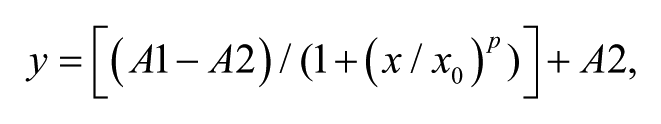Abstract
Since the cloning of its first member in 1998, transient receptor potential (TRP) cation channels have become one of the most studied ion channel families in drug discovery. These channels, almost all calcium permeant, have been studied in many different (patho)-physiological and therapeutic areas as diverse as pain; neurodegenerative, cardiovascular, and inflammatory diseases; and cancer. At the same time, implementation of automated electrophysiology screening platforms has significantly increased the tractability of ion channels, mainly voltage gated, as drug targets. The work presented in this article shows the design and validation of TRP screening assays using the IonWorks Quattro platform (Molecular Devices, Sunnyvale, CA), allowing a significant increase in throughput to support drug discovery programs. This new player has a direct impact on resources and timelines by prioritizing potential candidates and reducing the number of molecules requiring final testing by manual patch-clamp, which is still today the gold standard technology for this challenging drug target class.
Introduction
Transient receptor potential (TRP) channels are part of a large family that has been well described in recent years. TRP channels are expressed in many cell types and tissues (almost ubiquitously), they are diverse in function and many are important in sensory signal pathways. Their pathological implications also vary and include pain, respiratory reflex hypersensitivity, cardiac hypertrophy, and ischemic cell death. 1
Pharmaceutical companies are targeting TRP channels more and more in their drug discovery programs. Although ion channels have been successful drug targets, achieving subtype selectivity has always been a major challenge, particularly with voltage-gated sodium and calcium channels. Compared with other ion channel families, TRP channels share less homology, which increases the likelihood of identification of subtype-selective compounds. Several other factors make TRP channels appealing as drug targets. First, TRP channels act as integrators of several well-described signaling systems, including those that are mediated by cell surface receptors, thus facilitating assay development. Second, mutations in many of the genes that encode TRP channels are sufficient to cause disease in humans, making them validated targets in translational medicine models. 2
One of the channels that we focus on in this article is TRPA1, a cation channel that functions as a cellular sensor, detecting mechanical, chemical, and thermal stimuli. It is a component of neuronal, epithelial, blood, and smooth muscle tissues in mammals. TRPA1 is largely expressed in primary sensory neurons that mediate somatosensory processes and nociceptive transmission, supporting the involvement of the activation and blockade of TRPA1 in both inflammatory and neuropathic pain transmission. Consequently, this channel is currently being targeted in the search for new analgesic medicines. 3
Our study was extended to other subunits belonging to the TRPV and TRPM subfamilies, specifically TRPV1 and TRPM2. Both are considered putative targets in pain and inflammatory diseases. TRPV1 was coexpressed with TRPA1 in many small-diameter neurons that contain both substance P and calcitonin gene–related peptide (CGRP), highlighting it as a component in the propagation of noxious and inflammatory pain stimuli.1,4 TRPM2 is a nonselective calcium-permeable cation channel that acts as a sensor for reactive oxygen species. It is expressed abundantly in immune cells and is important in inflammatory processes. TRPM2 plays a crucial role in inflammatory and neuropathic pain. 5
In ion channel drug discovery projects, manual patch-clamp has been the bottleneck in identification of new chemical matter with low throughput and high expertise required. Thus far, there has been limited progress in significantly increasing the throughput of ligand-gated ion channel patch-clamp studies on mammalian cells using automated electrophysiology techniques. 6 A major limitation is the inability of most systems to rapidly apply ligands and simultaneously measure signals, as can be performed with conventional patch-clamp techniques. Here, we show that this can be successfully achieved with the highest throughput automated planar array electrophysiology IonWorks system (Molecular Devices, Sunnyvale, CA), although technically not designed for this purpose. 7 We found that measurements yield a highly acceptable pharmacological profile. Given the throughput of this format and the electrophysiological readout, we suggest that this approach may be valuable for focused set and secondary screening for TRP ion channel drug discovery programs.
