Abstract
Clavibacter michiganensis subsp. nebraskensis (Cmn) and Pantoea stewartii subsp. stewartii (Pss) are two plant pathogens that can cause tremendous agricultural economic losses. This novel method based on microsphere immunoreaction was developed for the simultaneous detection of Cmn and Pss in maize. This multiplex method was constructed based on microsphere immunodetection with fluorescent labels such as quantum dots (QDs) and R-phycoerythrin (R-PE) for the detection of Cmn and Pss. Captured QDs and R-PE serve as signal reporters for fluorescent readout. The principle of this method is based on a sandwich immunoreaction. Cmn and Pss captured by the microspheres were detected using flow cytometry. The limit of detection of this method was 10 times lower than the enzyme-linked immunosorbent assay (ELISA), and its analysis time (1 h) was much shorter compared with ELISA (6–8 h). The method, which has been proven to be an effective approach to multiplex detection of plant bacteria (Cmn and Pss as models), not only increased the varieties but also improved the sensitivity. The microsphere immunoreaction provides a universal method for the multiplex determination of microbes because of its high sensitivity, specificity, and speed. In the future, the method will be more fully validated in vivo to detect diversiform bacteria.
Introduction
Gross’s bacterial wilt and blight of corn, caused by the Gram-positive bacterium, Clavibacter michiganensis subsp. nebraskensis (Cmn), is a persistent and economically relevant plant disease. 1 Stewart’s wilt is a serious bacterial disease of corn caused by the bacterium Pantoea stewartii. Gross’s wilt (C. michiganensis subsp. nebraskensis) may be very similar to Stewart’s wilt (P. stewartii subsp. stewartii [Pss]). 2 They are usually not easily identified at an early stage of development, and extensive indexing is required to determine the incidence and distribution of infection. It is transmitted by the seed at a very low frequency. 3 To control the spread of the Cmn and Pss, several methods have been reported for the detection of Cmn and Pss, including traditional microbial assays, 4 an enzyme-linked immunosorbent assay (ELISA), 5 PCR, 6 rep-PCR, 7 and TaqMan-PCR. 8 Improving the currently available methods of detection of these plant pathogens might reduce economic losses.
Microsphere-based immunoassays have recently been developed and are already in use in medical diagnostics, bioassays, and drug screening. 9 In these assays, flow cytometry is used to analyze individual microspheres by fluorescence, distinguishing four fluorescent colors simultaneously: green (530 nm), orange (585 nm), red (605 nm), and deep red (675 nm). Typically, deep red fluorescence is used for microsphere classification, and green, orange, and red fluorescence are used for analyte measurement. By varying the ratio of the fluorophores, up to 100 different bead sets can be distinguished, and each bead set can be coupled to a different antibody. Beads of different sizes or colors, or a single bead with various fluorescent probes, can then be used for multiplexed immunoassays. These different combinations are purported to simultaneously measure up to thousands of analytes in a single tube.
The method presented here uses monoclonal or polyclonal antibodies bound to polystyrene beads (5.6 µm). The agent is then added together with monoclonal antibodies conjugated with quantum dots (QDs) or R-phycoerythrin (R-PE). The fluorescent-conjugated antibodies are bound to the monoclonal or polyclonal antibody-coated polystyrene beads, which settle to the bottom of centrifuge tubes and can be subsequently analyzed on a flow cytometer. To illustrate the feasibility of this novel application, Cmn and Pss were selected as practical analytes. To confirm its accuracy, we used the traditional ELISA as a comparable method. We believe that such assays, which combine the merits of chemical analysis and immunoassays, will provide an important new methodology in the determination of plant bacteria contamination.
