Abstract
The automated electrophysiology platform IonWorks has facilitated the medium-throughput study of ion channel biology and pharmacology. Electrical and chemical access to the cell is by perforated patch, afforded by amphotericin. Permeation of the amphotericin pore is limited to monovalent cations. We describe here the use of the saponin escin as an alternative perforating agent. With respect to the number and robustness of seals formed across a variety of cell and ion channel types, the performance of escin is equal to that of amphotericin. Escin also permits the permeation of larger molecules through its pore. These include nucleotides, important intracellular modulators of ion channel activity that can be used to prevent ion channel rundown of, for instance, CaV1.2. Furthermore, pharmacologic agents such as QX314 can also permeate and be used for mechanistic studies. Escin, in combination with IonWorks, increases the scope of ion channel screening and can facilitate the assay of previously difficult-to-assay targets.
Introduction
Ion channels are a diverse family of membrane-spanning proteins that conduct ions across biological membranes. In a pharmaceutical setting, ion channels represent important drug targets. Ion channels constitute 13.4% of currently available drug targets, and worldwide sales of ion channel–targeted drugs are in excess of $12 billion. 1 The path to new medicines necessitates the screening of large numbers of compounds against ion channel targets, for the purpose of lead generation and for the assessment of, for instance, the cardiac safety liability of a compound.2,3
Traditionally, the study of ion channels has relied on conventional patch-clamp electrophysiology. This technique yields high-fidelity data but is labor intensive, requires a highly skilled experimenter, and may be used to screen only a small number of compounds. By this technique, a high (gigaohm) resistance seal is formed between the cell membrane and a small-diameter glass electrode. 4 Subsequently, the membrane at the orifice of the pipette is broken, affording access to the interior of the cell (whole-cell recording). By this method, the investigator has control over extracellular and intracellular buffers. This is important because ion channels may be modulated by both extracellular and intracellular factors. 5
To facilitate a more high-throughput approach to electrophysiology, planar patch-clamp technology has been developed.6,7 Planar patch-clamp describes a technique whereby a cell resides in a hole at the bottom of a well separating two aqueous compartments. In the automated planar patch-clamp platforms currently available, the well may be constructed from glass (e.g., QPatch, PatchXpress, Patchliner) or plastic (e.g., IonWorks Quattro). With respect to IonWorks, there is a further advancement that each well may have a single hole, known as HT mode, 8 or 64 holes, known as population patch clamp (PPC 9 ). With the cell residing in the Patchplate well from an IonWorks system, the external extracellular compartment is accessible to the electronic recording apparatus. The intracellular compartment contains the ground electrode. To complete the circuit, a compound is introduced in the intracellular compartment to perforate the cell membrane and permit recording. Recordings on IonWorks are therefore made by perforated patch, first described for conventional patch-clamp electrophysiology using the polyene antibiotic amphotericin B. 10 Nystatin is another alternative to amphotericin. 11 Both polyenes confer permeability only to monovalent cations and anions. Divalent cations and larger molecules do not permeate the pore.10,11 There is, therefore, a lack of disruption of the intracellular milieu with perforated patch recording that may be considered positive or negative. In the positive sense, intracellular modulators may be retained by the cell. In the negative sense, these same modulators may become exhausted and need to be replenished. There is also a technical limitation to their use as polyenes are poorly soluble in water and require a DMSO vehicle.
Escin is a major constituent of the saponins obtained from the horse chestnut tree (Aesculus hippocastanum). Initially, it was used experimentally to permeabilize smooth muscle cells. 12 Escin has been used as a perforating agent with conventional patch-clamp and is reported to form holes permeable to high molecular weight (MW) substances. 13
We describe here the optimization and use of escin as the perforating agent on the IonWorks Quattro automated patch-clamp platform. Compared to amphotericin, escin creates larger pores in the membrane, improving intracellular access to the cell by nucleotides and small molecules.
Materials and Methods
Cell culture medium and serum were purchased from Invitrogen (Carlsbad, CA). All antibiotics, escin, amphotericin, buffer components, and drugs were purchased from Sigma (St. Louis, MO). QX314 was purchased from Tocris (Bristol, UK).
