Abstract
1,1′-Methylenebis[4-[(hydroxyimino)methyl]-pyridinium] (MMB4) dimethanesulfonate (DMS) is a bisquaternary pyridinium aldoxime that reactivates acetylcholinesterase inhibited by organophosphorus nerve agent. Time courses of MMB4 concentrations in plasma were characterized following 7-day repeated intramuscular (IM) administrations of MMB4 DMS to male and female Sprague-Dawley rats, New Zealand White rabbits, beagle dogs (single dose only), and rhesus monkeys at drug dose levels used in earlier toxicology studies. In general, there were no significant differences in MMB4 toxicokinetic (TK) parameters between males and females for all the species tested in these studies. After a single IM administration to rats, rabbits, dogs, and monkeys, MMB4 DMS was rapidly absorbed, resulting in average
Introduction
The clinical treatment of organophosphorus (OP) nerve agent poisoning involves an oxime that reactivates OP-inhibited acetylcholinesterase (AChE). 1 Although the oxime 2-pralidoxime (2-PAM), currently fielded for the US military population, has adequate efficacy against nerve agents such as GB and VX, it has shown less than optimal efficacy toward other nerve agents (GA, GD, GF, and VR). 2 –4 Numerous oximes have been synthesized and evaluated in search of more efficacious AChE reactivators. 5 1,1′-Methylenebis[4-[(hydroxyimino)methyl]-pyridinium] (MMB4) is one of several oximes that have shown better in vitro AChE reactivation potency or in vivo antidotal activity toward various OP nerve agents than 2-PAM. 6 –8
The ideal route of administration for the treatment of OP nerve agent poisoning in combat situations is an intramuscular (IM) injection using autoinjectors for self-administration and buddy aid. Several investigators evaluated MMB4 pharmacokinetics in various preclinical species following a single IM administration. In male pigs that received a single IM administration of MMB4 dimethanesulfonate (DMS) at a dose of 32.9 mg/kg, the elimination half-life, apparent volume of distribution, and apparent clearance values for MMB4 were 53.3 ± 8.7 minutes, 360 ± 46 mL/kg, and 282 ± 12 mL/h/kg, respectively. 9 In male guinea pigs after a single IM administration of MMB4 DMS at a dose of 29, 58, or 116 µmol/kg, MMB4 showed 34.4 to 43.6 minutes for the elimination half-life, 290 to 360 mL/kg for the apparent volume of distribution, and 347 to 352 mL/h/kg for the apparent clearance. 10 In male African Green monkeys after a single IM administration of MMB4 DMS, a dose-dependent difference in MMB4 elimination half-life was observed with 60.4 ± 10.4 minutes at a dose level of 19.3 µmol/kg (9.23 mg/kg) but 21.1 ± 7.8 and 25.0 ± 7.6 minutes at 58 and 116 µmol/kg, respectively. 11 On the other hand, apparent volume of distribution for MMB4 in African Green monkeys was not dependent on dose, with average values ranging from 348 to 417 mL/kg for the 3 dose levels tested. 11
Toxicological profiles of MMB4 DMS have been evaluated in various preclinical species for an investigational new drug (IND) application. Characterization of toxicokinetics (TK) of MMB4 DMS at various dose levels investigated in those species would help better understand the toxicological outcomes. In addition, MMB4 TK data in preclinical species can be used to predict important parameters in humans such as maximal MMB4 concentration (
This study was conducted following Good Laboratory Practice requirements so that experimental data generated from this study can be included in the IND application. The study was conducted in compliance with The American College of Toxicology Policy on the Use of Animals, under the guidance of the Battelle Institutional Animal Care and Use Committee and the Animal Care and Use Review Office of the US Army.
Materials and Methods
Dose
The test article for this study was MMB4 (CAS no. 61444-84-6) DMS and is described as an off-white to tan crystalline powder based on the Certificate of Analysis. Lot no. 1004 MMB4 DMS, with a manufacture date of July 2007. The source of the test article was Cambrex Charles City, Inc (Charles City, Iowa). The purity of the test article was >98%. The dose solution was formulated using 0.5% benzyl alcohol in water for IM injection. The dose formulation was stable when stored at room temperature for 14 days.
Experimental Animals
A total of 63 Sprague-Dawley rats per sex were purchased from Charles River Laboratories (Portage, Michigan). The rats were 9 to 13 weeks of age and weighed 250 ± 50 g at the start of the study. In all, 17 New Zealand White rabbits per sex were purchased from Myrtle Rabbitry, Inc. (Thompson Station, Tennessee). The rabbits were 13 to 15 weeks of age and weighed 2.2 to 3.0 kg at the start of the study. Four beagle dogs per sex were purchased from Covance Research Products, Inc (Kalamazoo, Michigan). The dogs were 17 to 20 months of age and weighed 6.9 to 14.2 kg at the start of the study. A total of 24 rhesus monkeys per sex were purchased from Primate Products Inc (Miami, Florida). The monkeys were 1 to 3 years of age and weighed 2.6 to 4.9 kg at the start of the study. All animals were individually housed in stainless steel cages with ad libitum access to feed and freshwater. The animal study rooms were maintained for temperature (61-84°F), humidity (30%-70%), and 12-hour light–12-hour dark cycle.
Dose Administration
The rats and rabbits received daily IM dose administrations of 100, 200, 400, or 800 mg/kg/d MMB4 DMS for 7 consecutive days. A dose volume of 0.25 mL/kg was administered based on the individual animal body weight recorded on the day of dose administration. The dogs received a single IM dose administration of 20 or 100 mg/kg MMB4 DMS. The dogs were used to evaluate safety pharmacology end points, for example, cardiopulmonary toxicity, but not as a test system to evaluate general toxicology, for example, systemic toxicity; for this reason, no repeat exposure testing was performed with the dogs. A dose volume of 0.25 mL/kg was administered based on the individual animal body weight recorded on the day of dose administration. The monkeys received daily IM dose administrations of 150, 300, 450, or 600 mg/kg/d MMB4 DMS for 7 consecutive days. A dose volume of 0.5 mL/kg was administered based on the individual animal body weight recorded on the day of dose administration. The dose levels for each species in this study were identical to those used in earlier toxicology studies. 12
Blood Sample Collections
For rats and rabbits, whole blood samples were collected predose and at 5, 10, 15, 20, 30, and 45 minutes and 1, 2, 4, 6, and 8 hours after the completion of dose administration on days 1 and 7. For dogs, whole blood samples were collected predose and at 5, 15, and 30 minutes and 1, 2, 4, 8, 12, 24, and 48 hours after the completion of dose administration on day 1. For monkeys, whole blood samples were collected predose and at 5, 15, and 30 minutes, and 1, 2, 4, 8, and 24 hours after the completion of dose administration on days 1 and 7. Blood samples were collected from the retro-orbital sinus for rats, ear artery for rabbits, jugular or peripheral vein for dogs, and femoral artery or vein for monkeys. All blood was collected in tubes containing lithium heparin as anticoagulant. After collection, blood samples from all animals were kept cold on wet ice until processed to plasma, which occurred within 60 minutes after blood collection. All blood samples were separated by centrifugation for at least 10 minutes using a refrigerated centrifuge (Allegra 21R; Beckman Coulter, Brea, California) set to maintain 5°C and 2200 rpm. Each sample of plasma was transferred into an appropriately labeled polypropylene vial and stored in a freezer set to maintain −30°C to −10°C until analysis.
