Abstract
Neurons derived from human-induced pluripotent stem cells were characterized using manual and automated patch-clamp recordings. These cells expressed voltage-gated Na+ (Nav), Ca2+ (Cav), and K+ (Kv) channels as expected from excitable cells. The Nav current was TTX sensitive, IC50 = 12 ± 6 nM (n = 5). About 50% of the Cav current was blocked by 10 µM of the L-type channel blocker nifedipine. Two populations of the Kv channel were present in different proportions: an inactivating (A-type) and a noninactivating type. The A-type current was sensitive to 4-AP and TEA (IC50 = 163 ± 93 µM; n = 3). Application of γ-aminobutyric acid (GABA) activated a current sensitive to the GABAA receptor antagonist bicuculline, IC50 = 632 ± 149 nM (n = 5). In both devices, comparable action potentials were generated in the current clamp. With unbiased, automated patch clamp, about 40% of the cells expressed Nav currents, whereas visual guidance in manual patch clamp provided almost a 100% success rate of patching “excitable cells.” These results show high potential for pluripotent stem cell–derived neurons as a useful model for drug discovery, in combination with automated patch-clamp recordings for high-throughput and high-quality drug assessments at human neuronal ion channels in their correct cellular background.
Introduction
Stem cell–derived neurons provide a novel and unique model for studying human drug targets in their physiologically relevant environment of terminally differentiated, postmitotic cells. It is becoming increasingly recognized that associated proteins modulate the physiology and pharmacology of neuronal proteins.1,2 Therefore, assays that investigate neuronal toxicity, drug effects, or basic cellular functions of neurons can mostly benefit from the development of human neurons derived from induced pluripotent stem cells (hIPS-neurons). Those assays need a thorough validation and comparison with previously published data sets to understand similarities and differences of hIPS-neurons with primary human cells, rodent primary neurons, or embryonic stem cells.
Several other biomedical studies rely on hIPS-neurons, such as recently published investigations of hIPS-neurons derived from patients with neurodevelopmental or neurodegenerative diseases.3,4 Many of the observed physiological differences were based on cellular excitability or other ion channel–based functions. Those studies can gain further credibility by a broad characterization of ion channels expressed in hIPS cells showing that their physiology, pharmacology, and function match those properties of ion channels expressed in human neurons in situ.
The aim of the present study was to perform a basic characterization of major ion channels expressed in hIPS-neurons. Neurons derived from human-induced pluripotent stem cells obtained from Cellular Dynamics International (CDI, Madison, WI), subsequently named iCell Neurons, were investigated in this study by manual and automated patch clamp. For chip-based, automated patch clamp, we used the Patchliner (Nanion Technologies, Munich, Germany), which allows recording from up to eight cells simultaneously. Until now, the iCell Neurons have been only partially characterized using conventional patch clamp (see http://www.cellulardynamics.com/products/neurons.html). In this study, we investigated the major voltage-dependent ion channels and a ligand-gated ion channel in these cells using manual and automated patch-clamp recordings. Stem cell–derived cardiomyocytes have already been successfully used on the planar patch-clamp workstations in which cells are maintained in suspension before recording.5,6 In contrast to cardiomyocytes, neurons develop highly arborized dendrites and axons during their maturation, and many of their ion channels are expressed in specific subcellular compartments. Therefore, a careful comparison of manual and automated patch-clamp data was an important prerequisite for the validation of this approach. Based on our experiments, hIPS-neurons present an excellent novel tool for drug discovery, and automated patch clamp is a well-suited method for their characterization at higher throughput while still maintaining the high quality of conventional patch clamp.
Materials and Methods
Cell culture and harvesting
Frozen stocks of iCell Neurons were obtained from CDI containing at least 2.5 million plateable cells per unit (catalogue number: NRC-100-010-001). Cells were thawed as per the manufacturer’s instructions at a density of 70 000 cells per well in a 24-well plate (manual patch clamp) or 50 000 cells per cm2 (automated patch clamp). Two different coating strategies were investigated, a poly-D-lysine coating or a poly-L-ornithine/laminin coating. In brief, for manual patch clamp, 24-well plates (Nunc, Penfield, NY) were coated with 50 µg/mL poly-D-lysine (Sigma-Aldrich, St. Louis, MO) for 1 h at 37 °C, aspirated after incubation, and rinsed twice with sterile distilled water. For automated patch clamp, four 12.5-cm2 culture flasks (BD Falcon, Franklin Lakes, NJ) were coated with 0.01% poly-L-ornithine (Sigma-Aldrich) for 1 h at room temperature; this was then removed, and the dishes were washed twice with sterile distilled water and coated with a 3.3-µg/mL solution of laminin (Sigma-Aldrich). The dishes were incubated in a 37 °C incubator for at least 1 h. The laminin was aspirated shortly before addition of the cell suspension. A vial containing 2.5 million plateable iCell Neurons was placed in a 37 °C water bath for exactly 3 min. The cells were transferred to a 50-mL Falcon tube, and 1 mL Complete Maintenance Media (containing iCell Neurons medium supplement and 1% penicillin/streptomycin; CDI) was added slowly (dropwise) to the cells. Cells were swirled gently. A further 8 mL Complete Maintenance Media (CDI) was added slowly to the centrifuge tube. The tube was swirled gently. Approximately 2.5 mL cell suspension was added to each of the four coated T12.5 culture flasks. The cells were kept in a 37 °C incubator (95% O2/5% CO2). After 24 h, media were exchanged for fresh Complete Maintenance Media (100% exchange). Cells could then be kept in a 37 °C incubator (95% O2/5% CO2) for up to 4 weeks. A 50% to 75% media exchange was made every 2 to 3 days.