Materials and Methods
General
The microorganisms used in this study are listed as follows. Among the tested 10 strains, P. stewartii subsp. stewartii (ATCC 8200) and C. michiganensis subsp. nebraskensis (ATCC 27822) were obtained from the ATCC (American Type Culture Collection Center, Manassas, VA). Xanthomonas campestris pv. holcicola (CMCC 1.1530), Acidovorax avenae subsp. avenae (CMCC 1.1726), Clavibacter michiganensis subsp. insidiosus (CMCC 1.1908), C. michiganensis subsp. michiganensis (CMCC 1.1909), and Clavibacter fangii (CMCC 1.2000) were purchased from the CMCC (China General Microbiological Culture Collection Center, Beijing, China). Pseudomonas syringae pv. syringae (CAAS 12056), C. michiganensis subsp. sepedonicus (CAAS 12057), and Erwinia carotovora subsp. carotovoraborgey (CAAS 12104) were from the CAAS (Institute of Plant Protection Chinese Academy of Agricultural Sciences, Beijing, China). All strains were grown and maintained on nutrient broth (NB) at 28 °C for 48 h. Then the bacterial cultures were counted by the colony counting method.
Bio-Plex microspheres (diameter 5.6 µm, 1.25 × 107 beads/mL) with surface carboxyl groups were used as solid carriers. The microspheres were internally dyed with a red and orange fluorophore. The microspheres were purchased from Bio-Rad Laboratories (Hercules, CA). Monoclonal and polyclonal antibodies against Cmn and Pss were prepared in our laboratory. Sulfo N-hydroxysulfosuccinimide (sulfo-NHS) and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDC) were purchased from Fluka (Buchs, Switzerland). Common reagents and solvents were analytical grade and purchased from Beijing Chemical Reagents Co. (Beijing, China). Aqueous solutions for immunoassay experiments were prepared using Millipore water (Elix-3+Milli-QA; Millipore, Molsheim, France). Buffers used were activation buffer (0.1 mol/L NaH2PO4, pH 6.2), storage/blocking buffer (phosphate-buffered saline [PBS], 1% bovine serum albumin [BSA], 0.05% NaN3, pH 7.4; Sigma, St. Louis, MO), washing buffer (PBS containing 0.05% Tween-20), dilution buffer (PBS, pH 7.4), and coupling buffer (0.05 mol L−1 2-(N-morpholino) ethanesulfonic acid [MES], pH 5.0; Sigma). Labeling results were determined by measuring absorbance at 633 and 488 nm by flow cytometry (ALTRA HyPerSort System; Beckman Coulter, Fullerton, CA) according to EXPO 32 MultiCOMP software (Beckman Coulter).
Covalent Coupling of Antibody to Carboxy-Modified Microspheres
Antibodies were coupled to the carboxylated microspheres using a two-step EDC/NHS coupling procedure. Briefly, after washing with 100 µL activation buffer, each 100 µL of carboxylated microspheres was pelleted (14 000 g for 4 min) in a 1.5-mL centrifuge tube using a microcentrifuge (3k30 super-speed bench; Sigma). The microspheres were resuspended by sonication (mini sonicator, KQ-250DE; Kunshan Ultrasonic Instruments, Kunshan City, China) and gently vortex mixed (Minishaker, MS1; IKA, Guangzhou, China) in 80 µL activation buffer, to which 10 µL activation buffer containing 5 mg/mL EDC and 10 µL activation buffer containing 5 mg/mL sulfo-NHS had been added. The mixture was incubated for 20 min at room temperature. The microspheres were then washed twice in 200 µL coupling buffer, and a solution of antibody conjugate in 100 µL coupling buffer was added and the mixture incubated with gentle shaking for 2 h at room temperature. The coupled microspheres were pelleted and washed twice with 500 µL of washing buffer, then resuspended in 500 µL storage/blocking buffer and blocked at 4 °C prior to use. The microsphere concentration was determined using a hemocytometer.
