Abstract
The present study was carried out to investigate the oral single and repeat toxicity of cross-linked β-cyclodextrin (β-CD) made with adipic acid. Ten each of female and male rats were orally administered a 5000 mg/kg single dose. At this dose, mortality or macroscopic changes of internal organs were not observed. Dose levels 500, 1000, or 2000 mg/kg of β-CD were given daily to 5 each of female and male rats by oral gavage for 14 days. Body and organ weights were not significantly different during the study (P < .05). Hematology and clinical chemistry did not show any toxicity from the cross-linked β-CD. Macroscopic or microscopic changes were not observed between the controls and the treated rats. Based on these results, the cross-linked β-CD did not produce any signs of toxicity or other adverse effects at dose levels up to 2000 mg/kg per d for 14 days.
Introduction
β-Cyclodextrin (β-CD) is a cyclic oligosaccharide composed of α-(1→4) linkages of 7 glucose units which has a wide variety of applications in foodstuffs, pharmaceuticals, pesticides, and cosmetics. β-Cyclodextrin is obtained from enzymatic hydrolysis of starch with cyclodextrin glucosyltransferases. The cross-linking of β-CD results in a ring complex having a macromolecular support which can incorporate various inclusion complexes such as cholesterol. 1 In the food industry, β-CD is used for the preparation of cholesterol-free products: the bulky and hydrophobic cholesterol molecule is easily incorporated inside β-CD rings, which are then removed. β-Cyclodextrin is nontoxic, edible, nonhygroscopic, chemically stable, and is easily separated from the complex. 2 Less than 1% of ingested β-CD is absorbed by the gastrointestinal tract in humans and rats, and the molecule is catabolized to glucose in the colon under the influence of bacterial microflora. 3 The oral LD50 values of β-CD for mice, rats, and dogs are 12.5, 18.8, and 5 g/kg body weight, respectively, 4 –6 and the compound causes no significant subchronic toxicity in rats at dietary levels 7 of up to 10%.
The LD50 by intravenous injection in rats is 0.8 g/kg. 7 β-Cyclodextrin is also used as a food additive approved by the food and drug administration (FDA) as generally recognized as safe (GRAS) for a flavor carrier and cholesterol remover. Specifically, β-CD removes over 90% of cholesterol from milk, cream, and so on. 8,9 Even though the β-CD treatment allows an effective removal of cholesterol from various foods, large amount of β-CD is consumed for the process due to ineffective recovery and no recycling. 8,9 To overcome these disadvantages of powdered β-CD, the cross-linking of β-CD has been extensively investigated. 9 –14 Cross-linking is a commonly used derivatization technique for manipulating starch functionality. Epichlorohydrin and adipic acid anhydride have been extensively used to produce cross-linked starch, in which inter- or intramolecular mono- and diethers are formed with hydroxyl groups of starch. 15 Of the cross-linking agents, adipic acid can be used in food since it is an additive that the FDA has approved as GRAS. The cross-linked β-CD forms an ester bond which is a linkage for the hydroxyl group of β-CD with the carboxyl group of adipic acid (Figure 1 ). In our previous studies, cross-linked β-CD using adipic acid exhibited over 90% cholesterol removal and a highly efficient recycling rate in milk, cream, lard, and egg yolk. 8,9,11,12 However, there is insufficient background information on the toxicological evaluation of cross-linked β-CD to give assurances of safety in developing food additives. Therefore, the objective of the present study is to investigate the acute oral toxicity and repeat-dose toxicity of cross-linked β-CD in rats.
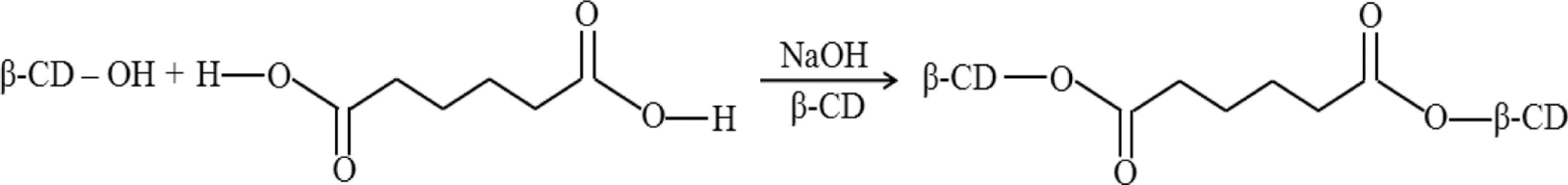
Chemical structure of cross-linked β-cyclodextrin (β-CD) reacted by adipic acid.