Cell Lines
CHO cells stably expressing hKV1.5 (KCNA5) were maintained in Ham’s F12 medium supplemented with 10% fetal calf serum (FCS) and 800 µg/mL G418.
HEK cells stably expressing hNaV1.7 (SCN9A) were maintained with DMEM supplemented with 10% FCS and 1% NEAA and 400 µg/mL G418.
hKV1.5 and hNaV1.7 cell lines were available in the AstraZeneca Global Cell Bank facility.
CHO cells stably expressing the hCaV1.2 calcium channel (α1c, β2a, and α2δ1 subunits; ChanTest, St. Louis, MO) were maintained in Ham’s F12 medium supplemented with 10% FCS, 10 µg/mL Blasticidin, 250 µg/mL G418, 750 µg/mL Zeocin, and 250 µg/mL Hygromycin. Twenty-four hours prior to assay, CaV1.2 α1c expression was induced by addition of 1 µg/mL doxycycline.
IonWorks Quattro
Prior to assay, cells in culture were washed twice with phosphate-buffered saline and dissociated with 0.1 % Trypsin-EDTA (Gibco). For experiments in HT mode (hKV1.5 and hNaV1.7), cells were counted and resuspended to ~0.5 × 106 cells/mL in extracellular buffer (in mM: 145 NaCl, 4.5 KCl, 1.8 MgCl2, 1 CaCl2, 10 HEPES, pH 7.4). For experiments in PPC mode (hCaV1.2), cells were counted and resuspended to ~1.5 × 106 cells/mL in Ba2+-containing extracellular buffer (in mM: 135 NaCl, 10 BaCl2, 4.5 KCl, 1.8 MgCl2, 10 HEPES, pH 7.4).
IonWorks uses two different intracellular buffers sequentially. Stable seals are first established in intracellular buffer 1 (also called internal buffer), and then the perforation agent is introduced in intracellular buffer 2 (also called access buffer). Intracellular buffer 1 was prepared as follows (mM): 100 potassium gluconate, 40 KCl, 3.2 MgCl2, 3 EGTA, and 5 HEPES pH 7.25. Intracellular buffer 2 buffer contained 145 KCl, 1 MgCl2, 1 EGTA, and 20 HEPES pH 7.25. Amphotericin (Sigma A4888) was first dissolved in DMSO at a concentration of 30 mg/mL before being diluted in intracellular buffer 2 to a final concentration of 100 µg/mL. Escin (Sigma E1378) was dissolved in water to a concentration of 5 mM before diluting in access buffer to the required concentration. All experiments were performed on the IonWorks Quattro (Molecular Devices, Sunnyvale, CA). Unless otherwise stated, the settings chosen for making a seal and establishing access were “pause for 200s before seal test and then obtain access - pause for 120s, introduce access 70s, circulate fluid 240s and pause 220s.” Voltage protocols for each channel are described in the figure legends.
Results and Discussion
For conventional, perforated patch-clamp, escin has been used at concentrations of 20 to 50 µM.13,14 With an escin concentration of 50 µM as our starting point, we first determined the optimal concentration range for use on IonWorks in combination with CHO-hKv1.5 cells. A number of parameters were considered; mean seal resistance, the number of successful seals formed, seal stability, and the mean Kv1.5 current. The data are compiled in Figure 1 .
