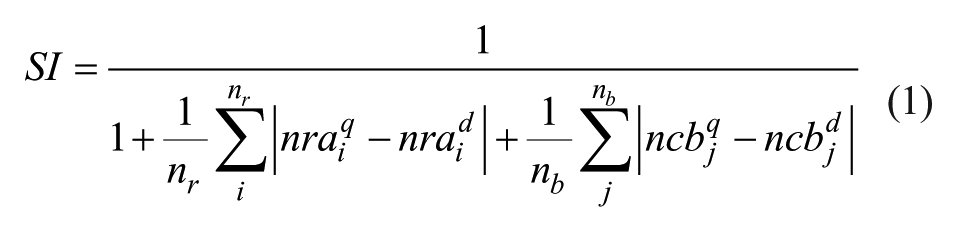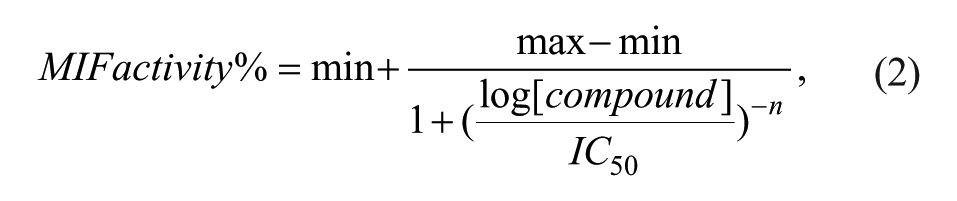Abstract
Macrophage migration inhibitory factor (MIF) is an autocrine- and paracrine-acting cytokine that is involved in several inflammatory, autoimmune, infectious, and oncogenic diseases. Clinical data have shown that inhibition of MIF, especially its tautomerase activity, with small compounds has been beneficial in some disease models. A virtual screening (VS) experiment is conducted for searching some active compounds to inhibit the tautomerase activity of MIF from the ZINC database. By using an x-ray–determined structure OXIM-11 as the query and an in-house developed two-dimensional scaffold comparing method designated as Sfilter, we have screened out some 1500 compounds for ranking by our previously published docking method ADDock. After further ranking by ADDock on 119 compounds screened, we have decided to choose 17 of them for measuring their inhibitory activity IC50 against the MIF tautomerase experimentally. The IC50’s are measured using both human monocytic THP-1 cell lysate and purified recombinant human MIF protein. We have found that the IC50’s measured for three searched compounds (namely, ZINC02693801, ZINC00141102, and ZINC12368346) are better than that determined for ISO-1, a known MIF tautomerase inhibitor and standard used throughout our VS experiment. Moreover, the scaffolds of most of our active compounds searched are also quite different from those searched and published by others previously.
Introduction
Macrophage migration inhibitory factor (MIF) is a versatile protein that is able to mediate the innate immunity and promote inflammation by inducing a wide range of proinflammatory cellular processes, including phagocytosis, T-cell proliferation, regulation of immunosuppressive action of glucocorticoids, activation of macrophages, and motivation of their survival through suppression of p53-dependent apoptosis. Although the underlying mechanisms of these pleiotropic functions of MIF are still unknown, it is believed that MIF exerts some of its cytokine activity through signal transduction by directly binding to the CD74-CD44 extracellular receptor complex. 1
MIF also exhibits some enzymatic activities to catalyze several biochemical reactions including D-dopachrome tautomerase, phenylpyruvate tautomerase, and thiol-protein oxidoreductase. 2 However, MIF also functions as an antagonist to counterregulate the inhibitory effects of endogenous or exogenous glucocorticoid-induced inflammatory and immune responses within the immune system. 3 In fact, glucocorticoids have various anti-inflammatory and regulatory effects on the host immune response system through a variety of mechanisms to inhibit the inflammation at multiple levels. 4 In other clinical studies, MIF is identified as a biomarker for different inflammatory-related diseases, including rheumatoid arthritis, inflammatory bowel disease, and atherosclerosis. In addition to its physiologic and pathophysiologic activities, MIF is also related to the tumor genesis process, including its effects on cell proliferation rate and tumor peripheral angiogenesis progress. The protein and its mRNA levels are overexpressed in a variety of human neoplasms, including pancreatic, breast, prostate, colon, brain, skin, and lung-derived tumors. 5 Using a functional screening assay, Hudson et al. 6 have observed that the MIF encoding cDNAs are capable of functionally inactivating the tumor suppressor p53. Other studies also show that the expression of MIF would influence both the hypoxic and normoxic neovascular processes within the microenvironment of a tumor, which are highly correlated with the aggressiveness and metastatic potential of the tumor. 7 These indicate that MIF might be a therapeutic target for developing a novel cancer chemotherapeutic strategy. Indeed, MIF has been aimed as a key therapeutic target for developing many small-molecule and antibody-based drugs. 8 Currently, developers for small-molecule inhibitors of MIF usually focus on rational structure-based design targeting the active site of tautomerase of MIF. 9 Since the finding by Zhang and Bucala 10 that a group of dopachrome analogs could inhibit the tautomerase activity of MIF with much lower concentrations than its native substrates, numerous MIF tautomerase inhibitors have been explored. The crystal structure determined for MIF has revealed that the protein is composed as a homotrimer by three identical monomers in which the catalytic site is located between each of two adjacent monomers. There are two α-helices packed against a four-stranded β-sheet on each monomer. An interior traversing channel in the center of the trimer is then formed by three β-sheets and six α-helices from each monomer. The N-terminal proline of each MIF monomer has an unusual pKa of 5.6 ± 0.1, which is thought to play a crucial role in the keto-enol tautomerization reaction. 11