Cells were cultured for at least 4 days and were typically used on the automated patch-clamp platform 4 to 14 days after plating. To harvest the cells, 1 × T12.5-cm2 culture flask was used at a time. Media were removed and the iCell Neurons were washed with 2 mL phosphate-buffered saline (PBS) containing Ca2+/Mg2+ and incubated in this buffer at room temperature (RT) for 10 min. After this time, the PBS was removed, and prewarmed (37 °C) 0.25% trypsin/EDTA (Invitrogen, Carlsbad, CA) was added. The cells were incubated in trypsin for 3 to 5 min at 37 °C. The majority of the trypsin was then removed, leaving a thin film over the bottom of the dish. The dish was tapped against the hand several times to dislodge the cells from the bottom of the dish. Then, 0.5 mL external recording solution (see below) and 0.5 mL Complete Maintenance Media were added and pipetted up and down a maximum of five times to ensure most of the cells were removed from the bottom of the dish. The cells were then placed in the cell hotel of the Patchliner, where they were periodically pipetted up and down to ensure viability and minimize clustering.
Patch-clamp solutions
For manual patch clamp of Nav, Kv, and γ-aminobutyric acid (GABA) currents, internal solution was composed of 10 mM NaCl, 125 mM KCl, 1 mM MgCl2, 5 mM EGTA, 10 mM HEPES, 0.1 mM GTP-Na3, and 5 mM ATP-Mg2, pH 7.2 (KOH). External recording solution contained 150 mM NaCl, 4 mM KCl, 1 mM MgCl2, 1.2 mM CaCl2, and 10 mM HEPES, pH 7.4 (NaOH). For recording of Cav currents, the internal solution contained 107 mM CsOH, 107 mM L-glutamate, 20 mM CsCl, 1 mM TBA-Cl, 11 mM BAPTA, 0.9 mM CaCl2, 1 mM MgCl2, 20 mM HEPES, 0.1 mM GTP-Na3, and 5 mM ATP-Mg2, pH 7.2 (CsOH). External recording solution contained 33.5 mM TEA-Cl, 5 mM BaCl2, 0.1 mM MgCl2, and 2.5 mM HEPES, pH 7.4 (TEA-OH). For automated patch clamp of Nav, Kv, and GABA currents, internal solution contained 50 mM KCl, 10 mM NaCl, 60 mM KF, 20 mM EGTA, and 10 mM HEPES/KOH, pH 7.2. External recording solution contained 140 mM NaCl, 4 mM KCl, 1 mM MgCl2, 2 mM CaCl2, 5 mM D-glucose monohydrate, and 10 mM HEPES/NaOH, pH 7.4. GABA (Sigma-Aldrich) was made as a 10-mM stock solution in external recording solution and diluted to the appropriate concentration in external recording solution. Bicuculline (Sigma-Aldrich) was made as a 1-mM stock in external solution and diluted to the appropriate concentration in external recording solution. Bicuculline was preapplied alone for at least 30 s before coapplication with 30 µM GABA. TTX (Tocris, Bristol, UK) was made as a 1-mM stock in external solution and diluted to the appropriate concentration in external recording solution. TEA was made as a 1-M stock in external solution and diluted to the appropriate concentration in external recording solution. 4-AP was made as a 10-mM stock solution in distilled water and diluted to the appropriate concentration in external solution. 4-AP (stock solution at 10 mM in distilled water) and nifedipine (stock solution 10 mM in 100% DMSO) were applied during manual patch clamp using a multi-to-one barrel pipette during continuous recording of voltage step responses.
