Abstract
Acetylcholinesterase (AChE) reactivation studies were conducted in guinea pigs (GPs) and nonhuman primates (NHPs) to determine the 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate (MMB4 DMS) dose that reactivated at least 20% of blood AChE within 15 minutes following cyclosarin (GF) dosing (used as the criterion for efficacy). Male GPs and male rhesus macaques (NHPs) were pretreated with atropine 15 minutes prior to GF administration (1 × median lethal dose [LD50]) and MMB4 DMS 15 minutes following GF administration. The GP survival was 5 of 8, 8 of 8, 8 of 8, and 6 of 8 for the 0.75, 3.0, 6.0, or 12.0 mg/kg MMB4 DMS treatment groups, respectively. In NHPs, survival was 6 of 6 at 0.5, 1.2, 3.0, or 9.3 mg/kg MMB4 DMS, respectively, 24 hours post-challenge, with the majority of animals noted as clinically normal by 24 hours. Pharmacokinetic/pharmacodynamic modeling revealed that 1.8 mg/kg in GPs or 0.013 mg/kg in NHPs would result in an average 20% reactivation; human equivalent doses were calculated as 0.39 mg/kg (based on GP data) and 0.004 mg/kg (based on NHP data). The model suggested that MMB4 plasma concentrations of 1000 ng/mL and AChE reactivation of 80% would be most effective. Although a 0.5 mg/kg MMB4 DMS dose in NHPs resulted in 100% survival and an average of 78% AChE reactivation, adverse effects associated with GF administration were still observed 24 hours post-challenge (tremors, mydriasis, and weakness were observed in 3 of 6 animals). In comparison, 6 of 6 animals treated with 1.2 mg/kg MMB4 DMS were observed as clinically normal 24 hours post-challenge.
Keywords
Introduction
Nerve agents have been a potential threat to military personnel since the Second World War. With the advent of recent Middle Eastern conflicts and the rise in terrorism, the potential for civilian exposure as well as the military threat is now of growing concern.
The toxicity of organophosphorus nerve agents is based on their ability to bind to acetylcholinesterase (AChE), resulting in the accumulation of acetylcholine at cholinergic nerve endings and their effector organs or postsynaptic sites. The build up of acetylcholine at cholinergic receptor sites is equivalent to continuous stimulation of these sites by the central nervous system (CNS) or peripheral nervous system (PNS). Common signs include increased secretions, fasciculations, seizures, convulsions, respiratory distress, and death. Antidotes used in treating organophosphorus toxicity include a combination of an anticholinergic drug (eg, atropine), an oxime that reactivates inhibited AChE, and an anticonvulsant.
The main mechanism of action for oximes include reactivation of AChE through the removal of the phosphoryl- or phosphonyl moiety from the serine active site of the enzyme, prior to irreversible AChE inhibition (eg, aging). The current US treatment for nerve agent poisoning is administration of atropine, 2-pralidoxime chloride (2-PAM), and an anticonvulsant. 2-PAM is a poor AChE reactivator for certain nerve agents 1 ; therefore, candidate replacements for 2-PAM are currently being investigated by the United States. 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate (MMB4 DMS; methoxime) is a bis-pyridinium oxime that has the potential to reactivate AChE following nerve agent poisoning. A number of laboratory studies indicate that MMB4 may be a more efficacious reactivator of AChE than 2-PAM, especially for certain highly toxic nerve agents. 2, 3 MMB4 DMS is being developed to replace the currently fielded 2-PAM in the Antidote Treatment Nerve Agent, Autoinjector (ATNAA). MMB4 DMS is intended to be used in conjunction with atropine at the first sign of nerve agent poisoning.
The objective of these guinea pig (GP) and nonhuman primate (NHP) studies was to determine the minimum effective dose of MMB4 DMS that would reactivate 20% of the blood AChE within 15 minutes following exposure to 1 × median lethal dose (LD50) cyclosarin (GF). This criterion for efficacy was an internally defined guideline for effectiveness, in which 20% AChE reactivation was associated with survival. In both the studies, the highest MMB4 DMS dose selected was less than or equal to the human equivalent dose (HED) of 3 mg/kg according to the US Food and Drug Administration. 4 This translated to high doses of 13.8 mg/kg and 9.3 mg/kg in the GP and NHP, respectively. Doses of 12 mg/kg in the GP and 9.3 mg/kg in the NHP were used as the upper doses for MMB4 DMS.
This study was conducted following Good Laboratory Practice requirements so that experimental data generated from this study can be included in the Investigational New Drug application. 5 The study was conducted in compliance with The American College of Toxicology Policy on the Use of Animals, under the guidance of the Battelle Institutional Animal Care and Use Committee and the Animal Care and Use Review Office of the US Army.
Materials and Methods
Animals
Guinea Pig Study
Male Hartley GPs (
Nonhuman Primate Study
Male Rhesus macaques of Indian origin (
Challenge Agent
The nerve agent GF ([fluoro-methyl-phosphoryl]oxycyclohexane) was administered to both GPs and NHPs at a target dose of 1.0 × LD50 (57 and 46.6 µg/kg, respectively). 6,7 The agent was administered by the subcutaneous route on the dorsum of the GP and by the intramuscular (IM) route on the left calf of the NHP. Dose confirmation samples were collected prior to and following the challenge to verify challenge agent.
Therapeutics
Atropine sulfate (Butler Animal Health Supply; 15 mg/mL) was administered IM in the GP in the left hind leg at a dose of 3 mg/kg (HED of 0.65 mg/kg). In the NHP, atropine sulfate was administered IM in the left thigh muscle at a dose of 1 mg/kg (HED of 0.32 mg/kg).
MMB4 DMS is described as an off-white to tan crystalline powder based on the Certificate of Analysis. Lot no. 1004 MMB4 DMS, with a manufacture date of July 2007, was used for this study. The source of the test article was Cambrex Charles City, Inc (Charles City, Iowa). The purity of the test article was >98%. MMB4 DMS was prepared in 5% benzyl alcohol in water, and methane sulfonic acid was used to adjust pH to 2.3 when required. The MMB4 DMS was administered IM in the right hind leg of the GP at doses of 0.75, 3.0, 6.0, or 12.0 mg/kg (HED of 0.16, 0.65, 1.30, and 2.61 mg/kg, respectively) and in the right thigh muscle of NHPs at 0.5, 1.2, 3.0, or 9.3 mg/kg (HED of 0.16, 0.39, 0.97, and 3.0 mg/kg, respectively). Dose confirmation samples were collected prior to and following challenge to verify therapeutic levels.
Experimental Design
Four groups of 8 GPs or 4 groups of 6 NHPs were injected with atropine sulfate 15 minutes prior to being dosed with GF. After GF administration (15 minutes for GPs or 5 minutes for NHPs), the animals were injected with MMB4 DMS. Blood was collected prior to administration of GF and prior to MMB4 DMS dosing and at target times of 5, 15, 30, 60, and 90 minutes after MMB4 DMS dose administration. The pre-GF administration blood sample was used to determine baseline AChE activity and MMB4 plasma levels, whereas the pre-MMB4 DMS administration blood sample was used to determine the maximum effect of GF on AChE activity prior to administration of the MMB4 DMS. The post-MMB4 DMS administration blood samples were taken to determine the change in AChE activity (relative to baseline) and MMB4 plasma concentration–time profile. Clinical observations were recorded prior to GF dosing and prior to and at 5, 15, 30, 60, and 90 minutes after MMB4 DMS administration. Additional clinical observations at 4, 6, and 24 hours were recorded for NHPs.
