Abstract
Various oximes are currently fielded or under investigation in the United States and other countries as a component of autoinjector emergency treatment systems for organophosphate nerve agent chemical weapons. Bis-pyridinium oximes in general have greater efficacy against a broad spectrum of nerve agents, but they have poor stability due to hydrolytic degradation at elevated temperatures. 1,1′-Methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate (MMB4 DMS) is a leading candidate for next-generation nerve agent treatment systems, because it is more stable than other bis-pyridinium oximes, but it still degrades quickly at temperatures often encountered during storage and field use. The primary goal is to increase the stability and shelf life of MMB4 while maintaining the desirable pharmacokinetic (PK) properties of the aqueous formulation. We have developed a formulation to be used in a phase 1 clinical trial consisting of MMB4 micro/nanoparticles suspended in cottonseed oil, a biocompatible vegetable oil. Through various milling techniques, the average particle size can be controlled from approximately 200 to 6000 nm to produce non-Newtonian formulations that are viscous enough to resist rapid particle sedimentation while remaining injectable at a range of concentrations from 5 to 400 mg/mL. The preliminary accelerated stability test shows that MMB4 in these formulations is stable for at least 2 years at temperatures up to 80°C. Preliminary preclinical in vivo studies have demonstrated that all concentrations and particle sizes have desirable PK properties, including high bioavailability and rapid absorption, which is critical to combat potent and fast-acting nerve agents.
Introduction
The US military is deployed on many global fronts and exposed to a large range of offensive weapons under a large range of climatic conditions. In order to combat a number of these arms, countermeasures have been developed. In the case of chemical weapons, the US military has specifically developed medical chemical countermeasures. Most of these countermeasures are delivered by autoinjectors carried by the war fighter and stockpiled in theater. Product stability and long shelf life at battlefield conditions are essential for these types of products. The task of keeping these drugs deployed in temperature-controlled conditions (cold chain) is difficult, and the cost, equipment, and manpower required would be large. Therefore, when developing medical chemical countermeasures, stability is paramount.
A range of medical chemical countermeasures of interest are used to treat organophosphate (OP) nerve agent exposure. The extreme toxicity of OP nerve agents results from the inhibition of the enzyme acetylcholinesterase (AChE) that hydrolyzes acetylcholine. 1,2 The accumulation of acetylcholine at muscarinic receptors may lead to rapid incapacitation and death; therefore, in the event of a nerve agent attack, prompt administration of antidotes is critical to save lives and minimize permanent physiological damage. Emergency treatment of OP intoxication typically employs 1 or more autoinjectors for intramuscular (IM) injection of an anticonvulsant such as diazepam, an antimuscarinic such as atropine, and an oxime compound to reactivate the inhibited AChE. 3 In the United States, this oxime has been the mono-pyridinium mono-oxime 2-pralidoxime chloride (2-PAM), but its lack of effectiveness against certain OP agents has led to the development of new oximes. 4 Bis-pyridinium bis-oximes are of particular interest, and MMB4 DMS (1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate) in particular is a leading candidate to replace 2-PAM. 5 –7 The MMB4 salts are generally very stable in solid form, but in aqueous formulations MMB4 is prone to hydrolytic cleavage, leading to poor stability (Figure 1). Elevated temperature is known to accelerate this hydrolytic cleavage reaction, leading to poor field stability and insufficient shelf life at temperatures above 25°C. 8
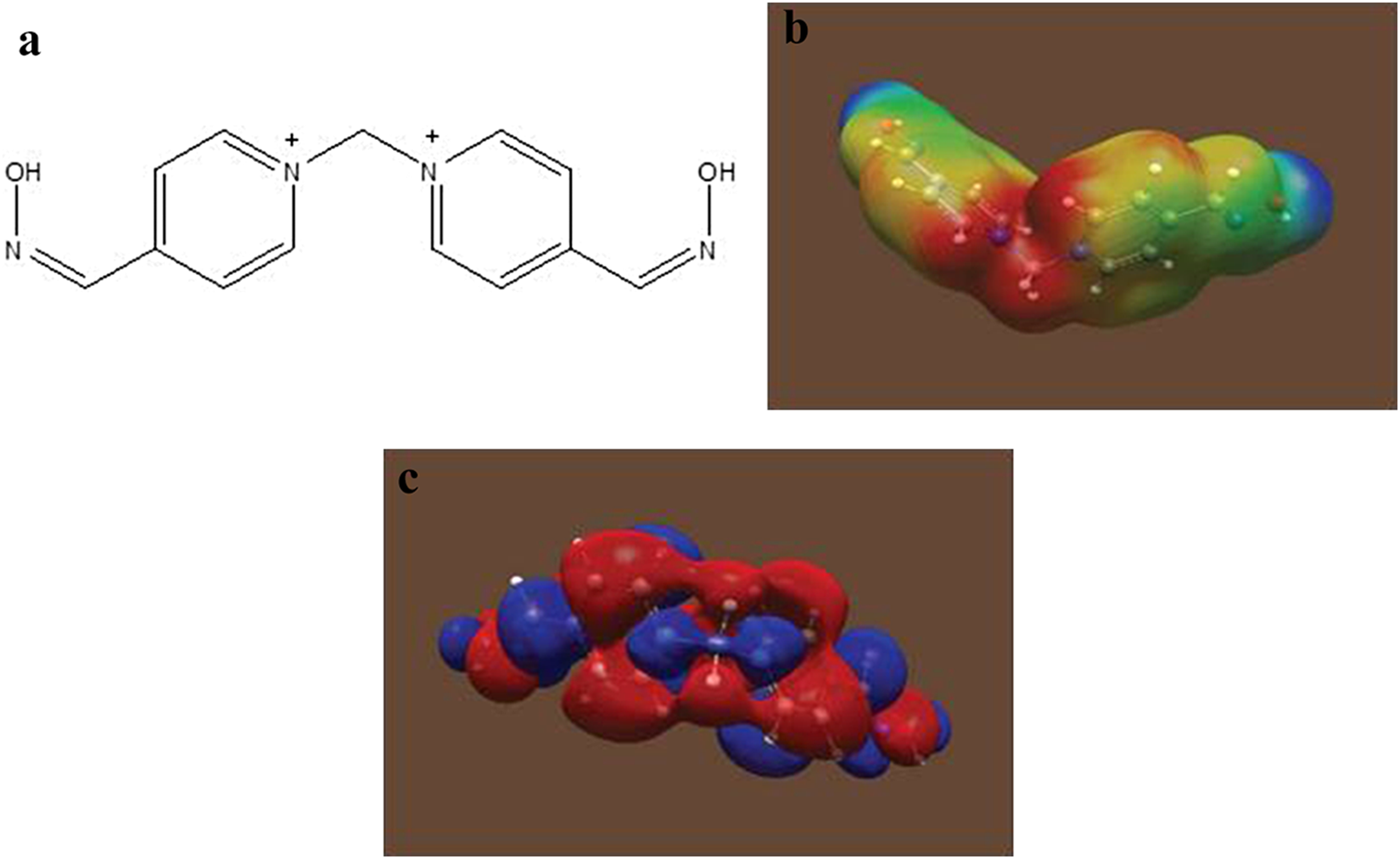
Explicit calculation of the electrostatic potential confirms that the central methylene group is an electropositive region (b, shown in red). However, this region of positive character is, in the equilibrium geometry, oriented away from the direction the ring faces and is instead more closely associated with the orientation of the central methylene hydrogen atoms. In terms of reactivity toward water or hydroxyl ion, one would expect attack in the regions of most positive potential. Note the expected orbital density on the back side of the methylene C-N bonds (c). This central blue region in the figure represents the overlap of the crossed C-N σ antibonding orbital lobes that are the most likely sites of an SN2-type nucleophilic attack. Their orientation largely determines the reaction geometry for an SN2 mechanism. a, Structure of MMB4 cation. b, Ab initio electrostatic potential of MMB4 cation. c, Ab initio calculation for LUMO of MMB4 cation. LUMO indicates lowest unoccupied molecular orbital; MMB4, 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium.