Materials and Methods
Cell Culture
Generation of stably transfected cell lines was achieved using the Flp-In T-Rex expression system (Invitrogen, Carlsbad, CA). For this purpose, complementary DNAs (cDNAs) encoding for the human (NM007332) as well as the mouse (NM177781) TRPA1, human TRPV1 (AY131289.1), and human TRPM2 (NM003307.3) were cloned into the Flp-In T-Rex expression vector (Invitrogen) and subsequently transfected into HEK-293 cell lines.
The cells were kept in culture under standard conditions (37 °C, air supplemented with 5% CO2) in Dulbecco’s modified Eagle’s medium (DMEM) (Invitrogen) supplemented with 10% fetal calf serum (Invitrogen), 50 µg/mL hygromycin (Invitrogen), and 15 µg/mL blasticidin (Invitrogen). Cells were subcultured every 3 to 4 days using accutase (Sigma, St. Louis, MO) to detach the cells by enzymatic dissociation. At least 12 to 24 h prior to assays, doxycycline (BD Biosciences, Franklin Lakes, NJ) was added to induce target expression.
Whole-Cell Patch-Clamp Recordings
The whole-cell configuration of the patch-clamp technique was used to validate each tested cell line. TRPA1-expressing human embryonic kidney (HEK) cells were prepared for conventional patch-clamp recordings by first plating them at low density onto glass coverslips. Twenty four hours later, a single coverslip was placed into a small recording chamber, which was constantly perfused (1–2 mL/min) with an external recording solution containing (in mM) 145 NaCl, 4.5 KCl, 3 MgCl2, and 10 HEPES (osmolarity set at 315 mOsm/L and pH 7.4 with NaOH) at room temperature (20–25 °C). Patch electrodes with tip resistances ranging from 4 to 7 MΩ were pulled and filled with a prefiltered intracellular solution containing (in mM) 140 Cs-methanesulfonate, 2.27 MgCl2, 1.91 CaCl2, 10 EGTA (free calcium estimated at 50 nM), 10 HEPES (osmolarity set at 307 mOsm/L and pH 7.4 with CsOH). Voltage-clamp and data acquisition were performed using an Axopatch 200B (Axon Instruments, Union City, CA) amplifier coupled to pClamp control software (v10; Axon Instruments) via an analog to digital converter (Digidata 1440; Axon Instruments). Signals were prefiltered at a 5-kHz bandwidth and sampled at 1 kHz. All experiments were performed on cells with seal resistances of 1 to 10 GΩ and at a holding potential of 0 mV. TRPA1 opener and compound solutions in external buffer were applied via multibarreled, pressure ejection pipettes controlled by electromagnetic switch valves (Warner Instruments, Holliston, MA).
IonWorks Quattro Recordings
Planar assay electrophysiology recordings were made on an IonWorks Quattro enabled for single-cell (HT) and population patch-clamp (PPC) measurements. 7 Cell suspensions were created immediately prior to an experiment by first aspirating the growth media from the T175 flask and washing with Ca/Mg-free phosphate-buffered saline (PBS) (Invitrogen). Cells were then treated with accutase as an enzymatic dissociation agent for 6 min at 37 °C.
Following a short centrifugation step (1000 rpm for 2 min), cells were resuspended in 5 mL prefiltered external buffer containing (in mM) 140 NaCl, 4 KCl, 1 MgCl2, 0.5 CaCl2, and 10 HEPES (osmolarity set at 302 mOsm/L and pH 7.4 with NaOH). Cells were gently triturated for 1 min to yield a final density of ~2 × 106 cells/mL, and cell suspension was then added to the cell reservoir on the instrument, from which it was dispensed onto the patch plate. All recordings were made using internal solution (in mM): 130 K-gluconate, 4 NaCl, 1 MgCl2, 10 EGTA, 5 CaCl2 (free calcium below µM range), and 10 HEPES (osmolarity set at 306 mOsm/L and pH 7.4 with KOH).
The permeabilizing reagent used for attaining the perforated patch-clamp configuration was amphotericin (A-4888; Sigma) which was solubilized in 100% DMSO to a concentration of 10 mM with the aid of sonication and vortexing. Amphotericin was added to a 50-mL aliquot of the internal buffer to give a final concentration of 10 µM.