In this report, we have conducted a virtual screening (VS) experiment for searching some active compounds from a publicly available database to inhibit the tautomerase activity of MIF. VS or in silico screening has become a popular technique in drug discovery research. 12 In finding some active antagonists, a large library of chemical structures is often docked into the active site of a targeted enzyme or protein through a molecular docking method. 12 Because only a small portion of searched compounds will be tested experimentally, a successful VS experiment should guarantee to rank the activity of compounds correctly. Therefore, a typical VS experiment may be conveniently divided into two phases, namely, early compound screening and late activity-ranking phases. 13 The techniques used in both screening and ranking phases should be accurate enough for allowing only 0.1% or fewer selected compounds to be tested experimentally. Here, we design a simple method for comparing the two-dimensional (2D) molecular scaffold between a query and every structure of a database for screening only a small portion of compounds for ranking by a docking method. We identify the number of atoms on each ring and count the number of chemical bonds connecting each pair of rings of a molecule first. Then, these numbers are plugged into an equation for computing the Manhattan metric 14 for comparing the structural similarity between the query and each structure of a database. Moreover, we employ Lipinski’s criteria 15 to screen out compounds that are unlikely to be a drug before proceeding with the molecular similarity screening process. The molecular docking program ADDock 16 is used for ranking the selected compounds from the database. Based on the docked energies computed by ADDock, we select out some 17 top-ranked compounds for testing their abilities to inhibit the MIF tautomerase experimentally. As revealed by in vitro IC50 measured, we have found that the inhibitory activities of some of these compounds measured are better than that of the standard, and the corresponding structures searched are also quite different from those published by others previously.17–19
Materials and Methods
VS Experiment
The x-ray structure of MIF (PDB code 2OOH 20 ) was downloaded from the RCSB protein data bank. The SYBYL 8.0 program (Accelery, Inc) was used to add all the hydrogen atoms and compute the Gasteiger-Hückel charges 21 for both the protein and a bound ligand OXIM-11. 20 The complete ZINC database (http://zinc.docking.org) consists of approximately 7,500,000 small molecular entries and is split into some 384 smaller subsets, with each carrying about 24,000 molecules were downloaded. Each of these subsets was stored as a consecutive SYBYL .mol2 file. Before conducting the VS experiment, compounds of each subset that did not fulfill Lipinski’s criteria 15 —namely (1) molecular weight falling below 500, (2) number of hydrogen (H)-bond donors less than 5, (3) number of H-bond acceptors less than 10, (4) polar molecular surface area computed below 140 Å2, and (5) logP < 5, where logP was the 1-octanol/water partition coefficient or a measure of lipophilicity—were excluded. The polar molecular surface of each compound was computed using a spherical polar coordinate system located on each atom and the atomic radii of AMBER GAFF force field. A total of 2592 surface points were generated for each atom treated like a sphere using an angle interval of 5°. The polar molecular surface area of each compound was computed by summing up the exposed surface area computed from each polar atom, which was a product of 4πr2 and the fraction of exposed surface points counted for the atom, where r was the atomic radius. An equation given by Xing and Glen 22 and parameters given by Wang et al. 23 were used to compute logP for each compound.
