Abstract
Angiogenesis, the formation of new vessels from preexisting vessels, involves multiple cell types acting in concert to cause endothelial cell proliferation, migration, and differentiation into microvascular arrays. Under pathologic conditions, microenvironment changes result in altered blood vessel production. Historically, in vitro angiogenesis assays study individual aspects of the process and tend to be variable, difficult to quantify, and limited in clinical relevance. Here, we describe a kinetic, quantitative, co-culture angiogenesis model and demonstrate its relevance to in vivo pharmacology. Similar to in vivo angiogenesis, a co-culture of human umbilical vein endothelial cells with normal human dermal fibroblasts remains sensitive to multiple cytokines, resulting in a concentration-dependent stimulation of tube formation over time. Treatment with axitinib, a selective vascular endothelial growth factor (VEGF) antagonist, inhibited VEGF-mediated tube length and branch point formation and was selective for inhibiting VEGF over basic fibroblast growth factor (bFGF), similar to previous studies. Conversely, an FGFR-1 selective compound, PD-161570, was more potent at inhibiting bFGF-mediated angiogenesis. These results demonstrate the cytokine dynamics, selective pharmacology, and translational application of this model system. Finally, combining quantitative angiogenic biology with kinetic, live-content imaging highlights the importance of using validated in vitro models in drug discovery research.
Introduction
The establishment and maintenance of the vascular system are required for the growth of both normal and pathogenic tissues (i.e., tumors). 1 Blood vessels form through one of two mechanisms, vasculogenesis and angiogenesis, resulting in the development of three layers: (1) the intima composed of endothelial cells, (2) the media composed of pericytes in microvessels, and (3) the adventitia composed mainly of connective tissue. 1 Angiogenesis, the formation of new vessels from preexisting vessels in the body, is an essential process during development, wound healing, and reproduction. In healthy tissues, the angiogenic process, consisting of endothelial cell (EC) proliferation, EC migration, and vessel morphogenesis, is tightly regulated by physiologic control of growth factors and inhibitors.2,3 Under steady-state conditions, the balance between pro- and anti-angiogenic factors tightly regulates new blood vessel growth. 2 However, under pathologic conditions, such as cancer, rheumatoid arthritis, and wet age-related macular degeneration, the balance is tipped to stimulate new vessel production, providing nutrients to the diseased area (i.e., tumor growth in cancer). 2
Clinically, anti-angiogenic therapies have proven effective at slowing disease progression. 3 However, due to either inherent or acquired resistance, these therapies have been only mildly beneficial to overall survival. This has led to the investigation of pathways such as Dll4/Notch, basic fibroblast growth factor (bFGF), and placenta growth factor as alternatives to, or combinations with, vascular endothelial growth factor (VEGF) therapy to increase therapeutic efficacy. Historically, in vivo models have been the preferred method to study the angiogenic process. However, these models are limited in their throughput, quantitation, cost, and, most importantly, translation to human pathologies.4,5 Thus, advancement of in vitro angiogenesis models to study more complex pharmacologic effects or to examine such phenomena as VEGF resistance are paramount to developing the next-generation therapeutics.
Over the past 10 years, various in vitro assays have been developed to study individual aspects of the angiogenic process, specifically proliferation, migration, and differentiation. Historically, these formats have proven unreliable and are limited in their ease of quantitative, reproducible results and translation to in vivo studies.4,5 This has limited the development of next-generation angiogenic therapeutic agents. When developing new methods to study angiogenesis in vitro, it is important that the model remains sensitive and responsive to a variety of cytokines, such as VEGF, bFGF, or epidermal growth factor (EGF), similar to what is observed in vivo.6,7 Further, as anti-angiogenic therapy is becoming increasingly important in the clinical setting, quantitating anti-angiogenic potential on developing tubes and/or established networks will strengthen the pharmacologic validation of an in vitro model.5–7 Here, we demonstrate that a method of human umbilical vein endothelial cells (HUVEC) in co-culture with normal human dermal fibroblasts (NHDF), when used in conjunction with a compact fluorescent imaging microscope, the IncuCyte, presents a reproducible, kinetic approach to studying angiogenesis in vitro. Further, the IncuCyte’s automated image acquisition and integrated angiogenesis software algorithm provides a sophisticated, straightforward approach to measuring angiogenic tube formation.
In addition to quantitating tube formation over the course of the assay, we investigate the ability of clinically evaluated compounds to disrupt growth factor–mediated angiogenesis. Specifically, we show that axitinib selectively inhibits VEGF-driven tube formation. Using a bFGF-selective compound, PD-161570, the converse effect is observed, namely, bFGF signaling exhibits higher sensitivity to PD-161570 treatment than VEGF. Further, a compound with minimal angiogenic activity, lestaurtinib, affected growth factor–mediated angiogenesis only at the higher concentrations, where toxicity can be an issue. Together, the ability to reproducibly quantify a test agent’s effect on angiogenesis in vitro allows for faster, more in-depth mechanistic studies and is exemplified using this co-culture approach.
Materials and Methods
Cell Culture and Reagents
HUVEC were grown and maintained in endothelial cell culture media according to manufacturer instructions (Caltag Medsystems, Buckingham, UK). NHDF (Caltag Medsystems) were grown and maintained in DMEM (ATCC, Manassas, VA) containing 10% fetal bovine serum (Invitrogen, Grand Island, NY). Recombinant VEGF-165, bFGF, and EGF were purchased from Peprotech (Rocky Hill, NJ). CytoLight Green–Lenti, cytomegalovirus (CMV), and NucLight Green–Lenti, EF1-α were generated at the University of Michigan Vector Core Facility (Ann Arbor, MI).
