Abstract
The dietary supplement Citicoline free-base (choline cytidine 5′-pyrophosphate) was toxicologically evaluated in Sprague-Dawley rats using oral gavage. In an acute 14-day study, 2000 mg/kg was well tolerated. In a 90-day study, 100, 350, and 1000 mg/kg/day doses resulted in no mortality. In males, slight significant increases in serum creatinine (350 and 1000 mg/kg/day), and decreases in urine volume (all treated groups) were observed. In females, slight significant increases in total white blood cell and absolute lymphocyte counts (1000 mg/kg/day), and blood urea nitrogen (BUN) (100 and 350, but not 1000 mg/kg/day) were noted. A dose-related increase in renal tubular mineralization, without degenerative or inflammatory reaction, was found in females (all treated groups) and two males (1000 mg/kg/day). Renal mineralization in rats (especially females) is influenced by calcium:phosphorus ratios in the diet. A high level of citicoline consumption resulted in increased phosphorus intake in the rats, and likely explains this result.
Keywords
Citicoline (CDP-choline; cytidine 5′-diphosphocholine; choline cytidine 5′-pyrophosphate) is a naturally occurring substance composed of cytidine and choline linked by a diphosphate bridge. 1,2 When taken orally, citicoline is hydrolyzed into cytidine and choline, which are readily absorbed. 3 As a choline donor, citicoline acts as a precursor for both phosphatidylcholine, an important structural component of cell membranes, and acetylcholine, a neurotransmitter vital for cellular communication in the brain. The clinical benefits of citicoline as a nootropic agent have been described in elderly patients with cognitive deficits, memory loss, or senile dementia. 4-6 Evidence also exists for the protective effects of citicoline in reducing ischemic injury to the brain 1,4 ; however, clinical trials in patients suffering acute ischemic infarction (stroke) have yielded mixed results. 7-9 This neuroprotective agent has been investigated in the treatment of glaucoma-induced optic nerve damage based on the agent’s potential role in repairing nerve cell membranes in the retina via enhanced phosphatidylcholine synthesis. 10,11 Recent research on citicoline showed that oral administration of citicoline produced regionally specific changes in neurochemistry of the anterior singulate cortex. 12 These results point to a potential mechanism for citicoline’s neuroprotective properties.
Citicoline is available as a drug in Europe and Japan and as a dietary supplement in the United States. It can be administered as a “free-base” or as a sodium salt. A lack of clarity exists in the scientific literature regarding the potential difference in efficacy or safety of these 2 forms of citicoline, as many of the published studies do not clearly identify the form used. Although some nonclinical toxicity studies on citicoline sodium salt have been published, 13,14 safety results of citicoline free-base taken orally have not been reported.
The LD50 of an acute single intravenous dose of citicoline was determined to be 4600 mg/kg and 4150 mg/kg in mice and rats, respectively. 15 A 30-day, subacute rat gavage study demonstrated that oral doses of 100 and 150 mg/kg/d sodium citicoline resulted in no signs of toxicity. 13 The chronic oral consumption of sodium citicoline was studied in dogs given a single 1.5 g/kg (limit) dose by esophageal cannulation daily for 6 months and was found to produce no physiological, biochemical, neurological, or histopathological abnormalities, aside from that attributed to normal individual variation. 14
To provide additional nonclinical toxicity assessment of citicoline free-base, this article presents the results of 2 oral gavage toxicity studies preformed in male and female Sprague-Dawley rats: a single-dose acute study with a 14-day observation period (limit test) and a 90-day repeat-dose oral toxicity study.
Materials and Methods
Studies were conducted under Good Laboratory Practice conditions at Pharmaceutical Control and Development Laboratory (PCDL), Budapest, Hungary.
Test Animals and Housing
SPF Crl:CD BR rats of Sprague-Dawley origin (Charles River Hungary, Budapest, Hungary) were used for these studies and kept under conventional conditions. The Institutional Animal Care and Use Committee of PCDL approved each study. Animals were quarantined, observed for 5 days, and randomly assigned to treatment groups prior to initiation of treatment.
On the day of randomization (the day prior to substance administration), rats in the acute study were 7 to 8 weeks of age weighing 211 to 227 g (males) and 180 to 193 g (females); rats in the subchronic study were 6 weeks of age weighing 120 to 157 g (males) and 120 to 153 g (females).
Rats of the same sex were housed in type II macrolone cages. Five rats were housed per cage in the acute study and 2 rats were housed per cage in the subchronic study. Rats were fed rodent diet S8106-S011 SM R/M-Z+H (Ssniff Spezialdiäten, Soest, Germany) ad libitum in both the acute and subchronic studies, except for the overnight fasting period prior to treatment, during treatment, and for the first 2 hours of the posttreatment observation in the acute study, and except for the overnight fasting period prior to blood sampling and necropsy in the subchronic study. Tap water (supplied by Waterworks of Budapest, average hardness 148 mg CaO/L) was available ad libitum via drinking bottles and was checked monthly for selected microbiological parameters at PCDL. The results met the requirements for potable water.
