Abstract
Methods for the diagnosis of parasitic infections have stagnated in the past three decades. Labor-intensive methods such as microscopy still remain the mainstay of several diagnostic laboratories. There is a need for more rapid tests that do not sacrifice sensitivity and that can be used in both clinical settings as well as in poor resource field settings. The fields of diagnostic medical parasitology, treatment, and vaccines are undergoing dramatic change. In recent years, there has been tremendous effort to focus research on the development of newer diagnostic methods focusing on serological, molecular, and proteomic approaches. This article examines the various diagnostic tools that are being used in clinical laboratories, optimized in reference laboratories, and employed in mass screening programs.
Introduction
Parasitic organisms are the causative agents of some of the world’s most devastating and prevalent infections. This group of pathogens includes members such as the protozoans Trypanosoma (Chagas disease and African sleeping sickness), Leishmania (leishmaniasis), Plasmodium (malaria), and the helminths such as Schistosoma (schistosomiasis), Wuchereria (filariasis), and Echinococcus (echinococcosis), among others. Many of these infections have always been described as being tropical or subtropical. However, the increase in international travel as well as the arrival of new immigrants has made some of these tropical diseases realities in developed countries as well. In addition, a new trend arose; individuals, who never traveled to endemic areas were being infected by “tropical” blood-borne protozoans. 1 This unusual observation could be traced back to blood transfusions and organ transplants. Asymptomatic carriers migrate and become part of the blood bank donor and transplant donor populations. Quick diagnosis has always been a priority to determine the appropriate treatment and prevent fatalities. In addition, now more than ever, advances in diagnostics can help prevent transmission and provide active surveillance. Unfortunately, there have been few major advances in diagnostic methods for parasitic infections. Efforts have stagnated, and the majority of definitive diagnoses still rely on labor-intensive and time-consuming methods such as microscopy. To have the most significant diagnostic impact, new techniques and assays should be simple and yield rapid results. Such characteristics could be achieved by reducing the number of steps to be performed in a procedure and making result interpretation obvious enough to avoid significant operator-dependent biases. An optimal diagnosis method would possess these favorable features while still maintaining a high level of sensitivity and specificity. Moreover, many of the tests that are used today cannot differentiate between current and past infections. Assays that are capable of making this distinction are necessary to properly determine disease prevalence, choose the appropriate treatment, and assess the effect of treatment. Currently, diagnostic and reference laboratories use several techniques, including microscopy, molecular assays, and serological assays. Each method has its advantages as well as disadvantages. Furthermore, many research laboratories are focusing on the development of new diagnostic methods as well as the improvement of old ones. There has especially been a focus on the development of molecular diagnostic techniques.2,3 Real-time polymerase chain reaction (PCR) procedures for the detection of various parasites are continuously being optimized. Recently, loop-mediated isothermal amplification (LAMP) has attracted much attention and seems to be the molecular tool of the future.4,5 The development of multiplex real-time PCR protocols has also been emphasized. These assays have the ability to detect mixed infections simultaneously. 6 Serology-based techniques have been complementing microscopy for many years; however, for many infections, they still cannot fully replace microscopic diagnosis. These assays rely on antigen or antibody detection from the provided patient samples. Rapid diagnostic tests have become some of the most popular serology-based assays.7–10 They are the focus of much research due to their potential to be used in field settings. Furthermore, major diagnostic advances have been made by the development of rapid tests that can detect mixed infections.3,11–14 Like the multiplex PCR procedures, these tests are of great relevance because they address the growing problem of parasitic coinfections in several regions. New and innovative diagnostic tools are constantly being tested; however, many of these methods still require optimization and standardization. The aim of this review is to highlight the various tools currently being used, as well as those under investigation, for the diagnosis of parasitic infections. For convenience, the various diagnostic tools that are presently in use and under investigation as well as those used in the past are summarized in different tables. Tools to diagnose protozoan infections, both blood borne and intestinal, are summarized in Tables 1 and 2 , respectively. Methods for diagnosis of helminth infections are summarized in Table 3 .
Diagnostic Tools for the Detection of Blood-Borne Protozoan Infections.

ELISA, enzyme-linked immunosorbent assay; ICT, immunochromatographic; PCR, polymerase chain reaction; NASBA, nucleic acid sequence–based amplification; CSF, cerebral spinal fluid; IIF, indirect immunofluorescent; ISAGA, immunosorbent agglutination assay; CATT, card agglutination test for trypanosomiasis; LAMP, loop-mediated isothermal amplification; LATEX; rapid latex agglutination test; SELDI-TOF, surface-enhanced laser desorption ionization time-of-flight mass spectrometry; MALDI-TOF, matrix-assisted laser desorption ionization time-of-flight mass spectrometry; IHA, indirect hemagglutination; TESA, trypomastigote excreted-secreted antigens; MS-MS, tandem mass spectrometry; RDT, rapid diagnostic test; LDMS, laser desorption mass spectrometry; RBC, red blood cell.
Diagnostic Tools for the Detection of Intestinal Protozoan Infections.
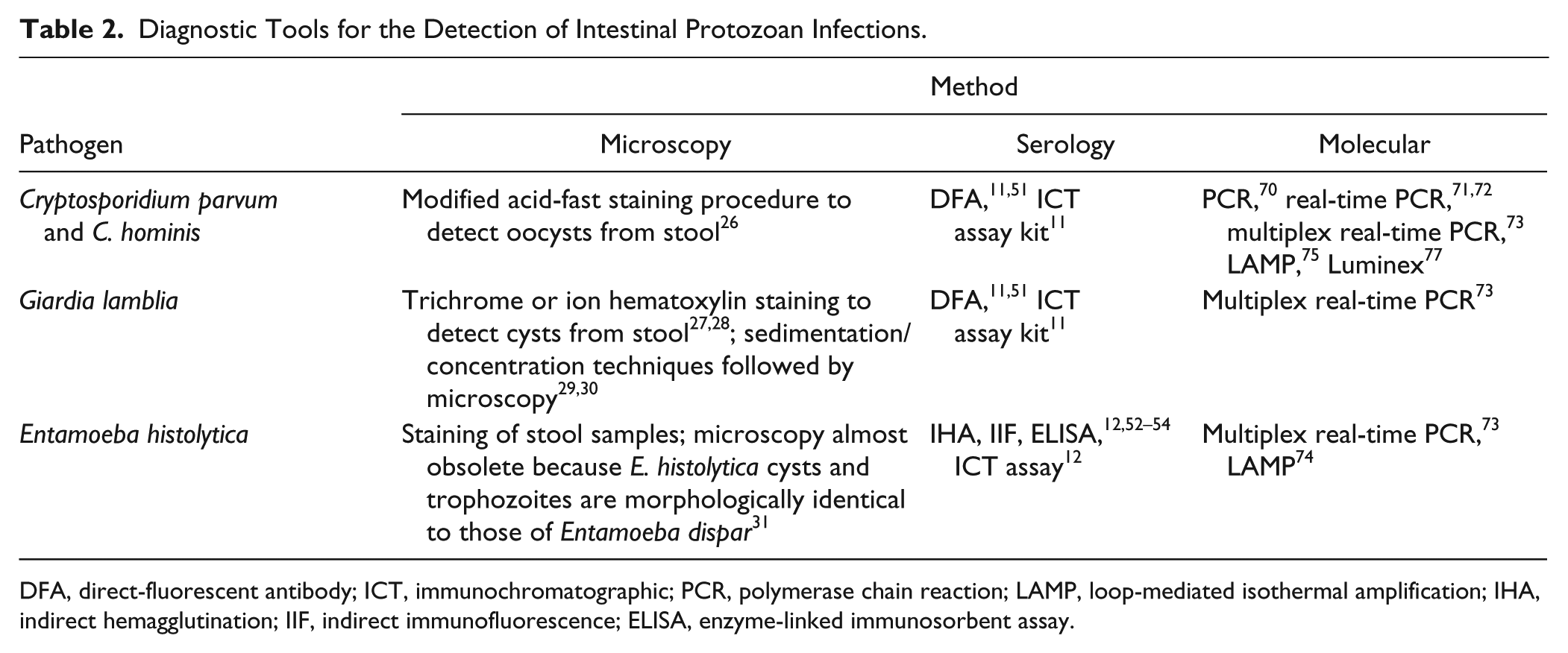
DFA, direct-fluorescent antibody; ICT, immunochromatographic; PCR, polymerase chain reaction; LAMP, loop-mediated isothermal amplification; IHA, indirect hemagglutination; IIF, indirect immunofluorescence; ELISA, enzyme-linked immunosorbent assay.