Lentiviral Infection of HUVEC
The lentivirus containing CytoLight Green is a third-generation HIV-based, vesicular stomatitis virus glycoprotein (VSV-G) pseudotyped lentivirus encoding soluble green fluorescent protein (GFP) driven off a CMV promoter. The lentivirus containing NucLight Green is a third-generation HIV-based, VSV-G pseudotyped lentivirus encoding a nuclear restricted GFP. Viruses used to carry out our studies include CytoLight Green and NucLight Green lentiviruses (Essen BioScience). Soluble GFP is restricted to the nucleus in a nonbinding manner. Infections were carried out using a multiplicity of infection (MOI) of six transducing units per cell. Transduction efficiencies of >95% were observed using fluorescence-activated cell sorting. Our extensive validation experiments have shown that the expression of soluble or nuclear-restricted GFP does not negatively alter functional cell biology (e.g., morphology, proliferation, migration, and differentiation). Following virus generation and titering, HUVEC infection was optimized by exposing 1.25 × 105 cells seeded in a T-25 flask with different concentrations of lentivirus. Briefly, cells were seeded 1 day prior to infection. On the day of infection, cells were washed with phosphate-buffered saline (PBS), and growth media containing the virus were added to the HUVEC flasks. Cells were grown for 48 to 72 h prior to harvest and analysis of infectivity by flow cytometry. To determine infection efficiency, the number of fluorescently expressing HUVEC was divided by the total cell count and expressed as a percentage. The optimal MOI chosen for final infection was one in which cell proliferation, as measured by confluence, was not affected compared with uninfected HUVEC ( Fig. 1B , D ) and ≥90% of the HUVEC were expressing GFP, as determined by flow cytometry analysis. For co-culture HUVEC, cells were infected with either CytoLight Green or NucLight Green at an MOI of 6 and frozen 48 h after infection in assay-ready aliquots.
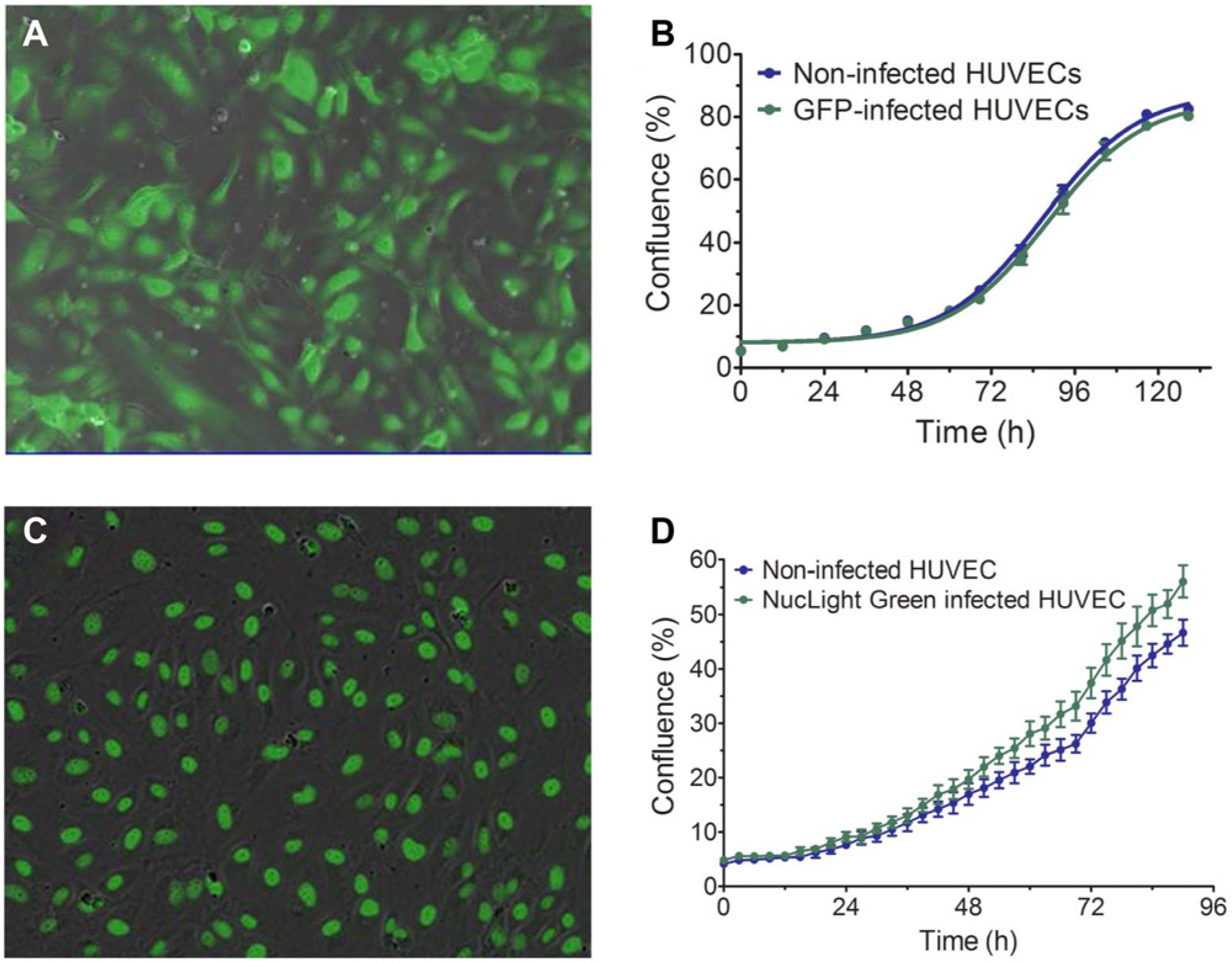
Lentiviral infection of human umbilical vein endothelial cells (HUVEC) does not alter cell properties. (
Angiogenesis PrimeKit (HUVEC/NHDF Co-culture)
The CellPlayer Angiogenesis PrimeKit is performed essentially as previously described, 6 while being optimized for 96-well format. The assay is initiated by thawing NHDF, rinsing, and plating in seeding media into the 96-well plate (Corning Life Sciences, Lowell, MA). On day 0, the NHDF were then incubated at room temperature in a tissue culture hood for 1 h to allow them to adhere to the plate. The HUVEC were then plated onto the NHDF feeder layers at a seeding ratio between 2:1 and 8:1. Following seeding of the HUVEC CytoLight Green, the plate was incubated at room temperature for 1 h prior to placing in the IncuCyte (Essen BioScience) for imaging. Treatment initiation occurred on day 4 of the assay, with a complete media change including growth factor and compound addition. Cultures were then fed on day 5 and every 2 to 3 days thereafter, at which time complete media changes occurred with fresh growth factor and compound addition.