We report the development of IM autoinjectable formulations of MMB4, which maintain the stability of the solid. These non-Newtonian liquid formulations consist of MMB4 DMS microparticles and/or nanoparticles suspended in biocompatible cottonseed oil (CSO). 9,10 The hypothesis is that the CSO will protect MMB4 from water thereby eliminating the hydrolytic degradation process. The suspension formulation will still allow quick release of MMB4 into the blood stream after IM injection, and it will maintain the desired pharmacokinetic (PK) properties of aqueous formulation. The viscosity of these non-Newtonian enhanced formulations (EFs) is strongly dependent on the concentration and size distribution of the suspended particles. This relationship must be maintained in a suitable range to minimize the effects of sedimentation on the delivery of the correct dose and allow the formulation to flow during injection. Therefore, the particle size distribution must be adjusted to match the desired solid concentration that could vary significantly for a range of dose levels. In this study, several different milling techniques have been used to produce different particle size distributions of MMB4 DMS, including a ball mill, comminutor impact mill, centrifugal impact mill, jet mill, and bead mill. The crystal structures were characterized before and after the size reduction, and the polymorph was conserved (Figure 2). The stability of the MMB4 DMS EF suspension was determined at ambient and elevated temperatures for 2 years, and the CSO suspensions showed superior stability to comparable aqueous solutions. The EFs maintain the favorable PK properties of comparable aqueous solutions, such as rapid absorption and high bioavailability. 11
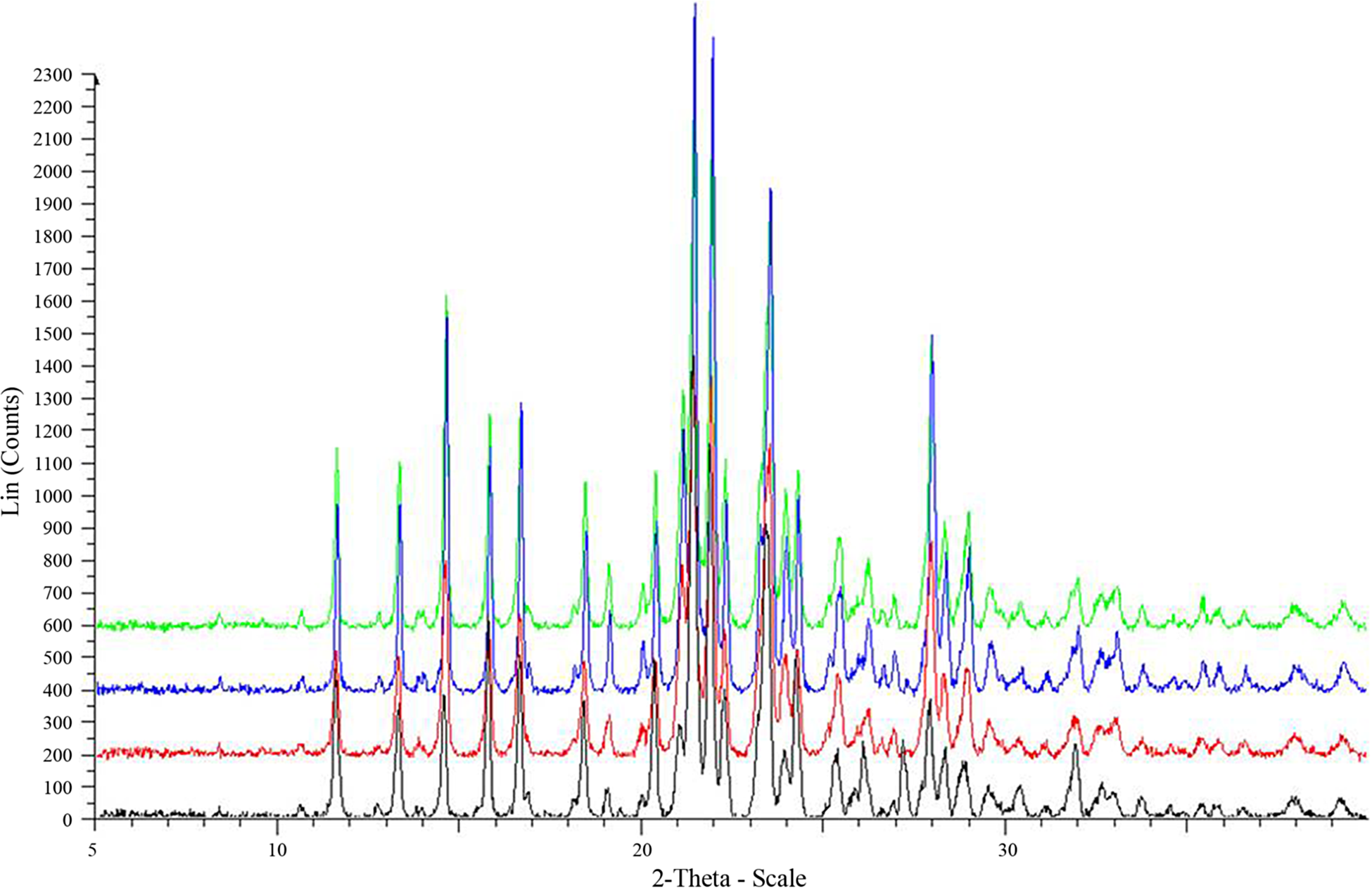
Powder X-ray diffraction patterns MMB4 DMS processed with different particle size reduction techniques: jet mill (green), pin mill (blue), bead mill (red), and unmilled MMB4 DMS (black). The diffraction patterns of the milled samples are similar to the unmilled sample, indicating that the crystal polymorph is not affected by milling. MMB4 DMS indicates 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate.
Materials and Methods
Materials
High-performance liquid chromatography (HPLC)-grade acetonitrile (99.9%), methanol (99.9%), and phosphate-buffered saline (PBS) were purchased from Fisher Scientific (Waltham, Massachusetts) and used as received. Reagent-grade methanesufonic acid (98%) and 4-pyridine aldoxime (4-PA, 98%) were purchased from Sigma-Aldrich (St. Louis, Missouri) and used as received. The CSO (National Formulary grade) was purchased from Spectrum Chemical Mfg Corp (New Brunswick, New Jersey). The MMB4 DMS (assay: 102%) was manufactured by SwRI (San Antonio, Texas).
High-Performance Liquid Chromatography
The quantification of MMB4 and its degradation products was performed by HPLC (Waters HPLC system; Waters Corporation). Separations were performed on reversed phase columns with ultraviolet (UV) or diode array detectors.
For MMB4, HPLC was performed on a Waters 2695 or Agilent 1100 system on a reversed-phase C18 column (Waters Atlantis HILIC, 4.6 × 150 mm2, 5 μm; Waters Corporation ) at a flow rate of 1.3 mL/min equipped with a UV detector set at 297 nm. The mobile phase consisted of 80/20 MeCN/0.05 mol/L ammonium formate buffer (pH 4). The major and minor peaks of MMB4 DMS and the impurity have retention times of 18, 21, and 25 minutes, respectively.
For quantification of the degradation product from MMB4 DMS, HPLC was performed on a reversed-phase C18 column (Discovery C-18, 3.5 × 150 mm2, Supelco; Supelco Inc, Bellefonte, Pennsylvania) at a flow rate of 1.5 mL/min with photodiode array detector set at 215 nm. The mobile phase consisted of a gradient of 90/10 (v/v) 25 mmol/L K2HPO4, pH 7.0/MeOH (solvent A2), and methanol (solvent B2) with the following conditions: 100% solvent A2 (0-20 minutes), 50% solvent A2/50% solvent B2 (20-25.5 minutes), and 100% solvent A2 (25.5-30 minutes). The 4-PA was eluted after 7 minutes.
Particle Characterization
The particle size distributions of MMB4 DMS particles were determined by 2 different techniques. Dry powders were characterized by laser diffraction using a Malvern Mastersizer 2000 (Malvern Instruments Ltd, Worcestershire, UK), and the results are reported as volume-weighted mean particle sizes. Nanoparticle suspensions were characterized by dynamic light scattering (DLS), also known as photon correlation spectroscopy, using a Brookhaven ZetaPALS 90Plus BI-MAS (Brookhaven Instruments Corporation, Holtsville, New York). The DLS experiments were conducted at a controlled temperature of 74°C after dilution of the samples with CSO to approximately 50 µg/mL, and the results are reported as unimodal intensity-weighted median particle sizes.
Environmental Scanning Electron Microscopy
Surface and morphology of the samples were examined with environmental scanning electron microscopy using a Carl Zeiss SMT EVO-50EP (Carl Zeiss, Oberkochen, Germany). The EVO-50 is equipped with a tungsten filament, extended pressure capabilities (10-3000 Pa), a 5-axis motorized stage, temperature control (−30 to 50°C), and 6 detection methods. In this study, the images were obtained at an accelerating voltage of 20 kV, under high vacuum, <1 × 10−3 Pa, constant temperature of 20°C, and with the variable pressure secondary electron detector and backscattered electron detector.