Diagnostic Tools for the Detection of Helminth Infections.
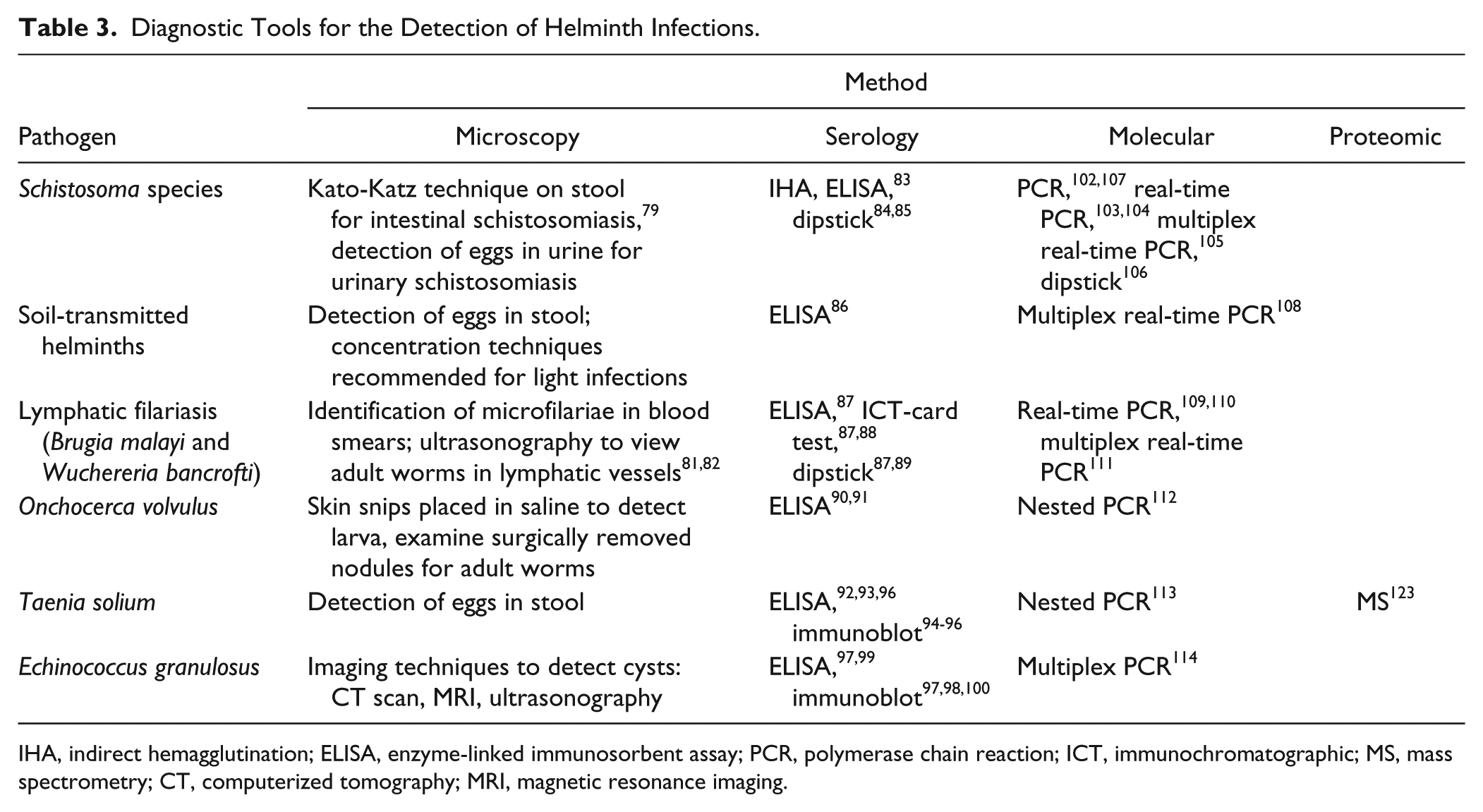
IHA, indirect hemagglutination; ELISA, enzyme-linked immunosorbent assay; PCR, polymerase chain reaction; ICT, immunochromatographic; MS, mass spectrometry; CT, computerized tomography; MRI, magnetic resonance imaging.
Protozoans
Microscopy
The introduction of the microscope had an influential effect on the field of medical diagnostics. For any new diagnostic method, whether it is molecular, serological, or proteomic, to have a meaningful impact in the field of diagnosis, it is likely to be compared with microscopy, which is still considered the gold standard. The development of newer diagnostic tools in the field of parasitology has stagnated over the past 20 to 30 years, thus strengthening the dependence on microscopic-based diagnosis. The popularity of this technique lies in its simplicity. Diagnosis via microscopy requires nothing more than the microscope itself, the specimen, and, for some techniques, a readily available dye. Microscopy has been the mainstay diagnostic tool for decades, and even though other techniques may exist, often results still require confirmation by microscopy since the newer diagnostic methods often lack standardization.