Materials and Methods
Materials
Commercial β-CD (purity 99.1%) was purchased from Nihon Shokuhin Cako Co Ltd. (Osaka, Japan). Adipic acid (purity 99%) was purchased from Shinyo Pure Chemicals Co Ltd (Tokyo, Japan).
Preparation of Cross-Linked β-CD
The method of cross-linking of β-CD was a modification of the method used by Han et al. 8 A 50-g sample of β-CD was dissolved in 100 mL of distilled water with 15% adipic acid and placed in a stirrer at 60°C with constant agitation for 2 hours. The pH was adjusted to 12.0 with 30% NaOH. The β-CD solution was stirred at 60°C for 24 hours and then readjusted to pH 5.0 with 30% HCl. The β-CD solution was stored at 4°C for 48 hours to induce crystallization. The cross-linked β-CD was recovered by filtering through a Whatman No. 2 filter paper and washing 3 times with 150 mL distilled water. The product was dried at 60°C in a Lab-Line mechanical convection oven (O-Sung Scientific Co, Seoul, Korea) for 20 hours and passed through a 200-mesh sieve.
Acute Toxicity Test
Animals and Diets
In total, 35 male and 35 female Sprague Dawley (SD) rats (specific pathogen free, 5 weeks of age), weighing 130 ± 20 and 110 ± 20 g, respectively, were purchased from Jung-Ang Laboratory Animal, Inc (Seoul, Korea). The rats were acclimated for 1 week with a chow diet and sterilized water ad libitum. Five rats of each sex were assigned to a group, were individually housed, and allowed free access to commercial basic diets (Purina, Seoul, Korea) and sterilized water. Cages were housed in a room with controlled temperature (22°C ± 3°C) and humidity (50°C ± 10°C) with the lights on from 19:00 to 7:00 hours. The study protocol was reviewed by the Korea Food and Drug Administration (KFDA) Guidelines. 16
Single-Dose Toxicity
A single-dose toxicity assessment was conducted following KFDA Guidelines. 16 The cross-linked β-CD was dissolved in 0.5% carboxymethyl cellulose (CMC; 10 mL/kg). The rats of the control group received the vehicle at the same volume of CMC. Five each of male and female rats were assigned into 2 groups (n = 5) with the weight range of 130 ± 20 and 110 ± 20 g, respectively. The rats in the treatment group were given the 5000 mg/kg cross-linked β-CD by oral gavage. Body weight, clinical signs, and mortality were observed for 14 days. At the end of the study, all rats were sacrificed and the internal sex organs and brain were examined.
Fourteen-Day Repeat-Dose Toxicity
Male and female rats, 25 each, were randomly divided into 5 groups with the weight range of 130 ± 20 and 110 ± 20 g, respectively, and were given powdered and cross-linked β-CDs orally for 14 consecutive days. Dose level of 2000, 1000, and 500 mg/kg per d were selected according to KFDA Guidelines. 16 The animals were dosed orally once a day for 14 days using a cannula attached to a 1-mL syringe. The powdered β-CD was dissolved in 0.5% CMC. As a negative control, 2000 mg/kg of CMC was given orally to the control group. Feeds and water were provided after 1 hour through oral administration.
Clinical Observations
Clinical observations include autonomic nervous function, home cage, and open-field behavior, response to simulation, alertness, and vigilance; and abnormalities of posture and gait were examined according to the functional observational battery (FOB) 17,18 daily throughout the experimental period.
Body Weight and Food Consumption
Body weights and food intake were recorded daily during the study.
Clinical Pathology and Macroscopic Examination
At the end of the experimental period and last day, all rats were fasted for approximately 18 hours and blood was collected from the orbital sinus via the eye to determine hematological and biochemical values. All surviving animals were subjected to terminal macroscopic examination. The animals were killed using an overdose of ether.
Organ Weights
Selected vital paired organs (including heart, liver, spleen, lung, kidney, thymus, testis/ovary, and brain) of control and treated rats were excised, blotted, and weighed together.
Clinical Chemistry
The blood sample was collected and centrifuged at 3000 rpm (Centrifuge 5415C; Eppendorf, Hamburg, Germany) for 15 minutes to extract the serum, and the serum sample was used to determine the total protein (TP), albumin (ALB), total cholesterol (CHOL), total bilirubin (T.BIL), alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP) by enzymatic methods using a biochemical blood analyzer (Hitachi 1780; Hitachi, Japan).
Hematology
The blood sample was used to determine the white blood cell (WBC) count, differential counts (neutrophils, lymphocytes, monocytes, eosinophils, and basophils), red blood cell (RBC) count, hemoglobin concentration (Hb), hematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), red blood cell distribution width (RDW), and platelet (PLT) using a blood cell counter (Hemavet 0950; CDC Tech, Irvine, CA).
