Abstract
This toxicological assessment evaluated the safety of a hydroethanolic extract prepared from Caralluma fimbriata (CFE), a dietary supplement marketed worldwide as an appetite suppressant. Studies included 2 in vitro genotoxicity assays, a repeated dose oral toxicity study, and a developmental study in rats. No evidence of in vitro mutagenicity or clastogenicity surfaced in the in vitro studies at concentrations up to 5000 μg of extract/plate (Ames test) or 5000 μg of extract/mL (chromosomal aberration test). No deaths or treatment-related toxicity were seen in the 6-month chronic oral toxicity study in Sprague-Dawley rats conducted at 3 doses (100, 300, and 1000 mg/kg body weight (bw)/d). The no observed effect level for CFE in this study was considered to be 1000 mg/kg bw/d. A prenatal developmental toxicity study conducted at 3 doses (250, 500, and 1000 mg/kg bw/d) in female Sprague-Dawley rats resulted in no treatment-related external, visceral, or skeletal fetal abnormalities, and no treatment-related maternal or pregnancy alterations were seen at and up to the maximum dose tested. CFE was not associated with any toxicity or adverse events.
Keywords
Introduction
Caralluma fimbriata, also known as Caralluma adscendens var. fimbriata, is an edible succulent cactus of the Asclepiadaceae family and has traditionally been used in India as an appetite suppressant and a famine food. 1 –4 The hydroethanolic extract of this perennial herb is currently sold as a botanical dietary supplement with potential anorectic properties. The fight against obesity and related health risk factors has historically focused on the quality and quantity of daily food consumption and the importance of physical activity, and recent incorporation of pharmaceuticals and natural herbal remedies has broadened the scope of treatment.
Plants of the Asclepiadaceae family contain pregnane glycosides, which are structurally diverse steroidal compounds, as part of their small molecule repertoire. 5,6 Active components isolated from several subspecies in the Hoodia genus, also in the Asclepiadaceae family, were characterized as pregnane glycosides and identified as therapeutic constituents with anorectic activity. 7 –9 Similarly, the Caralluma genus is also rich in pregnane glycosides and a large number of these metabolites have been isolated, characterized, and/or screened for biological activity. 6,10 –21
The potential appetite-suppressing activity of a hydroethanolic extract prepared from dried C. fimbriata has been examined in both animal and human studies. Two double-blind, placebo-controlled clinical trials on CFE extract are found in the literature; one was conducted in Australia, and the other in India on a total of 83 overweight or obese subjects. 22,23 The effects of 1 g/d of the extract in addition to diet and physical activity counseling were assessed over a period of 2 to 3 months. In both studies, a statistically significant decrease in waist circumference was noted in the experimental groups compared to the placebo groups at the end of the study periods. Additional findings included statistically significant reductions in waist-to-hip ratio compared to placebo, 22 palatability of the test meal (appeal, smell, and taste) compared to baseline (not seen in placebo group), 22 and hunger compared to placebo. 23 Although both the studies recorded significant decreases in body weight (bw), body mass index, and hip circumference compared to baseline in the groups taking the extract, these values did not reach statistical significance when compared to the placebo groups. Decreased consumption of several macro/micronutrients such as refined sugars, cholesterol, saturated fat, and sodium was recorded in the CFE groups, but the results were not consistent between the 2 studies so are difficult to interpret. The extract was well tolerated in both the studies. Only minor adverse events occurred, mainly related to gastrointestinal disturbances, and occurrences did not differ between the experimental and the placebo groups. One subject in the extract group experienced a skin rash, which was resolved after cessation of the test article.
In an animal study, researchers reported that male Wistar rats fed with a high-fat cafeteria diet and receiving CFE daily by oral gavage for 90 days exhibited appetite-suppressing effects. 24 Groups included a standard pellet chow control group, an untreated high-fat cafeteria diet control group, and 3 high-fat cafeteria diet groups receiving 25, 50, or 100 mg CFE/kg bw/d. The animals receiving CFE exhibited statistically significant (P < 0.05) dose-dependent reductions in food intake compared to the standard pellet chow and untreated high-fat cafeteria diet groups. CFE and standard pellet chow groups further exhibited statistically significant (P < 0.05) reductions in weight gains compared to the untreated high-fat cafeteria diet control group. Final bws of all CFE groups were similar to those of the standard pellet chow group.
