Abstract
Organophosphorus (OP) nerve agents pose tremendous threats to both military and civilian populations. The substance 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] (MMB4) is being developed as a replacement for the currently fielded 2-pyridine aldoxime, or pralidoxime (2-PAM) as a treatment for OP nerve agent–induced toxicity. The present study characterized pharmacokinetic (PK) profiles of MMB4 in male and female Sprague-Dawley rats, New Zealand White rabbits, and beagle dogs given a single intravenous (IV) administration of MMB4 dimethanesulfonate (DMS) at 55, 25, and 15 mg/kg dose, respectively. The plasma MMB4 concentration versus time profiles were biphasic for all species tested and fit a 2-compartment model with first-order elimination. There were no overt sex-related differences in the calculated PK parameters. For the rat, rabbit, and dog, the average systemic exposure parameters predicted Cmax (µg/mL) and AUC∞ (µg·h/mL) were 273 and 71.0, 115 and 48.1, and 87.4 and 39.6; the average volume of distribution (mL/kg) values to the central and peripheral compartments were 207 and 143, 242 and 172, and 198 and 213; and the average elimination half-life (hour) and clearance (mL/h/kg) values were 0.18 and 778, 0.29 and 577, and 0.32 and 430, respectively, when the PK parameters for males and females were combined. The current study revealed a similarity in the volume of distribution to the central compartment for MMB4 among the 3 species tested while demonstrating species-related differences in the elimination half-life and clearance of MMB4.
Introduction
Pyridinium aldoximes such as 2-pralidoxime (2-PAM), asoxime (HI-6), and 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] (MMB4) (see Figure 1 for chemical structures) reactivate acetylcholinesterase (AChE) inhibited by organophosphorus (OP) nerve agents. 1 –3 MMB4 was first synthesized in 1959 by Hobbiger and Sadler 4 and has been shown to be more potent than other oximes. 5 –8 A dimethanesulfonate (DMS) salt of MMB4 is being evaluated by the Department of Defense as a replacement for currently fielded 2-PAM as a treatment for OP nerve agent–induced toxicity.
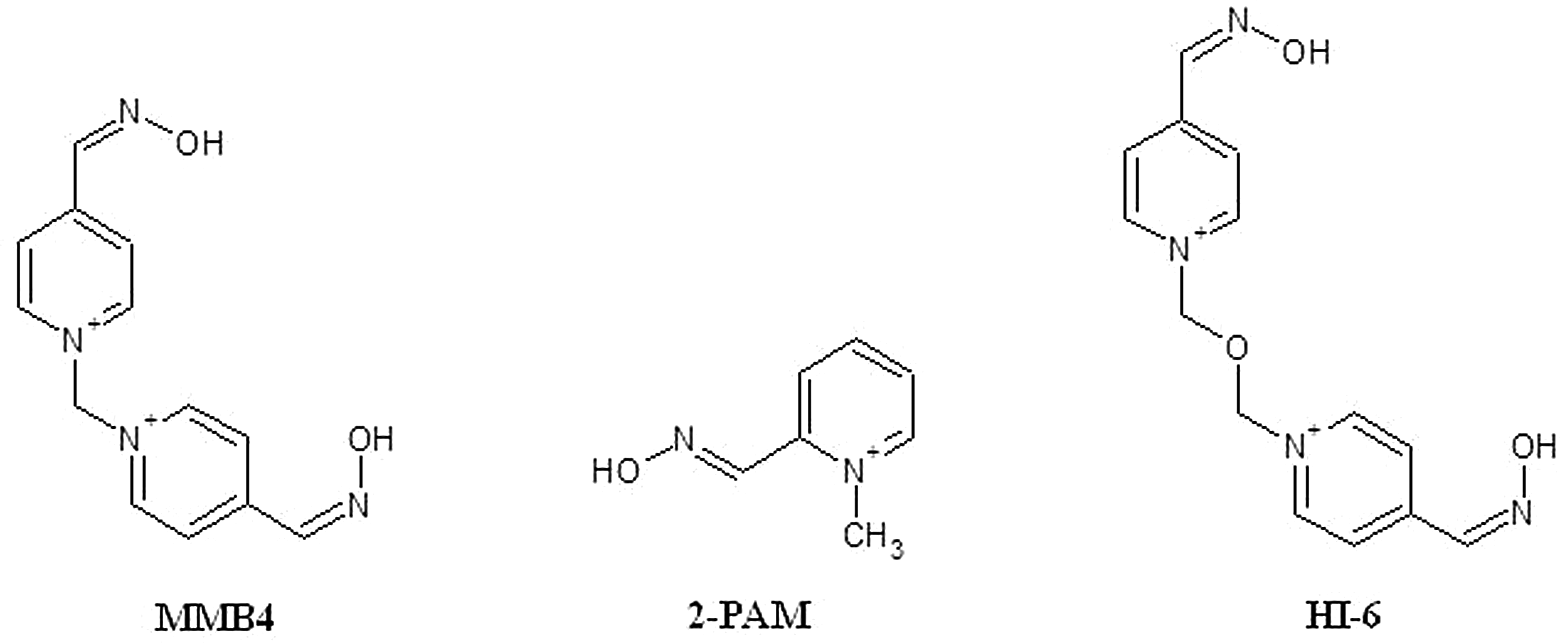
Chemical structures of MMB4, 2-PAM, and HI-6. MMB4 indicates 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium]; 2-PAM, 2-pralidoxime; HI-6, asoxime.
The antidotal efficacy toward nerve agent poisoning cannot be evaluated in humans for ethical reasons. Therefore, the pharmacology and toxicology of MMB4 DMS needs to be thoroughly examined in experimental animals so that the results can be translated to humans. The experimental results from the efficacy and toxicology studies with MMB4 DMS would be better understood by characterizing the time course of MMB4 concentrations. In addition, the efficacy and toxicology studies can be further refined using the pharmacokinetic (PK) data, because the systemic exposure to MMB4 can be calculated for different dose levels under consideration.