The temperature was maintained at 22°C ± 3°C and a relative humidity of 30% to 70%. Air was exchanged 15 times per hour, and artificial lighting was set for a 12-hour light-dark cycle.
Test and Control Articles
The test material consisted of citicoline free-base as a white-crystalline powder (Kyowa Hakko Bio, Chiyoda-ku, Tokyo, Japan; lot I43026 for the acute study and lot I930132 for the subchronic study) and was certified by analytical testing to be 99.85 to 99.9% pure. The heavy metal content (as lead) was not more than 10 ppm, ammonium not more than 0.05%, free phosphoric acid not more than 0.1%, 5′-cytidylic acid not more than 1%, and arsenic not more than 2 ppm.
Dosing suspensions for the single-dose toxicity study were formulated by mixing 10 g of citicoline free-base with 50 mL of distilled water. Three samples of 5 mL each were taken from the formulated test item immediately before the dosing procedure and transferred for analysis by Wessling Chemical Laboratory GmbH (Budapest, Hungary). The actual concentration was ±4% of the theoretical value, well within the acceptable limits of ±10%. Dosing suspensions for the subchronic study were prepared by mixing 1, 3.5, or 10 g of citicoline free-base with 100 mL of distilled water for the 100, 350, and 1000 mg/kg/d test articles, respectively. Solutions were stirred continuously during the treatment with a magnetic stirrer. Samples were analyzed by Wessling Chemical Laboratory immediately preceding the first dosing procedure, and the actual concentrations were reported as being ±5% of the theoretical.
Experimental Design
Fourteen-day acute oral toxicity study (limit test)
Ten male and 10 female rats were assigned to treatment groups. After an overnight fast, each rat received a single oral dose by gavage of 2000 mg/kg of citicoline free-base dissolved in distilled water at a volume of 10 mL/kg body weight (day 1). Animals were checked for mortality, general state, external appearance, behavior, and clinical symptoms twice daily at the beginning and end of the workday on weekdays and once on weekend days. Body weight was measured on days 1, 2, 8, and 15, and findings were calculated and tabulated. Euthanasia was performed on day 15 using intravenous administration of 0.1 mL/100 g body weight of T61 (Intervet International), which contains 0.2 g embutramide, 0.005 g tetracaine hydrochloride, and 0.05 g mebezonium iodide per milliliter. Gross necropsies were performed, and tissue changes were observed and recorded.
Ninety-day subchronic oral toxicity study
One hundred and sixty rats (80 males, 80 females) were divided into 4 groups of 20 males and 20 females each. The rats received a daily dose of either distilled water (control group) or 100, 350, or 1000 mg/kg/d of citicoline free-base (dissolved in distilled water) by gavage once daily for 90 consecutive days. Both the test and control articles were administered in a volume of 10 mL/kg, adjusted weekly for changes in the animals' body weight.
Animals were checked for mortality, general state, external appearance, behavior, and clinical signs twice daily at the beginning and end of the workday on weekdays and once on weekend days until the morning of the 91st day. They were weighed on the day of the first treatment (day 1) and weekly thereafter. Food consumption was measured weekly and the daily average food consumption was calculated. Daily (24-hour) water consumption was measured once per week.
Direct ophthalmoscopic examination was conducted on 10 males and 10 females of each dose group on the day before the first treatment and on all high-dose and control animals during week 11 (on days 75 or 76 for females and days 77 or 78 for males) by a veterinary ophthalmologist using a Welch Allyn Pan Optic Ophthalmoscope (Skaneateles Falls, NY).
Blood sampling was performed on day 91, prior to euthanasia. Animals were fasted overnight, and blood samples were drawn from the retro-orbital sinus under diethyl ether–induced anesthesia. For hematology analyses, an aliquot of approximately 0.5 mL was placed in a tube with anticoagulant (20 μL K-EDTA solution of 20%, dried in the tubes). Whole blood was collected for glucose determination; the remaining portion was placed in a plastic centrifuge tube without anticoagulant for collection of sera for clinical chemistry analyses.
The hematological parameters examined from blood samples included erythrocytes (red blood cell count) and leukocytes (white blood cell [WBC] count), hemoglobin concentration, hematocrit, and platelets (Coulter AcTdiff cytometer and Coulter reagent kit, Coulter Diagnostics, Miami, Fla). Differential leukocyte counts were performed by microscopic examination of blood smears stained with May-Grünwald-Giemsa stain.
Clinical chemistry parameters measured included aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, total cholesterol, total protein, albumin, blood urea nitrogen (BUN), chloride, total calcium, and creatinine. Blood glucose was measured in whole blood using the reagent obtained from Diagnosticum (Budapest, Hungary). Whole blood and sera were analyzed using a FP-901 Chemistry Analyzer (Labsystems Oy, Finland) and reagents from Reanal Finechemical Works (Budapest, Hungary). Sodium and potassium were determined using an IL 943 flame photometer (Instrumentation Laboratory, Lexington, Mass). Prothrombin time was determined from blood collected into tubes containing trisodium citrate with a dilution factor of 1:9 sodium citrate to 3.8% blood and was analyzed with a Coagulometer CM2 (Behnk Elektronik, Norderstedt, Germany).