Bioanalysis of MMB4
Plasma calibration standards for each species were prepared from 2 independently prepared stock solutions. The plasma calibration standards, plasma blanks, plasma quality control samples, and plasma samples were processed by protein precipitation. The resulting extracts were analyzed by high-performance liquid chromatography with mass spectrometry (HPLC/MS). The HPLC system consisted of a Shimadzu Prominence pump and detector (Shimadzu Scientific Instruments, Kyoto, Japan) for the monkeys and an Agilent pump and detector (Agilent Technologies, Palo Alto, California) for the rats, rabbits, and dogs. The MS system was Sciex API 3000 (AB SCIEX, Toronto, Ontario) for the monkeys and Sciex API 4000 (AB SCIEX, Toronto, Ontario) for the rats, rabbits, and dogs, with turbo spray and positive ion mode used for all MS systems. Samples were injected into 100 mm × 2 mm Hydro-RP column (Phenomenex, Torrance, California). The MMB4 concentrations were calculated using peak area response ratios, and a regression equation was constructed from the calibration standards. The lower limit of quantitation for this analytical method was 50 ng/mL for MMB4 in plasma using 100 µL of sample.
Toxicokinetic Analysis
The TK analysis was performed using the actual dose of MMB4 (mg/kg), actual sample collection time points (hours), and the measured concentrations of MMB4 in plasma (µg/mL). The MMB4 concentration–time profiles were evaluated by noncompartmental analysis using WinNonlin (Pharsight Corporation, Mountain View, California). Statistical differences in average TK values were determined using the Wilcoxon rank-sum test, a nonparametric method that does not require assumptions of normality and homogeneous variance, with a significance level of
Results
Toxicokinetics of MMB4 in Rats
The MMB4 was measurable in plasma at the first time point (5 minutes) up to and including 8 hours after a single or 7-day repeated IM administration of MMB4 DMS to male and female rats at 100, 200, 400, and 800 mg/kg/d (Figure 1). There was a secondary peak at 1 hour postdose in concentration versus time profile for the male rats after a single IM injection at 100 mg/kg. However, it was not observed with any other dose groups on day 1 or after the 7-day repeated administrations. The MMB4 TK parameters were generally similar for male and female rats, while several TK values in the 100 mg/kg dose groups on day 1 and in the 800 mg/kg/d dose groups on day 7 showed marginal sex-related differences (Table 1).
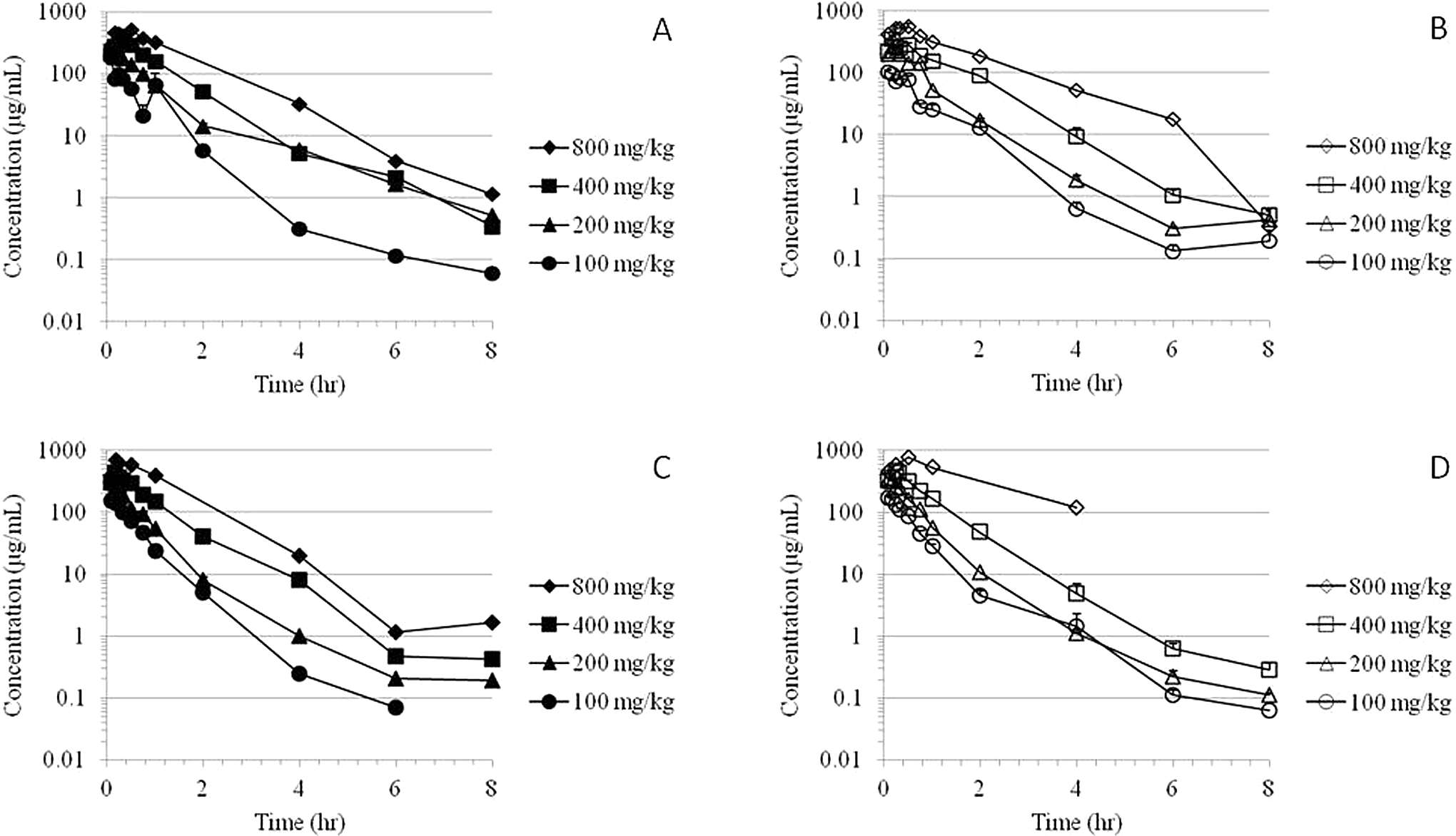
Time courses of group mean plasma MMB4 concentrations following a single IM administration of MMB4 DMS to male (A) and female (B) rats and following 7-day repeated administrations to male (C) and female (D) rats. DMS indicates dimethanesulfonate; IM, intramuscular; MMB4, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium].
Summary of TK Parameters for Rats Following IM Administration of MMB4 DMS.