Automated patch-clamp electrophysiology
Whole-cell patch-clamp recordings were conducted according to Nanion’s standard procedure for the Patchliner. 7 Equipment: The NPC-16 Patchliner Octo (Nanion Technologies) and PatchControlHT software (Nanion Technologies) were used for cell capture, seal formation, whole-cell access, and programming of the experiment. Using the Patchliner Octo, up to eight cells can be recorded simultaneously. Two EPC-10 quadro (HEKA Elektronik, Lambrecht/Pfalz, Germany) patch-clamp amplifiers with Patchmaster (HEKA Elektronik) software were used for data acquisition and basic analysis. Data were exported using functions in the PatchControlHT software and loaded into Igor (IGOR-Pro; WaveMetrics, Inc., Lake Oswego, OR) for automated analysis of concentration-response curves. Boroscillicate glass NPC-16 chips (single-use, disposable; Nanion Technologies) were used for automated patch-clamp recordings.
Manual patch-clamp electrophysiology
Whole-cell patch-clamp recordings were performed on iCell Neurons cultured on poly-D-lysine–coated glass coverslips using thick-wall, borosilicate glass patch pipettes with a tip resistance of 1.8 to 2.3 MΩ. Current or voltage amplification, data acquisition, and processing were as for automated recordings using an EPC-10 amplifier, PatchMaster, and Igor software.
Results and Discussion
The neurons derived from human-induced pluripotent stem cells could be maintained in culture for up to 6 weeks. After 24 h (days in vitro 1, DIV1), cells adhered and extended processes, creating a network of processes within days that usually resulted in spontaneously firing neuronal networks after DIV7. Figure 1A shows the cells at DIV7 before harvesting for use on the Patchliner. Figure 1B shows the cells after harvesting using the protocol described above. The cells still have clear processes attached; they do not have a smooth, round membrane that, for example, a HEK293 cell would have when in suspension. We suspect that these processes affect the success rate for capture on a planar patch-clamp chip. Although with the manual patch clamp, we obtained a Giga-seal success rate of about 80% (after a usual period of parameter optimization) and a stable whole-cell configuration (no change of access resistance by more than 20% in at least 3 min) in about 60% of all cells, the automated patch-clamp success rate was lower, with 40% successful seals. Figure 1C shows a bar graph for the success rate for sealing of iCell Neurons using manual patch clamp or on the Patchliner. We tested the relevance of different parameters for the seal success rate. Different harvesting techniques, particularly the use of different lifting enzymes, including TrypLE Select (1×; Invitrogen) or 0.05% trypsin (1×; PAA, Pasching, Austria), appeared to have little effect on the success rate. The cells adhered very tightly to the bottom of the dish, and only a relatively long (3–5 min) incubation with the enzyme and then tapping of the dish would dislodge the cells. On the other hand, the type of chips did affect success rate. The success rate was best when using NPC-16 chips with a resistance of 5 to 6 MΩ (Nanion Technologies). Typically, cell lines such as HEK293 cells work best with NPC-16 chips with a resistance of 2 to 3.5 MΩ. This indicates that the iCell Neurons are somewhat smaller than HEK293 cells, confirmed by the small cell capacitance measured in the whole-cell configuration (3.2 ± 0.2 pF, n = 198; see Table 1 ). In comparison, HEK293 cells typically exhibit a cell capacitance of around 8 to 18 pF, 6 and success rates of around 80% are achieved on the Patchliner. 6 However, since iCell Neurons are primary-like cells, we consider this success rate to be reasonable. The seal resistance was >1 GΩ for the 198 cells recorded ( Table 1 ). Table 1 also shows a summary of cell parameters, including average cell capacitance (Cm) and series resistance (Rs).
