Abstract
Male Hartley guinea pigs and male rhesus macaques were used to determine an efficacious dose of 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate (MMB4 DMS) that would result in 80% survival, 24 hours following a single exposure to cyclosarin (GF). The pharmacokinetic/pharmacodynamic relationship between acetylcholinesterase activity and MMB4 plasma concentrations relative to survival was evaluated. Guinea pigs and non-human primates (NHPs) were concurrently administered MMB4 DMS (guinea pigs: 0, 10, 30, or 40 mg/kg, intramuscular [IM] and NHPs: 0.1, 1, 5, 10, or 20 mg/kg, IM), atropine, and diazepam following a 3 × median lethal dose (LD50) GF challenge. Clinical observations were evaluated using a quality-of-life (QOL) scoring system. All GF-exposed animals exhibited typical signs of nerve agent poisoning immediately following challenge. In guinea pigs, 24-hour survival was 0%, 50%, 90%, and 90% for 0, 10, 30, and 40 mg/kg MMB4 DMS groups, respectively. In addition, nearly all animals surviving to 24 hours were clinically normal, with many in the 30 and 40 mg/kg MMB4 DMS dose group observed as normal by 4 hours post-challenge. In NHPs, survival was 100% for all treatment groups, with all animals noted as clinically normal by 48 hours. Following treatment with atropine/MMB4 DMS/diazepam, NHPs exhibited dose- and temporal-related decreases in incidence and duration of the clinical signs of toxicity. The QOL scores improved with increasing MMB4 DMS dose in both species. The estimated ED80s were 25.5 mg/kg MMB4 DMS (human equivalent dose [HED] of 5.5 mg/kg) and ≤0.1 mg/kg (HED of 0.03 mg/kg) in guinea pigs and NHPs, respectively.
Keywords
Introduction
Nerve agents continue to be a potential threat to both military and civilian personnel. Exposure to nerve agents has occurred in the past during wars, terrorist attacks, and chemical munitions destruction. 1 –4 With the growing unrest in the Middle East, the potential threat posed by chemical weapons continues to increase.
Organophosphorus (OP) nerve agents used in chemical warfare are potent cholinesterase (ChE) inhibitors. Their resultant toxicity is due to the accumulation of acetylcholine at cholinergic nerve endings and their effector organs or postsynaptic sites. This buildup of acetylcholine at cholinergic receptor sites is equivalent to continuous stimulation of these sites by the central nervous system or peripheral nervous system. Common signs include increased secretions, fasciculations, seizures, convulsions, respiratory distress, and death. The current US treatment for nerve agent poisoning is administration of atropine (an acetylcholine receptor antagonist), 2-pralidoxime chloride (2-PAM; an oxime that can reactivate inhibited acetylcholinesterase [AChE]), and an anticonvulsant. 2-PAM is a poor AChE reactivator for certain nerve agents; therefore, candidate replacements for 2-PAM are currently being investigated. 1,1′-Methylenebis{4-[(hydroxyimino)methyl-pyridinium} dimethanesulfonate (MMB4 DMS) salt is a bis-pyridinium oxime that has the potential to reactivate AChE following nerve agent poisoning. A number of laboratory studies indicate that MMB4 may be a more efficacious reactivator of AChE than 2-PAM, especially for certain highly toxic nerve agents. 5 The MMB4 DMS is being developed to replace the currently fielded 2-PAM in the Antidote Treatment Nerve Agent, Autoinjector (ATNAA). This replacement product is called the Improved Nerve Agent Treatment System (INATS). MMB4 DMS is intended to be used in conjunction with atropine at the first sign of nerve agent poisoning.
The objective of this study was to determine an efficacious dose of MMB4 DMS (when administered concurrently with atropine and diazepam) which would produce 80% survival in male Hartley guinea pigs and male rhesus macaques 24 hours following a single exposure to 3 × median lethal dose (LD50) of cyclosarin (GF). In addition, the pharmacokinetic/pharmacodynamic (PK/PD) relationship between AChE activity and MMB4 plasma concentrations was evaluated as they relate to survival.
The study was conducted in compliance with The American College of Toxicology Policy on the Use of Animals, under the guidance of the Battelle Institutional Animal Care and Use Committee and the Animal Care and Use Review Office of the US Army.
Materials and Methods
Animals
Male Hartley guinea pigs (Cavia porcellus), weighing between 250 and 475 g, were purchased from Charles River Laboratories, Inc (Stoneridge, New York). Animals used in the PK/PD phase of the study were surgically implanted with Culex (BASi, West Lafayette, Indiana) catheters in the carotid artery. Animals were single housed in polycarbonate cages on stainless steel racks and were on a 12-hour light/dark cycle.
Male rhesus macaques of Indian origin (Macaca mulatta), weighing 2.0 to 6.0 kg, were procured from National Institute of Health (Dickerson, MD). Monkeys were pair housed and quarantined for 5 weeks prior to placement on study. Once challenged on study, the animals were single housed. Animals were on a 12-hour light/dark cycle and were provided enrichment to enhance psychological well-being throughout the study. Prior to challenge, monkeys were tested and verified negative for tuberculosis and prescreened for simian immunodeficiency virus, simian T-lymphotrophic virus 1 (STLV-1), and Cercopithecine herpesvirus 1 (Herpes B virus) and negative for simian retrovirus (SRV1 and SRV2) by polymerase chain reaction.
Challenge Agent
The nerve agent GF ([fluoro-methyl-phosphoryl]oxycyclohexane) was administered to both guinea pigs and non-human primates (NHPs) at a target dose of 3.0 × LD50 (171 and 139.8 µg/kg, respectively). 6,7 Purity for GF was >95%. The agent was administered by the subcutaneous (SC) route on the dorsum of the guinea pig and by the intramuscular (IM) route on the left calf of the NHP. Dose confirmation samples were collected prior to and following the challenge to verify challenge agent identity and purity.
Therapeutics
Atropine sulfate was purchased from Butler Animal Health Supply (Dublin, Ohio). It was administered IM in the guinea pig in the left hind leg at a dose of 0.4 mg/kg (human equivalent dose [HED] of 0.09 mg/kg). In the NHP, it was administered IM in the left thigh muscle at a dose of 0.4 mg/kg (HED of 0.13 mg/kg). The formulation concentration was 0.54 mg/mL for the guinea pigs and 15 mg/mL for the NHPs.
MMB4 DMS is described as an off-white to tan crystalline powder based on the Certificate of Analysis. Lot No. 1004 MMB4 DMS, with a manufacture date of July 2007, was used for this study. The source of the test article was Cambrex Charles City, Inc (Charles City, Iowa). The purity of the test article was >98%. MMB4 DMS was prepared in 5% benzyl alcohol in water, and methane sulfonic acid was used to adjust pH to 2.3, when required. MMB4 was administered IM in the right hind leg of the guinea pig at doses of 0, 10, 30, and 40 mg/kg (HED of 2.2, 6.5, and 8.7 mg/kg, respectively). In NHPs, MMB4 DMS was administered in the right thigh muscle at 0.1, 1, 5, 10, or 20 mg/kg (HED of 0.03, 0.32, 1.6, 3.2, and 6.5 mg/kg, respectively). Dose confirmation samples were collected prior to and following challenge to verify formulation concentrations.
Diazepam (5 mg/mL; Webster, Carrollton, Kentucky) was administered IM in the guinea pig in the left hind leg at a dose of 2.16 mg/kg (HED of 0.47 mg/kg). In the NHP, it was administered IM in the left thigh muscle at a dose of 2.16 mg/kg (HED of 0.70 mg/kg).
