Abstract
Studies were conducted in Sprague-Dawley rats, New Zealand White (NZW) rabbits, and rhesus monkeys to characterize the toxicity of 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (MMB4 DMS) following intramuscular administration. Rats received MMB4 DMS once daily for 7 days at 100, 200, 400, and 800 mg/kg/d; rabbits received a range of dose levels in 3 separate 7-day studies from 3 to 800 mg/kg/d and in a single-dose study from 50 to 200 mg/kg; and monkeys received MMB4 DMS at 150 to 600 mg/kg/d. Mortality was noted in rats and rabbits administered ≥200 mg/kg. All monkeys survived until scheduled termination. Adverse clinical observations were noted in the rats at ≥400 mg/kg/d and in rabbits administered ≥200 mg/kg; no adverse findings were noted in the monkeys. Clinical pathology changes were noted in the rabbit related to cardiac and renal function. In the rabbit and monkey, elevations in myoglobin, alanine aminotransferase/aspartate aminotransferase, platelets, creatine kinase, and coagulation factors were related to local inflammation at the intramuscular administration site. Light microscopic examination at the injection sites revealed acute skeletal muscle necrosis in vehicle control and treated groups. Target tissues in the rabbit studies were identified as kidney, heart, and lungs at ≥100 mg/kg/d. All changes noted in all the species demonstrated partial to complete recovery comparable to control values or to a clinically irrelevant level of effect. The NZW rabbit was the most sensitive species, and the no observed adverse effect level (NOAEL) was determined as 50 mg/kg/d; the NOAEL in the rat was 100 mg/kg/d; and the NOAEL in rhesus monkeys was >600 mg/kg/d.
Introduction
Organophosphorus (OP) nerve agents are among the most lethal chemical weapons. They are chemically stable, easily dispersed, highly toxic, and have rapid effects when absorbed through the skin and via respiration. The pathophysiology of nerve agent intoxication is well understood. The OP nerve agents irreversibly bind to acetylcholinesterase (AChE), causing the phosphorylation and deactivation of AChE. The clinical effects are secondary to acetylcholine excess at cholinergic junctions (muscarinic effects) in the central nervous system and at skeletal nerve–muscle junctions and autonomic ganglia (nicotinic effects).
The United States Army Medical Research Institute of Chemical Defense is evaluating new drugs that are more efficacious and provide broader protection against the effects of nerve agents such as sarin that was used by terrorists in Matsumoto in 1994 and Tokyo in 1995. These and the other acts, including those of September 11, 2001, indicate that terrorists intend to maximize the lethality of their attacks, which may include the use of nerve agents. The anticipated treatment settings are during a military field combat situation following an exposure to nerve agents or in a first-echelon medical care unit (ie, follow-on medical treatment).
Countermeasures against nerve agents include a combination of up to 4 drug classes including carbamate (pyridostigmine bromide [PB]) pretreatment and an antimuscarinic (atropine), an oxime, and a benzodiazepine for postexposure treatment. Atropine and oxime are the first-line drug treatments for OP poisoning. The only oxime licensed in the United States for the treatment of nerve agent exposure is 2-pralidoxime chloride (2-PAM). Although 2-PAM has acceptable efficacy against certain nerve agents (eg, GB and VX), it lacks the desired level of efficacy against other agents (eg, GA, GD, GF, and VR), even when combined with PB pretreatment and atropine and diazepam postexposure. This lack of efficacy is being addressed by the development of a number of new countermeasures, among which is 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate (MMB4 DMS). The MMB4 DMS is intended for use as a treatment against the lethal effects of nerve agent intoxication at the first sign of poisoning, replacing the currently fielded oxime 2-PAM in the Antidote Treatment Nerve Agent Autoinjector. In this system, MMB4 DMS will be coadministered intramuscularly (IM) with atropine.
The Chemical Biological Medical Systems (CBMS) Medical Identification and Treatment System team designed and completed a preclinical evaluation program consisting of a battery of animal studies designed to demonstrate the efficacy, pharmacology, and toxicology of MMB4 DMS in support of a phase 1 study in human volunteers to evaluate the pharmacokinetics (PK) and safety of the test article in a dose-escalation paradigm. Companion articles chronicling the pharmacokinetics, efficacy, and safety pharmacology of MMB4 DMS in a variety of species appear elsewhere in this issue in order to share results with the scientific community for this safe and effective countermeasure against nerve agent intoxication. This article focuses on the comparative species safety results. The study was conducted in compliance with The American College of Toxicology Policy on the Use of Animals.
Materials and Methods
Materials
Test article
The test article for this study was MMB4 DMS and is described as an off-white to tan crystalline powder based on the Certificate of Analysis. Lot no. 1004 MMB4 DMS, with a manufacture date of July 2007 was used in this study. The source of the test article was Cambrex Charles City, Inc (Charles City, Iowa). The purity of the test article was >98%.
Vehicle
The vehicle consisted of 0.5% benzyl alcohol in sterile water for injection.
Formulation preparation
Dose formulations of MMB4 DMS were prepared according to a procedure developed by Battelle and formulated at the appropriate target concentrations for each of the studies discussed in 0.5% benzyl alcohol in water for injection. The dose formulations were adjusted to pH 2.3 using methane sulfonic acid (MSA) in water solution (1:50 MSA solution) and/or NaOH (0.1 N). Prior to administration, vehicle control and MMB4 DMS dose formulations were stored in a refrigerator set to maintain 2°C to 8°C.
Dose formulation analysis
The MMB4 DMS dose concentration of each formulation batch was determined by high-performance liquid chromatography using a validated procedure prior to and after dose administration. The postadministration analysis was performed in samples taken from bottles containing the vehicle control and MMB4 DMS formulations that were stored with the bottles used for dose administration.
Animals
Following approval by the test facility Institutional Animal Care and Use Committee and the sponsor’s Animal Care and Use Review Office, the animals were purchased from a qualified supplier. Upon receipt, all animals were observed while in quarantine for any abnormalities that would make them unfit for study. General procedures for animal care, housing, and environmental conditions conformed (as appropriate) to current Association for Assessment and Accreditation of Laboratory Animal Care International recommendations, current requirements stated in the
All animals were housed individually in stainless steel cages. The animals were housed in environmentally controlled rooms with at least 10 air changes per hour. The rooms were maintained at a temperature between 18°C and 26°C (64-79°F) and 30% to 70% relative humidity, in accordance with the USDA Animal Welfare Act.
3
A 12-hour light/12-hour dark photoperiod was maintained with timer-controlled lighting. Each cage met or exceeded specifications of the USDA Animal Welfare Act
3
as described in the
Rats
An appropriate number of Sprague-Dawley rats were procured from Charles River Laboratories (Raleigh, North Carolina) for the study. At initiation of dosing, males were approximately 12 weeks of age, and females were approximately 20 weeks of age. The males weighed between 328.0 and 435.2 g, and the females weighed between 243.0 and 316.2 g. The animals were individually housed in suspended stainless steel wire mesh cages, each containing a feeder, a source of water (pressure-activated water nipples), and 2 Cozee Pads (Lomir Biomedical, Inc, Malone, New York), which served the dual purpose of serving as a flat platform for the animal’s feet over the cage floor and/or as enrichment for each rodent. All animals were provided with Lab Diet Certified Rodent Diet 5002M. Each rat was individually identified by an ear tag.
Rabbits
The 4 rabbit studies were all conducted under the auspices of the US Food and Drug Administration (FDA) Good Laboratory Practice (GLP) Regulations. 4 The rabbit studies reported in this article consisted of 1 single-dose study in male and female New Zealand White (NZW) rabbits and three 7-day repeat dose intramuscular toxicity studies in male and female NZW rabbits with recovery. Where possible, information related to Materials and Methods is combined as relevant for all the 4 studies, while information specifically related to a particular study are reported as such. Results and specific data generated from each individual study are reported separately.
The NZW rabbits were supplied from Myrtle Rabbitry Inc (Thompson Station, Tennessee). The animals were 13 to 15 weeks of age and weighed 2.2 to 3.0 kg at study start (day 10). The animals were transitioned to a certified rabbit diet (Harlan Teklad Certified Global Diet 2031C) during the quarantine/pretest period and fed once daily according to the testing facility standard operating procedure (SOP). Enrichment feed stuffs, such as fruits or vegetables, were provided according to the testing facility SOPs. Animals had ad libitum access to freshwater from the Columbus municipal water supply using an automatic watering system except when the animal was removed from its cage. Each animal was identified by ear tattoo and cage card.
Nonhuman primates
Rhesus monkeys (6 macaques/sex/group) were supplied from Primate Products Inc (Miami, Florida). All animals were approximately 2 years of age and weighed 2.6 to 4.9 kg at study start. Cages were changed and sanitized according to the testing facility SOP. The animals were provided certified feed (Lab Diet 5048 C) twice daily according to the testing facility SOP and had ad libitum access to freshwater from the Columbus municipal water supply using an automatic watering system except when the animal was removed from its cage. The animals were also supplemented with fresh fruits and fresh vegetables in accordance with testing facility SOP.
Experimental Design
Rats
Sprague-Dawley rats (20 rats/sex/group) were administered the test article by intramuscular injection once daily for 7 consecutive days at 0, 100, 200, 400, and 800 mg/kg/d. Each animal received 2 or 4 intramuscular injections (depending on the dose group) of 0.25 mL/kg/injection site each day. A Hamilton microsyringe was used to achieve accuracy of dosing. The dose sites used for injection included the quadriceps, triceps, and the semimembranosus/semitendinosus muscles. The injection sites were alternated between these sites in order to minimize the injection site irritation throughout the study. The doses were administered at approximately the same time each day. General health observations were performed on all animals at least twice daily from study days 1 to 7. Observations included general physical appearance and behavior, abnormalities in food and water consumption, appearance of urine and feces, and an open field observation (observation of gait on a table top for the assessment of lameness). Body weights and food consumption were recorded prestudy and twice weekly thereafter. Terminal body weights were taken for those animals that died or were terminated due to moribund condition.
Blood was collected for hematology and clinical chemistry analysis prior to the initiation of dosing and on the day of necropsy (study day 8 or 28) or at unscheduled termination (moribund animals). Blood was collected via the retro-orbital sinus with the animals anesthetized under CO2. A sterile evacuated tube containing EDTA anticoagulant was used to collect blood for hematology, and a tube containing no anticoagulant was used to collect blood for serum preparation. On study day 8, half of the animals from each dose group were terminated via CO2 asphyxiation and then exsanguinated via cardiac puncture for the terminal blood collection. A gross necropsy, including tissue collection/weighing, was conducted under the supervision of a board-certified veterinary pathologist.
The remaining animals underwent a 21-day recovery phase. During the recovery phase, general health observations, body weights, and food consumption were recorded. On study day 28, the remaining animals were euthanized and necropsied as described previously. Any of the animals found to be moribund at any time during the study was terminated immediately and necropsied under the supervision of a board-certified veterinary pathologist. Histopathology was completed on the organs and tissues collected from the control and high-dose groups first. The mid-dose groups were assessed only if positive findings were identified in the high-dose group until a no-observed adverse effect level (NOAEL) was defined. For the pharmacokinetic phase, Sprague-Dawley rats were administered with the test article once daily for 7 days at 0, 100, 200, 400, and 800 mg/kg/d. General health observations were recorded throughout the study. Each animal was bled twice on study day 1 and twice on study day 7 for pharmacokinetic analysis, with 3 rats/sex/dose group bled at each time point.
Rabbits
In the acute toxicology study in rabbits, there were 4 groups (1 vehicle control group and 3 MMB4 DMS groups) of the 6 NZW rabbits/sex/group. Animals received vehicle control (0.5% benzyl alcohol in water adjusted to pH 2.3 with MSA) or test article (50, 100, or 200 mg/kg MMB4 DMS) via a single set of intramuscular (thigh muscle) injections (target of 0.25 mL/kg at each of the 2 injection sites). End points used to evaluate the potential toxicity of MMB4 DMS were mortality, clinical observations, body weights and food consumption, troponin I analysis (days 2, 3, and 14), clinical and anatomic pathology including organ weights (day 3 and 14), gross examination, and histopathology (days 3 and 14).
In each of the 3 repeat dose rabbit studies, the animals were randomly assigned to dose groups (1 vehicle control group and multiple MMB4 DMS groups). Dose levels ranged from 3 to 800 mg/kg/d (Supplemental Tables 1-4). Study animals received vehicle control or MMB4 DMS once daily (at approximately the same time daily) via a set of intramuscular injections (target of 0.25 mL/kg at each injection site) for 7 consecutive days. Dose administration sites included thigh, gluteal, triceps, and epaxial muscles and were alternated in order to minimize injection site irritation. The injection sites were shaved prior to the start of dose administration. A staggered start was used in some studies, with males starting dose administration on one day and the females the following day, each having a respective day 1 of study. The first cohort of rabbits/sex/group was necropsied on day 8. The remaining rabbits were necropsied on day 28 or later as was the case with the extended recovery study in rabbits. End points used to evaluate the potential toxicity of MMB4 DMS were mortality, clinical observations, body weights, food consumption, troponin I analysis, and clinical and anatomic pathology including organ weights, gross examination, and histopathology.