The clinical studies mentioned earlier highlighted a favorable effect of the extract in regard to reducing waist circumference. Benefits on hunger levels and food palatability were also noted. The animal study found CFE to be effective in preventing weight gain and reducing food intake. Rats fed CFE in addition to a high-fat cafeteria diet had significantly less weight gain compared to rats in the untreated high-fat cafeteria diet control group; rats receiving the high-fat cafeteria diet plus CFE also exhibited significantly reduced food intake compared to rats in the untreated high-fat cafeteria diet control group as well as compared to the standard pellet chow group not receiving CFE. It was also reported that CFE inhibited preadipocyte cell division in 3T3-L1 mouse embryo fibroblasts as well as cell growth. 25
With regard to safety, a 2006 abstract and 2007 book chapter briefly report on an acute oral toxicity and 90-day oral toxicity of CFE in Wistar rats; neither study was reported to be Good Laboratory Practice (GLP) compliant. 26,27 The acute oral toxicity study, conducted at St John’s Medical College (Bangalore, Karnataka, India), resulted in no deaths up to and at the highest dose tested (5 g/kg bw). No change in food intake, bw, hematology, or macroscopic histopathology was reported for male and female rats receiving a single dose of CFE at 5 g/kg bw. The 90-day oral toxicity study, conducted at Bombay College of Pharmacy (Mumbai, Maharastra, India), included 3 doses of CFE, 90, 270, and 900 mg/kg bw/d, and indicated no treatment-related hematological or clinical chemistry abnormalities. Four deaths occurred during that study (1 male in each of the 270, 900, and satellite 900 mg/kg bw/d groups and 1 female in the 900 mg/kg bw/d group) and were preceded by dramatic weight loss and weakness. Necropsy of the expired animals did not indicate any histopathological abnormalities or tissue damage, and the cause of death for these 4 animals was not established. Male rats in the 270 and both the 900 mg/kg bw/d groups further exhibited sparse hair (hair shedding/loss) and subcutaneous fat loss (loose skin) toward the end of the study; shedding gradually declined during the recovery period. All CFE-treated animals had organ weights and organ-to-bw ratios comparable to those of the control group, and no treatment-related morphological or histopathological alterations were detected upon histopathological examinations. These initial brief reports on preclinical non-GLP studies provide some insight into the safety of CFE in Wistar rats.
Herein, we report on a more comprehensive preclinical GLP-compliant safety assessment of a standardized hydroethanolic extract of CFE. The studies include an in vitro bacterial reverse mutation assay (Ames test), an in vitro chromosomal aberration assay, a 6-month chronic oral toxicity, and a prenatal developmental toxicity study in Sprague-Dawley rats. The outcome of the Ames test was previously briefly mentioned in an abstract and book chapter; 26,27 herein, we provide the first detailed description on the method and results of this study.
All studies were conducted by INTOX (Pune, Maharastra, India) in accordance with the studies’ respective Organization for Economic Co-operation and Development (OECD) Guidelines, and all studies adhered to the principles set forth in the 1998 OECD GLP principles.
Materials and Methods
Test Article
The test article, CFE, is a patented and proprietary hydroethanolic extract of the dried aerial parts of C. fimbriata prepared by Green Chem Herbal Extracts & Formulations (Bangalore, Karnataka, India). CFE is a brown powder and standardized to contain no less than 25% pregnane glycosides and 10% saponin glycosides. CFE batches used in the studies had a pregnane glycoside range of 27.9% to 28.7% and a saponin glycoside range of 18.2% to 18.7%. Based on its pregnane glycoside and saponin glycoside content, stability testing performed by Green Chem employing a high-performance liquid chromatography (HPLC) method indicated CFE to be stable under normal storage conditions at room temperature (30 ± 2°C) for at least 66 months. CFE is almost completely soluble in water (solubility of >98%), and its pregnane glycoside content (identified as the putative active content) is completely soluble in water as tested by the HPLC method. CFE formulated in distilled water is also stable at room temperature (25 to 35°C) and at low temperature (4 to 10°C) for at least 7 days (HPLC analysis). CFE formulated in distilled water was also found to be adequately homogeneous at 10 to 100 mg CFE/mL. CFE is currently sold by GE Nutrients (formerly Gencor Pacific; GE Nutrients, Inc., Anaheim, California, USA) as the trademarked ingredient Slimaluma.
Ames Test
CFE was assessed for mutagenicity in an Ames test performed with 5 strains of Salmonella typhimurium bacteria (obtained from Bruce N. Ames, PhD, Berkley, California) including TA97a, TA98, TA100, TA102, and TA1535. 28,29 The study was conducted in accordance with the OECD 471 Guidelines for the Testing of Chemicals, Bacterial Reverse Mutation Test (Section 4, No. 471, adopted July 21, 1997). 30
A preliminary cytotoxicity test using strain TA100 found concentrations of CFE ranging from 150 to 5000 µg/plate to be noncytotoxic in both the presence and the absence of metabolic activation (rat liver S9); dimethyl sulfoxide served as the vehicle in which CFE was completely soluble with no evidence of precipitation up to the highest concentration tested (50 mg/mL). For the main study, all strains were exposed to the test article in triplicate at 15, 50, 150, 500, 1500, and 5000 µg/plate, in both the presence and the absence of S9. Sterile water served as the negative control. Sodium azide (for strains TA1535 and TA100), ICR 191 (for strain TA97A), 2-nitrofluorene (for strain TA98), and cumene hydroperoxide (for strain TA102) served as the positive controls in the absence of S9, while 2-aminoanthracene (for strain TA1535), 2-aminofluorene (for strains TA97A, TA98, and TA100), and dantron (for strain TA100) served as the positive controls in the presence of S9. The number of histidine revertant colonies per plate was counted after 48 hours of incubation at 37°C, and the test article was considered mutagenic if a concentration-related and/or a reproducible increase in colonies was observed. A biologically significant increase was assumed if the average colony counts were double than that observed in the vehicle control for all strains except TA1535, for which the increase was considered if triple than that of the vehicle control. The test article was considered nonmutagenic if no increase in the number of revertant colonies was observed and if there was no indication of a dose relationship. The entire study was carried out twice to assess the reproducibility of the results.