Following seeding, co-cultures were placed in an IncuCyte live-content imaging system, and images were automatically acquired in both phase and fluorescence every 6 to 12 h for 10 to 14 days at 4× (single image) or 10× using the tiled field of view mosaic imaging mode. In this mode, six images (1.90 mm × 1.52 mm per image in a three-images-wide × two-images-high mosaic) are acquired per well and merged into a single, larger image (3.92 mm × 3.37 mm; total area = 13.21 mm2). Imaging in either 4× or 10× resulted in nearly 50% well coverage.
Experimental and Statistical Validation of PrimeKit Performance
The cell densities and growth conditions are optimized for assay performance, yielding a significant amount of VEGF-mediated tube and network formation over the 14-day time course. To ensure assay performance, tube length values had to meet or exceed the previously described values for VEGF stimulation.5,6 To do this, we imposed restrictions that the tubes formed upon stimulation with 4 ng/mL VEGF must obtain a length of at least 7 mm/mm2. Amassing data over multiple experiments and multiple cell batches more completely defined the performance of the PrimeKit over time (
Analysis of Vascular Network Development
Tube formation over the 14-day assay was quantified using the Essen BioScience Angiogenesis Analysis Module. This module provides multiple assay metrics, including tube length and branch point formation, which are used to assess both pro- and anti-angiogenic effects on network formation. Briefly, the fluorescent images were analyzed to generate a segmentation mask closely resembling the in vitro network. The mask was then refined to specifically identify tube-forming events, and the kinetic response was plotted using the IncuCyte and GraphPad Prism software (La Jolla, CA). In addition, the ability to export images and movies using the IncuCyte software enables the user to further verify and support the quantitative metrics.
For CD-31 end-point staining, co-cultures were grown and treated as described above. On day 14, cultures were fixed with 4% paraformaldehyde, washed with PBS, and permeabilized with 0.1% Triton X-100. Following permeabilization, cells were blocked in PBS containing 2% goat serum, followed by incubation with an anti-CD-31 antibody (R&D Systems, Minneapolis, MN) and goat anti-mouse AlexaFluor secondary antibody (Invitrogen). Cells were placed in the IncuCyte for imaging and quantitation.
Measuring Cell Confluence
Confluence is calculated using phase-contrast HD images in conjunction with a proprietary algorithm designed for use with the IncuCyte system (Essen BioScience). In general, the segmentation algorithm separates cells from background using brightness and texture features. The algorithm can adapt to different cell types during a training step. In this step, the user selects a collection of representative images, and the algorithm computes the features model used to segment the cells from the background. Here, HUVEC were seeded in a T-25 flask and placed in an IncuCyte for phase contrast and fluorescent imaging. To measure confluence, phase contrast images were collected over time, taking updates every 2 to 3 h, and confluence was measured, as described previously.8,9
Data Analysis
Data were exported from the IncuCyte software into Microsoft Excel. All data were then imported into GraphPad Prism software (La Jolla, CA) for graphing and statistical analysis. Raw data were imported into GraphPad Prism5 and graphed as the mean ± standard error for each sample. Statistical significance was determined by a one-way analysis of variance using a Tukey posttest to compare column pairs.
Results and Discussion
Fluorescent Nonperturbing HUVEC Labeling
The vascular networks formed in the HUVEC/NHDF co-culture approach closely mimic in vivo vasculature, both in tube length and network density.5,6 In the co-culture setting, although the phase images are useful for investigating the general health of each cell type, it is difficult to visualize HUVEC tube formation kinetically without a specific endothelial cell label. Lentiviral infection of HUVEC with a soluble GFP construct resulted in nearly 95% transduction efficiency, as measured by flow cytometry analysis ( Fig. 1A ). Importantly, the presence of GFP in HUVEC did not alter their growth properties compared with uninfected HUVEC ( Fig. 1B ). Although measuring tube formation over time is important, we were also interested in examining HUVEC proliferation in the co-culture setting. To do this, we developed a nuclear localized GFP construct that, when infected into cells, specifically labels cell nuclei with similar efficiency as soluble GFP ( Fig. 1C ). As with soluble GFP, the presence of the nuclear label did not alter the growth properties of HUVEC over time ( Fig. 1D ). Together, these tools provide the capability to measure HUVEC proliferation and differentiation kinetically in the presence of NHDF.
HUVEC Express Vascular Markers
When placed in co-culture with NHDF, the HUVEC proliferate, migrate, and differentiate into tubes with minimal network formation ( Fig. 2A ). To further characterize the tubes that formed, the cells were fixed and stained for endothelial and vascular markers. Despite being less complex, the HUVEC remain positive for two endothelial markers, CD-31 and the angiopoietin receptor Tie-2 ( Fig. 2B and E , respectively). When stimulated with cytokines, such as VEGF, the HUVEC differentiate into much more complex and extensive vascular networks ( Fig. 2G ). Similar to untreated cultures, VEGF stimulation maintains the staining with CD-31, where staining closely mirrored GFP expression ( Fig. 2H ). The merged image confirms the co-localization of CD-31 with the developing networks ( Fig. 2I ). Further investigation yields that the HUVEC maintain expression of the angiopoietin receptor protein, Tie-2 ( Fig. 2J - L ).