Walter Reed Army Institute of Research Assay
The Walter Reed Army Institute of Research whole blood ChE assay 8 was used to determine AChE and butyrylcholinesterase activity in the whole blood samples. Whole blood was added to a 96-well microplate, and the reaction initiated with the addition of acetylthiocholine iodide, propionylthiochoine iodide, s-butyr-ylthiocholine iodide, and 4,4′-dithiodipyridine. The absorbance was measured at 17-second intervals at 320 nm for approximately 4 minutes to determine cholinesterase activity. Additionally, the absorbance was measured at 450 nm following the kinetics evaluation to determine background levels for hemoglobin.
Pharmacokinetic/Pharmacodynamic Analysis
Pharmacokinetic/pharmacodynamic (PK/PD) analysis was performed using the sample collection time points (minutes) and the measured concentrations of MMB4 in plasma (ng/mL). The MMB4 concentration–time profiles were evaluated using noncompartmental analysis modules in the WinNonlin software program (version 5.0.1; Pharsight Corporation, Mountain View, California).
Group mean concentrations of MMB4 in plasma were evaluated against reactivation of AChE values at each sample collection time for all the dose groups combined. The PK parameters generated and evaluated were Cmax, Tmax, Tlast, and area under the plasma concentration–time curve from 0 time until the last (AUClast).
The concentration–reactivation profiles were analyzed using 2 PD modeling modules (Emax models with and without γ, the shape parameter) in the WinNonlin software program. The Emax, half maximal effective concentration (EC50), and γ, when appropriate, were reported along with goodness-of-fit measures such as Akaike Information Criterion and Schwartz Bayesian Criterion. Based on the PK/PD parameters, AChE reactivation EC20, EC50, and EC80 were derived and reported. The EC50 calculated from the WinNonlin represents the concentration that achieves 50% of Emax, not necessarily of control AChE activity levels.
Results
Survival
In the GP and NHP, 24-hour survival following 1 × LD50 GF challenge when pre-treated with atropine and post-treated with MMB4 DMS is summarized in Table 1.
The 24-Hour Survival Following GF Challenge and Treatment With Atropine and MMB4 DMS in Guinea Pigs and Monkeys.a
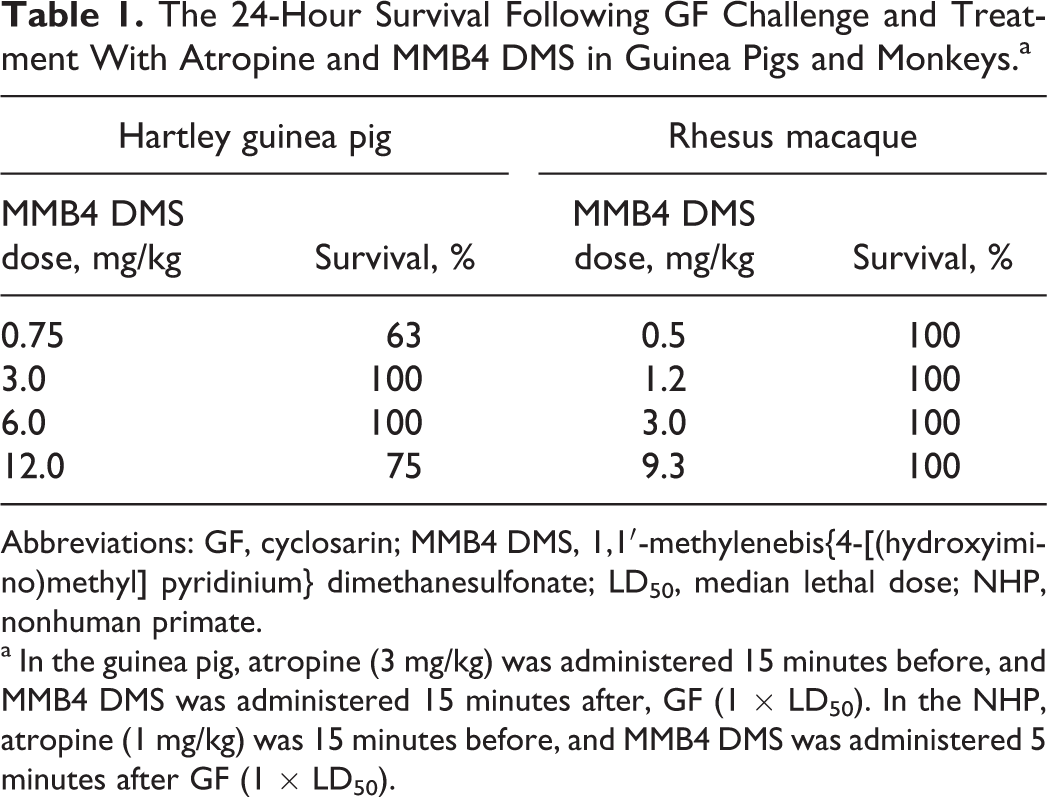
Abbreviations: GF, cyclosarin; MMB4 DMS, 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate; LD50, median lethal dose; NHP, nonhuman primate.
a In the guinea pig, atropine (3 mg/kg) was administered 15 minutes before, and MMB4 DMS was administered 15 minutes after, GF (1 × LD50). In the NHP, atropine (1 mg/kg) was 15 minutes before, and MMB4 DMS was administered 5 minutes after GF (1 × LD50).
Clinical Observations
In the GP and NHP studies, no overt effects were observed following atropine pretreatment. Following GF challenge, GPs exhibited signs of nerve agent toxicity including chewing and to a lesser extent weakness, lethargy, ataxia, fasciculations, tremors, prostration, and respiratory distress. It is suspected that the incidence and severity of signs was suppressed due to atropine pretreatment. MMB4 DMS did not appear to alleviate any GF-related signs through 90 minutes post-administration in the GP. Animals surviving to 24 hours post-challenge exhibited behavior similar to that observed pre-challenge.
In the NHP, clinical signs following GF exposure included fasciculations, mydriasis, tremors, and to a lesser extent weakness/lethargy, respiratory distress, and unresponsiveness. Following MMB4 DMS administration, a temporal-related decrease in the incidence and severity of GF-related clinical observations was apparent when evaluated by dose. In the 1.2 mg/kg MMB4 DMS dose group, cessation of fasciculations occurred within 4 hours post-challenge. In comparison, in the 3.0 and 9.3 mg/kg MMB4 DMS dose groups, fasciculations ceased within 90 and 60 minutes, respectively. When comparing the time it took for the animals to exhibit behavior consistent with that pre-challenge, a dose response was also noted. Although only 3 of 6 monkeys in the 0.5 mg/kg dose group appeared normal at 24 hours, 6 of 6, 5 of 6, and 4 of 6 appeared normal at 24 hours in the 1.2, 3.0, and 9.3 mg/kg dose groups, respectively.
Importantly, in the 2 highest dose groups, animals not recorded as normal were only observed to have mydriasis (one slight) or hunched posture. If mydriasis were not taken into account, the numbers of animals reported as normal in the 3.0 and 9.3 mg/kg dose groups would be 6 of 6 and 5 of 6, respectively, with the remaining animal at 9.3 mg/kg exhibiting only hunched posture. Up to 12 minutes following GF challenge, 8 of 24 monkeys on the study (across all dose groups) were observed to have stopped breathing, were unresponsive, moribund, or had respiratory distress. Remarkably, no later than 24 hours postchallenge, 6 of these 8 animals displayed normal behavior. The remaining 2 had mild signs (mydriasis and slight/intermittent tremors) and were in the lowest MMB4 DMS dose group.
Acetylcholinesterase Reactivation
In both the GP and the NHP studies, AChE activity was substantially inhibited by GF (91% average inhibition in the GP and 98% average inhibition in the NHP; Figures 1 and 2).