The 7-day repeat dose rabbit study with extended recovery periods was conducted to investigate the biomarkers and the extent of recovery over time following administration of MMB4 DMS. One vehicle control group and 2 MMB4 DMS groups each had 20 animals/sex. Animals in those groups received vehicle control (0.5% benzyl alcohol in water for injection) or test article (100 or 200 mg/kg MMB4 DMS) via a set of intramuscular injections (target of 0.25 mL/kg at each of 2 injection sites or a total of 0.5 mL/kg/d) once daily for 7 consecutive days. In general, in the repeat dose toxicology studies in rabbits, the end points used to evaluate the potential toxicity of MMB4 DMS were mortality (daily), clinical observations (daily), body weights (twice weekly), food consumption (twice weekly), troponin I and T analysis (days 2, 8, 60, and 91), urine N-acetyl-β-
All scheduled necropsies were conducted under the supervision of a board-certified veterinary pathologist. Each necropsy included examination of the external surface of the body and all orifices; the cranial, thoracic, abdominal, and pelvic cavities and their contents; and collection of tissues. Designated tissues were collected and processed from all animals and were placed in 10% neutral-buffered formalin (NBF), with the exceptions of testes, which were preserved in Bouin fixative and subsequently transferred to 70% ethanol; eyes with optic nerve, which were fixed in Davidson fixative, were subsequently transferred to 10% NBF. Protocol-specified tissues were weighed. All fixed tissues were processed to slides and stained with hematoxylin and eosin for histopathologic examination by a board-certified veterinary pathologist. Target organs identified in high-dose animals were examined from the next lower dose level in order to identify a no effect level. An internal peer review was performed.
Nonhuman primates
Six macaques/sex/group were IM administered with vehicle control or MMB4 DMS at 1 of the 4 dose levels (150, 300, 450, and 600 mg/kg/d) daily for 7 consecutive days at a total target dosing volume of 1 mL/kg (0.5 mL/kg for each injection). Dose volumes were based on individual body weights determined on day 1. In order to minimize injection site irritation throughout the study, daily intramuscular dose sites were alternated between limbs and included injections in the thigh and upper arm musculature. The order of daily (single site) dose administrations was as follows: left thigh, right thigh, left upper arm, right upper arm, left thigh, right thigh, and left upper arm. Four monkeys/sex/group were terminated on day 8, and the remaining 2 monkeys/sex/group were designated recpvery animals and were terminated on day 60.
Observations for mortality and moribundity were recorded twice daily, once in the morning and once in the afternoon. Cageside clinical observations for morbidity were conducted on all the study animals at least twice daily (once before dose administration and at a target time of 1 hour following each dose administration), at least once on the day of scheduled necropsy, and at least once daily during recovery. Skin irritation (modified Draize scale) was monitored at the most recent injection site for erythema and edema at 1 hour following dose administration, and any injection site areas previously used for dose administration were observed daily during the dose administration period.
Individual body weights were recorded from all the study animals on days 1 and 7 as well as weekly during recovery. Individual animal food consumption (semiquantitative) was recorded at least once daily on the days of dose administration and once weekly during recovery. Clinical pathology (hematology, coagulation, serum chemistry, and urinalysis assessments) was performed on all the animals during pretest and on all the study animals prior to necropsy on days 8 and 60 following an overnight fast. Blood (target of 6 mL/animal) was collected from a femoral vein or other suitable peripheral vein. The tubes for hematology contained EDTA as an anticoagulant, for coagulation parameters contained sodium citrate as the anticoagulant, and for serum chemistry determinations did not contain anticoagulant but contained serum separator gel.
Urine for routine urinalysis was collected overnight (at least 16 hours) prior to blood collection from all animals during pretest and on all study animals prior to necropsy on days 8 and 60. Additional blood specimens were collected to analyze the biomarkers of skeletal muscle, renal, and cardiac damage from all the animals during pretest and for study animals on days 1, 3, and 5 (6 hours postdose administration on each day) and prior to necropsy on days 8 and 60. Biomarkers consisted of creatine kinase (CK), creatinine, and urea nitrogen analysis (in a standard battery of serum chemistries). One collected blood specimen was allowed to clot and processed to serum, which occurred generally within 60 minutes after collection. The specimen was separated by centrifugation for at least 10 minutes using a refrigerated centrifuge set to maintain 5°C and set at a relative centrifugal force of 1300
The specimens were transferred to the Battelle Eastern Science Technology (BEST) Center (Aberdeen, Maryland) and processed for myoglobin, troponin I, and troponin T analysis. An aliquot (target 5 mL) of urine from each monkey was transferred to the BEST Center for NAG analysis. On day 8 (the day following the final dose administration), 4 animals/sex/group were weighed and humanely terminated via administration of barbiturate overdose followed by exsanguination, and a complete necropsy was performed. On day 60, the remaining 2 animals/sex/group were weighed and humanely terminated via administration of barbiturate overdose followed by exsanguination and a complete gross examination, organ weights, and tissue collection. Histological examination (slides stained with hematoxylin and eosin) was conducted on control- and high-dose animals as well as target organs from all groups (injection sites area, heart, kidneys, and lungs) that were identified in the rabbit during previous studies.
Statistical Analysis
Rats
Dose groups were compared to the control group for the following types of measurements: body weight, feed consumption, selected clinical observations related to “lameness,” labored breathing, porphyrin or urine stain, rough hair coat, lethargy, scab/sore, head tilt, thinness, and/or tremors. Analysis of variance (ANOVA) was used for overall dose effect on each continuous measurement. If a significant overall effect was found, pairwise comparisons of each dose to control were performed. For repeated measurements over time, an additional covariance structure in the ANOVA was included. Logistic regression was used to model the rate of adverse effects and other presence/absence types of measurements based on the dose level. Chi-square tests of independence in the adverse effect rate across dose levels were also performed, and relative risks of the dose levels were calculated. All statistical analyses were performed using SAS 9.1.3 (SAS Institute Inc, Cary, North Carolina).
Rabbits and nonhuman primates
All appropriate quantitative data collected at Battelle were analyzed statistically when n > 2. All data were analyzed for test article effects by ANOVA. For data in which variances were considered homogeneous across the test groups, as determined by Bartlett test for homogeneity at the 0.05 level, tests for differences between the vehicle group and the comparison groups were made using Dunnett test. For nonhomogeneous data, as determined by Bartlett test for homogeneity at the 0.05 level, tests for pairwise differences between the vehicle group and each of the comparison groups were made using Cochran and Cox modified 2-sample
Results
Mortality/Clinical Observations
Table 2 summarizes the incidence of mortality across all species and dose groups, for both males and females.
Mean and Standard Deviation of Body Weight Measurements in Rats (g) by Dose, Sex, and Study Phase (Predosing and Dosing Only).
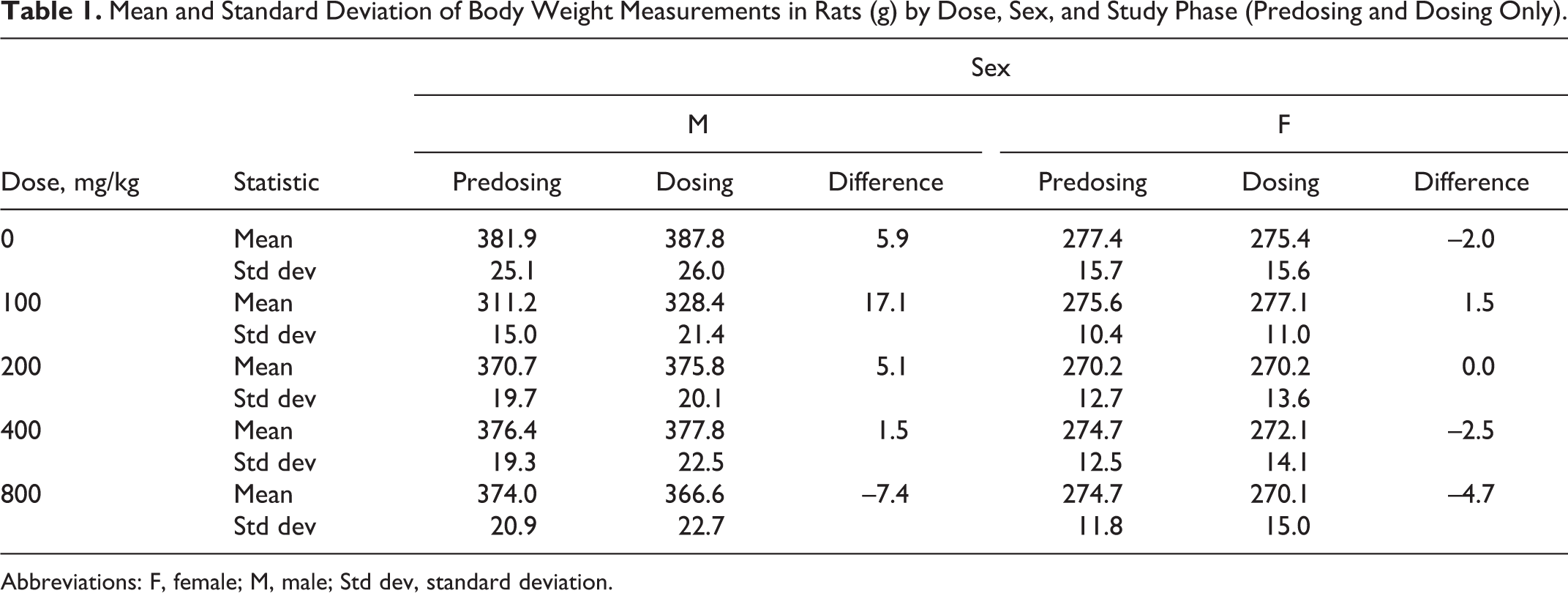
Abbreviations: F, female; M, male; Std dev, standard deviation.
Comparative Mortality Across Species by Dose Level (M; F).
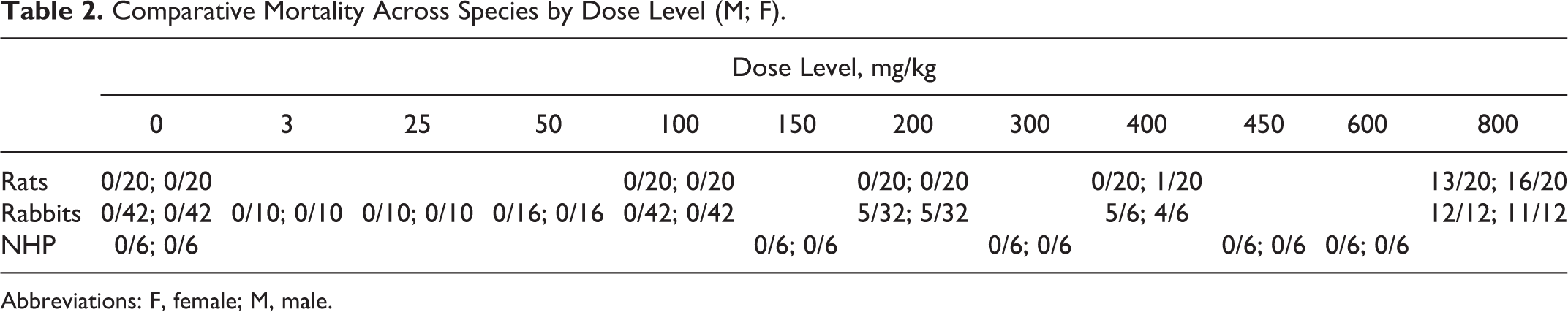
Abbreviations: F, female; M, male.
Rats
The majority of the animals in the 800 mg/kg/d dose group did not survive to either study day 8 or study day 28 due to early death or unscheduled termination (found moribund or in pain and/or suffering). Most of the unscheduled deaths in the 800 mg/kg/d dose group occurred on study day 1, the first day the rats received MMB4 DMS. The primary reason for early removal was due to moribundity, specifically labored, or difficult breathing. This observation was usually noted within approximately 4 hours or less of dose administration.
The predominant clinical observation noted in this study for the lower dose groups was “lameness,” which included holding the foot in an abnormal position (all toes curled in), splaying of the limb, high stepping, knuckling, and refusing to put weight on the limb. These clinical signs were first noted on study day 1. The observations related to lameness were most prevalent in the 2 highest dose groups (400 and 800 mg/kg/d) and showed a clear dose response, especially through the dosing phase of the study. Furthermore, the observations related to lameness resolved by about 7 days to the 21-day recovery period for most animals. Other clinical observations noted primarily during the dosing phase of the study included lethargy, head tilt, sores/self-mutilation of limbs, high stepping, labored breathing, rough hair coat, poor grooming (urine stain), alopecia (especially over the front limbs and flanks and injection sites), and tremors.
