Abstract
To investigate the effects of common nanosuspension-stabilizing excipients on the nature and temporal evolution of histopathological changes at intramuscular (i.m.) administration sites, 5 groups of 39 male rats per group received a single injection of 1 of the 5 analogous crystalline drug nanosuspensions containing 200 mg/ml of an antiviral compound with particle sizes of ±200 nm and identical vehicle compositions, except for the type of nanosuspension stabilizer. The investigated stabilizers were poloxamer 338, poloxamer 407,
Keywords
Long-acting injectable (LAI) or implantable drug delivery systems are able to release a drug in a controlled manner, hence, enabling effective drug plasma concentrations and exposure at the site(s) of pharmacologic activity to be maintained for long periods of time (i.e., up to several months) following a single administration (Burgess and Wright 2012). The steady plasma concentrations that are achieved can benefit the clinical outcome through a variety of principles, including an improved treatment adherence, less frequent disease relapses, a potential lowered risk of therapy resistance development, lower equivalent daily doses and lower peak plasma concentrations and, therefore, potentially reduced frequency and/or severity of treatment-related adverse events. Amid the different formulation concepts, nanotechnology, and especially drug nanocrystals, have become a well-established drug delivery platform, with several approved drug products on the market and many more currently in early and late development for applications in several therapeutic areas (Remenar 2014).
Intramuscular (i.m.) or subcutaneous LAI crystalline drug nanosuspensions are aqueous dispersions of nanosized crystalline particles of poorly water-soluble drugs (Shegokar and Műller 2010). Low concentrations of stabilizing excipients are added to the buffered dispersion vehicle to maintain the physical stability of the drug formulation, preventing irreversible nanoparticle agglomeration and improving the nanosuspension resuspendability prior to injection (Rabinow 2004). Only a limited number of nanosuspension-stabilizing excipients have been approved for parenteral use. These include, among others, sterically stabilizing polymers such as cellulose derivatives (e.g., hydroxypropyl [methyl]cellulose), polyvinyl pyrrolidone, and polyethylene glycols but also nonionic surfactants such as polysorbates (Tween®), poloxamers (i.e., triblock polyoxyethylene–polyoxypropylene–polyoxyethylene copolymers), and other polyethylene oxide-containing surfactants such as
Although parenteral grades of excipients listed by the Food and Drug Administration as “generally recognized as safe” are favored for the use in LAI formulations, the effect of these stabilizers on the host response and local tolerability after i.m. administration cannot always be predicted or extrapolated from existing data (i.e., mostly relying on oral dosing of immediate-release formulations). Therefore, a detailed characterization of the histopathological response to i.m. injections of LAI nanosuspensions containing different stabilizers is essential, not only in the assessment of the tolerability and safety profile of the drug product (including the functional excipients) but also to obtain fundamental insights in the processes contributing to the pharmacokinetics of LAI formulations (Darville, van Heerden, Erkens, et al. 2016). This was exemplified in a series of studies conducted by our group, which demonstrated that the granulomatous inflammatory reaction occurring upon i.m. injection of an LAI nano-/microsuspension in the rat is an important feature in the local disposition of the LAI formulation and a modulator of the i.m. drug release (Darville et al. 2014). The gradual yet extensive infiltration of CD68+ macrophages within the formulation depot formed at the i.m. injection site was accompanied by increasing amounts of LAI formulation being phagocytosed over time (Darville, van Heerden, Erkens, et al. 2016). This process of gradual uptake by macrophages and the intracellular relocation of large portions of the LAI dose meant that the intramacrophagic space became the primary site of drug dissolution and release. The injection site reaction, and macrophages in particular, was found to contribute to the slow drug release, hence, modulating the prolonged pharmacokinetics associated with these LAI drug delivery systems (Darville, van Heerden, Mariën, et al. 2016). Similar conclusions on the contribution of the formation of intracellular drug depots in monocyte-derived macrophages to the (very) long-acting properties of surface-modified antiretroviral drug nanocrystals were recently drawn by another group (Gnanadhas et al. 2017; Edagwa et al. 2017).
The purpose of this study was therefore to investigate the influence that a number of common nanosuspension-stabilizing excipients have on the histopathological and immunological changes at the i.m. injection site and primary draining lymph nodes following a single i.m. administration of LAI nanosuspensions. To this end, a series of analogous LAI crystalline antiviral drug (compound X) nanosuspensions with similar particle size distributions and identical formulation compositions, except for the type of the stabilizing excipient, were prepared. The stabilizers assessed were poloxamer 338, poloxamer 407, polysorbate 80, polysorbate 80 combined with egg phosphatidylglycerol (EPG), and TPGS. The nature, extent, and temporal evolution of the i.m. injection site reaction, following a single i.m. dose of each of the 5 analogous LAI nanosuspensions, were investigated in the rat according to a previously established procedure (Darville, van Heerden, Erkens, et al. 2016). Additionally, plasma samples were collected throughout the 12-week study duration to assess whether possible excipient-mediated differences in the local host response and formulation disposition could be reflected in differences in plasma pharmacokinetics.
Material and Method
Test Articles
The sterile poorly water-soluble, weakly basic, low-molecular-weight crystalline antiviral drug substance (compound X) utilized to produce the test articles was provided by Janssen Pharmaceutica NV (Beerse, Belgium). A series of 5 aqueous nanosuspensions of crystalline drug substance were manufactured aseptically on a low-shear roller mill (Peira Scientific Instruments bvba, Turnhout, Belgium) by milling the micronized drug substance at a concentration of 200 mg/ml in a vehicle composed of sodium hydrogen phosphate monohydrate (parenteral grade), citric acid monohydrate (parenteral grade), sodium hydroxide (ad pH 6.8–7.2), glucose monohydrate (parenteral grade; quantum satis 290 ± 30 mOsm/kg), and 50 mg/ml (1:4 ratio, cf. drug substance) of a nanosuspension-stabilizing excipient in water for injection. The investigated stabilizing excipients were poloxamer 338 parenteral grade (formulation 1 [F1], group 1), poloxamer 407 Ph.Eur. grade (formulation 2 [F2], group 2), polysorbate 80 parenteral grade (formulation 3 [F3], group 3), a combination of polysorbate 80 and 6.25 mg/ml EPG sodium salt parenteral grade (formulation 4 [F4], group 4), and TPGS Ph.Eur. grade (formulation 5 [F5], group 5). In summary, the formulation composition of all 5 test articles was identical, except only for the type of stabilizing excipient. Therefore, no separate vehicle groups were included as part of the study design (see Table 1).
Overview of the Study Design and Dosing Schemes.
The test articles (F1–F5) had similar monomodal particle size distributions as measured by disc centrifugation against a density gradient on a CPS Disc Centrifuge (CPS Instruments Europe, Oosterhout, the Netherlands). The median particle sizes were 246 nm (F1), 254 nm (F2), 266 nm (F3), 268 nm (F4), and 201 nm (F5), and the mean 10th and 90th percentile diameters were 151 ± 23 nm and 382 ± 41 nm, respectively (data not shown).
Animals
A total of 195 young, healthy, and specific pathogen free Sprague-Dawley (Crl:CD® [SD] IGS) male rats, aged 9 to 11 weeks and weighing 280 to 320 g at study initiation, were obtained from Charles River (Sulsfeld, Germany). The animals were assigned to 5 different groups (group 1–5) of 39 rats per group, using a balanced random allocation scheme based on body weight. They were kept in groups of 3 rats/group in individually ventilated polysulfone cages and housed in environmentally controlled rooms with a 12-hr light/12-hr dark cycle, a humidity range of 30% to 70%, and a temperature range of 20 to 24°C. Tap water and certified rodent pelleted maintenance diet (SM R/M-Z from SSNIFF® Spezialdiäten GmbH, Soest, Germany) were available
The study was conducted in accordance with the Belgian (Belgian Royal Decree KB 29.05.2013) and European (European Commission Directive 2010/63/EU) guidelines for the protection of vertebrate animals used for experimental and other scientific purposes.
Experimental Design
Each rat received a single i.m. injection of 60 mg/kg of one of the long-acting nanosuspensions in the hind leg (muscle biceps femoris) using a 27G × ½′′ needle. All but 6 animals per group received 1 injection of 60 mg/kg in the left hind leg, while the last 6 rats from each group were treated in both hind legs (i.e., total of 120 mg/kg). Batches of 3 or 6 animals from each group were then sacrificed at the following 10 time points postinjection; days 0 (2-hr postdose), 1, 3, 7, 14, 21, 28, 42, 56, and 84/85 as shown in Table 1. Three animals per group were sacrificed on days 0, 3, 14, 21, 28, 42, and 56 postdose, while 6 animals per group were sacrificed on days 1, 7, and 84/85 postdose. At the last time point, only 3 animals were examined microscopically as the administration sites from the other 3 animals were collected for pharmacokinetic analysis.
Pathology Procedures
At each scheduled day of necropsy, 3 or 6 animals per group were sacrificed by exsanguination via the carotid artery under isoflurane/oxygen anesthesia and after the last scheduled blood sampling (cf.