Conjugation of CdTe QDs or R-PE with Antibody
CdTe QDs were prepared as previously described. 10 CdTe QDs prepared in the aqueous phase were conjugated to proteins using EDC as a coupling reagent. First, a mixture of 0.1 mg/mL CdTe QDs, 0.2 mg/mL anti-Pss McAb, and 5 mg/mL EDC (containing 0.01M PBS, pH 7.4) was reacted for 0.5 h at room temperature. The reaction was then blocked by placing the mixture at 4 °C. Finally, the conjugated antibodies were purified using a Superdex 100 size exclusion column and stored at 4 °C prior to use. The R-PE conjugation protocol was based on a published protocol. 11
Preparation of Bacteria and Cmn-Spiked or Pss-Spiked Corn Seed Samples
The Cmn and Pss bacterial samples were pretreated at 28 °C for 48 h and harvested by centrifugation and washing (three times in PBS). PBS buffer without Cmn and Pss was used as a control. Samples of 500 healthy corn seeds, purchased from a local market, were surface sterilized for 1 min in 0.5% NaOCl, rinsed three times in sterile distilled water, and soaked overnight at 4 °C in 300 mL PBS. Seeds were blended in stomacher bags for 1 min and transferred into Eppendorf tubes. The suspension was centrifuged at 1000 rpm for 10 min. The supernatant was transferred and centrifuged again at 10 000 rpm for 15 min. Cmn-spiked or Pss-spiked corn seed samples were prepared by spiking a series of different concentrations of standard Cmn or Pss in the extract solution of corn seed. The extract solution without addition of standard Pss served as a control.
Microsphere-Based Fluorescent Immunoassay
The 1.0 × 104 coupled beads were placed in reaction tubes.12,13 MultiScreen 96-well filtration plates (Millipore, Bedford, MA) were first prewetted with 100 µL PBS (0.01M, pH 7.4) in each well, and then 1 µL of the antibody-coated beads was added to each of the washed wells. Then, 100 µL sample per well was added, and the mixture was rotated at 37 °C for 40 min. The beads were then washed with 100 µL PBS per well. Cmn McAb-PE and Pss McAb-QDs were diluted with PBS to an optimal concentration. Next, 100 µL of this solution was added to the wells after the filtrates had been removed. This mixture was incubated in the dark for another 40 min at 37 °C before washing twice with PBS. The beads were resuspended in 100 µL PBS.
Flow Cytometry
A Coulter Epics Altra HYPerSort system (Beckman Coulter) equipped with an air-cooled argon ion laser (488 nm) and HeNe ion laser (633 nm) was used for this study. The 633-nm red diode laser excited the fluorochrome held within the microsphere, and the 488-nm laser excited fluorescent molecules bound to the microsphere surface. The microspheres, R-PE, and QDs were measured on PMT5 (675 nm), PMT3 (575 nm), and PMT2 (525 nm) detectors, respectively.12,13 Fluorescence of thousands of microspheres was reported by EXPO 32 MultiCOMP software. The fluorescence intensities of individual microspheres were measured and histograms were used to calculate the adhesion ratio.
If the samples contained Pss, the fluorescence signals of the COOH beads and the QDs were expected to be found by flow cytometry, whereas if the analyte contained Cmn, the fluorescence signals of the COOH beads and the R-PE were expected to be found. Similarly, if the analyte contained both Pss and Cmn, fluorescence signals from the COOH beads, the QDs, and the R-PE were expected.
Results and Discussion
Determination of Microsphere-Based Fluorescent Immunoassay Parameters
The microsphere-based flow cytometric immunoassay used two specific reagents: a microsphere-PcAb/McAb conjugate and a QD/R-PE reporter molecule. Flow cytometric analysis of the functionalized fluorescent microspheres bound with antibody-QD or antibody-PE found a high adhesion ratio signal in the absence of Cmn or Pss.
To establish the optimal captured antibody concentration, microspheres were coupled with varying concentrations of the antibody. The antibody conjugate ( Fig. 1 ) was bound to the surface carboxyl groups of the microsphere after microsphere activation. The functionalized microspheres were then blocked using a BSA solution to diminish nonspecific binding in the immunoassay procedure.