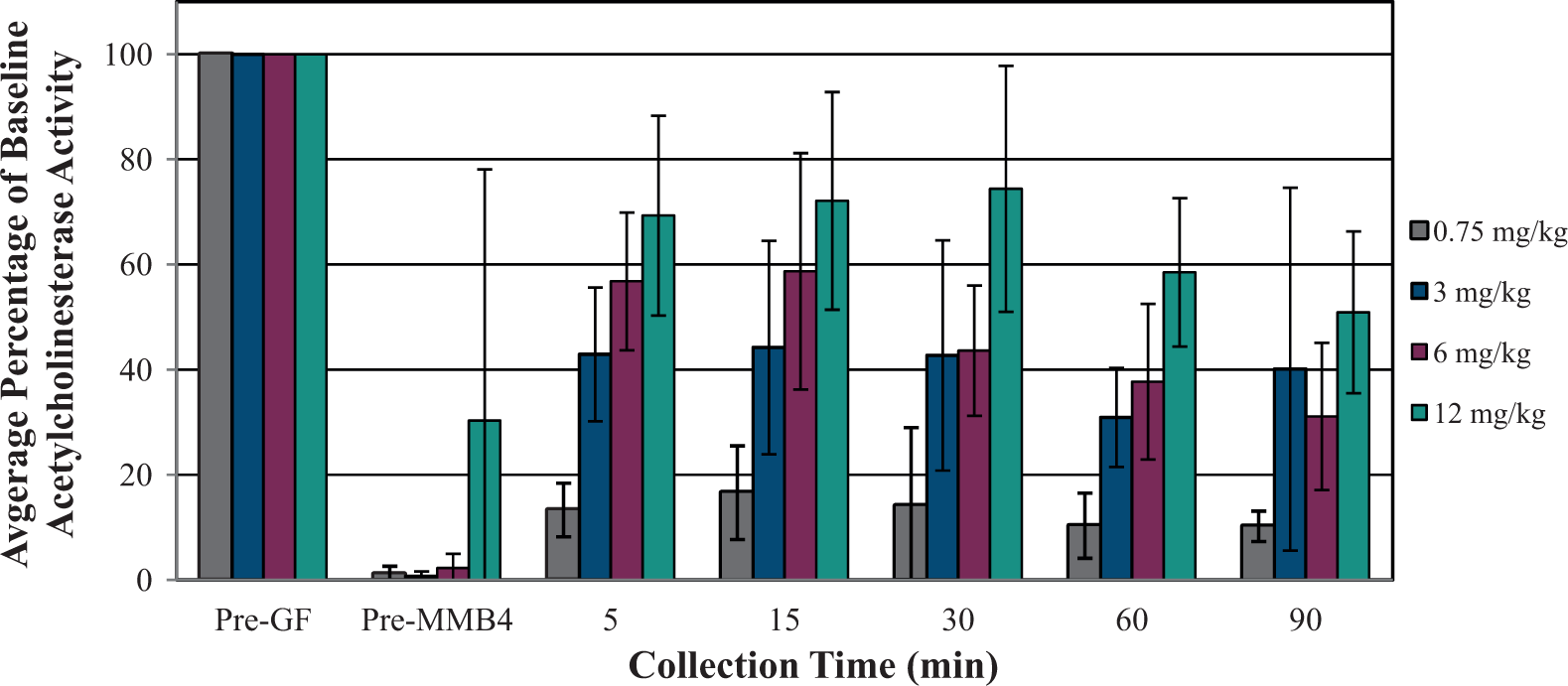
Blood acetylcholinesterase (AChE) reactivation after inhibition by cyclosarin (GF; 1 × median lethal dose [LD50], subcutaneous [SC]) and treatment with 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate (MMB4 DMS) in atropinized male guinea pigs.
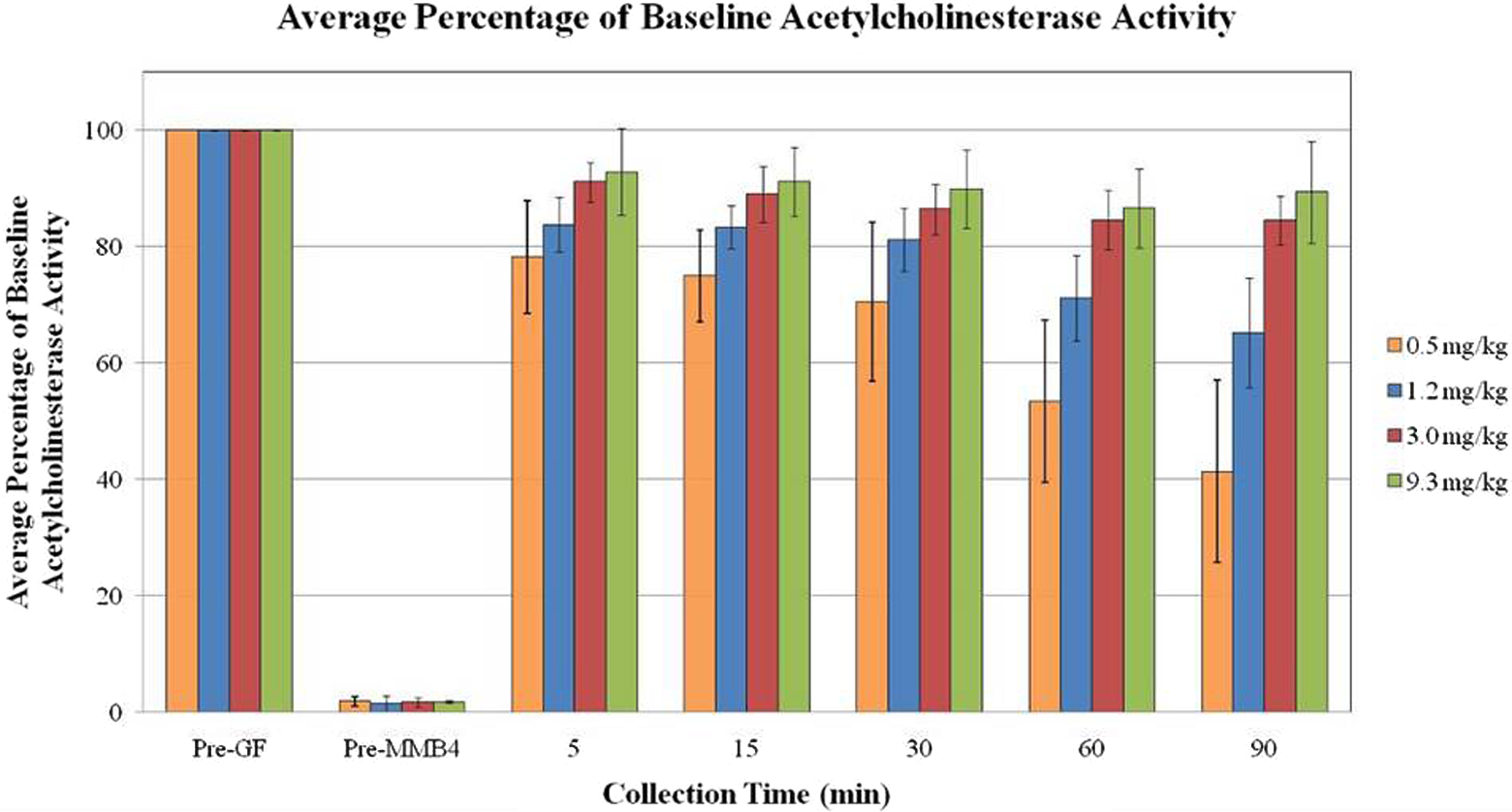
Blood acetylcholinesterase (AChE) reactivation after inhibition by cyclosarin (GF; 1 × median lethal dose [LD50], intramuscular [IM]) and treatment with 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate (MMB4 DMS) in atropinized male monkeys.