Histology, Histopathology, and Immunohistochemistry
All left administration sites that were collected at the 10 sacrificial time points were processed histologically for histopathological evaluation and immunohistochemistry analysis, except for 3 of the 6 animals killed on day 84/85. Lymph nodes for histopathology were collected from all animals sacrificed on the following time points postdose: day 0, 3, 14, 28, 56, and 84. Therefore, histopathology and immunohistochemistry examination of administration sites were carried out on 36 of the 39 rats per group, while histopathology evaluation of lymph nodes was carried out on 21 of the 39 rats per group. After fixation in NBF (for a minimum period of 48 hr and a maximum of 7 days), tissues were embedded in paraffin wax, sectioned to a 3–4 µm thickness, and stained with hematoxylin and eosin. Additionally, paraffin sections of the administration sites were stained immunohistochemically with CD68, CD31, CD3, and CD79b markers for macrophages, vascular endothelium, T cells, and B cells, respectively. All microscopic slides were examined histopathologically by a board-certified pathologist and the findings entered directly into a computerized database (Ascentos, Pathology Data Systems Limited, Pratteln, Basel, Switzerland).
Qualitative and Semiquantitative Histopathological Evaluation of Administration Sites
Histology slides of the administration sites were subjected to qualitative and semiquantitative evaluation that focused on the following: identification of the location of the formulation depot, a semiquantitative assessment of the overall inflammatory response and its various components, and a semiquantitative assessment of the extent of macrophage infiltration of the formulation depot.
The localization of the formulation depots within the muscle was broadly classified into the following 3 categories: deep i.m. (i.e., truly i.m., formulation present between muscle fibers or in endomysium), part endomysium/part perimysium (partially i.m., i.e., formulation present between muscle fibers or endomysium and in loose interstitium [peri- or epimysium]), and interfascial (i.e., exclusively present in larger peri- or epimysial interstitium).
The semiquantitative assessment of the inflammatory process at the administration site included: grading of the overall inflammatory response; diagnosis and grading of the type and extent of the local tissue reaction and/or damage, such as myofiber degeneration/necrosis or myofiber regeneration; grading of edema, hemorrhage, angiogenesis, fibrosis/fibroplasia, or formation of granulation tissue; and diagnosis and grading of the cellular infiltrates, mainly granulocytes, macrophages, multinucleated giant cells (MNGC), and T or B lymphocytes.
The grading of the overall inflammatory response was largely based on the severity of the various key components of the acute or “active” inflammatory response, such as hemorrhage and edema, granulocytic infiltrates, fibrin, extensive granulation tissue or fibroplasia, and tissue damage, present at the administration site.
The histopathology findings were graded on a scale of 0 to 5 represented by, not present, minimal, mild, moderate, marked, and severe, respectively. Mean scores (
Pharmacokinetics
Blood samples for drug plasma concentration determination were obtained at scheduled preterminal time points from the 6 animals per group that were sacrificed on the last day of the study (
Results
General Progression of Administration Site Findings across All Groups
The intergroup time course comparisons of the histopathological progression of each of the main components of the inflammatory response at the administration site treated with formulations containing 5 different stabilizing excipients are shown in Figure 1a–h. The data show that although the general trend of the progression of inflammatory changes at the administration sites was more or less similar across the groups with different excipients, there were some considerable differences between the groups at each time point in the following parameters: the exact timing of the appearance of inflammatory cell subtypes, the severity grades of the overall inflammatory change, and the nature of the inflammatory response.
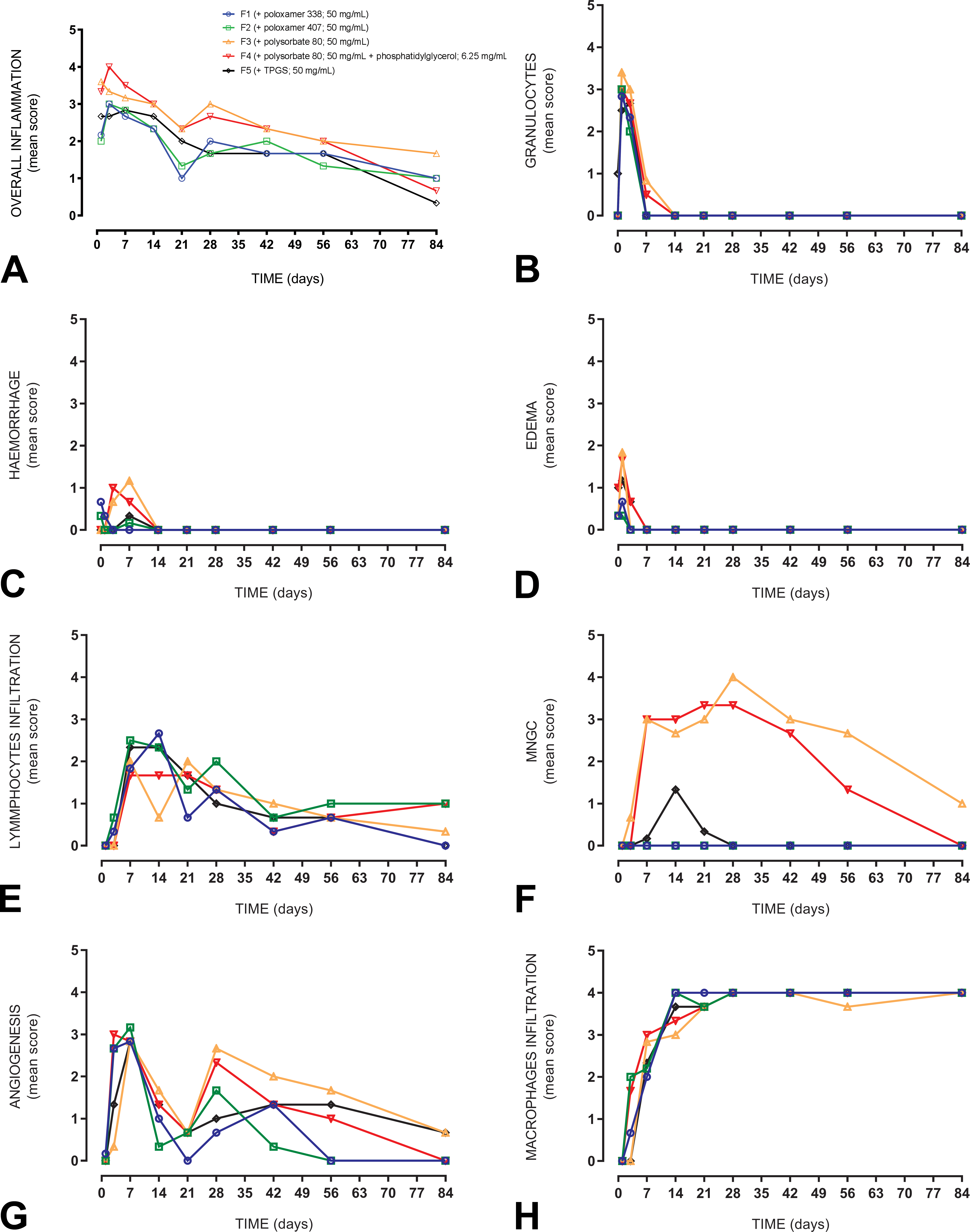
(a–h) A comparison of the temporal evolution of the main components of the inflammatory response at the administration sites, following a single intramuscular injection of formulations containing 5 different stabilizing excipients, in the rat. Criteria for scoring are provided in the text. All parameters are expressed as mean scores on a semiquantitative scale (0–5) +
The general temporal evolution of histopathological findings at administration sites across all 5 groups, from acute to chronic inflammatory changes (Figure 2a–h), was relatively similar to those previously described for long-acting i.m. drug nanosuspensions (Darville, van Heerden, Vynckier, et al. 2014; Darville, van Heerden, Erkens, et al. 2016). Findings progressed over the 10 sacrificial time points from minimal edema and/or hemorrhage at 2-hr postdose to mainly granulocytic infiltrates between days 1 and 3 postdose, angiogenesis and CD68+ macrophage/histiocytic infiltrates, including multinucleate giant cells (MNGCs), from day 3 postdose; CD3 positive lymphocytic infiltrates, from day 3 or 7 postdose; and peaking of the inflammatory response between days 7 and 14 postdose, leading to a gradual reduction in the inflammatory response from day 14 postdose onward. The resolution of the active inflammatory response between days 14 and 21 was associated with a progressive loss of CD68 immunostaining of macrophages and the appearance of a few scattered CD79+ lymphocytes. Complete infiltration of the depot formulation by macrophages was observed as early as day 14 and present in all groups by day 28 postdose. Days 56 to 84/85 postdose were characterized by a further resolution of the inflammatory reaction to just a few residual solid nodules of eosinophilic staining and weakly CD68 positive macrophages with fewer lymphocytes that were both CD3 and CD79 positive at the administration sites.