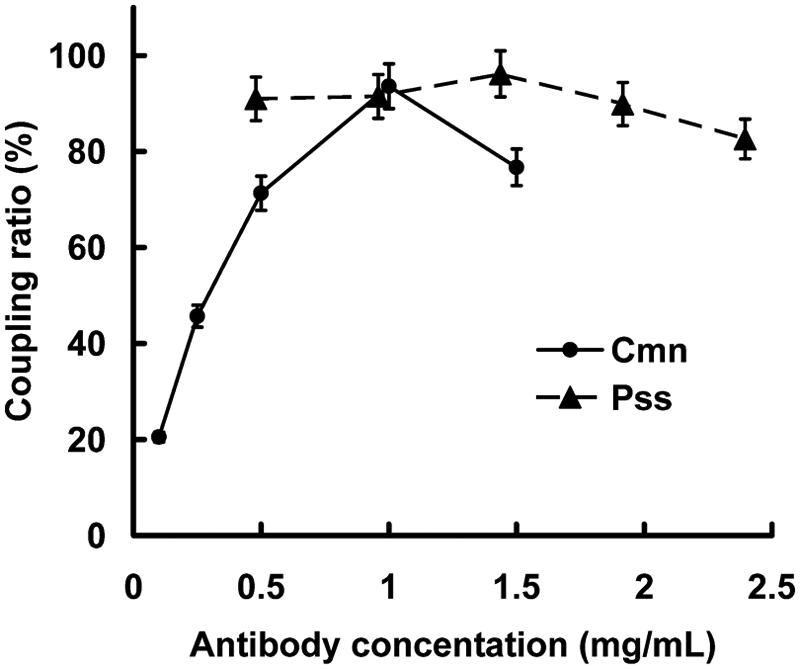
Optimization of captured antibody concentrations: Clavibacter michiganensis subsp. nebraskensis (Cmn, ●) and Pantoea stewartii subsp. stewartii (Pss, ▲).
We chose a concentration of the McAb-PE/QD conjugate with higher fluorescence intensity and lower nonspecific binding as our optimal concentration ( Fig. 2 ). The effect of the antibody-PE/QD conjugate was to enhance the fluorescence signal and thereby improve the sensitivity of the immunoassay.
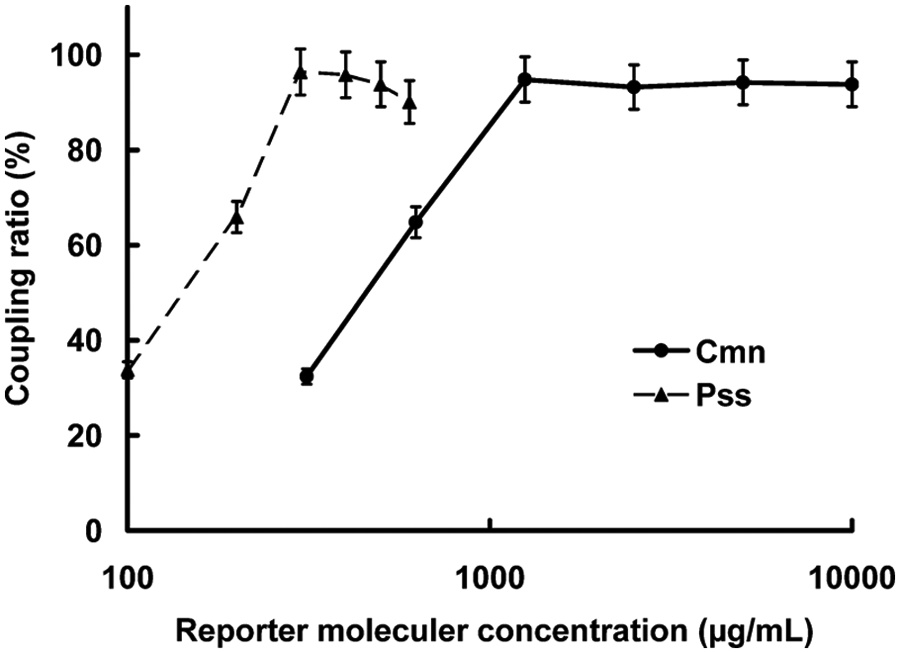
Optimization of reporter molecular concentrations: Clavibacter michiganensis subsp. nebraskensis (Cmn, ●) and Pantoea stewartii subsp. stewartii (Pss, ▲).
We deemed 0.1 to 1.5 mg/mL anti-Cmn McAb and 0.5 to 2.5 mg/mL anti-Pss PcAb to provide optimal sensitivity and a high adhesion ratio signal. Because of test costs, 1 mg/mL anti-Cmn McAb and 1.5 mg/mL anti-Pss PcAb coupled to microspheres were used in the protocol.
Simultaneous Detection of Cmn and Pss
The encoded microspheres were used to capture antibodies to the plant diseases Cmn and Pss. Flow cytometric analysis showed that a high adhesion ratio of microspheres bound with both antibody-QD and antibody-PE could be used to detect the presence of Cmn and Pss in samples.