The HED for all therapeutics was determined using body surface area conversions from the Food and Drug Administration’s Guidance for Industry, July 2005. 8
Experimental Design
In all, 112 guinea pigs were divided into 2 phases (76 animals for the efficacy study and 36 for the PK/PD study). A total of 24 NHPs were used in a combined efficacy and PK/PD study. For all studies, the animals were challenged with GF (3 × LD50; SC in guinea pigs; IM in NHPs) 1 minute prior to administration of atropine sulfate, MMB4 DMS, and diazepam. In guinea pigs, a control group in the efficacy study was administered atropine sulfate, diazepam, and MMB4 DMS vehicle control 1 minute post-GF challenge. No control group was included in the NHP study to reduce unnecessary pain and distress to NHPs. Clinical observations for the guinea pig efficacy study and the NHP study were recorded prior to GF challenge and at 5, 15, 30, 60, and 90 minutes and at 4, 6, and 24 hours postchallenge. An additional clinical observation at 48 hours was recorded for the NHPs. Minimal clinical observations were collected on the guinea pig PK/PD study.
Quality- of-life (QOL) scores were calculated as a weighted sum of the observed signs at 5 and 90 minutes and at 6 and 24 hours postchallenge. An additional QOL score was determined at 48 hours postchallenge for NHPs. The signs and scores associated with the signs are described in Table 1. For the impaired and mild signs, if any of the listed signs were present for that classification (eg, hunched posture, lethargic, etc), then the score for that classification was assigned a value of 1 regardless of the presence/absence of other signs in that classification. Otherwise, if none of the signs in the group were present, the entire group was assigned the value of 0. Moderate and severe signs were scored individually and not as a group. This allowed for the progression of signs among the animals, since more severe signs were weighted greater than mild signs. For any time period in which the animal was still alive, the highest score that an animal could attain was 11. If death was recorded in a particular time period, the total score for that period was assigned a value of 12 and that score of 12 was carried through to the end of the clinical observation period. The score of 12 allowed for the progression of severe signs to death to be shown. The lower the animal’s QOL score, the closer its exhibited behavior was to that prior to challenge.
Quality-of-Life (QOL) Scoring System.
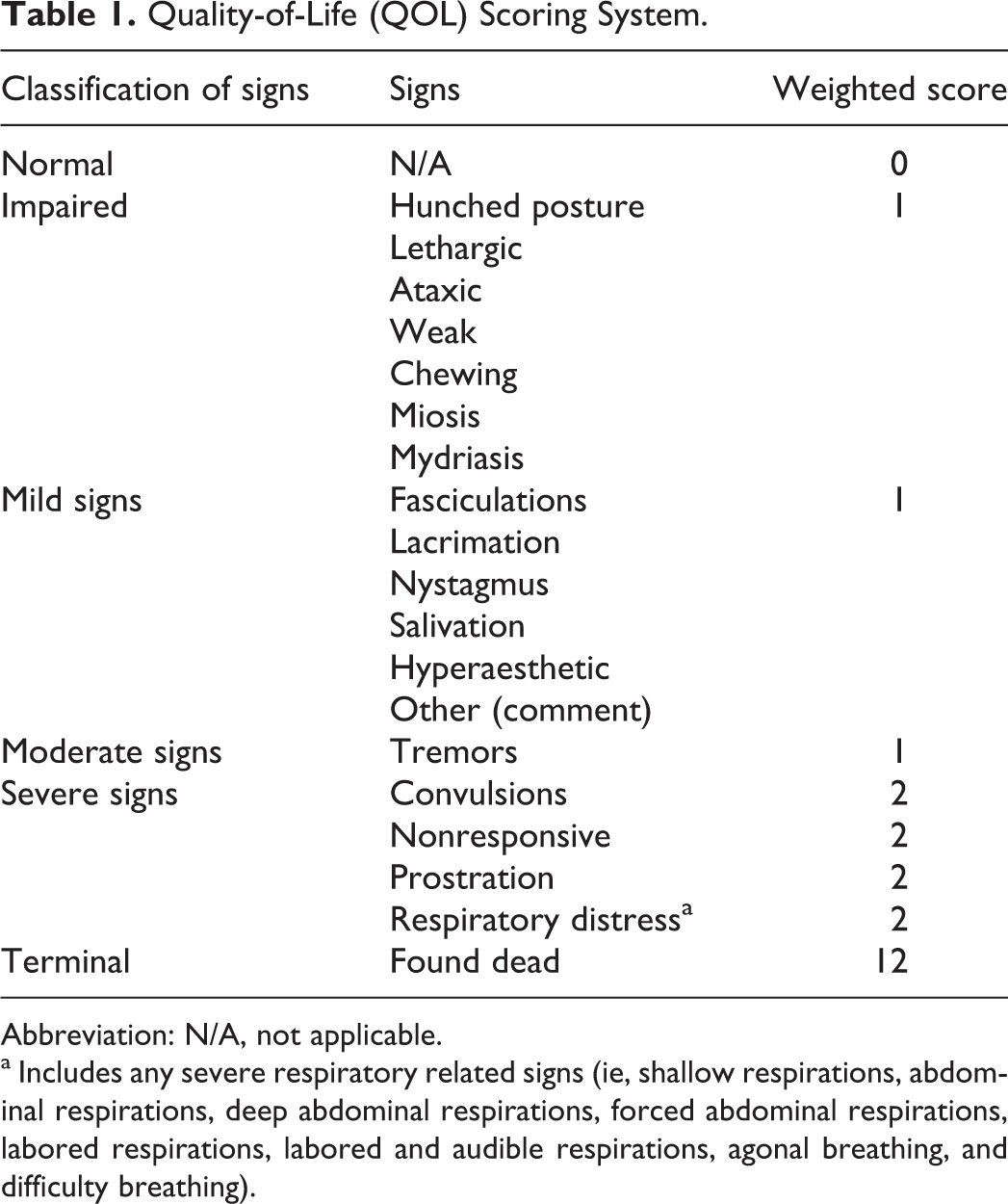
Abbreviation: N/A, not applicable.
a Includes any severe respiratory related signs (ie, shallow respirations, abdominal respirations, deep abdominal respirations, forced abdominal respirations, labored respirations, labored and audible respirations, agonal breathing, and difficulty breathing).
Blood was collected prior to GF challenge and at target times of 5, 15, 30, 60, and 90 minutes after MMB4 DMS dose administration. The pre-GF administration blood sample was used to determine baseline AChE activity. The post-MMB4 DMS administration blood samples were taken to (1) determine the change in AChE activity (relative to baseline) and (2) determine the MMB4 plasma concentration time profile.
Walter Reed Army Institute of Research (WRAIR) Assay
The WRAIR whole blood ChE assay 9 was used (with permission from the US Army) to determine AChE and butyrylcholinesterase activity in the whole blood samples. Aliquots of diluted whole blood were added to a 96-well microplate, and the reaction initiated with the addition of acetylthiocholine iodide (1 mmol/L), propionylthiocholine iodide (1 mmol/L), S-butyrylthiocholine iodide (1 mmol/L), and 4,4′-dithiodipyridine (0.2 mmol/L). The absorbance was measured at 17-second intervals at 320 nm for approximately 4 minutes. Additionally, the absorbance was measured at 450 nm following the kinetics evaluation to determine background levels.
MMB4 Plasma Analysis
Guinea pig and NHP plasma calibration standards were prepared from stock solutions. The plasma calibration standards, plasma blanks, plasma quality control samples, and plasma samples were processed by protein precipitation. The extracts were analyzed by liquid chromatography with mass spectrometry. The MMB4 concentrations were calculated using peak area response ratios and a regression equation constructed from the concentrations and peak area response ratios of the calibration standards.
Pharmacokinetic/Pharmacodynamic Analysis
The PK analysis was performed using the sample collection time points (minutes) and the measured concentrations of MMB4 in plasma (ng/mL). MMB4 concentration time profiles were evaluated using noncompartmental analysis modules in the WinNonlin software program (version 5.0.1; Pharsight Corporation, Mountain View, California). The PK parameters generated and evaluated were Cmax, Tmax, Tlast, and area under the plasma concentration–time curve from 0 time until the last (AUClast).
