Abstract
In early drug discovery (e.g., in fragment screening), recognition of stereoisomeric structures is valuable and guides medicinal chemists to focus only on useful configurations. In this work, we concurrently screened mixtures of stereoisomers and estimated their affinities to a protein target (thrombin) using weak affinity chromatography–mass spectrometry (WAC-MS). Affinity determinations by WAC showed that minor changes in stereoisomeric configuration could have a major impact on affinity. The ability of WAC-MS to provide instant information about stereoselectivity and binding affinities directly from analyte mixtures is a great advantage in fragment library screening and drug lead development.
Keywords
Introduction
Fragment screening is an early hit identification strategy in drug discovery that has gained much attention during the past decade. 1 Several advantages have been recognized in fragment screening when compared with traditional high-throughput screening (HTS) of drug-like compounds. These include higher hit rate (especially for difficult targets) and higher quality of identified hits. Due to their small sizes, fragments suffer less from steric hindrance in the site-binding process. Fragment libraries, for the same reason, can sample the chemical space more efficiently even though they are quite small (about 1000–3000 compounds).1,2 At present, most fragment libraries are dominated by planar aromatic compounds due to the popularity of sp 2 -coupling reactions. 3 Another reason is that planar aromatic structures serve as hydrophobic frames that stabilize the molecules and subsequently facilitate interactions. However, the mainly planar feature of fragment libraries reduces the chemical variability, and by including more 3D characters (sp 3 character and chirality), a higher diversity can be achieved. 4 As a result, this will, among other things, increase the number of compounds with one or several stereocenters, which may be important as chiral compounds can be more specific. 3 Information about enantioselectivity (stereoselectivity) in target binding in the early stages of drug development is therefore of high importance. Recording chiral selectivity is also essential in the development of an initial fragment hit into a more drug-like lead compound. In this case, a selected structure is elaborated in different directions, and collections of related compounds are created in the process. The synthesis of these elaborated fragments often produces chiral or diastereomeric mixtures of the desired product that has to be evaluated.
In fragment screening, the affinity of identified initial fragment hits and first round of evolved fragments is normally in the range of 10 mM to 100 µM in the dissociation constant (KD). Sensitive screening techniques such as X-ray crystallography, nuclear magnetic resonance, surface plasmon resonance, affinity selection–mass spectroscopy, frontal affinity chromatography–mass spectroscopy, and weak affinity chromatography (WAC) have been used to detect such low-affinity binders. 5 WAC, when incorporated with mass spectrometry (MS), is perfectly suited for measuring the binding of individual fragments in mixtures because it is based on separation. Another benefit with this technique is that a high throughput of thousands of fragments per day can be achieved. 6
The present work demonstrates the applicability of WAC with MS detection to determine the affinity of stereoisomeric fragments to thrombin, an important serine protease in the coagulation cascade. Screening was performed with enantiomeric as well as diastereomeric mixtures of fragments and elaborated fragments, respectively.
Materials and Methods
Chemicals
All chemicals, including water, were of analytical grade or higher and purchased from Sigma Aldrich (St. Louis, MO), if not otherwise stated. Human α-thrombin (2400 IU/mg) was a gift from Octapharma (Stockholm, Sweden).
WAC with MS Detection
Human α-thrombin, purified by published methods, 7 was in situ immobilized onto porous diol-based spherical silica particles (Kromasil, 5 µm in diameter, 300-Å pore size; Eka Chemicals, Bohus, Sweden) that had been prepacked into capillary columns (35 mm × 0.5 mm i.d.; Agilent Techno-logies, Waldbronn, Germany). The immobilization procedure, which has been described in detail previously,6,8 included oxidation of diol silica to aldehyde silica followed by covalent coupling to primary amino groups of thrombin by reductive amination chemistry (Schiff base method). The reaction creates covalent bonds (aminolinks) between the high-performance silica support and the protein. 9 Two reference columns, with the same size and silica material as the active ones, were used to estimate nonspecific binding. The first of these reference columns contained PPACK-inhibited thrombin (inhibition procedure has been reported previously 6 ), and it was used to quantify binding to noncatalytic sites of the protein. The second reference column was a blank diol-silica column that was used to determine binding to the supporting silica material.
