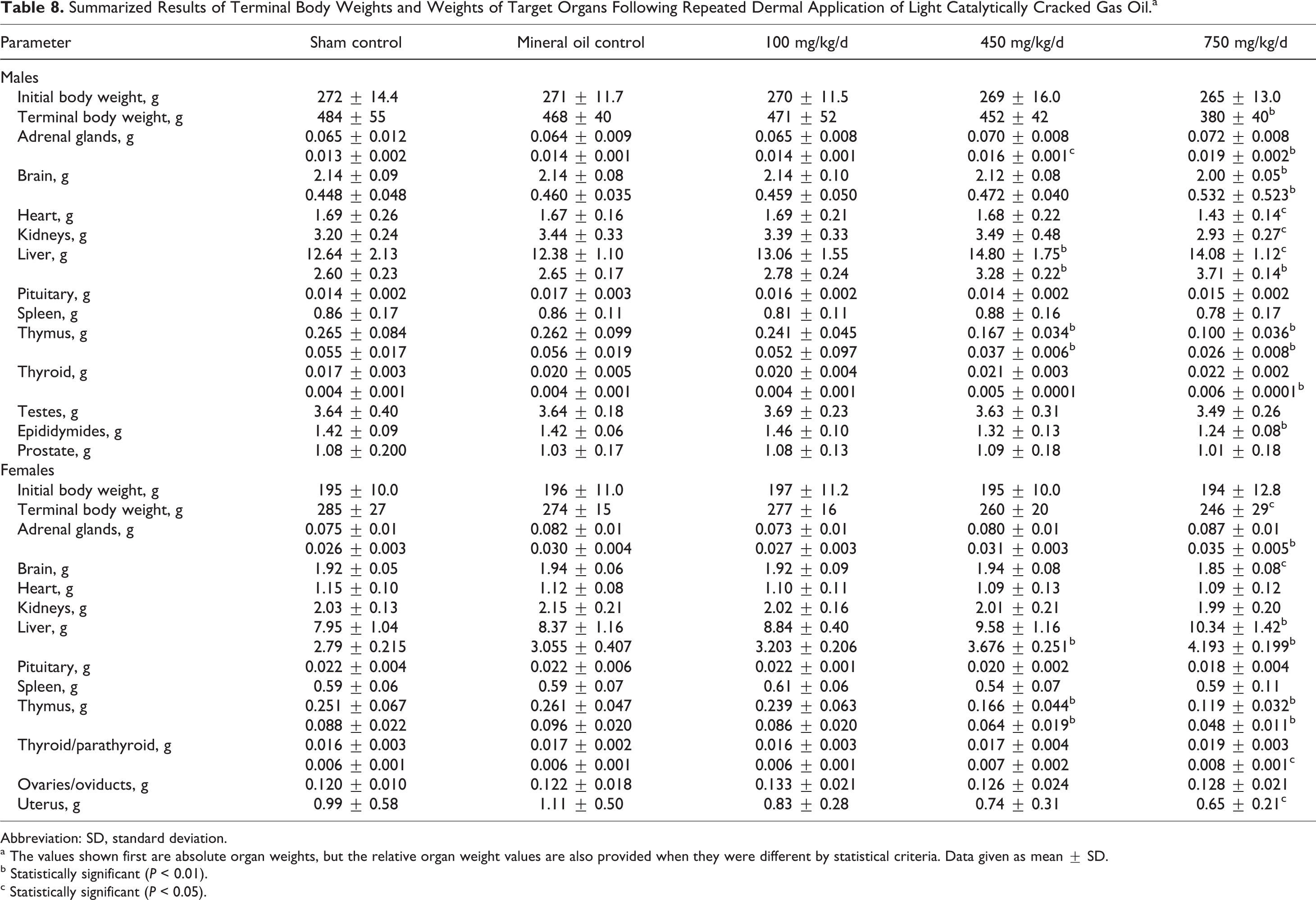
Abbreviation: SD, standard deviation.
a The values shown first are absolute organ weights, but the relative organ weight values are also provided when they were different by statistical criteria. Data given as mean ± SD.
b Statistically significant (P < 0.01).
c Statistically significant (P < 0.05).
