Abstract
1,1′-Methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (MMB4 DMS) is currently under development for the treatment of chemical warfare organophosphorus nerve agent poisoning. The present study evaluates the absorption, distribution, metabolism, and excretion of 14C-MMB4 DMS administered intramuscularly to rats and rabbits. The formulated mixture of radiolabeled and nonradiolabeled MMB4 DMS was administered as a single or 7-day repeated dose. Rat doses were 55 or 220 mg/kg (100 µCi/kg), and rabbit doses were 25 or 100 mg/kg (31.25 and 62.5 µCi/kg, respectively). Urine, bile (rats only), feces, blood, and tissues were collected for up to 72 hours. Metabolic profiling using high-performance liquid chromatography with radiodetection was performed on selected urine samples. For both animal species, the majority of the total radioactivity was excreted in the urine (74%-94%) by 72 hours after dosing with greater than 90% of the radioactivity measured in the urine within 8 to 12 hours after dosing. There were no apparent species or dose differences in the urine excretion pattern. The distribution of 14C-MMB4 DMS-derived radioactivity was rapid and generally reached the highest concentration by the first collection time point (0.25 hours). The tissue–blood concentration ratios were highest at the injection sites and in the kidneys and gastrointestinal tract contents for both the species. Two metabolites of MMB4 DMS were detected in rat and rabbit urine; their structure was confirmed by liquid chromatography with tandem mass spectrometry as 4-pyridine aldoxime and isonicotinic acid (pyridine-4-carboxylic acid).
Introduction
Currently, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (MMB4 DMS; Figure 1) is under investigation in the United States as a potential replacement for 2-pralidoxime, the current oxime therapy for organophosphorus (OP) nerve agent poisoning. 1 Oximes currently available and licensed for human clinical use exhibit poor protective effects against the OP nerve agents soman, cyclosarin, and so-called “Russian VX” (O-ethyl S-[2-(diisopropylamino)ethyl] methylphosphonothioate), so there is a great need to develop more efficacious oxime therapies. Evaluation of the absorption, distribution, metabolism, and excretion (ADME) properties of MMB4 DMS was conducted to characterize the drug and metabolite fates. To our knowledge (supported by an extensive literature search), this is the first published data related to MMB4 DMS ADME properties. No MMB4 ADME studies were found in the literature. However, a search for oxime-related ADME studies did uncover limited information. Ecobichon et al investigated the oxime HI-6 (Asoxime) in the rat, dog, and rhesus monkey. 2 Following intramuscular injection, HI-6 was rapidly absorbed and distributed into tissues. The parent and its metabolites were eliminated primarily in the urine. Much of the administered dose was excreted unchanged and within a few hours of administration.
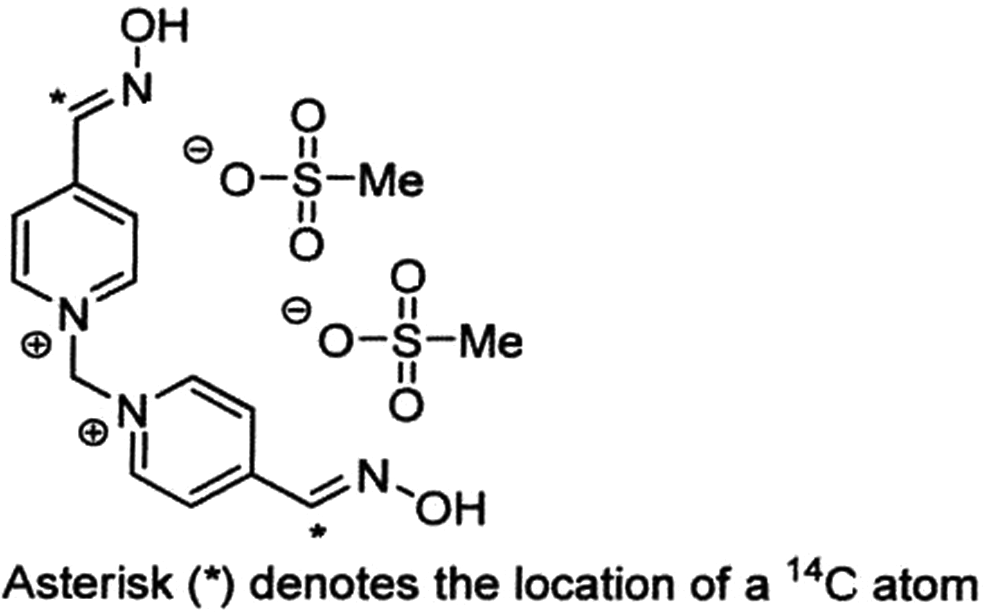
Structure of 14C-1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (14C-MMB4 DMS).
This study investigated the ADME of 14C-MMB4 DMS in male Sprague-Dawley (SD) rats and male New Zealand White (NZW) rabbits following a single intramuscular (IM) administration of 14C-MMB4 DMS and a 6-day repeated IM administration of MMB4 DMS followed by a single administration of 14C-MMB4 DMS (on day 7). The main objectives of this ADME study were to use a radiolabeled form of the drug candidate to determine the routes of elimination and the extent of absorption, determine mass balance, identify circulatory and excretory metabolites, evaluate clearance mechanisms, and characterize the distribution of the drug into tissue and organs.
The study was conducted in compliance with The American College of Toxicology Policy on the Use of Animals, under the guidance of the Battelle Institutional Animal Care and Use Committee and the Animal Care and Use Review Office of the US Army. The study was conducted following the Food and Drug Administration Good Laboratory Practice requirements.
Materials and Methods
Chemicals
The nonradiolabeled MMB4 DMS (98% purity) was supplied by Cambrex Charles City (Charles City, Iowa). The 14C-MMB4 DMS was supplied by Southwest Research Institute (San Antonio, Texas). The radiochemical purity, as determined by the supplier using proton-nuclear magnetic resonance (1H-NMR) spectroscopy, was 100% (up to 1% error in NMR resolution). Sterile water for injections (US Pharmacopeial Convention) was purchased from Baxter (Deerfield, Illinois); benzyl alcohol was purchased from Sigma (St Louis, Missouri). Harlan Certified Diet feed for rats and Harlan Diet 2031C feed for rabbits were obtained from Harlan (Indianapolis). Ultima Gold scintillation cocktail was from Perkin Elmer (Waltham, Massachusetts). All other chemicals and reagents used were the highest grade available and were purchased from Sigma or Fischer Scientific (Pittsburgh, Pennsylvania).
Animals and Methods of Treatment
Male and female CD SD rats for the pilot study and male CD (SD) rats for the definitive study were supplied by Charles River Laboratories (Portage, Michigan) and were 7 to 9 weeks old and weighed approximately 215 to 256 g at the start of the study. The animals for the ADME groups (1-4) were surgically implanted with bile duct catheters at the supplier. Male NZW rabbits supplied by Myrtle Rabbitry Inc (Thompson Station, Tennessee) were 10 to 12 weeks old and weighed 2.18 to 2.62 kg at the start of the study. General procedures for animal care, housing, and environmental conditions conformed (as appropriate) to current Association for Assessment and Accreditation of Laboratory Animal Care recommendations, requirements stated in the current
All animals were acclimated to the environmental conditions that were used for the study, were individually housed in polycarbonate cages, and were maintained at 18 to 26°C with 70% humidity on photoperiod 12 light:12 dark (0600-1800 hours,) and fresh air supplied to the housing room at the rate providing a minimum of 10 changes of air per hour. The animals for the ADME groups were subsequently transferred to metabolism cages (designed to separate urine and feces) prior to dose administration. All animals were observed for signs of abnormalities that would make them unfit for study. The health status of each animal was assessed by a laboratory veterinarian during quarantine. Food and tap water were provided ad libitum.
In the pilot study, 4 rats (2 males and 2 females) were administered a single IM dose (0.1 mL) of 55 mg/kg of MMB4 DMS at a concentration of 137.5 mg/mL and a radioactivity of 250 µCi/mL. The MMB4 DMS (mixture of unlabeled and 14C-labeled) was formulated in 0.5% benzyl alcohol in water for injections adjusted to a pH of 2.2 to 2.3 with methane sulfonic acid.