Group mean concentrations of MMB4 in plasma were also evaluated against reactivation of AChE values at each sample collection time for all the dose groups combined. The concentration–reactivation profiles were analyzed using 2 PD modeling modules (Emax models with and without γ, the shape parameter) in the WinNonlin software program. The Emax, half maximal effective concentration (EC50), and γ, when appropriate, were reported along with goodness-of-fit measures such as Akaike Information Criterion and Schwartz Bayesian Criterion. Based on the PK/PD parameters, AChE reactivation EC20, EC50, and EC80 were derived and reported. The EC50 calculated from WinNonlin represents the concentration that achieves 50% of “Emax,” not necessarily of control AChE activity levels.
Statistical Analysis
Dose–response trends were determined using a Cochran-Armitage trend test, and dose comparisons for survival were based on a Fisher exact test. In addition, QOL score data were analyzed using a nonparametric approach (Kruskal-Wallis test), and the Wilcoxon rank sum test was used to compare among the MMB4 DMS treatment groups.
Results
Survival
For the guinea pig PK/PD study, 24-hour survival is summarized in Supplemental Table 1, and 24-hour survival in the guinea pig efficacy study and monkey study, each following a 3 × LD50 GF challenge, and administration of atropine, MMB4 DMS, and diazepam, is summarized in Supplemental Table 2. When the guinea pig PK/PD and efficacy results were combined, statistical analysis (using a Fisher exact test comparison) revealed no difference between the 10 mg/kg MMB4 DMS group and the control group (P = 0.054). However, the 30 and 40 mg/kg MMB4 DMS groups, although not statistically different from each other, had significantly greater survival than the control (P < 0.001) and the 10 mg/kg dose group (P = 0.023). In addition, there was a statistically significant dose–response trend (P < 0.001) in the guinea pigs. Guinea pig survival was greater than 80% for the animals given MMB4 DMS at doses ≥30 mg/kg together with atropine and diazepam, whereas NHP survival was 100% for all the groups.
Average QOL Scores Following GF Exposure and Treatment With Atropine, MMB4 DMS, and Diazepam in Guinea Pigs.

Abbreviations: Avg, average; GF, cyclosarin; MMB4 DMS, 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate; QOL, quality of life.
Clinical Observations and QOL Scoring
Minimal clinical observations and no QOL scoring were performed on the guinea pig PK/PD study. All clinical observations recorded were in concurrence with those observed on the challenge study. As a result, clinical observations are provided for the guinea pig efficacy and NHP efficacy/PK/PD studies.
Guinea Pig Efficacy Study
Following GF challenge, guinea pigs exhibited signs of nerve agent toxicity including chewing, lethargy, ataxia, fasciculations, tremors, prostration, and respiratory distress. Of the animals that received atropine and diazepam, but only the vehicle for MMB4 DMS post-GF exposure, all were nonresponsive at 5 minutes, 6 of 8 were dead prior to the 15-minute observation, and none survived to 30 minutes. A clear dose–response effect with MMB4 DMS was observed when comparing nonresponsiveness among the groups. While 8 of 8 animals that received MMB4 DMS vehicle were nonresponsive 5 minutes post-GF exposure, only 4 of 10, 2 of 10, and 0 of 10 were nonresponsive in the 10, 30, and 40 mg/kg MMB4 DMS dose groups, respectively, at this same time point.
Other dose-related clinical effects were observed when comparing the low dose of MMB4 DMS (10 mg/kg) to the high dose (40 mg/kg). For example, fasciculations and tremors did not stop in all animals until the 4- and 6-hour observation, respectively, at 10 mg/kg, yet none were seen in any animal administered 40 mg/kg MMB4 DMS at either the 90-minute or 4-hour time points. In addition, at least half of all surviving animals were free of signs at the 30 and 40 mg/kg MMB4 DMS dose by 4 hours; however, the majority of animals in the 10 mg/kg dose group did not return to normal until 24 hours. During the first 5 minutes post-GF challenge, convulsions were observed in 30 of 38 animals. However, at the 10, 30, and 40 mg/kg MMB4 DMS dose levels, convulsions in all animals ceased by 15 minutes post-MMB4 DMS administration.
As for the other clinical observations, no other consistent relationships between incidence and dose/time were identified. Thus, clinical observations related to GF toxicity in guinea pigs were generally present 90 minutes after dosing with MMB4 DMS at all dose levels tested, with the 30 and 40 mg/kg MMB4 DMS dose levels demonstrating dramatic improvements in appearance and behavior noted 4 hours post agent exposure.
The QOL scores in guinea pigs were determined at the clinical observation time points. The lower the QOL score, the better the animal’s QOL. Substantial improvements in the QOL score can be seen when comparing animals dosed with MMB4 DMS vehicle to those dosed with MMB4 DMS (Table 2). For example, at 24 hours post dose, a decrease from a QOL score of 12 (dead) at 0 mg/kg MMB4 DMS (atropine, diazepam, and MMB4 DMS vehicle only) to 6.0 at 10 mg/kg and then approximately 1.0 at 30 and 40 mg/kg indicates a better QOL with increasing MMB4 DMS dose. Significantly lower QOL scores were determined in all MMB4 DMS dose groups compared to control, at all clinical observation times (P < 0.05). At 6 hours post-challenge, there was a significant difference between the 10 mg/kg MMB4 DMS group and the 30 or 40 mg/kg groups (P = 0.003). There were no significant differences at any of the other time points.
NHP Efficacy and PK/PD Study
In the NHP efficacy and PK/PD study, clinical signs following GF exposure included fasciculations, nystagmus, tremors, prostration, convulsions, respiratory distress, and nonresponsiveness. Treatment with increasing doses of MMB4 DMS, in combination with atropine and diazepam, resulted in a dose–response and temporal–response effect, as it pertained to the clinical observations exhibited. Regarding dose response, the peak incidence (initial and highest incidence recorded for the clinical observation) of hunched posture, nystagmus, convulsions, nonresponsiveness, prostration, and respiratory distress decreased with increasing dose of MMB4 DMS. The other clinical observations, that is, weak/lethargic, ataxia, fasciculations, and tremors, did not show a reduction in the peak incidence. Regarding a temporal response, the time to onset for a given clinical observation was similar over the MMB4 DMS dose range tested, except for hunched posture and weakness/lethargy, when onset occurred earlier with increasing dose. Hunched posture and weakness/lethargy was observed during recovery of the animals from GF exposure, with an earlier onset indicating a quicker recovery with increasing MMB4 DMS doses. In general, duration of a clinical observation decreased with increasing dose of MMB4 DMS. The most substantive decrease in signs of toxicity occurred when the MMB4 DMS dose was increased from 0.1 to 1 mg/kg. Doses of 1 to 20 mg/kg did not result in noticeable delays in onset or shorter durations, thereby suggesting a threshold effect.
The QOL scores in NHPs indicated that MMB4 DMS generally reduced GF-related toxicity, as the dose was escalated (Table 3). At the 5-minute time point, there was a trend toward decreasing QOL scores and increasing dose of MMB4 DMS. At the 90-minute time point and beyond, the QOL scores decreased with increasing dose. Statistical differences were found among the MMB4 DMS dose groups at all observation time points through 24 hours post-challenge (5 minutes, P = 0.006; 90 minutes, P = 0.024; 6 hours, P < 0.001; and 24 hours, P = 0.01) using the Kruskal-Wallis test. In addition, at 6 and 24 hours, significant differences were observed between the 0.1 mg/kg MMB4 DMS dose group and the higher MMB4 DMS doses (P < 0.05). At earlier time points (5 and 90 minutes), QOL scores in the 0.1 and 1.0 mg/kg dose groups were significantly higher than those in the 20 mg/kg dose groups (P < 0.05). All animals appeared normal (QOL score of 0) by 48 hours post-GF challenge.
Average QOL Scores Following GF Exposure and Treatment With Atropine, MMB4 DMS, and Diazepam in NHPs.