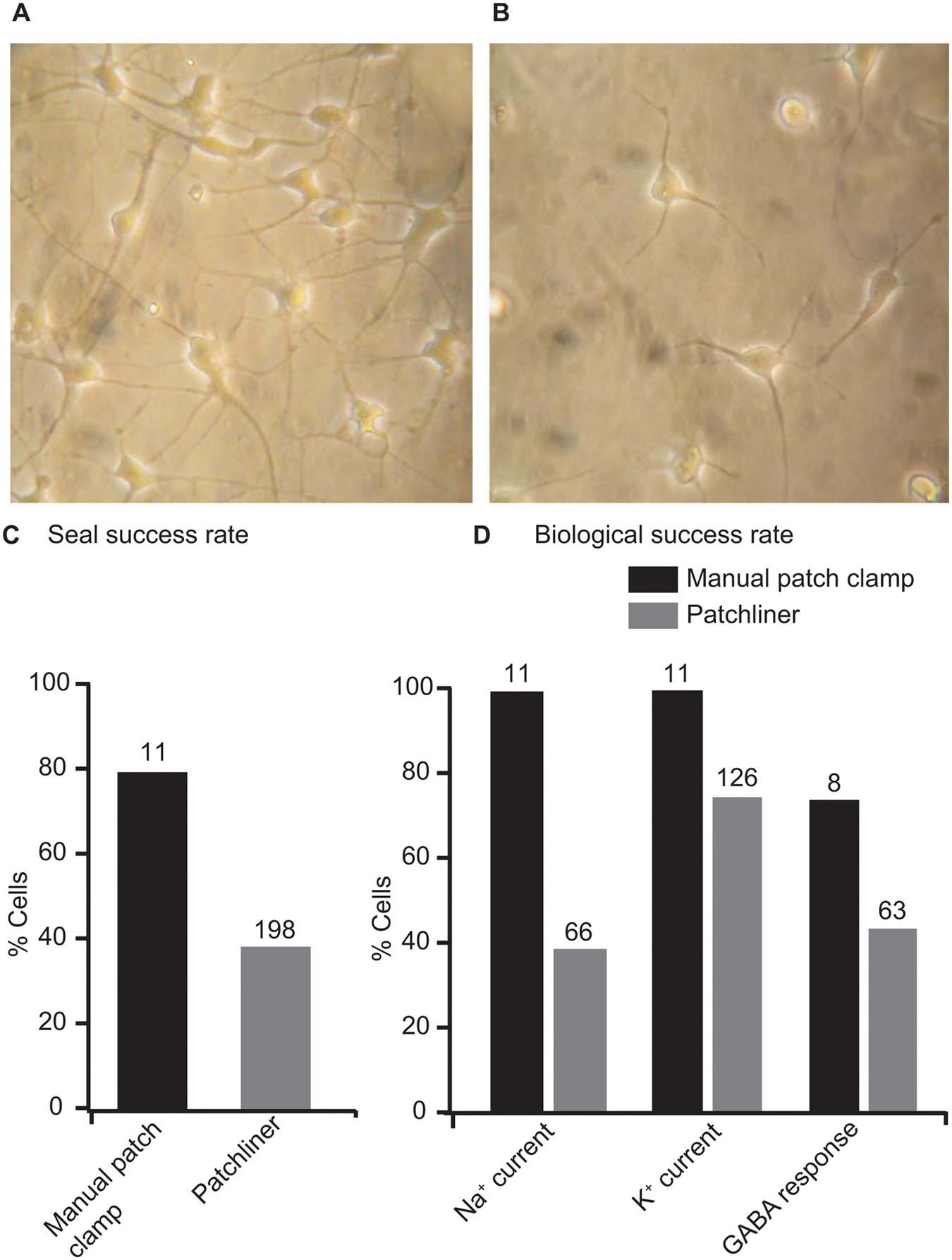
iCell Neurons as used for manual patch clamp and the Patchliner. Panel
Comparison of Cell Parameters and Current Amplitudes for iCell Neurons Recorded at Stages between DIV4 and DIV15 on the Patchliner (Upper Row) and in Manual Patch-Clamp Configuration (Lower Row)

Data are shown as mean ± SEM (number of cells) or value ranges. Values are the maximal inward currents for Na peaks, at Vhold = +60 mV for K currents and at Vhold = −70 mV for GABA currents.
After reaching the whole-cell configuration, voltage step protocols were applied to each cell in manual and automated patch clamp to investigate the presence of Nav, Cav, and Kv channels in each cell. To some cells, GABA (30−100 µM) was applied to the cell for 0.6 to 1.0 s to find out if GABA receptors are expressed in these cells.