In the case of blood-borne protozoan infections, diagnosis via microscopy, often of blood smears, is the method of choice. Common blood-borne protozoan infections diagnosed by microscopic examination include malaria, babesiosis, African trypanosomiasis, Chagas disease, and leishmaniasis. Malaria is the most important parasitic infection in the world due to its high mortality. Quick diagnosis is essential to determine the appropriate treatment as well as to prevent further transmission. Microscopy is the gold standard for laboratory diagnosis. This method involves detecting parasites in Giemsa-stained thick and thin blood smears. However, microscopic results are operator dependent, thereby causing the sensitivity to vary. This variability between operators and the requirement for adequately qualified personnel to prevent misdiagnosis represent major disadvantages of this technique. Microscopy is the gold-standard method to diagnose infections caused by members of the Leishmania genus. This method has high specificity but varying sensitivity. Direct detection of parasites is performed by microscopic examination of aspirates from spleen, bone marrow, or lymph nodes. 15 Using spleen samples increases sensitivity, but the procedure to obtain the aspirates risks internal bleeding. Bone marrow aspirates may also be performed; however, splenic ones are preferred because they are less painful. 16 Trypanosoma brucei, another blood-borne pathogen, is the causative agent of African trypanosomiasis. During the first stage of the disease, parasites can be found circulating in the peripheral blood. The second stage is marked by parasites crossing the blood-brain barrier and infecting the central nervous system (CNS). The parasitic subspecies dictates geographic distribution, prognosis, and diagnosis. T. b. gambiense causes West African trypanosomiasis, which is a slow progressing disease characterized by low parasite loads. 17 Definite diagnosis is carried out by microscopic observation of blood, lymph node aspirate, or cerebral spinal fluid (CSF) for the presence of parasites. Positive results by other tests are always confirmed by microscopy. T. b. rhodesiense causes East African trypanosomiasis, which progresses quickly and is characterized by high parasite loads. 17 Diagnosis by microscopic observations of thick and thin smears is simple due to the elevated parasite load associated with T. b. rhodesiense. Currently, second-stage infections are diagnosed by microscopic observation of CSF. 18
The need for invasive procedures to collect samples for microscopic examinations represents another important disadvantage of this method. As previously mentioned, parasite detection in the CSF is the only way to diagnose the second stage of African trypanosomiasis, and, in the case of leishmaniasis, spleen aspirates greatly increase the sensitivity of parasite detection. The fear and worry of undergoing such invasive procedures often discourage patients from being tested. Consequently, these undiagnosed individuals contribute to increases in disease transmission by acting as parasite reservoirs. In addition, the requirement for such painful procedures further hampers the already poor rapport between individuals living in disease-endemic areas and the health care professionals. Too often, there is a lack of communication between these two groups, which leads to unsuccessful control program outcomes. For instance, control programs set up in the Democratic Republic of Congo suffered several setbacks due to the participants’ refusal to attend posttreatment follow-ups. 19 These follow-up sessions require repeated lumbar punctures to collect CSF. In addition to being uncomfortable, participants believed that this procedure caused decreased libido in men and sterility in women. 19 These observations from the field stress the importance of developing diagnostic techniques that do not require invasive procedures.
Chagas disease is the result of an infection with the blood-borne protozoan Trypanosoma cruzi. The parasite is transmitted by the triatomine bug. The second most important mode of transmission is via contaminated blood. This includes blood transfusions, organ transplants, and congenital transmission.1,20–22 During the acute stage of Chagas disease, parasites can be observed in the blood. For this reason, diagnosis is carried out by direct microscopic viewing of Giemsa-stained thin and thick blood smears. 23 Screening of neonates can be performed via direct methods, such as microscopy using venous or cord blood samples from the newborn. Babesiosis is caused by parasites belonging to the Babesia genus that are spread by certain ticks commonly found in North America and sporadically in Europe, Asia, Africa, and South America. 24 The parasites infect red blood cells (RBCs) and consequently cause hemolytic anemia. The disease can be fatal in splenectomy patients, immunocompromised individuals, and the elderly. The gold standard of babesiosis diagnosis relies on detecting the parasites in the patients’ RBCs. This is achieved by microscopic observation of thick and thin blood smears. Babesia infections can be easily mistaken for Plasmodium falciparum infections. 25 In addition, false negatives are common in immunocompetent individuals whose parasitemia can be lower than 1%. 25
Microscopy can also be used for the diagnosis of infections with intestinal protozoans, but the process is very labor intensive and time-consuming. These procedures involve collecting several stool samples from patients on different days and identifying the parasite within the sample. Fecal collection over several days increases the sensitivity of microscopic examination. Cryptosporidium infections can be diagnosed by microscopic examination of patient stool samples. The modified acid-fast staining procedure is used to detect parasite oocysts. The sensitivity of detection has been shown to vary according to the state of the specimens. 26 In watery diarrheal stool specimens, 10,000 oocysts per gram of stool were necessary to achieve 100% detection. Differently for formed specimens, 50,000 to 500,000 oocysts per gram of stool were needed for 100% detection. 26 Similarly to cryptosporidiosis, Giardia infection can be diagnosed by the examination of patient stool samples collected over several days. Giardia cysts are excreted intermittently in the stool, and their concentration varies greatly; therefore, making diagnosis by microscopy difficult. As previously mentioned, collection of several samples increases the sensitivity of microscopic examination. However, collection of samples over several days represents an inconvenience. Trichrome or ion hematoxylin staining is performed to visualize the parasite. However, these staining procedures yield sensitivity rates of merely 50% to 70%.27,28 Several diagnostic laboratories also perform sedimentation/concentration followed by light microscopy. Concentrated fecal preparations may be obtained by formalin-ether or formalin-ethyl acetate procedures.29,30
Microscopic examination to diagnose amebiasis is becoming obsolete. Entamoeba histolytica, which causes amoebic dysentery, is morphologically identical to Entamoeba dispar, which does not cause disease. The two species cannot be distinguished by microscopy. 31 E. dispar is 10 times more common than E. histolytica. 32 However, both infections are usually treated as a precaution. 31 This often leads to useless treatment, or it may distract from another underlying condition. In addition to this lack of specificity, another disadvantage of microscopy for amebiasis diagnosis is poor sensitivity. Approximately half to two-thirds of E. histolytica culture-confirmed cases are missed by microscopic examination. 33 It has become obvious that less labor-intensive as well as more specific and sensitive tools are better suited for the diagnosis of intestinal protozoan infections. Furthermore, the risk of misdiagnosis is high when using microscopy to diagnose intestinal protozoan infections. It has been shown that in some cases, laboratory technicians struggle to differentiate between artifacts and parasites present in the stool specimen.
Serology
The development of serology-based techniques has helped improve the diagnosis of blood-borne protozoan infections. Extensive research focused on the development of Leishmania serological assays has uncovered a myriad of candidate diagnostic antigens. The most promising antigens were the kinesin-related proteins. From this group, rK39 was the most tested antigen.34–36 The rK39 antigen has been used to develop an immunochromatographic strip test (ICT)–based rapid diagnostic test, which is advantageous for mass screening in endemic areas. This test requires a drop of peripheral blood and can be completed in approximately 15 min. 35 Although the rK39 ICT rapid test was quite successful in Asia, it was often unable to detect Leishmania infections in African patients. 15 In addition, rapid diagnostic tests still need standardization to become a regular practice in clinical laboratories.