Physical Properties
Dynamic viscosity of the formulations was measured using 2 different techniques: a Brookfield rotational spindle viscometer (Brookfield Digital Viscometer, Model DV-I+; Brookfield Engineering Laboratories, Middleboro, Massachusetts) and a strain-controlled rotational rheometer (Rheometrics Fluids Spectrometer Model RFS2; Rheometric Scientific, Inc, Piscataway, New Jersey) with a 25-mm diameter parallel plate accessory. Injection forces were measured using a load cell and an electrodynamic test system (MTS Insight; MTS Systems Corporation, Eden Prairie, Minnesota).
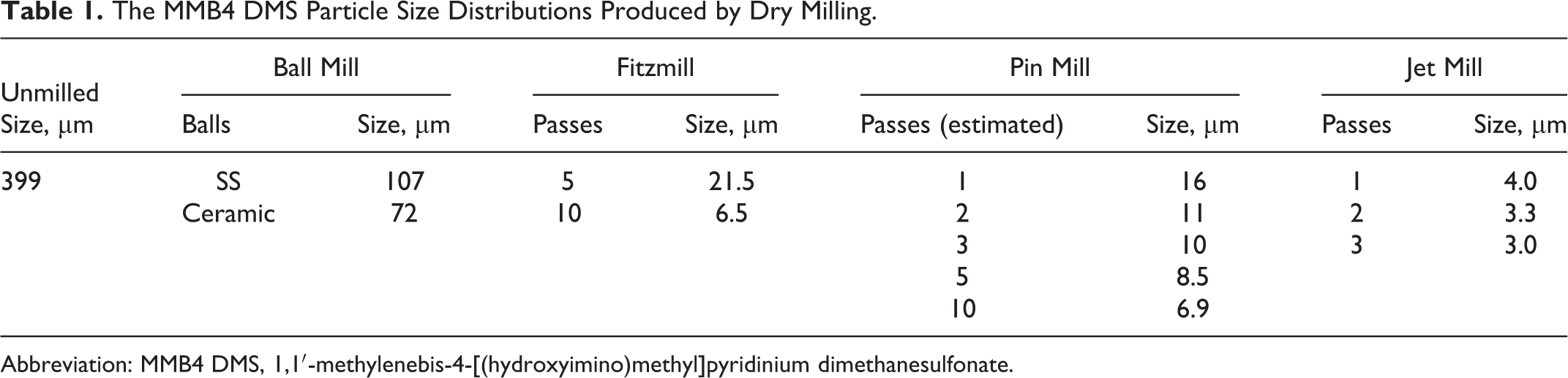
Milling
Four different types of mills were used to reduce the particle size of MMB4 DMS in dry powder form. A benchtop ball mill (Retsch PM100) was used in a batch process; stainless steel and ceramic balls were both used separately to mill the MMB4 DMS. Particle sizes of approximately 72 to 107 µm were achieved using this method. A comminutor impact mill or Fitzpatrick mill (Model LA1 Fitzmill; The Fitzpatrick Company, Elmhurst, Illinois) was used in a batch process with up to 10 consecutive passes through the mill to produce volume-weighted average particle sizes from approximately 6 to 60 µm. A centrifugal impact mill or pin mill (CIM-18-SS; Munsen Machinery, Utica, New York) was used in a batch process and was also modified to use a closed-loop system for continuous recirculation. Volume-weighted average particle sizes of approximately 7 to 8 µm were achieved using this technique. Another type of impact mill, a jet mill (2-in Micro-Macinazione [M&M] Micronizing Jet Mill, M-3855; Micro-Macinazione SA, Molinazzo di Monteggio, Switzerland), produced volume-weighted average particle sizes of approximately 3 to 4 µm. All the previously discussed mills operated with MMB4 DMS as a dry powder. To reduce the particle size to submicron levels, an agitator bead mill (Glen Mills Dyno-Mill Multi-Lab; Glen Mills Inc, Clifton, New Jersey) was used in a wet milling process. This technique produced intensity-weighted median particle sizes of approximately 200 to 500 nm.
Homogenizer
The MMB4 DMS powder was dispersed in CSO with a rotor-stator dispersion tool, either an ULTRA-TURRAX T-25 digital (IKA, S25N-8G) at 24 000 rpm or an ULTRA-TURRAX T-50 (IKA, S50N-G45F) at 10 000 rpm.
In Vivo Studies
In vivo studies were conducted at Battelle (Columbus, Ohio). The dose proportionality study of MMB4 DMS EF in the Male New Zealand White rabbit has been described elsewhere. 11
Results and Discussion
The MMB4 DMS was synthesized by a process 12 involving a final crystallization step, which produced particles with a volume-weighted average size of approximately 400 μm (Figure 3). This is approximately the same size as the nominal inner diameter of a 22-gauge needle (413 µm), which is commonly used in autoinjectors. To be injectable as a suspension of particles in CSO, the particle size must be reduced significantly to allow the formulation to flow freely through a needle of this size. In addition, as will be discussed subsequently, the particle size has a profound effect on the physical properties of the suspension. The solid concentration of MMB4 DMS in the suspension also strongly affects the viscosity. As the particle size is reduced and/or the concentration is increased, the formulation becomes increasingly viscous and non-Newtonian, displaying shear-thinning behavior. A high zero-shear viscosity allows the formulation to resist sedimentation of the suspended particles, while still allowing flow through a needle under the high shear forces induced during injection. A key goal of this project was to enable production of injectable suspensions at a wide range of dose levels, all using the same liquid volume. Therefore, it was necessary to produce injectable formulations with concentrations from 400 to 100 mg/mL or below. Several different particle size reduction technologies were used to reach these targets.

Particle size and shape of unmilled 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate (MMB4 DMS) by environmental scanning electron microscopy (ESEM).
Dry Milling
The ball mill operates by introducing the MMB4 DMS into grinding jars containing stainless steel or ceramic balls. These grinding jars are subjected to superimposed rotational movements, and the difference in speeds between the balls and grinding jars produces an interaction between frictional and impact forces, which releases high dynamic energies. The MMB4 DMS was cooled by liquid nitrogen and ball milled for a total of 20 minutes with intermittent liquid nitrogen cooling to keep the temperature of the milled sample below 20°C. Specifically, about 30 g of MMB4 DMS was cryogenically ball milled (with eight 3/4-in-diameter stainless steel balls) in a stainless steel milling bowl for a total of 20 minutes at 500 rpm. About 5 g of MMB4 DMS was cryogenically ball milled (with three 3/4-in-diameter and three 3/8-in-diameter ceramic balls) in a ceramic milling bowl for a total of 20 minutes at 450 rpm. This technique produced volume-weighted average particle size of approximately 100 μm as shown in Table 1 and Figure 4.
A comminutor impact mill (Fitzmill) operates by introducing the material into a chamber in which a rotating blade assembly reduces the size of the particles by cutting or impacting them. The particles discharge through a screen that determines the final particle size. The MMB4 DMS was milled at the 50-g MMB4 DMS scale. The milling was conducted at 9000 rpm rotor speed. It was determined that ambient temperature milling of MMB4 DMS repeatedly for up to 10 times through the Fitz mill at 9000 rpm rotor speed significantly reduced the particle size of MMB4 DMS. This technique produced volume-weighted average particle sizes down to approximately 6 µm, as shown in Table 1 and Figure 4.