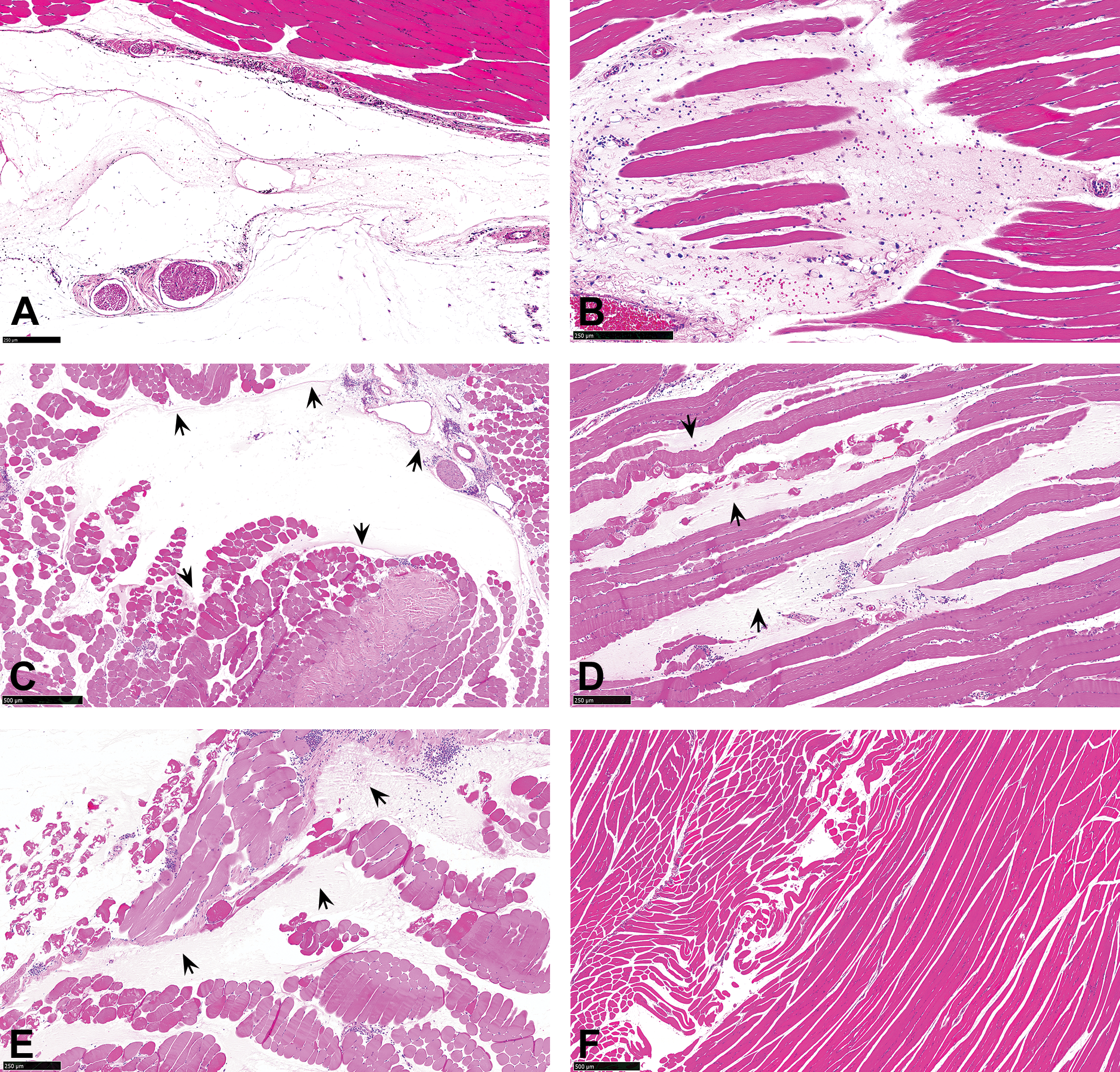
(a–f) Administration sites from animals sacrificed at 2 hr postdose, showing test material (arrows) deposited in the fascia (a) and intramuscularly (b–e), with minimal edema and hemorrhage, myofiber degeneration (c–e), and needle track damage (f). Hematoxylin and eosin.
Despite these general similarities in the temporal progression of the administration site findings, there were some considerable intergroup differences observed at each time point; and they were mainly between groups treated with formulations containing polysorbate 80 (groups 3 and 4), on one hand, and groups injected with formulations containing poloxamer 338, poloxamer 407, and TPGS (groups 1, 2, and 5, respectively), on the other hand. The main differences were in the time of the appearance or disappearance of inflammatory cell subtypes, such as granulocytes or lymphocyte subsets, the severity grades of the overall inflammatory response, and the nature of the inflammatory response, including the presence or absence of MNGCs.
Administration Site Findings after 2 hr and at Day 1 Postdose
Administration sites from all animals showed variable amounts of deposited test material (depot), with minimal edema and/or hemorrhage (Figure 2a–e), minimal myofiber degeneration/necrosis, mostly caused by the needle track (Figure 2d–f), and very little to no evidence of inflammation (overall inflammatory grade 0). The depot location was recorded as deep i.m. (Figure 2b, d, and e), part endomysium/part perimysium (Figure 2c), or interfascia/perimysium (Figure 2a). There were no differences in the depot location, severity, or nature of findings across the groups.
In all animals, administration sites from day 1 postdose were characterized by mild to marked granulocyte infiltrates, minimal to marked myofiber degeneration/necrosis, minimal to mild edema and/or hemorrhage, and up to marked grades of overall inflammation (Figure 3a–f). The polymorphonuclear infiltrates formed a distinct rim around the formulation depot (Figure 3a and b) as well as characteristic streams or tracks of granulocytic cellular infiltrates that projected into the depot (Figure 3b inset, e, and f).
(a–f) Administration sites from animals sacrificed on day 1 postdose, showing test material deposited in the interfascial (a) and deep intramuscular (c–e) space, with minimal to moderate grades of acute inflammation and the characteristic granulocytic tracks (arrows) and minimal to moderate myofiber degeneration/necrosis (c). Figures d and e show administration sites with minimal to no evidence of myofiber degeneration. Hematoxylin and eosin.
On average, higher severity grades of overall inflammation were present in animals treated with formulations containing polysorbate 80 (formulation 3, group 3) and the combination of polysorbate 80 and EPG (formulation 4, group 4; Figure 3c), when compared to those injected with formulations containing poloxamer 338, poloxamer 407, and TPGS (groups 1, 2, and 5, respectively; Figure 3d), while higher severity grades of myofiber degeneration/necrosis were present in groups treated with formulations containing polysorbate 80 (groups 3 and 4) and TPGS (group 5).
Administration Site Findings Day 3 Postdose
In all groups, administration sites were characterized by the appearance of a rim of CD68 positive macrophages around the formulation depot (Figure 4a–e), which also formed distinct infiltrating streams or radial migration tracks into the depot, intermixed with the streaming granulocyte infiltrates (Figure 4a and b insets). In addition, there were mild to marked CD68+ macrophage accumulations within the vicinity of the administration site (Figure 4f), minimal to marked myofiber degeneration and/or regeneration (Figure 4a and b), minimal to moderate angiogenesis and/or fibrosis/fibroplasia (Figure 4a and b), and moderate grades of overall inflammation (Figure 4a). The depot-infiltrating and locally accumulating macrophages were enlarged due to the extensive phagocytosis of the formulation material.
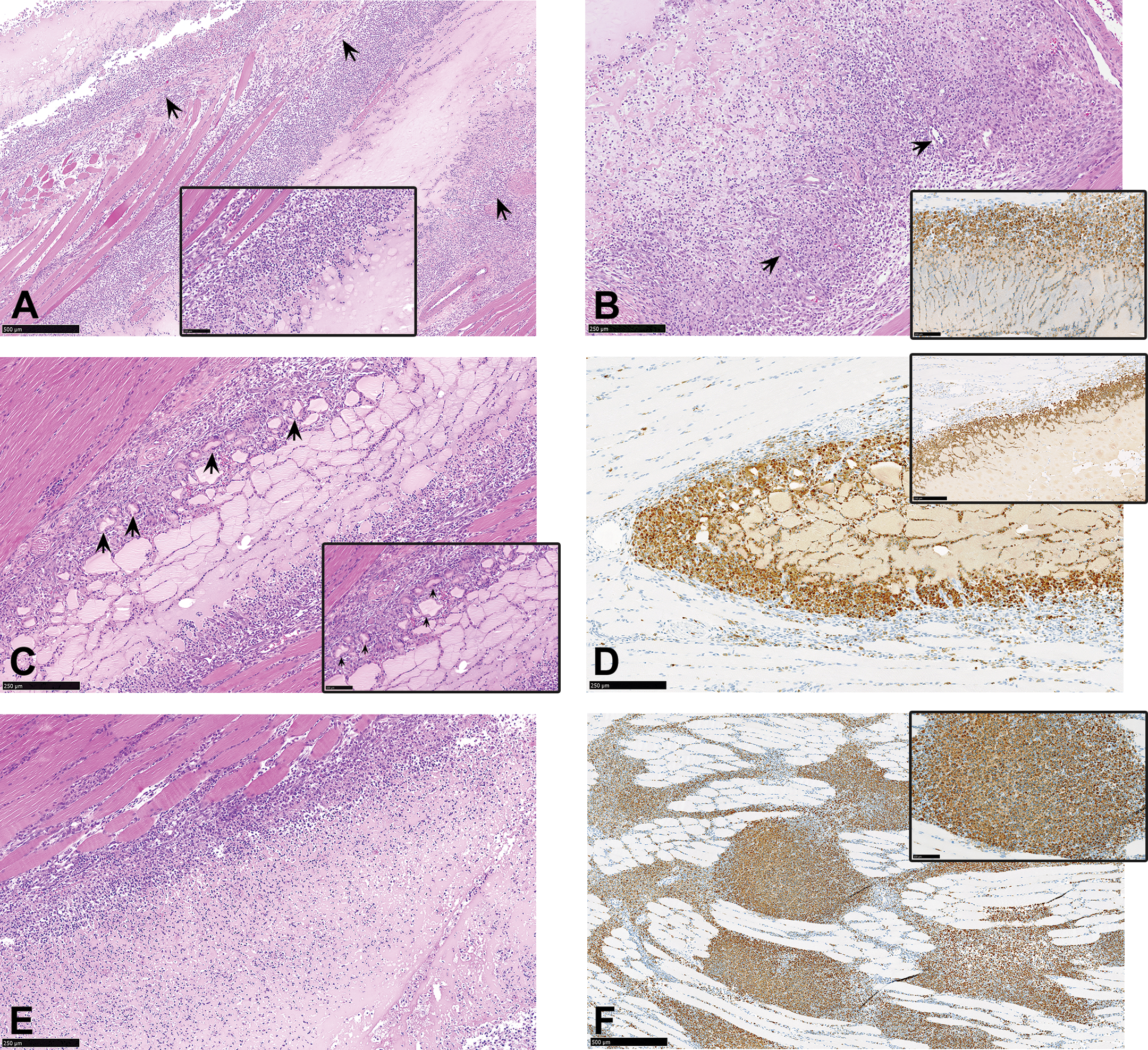
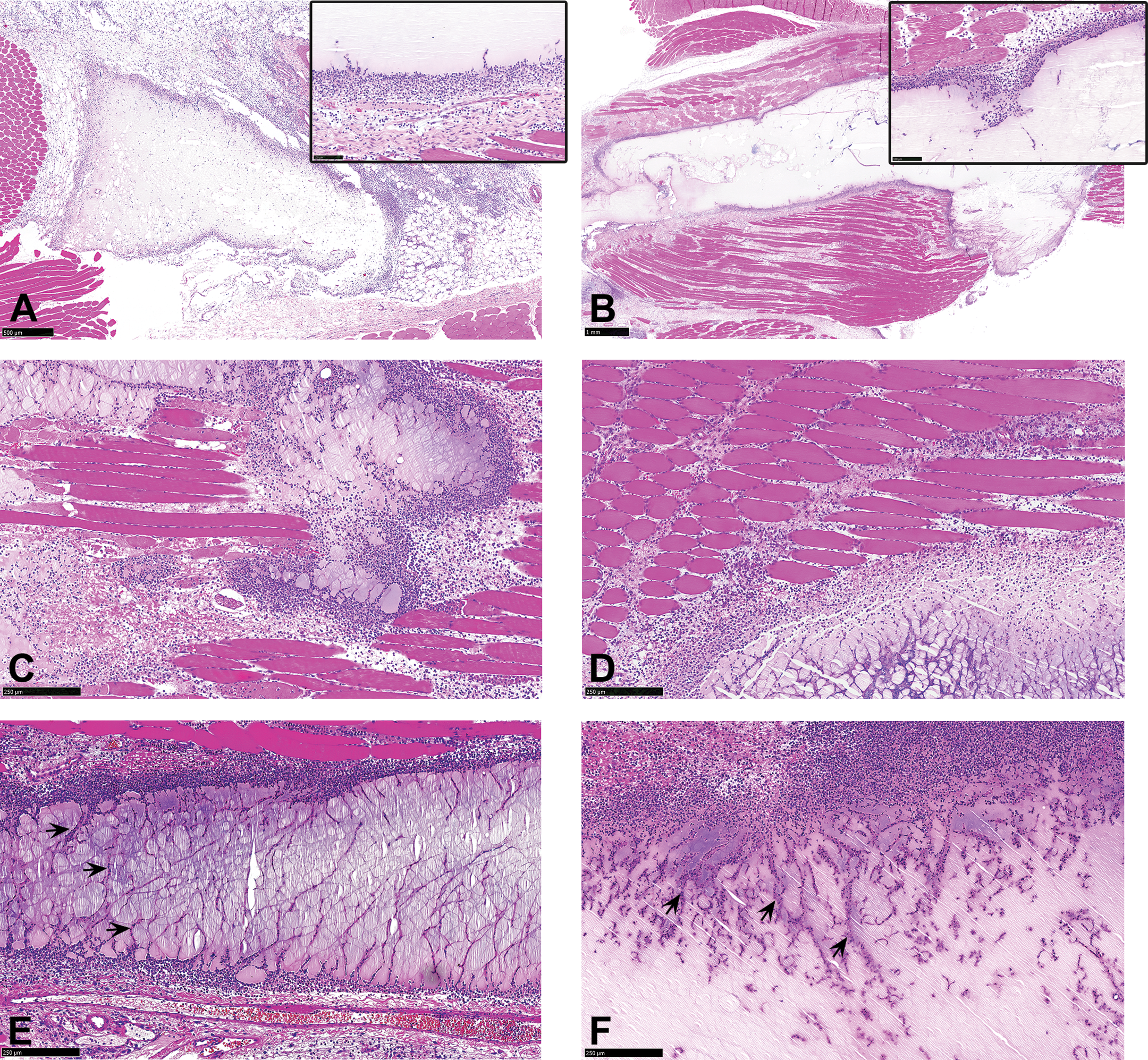