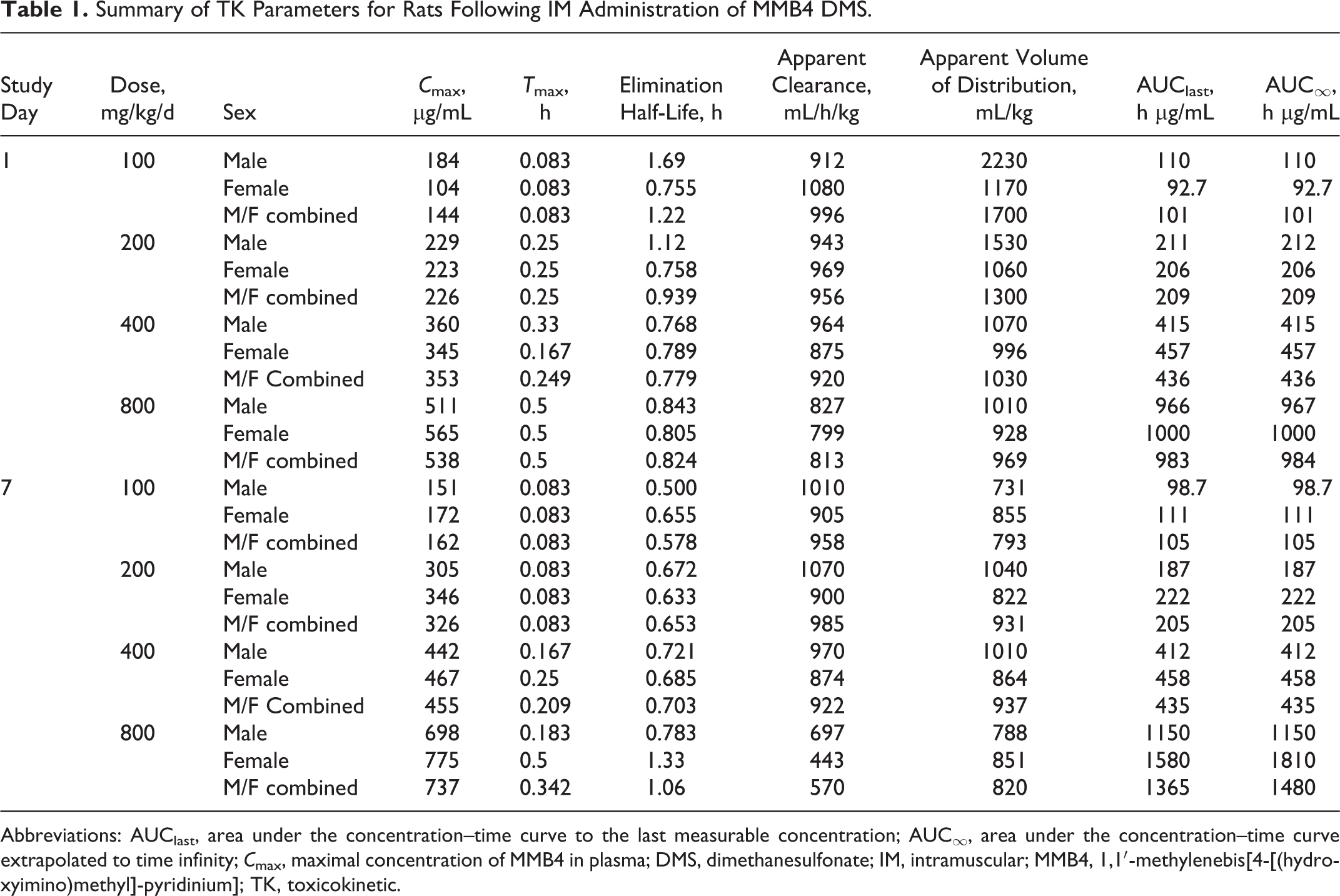
Abbreviations: AUClast, area under the concentration–time curve to the last measurable concentration; AUC∞, area under the concentration–time curve extrapolated to time infinity;
After a single IM injection of MMB4 DMS, the maximal concentration of MMB4 (
The repeated administrations of MMB4 DMS for 7 days did not result in overtly different TK values compared to the single-dose TK data (Table 1). The
Toxicokinetics of MMB4 in Rabbits
The MMB4 was measurable in plasma at the first time point (5 minutes) up to and including 8 hours after a single- or 7-day repeated IM administration of MMB4 DMS to male and female rabbits at 100, 200, 400, and 800 mg/kg/d (Figure 2). The MMB4 TK parameters were similar for male and female rabbits after a single IM injection or 7-day repeated administrations (Table 2). There were no statistically significant sex-related differences in TK parameters except for the
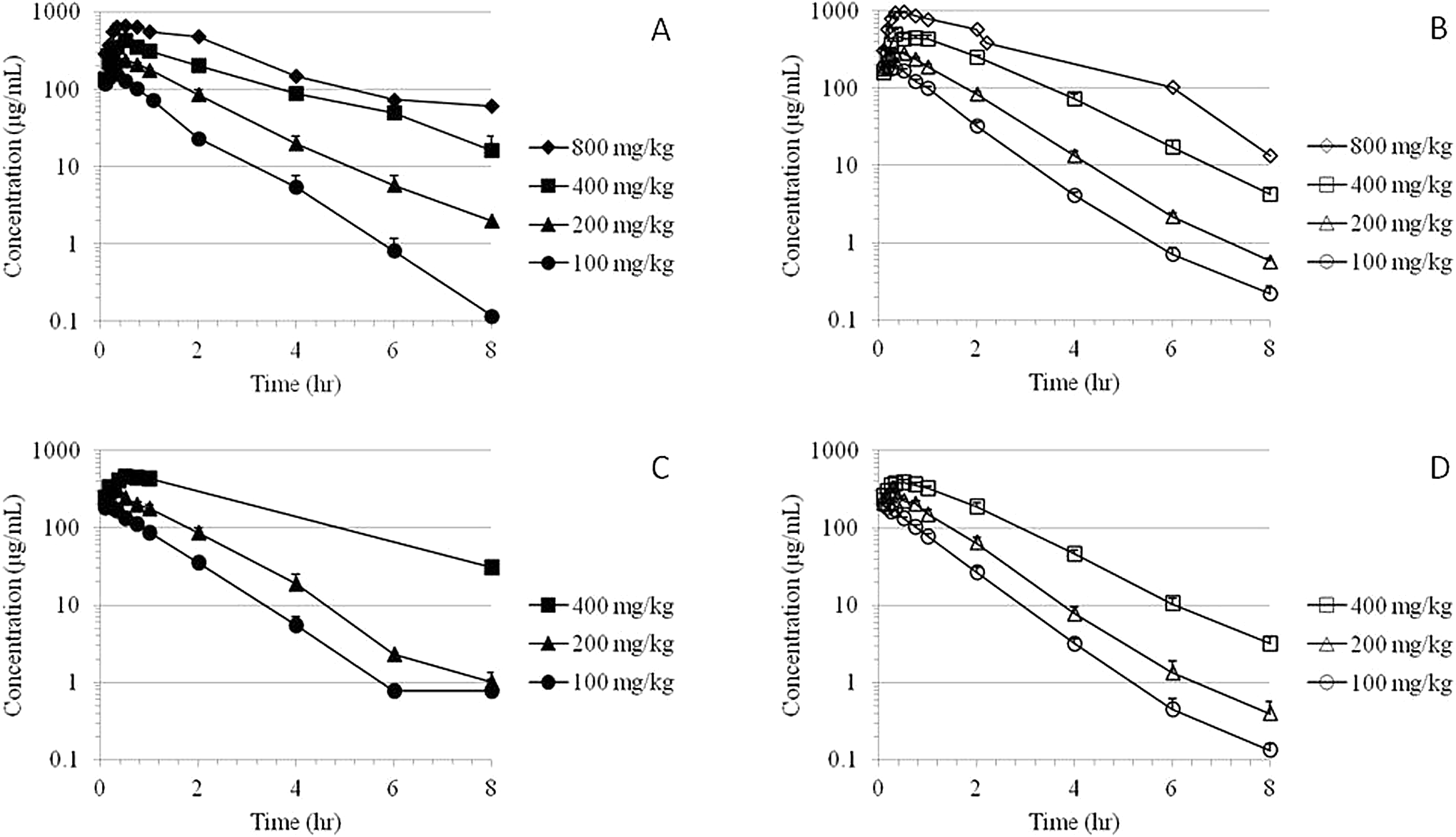
Time courses of group mean plasma MMB4 concentrations following a single IM administration of MMB4 DMS to male (A) and female (B) rabbits and following 7-day repeated administrations to male (C) and female (D) rabbits. DMS indicates dimethanesulfonate; IM, intramuscular; MMB4, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium].