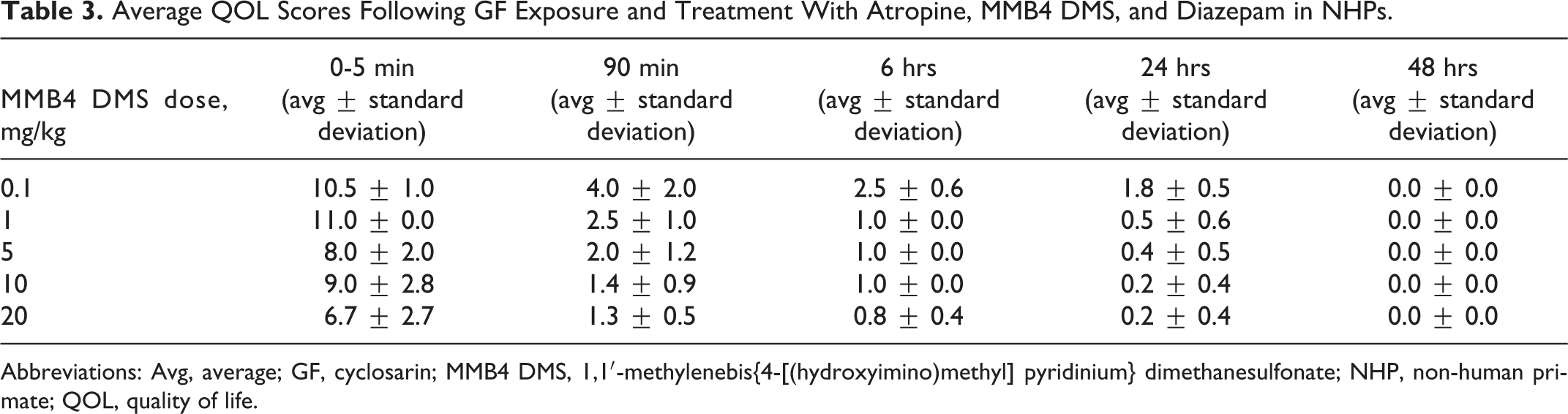
Abbreviations: Avg, average; GF, cyclosarin; MMB4 DMS, 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate; NHP, non-human primate; QOL, quality of life.
Acetylcholinesterase Reactivation
No AChE reactivation analysis was performed in the guinea pig PK/PD study due to microclots in the samples.
In the NHP study, analysis of blood samples collected prior to GF administration resulted in a baseline AChE level of 3.44± 0.54 U/mL (expressed as an overall average [±standard deviation]). Levels of AChE were measured in the NHP study, and oxime reactivation of AChE was inferred from that data. Minimal inhibition of blood AChE was observed 5 minutes post-GF challenge due to the administration of MMB4 DMS just 1 minute following agent administration (Figure 1 and Supplemental Table 3). However, based on the severe clinical signs of nerve agent intoxication observed in the animals and the degree of AChE inhibition at the low dose of MMB4 DMS (0.1 mg/kg), it is likely that GF did cause inhibition of AChE.
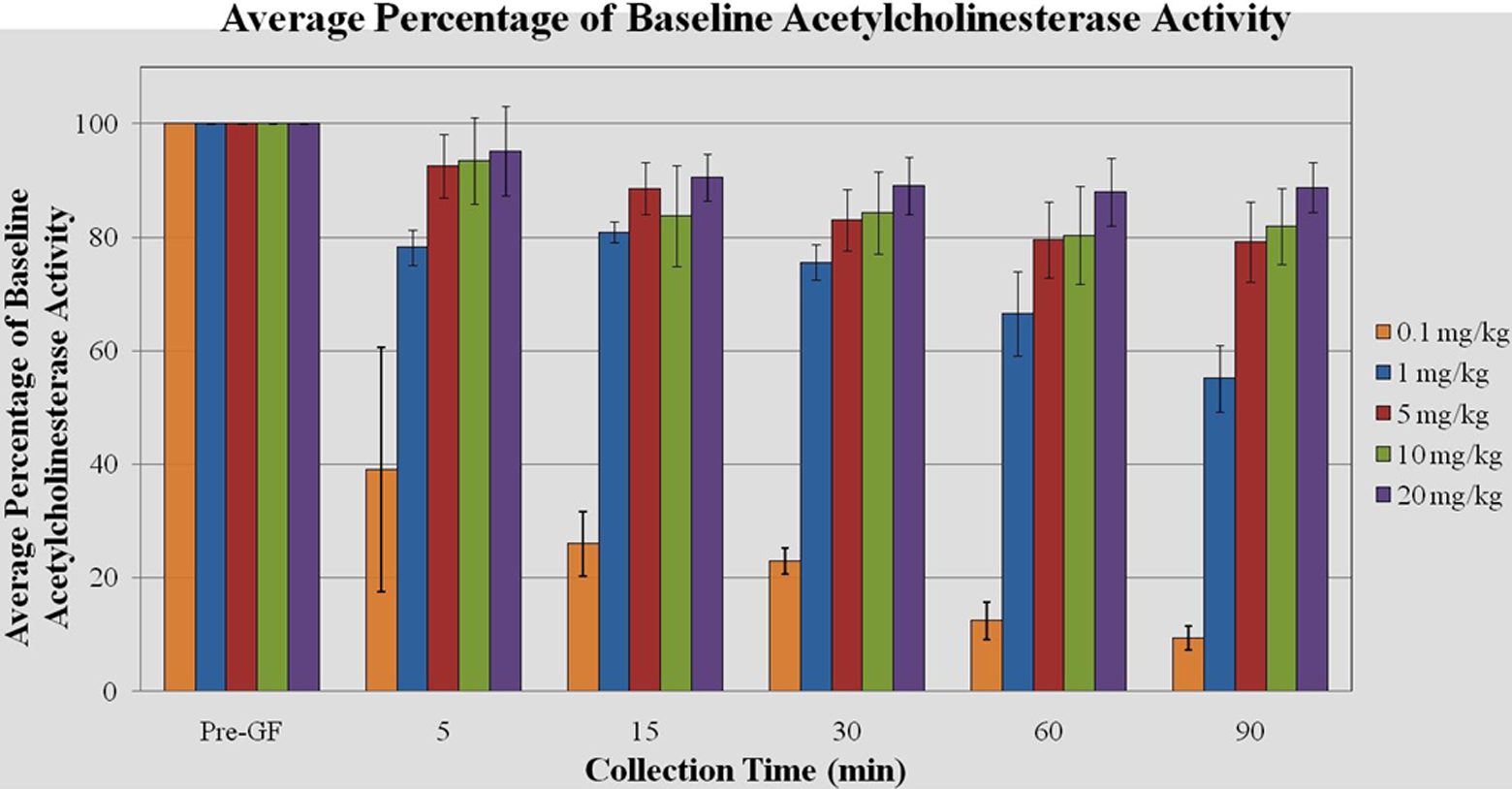
Blood acetylcholinesterase (AChE) reactivation after inhibition by cyclosarin (cyclosarin [GF], 3 × median lethal dose (LD50), intramuscular [IM]) and treatment with atropine (0.4 mg/kg), 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate (MMB4 DMS), and diazepam (2.16 mg/kg) in male monkeys.
Blood AChE activity levels remained approximately at or above 80% of baseline through 90 minutes post 3 × LD50 GF challenge for the 5, 10, and 20 mg/kg MMB4 DMS dose group when administered in combination with atropine and diazepam. The 1 mg/kg MMB4 DMS dose initially resulted in near 80% activity levels, however decreased to approximately 55% of baseline values by 90 minutes. The lowest AChE reactivation was observed with the 0.1 mg/kg MMB4 DMS dose group, with the highest reactivation for that group observed at 5 minutes (39%) and declining through 90 minutes to less than 10% of baseline values.
There was some individual animal variability associated with the MMB4 DMS-related AChE reactivation, with the greatest variability seen in the 0.1 mg/kg MMB4 DMS dose group. For example, although the group mean AChE activity was approximately 40% for the 0.1 mg/kg MMB4 DMS dose group 5 minutes postadministration, 2 of 4 animals exhibited equal to or less than 25% reactivation at this time, and the 2 animals with the highest level of AChE displayed between 40% and 70% reactivation. Thus, group mean AChE reactivation without consideration of the variability could under- or overestimate the efficacy of MMB4 DMS.