Rabbits
In the acute toxicity study in rabbits, all rabbits survived until scheduled termination except 1 male in the 200 mg/kg dose group that was found dead 4 days after dose administration (day 5). Death of this animal was ascribed to the combined renal (nephrosis of renal tubules and protein casts) and pulmonary (bronchial epithelial necrosis/restitution/proliferation and proliferative bronchiolitis) toxicity related to MMB4 DMS administration. All clinical observations in the vehicle control, 50 mg/kg, and 100 mg/kg dose groups were normal. Clinical abnormalities related to MMB4 DMS were limited to the 200 mg/kg dose group. In all, 4 rabbits were noted with orange urine; 2 rabbits were noted with abnormal posture characterized as intermittent hyperextension of the neck; 4 animals were noted with clear nasal discharge; and 1 rabbit was noted with decreased activity. All observations were noted after dose administration on day 1 only. The rabbit that was found dead on day 5 was noted with progressively worsening clinical observations ranging from soft feces, clear eye discharge, decreased activity, and finally reduced feces, labored respiration, and lethargy before death.
In the 7-day repeat dose rabbit studies, all rabbits administered the vehicle control and the 100 mg/kg/d MMB4 DMS survived until scheduled termination of the study. Mortality at dose levels ≥200 mg/kg/d displayed a dose-related incidence but without apparent gender effects. Clinical observations of decreased activity, abnormal posture characterized as an intermittent hyperextension of the neck, decreased activity/lethargy, and orange urine were noted (in different frequencies) in the 200, 400, and 800 mg/kg/d dose groups and were attributable to test article administration. Abnormal clinical observations attributable to test article administration in the 50 and 100 mg/kg/d dose groups were limited to orange urine in a few animals. There were no clinical observations noted in the vehicle control group. With the exception of a few adverse observations at the start of recovery, all recovery animals were normal.
Nonhuman primates
All animals survived to the scheduled necropsy on days 8 and 60. There were no adverse clinical observations attributed to vehicle control administration during the 7-day dosing period. Intermittent decreased activity after each dose administration was the most common abnormal observation noted in 1 female administered 300 mg/kg/d MMB4 DMS, and in 5 of 6 males and 6 of 6 females in both 450 and 600 mg/kg/d MMB4 DMS dose groups. Generally, the decreased activity was observed approximately 1 hour after dose administration with normal activity resuming within 4 hours after dose administration. All animals were normal the following day before the next dose administration. Based on the transient nature of this finding and rapid recovery with no apparent lingering effects, the decreased activity was not considered an adverse finding.
The vehicle control group had a few grade 1 observations of edema/erythema noted at the area of injection. In contrast, there was a higher incidence and severity of erythema and edema noted at the area of MMB4 DMS injection at all dose levels. The highest incidences noted in the treated animals were grades 1 and 2 without an obvious dose or gender effect. In addition, edema was more commonly observed than erythema. Most injection site swellings were intermittent and decreased in size the day following injection.
Body Weight and Food Consumption
Rats
There was a decrease in body weight gain for male rats in a dose-dependent manner at the 400 and 800 mg/kg/d doses when compared to control and 200 mg/kg/d animals (Table 1). Female rats at 400 and 800 mg/kg/d had larger differences, respectively, and decreased body weight gain when compared to 0, 100, or 200 mg/kg/d females. Due to the limited availability of male rats for the 100 mg/kg/d add-on group from the vendor at the time of purchase and the timeline of the study, these rats were slightly younger than those for all of the other dose groups and, therefore, had a lower body weight at the beginning of the study. The increase in body weight observed in the 100 mg/kg/d males was attributed to the rapid growth rate of younger animals procured separately from the other groups and are displayed for reporting purposes only.
There was a statistically significant decrease (
Mean and Standard Deviation of Food Consumption Measurements (g) in Rats by Dose, Sex, and Study Phase.
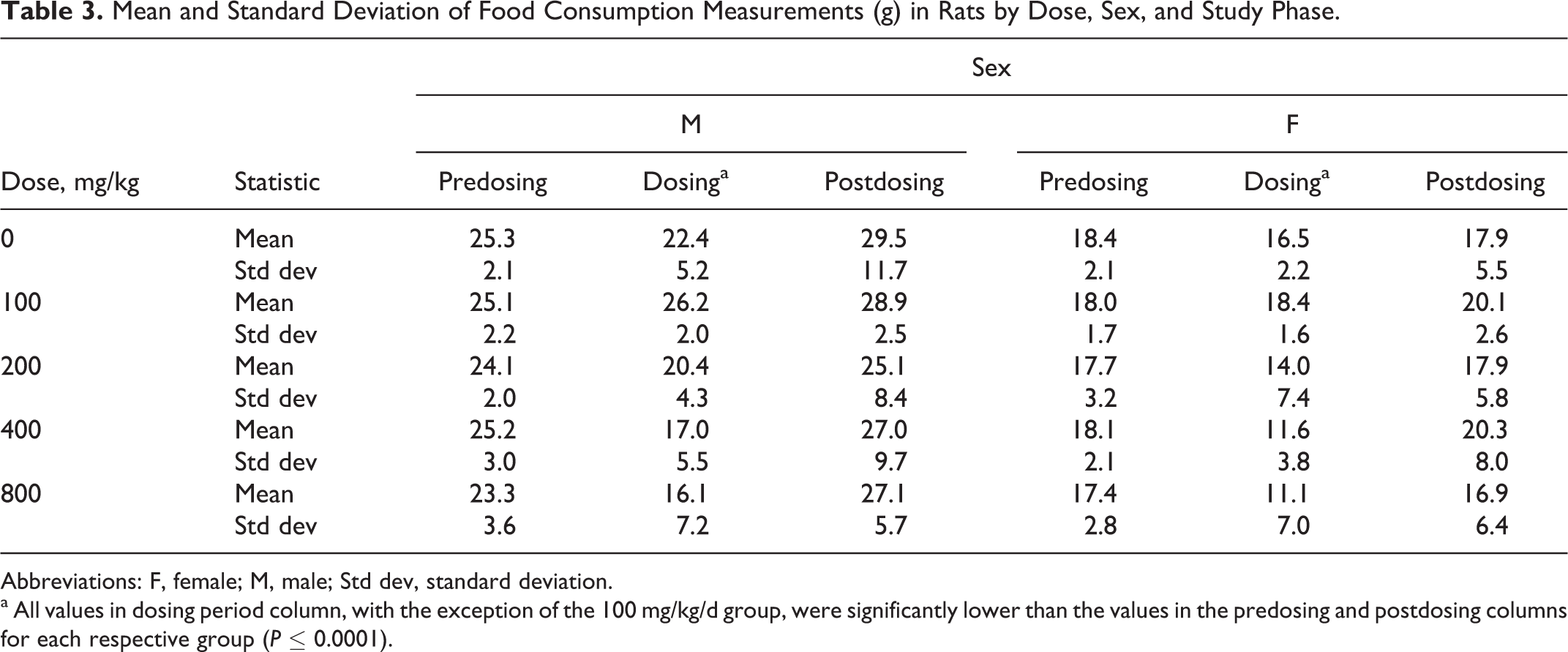
Abbreviations: F, female; M, male; Std dev, standard deviation.
a All values in dosing period column, with the exception of the 100 mg/kg/d group, were significantly lower than the values in the predosing and postdosing columns for each respective group (
Rabbits
In general, there were no significant differences in body weights or food consumption values attributable to the administration of MMB4 DMS at up to 100 mg/kg/d; however, minor body weight loss and decreases in food consumption were noted during a few intervals in the 200 and 400 mg/kg dose group animals. All abnormal clinical observations, body weight losses, and food consumption decreases resolved before the conclusion of the recovery period.
Nonhuman Primates
There were no statistically significant differences in body weight or notable differences in food consumption measurements for either group. All animals maintained their body weight during the course of the study.
Clinical Pathology Evaluations
Rats
On study day 8, there were a few minor clinical pathology abnormalities found. The 400 mg/kg/d females had an elevated mean aspartate aminotransferase (AST) value; however, the standard deviation for this group was very high (358.2), and many of the samples for this group were hemolyzed, which can falsely elevate AST.
Unscheduled deaths in the 800 mg/kg/d dose group revealed a number of clinical pathology differences; however, none were suggestive of cause of death. There were slight elevations in both AST and CK, elevated glucose associated with stress and moribundity, and some animals had a slightly elevated blood urea nitrogen (BUN) and hematocrit due to dehydration. In addition, many of the animals had a decreased total protein and albumin, as well as decreased sodium values, and some had slightly decreased chloride values.
Rabbits
In the acute toxicity study, elevated troponin I levels, a marker for cardiac muscle damage, were detected in the 200 mg/kg dose group only, which is consistent with the absence of cardiac pathology findings in the lower dose groups. Four of the 6 animals on day 2 and 1 of the 3 animals on day 3 had elevated levels of troponin I when compared to the prestudy levels. No increases were detected on day 14. There were no hematology results that indicated effects due to MMB4 DMS or vehicle control. Clinical chemistry evaluations revealed increased BUN, creatinine, and AST values as well as increased globulin, triglycerides, and potassium and decreased albumin/globulin ratio in 1 male rabbit in the 200 mg/kg MMB4 DMS group on day 3. This animal was necropsied as scheduled on day 3, and its microscopic findings included moderate MMB4 DMS-induced nephrosis and protein casts in the kidneys, which was consistent with the increased BUN and creatinine values noted. Moderate necrotizing inflammation at the injection sites, minimal necrotizing inflammation of the myocardium, and mild hepatic necrosis were also found microscopically in this animal, all of which could account for the increased AST. Elevated CK levels were noted in all the dose groups on day 3, when compared with prestudy results (Figure 1). The elevations were greater in the 100 and 200 mg/kg dose groups than the 50 mg/kg/d and vehicle control groups; the effects were reversed by day 14. The CK is a marker of striated muscle (skeletal or cardiac) damage. Due to the extent of injection site muscle inflammation, it was concluded that the enzyme elevations were principally sequelae of the local injection site reaction to both the vehicle and MMB4 DMS.
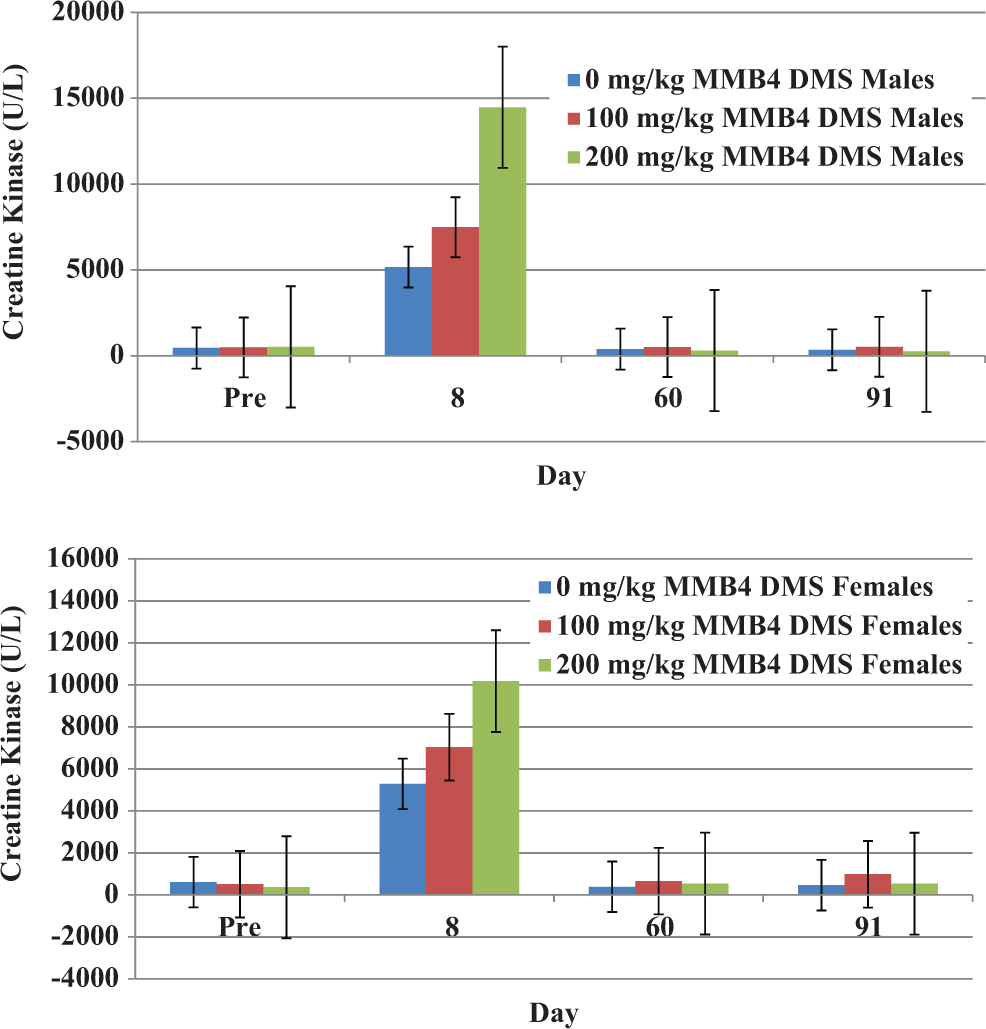
Mean male and female creatine kinase levels (U/L) over time in rabbits.