A liquid chromatography–mass spectrometry (LC-MS) instrument dedicated for capillary columns (1200 LC system/6130 single quadrupole mass spectrometer; Agilent Technologies) constituted the WAC-MS platform. Mobile phase was 20 mM ammonium acetate, pH 6.8–6.9 (AmAc). Flow rate was 14 µL/min and injection volume was 0.4 µL, if not otherwise stated. The system was controlled by ChemStation software (Rev. B.04.01 SP1; Agilent Technologies). The columns were thermostated at 22 14;°C during analysis. Ionization was carried out by electrospray ionization employing a nebulizer pressure of 20 psig and a capillary voltage of +3500 V in positive phase when analyzing diastereomers or in alternating positive (+3500 V) and negative (–2200 V) phases when analyzing fragments from the TimTec library (TimTec LLC, Newark, DE; see below for more details). The nitrogen drying gas flow was 7 L/min, and gas temperature was 300 14;°C. Fragmentor voltage was 100 V and quadrupole temperature was 100 14;°C. The MS detector was run in selected ion monitoring (SIM) acquisition mode. Target masses for SIM detection were selected to match the components in the analyzed mixture. Alternating SIM and scan acquisition modes (m/z range of 250–290) were used in one case for detection of the enantiomeric mixture ST058742.
TimTec fragments were analyzed by single injections of three 10-component mixtures (see below) on the active and PPACK-inhibited thrombin columns. The binding of the TimTec hit ST058742 was confirmed by analyzing it separately both on the active thrombin column and the reference columns (PPACK-inhibited thrombin and diol-silica columns). Analysis of the diastereomers was carried out by a single injection of a mixture including all the compounds (see below) on the active thrombin column. The mixture was analyzed in triplicate also on the reference columns. In each series of injections, the known thrombin binders benzamidine (BZA), 3-aminobenzamidine (3-ABA), and 4-aminobenzamidine (4-ABA), each at a concentration of 0.1 mM in water, were included as positive binding references. Dimethyl sulfoxide (DMSO) (0.1 mM) was employed as a void marker. The data collection window was 20 min for TimTec fragments and 900 min for the diastereomer mixture.
Selection of Chiral Racemates for WAC-MS Analysis
The commercial TimTec fragment library PO6300-FBL-3200 (TimTec LLC) was used to evaluate the ability of WAC to separate enantiomers. This is a general fragment library that comprises 3200 fragments with a molecular weight (MW) between 75 and 289 with an average of 216. The number of nonhydrogen atoms of the fragments is between 5 and 21, and clogP values are from −4.24 to 2.52. The concentration of each fragment in the library is 1.5 mM in 100% DMSO.
The TimTec library was manually searched for compounds that included chiral centers. The fragments that contained one chiral center (433 fragments) were in silico desalted and the tautomers were generated of all possible stereoisomers using LigPrep (v.2.4, Schrödinger Suite 2010; Schrödinger, LLC, New York, NY). The resulting structures were docked into two different crystal structures of human thrombin using Glide (v.5.6, Schrödinger Suite 2010) in extra precision mode. The protein structures were downloaded from the Protein Database Bank (PDB ID: 3DA9, 10 resolution of 1.80 Å and PDB ID: 1K22, 11 resolution of 1.93 Å). The most negative GlideScores of each fragment docked to the different thrombin structures were averaged and the top 30 fragments were chosen for experimental evaluation. Equal volumes of the selected fragments were distributed into three mixtures, each comprising 10 fragments having nonoverlapping molecular weights. The resulting mixtures were diluted 20 times with water to a concentration of 7.5 µM of each fragment and a DMSO concentration of 5%. All mixtures were analyzed on the active thrombin column as well as on the PPACK-inhibited column as described above.