The diagnosis of toxoplasmosis relies on serology. Toxoplasmosis, caused by Toxoplasma gondii, has a worldwide distribution. In immunocompetent individuals, more than 80% of primary Toxoplasma infections are asymptomatic. 37 Toxoplasmosis becomes a problem when an individual is immunocompromised or when a woman is pregnant. Early antibody responses can be detected via methods such as the dye test, immunofluorescent assay, and agglutination test, whereas later, immunoglobulin G (IgG) titers are detected by enzyme-linked immunosorbent assay (ELISA). For many years, the Sabin-Feldman dye test was the gold-standard diagnostic technique due to its sensitivity and specificity. 38 This test involves incubating patient serum with Toxoplasma trophozoites. If the patient possesses parasite-specific antibodies, they will activate complement, which will lead to parasite membrane lysis. After incubation, the methylene blue dye is added. Blue-stained parasites represent a negative result, indicating an intact membrane due to the lack of Toxoplasma-specific antibodies, 38 while unstained parasites represents a positive result. In recent years, few laboratories have continued to use this method and rather focused on newer techniques such as indirect immunofluorescent (IIF) antibody tests, hemagglutination tests, capture ELISAs, and immunosorbent agglutination assays (ISAGAs). Unfortunately, serological assays lack the capacity to differentiate between recent and older infections; IgM levels can persist for over 2 years. 39 To determine whether an infection is recent, avidity ELISA is performed. This assay verifies IgG avidity and is based on the concept that as the immune response progresses, an immunoglobulin’s affinity for a specific antigen will increase. 40 Toxoplasma infection during pregnancy is cause for concern because it can lead to congenital toxoplasmosis. Upon delivery, ISAGAs can be performed using placenta or cord blood serum. If the tests are positive, cord blood samples at 1 week of life are sent to a reference laboratory. 37 Follow-up serology is again performed at 1 month and then every 2 to 3 months. There have been recent advances in the field of toxoplasmosis postnatal diagnosis. An ELISA assay that measures interferon-γ levels upon stimulation of whole blood cells with Toxoplasma crude antigens has been developed. This method has proven to be both sensitive and specific but will require standardization. 41
Serology-based field tests have also been popular for the diagnosis of West African trypanosomiasis. In the field, the card agglutination test for trypanosomiasis (CATT/T. b. gambiense) has been widely used since its development in 1978. 42 Whole blood is used, and the assay directly detects T. b. gambiense–specific antibodies. CATT/T. b. gambiense is cheap, quick, and highly sensitive. However, the test can give rise to false positives in individuals who are coinfected with malaria. 42 Similar tests, such as micro-CATT and a rapid latex agglutination test (LATEX)/T. b. gambiense, can also be used. 43 Although these tests are used for mass screening of populations living in endemic regions, confirmatory diagnosis still relies on microscopic identification of the parasite. 44 Differently for East African trypanosomiasis, T. b. rhodesiense, there is no diagnostic equivalent to the CATT/T. b. gambiense. However, diagnosis by microscopic observations of thick and thin smears is simple due to the elevated parasite load associated with T. b. rhodesiense. For the diagnosis of second-stage disease, research is currently being conducted to test various cytokines and antibodies as biomarkers for CNS infection. 17
As in the case of toxoplasmosis, diagnosis of chronic Chagas disease relies on serology. However, the tests employed for chronic Chagas diagnosis often yield results that are difficult to interpret; thus, these assays are normally repeated at least twice. 23 Standardized serological assays include IIF, indirect hemagglutination (IHA), and ELISA. IIF and IHA are commonly used due to their good sensitivity; however, their results are operator dependent, and there is a lack of studies that analyze their reproducibility. 23 Currently, the immunoblot and radioimmunoprecipitation assays are in the process of being standardized. Both tests showed promise in early studies. Excretory/secretory antigens of T. cruzi also seem to have potential to be used for serodiagnosis.45,46 Furthermore, Umezawa et al. 47 demonstrated that a T. cruzi mix-ELISA, which combines different parasite recombinant antigens, could improve serology-based diagnosis. The recombinant antigens used for this assay included B13, 1F8, and H49. The sensitivity and specificity of this mix-ELISA were shown to be 99.7% and 98.6%, respectively. 47
As seen with leishmaniasis and African trypanosomiasis, a great deal of effort has been focused on developing rapid diagnostic tests (RDTs) that can be used in the field. These tests can supplement microscopy, but they cannot replace it yet. For malaria, current RDTs are serology based and use three different Plasmodium antigens: Plasmodium histidine-rich protein, Plasmodium lactate dehydrogenase, or Plasmodium aldolase. 48 These tests are quick, are easy to perform, and require minimal patient samples. However, they are not specific for species such as P. malariae, P. ovale, and P. knowlesi. Furthermore, false positives may be observed due to cross-reactions in patients with Schistosoma mekongi or rheumatoid factor. 49 In addition, the tests inefficiently detect P. falciparum infections from South America since this species does not produce the common histidine-rich proteins. 50
For diagnosis of babesiosis, microscopy remains the gold-standard technique; however, samples are often sent to reference laboratories to confirm ambiguous results. IIF is used to detect antibabesial IgM and IgG. 25 This test is sensitive, specific, and reliable. ELISAs and immunoblots, although not standardized, can be performed to confirm the IIF results. However, compared with IIFs, Babesia detecting ELISAs require higher concentrations of antigen and have varying sensitivity. 25
Immunoassays have become the main tool for diagnosis of intestinal protozoan infections in both hospital and public health laboratories. These methods are preferred because they reduce both labor and time, thereby, increasing the laboratory’s efficiency. Some commercially available immunoassays simultaneously test for Giardia and Cryptosporidium. This combinatorial style of testing is advantageous because coinfections involving these two parasites are very common. The commercially available kit MERIFLUOR C/G (Meridian Life Science, Memphis, TN) uses the direct fluorescent antibody procedure for the diagnosis of both giardiasis and cryptosporidiosis using stool specimens. This assay simultaneously detects Giardia cysts and Cryptosporidium oocysts by using FITC-labeled monoclonal antibodies targeting cell wall antigens. The kit’s sensitivity for Cryptosporidium and Giardia is 99.8% to 100% and 96% to 100%, respectively.11,51 Its specificity is 94% to 100% for Cryptosporidium and 100% for Giardia (according to the manufacturer’s booklet). Meridian Life Science also provides ImmunoCard STAT!, which is an immunochromatographic assay kit. It is a rapid and qualitative assay in card format that can detect both Giardia and Cryptosporidium. The specificity of the assay is 100% for both parasites, and the sensitivity is 97.3% for Cryptosporidium and 100% for Giardia (according to the manufacturer’s booklet). The assay is performed in 20 min, and it is simple to interpret. The interaction of the labeled monoclonal antibodies with their target results in the formation of a band at either the Giardia or Cryptosporidium positions on the device depending on the infection status of the patient. 11
For the diagnosis of E. histolytica infections, IHA and IIF may be performed, but ELISAs are the most popular antibody detection assays. 12 The microtiter ELISA from LMD Laboratories (Carlsbad, CA) detects antibodies to E. histolytica. Using samples from patients with amebic live abscesses, this ELISA has a sensitivity of 97.9% and specificity of 94.8%. 52 There are several commercially available antigen-based ELISA kits specific for E. histolytica diagnosis. These assays allow for the differentiation between E. histolytica and E. dispar, which cannot be achieved by microscopy. The E. histolytica II kit (TechLab, Blacksburg, VA) uses either monoclonal antibodies against Gal/GalNAc-specific lectin or serine-rich antigen of E. histolytica. Stool samples from patients are commonly used for these kits. 12 The sensitivity of the assay is 86% to 95%, and its specificity is 93% to 100%. 12 Besides stool specimens, serum samples may also be used in the assay as well as liver abscess puss from patients with liver disease. The Optimum S kit (Merlin Diagnostika, Bornheim-Hersel, Germany) and the Entamoeba CELISA PATH kit (Cellabs, Brookvale, Australia) may also be used to diagnose E. histolytica. The Optimum S kit has a sensitivity of 100%. 12 The CELISA PATH kit has been shown to have a sensitivity of 95% to 100% and a specificity of 93% to 100%. 12 Besides the commercially available kits, several research laboratories have developed procedures involving monoclonal antibodies, which use various E. histolytica antigens such as lectin-rich surface antigen,12,33 lipophosphoglycan, 53 and a 170-kDa amebic adherence lectin detected in saliva. 54 Biosite Diagnostic (San Diego, CA) provides an immunochromatographic assay called Triage Parasite Panel, which can simultaneously detect Giardia lamblia, E. histolytica/E. dispar, and C. parvum. This assay uses a strip that is coated with monoclonal antibodies against the 29-kDa surface antigen of E. histolytica/E. dispar, the α-1-giardin of G. lamblia, and the protein disulfide isomerase of C. parvum. 12 This assay has good sensitivity (96%–100%) and specificity (99.1%–100%), and it can be performed in 15 min.12–14 However, it is often not the assay of choice in diagnostic laboratories because it does not distinguish between E. histolytica and E. dispar.