Information on the pharmacokinetics of MMB4 is scant while the PK profiles of 2-PAM and HI-6 have been evaluated in various preclinical species. 9 –13 To the best of our knowledge, there are no published results that describe the pharmacokinetics of MMB4 after intravenous (IV) administration. On the other hand, several investigators examined MMB4 PK profiles after intramuscular (IM) administration. 14 –16 While the intended route of administration for MMB4 DMS for clinical use is an IM route, it is important to evaluate MMB4 pharmacokinetics after an IV administration. Without the IV PK data, it is impossible to evaluate how much MMB4 would be absorbed and enter the systemic circulation after IM administration.
The current study characterized PK profiles of MMB4 in male and female Sprague-Dawley rats, New Zealand White rabbits, and beagle dogs given a single IV administration of MMB4 DMS at 55, 25, and 15 mg/kg dose, respectively. The PK parameters were compared to evaluate whether there were sex-related or species-specific differences in pharmacokinetics of MMB4 DMS.
This study was conducted following Good Laboratory Practice (GLP) requirements so that experimental data generated from this study can be included in the Investigational New Drug (IND) application. The study was conducted in compliance with The American College of Toxicology Policy on the Use of Animals, under the guidance of the Battelle Institutional Animal Care and Use Committee (IACUC) and the Animal Care and Use Review Office (ACURO) of the US Army.
Materials and Methods
Dose
The test article for this study was MMB4 DMS and is described as an off-white to tan crystalline powder based on the Certificate of Analysis (CoA). Lot No. 1004 MMB4 DMS, with a manufacture date of July 2007. The source of the test article was Cambrex Charles City, Inc (Charles City, Iowa). The purity of the test article was >98%. The MMB4 DMS dose solution was formulated at a concentration of 27.5, 50, and 100 mg/mL for rats, rabbits, and dogs, respectively, in 0.5% benzyl alcohol and sterile water for injection. The dose formulations were stable for 14 days at room temperature (data not shown).
Experimental Animals
Eighteen Sprague-Dawley rats per sex were purchased from Charles River Laboratories (Portage, Michigan). The rats were 13 weeks of age and weighed 222.3 to 398.8 g at the start of the study. The rats were individually housed in polycarbonate cages with hardwood bedding. Three New Zealand White rabbits per sex were purchased from Covance Research Products, Inc (Denver, Pennsylvania). The rabbits were 7 months of age and weighed 2.9 to 3.4 kg at the start of the study. The rabbits were individually housed in stainless steel cages. Three beagle dogs per sex were purchased from Covance Research Products, Inc (Kalamazoo, Michigan). The dogs were 11 to 12 months of age and weighed 8.9 to 11.6 kg at start of the study. The dogs were individually housed in stainless steel cages. All animals had ad libitum access to feed and fresh water from the Columbus municipal water supply. The study rooms to study animals were maintained at a temperature range of 64°F-84°F, humidity of 30% to 70%, and 12-hour light/12 -hour dark cycle.
Dose Administration and Blood Sample Collections
The rats received a single IV dose administration of 55 mg/kg MMB4 DMS via an indwelling jugular catheter. A dose volume of 2 mL/kg was administered based on the individual animal body weight recorded on the day of dose administration. The rabbits received a single IV dose administration of 25 mg/kg MMB4 DMS via an indwelling femoral vein catheter. A dose volume of 0.5 mL/kg was administered based on the individual animal body weight recorded on the day of dose administration. The dogs received a single IV dose administration of 15 mg/kg MMB4 DMS via a percutaneous catheter placed in the cephalic vein. A dose volume of 0.15 mL/kg was administered based on the individual animal body weight recorded on the day of dose administration. The doses for rats, rabbits, and dogs were selected based on toxicology study results and the human equivalent dose of 8.5 mg/kg that was adjusted by surface area 17 and rounded for each species. Whole blood samples were collected predose and at 1, 3, 6, 10, 15, 20, and 30 minutes, and at 1, 1.5, 2, and 4 hours after the completion of dose administration. Three blood samples were taken from rabbits and dogs at each time point. (Two samples were collected from each rat.) Blood samples were collected from the retro-orbital sinus for rats, via jugular vein catheter for rabbits, and via jugular or cephalic vein (different from the cephalic vein used for dose administration) for dogs into tubes containing lithium heparin as anticoagulant. After collection, blood samples from all animals were kept cold on wet ice until processed to plasma, which occurred within 60 minutes after collection. All blood samples were separated by centrifugation for at least 10 minutes using a refrigerated centrifuge set to maintain 5°C and 2200 rpm. Each sample of plasma was transferred into an appropriately labeled polypropylene vial and stored in a freezer set to maintain −30° to −10°C until analysis.
Bioanalysis of MMB4
Plasma calibration standards for each species were prepared from 2 independently prepared stock solutions. The plasma calibration standards, plasma blanks, plasma quality control samples, and plasma samples were processed by protein precipitation. The resulting extracts were analyzed by high-performance liquid chromatography with mass spectrometry (HPLC/MS). The HPLC system was composed of a Shimadzu prominence pump and detector (Kyoto, Japan). The MS system was a Sciex API 4000 (Toronto, Ontario) with turbo spray and positive ion mode. Samples were injected into 100 c× 2 mm Hydro-RP column (Phenomenex, Torrance, California). MMB4 concentrations were calculated using peak area response ratios and a regression equation constructed from the calibration standards. The lower limit of quantitation (LLOQ) for this analytical method was approximately 50 ng/mL for MMB4 in plasma using 100 µL of sample.