MMB4 Plasma Concentration and PK/PD Analysis
In both the guinea pig and the NHP study, prechallenge administration blood samples had MMB4 plasma concentrations below the limit of quantitation, thereby indicating there was no test article contamination. The MMB4 plasma concentrations were measureable out to the last serial sampling time point, that is, 90 minutes postdose, for all MMB4 DMS dose groups (with the exception of the 60- and 90-minute time points at the 0.1 mg/kg dose level in the NHP study), thereby indicating that the level of quantitation (approximately 50 ng/mL) was appropriate for this study.
Guinea Pig PK/PD Study
The MMB4 Cmax values approximated a dose-proportional increase over the MMB4 DMS dose range tested in animals challenged with GF (Table 4). Group mean Tmax values ranged from 7 to 10 minutes and were similar for all dose groups, which is expected when the range of doses are dose proportional. The MMB4 plasma concentration–time results are illustrated in Figure 2.
The MMB4 PK Parameters Following 3 × LD50 GF Challenge in Guinea Pigs.a
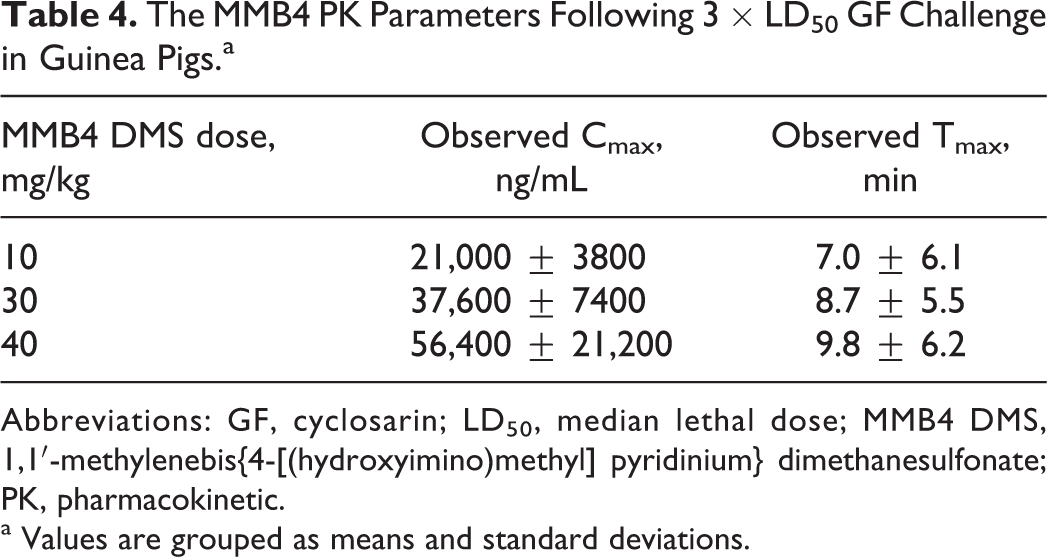
Abbreviations: GF, cyclosarin; LD50, median lethal dose; MMB4 DMS, 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate; PK, pharmacokinetic.
a Values are grouped as means and standard deviations.
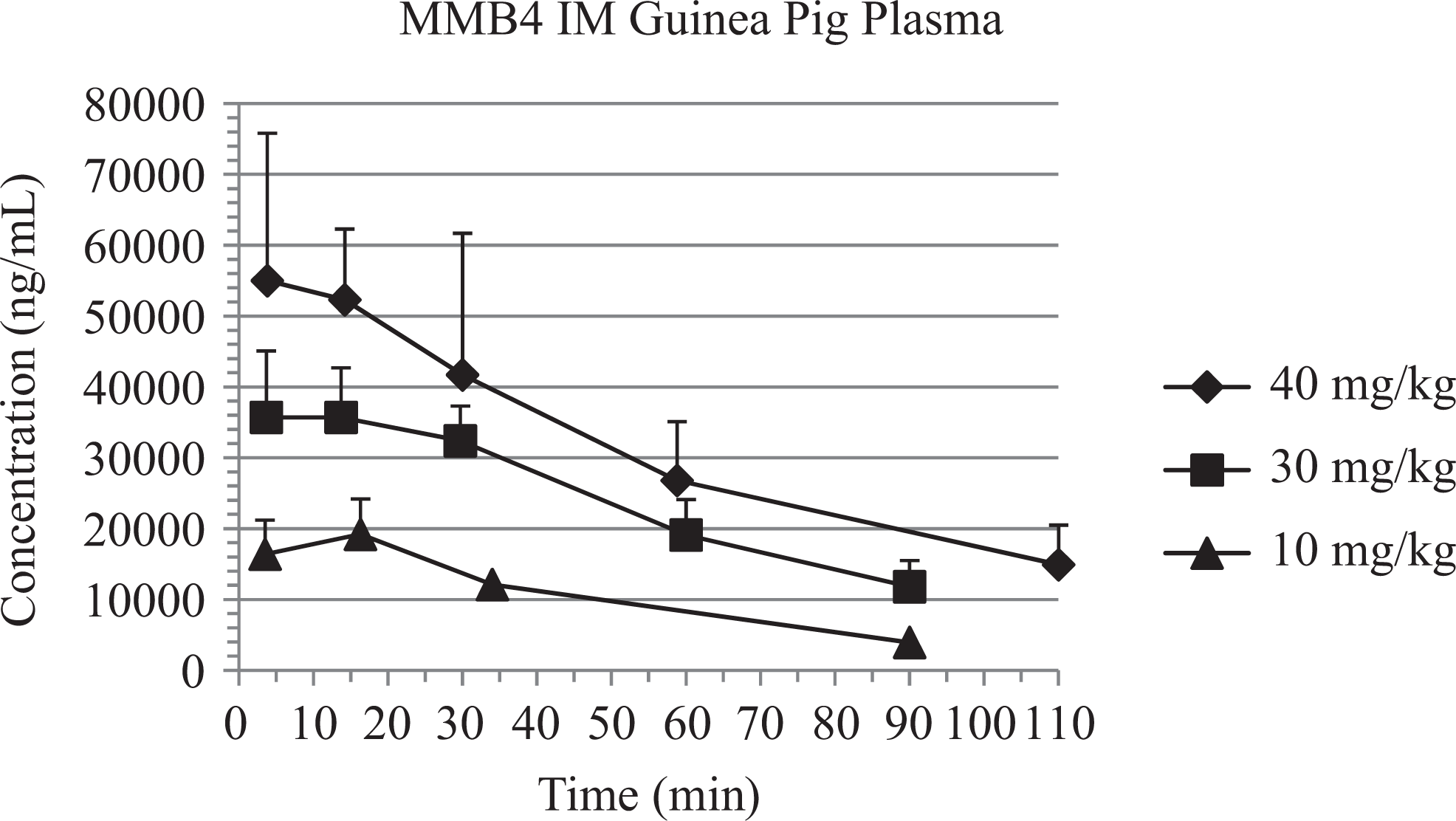
MMB4 plasma concentrations (mean ± standard deviation) after cyclosarin (cyclosarin [GF], 3 × median lethal dose [LD50]) and treatment with atropine (0.4 mg/kg), 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate (MMB4 DMS), and diazepam (2.16 mg/kg) in guinea pigs.
