Abstract
Dinitrotoluene (DNT) is a nitroaromatic explosive used in propellant mixtures and in the production of plastics. Isomers of DNT were administered daily via oral gavage to male Sprague-Dawley rats for 14 days to determine the subacute toxicity of individual isomers of DNT. The 3,5-DNT isomer was the most toxic isomer, inducing weight loss and mortality within 3 days. Cyanosis and anemia were observed for all isomers. Exposure to 2,4-, 2,6-, and 3,5-DNT resulted in decreased testes mass and degenerative histopathological changes. Increased splenic mass was observed for 2,4-, 2,6-, and 2,5-DNT. Extramedullary hematopoiesis of the spleen was noted for all isomers, while lymphoid hyperplasia of the spleen was noted for all isomers except 2,5-DNT. Increased liver mass was observed for 2,3-DNT and 3,4-DNT. Hepatocellular lesions were observed for 2,6-DNT and 2,4-DNT. Neurotoxic effects were noted for 3,4-DNT, 2,4-DNT, and 3,5-DNT.
Introduction
Dinitrotoluene (DNT) is used to make polyurethane foams, dyes, and munitions. The DNTs are a primary product in propellants and are also produced as by-products in the manufacture of the common military explosive, 2,4,6-trinitrotoluene (TNT). The DNT is produced by mixing toluene with nitric acid and can exist as any of 6 isomers (2,4-, 2,6-, 3,4-, 2,3-, 2,5-, and 3,5-DNT). Technical grade DNT (TG-DNT), a mixture of the 6 isomers, is composed of approximately 76.5% 2,4-, 18.8% 2,6-, and 4.7% minor isomers (2.43% 3,4-, 1.54% 2,3-, 0.69% 2,5-, and 0.04% 3,5-DNT). The DNTs are often found in soils associated with munitions manufacturing sites, open burning/incineration, landfills, wastewater holding facilities, and buried ammunition wastes. 1,2 The major isomers have been detected in groundwater at military installations at concentrations as high as 1788 µg/L (2,4-DNT) and 651 µg/L (2,6-DNT). Near production sites, the Chemical Manufacturers Association reported concentrations in surface and groundwater of 32 µg/L and 360 to 20 000 µg/L, respectively, for 2,4-DNT and 49 µg/L and 7 to 20 000 µg/L, respectively, for 2,6-DNT. 3 Detection of the major isomers in surface and groundwaters, however, is relatively infrequent (1.1%-1.6% of samples positive) and when detected, the concentrations are low (mean <10 µg/L). 4
Interest in the toxicity of DNT has generally focused on the major isomers and has specifically focused on the potential carcinogenicity of DNT. Numerous studies, ranging from acute to chronic, have been conducted in several mammalian species with the major isomers and TG-DNT. These studies revealed generally similar toxicological effects. Clinical signs of neurotoxicity reported for 2,4-DNT included muscular incoordination, a characteristic widespread stiff legged gait, and rigid paralysis of the hind legs. 5,6 Associated central nervous system (CNS) lesions were noted in the dog. 7 Altered hematological parameters (anemia, Heinz bodies, and reticulocytosis) consistent with a mild regenerative anemia or methemoglobinemia were reported for 2,4-DNT, 2,6-DNT, and TG-DNT. 5,8,9 Exposure to the major isomers also resulted in impairment of the male reproductive system (testicular atrophy and aspermatogenesis) in rats, mice, and dogs. 5,7,8,10 Hepatic effects are perhaps the most extensively studied effects of DNTs. As a result of differences in the hepatocarcinogenicity reported in 3 bioassays conducted with 2,4-/2,6-DNT and TG-DNT, 7,9,11 a wide range of studies have been conducted to investigate the carcinogenic potential of the major isomers. 12 –15
In contrast to the breadth and depth of studies conducted on the major isomers, studies on the minor isomers are lacking. Toxicity data for the minor isomers are limited to in vitro and acute studies. No subacute, subchronic, or chronic mammalian toxicity studies have been conducted. To evaluate the subacute toxicity of the individual isomers of DNT, a repeated dose oral toxicity study was conducted in Sprague-Dawley rats.
Materials and Methods
These studies were conducted at the US Army Public Health Command in compliance with Good Laboratory Practice standards as described in Title 40, Code of Federal Regulations, Part 792.
Test Substances
Neat 2,4-DNT (CAS # 121-14-2; lot#03718EH; purity 99.0%), 2,3-DNT (CAS # 602-01-7; lot#04918JQ; purity 99.9%), 3,4-DNT (CAS # 610-39-9; lot#05713CA; purity 99.9%), and 2,6-DNT (CAS # 606-20-2; lot#08328CR; purity 99.8%) were purchased from Sigma-Aldrich (St Louis, Missouri). 2,5-DNT (CAS # 619-15-8; lot#15317-24; purity 99.3%) and 3,5-DNT (CAS # 618-85-9; lot#15317-8; purity 99.8%) were produced by SRI International (Menlo Park, California).
Test Animals
The study was conducted in 2 phases, testing 3 isomers in each phase. One hundred and twenty-six male Sprague-Dawley rats (43-45 days old, 151-175 g) were obtained from Charles River Laboratories, Wilmington, Massachusetts, for each phase. The animals were acclimatized for 7 days upon arrival. For each phase, animals were randomly distributed using the LABCAT randomization program into 6 treatment groups per isomer and 3 control groups (corn oil control, negative control, and positive control) consisting of 6 rats each. Rats were distributed into groups according to body mass the day before the start of dosing. To facilitate scheduling of necropsies, the animals were then divided into 6 experimental start dates, with 1 animal from each treatment group per start date. The rats were 7 weeks old and weighed 262 ± 29.7 and 260 ± 17.9 g at the start of the study for phases I and II, respectively; body mass did not differ between treatment groups at the start of the study. Housing conditions were maintained at 64°F to 79°F, 30% to 70% relative humidity with a 12-hour light/dark cycle. A certified pesticide-free rodent chow (Harlan Teklad, 8728C Certified Rodent Diet) and drinking quality water were available ad libitum. Rats were housed individually in suspended polycarbonate boxes with Harlan Sani-Chip bedding. Animal use procedures were approved by the Animal Care and Use Committee at the US Army Public Health Command (USAPHC). Animal care and use was conducted in accordance with the principles stated in the Guide for the Care and Use of Laboratory Animals 16 and in accordance with all applicable Federal and Department of Defense (DOD) regulations. The USAPHC animal care program is fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International.
Test Article Administration
Three solutions/suspensions in corn oil were prepared for each isomer and analyzed to verify concentration, homogeneity, and stability. The test articles were administered once daily for 14 days using a syringe attached to a 16-gauge gavage needle. Dosing volume was calculated based on the most recent body mass measurement. The corn oil control group was given an equivalent volume as the highest dose group for each phase (6.9 and 9 mL/kg, respectively). Dose levels were selected based on reported LD50 values and on toxicity observed in an acute range finding study and were set at a maximum of ½ or ¼ of the LD50. The test groups were as shown in Table 1.