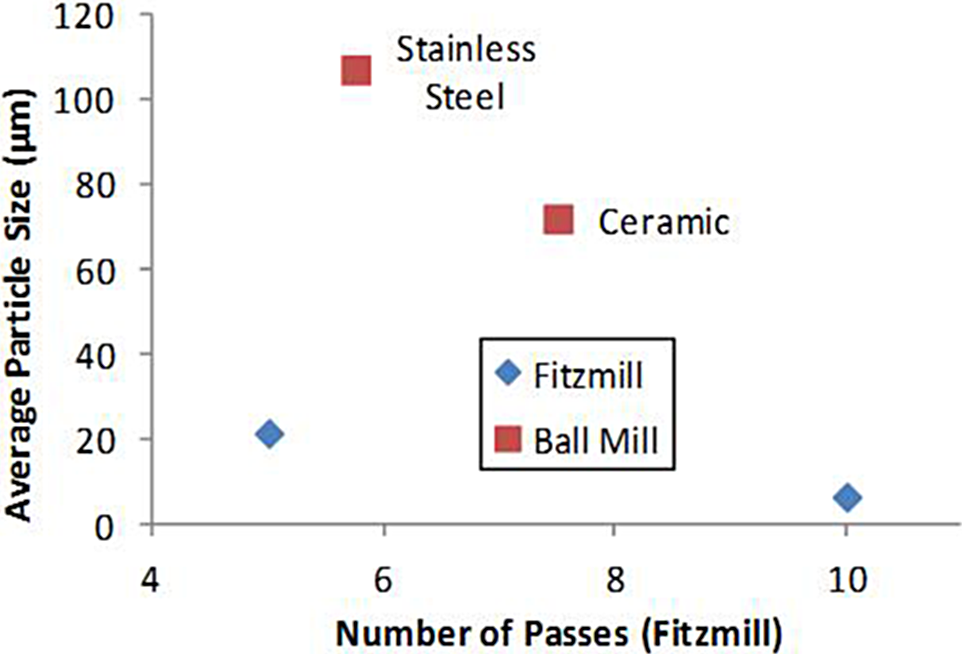
Volume-weighted average particle sizes of 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate (MMB4 DMS) samples produced by the ball mill and Fitzmill. Both ball-milled samples were produced with a milling time of 20 minutes but with different balls as shown.
The MMB4 DMS Particle Size Distributions Produced by Dry Milling.
Abbreviation: MMB4 DMS, 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate.
A centrifugal impact mill (pin mill) operates by introducing the particles into a chamber containing multiple pins arranged in concentric circles. As the pins move radially, they accelerate the particles that impact the pins and the chamber walls, thereby reducing the particle size. This mill was modified to use a closed-loop system for continuous recirculation. Therefore, the number of circulation loops (passes) was estimated by the time and feed rate. A scanning electron micrograph of a typical pin-milled sample is shown in Figure 5. Using a rotor speed of 90 Hz (5328 rpm), volume-weighted average particle sizes of approximately 7 µm were achieved, as shown in Table 1 and Figure 6.

Particle size and shape of milled 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate (MMB4 DMS; after centrifugal impact milling process) by environmental scanning electron microscopy (ESEM).
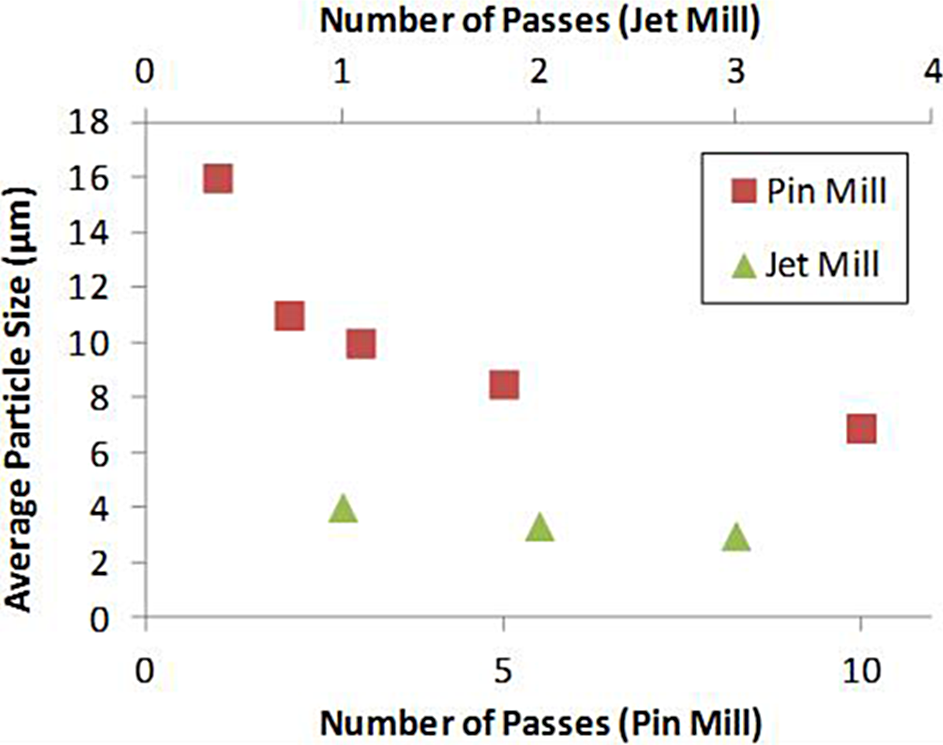
Volume-weighted average particle sizes of 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate (MMB4 DMS) samples produced by the pin mill and jet mill. Number of passes for the pin mill is estimated because it was run in closed-loop recirculation mode.
A jet mill operates using pressurized air or nitrogen to introduce the particles into a chamber. The particles are accelerated to high velocity by a series of gas jets around the chamber perimeter in a spiral pattern. Particle size reduction occurs when fast-moving particles in the spiral path collide with slow-moving incoming particles. Centrifugal force retains the larger particles around the chamber periphery, while the smaller particles exit with the exhaust gas from the center of the chamber. The jet mill was charged with approximately 200 g of MMB4 DMS, and the milled material was processed through the mill in a batch process a total of 3 times. This technique produced volume-weighted average particle sizes of approximately 3 to 4 µm, as shown in Table 1 and Figure 6.
The 4 different dry milling techniques produced a wide range of particle sizes (Table 1). The Fitzmill and pin mill produced similar minimum average particle size distributions (6-7 µm). The jet mill produced particles of approximately half that size (3-4 µm). The Fitzmill was the most versatile (especially if larger particles are required), but the jet mill produced the smallest particles.
Compounding
The dry-milled MMB4 DMS was compounded in CSO using a rotor stator homogenizer. The homogenizer consists of a high-speed rotor with a very close tolerance for the stator, which results in thorough and efficient mixing. The mixture is drawn by the turbine of the rotor and forced through the 2 rows of the stator, which generate a high shearing action. Homogenization ensures that a product has the same composition, structure, or characteristics throughout. The dry-milled MMB4 DMS was added to the vehicle of CSO with low-intensity mixing followed by homogenization to ensure uniform dispersion of MMB4 DMS at up to 400 mg/mL MMB4 DMS in CSO.
Wet Milling
To develop formulations with concentrations in the range of 100 mg/mL or below, a smaller particle size distribution was necessary to maintain the suspension stability. A wet mill was used to reduce the particle size into the nanometer regime. This mill operates by introducing the particles and CSO into a chamber containing zirconia grinding media (beads). The chamber also contains an agitator shaft that rotates, imparting kinetic energy to the mixture of MMB4 DMS particles, CSO, and beads, resulting in shear and impact forces that reduce the particle size distribution. A gap separator retains the beads while allowing the product to discharge from the mill. This mill can be used in a single-batch process and can also be used with a closed-loop recirculation system to reduce the particle size further. The final particle size of the milled product depends on the properties of the solids and can be optimized by several process parameters, including the solid concentration in the suspension, the diameter of the grinding beads, the rotational speed of the agitator shaft, the milling time, and the suspension feed rate. In our experiments, the zirconia beads ranged in diameter from 0.1 to 1.5 mm, the MMB4 DMS concentration in the suspension ranged from 1% to 12% (w/v), and the rotor speed ranged from approximately 2000 to 6000 rpm. The milling chamber was cooled by circulating coolant through a jacket that kept the temperature of the suspension sample below 70°C inside the milling chamber.
In a typical milling experiment, CSO was first charged into a stainless steel vessel equipped with overhead agitator and blanketed with nitrogen, followed by addition of MMB4 DMS. The mixture was pumped using a peristaltic pump into the grinding chamber, and the milled material exiting the chamber was collected in a second stainless steel vessel (also equipped with an overhead agitator and blanked with nitrogen). When the first vessel was empty, a 3-way valve was switched to continue pumping the mixture from the second vessel into the grinding chamber. Using this procedure and switching back and forth between the 2 collection vessels, the mixture was continuously recirculated through the mill. A scanning electron micrograph of a typical wet-milled EF sample is shown in Figure 7.

Scanning electron micrograph of wet-milled 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate (MMB4 DMS).