Summary of TK Parameters for Rabbits Following IM Administration of MMB4 DMS.
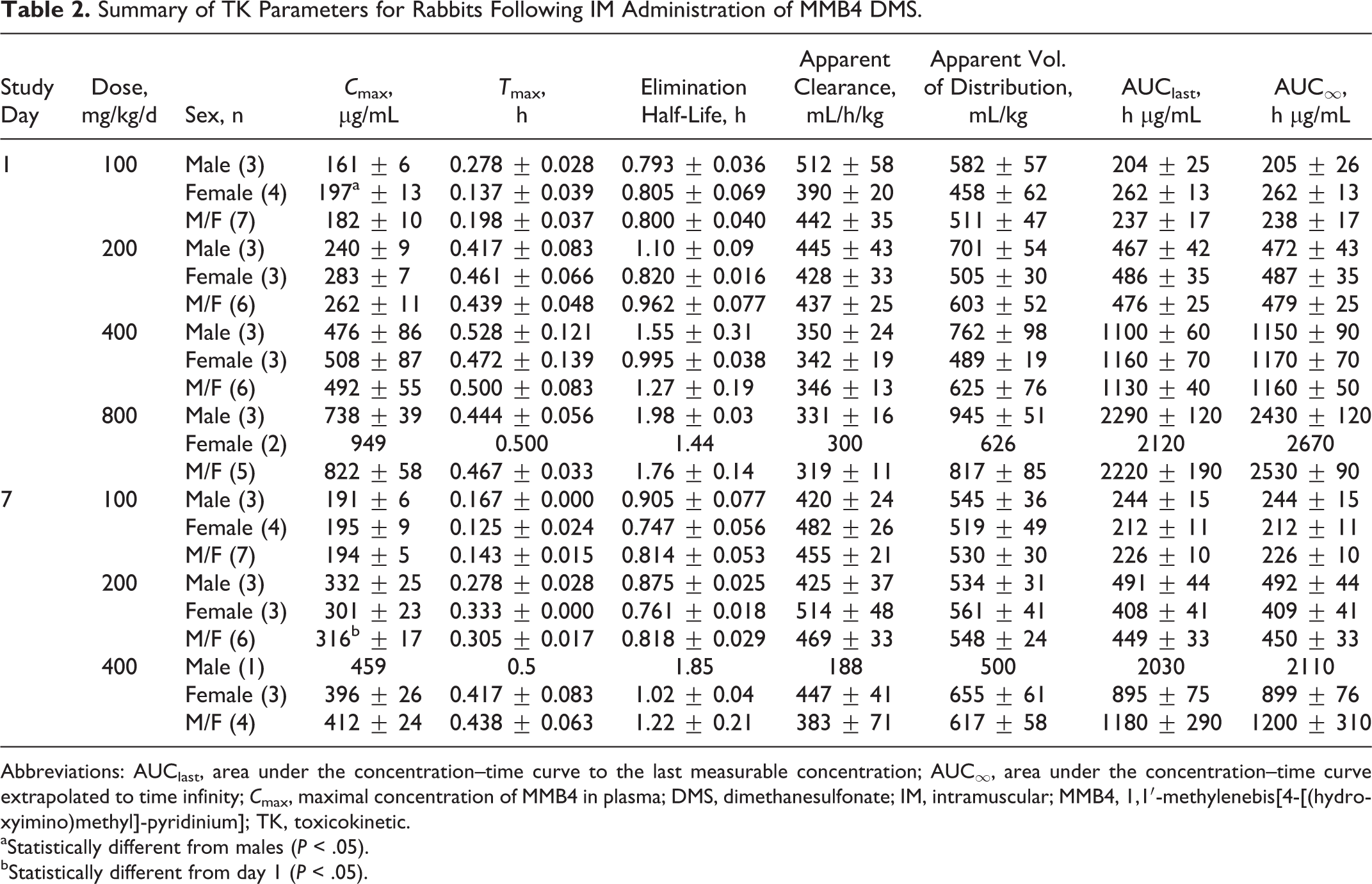
Abbreviations: AUClast, area under the concentration–time curve to the last measurable concentration; AUC∞, area under the concentration–time curve extrapolated to time infinity;
aStatistically different from males (
bStatistically different from day 1 (
After a single IM injection of MMB4 DMS at 100 mg/kg dose, the maximal concentration of MMB4 in plasma was observed at 12 ± 2 minutes, whereas higher dose groups showed delayed
The TK parameters of MMB4 after the repeated administrations for 7 days at 100, 200, and 400 mg/kg/d dose levels were very comparable with the TK results obtained after a single IM injection to rabbits at respective doses of MMB4 DMS (Table 2). There were no statistically significant differences in day 1 versus day 7 TK parameters except for
Toxicokinetics of MMB4 in Dogs
The MMB4 was measurable in plasma at the first time point (5 minutes) up to and including 12 hours after a single IM administration of MMB4 DMS to male and female dogs at 20 and 100 mg/kg (Figure 3). There were no statistically significant sex-related differences in TK parameters except for apparent clearance and AUC values at the 100 mg/kg dose (Table 3).
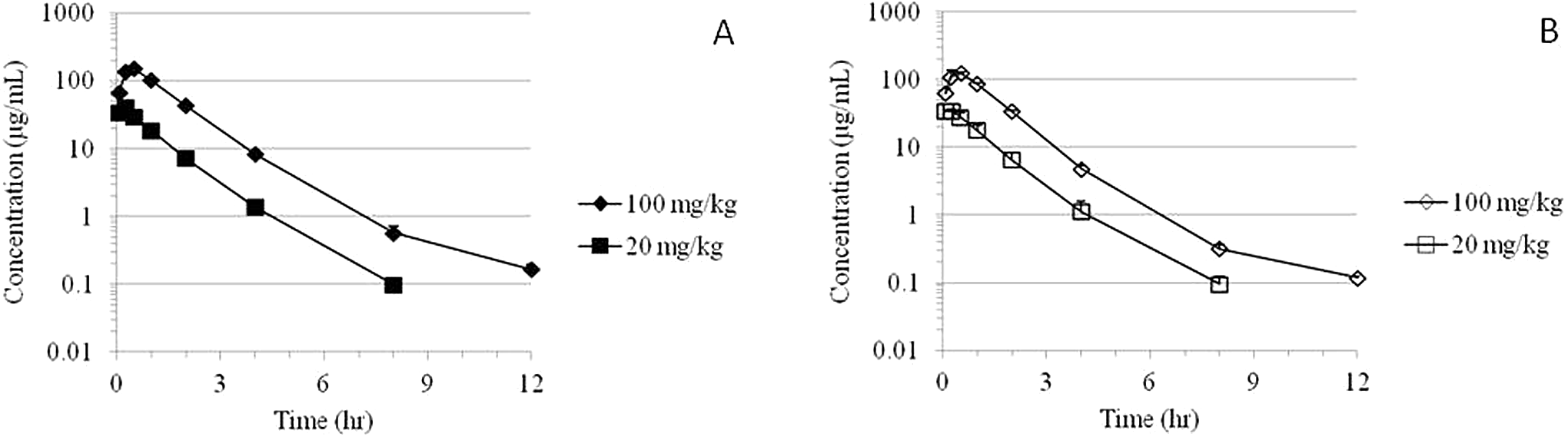
Time courses of group mean plasma MMB4 concentrations following a single IM administration of MMB4 DMS to male (A) and female (B) dogs. DMS indicates dimethanesulfonate; IM, intramuscular; MMB4, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium].