In the definitive study, 42 male rats were randomly assigned to 1 of the 5 dose groups (6 rats in groups 1-4 and 18 rats in group 5) and were administered a single or repeated IM dose of MMB4 DMS. Rats in groups 1 and 2 received a single IM dose of 55 or 220 mg/kg (100 µCi/kg), respectively, and rats in groups 3 and 4 were administered a repeat IM dose of 55 or 220 mg/kg of unlabeled MMB4 DMS for 6 days, respectively. On the seventh day, a single IM dose of 55 or 220 mg/kg (100 µCi/kg) was administered to rats in groups 3 and 4. Rats in group 5 received a single IM dose of 220 mg/kg (100 µCi/kg) of MMB4 DMS (mixture of labeled and unlabeled). Dose volumes were based on the individual body weights on the day of dosing. For the repeated dosing, IM dose sites included left and right thigh muscle and were alternated between those sites in order to minimize the injection site irritation.
A total of 30 male rabbits were randomly assigned to 1 of the 5 groups (3 rabbits in groups 1-4 and 18 rabbits in group 5) and were administered a single or repeated IM dose of MMB4 DMS. Rabbits in groups 1 and 2 received a single IM dose of 25 or 100 mg/kg (31.25 or 62.5 µCi/kg, respectively), and rabbits in groups 3 and 4 were administered a repeated IM dose of 25 or 100 mg/kg of unlabeled MMB4 DMS for 6 days, respectively. On the seventh day, a single IM dose of 25 or 100 mg/kg (31.25 or 62.5 µCi/kg) of MMB4 DMS (mixture of labeled and unlabeled) was administered. Rabbits in group 5 received a single IM dose of 100 mg/kg (62.5 µCi/kg) of MMB4 DMS. Dose volumes were based on the individual body weights on the day of dosing. For the repeated dosing, IM dose sites included left and right thighs, gluteal, triceps, and epaxial musculature and alternated among those sites in order to minimize injection site irritation throughout the study.
Sample Collection and Analysis
For the rat pilot study, expired gas was spurged through aqueous (2 mol/L NaOH) and organic (ethylene glycol) scrubbing solutions at predose (overnight) and at the following intervals: 0 to 4, 4 to 8, 8 to 12, 12 to 24, and 24 to 48 hours after dosing. Each solution was weighed prior to being stored and measured for radioactivity. Urine, cage rinse, and feces samples were collected predose and at the following intervals: 0 to 4, 4 to 8, 8 to 12, 12 to 24, 24 to 48, 48 to 72, 72 to 96, 96 to 120, 120 to 144, and 144 to 168 hours after dosing. Each sample was weighed prior to being stored and measured for radioactivity. Duplicate aliquots of each scrubbing solution, urine, and cage rinse were analyzed for radioactivity. Each feces sample was mixed with water (1:1, w/v) and homogenized. A single-weighed aliquot of each sample was combusted using an oxidizer (Perkin Elmer, Waltham, Massachusetts) and subsequently analyzed for radioactivity content. Blood, liver, kidney, spleen, brain, heart, lung, bone, fat (perirenal), muscle (gluteal), muscle (injection site), testes (male), uterus/ovaries (female), gastrointestinal (GI) tract, GI tract contents, and residual carcasses were collected at termination (168 hours postdose) and weighed. Each tissue sample was homogenized (with water if needed), and a single-weighted aliquot of each sample was combusted and subsequently analyzed for radioactivity.
For the definitive rat study (groups 1 and 2), bile, urine, cage rinse, and feces were collected predose and at the following intervals: 0 to 4, 4 to 8, 8 to 12, 12 to 24, 24 to 48, and 48 to 72 hours after dosing; bile was collected during the first 30 minutes of each collection interval. For groups 3 and 4, this procedure was performed on day 7. Duplicate aliquots of each bile, urine, and cage rinse were analyzed for radioactivity content. Aliquots of selected urine samples were analyzed by high-performance liquid chromatography (HPLC) and liquid chromatography with tandem mass spectrometry (LC-MS/MS) for metabolite profiling and identification.
Each feces sample was mixed with water (1:1, w/v) and homogenized. Duplicate weighed aliquots of each sample were combusted and subsequently analyzed for radioactivity content. Prior to termination, a final blood sample was collected via cardiac puncture under CO2/O2 anesthesia into tubes with lithium heparin. The following tissues or specimens were collected and weighed: liver, kidney, spleen, brain, heart, lung, bone (femur), fat (perirenal), muscle (gluteal), muscle (injection site), testes, GI tract, GI tract contents, and residual carcass. Each sample was homogenized (with water if needed), and duplicate weighed aliquots were combusted and subsequently analyzed for radioactivity content. Collected excreta samples were stored frozen until analysis; collected blood samples were stored refrigerated until analysis.
For the rabbit study (groups 1 and 2), urine, cage rinse, and feces were collected predose and at the following intervals: 0 to 4, 4 to 8, 8 to 12, 12 to 24, 24 to 48, and 48 to 72 hours after dosing on day 1. For groups 3 and 4, this procedure was performed on day 7. Collected rabbit excreta sample were stored and analyzed as described above. The same tissues or specimens as listed above were collected for the rabbit study.
Quantitation of Radioactivity
Quantitative measurements of radioactivity were conducted by liquid scintillation counting (LSC) using scintillation counters (Beckman Coulter, Brea, California). Conversion of the counts per minute to disintegrations per minute (dpm) was performed directly by the microprocessor in the LSC using a quench curve stored in the instrument memory; each sample radioactivity was automatically corrected for a background radioactivity. Sample counting was terminated after 10 minutes or when twice the standard deviation was ≤2% of the mean, whichever occurred first. All samples were analyzed in duplicate if sample size allowed. If results from sample duplicates differed by more than 10% from the mean value, the samples were reanalyzed (if sample size permitted).
High-Performance Liquid Chromatography and LC-MS/MS Analysis
The dosing formulations were quantitatively analyzed by a Waters HPLC system (Waters, Milford, Massachusetts) with Prodigy ODS-3 column (5 µm, 250 × 4.6 mm; Phenomenex, Torrance, California) at a flow rate of 1 mL/min and ultraviolet detector (290 nm). The mobile phase consisted of 2 mmol/L heptanesulfonic acid sodium salt and 0.5% formic acid in 10% aqueous acetonitrile (A) and 0.5% formic acid in 90% aqueous acetonitrile (B). In gradient elution, the mobile phase A was eluted for 5 minutes, followed by a linear gradient to 50% of the mobile phase B at 20 minutes, to 88% of the phase B at 21 minutes, and then with an isocratic elution for 9 minutes, followed by a linear gradient to 0% of the mobile phase B at 31 minutes and consecutive elution with 100% of the mobile phase A up to 45 minutes.
Metabolite profiling of urine samples was carried out using a Waters HPLC system (Waters) with Hydro-RP column (4 µm, 100 × 2.0 mm; Phenomenex) at a flow rate of 0.2 or 0.3 mL/min, radiochemical detector (β-RAM Model 4; IN/US Systems, Inc, Tampa, Florida), and Atlas Version 8.2 (Fisher Scientific, Pittsburgh, PA) control and data acquisition system. The mobile phase consisted of 0.1% acetic acid with 0.5% perfluoropentanoic acid in water (A) and 0.1% acetic acid in acetonitrile (B). The 80% of the mobile phase A was eluted for 2.5 minutes (at 0.2 mL/min), followed by a linear gradient to 60% of the mobile phase B (at 0.2 mL/min) at 4.5 minutes and a linear gradient to 20% of the mobile phase B (at 0.2 mL/min) at 8.5 minutes, and then with an isocratic elution for 2.9 minutes at a flow rate of 0.2 mL/min and 0.1 minutes at a flow rate of 0.3 mL/min with continuous elution at 0.3 mL/min for up to 17 minutes. Urine metabolite identification was carried out using an LC-MS/MS system (Sciex API 4000Q; AB Sciex, Framingham, Massachusetts) with Turbo Spray in positive ion mode, Hydro-RP (4 µm, 100 × 2.0 mm, Phenomenex) column, and Analyst Version 1.4.2 (AB Sciex) control and data acquisition system. The same mobile phase gradient as for radioprofiling (as described above) was used for LC-MS/MS analysis.