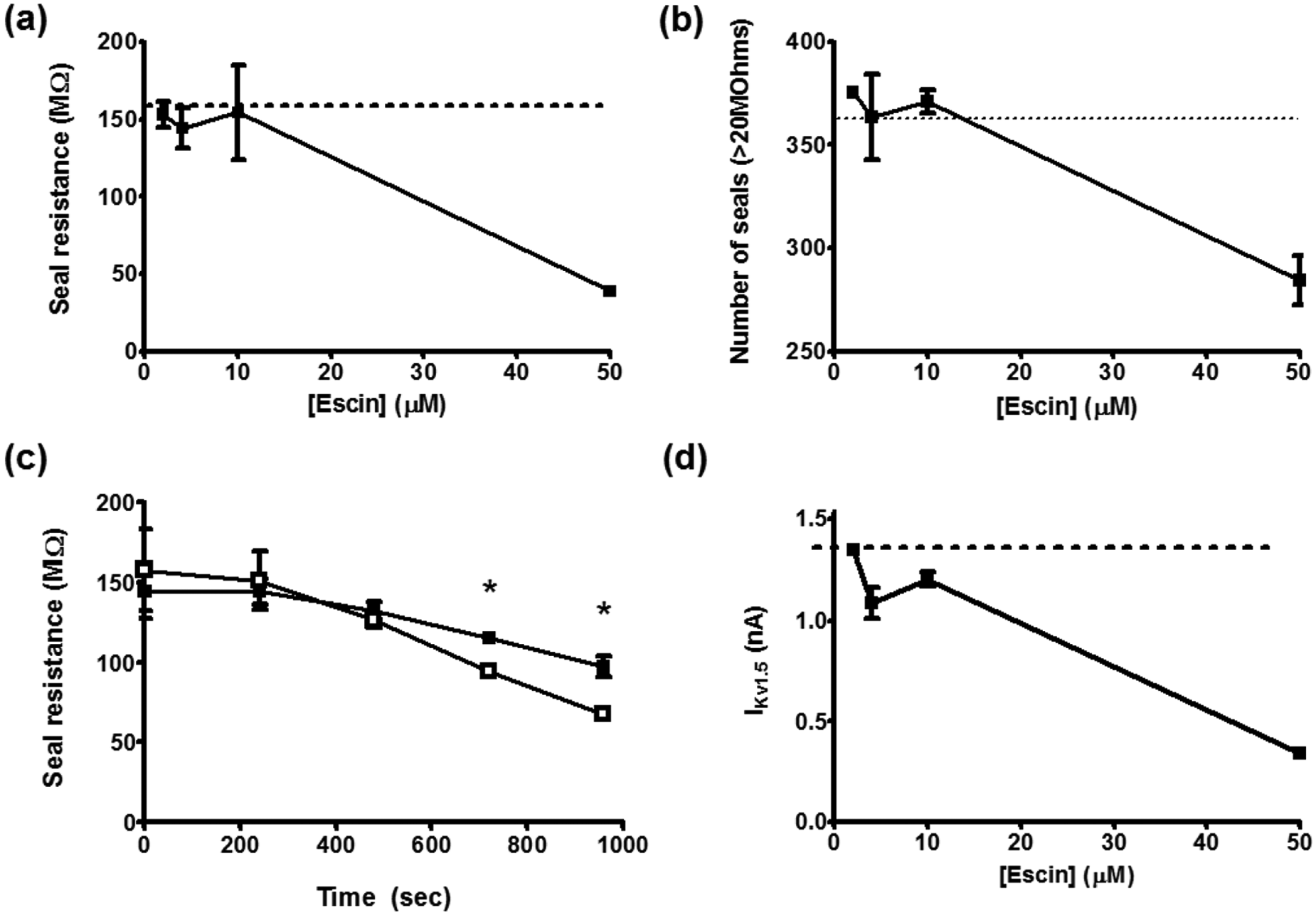
Seal resistance parameters were measured in HT mode between CHO-Kv1.5 cells and the IonWorks Patchplate in access buffer containing different concentrations of escin. The dotted line indicates those measured with amphotericin as perforating agent. All data are the mean ± standard deviation of two runs performed on different IonWorks machines. (
The mean seal resistance achieved with amphotericin was 157 ± 25 MΩ (all data are the mean ± standard deviation [SD] of two runs performed on different IonWorks). Mean seal resistances of 153 ± 11 MΩ were achieved at an escin concentration of 2 µM, equivalent to those seen with amphotericin ( Fig. 1a ). At this concentration of escin, 375 successful seals were formed (out of 384). With amphotericin in the access buffer, the number was 361 ( Fig. 1b ). Seal stability was recorded over a period of 16 min. Over this time period, the seal was more stable when escin was used as perforating agent than amphotericin ( Fig. 1c ). At the final two time points, there was a statistically significant difference between seal resistances with escin as perforating agent versus amphotericin (p < 0.05).
Mean Kv1.5 currents recorded with amphotericin were 1.39 ± 0.32 nA. Maximal currents recorded with escin were 1.34 ± 0.04, equivalent to those seen with amphotericin. Four micromolar escin was chosen as the optimal concentration for further experiments.