Figure 8 shows the reproducibility of the wet milling process for the preparation of MMB4 DMS nanoparticle suspension formulations. Two process conditions were selected, and the experiments were repeated 4 times. The 30 mg/mL formulation was obtained with 0.65-mm diameter zirconia beads and approximately 2400 rpm agitator speed, and the 100 mg/mL formulation was obtained with approximately 4200 rpm agitator speed. The milling lasted 2 hours for each process. Changes in particle size as a function of processing time are shown in Figure 8. In all the 8 experiments, the particle size decreased rapidly within 30 minutes from the initial 400 µm to about 400 nm and with much smaller change observed during the next 90 minutes. Prolonged milling times were not beneficial, because the particle size did not decrease further.
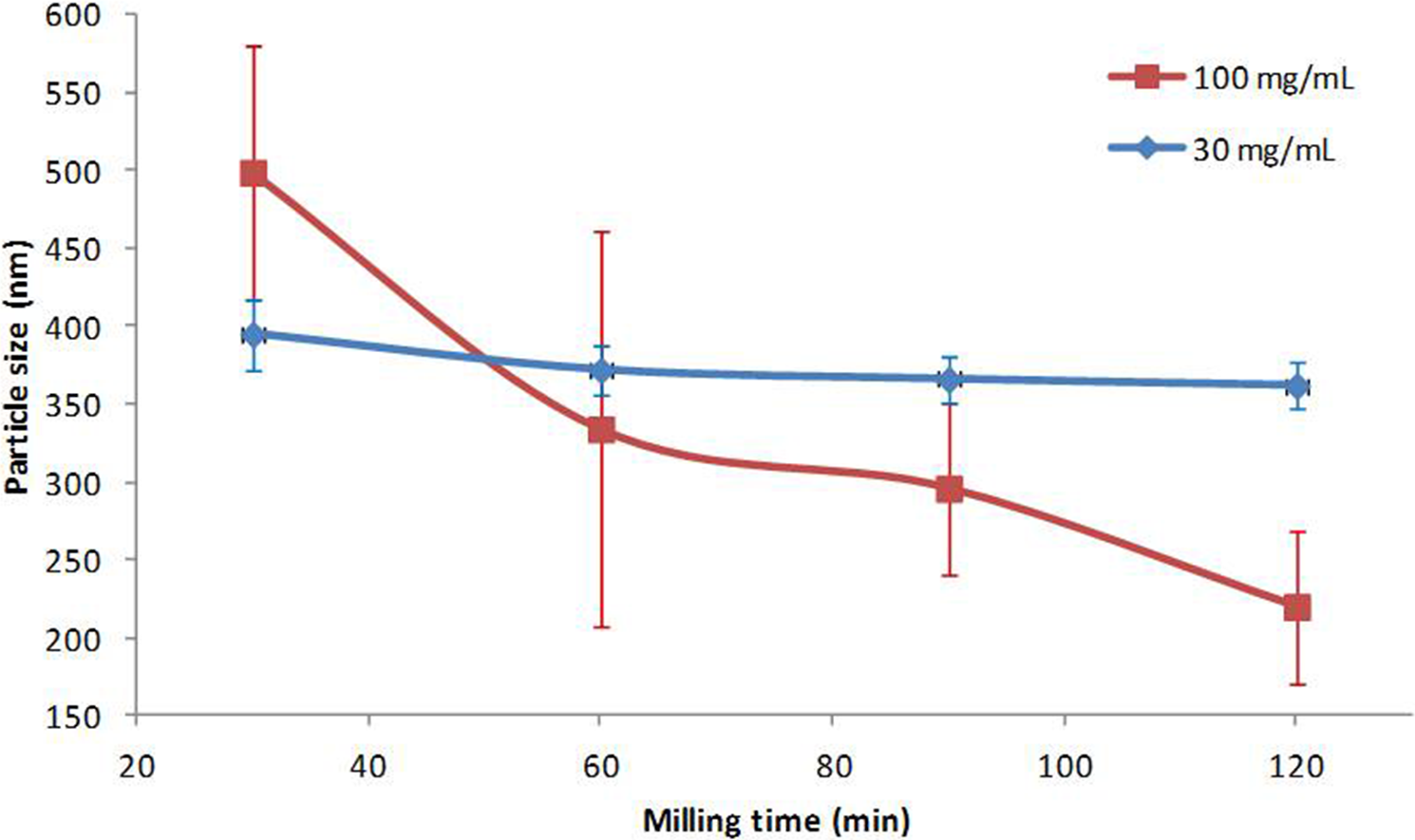
Particle size reduction during wet milling of 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate (MMB4 DMS) formulations at different concentrations. The majority of particle size reduction occurred in the first 30 minutes before the first data point (unmilled powder is approximately 400 µm). Each data point represents an average of 4 experiments, with error bars representing the standard deviation of the median particle sizes of the 4 experiments.
As particle size decreases, interparticle interactions become stronger due to the increase in total surface area, leading to an increase in bulk viscosity of the suspension. This relationship is evident in Figure 9.
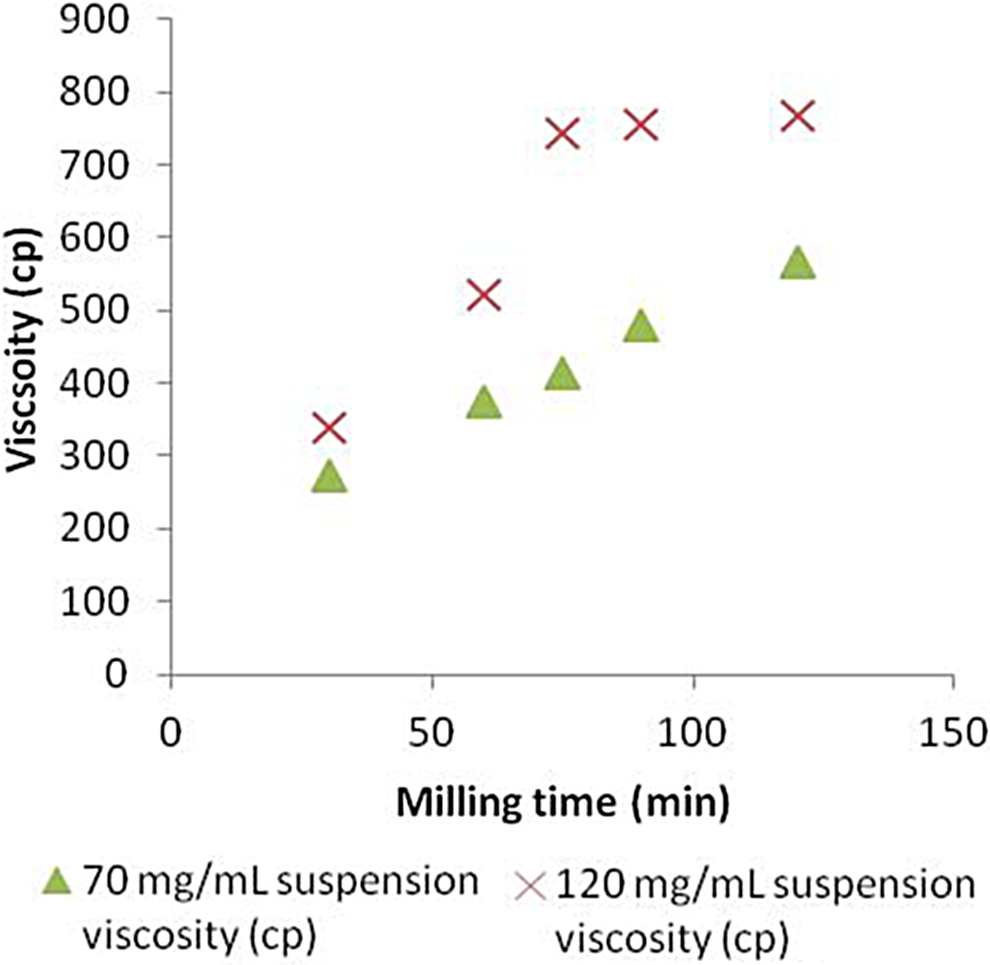
Viscosity as a function of milling time.
Resistance to Degradation of MMB4 DMS During Milling
The MMB4 DMS samples were analyzed before and after milling to assess the effect of milling on the drug stability. As demonstrated in Table 2, the milling processes caused negligible drug degradation.
The MMB4 DMS Particle Size and Purity After Milling Process.

Abbreviation: MMB4 DMS, 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate.
Injectability
The MMB4 DMS EFs are designed to be physically stable and injectable over a wide range of particle sizes and concentrations. Rheological data (Figure 10) show the shear thinning behavior over a range of temperatures from 0°C to 70°C.