Summarized Results of Hematological Parameters Following Repeated Dermal Application of Light Catalytically Cracked Gas Oil.a
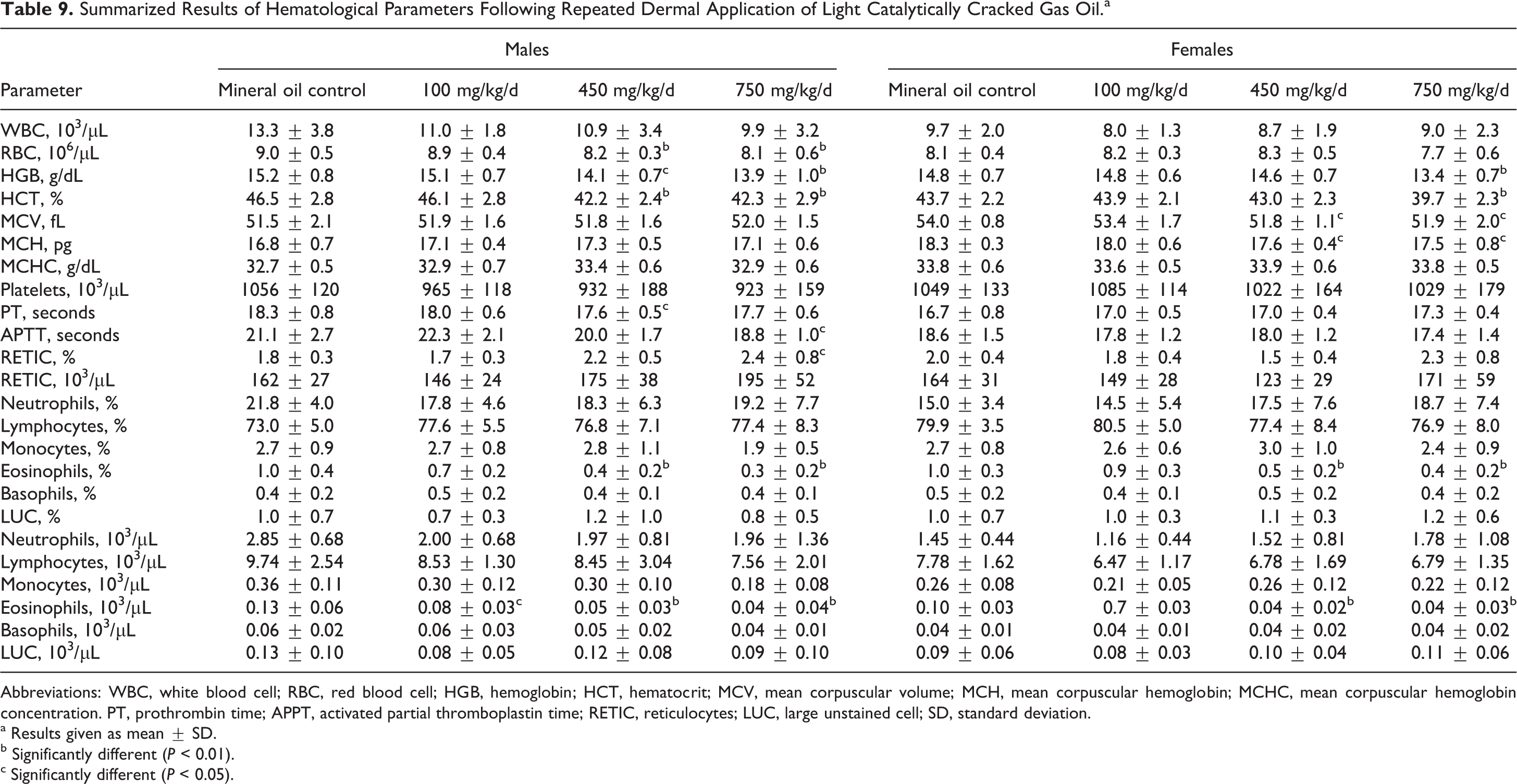
Abbreviations: WBC, white blood cell; RBC, red blood cell; HGB, hemoglobin; HCT, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration. PT, prothrombin time; APPT, activated partial thromboplastin time; RETIC, reticulocytes; LUC, large unstained cell; SD, standard deviation.
a Results given as mean ± SD.
b Significantly different (P < 0.01).
c Significantly different (P < 0.05).
The serum chemistry investigation revealed treatment-related increases in urea nitrogen, albumin, A/G ratio, cholesterol, GGT, and creatinine levels and lower globulin and chloride levels. However, although these differences were statistically significant, they were small and within the historical ranges of the laboratory. None of the differences in the clinical chemistry investigation was considered to have been toxicologically important (data not shown).
The gross examination revealed a number of differences in absolute and/or relative organ weights (Table 8) in the 450 and 750 mg/kg/d groups. In some cases, these were most likely a consequence of the reductions in body weight gain as few of these differences were significant when the comparisons were made on a body weight or brain weight basis. The increases in adrenal gland and liver weights were significant when the comparisons were made on a relative basis for adrenals and absolute and relative basis for liver and were considered to have been treatment related. Statistically significant reductions in absolute and relative thymus weights were also noted. No absolute or relative organ weight changes were seen in the 100 mg/kg/d group. None of the weight differences was correlated with histological changes. The only pathological findings were reports of epithelial hyperplasia and hyperkeratosis associated with chronic active inflammation in the nonglandular stomach of 1 female from the 750 mg/kg/d group and in the tail, ear, and paws of another. These pathological findings were incidental, and the relationship to treatment was unclear.
Prenatal Developmental Toxicity Study of LCCO
Two females in the 750 mg/kg/d group died (1 euthanized on GD 13 and 1 found dead on GD 16) during the gestational period; all others survived to scheduled termination. Clinical observations that were confined to the females in the 450 and 750 mg/kg/d groups included yellow and/or red material around the urogenital area, and red vaginal discharge during the latter part of the gestational period. There were also dermal observations in the high-dose females including slight to moderate erythema, very slight to slight edema and desquamation. There were no dermal effects in the females from the 100 and 450 mg/kg/d groups.
As shown in Table 10, weight gain during gestation was significantly reduced in females from the 450 and the 750 mg/kg/d groups. When analyzed separately it was apparent that the uterine weights in this group were significantly different from control values in part due to the reduced number of viable fetuses and lower fetal body weights, but the nonuterine weight gain was also significantly reduced. Brain and liver weights were similar across groups, but the thymus weights were reduced and significantly different from control values in the 750 mg/kg/d group (Table 11).