The base protocol allowed 3 min for the cells to seal to the substrate followed by a further 10 min to obtain the stable perforated patch-clamp configuration. All recordings were made at room temperature (20–25 °C). Cells/wells were clamped using the electronics (E-) head held at −60 mV (with reference to a common “intracellular” ground electrode) for 10 s. Ramp (500 ms from −80 to +80 mV) command was then applied and the resulting currents sampled at 2.5 kHz. Leak subtraction was not employed. After this initial signal measurement (in the absence of ligand), the E-head was removed from the recording well, and a solution containing the channel opener (or test compound) was added via a fluidics (F-) head dispenser. The F-head must then be moved away before the E-head can be repositioned to clamp again and record from the cells. By using the “eighth of a plate” at once software configuration, it was possible to measure compound-evoked currents only 8 s after the compound addition.
Reagents
Compounds were obtained from the following sources: allyl isothiocyanate (AITC) AP-18, ruthenium red, capsaicin, hydrogen peroxide (H2O2), and N-(p-amylcinnamoyl) anthranilic acid (ACA) were purchased from Sigma. Specific TRPA1 (HC-030031) and TRPV1 (A-784158) blockers were purchased from Tocris (Ellisville, MO).8,9 TRPA1 antagonist, A-967079, was synthesized at Sanofi (Paris, France). 10
Unless otherwise stated, all compounds were initially solubilized to a concentration of 10 mM in 100% DMSO. Serial dilutions were made by hand in appropriate external buffer. For the IonWorks experiments, low-volume (1 µL), 3-fold serial dilutions were performed on a 384-well microplate (Greiner, Monroe, NC) in DMSO using a Mosquito (TTP Labtech, Melbourn, UK) liquid-handling device. O and P rows were used as high (agonist at Emax) and low (DMSO vehicle) controls in all cases. Then, 300-nL plate copies were stamped out onto fresh 384-well plates. Immediately prior to the experiment, 100 µL bulk volume of the external recording solution was added across the plate using a Multidrop Combi (Thermo, Waltham, MA) to yield serial dilutions in 3% DMSO. Allowing for the final 1:3 dilution in the patch plate, the final assay DMSO concentrations were 1%.
Data Handling and Analysis
Data analyses and graphical presentation were performed using a combination of IonWorks (v2.0; Molecular Devices), IDBS XLfit (v4.2; IDBS, Guildford, UK), Microcal Origin (v8.6; OriginLab Corporation, Northampton, MA), and pClamp (v10; Axon Instruments) software. All data are given as mean ± standard deviation.
For the IonWorks data, current amplitudes were measured from the ramp protocol at +50 mV from each read and the drug-evoked signal calculated by subtracting the precompound from the postcompound amplitude. Concentration-response curves were fitted using a four-parameter logistic equation of the form
where A1 is the maximum asymptote, A2 is the minimum asymptote, x0 is the XC50, and p is the Hill slope.
Results and Discussion
Pharmacological Validation Using Conventional Patch-Clamp Technique and Transfer onto the Automated Patch-Clamp System IonWorks Quattro for an hTRPA1 Agonist HT Assay
A human TRPA1 stable cell line was generated using an inducible expression system allowing transient expression of the gene of interest while decreasing toxicity due to channel expression. One day after doxycycline addition to the culture media, TRPA1 protein induction occurred and recordings were performed using the manual whole-cell patch-clamp technique. In voltage-clamp studies, perfusion of 5 µM AITC induced reversible strong currents ( Fig. 1A ) in human TRPA1-expressing cells, which was not observed in noninduced cells (data not shown). Coapplying the TRPA1 blocker A-967079 at increasing concentration (from 30 nM to 3 µM) induced a dose-dependent inward and outward current block. In Figure 1B , the time course of recorded hTRPA1 currents at −50 and +50 mV is shown (color coding as for Fig. 1A ). After full block was obtained, a washout step was performed. The TRPA1 current was only partially recovered. Experiments with double AITC application highlighted channel desensitization (data not shown). The difference in current amplitude before and after compound application could be the result of this desensitization process. These experiments provided strong validation of the biological materials used in further experiments on the automated patch-clamp system IonWorks Quattro as well as valuable information on channel biophysics.
Biophysical and pharmacological validation of the recombinant human TRPA1 cell line using manual and automated patch-clamp techniques. (
Despite being the gold standard technology, manual patch-clamp is often a bottleneck in a drug discovery program. To increase throughput and support structure-activity relationship (SAR) studies with an electrophysiological system, we had an opportunity to assess the automated patch-clamp platform, IonWorks Quattro, to perform TRPA1 experiments.