Summary of Study Design
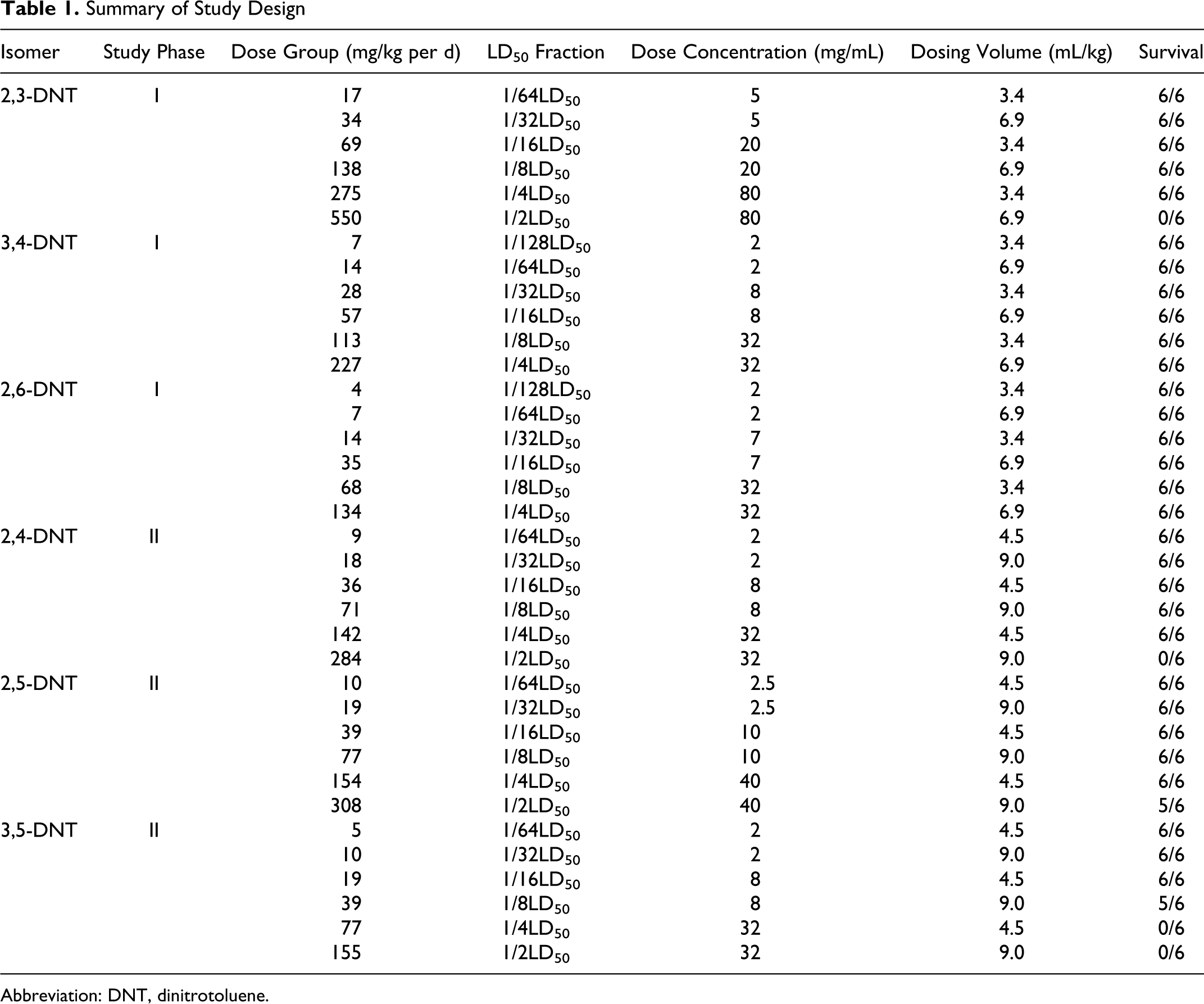
Abbreviation: DNT, dinitrotoluene.
Observations, Body Mass, Food Consumption
All animals were observed twice daily for signs of toxicity and morbidity. Body mass and food consumption were measured on days 0, 1, 3, 7, and 14.
Hematology and Blood Chemistry
Blood samples were collected from all animals prior to scheduled necropsy at the end of the study period via intracardiac puncture under carbon dioxide (CO2) anesthesia. For hematology analysis, a portion of the blood was treated with K3-EDTA and used to determine (Cell-Dyn 3700 Hematology Analyzer, Abbott Laboratories, Abbott Park, Illinois) the following parameters: white blood cell (WBC) count, WBC differential (% neutrophils [NEU %N], % lymphocytes [LYM %L], % monocytes [MONO %M], % eosinophils [EOS %E], % basophils [BASO %B]), red blood cell (RBC) count, hemoglobin (HGB), hematocrit (HCT), mean cell volume (MCV), mean cell hemoglobin (MCH), mean cell hemoglobin concentration (MCHC), red blood cell distribution width (RDW), platelet count (PLT), and mean platelet volume (MPV).
For clinical chemistry parameters, blood samples were treated with lithium heparin and centrifuged to obtain plasma for analysis (VetTest 8008 Chemistry Analyzer and VetLyte Na, K, Cl Analyzer, IDEXX Laboratories, Inc, Westbrook, Maine). The following parameters were measured: albumin (ALB), alkaline phosphatase (ALKP), alanine aminotransferase (ALT), aspartate aminotransferase (AST), blood urea nitrogen (BUN), calcium, cholesterol (CHOL), creatinine, glucose (nonfasting), globulin, lactate dehydrogenase, total bilirubin (TBIL), total protein (TP), sodium, potassium, and chlorine (Cl).
Necropsy
All animals were euthanized at the end of the study using CO2. Gross necropsies were completed on all terminal animals. The following organs were removed and absolute and relative (organ/body and organ/brain) organ weights were determined: brain, heart, kidneys, epididymides, liver, spleen, and testes. Bilateral organs were weighed together. Tissues were fixed and preserved in 10% buffered formalin.
Histopathology
Tissues were packaged and shipped to Comparative Biosciences, Inc (Sunnyvale, California) for processing. Cassettes containing tissues were processed through a graded series of alcohols using an automated processor, then oriented and embedded in paraffin, sectioned at approximately 3 to 5 µm thickness, stained with hematoxylin and eosin and coverslipped. Organs with apparent test article-related weight effects were selected for microscopic evaluation. The control group and all dose groups with weight effects were evaluated for each tissue for each isomer. Evaluation was performed by an American College of Veterinary Pathology board certified military veterinary pathologist. The severities of lesions per tissue were recorded as: grade 0 = normal, grade 1 = minimal/trace, grade 2 = mild, grade 3 = moderate, and grade 4 = severe. Specifically, mitotic frequency grading in the liver was determined by total mitotic figures counted within ten, ×20 powered fields (five ×20 fields were counted per liver section). Grades were recorded as: grade 0 = normal (0-9 mitoses/10 fields as determined by controls), grade 1 = trace (10-15 mitoses/10 fields), grade 2 = mild (16-20 mitoses/10 fields), grade 3 = moderate (21-30 mitoses/10 fields), grade 4 = severe (>30 mitoses/10 fields).
Statistical Analyses
Results are presented as means ± standard error of the mean for each treatment group. Statistical analyses were performed using SPSS 16.0 software. For body mass changes and food consumption over the study period, a repeated measures analysis of variance (ANOVA) with time as the repeated factor and dose group as between-group factor was used to assess changes in times and dose groups. One-way ANOVA was used to test for differences among treated and control groups. If the ANOVA showed significant differences, post hoc analysis was conducted using a Tukey multiple comparison test. All analyses were conducted within each phase. Statistical significance was defined as
Results
Clinical Observations
2,3-DNT
Clinical signs of toxicity, including cyanosis, lethargy, squinting, rapid respiration/labored breathing, hunched posture, dark urine, chromodacryorrhea, and yellow diarrhea were observed in the 550 mg/kg per d dose group. Signs of cyanosis were apparent within 1 hour of dosing and persisted for several hours but showed marked improvement or complete recovery after approximately 7 hours. All animals in the 550 mg/kg per d dose group died or were humanely sacrificed due to overt toxicity after approximately 2 (2.3 ± 0.82 days) days of dosing.
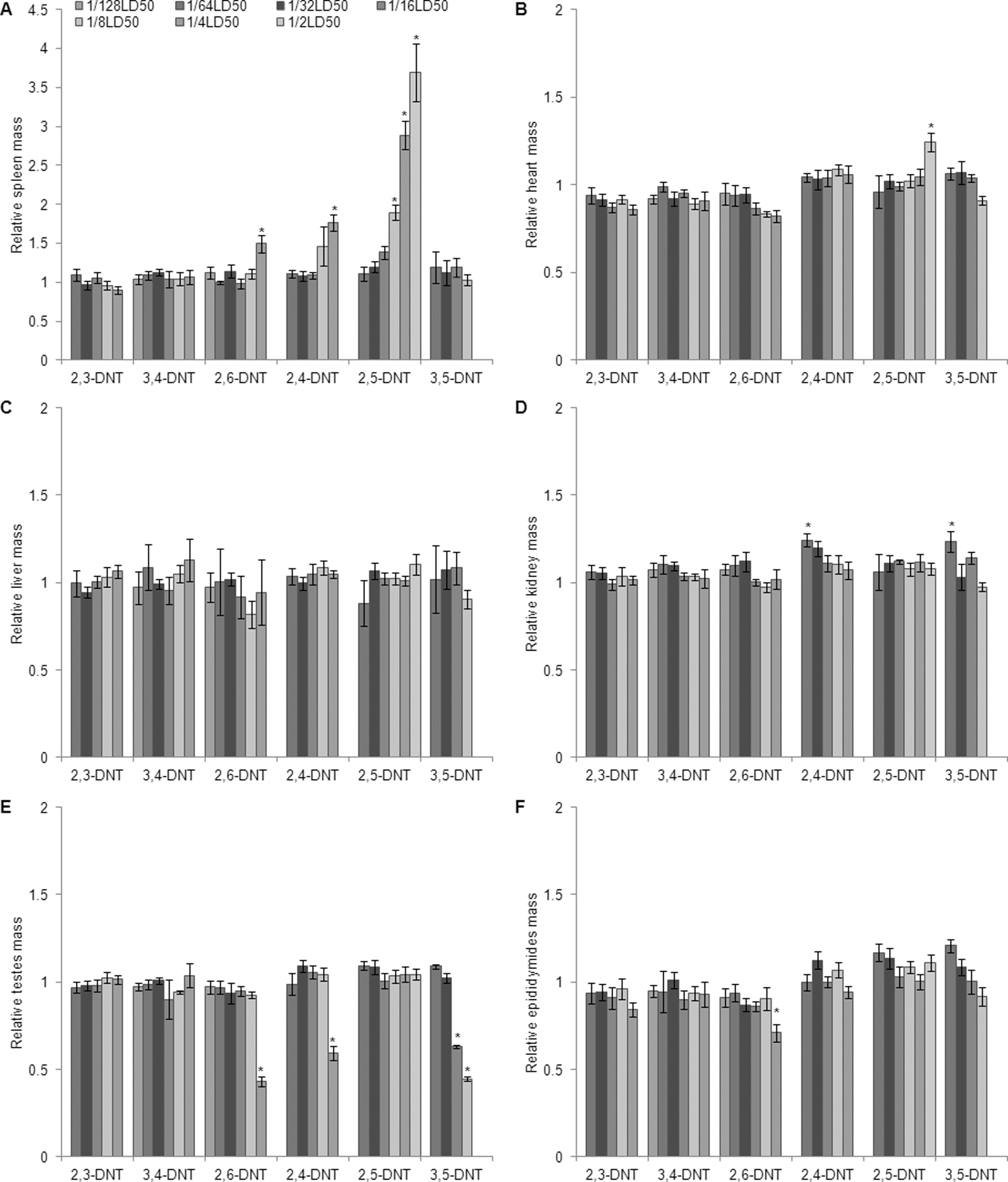
Fold change relative to controls in organ mass/brain mass in male Sprague-Dawley rats treated orally with isomers of DNT for 14 days. (A) spleen; (B) heart; (C) liver; (D) kidneys; (E) testes; (F) epididymides. Data are presented as means ± SEM for each dose group. The study was conducted in 2 phases; statistical analyses were conducted within phases between treated groups and their respective control group. Values marked with an asterisk (*) are significantly different (
3,4-DNT
Clinical signs of toxicity, including cyanosis, lethargy, dark urine, facial twitching, and hypoactivity/staring were observed in the 227 mg/kg per d dose group. Dark urine was observed in 1 rat dosed with 113 mg/kg per d.