Urinalysis was performed once during the 12th treatment week (on days 82 or 83 of the study) for 10 males and 10 females from each dose group. Approximately 20 hours after the previous treatment, animals were given 10 mL/kg distilled water by gavage and placed into stainless steel cages especially constructed for separating urine from feces. Urine was collected prior to the next treatment for 3 hours. Appearance, volume, and specific gravity (by refractometry) were measured, and pH, protein, glucose, and blood were analyzed using test strips (Hepta phan, Lachema, Brno, Czech Republic). To prevent degradation and loss caused by evaporation, test strips were applied to urine collected during the first hour, and samples from each animal were collected in tubes with tight fittings. Volume and specific gravity were measured at the end of the collection time.
Necropsy was performed on day 91 of the study. All rats were deprived of food for 16 hours, anesthetized using diethyl ether, exsanguinated, and necropsied. Tissues and organs listed as follows were observed, collected, and preserved in 8% neutral buffered formalin: liver, kidneys, adrenals, testicle (left only), spleen, brain, spinal cord, eye (left only), thymus gland, heart, mesenteric lymph node, submandibular lymph node, sternum, sciatic nerve with skeletal muscle, stomach, duodenum with pancreas, lungs, pituitary gland, submandibular salivary gland (left), trachea, esophagus, thyroid (including parathyroids), epididymis (right only), prostate, female mammary gland, uterus, ovaries, colon, urinary bladder, aorta, and all gross lesions. Organs fixed in Bouin’s solution included the right testicle and the right epididymis. The following organs were weighed: liver, kidneys, adrenals, testicles, epididymides, uterus, ovaries, thymus, spleen, brain, and heart. Ratios of body weight to organ weight were calculated.
Following dehydration and embedding in paraffin, tissues were sectioned at 5 microns, stained with hematoxylin and eosin, and then examined microscopically. Histopathological examination was performed on all collected tissues from all males and females from the 0 and 1000 mg/kg/d groups. Because increased degrees of renal mineral deposits were found in the 1000 mg/kg/d female group, preparation of slides and histopathological examination of the kidneys were also performed in the 100 and 300 mg/kg/d groups of both sexes.
Liver, heart, and kidney sections of the 0 and 1000 mg/kg/d groups and kidney sections of the 100 and 350 mg/kg/d groups of both sexes were stained using periodic acid-Schiff (PAS) reaction combined with diastase digestion. Sections of liver, heart and kidney, previously fixed in formalin, were cut on a freezing microtome (Cryostate) at 10 to 15 micrometers and stained for fat with Fat Red.
Statistical Analyses
Statistical analyses were performed with Statistica 5.5 (edition 99, StatSoft, Tulsa, Okla). Data were analyzed for homogeneity of variance by the Bartlett test. If the variances proved to be homogeneous, 1-way analysis of variance (ANOVA) was performed. If ANOVA detected significant differences (P < .05), the Tukey test was used to compare the treatment groups versus the control group. In the case that the variances failed the Bartlett’s homogeneity test, the Kruskal-Wallis nonparametric 1-way ANOVA was performed followed by the Kolmogorov-Smirnov test. In the case of nonparametric values, the Kruskal-Wallis nonparametric 1-way ANOVA was performed followed by the Kolmogorov-Smirnov test.
Results
Acute Toxicity
No mortality was observed following single oral administrations of 2000 mg/kg citicoline free-base. There were no treatment-related effects on clinical signs, behavior, or body weights. No gross lesions were noted in any organs of the animals on day 15.
Subchronic Toxicity
No mortality was observed. No treatment-related changes in either the general condition or external appearance were observed in any of the male and female dose groups treated with citicoline free-base. There were no significant differences in body weight or body weight gain among groups treated with citicoline free-base compared with the control group at any time point during the experimental period. All male and female groups that were treated with citicoline free-base consumed similar amounts of food compared with the corresponding control groups throughout the study. A minimal (6%) yet statistically significant increase in water consumption that occurred in the male group treated with 350 mg/kg/d was attributed to sporadic deviation within the groups and has no biological significance.
No ophthalmological changes were observed in males or females from the 0 or 1000 mg/kg/d groups at the 12th week of treatment.
In females, slight but statistically significant increases in WBC and absolute lymphocyte counts were noted in the 1000 mg/kg/d group; corresponding elevation did not occur in the male treatment groups. No other treatment-related changes in hematology parameters were noted in males or females (Tables 1 and 2).