In Vitro Chromosomal Aberration Assay
Cytogenotoxicity of CFE was assessed in an in vitro chromosomal aberration assay performed with human peripheral blood lymphocytes in accordance with the OECD 473 Guidelines for the Testing of Chemicals, In Vitro Mammalian Chromosome Aberration Test (Section 4, No. 473, adopted July 21, 1997). 31
Preliminary cytotoxicity testing with cultured human lymphocytes at 500, 1500, and 5000 µg/mL found CFE to be noncytotoxic in both the presence and the absence of S9. Solubility testing of CFE at 255 and 267.5 mg/mL in distilled water (test vehicle) resulted in a suspension, and any undissolved components were removed by filtration through a 0.2-µm syringe filter. Analysis of the filtrate by HPLC indicated the test article’s glycoside concentration remained unaltered, with no evidence of precipitation at the highest concentration tested. For the main study, lymphocytes were exposed to 500, 1500, and 5000 µg/mL of the test article, in both the presence and the absence of S9. The cells were incubated for 3 hours at 37°C in either the presence or the absence of S9 and sampled at 24 hours (1.5 cell cycles). An additional test was conducted in which the cells underwent continuous 24-hour exposure to CFE in the absence of S9. Methyl methane sulfonate served as the positive control without S9, cyclophosphamide monohydrate served as the positive control with S9, and distilled water served as the negative control. Upon treatment with colchicine, 200 metaphases were evaluated microscopically and analyzed for chromosomal aberrations at each test concentration. The number of chromosomal aberrations (gaps, breakage, fragments, and exchanges) per cell was calculated, and the mitotic index was also determined. Experiments were conducted in duplicate, and the test article was to be considered clastogenic if it resulted in a concentration-dependent increase and/or a reproducible increase in the number of cells with chromosomal aberrations in the presence or absence of S9 over the tested concentration range. A P value of <0.05 was considered statistically significant.
Animal Studies
Animal studies were performed in accordance with recommendations set forth by the Institution of Laboratory Animal Research, National Research Council in the Guide for the Care and Use of Laboratory Animals, 32 and under the conditions recommended by the Committee for the Purpose of Control and Supervision of Experiments on Animals in Guidelines for Laboratory Animal Facility. 33
Chronic (6-Month) Oral Toxicity Study in Rats
The repeated dose oral toxicity of CFE was assessed in rodents in accordance with the OECD 408 Guidelines for the Testing of Chemicals, Repeated Dose 90-day Oral Toxicity Study in Rodent (Section 4, No. 408, September 21, 1998), 34 OECD 452 Guidelines for the Testing of Chemicals, Chronic Toxicity Studies (Section 4, No. 452, adopted May, 12 1981), 35 and US FDA Redbook 2000 standards, IV.C.4.a, Subchronic Toxicity Studies with Rodents. 36
Male and female Sprague-Dawley rats (bred at INTOX), aged 8 weeks and acclimated to the experimental room conditions, were housed in polypropylene cages, with 2 animals of the same gender and same dose level per cage, at 19 to 25°C, 30% to 70% relative humidity, and a 12-hour light–dark cycle. Animals received Nutrilab rodent pellet feed and potable water (filtered and ultraviolet irradiated through an Aqua guard water filter [Eureka Forbes Aquaguard Classic]) was provided ad libitum. All animals were subjected to veterinary examination to ensure good health prior to treatment.
Three treatment doses, for administration by oral gavage, were selected based on the OECD guidelines and included the limit dose of 1000 mg/kg bw/d. The treatment dosages were 100, 300, and 1000 mg/kg bw/d and consisted of 40 animals (20/gender) each. Two satellite groups, a control and high-dose group with 20 animals (10/gender) each, were included in which the animals received an additional 28-day recovery period after treatment termination. The test article was formulated and prepared daily in distilled water; stability and homogeneity analyses were performed on the formulated test article by HPLC. A single daily administration dose volume of 5 mL/kg bw was employed, and control group animals received distilled water at the same dose volume. The treatment continued daily for 180 days, and the satellite groups were observed for an additional 28 days to assess reversibility of effects and persistent or delayed toxicity. The bw range at the start of the study was 210 to 292 g for males and 167 to 222 g for females.
All examinations and observations were in accordance with OECD and Redbook guidelines mentioned previously. The animals were observed twice daily for signs of mortality and morbidity, while general cage-side clinical examinations were conducted daily throughout the treatment period. All animals were subjected to detailed clinical examinations prior to the study and weekly thereafter, including manipulative examination and behavior observations to assess potential neurotoxicity. Ophthalmological observations were performed at the start and the end of the study on rats in the control and the 1000 mg/kg bw/d groups. Sensory, grip strength, and motor activity assessments were made on these rats after 22 to 23 weeks of treatment. The bws were recorded at the start of the study (day 1), weekly thereafter, and at necropsy. The amount of food consumed by the rats per cage was recorded weekly and used to calculate daily food intake. Weekly food intake observations for the satellite groups continued until the end of the recovery period.