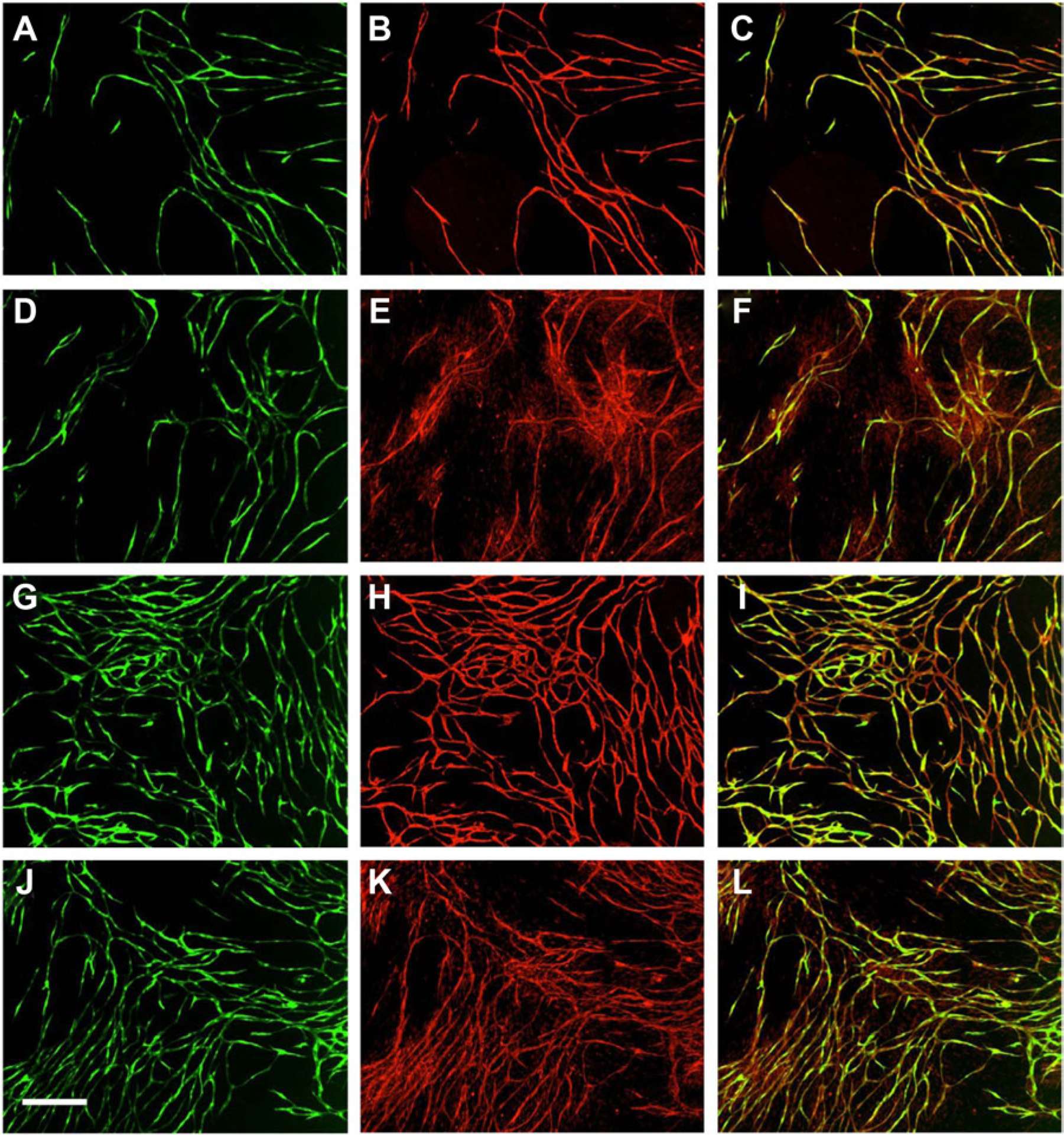
Expression of endothelial markers in human umbilical vein endothelial cells (HUVEC). (
Quantitating Tube Formation
One of the most difficult and time-consuming aspects of an in vitro angiogenesis assay is quantifying angiogenic events. The Angiogenesis Analysis Module in the IncuCyte live-content imaging system quantifies multiple metrics, including tube length, tube area, and branch point formation, over the course of each experiment (
Fig. 3B
,
E
) and has been optimized for providing a kinetic readout using the HUVEC/NHDF co-culture. In the first step of the process, the angiogenesis algorithm analyzes each fluorescent image (
Fig. 3A
,
D
) and assigns a segmentation mask that closely resembles the in vitro network (
Fig. 3B
,
E
). From here, the mask can be refined and filtered to exclude non–tube-forming events and measure angiogenesis over time. As an example shown in
Figure 3
, during network development, tube length increases from 0.09 mm/mm2 to 9.29 mm/mm2 (
Fig. 3B
,
E
). Kinetic plots of two of the angiogenesis metrics (tube length and branch points) can be generated, allowing for a direct and straightforward comparison of the wells of interest to validated control conditions (
Fig. 3C
,
F
, respectively, compared with
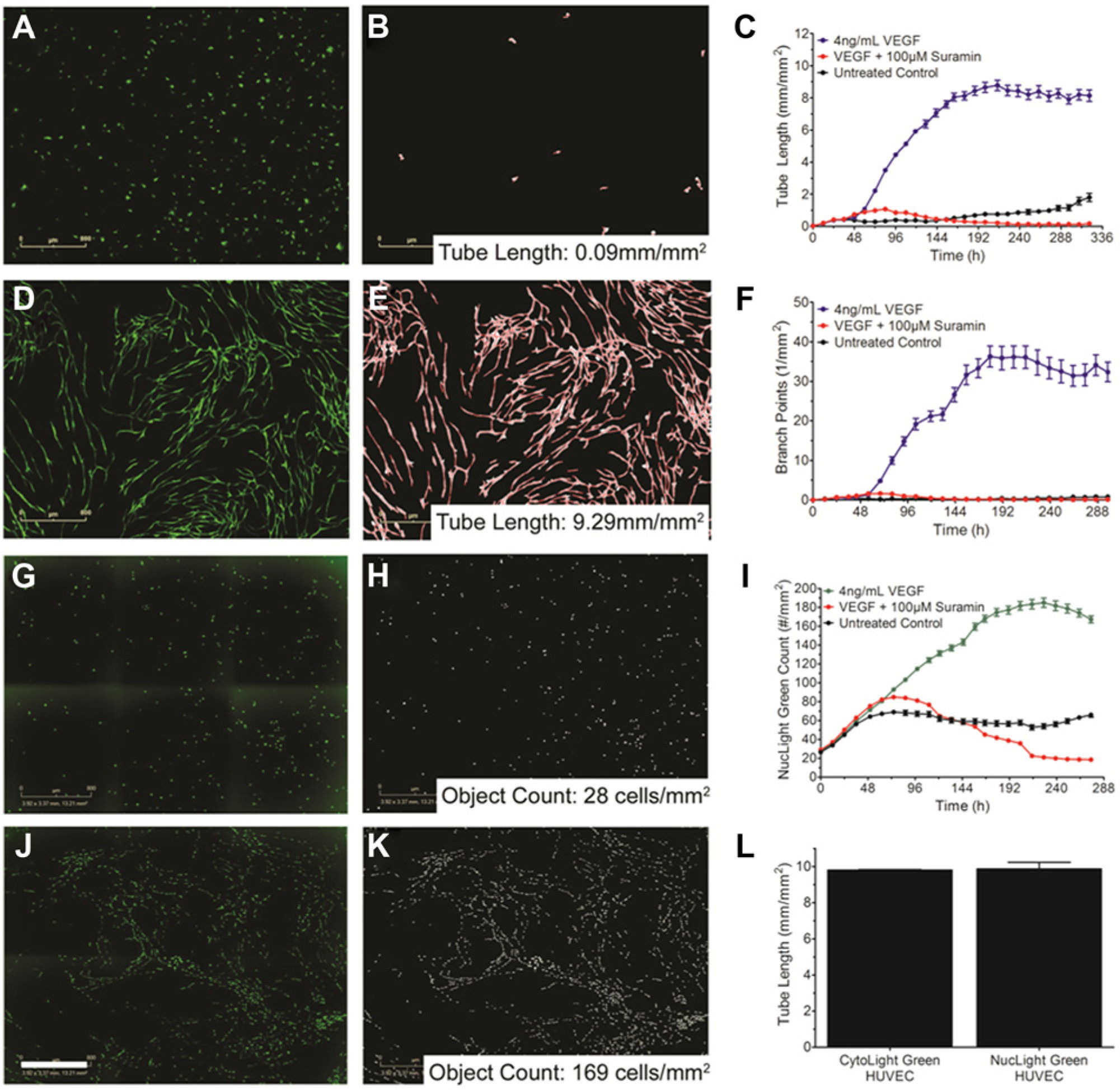
Quantifying tube formation and cell number using the live-content imaging system, IncuCyte. (
To investigate HUVEC proliferation, a similar approach segmenting the images to identify fluorescent objects was used to quantify HUVEC cell numbers over time. VEGF treatment increases the number of HUVEC from 28cells/mm2 to 169cells/mm2 by the end of the assay ( Fig. 3G , J ). Interestingly, as the assay progresses, the HUVEC nuclei align such that tube formation can be followed over time. Nuclear count over time confirms that VEGF stimulates extensive proliferation over the first 190 h of the assay ( Fig. 3H , K ). After the 190 h time point, the HUVEC appear to reach steady state and no longer are undergoing proliferation ( Fig. 3I ). When no exogenous growth factor is added, the HUVEC reach steady state within the first 48 h and maintain throughout the assay, whereas treatment with Suramin results in a nearly complete loss of HUVEC ( Fig. 3I ). To quantify the amount of tube formation in the NucLight HUVEC, cells were fixed on day 14 and stained for the endothelial cell marker CD-31. The same algorithm and segmentation parameters for quantifying kinetic tube formation in CytoLight Green HUVEC were used to quantify endpoint CD-31 stained cultures. CD-31 staining at the end of the assay revealed that the nuclear marker did not affect tube formation ( Fig. 3L ). Together, these models can be used to investigate HUVEC proliferation and tube formation in a co-culture setting.
Measuring Growth Factor Promotion of Angiogenesis