In the GP, AChE inhibition was substantially reversed by MMB4 DMS treatment at all dose levels evaluated. At all but the lowest MMB4 DMS dose level (0.75 mg/kg), at least 20% AChE reactivation was achieved within 15 minutes post-administration, and AChE activity remained above 20% through the last blood draw time point at 90 minutes post-challenge.
In the NHP, AChE inhibition was substantially reversed by MMB4 DMS treatment at all dose levels evaluated. All MMB4 DMS dose levels resulted in ≥78% AChE reactivation within approximately 5 minutes after administration. At all dose levels, the average percentage of baseline AChE activity remained above 20% through approximately 90 minutes after MMB4 DMS administration, which was the last collection time point. In addition, the 3.0 and 9.3 mg/kg MMB4 DMS dose levels resulted in a sustained AChE activity above 84% through at least 90 minutes post-administration.
MMB4 Plasma Concentration and PK/PD Analysis
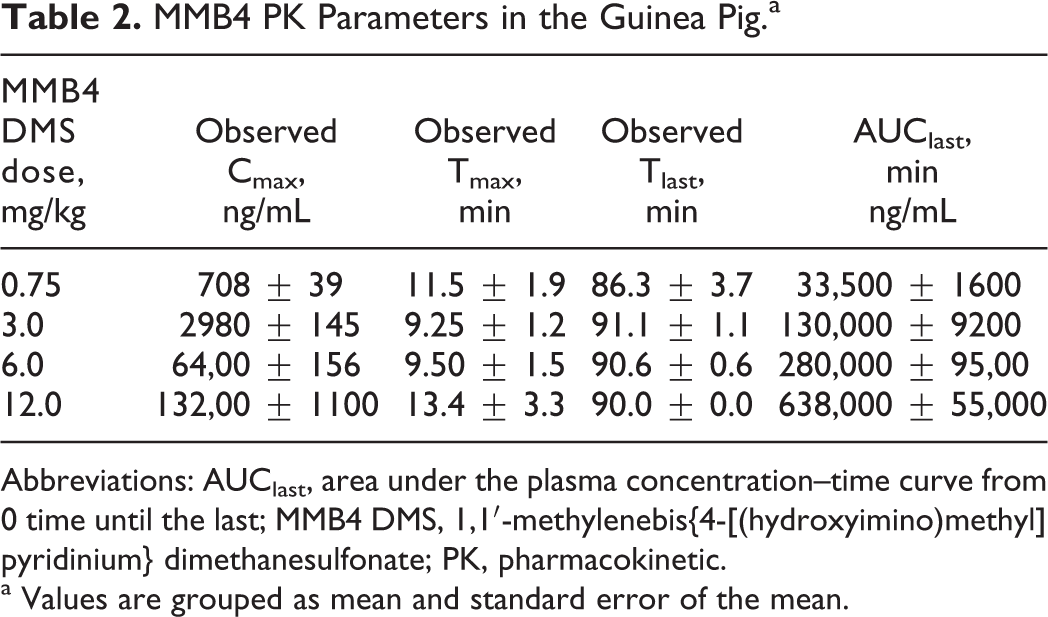
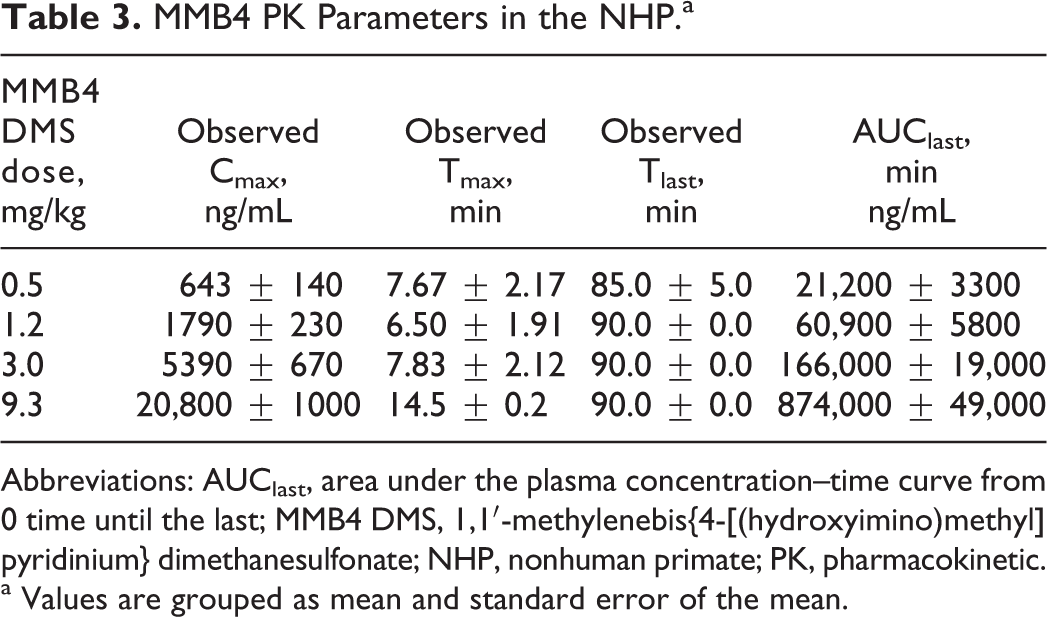
The MMB4 plasma concentration–time profiles for the GP and NHP are found in Figures 3 and 4. The MMB4 PK parameters were determined based on the concentration–time profiles and in both the GP and the NHP increased in a dose-proportional manner based on Cmax and AUClast results (Tables 2 and 3).
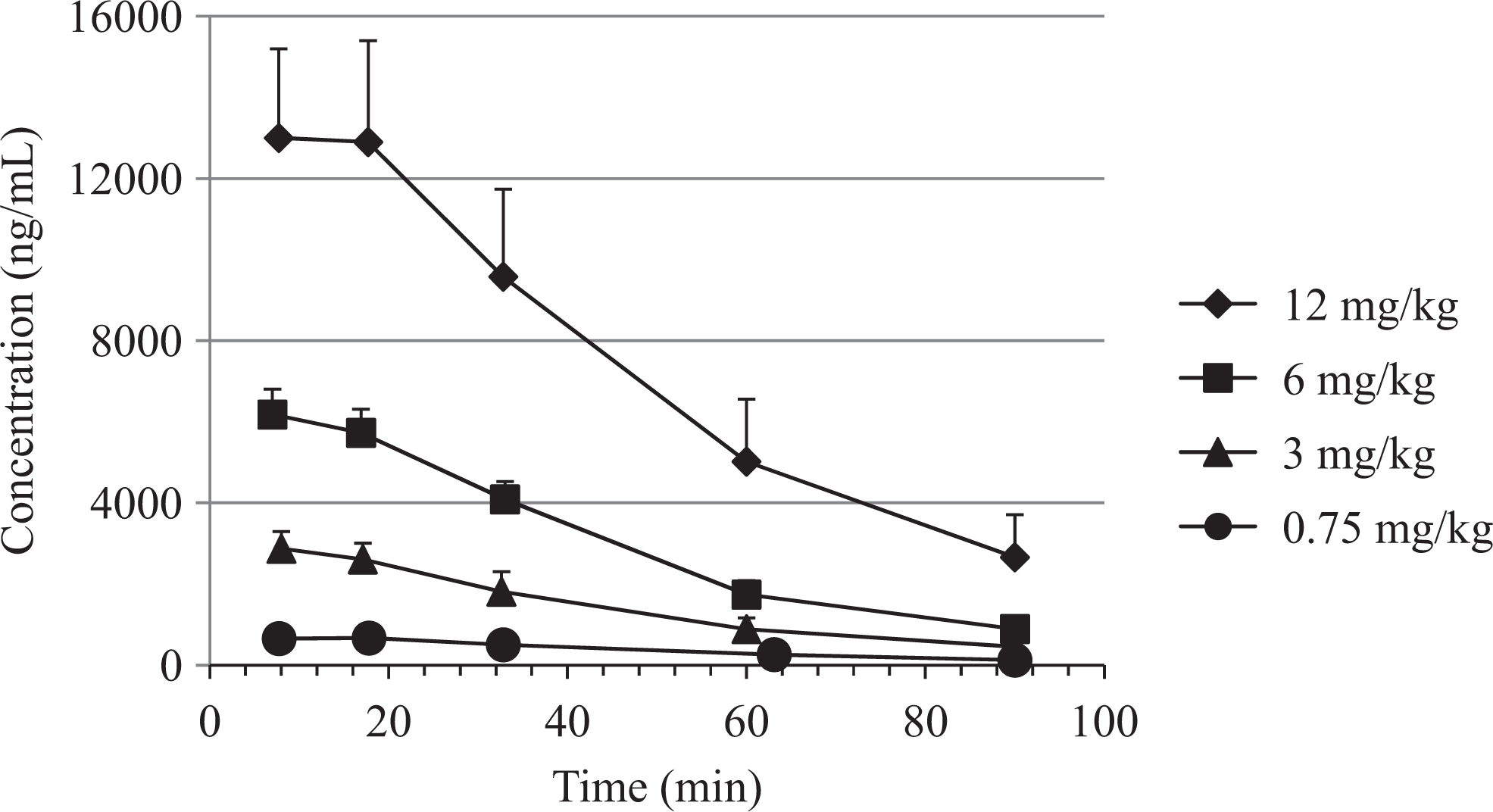
MMB4 plasma concentrations (mean ± standard error of the mean) after cyclosarin (GF; 1 × median lethal dose [LD50]) and treatment with MMB4 DMS in atropinized guinea pigs. MMB4 DMS indicates 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate.