To further screen compounds from each subset, we identified all the atoms on each ring of a compound first and then the connecting atoms through which two rings were connected. The number of chemical bonds (ncb) or path length between each pair of connected rings was counted through the two ring connecting atoms. The number of atoms on each ring or the number of ring atoms (nra) on each ring were also counted. These were the two 2D molecular descriptors used in comparing the structure similarity between the query and each structure of a subset. A Manhattan distance or the Similarity Index (SI) between each 2D molecular descriptor of the query and each structure of a subset was computed as follows for screening the compounds:
where nr and nb were number of rings and number of path lengths counted between every pair of rings on a structure, and superscripts q and d were used for denoting the query and database structures, respectively. This in-house developed program was designated as Sfilter. The crystal ligand OXIM-11 of 2OOH was used as the query throughout the screening process. The largest SI computed was 1 if the two structures compared were similar to each other; otherwise, it would descend to smaller values if the two structures compared were less similar to each other. We collected compounds with larger SI computed from each subset and then pooled them together for ranking by ADDock. All of the crystal waters or cofactors identified inside the docking box of 2OOH were removed before docking. The docking by ADDock for a ligand into the active site of 2OOH includes the following steps: (1) selecting a terminal atom as an anchor, (2) counting the topology for the entire molecule based on the anchor selected, (3) defining the docking parameters and docking box based on the topology coordinates computed for the ligand, and (4) evolving the parameters using a genetic algorithm and then scoring the interactions between the docked ligand and protein target. ADDock used a piecewise linear potential function to score the interaction between a docked ligand and protein target, and both distance and geometry involved in the hydrogen bonding interaction were accounted for. The electrostatic interaction energy was computed from the formal charge assigned for each atom and scaled by a distance-dependent dielectric constant computed. Further, ADDock could systematically change the anchors for all the terminal atoms or anchors identified on a ligand. In other words, a series of conformations were automatically generated at the beginning based on all the anchors chosen, and then the best anchor or conformation was determined as the best docked energy computed among all the anchors or conformations generated. There were 17 best-docked or top-ranked molecules by ADDock selected for experimental testing.
Experimental Testing
The human monocytic cell line THP-1 (ATCC, Manassas, VA) was cultured in RPMI 1640 medium (Gibco BRL, Pasley, UK) supplemented with 10% heat-inactivated fetal calf serum (JRH, Australia), 100 U/mL penicillin (Sigma Aldrich, St. Louis, MO), and 100 µg/mL streptomycin (Sigma Aldrich). For preparing the cell lysate, THP-1 cells were harvested and washed extensively by a phosphate-buffered saline. The cell pellet was lysed by 100 µL/mL mammalian cell lytic RIPA (Sigma Aldrich, radio-immunoprecipitation assay; 50 mM Tris-HCl, pH 8.0, 150 mM sodium chloride, 1.0% Igepal CA-630, 0.5% sodium deoxycholate, and 0.1% sodium dodecyl sulfate) plus 10 µL/mL mammalian cell protease inhibitor cocktail (AMRESCO Inc., Solon, OH). The MIF protein could be secreted into culture medium by the lysed THP-1 cells. We followed a published protocol 24 to perform the tautomerase activity assay for MIF. The cell lysate equivalent to 5 × 105 THP-1 cells was diluted in 1 mL assay buffer (50 mM potassium phosphate, pH 6.0, and 1 mM EDTA). Subsequently, 200 µL of the diluted lysate was aliquoted into several different wells in a 96-well plate and treated with varying concentrations (200, 100, 50, 25, 12.5, 6.25, 3.125, 1.5625, 0.78125, 0.390625, and 0 µM) of the selected compounds for 30 min at room temperature. An anti-inflammatory MIF inhibitory drug ISO-1 (S,R-3-(4-hydroxyphenyl)-4,5-dihydro-5-isoxazole acetic acid methyl ester; Merck) 25 was used as a control. Some cell lysates were treated only with the vehicle DMSO or without addition of any cell lysate. The tautomerase activity of MIF was then measured by adding 20 µL of freshly prepared dopachrome methyl ester into each well. The dopachrome methyl ester was made by mixing an equal volume of 4 mM L-3,4-dihydroxyphenylalanine methyl ester with 8 mM sodium periodate on ice. The absorbance of dopachrome methyl ester at 475 nm was measured using a microwell plate reader (Molecular Devices, Sunnyvale, CA). The inhibitory activity against MIF tautomerase was measured at least nine times (n ≥ 9) for each compound. The aforementioned procedures were also used for determining the inhibitory activity for the same selected compounds using purified recombinant human MIF (50 nM; Abcam, Cambridge, UK). In each of these assays, 200 µL of diluted cell lysate was replaced by the same amount of purified recombinant human MIF. Each of these measurements was repeated at least three times (n ≥ 3). The IC50 of each selected compound was determined from eight concentrations ranging from 0 to 200 µM prepared for each compound. The percentage of inhibition was calculated as MIF activity % = 100 – (100 × [test compound value – average of blank control]/[average of DMSO control – average of blank control]). Then, the IC50 was determined through fitting MIF activity % to the following equation using the Sigmaplot software:
where min is the average of blank control, max is the maximum MIF activity % measured, and n is the Hill coefficient. Here, we have focused only on screening the MIF antagonists; hence, only the antagonistic model compound OXIM-11 and standard ISO-1 and assays are employed during the screening process. It is also feasible to use ADDock to screen some agonists if agonistic standards and assays are used.
Statistical Analysis
The mean and standard deviation (SD) of activity measurements performed for each compound was calculated such that all the activity data were presented as mean ± SD. The statistical significance defined as p < 0.05 was also computed using the Student t test.