PK Analysis
PK analysis was performed using the actual dose of MMB4 (mg/kg), actual sample
collection time points (hours), and the measured concentrations of MMB4 in plasma
(µg/mL). PK parameters for MMB4 were calculated using WinNonlin (Pharsight
Corporation, Mountain View, California). The concentration–time profile demonstrated
2 exponential phases, that is, an initial distributional phase followed by a terminal
elimination phase (Figure 2),
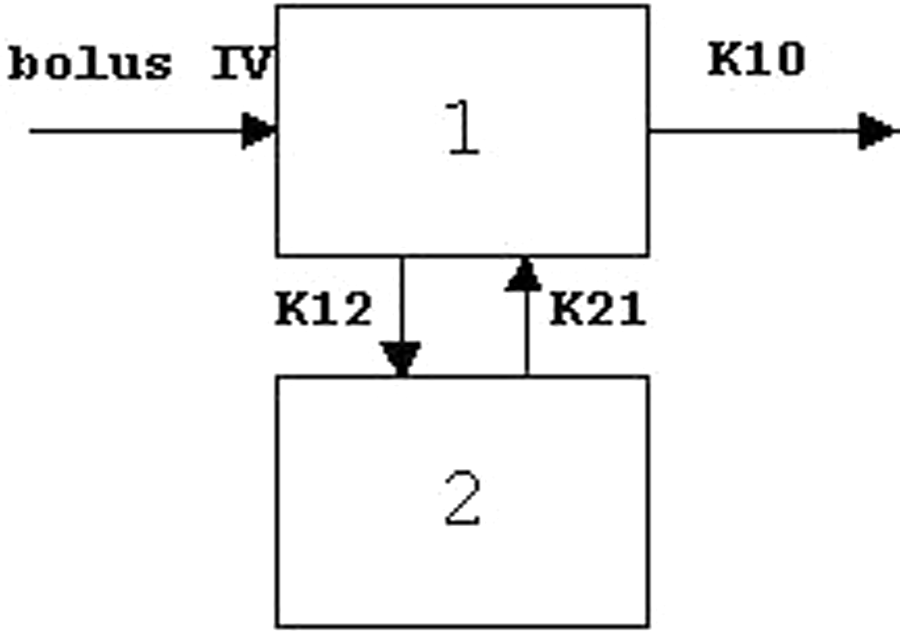
which would be best described by a 2-compartment model. Therefore, a 2-compartment
open model with IV bolus input and first-order elimination (see Figure 3 for the model structure) was used to
fit the following equation to the experimental data generated in this study: Time course of plasma concentrations (±SE) of MMB4 following a single IV
administration of MMB4 DMS to rats at 55 mg/kg (A), rabbits at 25 mg/kg (B),
and dogs at 15 mg/kg (C). MMB4 indicates
1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium]; DMS,
dimethanesulfonate; SE, standard error. Two-compartment model with IV bolus input and first-order elimination. IV
indicates intravenous; 1, central compartment; 2, peripheral compartment;
K12, a rate constant for distribution from central to peripheral
compartment; K21, a rate constant for distribution from peripheral to
central compartment; K10, a rate constant for elimination from central
compartment.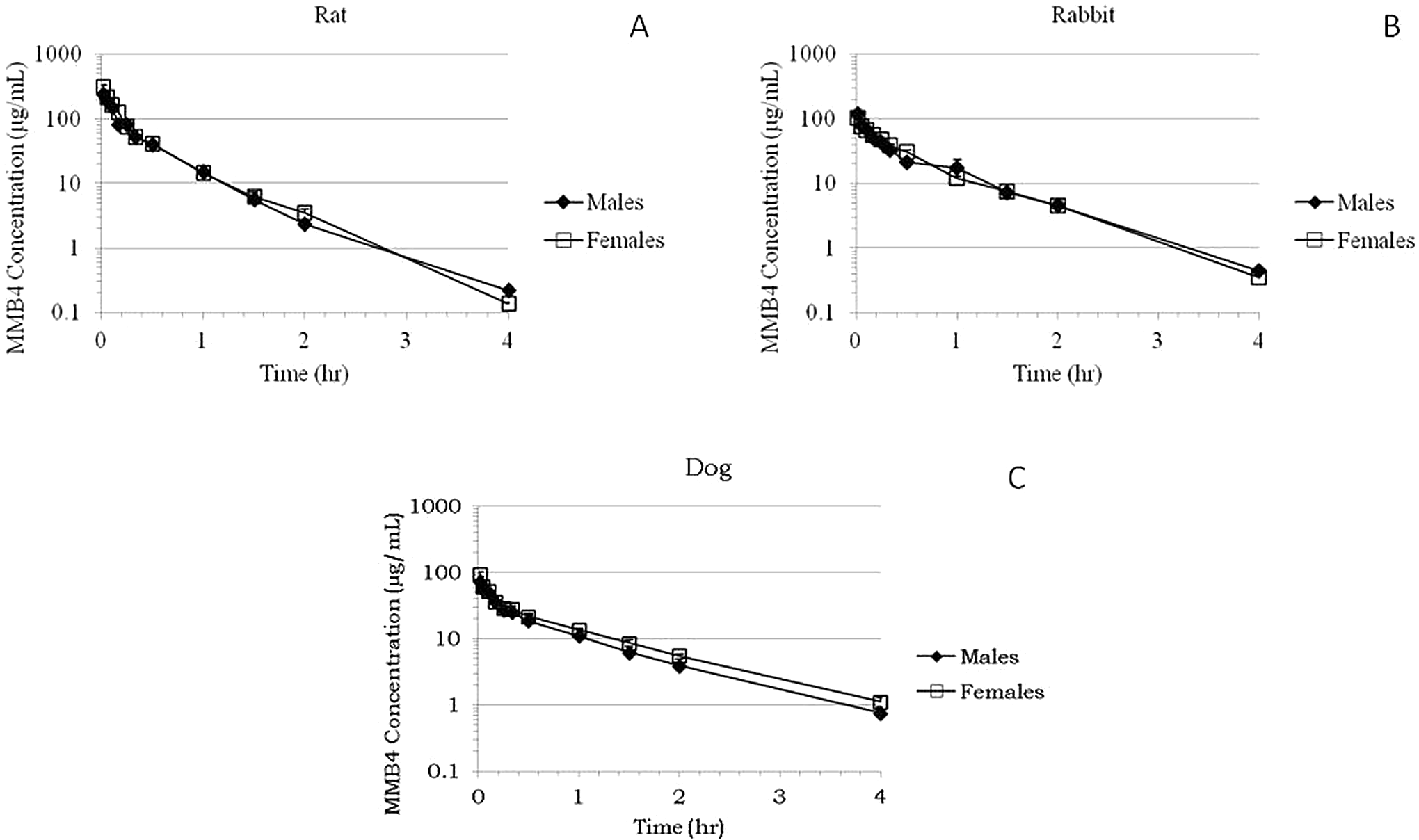
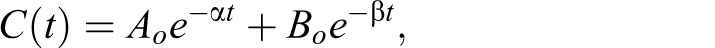
where
Statistical differences in average PK values were determined using a 2-sided equal
variance 2-sample
Results
Bioanalysis of MMB4
The plasma calibration standards used to form the calibration curve met all acceptance criteria, with the correlation coefficients greater than 0.99 and the average errors within 15% of nominal concentrations (data not shown). In addition, the plasma blanks and quality control samples met the acceptance criteria as the average responses of the blanks were less than 20% of the average response of the lowest acceptable standard, the average determined concentrations of the control samples were within 15% of the nominal concentration, and relative standard deviations were less than 15% (data not shown).