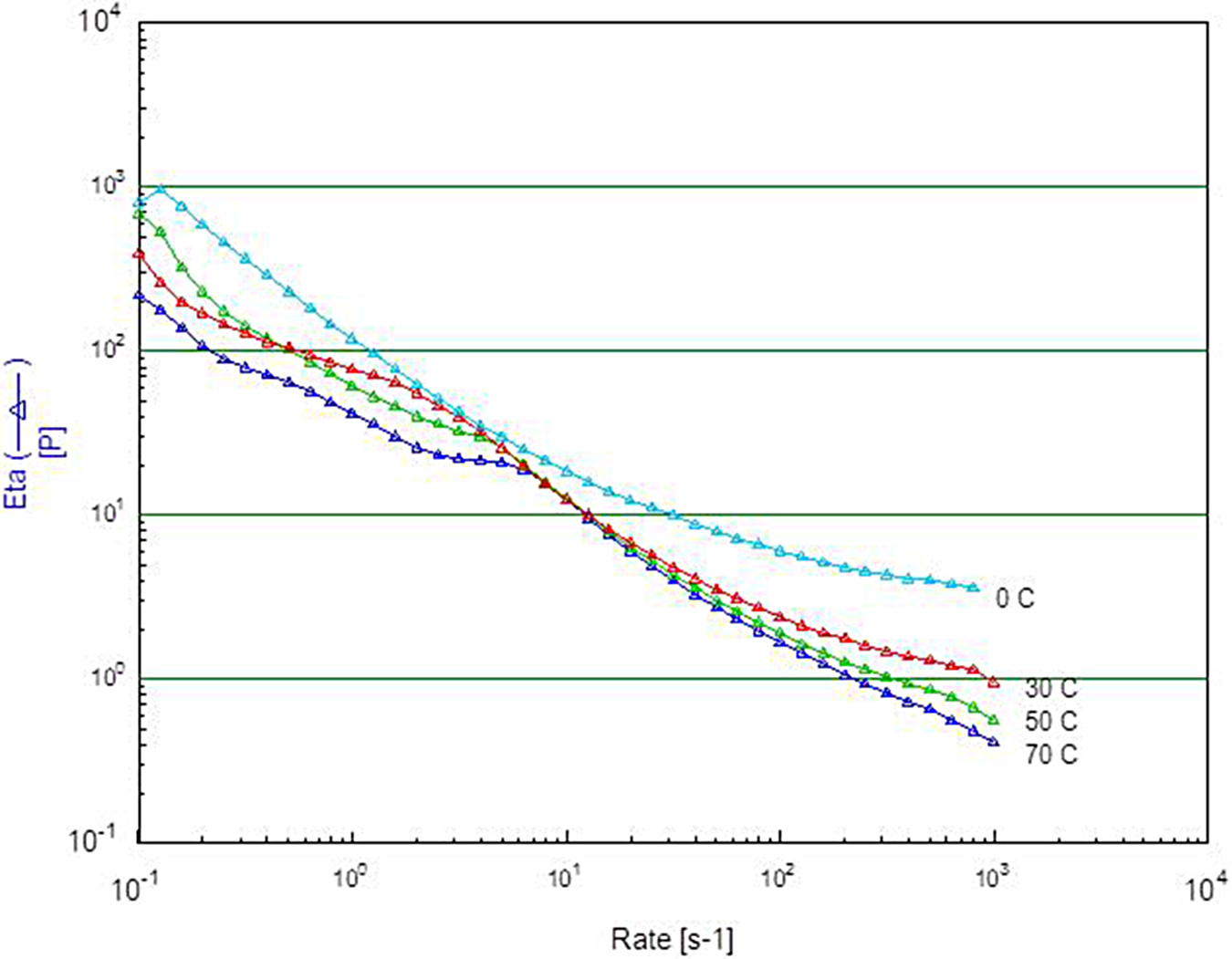
Dynamic viscosity of a 110 mg/mL 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate (MMB4 DMS)/cottonseed oil (CSO) suspension formulation.
The shear thinning shown in Figure 10 allows the viscous formulations to be delivered through syringes. After storage at room temperature (20°C-25°C) for 2 months, the milled MMB4 DMS EF demonstrated excellent physical stability as assessed by the injectability of the formulations (Table 3). Approximately 2 mL of MMB4 DMS EF was loaded into each of the autoinjector syringes (400 mg/mL samples were loaded in 5-mL syringes; whereas 100 mg/mL samples were loaded in 2-mL syringes). The syringe and plunger assembly with a 1-in, 22-gauge, luer-lock needle was mounted on an electrodynamic test system vertically with the needle pointed downward at room temperature. The syringe plunger was compressed at a constant speed, and the compressive stress was measured by a load cell. This study confirmed that MMB4 DMS in CSO suspension formulation is suitable for use in autoinjector syringes with 22-gauge needles, when 2 mL could be delivered within 6 seconds with a maximum load of 40 pounds.
Syringe Test Results.
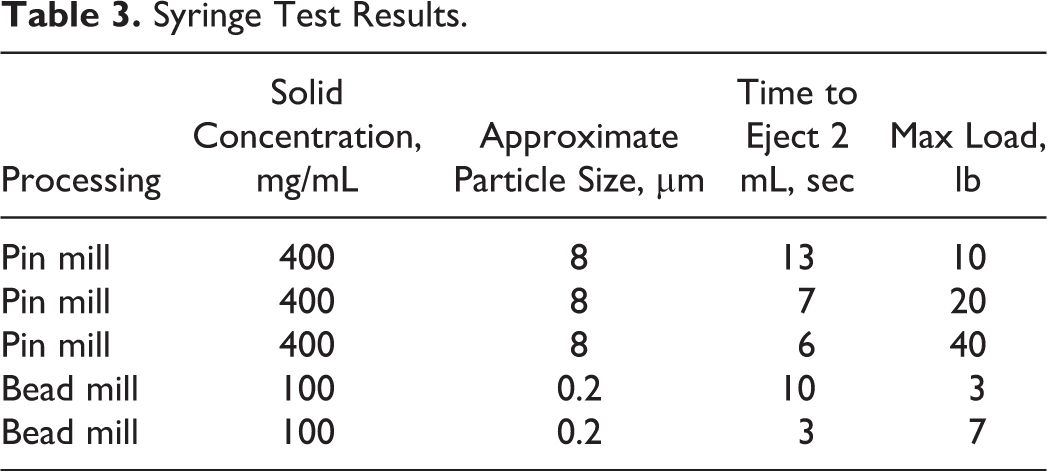
The MMB4 DMS EF samples were prepared with various drug concentrations by dilution or centrifugation/decantation from a wet-milled product. Table 4 shows an example of the formulations obtained from a wet milling process in support of a preclinical study. 11 The formulations were loaded into syringes and ejected to test dose delivery. Results shown in Table 4 indicated that the delivered doses were within 10% of the target doses.
Composition of Preclinical MMB4 DMS Suspension Formulations.
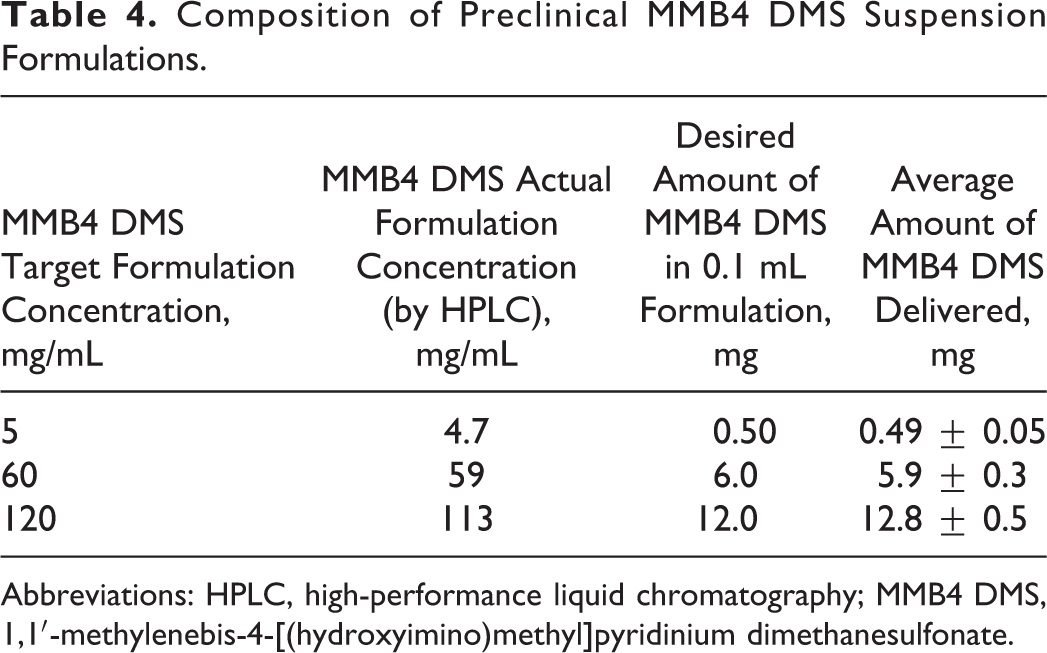
Abbreviations: HPLC, high-performance liquid chromatography; MMB4 DMS, 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate.