Selected Data From the Subchronic Study of Male Crl:CD BR Sprague-Dawley Rats Exposed to Citicoline Free-Base By Gavage
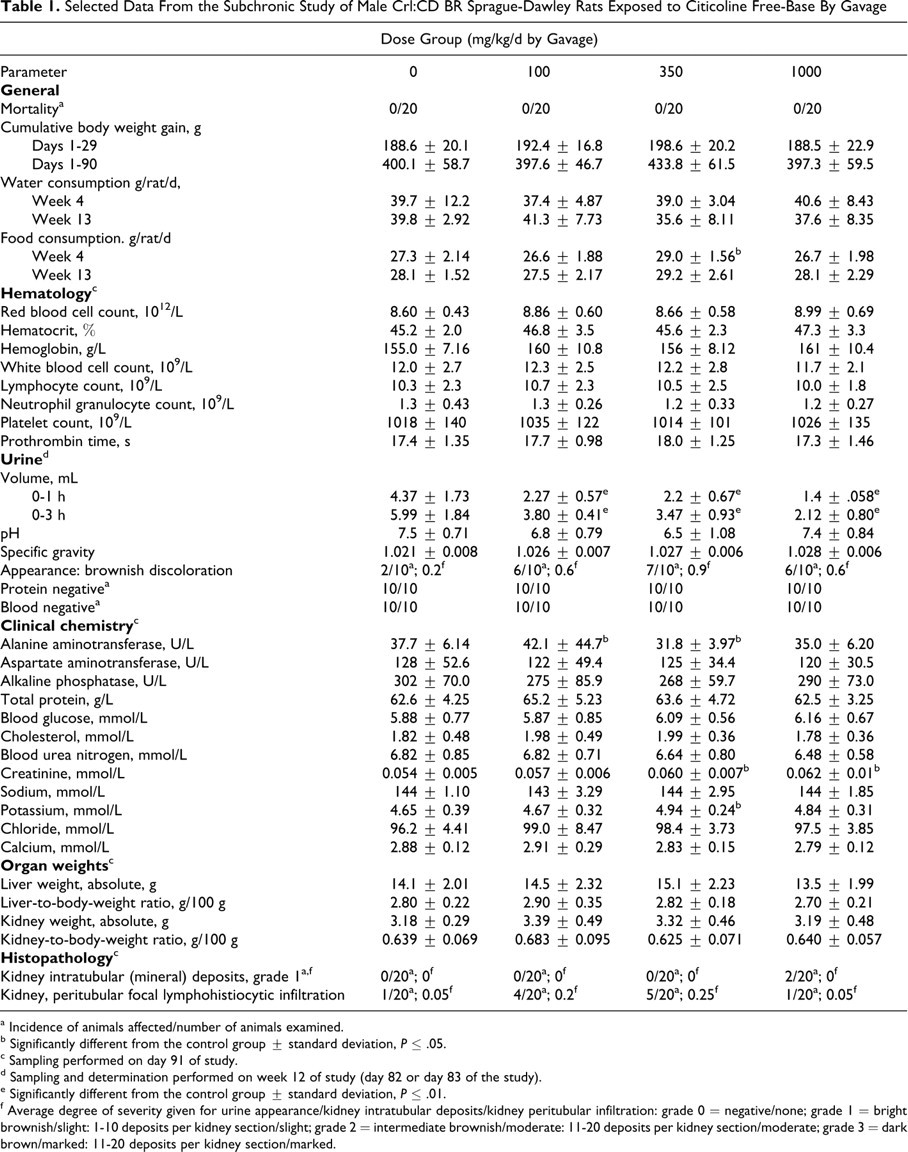
a Incidence of animals affected/number of animals examined.
b Significantly different from the control group ± standard deviation, P ≤ .05.
c Sampling performed on day 91 of study.
d Sampling and determination performed on week 12 of study (day 82 or day 83 of the study).
e Significantly different from the control group ± standard deviation, P ≤ .01.
f Average degree of severity given for urine appearance/kidney intratubular deposits/kidney peritubular infiltration: grade 0 = negative/none; grade 1 = bright brownish/slight: 1-10 deposits per kidney section/slight; grade 2 = intermediate brownish/moderate: 11-20 deposits per kidney section/moderate; grade 3 = dark brown/marked: 11-20 deposits per kidney section/marked.