Pharmacokinetics of MMB4 in Rats
The time course of MMB4 concentrations in plasma exhibited a biexponential decline after a single IV administration of MMB4 DMS to male and female rats at a dosage of 55 mg/kg (Figure 2A). The concentration versus time profiles were similar for male and female rats, suggesting that there was no overt sex-related difference in MMB4 pharmacokinetics. This was further substantiated by similar PK parameters for male and female rats (Table 1). The maximal concentration of MMB4 (Cmax) in plasma, which represented a concentration in plasma immediately following the IV administration as predicted by the 2-compartment model, was marginally higher in females than in males with an average value of 273 µg/mL when males and females were combined. Average elimination half-life was 0.183 hour (11 min) and average clearance in rats was 778 mL/h/kg. Average values for volume of distribution to central and peripheral compartments were 207 and 143 mL/kg, respectively. Average area under the curve to the last measurable concentration (AUClast) and to infinity predicted by the 2-compartment model (AUC∞) were 75.1 and 71.0 µg·h/mL, respectively.
Summary of PK Parameters for Rats Following a Single IV Administration of MMB4 DMS at a Dosage of 55 mg/kg.
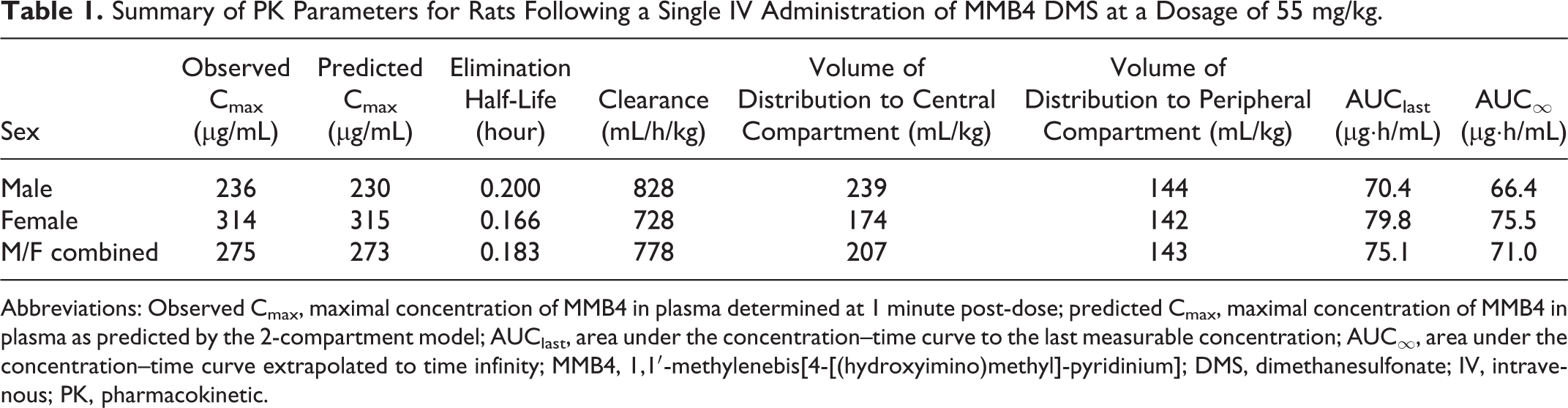
Abbreviations: Observed Cmax, maximal concentration of MMB4 in plasma determined at 1 minute post-dose; predicted Cmax, maximal concentration of MMB4 in plasma as predicted by the 2-compartment model; AUClast, area under the concentration–time curve to the last measurable concentration; AUC∞, area under the concentration–time curve extrapolated to time infinity; MMB4, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium]; DMS, dimethanesulfonate; IV, intravenous; PK, pharmacokinetic.
Pharmacokinetics of MMB4 in Rabbits
The concentrations of MMB4 in plasma declined in a biexponential manner after a
single IV administration of MMB4 DMS to male and female rabbits at a dosage of 25
mg/kg (Figure 2B). There was
no apparent sex-related difference in the concentration versus time profiles. In
addition, there were no statistically significant differences (
Summary of PK Parameters for Rabbits Following a Single IV Administration of MMB4 DMS at a Dosage of 25 mg/kg.