Blood samples were collected from 10 randomly selected animals per sex per group at set intervals for analysis during the first 2 weeks of treatment initiation, midway through the study (week 13), and at the end of the treatment period. During scheduled blood sampling, the animals were fasted overnight, and for each sampling interval the blood samples were collected from the same 10 animals. Clinical pathological examinations, including hematology and clinical chemistry screens, were conducted on the same animals as was a urinalysis (collected over 4 hours) during the last week of the study.
Upon termination of the treatment, all the animals were killed by exsanguination under carbon dioxide (CO2) anesthesia for gross pathological evaluations. At the end of the study, necropsy was staggered during weeks 26 and 27 and was accompanied with extended dosing. Terminally killed animals and animals that expired during the study underwent complete necropsy. Organ weight measurements were recorded for all the animals. Histopathological examinations and evaluations of the tissue were conducted on all rats in the control and the high-dose groups (tissue was stained with hematoxylin and eosin) and included evaluation of the gastrointestinal tract.
Neurotoxicity assessment of the test article was made based upon specific histopathological examination, and gross functional deficits were assessed by means of a functional battery of quantifiable observations and manipulative tests. Immunotoxicity was evaluated based on Basic Type 1 Tests, namely, hematology, clinical chemistry, histopathological, and organ/bw indicators.
Bartlett test was used to compare bws, food intake, hematology, clinical chemistry, and organ weights for homogeneity. Data with homogenous intragroup variances were subjected to 1-way analysis of variance (ANOVA). Dunnett pairwise comparison was used for statistically significant results. A P value of <0.05 was considered statistically significant.
Developmental Toxicity Study in Rats
A prenatal developmental toxicity study of the test article was conducted in Sprague-Dawley rats in order to determine the effects of the test article on pregnant females and on embryo-fetal development. The animals were monitored for maternal toxicity and the effects of the test article on the developing organism, including death, structural anomalies, and altered or retarded growth. This study was conducted in accordance with the OECD 414 Guidelines for the Testing of Chemicals, Prenatal Development Toxicity (Section 4, No. 414 adopted January 22, 2001) 37 and US FDA Redbook 2000 standards, IV.C.9.b., Guidelines of Developmental Toxicities Studies (July 2002). 38
Male and nonpregnant nulliparous female rats (bred at INTOX), aged 13 to 14 weeks, were subjected to veterinary examination to ensure they were healthy. The weight variation in animals prior to mating did not exceed ±20% of the mean weight (both genders). Animals were housed in polypropylene cages at 19 to 25°C and 30% to 70% relative humidity with a 12-hour light–dark cycle. The animals received Nutrilab rodent pellet feed and potable water (passed through Aqua guard water filter) was provided ad libitum. A 5-day laboratory acclimation period preceded the initiation of the study. Three animals of the same sex and dose group were housed per cage during the premating stage. During the mating period, the animals were housed 1 male and 3 females per cage. Mating was confirmed if sperm was detected in vaginal smears (gestation day 0) and female rats were caged individually in postmating period.
A preliminary dose range-finding study on pregnant rats (10 female rats per group with a minimum of 7 pregnant females at the end of gestation) indicated that exposure to the test article at dose levels of 250, 500, and 1000 mg/kg bw/d from gestation days 6 to 19 did not result in any maternal or fetal toxicity. Mated female animals were assigned to control and treatment groups, 25 animals per group. The test article was freshly prepared daily in water and administered by oral gavage at a dose level of 10 mL/kg bw/d from gestation day 6 to 19 (1 day prior to the expected parturition) at dose levels of 250, 500, and 1000 mg/kg bw/d. The vehicle control groups received distilled water.
All examinations and observations were in accordance with the OECD and Redbook guidelines mentioned previously. The animals were examined twice daily during the gestation period for mortality and morbidity and observed for toxicity and clinical abnormalities. On nonweek days, all observations were made once daily. Dead animals were subjected to necropsy examination. The amount of food consumed was recorded daily for each animal, and the bws of the female rats were recorded on gestation day 0, before the first treatment was administered, and then on days 6, 9, 12, 15, 18, and 20. At the end of the treatment period (day 20 of gestation), all the animals were killed by CO2 euthanasia and subjected to necropsy examination and observed for gross pathological changes. Fetuses were delivered by cesarean delivery. The intact uterus was removed from each animal and weighed to determine the adjusted bw gain. The ovaries of the females were examined, and the uterus content was examined for embryonic or fetal deaths. The weight and sex of each fetus were determined. Fetuses were examined for gross external abnormalities and alternately assigned for soft tissue or skeletal examination. Following external examination, the fetuses selected for visceral organ/soft tissue examination were preserved in 70% alcohol for evaluation of soft tissue abnormalities. Fetal abnormalities were recorded as follows: normal variants for incidental abnormalities, minor abnormalities for malformations not expected to directly affect fetus survival and major malformations, which were considered to be of teratological significance and/or life threatening. Fetuses selected for skeletal examination were eviscerated, skinned, and stained with Alizarin red for 24 hours in order to detect skeletal abnormalities.
Statistical analysis was performed with litter as the basic sampling unit (litter incidence) and by fetal incidence. Bartlett test followed by ANOVA was used to compare maternal bws, weight gains, food intake, number of corpora lutea, number of implantations, and mean fetal weights. The Mann-Whitney U test was used for the number and percentage of early and late resorptions, fetus mortality, and percentage of pre- and postimplantation loss. Statistical analysis on the litter size was performed using the Student’s t test. A P value of <0.05 was considered statistically significant.