Microscopic Assessment of the Liver and Kidney
For the histopathology assessment of the liver and kidney, tissues were fixed in 10% buffered formaldehyde solution (Hedwin, Baltimore, Maryland) and embedded in paraffin. The paraffin waxes were cut into 2 μm sections, stained with hematoxylin and eosin, and then examined under the light microscope (Nikon eclipse 80i, Tokyo, Japan).
Analysis of Data
All statistical analyses were performed using SAS version 9.0 (SAS Institute Inc, Cary, North Carolina). An analysis of variance (ANOVA) was performed using the general linear models (GLMs) procedure to determine significant differences among treatment groups and between males and females. Means were compared using Fisher least significant difference (LSD) procedure. Significance was defined at the 5% level.
Results
Single-Dose Toxicity
The mortality of the rats orally administered with the cross-linked β-CD for single-dose toxicity was tested. The cross-linked β-CD at the dose of 5000 mg/kg did not cause death in any of the rats during the 14-day observation. The rats did not show any sign of toxicity or changes in general behavior or other physiological activities. There were no differences in body weight changes and gross observation between the control and the treatment groups. The macroscopic examination of the internal organs revealed no sign of abnormalities. Thus, it can be concluded that the cross-linked β-CD has no significant toxicological effects.
Repeat-Dose Toxicity
The mortality and clinical signs of rats were observed for repeated oral exposure for 14 days to cross-linked β-CD (500, 1000, or 2000 mg/kg d−1). No mortality was observed during the experimental period. There were also no clinical signs of toxicity in any treated rats at any dose level throughout the 14-day study period.
Body Weight and Food Consumption
The changes in body weight in rats orally administered powdered and cross-linked β-CD for 14 days are shown in Figure 2 . The changes in body weight for the 14-day period were not significantly different (P < .05) among treatment groups. The body weights of all the groups gradually increased during the study duration. Food consumption was not significantly different (P < .05) among the groups.
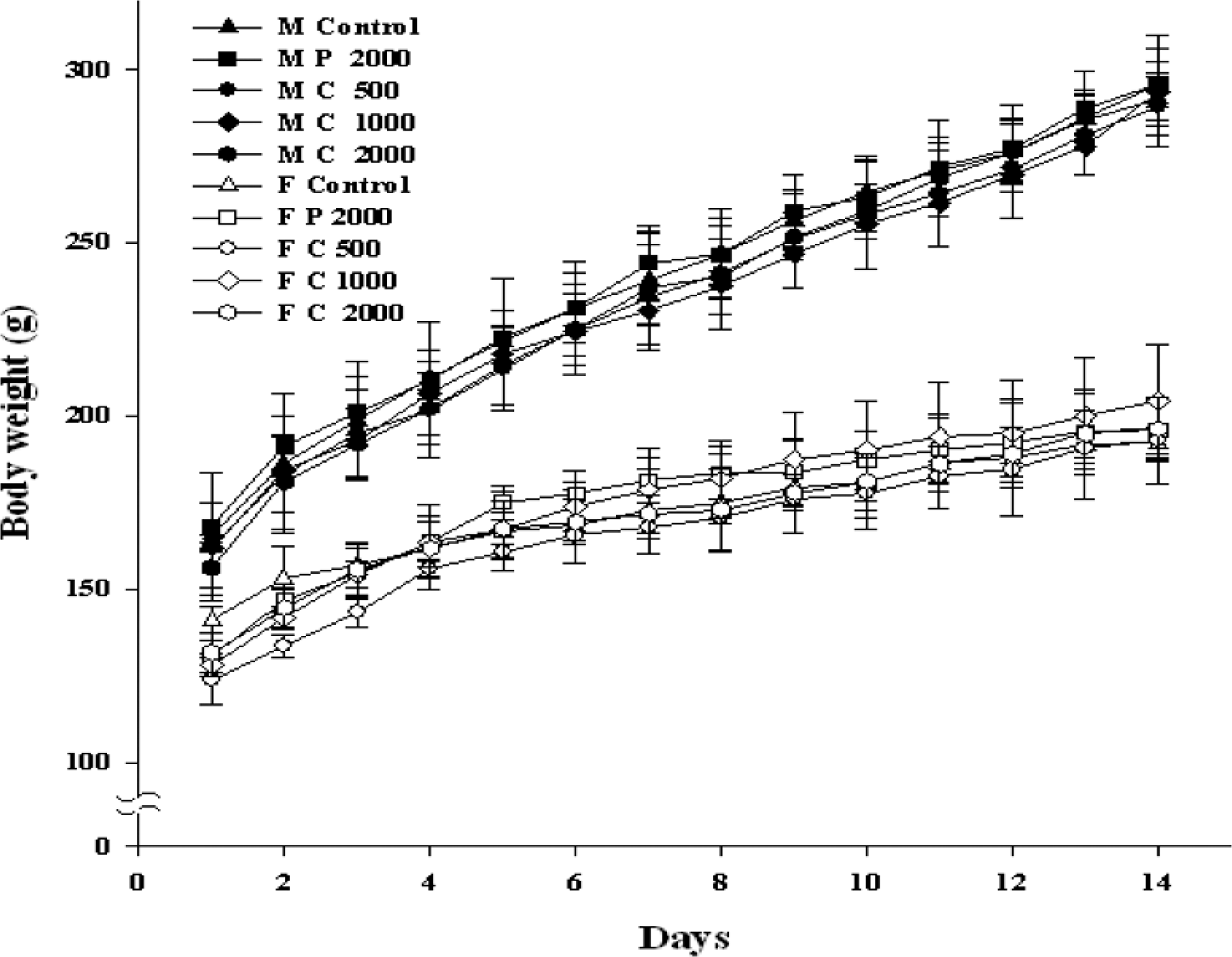