We next addressed the question as to whether escin would confer intracellular access to larger molecules. The nucleotides ATP and GTP are commonly added to intracellular (pipette) buffers in conventional patch-clamp to activate channels and/or prevent rundown. 15 To determine whether ATP and GTP can permeate the escin pore, we measured currents carried by the cardiac L-type calcium channel, hCaV1.2, before and after the addition of vehicle, DMSO, to a final concentration of 0.33% ( Fig. 2 ). Calcium currents with access buffer containing 4 µM escin, 2 mM ATP, and 300 µM GTP were 450 ± 18 pA. With amphotericin, they were significantly lower, 239 ± 9 pA (p < 0.05). The same currents after the addition of DMSO with escin as perforating agent were 434 ± 19 pA, a decrease in current (i.e., rundown) of <4%. By contrast, currents with amphotericin were 130 ± 5 pA, a significant rundown of ~46% (p < 0.05).
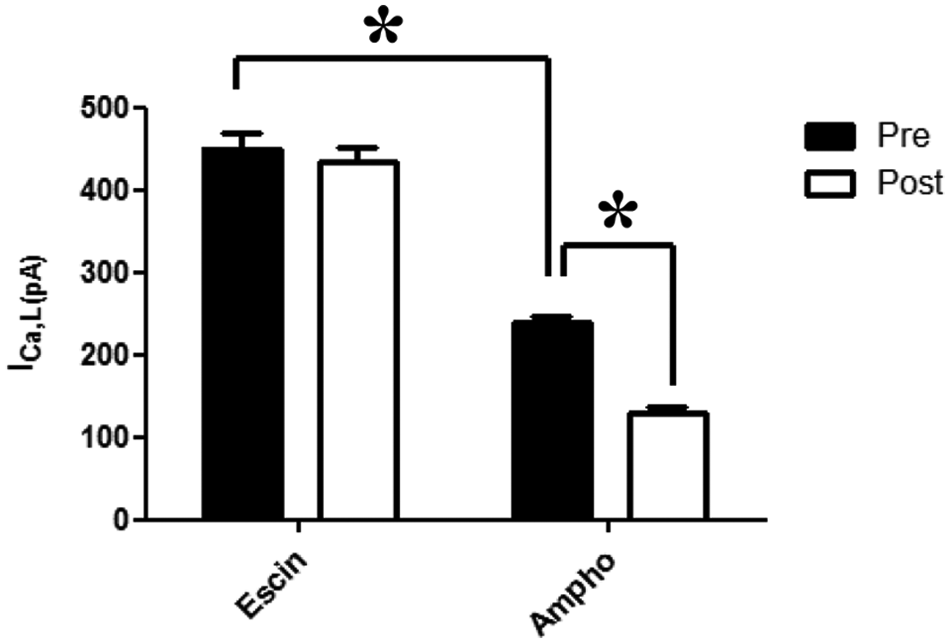
hCaV1.2 currents were compared between recordings using escin or amphotericin as perforating agent before (solid bars) and after the addition of DMSO to a final concentration of 0.33% (clear bars). Currents were evoked by depolarizing from a holding potential of −60 mV to 0 mV for 500 ms. All data are mean ± SD from two runs performed on different IonWorks machines. Statistical significance (p < 0.05) is denoted by an asterisk.
Potencies of four L-type calcium channel blockers were tested using escin as a perforating agent ( Fig. 3 ). Mean values are given with 95% confidence interval in brackets. Verapamil 3.3 µM (2.2–4.9 µM), isradipine 10.9 nM (7.9–14.9 nM), nifedipine 60.7 nM (46.1–80.0 nM), and bepridil 3.16 µM (2.3–4.4 µM). These values are in good agreement with the literature values from Balasubramanian et al. 16 : verapamil 15 µM, isradipine 8 nM, nifedipine 16 nM, and bepridil 4.4 µM.