Molecular
A great deal of work is being focused on the development and standardization of molecular methods, which would be useful in diagnosing parasitic infections as well as monitoring chronic phases of disease, reactivation, and treatment responses. Over the past few years, the importance of molecular diagnostic tools has become more obvious. Quantitative PCR is useful because it allows for the quantification of parasites as well as species typing. Furthermore, this technique can be used to monitor treatment efficiency. Unfortunately, equipment requirements as well as the high cost limit the use of PCR for mass screening purposes in the field. The introduction of LAMP could facilitate the use of molecular techniques for diagnostics. LAMP is highly specific, carried out under isothermal conditions, and quick, and it requires less complicated equipment.3,4,15 Moreover, reagents can be kept at room temperature, and there are no post-PCR steps. PCR is the main molecular tool for Leishmania diagnosis due to its high sensitivity and reliability. Different PCR target sequences that are commonly used include ribosomal RNA genes, kinetoplast DNA, mini-exon derived RNA, internal transcribed spacer regions, and so on. 15 Furthermore, assessment of leishmaniasis drug treatment and determination of clinical outcome can be carried out through the use of nucleic acid sequence–based amplification (NASBA), which amplifies RNA sequences under isothermal conditions. Coupled to oligochromatography, NASBA can be used to monitor the progression from active disease to cure. 55 Quantitative NASBA using skin biopsy specimens from patients with cutaneous leishmaniasis has been shown to have a sensitivity of 97.5% and specificity of 100%. 56 In addition, the lower detection limit of the assay was determined to be two parasites per biopsy sample. Therefore, this method can detect parasite levels that are 100-fold lower than those detectable by conventional PCR. 56 This capability to detect such a low number of parasites per sample makes this technique very useful for monitoring treatments and clinical outcome. Recently, NASBA has been used in combination with gold nanorods to develop a colorimetric assay targeting the 18S rRNA of Leishmania major. 57 This assay makes use of the electrostatic interactions occurring between the phosphate group of the amplified RNA and the cethyl trimethylammonium bromide layer on the gold nanorods, which leads to aggregation and a color change from red to purple that can be visualized. 57 This method does not require a gel electrophoresis step to visualize the results. Moreover, the assay has 100% sensitivity and 80% specificity. 57
PCR is also commonly used for the prenatal diagnosis of toxoplasmosis. The assay is carried out using amniotic fluid. The PCRs used often target the B1 gene of the parasite.37,58–60 Upon delivery, PCR is performed on either the placenta or the cord blood serum to detect parasites. If the tests are positive, cord blood samples at 1 week of life are sent to a reference laboratory. 37 In the case of immunocompromised patients, a quick diagnosis is essential because Toxoplasma infection can be fatal. Diagnosis relies on detecting parasites either by PCR or microscopy. Microscopic examination of Giemsa-stained tissues or smears is the quickest and most inexpensive method for diagnosing toxoplasmosis. However, poor sensitivity is the major pitfall of this method. PCR can also be performed on blood or CSF samples to detect Toxoplasma DNA. However, the degree of sensitivity attained by the PCRs is questionable and requires further investigations. 37 Molecular methods would also substantially improve the diagnosis of African trypanosomiasis. PCR techniques have been developed to screen the CSF of patients. The discovery of the serum resistance-associated (SRA) gene in T. b. rhodesiense has proven to be a breakthrough for the promotion of both PCR and LAMP techniques. Reactions targeting this gene have the potential to identify a single trypanosome. 17 Real-time PCR has shown to be specific and sensitive for T. brucei DNA in blood samples. Becker et al. 61 developed an assay using primers designed against the 17 base-pair repeat satellite DNA in the parasite. A LAMP procedure is also being developed for the diagnosis of African trypanosomiasis.62,63 Like in the case of toxoplasmosis, PCR may also be used to screen neonates for congenital Chagas by using venous or cord blood samples from the newborn. These tests have very high sensitivity when performed during the first month of life. 23 Optimization and standardization of molecular tools to diagnose Chagas disease is still ongoing. Moser et al. 64 developed a PCR procedure to detect T. cruzi DNA. Primers were used to amplify a segment of the parasite repetitive 195 base-pair nuclear sequence. The sensitivity of the assay was increased by including radiolabeled probes that hybridized to the PCR products. 64
Currently, there are no commercially available molecular assays for malaria. Although some reference and government laboratories have developed their own molecular assays, their availability is limited. LAMP is currently in the spotlight. Poon et al. 65 developed a LAMP test that detected the target sequence of the P. falciparum 18S ribosomal RNA gene. They stated that the price of this test was one-tenth that of a conventional PCR. Recently, LAMP was further simplified in the form of a card test. It was used in combination with DNA filter paper and melting curve analysis. This system was shown to be highly specific and sensitive. 66 Improvement of the LAMP technique for malaria diagnosis should be geared toward the development of rapid diagnostic tests that could potentially be used in the field. There has been work involved in the optimization of LAMP for the four main Plasmodium species infecting humans. 67 Making molecular techniques more valuable, it was shown that PCR can also be used to detect infections with drug-resistant parasites. Farcas et al. 68 developed a real-time PCR procedure that could detect P. falciparum and its chloroquine-resistant determinants in returning travelers. The assay targets the K76T mutation in the parasite. 68
Future research on babesiosis diagnosis is aimed at improving Babesia detecting PCR procedures 69 and developing multiplex PCR assays that will be able to detect several tick-borne infections. This would be an advantageous diagnostic feature because it is often difficult to distinguish between different tick-borne infections. PCR assays have the potential to yield positive results from 100-µL blood samples containing as little as three parasites, elucidating the incredible advantage that molecular techniques could contribute to diagnosis of this parasitic disease. 25