Calculations
Data from the dosing of each animal, weight of samples collected (eg, urine, feces, and tissue), and the concentration of radioactivity in samples were used to calculate the dose recovery. The amount of dose formulation administered was determined by weighing the full-dosing syringe prior to dose administration and the emptied syringe after dosing.
Concentration of radioactivity in individual samples was based on weight or volume. Samples with dpm less than twice the concurrently run background were considered to contain insufficient radioactivity for reliable quantification. When appropriate, results are expressed as the mean ± standard deviation. The total radioactivity recovered from each animal was calculated as the sum of the radioactivity found in urine, cage rinse, bile (rats only), feces, tissues (including blood), and carcass (rats only).
Results
The 14C-MMB4 DMS radiochemical purity was determined before each dosing formulation preparation and was in the range of 95.4% to 97.8%. All prepared dosing formulations were within 10% of the specified mass and radioactivity concentrations.
The group mean percentage of recoveries of the total administered 14C-MMB4 DMS-derived radioactivity in all treatment groups are presented in Tables 1 to 3. Group mean mass balance values were 85% or higher for all groups, except for the rat high-dose groups that had values ranging from 75% to 79%. For these 2 groups, the lower mass balance was attributed to the lower percentage of dose recovered in the urine.
Recovery of Radioactivity in Rat (Pilot Study).
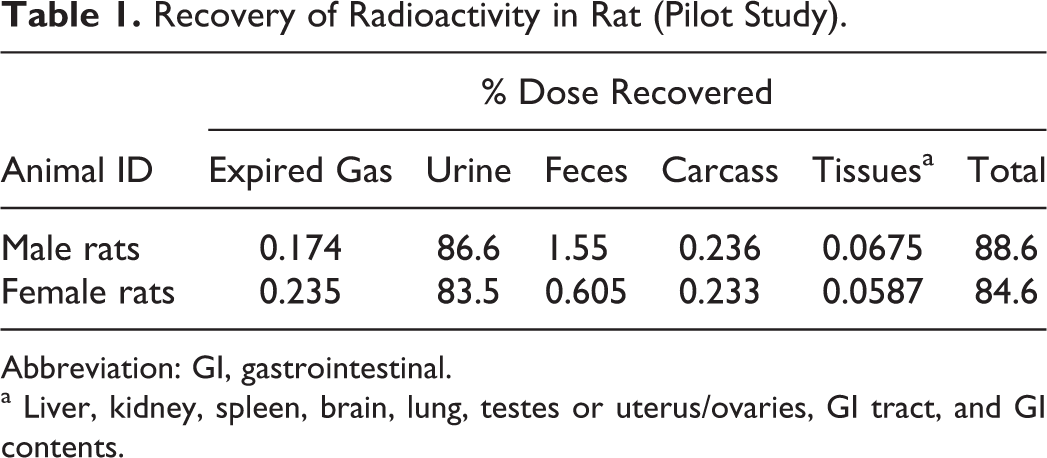
Abbreviation: GI, gastrointestinal.
a Liver, kidney, spleen, brain, lung, testes or uterus/ovaries, GI tract, and GI contents.
Recovery of Administered Dose in Rat (Definitive Study).
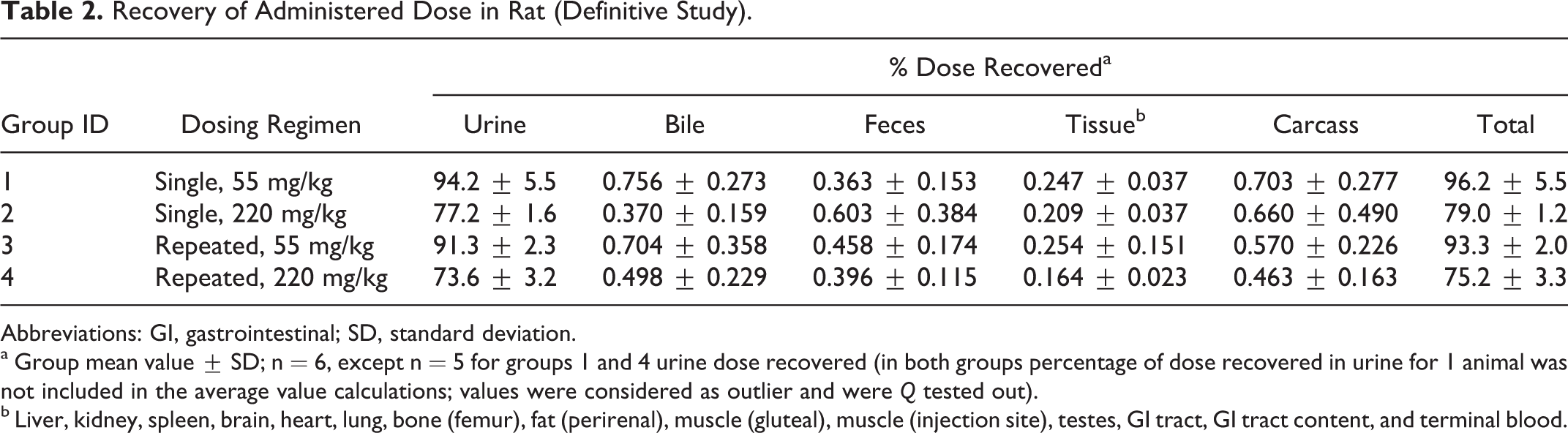
Abbreviations: GI, gastrointestinal; SD, standard deviation.
a Group mean value ± SD; n = 6, except n = 5 for groups 1 and 4 urine dose recovered (in both groups percentage of dose recovered in urine for 1 animal was not included in the average value calculations; values were considered as outlier and were
b Liver, kidney, spleen, brain, heart, lung, bone (femur), fat (perirenal), muscle (gluteal), muscle (injection site), testes, GI tract, GI tract content, and terminal blood.
Recovery of Administered Dose in Rabbit.
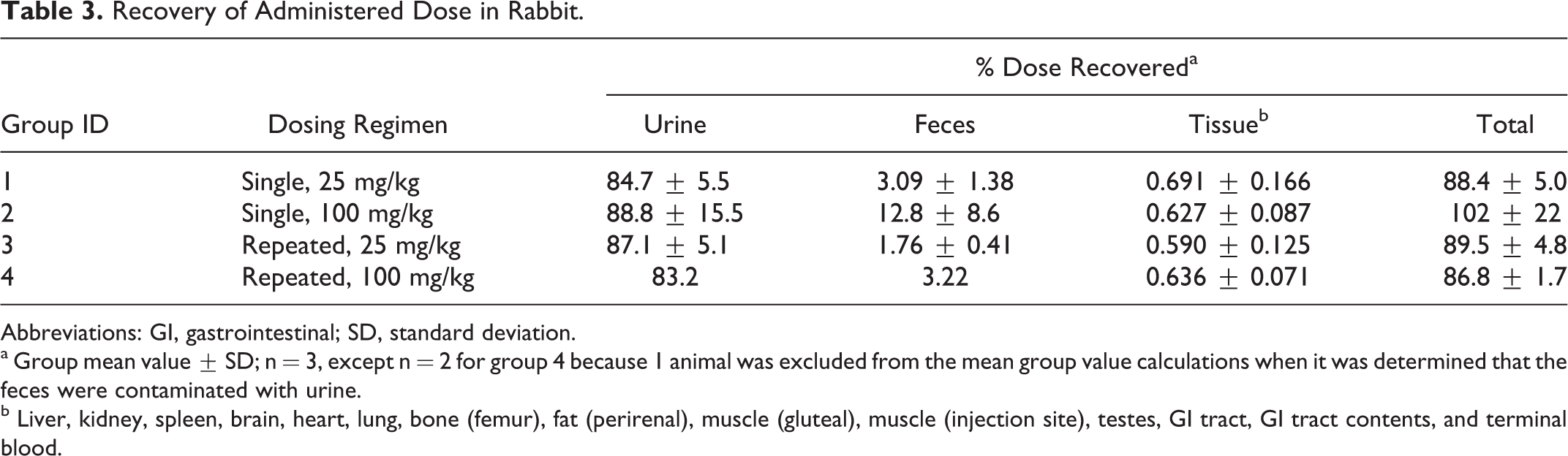
Abbreviations: GI, gastrointestinal; SD, standard deviation.
a Group mean value ± SD; n = 3, except n = 2 for group 4 because 1 animal was excluded from the mean group value calculations when it was determined that the feces were contaminated with urine.
b Liver, kidney, spleen, brain, heart, lung, bone (femur), fat (perirenal), muscle (gluteal), muscle (injection site), testes, GI tract, GI tract contents, and terminal blood.