A region of interest (ROI) was drawn around the microspheres (see Fig. 3a ). The encoded microspheres capturing anti-Cmn McAb and anti-Pss PcAb are shown in Figure 3b . Antibody-QD signals were observed in PMT2 (525 nm) and antibody-PE signals were observed in PMT3 (575 nm). In Figure 3c , d , the low adhesion ratios in PMT2 and PMT3 indicate that there was no Cmn and Pss in the sample. In contrast, a high adhesion ratio in PMT2 indicates there was Pss in the sample, and a high adhesion ratio in PMT3 indicates the presence of Cmn ( Fig. 3e , f ). A high adhesion ratio in both channels indicates the presence of both Pss and Cmn.
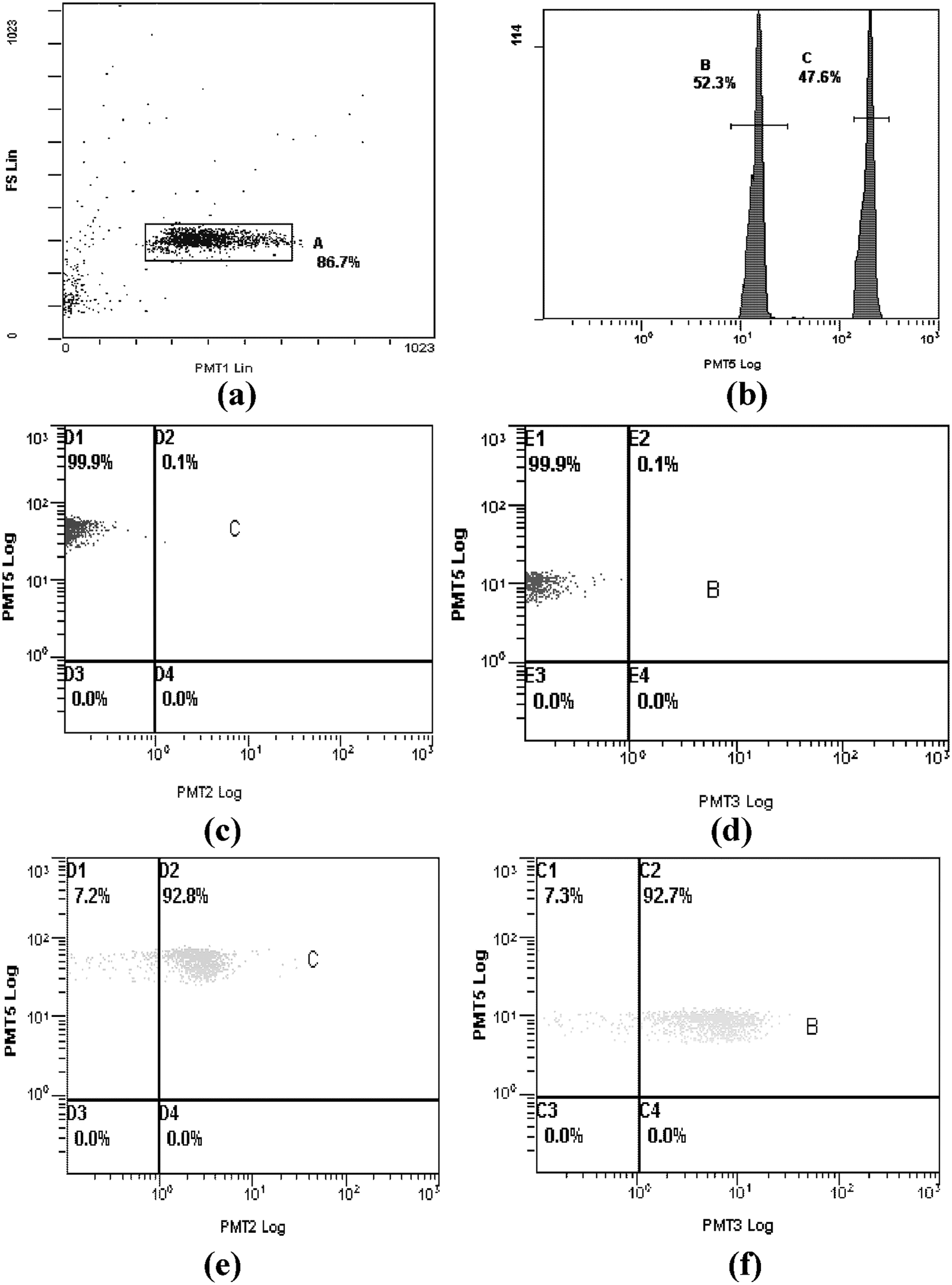
Analysis of Clavibacter michiganensis subsp. nebraskensis (Cmn) and Pantoea stewartii subsp. stewartii (Pss) by flow cytometry. The 633-nm laser excites the fluorochrome held within the microsphere, and the 488-nm laser excites fluorescent molecules bound to the microsphere surface. The fluorescence of microspheres, R-phycoerythrin (R-PE), and quantum dots (QDs) was measured on PMT5 (675 nm), PMT3 (575 nm), and PMT2 (525 nm) detectors, respectively. (