In the repeat dose rabbit studies, periodic elevations in platelet and/or reticulocyte values (with concomitant minimal decreases in red blood cell mass) were noted in all the dose groups ≥200 mg/kg/d on day 8, when compared to the respective group prestudy values and were interpreted to be due to irritation at the injection sites. In addition, slight decreases in white blood cell/differentials in the 200 and/or 400 mg/kg/d groups were interpreted to be due to MMB4 DMS administration. By day 28, hematology parameters were similar to prestudy results, indicating complete recovery. The CK levels were elevated in all the dose groups on day 8 in comparison to prestudy values; the elevations of CK values were slightly higher in the 50 and 100 mg/kg/d groups than in the lower dose-treated groups and vehicle control groups. By day 28, the CK results of all the groups were lower than that had been found in prestudy sampling.
Compared to vehicle control values on day 8, decreased total protein, albumin, and/or albumin–globulin ratios were related to protein loss from MMB4 DMS-induced nephrosis as well as due to loss from the injection site and related body weight decreases in the ≥200 mg/kg/d groups. At day 28, the results for clinical chemistry parameters of the surviving-treated groups were similar to those of the vehicle rabbits, indicating reversibility of clinical chemistry effects. Exceptions were the significantly elevated triglycerides in the 400 mg/kg/d males.
There were no changes in NAG values that would indicate renal dysfunction in treated animals. Troponin I was generally below measurable limits with the exception of day 2, when 15 of 39 animals in the 200 mg/kg/d group had measureable troponin I levels. By day 8 and thereafter, measureable troponin I was found in 1 of 84 animals. Troponin T was measurable in 6 of the 40 vehicle control animals and all MMB4 DMS animals (except 1) by day 2. By day 8, all vehicle control and MMB4 DMS animals had measureable troponin T levels. By day 60 and thereafter, measurable troponin T was found in 3 of the 54 animals.
Nonhuman primates
Serum myoglobin levels on days 1, 3, and 5 were found to be consistently elevated in comparison to pretest myoglobin results (Figure 2). However, the levels did not escalate on day 8 even in the presence of continued daily intramuscular administrations through day 7. The 600 mg/kg/d MMB4 DMS group had consistently higher myoglobin levels compared to the lower dosage groups, which was consistent with the greater severity of local inflammation described histologically.
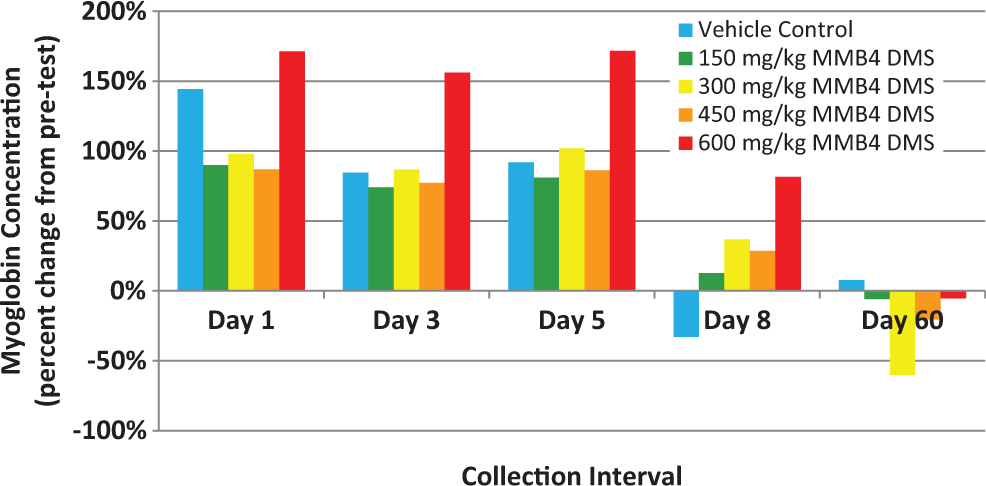
Myoglobin concentration (ng/mL) over time in rhesus monkeys.
On day 1, there were small but detectable troponin I and T levels (Figures 3 and 4), but there was no dose response in either parameter. Subsequent time points evaluated (days 3, 5, and 8) demonstrated troponin I and T values that were comparable to pretreatment values in spite of continued dose administrations of vehicle and MMB4 DMS. Histological examination of the hearts on day 8 from all the study animals was within normal limits.
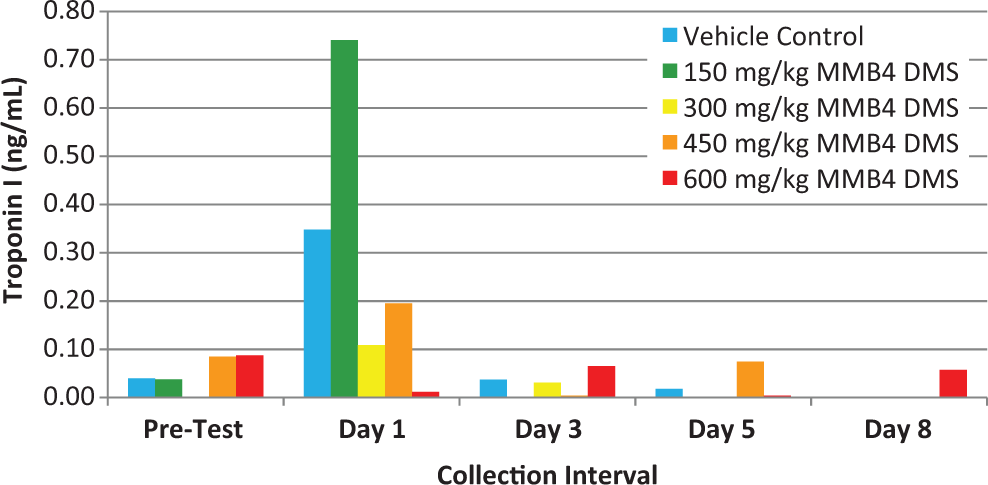
Troponin I (ng/mL) over time in rhesus monkeys.
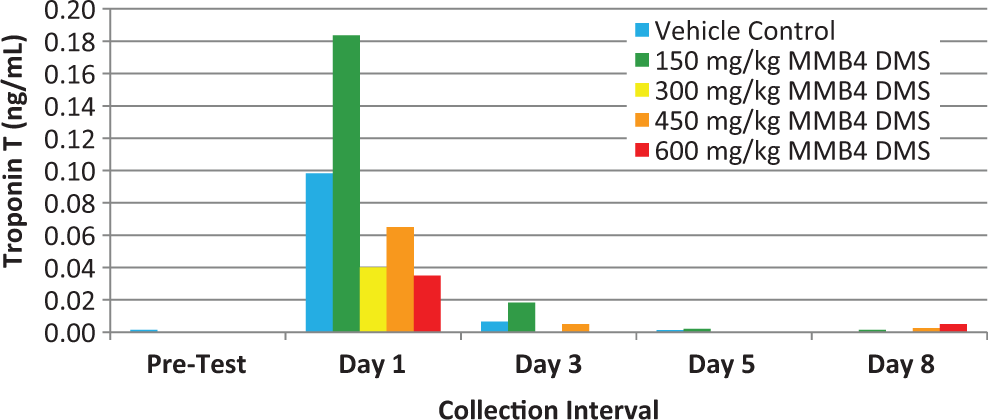
Troponin T (ng/mL) over time in rhesus monkeys.
Urine NAG (Figure 5) was elevated on day 8 compared to pretest in 2 of the 4 groups; however, there was no dose response. The difference in the day 8 NAG values when compared to pretreatment values was likely a reflection of biological variability. Histological examination of the kidneys on day 8 from all study animals was within normal limits.
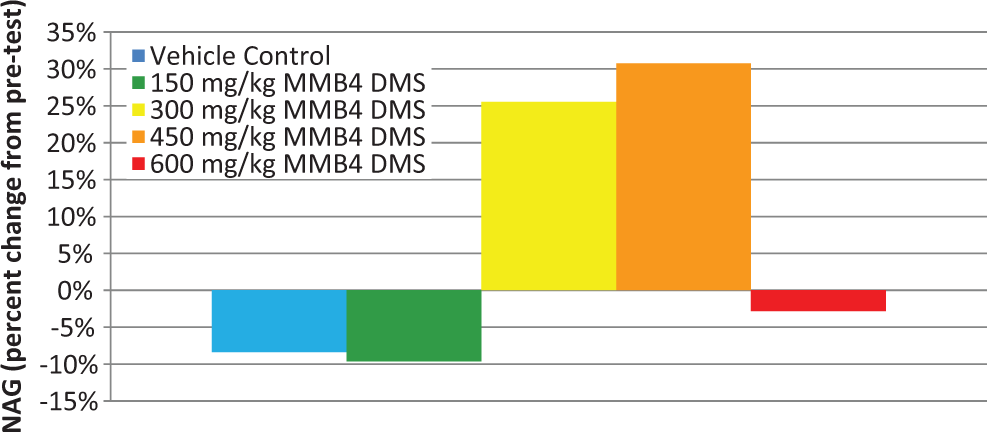
N-acetyl-β-
Each study animal, including vehicle controls, had marked increases (2- to 67-fold) in CK (Figure 6; Table 4) activity following initiation of dose administration; however, similar to the temporally decreasing trend noted in myoglobin values, the CK also displayed a decrease over time, even though animals continued with daily MMB4 DMS dose administrations through day 7. The peak values and fold increase values are presented in Table 4. The increased CK was interpreted to be due to the skeletal muscle damage from intramuscular injections of MMB4 DMS, including effects of the vehicle. There was no notable change in BUN during the dose administration period compared to pretest.
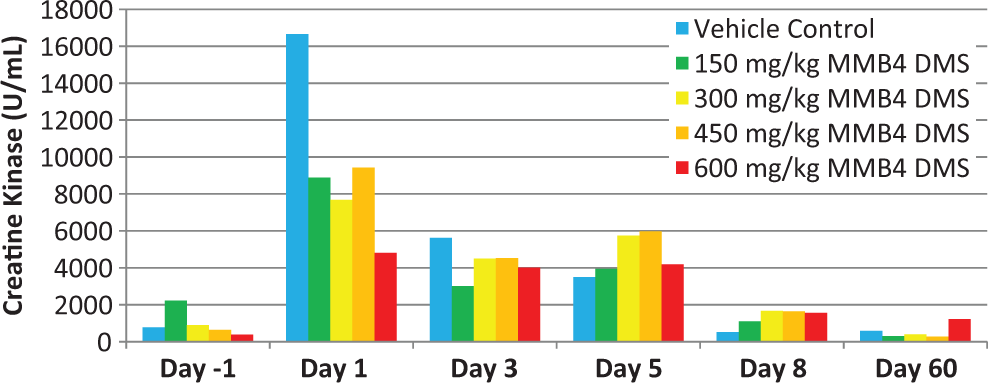
Creatine kinase (mg/dL) over time in rhesus monkeys.
Peak CK Values and Fold Increase Values in Rhesus Monkeys.