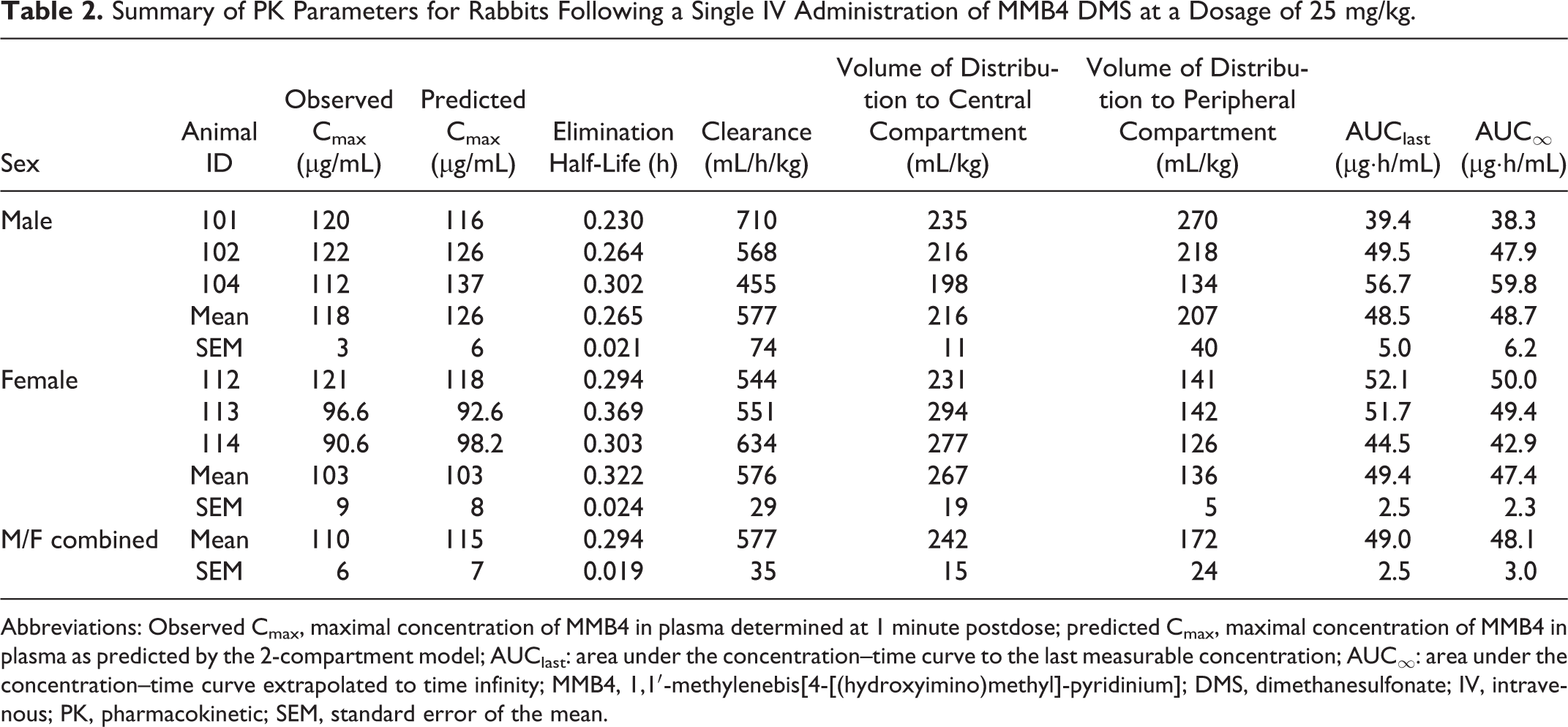
Abbreviations: Observed Cmax, maximal concentration of MMB4 in plasma determined at 1 minute postdose; predicted Cmax, maximal concentration of MMB4 in plasma as predicted by the 2-compartment model; AUClast: area under the concentration–time curve to the last measurable concentration; AUC∞: area under the concentration–time curve extrapolated to time infinity; MMB4, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium]; DMS, dimethanesulfonate; IV, intravenous; PK, pharmacokinetic; SEM, standard error of the mean.
Pharmacokinetics of MMB4 in Dogs
The plasma concentrations of MMB4 declined in a biexponential manner after a single
IV administration of MMB4 DMS to male and female dogs at a dosage of 15 mg/kg (Figure 2C). The average
concentration versus time profiles did not indicate sex-related differences in MMB4
PK. In addition, there were no statistically significant differences
(
Summary of PK Parameters for Dogs Following a Single IV Administration of MMB4 DMS at a Dosage of 15 mg/kg.
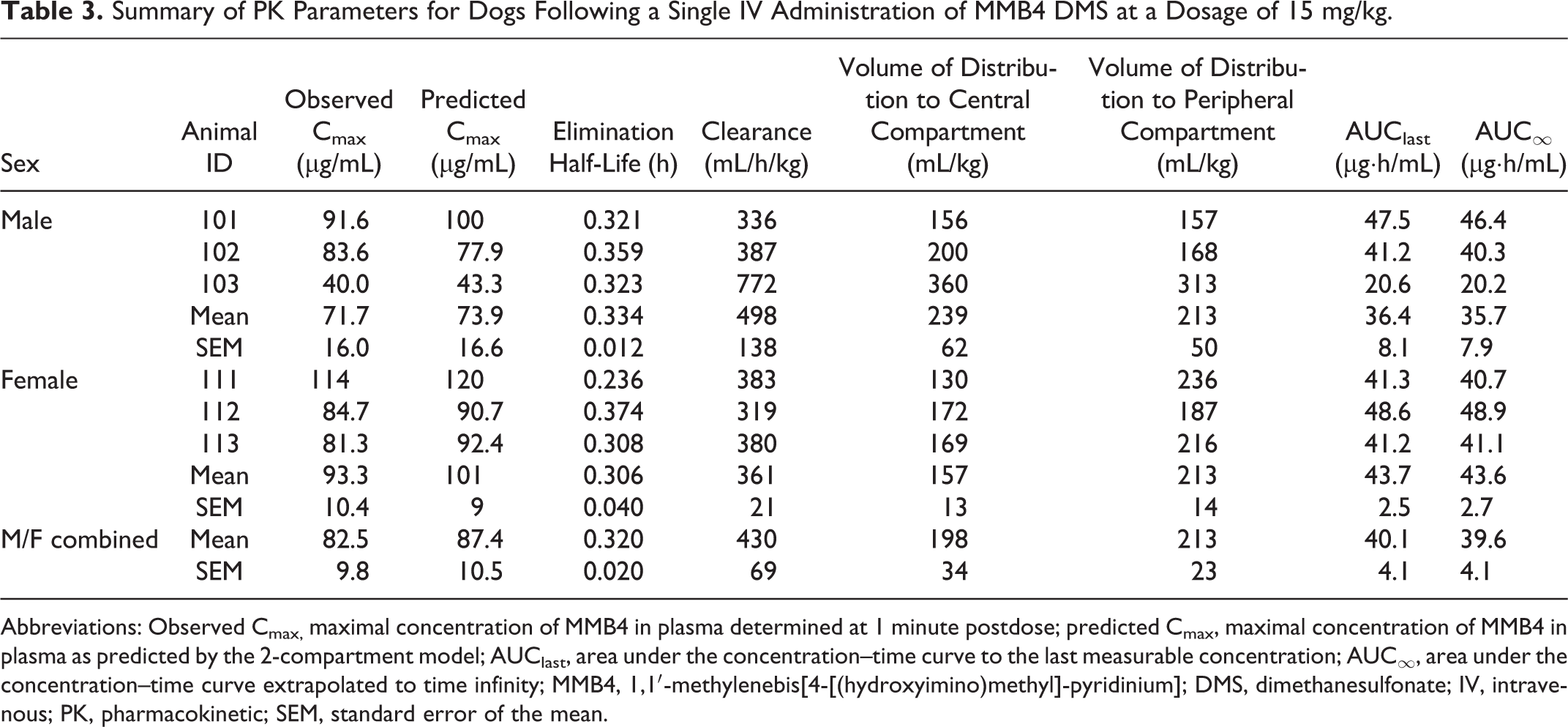
Abbreviations: Observed Cmax, maximal concentration of MMB4 in plasma determined at 1 minute postdose; predicted Cmax, maximal concentration of MMB4 in plasma as predicted by the 2-compartment model; AUClast, area under the concentration–time curve to the last measurable concentration; AUC∞, area under the concentration–time curve extrapolated to time infinity; MMB4, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium]; DMS, dimethanesulfonate; IV, intravenous; PK, pharmacokinetic; SEM, standard error of the mean.