2,6-DNT
No overt clinical signs of toxicity were observed in any dose group. Dark urine was observed on a single observation day in 2 rats dosed with 134 mg/kg per d.
2,4-DNT
Clinical signs of toxicity, including cyanosis, squinting, lethargy, prostrate posture, rapid respiration/labored breathing, hunched posture, dark urine, red discharge around nose/dried red material around nose, orange feces, and limb paralysis were observed in the 284 mg/kg per d dose group. All animals in the 284 mg/kg per d dose group died after approximately 3 (3.0 ± 1.5 days) days of dosing.
2,5-DNT
Clinical signs of toxicity, including cyanosis, squinting, prostrate posture, rapid respiration/labored breathing, hunched posture, dark urine, red discharge around nose/dried red material around nose, orange feces, and soft feces or diarrhea were observed in the 154 and 308 mg/kg per d dose groups.
3,5-DNT
Clinical signs of toxicity, including cyanosis, squinting, prostrate posture, rapid respiration/labored breathing, hunched posture, dark urine, red discharge around nose/dried red material around nose, orange feces, and soft feces were observed in the 77 and 155 mg/kg per d dose groups. In addition, animals in the 39, 77, and 155 mg/kg per d dose groups exhibited signs of neurological impairment that progressed rapidly from facial twitching, head bobbing, tilted head carriage, and uncoordinated movement, to loss of motor control, and paralysis of the thoracic limbs. All animals in the 77 and 155 mg/kg per d dose groups died or were humanely sacrificed after approximately 8 (8.0 ± 2.2 days) and 3 (2.8 ± 0.4 days) days of dosing, respectively.
Body Mass and Food Consumption
2,3-DNT
Body mass increased with time for all dose groups except the 550 mg/kg per d group, animals in this group lost body mass and died before the end of the study. Body mass and food consumption did not differ significantly among surviving treatment groups at any time point.
3,4-DNT
Body mass increased with time for all dose groups. Rats dosed with 227 mg/kg per d weighed less than all other dose groups at every time point, weighing 10% less than corn oil controls on day 14. However, this difference was not statistically significant. Food consumption was reduced in the 227 mg/kg per d dose group; however, food consumption did not differ significantly between treated and control groups at any time point.
2,6-DNT
Body mass increased with time for all dose groups. Rats dosed with 35, 68, and 134 mg/kg per d weighed less than all other dose groups starting at day 3 of dosing. At day 14, rats dosed with 35, 68, and 134 mg/kg per d weighed 10%, 15%, and 17% less than the rats in the corn oil control group, respectively. However, this body weight difference was only statistically significant (
2,4-DNT
Body mass increased with time for all dose groups except the 284 mg/kg per d group, animals in this group lost weight and died before the end of the study. At the end of the study, body mass in the 142 mg/kg per d dose group was reduced (
2,5-DNT
Body mass increased with time for all dose groups and did not differ between treated and control groups at any time during the study. Food consumption was reduced (
3,5-DNT
Body mass increased with time for all dose groups except for the 39, 77, and 155 mg/kg per d groups. Animals in the 77 and 155 mg/kg per d groups, lost weight and died before the end of the study, with the treated groups weighing (16% and 14%, respectively) less (
Hematology and Clinical Chemistry
Hematology results are shown in Tables 2 and 3. Statistically and/or biologically significant hematology and clinical chemistry changes related to treatment with DNTs are discussed below.
Hematology Results for Erythroid Parameters for Male Sprague-Dawley Rats Administered Isomers of DNT for 14 days
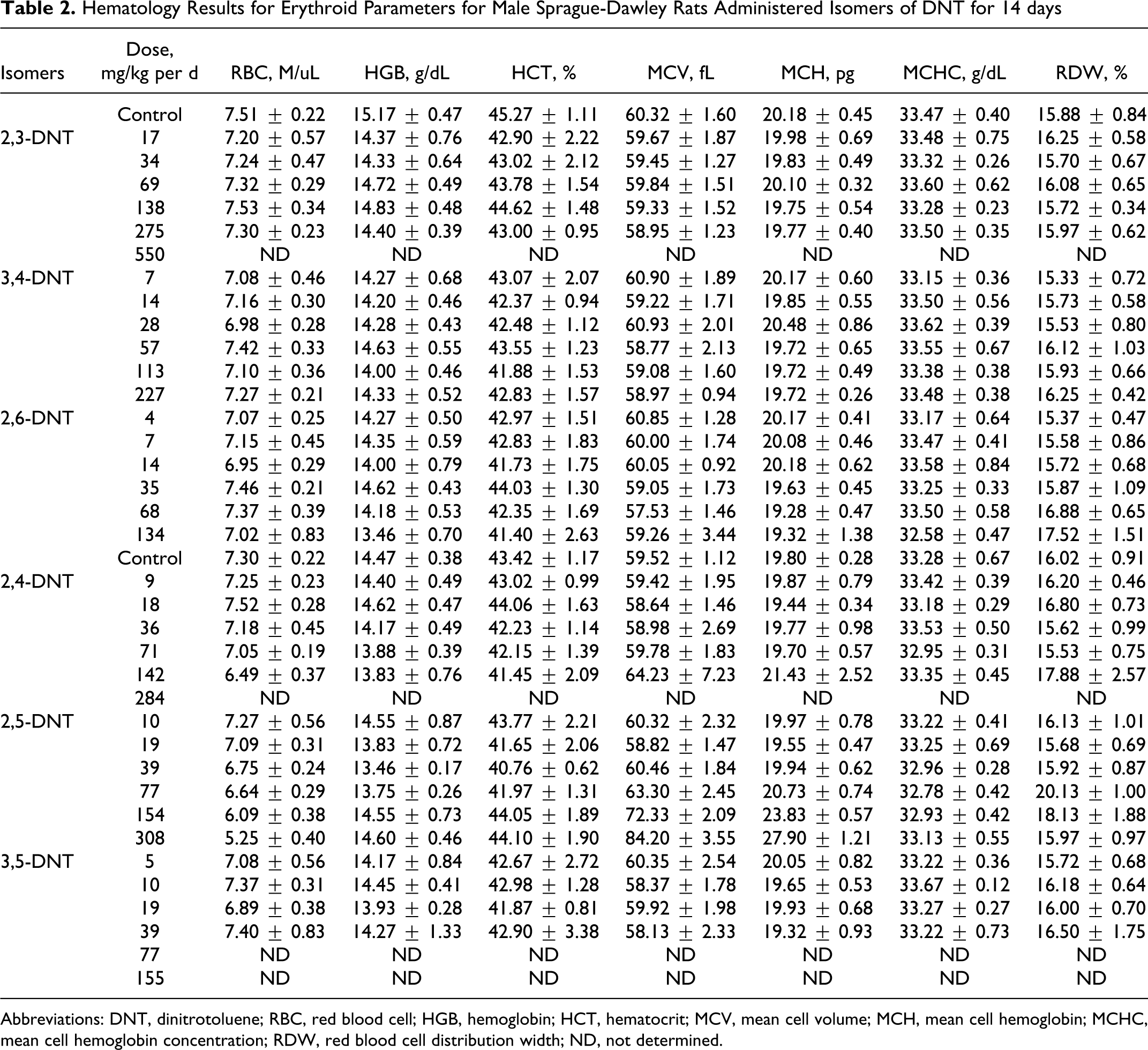
Abbreviations: DNT, dinitrotoluene; RBC, red blood cell; HGB, hemoglobin; HCT, hematocrit; MCV, mean cell volume; MCH, mean cell hemoglobin; MCHC, mean cell hemoglobin concentration; RDW, red blood cell distribution width; ND, not determined.