Gestational Body Weights of Rats Following Dermal Treatment With Light Catalytically Cracked Gas Oil.a
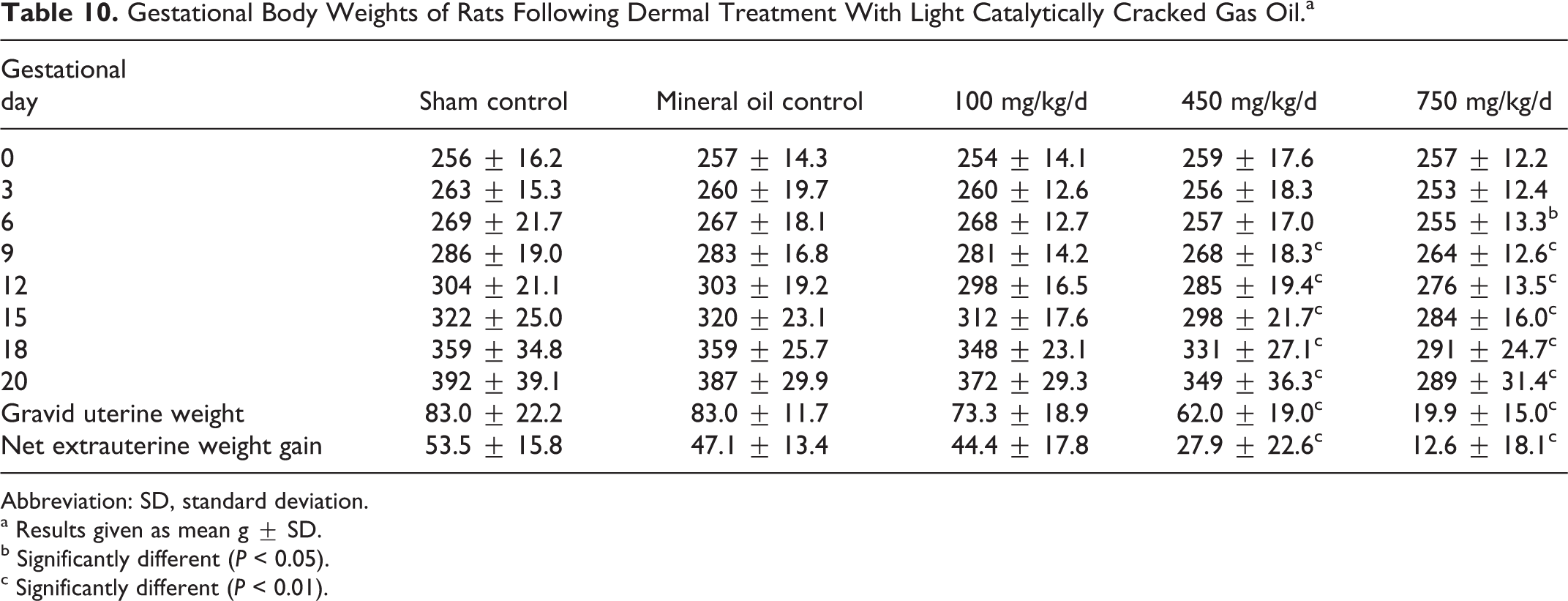
Abbreviation: SD, standard deviation.
a Results given as mean g ± SD.
b Significantly different (P < 0.05).
c Significantly different (P < 0.01).
Summary of Maternal Organ Weights Following Treatment With Light Catalytically Cracked Gas Oil on Days 0 to 20 of Gestation.a
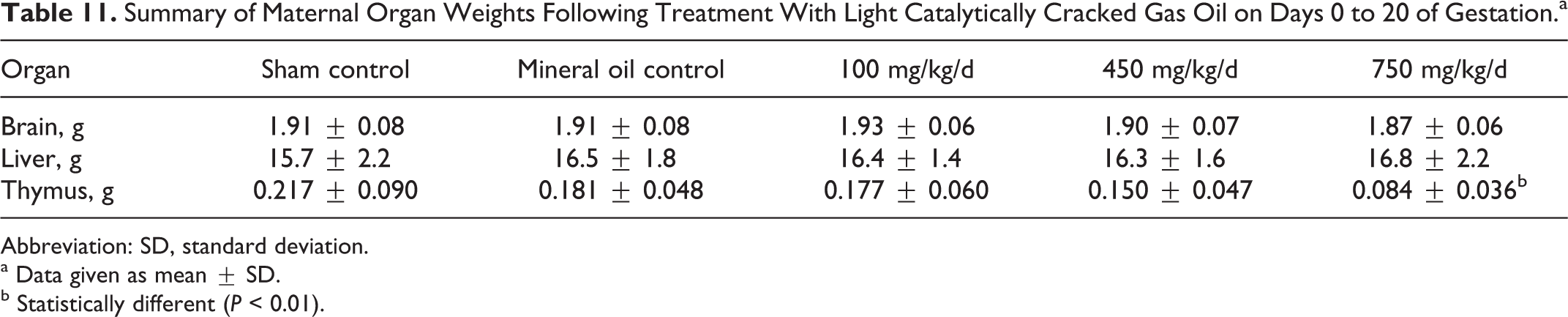
Abbreviation: SD, standard deviation.
a Data given as mean ± SD.
b Statistically different (P < 0.01).
The examination of uterine contents revealed that the number of gravid females was similar across groups, and that there were no differences in numbers of corpora lutea or implantation sites. However, the number of early resorptions was significantly increased, and the number of viable fetuses was significantly decreased in the 750 mg/kg/d group. Fetal weights were significantly decreased in the 450 and 750 mg/kg/d groups (Table 12).
Results of Fetal Examination Following Dermal Treatment of Dams by Light Catalytically Cracked Gas Oil.