Discussion
MMB4 DMS is being developed as one of the countermeasures against nerve agent poisoning
under the Food and Drug Administration (FDA) guidance “Animal Rule” since humans cannot
be used in demonstrating the antidotal efficacy due to safety concerns.
18
Therefore, it is critical to examine the similarities and differences in
pharmacokinetics of MMB4 among different species, which will help better understand
species-related differences in efficacy and toxicology results. Following a single IV
administration of MMB4 DMS, the plasma concentration versus time profiles demonstrated
an early distribution phase followed by a linear elimination phase, regardless of the
species tested. As such, a 2-compartmental model with bolus input and first-order
elimination adequately described the experimental MMB4 PK data collected from rats,
rabbits, and dogs. Male and female rats showed similar PK profiles without any overt
sex-related differences in the calculated PK parameters. In addition, there were no
statistical differences (
The present study demonstrated that there were no overt species-related differences in
the volume of distribution into central or peripheral compartments for MMB4 (Figure 4). These results suggest
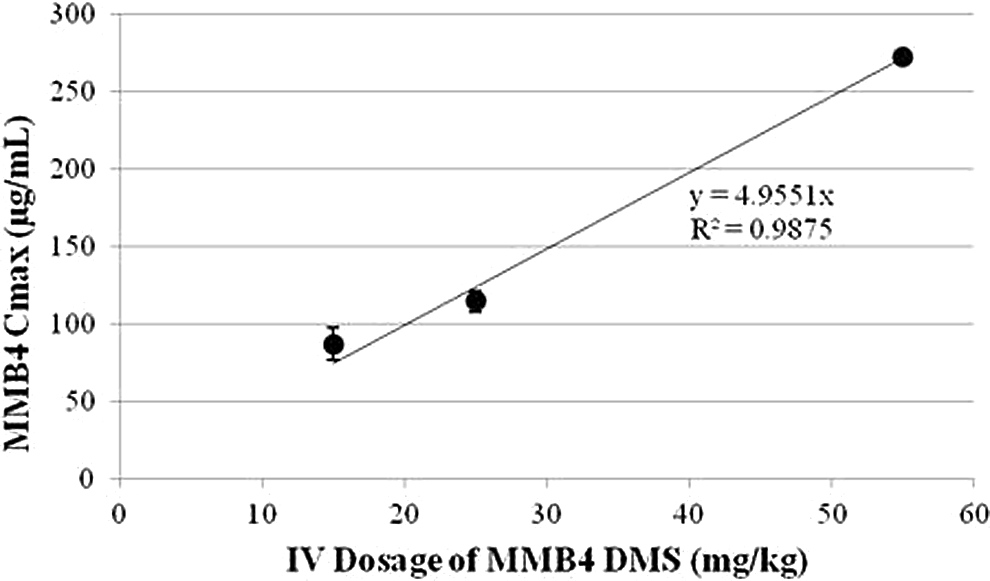
that Cmax levels in these preclinical species would correlate well to dosages
regardless of species, since Cmax is dependent on dosage and volume of
distribution. This was corroborated by a strong correlation ( Species comparison of volume of distribution for MMB4. There was no measure of
variability calculated for the rat data since multiple animals were used to
generate a single PK profile. MMB4 indicates
1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium]; PK,
pharmacokinetic. Relationship between MMB4 Cmax and dosage for rat, rabbit, and dog.
Rats, rabbits, and dogs received 15, 25, and 55 mg/kg MMB4 DMS, respectively.