Body weight changes in Sprague Dawley rats orally administered with cross-linked β-cyclodextrin (β-CD) for 14 days. Values are presented as mean ± standard deviation (SD) for 5 rats. M control indicates male rat control; M P, male rat dose with powdered β-CD (without cross-linking); M C, male rat dose with cross-linked β-CD; F control, female rat control; F P, female rat dose with powdered β-CD (without cross-linking); F C, female rat dose with cross-linked β-CD.
Macroscopic Examination and Organs Weights
The organ weights in the rats orally administered powdered and cross-linked β-CDs for 14 days are shown in Table 1 . At the scheduled necropsy, there were no treatment-related gross findings in any of the treated animals. There were no statistically significant differences (P < .05) in weights of the organs (heart, lung, thymus, liver, spleen, kidney, testis/ovary, and brain) between control and treatment groups.
Organ Weight in Sprague Dawley Rats Orally Administered With Cross-Linked β-Cyclodextrin (β-CD) a
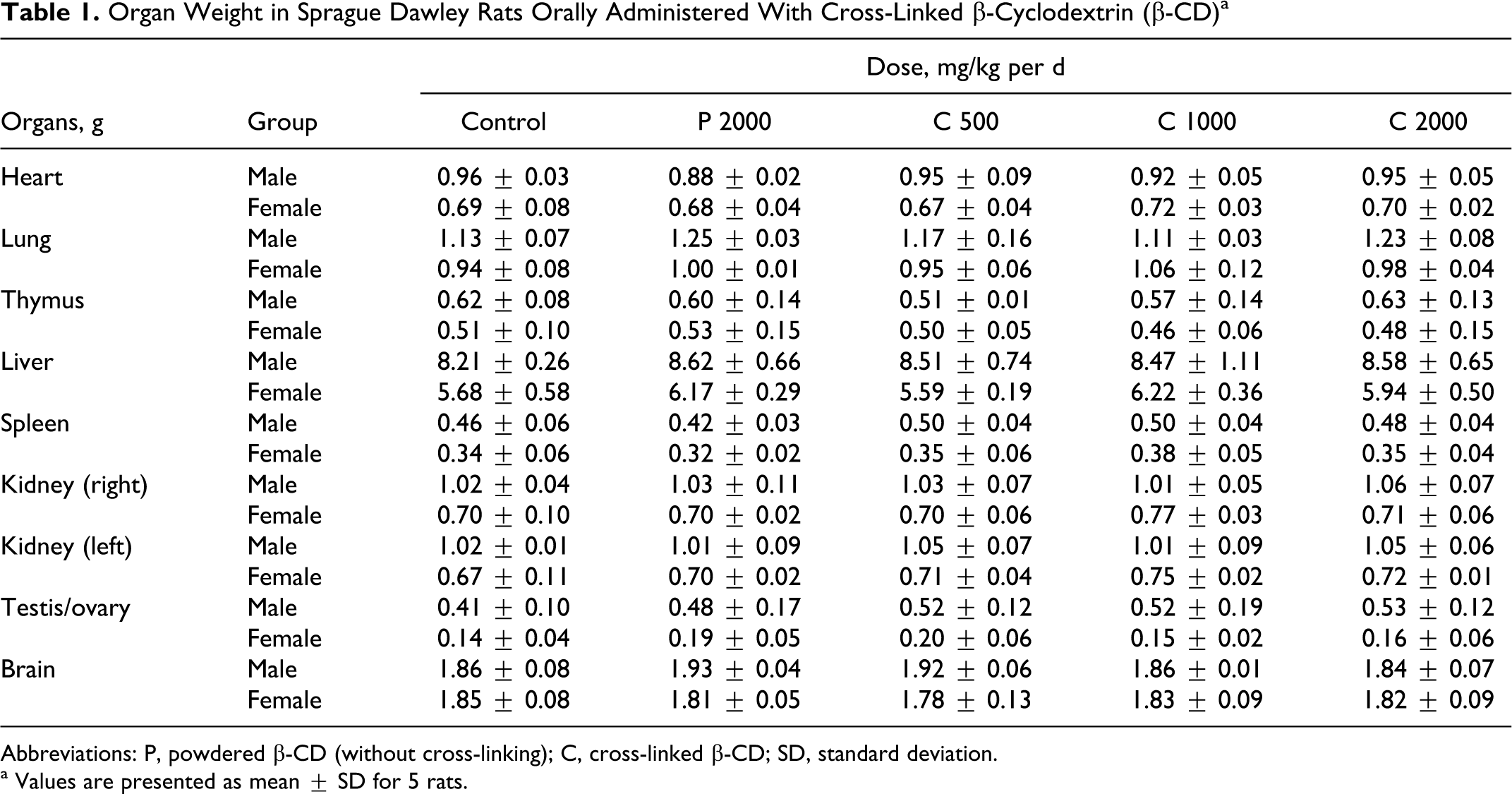
Abbreviations: P, powdered β-CD (without cross-linking); C, cross-linked β-CD; SD, standard deviation.