Molecular techniques could help improve the diagnosis of intestinal protozoan infections. A PCR assay targeting the 18S rRNA has been developed to detect Cryptosporidium oocysts in environmental samples. 70 The sensitivity of this assay was increased by including a step where the PCR product is probed on dot blots with oligonucleotide probes that can be detected via chemiluminescence. 70 Limor et al. 71 developed a LightCycler real-time PCR assay using primers for the small-subunit rRNA gene. The use of melting curve analysis of the PCR products with fluorescence-labeled hybridization probes allowed for the differentiation of five common Cryptosporidium species. Jothikumar et al. 72 developed a real-time PCR TaqMan procedure for the detection of C. parvum and C. hominis in patient stool samples. The detection limit was shown to be 1 to 10 oocysts in a 300-µL stool sample. The procedure was also tested on specimens obtained from outbreaks that occurred in the United States and Botswana, and the specificity was shown to be 94%. 72 A multiplex real-time PCR assay for E. histolytica, G. lamblia, and C. parvum/C. hominis was shown to be comparable to microscopy. 73 Multiplex assays and LAMP are currently the focus of molecular research. Liang et al. 74 have been working on the development of a procedure for E. histolytica LAMP detection. This is a novel, one-step procedure with a sensitivity of one parasite per reaction. The assay was also specific as it did not detect DNA from other Entamoeba species, bacteria, or viruses. 74 LAMP is also being developed to detect Cryptosporidium oocysts in fecal and environmental water samples. 75 Karanis et al. 75 developed a LAMP procedure using primers designed from the 60-kDa glycoprotein gene of C. parvum. Further strengthening the support for LAMP, the procedure was also shown to be better than nested PCR for the diagnosis of veterinary cryptosporidiosis. 76 Luminex-based assays are newer molecular methods that can be used to differentiate between C. parvum and C. hominis. The probes used in this assay are species specific, and they are conjugated to carboxylated Luminex microspheres. 77 These probes hybridize to the Cryptosporidium microsatellite-2 region that differ by one nucleotide substitution. The specificity of the assay is 100%, and it was shown to be more sensitive than the direct fluorescent antibody test. 77 All of the work performed by the different research groups demonstrates the significant focus being dedicated to the development of optimal molecular tools for the diagnosis of parasitic infections.
Helminths
Microscopy
Helminths infect billions of individuals worldwide. 78 They have a substantial impact on public health due to the high morbidity associated with the diseases they cause. The standard methods involved in diagnosing helminth infections rely on direct examinations of a parasite structure either by microscopy or other imaging techniques. Direct observation via microscopy often involves the identification of parasite eggs in a provided specimen. In the case of intestinal schistosomiasis (Schistosoma mansoni and Schistosoma japonicum), diagnosis is achieved by observing parasite eggs in stool samples using the Kato-Katz technique. 79 This method involves sieving the stool sample before staining it and performing microscopic examination. It continues to be used because it is quantitative, simple, and inexpensive. However, the sensitivity of the Kato-Katz technique suffers when infection intensity is low. 80 Differently, urinary schistosomiasis (Schistosoma haematobium) is diagnosed by microscopically detecting the parasite eggs in the patient’s urine. Similarly to intestinal schistosomiasis, infections with soil-transmitted helminths are also diagnosed by detection of eggs in the stool. These include infections with ascaris (Ascaris lumbricoides), hookworm (Ancylostoma duodenale and Necator americanus), and whipworm (Trichuris trichiura). For light infections, it is recommended to concentrate the stool specimen to identify parasite eggs. Taenia solium diagnosis is also achieved by identifying the eggs in the stool. Specimen examination is done over 3 days on different samples to account for intermittent excretion of eggs. T. solium diagnosis is imperative because this infection can lead to neurocysticercosis, which is represented as cysts in the brain that can cause seizures. Lymphatic filariasis, caused by the nematode worms Wuchereria bancrofti and Brugia malayi, causes obstruction of the lymphatics, which leads to excessive swelling of the lower limbs and devastating morbidity. The standard method of diagnosis involves microscopic identification of the microfilariae in a blood smear. It is best to collect the blood sample at night due to the nocturnal periodicity of the microfilariae. Brazilian researchers also demonstrated that ultrasonography could be used to visualize live W. bancrofti adult worms in the lymphatic vessels.81,82 In the case of onchocerciasis, an infection caused by the nematode Onchocerca volvulus, the larval form of the parasite travels in the skin, whereas the adult form is found in subcutaneous nodules. Diagnosis is performed by collecting skin snips from patients and placing them in normal saline as this results in the emergence of the larva from the skin. Another diagnostic option would be to surgically remove a nodule and examine it for adult worms. These methods may turn away patients because they involve surgery and collection of skin biopsy specimens. Infections with parasites belonging to the Echinococcus genus lead to the development of hydatid disease. This infection causes the development of parasite cysts that can be life-threatening to humans. Diagnosis is carried out by the use of various imaging techniques such as computerized tomography (CT) scans, magnetic resonance imaging (MRI), and ultrasonography to detect the cysts. All of these methods of direct observation are the standard techniques used for the diagnosis of helminth infections. Examining samples for parasite eggs is time-consuming and requires a skilled technician. The development and standardization of other techniques would greatly improve the field of diagnostics.