Leukocyte Count and 5-Part Differential Results for Male Sprague-Dawley Rats Administered Isomers of DNT for 14 days

Abbreviations: DNT, dinitrotoluene; WBC, white blood cell; NEU, neutrophils; LYM, lymphocytes; MONO, monocytes; EOS, eosinophils; BASO, basophil.
2,3-DNT
Erythroid parameters (RBC, HGB, HCT, MCV, MCH, and RDW) were not altered by treatment with 2,3-DNT, with values for all dose groups approximating those of the control group. Similarly, the WBC and differential leukocyte counts (NEU, LYM, MONO, EOS, BASO) did not differ significantly between 2,3-DNT-treated and control groups. Levels of ALB were lower (
Liver from a male Sprague-Dawley rat treated orally with 2,3-dinitrotoluene (DNT) for 14 days. Multifocally, hepatocytes are necrotic, characterized by shrunken, hypereosinophilic cells with pyknotic nuclei (large arrows). Multifocally necrotic cells contain deeply basophilic granular material, mineralization (*). Few hepatocytes contain lipid vacuoles (small arrows).
3,4-DNT
Both HGB and HCT were decreased (
2,6-DNT
Both HGB and HCT were reduced (
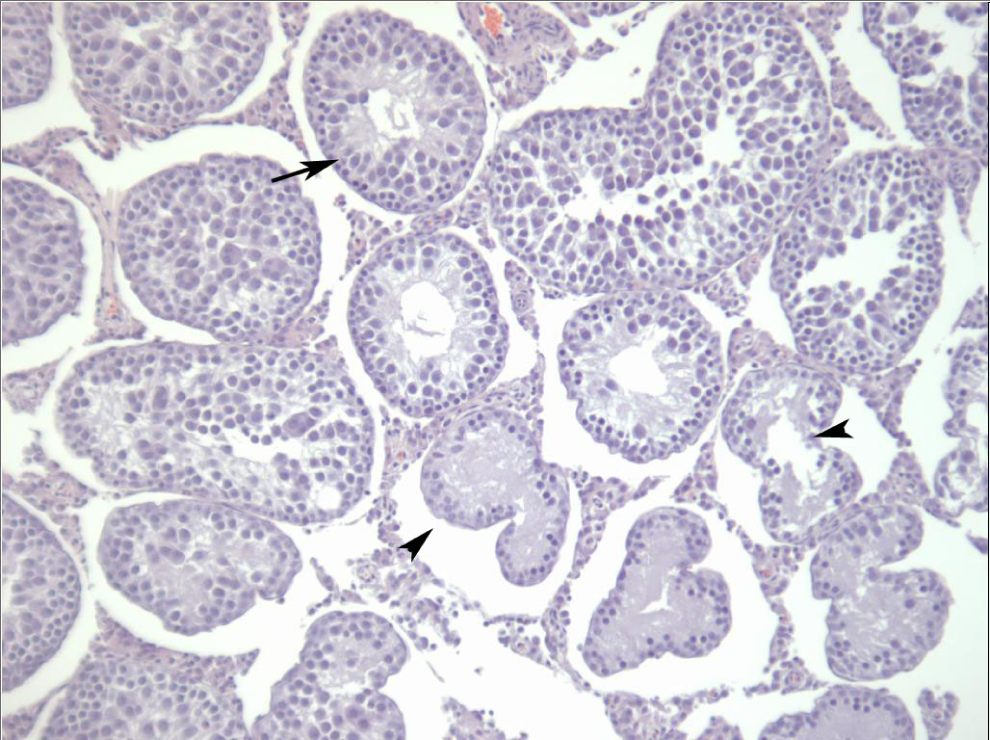
Testis from a male Sprague-Dawley rat treated orally with 2,6-dinitrotoluene (DNT) for 14 days. Seminiferous tubules are shrunken and have lost multiple germ cell layers (long arrows). Often tubules are atrophic and lined by a remaining cell layer of Sertoli cells (short arrows). All tubules within the photomicrograph are affected in varying degrees.
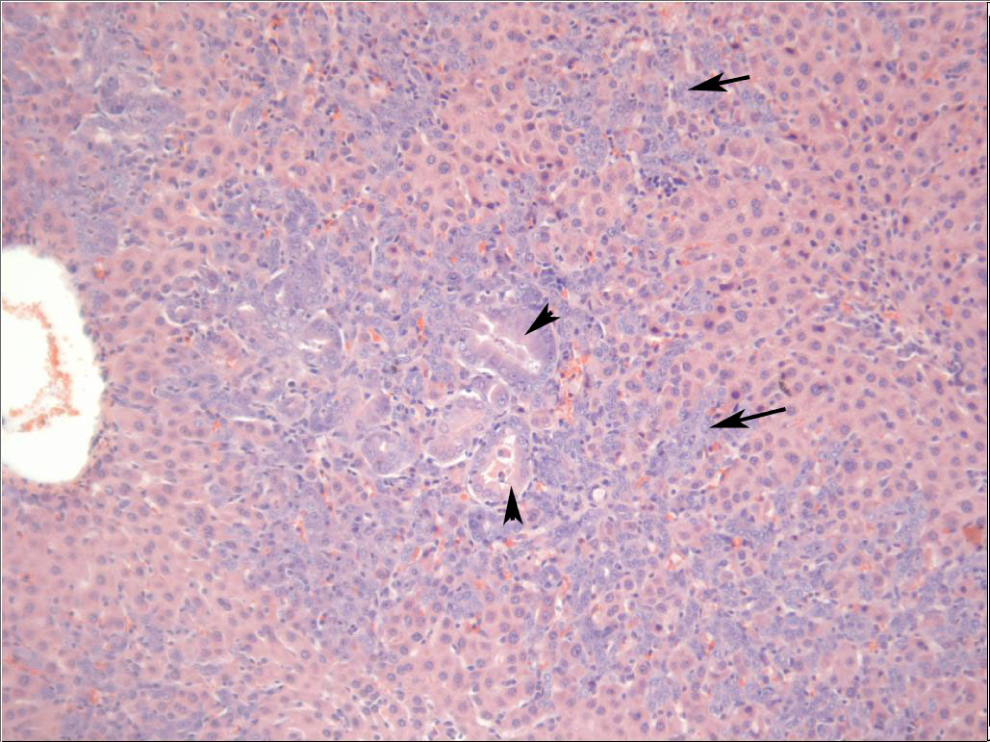
Liver from a male Sprague-Dawley rat treated orally with 2,6-dinitrotoluene (DNT) for 14 days. There is karyocytomegaly characterized by hepatocytes enlarged 10 to 20 times normal (arrows). Rarely, enlarged cells demonstrate mitotic figures (arrowhead).
2,4-DNT
The RBCs were reduced (
2,5-DNT
The RBCs decreased with increasing dose and were reduced (
3,5-DNT
Treatment with 3,5-DNT did not significantly alter the erythroid parameters (RBC, HGB, HCT, MCH, MCV, and RDW). Similarly, the WBC and differential leukocyte counts (NEU, LYM, MONO, EOS, and BASO) did not differ significantly between 3,5-DNT-treated and control groups. Levels of ALB, TBIL, and TP were elevated (
Necropsy
2,3-DNT
Animals in the 550 mg/kg per d dose group had mottled coloration of the liver, pinpoint sized pitting in the surface of the kidneys, and dark red–brown blood. One animal in each of the 17 and 275 mg/kg per d dose groups had a small cyst(s) on the kidney. A diaphragmatic hernia and a splenic abscess were noted in 2 animals in the 69 mg/kg per d dose group.
3,4-DNT
No macroscopic findings related to treatment were noted in any dose group.
2,6-DNT
One animal in the 7 mg/kg per d dose group had a small cyst on the right kidney. Small testes were noted for all the animals in the 134 mg/kg per d dose group.
2,4-DNT
Dark spleen was noted in 3 animals in the 142 mg/kg per d dose group. Small testes were noted in 2 animals in the 142 mg/kg per d group. A splenic cyst was noted in 1 animal in the 36 mg/kg per d dose group. A tracheal/esophageal mass was noted in 1 animal in the 9 mg/kg per d group. Hydronephrosis and mucous plugs of the bladder were noted in 1 control animal.