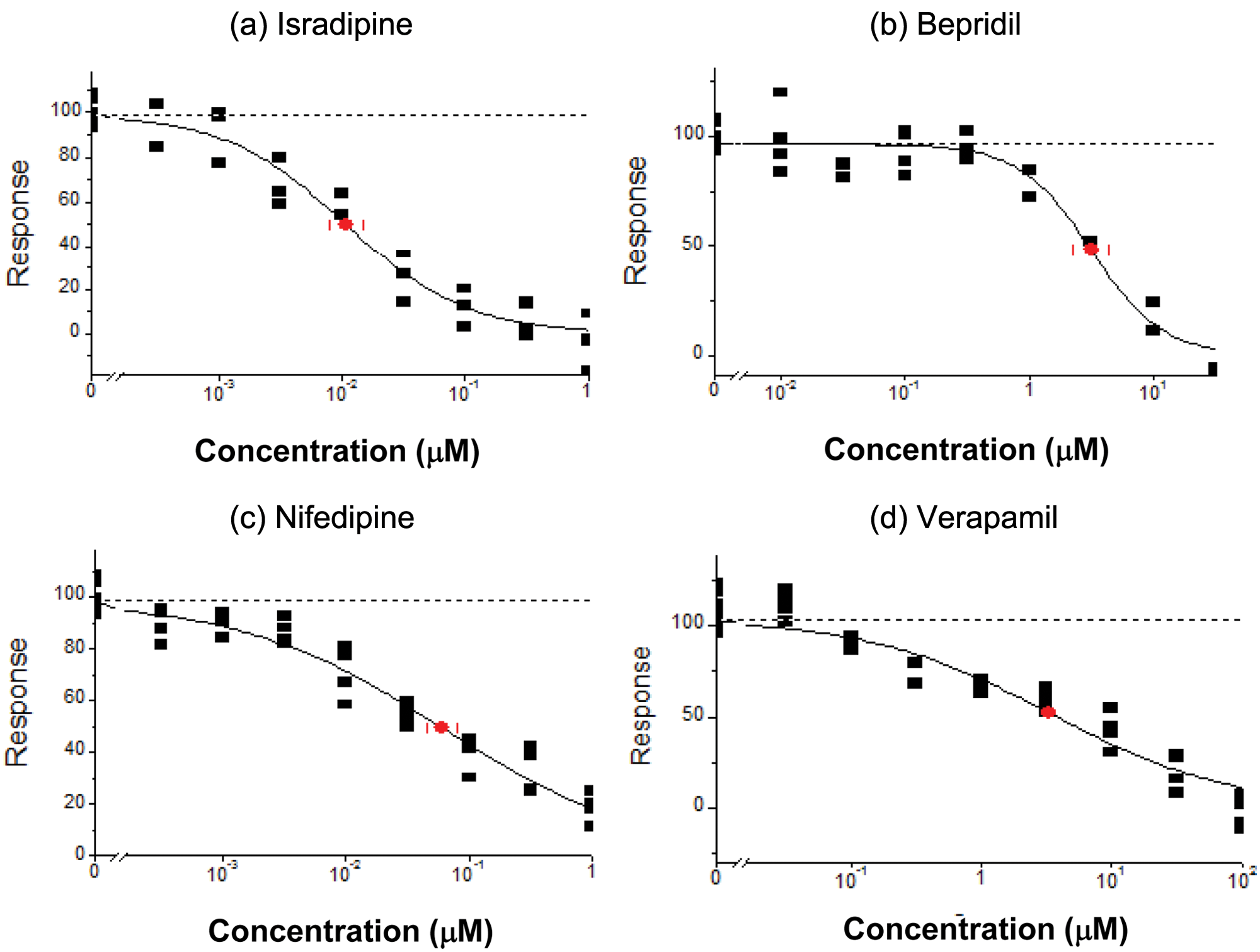
Pharmacology of four common L-type calcium channel blockers. IC50s were generated for isradipine, bepridil, nifedipine, and verapamil from eight-point concentration-response curves (Microsoft Origin).
We next investigated whether drugs with a low MW could also permeate the escin pore. N-ethyl lidocaine (QX314) is a membrane-impermeant lidocaine derivative that blocks NaV1.7 sodium channels from the intracellular side only. 17 We added 5 mM QX314 to the access buffer and compared its effect on NaV1.7 currents with amphotericin or escin as a perforating agent. In combination with escin, QX314 caused a statistically significant inhibition of 97% ± 3% compared with control experiments in the absence of QX314 (data are mean ± SD from two runs on different IonWorks machines, Student’s t-test p < 0.01; Fig. 4 ). No statistically significant effect of QX314 was observed when amphotericin was used as perforating agent (p > 0.05). Furthermore, no statistically significant effect of QX314 was observed when added extracellularly (p > 0.05).