(a–f) Administration sites from animals sacrificed on day 3 postdose. (a) Administration site from animal treated with a formulation containing polysorbate 80, showing marked inflammation with macrophage infiltration, minimal myofiber degeneration, and fibroplasia (arrows). Hematoxylin and eosin. (a, inset) Higher magnification of figure a showing streams or tracks of depo-infiltrating macrophages. Hematoxylin and eosin. (b) Administration site from animal treated with a formulation containing polysorbate 80 showing rim of infiltrating macrophages, angiogenesis (arrow), fibroplasia, and minimal myofiber regeneration. Hematoxylin and eosin. (b, inset) Immunohistochemistry staining of figure b showing positive CD68 staining of depot-infiltrating macrophages. (c) Macrophage agglomeration and fusion (arrows) to form multinucleate giant cells at the administration site of an animal treated with a formulation containing polysorbate 80. Hematoxylin and eosin. (c, inset) Higher magnification of figure c. Hematoxylin and eosin. (d) Immunohistochemistry staining of figure c, showing positive CD68 staining of macrophage agglomerates. (e) Administration site from animal treated with a formulation containing poloxamer 338 showing reduced inflammation and myofiber degeneration in comparison to a. Hematoxylin and eosin. (f and inset) Positive CD68 immunohistochemistry staining of macrophages accumulating between muscle fibers within the vicinity of the administration site.
Intergroup differences were again observed between the 2 sets of administration sites: those from animals injected with formulations containing poloxamer 338, poloxamer 407, and TPGS (groups 1, 2, and 5, respectively) and those injected with formulations containing polysorbate 80 (groups 3 and 4). Higher severity grades of the overall inflammatory response and fibrosis/fibroplasia were observed in groups injected with formulations containing polysorbate 80 (groups 3 and 4; Figure 4a and c), when compared to those from animals that were injected with formulations containing stabilizing excipients other than polysorbate 80 (groups 1, 2, and 5; Figure 4e), while a minimal increase in the severity of myofiber degeneration and regeneration and granulocytic infiltration was observed in the groups injected with formulations containing polysorbate 80 (group 3 and 4) and TPGS (group 5). However, in contrast to the above changes (associated with acute inflammation), the initial appearance of CD3 positive lymphocytic infiltrates occurred only in animals injected with formulations containing poloxamer 338 or poloxamer 407 (groups 1 and 2).
Interestingly, one administration site from the polysorbate 80 group (group 3) presented with the initial stages of MNGC formation (Figure 4c), which were characterized by the agglomeration of macrophages into several single- or double-lined macrophage rings that completely surrounded the formulation depot to form follicular-like structures that later fused into typical foreign body MNGCs (Figure 4c inset).
Administration Site Findings at Day 7 Postdose
Across all groups, administration sites were generally characterized by a substantial (up to moderate) infiltration of CD3 positive lymphocytes (Figure 5a and b), an increased amount and thickness of radiating and depot-infiltrating and enlarged CD 68+ macrophages (up to moderately severe; Figure 5a and c), more prominent angiogenesis (Figure 5c inset), myofiber degeneration/necrosis, myofiber regeneration and fibrosis/fibroplasia, and increased accumulation of enlarged CD68+ macrophages in the outer rim of the administration site (Figure 5c). This time point therefore marked the start of a gradual resolution of the acute or “active phase” of the inflammatory response, typified by the reduction in (viable) granulocytic infiltrates, granulocytic apoptosis (Figure 5a), decreased severity grades and incidence of edema and hemorrhage, and the increased severity and incidences of cellular infiltrates and processes associated with the reparative/healing phase of the inflammatory response (increased macrophages, lymphocytes, fibroplasia, and angiogenesis).