2,5-DNT
Dark, enlarged spleen with granular surface was noted in 4 animals in the 308 mg/kg per d dose group and all the animals in the 154 mg/kg per d group. Dark spleen was noted in 3 and 1 animals in the 77 and 39 mg/kg per d dose groups, respectively. Pale liver was noted in 2 animals in the 19 mg/kg per d dose group. Mucous plug of the bladder was noted in 1 high-dose animal.
3,5-DNT
Extremely full stomach with empty intestines was noted for animals in the 77 and 155 mg/kg per d dose groups that died preterm. Small testis size was noted in 5 animals in the 39 mg/kg per d group and three animals in the 19 mg/kg per d group.
Organ Mass
2,3-DNT
Kidney and liver-to-body mass ratios were increased (
3,4-DNT
Liver-to-body mass ratio was increased (
2,6-DNT
Kidney-to-body mass ratios were increased (
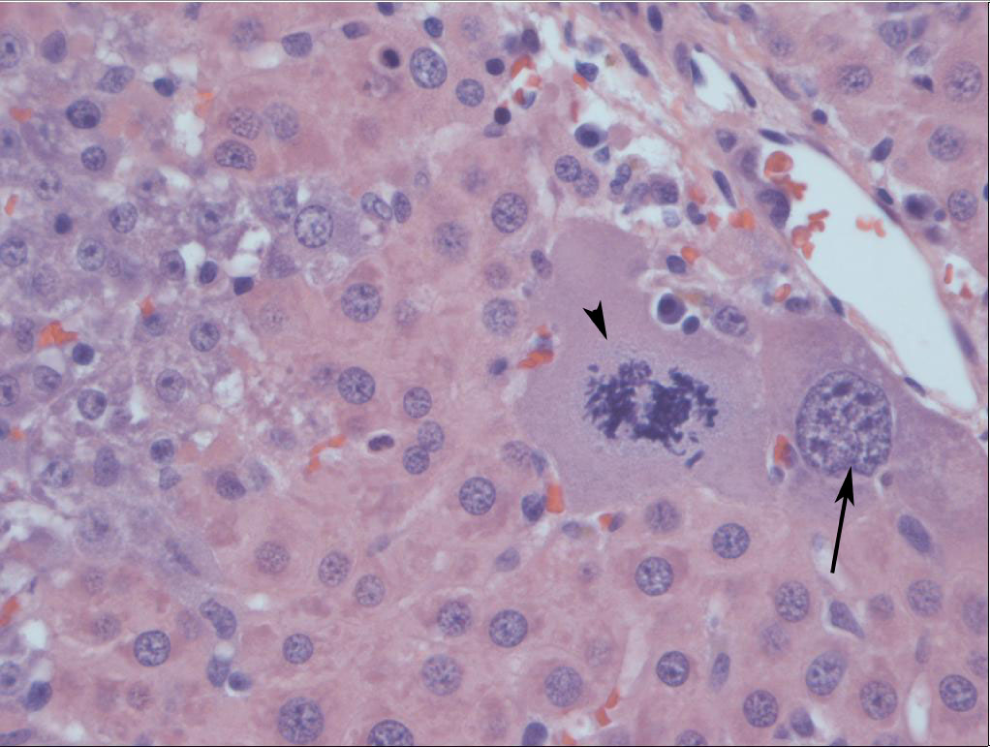
Liver from a male Sprague-Dawley rat treated orally with 2,6-dinitrotoluene (DNT) for 14 days. Separating and surrounding hepatocytes are numerous basophilic spindle cells, oval cells (arrows), that often form bile ducts (arrowheads).
2,4-DNT
Kidney mass, kidney to body mass, and kidney to brain mass were increased (
2,5-DNT
Heart mass and heart-to-body and brain mass ratios were elevated (
3,5-DNT
Testes mass and testes-to-body and brain mass ratios were reduced (
Histopathology
2,3-DNT
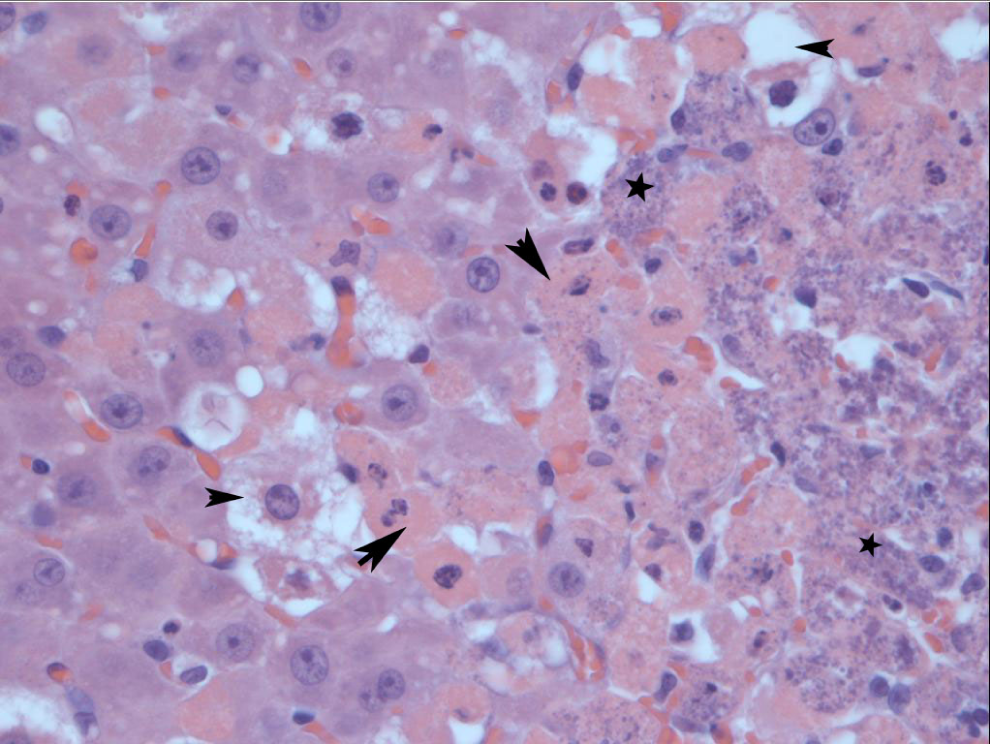
Trace-to-moderate bilateral degenerative changes in the testes characterized by tubular degeneration (1 of 5), multinucleated giant cell formation (2 of 5), and interstitial atrophy (5 of 5) were present in rats at 550 mg/kg per d. In the liver, trace mitotic activity was noted in the 34 mg/kg per d (1 of 6) and 69 mg/kg per d (1 of 5) groups. Additional liver lesions, considered adverse, were noted only in the highest dose group (550 mg/kg per d). Moderate-to-severe centrilobular necrosis was noted in 3 of 5 rats and was accompanied by mineralization. Additionally, individual necrotic hepatocytes were noted throughout the parenchyma of 3 of 5 rats (Figure 2). Trace-and-mild proximal tubule degeneration (3 of 5), mild tubular dilatation (3 of 5), moderate mineralization (2 of 5), and tubular necrosis (1 of 5) were present in the kidney of the 550 mg/kg per d group. Extramedullary hematopoiesis (EMH) was present in the spleen of 3 of 6 animals in the 275 mg/kg per d dose group. Lymphoid hyperplasia of the spleen was present in the 275 mg/kg per d (1 of 6) and 550 mg/kg per d (3 of 5) dose groups. Lymphoid depletion was present in the 275 mg/kg per d (1 of 6) and 550 mg/kg per d (1 of 5) dose groups (Figure 6).
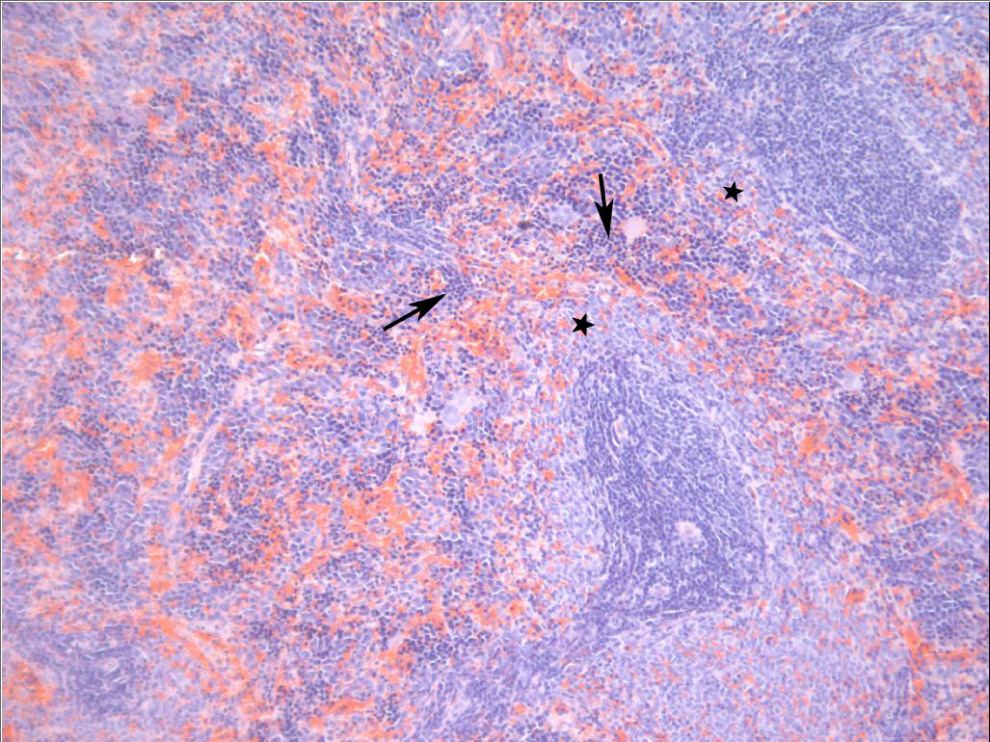
Spleen from a male Sprague-Dawley rat treated orally with 2,6-dinitrotoluene (DNT) for 14 days. Lymphocytes are decreased in the marginal zone (stars). Scattered throughout the red pulp is moderate extramedullary hematopoiesis characterized by cells with dark nuclei (arrows).