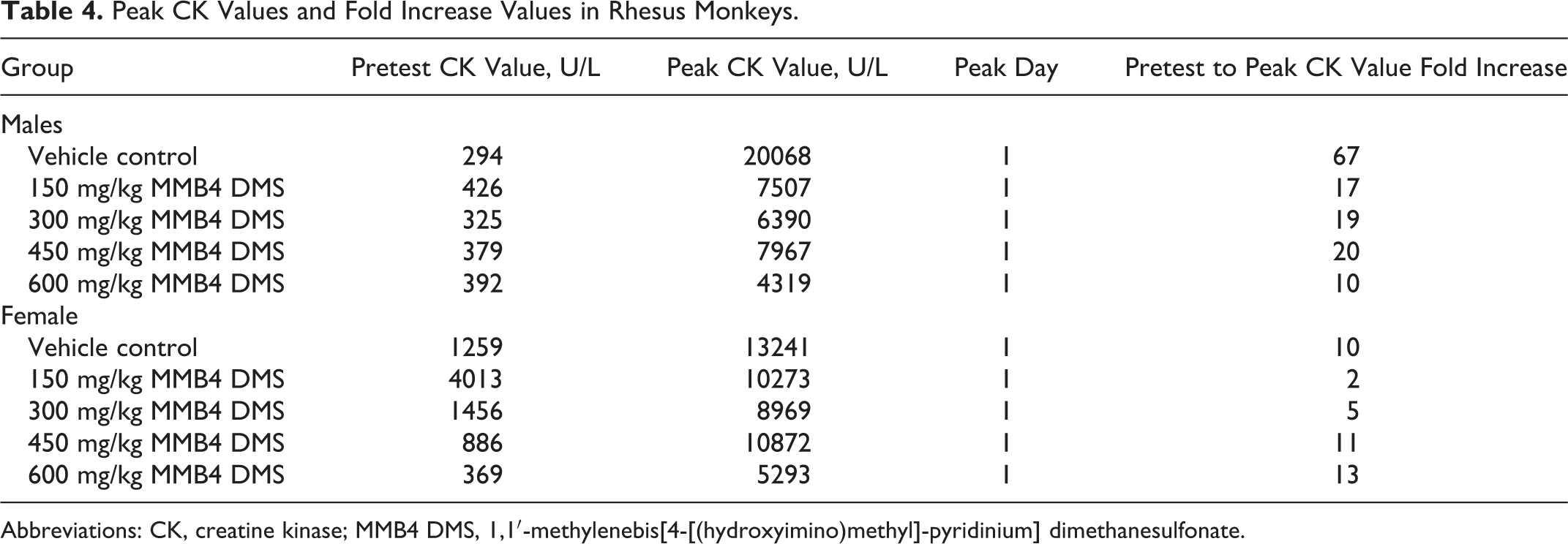
Abbreviations: CK, creatine kinase; MMB4 DMS, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate.
On day 8, increased platelet values as well as increased mean activated partial thromboplastin time, prothrombin time, and/or fibrinogen concentrations, compared with prestudy results, were seen in vehicle control and treated groups. The means of these 3 parameters were frequently increased to a greater extent in MMB4 DMS groups on day 8 than were the vehicle control values. Increases in these parameters are often noted during an acute inflammatory phase as was reflected by the local reaction to the vehicle and MMB4 intramuscular injections. The results of day 60 indicated recovery from the effects that had been noted on day 8.
On day 8, increased alanine aminotransferase (ALT) and AST results, compared with same-group prestudy results, were noted in all MMB4 DMS groups and vehicle controls of both sexes. The increases in ALT and AST were interpreted to be due to skeletal muscle damage from intramuscular injections of MMB4 DMS, including possible effects of the vehicle. No apparent treatment-related histological changes were noted in the liver. The decreased albumin, total protein, albumin–globulin ratio, cholesterol, and inorganic phosphorus found on day 8 in vehicle and treated groups were not related to skeletal muscle damage but were of minimal clinical relevance, based on their small size and absence of abnormal clinical findings.
The results of day 60 indicated recovery from the effects that had been noted on days 1, 3, 5, and 8.
Organ Weight Evaluations
Rats
No apparent differences.
Rabbits
On study day 8, the absolute liver weights of 200 and 400 mg/kg/d males and females and the single surviving 800 mg/kg/d female were increased, when compared to those of the vehicle rabbits; these increases were statistically significant in the 200 mg/kg/d males and 400 mg/kg/d rabbits of both sexes. Also on day 8, the absolute thymus weights in the 200 mg/kg/d males, the 400 mg/kg/d rabbits of both sexes, and the single surviving 800 mg/kg/d rabbit were decreased when compared to those of the vehicle rabbits; these decreases were statistically significant in the 200 and 400 mg/kg/d males.
Nonhuman primates
Mean absolute thymus weights of the 300 and 600 mg/kg/d male and 150, 450, and 600 mg/kg/d females were significantly decreased from comparable control groups. The remaining test article groups were also decreased compared to controls, although the difference did not reach statistical significance. These decreases were correlated with the thymic atrophy noted microscopically in the 300 to 600 mg/kg/d males and females. Similar decreases in mean thymus to terminal body weight and thymus to brain weight values were present in treated animals necropsied on day 8. The differences in terminal thymus weight, thymus to terminal body weight, and thymus to brain weight were no longer present in any of the day 60 animals.
Macroscopic Findings
Rats
Test article-related gross pathology findings were observed at the injection sites at all dose levels, consisting of various muscle discolorations that correlated microscopically with test article-related findings of myofiber degeneration, necrosis, regeneration, hemorrhage, fibrosis, mononuclear cell infiltrations, myofiber mineralization, and pigmentation.
Test article-related gross pathology findings observed at terminal euthanasia (muscle discoloration that correlated microscopically with test article-related findings) were not observed at the end of the recovery period (day 28). All other gross findings observed were considered incidental, of the nature commonly observed in the age and strain of the test species, and/or were of similar incidence in control and treated animals and were therefore considered unrelated to administration of the test article.
Rabbits
Gross observations due to MMB4 DMS were limited to the injection sites. Gross findings at necropsy included red or dark discoloration of the injection sites interpreted to indicate injection trauma and/or irritation of the muscle. These were found in vehicle and MMB4 DMS-treated rabbits at the end-of-dosing necropsy; based on the higher incidence rates of findings in MMB4 DMS-treated rabbits at the injection sites when compared with vehicle rabbits, gross pathology results indicated that MMB4 DMS was more irritating than the vehicle.
Gross necropsy findings related to MMB4 DMS dose administration were present at the sites of intramuscular injection in animals that died or were terminated due to moribund condition and in the animals sent for necropsy on day 8. Generally, the lesions were described as red discolorations; they corresponded microscopically to skeletal muscle necrosis and displayed an increased incidence for animals treated with MMB4 DMS when compared to those administered vehicle control alone.
Nonhuman primates
Gross necropsy findings related to MMB4 DMS administration were present at the areas of intramuscular injection in at least 1 animal terminated on study day 8 from each of the treated groups. The lesions in these animals were described as red, dark, mottled, or tan discolorations at the injection sites. These gross findings corresponded microscopically to acute skeletal muscle necrosis. There were no gross findings related to MMB4 DMS administration in the animals evaluated from day 60.
Histopathology
Rats
Test article-related histopathology findings were limited to the muscle injection sites from males and females in the 100, 200, 400, and 800 mg/kg/d dose groups. Primary injection site alterations due to test article administration were comprised of myofiber degeneration/necrosis (variably sized, often enlarged, hyalinized [pink] myofibers that were often void of nuclei) and myofiber regeneration (small, hyalinized [pink], or basophilic [purple] myofibers with centrally located nuclei that were characteristic of the regenerative or reparative process).
These 2 alterations were considered to be the primary muscle alterations due to test article administration, which in turn led to secondary processes of inflammation (mononuclear cell infiltration), fibrosis, hemorrhage, mineralization, and pigmentation. The characterization of myofiber degeneration/necrosis (more acute) versus myofiber regeneration (more chronic) was consistent with the time-related nature of the irritating event.
Nearly, all of the primary muscle alterations in the recovery animals were regenerative in nature, minimal-to-mild in severity, and lower in incidence compared to the terminal euthanasia cases. There were no primary muscle alterations in the control recovery-euthanasia animals, and only 1 case of primary muscle alteration in a 100-mg/kg/d male, indicating nearly complete recovery at this dose level.
Rabbits
In the acute toxicity study in rabbits, MMB4 DMS-related cardiac, renal, and pulmonary findings were noted in several rabbits in the 100 and/or 200 mg/kg/d dose groups. Cardiac changes consisted of minimal myocardial necrotizing inflammation (Supplementary Table 5) and occurred on day 3 necropsy in one of the three 200 mg/kg/d males, and in two of the three 200 mg/kg/d females, and a finding coded as mild in one of the two 200 mg/kg/d males from the day 14 necropsy.
Renal changes (nephrosis and protein casts) were noted in one 200 mg/kg/d male on the day 3 necropsy (Supplementary Table 6). Nephrosis of renal tubules and protein casts were also noted in a male animal in the 200 mg/kg/d group and were believed to be associated with its early death. In terminal necropsy rabbits, nephrosis with protein casts was noted in one 200 mg/kg/d male, and mild nephrosis without protein casts was noted in the other 200 mg/kg/d male and one of the three 200 mg/kg/d females. A similar morphologic finding (minimal renal tubular nephrosis) was noted microscopically in a day-14 necropsy vehicle control female; this finding in this female was interpreted to be an incidental degenerative change.
The MMB4 DMS-related pulmonary findings consisted of bronchial epithelial necrosis, bronchial epithelial restitution/proliferation, and proliferative bronchiolitis noted at ≥100 mg/kg/d (Table 5). This series of morphological changes noted in the lungs represents a temporal change beginning with the initial damage followed rapidly with a repair process.
Incidence Summary of Pulmonary Findings Attributed to MMB4 DMS With Average Severity, Acute Rabbit Study.
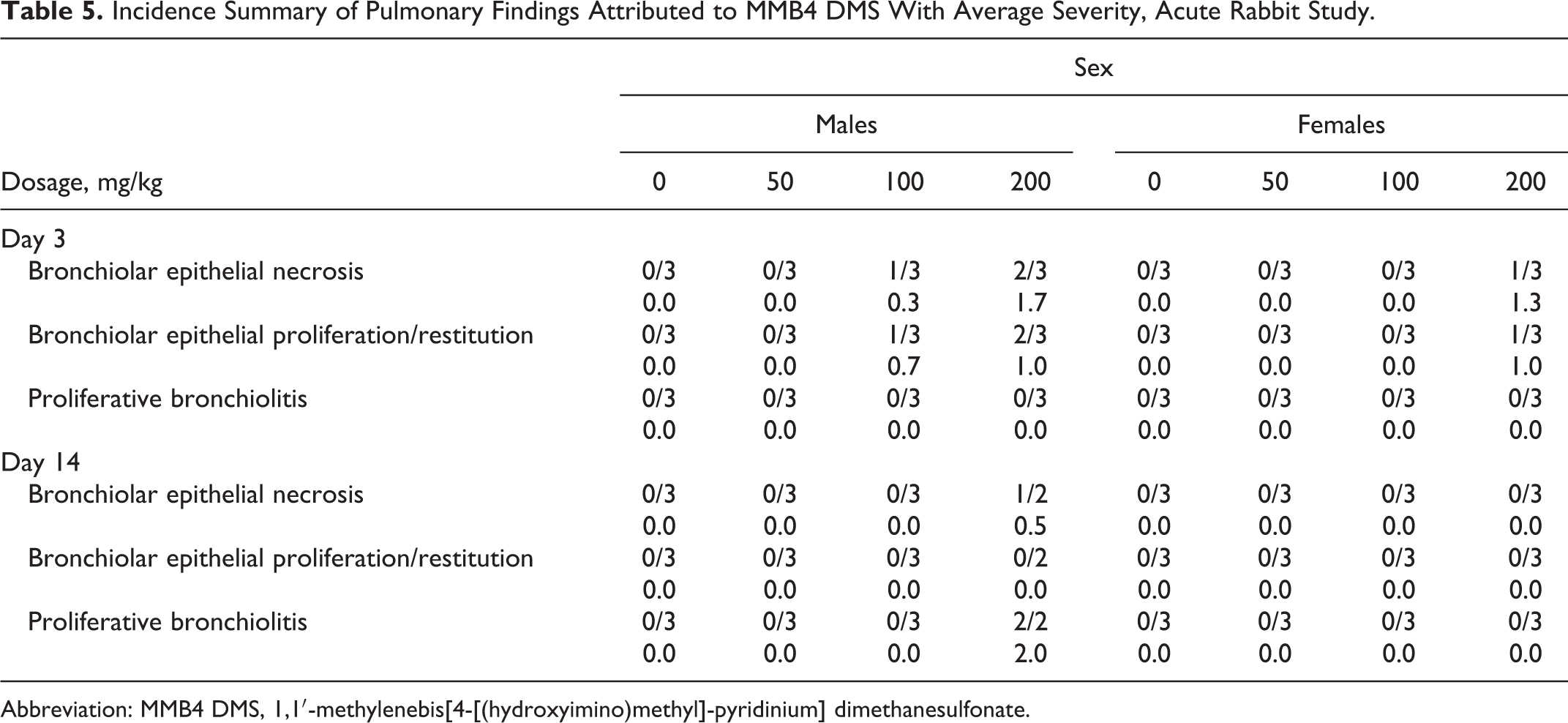
Abbreviation: MMB4 DMS, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate.