Cmax indicates maximal concentration of MMB4 in plasma as
predicted by the 2-compartment model; MMB4,
1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium].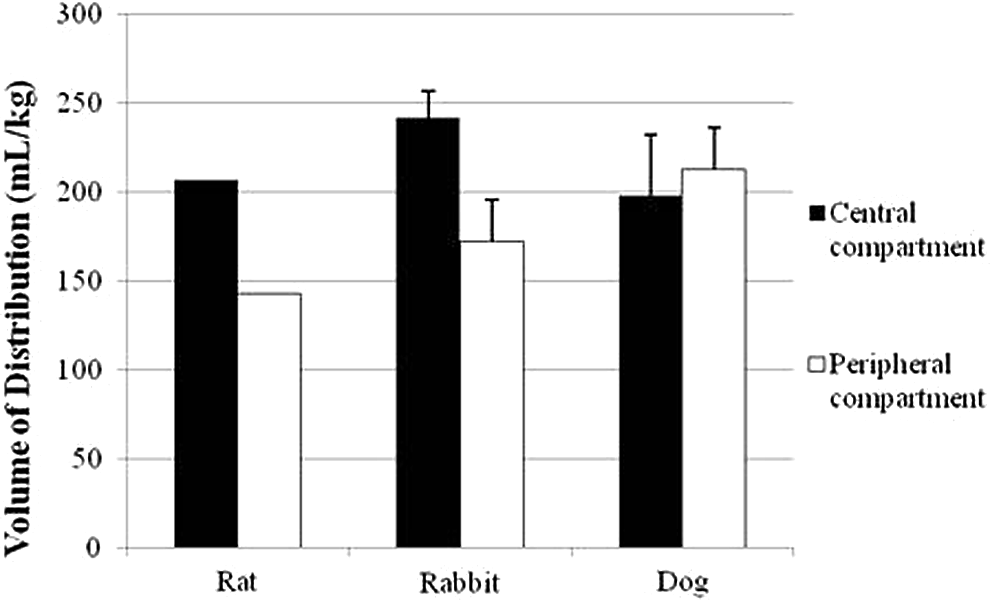
There were species-dependent differences in elimination and clearance of MMB4 following
IV administration. The elimination half-life for MMB4 in rats (11 minutes) was shorter
than those in rabbits (18 minutes) and dogs (19 minutes). In addition, the clearance of
MMB4 in rats was apparently faster than in rabbits and dogs, while there was no
statistical difference (
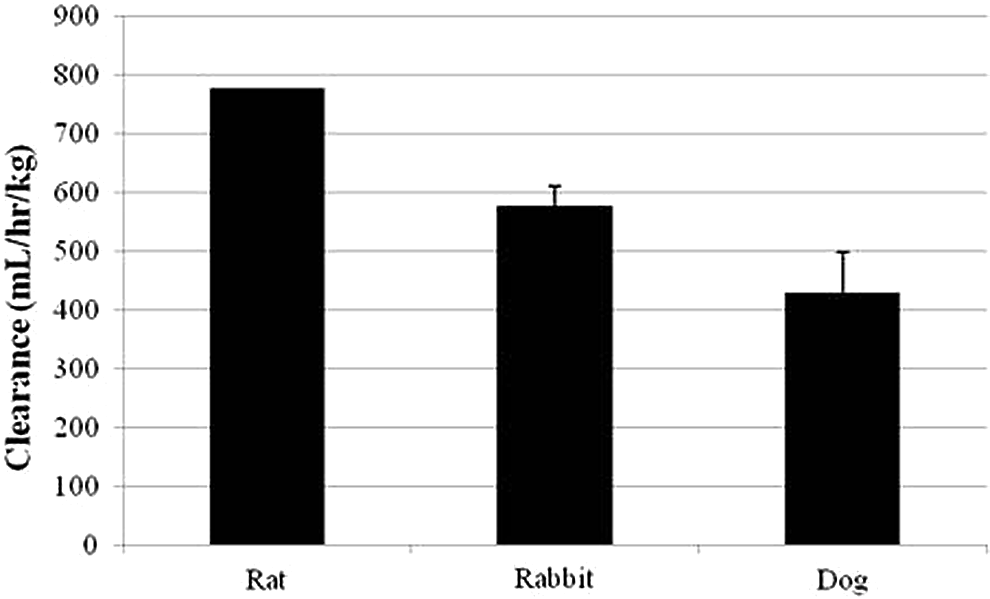
Species comparison of clearance for MMB4. there was no measure of variability calculated for the rat data since multiple animals were used to generate a single PK profile. MMB4 indicates 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium].
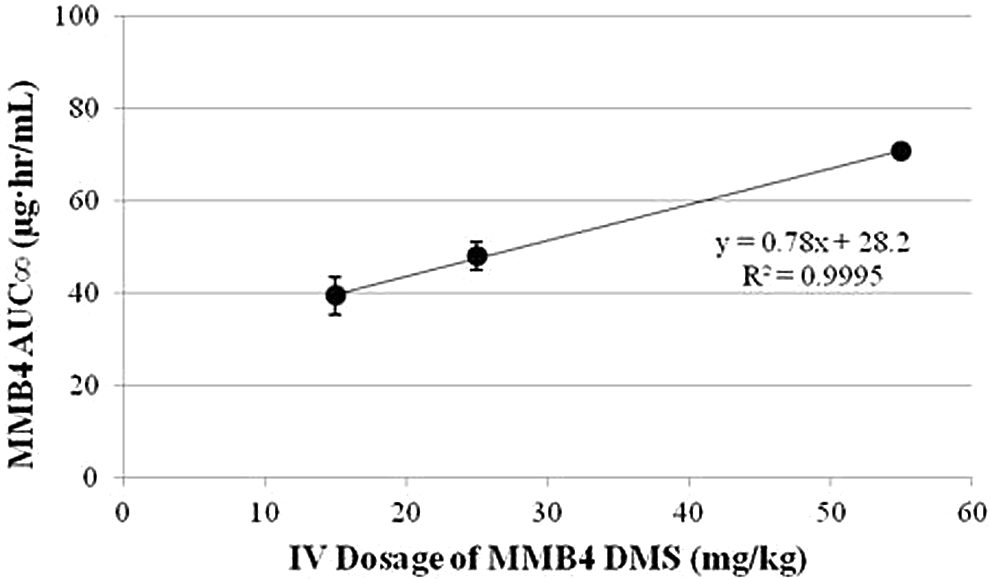
Relationship between MMB4 AUC∞ and dosage for rat, rabbit, and dog. Rats, rabbits, and dogs received 15, 25, and 55 mg/kg MMB4 DMS, respectively. MMB4 indicates 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium]; AUC∞, area under the concentration–time curve extrapolated to time infinity; DMS, dimethanesulfonate.
Using the calculated PK parameters for MMB4 in rat, rabbit, and dog, MMB4 concentration versus time profiles in humans after a single IV administration of MMB4 DMS were simulated (Figure 8). Following an IV administration of MMB4 DMS at 1.0 mg/kg to humans, the Cmax levels would be similar with the predicted values of 4.83, 4.13, and 5.05 µg/mL when the rat, rabbit, and dog PK data were used, respectively. However, the simulated AUC∞ values in humans given 1.0 mg/kg IV dose were somewhat variable depending on which animal PK data were used in simulation and resulted in the predicted values of 1.26, 1.71, and 2.29 µg·h/mL when the rat, rabbit, and dog PK data were used, respectively.