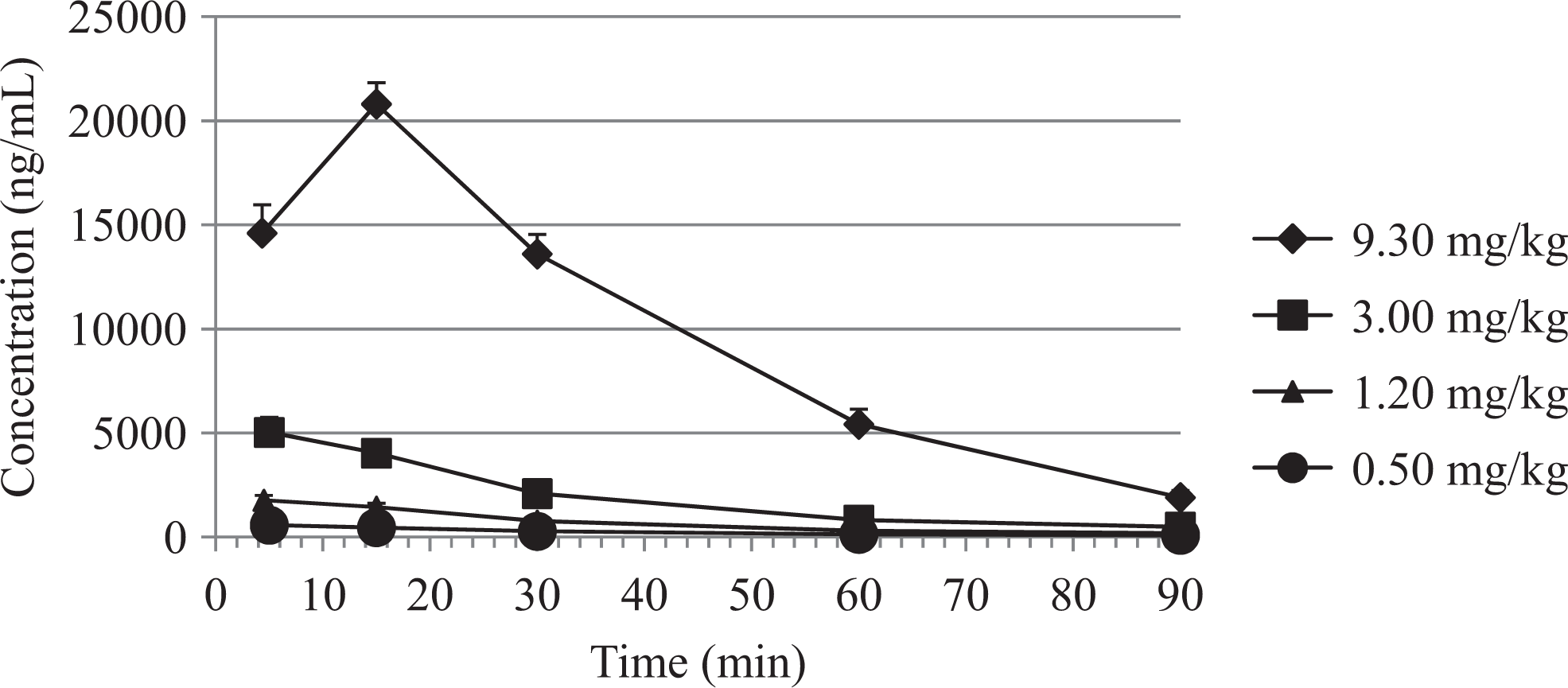
MMB4 plasma concentrations (mean ± standard error of the mean) after cyclosarin (GF, 1 × median lethal dose [LD50]) and treatment with MMB4 DMS in atropinized male monkeys. MMB4 DMS indicates 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate.
MMB4 PK Parameters in the Guinea Pig.a
Abbreviations: AUClast, area under the plasma concentration–time curve from 0 time until the last; MMB4 DMS, 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate; PK, pharmacokinetic.
a Values are grouped as mean and standard error of the mean.
MMB4 PK Parameters in the NHP.a
Abbreviations: AUClast, area under the plasma concentration–time curve from 0 time until the last; MMB4 DMS, 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate; NHP, nonhuman primate; PK, pharmacokinetic.
a Values are grouped as mean and standard error of the mean.