Multiple growth factors, including VEGF and bFGF, play an integral role in the stimulation, differentiation, and development of endothelial and mural cells into angiogenic networks in vivo. 3 As VEGF is one of the major growth factors in this process, inhibition of VEGF signaling is a prevalent therapeutic target in diseases such as cancer, macular degeneration, rheumatoid arthritis, and other pathologies.3,10 More recently, proangiogenic therapies are also being developed to treat critical limb ischemia and cardiovascular diseases.3,10 Thus, investigating the complexities and sensitivities of cytokine signaling is important to establishing a robust in vitro angiogenesis model. To this end, we examined how increasing VEGF concentrations could induce tube formation over the 14 day assay period. In this co-culture model, the media conditions have been optimized to maximize the signal window above baseline. In the absence of exogenous growth factor addition, the extent of tube formation and branching remains low ( Fig. 3C , F ). To measure the effect of VEGF, the indicated concentrations were added on day 2 and maintained throughout the assay ( Fig. 4A , B ). As soon as VEGF is introduced, the HUVEC respond, begin to elongate, and differentiate into tubes. The extent of overall tube length and branch point formation is dependent on the VEGF concentration, with 1 ng/mL exhibiting a 50% response whereas 4 ng/mL VEGF reaches maximal stimulation ( Fig. 4A , B ).
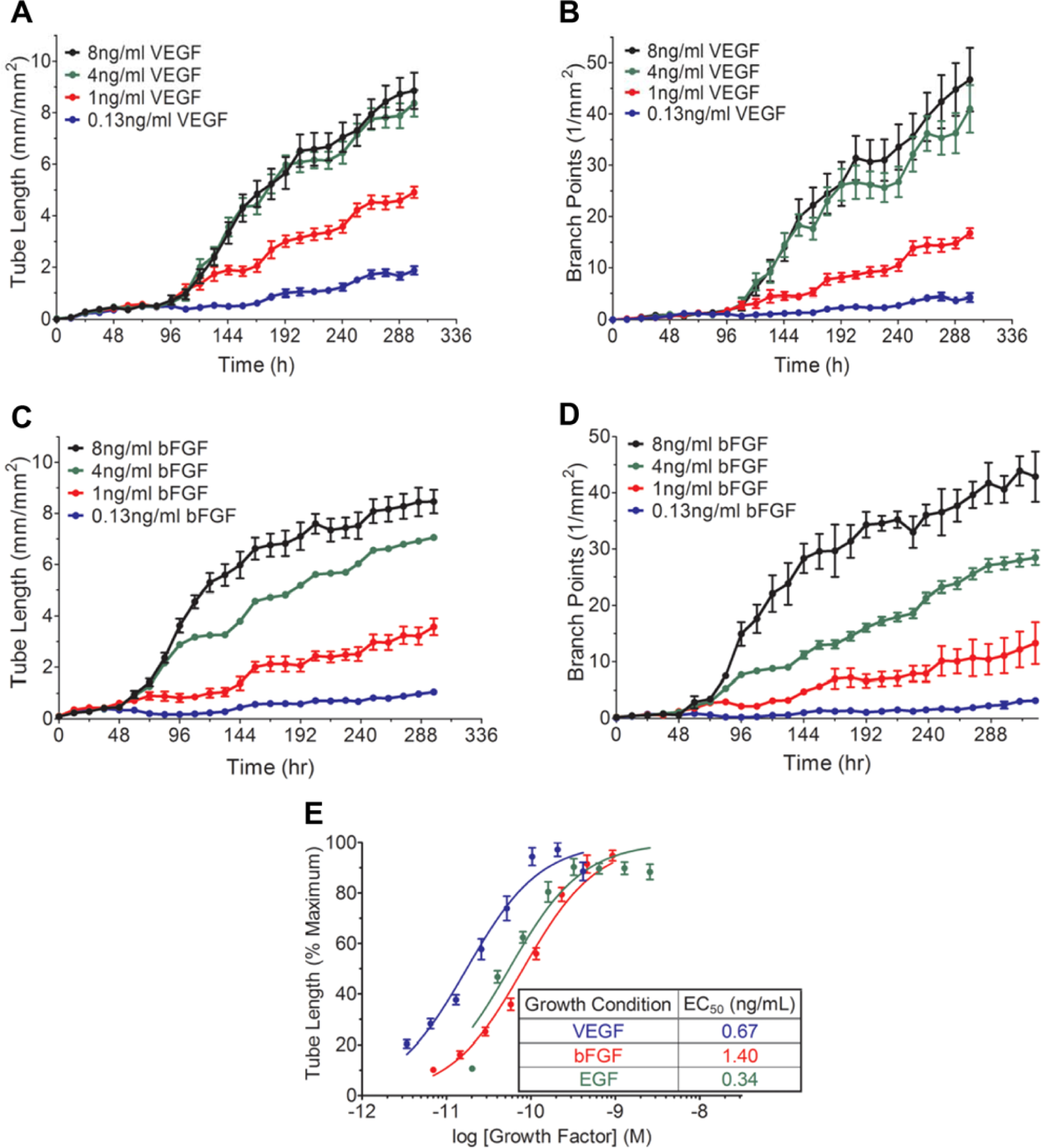
Growth factor stimulation of tube formation. (
Although VEGF is an integral proangiogenic cytokine, it has been well established that multiple growth factors can support angiogenesis. 11 Accordingly, we have also investigated the angiogenic effects of bFGF and EGF. Cultures were seeded and either bFGF or EGF treatment was initiated on day 2 at the indicated concentrations ( Fig. 4C , D , EGF not shown). As can be seen in Figure 4 , cultures responded to bFGF, resulting in a concentration-dependent increase in both tube length and branch point formation ( Fig. 4C , D ). Interestingly, the bFGF response in this assay is, to a small degree, mediated through a VEGF signaling pathway, as anti-VEGF antibodies can moderately affect bFGF-mediated angiogenesis. 12 Perhaps a more indicative measure of the growth factor efficacy and potency is to examine the EC50 values for each cytokine. Replotting the data at the endpoint (300 h) as a function of concentration revealed nearly identical EC50 values for VEGF, bFGF, and EGF indicating, that the HUVEC/NHDF co-culture responds to a variety of proangiogenic stimuli ( Fig. 4E ).
Differentiating VEGF- and bFGF-Mediated Angiogenesis
Studies have indicated that VEGF and bFGF play different roles in the angiogenic process.13,14 Specifically, bFGF has been shown to have a more profound effect on endothelial cell survival.