Preparation of Diastereomers for WAC-MS Analysis
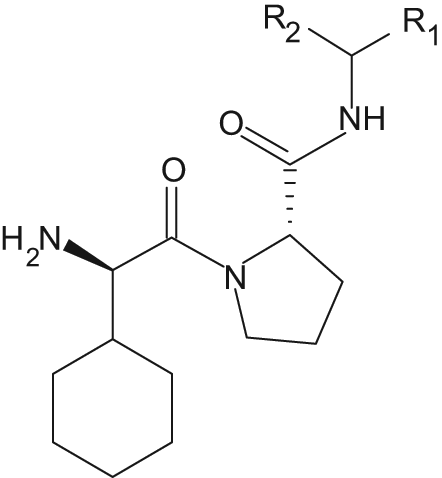
A collection of compounds, with an expected affinity toward thrombin, was synthesized according to
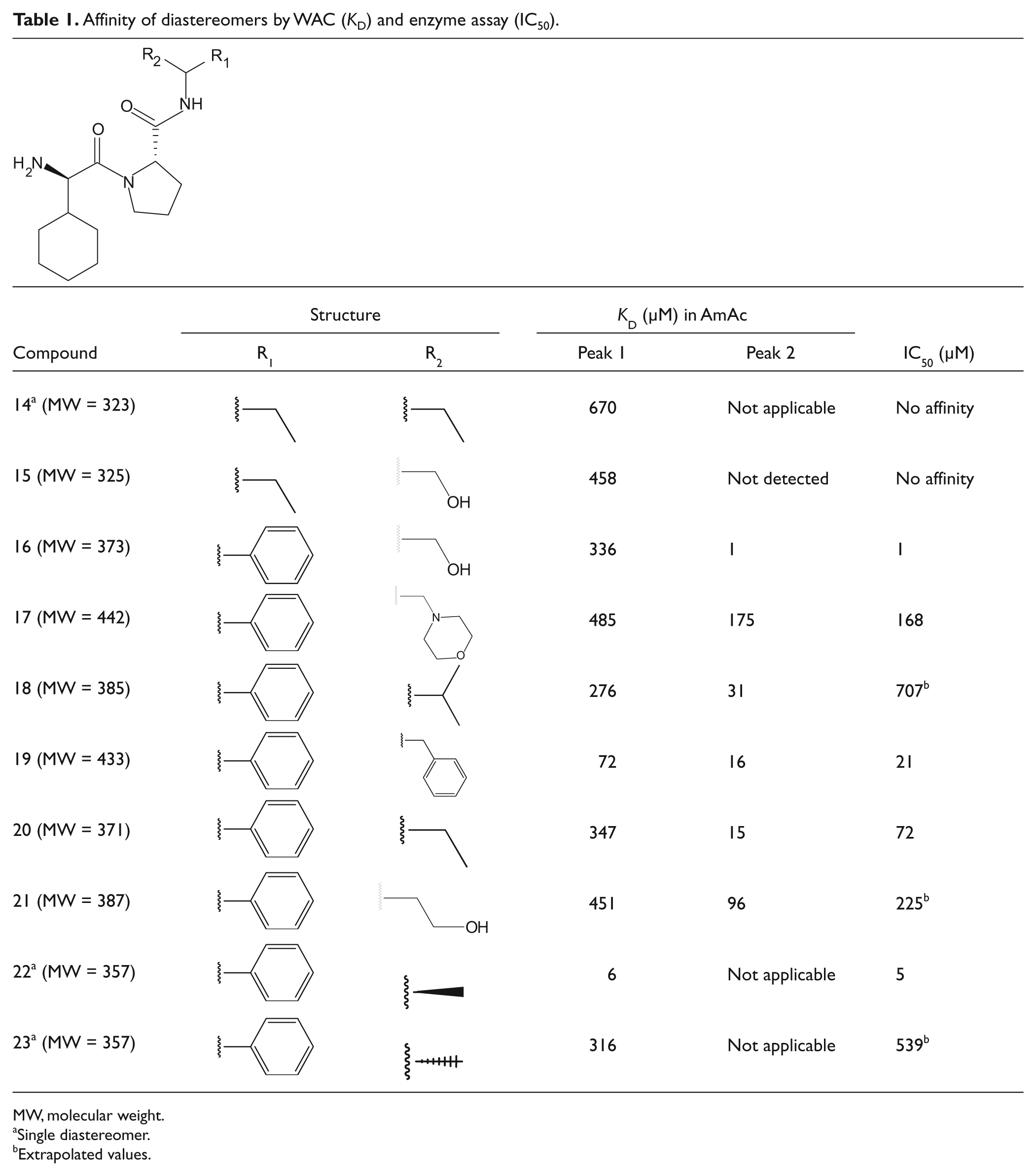
Affinity of diastereomers by WAC (KD) and enzyme assay (IC50).
MW, molecular weight.
Single diastereomer.
Extrapolated values.
All compounds (14–23) were mixed and diluted with water to a concentration of approximately 0.05 mM of each diastereomer (except for 14, 22, and 23, which were 0.1 mM). The resulting DMSO concentration of the mixture was 10%. The obtained mixture was analyzed on the active thrombin column as well as the reference columns (PPACK-inhibited column and diol-silica column) as described above.