Chemical Stability
Solid MMB4 DMS is relatively stable in air, but it degrades much more quickly when dissolved in water and exposed to elevated temperatures (Figure 11). Since the EF retains the MMB4 DMS in solid form in a highly hydrophobic dispersion in CSO, the EF is very stable in air (Figure 11). Degradation is quantified by measuring the amount of 4-PA, which is the primary degradation product for hydrolytic cleavage of MMB4.
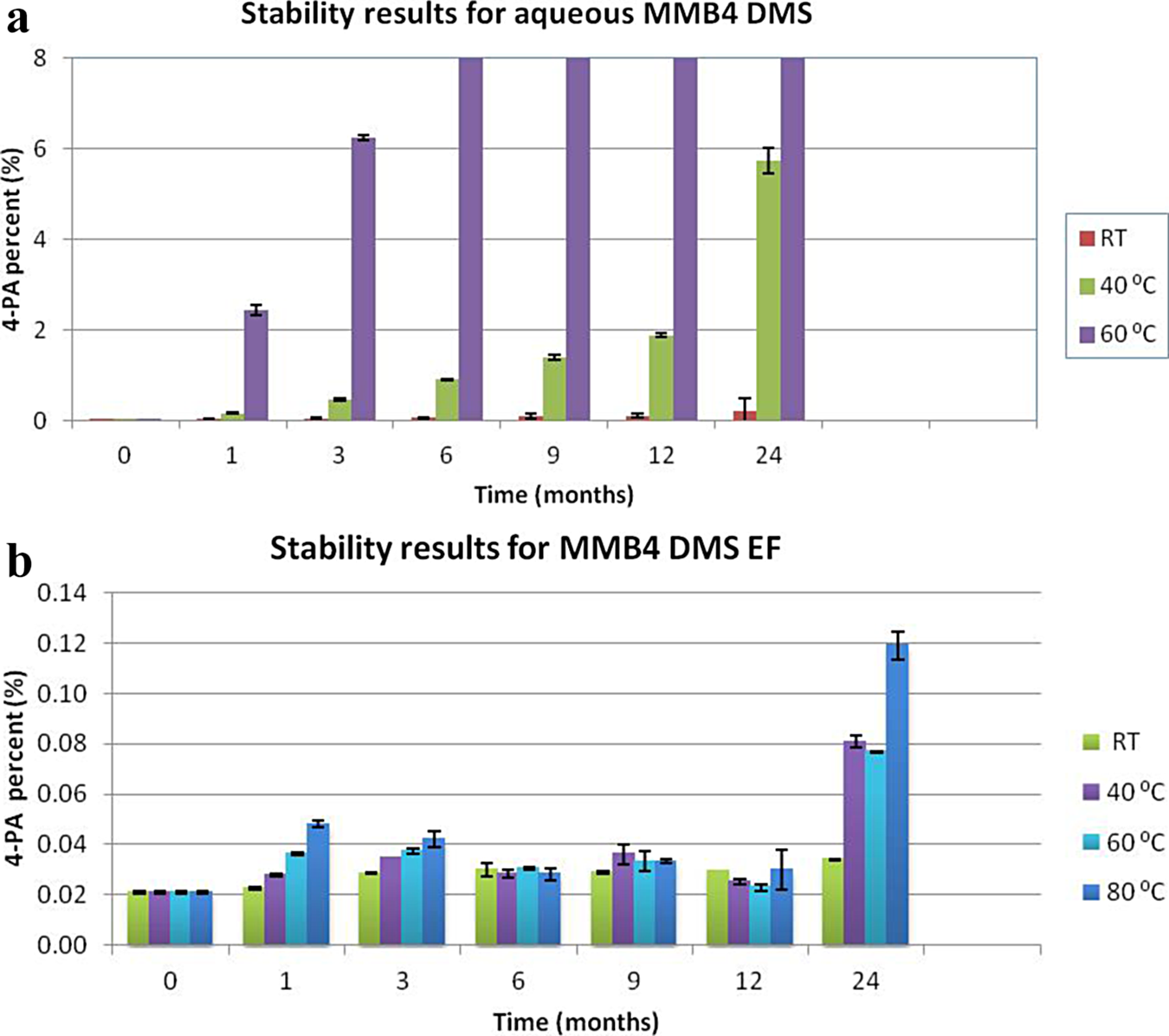
Stability of 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium dimethanesulfonate (MMB4 DMS) suspension formulation versus aqueous formulation.
Preclinical Studies
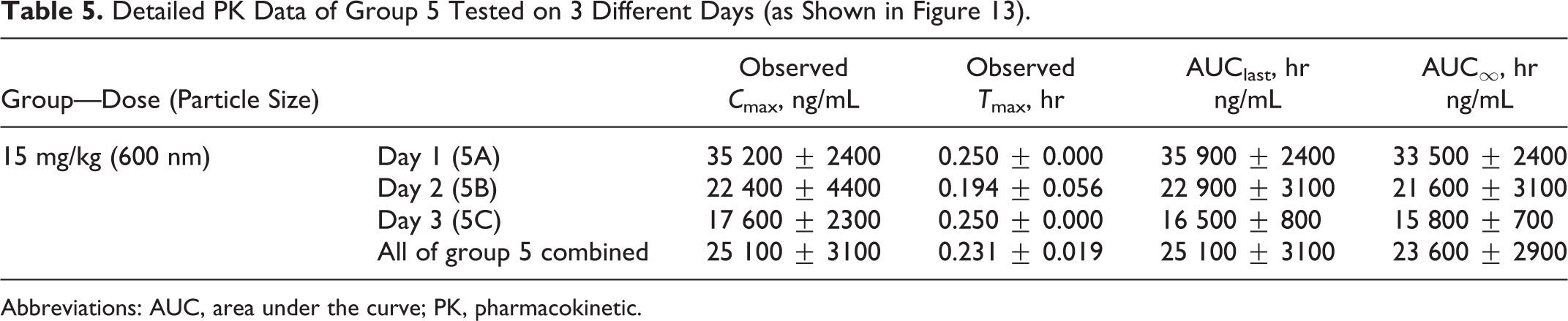
Range-finding preclinical studies were conducted to determine the dose proportionality of multiple combinations of particle sizes and concentrations of MMB4 DMS EF by evaluating PK parameters following a single IM injection in male New Zealand White rabbits. 11 Three dose levels were varied using different combinations of concentrations and particle sizes as follows: 1.25 mg/kg (5 mg/mL at 200, 600, and 6000 nm), 15 mg/kg (60 mg/mL at 200 and 600 nm), and 30 mg/kg (120 mg/mL at 400 and 6000 nm). The goal of the study was to test the effects of particle size and concentration of the micro/nanoparticle suspension formulation of MMB4 DMS on bioavailability and to select the formulation with the most favorable PK properties for subsequent development. Figures 12, 13, and 14 show the plasma MMB4 concentration–time data following IM injections in rabbits at 1.25, 15, and 30 mg/kg, respectively. Table 5 shows the detailed and representative PK data of group 5 animals tested on 3 different days as shown in Figure 13.