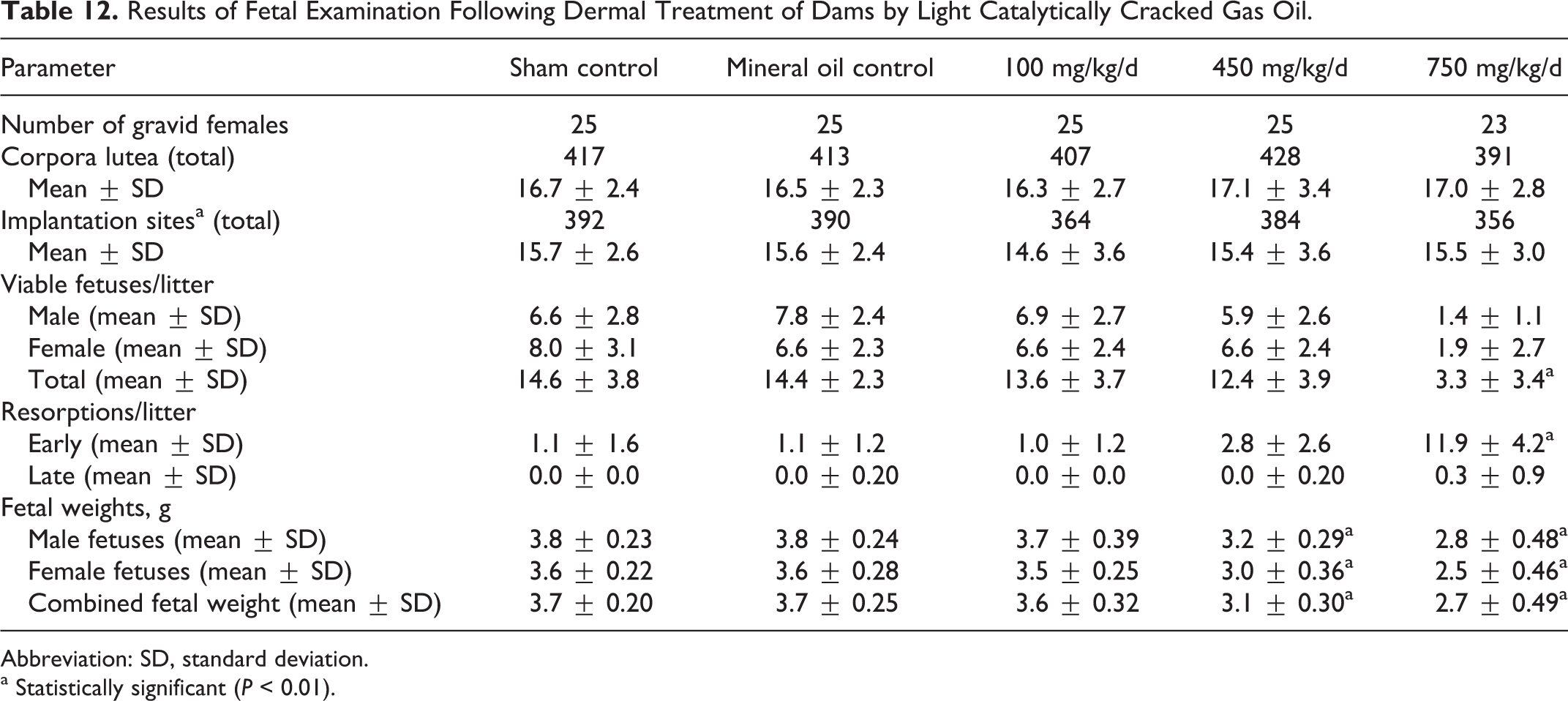
Abbreviation: SD, standard deviation.
a Statistically significant (P < 0.01).
There were no test substance-related external or visceral malformations (Table 13) or variations (data not shown). There were also no test substance-related skeletal malformations, but there were a number of skeletal variations (Table 14). More specifically, reduced ossification of the skull, unossified pubis, reduced ossification of the vertebral arches, metacarpal(s) and/or metatarsal(s) unossified, sternebra(e) nos. 5 and/or 6 unossified, and un/coossified vertebral centrum were noted to be present at higher incidence and at higher mean litter proportions than in the corresponding control groups. In addition, these variations were found at levels outside the historical controls of the testing laboratory. The increased incidence of skeletal variations may have been an indication of delayed development and related to the decreased fetal weights.
Summary of Examinations of Fetuses from Dams Treated by Dermal Application with Light Catalytically Cracked Gas Oil.
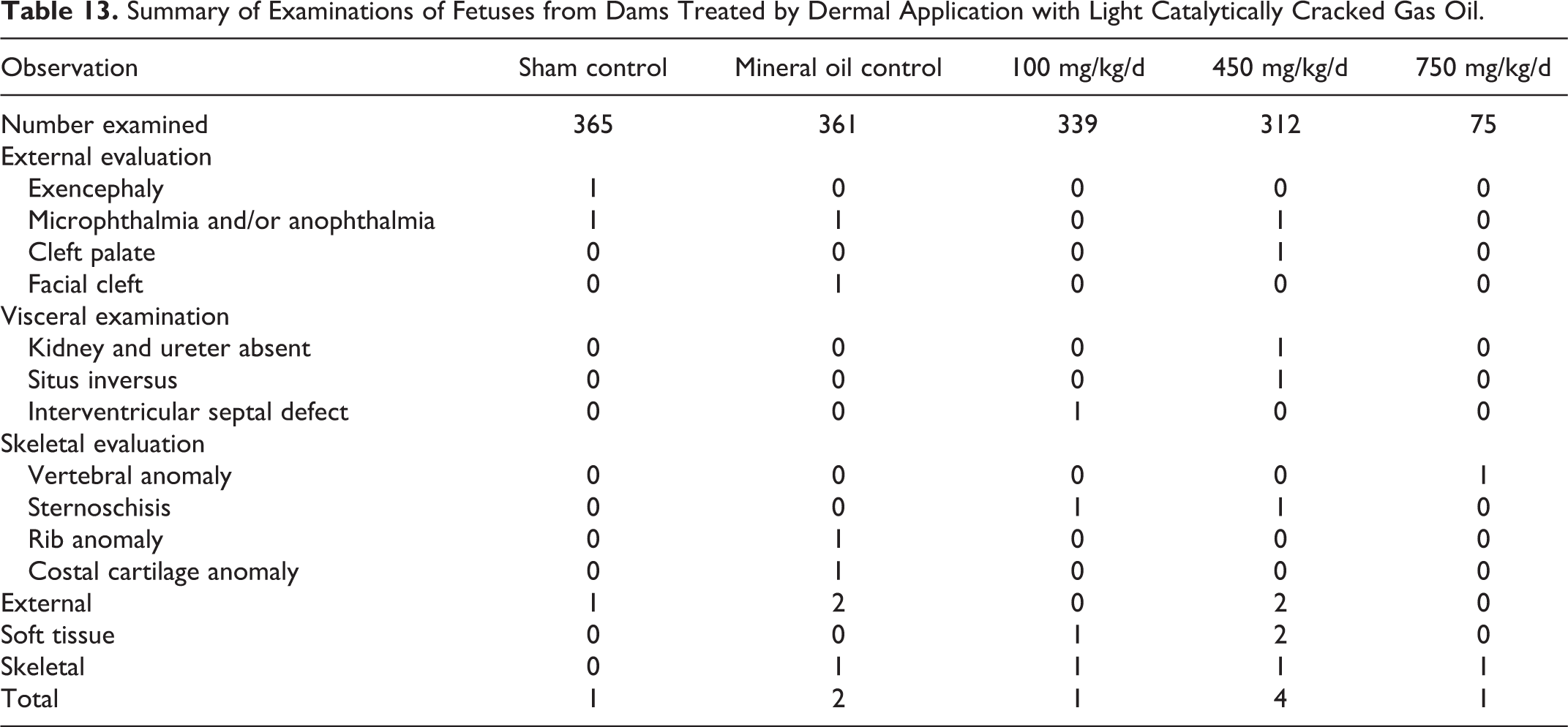
Skeletal Variations in Offspring from Dams Treated by Dermal Application With Light Catalytically Cracked Gas Oil.a