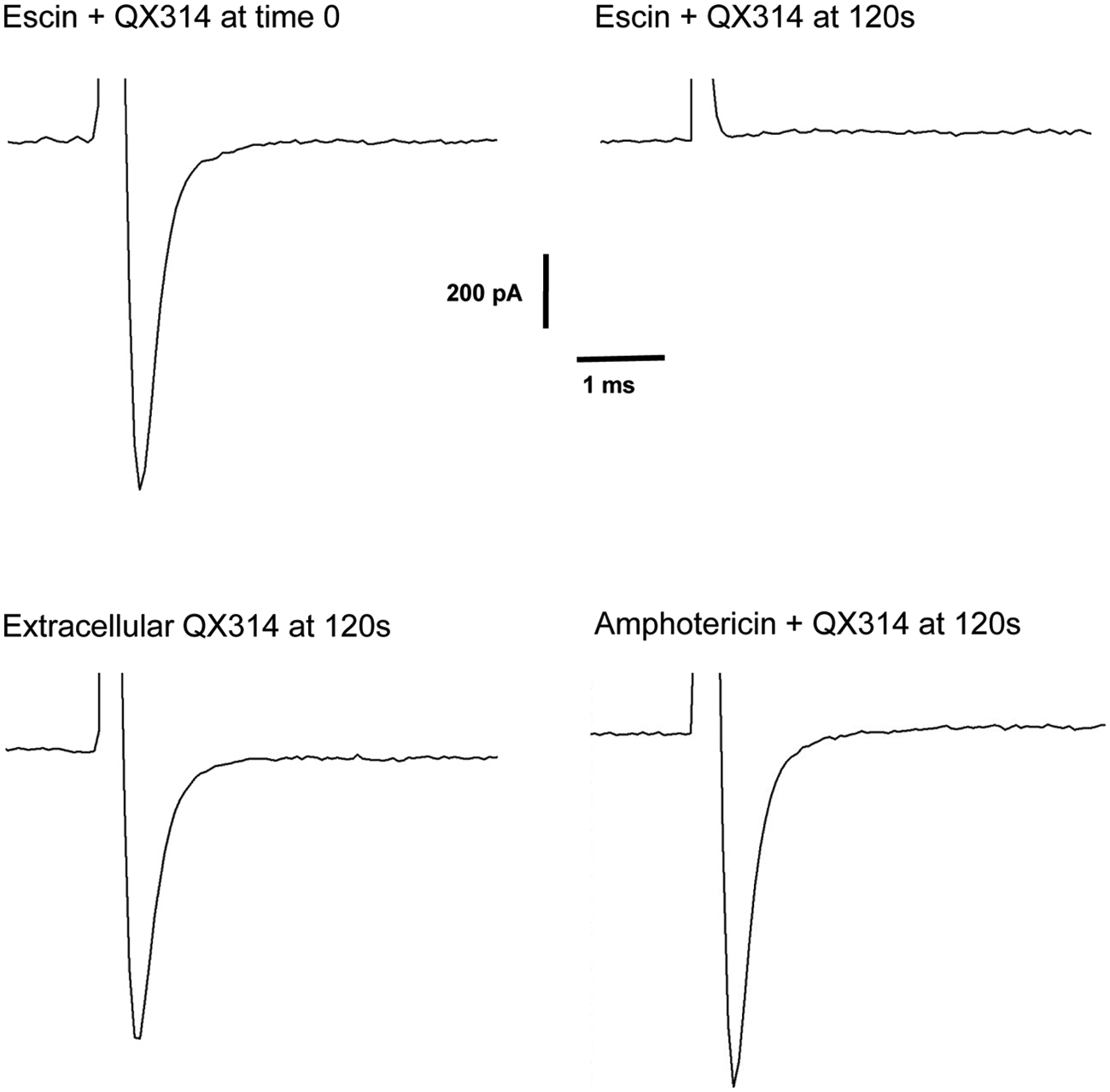
hNaV1.7 currents were recorded comparing the effects of 5 mM intracellular QX314 with escin or amphotericin as perforating agent or the effect of QX314 added extracellularly. Sodium currents were evoked by depolarizing from a holding potential of −90 mV to 0 mV for 50 ms.