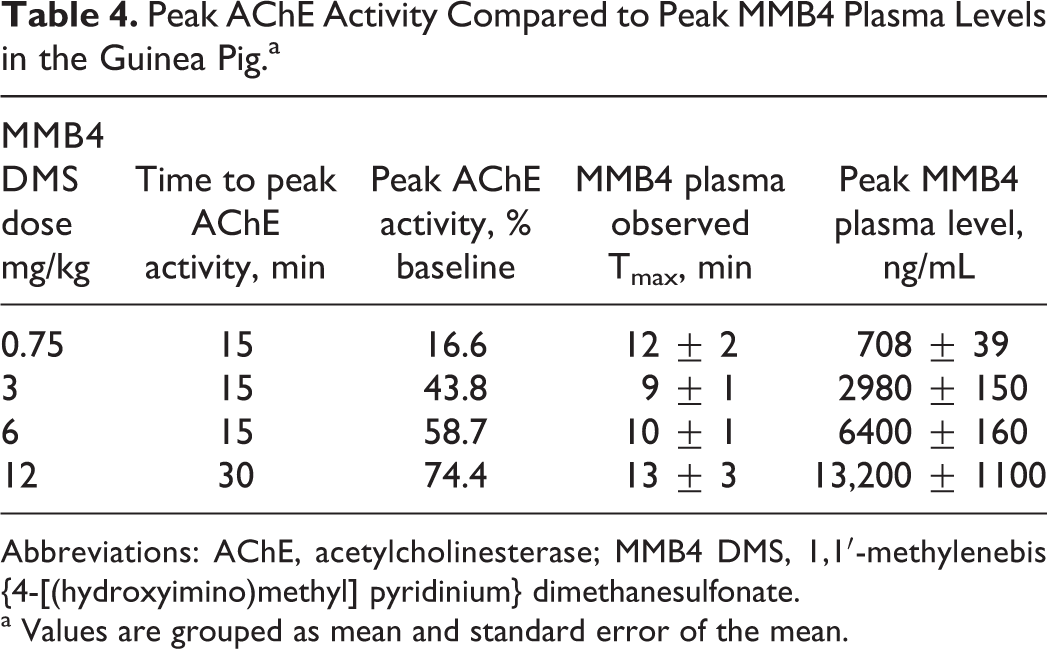
The MMB4 DMS dose and MMB4 plasma concentrations were directly correlated with AChE activity in the GP and NHP (Tables 4 and 5). A dose level of 0.75 mg/kg MMB4 DMS in the GP produced an MMB4 plasma level of 670 ng/mL, which resulted in a peak group mean AChE activity level of 16.6% of baseline. This was insufficient to meet the efficacy criterion of 20% AChE reactivation within 15 minutes, although this dose still produced 100% survival in all animals. At dose levels of ≥3 mg/kg MMB4 DMS, peak MMB4 plasma concentrations averaged greater than approximately 2900 ng/mL, resulting in peak group mean AChE activity values of greater than 43%, exceeding the stated criterion for efficacy. Thus, by extrapolation, 1800 ng/mL is the minimum effective concentration (MEC) of MMB4 that would provide 20% reactivation of AChE activity, as related to baseline values, within 15 minutes. An MMB4 DMS dose of 1.8 mg/kg in the GP is predicted to achieve the MEC.
Peak AChE Activity Compared to Peak MMB4 Plasma Levels in the Guinea Pig.a
Abbreviations: AChE, acetylcholinesterase; MMB4 DMS, 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate.
a Values are grouped as mean and standard error of the mean.
Peak AChE Activity Compared to Peak MMB4 Plasma Levels in the NHP.a
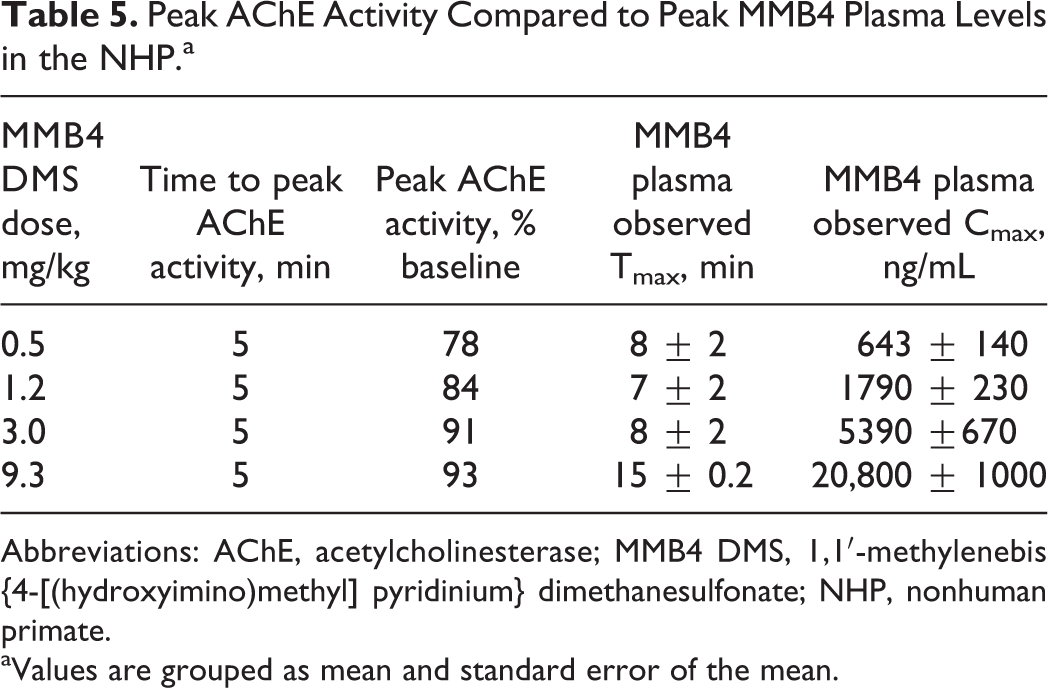
Abbreviations: AChE, acetylcholinesterase; MMB4 DMS, 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} dimethanesulfonate; NHP, nonhuman primate.
aValues are grouped as mean and standard error of the mean.
The MMB4 DMS dose and MMB4 plasma concentrations were directly correlated with AChE activity in the monkey (Table 5). A dose level of 0.5 mg/kg MMB4 DMS produced an MMB4 plasma level of approximately 643 ng/mL that resulted in a peak group mean AChE activity level of 78% of baseline, the lowest observed among the groups. All dose levels exceeded 20% AChE reactivation within 5 minutes. At dose levels of ≥3 mg/kg MMB4 DMS, peak MMB4 plasma concentrations were greater than 5000 ng/mL, resulting in peak group mean AChE activity values of greater than 90%. Differences in AChE activity between the 2 highest MMB4 DMS dose groups was minimal (91% vs 93%), yet the peak MMB4 plasma concentrations differed by nearly 4-fold (approximately 5000 to 20000 ng/mL), indicating that additional increases in MMB4 plasma concentration did not substantially affect AChE reactivation.
Figures 5 and 6 illustrate the dose–response relationship among peak AChE reactivation values (percent baseline), observed MMB4 plasma Cmax values (ng/mL), and dose of MMB4 DMS administered to each group of male GPs and male monkeys, respectively. As shown, AChE reactivation values increased with increasing MMB4 DMS dose, and MMB4 plasma concentrations increased in a dose proportional manner in both the GP and the NHP. Development of a maximal effect (Emax) model in GPs and NHPs allowed for the estimation of a plasma level and corresponding MMB4 DMS dose level that would reactivate 20% AChE in 10 to 15 minutes. It is estimated that 1800 and 25.6 ng/mL MMB4 in the plasma would be needed to achieve an average of 20% AChE reactivation in GPs and NHPs, respectively.
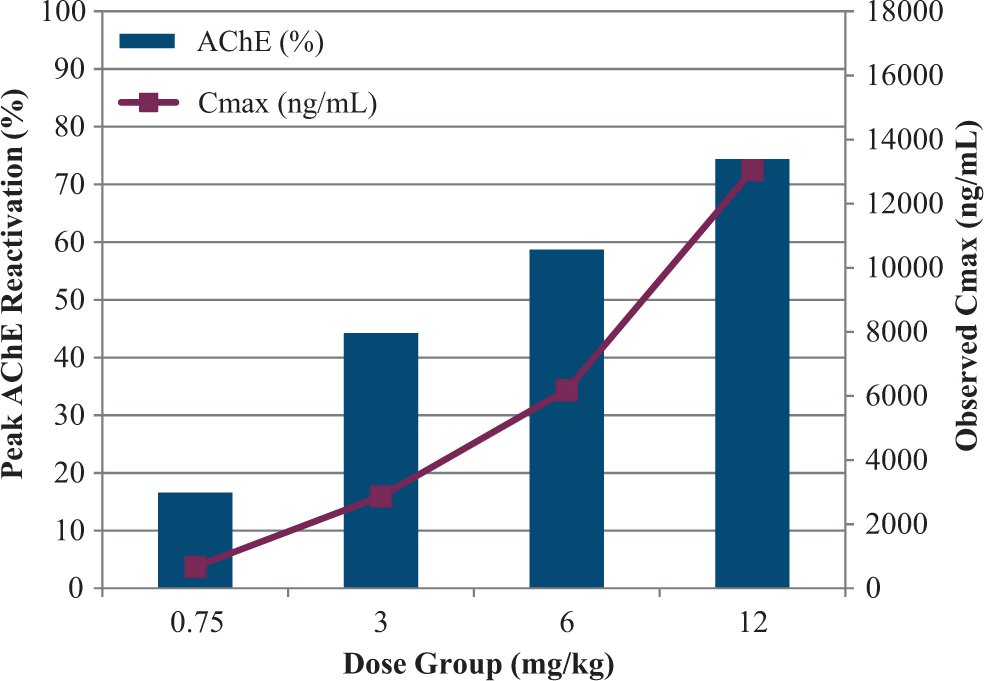
Relationship between peak acetylcholinesterase (AChE) reactivation and 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} (MMB4) Cmax in guinea pigs.