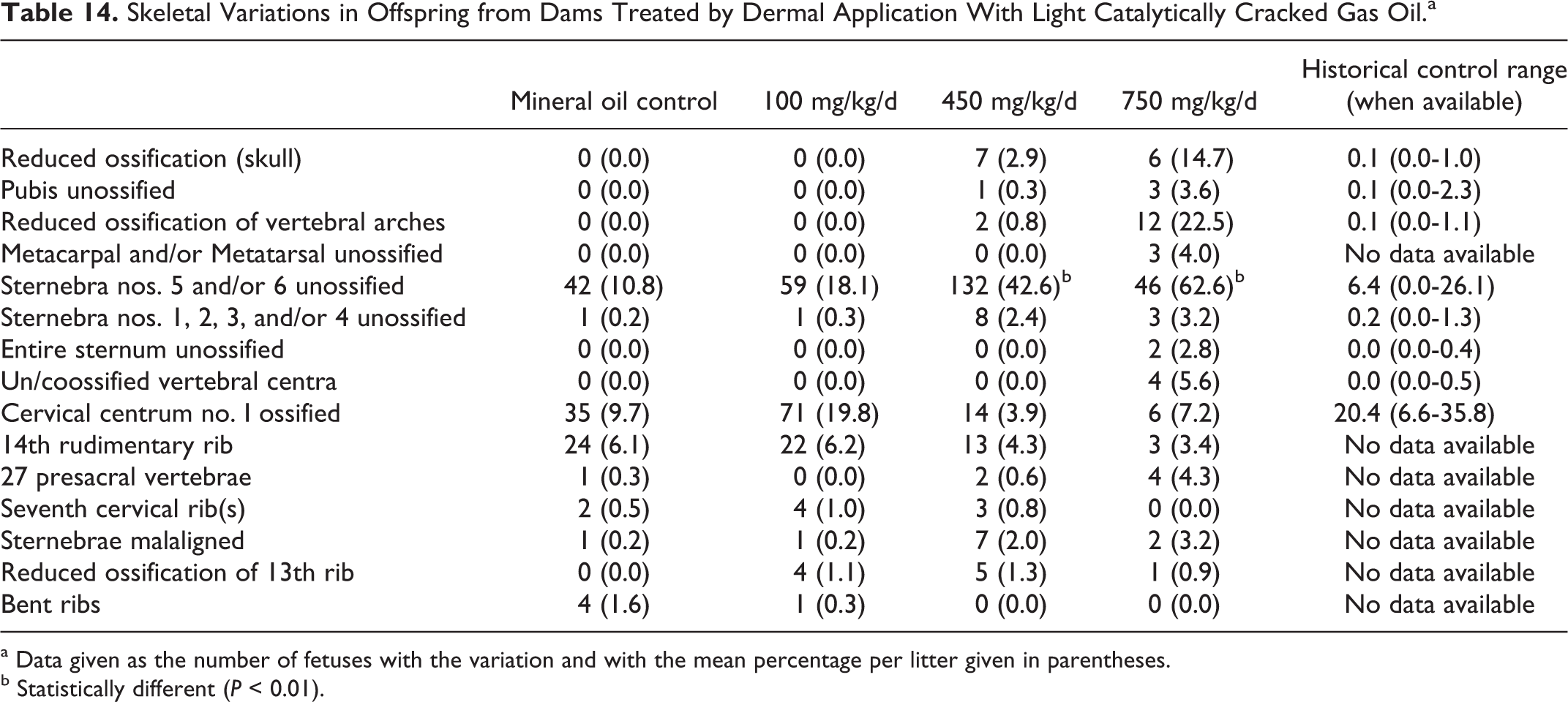
a Data given as the number of fetuses with the variation and with the mean percentage per litter given in parentheses.
b Statistically different (P < 0.01).
Discussion
The specific objective of this study was to compare the potential for systemic and developmental toxicity of the 2 types of gas oils with differing aromatic contents. One of the test substances (ULSD fuel) was a blend of commercial fuels and contained primarily 1- and 2-ring aromatic constituents. The other test samples were from catalytic cracking processes and contained much higher levels of aromatics including 3-ring aromatics than did the ULSD fuel. The hypothesis was that among gas oils (as well as other high-boiling petroleum substances), the potential for systemic and developmental effects is associated with the types and levels of the aromatic constituents that they contain. Based on the previous experience, 12 the systemic effects associated with exposure to aromatic constituents were increased liver weights, decreased thymus weights, and reductions in hematological parameters. The developmental effects that were associated with aromatic content included increased frequencies of fetal death and resorption and reductions in fetal weight.