Selected Data From the Subchronic Study of Female Crl:CD BR Sprague-Dawley Rats Exposed to Citicoline Free-Base By Gavage
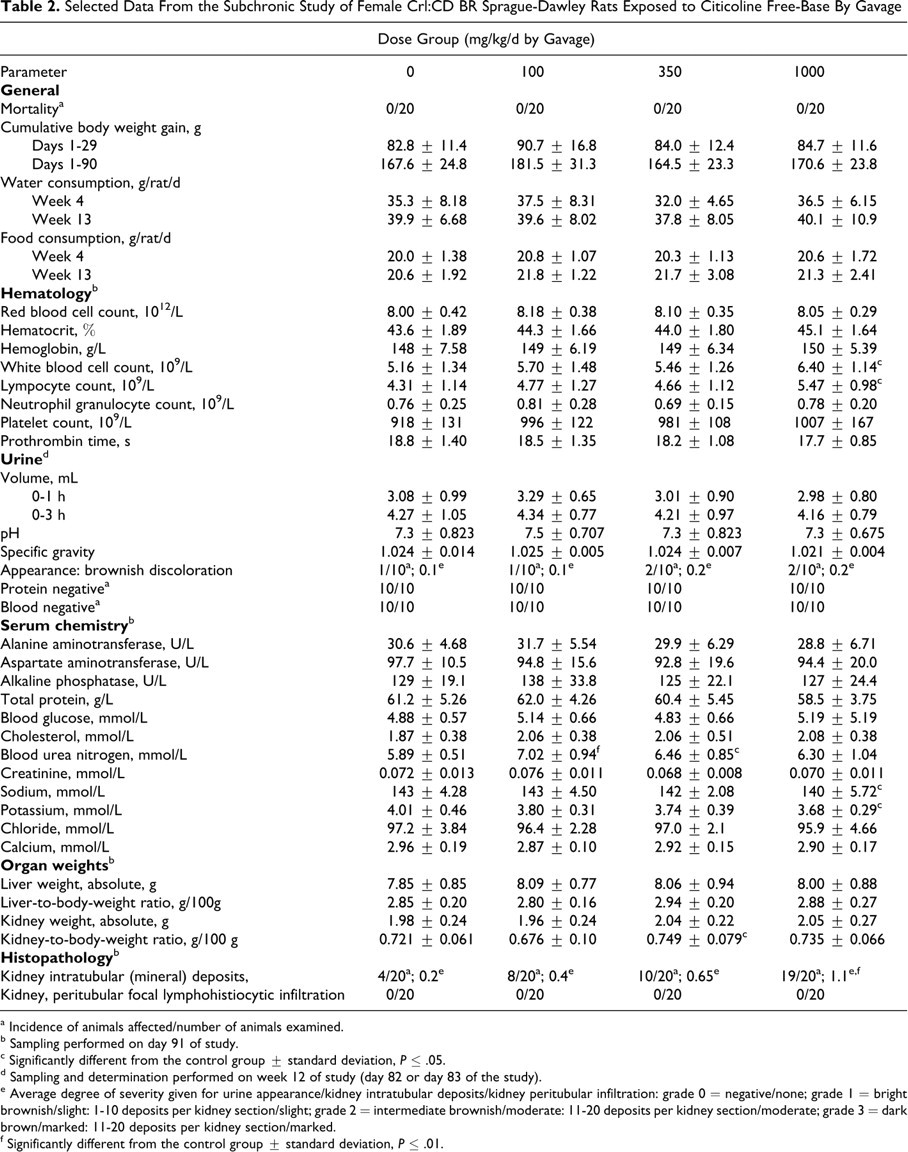
a Incidence of animals affected/number of animals examined.
b Sampling performed on day 91 of study.
c Significantly different from the control group ± standard deviation, P ≤ .05.
d Sampling and determination performed on week 12 of study (day 82 or day 83 of the study).
e Average degree of severity given for urine appearance/kidney intratubular deposits/kidney peritubular infiltration: grade 0 = negative/none; grade 1 = bright brownish/slight: 1-10 deposits per kidney section/slight; grade 2 = intermediate brownish/moderate: 11-20 deposits per kidney section/moderate; grade 3 = dark brown/marked: 11-20 deposits per kidney section/marked.
f Significantly different from the control group ± standard deviation, P ≤ .01.
The serum creatinine levels in the 350 and 1000 mg/kg/d males exhibited statistically significant increases (11% and 15%, respectively), but the levels were unchanged in the females. Blood urea nitrogen levels exhibited statistically significant increases in the 100 and 350 mg/kg/d females (19% and 10%, respectively) but were not elevated in the 1000 mg/kg/d females or in any male groups. There was a very mild, statistically significant decrease in the 1000 mg/kg/d female serum sodium level (–2%) (Tables 1 and 2).
A dose-related decrease in urine volume was found in all treated male groups (Table 1). The differences were statistically significant in all groups during the first hour of urine collection as well as during the 0- to 3-hour period. Although dose-related increases in specific gravity were noted in all treated male groups, these increases were not statistically significant. There were no differences in the urine pH, protein, glucose, or blood in any of the treated male groups compared with controls. Brownish-colored urine samples were more often noted in the treated males of all dose groups than in the controls (Table 1). In females, no treatment-related differences in urinalysis parameters were noted (Table 2).