3,4-DNT
Mild myocardial fibrosis, inflammation, and necrosis were present in 1 of 5 animals from the high-dose group (227 mg/kg per d). In the spleen, trace-to-mild EMH was present in the 57 mg/kg per d (2 of 6), 113 mg/kg per d (2 of 5), and 227 mg/kg per d (3 of 6) groups. Trace-to-mild lymphoid hyperplasia was present in the 57 mg/kg per d (3 of 6), 113 mg/kg per d (2 of 5), and 227 mg/kg per d (2 of 6) groups. Lesions in the kidney consisted of trace-to-moderate proximal tubule degeneration (1 of 6), renal tubular basophilia (4 of 6), and lymphocytic infiltration (5 of 6) animals in the high dose group (227 mg/kg per d).
2,6-DNT
Trace-to-severe degenerative changes characterized by tubular degeneration (4 of 6 and 6 of 6), multinucleated giant cell formation (4 of 6 and 4 of 6), and interstitial atrophy (3 of 6 and 6 of 6) were noted in the testes of rats from the 2 highest dose groups (68 and 134 mg/kg per d, respectively) (Figure 5). Trace-to-moderate hepatocellular hyperplasia was present in 1 of 6, 3 of 6, and 4 of 6 rats at 35, 68, and 134 mg/kg per d, respectively. Trace-to-severe oval cell hyperplasia was present in 1 of 6, 5 of 6, and 6 of 6 rats at 35, 68, and 134 mg/kg per d, respectively, with severity increasing with increasing dose (Figure 3). Hepatocellular hypertrophy was also present as trace to mild in the 35 mg/kg per d (6 of 6), mild to moderate in the 68 mg/kg per d (4 of 6), and trace to mild in the 134 mg/kg per d (4 of 6) dose groups. Mild to severe increases in mitotic activity were observed in 4 of 6 and 5 of 6 rats in the 68 and 134 mg/kg per d dose groups, respectively. Trace-to-moderate single cell necrosis was also observed in the 68 mg/kg per d (5 of 6) and 134 mg/kg per d (5 of 6) dose groups. Karyocytomegaly was noted as trace in 3 of 6 of the 68 mg/kg per d group and mild to trace in 3 of 6 and moderate in 1 of 6 animals in the 134 mg/kg per d group (Figure 4). Trace-to-mild proximal tubule degeneration and renal tubular basophila were present in 4 of 6 animals in the high-dose group (134 mg/kg per d). Trace-to-mild lymphoid hyperplasia was noted in 2 of 5, 1 of 6, and 1 of 6 rats in the 35, 68, and 134 mg/kg per d dose groups. Mild-to-moderate EMH was present in the 68 mg/kg per d (1 of 6) and 134 mg/kg per d (6 of 6) groups (Figure 6). Lymphoid depletion was noted in 1 of 6 rats in the highest dose group (134 mg/kg per d).
2,4-DNT
Moderate-to-severe degenerative changes characterized by tubular degeneration (6 of 6 and 1 of 1), multinucleated giant cell formation (3 of 6 and 0 of 1), and interstitial atrophy (3 of 6 and 1 of 1) were noted in the testes of rats in the 2 highest dose groups (142 and 284 mg/kg per d, respectively). In the liver, trace-to-mild single cell necrosis was noted in the 18 mg/kg per d (1 of 6), 36 mg/kg per d (3 of 6), 71 mg/kg per d (1 of 6), and 142 mg/kg per d (4 of 6) groups. Trace apoptosis was noted in 1 of 6 rats in each of the 36 and 71 mg/kg per d dose groups. In the spleen, mild to moderate increases in EMH were noted in the 36 mg/kg per d (1 of 6), 71 mg/kg per d (3 of 6), and 142 mg/kg per d (6 of 6) groups. Lymphoid hyperplasia was present in the 36 mg/kg per d (1 of 6) and 71 mg/kg per d (3 of 6) groups. Lymphoid depletion was present in 1 of 1 rats examined at 284 mg/kg per d.
2,5-DNT
In the spleen, trace-to-mild EMH was present in the 39 mg/kg per d (3 of 6), 77 mg/kg per d (5 of 5), 154 mg/kg per d (6 of 6), and 308 mg/kg per d (5 of 5) groups. Mild-to-moderate fibrosis (4 of 5) and trace-to-moderate inflammation (2 of 5) were present in rats in the 308 mg/kg per d group.
3,5-DNT
Bilateral degenerative changes characterized by tubular degeneration, multinucleated giant cell formation, and interstitial atrophy were present in rats at 19, 39, 77, and 155 mg/kg per d. Mild-to-severe tubular degeneration was present in all rats at 19, 39, 77, and 155 mg/kg per d. Trace-to-severe multinucleated giant cell formation was present in the 19 mg/kg per d (6 of 6), 39 mg/kg per d (5 of 6), 77 mg/kg per d (3 of 3), and 155 mg/kg per d (2 of 3) groups. Mild-to-severe atrophy was present in the 77 mg/kg per d (3 of 3) and 155 mg/kg per d (2 of 3) groups. In the spleen, trace-to-mild EMH was present in the 77 mg/kg per d (1 of 3) and 155 mg/kg per d (1 of 3) groups. Trace lymphoid hyperplasia was present in 2 of 6 rats at 19 mg/kg per d. Trace-to-mild lymphoid depletion was present in the 10 mg/kg per d (1 of 6), 19 mg/kg per d (1 of 6), and 77 mg/kg per d (2 of 3) groups. Inflammatory infiltrates were present in the brains of 3 of 6 rats at 39 mg/kg per d (1 of 6 mild, 2 of 6 moderate) and moderate infiltrates in 2 of 3 rats at 77 mg/kg per d (Figure 7).