The outcomes of the studies were consistent with expectation. The diesel fuel blend did not produce any target organ or developmental effects at the highest dose tested (600 mg/kg/d). As noted, the highest dose was chosen based on the preliminary studies and previous experience that suggested that repeated application of diesel fuels at higher levels was likely to produce dermal irritation to a degree sufficient to cause secondary effects as well as being inhumane. However, the level of dermal irritation observed at termination of this study was lower than expected and unlikely to have been associated with any untoward effects.
The LCCO produced the types of systemic effects that had been anticipated. Target organ effects included significant increases in liver weights, significant reductions in thymus weights, and significant reductions in hematological parameters, although the reduced thymus weights were not significantly different when compared on a body weight basis, and there were no pathological findings that correlated with the organ weight data. In males, there were significant differences in most of these parameters in both the 450 and the 750 mg/kg/d groups. In females, significant differences were found in the same parameters, but in most cases, statistical significance was only found in the 750 mg/kg/d groups. No other target organ effects were observed; in particular, there were no weight or histological changes in the reproductive organs. The overall no observed adverse effect level (NOAEL) for systemic effects was 100 mg/kg/d.
In the developmental toxicity study, the frequency of resorptions was significantly increased in the 750 mg/kg/d group, and fetal body weights were significantly reduced in the 450 and 750 mg/kg/d groups. The overall no effect level for developmental effects was 100 mg/kg/d.
When the data from the current study were compared to those from previous studies, it was apparent that there was a relationship between the content of aromatics and the various target organ and developmental parameters that were investigated. As a means of comparing outcomes across studies, 2 parameters were selected, reductions in fetal weight and reductions in the number of live fetuses per litter. The results from the 2 samples reported in the present study and the other 4 samples (HAGO, VTO, LCGO, and another sample of LCCO) were previously reported in the summary form. 12 There were no significant reductions in fetal body weights in the studies of ULSD and LCGO (Figure 1) but more pronounced effects in the studies of HAGO, VTO, and one of the LCCO samples. Significant reductions in live fetuses/litter were produced by HAGO, VTO, and one of the LCCO samples (Figure 2). One of the distinguishing characteristics of the LCGO is that while it contained 44% aromatics, these constituents were almost entirely 1- and 2-ring compounds, whereas the LCCO, VTO, and HAGO samples had similar levels of total aromatics but with higher levels of higher molecular weight compounds including 5% to 10% aromatics with 3 or more rings. The most straight forward explanation for these data is that although the target organ effects may be a more general effect of aromatics, the developmental effects, particularly the increased incidence of fetal death and resorption are associated with aromatics containing 3 or more rings and that other constituents of these substances (aliphatic constituents and 1- and 2-ring aromatics) do not make any important contribution to the developmental toxicity of these substances.
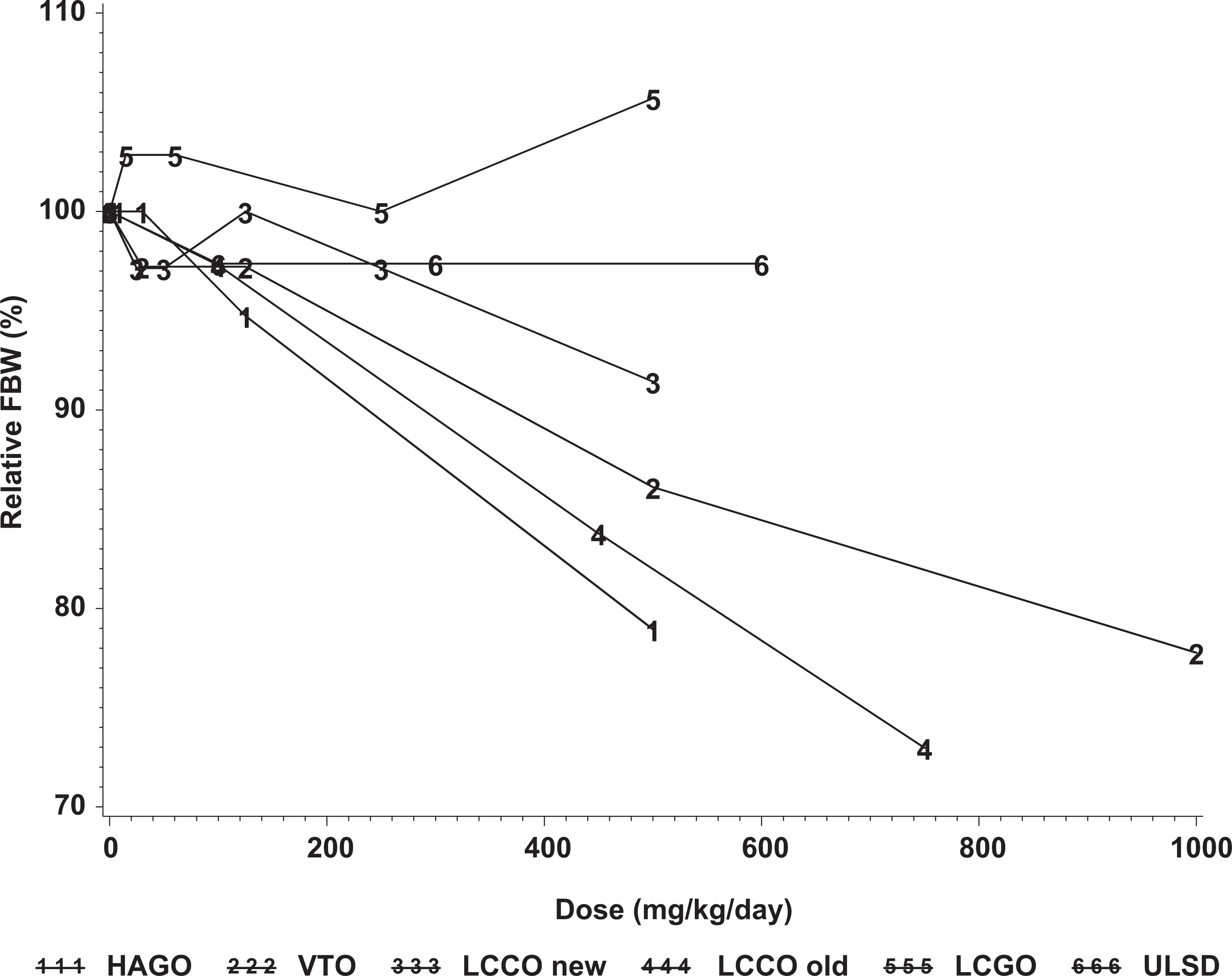
Effects of gas oils on fetal body weights (FBW) in developmental toxicity studies in rats exposed by repeated dermal administration. LCCO new refers to the sample tested in the present study; LCCO old refers to a sample tested previously and described in Feuston et al. 12 LCCO indicates light catalytically cracked gas oil; HAGO, heavy atmospheric gas oil; LCGO, light coker gas oil; VTO, vacuum tower overhead; ULSD, ultralow sulfur diesel.