NHP Efficacy and PK/PD Study
Kinetic parameters evaluated for MMB4 after IM administration of MMB4 DMS in NHPs were Tmax, Cmax, and AUClast (Table 5). Regarding Tmax, a majority of the animals from the 5 dose groups (ie, 18 of 24) showed maximal plasma concentrations of MMB4 at the first collection time point (ie, 5 minutes). Group mean Tmax values were similar for the 5 dose groups ranging from 5.0 to 8.5 minutes. Group mean Cmax values increased by 8.2-, 112-, 249-, and 324-fold after an increase in dose by 10-, 50-, 100-, and 200-fold, respectively. One animal administered 5 mg/kg and 1 animal administered 10 mg/kg MMB4 DMS displayed at least 3 times higher Cmax values than the other animals within their respective groups. If these animals were excluded as outliers, then the 5 and 10 mg/kg group mean Cmax values would be 10,800 and 21,800 ng/mL, respectively, resulting in increases in the respective group mean Cmax values of 60- and 121-fold after a 50- and 100-fold increase in dose, respectively. There was large variability in Cmax values for the 5, 10, and 20 mg/kg dose groups, which precluded conclusive evidence for dose-proportionality based on Cmax values. However, AUClast values showed dose-proportional increases from 0.1 to 20 mg/kg with tighter variability than those associated with Cmax values, thereby indicating that the range of doses tested did result in dose-proportional increases in MMB4 plasma concentration. Figure 3 illustrates the MMB4 plasma concentration time profiles.
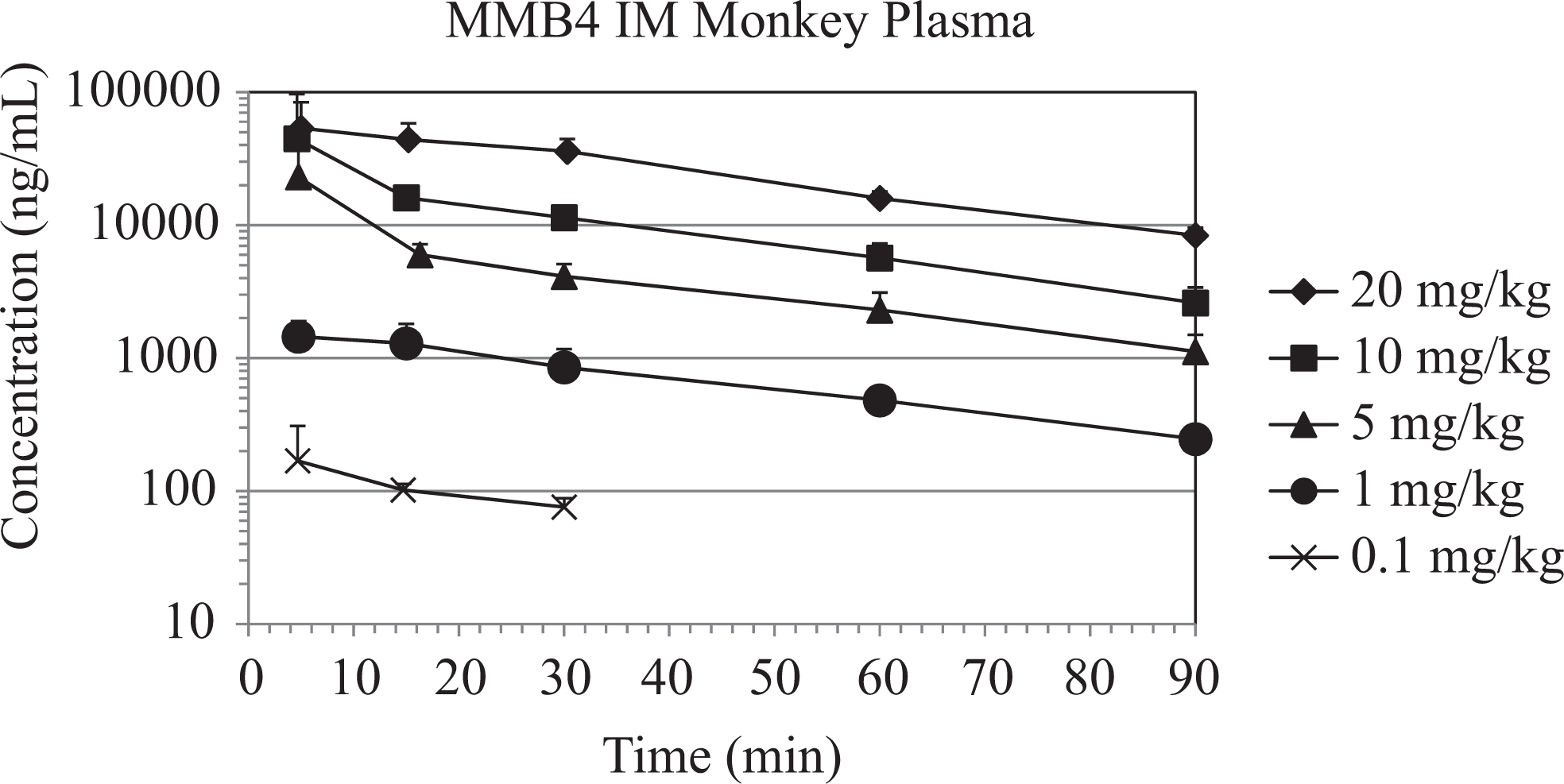
The MMB4 plasma concentrations (mean ± standard deviation) after cyclosarin (cyclosarin [GF], 3 × median lethal dose [LD50]) and treatment with 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate (MMB4 DMS), atropine (0.4 mg/kg), and diazepam (2.16 mg/kg) in male rhesus monkeys.
The MMB4 PK Parameters in the NHP Study.a
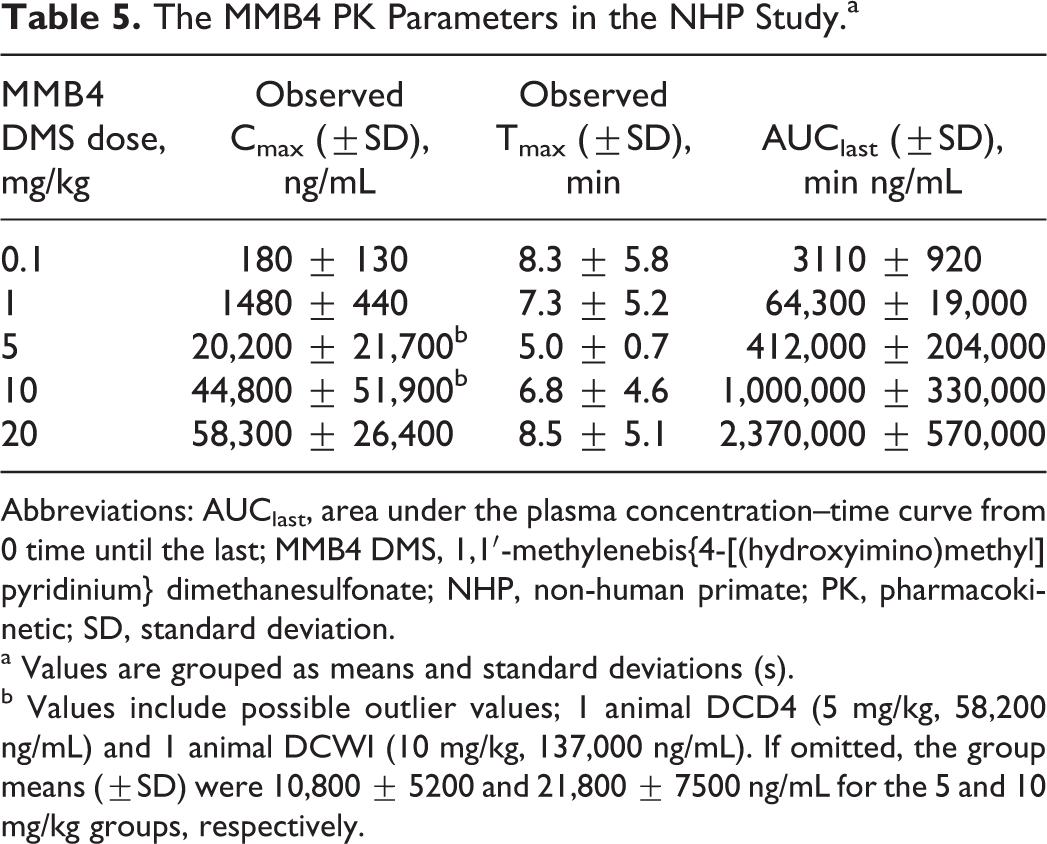
Abbreviations: AUClast, area under the plasma concentration–time curve from 0 time until the last; MMB4 DMS, 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate; NHP, non-human primate; PK, pharmacokinetic; SD, standard deviation.
a Values are grouped as means and standard deviations (s).
b Values include possible outlier values; 1 animal DCD4 (5 mg/kg, 58,200 ng/mL) and 1 animal DCWI (10 mg/kg, 137,000 ng/mL). If omitted, the group means (±SD) were 10,800 ± 5200 and 21,800 ± 7500 ng/mL for the 5 and 10 mg/kg groups, respectively.