Results and Discussion
Our first compound-screening stage is a ligand-based VS (LBVS)
26
because it compares the structure similarity using some 2D molecular descriptors computed between the query and each structure of a database.
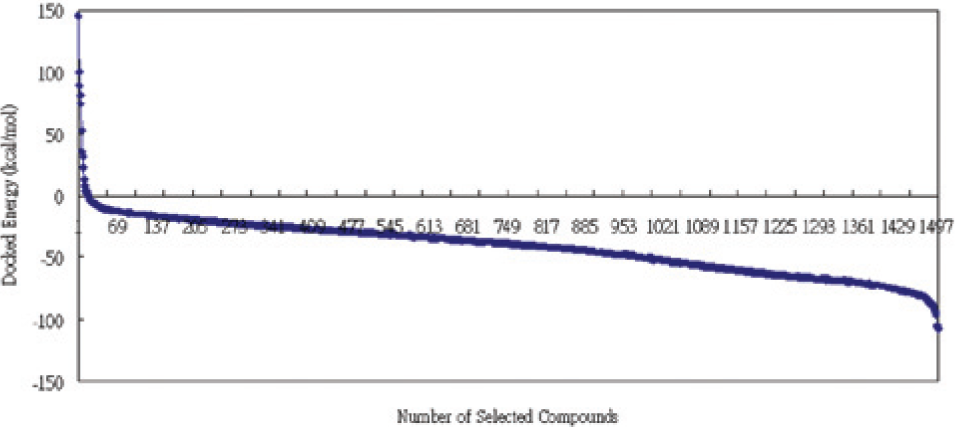
The total number of compounds screened after ligand-based virtual screening from all of the 384 subsets is 1500. All of these 1500 compounds are then docked into the active site of tautomerase of 2OOH using ADDock. The docked energies are sorted in order of magnitude for plotting.
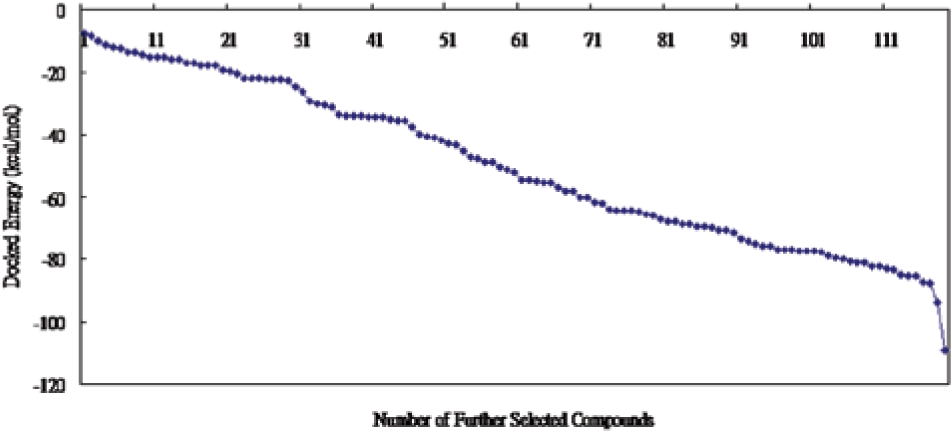
Compounds with docked energy better than −70 kcal/mol, a tentative criterion set for choosing compounds for further screening, are selected for further ranking using ADDock. There are 119 compounds thus selected and docked into the active site of tautomerase of 2OOH using ADDock again. The docked energies of all the 119 further screened compounds are also sorted in order of magnitude for plotting.