Chromatography Evaluation
The retention time of each compound (injected as a single solution or in a mixture) was determined from the peak apex in the chromatogram. The specific retention time (tspec) was defined as the difference in retention on the active column and the PPACK-inhibited column (tspec = tactive – tPPACK). The affinity of each compound (KD) was calculated from equation (1). Btot is the total number of active sites in the WAC column and F is the mobile phase flow rate. Retention (tspec) of three reference binders (0.1 mM BZA, 3-ABA, and 4-ABA) was used to determine Btot of thrombin columns 1 and 2. It was done by comparing the average retention (tspec) of the reference compounds with previously obtained values, under identical conditions, using thrombin columns with a known Btot (determined with frontal chromatography 6 ). Assuming that equation (1) is valid, tspec is directly related to Btot for a specific compound and Btot can be estimated from the tspec ratio of the “calibrated” and “unknown” columns. The resulting Btot was 7.5 nmol for column 1 and 0.8 nmol for column 2.
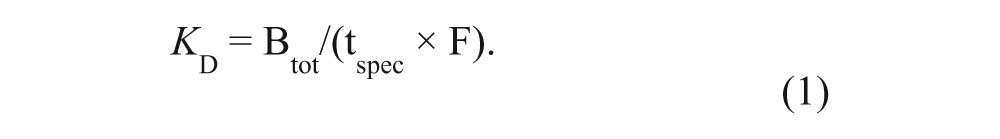
Equation (1) is a simplification that is valid for chromatography under linear conditions, where the concentration of the injected analyte should be much smaller than the KD of the interaction. 8
Thrombin inhibitory activity of the diastereomeric collection was measured by an enzyme inhibitory assay. Experimental details of the assay can be found in the Supplementary Information.
Results and Discussion
Characteristics of the WAC-MS Platform
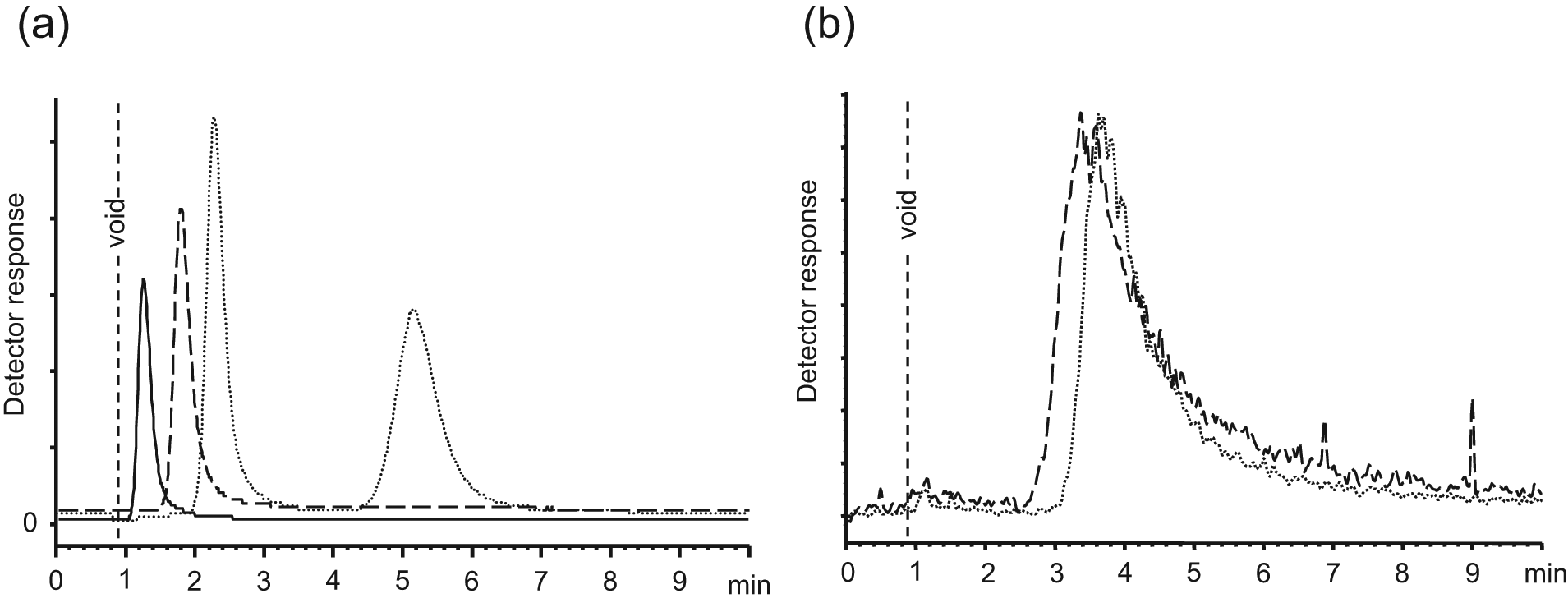
High-throughput screening with WAC-MS is normally performed by the evaluation of compounds, such as fragments, in mixtures. In our studies, about 10 enantiomeric or diastereomeric pairs were included in each mixture, but higher numbers are possible. 6 Fragments are considered hits if the retention is considerably higher on the target (active protein) column as compared with suitable reference columns. Introduction of reference columns is vital since many fragments can be retained to some extent by weak interactions to off-site target protein surface or silica support ( Fig. 1a , b ).