Although Kv currents could be recorded in almost 100% of the cells using both methods, there was a clear difference in the presence of inward Nav currents between automated and manual patch-clamp experiments: On the Patchliner, only approximately 40% of cells showed a significant Nav current ( Fig. 1D ). After a day of training, visually guided manual patch clamp reached an almost 100% success rate in patching cells that show large Nav currents ( Fig. 1D ). Comparing those cells that showed Nav currents, we observed a similar voltage to elicit half-maximal peak amplitudes, which was in the range of −30 and −10 mV. However, the maximum current amplitudes were quite different, ranging from −20 pA to >−1 nA with an average of −225 ± 32 pA (n = 66) using the Patchliner and from −1500 to −6700 pA in manual patch clamp with an average of −2800 pA ( Table 1 ). Figure 2A , D shows for comparison current responses of example cells expressing a Nav current in manual ( Fig. 2A ) and automated ( Fig. 2D ) patch clamp. In the example shown, the Nav current on the Patchliner is very large. The current-voltage (I-V) relationships are well comparable between both recording techniques ( Fig. 2B , C for manual patch clamp and Fig. 2E , F for Patchliner). Note that these I-V relationships show maximal inward currents at a very early phase after depolarization, which could already contain (under the ionic conditions used) some Kv currents at stronger depolarizations. These outward Kv currents can be already seen at positive potentials in the raw data traces. A Kv current was by far the most prevalent current that we recorded in these cells ( Fig. 1D ). Interestingly, in both settings, there appeared to be two different populations of Kv channel expressed in these cells. Some cells expressed “only” a noninactivating type Kv current, as shown in Figure 2Diii , and some cells expressed a clearly inactivating type Kv current, as shown in Figure 2Dii . In most cells, however, it was difficult to clearly categorize the currents, and these most probably expressed a mixed population of Kv currents. The average current-voltage plot for 121 cells as measured using the Patchliner is shown in Figure 2F . The maximum current amplitude at 40 mV ranged from 40 pA to almost 2 nA with an average of 368 ± 25 pA (n = 126). The I-V of Kv currents is well comparable with those measured in manual patch clamp, shown in Figure 2C . As observed with Nav currents, Kv currents in manually patched cells were about 10-fold larger, with an average steady-state level 50 ms after depolarization of 3070 pA (range, 1020–4550 pA, n = 11).
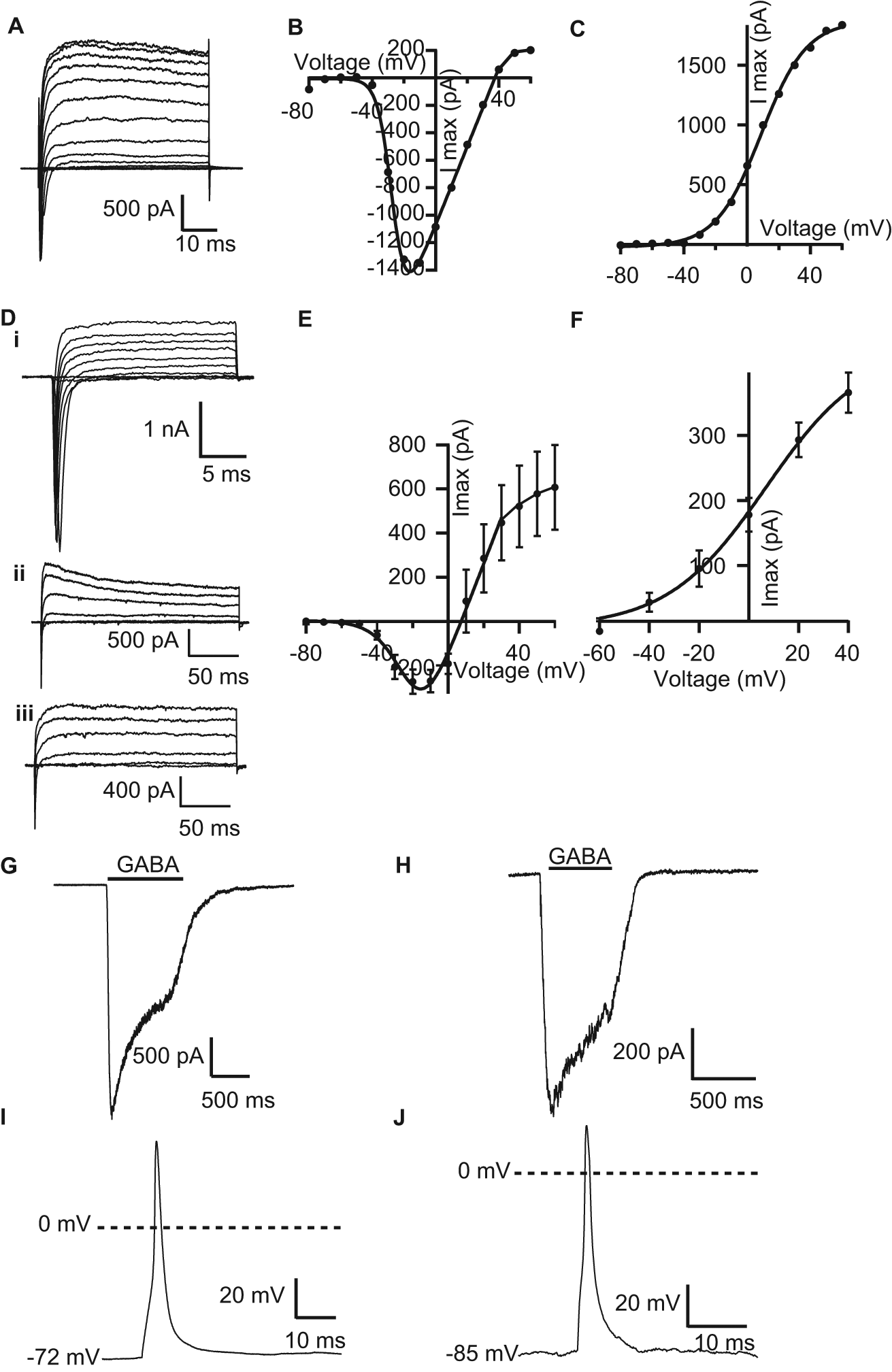
Comparison of manual and automated patch-clamp recordings from iCell Neurons. Panel
In manual patch-clamp recordings, we also investigated the presence of voltage-gated calcium channels. Large Cav currents could be recorded in a BaCl2-containing extracellular solution ( Figure 3H ). These currents were fast activating, starting at depolarizing steps of −20 mV, and inactivated rapidly when stepping back to −60 mV. These properties are indicative for high-voltage activated Cav channels such as N-, P/Q-, and L-type channels. Application of 10 µM nifedipine, a selective L-type channel blocker, inhibited Cav currents by about 50% ( Fig. 3H ), indicating that the observed current is likely a mixture of different types of Cav channels.