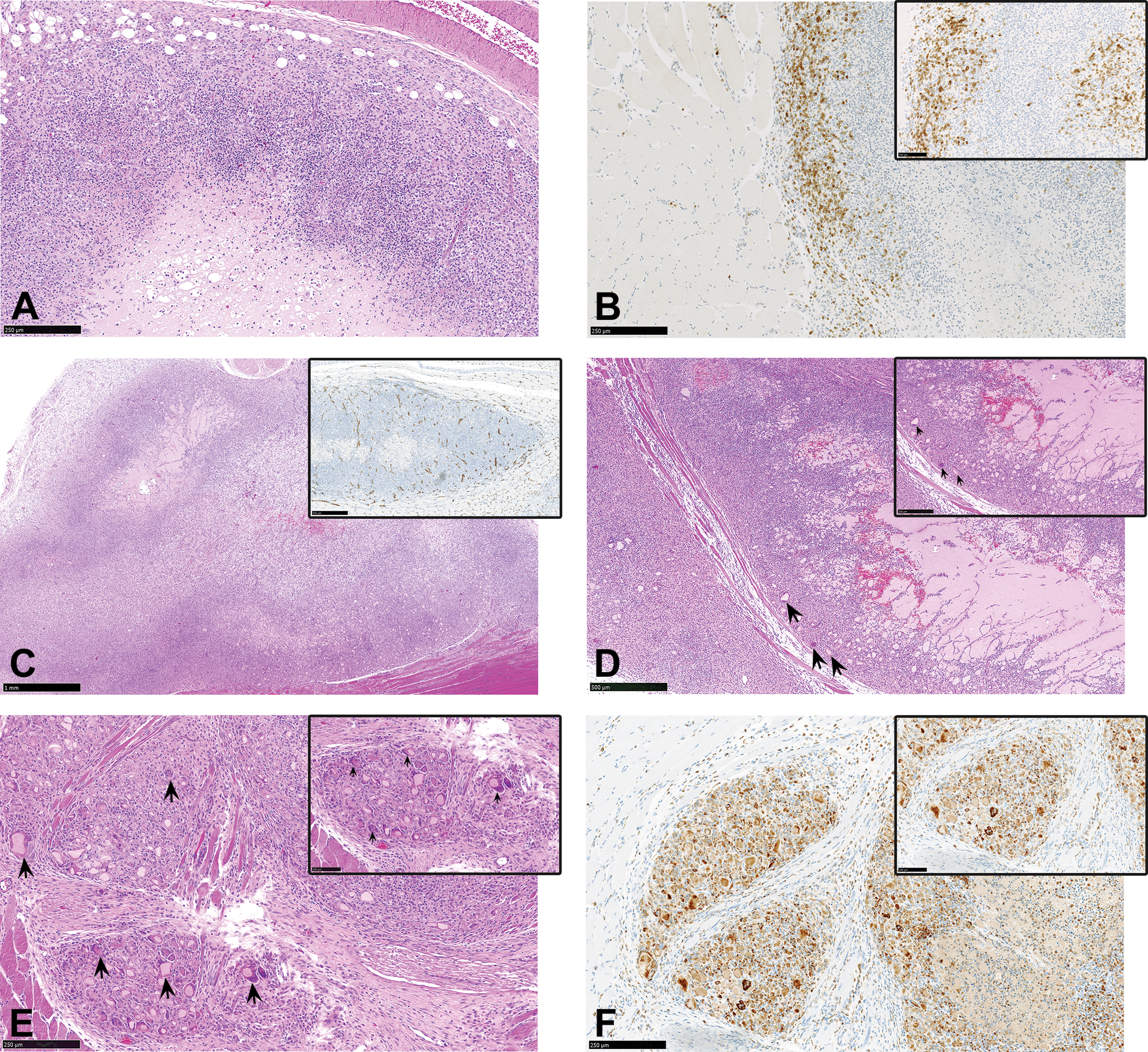
(a–f) Administration sites from animals sacrificed on day 7 postdose, showing (a) an increase in the amount and thickness of radiating and depot-infiltrating macrophages, granulocytic apoptosis, more prominent angiogenesis, and lymphocytic accumulation in the periphery of the lesion. Hematoxylin and eosin. (b and inset) Positive CD3 immunohistochemistry staining of T lymphocytes in the periphery of the lesion. (c) Chronic or healing phase of the inflammatory response characterized by increased macrophages, lymphocytes, fibroplasia, and angiogenesis. Hematoxylin and eosin. (c, inset). CD31 immunohistochemistry staining to demonstrate prominent angiogenesis. (d and inset) Animal treated with a formulation containing polysorbate 80 showing some pockets of “active” inflammation (hemorrhage), multinucleate giant cells (MNGCs; arrows), and reduced depot infiltration. Hematoxylin and eosin. (e and inset) MNGCs (arrows) at the administration site of an animal treated with a formulation containing polysorbate 80. Hematoxylin and eosin. (f and inset) Positive CD68 immunohistochemistry staining of MNGCs in figure e.
Slight differences were present between administration sites injected with formulations containing poloxamer 338, poloxamer 407, and TPGS (groups 1, 2, and 5, respectively), when compared to those that received formulations containing polysorbate 80 (groups 3 and 4). Administration sites from groups that received formulations containing polysorbate 80 (groups 3 and 4) were characterized by mild to moderate grades of MNGCs (at 100% incidence; Figure 5d–f), higher severity grades of granulocytic infiltrates, and slightly increased severity grades of overall inflammation (Figure 5d). The severity grades of lymphocytic infiltrates were, however, higher in the groups that were injected with formulations containing poloxamer 338, poloxamer 407, and TPGS (groups 1, 2, and 5), mirroring the trend observed on day 3 postdose. The CD3-expressing T lymphocytes were found either scattered throughout the inflammatory zone or distributed peripheral to the macrophage aggregates (Figure 5b), where they tended to form small distinct lymphoid follicle-like nodules (mostly perivascular). No substantial amounts of B lymphocytes were detected at day 7 postdose.
Interestingly, although the presence of numerous MNGCs (of up to moderate severity grades) was a key feature of the macrophage response at all administration sites treated with formulations containing polysorbate 80 (groups 3 and 4; Figure 5d) from day 7 postdose onward, minimal grades of MNGCs were also observed in a single animal treated with formulations containing the TPGS stabilizing excipient (group 5) on day 7.
Administration Site Findings at Day 14 Postdose
Compared to day 7 postdose, inflammatory reactions at the administrations sites of animals sacrificed on day 14 postdose generally showed the following differences: a moderate increase in the severity of CD3 positive lymphocytic infiltrates, with increased formation of lymphoid follicle-like nodules that were mostly distributed perivascularly (Figure 6a); the appearance of scattered CD79 positive lymphocyte infiltrates (Figure 6b); and a mild to marked increase in the macrophage infiltration of the formulation depot that achieved complete (100%) or almost complete depot infiltration (Figure 6c). At day 14, peripherally accumulating macrophages assumed a more eosinophilic cytoplasmic staining (Figure 6a and inset) and a reduction in the amount of cytoplasm, which were associated with a decrease in the intensity of the CD68 immunostaining (Figure 6d). In contrast, macrophages closer to or infiltrating the depot still showed phagocytic enlargement and a more intense CD 68 immunostaining. There was, in addition, a general complete absence of findings associated with the acute or active phase of the inflammatory response, such as viable granulocytic infiltrates, edema and/or hemorrhage, and myofiber degeneration/necrosis on day 14.
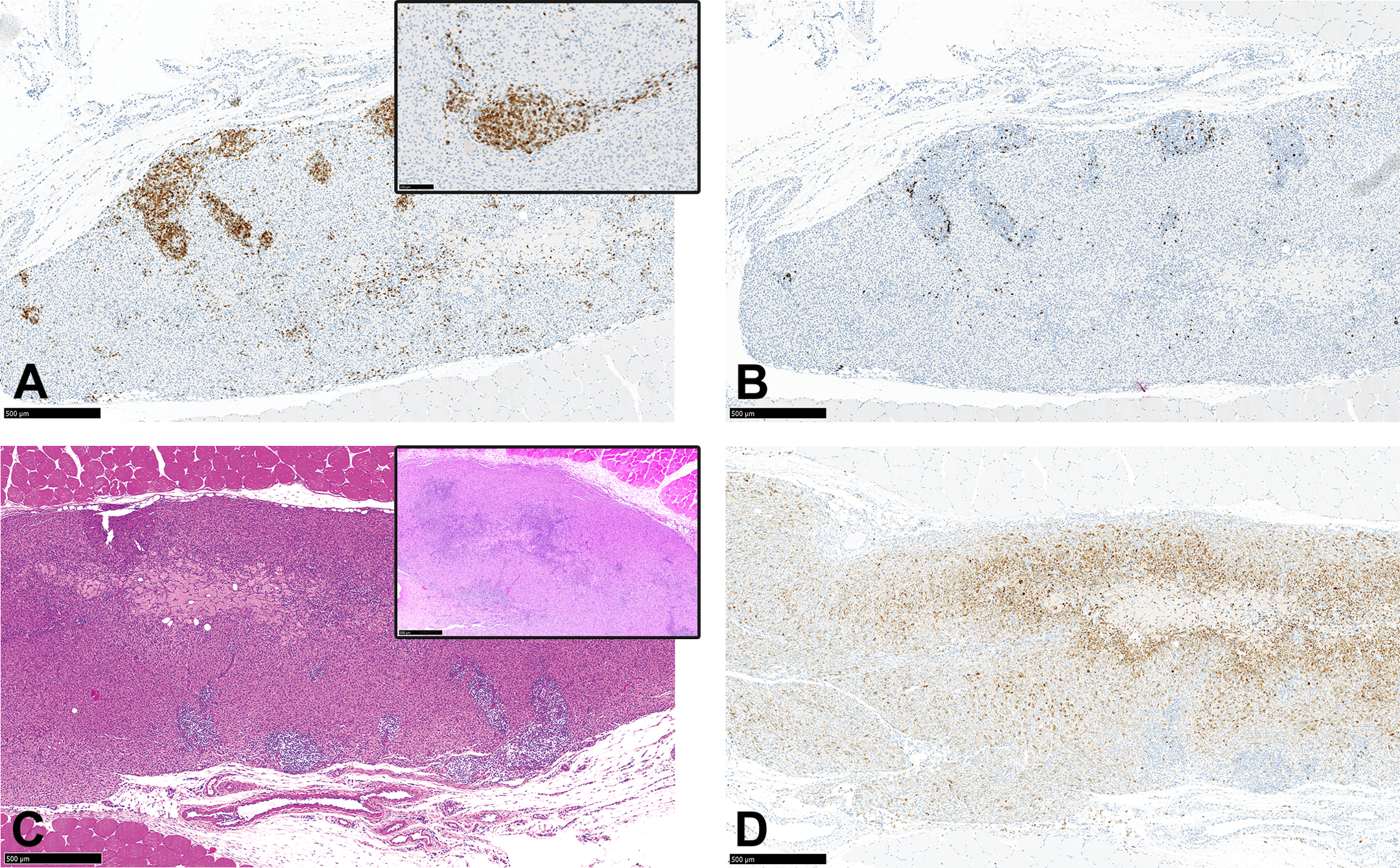
(a–d) Administration site from an animal sacrificed on day 14 postdose showing (a and inset) positive CD3 immunohistochemistry staining of T lymphocytes demonstrating a moderate increase in the severity of lymphocytic infiltrates, with increased formation of lymphoid follicle-like nodules. (b) Positive CD79 immunohistochemistry staining demonstrating scattered B lymphocytes. (c and inset) Marked increase in the extent of macrophage infiltration of the formulation depot to almost complete depot infiltration. (c, inset) Macrophages are smaller and assume a more eosinophilic cytoplasmic staining. Hematoxylin and eosin. (d) CD68 immunohistochemistry staining of macrophages showing a reduction in the staining intensity particularly in the peripheral macrophages.