13
To investigate VEGF and bFGF effects on HUVEC proliferation in a co-culture setting, we infected HUVEC with a nuclear-localized GFP marker. Segmentation of these nuclei over time provides an accurate quantification of cell count throughout the assay.
Correlations between In Vitro Results and In Vivo Pharmacology
Due to the increasing clinical emphasis on anti-angiogenic therapies, the ability to compare in vitro results to in vivo pharmacology is imperative for developing the next generation of anti-angiogenic therapies. Here, we investigated the effects of two compounds, Axitinib and PD-161,570, which have shown to be growth factor selective and anti-angiogenic both in vitro and in vivo.15–18 Axitinib is currently used in the clinic for treatment of patients with renal cell carcinoma who have failed to respond to other interventions. Using our in vitro model, axitinib treatment in the presence of VEGF potently inhibited VEGF-mediated angiogenesis ( Fig. 5A ). Although axitinib inhibited bFGF-mediated angiogenesis ( Fig. 5B ), it was much less potent and did not reach complete inhibition at the concentrations tested ( Fig. 5B , C ). Effects of axitinib on both VEGF- and bFGF-mediated branch point formation followed the same trends as their respective inhibition of tube length (data not shown). When data at the 300 h time point were regraphed as a function of axitinib concentration, a rightward shift in the concentration-response curve of axitinib is observed in the presence of bFGF compared with VEGF ( Fig. 5C ). This resulted in a nearly fourfold shift in the IC50 from 15 nM to 50 nM for VEGF and bFGF-mediated angiogenesis, respectively, demonstrating the moderate selectivity of axitinib for inhibiting VEGF-mediated signaling ( Fig. 5C ). These results reflect the previously characterized in vitro biochemical studies, in which a shift in the IC50 of axitinib was observed in the presence of various growth factors. 15 Although the selective effects on VEGF versus bFGF signaling observed in the biochemical studies were larger, 15 this is likely due to the isolated nature of the receptor environment in a binding assay versus the complex molecular interactions of a co-culture angiogenesis approach. As indicated above, a portion of the proangiogenic response to bFGF is mediated through VEGF signaling. 12 Thus, it is not surprising that axitinib would have an effect on co-cultures treated with bFGF, an outcome that would be missed in isolated biochemical studies. Importantly, these data reflect previously published in vivo studies, 16 in which axitinib treatment was shown to induce partial remission of renal cell carcinomas and reduce lung metastases in humans. 16 Taken together, these studies indicate that results from this in vitro model correlate well with in vivo pharmacology.