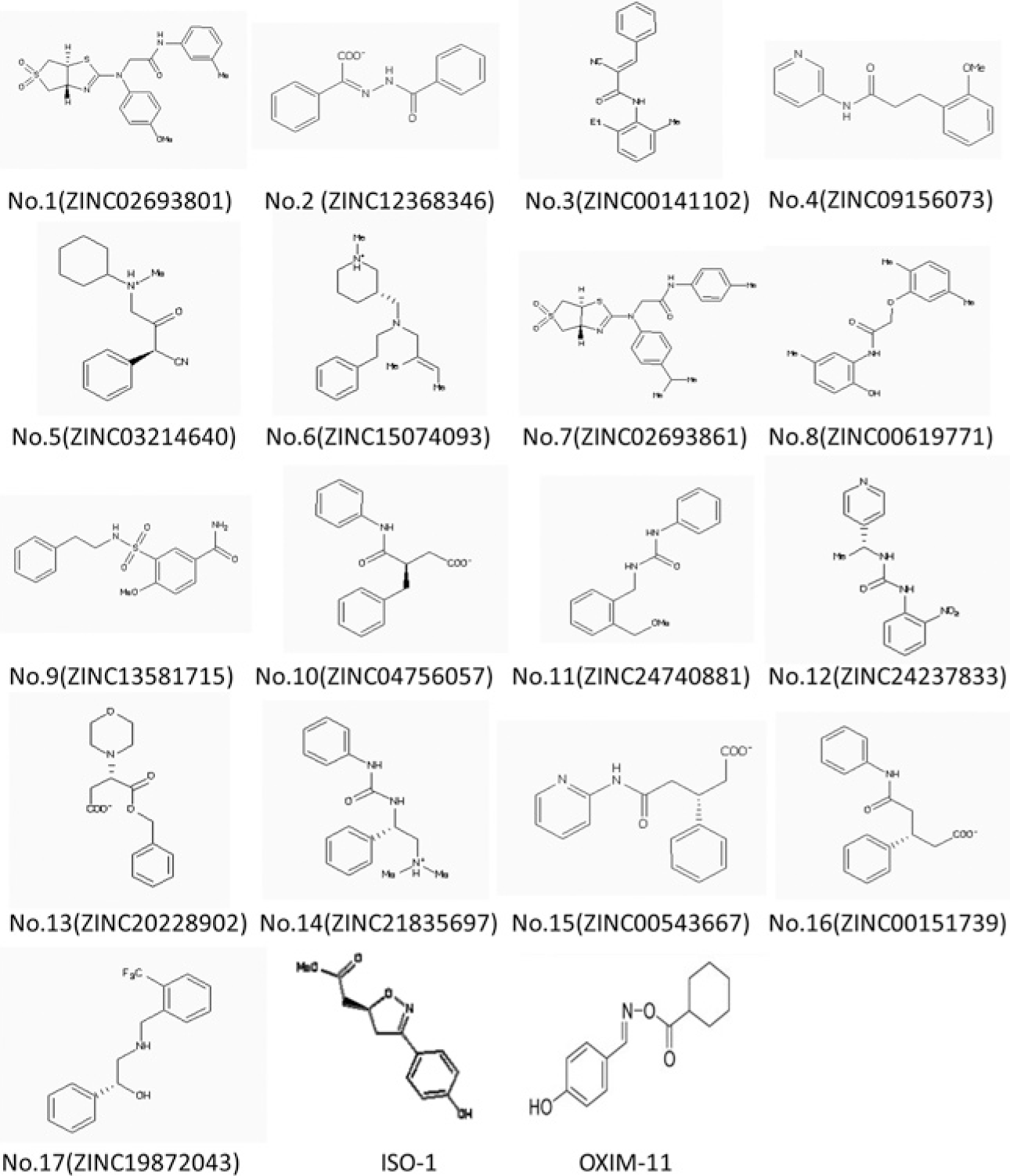
The two-dimensional (2D) structures and corresponding ZINC codes of the 17 selected compounds for expermiental testing. The 2D structures of ISO-1 and OXIM-11, the standard, and query used are also shown.
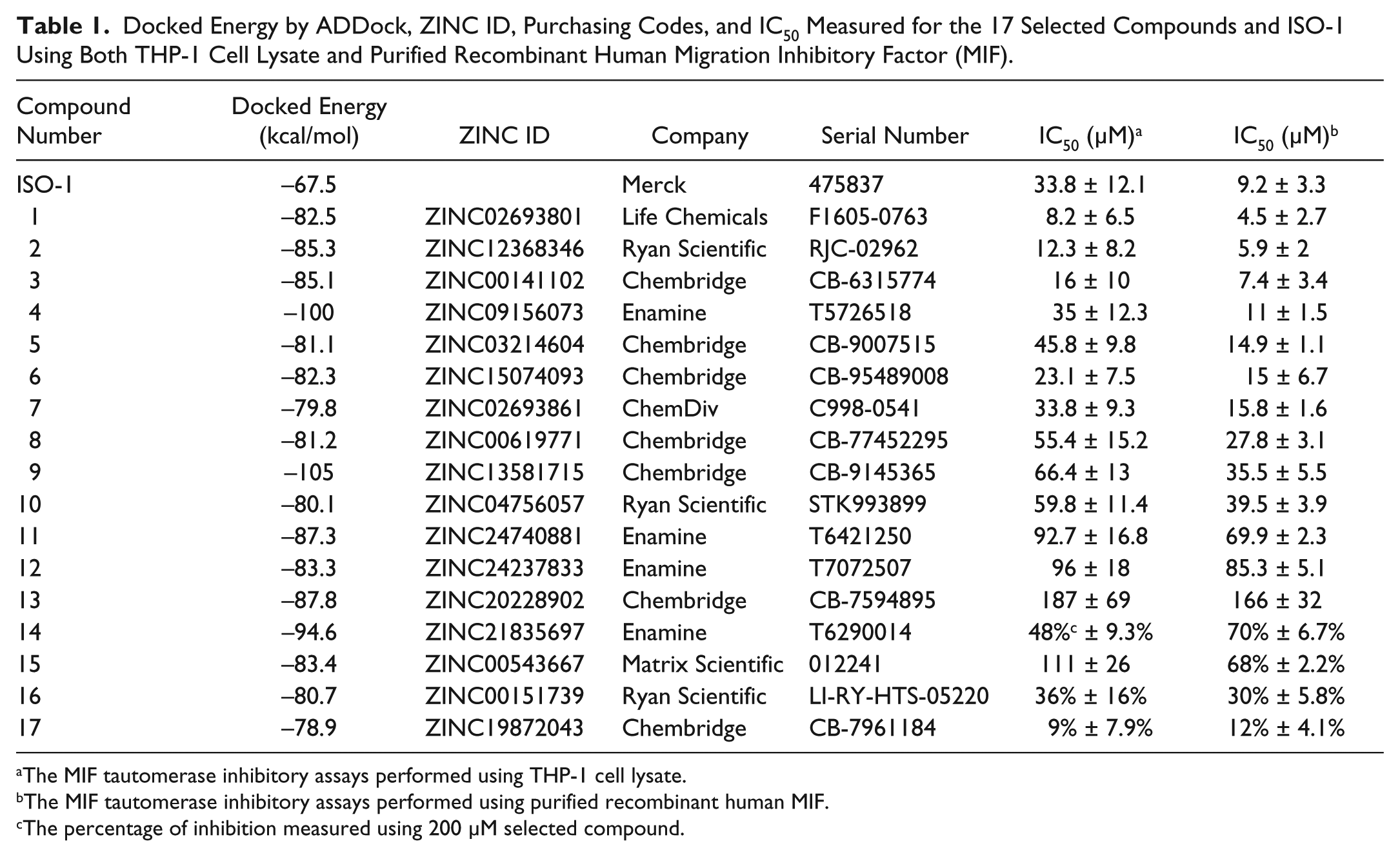
We perform the in vitro dopachrome tautomerase activity assays to measure the MIF inhibitory activity IC50 for the 17 selected and purchased compounds (
Table 1
) using both human monocytic THP-1 cell lysate and purified recombinant human MIF protein (Abcam). We also employ ISO-1, the first MIF inhibitory drug approved by the Food and Drug Administration (FDA) as a standard in our experimental measurements. The 2D structure of ISO-1 is also shown in
Figure 3
. The IC50 measured for ISO-1 using either THP-1 cell lysate or purified recombinant human MIF are 33.8 and 9.2 µM, respectively (
Table 1
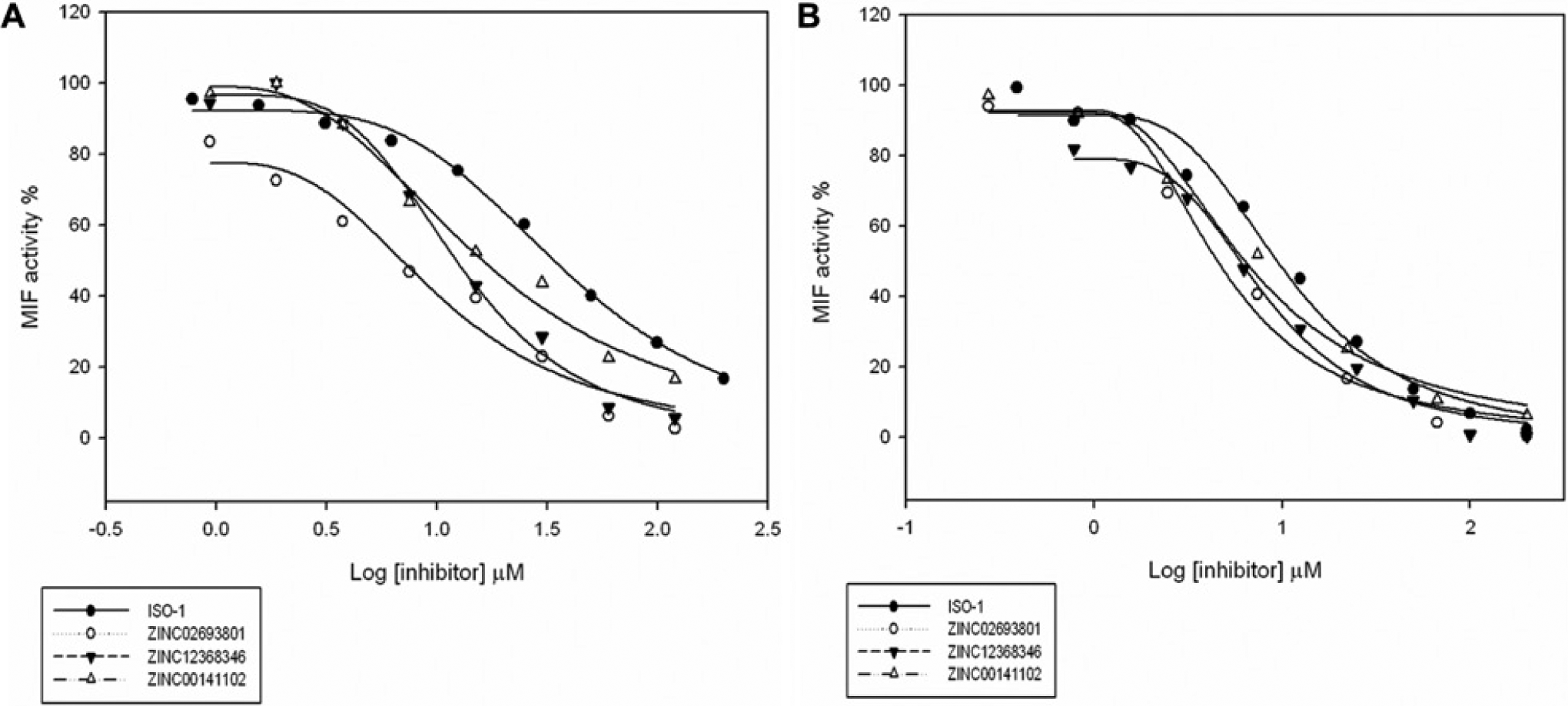
). However, we find that the IC50 measured using both MIF protein preparations for our three selected and purchased compounds are better than those measured for the standard. As shown in
Table 1
and
Figure 4
, whereas the best three IC50’s measured using THP-1 cell lysate for our selected and purchased compounds are ZINC02693801 (IC50 =8.2 µM), ZINC12368346 (IC50 = 12.3 µM), and ZINC00141102 (IC50 = 16 µM), the top three IC50’s measured using purified recombinant human MIF are also for the same three compounds, ZINC02693801 (IC50 = 4.5 µM), ZINC12368346 (IC50 = 5.9 µM), and ZINC00141102 (IC50 = 7.4 µM;
Table 1
;
Figure 4A
,
B
). A cytotoxicity assay for each selected compound has also been performed using the noncancerous human gland/breast epithelium H184B5 (ATCC CRL-8799) cell line. The cytotoxicity of H184B5 cells in the presence of each of the 17 screened compounds is estimated using the WST-1 cell proliferation assay. Compared with those treated with the same volume of DMSO, the cytotoxicity due to the presence of most of the 17 screened compounds is negligible because the corresponding EC50’s are undetectable, even though the concentration has been raised to 200 µM (