The MMB4 DMS dose and MMB4 plasma concentrations in NHPs were directly correlated with AChE activity (Supplemental Table 4). A dose level of 0.1 mg/kg MMB4 DMS produced a peak MMB4 plasma level of approximately 180 ng/mL, which resulted in a peak, group mean AChE activity level of 39% of baseline, the lowest observed among the groups. At dose levels of ≥5.0 mg/kg MMB4 DMS, peak MMB4 plasma concentrations were greater than 20,000 ng/mL resulting in peak group mean AChE activity values of greater than 90%. The AChE activity between the 1.0 and 20.0 mg/kg MMB4 DMS dose groups only increased from 81% to 95%, despite a 20-fold increase in MMB4 DMS dose. In addition, differences in AChE activity among the 3 highest MMB4 DMS dose groups were minimal (93%, 93%, and 95%), yet the peak MMB4 plasma concentrations differed by nearly 3-fold (approximately 20,000 to 60,000 ng/mL), indicating that additional increases in MMB4 plasma concentration did not substantially affect AChE reactivation.
Figure 4 illustrates the dose–response relationship among peak AChE reactivation values (percentage of baseline), observed MMB4 plasma Cmax values (ng/mL), and dose of MMB4 DMS administered to each group of male monkeys. As shown, AChE reactivation values increased with increasing MMB4 DMS dose, appearing to plateau around 93% to 95% and MMB4 plasma concentrations increased in a dose-proportional manner with increasing doses of MMB4 DMS doses from 0.1 to 20.0 mg/kg. Development of an Emax model in NHPs allowed for the estimation of a plasma level and corresponding MMB4 DMS dose level that would predict MMB4 plasma concentrations needed to achieve various AChE reactivation levels (Figure 5).
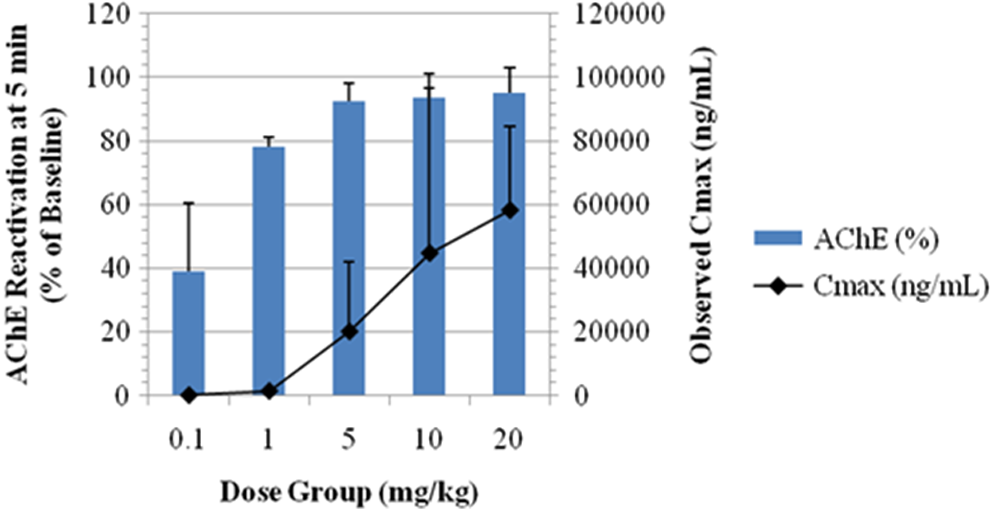
Relationship between peak acetylcholinesterase (AChE) reactivation and MMB4 Cmax .
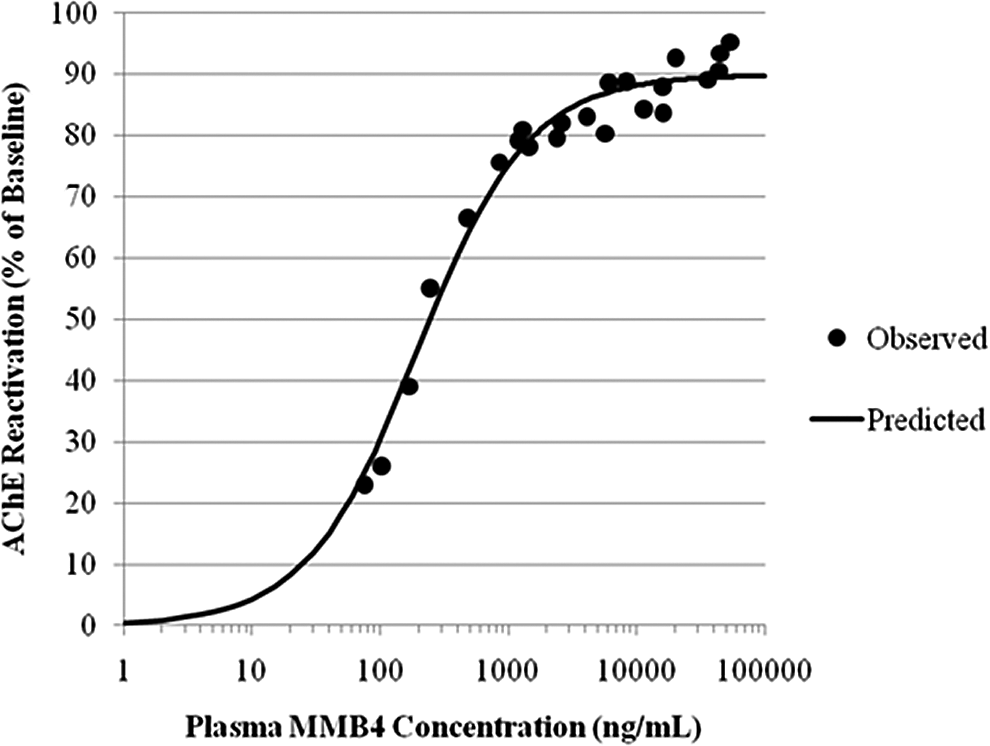
Relationship between acetylcholinesterase (AChE) reactivation and MMB4 plasma concentrations in male rhesus monkeys—all dose groups combined.
Discussion
Organophosphates inhibit AChE at muscarinic and nicotinic synapses by phosphorylation of the serine hydroxyl group enzyme’s active site, resulting in an accumulation of acetylcholine and uncontrolled activation of cholinergic synapses. Atropine works at muscarinic synapses only, competitively antagonizing the accumulated acetylcholine. Atropine is not effective at nicotinic receptors. Oximes reactivate AChE by removing the phosphoryl- or phosphonyl-moiety group, prior to irreversible AChE inhibition (eg, aging). The AChE that is irreversibly inhibited with its negatively charged phosphate can no longer be attacked by a negatively charged nucleophile (oxime), and regeneration is no longer possible. The effectiveness of an oxime is based on the reactivity and affinity of the oxime for the reactive site on the AChE molecule, the speed with which the aging process occurs, which is dictated by the specific OP, the dose, and route of administration/persistence in the body. 10,11
In our studies, MMB4 DMS was evaluated in guinea pigs and rhesus monkeys administered a 3 × LD50 challenge of GF in an attempt to identify a dose level of MMB4 DMS that would meet the criteria for effectiveness, that is, 80% survival following a 3 × LD50 challenge of a given nerve agent (ED80). A standard dosing regimen was executed in this study with regard to the timing of the challenge and therapeutic agents relative to one another. In addition, we investigated a PK/PD model that would allow us to link AChE activity with MMB4 plasma concentrations as they relate to survival.