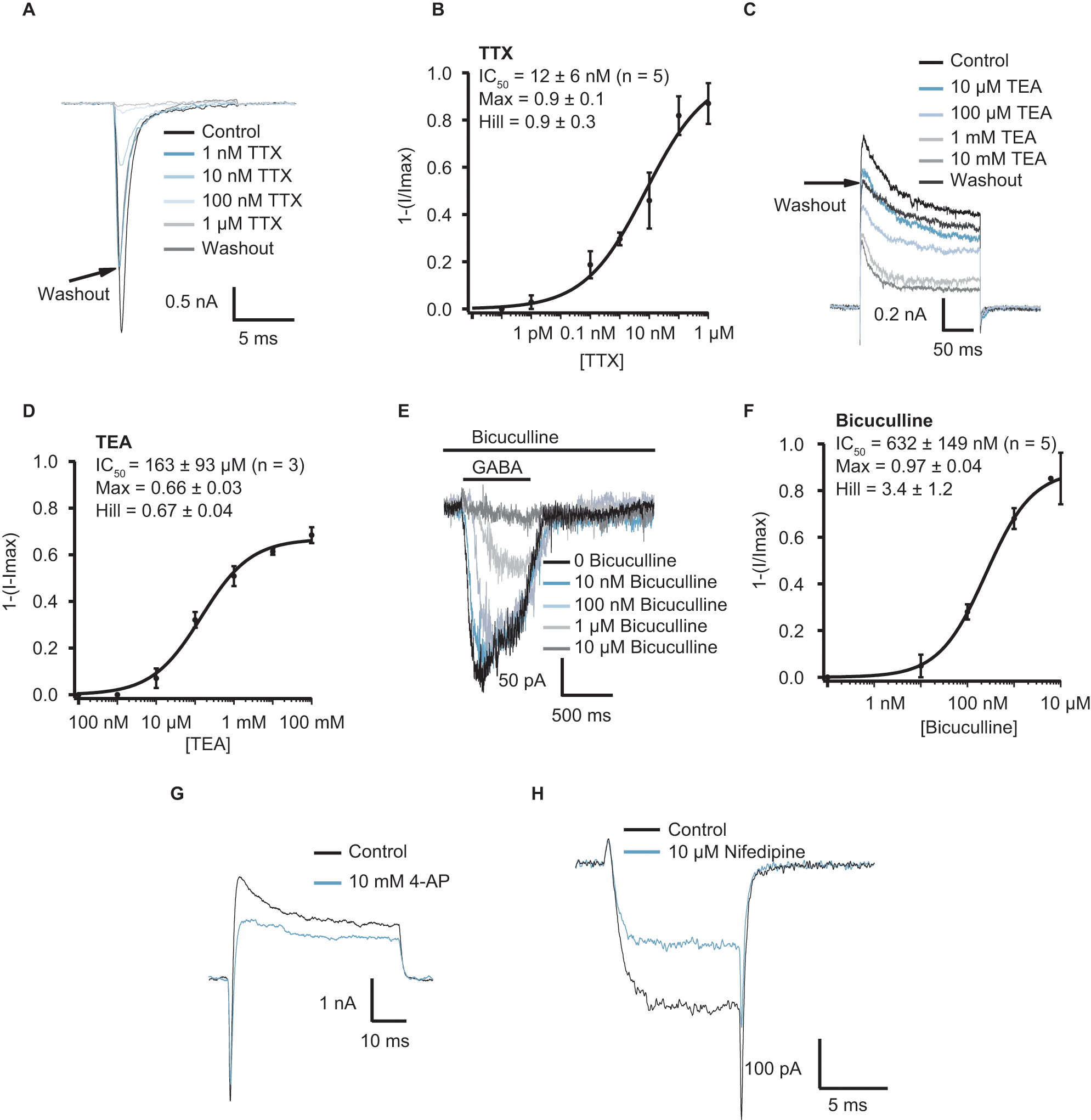
Pharmacology of iCell Neurons as measured with automated and manual patch-clamp recordings. Panel
The ligand GABA (30−100 µM) activated a transient, slowly desensitizing inward current response at a holding potential of −70 mV in 8 of 11 cells tested in manual patch clamp (example current trace in Fig. 2G ) and about 45% of cells tested on the Patchliner ( Fig. 1D , example current trace in Fig. 2H ). There, the current amplitude ranged from as little as −5 pA up to almost −1 nA. The average peak current amplitude elicited with 30 µM for an average of 63 cells was −167 ± 26 pA. These currents are, again, much smaller than those measured in manual patch clamp, where 100 µM GABA elicited inward currents at a holding potential of −70 mV from −1.5 to −5.5 nA with an average peak amplitude of −3.9 nA ( Table 1 ). In the manual patch experiments, we applied GABA longer to follow the desensitization phase of GABA-induced currents. This enabled us to fit a desensitization time constant (τdes) to the data. Cells showed a rather heterogeneous desensitization of GABA-induced currents with an average τdes of 1.6 s, ranging from 0.6 to 2.6 s (n = 8), pointing to a mixed expression of different GABA receptor subunit combinations.
High-quality patch-clamp amplifiers (EPC10; HEKA Elektronik) are incorporated into the Patchliner for electrophysiology measurements. Since these amplifiers have current clamp as well as voltage clamp capabilities, action potentials in the current clamp mode could be recorded in the iCell Neurons. This allowed us to also compare the generation of action potentials in both recording configurations, elicited by a short depolarizing pulse. In the example shown in Figure 2I , a short (3-ms) depolarizing pulse elicited an action potential in manually patched cells. In the example shown in Figure 2J , a short (2-ms) depolarizing pulse elicited an action potential on the Patchliner. Spontaneous action potentials were not observed using the Patchliner, although only a few current clamp experiments have been attempted thus far. In manual patch clamp, cells are still connected to a large network of other neurons. In those conditions, spontaneous action potentials could sometimes be observed.
The comparison of physiological parameters of neuronal ion channels measured either in manual or automated patch clamp showed that chip-based recording of neuronal cells is feasible and comparable in kinetics and voltage dependence of channels. However, the amplitude of the elicited currents is about 10 times smaller than when patching manually adherent, well-differentiated neurons. If corrected for the smaller cell volume that we estimated from the membrane capacitance (Cm), there is still a difference by a factor of about 3 between the two configurations. This observation may be explained by the subcellular localization of those ion channels. If, during the detachment of cells for automated recordings, more distal cell regions are detached from their somata, channels with a higher density there would be underrepresented when cells are measured in suspension.