Docked Energy by ADDock, ZINC ID, Purchasing Codes, and IC50 Measured for the 17 Selected Compounds and ISO-1 Using Both THP-1 Cell Lysate and Purified Recombinant Human Migration Inhibitory Factor (MIF).
The MIF tautomerase inhibitory assays performed using THP-1 cell lysate.
The MIF tautomerase inhibitory assays performed using purified recombinant human MIF.
The percentage of inhibition measured using 200 µM selected compound.
(
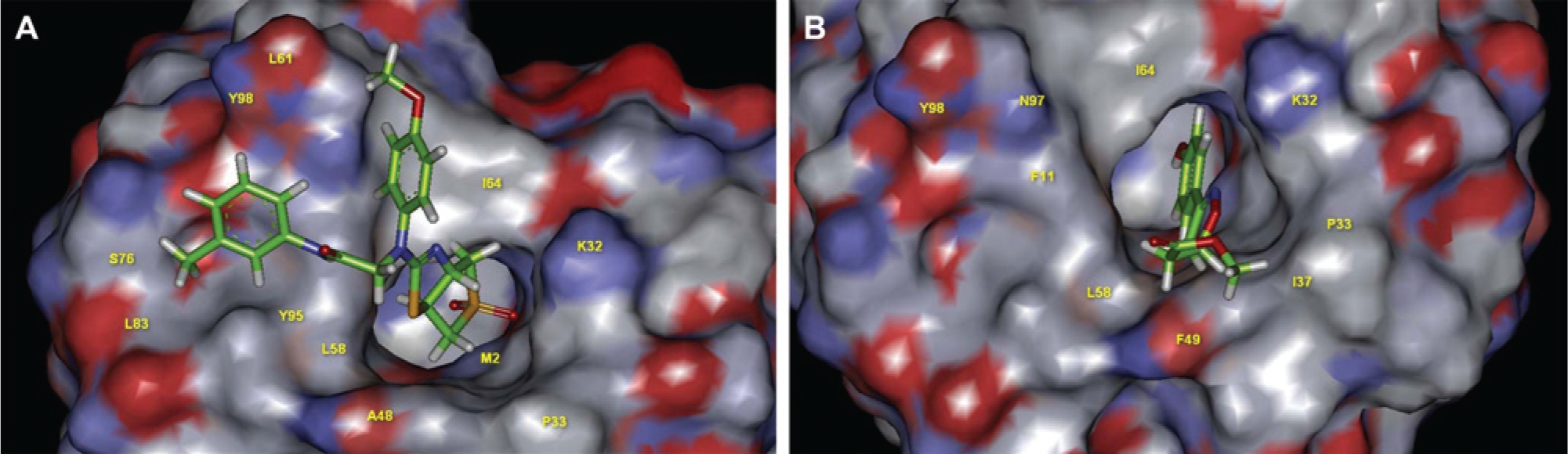
In fact, there are 10 compounds that can be considered as active because the corresponding IC50 measured using purified recombinant human MIF are smaller than 50 µM ( Table 1 ). The hit rate of our VS experiment is then 58.8% (10/17), which is higher than some recently published VS results, namely, 22.0% (9/41), 17 15.4% (4/26), 18 and 0.8% (8/1000). 19 However, there are some more highly active hits with IC50’s about 0.05 µM searched by these published VS results than that presented here. Note that the IC50 of the standard ISO-1 measured here using purified recombinant human MIF is 9.2, which falls within the published range of 7 to 100 µM.27–29 With the best docked energy −105 kcal/mol computed by ADDock, compound ZINC13581715 is also considered as active because the corresponding IC50 measured is 35.5 µM ( Table 1 ). Moreover, the docking scores given by ADDock for some less active compounds measured also declines apparently ( Table 1 ). The scaffolds of most active compounds searched, namely, ZINC02693801, ZINC12368346, ZINC00141102, ZINC09156073, ZINC03214640, ZINC1-5074093, and ZINC02693861 ( Fig. 3 ), are also quite different from those published by others.22–24 However, the