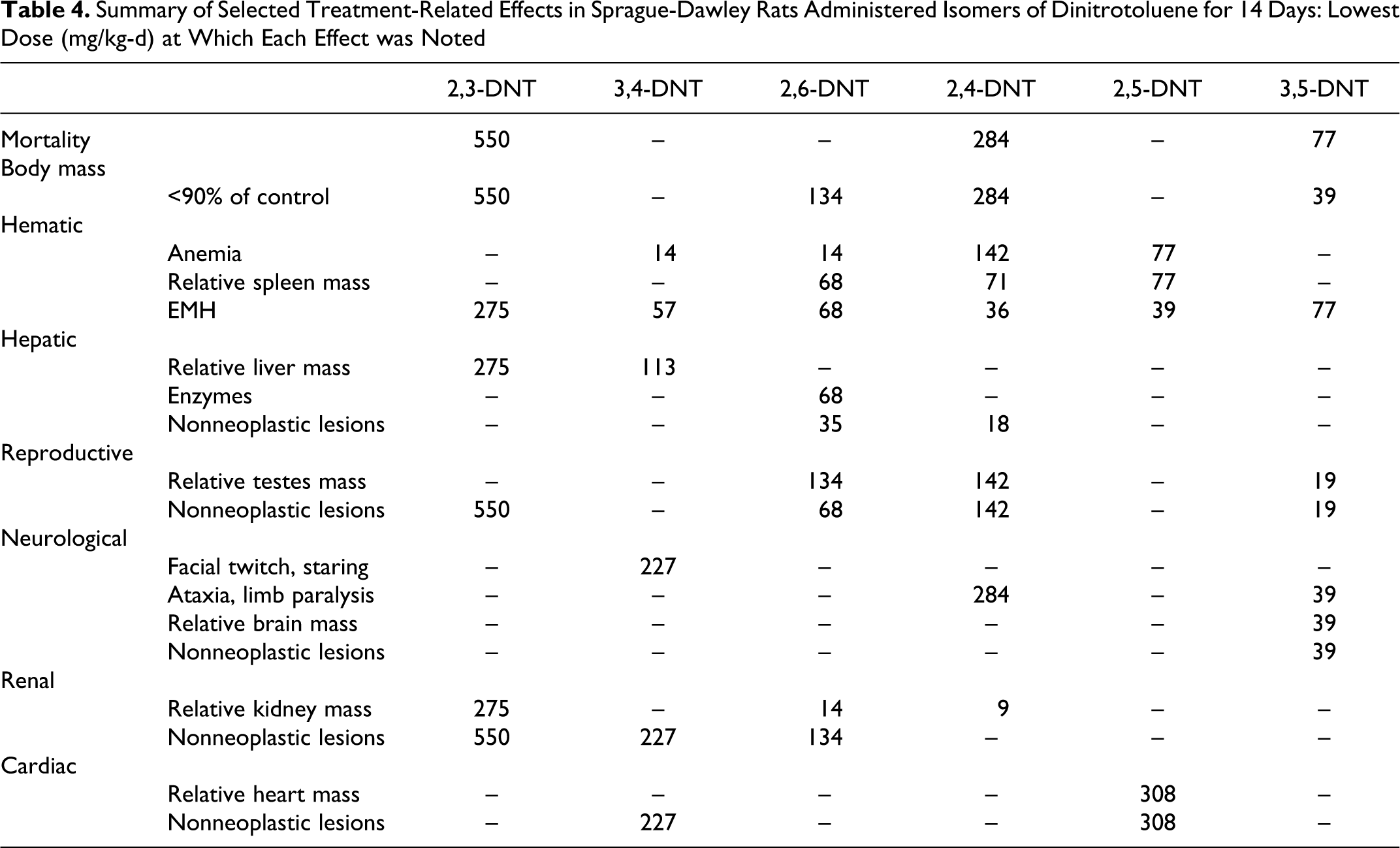
Summary of Selected Treatment-Related Effects in Sprague-Dawley Rats Administered Isomers of Dinitrotoluene for 14 Days: Lowest Dose (mg/kg-d) at Which Each Effect was Noted
Discussion
In this study, isomers of DNT caused an overall pattern of toxicity in several organ systems that was qualitatively similar to the toxicity reported for TG-DNT, with effects being noted in the blood, spleen, liver, testes, brain, kidney, and heart. The organ systems affected, the pattern of lesions and the dose level at which effects were observed, however, differed among the isomers (Table 4).
The 3,5-DNT isomer was the most toxic when administered repeatedly, with weight loss and mortality occurring at 39 mg/kg per d which was one eighth of the LD50. Weight loss and mortality were also observed for the 2,3- and 2,4-DNT isomers at ½ the LD50 (550 and 285 mg/kg per d, respectively). Repeated administration of the 2,6-DNT isomer up to one sixteenth the LD50 (35 mg/kg per d) resulted in reduced weight gain. Mortality and weight loss/reduced weight gain were not observed for 3,4-DNT and 2,5-DNT up to ¼ (227 mg/kg per d) and ½ (308 mg/kg per d) the LD50, respectively.
Hematological effects associated with anemia were observed for all isomers. Although the most apparent and extreme effect was the severe cyanosis in the 2,5-DNT isomer at doses up to 77 mg/kg per d, anemia was observed at lower doses for other isomers. Mild anemia was seen in both the 3,4-DNT and 2,6-DNT isomers at 14 mg/kg per d. The presence of cyanosis and anemia are consistent with methemoglobinemia reported to occur following exposure to DNTs. 17 Increased spleen mass was observed for 2,5-DNT, 2,4-DNT, and 2,6-DNT. The EMH was noted for all isomers, occurring at the lowest doses for 2,5-DNT and 2,4-DNT. The increase in spleen size and EMH of the spleen suggest enhanced red cell turnover and a compensatory response to anemia. Increased reticulocyte production was also noted for 2,5-DNT, 2,4-DNT, and 2,6-DNT,18 further indicating a compensated anemia. Although all isomers were hematotoxic, the severity differed greatly among isomers such that the toxicity of 2,5-DNT > 2,4-DNT > 3,4-DNT > 2,6-DNT > 3,5-DNT > 2,3-DNT. The DNTs or their metabolites produce methemoglobin by oxidizing the ferrous ion in HGB. An intermediate in the reduction of nitros to amines, hydroxylamine, has been proposed as a likely oxidizing species. 7 Isomeric differences in hematotoxicity may be due to differences in production of reduced metabolites. Reduction potentials of DNT isomers indicate that 2,5-DNT is the easiest to reduce; however, 2,6-DNT is the most difficult to reduce, indicating that ease of reduction does not fully explain the isomeric differences. 19
The DNTs are commonly reported to be hepatotoxic, demonstrating both proliferative and degenerative lesions, based on studies with TG-DNT, 2,4-DNT, and 2,6-DNT. 7,9,11 The present study, however, demonstrated marked differences in the hepatotoxicity of the isomers. Two isomers, 2,5-DNT and 3,5-DNT, were shown to be nonhepatotoxic. The 3,4-DNT isomer showed only relative organ weight effects at 113 and 227 mg/kg per d. Hepatotoxicity (centrilobular necrosis) was noted in animals in the 550 mg/kg per d 2,3-DNT group, all of which died after 2 to 4 days of dosing. Although 2,3-DNT was hepatotoxic in the highest dose group, treatment-related hepatic lesions were not noted in lower dose groups. Hepatotoxicity was most pronounced for 2,4-DNT and 2,6-DNT, with altered histopathology at 18 and 35 mg/kg per d, respectively. Although changes were observed in the present study for 2,4-DNT at doses as low as 18 mg/kg per d, the lesions were limited to single cell necrosis and apoptosis. These results are not atypical for 2,4-DNT as subacute and subchronic exposures have previously failed to demonstrate hepatic lesions, with evidence of hepatotoxic effects being limited to increases in CHOL and ALT levels. 6,7 In contrast, severe lesions associated with hepatocellular proliferation and neoplasms were noted for 2,6-DNT at doses down to 35 mg/kg per d. Similar hepatocellular lesions have been reported for 2,6-DNT following subacute exposures. 8 Subchronic and chronic exposure at the same dose (35 mg/kg per d) resulted in hepatocellular carcinomas. 15
Isomer-specific hepatocarcinogenic effects of DNTs have been the focus of considerable study as a result of conflicting results obtained in 3 bioassays conducted with varying mixtures of TG-DNT and 2,4-DNT/2,6-DNT. Initial investigations conducted in vitro conflicted with the known hepatocarcinogenicity of DNT as isomers were not genotoxic and did not induce morphological transformation in mammalian cells. 5 –8,20 –22 In contrast, in vivo assessment of the genotoxicity of DNT using the in vivo–in vitro hepatocyte DNA repair assay indicated that TG-DNT is a potent genotoxic agent, with 2,6-DNT being primarily responsible for this effect. 23 –25 These results were interpreted as indicating that 2,6-DNT activation in vivo requires processes other than hepatic metabolism. Subsequent work with axenic rats demonstrated that metabolism by gut flora is necessary for the formation of carcinogenic metabolites of DNT. 5,27 The 2,6-DNT isomer was further demonstrated to be a complete liver carcinogen in in vivo hepatic initiation–promotion assays. 12 –15 The 2,4-DNT isomer demonstrated only weak promoting activity and the minor isomers demonstrated no initiating activity. 12 –14 In assays conducted in concert with this study, the 2,6-DNT isomer was the only isomer shown to be genotoxic in the liver Comet assay.18
Taken together, these data suggest that although hepatotoxicity may be indicated for several isomers, severe hepatotoxicity is limited to the major isomers, with hepatocarcinogenicity being limited to the 2,6-DNT isomer. This is in contrast to in vitro results which indicated the ortho and para substituted isomers are more hepatotoxic than the meta-substituted isomers. 20 The in vitro work suggested a relationship between ease of reduction of substituent nitro groups and hepatotoxicity. The in vitro study suggests that metabolism of DNT isomers by gut microflora is not necessary to induce hepatotoxicity. It remains, however, that the metabolism of DNTs in vivo includes both oxidative and reductive metabolism, resulting in different metabolic profiles and different exposure scenarios in vivo versus in vitro. In vitro assays have been labeled as not reflective of the carcinogenicity of DNTs due to the enterohepatic circulation necessary to produce the active metabolite. 22 It may be that in vitro assays are similarly not reflective of hepatotoxicity due to differential toxicity of the metabolites produced.