Summary of TK Parameters for Dogs Following IM Administration of MMB4 DMS.
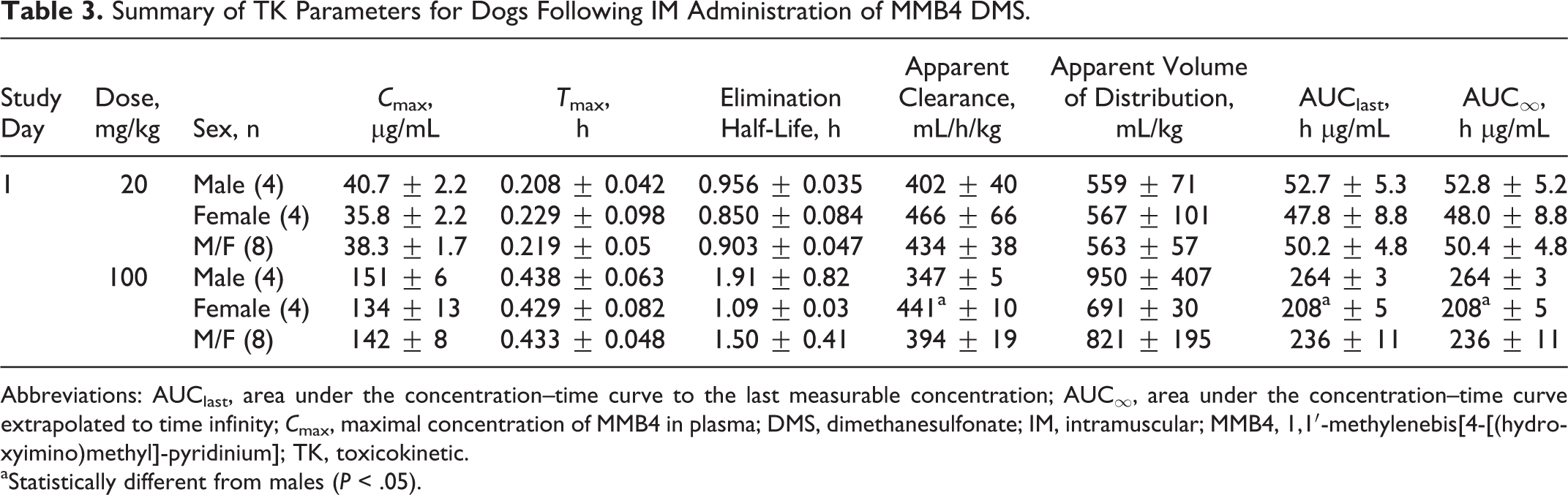
Abbreviations: AUClast, area under the concentration–time curve to the last measurable concentration; AUC∞, area under the concentration–time curve extrapolated to time infinity;
aStatistically different from males (
After a single IM injection of MMB4 DMS at 20 mg/kg dose, the maximal concentration of MMB4 in plasma was observed at 13 ± 3 minutes, whereas a delayed
Apparent clearance values were not dependent on dose, with average clearance of 434 ± 38 and 394 ± 19 mL/h/kg for the 20 and 100 mg/kg dose groups, respectively, when data from males and females were combined. Average apparent volume of distribution for male dogs was higher than that of females at the 100 mg/kg dose level due to the outlier male dog (2170 mL/kg). When this animal was excluded, the average volume of distribution for male dogs (543 ± 12 mL/kg) was comparable to that of female dogs (691 ± 30 mL/kg). Furthermore, the average volume of distribution for males and females combined excluding the outlier male dog at the 100 mg/kg dose level (628 ± 34 mL/kg) was similar to 20 mg/kg dose level (563 ± 57 mL/kg). Average AUClast accounted for greater than 99% of average AUC∞. The average AUC∞ value increased by 4.7-fold when the dose of MMB4 DMS increased by 5-fold.
Toxicokinetics of MMB4 in Monkeys
The MMB4 was measurable in plasma at the first time point (5 minutes) up to and including 24 hours after a single- or 7-day repeated IM administration of MMB4 DMS to male and female monkeys at 150, 300, 450, and 600 mg/kg/d (Figure 4). In general, MMB4 TK parameters were similar for male and female monkeys after a single IM injection or 7-day repeated administrations (Table 4). There were no statistically significant sex-related differences in TK parameters except for apparent volume of distribution and AUC values at the 450 and 600 mg/kg/d dose levels on day 1 and
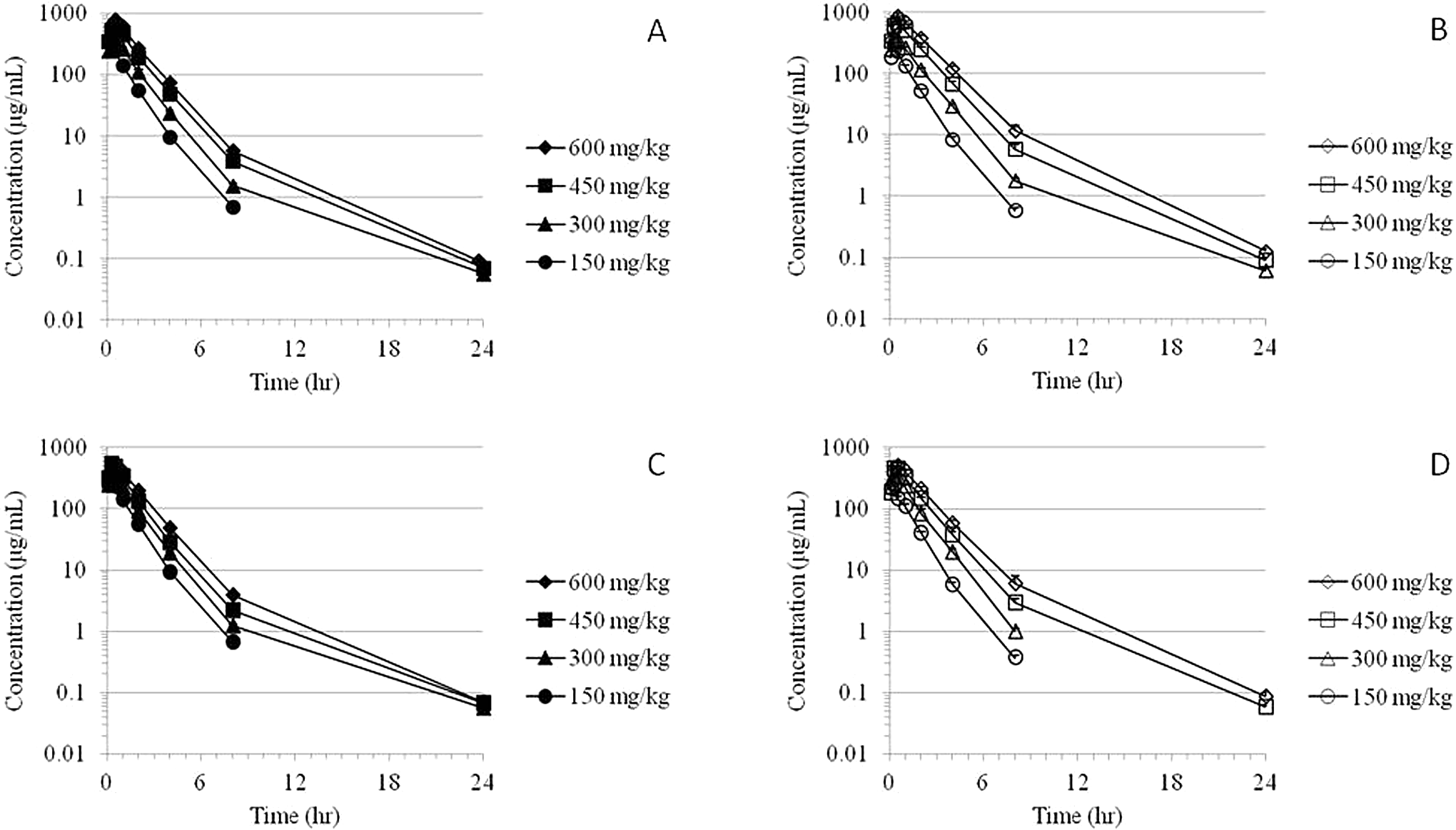
Time courses of group mean plasma MMB4 concentrations following a single IM administration of MMB4 DMS to male (A) and female (B) monkeys and following 7-day repeated administrations to male (C) and female (D) monkeys. DMS indicates dimethanesulfonate; IM, intramuscular; MMB4, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium].