(
The affinity of each fragment in a mixture was determined according to equation (1) to estimate specific binding. The concentrations of analytes used were 7.5 µM and 0.1 mM in TimTec and the diastereomeric collections, respectively. The higher concentration of the diastereomers was necessary to detect some of the tighter binders in this group, which, due to long retention times, gave wide peaks (band broadening) ( Fig. 2 ). These tighter binding compounds were in fact analyzed under conditions where equation (1) will underestimate the affinity of the interaction. The deviation is, however, of minor importance as the essential information is the relative affinity of the compounds in the collection, rather than the absolute affinity. By adjusting the protein amount on the affinity column to fit the expected affinity of a specific collection of compounds, extreme retention times can be avoided. Using capillary columns, the amount of target protein needed for fragment library screening, with an expected KD from 10 mM to 100 µM, is in the nanomolar range. The capillary columns used in the present study contained 7.5 and 0.8 nmol of active thrombin, respectively. Column 2 was created for the study of tight binders with a KD of about 1 µM (see below), and therefore a lower amount of thrombin was applied.
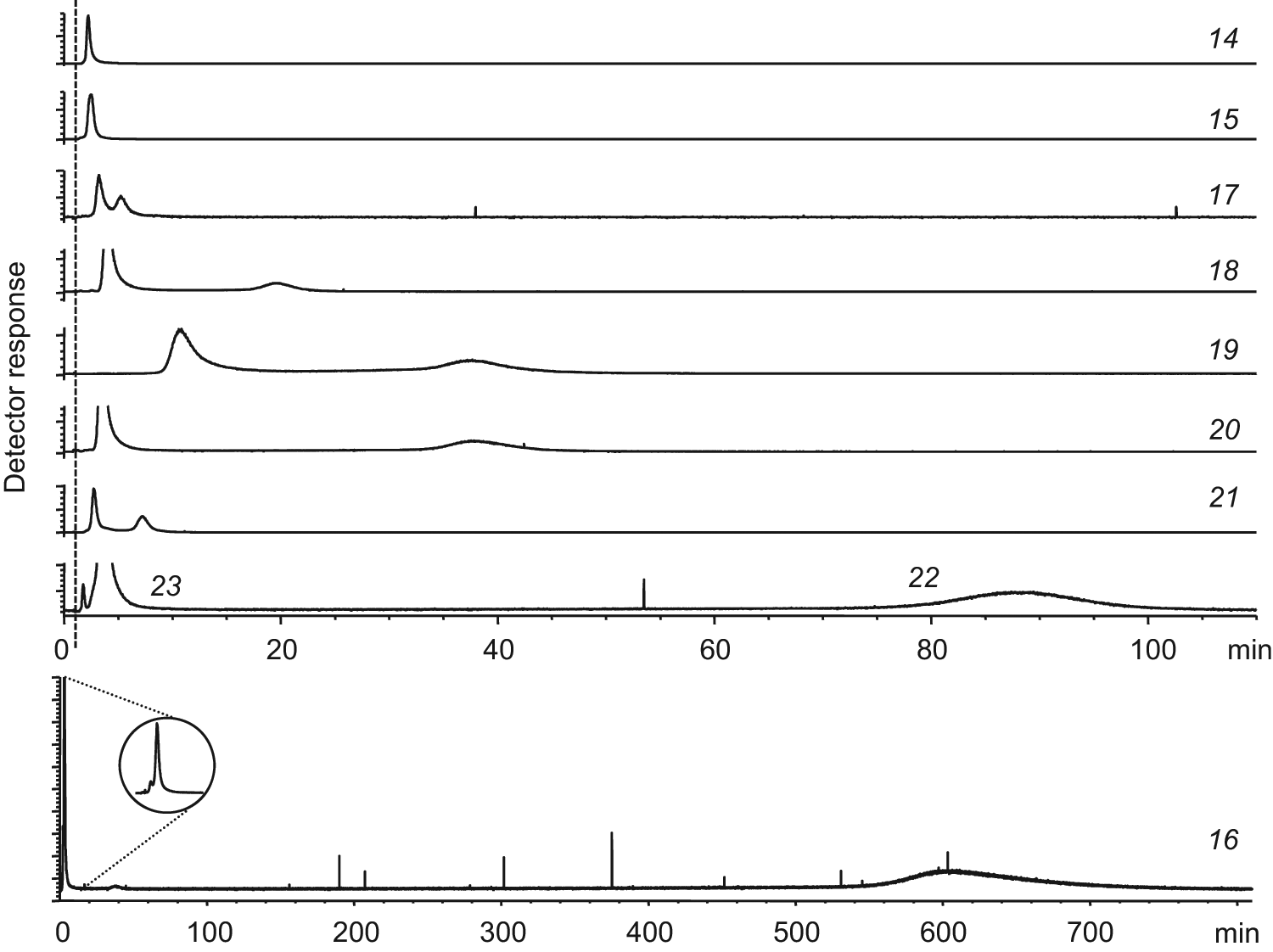
Extracted ion chromatograms of the mixture containing eight pairs of diastereomers and one single compound (14). The dashed line marks the void time. Please note the different time scale of diastereomeric pair (16) that contained a tight binder. The inset on chromatogram 16 is the full view of its first peak.