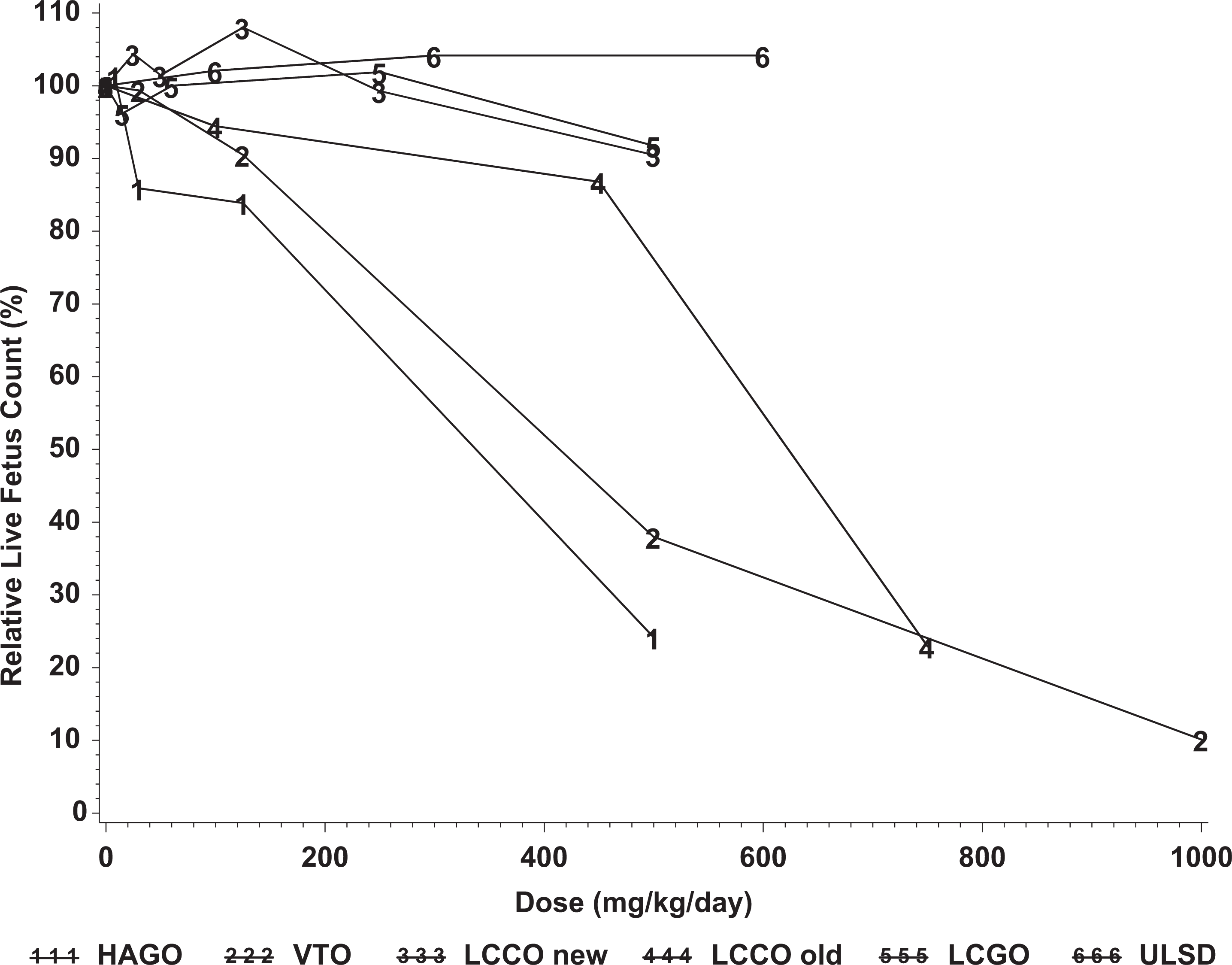
Effects of gas oils on numbers of live fetuses/litter in developmental toxicity studies in rats exposed by repeated dermal administration. LCCO new refers to the sample tested in the present study; LCCO old refers to a sample tested previously and described in Feuston et al. 12 LCCO indicates light catalytically cracked gas oil; HAGO, heavy atmospheric gas oil; LCGO, light coker gas oil; VTO, vacuum tower overhead; ULSD, ultralow sulfur diesel.