a Values are presented as mean ± SD for 5 rats.
Hematology
The hematological values in the rats orally administered with cross-linked β-CD as well as powdered β-CD are presented in Table 2 , and the change in WBC was counted as shown in Table 3 .
Hematological Values in Sprague Dawley Rats Orally Administered With Cross-Linked β-Cyclodextrin (β-CD) a
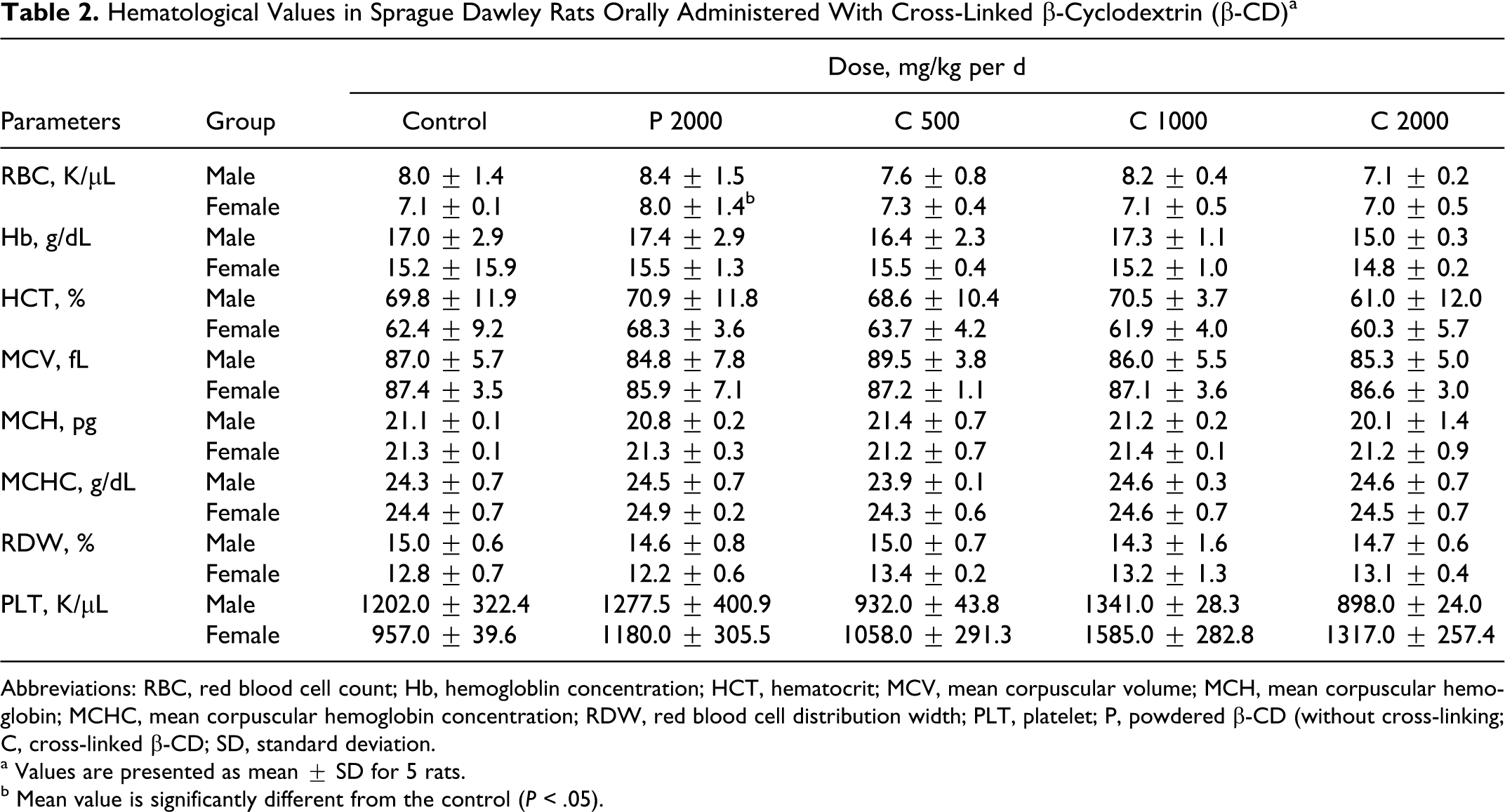
Abbreviations: RBC, red blood cell count; Hb, hemogloblin concentration; HCT, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; RDW, red blood cell distribution width; PLT, platelet; P, powdered β-CD (without cross-linking; C, cross-linked β-CD; SD, standard deviation.
a Values are presented as mean ± SD for 5 rats.
b Mean value is significantly different from the control (P < .05).
Change in WBC Values in Sprague Dawley Rats Orally Administered Cross-Linked β-Cyclodextrin (β-CD) a
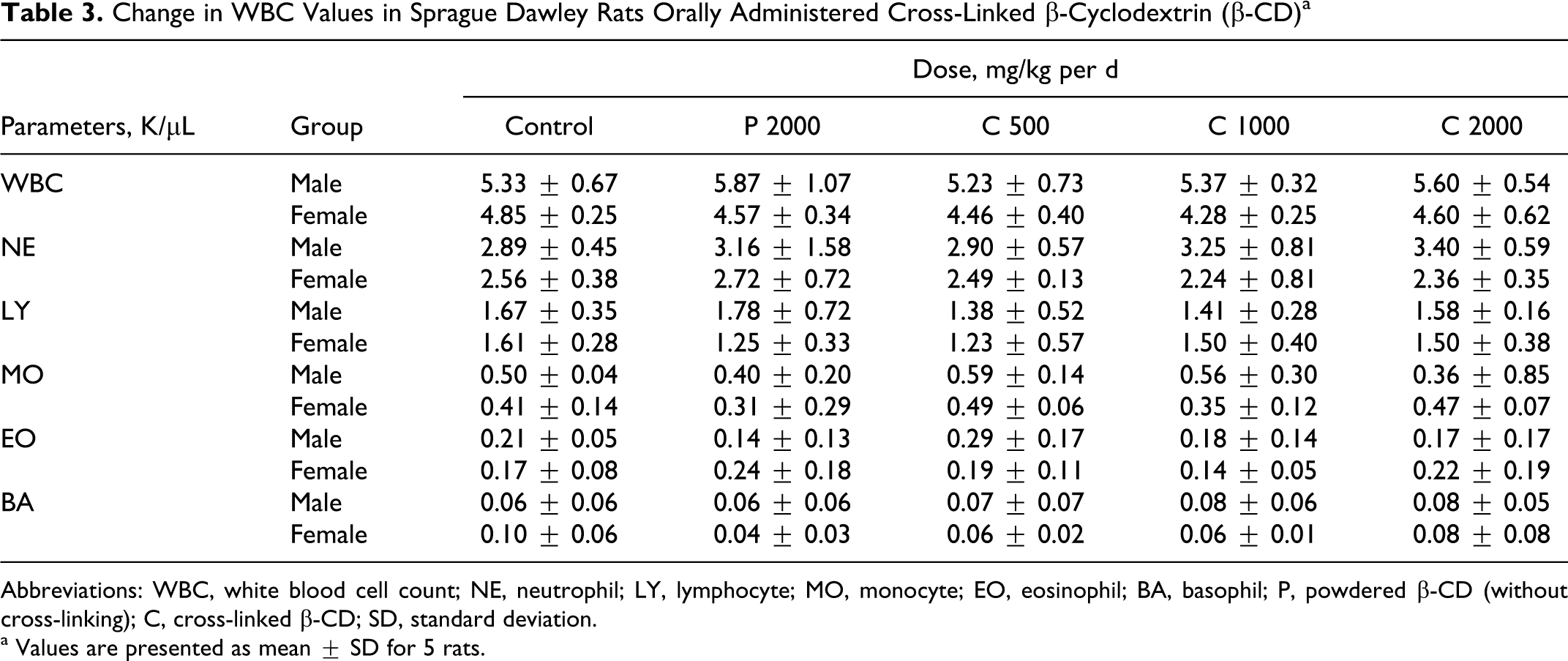
Abbreviations: WBC, white blood cell count; NE, neutrophil; LY, lymphocyte; MO, monocyte; EO, eosinophil; BA, basophil; P, powdered β-CD (without cross-linking); C, cross-linked β-CD; SD, standard deviation.