Serology
Serology-based diagnostic tools would allow for faster diagnosis of helminth infections, and in situations where microscopic results are confounding, serological examinations could provide additional insight. Immunodiagnostic assays have been tested for the detection of imported schistosomiasis. 83 IHA using erythrocytes coated with S. mansoni worm antigens has been evaluated using sera from individuals with egg-confirmed schistosomiasis (S. mansoni or S. haematobium) as well as patients with other parasitic infections as controls for specificity. The IHA had a sensitivity of 88% for S. mansoni and 80% for S. haematobium. The sensitivity to detect coinfections was 86%. The assay had a specificity of 94.7%. 83 The same group that tested the IHA assay also evaluated the diagnostic potential of using an ELISA coated with S. mansoni egg antigens. The ELISA had a sensitivity of 93.3% for S. mansoni and 92.0% for S. haematobium. The sensitivity to detect coinfections was 93.0%. The specificity of the assay was 98.2%. 83 These two assays were also performed in combination, which increased the overall diagnostic potential. The combination of the two assays resulted in a sensitivity of 98.7% for S. mansoni, 96.0% for S. haematobium, and 98.0% for coinfections. The specificity of the assay was 97.2%. 83 These results demonstrate the high level of sensitivity and specificity that can be achieved in diagnosing schistosomiasis with a serology-based approach. Dipstick assays have also been developed for the diagnosis of schistosomiasis. One available dipstick that employs a monoclonal antibody against S. mansoni adult worm tegumental antigen was compared with an ELISA sandwich assay that uses the same monoclonal antibody. 84 The sensitivity and specificity of the dipstick test using urine samples were 86.7% and 90.0%, respectively. Using serum samples, the sensitivity and specificity of the dipstick assay were 88.3% and 91.7%, respectively. 84 The dipstick assay was compared with the sandwich ELISA, and their diagnostic powers were statistically comparable. 84 Therefore, in a field setting, the dipstick method would be a more practical choice due to its rapid nature and simplicity. The urine circulating cathodic antigen (CCA) dipstick represents another rapid diagnostic method for schistosomiasis. This assay yielded sensitivity and specificity ranging from 52.5% to 63.2% and 57.7% to 75.6%, respectively. 85 Sousa-Figueiredo et al. 85 demonstrated that this dipstick test achieved much higher sensitivity levels than that observed with microscopy (16.7%). Therefore, the dipstick assay may be a better choice in areas of low transmission. Advances in serological assays have also been ongoing for hookworm diagnosis. Bungiro and Cappello 86 have shown, through a hamster experimental hookworm model, the possibility of using fecal ELISA to detect excretory/secretory coproantigens. Detection occurred as early as 4 days postinfection, and the excretory/ secretory levels correlated with the intestinal worm burden. 86 These new insights could contribute to future diagnostic approaches.
In the case of lymphatic filariasis, there are some serodiagnostic kits that are commercially available. The discovery of circulating filarial antigen (CFA) had an important impact on the diagnosis of bancroftian filariasis. TropBio Antigen ELISA (James Cook University, Tropical Biotechnology Pty Ltd, Townsville, Queensland, Australia) and the immunochromatographic card test Binax NOW Filariasis (Alere Inc., Waltham, MA, USA) are two commercially available and widely used kits that make use of CFA.87,88 The ELISA kit uses IgM monoclonal antibodies, produced using the bovine parasite Onchocerca gibsoni, to detect the Og4C3 antigen.87,88 This assay does not show cross-reactivity with other helminth infections; however, it is time-consuming due to long incubations, serum pretreatment, and many steps. Differently, the immunochromatographic card test provides almost immediate results. This assay uses the monoclonal antibody AD12, which recognizes a filarial antigen of 200 kDa. 88 The CFA-based tests have high sensitivity as well as specificity, and they are not affected by the microfilariae periodicity. 87 Brugian filariasis can be diagnosed using the Brugia Rapid antibody-detection dipstick (Reszon Diagnostics International Sdn. Bhd., Selangor, Malaysia). This assay detects IgG4 antibodies and is also available in an ELISA version for the use of batch testing in reference laboratories. 87 This kit has been tested in endemic areas of Malaysia, and it was shown that the dipstick test, compared with microscopy, could detect 10 times more cases of filariasis. 89 For onchocerciasis, serodiagnosis can be achieved by using the Ov16 ELISA. Antigen recognition occurs by IgG4 antibodies. This assay is used during control programs to monitor elimination of infection. 90 The OvH3 ELISA has been tested in nonendemic areas to detect imported cases of onchocerciasis. The sensitivity of the OvH3 ELISA was shown to be 93.2% and its specificity 93.5%. 91
A great amount of effort has been focused on optimizing an ELISA to diagnose the disease cysticercosis, which is caused by the parasite T. solium. Rosas et al. 92 demonstrated that ELISAs based on IgM against the cysticercus could detect active neurocysticercosis using CSF samples from the patient. The assay had a sensitivity of 87% and specificity of 95%. Allan et al. 93 also showed that stool samples could be used for ELISA that would detect coproantigens. This assay did not show any cross-reactivity with other parasites. For a long time, the serodiagnostic gold standard for T. solium has been considered a Western blot assay using the lentil lectin-purified glycoprotein.94–96 Bueno et al. 96 derived two antigens from this molecule and tested them in ELISAs to determine their diagnostic potential. The first antigen is synthetic TS18var1. In ELISAs using patient serum samples, it had a sensitivity of 90.4% and a specificity of 90.3%. When CSF samples were used instead, the assay’s sensitivity was 90.2% and its specificity was 98.0%. 96 The second antigen tested was recombinant GP50. Using serum, the sensitivity of this ELISA was 94.7% and its specificity was 93.8%. Using CSF instead yielded both sensitivity and specificity of 100%. 96 In the case of echinococcosis, another cestode parasite, the lipoproteins antigen B and antigen 5 are the most widely used antigens for serodiagnosis of Echinococcus granulosus infections. 97 These two antigens are major components of hydatid cyst fluid. The standardization of assays using these antigens has been challenging due to cross-reactivity problems with other parasites as well as varying assay sensitivities and specificities. 97 Several other antigens have also been tested for possible use in serodiagnostic assays. Some of these antigens include EpC1, 98 malate dehydrogenase (EgMDH), 99 EF-hand calcium-binding protein (Eg-CaBP2), 99 full-length and truncated forms of actin filament fragmenting protein (EgAFFPf and EgAFFPt), 99 and thioredoxin peroxidase (TPxEg). 100 Al-Sherbiny et al. 101 developed a dipstick assay using camel hydatid fluid. The test was shown to have a sensitivity of 100% and specificity of 91.4%. Furthermore, visual results are obtained within 15 min. Innovative serology-based techniques could potentially improve the diagnosis of helminth infections. These methods yield results quickly, and they are not labor intensive. Moreover, unlike microscopy, they do not require highly skilled professionals to interpret the results.