The total 14C-MMB4 DMS-derived radioactivity recovered in the urine included the radioactivity measured in the cage rinse. The recovery of 14C-MMB4 DMS-derived radioactivity in rat urine per collection period in groups 1 to 4 is presented in Figure 2. For all the treatment groups (1-4), the highest amounts of radioactivity were excreted during the first 8 hours after dose administration, and greater than 90% of the radioactivity found in the urine was eliminated within 12 hours after dose administration.
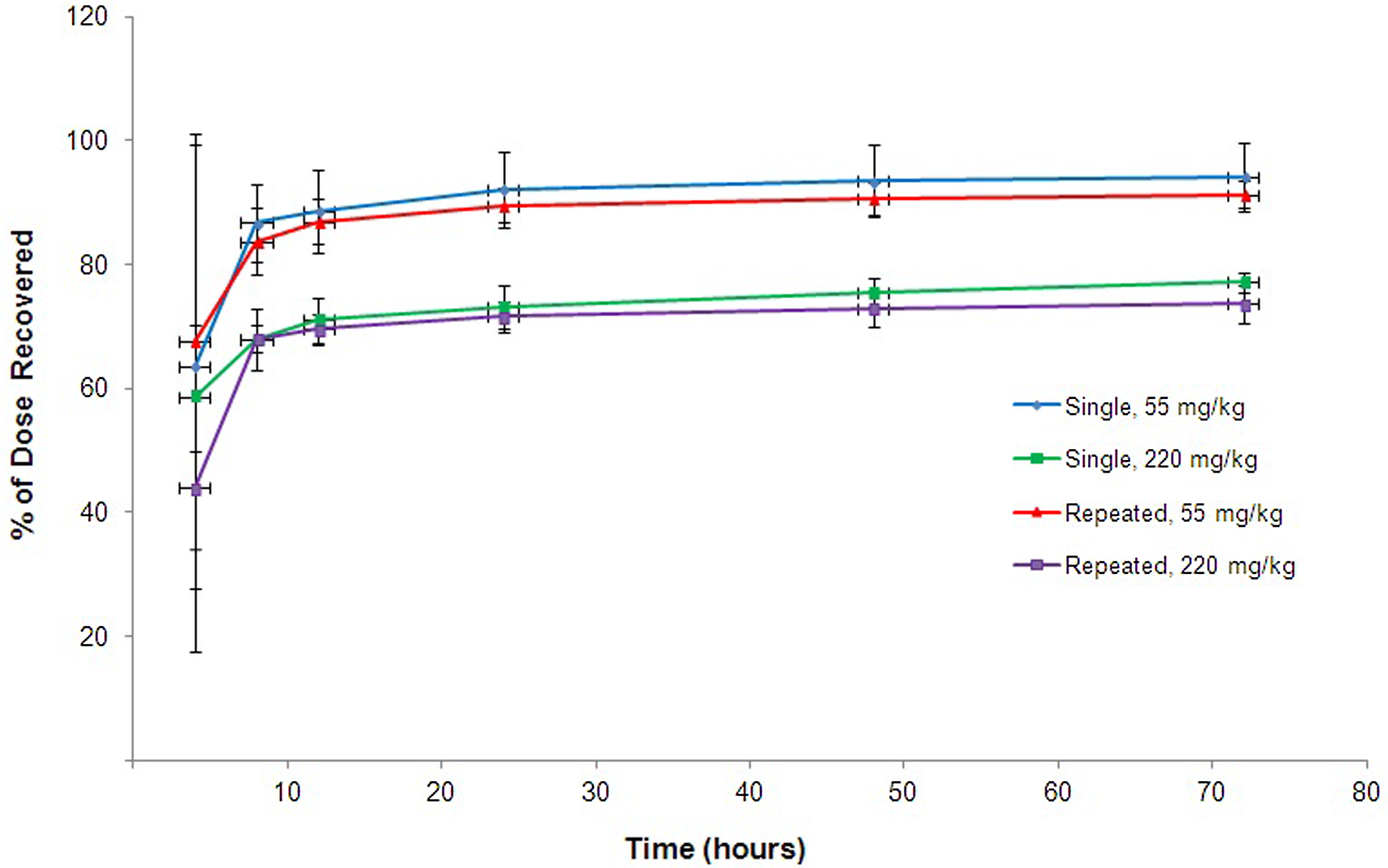
Average cumulative percentage of administered dose at specified collection intervals determined in rat urine after intramuscular (IM) administration of 14C-1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (14C-MMB4 DMS).
The recovery of 14C-MMB4 DMS-derived radioactivity in rabbit urine per collection period in groups 1 to 4 is presented in Figure 3. For all the treatment groups (1-4), the highest amounts of radioactivity were excreted during the first 8 hours after dose administration, and greater than 90% of the radioactivity found in the urine was eliminated within 8 hours after dose administration.
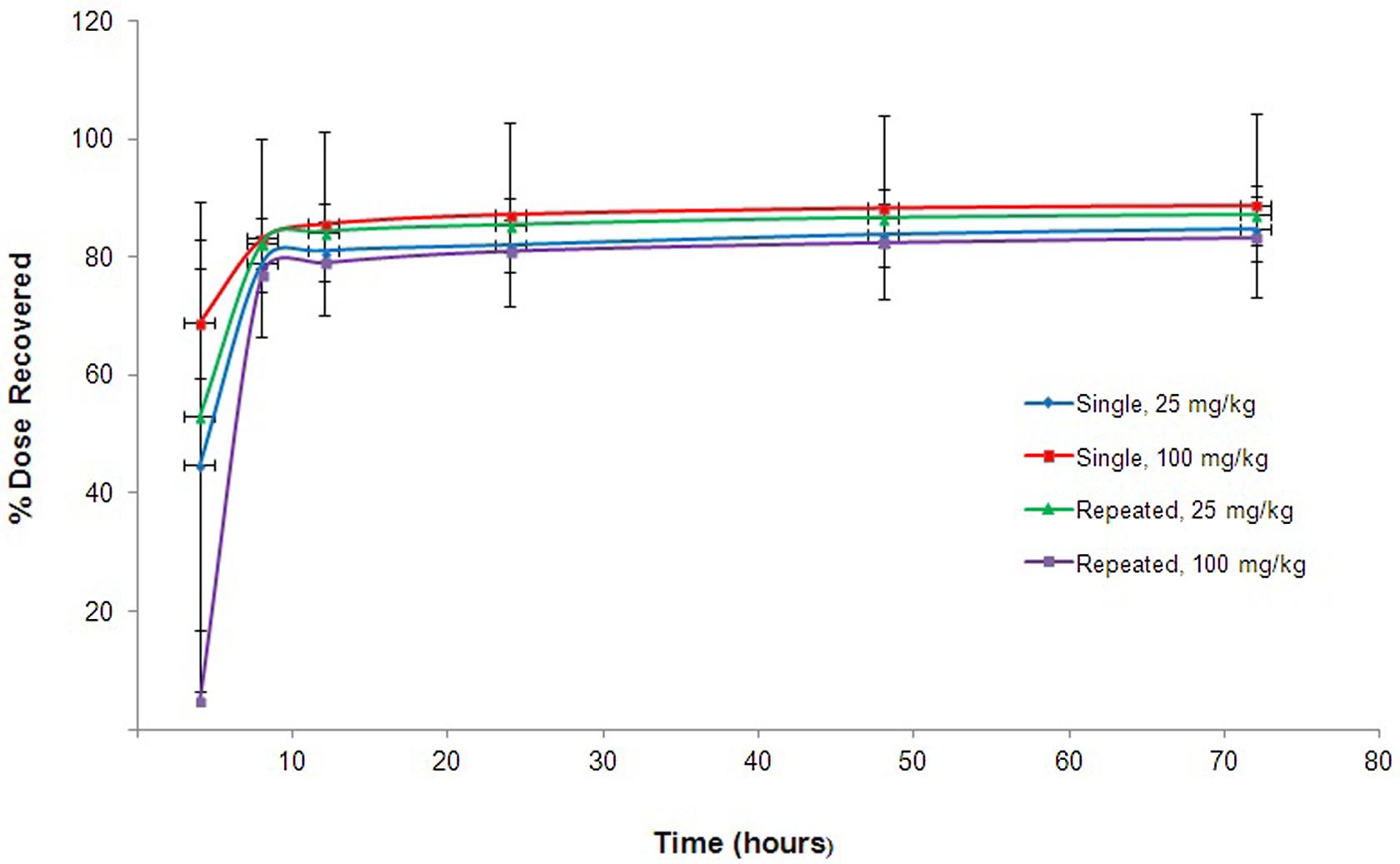
Average cumulative percentage of administered dose at specified collection intervals determined in rabbit urine after intramuscular (IM) administration of 14C-1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (14C-MMB4 DMS).