a Values are presented as mean ± SD for 5 rats.
No significant changes were observed between the administered cross-linked β-CD, powdered β-CD, and the control in Hb, HCT, MCV, MCH, MCHC, RDW, and PLT (Table 2). However, the RBC count in powdered β-CD was statistically significantly (P < .05) increased, but it was within normal reference limit range (4.60-9.19 K/μL). 19,20 Similarly, WBC counts in the administered groups were not significantly different from those in the powdered β-CD and control groups (Table 3). The changes in neutrophil (NE), lymphocyte (LY), monocyte (MO), eosinophil (EO), and basophil (BA) cell values included in the WBC were also not significantly different from those in the powdered β-CD and cross-linked β-CD when compared with the controls.
Clinical Chemistry
Table 4 shows the serum biochemical values. No significant changes were observed among the administered cross-linked β-CD, powdered β-CD, and the control groups in TP, ALB, CHOL, TBIL, ALT, AST, and ALP.
Serum Biochemical Values in Sprague Dawley Rats Orally Administered Cross-Linked β-Cyclodextrin (β-CD) for the 14-day Repeat-Dose Toxicity Test a
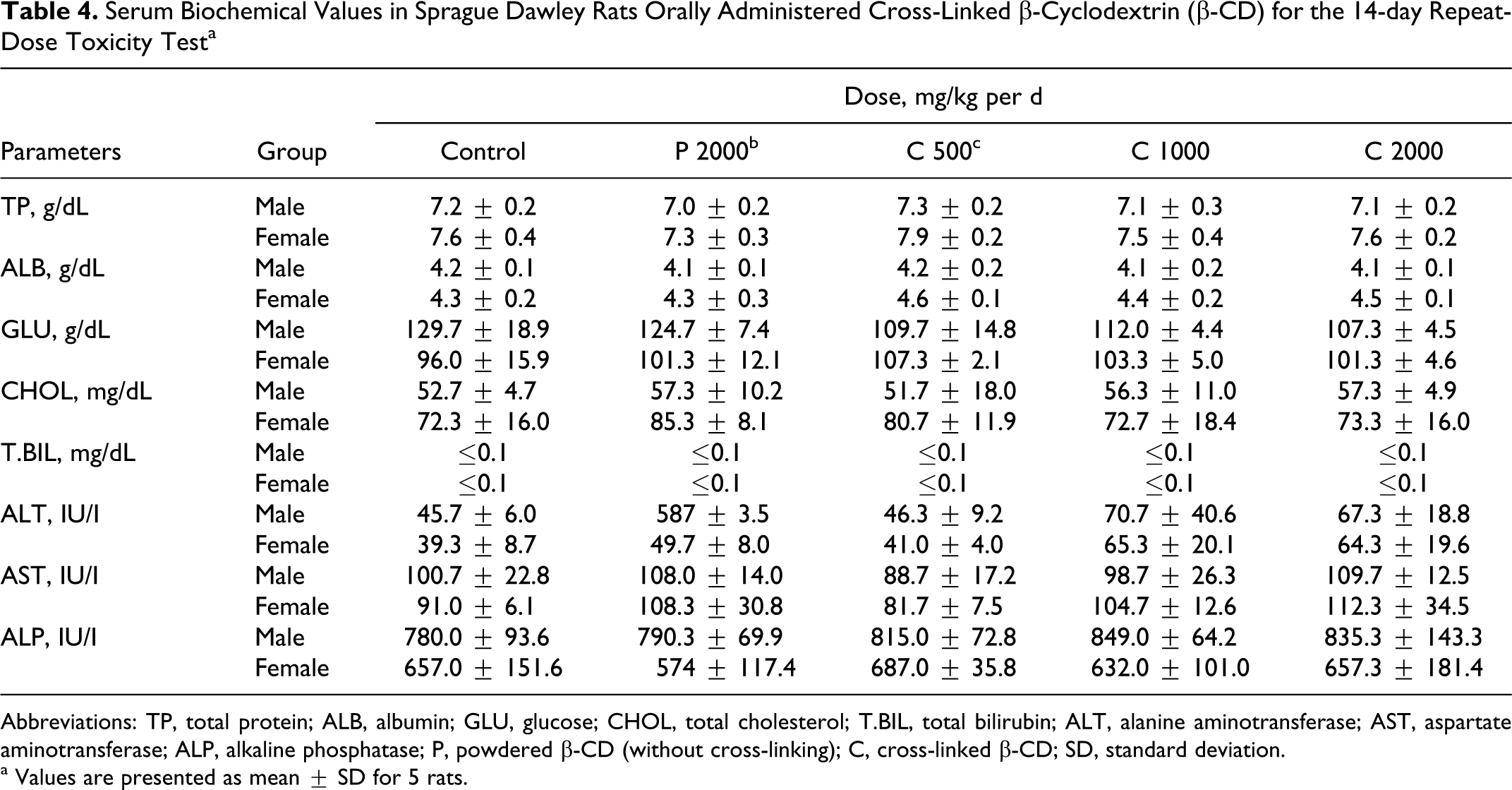
Abbreviations: TP, total protein; ALB, albumin; GLU, glucose; CHOL, total cholesterol; T.BIL, total bilirubin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; P, powdered β-CD (without cross-linking); C, cross-linked β-CD; SD, standard deviation.
a Values are presented as mean ± SD for 5 rats.
Microscopic Assessment of the Liver and Kidney
Oral exposure to 14-day repeat doses of cross-linked β-CD did not cause any observable histopathological changes in the liver tissue or kidney tissue. No histopathological changes were observed from the powdered β-CD or the control groups either.
Discussion
The main objective of this study was to investigate oral acute and repeat-dose toxicity of cross-linked β-CD by adipic acid in rats. The toxicity of β-CD was found in the earlier study conducted by Bellringer et al 21 before investigating the cross-linking. These studies included 52-week toxicity studies in rats and dogs, carcinogenicity studies in mice and rats, and a 3-generation reproductive toxicity study in rats with a teratology phase. These data have previously been reviewed by the joint Food and Agriculture Organization/World Health Organization's (FAO/WHO) joint expert committee on food additives (JECFAs). 