Results
Bacterial Reverse Mutation (Ames) Assay
The test results indicated that the revertant frequencies in treated S. typhimurium strains TA97a, TA98, TA100, TA102, and TA1535 were comparable to those observed in the vehicle controls at all CFE test concentrations. Plate counts for histidine revertants in the control/vehicle groups were within the frequency ranges expected from the historical control data at INTOX laboratory and the reported literature ranges. 29 Concurrent positive controls in the presence and absence of S9 demonstrated sensitivity of the assay. Therefore, the test article had no mutagenic effect on the Ames test up to the maximum tested level of 5000 µg/plate.
In Vitro Chromosomal Aberration Assay
The incidence of cells with aberrations at all CFE test concentrations was not dose-dependent and did not differ significantly from that in the vehicle control group. The number of cells with chromosomal aberrations was 3, 4, 3, and 6 in cells incubated for 3 hours in the presence of S9 at CFE concentrations of 0, 500, 1500, and 5000 µg/mL, respectively, and indicated no significant toxicity since no statistically significant increases were detected compared to control (0 µg/mL). This value was 18 for the positive control (P < 0.05). The number of cells with aberrations was 2, 4, 3, and 4 in cells incubated for 3 hours in the absence of S9 at CFE concentrations of 0, 500, 1500, and 5000 µg/mL, respectively, and also indicated no significant toxicity compared to control (0 µg/mL). The aberration value was 14 for the positive control (P < 0.05). Cells subjected to 24-hour exposure to CFE in the absence of S9 had aberration values of 3, 3, 3, and 5 at CFE concentrations of 0, 500, 1500, and 5000 µg/mL, respectively, and indicated no significant toxicity compared to control (0 µg/mL). This value was 16 for the positive control (P < 0.05).
The number of gaps, breaks, and acentric fragments in cells treated with CFE at and up to the highest CFE concentrations did not differ with statistical significance from that observed for the solvent/vehicle control. Mitotic indices at all CFE concentrations compared to that observed for the solvent/vehicle control. Under the experimental conditions described, CFE at and up to the concentration of 5000 µg/mL did not induce any increase in the incidence of cultured mammalian cells with structural chromosome aberrations, either in the presence or absence of metabolic activation. The concurrent positive controls induced a significant increase (P < 0.05) in the incidence of cells with structural chromosome aberrations over that in the concurrent control groups, thereby validating the test system. The test article is therefore considered as nonclastogenic in human peripheral blood lymphocytes.
Chronic (6-Month) Oral Toxicity Study