Gross necropsy examination revealed no treatment-related lesions. No treatment-related differences in the organ weights or relative organ weights were found. Mineralization occurred with dose-related increased incidence and severity in the kidney of female rats (Table 2). In males, similar deposits were seen only in the kidneys of 2 rats from the 1000 mg/kg/d treated group (Table 1). Mineralization that consisted of lamellated basophilic concretions located intraluminally in the descending proximal tubules (corticomedullary junction region) were seen without apparent degenerative or inflammatory reaction (Figures 1 and 2). No other treatment-related changes were noted in the kidneys or other tissues and organs. Special stains (PAS, Fat Red) of kidneys, livers, and/or hearts did not reveal any treatment-related changes. Sporadic focal lymphohistiocytic infiltration between the tubuli of the renal cortex was noted in a few control and treated males but was not considered treatment related (Table 1).
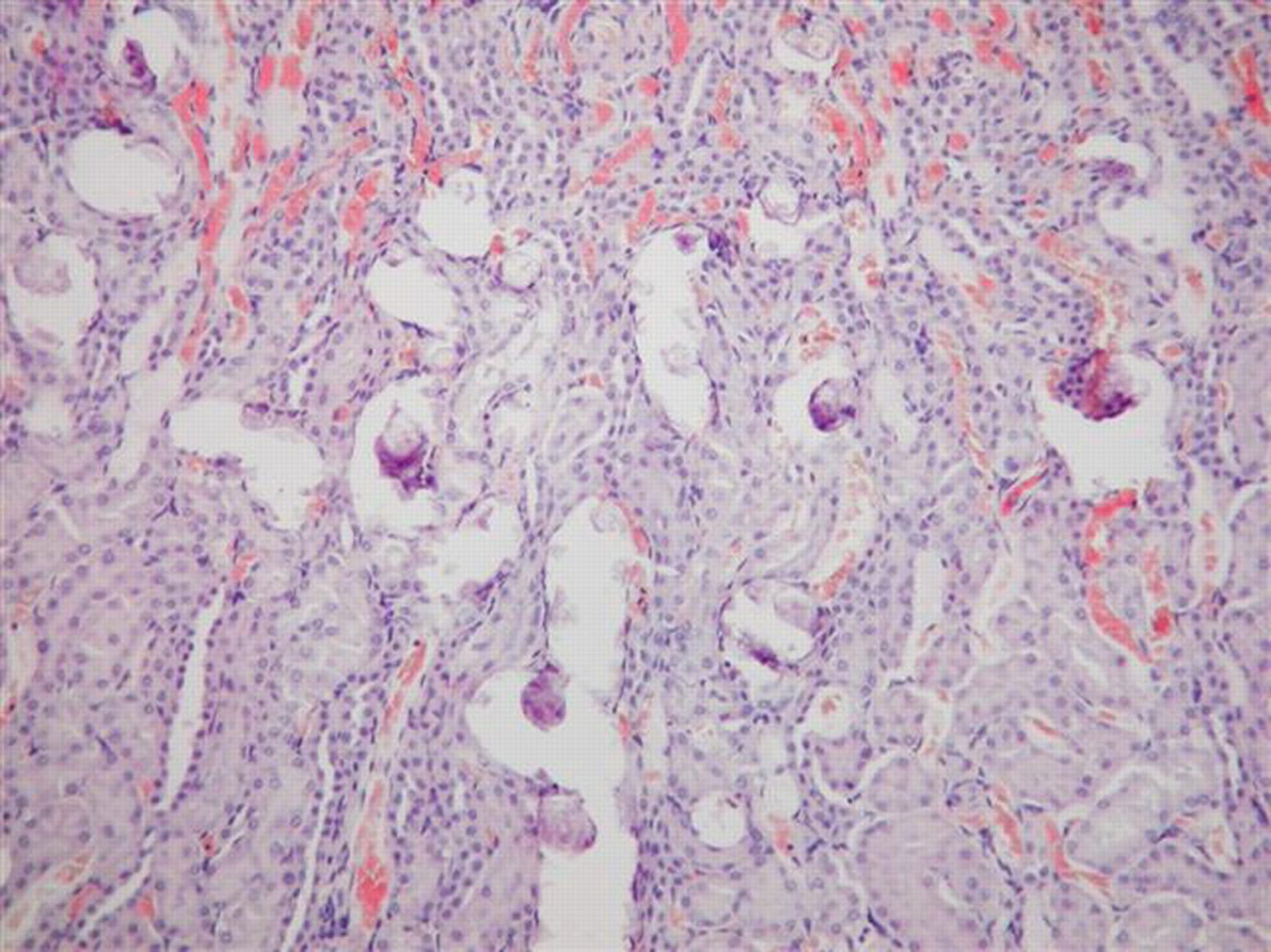
Marked intraluminar mineral deposits in tubules at the corticomedullary junction. Kidney of female rat, 350 mg/kg/d dose group. Hematoxylin and eosin, ×100.