The Cmn- and Pss-containing samples were diluted from 108 to 10 colony-forming units (cfu)/mL in PBS (pH 7.8) and tested using this method. Under the optimal conditions, the performance of multiplex detection for Cmn and Pss was further evaluated. The typical fluorescence signals increased with the increase of Cmn and Pss concentrations ( Fig. 4 ). According to Figure 4 , the detection limit of Cmn and Pss was 104 cfu/mL. Corresponding to three times the standard deviation (SD) of the background signal, 14 the detection limit calculated was 104 cfu/mL. Results demonstrate that the method has potential for a real application.
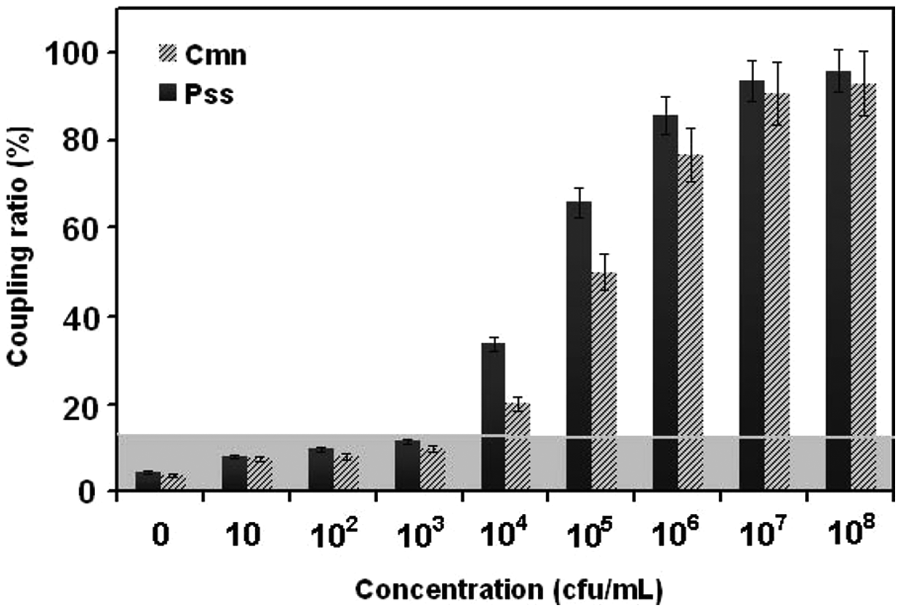
Detection of Clavibacter michiganensis subsp. nebraskensis (Cmn) and Pantoea stewartii subsp. stewartii (Pss) using a mulitplex detection based on microsphere immunoreaction. Detection signals exceeding three times the background value (shaded areas) are considered a positive signal.