Clinical signs of GF intoxication in guinea pigs responded well to MMB4 DMS administration. Alleviation of the clinical signs was dose dependent, with the higher dose animals recovering more quickly than the lower dose animals. When survival results from the guinea pig challenge and PK/PD studies were combined following GF exposure, survival was approximately 44%, 88%, and 88% for the 10, 30, and 40 mg/kg MMB4 DMS dose groups, respectively (Table 6). The MMB4 Cmax associated with the lowest dose used (30 mg/kg MMB4 DMS) that resulted in at least 80% survival was 37,600 ± 7400 ng/mL. The overall study ED80 for MMB4 DMS following a 3 × LD50 challenge of GF was calculated to be 25.5 mg/kg in the guinea pig (HED of 5.5 mg/kg).
Combined Survival for GF Guinea Pigs Administered MMB4 DMS/Atropine/Diazepam.

Abbreviations: GF, cyclosarin; MMB4 DMS, 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate; PK/PD, pharmacokinetic/pharmacodynamic.
The lowest MMB4 DMS dose used that resulted in at least 80% survival and blood AChE reactivation in the guinea pig was 30 mg/kg. The corresponding NHP dose, based on a surface area conversion factor, was 20 mg/kg; therefore, this was the dose selected as the starting dose in the NHP study. The results from the NHP study indicated that the doses selected exceeded the criteria for determining the ED80, reflecting the efficacy of MMB4 DMS. Although lower dose levels may have contributed to the identification of the ED80, the severe clinical signs observed following the administration of 0.1 mg/kg of MMB4 DMS may have suggested that slightly lower dose levels would have resulted in mortality. Further evidence for this conclusion would have been supported by a control group included in the experimental design. Thus, although the minimal effective dose of MMB4 DMS needed to achieve the efficacy criteria was not determined, the set of doses that were tested did generate data that allowed an effective dose at a relatively high level of GF challenge to be identified.
Similar to those of the guinea pigs, an improvement in clinical signs of NHPs following GF challenge occurred following the administration of a combination of MMB4 DMS/atropine/diazepam. At doses ranging from 0.1 to 20 mg/kg of MMB4 DMS, 100% of the animals survived. Duration and severity of the clinical signs showed dose and temporal dependence, with the higher dose group animals showing a lower incidence and recovering sooner than the lower dose group animals. However, even at the lowest dose of MMB4 DMS administered (0.1 mg/kg), all animals were responsive, no longer prostrate, not exhibiting respiratory distress, nor convulsing at 4 hours postadministration. Although there were some clinical observations, for example weakness/lethargy, fasciculations, and tremors, which showed delayed recovery, for example <24 or 48 hours, for the animals in the 0.1 mg/kg MMB4 DMS dose group, the animals treated with ≥1 mg/kg MMB4 DMS showed remarkably faster recovery, for example <4 to 24 hours, from these latter clinical observations. These results indicate that while survival could not be used to discriminate between the effectiveness of MMB4 DMS doses tested, clinical observations did show a substantive improvement between 0.1 and ≥1 mg/kg MMB4 DMS. Thus, based on the criteria for efficacy, that is, 80% survival, an ED80 was unable to be generated; however, it was demonstrated to be <0.1 mg/kg MMB4 DMS.
This study provided information about the minimum effective MMB4 plasma level and AChE activity as they pertain to survival. At least 80% survival occurred when the MMB4 Cmax was 180 ± 130 ng/mL, the peak blood AChE activity was 39%, and Tmax/time to peak was 5 minutes, which corresponded with an MMB4 DMS dose of 0.1 mg/kg. The HED is 0.03 mg/kg, or a total administration of 2.1 mg MMB4 DMS (70 kg adult).
In the PK/PD analysis phase of the study, microclots in the blood taken from the guinea pigs were found that nullified our ability to accurately quantitate AChE reactivation. As a result of the rapid reactivation of AChE by MMB4 in the monkeys, minimal inhibition of AChE was observed at the first scheduled blood draw (5 minutes postchallenge). As a consequence, the precise recovery from the nadir of AChE activity cannot be ascertained; however, we are certain of 3 facts (1) nerve agents generally result in AChE inhibition, which is measurable in blood samples; (2) nerve agent-induced clinical observations are typically aligned with AChE inhibition; and (3) based on the severe clinical signs of nerve agent intoxication, we know that GF was administered and actively inhibited AChE.
This last point is supported by investigations from a previous study using 1 × LD50 GF in guinea pigs and rhesus monkeys. 12,13 In that study, atropine was administered to the investigational animals 15 minutes prior to challenge agent administration, and MMB4 was administered at 5 minutes (NHP) or 15 minutes (guinea pigs) postchallenge agent administration, rather than 1-minute postchallenge as in this study. Results revealed that in both the guinea pig and the NHP studies, AChE activity was substantially inhibited by GF by a mean of 91% in the guinea pig and 98% in the NHP. A significant level of AChE reactivation was noted under both experimental paradigms and in this study, following a 3 × LD50 challenge of GF, AChE activity levels remained approximately at or above 80% of baseline through 90 minutes for the 5, 10, and 20 mg/kg MMB4 DMS dose group when administered in combination with atropine and diazepam. The 1 mg/kg MMB4 DMS dose initially resulted in near 80% activity levels; however, these levels decreased to approximately 55% of baseline values by 90 minutes; thereby displaying a dose-response PD activity model. The decrease in AChE activity from 80% to 55% may be due to short half-life of MMB4 in comparison to the nerve agent.
Although a 2-PAM control was not investigated in this study, historical data have shown that it is not a good AChE reactivator following GF exposure. 14 –16 In a study by Shih et al, for instance, a number of oximes were evaluated for their ability to reactivate AChE in the central and peripheral compartments. 16 Results from this study revealed that although all oximes studied, including 2-PAM, significantly reactivated ChE activity inhibited by GB, MMB4 significantly reactivated more whole blood AChE than did 2-PAM. In addition, 2-PAM was unable to significantly reactivate whole blood ChE activity inhibited by GF or Russian VX; however, MMB4 showed the greatest reactivating capability following exposure of both nerve agents in guinea pigs. Finally, all oximes evaluated significantly reactivated whole blood ChE activity inhibited by VX, with MMB4 displaying the highest levels of reactivation.
In summary, remarkable efficacy with MMB4 DMS was observed, specifically in NHPs, at doses ranging from 0.1 to 20 mg/kg MMB4 DMS, with faster recovery observed at higher MMB4 DMS dosages. The efficacy and PK/PD studies in male guinea pigs and male rhesus monkeys successfully characterized the relationships among MMB4 DMS dose, MMB4 plasma concentration, and AChE activity following a 3 × LD50 challenge of GF. In addition, results from this study were able to determine an efficacious HED of MMB4 DMS of 5.5 or 385 mg/70 kg man (based on the guinea pig data) and 0.03 mg/kg or 2.1 mg/70 kg adult (based on the NHP data). Differences between the guinea pig and NHP highlight species differences with regard to MMB4 DMS efficacy following nerve agent exposure. The differences observed may partially be attributed to species differences in reactivation 17 and AChE sequences. 18
Footnotes
Acknowledgments
The authors wish to thank Dr Irwin Koplovitz, US Army Medical Research Institute of Chemical Defense (USAMRICD), for his technical assistance during these studies. The authors acknowledge the editorial assistance of Dr Vincent Brown.
Authors’ Note
The opinions and assertions contained herein are the private views of the author and are not to be construed as official or reflecting true views of the Department of the Army or the Department of Defense.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the United State Army; Chemical Biological Medical Systems (CBMS)/Medical Identification and Treatment Systems (MITS) under contract SP0700-00-D-D3180, Delivery Order 0599 (Task 771) and Delivery Order 0600 (Task 789).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