The recovery of 14C-MMB4 DMS-derived radioactivity in bile was determined only in rats, as shown in Supplementary Table 1. For all the treatment groups, the highest amounts of radioactivity were excreted during the first 8 hours after dose administration, and greater than 90% of the radioactivity found in the bile of all the rats (groups 1-4) was eliminated within 24 hours after dose administration.
The recovery of 14C-MMB4 DMS-derived radioactivity in rat and rabbit feces per collection period is presented in Supplementary Tables 2 and 3, respectively. Greater than 80% of the radioactivity found in the rat and rabbit feces from all the treatment groups was eliminated within 48 hours after dose administration.
The group average 14C-MMB4 DMS-derived radioactivity recovered in blood and tissues collected at the 72-hour termination time point was below 0.3% and 0.7% in rats and rabbits, respectively, as shown in Supplementary Tables 4 and 5. Liver was the tissue with the highest concentration of radioactivity at approximately 30% of the total radioactivity detected in all the tested rat and rabbit tissues.
The concentrations of radioactivity (µg equivalents 14C-MMB4 DMS/g) in blood, plasma, and tissues collected at 0.25, 2, 8, 24, 48 (rats), and 72 (rabbit) hours following IM administration (group 5) are presented in Supplementary Tables 6 and 7 for rat and rabbit, respectively.
The
Drug-derived radioactivity reached
The tissue–blood concentration ratios at the 0.25, 2, 8, 24, 48 (rats), and 72 (rabbits) hours collection time points are presented in Tables 4 and 5. For both the species, tissue–blood ratios were always higher than 1 for kidneys, injection site muscle(s), and liver for all collection time points except 0.25 hours and always lower than 1 for brain, heart, and fat.
The radioactivity levels for the purpose of metabolic profiling were adequate only for rat and rabbit urine samples collected between 0 and 24 hours. Feces, plasma, bile, and terminal blood samples were not subjected to metabolic profiling due to low total radioactivity recovered in those samples. The representative radiochromatographic profile for rat and rabbit urine is presented in Figure 4.
Tissue–Blood Concentration Ratios at Specified Collection Times After a Single Dose Administration of 14C-MMB4 DMS to Rats (Group 5, 220 mg/kg).
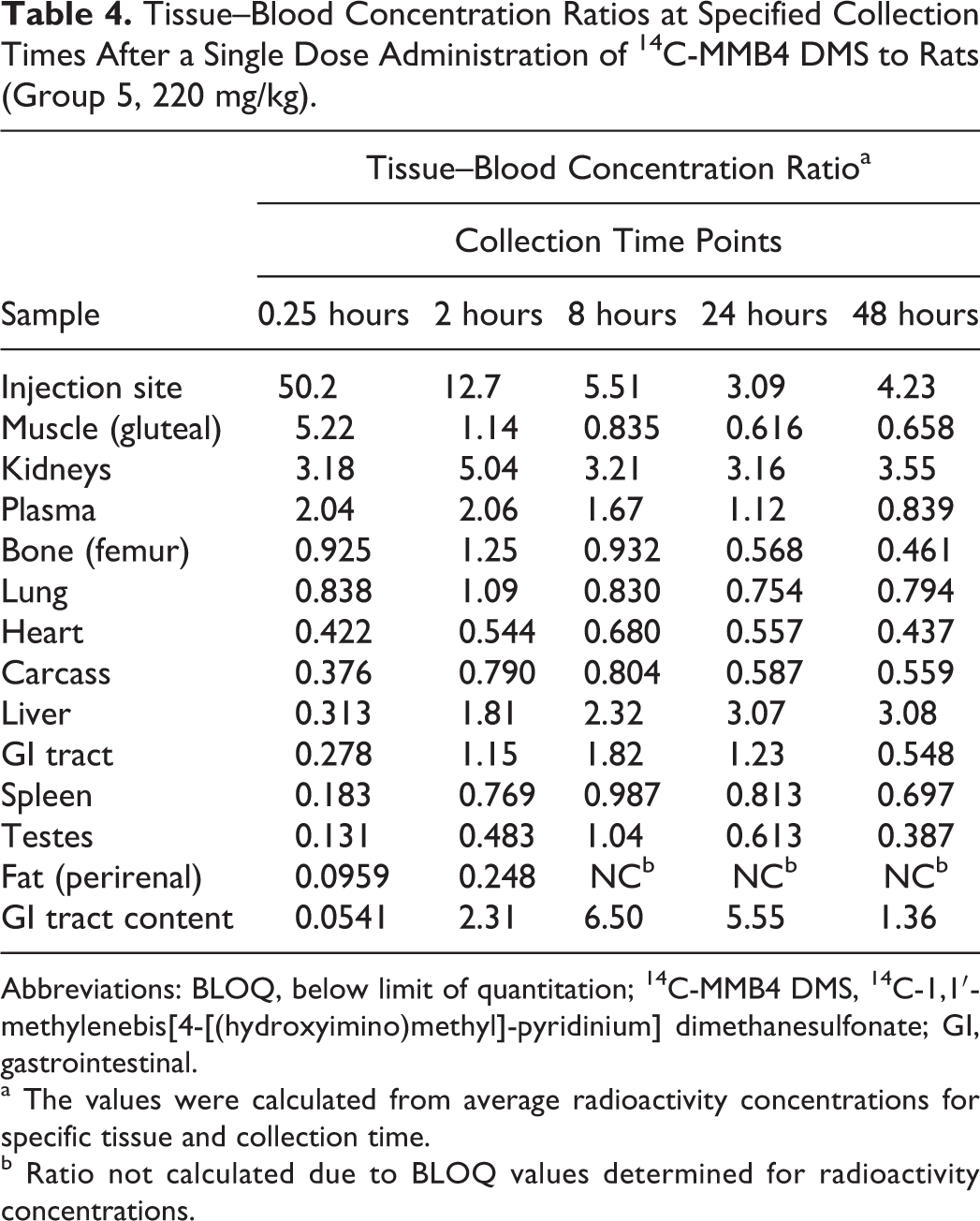
Abbreviations: BLOQ, below limit of quantitation; 14C-MMB4 DMS, 14C-1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate; GI, gastrointestinal.
a The values were calculated from average radioactivity concentrations for specific tissue and collection time.
b Ratio not calculated due to BLOQ values determined for radioactivity concentrations.
Tissue–Blood Concentration Ratios at Specified Collection Times After a Single Dose Administration of 14C-MMB4 DMS to Rabbits (Group 5, 100 mg/kg).