During the 180-day treatment period, and the additional 28-day recovery period for the satellite groups, no incidences of treatment-related mortality and morbidity were observed at any of the tested dosages. A single death of a male rat in the 300 mg/kg bw/d group occurred during week 25 and was caused by gavage error, unrelated to the test article. Throughout the study, no significant clinical, neurological, or ophthalmological abnormalities were observed. A few cases of diarrhea, nasal discharge, increased lacrimation, corneal opacity, circling disorder, and hypoactivity were observed in male rats only. However, these isolated instances occurred without any dose-dependent trend and were considered unrelated to treatment and regarded as incidental. No significant changes in bw or average daily food intake were observed for any animals in any of the treatment groups (refer to Table 1 for a summary of the bw and food intake data).
Summary of Body Weight and Food Intake Data in the 6-Month Chronic Oral Toxicity Study.a
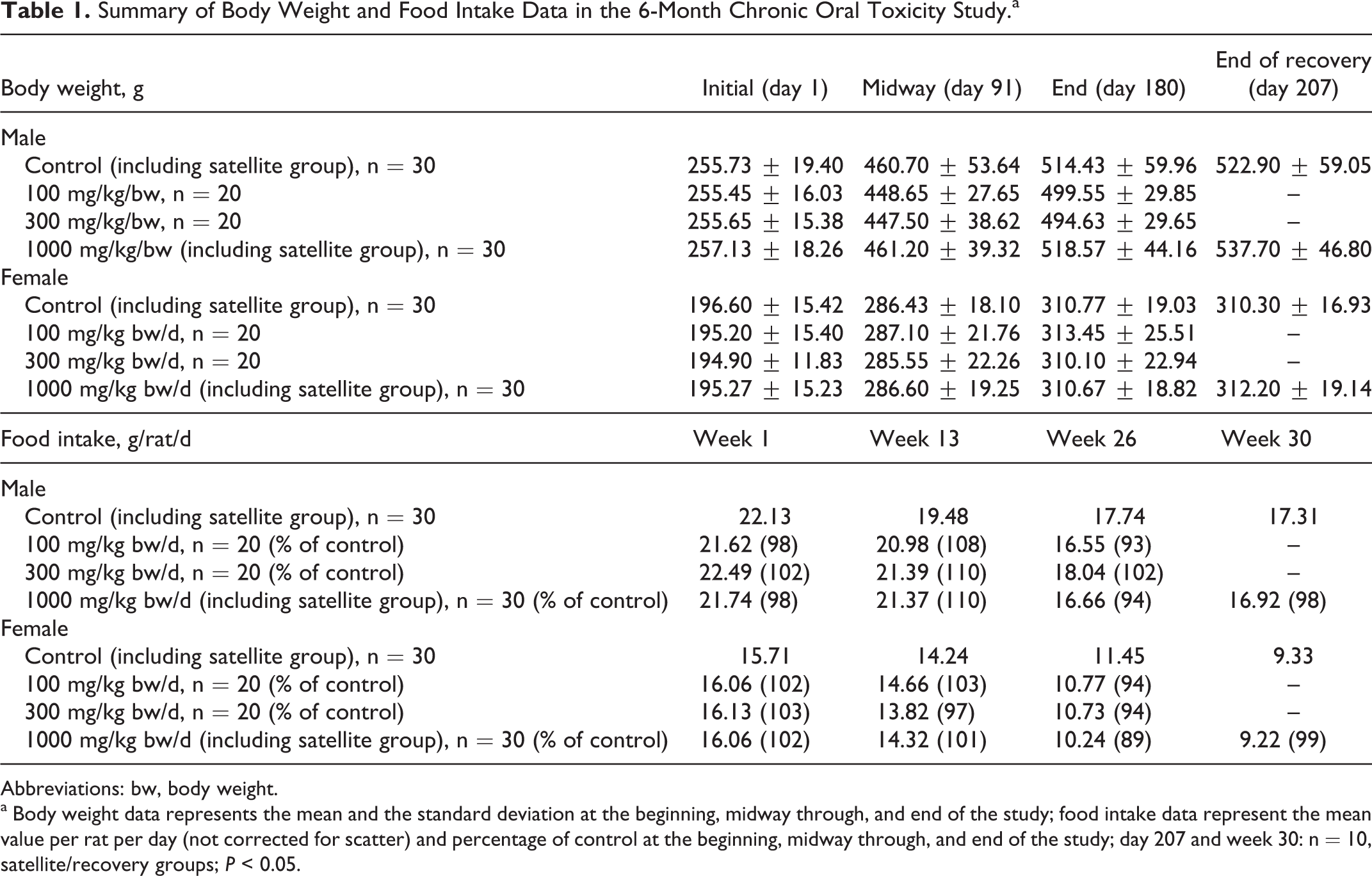
Abbreviations: bw, body weight.
a Body weight data represents the mean and the standard deviation at the beginning, midway through, and end of the study; food intake data represent the mean value per rat per day (not corrected for scatter) and percentage of control at the beginning, midway through, and end of the study; day 207 and week 30: n = 10, satellite/recovery groups; P < 0.05.
Hematological and clinical chemistry examinations conducted during the second week, midway through the study, and at the termination of the treatment did not indicate any remarkable alterations in the measured parameters as a result of the treatment at any dose level (supplementary Tables 1 and 2, respectively). Some statistically significant fluctuations were observed throughout the treatment period but all values remained within historical INTOX laboratory ranges and literature reported historical/control ranges for Sprague-Dawley rats 39 –45 and were not dose-dependent. As such, the observed hematological and clinical chemistry fluctuations were not considered to be of toxicological significance or concern.
Summary of the Developmental Study.a
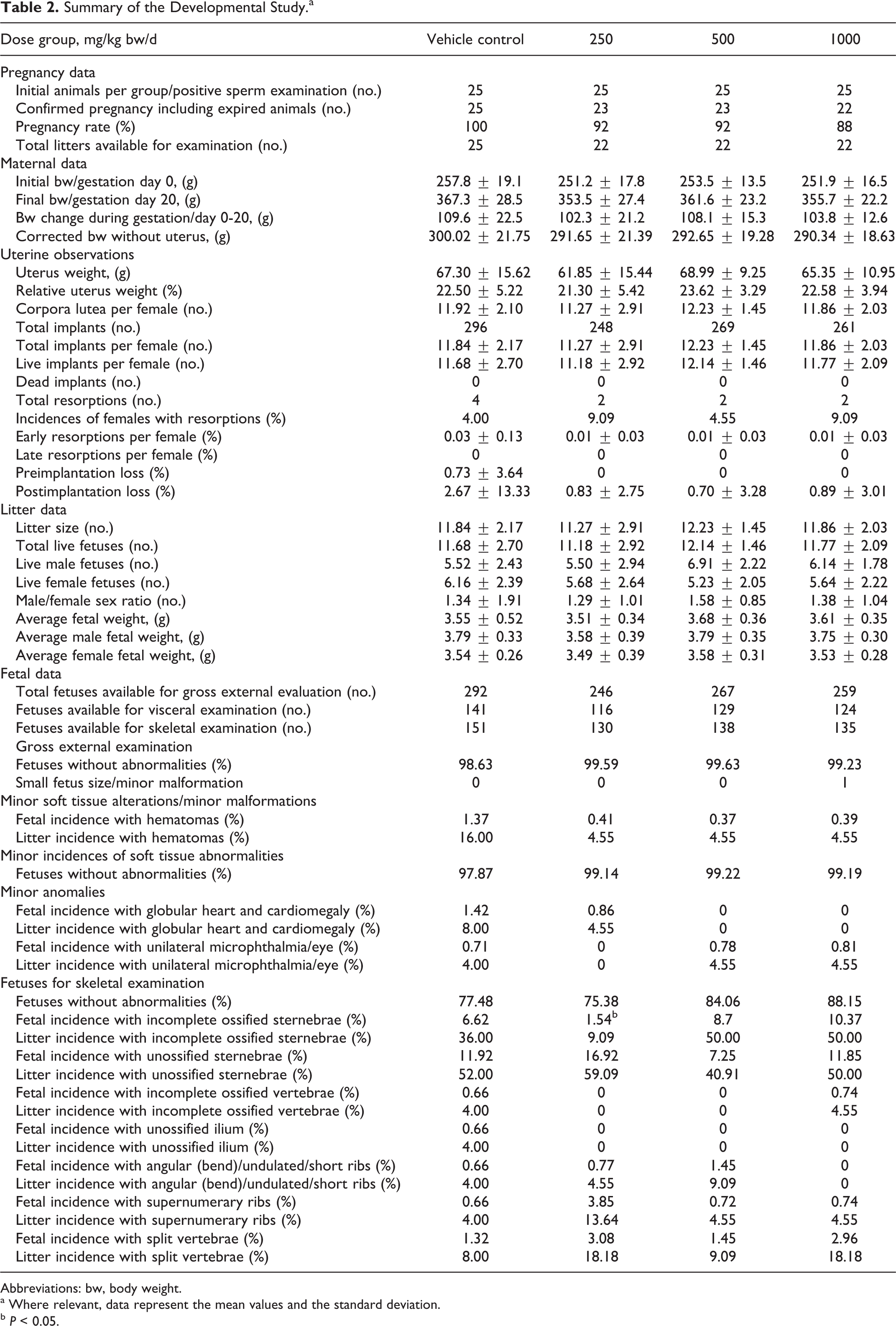
Abbreviations: bw, body weight.
a Where relevant, data represent the mean values and the standard deviation.
b P < 0.05.
Urinalysis performed during the last week of the treatment period did show an increased pH for all the animals in the 100 mg/kg bw/d group and for males in the 300 mg/kg bw/d group but not for those in the highest dose groups (data not shown). No indication of treatment-related deviations was detected, as all measured parameters were comparable to those of the controls with no dose-related alterations.
Organ weights recorded for animals in all dose groups at the end of treatment were found to be comparable to the values recorded for control groups with 1 exception (Supplemental Table 3). Males in the mid-dose group had statistically significantly higher mean weight of epididymides relative to bw. No treatment-related gross pathological or histopathological alterations in any of the organs or tissues of treated animals were detected.