The injection sites of all rabbits on day 3 had grossly evident red or dark discoloration; some day 3 necropsy female vehicle control and 200 mg/kg/d rabbits also had yellow or tan foci of the injection site. Discoloration was only noted grossly in the 200 mg/kg/d dose group by day 14 necropsy. Microscopically, necrotizing inflammation, hemorrhage, and fibrosis were noted in the injection sites of all the rabbits; fibrosis was only noted at the day 14 necropsy (Table 6). The severity of necrotizing inflammation and hemorrhage of the injection sites was greater in the 100 and 200 mg/kg/d MMB4 DMS groups than vehicle control groups, which was frequently, but not always, increased over the vehicle control rabbits in the 50 mg/kg/d groups. The severity decreased at the day 14 necropsy compared to day 3 in all the groups except in the 200 mg/kg/d group.
Incidence Summary of Microscopic Findings Attributed to MMB4 DMS in the Injection Site with Average Severity, Rabbits.
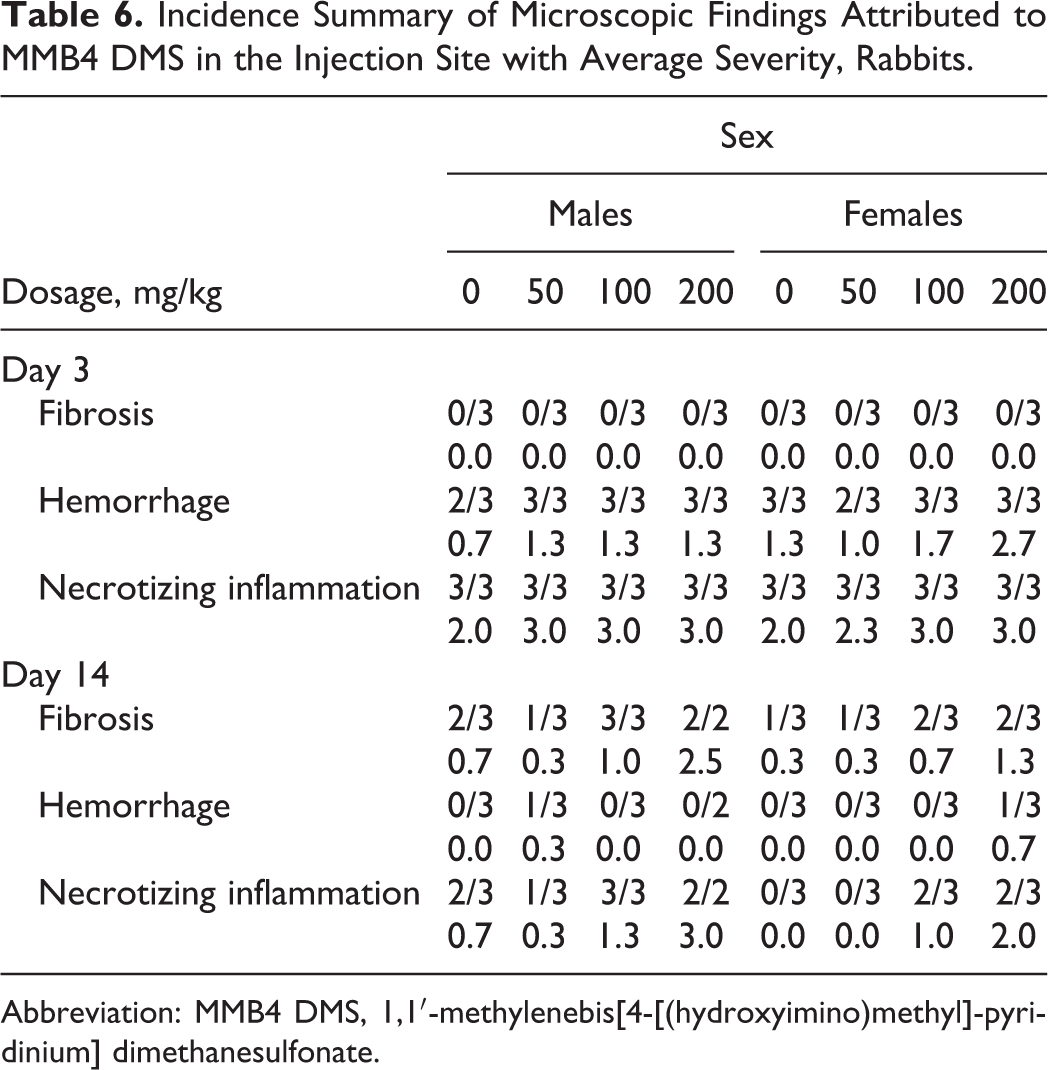
Abbreviation: MMB4 DMS, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate.
In the repeat dose rabbit studies, microscopic changes were noted in the heart and kidneys (≥200 mg/kg/d), lungs (≥100 mg/kg/d), and at the injection sites (all groups including vehicle control animals). At the administration sites, necrotizing inflammation, hemorrhage, and fibrosis were noted at the day 8 necropsy in the vehicle control and MMB4 DMS rabbits, with marginally greater severity noted in the ≥50 mg/kg/d/d rabbits. Elevated CK levels on day 8 correlated with macroscopic and microscopic evidence of vehicle control/test article effects noted at the injection sites of all rabbits and were interpreted to be due to muscle damage at the injection sites. The greater sized increases in the 50 and 100 mg/kg/d males and females indicated that a greater level of muscular damage occurred in the injection sites of these groups than that was found at lower MMB4 DMS doses and in vehicle controls. By day 28, hemorrhage was not noted in the injection sites and only a few indications of muscle cell damage and fibrosis were noted microscopically. Thus, recovery from local irritating effects of the injections was well underway.
Microscopic findings due to MMB4 DMS administration in unscheduled-death rabbits and end-of-dosing necropsy rabbits included necrotizing inflammation in the heart, tubular nephrosis and protein casts in the kidneys, and necrotizing bronchiolitis, bronchiolar epithelial restitution/proliferation, and proliferative bronchiolitis in the lungs, present at various dose levels ≥100 mg/kg/d/d. Serum biomarkers for potential renal injury were noted in 1 animal that died early which had elevated serum BUN and creatinine values on day 2 that was consistent with the increased severity of microscopic nephrosis. Necrotizing myocarditis and nephrosis with protein casts were found in the 200 mg/kg/d animals terminated at day 8. However, the average severity of the finding was less when compared to those animals that died at an unscheduled interval or were terminated due to moribund condition.
Altogether, the results of the 4 rabbit studies suggest that the NOAEL for lung histopathologic findings in rabbits after intramuscular injection of MMB4 DMS was 50 mg/kg/d.
In the extended recovery periods in the rabbit study, residual fibrosis with myocyte regeneration from the injection sites was present in a small number of 100 and 200 mg/kg/d MMB4 DMS-treated animals necropsied on day 60, but these changes had resolved in all animals examined by day 91. On days 60 and 91, platelet counts of vehicle control and MMB4 DMS groups were similar and decreased when compared with pretest results; the clinical chemistry alterations found in treated rabbits on days 2 and 8 were no longer present, indicating recovery from MMB4 DMS-related effects. The heart and kidney changes had completely resolved in animals sent for necropsy on day 60, and resolution of these lesions was subsequently confirmed upon microscopic examination of animals terminated on day 91. In addition, serum biomarkers associated with heart, kidney, and skeletal muscle damage were elevated on days 2 and 8 but were comparable to baseline values on days 60 and 91.
In summary, the nature of the histopathological lung lesions, the lack of gross observations that would be expected with diffusion impairment or obstruction of ventilation, the lack of significant lung lesions in some of the unscheduled deaths in the rabbit studies, and the lack of lung lesions in the monkey study suggest that the lung lesions were not the sole cause of death in these studies. Similarly, subject matter experts in cardiac and renal toxicology believed that the minimal-to-mild lesions in the heart and kidneys may have contributed but were not the cause of death of those rabbits that died on study.
Nonhuman primates
Acute skeletal muscle necrosis, characterized by widespread myocyte necrosis and scattered acute mixed cell inflammation and hemorrhage, was present at all injection sites in nearly all animals necropsied on day 8 from all the groups, including controls (Tables 7 and 8). The extent of the necrosis was generally restricted to the area examined microscopically, with the majority of the affected tissue present within the approximately 1 cm2 area of the section. Average weighted severity of injection site necrosis generally showed dose-dependent increases at each site. Average weighted severity was calculated as the sum of values for each finding divided by the number of animals affected.
Thymic atrophy was present in both males and females from groups 3 to 5 (300-600 mg/kg/d/d MMB4 DMS), with dose-dependent increases in incidence and average weighted severity in males and dose-dependent increases in incidence but stable average weighted severity in females (Tables 7 and 8). This finding was considered likely to be an effect of stress hormone release due to ongoing intramuscular injection with substantial local tissue damage and not a direct or immunomodulatory effect of MMB4 DMS. In animals necropsied on day 60, findings associated with recovery from the acute tissue necrosis at the injection site were still present, although the incidence and severity of these remaining changes were decreased compared to the findings present at day 8. Findings, when present, included foci of fibrosis, along with scattered areas of removal of remnant necrotic skeletal muscle fibers. There was no evidence of thymic atrophy in any of the day 60 animals (Tables 9 and 10).
Incidence Summary of Microscopic Graded Observations With Average Weighted Severity: Males, Day 8 Scheduled Necropsy, and Rhesus Monkeys.a
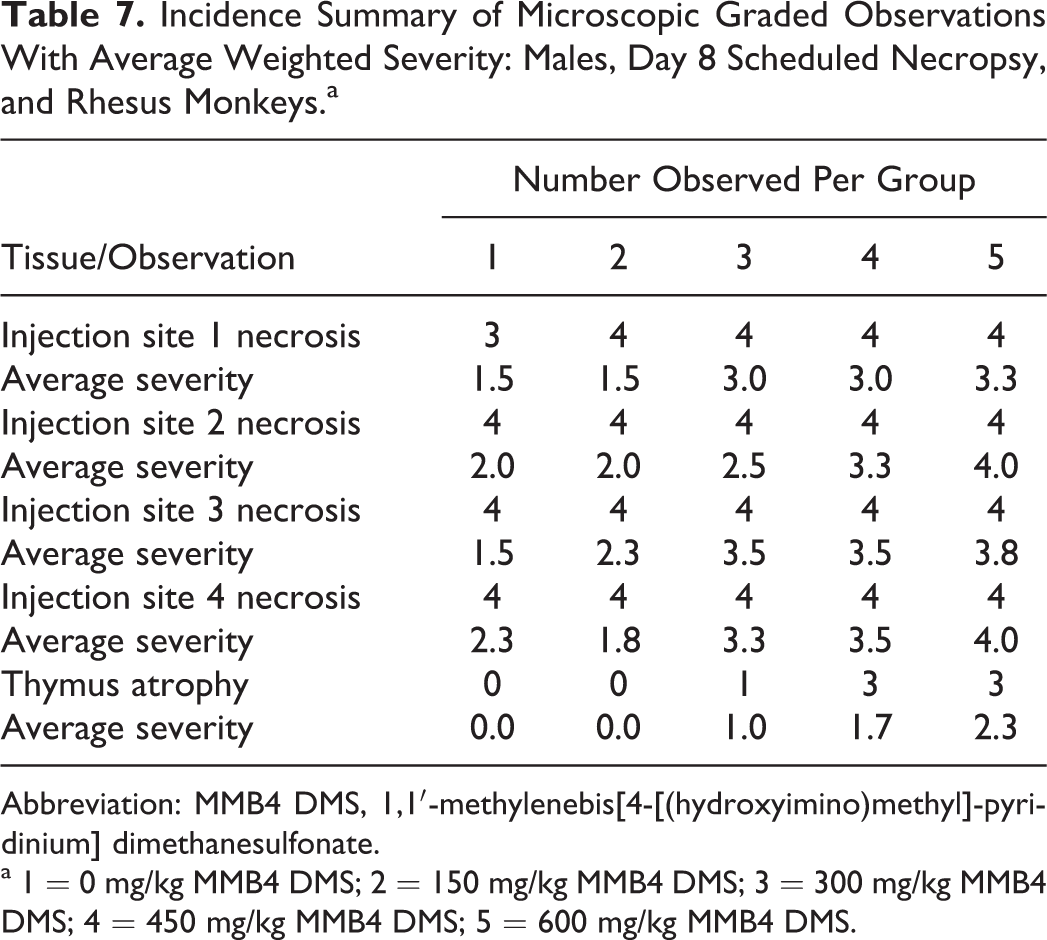
Abbreviation: MMB4 DMS, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate.
a 1 = 0 mg/kg MMB4 DMS; 2 = 150 mg/kg MMB4 DMS; 3 = 300 mg/kg MMB4 DMS; 4 = 450 mg/kg MMB4 DMS; 5 = 600 mg/kg MMB4 DMS.