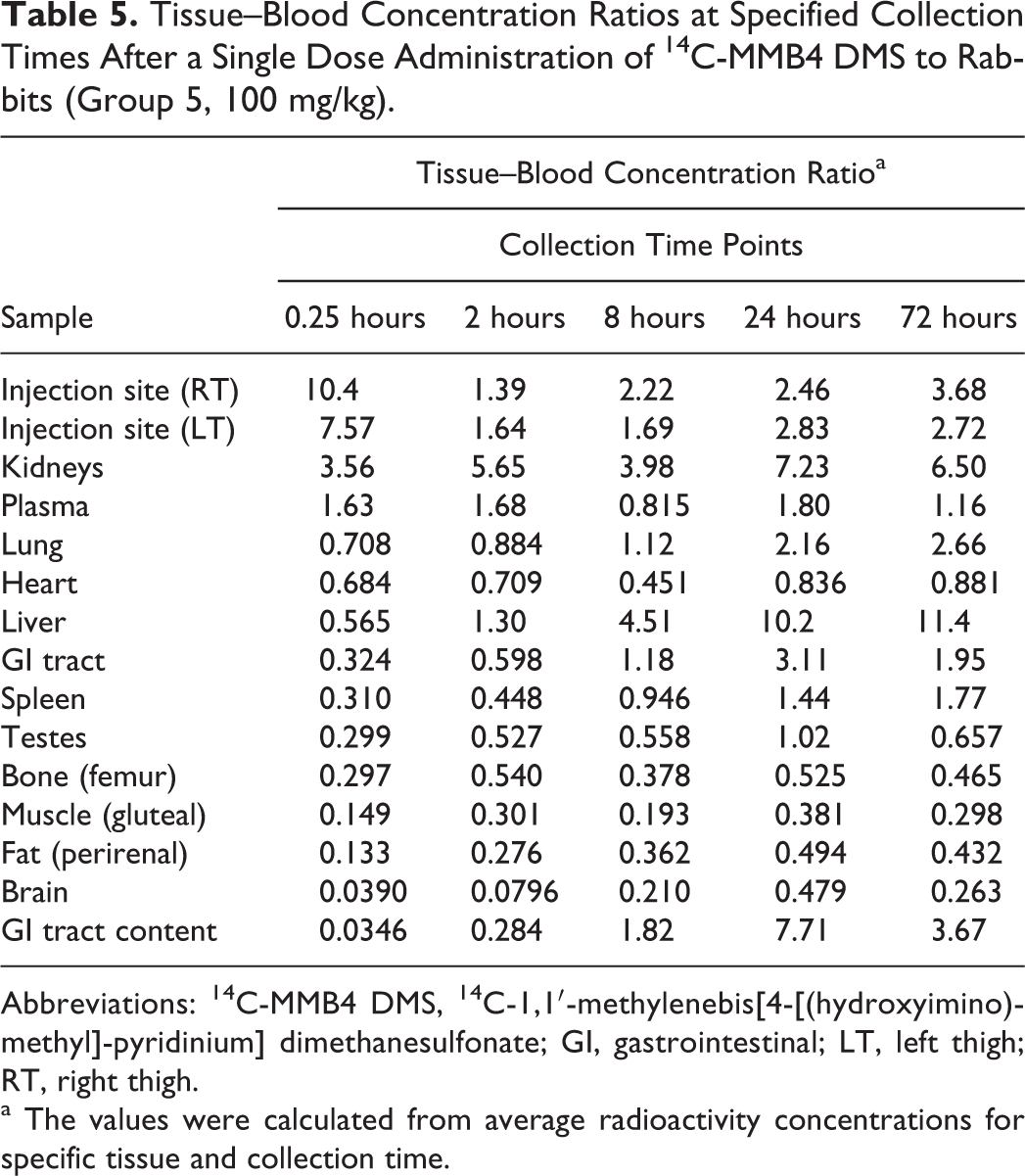
Abbreviations: 14C-MMB4 DMS, 14C-1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate; GI, gastrointestinal; LT, left thigh; RT, right thigh.
a The values were calculated from average radioactivity concentrations for specific tissue and collection time.
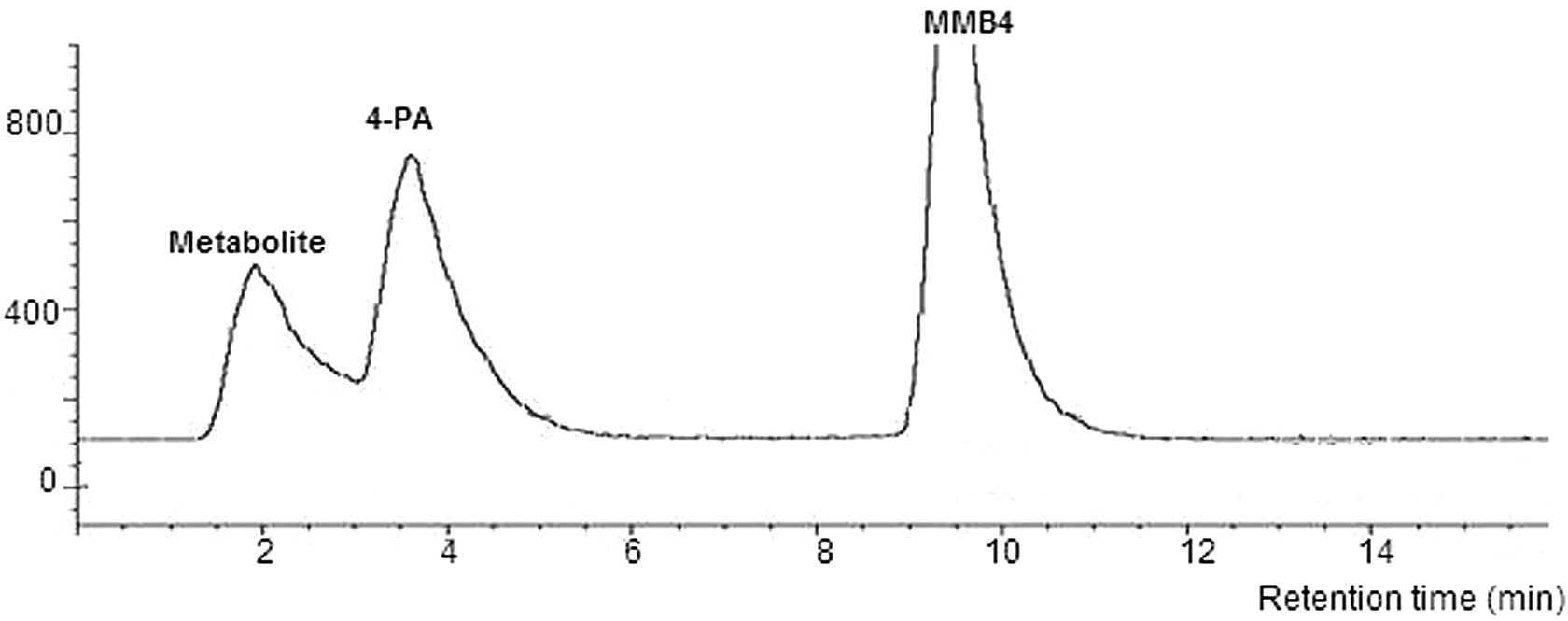
Radiochromatographic profile of representative urine sample.
The retention time of the MMB4 peak was approximately 10 minutes. Two additional peaks were observed at approximately 4 and 2 minutes. The peak with the retention time of approximately 4 minutes corresponded with that of 4-pyridine aldoxime (4-PA) standard, a synthetic starting material and a breakdown product of MMB4 DMS. 5 The structure of the metabolite with the approximate retention time of 2 minutes was assigned as isonicotinic acid (pyridine-4-carboxylic acid, or INA) based on the LC-MS/MS analysis for unknown metabolite and INA standard.