The performance in terms of operational stability of thrombin column 1 was studied by recording the retention time of a reference compound (0.1 mM BZA) for a period of time with an intensive workload. The retention was reduced about 20% after 65 injections of mixtures during 71 h using AmAc as the mobile phase. A total recovery of activity, however, was achieved by storing the column in a physiological buffer (10 mM sodium phosphate, 150 mM sodium chloride, 0.02% sodium azide, pH 7.4) at 4 14;°C for 60 days. The operational stability of this reconditioned column was higher, and during the next analysis campaign, which included more than 100 injections during 194 h of operation, only 17% of the retention was lost. To keep track of column performance, it is good practice to incorporate reference compounds as internal standards in the fragment mixtures.
WAC-MS in Chiral Separations
A binding site of a protein normally has a 3D structure and is therefore able to recognize and differentiate between stereoisomers. This property is used in LC separations of enantiomers based on proteins, such as albumin and α-1-acid glycoprotein, as chiral selectors. 12 Using WAC-MS for the analysis of a pair of enantiomers (a racemate) in a similar way will produce two peaks with an identical m/z value in the chromatograms, provided the enantiomers are recognized differently by the protein’s binding site. Identification of which peak belongs to the (R)- or (S)-enantiomer has to be accomplished in a separate experiment by analysis of the pure stereoisomers.
The potential prevalence of stereoisomers in the evaluated fragment library was determined by manually searching the 3200 fragments of the TimTec library for fragments that possessed at least one chiral center. In this search, 593 fragments were found, which constituted 18.5% of the whole library. The vast majority, 433 fragments, contained only one chiral center. According to the TimTec library manufacturer, these compounds are considered racemates, except for a few cases where the configurations were known (compounds ST024707 and ST060195 in
Only one fragment (ST058742) of the 30 evaluated racemates was separated into two peaks when analyzed on the thrombin column ( Fig. 3 ). The binding of ST058742 was confirmed to be thrombin specific as only a single, less retained peak appeared applying the PPACK reference column ( Fig. 1a ). Obviously, the (R)- and (S)-enantiomers of ST058742 interacted differently with the active site of thrombin, thereby producing two retained peaks on the active thrombin column, whereas the PPACK-blocked column was unable to separate the enantiomers. The tspec values of the enantiomers on column 1 were 0.5 min and 3.4 min, resulting in KD values of 1.07 mM and 0.16 mM, respectively. The enantiomers were baseline separated, having a resolution (Rs) of 3.5. In one experiment, the mass spectra were collected from m/z 250 to m/z 290 in scan mode. Here an identical fragmentation pattern was obtained, which supports the conclusion that the two peaks are from the enantiomers ( Fig. 3c , d ).