Although no formal reproductive toxicity studies of gas oils have been conducted, there are data from several sources, which support the view that these substances are unlikely to have effects on fertility. First, there have been 6 repeated-dose studies (the 2 studies summarized in the present publication as well as the previously unpublished data on the 4 gas oil samples summarized in Feuston et al 12 ) that have included both gross and microscopic evaluations of male and female reproductive organs. There were no gross or histological effects on reproductive organs in the repeated-dose study of ULSD fuel. In the study of LCCO, there were reductions in absolute weights of epididymis and uterus in high-dose group animals, but the differences in organ weights were not significantly different when expressed on a “relative to body weight” basis, and there were no pathological changes in these organs. Among the previously unreported studies, there were no differences in reproductive organs among rats repeatedly exposed to VTOs. In the HAGO, absolute prostate weights were significantly below control values but were not different when expressed on a relative to body weight basis. In a study in which rats were exposed for 90 days to LCGO, the absolute weights of testes or prostate were comparable to control values. Small increases in relative weights of these organs were associated with reductions in body weights of high-dose males. Because there were no differences in absolute organ weights, the differences in relative weights were not considered toxicologically important. In summary, these data provide evidence that the reproductive system is not a target for gas oils.
In addition to the studies of gas oils, there have been several studies of reproductive parameters of related substances, which effectively cover the range of aromatic constituents that might be found in gas oils, and none of these studies provided any evidence of reproductive toxicity. The first of these was a reproductive toxicity study of jet A, an aviation fuel that is blended to meet performance specifications. The jet A test sample was comprised of hydrocarbon constituents with carbon numbers ranging from approximately C9 to C16 of which approximately 20% were 1-ring aromatics and approximately 1% 2-ring compounds as assessed by ASTM D2425. As reported by Mattie et al 31 in separate studies to assess male and female fertility, there were no effects on pregnancy rate, gestation length, or sperm parameters in studies in which males were treated for 70 days at levels up to 3000 mg/kg/d and mated to naive females; in studies in which females were treated for 90 days prior to mating and then through the mating, gestation, and lactation (postnatal day 21) at doses up to 1500 mg/kg/d, there were no effects on gestation length, pregnancy rate, or number of pups/litter. A similar substance, hydrodesulfurized kerosene (CAS number 64742-81-0), was tested in a reproductive/developmental toxicity screening test in which the test substance was applied dermally at levels up to 495 mg/kg/d. There were no treatment-related effects in any of the reproductive or developmental parameters. 32
The potential for aromatics with 3 or more rings to produce reproductive effects was assessed in a dermal study of clarified slurry oil (CAS number 64741-62-4), a high-boiling aromatic substance that is considered to be a reasonable worst case for all petroleum-derived materials. 33 Compositional information on a similar substance indicated approximately 14% aliphatic constituents with carbon numbers ranging from C10 to C30, with the remaining 86% being comprised of 10% 2-ring aromatics, 26% 3-ring aromatics, 21% 4-ring aromatics, and 11% 5-ring aromatics along with 22% carbazoles. 34 Males were treated daily by dermal administration for 70 days prior to cohabitation with naive females at levels ranging from 0.1 to 250 mg/kg/d. Females were treated daily for 14 days premating and continuing through a 7-day cohabitation period to day 0 of gestation. There were no effects on mating, fertility, or testicular end points in the males and no adverse effects on gonadal function, estrous cycles, mating behavior, conception rates, or reproductive organ weights in the females at the highest dose tested (250 mg/kg/d). Accordingly, the NOAEL for both male and female reproductive parameters was 250 mg/kg/d, the highest dose tested. 33 In contrast, in developmental toxicity tests run under similar conditions, significant effects, particularly reductions in viable fetuses, were observed at levels as low as 1 mg/kg/d. 35 These studies together provide evidence that the aromatic hydrocarbon constituents of gas oils do not affect fertility but may affect fetal survival and development during gestation.
In summary, gas oils are not acutely toxic, are not irritating to the eyes, and do not produce allergic contact dermatitis, although they can produce dermal irritation, particularly with repeated contact 4 and can also cause chemical pneumonitis if aspirated into the lung. In repeated-dose dermal studies, some gas oils produced systemic effects including liver enlargement, thymus weight reduction, and reductions in certain blood cell parameters. 12 In developmental toxicity studies, some gas oils increased the incidence of fetal death and resorption and caused developmental delays resulting in reduced fetal weight and increased frequencies of some developmental variants, 12 but there is evidence that gas oils of different types are not reproductive toxicants. 31 –33 Some gas oils were mutagenic under in vitro conditions when tested in “optimized” Salmonella assays, 3 but they did not produce chromosomal effects in bone marrow when tested under in vivo conditions.9 The gas oils that were mutagenic in “optimized” Salmonella assays also initiated skin tumors. 8 Studies of several representative gas oils provided evidence that the constituents that are associated with target organ toxicity, developmental toxicity, as well as being the mutagenic and carcinogenic components were aromatics. However, this association is generic; it is not known which aromatics are associated with these outcomes or even whether all of the outcomes are associated with the same aromatics. But what is evident is that the aliphatic constituents do not contribute to these effects. Finally, the gas oil samples tested in this set of studies reasonably approximate the boundary conditions, that is, the ULSD sample that contained very low levels of aromatics with 3 or more rings did not produce systemic or developmental toxicity at the highest levels tested (600 mg/kg/d), whereas gas oils with significant levels of aromatics with 3 or more rings produced both systemic and developmental effects with an NOAEL of 100 mg/kg/d.
Footnotes
Acknowledgments
The authors would like to thank Chris Sexsmith for quality assurance support and Lynn Bennett for assistance in manuscript preparation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors of this article are employed by companies that manufacture petroleum products and contractors working on behalf of the petroleum industry HPV program.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was sponsored and funded by the PHPVTG, an unincorporated group of manufacturers affiliated by contractual obligation to fund a voluntary data disclosure and toxicity testing program on certain petroleum-related chemical substances in response to EPA’s HPV Challenge Program. The American Petroleum Institute (API) manages the PHPVTG’s activities.