At first, a classic protocol was followed to estimate TRPA1 assay feasibility on such a platform. AITC 10 µM was used as reference agonist and added in an HT mode assay (entire process described in Fig. 1C ). Seal success was larger than 90% and seal resistance was around 90 ± 28 MΩ (n = 350). Increase in current amplitudes (difference between pre- and postrecordings) was variable mainly due to cell line heterogeneity. As shown in Figure 1D , a large number of cells (about one-third) displayed a TRPA1 current smaller than 200 pA. However, currents larger than 1 nA were recorded in more than 20% of cells. In addition, the desensitization process highlighted during the manual patch-clamp experiments was monitored by adjusting the time windows to estimate the maximum amplitude obtained following agonist application. Ramp protocols run on a single cell at different time intervals showed that currents reached maximum amplitude after 40 s (around 800 pA at +50 mV in the representative shown recording), and then desensitization occurred ( Fig. 1E ). Currents decreased by 50% compared with maximum current when recorded 60 s following AITC application. Statistical analysis performed on a complete patch-plate (384 recordings) using AITC was investigated. Vehicle (DMSO)–treated wells were measured with no significant response. AITC elicited the largest TRPA1 current 40 s after application ( Fig. 1F ). This parameter was used in further described assays. Finally, attempts to generate concentration-response curves to reference agonists in HT single-hole mode were confounded by the cell-to-cell variation. It was decided to investigate a PPC (population) mode approach.
Transfer onto the Automated Patch-Clamp System IonWorks Quattro of an hTRPA1 Agonist PPC Assay and Set Up of an Ortholog Mouse TRPA1 Agonist PPC Assay
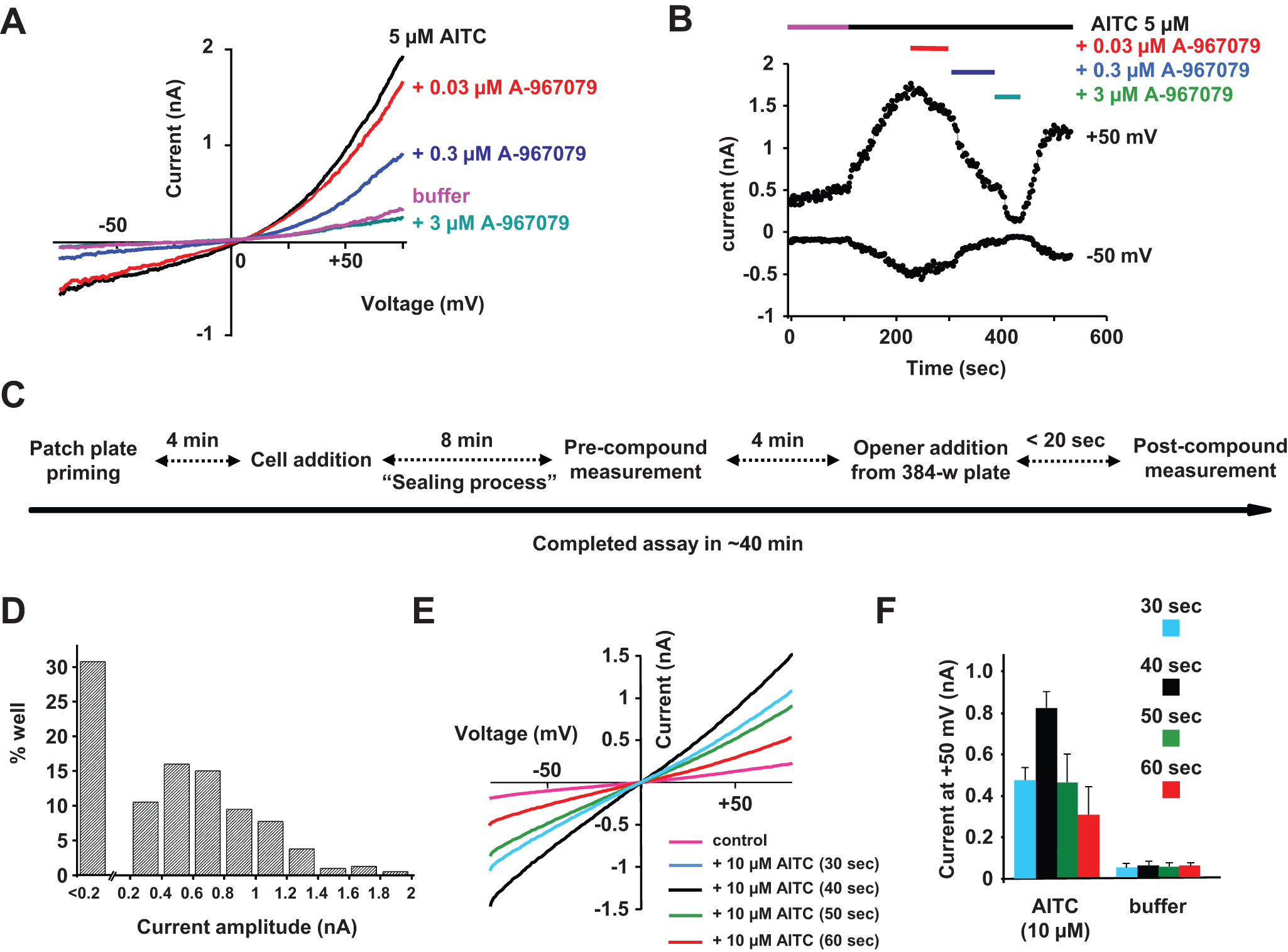
In PPC mode, the seal resistances were lower (as normally seen for other assays) than HT mode, with mean resistance being 35 ± 4 MΩ (n = 8 plates). As anticipated, measurement of an ensemble “average” signal from a population of (up to 64) cells yielded measurable TRPA1-evoked currents, which were highly consistent well to well. When measured 40 s after opener application (as highlighted during HT experiments), a maximum current amplitude was reached with an average current of 780 ± 80 pA (n = 8) at +50 mV for the 10-µM AITC application ( Fig. 2A ). As with AITC, robust