Administration sites on day 14 showed more intergroup similarities than at earlier time points, with minimal differences, mainly between administration sites injected with nanosuspensions containing poloxamer 338 and poloxamer 407 (groups 1 and 2) and those from animals dosed with polysorbate 80–containing nanosuspensions (groups 3 and 4). The differences between the 2 broad groups include the presence of MNGCs, residual myofiber regeneration, and a slight increase in the severity of the overall inflammation in the groups injected with formulations containing polysorbate 80 (groups 3 and 4). At this time point, administration sites treated with formulations containing TPGS (group 5) showed some similarities with polysorbate groups, such as myofiber regeneration and MNGCs, but at a lower incidence and severity than in the polysorbate groups (groups 3 and 4). Analysis of the exact deposition site of the formulation material on day 14 postdose (data not shown) revealed more deep i.m. deposition in the polysorbate 80 and TPGS groups (when compared to the poloxamer 338 and 407 groups), which was considered to account for the slight increase in the incidence of myofiber regeneration in these groups.
Administration Site Findings at Days 21, 28, and 42 Postdose
Administration sites of animals sacrificed on day 21 onward showed a marked reduction in the severity grades of the active inflammatory response and myofiber lesions, but very little change in the severity of lymphocytic infiltrates, which showed an increased tendency to form more lymphoid follicle-like nodules.
In addition, there was a marked increase in the infiltration of the formulation depot by macrophages, with some administration sites showing evidence of complete macrophage infiltration of the depot, so that on most sections, only solid nodules of more eosinophilic stained macrophages without a visible depot were present (Figure 7a). Macrophages at the injection sites showed further progressive loss of CD68 reactivity between days 21 and 42 (Figure 7b) but less so in others (Figure 7c).
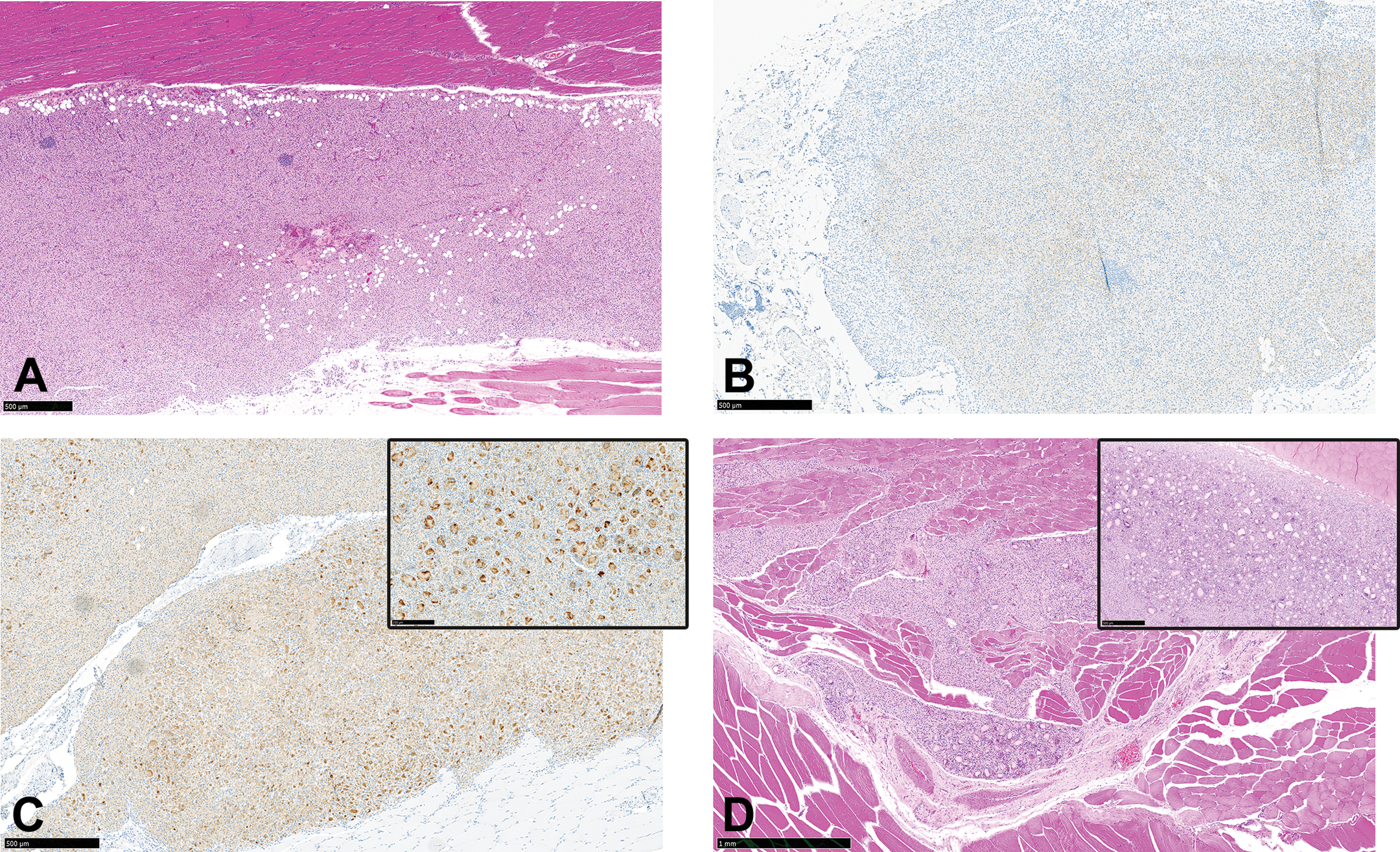
(a–d) Administration sites from animals sacrificed between days 21 and 56 postdose, showing (a) marked reduction of the inflammatory lesions and complete depot infiltration (no visible depot material) on day 56 postdose, with only solid nodules of more eosinophilic-stained macrophages present. There is increased adipocyte infiltration and smaller lymphoid nodules. Hematoxylin and eosin. (b) A diffuse loss of CD68 immunohistochemistry staining of macrophages on day 42 postdose in an animal injected with a formulation containing poloxamer 338. (c) A slightly less “loss” of CD68 immunohistochemistry staining of macrophages on day 42 postdose in an animal injected with a formulation containing polysorbate 80. (c, inset) A higher magnification of figure c showing that CD68 staining is mostly concentrated within areas of multinucleate cells giant cells (MNGCs). (d) Macrophage agglomerates and MNGCs in an animal injected with a formulation containing polysorbate 80 and sacrificed on day 42. (d, inset) Some depot material is still present within the macrophage agglomerates.
Compared to those from animals injected with formulations containing poloxamer 338, poloxamer 407, and TPGS (groups 1, 2, and 5), administration sites from groups 3 and 4 (formulations containing polysorbate 80) showed a slightly lower reduction in overall inflammation by day 42 postdose (therefore showed higher severity grades of inflammation), and a higher number of injection sites with ongoing macrophage infiltration of the depot, indicating a slower or more prolonged macrophage infiltration of the formulation depot (Figure 7d). There was also a slight increase in the severity of MNGC in the groups injected with formulations containing polysorbate 80 (groups 3 and 4) and sacrificed on days 21–42 postdose, when compared to earlier time points, and the areas with MNGC consistently retained strong CD68 positive staining (Figure 7d and inset). Therefore, administration sites from groups 3 and 4 showed a slight persistence of the macrophage response up to day 42 and generally took longer time to completely infiltrate the depot than groups 1, 2, and 5.
Across all groups, the moderate drop in the severity grades of overall inflammation and angiogenesis that occurred on day 21 postdose (Figure 1a and g) was unexpectedly followed by a mild increase in the inflammatory response (and angiogenesis), back to day 14 postdose levels, which then showed a more gradual decrease from day 28 onward. The reason for this observed trend could not be explained.
Administration Site Findings at Days 56 and 85 Postdose
Administration sites were characterized by a profound reduction in the size and severity of the inflammatory lesion, which now consisted of bland, compact nodules of smaller, intensely eosinophilic, and weakly CD68 positive macrophages, and fewer lymphocytic infiltrates that formed smaller follicle-like structures, and stained positive for both CD3 and CD79. The solid histiocytic infiltrates from day 56 onward were considered to represent a complete invasion/infiltration of the formulation depot by macrophages, and these bland granulomatous nodules were often infiltrated by adipose tissue.