In this article, we report that escin can be used as a perforating agent on the IonWorks automated patch-clamp platform. In many respects, performance of escin is equal to that of amphotericin. However, given the permeability of escin pores to large molecules such as ATP and GTP, there are important advantages to using escin over amphotericin.
The optimal concentration of escin was in the range of 2 to 10 µM. At this concentration, the seals formed between Patchplate and cell, with respect to seal resistance, were equivalent to those observed when using amphotericin. With respect to seal stability, the seals are more stable when using escin as a perforating agent compared with amphotericin. Like amphotericin, we demonstrate here that escin works with commonly used cell lines such as CHO and HEK. We have successfully used escin with a variety of ion channel targets, namely, K+, Na+, and Ca2+ channels. Furthermore, escin works in both HT and PPC modes on IonWorks. Taken together, it is clear that escin affords as much flexibility and as widespread application as amphotericin. However, escin poses a number of advantages over amphotericin. The first is its solubility in water. Polyene antibiotics such as amphotericin and nystatin are poorly soluble in water. The end result is access buffer with a heavy precipitate that may result in uneven mixing and block tubing, with long-term maintenance implications. Escin is water soluble, and no such problems arise. Escin is also cheaper. At the required concentrations, escin is approximately 1% of the cost of amphotericin.
Amphotericin is permeable only to monovalent ions. This does not permit close control or buffering of intracellular free calcium, an important intracellular regulator of some ion channels, and is therefore a hindrance to the pharmacologic screening of such channels. 18 We did not address Ca2+ permeability here, but it is clear from early work with permeabilized skeletal muscle that escin in permeable to Ca2+. 19 Also previously addressed in the literature is the permeability of escin to larger MW molecules (e.g., ATP) at concentrations of escin as low as 5 µM. 20 Furthermore, escin has previously been shown to be permeable to heparin (MW ~500012) and Fluo3 (MW 85513). The permeability to these species appears time and concentration dependent. Even molecules as large as 10 kDa start to permeate after approximately 20 min. 13 We have demonstrated here, in the context of an automated electrophysiology platform, that the escin pore is permeable to nucleotides and drugs such as QX314, with a MW of ~620 and ~343, respectively. A number of ion channels exhibit run down during the course of an experiment, most notably the L-type calcium channel hCaV1.2. 16 This is a problem not unique to automated patch-clamp but also arises in conventional patch-clamp. With conventional patch-clamp, however, the experimenter may add ATP to the patch pipette solution. We report here that it is possible, with escin as a perforating agent, to add nucleotides directly to the IonWorks access buffer. In the case of CaV1.2, we show that the inclusion of escin in the access buffer increased the size of the currents and largely prevented channel run down. Small currents and channel run down were cited as limiting the achievable throughput of the CaV1.2 assay on the PatchXpress platform 16 to two to four compounds per day per FTE. The combination reported here of IonWorks platform and escin can achieve a throughput of >40 compounds per day per FTE (eight-point dose-response curve).
Escin also introduces the possibility of studying the actions of membrane-impermeant ion channel modulators. The example cited here is the membrane-impermeant lidocaine derivative QX314. The escin pore was clearly permeable to QX314, which blocked NaV1.7 from the intracellular side only. There are many examples in which such intracellular access to membrane-impermeant compounds might be useful (e.g., when using thiol-modifying reagents such as MTSET to study ion channel topology 21 or to study the sidedness of a compound’s action, e.g., ethylbromide tamoxifen on BKCa 22 ).
To conclude, this article demonstrates, for the first time, the use of escin as an alternative perforating agent on the automated electrophysiology platform IonWorks. Escin confers the ability to add important ion channel modulators to the intracellular buffer. The use of escin can facilitate the screening of previously difficult targets, such as hCaV1.2, or be useful for mechanistic studies.
Footnotes
Acknowledgements
The authors thank Dr Helen Garside for useful discussions during the preparation of this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