Representative urine LC-MS/MS chromatograms and mass spectra are presented in Figures 5 to 7. Panel A of Figure 5 represents a full total ion chromatogram (TIC) with the scan range of 100 to 300
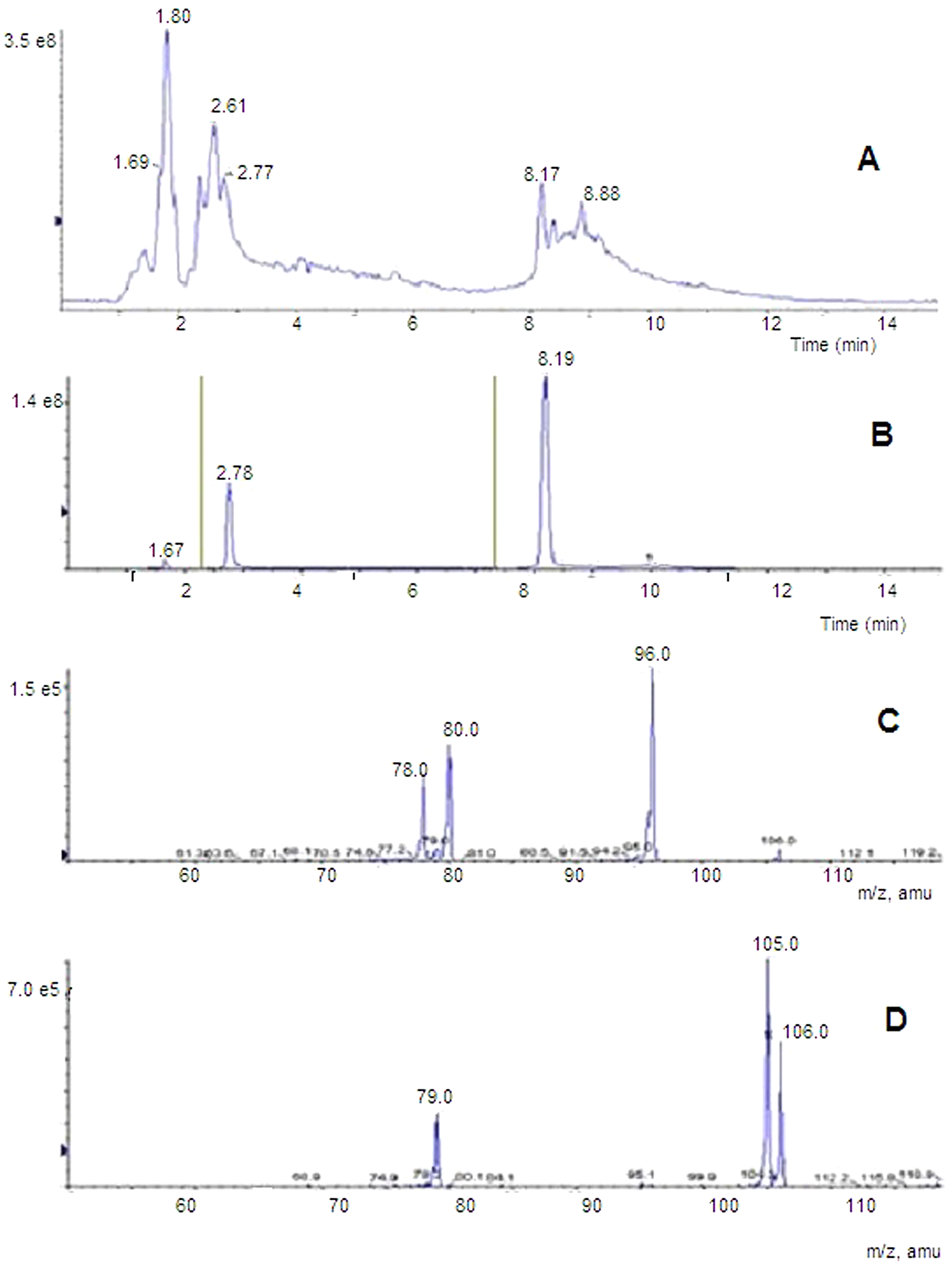
LC-MS/MS chromatogram of representative urine sample. A, LC-MS full-scan total ion chromatogram (TIC) with scan range 100 to 300
Panel C of Figure 5 shows the composite mass spectrum of the peak at approximately 1.7 minutes. Panel D of Figure 5 shows the composite mass spectrum of the peak at approximately 2.8 minutes. To confirm the structures of the detected metabolites (4-PA and INA), the TICs for both standard compounds (commercially obtained 4-PA and INA) were recorded (Figures 6A and 7A), and the respective mass spectra for both compounds were generated (Figures 6B and 7B). The mass spectra for standards 4-PA and INA were identical with those created for respective peaks from urine chromatograms (compare Figures 5C with 7B and Figures 5D with 6B).
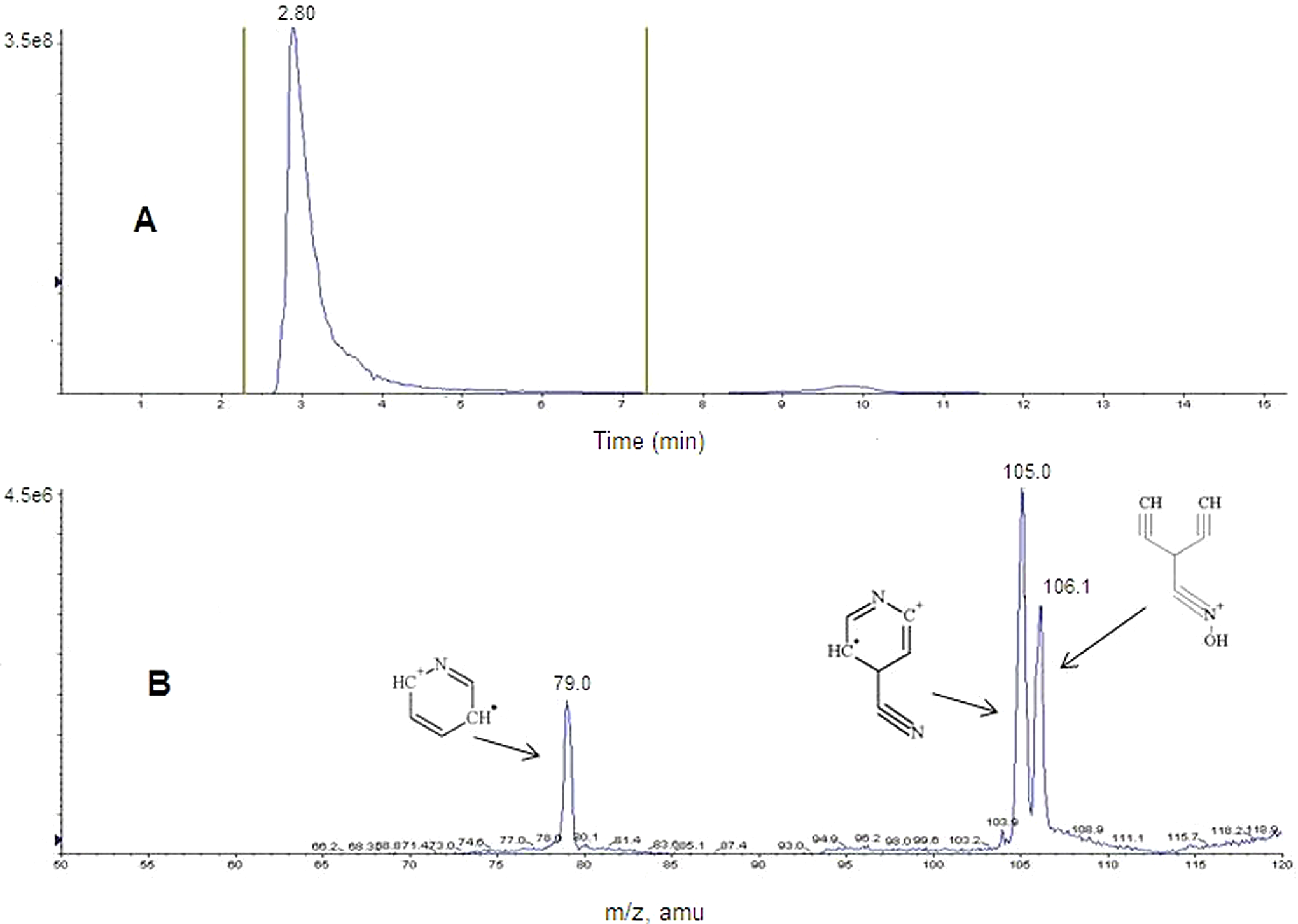
Liquid chromatography with tandem mass spectrometry (LC-MS/MS) chromatogram and mass spectrum of 4-PA. A, Total ion chromatogram (TIC) of the mass 123
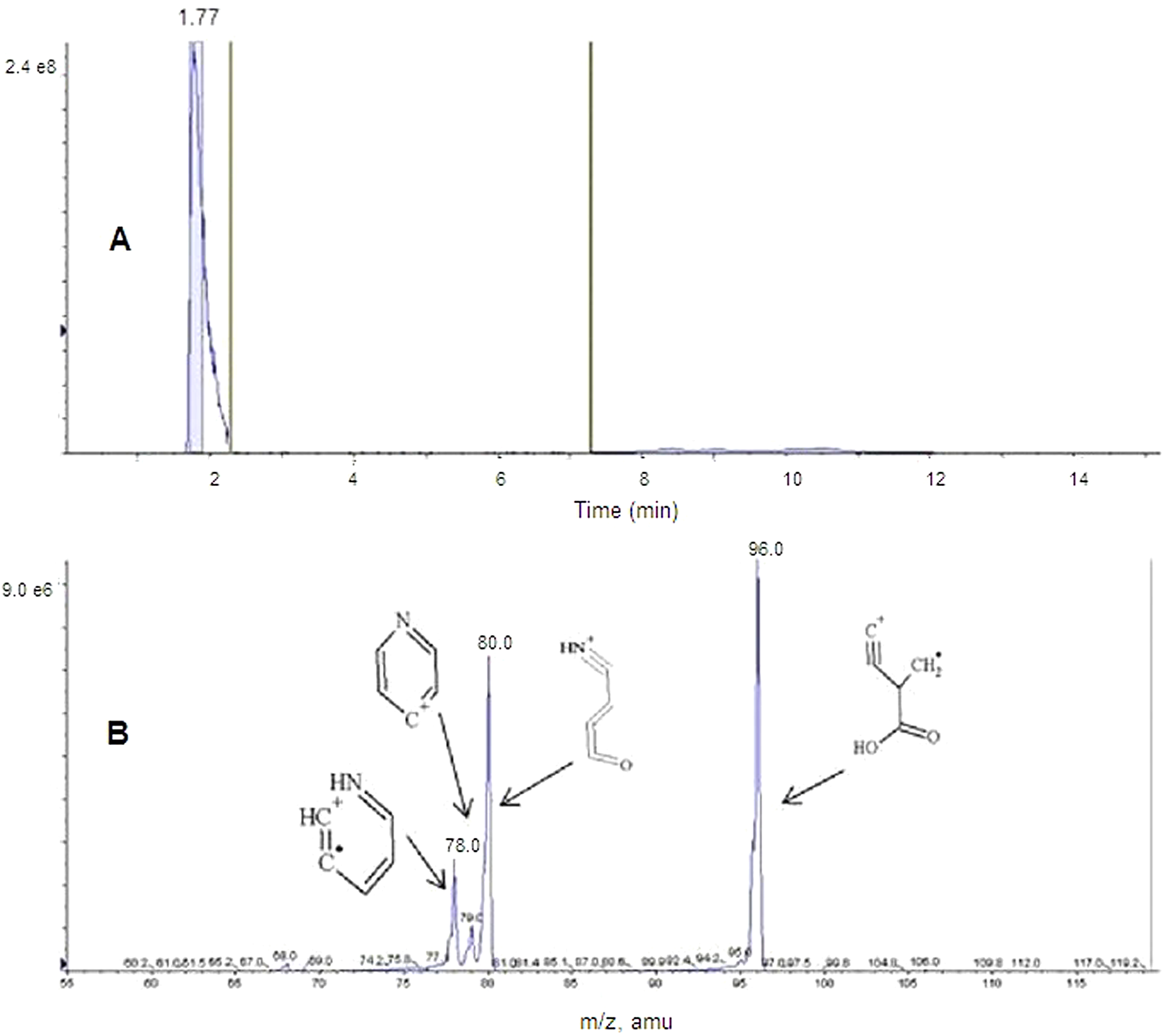
Liquid chromatography with tandem mass spectrometry (LC-MS/MS) chromatogram and mass spectrum of isonicotinic acid (INA). A, Total ion chromatogram (TIC) of the mass 124
Discussion
In the rat pilot study, the extent of the administrated radioactivity excreted as expired gases was lower than 0.25% (Table 1), the dose recovered in male and female rat urine was negligible in all urine samples collected between 72 and 168 hours postdose administration (data not shown), and no gender difference was observed in the radioactivity excretion pattern. Based on those results, experimental design considerations for the definitive study were to test only male rats, collect no expired gases, and collect excreta up to 72 hours.
There were no apparent differences in mass balance results between different species (rat and rabbit), gender (male and female rat, pilot rat study), single or repeated dose, and MMB4 DMS dosing concentration levels. A slight difference (approximately 18%) was observed in rats in the overall dose recovered between low (55 mg/kg, single and repeated) and high (220 mg/kg, single and repeated) treatment groups. An investigation into this finding did not result in any definitive explanation. Also, based on the findings, there was no evidence to support the possibility that this was a dose-related effect. No difference between different dosing concentration levels was observed in rabbits. The overall radioactivity recovery was approximately 88% (across both the species and all the treatment regimens).
The major elimination pathway of MMB4 DMS-derived radioactivity in rats and rabbits occurred via the kidney with an average greater than 97% (rat) and 93% (rabbit) of the eliminated radioactivity recovered in the urine. Peak recovery of the radioactivity excreted in the urine occurred within 8 hours after dose administration, and virtually all were recovered within 24 hours after administration (both rats and rabbits), thereby suggesting that MMB4 is rapidly removed from the body. The liver represented a minor pathway of elimination as the percentage of eliminated radioactivity was low: below 0.7% on average in both bile and feces in rats and below 6% in rabbit feces. This finding helps in explaining the presence of measurable radioactivity in the GI tract following IM administration. The average total amount of radioactivity determined in all the tissue samples (rat and rabbit, groups 1-4, 72 hours termination collection time point) was below 0.3% (rat) and 0.7% (rabbit). In rats, more than 29% of this radioactivity was detected in the liver. In rabbits, more than 47% was detected in the GI and GI tract contents, and liver was the tissue with the second highest concentration of radioactivity.