The severity grades of the overall inflammation were still higher in the groups treated with the formulations containing polysorbate 80 (groups 3 and 4) than in those injected with formulation containing poloxamer 338, poloxamer 407, and TPGS (groups 1, 2, and 5) due to the persistence of MNGCs and their increased CD68 staining intensity.
Lymph Node Weights and Histopathology
A transient increase in wet weights of the draining lymph nodes (popliteal and medial iliac), normalized for body weights, was recorded in all groups with no differences between the groups (Figure 8a–d). However, the increase in lymph node weights was more pronounced in the medial ILN when compared to the PLN, and, in general, the organ weight increase was more evident in the left (i.e., ipsilateral side in comparison to the i.m. site of administration of the nanosuspensions) lymph nodes as opposed to the right ones.
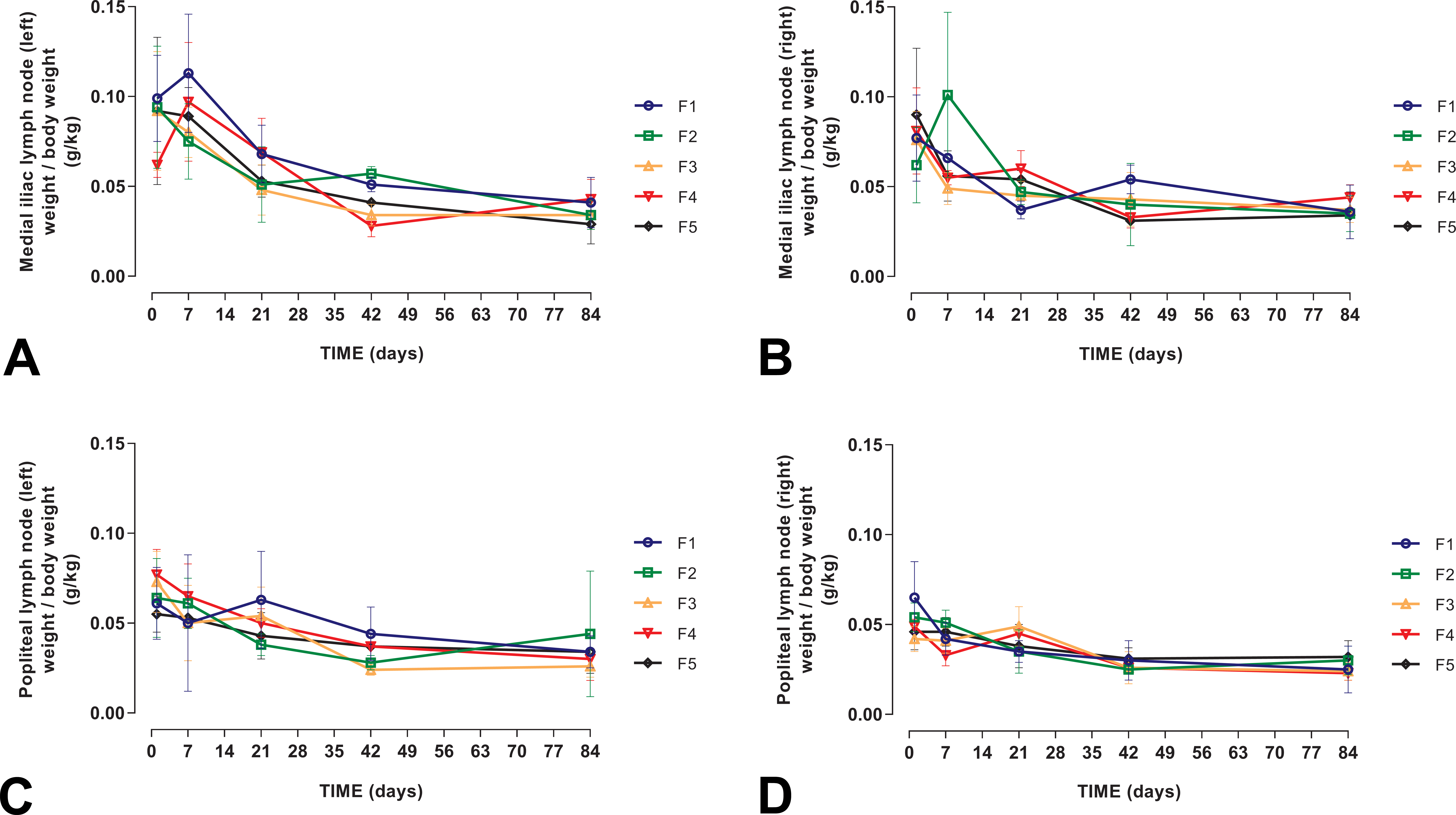
(a–d) A comparison of the temporal change in the body weight–normalized wet weights of the draining lymph nodes, left medial iliac (a), right medial iliac (b), left popliteal (c), and right popliteal (d), following a single intramuscular injection of formulations containing 5 different stabilizing excipients in the rat.
The main findings observed in the local lymph nodes (popliteal and medial iliac) were histiocytic infiltrates in the medullary sinus and increased cellularity (Figure 9). The findings were present from day 1 postdose and showed a general increase up to day 28, from which there was a gradual decrease in the incidence and severity of the findings as lymph nodes returned to normal. There was a slight increase in the total incidences of histiocytic infiltrates (in all lymph nodes) in groups treated with the formulations containing polysorbate 80 (groups 3 and 4) when compared to those injected with formulation containing poloxamer 338, poloxamer 407, and TPGS (groups 1, 2, and 5; data not shown). There was no difference in the incidence or severity of increased cellularity among the groups.
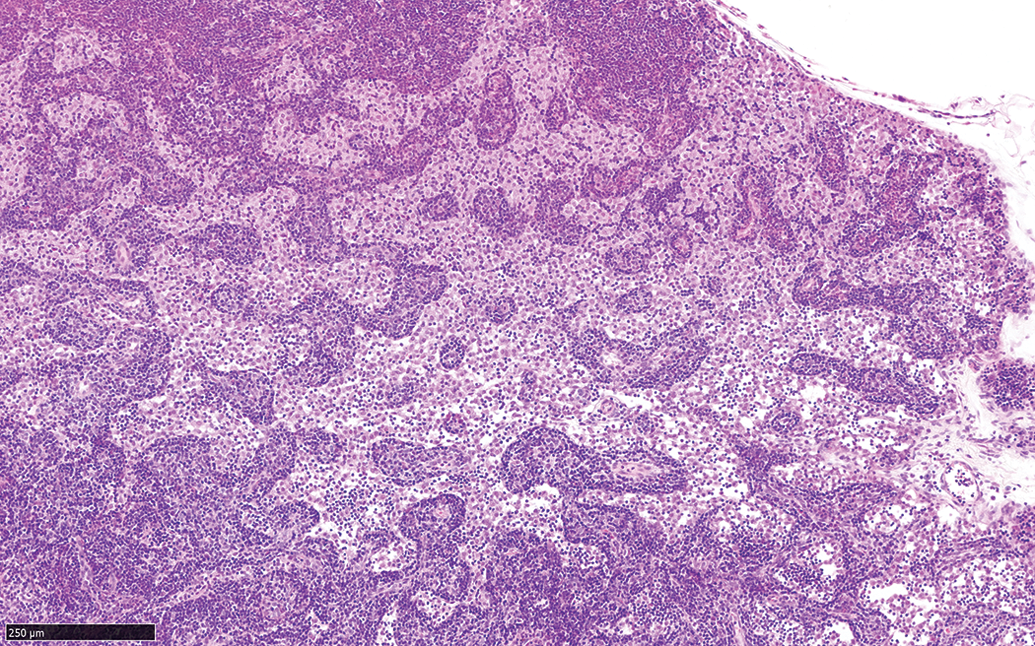
The left medial iliac lymph node from an animal treated with a formulation containing polysorbate 80 and sacrificed on day 3 postdose, showing histiocytic infiltrates in the medullary sinus and increased cellularity. Hematoxylin and eosin.
Plasma Pharmacokinetics after i.m. Administration in the Rat
Following single i.m. administrations of the 5 compound X nanosuspensions with similar quantitative compositions, but containing different stabilizing excipients, sustained compound X plasma concentrations above the lower limit of quantification (1 ng/ml) were observed in all animals of all groups for the duration of the study (85 days for groups 1 and 2, and 84 days for groups 3–5). In general, mean compound X plasma concentration versus time profiles were remarkably similar from day 14 onward for all groups. Most differences between the concentration versus time profiles in groups dosed with the different variants of compound X nanosuspensions were observed during the first 7 to 14 days after i.m. dosing (Figure 10). From day 14 onward, drug plasma concentrations converged in all groups, although slightly faster terminal decline was noticed in animals given formulations containing poloxamer 338 and poloxamer 407 (groups 1 and 2).