With this data set, we found the assay in Patchliner sufficiently validated to perform pharmacological experiments in this configuration. Full concentration-response curves with at least four concentrations of a compound could be completed on a single cell. Figure 3A – F shows current responses and concentration-response curves for TTX block of Nav, TEA block of Kv, and bicuculline block of GABA currents. Figure 3A shows Nav currents activated using a single voltage step protocol to −20 mV and block of these currents by increasing concentrations of TTX. TTX was applied for at least 100 s before peak current amplitude at −20 mV was evaluated. Figure 3B shows the corresponding concentration-response curve for an average of five cells, IC50 = 12 ± 6 nM (n = 5), consistent with a TTX-sensitive channel expressed in these cells. Full block was achieved with the highest concentration (1 µM) of TTX. This rules out the presence of the neuronal TTX-insensitive channels, Nav1.8 and Nav1.9, and the primarily cardiac TTX-insensitive channel, Nav1.5. However, further analysis would be required to discover the exact nature of the Nav channel present in the iCell Neurons. Figure 3C shows Kv currents (inactivating, A-type) elicited using a single voltage step to 40 mV and block of these currents by increasing concentrations of TEA. TEA was applied for at least 100 s before peak current amplitude at 40 mV was evaluated. Figure 3D shows the corresponding concentration-response curve for an average of three cells, revealing an IC50 of 163 ± 93 µM (n = 3). This is consistent with a TEA-sensitive A-type Kv channel. 8 Full block was not achieved even with the highest concentration (10 mM) of TEA; maximum block achieved was 66%. The GABA-activated currents were blocked by bicuculline in a concentration-dependent manner ( Fig. 3E ). Bicuculline was preincubated for at least 30 s before coapplication with 30 µM GABA. Bicuculline blocked the GABA-activated currents with an IC50 = 632 ± 149 nM (n = 5; Fig. 3F ), proving the existence of a GABAA receptor in these cells as opposed to a bicuculline-insensitive GABAB or GABAC receptor type (for review, see Barnard et al. 9 ). In manual patch clamp, no concentration-response curves were generated due to the much higher workload. We tested the pharmacology of the observed currents at single concentrations of drugs using a focal application system. Nav currents were fully blocked by 1 µM TTX as in the automated system (data not shown). We also tested the sensitivity of A-type Kv currents to the selective blocker 4-AP and observed a relatively selective inhibition of the initial, transient phase of outward Kv currents elicited by a depolarizing voltage step to +20 mV in presence of 10 mM 4-AP ( Fig. 3G ).