The distribution of 14C-MMB4 DMS-derived radioactivity was rapid. The drug-derived radioactivity was quantifiable in all analyzed matrices (except some rat perirenal fat and brain samples as shown in Supplementary Table 6) and reached the highest concentration at the first collection time point (0.25 hour), for all the analyzed matrices except GI track content (rat and rabbit, 2 hours) and liver (rat, 2 hours). The concentration of radioactivity decreased with time, was quantifiable, and was below 3% at 72 hours postdose (rabbit) and at 48 hours postdose (rat) when compared to its highest concentration (0.25 or 2 hours).
In both the studied species, 14C-MMB4 DMS-derived radioactivity was observed at low levels in testes and brain, suggesting that drug-derived radioactivity crossed the blood–testes and blood–brain barriers.
The tissue–blood concentration ratios were smaller than unity for the following rat and rabbit tissues: brain, fat (perirenal), and heart at all collection time points. High ratios were observed in injection sites (rat and rabbit), kidney (rat and rabbit), GI tract content (rat and rabbit), and liver (rabbit).
The urine metabolic profiles indicated the formation of 2 metabolites: one was positively identified as 4-PA (product of nonenzymatic degradation of MMB4 DMS) and the second was identified by LC-MS/MS (based on the comparison with the standard) as INA (pyridine-4-carboxylic acid). The identification of INA is in good agreement with the published data. 6 In the proposed mechanism of the INA formation, the aldoxime (4-PA) is enzymatically reduced under aerobic conditions into the respective intermediate imine, which is subsequently readily hydrolyzed to the aldehyde and then oxidized to the corresponding acid. 7
Regarding class effects, there were similarities in the ADME of MMB4 (bis-pyridinium bis-oxime) and HI-6 (bis-pyridinium mono-oxime). The MMB4 and HI-6 are structural analogs and, for this reason, it is not unexpected to find similar ADME properties. In the study published by Ecobichon et al, HI-6 absorption in rats, dogs, and rhesus monkeys following IM injection was fast, and distribution to the tissues was rapid. 2 The majority of the administered oxime dose was excreted within a few hours in the urine unchanged, although other products were detected. The highest concentrations of HI-6 were detected in the kidneys and the lowest in the brain. Three major metabolites of HI-6 were detected in plasma and urine, which included a spontaneous breakdown product (also detected in a buffered solution), picolinic acid (an analog of HI-6), and an unidentified third product. The picolinic acid analog of HI-6 was postulated to be an enzyme-catalyzed degradation product of HI-6. The picolinic acid analog is structurally related to the MMB4 metabolite, INA.
Footnotes
Acknowledgments
The authors thank D. Burnham, C. Mickolick, T. Poliquin, J. Smith, and J. Ross for their technical assistance and contributions to the conduct of the studies and Dr. V. J. Brown for his editorial review of the manuscript.
Authors’ Note
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or reflecting true views of the Department of the Army or the Department of Defense.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the United States Army; Chemical Biological Medical Systems (CBMS)/Medical Identification and Treatment Systems (MITS) under contract SP0700-00-D-D3180, Delivery Order 0599 (Task 771), and Delivery Order 0600 (Task 789).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