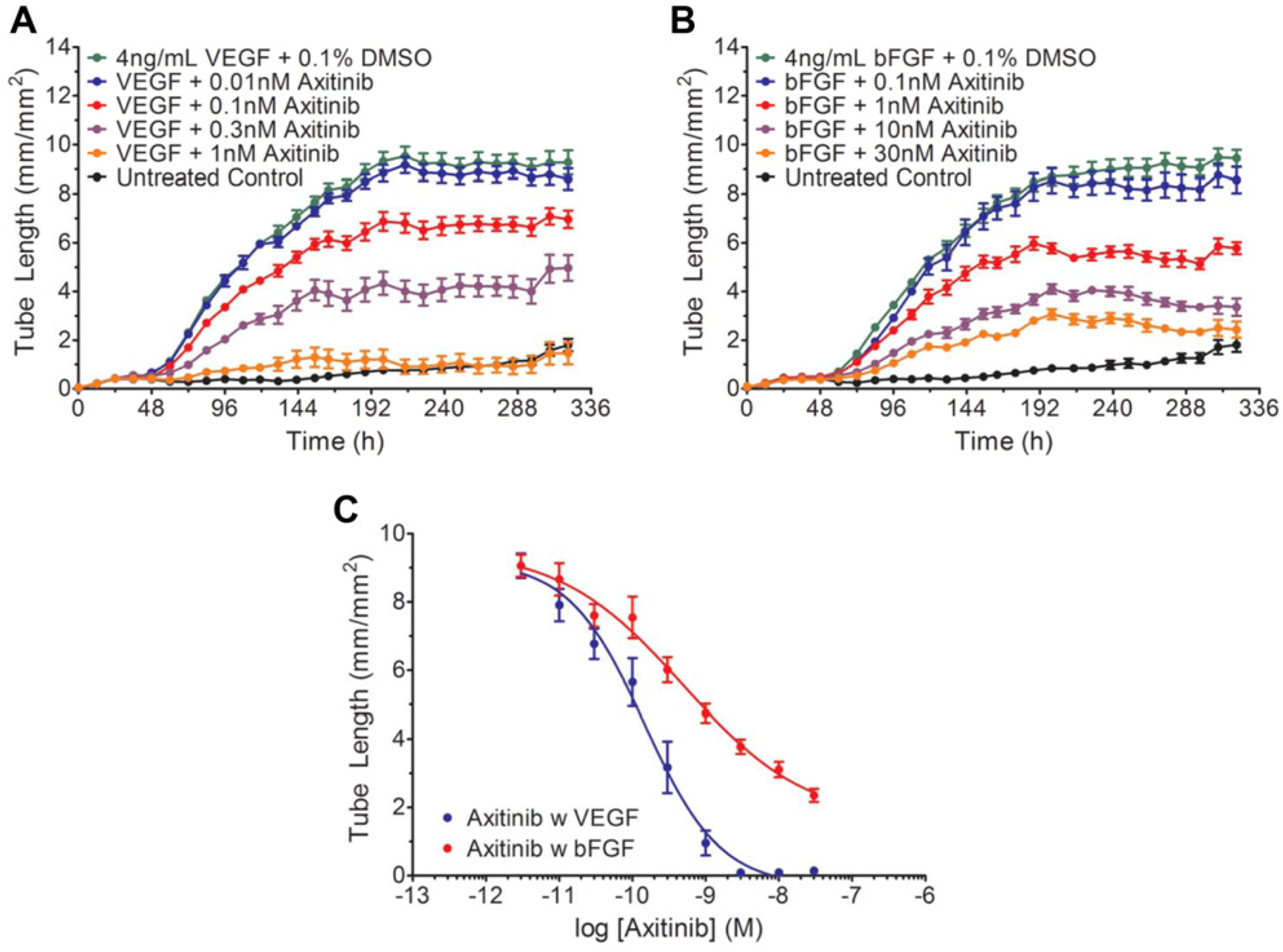
Treatment with axitinib selectively inhibits vascular endothelial growth factor (VEGF)–driven angiogenesis. (
Interestingly, when we investigated the effects of PD-161570, we observed the converse effect, namely, that PD-161570 was more effective in inhibiting bFGF- than VEGF-mediated angiogenesis ( Fig. 6A , B ). Here, PD-161570 treatment in the presence of VEGF resulted in a concentration-dependent inhibition of angiogenesis ( Fig. 6A ). However, only at the highest concentration, 3 µM, did we observe complete inhibition. When PD-161570 was added in the presence of bFGF, however, a more potent inhibition was observed ( Fig. 6B ). Under these conditions, 0.1 µM PD-161570 resulted in a 50% inhibition of bFGF-mediated angiogenesis, and complete inhibition was observed at 0.3 µM PD-161570 and greater. This is reflected in the concentration-response curves, in which a nearly eightfold rightward shift in the IC50 of PD-161570 is observed in the presence of VEGF ( Fig. 6C ; 100 nM IC50 for bFGF and 800 nM IC50 for VEGF). These results support the previous biochemical findings, in which PD-161570 was shown to be selective for inhibiting bFGF-mediated signaling through the FGFR-1 receptor.17,18 As described previously, the higher cross-reactivity of PD-161570 on VEGF signaling is likely due to the complex molecular interactions of the co-culture environment. Notably, a compound exhibiting this type of selectivity may be important clinically in such diseases as diabetic retinopathy, atherosclerosis, and tumor neovascularization, in which abnormal FGF receptor expression has been implicated in disease progression. 17
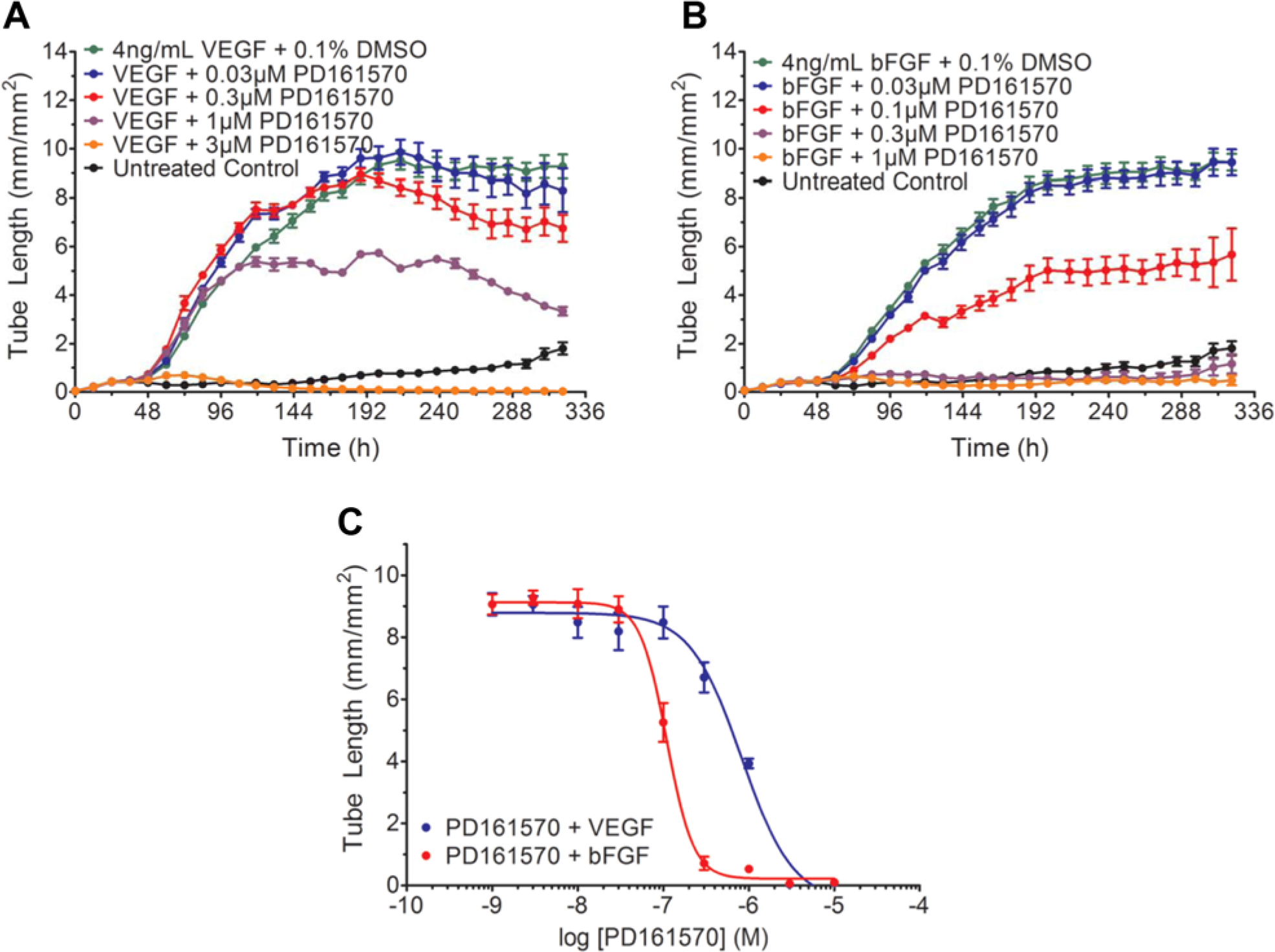
Treatment with PD-151670 selectively inhibits basic fibroblast growth factor (bFGF)–mediated angiogenesis. (
In addition to showing growth factor selectivity for small-molecule inhibitors, it was important to demonstrate that not all tyrosine kinase inhibitors are active in this model. Lestaurtinib, a potent JAK2, FLT3, and TrkA inhibitor that exhibits antiproliferative activity in vitro and is used for myeloproliferative disorders in vivo, has not been shown to be active in affecting angiogenesis.19,20 When added in the presence of either VEGF or bFGF, lestaurtinib did not significant alter angiogenesis (
Angiogenesis and vasculogenesis are critical physiological processes that occur during development, wound healing, and pregnancy, to name a few.3,21,22 Under pathological conditions, such as tumorigenesis, intraocular disease, cardiovascular disease, and rheumatoid arthritis, extensive, yet incomplete, angiogenesis becomes driven by the microenvironment of the particular pathology.21,23,24 Despite an increasing understanding of the molecular drivers of vascular network formation, many angiogenic processes controlling morphologic events, and the factors that regulate them, remain unclear. 21 In addition, although combination therapies attacking the tumor microenvironment and vasculature have shown promise, the increasing occurrence of vascular resistance or refractoriness calls for the development of novel therapeutic strategies to attack angiogenic pathologies.