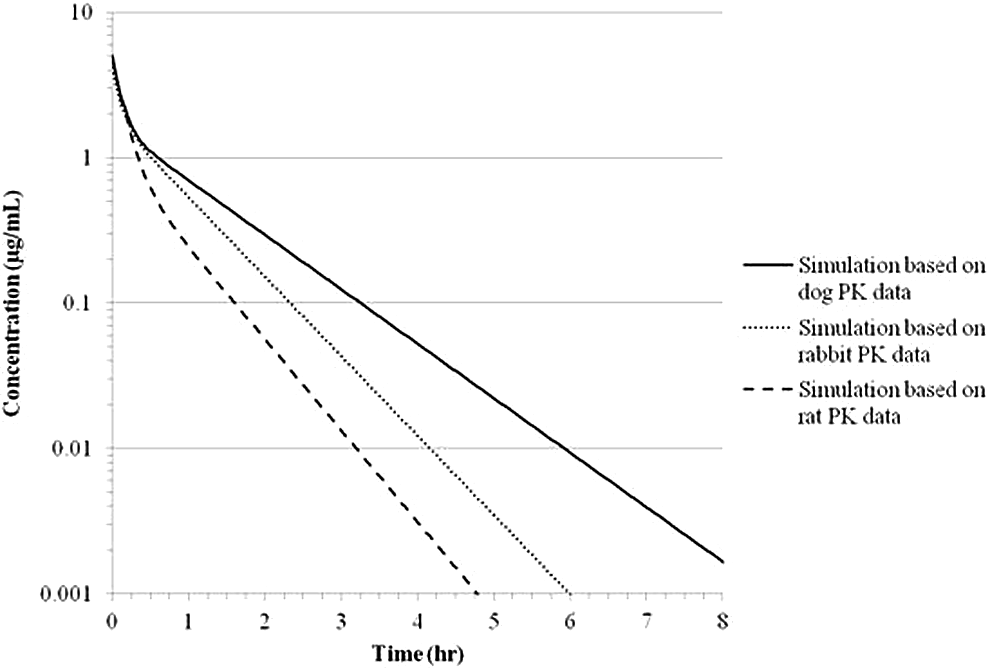
Simulated plasma MMB4 concentration versus time curves in humans following a single IV administration of MMB4 DMS at 1.0 mg/kg dose. MMB4 indicates 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium]; DMS, dimethanesulfonate; IV, intravenous.
While no previous PK information was available for MMB4 after IV administration, pharmacokinetics of MMB4 have been evaluated in several preclinical species following IM injection of MMB4 DMS. Stemler et al reported 53.3 ± 8.7 minutes for the elimination half-life, 360 ± 46 mL/kg for the apparent volume of distribution, and 282 ± 12 mL/h/kg for the apparent clearance after a single IM administration of MMB4 DMS at a 32.9 mg/kg dose to male pigs. 14 In male guinea pigs, it was found that MMB4 showed 34.4 to 43.6 minutes for the elimination half-life, 290 to 360 mL/kg for the apparent volume of distribution, and 347 to 352 mL/h/kg for the apparent clearance after a single IM administration of MMB4 DMS at 29, 58, or 116 µmol/kg dose. 15 Myers demonstrated dose-dependent difference in MMB4 elimination half-life after a single IM administration of MMB4 DMS to male African Green monkeys with 60.4 ± 10.4 minutes at a 19.2 µmol/kg dose level but 21.1 ± 7.8 and 25.0 ± 7.6 minutes at 58 and 116 µmol/kg, respectively. 16 On the other hand, apparent volume of distribution for MMB4 in African Green monkeys was not dependent on dose, with average values ranging from 348 to 417 mL/kg for the 3 dose levels tested. 16 These results supported the findings from the present study, showing species-related differences in the rate of MMB4 elimination. The apparent volume of distribution and clearance from these studies that employed the IM route of administration could not be directly compared to the IV PK data due to the lack of information on the absolute bioavailability for the IM route in those species.
Dispositional characteristics such as elimination half-life, volume of distribution, and clearance are important factors in evaluating drug candidates against existing therapeutic agents. Therefore, PK parameters for other oximes such as 2-PAM and HI-6 were compared to those for MMB4 DMS. Green et al reported 34.8 ± 12.4 minutes of elimination half-life, 2.21 ± 0.45 L/kg of volume of distribution, and 2.46 ± 0.91 L/h/kg of clearance in male rats given a single IM administration of 2-PAM at 40 mg/kg dose. 9 Similar PK parameters for 2-PAM were obtained by Kayouka et al who reported 38.2 ± 3.8 minutes of elimination half-life, 2.2 ± 0.3 L/kg of volume of distribution, and 3.4 ± 0.3 L/h/kg of clearance after a single IM administration of 2-PAM at 50 mg/kg dose to male Sprague-Dawley rats. 10 These results suggest that MMB4 is eliminated and cleared of the systemic circulation faster than 2-PAM, while MMB4 has a smaller volume of distribution, indicating that there is less sequestration of MMB4 into various tissues than 2-PAM. On the other hand, the elimination rate and clearance of MMB4 in rats were comparable to HI-6, because HI-6 exhibited 14 minutes of elimination half-life and 722 mL/h/kg of clearance after a single IV administration to rats at a 20 mg/kg dose. 11 However, the volume of distribution to central compartment for MMB4 (207 mL/kg) in rats was smaller than for HI-6 (320 mL/kg). 11 Furthermore, the rate of elimination and clearance of MMB4 were faster than those of HI-6 in dogs. Simons and Briggs reported 48.2 ± 17.7 minutes of elimination half-life and 310 ± 49 mL/h/kg of clearance for HI-6 in dogs following a single IV administration at 20 mg/kg dose. 12 These results were consistent with 46.5 ± 7.8 minutes of elimination half-life and 221 ± 41 mL/h/kg of clearance for HI-6 in dogs reported by another group of investigators. 13 The volume of distribution to central compartment for MMB4 in dogs (198 ± 34 mL/kg) appeared to be comparable to that for HI-6 (125 ± 54 mL/kg) 13 even though the average value for MMB4 was slightly larger than that for HI-6. These results also demonstrated species-related differences in HI-6 disposition kinetics, including the volume of distribution to the central compartment that was more conserved for MMB4 among different species.
The current study revealed a similarity in the volume of distribution to the central compartment for MMB4 among the 3 species tested, while demonstrating species-related differences in the elimination half-life and clearance of MMB4. In addition, it provides necessary PK information to evaluate the absolute bioavailability of MMB4 DMS in a follow-on study involving the intended route of administration for nerve agent antidotes.
Footnotes
Acknowledgments
The authors wish to thank Dr. Vincent Brown for the editorial assistance.
Authors’ Note
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or reflecting true views of the Department of the Army or the Department of Defense.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the United States Army; Chemical Biological Medical Systems (CBMS)/Medical Identification and Treatment Systems (MITS) under contract SP0700-00-D-D3180, Delivery Order 0599 (Task 771) and Delivery Order 0600 (Task 789).