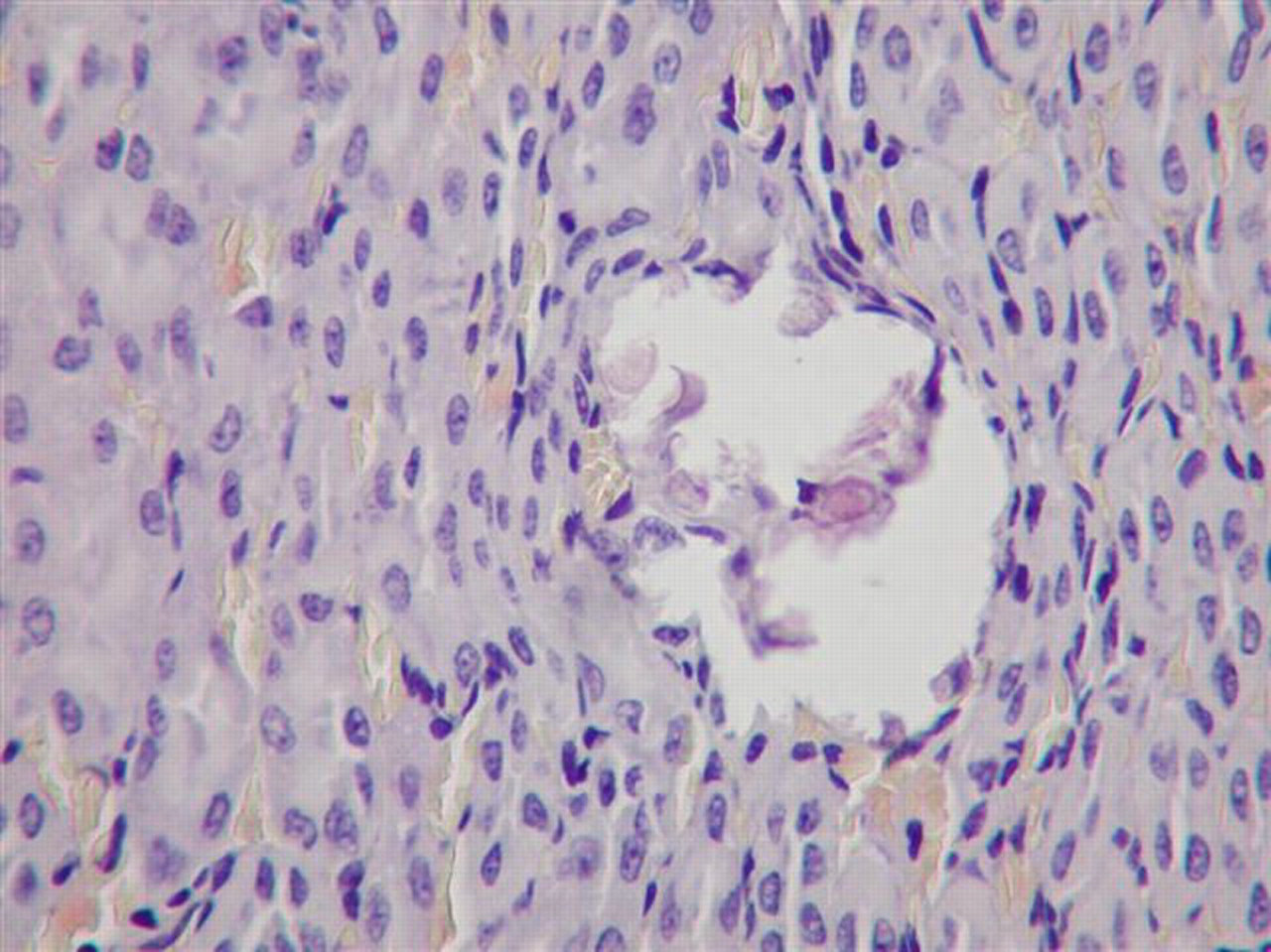
Solitary mineral deposit at the corticomedullary junction. Kidney of female rat, 1000 mg/kg/d dose group. No associated necrosis or inflammation is present. Hematoxylin and eosin, ×400.
Discussion
Kidney-related changes were noted in this subchronic study in both male and female animals despite an absence of apparent toxicity. In males, all treated groups had dose-related decreased urine output, which was not accompanied by significant increases in specific gravity. Creatinine levels slightly increased with dose. These signs occurring together suggest a mild renal functional disorder. They were not accompanied by any other signs of kidney changes, such as increased urinary protein, BUN level, or kidney weight, or by relevant histopathological findings, such as thickening of the basement membranes detected by the PAS reaction; no fibrosis or destruction of the tubular or glomerular elements was noted.
Brownish discoloration of the urine occurred more often in the 100, 350, and 1000 mg/kg/d male groups. The limited semiquantitative urinalysis data (no bilirubin or UBG determinations were done) did not reveal any explanation for this finding. Additionally, liver damage was not noted: serum aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, and protein were unchanged; liver weights were not increased; and no treatment-related microscopic lesions were seen. Histological lesions were not seen in the urinary bladders, nor was blood detected in the urine. The limited occurrence of minimal brown urine discoloration in a few control animals suggests that the finding was not a colored metabolite or conjugate of the test substance. Thus, although the brown urine discoloration in the male rats was considered treatment-related, its cause remains undetermined.