Given a highly reproducible, quality-controlled format, an in vitro angiogenesis assay can prove to be a useful tool in studying complex mechanisms and therapeutic interventions of vascular formation.5–7 Importantly, these models must try to accurately re-create the physiology of the tissue environment. Although the NHDF/HUVEC co-culture is limited by the absence of pericyte biology, this model, contrary to previous models, 7 recapitulates all phases of the angiogenic process, including HUVEC proliferation. Further, in developing in vitro models, it is essential to correlate in vitro results to in vivo pharmacology. As shown here, the pharmacological and physiological effects obtained using the HUVEC/NHDF co-culture assay format are highly correlative to previous in vivo characterizations. Despite the length of the assay, the kinetic nature, in conjunction with high-resolution images, increases the depth of knowledge obtained from a single experiment. Further, it is becoming clear that cytokines outside of VEGF are important players in vascular development and maintenance.3,10,25 The ability to investigate cytokine dynamics in an in vitro setting strengthens the relevance and correlative aspect of this co-culture model. This is exemplified by the axitinib and PD-161570 studies, in which not only do we observe differing sensitivities to VEGF- and bFGF-driven angiogenesis but the results also replicate trends demonstrated in in vivo clinical and pharmacologic studies.
Similar co-culture models have been described yet are not as information rich as this live content approach to investigating angiogenesis. 7 Although other models must rely on either endpoint imaging or removing plates from their incubator to image using a high-content imaging system, 7 combining the NHDF/HUVEC model with the IncuCyte live content imaging system provides kinetic, time-course data collected on the same cell population maintained under physiologic conditions throughout the course of the experiment. Because no initial time course optimization is required, retrospective analysis of the kinetic data can be performed at any point during the assay, enabling a flexible approach to determine pharmacologic responses. As well as being able to measure complex biological responses, this in vitro co-culture model is a robust and reproducible assay, enabling a higher-throughput approach to investigating angiogenic responses. Finally, the IncuCyte allows for automated data acquisition of phase contrast and fluorescent images that can be used in combination with the Angiogenesis Analysis Module to more completely describe the angiogenic response. Together, the family of angiogenesis assays provides quantitative and powerful approaches for studying angiogenesis in vitro.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