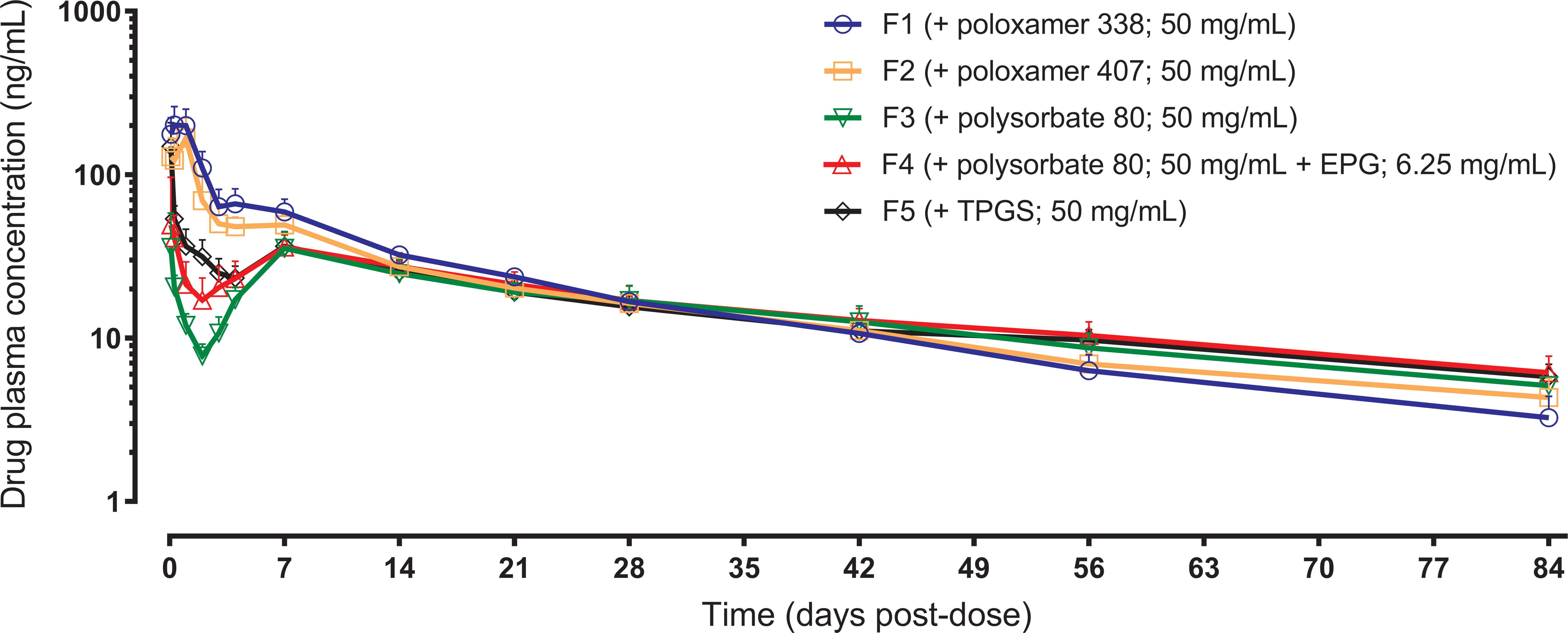
Mean plasma concentration versus time profiles in groups dosed with formulations containing 5 different stabilizing excipients in the rat. Differences were observed during the first 7 to 14 days after intramuscular dosing. From day 14 onward, drug plasma concentrations converged in all groups.
In all groups, 2 plasma concentration peaks/shoulders were observed within the first 14 days after administration and prior to the slow first-order decline in plasma concentration (reflecting the slow systemic input and flip-flop pharmacokinetics). In all animals, the first plasma concentration peak was observed within the first 24 hr after dosing. In contrast to the animals from groups 1 and 2, the first peak plasma concentrations in all animals from groups 3 and 5, and most animals in group 4 were measured at the first sampling time point of 2 hr, indicating a faster initial drug release and absorption in those animals. The relative extent of the peak plasma concentrations reached during these 2 systemic input events varied between the different groups. In animals in group 1, 2, and 5, approximately 2- to 4-fold higher peak drug plasma concentrations were reached compared to animals in groups 3 and 4.
In conclusion, and based on nonstatistical assessment of the individual and mean compound X plasma concentration versus time profiles, the 5 nanosuspensions could be clustered as follows: (i) poloxamer-containing nanosuspensions (group 1 [F1] + group 2 [F2]), (ii) polysorbate 80–containing nanosuspensions (group 3 [F3] + group 4 [F4]), and (iii) the TPGS-containing nanosuspension (group 5 [F5]).
Discussion
We investigated the nature and temporal evolution of the administration site reaction following a single i.m. injection with 5 different formulations of a long-acting antiviral nanosuspension containing different stabilizing excipients in the rat. Since the characterization of the nature and dynamics of the local inflammatory response to long-acting nanosuspensions in general, and the critical time points at which significant histopathological changes are observed have been previously studied and reported by our group (Darville, van Heerden, Vynckier, et al. 2014; Darville, van Heerden, Erkens, et al. 2016), the overall objective of the present study was to determine whether such a response can be modulated by the type of stabilizing excipient. To meet this objective, all formulation properties were kept constant as much as practically possible during preparation, apart from the type of stabilizing excipient.
After a single i.m. injection, the general progression of the administration site inflammatory response was relatively similar to that previously described and followed the same general temporal evolution from a short-lived acute inflammation with granulocyte infiltrates, followed by the formation of a rim of depot-infiltrating enlarged macrophages with radial migration tracks, lymphocytic infiltration, angiogenesis and granulation tissue formation (fibrosis/fibroplasia) and, eventually, the slow resolution of both the formulation depot and the chronic inflammatory reaction (Darville, van Heerden, Vynckier, et al. 2014; Darville, van Heerden, Erkens, et al. 2016). However, some subtle differences in the temporal spread of the inflammatory phases and the appearance of inflammatory cell subtypes, the severity grades of the overall inflammatory change, and the nature of the inflammatory response were observed between groups treated with nanosuspensions containing different stabilizing excipients. Administration sites from animals treated with formulations containing polysorbate 80, and polysorbate 80/EPG, and to a lesser extent, those treated with TPGS generally showed higher severity grades of inflammatory components that are associated with the “active” phase of the inflammatory response, combined with a slight delay in the establishment of the chronic phase of the inflammatory response, and its eventual resolution. The slight delay in the onset of the chronic phase of the inflammatory response in these 2 groups was associated with the delayed appearance of lymphocytic infiltrates and the more prolonged resolution of the “active” phase of the inflammation (persistence of viable granulocytic infiltrates, edema and/or hemorrhage). These differences were mostly apparent in the first 7 days (days 1–7) and were less obvious on day 14, where the severity and nature of the inflammatory response appeared to become similar across all groups.
In the chronic phase of the inflammatory response, administration sites of animals injected with formulations containing polysorbate 80 showed a slight delay in the complete infiltration of the formulation depot compared to those injected with formulations containing poloxamer 338, poloxamer 407, and, to a less extent, TPGS. The delayed resolution of the chronic inflammatory response was considered to be most likely due to the surrounding of the formulation depot by macrophage agglomerates that later fused to form MNGCs, their prolonged presence, and the continued phagocytic activity even after the complete infiltration of the formulation depot. The formation of foreign body MNGCs in the groups injected with formulations containing polysorbate 80 was associated with a prolonged strong CD68 positive staining, which suggests that active phagocytosis at the administration sites in these groups continued beyond day 42, since CD68 immunoreactivity is often considered as a marker for active phagocytosis (Chistiakov et al. 2017; de Beer et al. 2003; Perego, Fumagalli, and De Simoni 2011).
The prolonged phagocytosis and prolonged persistence of the formulation depot at the administration sites of animals treated with polysorbate 80–containing nanosuspensions was considered to account for the prolonged tracking of the macrophages to the local lymph nodes.
Although not the primary objective of this study, an empirical assessment of the plasma pharmacokinetics was conducted to evaluate if (any) observed effects on the nature and temporal evolution of the inflammatory changes at the i.m. administration sites could translate into differences in plasma pharmacokinetics. Clear correlations or causalities could (and should) not be established based on the presented data, since a detailed analysis of the pharmacokinetics associated with the 5 different long-acting nanosuspensions fell outside the scope of this report, and additional formulation characterization data, both
This work indicates that subtle vehicle-mediated differences in the injection site reaction following the i.m. administration of LAI crystalline drug nanosuspensions could influence the overall tolerability profile and potentially even the drug pharmacokinetics of these drug delivery systems in the rat and in other species.
Conclusion
Administration sites treated with polysorbate 80–containing nanosuspensions revealed a more severe acute inflammatory response, a slightly delayed and muted lymphocytic response, development of MNGCs, higher severity grades of overall inflammation, and a slower resolution of the inflammatory reaction which was associated with a slightly increased persistence of the formulation depot, when compared to administration sites treated with other stabilizing excipients. Administration sites from the group treated with TPGS-containing formulation appeared to lie somewhere in between the 2 sets (the polysorbate and the poloxamer groups) and shared certain characteristic with both. It can be concluded therefore that the nature and dynamics of the local inflammatory response to long-acting nanosuspensions and the critical time points at which significant histopathological changes are observed can be modulated by the type of stabilizing excipient. This in turn can influence the plasma pharmacokinetics, which in this study showed a good correlation to the histopathological clustering described above.
Footnotes
Acknowledgments
The authors wish to thank the Product Development and Manufacturing organization at Janssen Pharmaceutica NV and especially Michiel Peeters for the manufacturing and characterization of the test articles, the In Vivo Sciences group for the dosing and
Author Contribution
Authors contributed to conception or design (RC, ND); data acquisition, analysis, or interpretation (RC, ND, MH, SD); drafting the manuscript (RC, ND); and critically revising the manuscript (RC, ND, MH, SD). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was provided by the Agency for Innovation by Science and Technology in Flanders/Flanders Innovation & Entrepreneurship agency, Belgium (IWT O&O/AIO R&D grant no. IWT150002).
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