A dose-related increased incidence and severity of kidney mineralization occurred in 100, 350, and 1000 mg/kg/d female groups; kidney mineralization was also seen in 2 male rats receiving 1000 mg/kg/d. The microscopic picture of the mineralization—tiny lamellated basophilic concretions located intraluminally in the descending proximal tubules (corticomedullary junction region) without apparent degenerative or inflammatory host reaction—is similar to the published description of nephrocalcinosis (NC) in female rats. 16,17 However, no specific (chemical or histochemical) identification of the composition of the intraluminar deposits was determined in the present study.
Mineralization (NC) in female rats is a common incidental finding. It has been widely investigated in all rat strains generally used for repeated-dose toxicity studies (Fischer 344, 18 Wistar, 19-21 and Sprague-Dawley 21-23 ). Similar histopathological lesions found in different laboratories were considered related to the calcium (Ca), phosphorus (P), and magnesium levels consumed in the diet. The ratio of Ca to P especially influences the occurrence of renal mineral deposits: a Ca:P ratio in the diet of less than 1 increases the incidence and severity of this lesion, whereas increasing the ratio to greater than 1 prevents development of the lesion. 24-26
The Ssniff rodent feed used in the present study contained Ca:P at a molar ratio of 0.97. However, citicoline free-base is composed of cytidine and choline linked by a diphosphate bridge, yielding a 12.69% phosphorus content for the test article. The phosphorus contributed by the addition of citicoline free-base to the diet decreased the total dietary Ca:P ratio throughout the study; for example, at week 12, the ratio was decreased to 0.92 (100 mg/kg/d group), 0.87 (350 mg/kg/d group), and 0.76 (1000 mg/kg/d group), resulting in a dose-related increasing imbalance that may have favored the accumulation of calcium in the kidneys of female rats. We suggest that this imbalance led to the dose-dependent increased incidence and severity of NC in the treated female rat groups. Male rats are generally less susceptible to NC induced from the diet. 18,26,27 In accordance with this, only 2 males of the 1000 mg/kg/d group exhibited slight NC in the present study.
The effect of NC on kidney function is not clear. Increased concentration of serum urea and/or creatinine was reported in some studies, 24,28,29 whereas other studies failed to find such an association. 24 Albumin excretion has been found in early stages, 30,31 but other authors 23,28,32 found that kidney function was largely unaffected by NC. In the present study, slightly increased BUN was found in the females treated with 100 and 350 mg/kg/d, and slightly increased creatinine was found in the males that received 1000 mg/kg/d; however, protein was not detected in the urine, nor were there any changes in kidney weights. Further studies in this test model with adequately balanced P levels could verify whether the increased NC and the slight alterations in BUN and creatinine could be prevented by a Ca:P ratio greater than 1.0.
A slight tendency toward an increase in the absolute lymphocyte and total white blood cell counts was noted in the females only, which was statistically significant in the 1000 mg/kg/d group (26%). It is possible that this could be the result of a secondary reaction accompanying NC, the only treatment-related alteration found in these groups. However, this has not been previously shown, and no local lymphocytic reaction was detected by routine histological methods in the kidneys. Alternatively, the changes could be due to normal physiological variability, as the elevated counts still fall within published normal values for WBCs (2.0-11.5 × 109/L) and lymphocytes (1.0-10.00 × 109/L) in female adult Sprague-Dawley rats. 33
If our investigation suggests that the tiny lamellated basophilic concretions located in the intraluminar kidney of citicoline free-base-treated rats correspond to nephrocalcification, the finding has limited toxicological significance, because Ca:P ratio–related calcium depletion is known to be a rat-specific finding.
Humans are less susceptible to the Ca:P imbalance. The reference daily intake of P for adult humans is 0.7 to 1.0 g. 34,35 If this daily intake provided by food is complemented by the P content of the highest published daily human dose of 2.0 g citicoline free-base (ie, 0.254 g/d P), 1 the phosphate intake increases by 25% to 36%. However, it remains far below the Tolerable Upper Intake Level of 4.0 g determined by the US Food and Nutrition Board, Institute of Medicine. 35 On the other hand, Wurtman’s group investigated the metabolism of citicoline and found that the metabolism in humans differs from that in the rat. In the rat, oral citicoline administration increases blood plasma levels of citidine and choline, 36 whereas, in humans blood plasma levels of uridine but not cytidine are increased because of cytidinedeaminase in the gastrointestinal tract. 37 This difference in metabolism may also influence the extrapolation of the present data to humans.
Footnotes
Acknowledgments
We thank Krisztina Finta, Erzsebet Palmai, Iren Schön, and Margit Gal for their technical assistance.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Kyowa Hakko USA, Inc., New York, NY, and Healthcare Products Development Center, Kyowa Hakko Bio Co., Ltd., Ibaraki, Japan, with support from the Natural and Medicinal Products Research Division of AIBMR Life Sciences in Puyallup, Washington, DC, USA.