Incidence Summary of Microscopic Graded Observations With Average Weighted Severity: Females, Day 8 Scheduled Necropsies, and Rhesus Monkeys.a
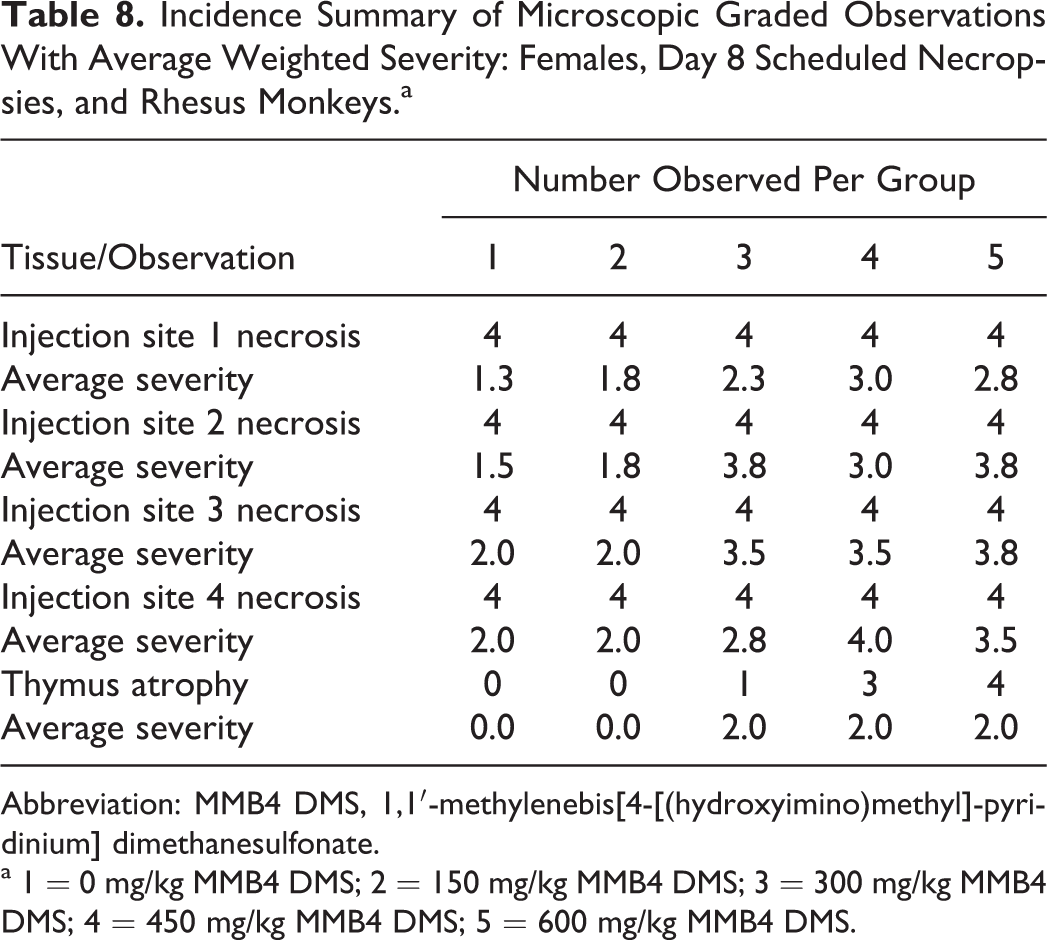
Abbreviation: MMB4 DMS, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate.
a 1 = 0 mg/kg MMB4 DMS; 2 = 150 mg/kg MMB4 DMS; 3 = 300 mg/kg MMB4 DMS; 4 = 450 mg/kg MMB4 DMS; 5 = 600 mg/kg MMB4 DMS.
Incidence Summary of Microscopic Graded Observations With Average Weighted Severity: Males, Day 60 Scheduled Necropsy, and Rhesus Monkeys.
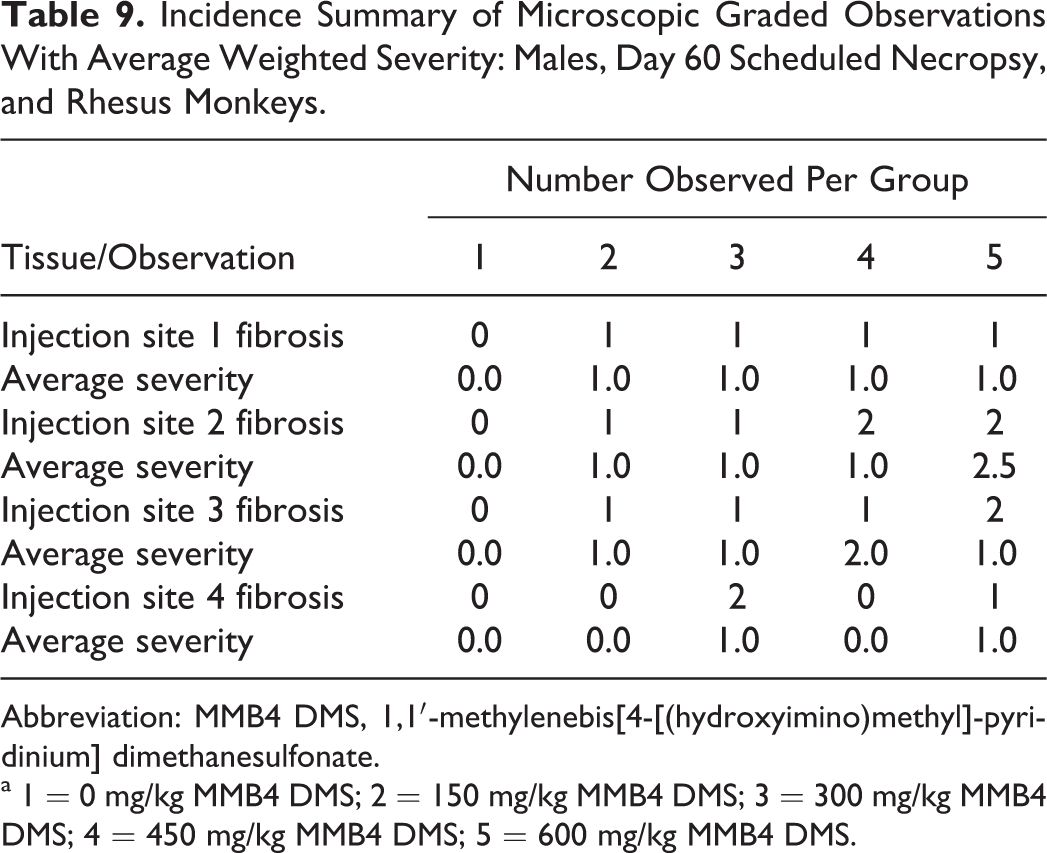
Abbreviation: MMB4 DMS, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate.
a 1 = 0 mg/kg MMB4 DMS; 2 = 150 mg/kg MMB4 DMS; 3 = 300 mg/kg MMB4 DMS; 4 = 450 mg/kg MMB4 DMS; 5 = 600 mg/kg MMB4 DMS.
Incidence Summary of Microscopic Graded Observations With Average Weighted Severity: Females, Day 60 Scheduled Necropsy, and Rhesus Monkeys.a
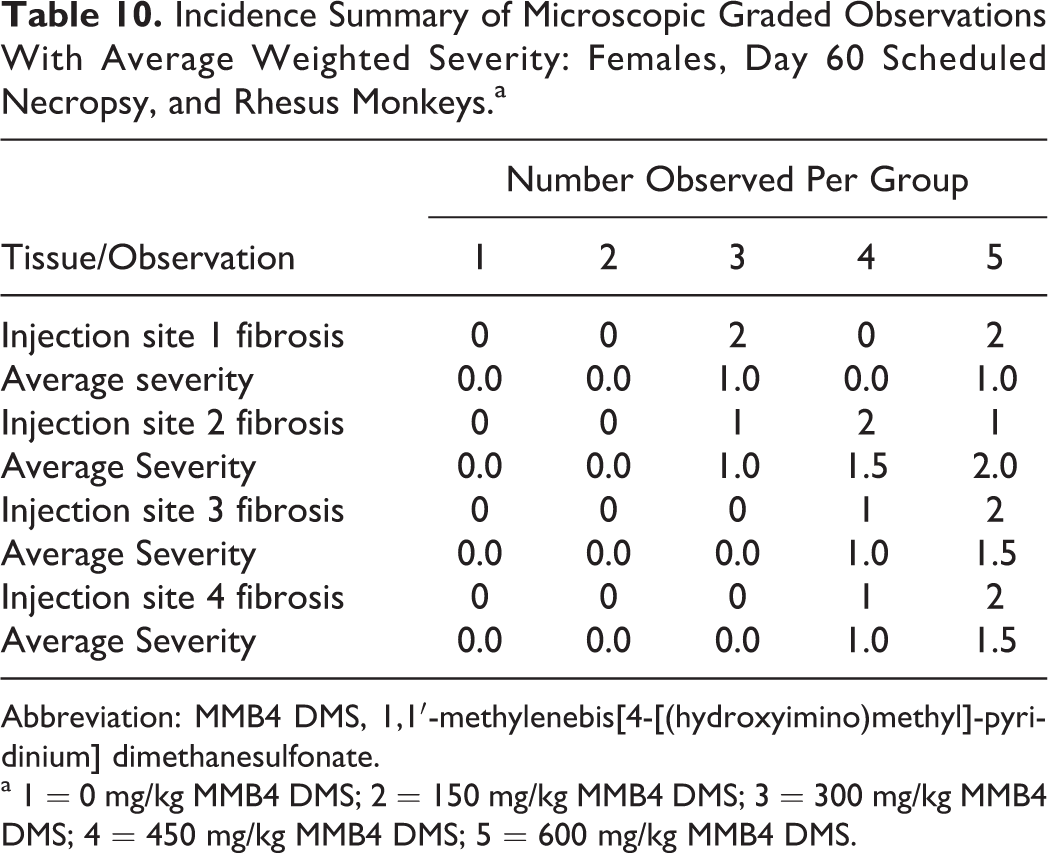
Abbreviation: MMB4 DMS, 1,1′-methylenebis[4-[(hydroxyimino)methyl]-pyridinium] dimethanesulfonate.
a 1 = 0 mg/kg MMB4 DMS; 2 = 150 mg/kg MMB4 DMS; 3 = 300 mg/kg MMB4 DMS; 4 = 450 mg/kg MMB4 DMS; 5 = 600 mg/kg MMB4 DMS.
Discussion
The MMB4, a member of the group of bis-quaternary pyridinium aldoximes, was first synthesized by Hobbiger and Sadler in 1959 5 and has been shown to be more potent than 2-PAM for in vitro reaction of OP compound pesticide-inhibited AChE. Based on the pharmacology studies which suggest that MMB4 may be an effective treatment for OP nerve agent exposure, the development of MMB4 DMS has been ongoing over the past decade. The preclinical development program conducted in support of an Investigational New Drug (IND) submission to the FDA consisted of a series of pharmacology, pharmacokinetic, and toxicology studies that investigated the characteristics of MMB4 DMS from the standpoint of both pharmacology and efficacy as well as the toxicologic/toxicokinetic properties of the drug. 6 The toxicology animal models investigated were the Sprague-Dawley rat, the NZW rabbit, and the rhesus/cynomolgus monkeys. The pivotal IND-enabling studies described in this article were all conducted in compliance with the FDA’s GLP Regulations. 4 The intramuscular route of administration was used in the nonclinical studies because this is the intended route of administration in humans. In addition, the intent is to administer MMB4 DMS once; therefore, the 7-day repeat dose administration was acceptable by the FDA for evaluating the safety of the drug. Of the toxicology species tested in the repeated dose studies used to calculate the maximum recommended starting dose (MRSD) and appropriate safety factors, rabbits were the most sensitive species to the effects of MMB4 DMS at equivalent doses based on the survival and the presence of target organs, other than injections sites, noted only in the rabbit.
In the current MMB4 DMS development program, Sprague-Dawley rats, NZW rabbits, and rhesus monkeys were administered with MMB4 DMS via the intramuscular route once daily for 7 consecutive days and subjected to a full toxicological evaluation. Rats received MMB4 DMS at dose levels ranging from 100 to 800 mg/kg/d, rabbits received dose levels of 3 to 800 mg/kg/d (across multiple studies), and rhesus monkeys received MMB4 DMS at dose levels of 150 to 600 mg/kg/d/d. Of the 3 species evaluated, the NZW rabbit appeared to be the most sensitive in terms of toxicological consequences of MMB4 DMS dose administration. Mortality was noted in the rats at dose levels of ≥400 mg/kg/d and in the rabbits at ≥200 mg/kg/d; all monkeys administered up to 600 mg/kg/d survived until their scheduled termination. The cause of death in the rats and rabbits has not been determined; without histopathological changes that could indicate specific organ effects, mortality in the rats would be very difficult to elucidate.