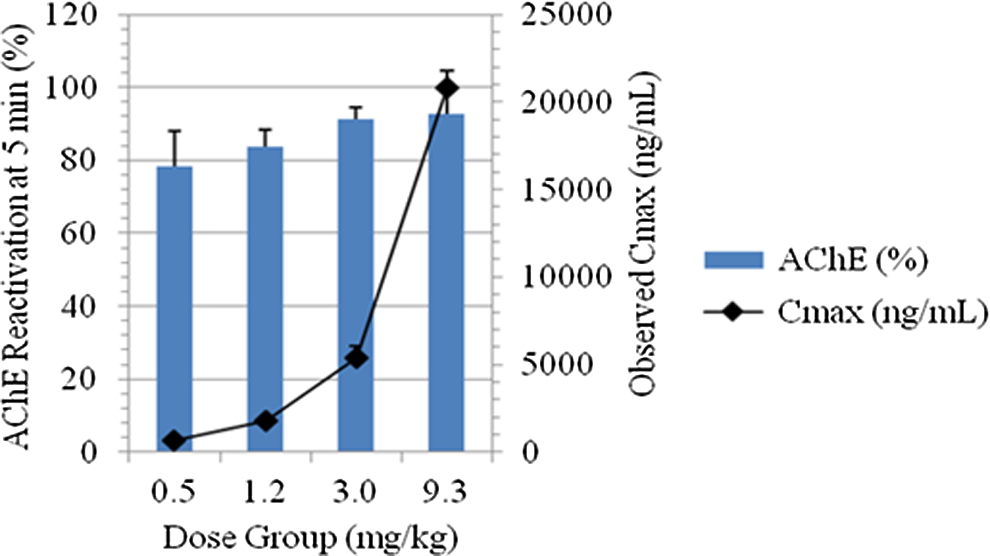
Relationship between peak acetylcholinesterase (AChE) reactivation and 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} (MMB4) Cmax in nonhuman primates (NHPs).
Pharmacokinetic/Pharmacodynamic Comparison Between GPs and Non-Human Primates
Results of the GP and NHP studies allowed a well-characterized PK/PD model to be developed which demonstrated the relationship among MMB4 plasma concentration, AChE reactivation, and MMB4 DMS dose in GPs and NHPs challenged with 1 × LD50 of GF. Comparison of the GP model with the NHP model developed resulted in the determination that MMB4 DMS is approximately 25- to 30-fold more effective in monkeys than in GPs toward reactivating GF-inhibited AChE (Figure 7; Table 6). For example, a plasma concentration of 3000 ng/mL would be required to achieve 50% reactivation of AChE in GPs after being challenged with GF at a dose level of 1.0 × LD50. In comparison, a calculated MMB4 plasma concentration of 110 ng/mL would be required to achieve 50% reactivation of AChE in monkeys after being challenged with GF at a dose level of 1.0 × LD50. Therefore, these results demonstrate that 50% AChE reactivation can be achieved at a plasma MMB4 concentration that is approximately 30 times lower in NHPs than in GPs.
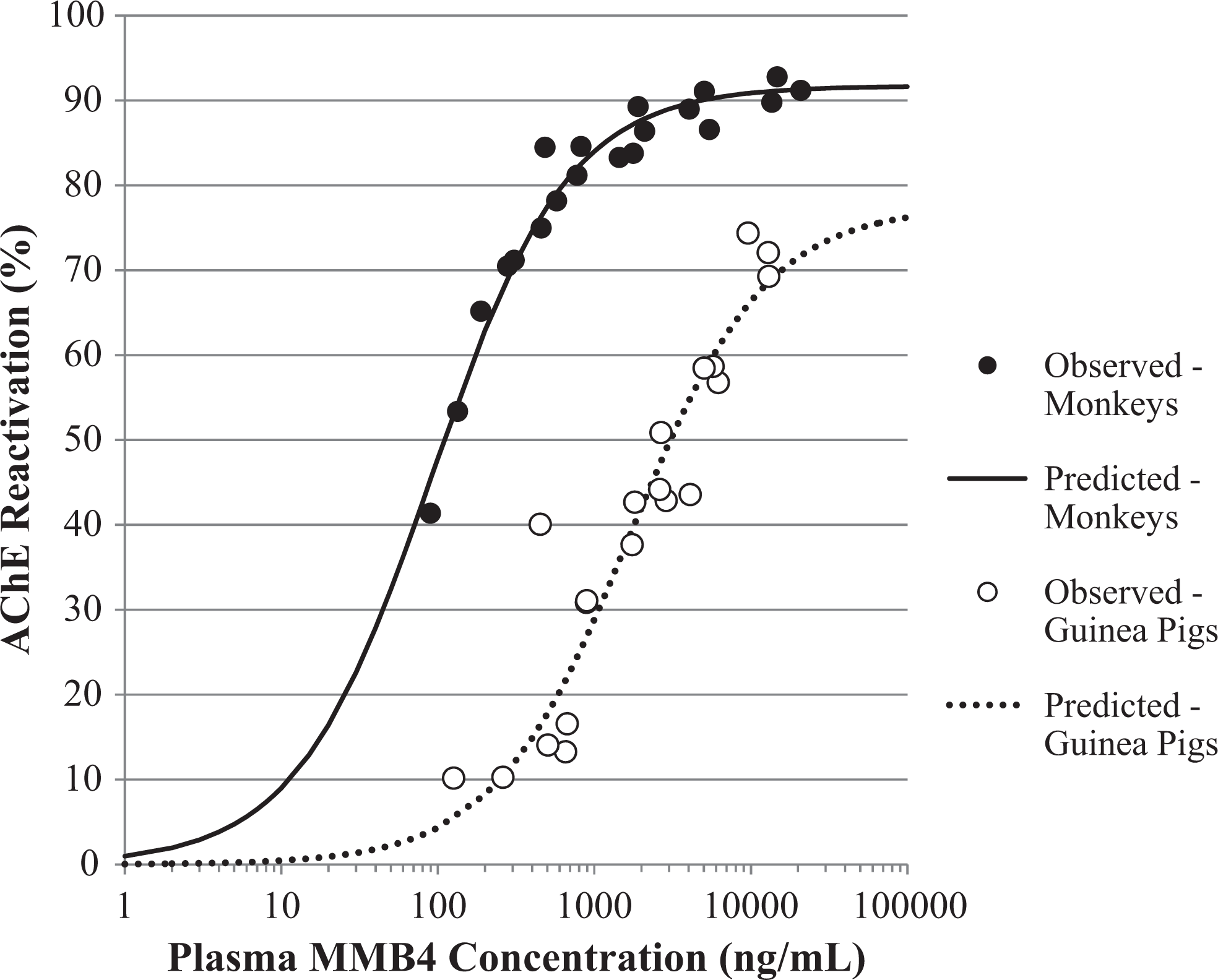
Relationship between acetylcholinesterase (AChE) reactivation and 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium} (MMB4) plasma concentrations in monkey versus guinea pig.