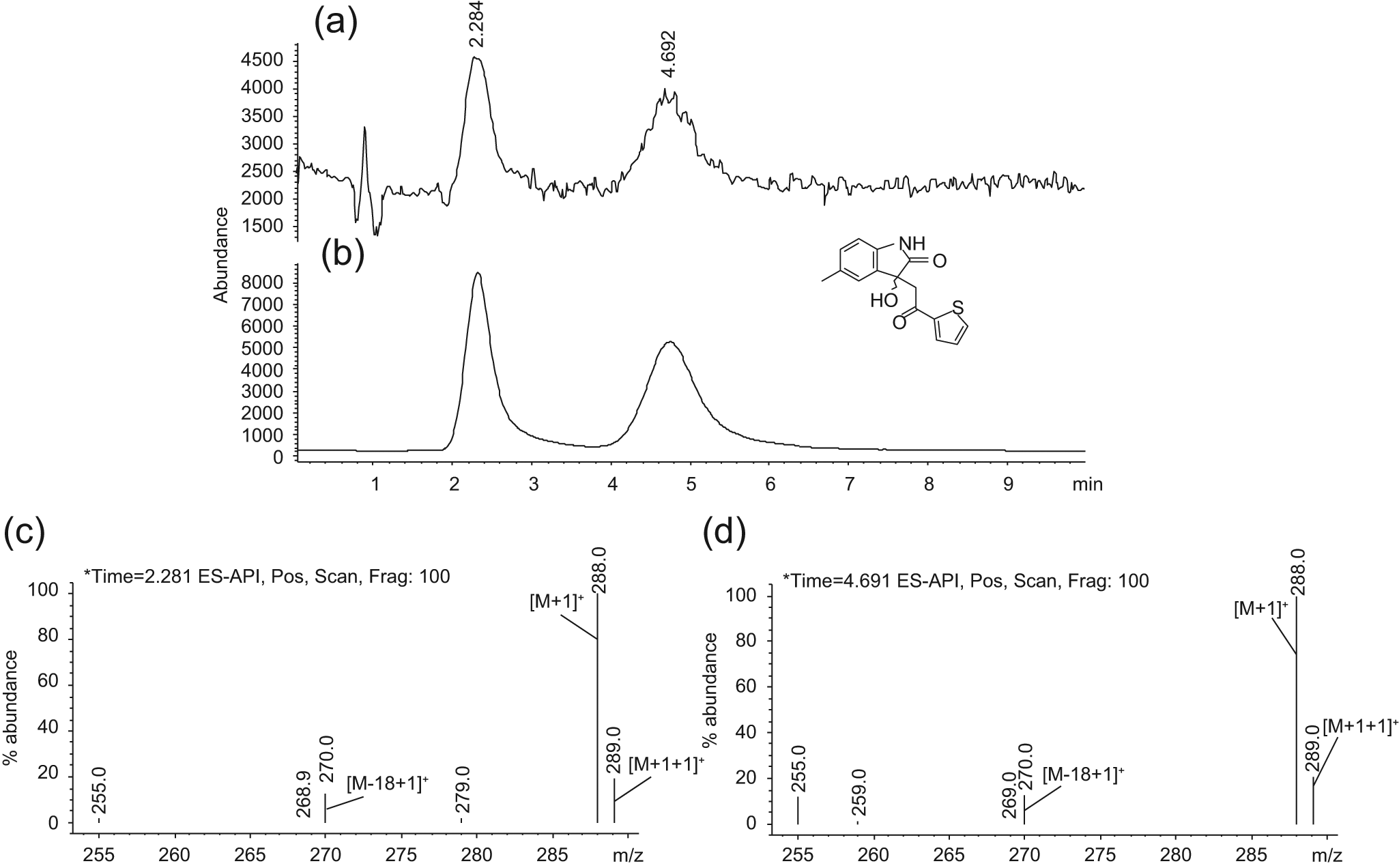
Chromatograms of ST058742 (0.1 mM) in scan mode (
Five fragments (17%) of the remaining 29 potential racemates were impossible to detect by MS (ST075161, ST060195, ST094899, ST024707, and ST084615). The most probable reason for these compounds to be “invisible” is that the ionization was suppressed by coelution with DMSO in the void or they were difficult to ionize in MS as observed in previous studies.6,13 The suppressing action of DMSO can be noticed in
Figure 1a
, where the peaks eluting closer to the void were smaller although the same sample was injected. The remaining 24 detectable racemates appeared as single peaks that had almost the same retention on the active thrombin and the PPACK-inhibited column. For this reason, the binding of these compounds was considered nonspecific (
WAC-MS in Separation of Diastereomers
As demonstrated above, WAC-MS is especially useful in fragment library screening as it is based on separation and therefore it can analyze mixtures or even crude mixtures. The development of fragment hits into more drug-like leads includes linking, merging, or growing the fragment in different ways. Frequently, these synthetic procedures introduce new chiral centers into the resulting compounds, and if not performed stereoselectively, diastereomeric mixtures are obtained. Such diastereomeric mixtures can be analyzed directly by WAC-MS without further purification to determine which ones might be of interest to purify and develop further. A collection of closely related pairs of diastereomers was analyzed by WAC-MS to demonstrate this kind of application.