Several insignificant necropsy findings included 1 instance of an undersized testis in a control male, a single instance of scattered white foci in lungs in a control female, multiple cysts in the kidney of 1 female in the 100 mg/kg bw/d group, and 1 female in the 300 mg/kg bw/d group with a white round nodular mass in its glandular stomach (confirmed microscopically to be a keratinized cyst). However, these necropsy results were isolated, not dose-dependent, and considered unrelated to the treatment. All microscopic changes noticed in this study appeared to be incidental, as their frequency and severity remained identical for the control and the treated animals.
The test article did not affect the structural or functional integrity of the central nervous system based on specific histopathological examination of the brain, spinal cord, and sciatic nerve. Histopathological examinations did not reveal any microscopic abnormalities and functional battery observations and manipulative tests did not detect any neurological, behavioral, or physiological dysfunctions. Through all examinations, serum and organ/tissue analyses and immune toxicity testing, tests failed to indicate the potential for any treatment-related neurotoxic or immunotoxic effects. Evaluation of hematological, clinical chemistry, and histopathological parameters, organ and bws, and immunotoxicity indicators revealed no alterations or abnormalities. Frequency of incidences of scattered focal mononuclear cell infiltration in nonlymphoid organs was comparable to that of the controls. With the absence of significant toxicological abnormalities, the no observed effect level (NOEL) was considered 1000 mg/kg bw/d, the highest dose tested.
Developmental Toxicity Study in Rats
Maternal, pregnancy, litter, and fetal data from the developmental toxicity study are summarized in Table 2. No evidence of maternal toxicity was observed in either the treatment (250, 500, and 1000 mg/kg bw/d) or the control groups. General toxicity measurements did not indicate any treatment-related alterations in any of the toxicity parameters, including mortality, clinical signs, bw gain, food intake, and gross pathology. All animals survived with the exception of 3 females that expired during the gestation period. Two females, 1 each in the low- and mid-dose groups, died due to gavage-related errors evident upon necropsy (frothy lungs and trachea content). The third female in the low-dose group exhibited nasal discharge and increased lacrimation the day prior to death, but necropsy did not reveal any abnormalities. As such, the deaths were regarded as incidental and not a result of test article exposure. CFE had no effect on food intake during gestation in any of the groups (data not shown) and bws for all treatment groups compared to that of the control; initial and final values are listed in Table 2. No remarkable alterations or observations surfaced during the uterine examinations and the uterus weights of animals in the CFE groups compared to those measured for the control animals. The test article also did not appear to have an effect on the rate of pregnancy or the percent incidence of miscarriages.