Summary of TK Parameters for Monkeys Following IM Administration of MMB4 DMS.
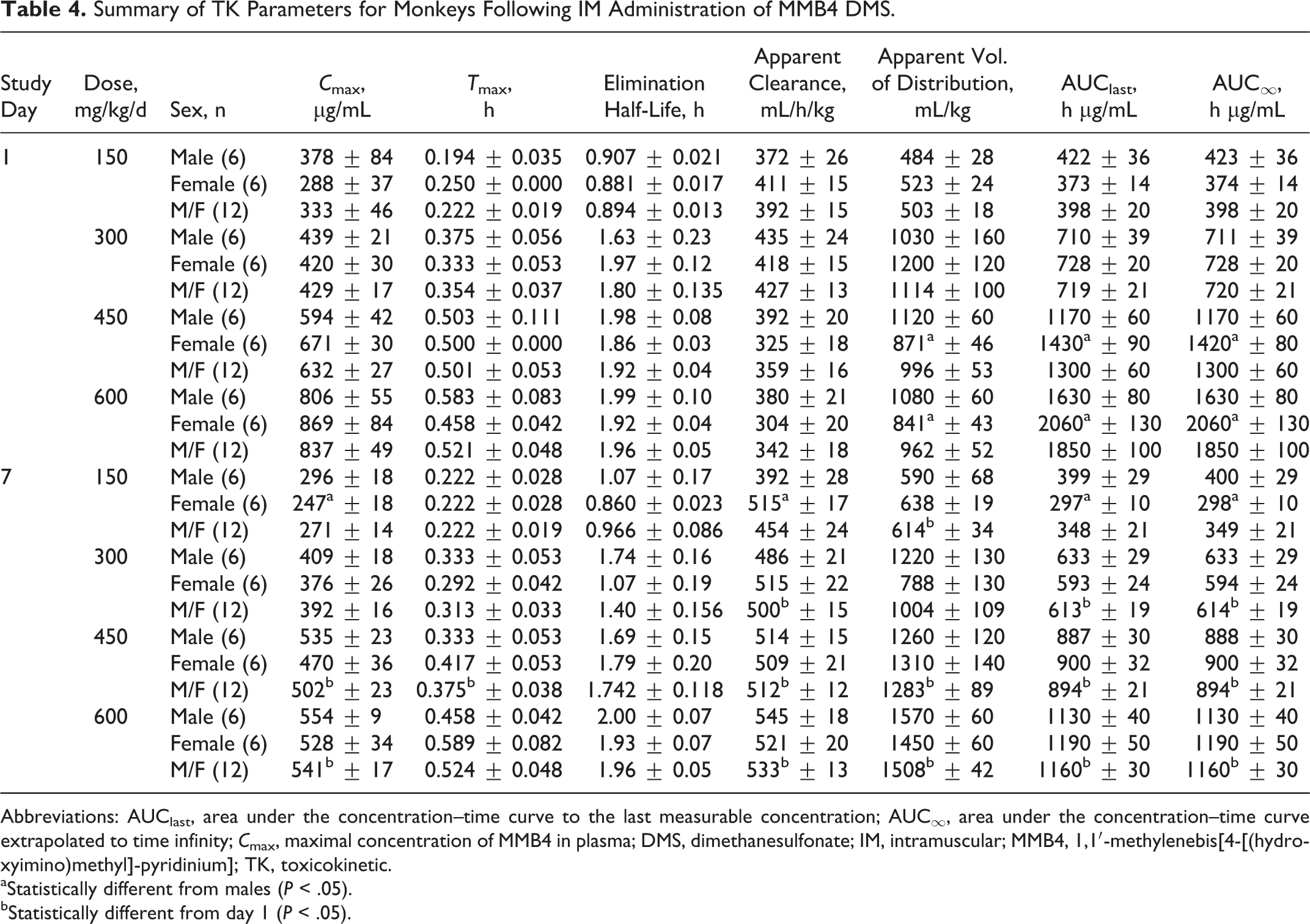
Abbreviations: AUClast, area under the concentration–time curve to the last measurable concentration; AUC∞, area under the concentration–time curve extrapolated to time infinity;
aStatistically different from males (
bStatistically different from day 1 (
After a single IM injection of MMB4 DMS at 150 and 300 mg/kg dose, the maximal concentration of MMB4 in plasma was observed at 13 to 21 minutes, whereas higher dose groups showed slightly delayed
After the repeated administrations for 7 days at the 150 and 300 mg/kg/d dose levels, TK parameters for MMB4 were generally consistent with the results obtained after a single IM injection of MMB4 DMS to monkeys at respective doses (Table 4). However, the 7-day repeated administrations of MMB4 DMS led to statistically significant differences in several TK parameters but not for all the dose levels. The following parameters were found to be statistically significant after the repeated administration:
Discussion
The current study characterized the TKs of MMB4 in rats, rabbits, dogs, and monkeys following a single- and 7-day repeated IM administration at various dose levels used in toxicology evaluations. In general, there were no statistically significant differences in MMB4 TK parameters between males and females after a single IM administration or 7-day repeated injections of MMB4 DMS to all the species tested in this study. Although several TK parameters showed sex-related differences, there were no coherent patterns suggesting that males and females exhibited different TKs of MMB4 at the dose levels investigated in this study.