Molecular
Molecular techniques could render the diagnosis of helminth infections more sensitive and specific. PCR protocols have been developed to diagnose schistosomiasis. Abath et al. 102 were able to detect S. mansoni in fecal specimens by using primers designed to amplify a tandem repeat DNA sequence. No amplification was observed with other helminths, thereby demonstrating the specificity of the reaction. Furthermore, the detection limit of the PCR was shown to be 2 eggs/g of sample. 102 It would not be possible to detect such a low concentration of parasite eggs by the Kato-Katz technique. A real-time PCR protocol has also been developed using primers that target the small subunit rRNA of S. mansoni. 103 The specificity of the reaction was shown by the lack of amplification of nonschistosome trematode DNA. Moreover, the limit of detection was determined to be 10 fg of purified genomic DNA. This amount represents less than one parasite cell. 103 For S. haematobium, a real-time PCR procedure targeting the Dra1 sequence of the parasite has been tested. 104 This procedure could amplify parasite DNA from urine, stool, and serum samples. A multiplex real-time PCR that could detect S. mansoni and S. haematobium DNA from fecal samples collected in northern Senegal was developed by ten Hove et al. 105 The species-specific primers and probes used for this assay were designed to target the cytochrome c oxidase gene. This multiplex real-time PCR was shown to have specificity of 100% and a detection rate of 84.1%. 105 The utilization of this technique can be advantageous due to its high-throughput potential. The need for a gel electrophoresis step to visualize PCR products represents a disadvantage of this molecular technique for diagnosis. Additional steps are time-consuming and not practical for rapid diagnosis of an infection. Akinwale et al. 106 have developed a technique that allows for the visualization of S. mansoni and S. haematobium DNA amplicons through the use of an oligochromatographic dipstick. Parasite tandem repeat sequences are amplified and then hybridized with gold conjugated probes. This method is species specific, it has a low detection limit (10 fg of genomic DNA), and results become visible within 10 min. 106 The dipstick also contains a control line, which is essential to validate the migration occurring in the dipstick. Recently, Lodh et al. 107 have shown that S. mansoni and S. haematobium DNA could be detected in urine sediment obtained by filtration. The DNA was extracted from urine sediment samples collected in southern Ghana, and the PCR procedure was performed using species-specific primers targeting repeat fragments. The assay was shown to have a sensitivity of 99% to 100% and specificity of 100%. 107 Assays that can simultaneously detect S. mansoni and S. haematobium are greatly coveted because infections with these two schistosome species can overlap geographically. Being able to simultaneously detect different parasite species in one diagnostic assay is a favorable characteristic; results are obtained rapidly, less assays need to be performed, and consequently, the use of reagents is reduced. Basuni et al. 108 developed a pentaplex real-time PCR procedure to detect four species of soil-transmitted helminths (Ancylostoma, N. americanus, A. lumbricoides, and Strongyloides stercoralis). This assay was shown to be more successful than microscopy in detecting soil-transmitted helminth infections.
For lymphatic filariasis, PCR protocols have been developed for both W. bancrofti and B. malayi infections. For W. bancrofti infections, real-time PCR can be performed to detect parasite DNA in blood samples by using primers designed to target the long DNA repeat element. 109 In the case of B. malayi, the real-time PCR assay involves the amplification of the parasite HhaI repeat element. 110 Pilotte et al. 111 have developed a TaqMan-based multiplex real-time PCR that can simultaneously detect W. bancrofti and B. malayi DNA using the primers used in the previously mentioned single PCR reactions. For this multiplex real-time PCR, DNA was extracted from human bloodspots. The use of simultaneous diagnostic tools has also been applied to the detection of filarial parasites from the Amazon region. Tang et al. 112 developed a nested PCR to detect three filarial species—O. volvulus, Mansonella ozzardi, and M. perstans—by amplifying the first internal transcribed spacer rDNA of the different species. The assay was shown to be specific as there was no cross-amplification with other blood-dwelling parasites. The test can be performed using whole-blood, blood spots, and skin biopsy specimens.
Molecular tools have also been developed for the detection of T. solium DNA. A nested PCR targeting the Tso31 gene has been tested. 113 Using archived samples, the assay’s sensitivity and specificity were 97% and 100%, respectively. When the nested PCR was tested in the field, both the sensitivity and the specificity of the assay were 100%. 113 For E. granulosus, a different cestode parasite, molecular tools can help decipher the species variability. Boubaker et al. 114 developed a multiplex PCR that can detect parasites based on the genus (Echinococcus), the species (E. granulosus), and the genotype within the E. granulosus complex (G1–G10). This can become an invaluable tool in the study of E. granulosus epidemiology.
Discussion
Although for many years, parasitic infections had been neglected, the issue cannot be ignored any longer. In low-income countries, infectious and parasitic diseases are the leading cause of years lost because of disability. Furthermore, these diseases are not restricted to the elderly as the young populations are greatly affected as well. 115 Although the disease burden attributed to parasitic infections is greatest in the developing world, this does not restrict the problem to the tropics. Moreover, some parasitic infections do not require international travel as they are already present in our own backyards. The Centers for Disease Control and Prevention (CDC) has targeted five neglected parasitic infections, commonly found in the United States, for public health intervention. Some of these include Chagas disease, cysticercosis, and toxoplasmosis.116–118 Parasitic infections are a reality, and it is of great importance to stress the quick and correct diagnosis of these infections to avoid deaths and further disease transmission.
Diagnostic methods include parasitological techniques, such as microscopy and culturing, serological assays, and molecular tests. Although several serological and molecular diagnostic tools are being tested and used by certain reference laboratories, results are often confirmed by microscopy, which remains the gold standard. Many newer assays have not been standardized yet, thus forcing diagnosticians to rely on microscopic observations. The evolution of diagnosis in the field of parasitology has been slow to progress. However, in recent years, several groups have focused their research on the improvement of diagnostics. The use of serology-based techniques has revolutionized clinical diagnostics. Many different assays, such as ELISA, are being used in reference laboratories. Furthermore, several commercially available kits rely on antigen and antibody detection methods. Some assays also offer formats that can simultaneously detect more than one parasite. This feature, which is common for the detection of intestinal parasites, is advantageous as coinfections involving these pathogens are widespread. The introduction of rapid diagnostic tests represents a bridge that can connect the laboratory and the field. These tests could allow for the mass screening of endemic populations and for the monitoring of control programs, thereby improving quick diagnosis and helping to reduce disease transmission. Current research emphasizes the development and optimization of molecular techniques such as PCR and LAMP. These assays maintain high sensitivity as well as specificity levels, and they do not require a large amount of patient sample to be effective. Furthermore, some assays, such as multiplex real-time PCR, can simultaneously detect several parasites. As previously mentioned, assays able to detect different species have been developed for schistosomes, soil-transmitted helminths, and filarial species. Molecular methods are promising tools for the future of diagnostics; however, additional work must concentrate on rendering molecular diagnostics more accessible.
Although relatively new at the moment, proteomic platforms seem to have an important place in the future of parasitic diagnosis. These new techniques can identify biomarkers that can categorize susceptible individuals, distinguish between the different stages of an infection, and monitor whether treatments lead to cure. Some techniques that can be used to analyze protein expression include matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS), surface-enhanced laser desorption ionization time-of-flight mass spectrometry (SELDI-TOF MS), liquid chromatography combined with mass spectrometry, isotope-coded affinity tags, and isobaric tags for relative and absolute quantification. 3 Proteomic work is ongoing for a number of parasitic diseases such as African trypanosomiasis, 119 malaria,120–122 taeniasis, 123 and Chagas disease.124,125
There are many different avenues to be investigated in the field of parasitic diagnostics. To achieve major advances in this field, knowledge and collaborations are essential. Clinicians should be familiar with the “tropical” diseases to report them efficiently. Time and effort should be dedicated to educating and communicating with individuals living in endemic areas to ameliorate the impact of control programs. Finally, multicenter studies involving clinical diagnostic laboratories as well as reference laboratories could accelerate the standardization of the assays. Diagnostic research has made much progression; however, there is still a lot of work to be done and improvements to be made. To better diagnose parasitic infections, research plus communication is the answer.
Footnotes
Acknowledgements
We thank the research institute of the McGill University Health Center, the Public Health Agency of Canada, and the National Microbiology Laboratory for supporting the National Reference Center for Parasitology.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