Our studies aimed to address two questions for human neurons derived from pluripotent stem cells: (1) Do they faithfully express voltage-gated ion channels that are underlying neuronal excitability? (2) Can we use automated patch-clamp recording devices to study pharmacological and physiological parameters of ion channels expressed in these cells?
The presented preliminary results indicate that iCell Neurons indeed show the three major voltage-dependent currents that underlie neuronal excitability with the expected physiological and pharmacological properties. Our study is not yet sufficient to compare in detail if all relevant properties of these currents are similar to or different from those from human primary neurons. We did not observe any clear difference between published and our data, neither in physiological nor pharmacological parameters measured. In addition to voltage-gated currents, these cells also express GABAA receptors in significant amounts, measurable in both recording devices. It seems that the iCell Neurons that we studied are a heterogeneous population of different neuronal and even nonneuronal phenotypes. This could be used as an advantage in drug screening campaigns but should be clearly taken into account when comparing results from single-cell experiments of neurons derived from different donors. A sufficiently high number of cells would need to be characterized before making statements that the observed differences are due to donor-specific differences and not due to the heterogeneous cell population. Nevertheless, our characterization clearly shows the potential of pluripotent stem cell–derived neurons for studying cellular function, disease states, and neurotoxicity as a viable alternative to primary neuronal cell culture.
To our knowledge, our study is the first to characterize ion channels of hIPS-neurons in an automated patch-clamp device. We have been surprised about the acceptable success rate despite the nonoptimal shape of the cells in suspension. It is possible to generate data from about 40% of the wells used, about half of the relatively high success rates of manual patch clamp in those cells (J. Mosbacher, personal communication, 2012). Comparing current responses from manual and automated patch clamp, we found some specific differences. A major difference are the smaller currents measured in the automated patch-clamp device. This is likely based on the cell preparation rather than the recording technology. If, during the detachment of cells for automated recordings, more distal cell regions are detached from their somata, channels with a higher density there would be underrepresented when cells are measured in suspension. Cells in suspension were about three to four times smaller in membrane capacitance, which already would explain a factor of 3 to 4 in the current amplitudes. A second difference was the frequency of cells showing significant Nav currents. When visually guided by cell morphology, manually patched cells always showed transient, fast inactivating sodium currents, indicative of excitable, neuronal cells. Automated patch-clamp recordings come from more randomly patched cells. In this configuration, only 40% of patched cells showed significant Nav currents. Either the trypsinization of cells affects sodium channels in particular, or the iCell Neurons are a heterogeneous mix of different cell types, including nonexcitable cells such as astrocytes. Indeed, there are observations that iCell Neuron cultures express the astrocyte marker GFAP (J. Mosbacher, personal communication, 2012) but not to such a large extent. Further expression studies may be needed to clarify this point further.
It is very feasible to produce concentration-response curves using the Patchliner, seal conditions are stable over time, and the ability to wash in and wash out drugs in a relatively short time allows a thorough differentiation of drug effect and other effects such as current rundown. It will be interesting to see if also currents from more distal cell compartments, such as GABAB-mediated modulation of Kir or N-type channel currents, can be recorded in suspension. Based on the presented data, planar patch-clamp devices (e.g., the Patchliner, which was used in this study) provide an easier and more user-friendly alternative to conventional patch clamp, bringing patch-clamp electrophysiology to a wider audience while maintaining quality of data.
The combination of a more primary-like neuronal population of cells, such as stem cell–derived neurons, and the potential of stem cell–derived neurons to provide critical models for disease states, coupled with voltage and current clamp recordings from several cells in parallel using an automated patch-clamp device, will be a major advantage for disease research and drug discovery.
Footnotes
Acknowledgements
We highly appreciate excellent expert support and input from C. Deymier, E. Ertel, M. Aubert, R. Moon, and W. Fischli.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