Observations made on the litters from the treatment groups did not reveal any remarkable differences compared to controls. The external examination did not indicate any treatment-related malformations. Isolated instances of hematomas, classified as minor malformations, were observed in all the groups, and 1 incidence of a small-sized fetus was observed for the high-dose group; these events were considered as isolated and incidental due to the absence of any statistical significance and dose-dependence. Investigation of the organs and soft tissue detected no treatment-related alterations. Minor soft tissue abnormalities included globular heart/cardiomegaly with a fetal incidence of 1.42% for control group and 0.86% in the low-dose group, with a litter incidence of 8.00% for control and 4.55% in the low-dose group. Unilateral microphthalmia was recorded with a fetal incidence of 0.71% for control, 0.78% for the mid-dose group, and 0.81% for the high-dose group; litter incidence was 4.00% for controls, 4.55% for the mid-dose group, and 4.55% for the high-dose group. These incidences of soft tissue abnormalities were considered incidental, insignificant, and not treatment-related, since they were isolated and comparable to that in the control group. Skeletal examination of the litters in the CFE groups indicated no treatment-related abnormalities or unusual variations (Table 2). All variations were comparable to those of the control and without any statistical significance except for 1; the fetal incidence of incompletely ossified sternebrae was significantly lower (P < 0.05) for the low-dose group (1.54%) compared to control (6.62%). No treatment-related abnormalities were detected in any of the treated or control fetuses. With the absence of maternal, litter, and fetal toxicological findings, the NOEL for pregnancy and fetal developmental effects in Sprague-Dawley rats was determined to be 1000 mg/kg bw/d, the highest dose tested.
Discussion
The hydroethanolic extract of dried aerial parts of C. fimbriata has been shown to significantly reduce waist circumference, hunger levels, and food palatability 22,23 and is sold as an appetite-suppressing dietary supplement, Slimaluma. Until now a comprehensive preclinical safety assessment has not yet been published on this extract. An acute oral toxicity study of CFE in Wistar rats (reported in both an abstract and book chapter) stated that no deaths occurred at up to 5 g/kg bw, the highest dose tested. 26,27 The same authors reported on a 90-day oral toxicity, which further indicated CFE (up to 900 mg/kg bw/d, the highest dose tested) to be relatively nontoxic in Wistar rats. These rodent studies provided some initial insight into the safety of CFE. The research herein assessed the genotoxicity of CFE in an Ames and chromosomal aberration test and evaluated the test article’s toxicological potential in 2 GLP-compliant animals toxicity studies, namely, a 6-month repeated dose chronic oral toxicity study and a developmental toxicity study in Sprague-Dawley rats.
No evidence of in vitro mutagenicity or clastogenicity of CFE was detected in the Ames test at concentrations up to 5000 μg of extract/plate or in the in vitro chromosomal aberration assay at concentrations up to 5000 μg of extract/mL, respectively. Both in vivo assessments revealed CFE to be nongenotoxic up to the highest concentrations tested in the presence and absence of metabolic activation.
A single death, caused by gavage error, occurred in the 6-month study in Sprague-Dawley rats. Minor and isolated events related to general clinical observations were recorded but considered incidental and unrelated to CFE consumption. No indication of toxicity was observed throughout the study at and up to 1000 mg/kg/bw, the highest dose tested. The bw and food intake data for all treated animals compared to controls (Table 1), indicated that CFE did not have any effect on the weight or appetite of Sprague-Dawley rats over the course of 6 months. Although this finding is of interest due to the nature of this ingredient, this study was not designed to study efficacy. Additionally, the study design of the published efficacy studies differed from the current study in subject selection (overweight humans versus normal weight rats) and in diet (dietary restrictions and/or high-fat diet versus normal rat chow). 22 –24 Reviews of the efficacy/potential mechanisms of action of CFE have also already been conducted. 3,25,46
Several statistically significant fluctuations in various hematological and clinical chemistry measurements were observed in the 6-month chronic oral toxicity study, but remained within historical INTOX laboratory ranges and literature reported ranged for Sprague-Dawley rats, and were not considered treatment-related or of toxicological concern. No targeted tissue or organs were identified, and microscopic examination of the upper and lower gastrointestinal tract organs and tissue did not indicate any evidence of treatment-related abnormalities.
The previously published 90-day oral toxicity in Wistar rats reported 4 unexplained deaths as well as sparse hair (hair shedding/loss) and subcutaneous fat loss (loose skin) in male rats toward the end of the treatment period; 26,27 none of these events occurred in the longer 6-month chronic oral toxicity study described herein, and prolonged oral administration of CFE to Sprague-Dawley rats resulted in no toxicological findings.
Finally, no maternal or embryonic-fetal development abnormalities surfaced in the prenatal developmental toxicity test at levels up to 1000 mg/kg bw/d conducted in Sprague-Dawley rats. The results revealed no evidence of maternal, pregnancy, litter, or fetal toxicity at and up to the highest dose tested.
In conclusion, the compilation of toxicological studies supports the safety for the repeated oral consumption of the hydroethanolic extract of C. fimbriata. Based on the results of the 6-month chronic oral consumption study in Sprague-Dawley rats, the NOEL is considered 1000 mg/kg bw/d, the highest dose tested.
Footnotes
Authors’ Note
Declaration of Conflicting Interests
The author(s) declared no conflicts of interest in regard to the research, authorship, and/or publication of this article.
Funding
The author(s) disclose that financial support for the research, authorship, and/or publication of this article: GE Nutrients (formerly Gencor Pacific).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