After a single IM administration to rats, rabbits, dogs, and monkeys, MMB4 DMS was rapidly absorbed, resulting in measurable levels of MMB4 in plasma at the first sample collection time point (ie, 5 minutes). In addition, maximal concentration of MMB4 in plasma was achieved early with average
Considering the rapid elimination half-life of MMB4 in these species, with average values ranging from approximately 0.8 to 1.2 hours, the less than dose-proportional increase in
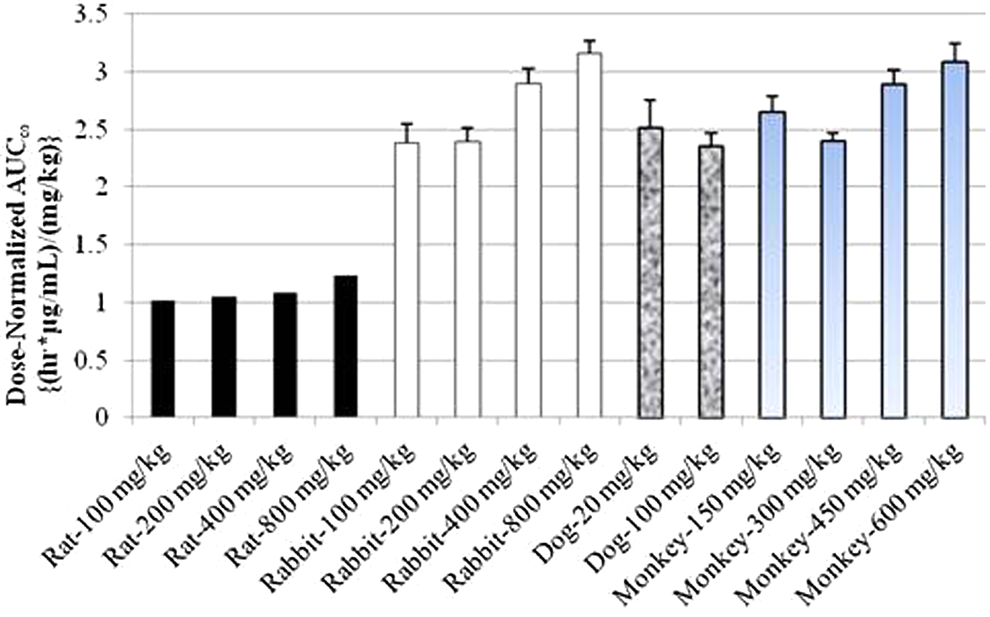
Comparison of dose-normalized AUC∞ after single IM administration of MMB4 DMS to rats, rabbits, dogs, and monkeys. AUC∞ indicates area under the concentration–time curve extrapolated to time infinity; DMS, dimethanesulfonate; IM, intramuscular; MMB4, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium].
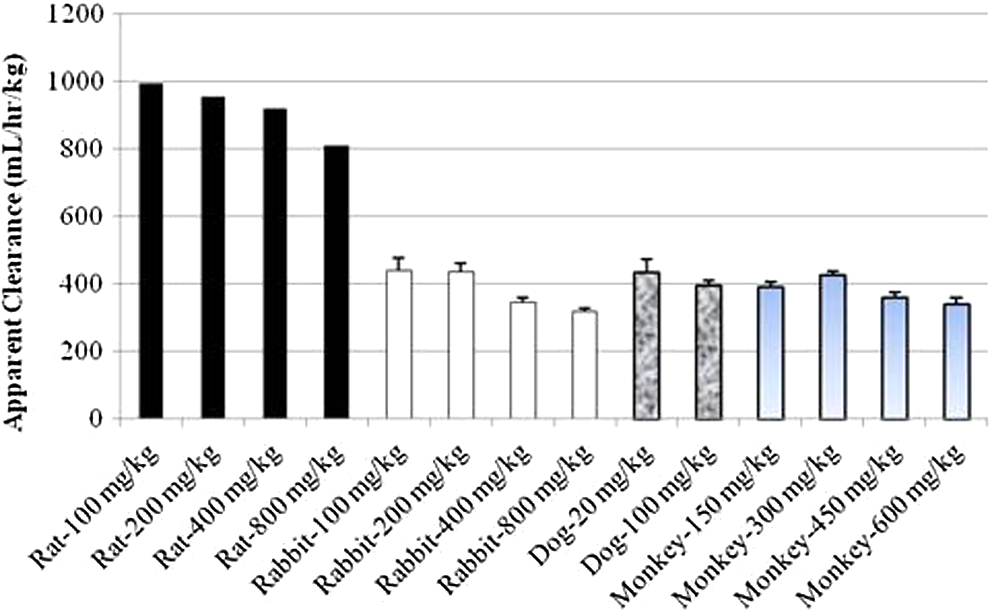
Comparison of apparent clearance after single intramuscular (IM) administration of 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (MMB4 DMS) to rats, rabbits, dogs, and monkeys.
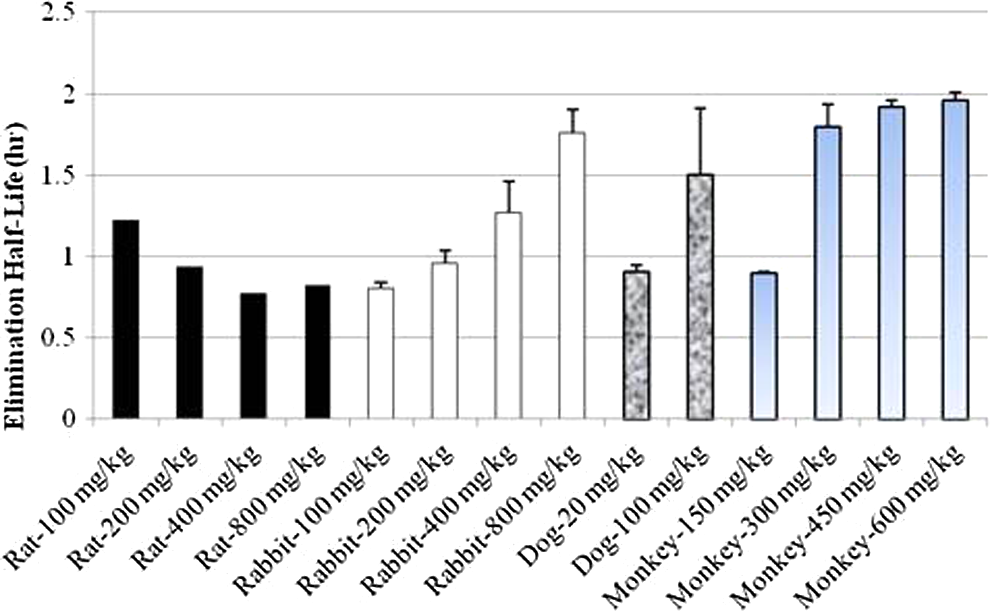
Comparison of elimination half-life after single intramuscular (IM) administration of 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (MMB4 DMS) to rats, rabbits, dogs, and monkeys.