22 ,23 The JECFA considered the no-observed-adverse-effect level (NOAEL) in a 1-year toxicity study in dogs to be 1.25% β-CD in the diet (approximately equal to 470 mg/kg). A 100-fold safety factor was applied by the JECFA to this NOAEL and determined an acceptable daily intake (ADI) of 0 to 5 mg/kg per d for β-CD, equivalent to 300 mg/person per d for a 60-kg person. It was also noted that adipic acid, the cross-linking agent used in this study, was previously shown to cause no developmental toxicity in mice, rats, rabbits, or hamsters when administered orally at a concentration of 5%. Adipic acid has a low acute toxicity in rats with an LD50 greater than 5000 mg/kg. 24
In the present study, a single dose of 5000 mg/kg per d and 14-day repeat-dose levels of up to 2000 mg/kg per d administered with cross-linked β-CD in male and female rats did not produce any significant mortality, clinical signs, macroscopic, or microscopic changes. Clincial signs of red material around the eyes, loss of fur, and/or eschar formation in the treatment groups were not considered to be related to treatment since the findings occurred at a low incidence and did not show a dose–response relationship. Also, these clinical signs are common findings in toxicity studies and can result from such factors including stress by group housing. 25 ,26 Even though there was a slight increase in food consumption observed in the male of 2000 mg/kg per d cross-linked β-CD group and a slightly decreased food consumption observed in the female of 500 mg/kg per d group, these changes were not considered biologically relevant. It was mostly sporadic and was not concerned with the dose–response relationship. Under these experimental conditions, the NOAEL of cross-linked β-CD was considered to be over 2000 mg/kg per d and provide the development possibility as food additives.
In conclusion, the present study showed that acute oral administration with cross-linked β-CD and 14-day repeat-dose administration in rats showed no administration-related toxicological or histopathological abnormalities. Our study suggests that addition of cross-linked β-CD into food system such as milk and dairy products in order to lower cholesterol levels could not cause any adverse health effects.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by a grant from the Small and Medium Business Administration in Seoul, Republic of Korea.