structures of most active hits searched are characterized by conjugated ring systems similar to that of OXIM-11 ( Fig. 3 ), the query used throughout our LBVS. These ring systems can be fitted inside the MIF tautomerase active site, where they can be stabilized by the aryl-aryl interactions provided by Tyr95 and/or Phe113. The catalytic pocket of MIF tautomerase is known to be hydrophobic and consisted of Pro1, Met2, Ile64, Tyr95, Val106, and Phe113. 30 This catalytic pocket is also surrounded by a hydrophobic rim consisting of Pro33, Tyr36, Trp108, and Phe113. 30 Because this second hydrophobic region is very close to the catalytic pocket, it is expected that compounds capable of binding with both the active site and adjacent hydrophobic surface may enhance the potency. As shown in Figure 5A , while the thiazole ring of the most active compound ZINC02693801 (IC50 = 4.5 µM; Fig. 3 ) searched is docked into the tautomerase pocket of MIF, both methylphenyl and methoxyphenyl rings of the compound are apparently docked into the surrounding rim regions by ADDock, which would fulfill the criterion that both cavity and rim regions are bound simultaneously by the docked compound. However, the docking score of −88.2 kcal/mol computed for the compound ( Table 1 ) is not the best one obtained, which may reflect the fact that the thiazole ring of the compound is not deeply docked into the MIF tautomerase pocket ( Fig. 5A ). On the other hand, although both phenol and isoxazolyl rings of ISO-1 are deeply docked into the MIF tautomerase pocket ( Fig. 5B ), both the corresponding docked score of −67.5 kcal/mol computed and the IC50 of 9.2 µM measured for the compound are worse than those determined for compound ZINC02693801 searched ( Table 1 ).
(