Histomorphological changes in the rabbit were noted and were consistent across studies. Specific organ toxicity included necrotizing inflammation in the heart and tubular nephrosis and protein casts in the kidneys (≥200 mg/kg/d), and necrotizing bronchiolitis, bronchiolar epithelial restitution/proliferation, and proliferative bronchiolitis in the lungs, present at various dose levels ≥100 mg/kg/d. In order to gain a better perspective on the potential relationship between these histological changes and cause of death of the rabbits, subject matter experts were consulted and requested to review the slides from these studies and other selected studies. Dr Carl Alden (Millennium Pharmaceuticals) reviewed the kidney slides, Dr John Van Vleet (Purdue University) reviewed the heart slides, and Dr Kristin Nikula (Seventh Wave) reviewed the lung slides. Although each expert reviewed his or her respective slides individually, discussions were maintained so that the experts could compare their findings in case the changes in the organ they were examining may have had an impact on the other organs being examined as well.
All 3 experts agreed on 2 basic conclusions. First, the changes originally noted in the kidneys, hearts, and lungs of the rabbits appeared to be species specific, as these histological changes were not noted in the rat study or any other monkey study histologically examined. Second, the histopathological changes noted in these 3 organ systems may have been associated with the cause of death of the rabbits but were not primarily responsible for the mortality. This finding was based on the fact that the severity of the changes noted was generally minimal-to-mild in nature. In some cases, similar histopathological changes noted were also seen in control vehicle animals.
The 1 lesion that was noted histologically across all the species tested was at the injection site. Microscopic examination revealed necrotizing inflammation, hemorrhage, and fibrosis at the day 8 necropsy in the vehicle control and MMB4 DMS animals, which tended to increase in severity with increasing dose level. Elevations in CK, myoglobin, ALT, AST, and platelets were noted, which supported the inflammation at the administration sites; however, to maintain perspective regarding the size of the muscle tissue involved, the lesions generally affected no more than a 1-cm2 area of the section examined. In the rhesus monkeys, CK displayed maximum increases in all the groups, including vehicle controls, on day 1 of dosing and subsequently decreased on days 3 and 5, with levels close to baseline by day 8. Similarly, in the rabbits, CK values were significantly higher in all the groups on day 8 compared to pretreatment, with full recovery observed by day 28 or 60. It appears that the vehicle control is associated with elevated CK values, principally from inflammation at the administration site, and that this reaction is exacerbated by the presence of MMB4 DMS. In all cases, recovery was noted in clinical pathology and histomorphology by day 28. Hemorrhage was not noted in the injection sites, and only a few indications of muscle cell damage and fibrosis were noted microscopically. In the extended recovery periods in the rabbit study, residual fibrosis with myocyte regeneration from the injection sites was present in a small number of 100 and 200 mg/kg/d MMB4 DMS-treated animals necropsied on day 60, but these changes had resolved in all the animals examined by day 91.
Kidney injury, characterized primarily by proximal tubular necrosis with predilection for the pars recta, was associated with intramuscular injection of MMB4 DMS in the NZW rabbit. No necrosis was observed following 14 or 21 days of recovery. In addition, regenerative effects were prominent, characterized by a basophilic tinctorial change in tubular epithelium of mild-to-moderate severity based on the percentage of parenchyma affected. This regenerative change occurred rapidly, noted at the earliest sacrifice day (day 3) and, after the recovery periods, was the primary finding at dose levels that caused tubular necrosis observed in early killing periods. The affected doses were those ≥200 mg/kg/d MMB4 DMS in males and 400 mg/kg/d in females in single as well as repeat dose regimens.
Associated with these changes in the kidney were decreases in some clinical pathology parameters (ie, protein and albumin) which may have reflected some loss through compromised functionality as well as decreases in food consumption and loss from inflammation at the administration sites. As with other changes elicited by the administration of MMB4 DMS in the rabbit, rapid recovery was noted following a nontreatment period. A dose of 100 mg/kg/d was the NOAEL for both males and females in both single and repeat dose regimens. It was suggested that the kidney injury might be driven by acute events rather than events occurring over time, since the findings were quite comparable in single versus repeat dose exposures. It was also apparent that the males were slightly more sensitive to the renal effects than the females. The primary kidney effects may have been secondary to hypoxia based on a number of observations including the fact that the pars recta was primarily affected and represents the most vulnerable site in the kidney to systemic hypoxia and is the site with the highest metabolic rate and the highest oxygen consumption. This is the site of maximal metabolizing enzyme activity and of maximal organic ion transporter activity, and although the necrosis in the pars recta was not pathognomonic for ischemic injury, it was characteristic. The 100 mg/kg/d dose group was identified as the NOAEL, and effects at 200 mg/kg/d in males were comparable for both single dose and repeated dose administration regimens, which suggests that the renal injury might be more related to
In the heart, 2 types of cardiac lesions were present; background lesions consisted of focal to multifocal infiltrations of either mononuclear leukocytes or mononuclear leukocytes admixed with granulocytic leukocytes and with occasional single necrotic cardiac muscle cells present, noted in both control vehicle and test article animals. The second type of cardiac lesion was attributed to MMB4 DMS and was characterized by limited acute/subacute myocardial focal necrosis that had infiltration of macrophages into the damaged tissue. The cellular infiltration is the result of chemotactic attraction of macrophages from the blood into necrotic tissue. The lesions of necrosis were generally either minimal or mild in severity. The lesions were consistently concentrated in the left ventricular papillary muscle and/or in the subendocardial or subepicardial myocardium of the left and right ventricular walls; however, based on the low level of severity, these lesions were not considered to be the cause of death.
Nearly, one-quarter of the unscheduled death rabbits did not have any evidence of lesions of myocardial necrosis. The myocardial lesions were similar in rabbits with unscheduled deaths versus those with scheduled deaths. The occurrence of a low frequency and severity of myocardial necrosis in both the control and the 100 mg/kg/d groups indicated that the NOAEL for MMB4 DMS was 100 mg/kg/d with respect to the aforementioned heart changes. Thus, Dr Van Vleet concluded that the cardiac lesions were not the cause of the unscheduled deaths. The distribution and character of the lesions of myocardial necrosis attributed to MMB4 DMS were similar to those reported in rabbits following endogenous catecholamine release 7 and from exogenous catecholamine administration. 8, 9 Also, similar lesions of myocardial necrosis were associated with various forms of stress such as overcrowding in rabbits and rats and restraint in rats. 10 These stress-related cardiac lesions may be due to endogenous catecholamine release.
Pathophysiologic alterations associated with catecholamine toxicosis include tachycardia and myocardial hypoperfusion. The inconsistency across group with respect to troponin values may have been a reflection of the inconsistency of the lesions and the alternate etiology. Measureable troponin values were noted in some groups (including vehicle controls) pretreatment and subsequently demonstrated peak values on day 1, after which they decreased close to baseline values in spite of continued MMB4 DMS administration. During this time, no association to dose level was noted, and animals that displayed measurable values on day 2 or 3 no longer had measurable troponin values at subsequent time points. Similar results were noted in the rhesus monkey study as well. Whether troponins are a sensitive biomarker for cardiotoxicity in these species cannot be determined from these studies, but it may be concluded that in these particular studies, troponin I and T do not correlate with cardiac changes.
In the lungs, the results of the 4 rabbit studies suggest that the NOAEL for lung histopathologic findings in rabbits after intramuscular injection of MMB4 DMS was 50 mg/kg/d. The cause of unscheduled deaths was not clearly established by this examination of lung histopathology, as there were a number of cases where lung lesions were not observed microscopically in the unscheduled deaths; in other cases, animals were terminated as scheduled and were found to have lung lesions similar in severity to the unscheduled deaths. The test article-related bronchiolar lesions, which were the main test article-related lesions in the lung, would not be expected to interfere with diffusion of oxygen from the alveoli to the blood in capillaries and thus would not be expected to lead to hypoxemia. Lesions in the lungs of the rats and rhesus monkeys (or cynomolgus monkeys from one pilot study) were not observed.
The test article-related findings affected the bronchioles in rabbits. The incidence and severity of the findings in rabbits over time in the unscheduled death and scheduled sacrifices suggested that the initial damage was necrosis of the bronchiolar epithelium. Repair/proliferation of the bronchiolar epithelium occurred in response to the bronchiolar epithelial necrosis. In response to denuding of the epithelium, adjacent bronchiolar epithelial cells flattened and spread along an intact basal lamina, when present. These cells and adjacent cells proliferated (regenerative hyperplasia), leading to reepithelialization of the bronchioles. In some cases, small amounts of fibrinous or cellular exudates that were attached to the denuded bronchiolar surface were covered during the reepithelialization leading to small papillary projections into the bronchiolar lumens. Over time, regenerative hyperplasia may resolve completely, and the morphology of the bronchiole may return to normal. In areas of more severe damage to the bronchiolar wall, bronchiolitis with remodeling occurred. This finding suggested that the damage breached the basal lamina in some foci and extended deeper into the bronchiolar wall and/or that the amount of exudation was focally greater than the more common small exudates that led to small papillary projections during the repair process. The lumen was partially or completely obstructed with subsequent outcomes of fibrosis, recanalization of the lumen with stenosis, or obstruction without recanalization of the lumen. Bronchiolitis with remodeling would not be expected to completely resolve over time in all the cases because the remodeling involved fibroplasia and obstruction of the bronchiolar lumen. Recanalization and reepithelialization of the remodeled bronchiole can occur, but some fibrosis and evidence of a remodeled bronchiole would be expected to remain, in some foci. Male rabbits appeared to be slightly more susceptible to MMB4 DMS-induced bronchiolar damage than female rabbits. However, the NOAEL for MMB4 DMS, based on lung histopathology, was 50 mg/kg/d for both genders.
Serum biomarkers, when compared to respective control or pretest values, included elevations in CK, which was generally increased in all the groups on day 8, and AST and lactate dehydrogenase, which were increased in the 200 mg/kg/d MMB4 DMS group. Elevations in these specific biomarkers are indicative of striated muscle damage principally derived from skeletal muscle with a small contribution possible from cardiac muscle. In addition, increases in the cardiac-specific biomarkers troponin I and T, typically correlated with histologic evidence of muscle damage in the heart, were elevated on the day-2 and day-8 rabbits and returned to baseline values in the day-60 and day-91 rabbits, indicating recovery and the absence of continuing cardiac damage.
Therefore, the overall NOAEL in the rabbit was determined as 50 mg/kg/d. The NOAEL in the rat study was 100 mg/kg/d and the NOAEL for the rhesus monkey has been subsequently determined to be 600 mg/kg/d. Calculations for the MRSD were determined using rabbit NOAEL data; and in general, the blood levels of MMB4, at this dose level, were approximately 100 μg/mL (
In summary, MMB4 DMS has been evaluated for toxicity in a preclinical regimen in Sprague-Dawley rats, NZW rabbits, and rhesus monkeys. In our studies, the NZW rabbit was determined to be the most sensitive species following the intramuscular administration of MMB4 DMS daily for 7 consecutive days. The NOAEL in these species was 50 mg/kg/d in the rabbit, 100 mg/kg/d in the rat, and 600 mg/kg/d in the monkey. With the exception of the intramuscular administration sites in all species, no other specific target organs of toxicity were noted in the rats or monkeys; however, it appeared that the lung, heart, and kidneys did present with clinicopathologic changes indicative of test article toxicity. With this information and an adequate safety factor imposed, a clinical monitoring plan was developed for a phase 1 study in humans.
Footnotes
Authors’ Note
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or reflecting true views of the Department of the Army or the Department of Defense.
Declaration of Conflicting Interests
The studies and results summarized in this manuscript are a compilation of a number of studies conducted at Midwest Research Institute (now MRIGlobal, Kansas City, Missouri) and Battelle (Columbus, Ohio). The results reflect the views of the contract research organizations’ staff, including the board-certified clinical/anatomic pathologists, study directors, and expert support staff. In addition, subject matter experts in renal (Dr Carl Alden, Millennium Pharmaceuticals, Cambridge, Massachusetts), cardiac (Dr John Van Vleet, Purdue University, West Lafayette, Indiana), and pulmonary (Dr Krstin Nikula, Seventh Wave, St Louis, Missouri) pathology were instrumental in providing unbiased and expert information related to the etiology, pathogenesis, and sequelae of the changes noted in these studies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the United States Army; Chemical Biological Medical Systems (CBMS)/Medical Identification and Treatment Systems (MITS) under contract SP0700-00-D-D3180, Delivery Order 0599 (Task 771), and Delivery Order 0600 (Task 789).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