Comparison of Species Differences Between Rhesus Monkeys and Hartley Guinea Pigs.

Abbreviations: AChE, acetylcholinesterase; MMB4, 1,1′-methylenebis{4-[(hydroxyimino)methyl] pyridinium}.
a Reflects predicted concentration to achieve 70% AChE reactivation.
Discussion
Exposure to nerve agents, such as GF, can result in inhibition of AChE. This AChE inhibition is thought to be the main factor in the onset of a cholinergic crisis that can follow nerve agent exposure, due to an excess of acetylcholine at the synaptic junction of the cholinergic CNS and PNS. An oxime capable of reactivating inhibited AChE is crucial to the successful treatment of exposed individuals. The use of the anticholinergic atropine in conjunction with an oxime in nerve agent poisonings serves to reduce secretions and bronchoconstriction through competitive blocking of the muscarinic receptors from acetylcholine.
Prior to these studies, the relationships among GF exposure, AChE activity, MMB4 DMS dose, MMB4 plasma concentration, and survival in the GP and NHP were undetermined or equivocal. These studies investigated a range of MMB4 DMS dose levels and their corresponding plasma concentrations that would reactivate GF-inhibited AChE. Effectiveness was defined as the dose that reactivated at least 20% of blood AChE within 15 minutes following administration. These studies provided the information needed to characterize the relationships among the previously listed end points and, from that, allowed a PK/PD model to be constructed.
The priority of these studies was to optimize animal survival in order to obtain blood samples for measuring AChE activity and MMB4 plasma concentrations over the MMB4 DMS dosage range tested. For this reason, GF exposure was limited to 1 × LD50, and animals were pretreated with 3 mg/kg (GPs) or 1 mg/kg (NHPs) atropine approximately 15 minutes before GF. Although atropine was administered at a fairly high dose (eg, HED in NHPs was 0.32 mg/kg, yet Centers for Disease Control and Prevention/Agency for Toxic Substances and Disease Registry, or CDC/ATSDR, recommendations 9 for severe nerve agent intoxication in adults are approximately 0.09 mg/kg initially and 0.03 mg/kg every 5 to 10 minutes afterward to effect), this dosage was necessary to meet the objectives of the study. In addition, these dosages are still reflective of a possible total atropine administration following severe intoxication.
Atropine dosages as high as 15 to 30 mg (0.21 to 0.43 mg/kg) may be required within the first few hours of therapy. 10, 11 In addition, MMB4 DMS was not administered until approximately 15 minutes (GP) or 5 minutes (NHP) after GF to obtain the maximum GF-induced AChE inhibition possible without causing death. This allowed the most complete MMB4-related reactivation profile possible to be measured. In the previous studies, atropine alone was found to have minimal to no beneficial effect on survival when an oxime was not present, 12 and the importance of conjunctive therapy using both atropine and oxime (MMB4 or HI-6) was demonstrated. Our findings are supplementary to those of Koplovitz et al 7 and Ligtenstein and Moes. 12 Therefore, in our study a consistent effective dose of atropine was administrated with varying dose levels of MMB4 DMS. Results provided important information regarding blood AChE reactivation and survival albeit a direct link between these biomarkers has not been established. In fact, it would be essential in the overall understanding of the mechanism of action to fully elucidate the ability of MMB4 to reactivate neuromuscular/neuroreceptor function at the organs of survival including the diaphragm and brain.
Nonetheless, our study clearly demonstrated the effectiveness of MMB4 to reactivate AChE in the peripheral blood compartment and to provide protection against a 1 × LD50 of GF in terms of survival. At the highest dose of MMB4 DMS administered in the GP study (12 mg/kg), approximately 74% reactivation was achieved. It was unclear whether this was due to a ceiling effect that exists in GPs or whether 100% reactivation (as a percent of baseline) is attainable. However, as the MMB4 DMS dose was increased, there was a dose-proportional increase in MMB4 plasma concentration, which resulted in a positive correlation with AChE reactivation. In comparison, a ceiling effect is likely to exist in NHPs, where greater reactivation may not be attainable in GF-exposed animals. For example, the 3.0 mg/kg MMB4 DMS dose level resulted in a 91% AChE return to baseline, with corresponding MMB4 plasma levels of 5025 ng/mL. However, when the MMB4 DMS dose was increased to 9.3 mg/kg, AChE return to baseline was 93%, yet MMB4 plasma concentrations had increased nearly 4-fold to 20 833 ng/mL.
Whether this represents a ceiling for reactivation due to an early “aging” effect upon a certain fraction of AChE or simply a method variability for recovery cannot be ascertained from the data; however, biological or toxicological relevance of the pre- and postexposure values are questionable in light of the significant reactivation noted and the survival results following MMB4/atropine therapy. A 2-PAM control for comparison was not investigated in this study; however, historical data have shown that while 2-PAM is an effective AChE reactivator for sarin or VX, 13, 14 it does not significantly reactivate AChE following a GF exposure. 15, 16
In vitro study estimates of an effective MMB4 DMS concentration against GF in humans were compared to the predictions made from the present in vivo studies. Worek et al reported that a 10 µmol/L concentration (2580 ng/mL) of MMB4 DMS reactivated 20% of the AChE in human erythrocyte ghosts within 10 minutes following 95% to 98% AChE inhibition in the absence of excess GF. 17 In a current, ongoing MMB4 DMS Phase I clinical trial, MMB4 DMS was found to produce a Cmax plasma concentration of 2420 ng/mL following a single 1.5 mg/kg administration of MMB4 DMS. 18 Using the PK/PD models developed, 20% AChE reactivation in vivo in the GP and NHP was extrapolated to be 1800 and 25.6 ng/mL, respectively (Table 6).
Based on the in vitro data, the predicted human dose of MMB4 DMS that would reactivate 20% AChE is 1.5 mg/kg. This predicted dose assumes that the apparent volume of distribution for MMB4 is fairly well conserved across species, since it has been shown to be approximately 0.338 L/kg based on PK studies in the rat, GP, rabbit, dog, and NHP. 19 –23 The corresponding extrapolated MMB4 DMS dose is 0.013 mg/kg in the NHP, which converts to a human dose of 0.004 mg/kg and a total dose of 0.28 mg in a 70-kg adult. Comparing the predicted human effective MMB4 DMS doses in the NHP based on in vitro and in vivo findings results in a 375-fold difference in the predicted effective dose. Thus, there is a large disparity in predicting the estimated human effective dose to achieve 20% AChE reactivation when using the in vitro or in vivo approach.
In addition to demonstrating the ability of IM administered MMB4 DMS to reactivate significant fractions of AChE and to improve survival, through the PK/PD model that was developed, these studies were able to successfully characterize the relationships among MMB4 DMS dose, MMB4 plasma concentration, and AChE activity following a 1 × LD50 challenge of GF in the GP and NHP. This allowed the determination of the dose of MMB4 DMS that would achieve 20% AChE reactivation within 15 minutes after administration of a 1 × LD50 dose of GF. However, the direct relationship between survival and AChE activity in the blood compartment has not been established. Whether AChE can serve as a biomarker for activity at a tissue that may be responsible for survival (eg, diaphragm or brain) is a topic for further investigation.
Footnotes
Acknowledgments
The authors wish to thank Dr Irwin Koplovitz, USAMRICD, for his technical assistance during these studies. The authors acknowledge the editorial assistance of Dr Vincent Brown.
Authors’ Note
The opinions and assertions contained herein are the private views of the author and are not to be construed as official or reflecting true views of the Department of the Army or the Department of Defense.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by CBMS/MITS through CBRNIAC contract SP0700-00-D-3180.