Specificity confirmation was also carried out using the 10 standard strains. Test bacteria included (1) P. stewartii subsp. stewartii, (2) X. campestris pv. holcicola, (3) A. avenae subsp. avenae, (4) C. michiganensis subsp. insidiosus, (5) C. michiganensis subsp. michiganensis, (6) C. fangii, (7) P. syringae pv. syringae, (8) C. michiganensis subsp. sepedonicus, (9) E. carotovora subsp. carotovoraborgey, and (10) C. michiganensis subsp. nebraskensis. The specificity of the antibodies used was proven in ELISA and this method. ELISA enabled specific detection of Cmn and Pss. A similar specificity can be expected in both ELISA and the microsphere-based assay. The results of these two methods for Cmn and Pss did not show cross-reactivity with other strains (see
Comparison of Microsphere-Based Fluorescent Immunoassay with ELISA
Because the ELISA assay is the traditional immunoassay for the measurement of plant diseases, the sensitivity of the microsphere-based fluorescent immunoassay was compared with that of ELISA assays. A comparable ELISA assay setup using the same antibody concentrations was used, and a good correlation between the two assays was found. The ELISA method with the same pair of antibodies had a detection limit of 105 cfu/mL.
The qualitative detections of Cmn and Pss were 10 times lower than ELISA. Its analysis time (1.5 h) was much shorter compared with that of the enzyme-linked immunosorbent assay (6–8 h). The bacteria can be detected in a multiplex assay, which reduces sample preparation time and requires less sample material, regents, and disposables.
The reliability of the microsphere-based assay was clearly demonstrated in comparative tests, including positive control samples, 102 spiked samples, and 18 negative samples. A correlation of 100% was found positive for Cmn and Pss. When the samples were analyzed with both ELISA and the microsphere-based assay, detection signals exceeding three times the negative control were considered a positive signal (n = 3). Based on the signals, the two methods gave completely consistent results for all samples.
Field Sample Tests
The multiplex microsphere-based assay was evaluated using a set of naturally infected and bacteria-free extracts from maize seed lots, selected based on ELISA results. ELISA and the microsphere-based assay produced the same result for these field samples. A correlation of 100% was found for Cmn and Pss. In total, 26 samples were tested for Cmn. Among them, 20 samples were positive and 6 were negative in both assays (see
In future work, the method can be extended for other maize bacteria, thus increasing the benefit of microsphere-based fluorescent immunoassay compared with ELISA. The precision of our microsphere-based assays would be increased by using arrays with higher microsphere density, improved immobilizing chemicals, optical imaging fibers, and an imaging system better suited for these analyses. It can be anticipated that this strategy will be developed into a compact device using the principle of flow cytometry suitable for in situ analysis of bacteria. This compact device devised by our group will decrease instrumentation costs and extend the scope of applications.
We have developed an aqueous system microsphere-based fluorescent immunoassay for the detection of plant diseases and showed its applicability to the detection of Cmn and Pss. The most significant advantage of this assay is its capability to analyze multiple bacteria. Using precise amounts of each fluorochrome, an assay is created consisting of 1000 different microsphere sets with specific spectral addresses. Each individual analyte is encoded by one set of microspheres. We used the new QD fluorochromes as they are more stable than FITC during light exposure. R-PE, a fluorescent protein from the phycobiliprotein family, has broad absorption bands with peaks at 565 nm, 498 nm, and 539 nm and can therefore be excited with versatile excitation sources.
The broad excitation spectrum also provides the advantage of multicolor immunofluorescent staining or cell sorting. The optically encoded microspheres and QDs or R-PE can be combined to test hundreds of different materials (such as bacteria, residues, and pathogens) simultaneously. Our laboratory is currently using this assay in a high-throughput screen of multiple bacteria in single samples. The microsphere-based fluorescent immunoassay has superior sensitivity to a conventional ELISA. Finally, we evaluated the qualitative ability of this method using 26 maize samples previously shown to be positive on ELISA. The two methods yielded concordant results for all samples, indicating that the Pss strip was at least as sensitive as ELISA. Furthermore, the assay also offers the advantage of multiplex analysis. We believe this multiplex detection method might be developed to form a simple and rapid detection platform for bacteria.
Footnotes
Acknowledgements
We gratefully acknowledge the direction and assistance of Dr. Yong Jin and Dr. Shiming Tian in the preparation of antibody.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Commonweal Scientific Foundation for Industry of Chinese Inspection and Quarantine (201010022) and the basic research foundation of Chinese Academy of Inspection and Quarantine (2010JK018, 2010JK019).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