Seven pairs of diastereomers were synthesized from a pure starting material that was reacted with a racemic amine. The (R)- and (S)-enantiomers of the amine might have differed in reactivity and the obtained amount of each diastereomer in a pair varied, but both diastereomers were achieved in all reactions. Three diastereomeric pure compounds (compounds 14, 22, and 23) were produced by the same methodology using nonracemic amines in the reactions (see Supplementary Information). Two of these, compounds 22 and 23, constituted a diastereomeric pair, and in total, eight diastereomeric pairs and one closely related compound were analyzed. IC50 values were determined for the compounds by an enzyme inhibition assay (see Supplementary Information for details), and all compounds except 14 and 15 were found to have affinity for thrombin (
Table 1
). A mixture of all compounds (
Table 1
; compounds 14–23) was prepared and analyzed by WAC-MS, and the obtained KD values were compared with IC50 values from the thrombin inhibition assay. Each pair of diastereomers (
Table 1
; compounds 15–23) and the single compound (
Table 1
; compound 14) had a unique mass and could be individually detected after separation on thrombin column 1. Some starting material of the synthesis was present in the samples, which eluted close to the void marker of DMSO (chromatograms not shown). With one exception (compound 15), all the diastereomeric pairs were separated into two peaks (
Fig. 2
) when analyzed on the active thrombin column. Compound 14, the single diastereomer, gave a single peak as expected. Compounds 14 and 15 were the least retained compounds in the collection and corresponded well to data from the enzyme assay, which reported no detectable affinity of these compounds (
Table 1
). All compounds analyzed on the PPACK-inhibited reference column produced single peaks that were significantly less retained compared with those on the thrombin column (see
The diastereomeric pair constituted by compounds 22 and 23 was also analyzed separately. The (R)-form (compound 22) was considerably more retained compared with the (S)-form (compound 23). The behavior was in accordance with enzyme inhibition data ( Table 1 ). The highest affinity was obtained for one of the diastereomers of compound 16, which had a retention time exceeding 600 min. The apparent KD value of this diastereomer was 1 µM, whereas the diastereomeric counterpart eluted close to the void marker ( Fig. 2 ) with a KD value of 0.34 mM.
Analyzing structures of pairs of diastereomers ( Table 1 ) showed that changes in the configuration of a stereoisomeric center could have a great impact on the binding into the S1′ pocket of thrombin. Diastereomers 22 and 23 are examples of this effect. The difference in the arrangement of the groups caused a change of two orders of magnitude in affinity measured by both WAC (from KD = 6 to 316 µM) and the enzyme assay (IC50 = 5 to 539 µM) ( Table 1 ). Similarly, the members of pair 16 had affinities that differed more than 300 times in KD. These dramatic changes emphasize the importance of stereoisomeric configuration when studying ligand-protein interaction even when the ligand is small in size.
As illustrated by compound 16, it can be difficult to identify a tight binder due to extremely long retention times. An elution procedure was therefore developed to elute such compounds without denaturing the immobilized protein. It was found that an injection of 0.2 µL of 10% acetic acid in water 3 min after the injection of the sample mixture onto thrombin column 2 eluted all compounds still remaining in the column, even the tightest binding diastereomer of compound 16. The procedure was evaluated with the reference compound 4-ABA that was eluted by injecting 10% acetic acid as described above. The thrombin column showed full recovery of retention after 30 min of equilibration with the mobile phase. An optimal concentration and injection volume of acetic acid was found to be critical to avoid loss of binding ability of thrombin. By using this elution procedure, we were able to confirm the absence of tight binders in the pair of compound 15. We believe that this elution procedure is useful in fragment screening by WAC-MS to confirm the presence or absence of tight binders.
In conclusion, WAC is a valuable approach for high-throughput fragment screening in early stages of drug development, as evidenced by its applicability on several targets such as kinases and proteases.6,14 The present work demonstrates another important feature of this technique: to assist in selection and screening of stereoisomeric structures. Furthermore, since WAC is based on separation according to affinities to target, the presence of impurities does not affect the affinity measurements of compounds of interest. In combination with MS as the detection unit, WAC is a powerful tool for analysis and screening of mixtures whether it is stereoisomeric or crude synthetic reactions. Consequently, WAC-MS allows compounds with different affinities to be separated and analyzed without the need of elaborative purification.
Footnotes
Acknowledgements
The authors thank Astra&Zeneca for support in synthesis of the diastereomers, Göran Karlsson and Stefan Winge from Octapharma for production of thrombin, and Agilent Technologies for providing access to the LC-MS platform. Linnaeus University is also acknowledged for giving financial support to the project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Linnaeus University is also acknowledged for giving financial support to the project.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.