Effects on the reproductive system including reduced testes mass and testicular atrophy were noted for 2,3-DNT, 2,6-DNT, 2,4-DNT, and 3,5-DNT. Testicular effects, including testicular degeneration, atrophy, altered Sertoli cell morphology, and oligospermia have been reported previously following exposure to 2,4-DNT and 2,6-DNT. 5 –8 These testicular effects have been reported to be due to direct toxic effects on Sertoli cell morphology. However, effects on serum luteinizing hormone and follicle-stimulating hormone also suggest that DNT affects pituitary function, which could attribute at least some of the testicular effects to hormone-mediated causes. 10 In vitro work with 4 isomers of DNT suggests that the isomers are Sertoli cell toxicants. 28 The in vitro work, however, indicates a ranked Sertoli cell toxicity of 3,4-DNT > 2,3-DNT > 2,4-DNT ≥ 2,6-DNT which is in contrast to the present in vivo testicular toxicity of 3,5-DNT > 2,6-DNT > 2,4-DNT > 2,3-DNT. This contrast suggests that the effects on the testes are likely not limited to direct effects on the Sertoli cells. As was evident in the hepatocarcinogenicity work, differences between the in vitro and in vivo results may also be due to differences in metabolism. Isomeric differences in testicular effects may also be due to differences in metabolism. Dinitrobenzene (DNB), a structurally similar nitroaromatic compound, demonstrates a similar pattern of isomer specificity, with testicular effects being found only with the meta isomer. 29 For DNB, a series of in vitro and in vivo studies with parent compounds and metabolites demonstrated that the parent compound was the testicular toxicant. The lack of testicular toxicity of the ortho and para isomers was attributed to their more complete metabolism via glutathione conjugation and nitroreduction at extragonadal sites. 29 In the present study, similar to DNB, the meta isomers exhibited testicular toxicity while the ortho and para isomers were largely nontoxic. The theory that ortho and para DNTs are not testicular toxicants as a result of extragonadal metabolism leading to detoxification warrants study. The testicular toxicity observed in the high dose of 2,3-DNT may have resulted from the associated hepatotoxicity and a reduced ability to fully metabolize the parent compound leading to toxic concentrations in the testes. Additionally, 2,3-DNT-associated reductions in weight may have also affected testicular histology. 30
Neurotoxic effects have been reported previously for 3,4-DNT, 2,4-DNT, and 2,6-DNT. 5,6,8,17 In the present study, effects were noted for 3,4-DNT, 2,4-DNT, and 3,5-DNT. Convulsions have been reported following acute administration of 3,4-DNT. 17 Similar results were noted for this isomer in a range finding study at 1000 mg/kg. When administered repeatedly at doses as high as 227 mg/kg per d, no convulsions were noted; however, hypoactivity/staring similar to an absence seizure was observed. For 2,4-DNT, the signs of neurotoxicity were consistent with previous reports of muscular incoordination and rigid paralysis of the hind legs. 7 The clinical signs of neurotoxicity reported for 3,5-DNT differed from those reported for the other isomers in the addition of facial twitching, altered head carriage and that paralysis was restricted to the front limbs. Lesions of the cerebellum (inflammatory infiltrates: gliosis and astrocytosis with gitter cells and spongiosis) were also noted for 3,5-DNT but differed from the cerebellar lesions previously noted in dogs given 2,4-DNT (vacuolization, hypertrophy, endothelial mitosis, focal gliosis, and perivascular hemorrhage; Figure 7). 7 Similar neurological signs and brain lesions have been reported in the rat following exposure to other nitroaromatic compounds. Exposure to 1,3-DNB causes ataxia as well as formation of gliovascular lesions in the brain stem, particularly the nuclei of the auditory pathway, vestibular system, and deep cerebellar roof nuclei. 31 1,3,5-Trinitrobenzene (TNB) and nitrobenzene (NB) produce similar histological, neuroanatomic, and clinical findings; however, the required dose and timing of onset of the lesions varied. Brain lesions were apparent in NB- and 1,3-DNB-treated rats within 24 hours of exposure. Whereas in TNB-treated rats, 10 days of exposure to 71 mg/kg per d was required to produce lesions; lower doses and shorter durations did not induce lesions. Similar results were observed for 3,5-DNT in which neurological signs were first noted in the 155 mg/kg per d group after 3 days, in the 77 mg/kg per d group after 7 days, and in the 39 mg/kg per d groups after 12 days, suggesting a time–concentration threshold for neurotoxicity similar to that observed for 1,3-DNB. 33 The 1,3-DNB neurotoxicity occurs via nitroreduction, primarily in endothelial cells of the CNS, resulting in production of reactive intermediates and induction of oxidative stress. 31 A similar mechanism may be possible for DNTs given that nitroaromatic compounds undergo redox cycling to form free radicals. However, little is known about the mechanism of DNT-induced neuropathology.
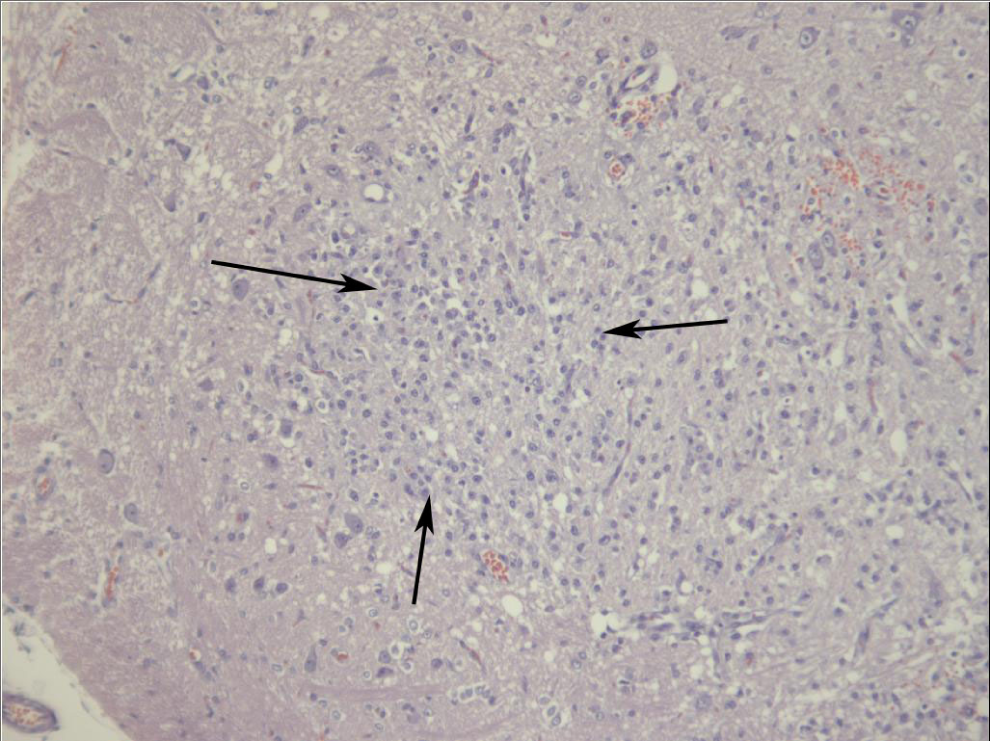
Cerebellum from a male Sprague-Dawley rat treated orally with 3,5-dinitrotoluene (DNT) for 14 days. There is a focal area of increased cellularity within the grey matter (arrows bound an area of increased cellularity).
Limited treatment-related effects were noted for individual isomers in the kidney and heart. The kidney has been reported to be an insensitive target for DNT toxicity as reported renal effects have been limited and primarily restricted to 2,4-DNT in mice and 2,6-DNT in dogs. 7,8 Renal effects were noted for 3 isomers in this study; relative organ weight and microscopic lesions were observed for the 2,3-DNT, 3,4-DNT, and 2,6-DNT isomers. With the exception of the weight effect in 2,6-DNT, these effects were limited to the highest dose groups, making the kidney less sensitive than other affected organ systems. Cardiac effects were limited to 2 isomers, 3,4-DNT and 2,5-DNT. Effects for 3,4-DNT were limited to a single animal. With the exception of myocarditis observed after subchronic exposure to TG-DNT, 9 histopathological effects on the cardiovascular system have not been noted for DNTs. The myocardial damage observed for 3,5-DNT was likely a result of the intermittent hypoxia experienced by these animals. 34
In summary, the 3,5-DNT isomer was the most toxic isomer in a repeated dose setting. Overt toxicity was generally highest for the meta-substituted isomers followed by the ortho- and was lowest for the para-substituted isomer. The characteristic effects associated with each isomer were neurotoxic effects in 3,4-DNT and 3,5-DNT, severe cyanosis and anemia in 2,5-DNT, hepatotoxicity in 2,6-DNT, a combination of effects in 2,4-DNT, and limited toxicity in 2,3-DNT.
Footnotes
Acknowledgments
The authors gratefully acknowledge the following people for the assistance: Matt Bazar, Patricia Beall, Shenell Bolden, LTC Kenneth Despain, Mike Hable, Terry Hanna, John Houpt, Craig McFarland, Will McCain, Curtis Oliver, Arthur O’Neill, and Mark Way.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: funded by the US Army Environmental Center Installation Restoration Program (MIPR numbers MIPRMIPR8BDAT48114, 2008 and MIPR9ADAT48134, 2009).