The 2D molecular descriptors computed by Sfilter are strictly dependent on the ring scaffold compared. However, they are capable of discriminating the size and path length difference between any pair of rings of a query against those of database compounds. Although the 2D molecular descriptors computed are somewhat insensitive to the structural details of the structures compared, they have benefit for quickly screening out potentially active compounds carrying more diversified structural features. Moreover, ADDock has the advantage of anchoring all the docked compounds at the same position in an active site such that the corresponding docked energies computed are all compared on the same ground. This may be advantageous for searching active compounds with a high degree of structural similarity and targeted at the same active site. For example, the scaffolds of compounds ZINC12368346, ZINC00141102, ZINC09156073, and ZINC03214604 searched are quite similar to each other, and the corresponding IC50’s measured are also close to each other ( Table 1 ). Although it is somewhat hydrophobic, the tautomerase active site of MIF seems to be more versatile than what we expect because active compounds of quite different scaffolds are searched by different groups. The scaffolds of most of the active compounds searched here are also quite different from those published by others.17–19 Moreover, we have shown that a better IC50 is measured for a compound searched that is able to bind across the tautomerase active site and surrounding rim regions of MIF.
Footnotes
Acknowledgements
We thank the National Center for High Performance Computing of Taiwan, R.O.C., for providing us the computational facilities and the computer package SYBYL 8.0 described herein to complete the work.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The experimental part of this work was supported by a grant NSC99-2628-B007-001-MY3 from the National Science Council, Taiwan, R.O.C.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.