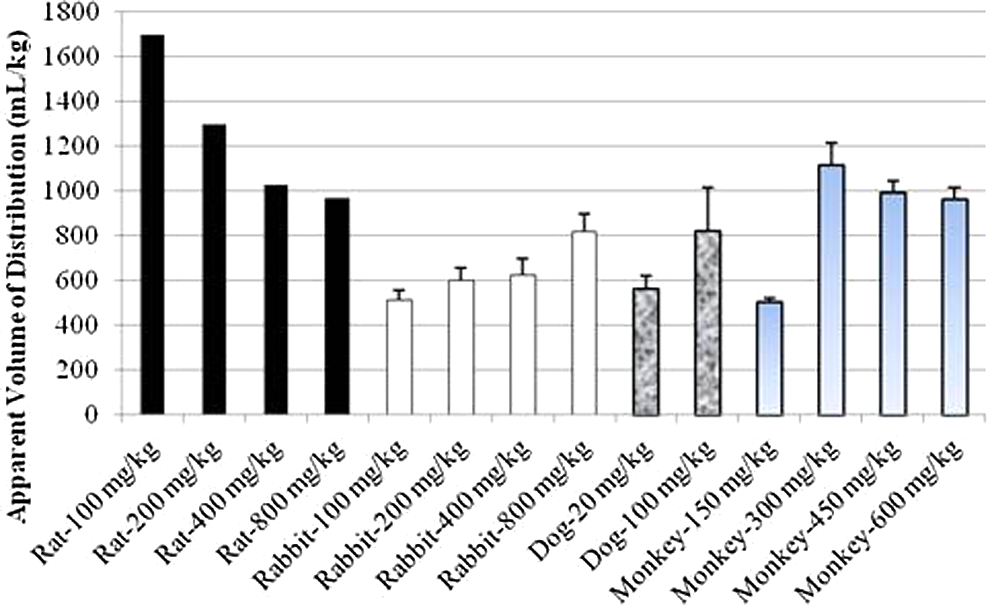
Comparison of apparent volume of distribution after single intramuscular (IM) administration of 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (MMB4 DMS) to rats, rabbits, dogs, and monkeys.
The absolute bioavailability after a single IM injection of MMB4 DMS to rats, rabbits, and dogs could be calculated using the experimental data obtained from the current study and a previous study that examined MMB4 pharmacokinetics after a single intravenous (IV) administration. 13 The absolute bioavailability values ranged from 79% to 95% for rats, 124% to 164% for rabbits, and 89% to 95% for dogs. These results suggest that MMB4 is extensively absorbed into the systemic circulation after the IM administration. However, it is not clearly understood for the absolute bioavailability values of rabbits exceeding a theoretical limit (ie, 100%), since AUC∞ values of rabbits increased in a dose-proportional manner. This might be caused by an underestimated AUC value from the intravenous (IV) study if the dosage of MMB4 DMS (25 mg/kg) was out of dose-proportional range. Additional investigation for IV administration at multiple dose levels would be required to test this hypothesis.
The repeated administrations of MMB4 DMS for 7 days did not overtly alter TK parameters for MMB4 in rats and rabbits. A few TK parameters were affected by the repeated administrations, but there were no consistent changes across different TK parameters for a given dose level or among different dose levels for a given TK parameter. On the other hand, the majority of TK parameters in monkeys given a 450 and 600 mg/kg/d IM dose of MMB4 DMS changed following the repeated administrations of MMB4 DMS for 7 days.
Following a single IM administration, other oximes such as 2-PAM and HI-6 (asoxime) exhibited differences in some PK parameters when compared to those for MMB4 DMS. The 2-PAM showed 14.0 ± 3.5 minutes of
When compared to HI-6, the rate of MMB4 elimination in rats was comparable, since HI-6 showed 59.5 ± 25.5 minutes of elimination half-life after a single IM administration to rats at a 20 mg/kg dose. 16 On the other hand, MMB4 showed a faster clearance and a larger volume of distribution to the central compartment in rats than HI-6 (297 ± 137 mL/h/kg, and 370 ± 120 mL/kg, respectively). 16 In dogs, the rate of elimination, volume of distribution, and clearance of MMB4 were modestly larger than those of HI-6 that showed 41.6 ± 7.8 minutes of elimination half-life, 320 ± 40 mL/kg of volume of distribution, and 320 ± 32 mL/h/kg of clearance following a single IV administration at a 20 mg/kg dose. 17
The MMB4 pharmacokinetic data obtained from various preclinical species provide useful insight in predicting MMB4 kinetics in humans. Using the TK results obtained from the current study and published investigations,
9
–11
the relationships between the body weights of preclinical species and the apparent volume of distribution or the apparent clearance were examined (Figure 9). It was notable to observe conserved apparent volume of distribution and clearance values among various preclinical species, except for the rat that exhibited 3- to 5-fold larger volume of distribution and 2- to 4-fold faster clearance. When the rat values were excluded, the average apparent volume of distribution and clearance values was 444 ± 92 mL/kg and 395 ± 70 mL/h/kg, respectively. If humans had similar volume of distribution and clearance values, the predicted
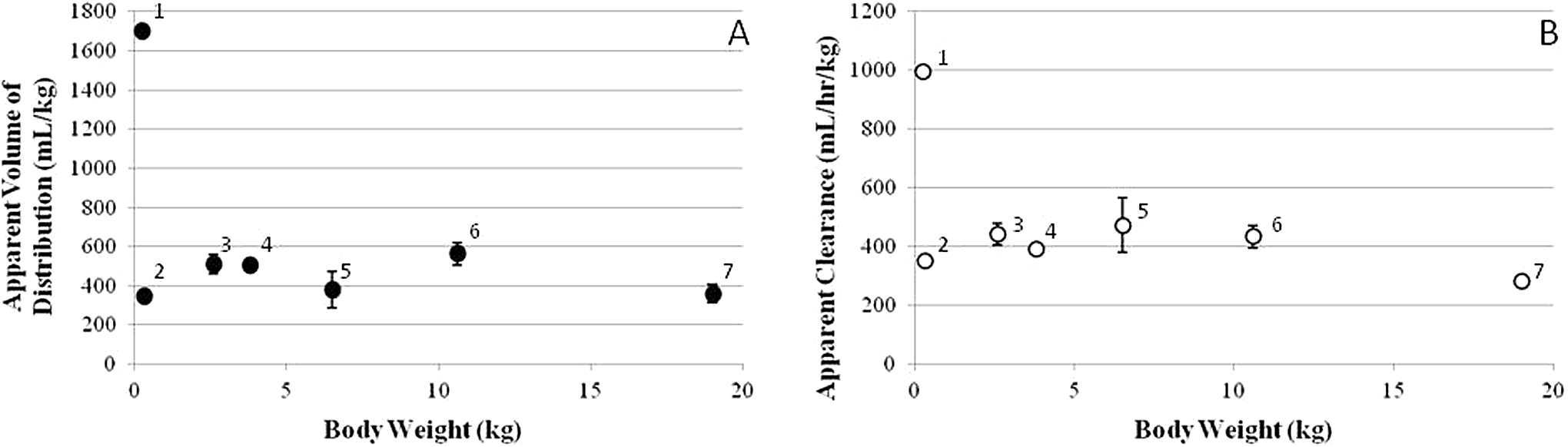
Relationship between body weight of preclinical species used for toxicokinetic (TK) evaluation and apparent volume of distribution (A) or apparent clearance (B) for 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] (MMB4). 1, Rat data from the current study. 2, Guinea pig data from Capacio. 10 3, Rabbit data from the current study. 4, Monkey data from the current study. 5, Monkey data from Myers. 11 6, Dog data from the current study. 7, Pig data from Stemler et al. 9
Footnotes
Acknowledgments
The authors wish to thank Dr. Vincent Brown for the editorial assistance.
Authors’ Notes
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or reflecting true views of the Department of the Army or the Department of Defense.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the United States Army; Chemical Biological Medical Systems (CBMS)/Medical Identification and Treatment Systems (MITS) under contract SP0700-00-D-D3180, Delivery Order 0599 (Task 771), and Delivery Order 0600 (Task 789).