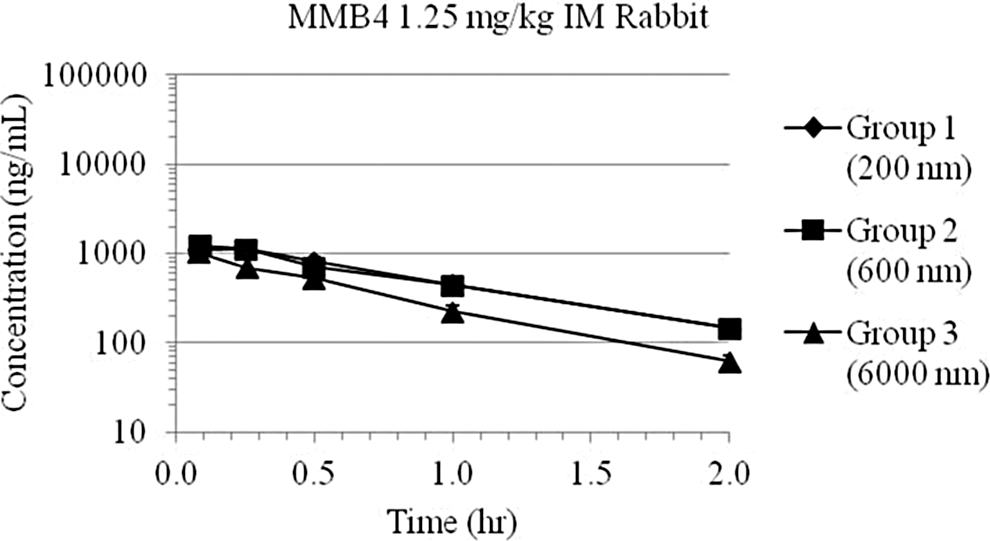
Plasma 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium (MMB4) day 1 concentration–time data following intramuscular (IM) injection of MMB4 dimethanesulfonate (DMS)-enhanced formulation (EF; 1.25 mg/kg) to rabbits, for a 1.25-mg/kg dose delivered at a concentration of 5 mg/mL, using 3 separate nanoparticle formulations (200, 600, and 6000 nm). Groups 1 and 2 data points and trendlines overlap.
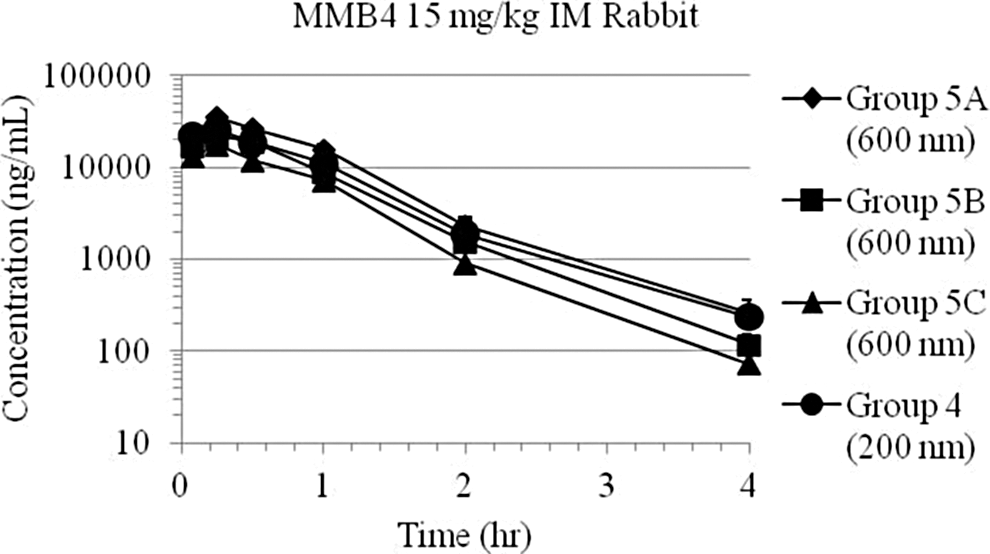
Plasma 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium (MMB4) day 1 concentration–time data following intramuscular (IM) injection of MMB4 dimethanesulfonate (DMS)-enhanced formulation (EF; 15 mg/kg) to rabbits, for a 15-mg/kg dose delivered at a concentration of 60 mg/mL, using one 600 nm nanoparticle formulations and one 200 nm formulation (the 600 nm nanoparticle formulation was also tested with different rabbits on days 2 and 3).
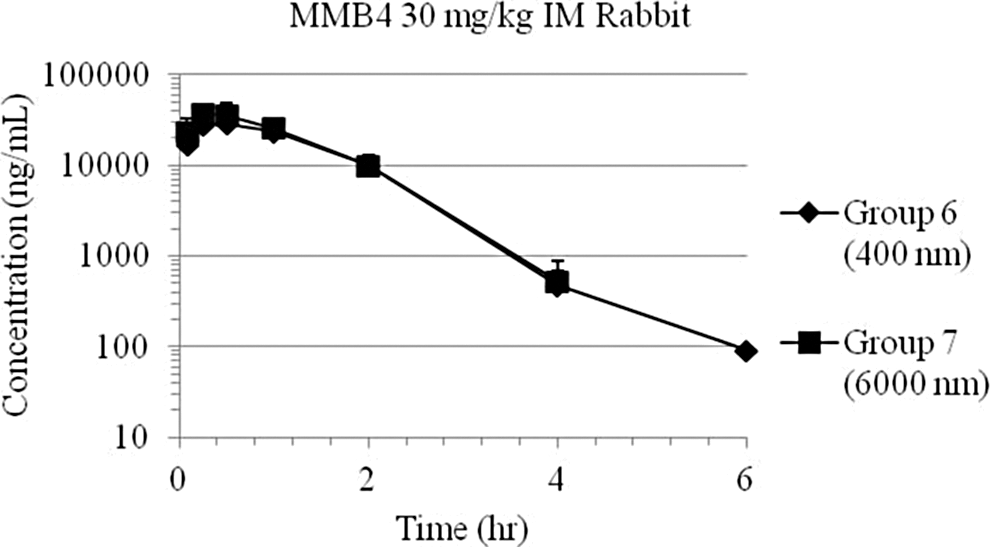
Plasma 1,1′-methylenebis-4-[(hydroxyimino)methyl]pyridinium (MMB4) day 1 concentration–time data following intramuscular (IM) injection of MMB4 dimethanesulfonate (DMS)-enhanced formulation (EF; 30 mg/kg) to rabbits, for a 30-mg/kg dose delivered at a concentration of 120 mg/mL, using 2 separate nanoparticle formulations (400 and 6000 nm). Groups 6 and 7 data points and trendlines overlap.
Detailed PK Data of Group 5 Tested on 3 Different Days (as Shown in Figure 13).
Abbreviations: AUC, area under the curve; PK, pharmacokinetic.
These range-finding preclinical studies showed that the MMB4 DMS EF nanoparticle suspension formulations were bioequivalent across a particle size range of 200 to 6000 nm. The MMB4 DMS EF was found to be bioequivalent to the systemic circulation following a single IM injection across concentrations that ranged from 5 to 120 mg/mL and particle sizes that ranged from 200 to 6000 nm within the in vivo experimental variability. 11
Conclusion
We have demonstrated the development of nanoparticle suspension formulations through a series of millings designed to produce particles in the range of 200 to 6000 nm. Dry milling produced particles down to 3000 nm. This dry-milled material requires compounding with CSO. Wet milling produced particles down to 200 nm. By using CSO as the liquid in the bead (wet) mill, the wet-milled material does not require compounding. In general, bead (wet) milling offers a convenient process for the production of suspensions of drug nanoparticles with ease of scale-up for commercial manufacturing. We consistently produced 200- to 400-nm material. Under a variety of milling conditions, MMB4 remained stable, and the crystal polymorph was conserved. Chemical stability studies for 2 years at temperatures up to 80°C demonstrated superior thermal stability compared to the aqueous formulations.
As expected, smaller particle sizes and higher particle concentrations lead to higher viscosities of the suspension formulations. The precise concentration and particle size combination produced stable, injectable formulations over a range of 5 to 400 mg/mL and particle sizes of 200 to 6000 nm. Preclinical studies demonstrated PK bioequivalence across a particle size range (200-6000 nm). This result indicates that particles manufactured in a relatively large range are absorbed in vivo similarly. Therefore, a wide particle size specification can be set, leading to a robust manufacturing process.
The operationally stable non-Newtonian shear thinning formulation is an exciting technology for not only improving the stability of chemical and biological countermeasures in advanced development but also as a promising application for stabilization of biologics, potentially removing refrigeration requirements for these countermeasures as well.
Footnotes
Acknowledgment
The authors acknowledge the editorial assistance of Dr Vincent Brown.
Authors’ Note
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or reflecting true views of the Department of the Army or the Department of Defense.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the US Army Chemical Biological Medical Systems under SwRI grant number W9113M0810001 and Battelle contract number SP070000D3180DO0599.