TRPA1 currents were also obtained for two TRPA1 agonists already described in the literature, menthol and hydrogen peroxide, with maximum current amplitudes of 740 ± 80 pA (n = 16) and 420 ± 100 pA (n = 16), respectively ( Fig. 2A ).11,12 Note that the kinetics of the responses was modulated and a much slower response was obtained when using H2O2. This correlates with published data that describe an indirect mode of TRPA1 activation by this agonist. 11
Agonist TRPA1 recordings on IonWorks Quattro (IWQ) in population patch-clamp (PPC) mode. (
Mean Z′ value determined with the high (only calculated with AITC) and low (vehicle only—0.3% DSMO) control well currents was greater than 0.7. Assay stability and robustness were assessed over 10 weeks. Application of the 10-µM AITC-triggered current amplitude was consistent, and variations were small from assay to assay (data not shown).
With the added consistency and PPC precision, it proved possible to construct robust agonist concentration-response curves. As shown in Figure 2B , agonist elicited concentration-dependent currents that could be fitted with a four-parameter logistic equation. Concentration-response curves were obtained and the rank order of agonist potency was AITC > menthol > H2O2, with mean effective half-maximum concentration (EC50) values of 2.2 ± 0.4 µM (n = 16), 64 ± 5 µM (n = 3), and 174 ± 12 µM (n = 3), respectively. These EC50 values are comparable to values from the literature.11,12
The feasibility of an IonWorks Quattro in PPC mode assay was also evaluated using an HEK cell line expressing the mouse TRPA1 channel. Seal resistances were measured at 32.8 ± 5.7 MΩ (n = 3 plates). Like hTRPA1, measurement of an ensemble “average” signal from a population of (up to 64) cells provided consistent well-to-well current amplitude measurements. When measured 40 s following opener application at +50 mV, an average current of 845 ± 115 pA (n = 3 plates) was obtained for 30 µM AITC. No current was detected when buffer alone was applied or when 1 µM A-967970 was coapplied ( Fig. 2C ). The well-to-well current reproducibility allowed dose-response experiments, and an EC50 value of 10.3 ± 1.9 µM (n = 8) was determined for AITC ( Fig. 2D ). This shift between orthologs in the EC50 values for agonists such as AITC has already been described in the literature. 13
Set Up of an Antagonist PPC Mode TRPA1 Assay on the Automated Patch-Clamp System IonWorks Quattro
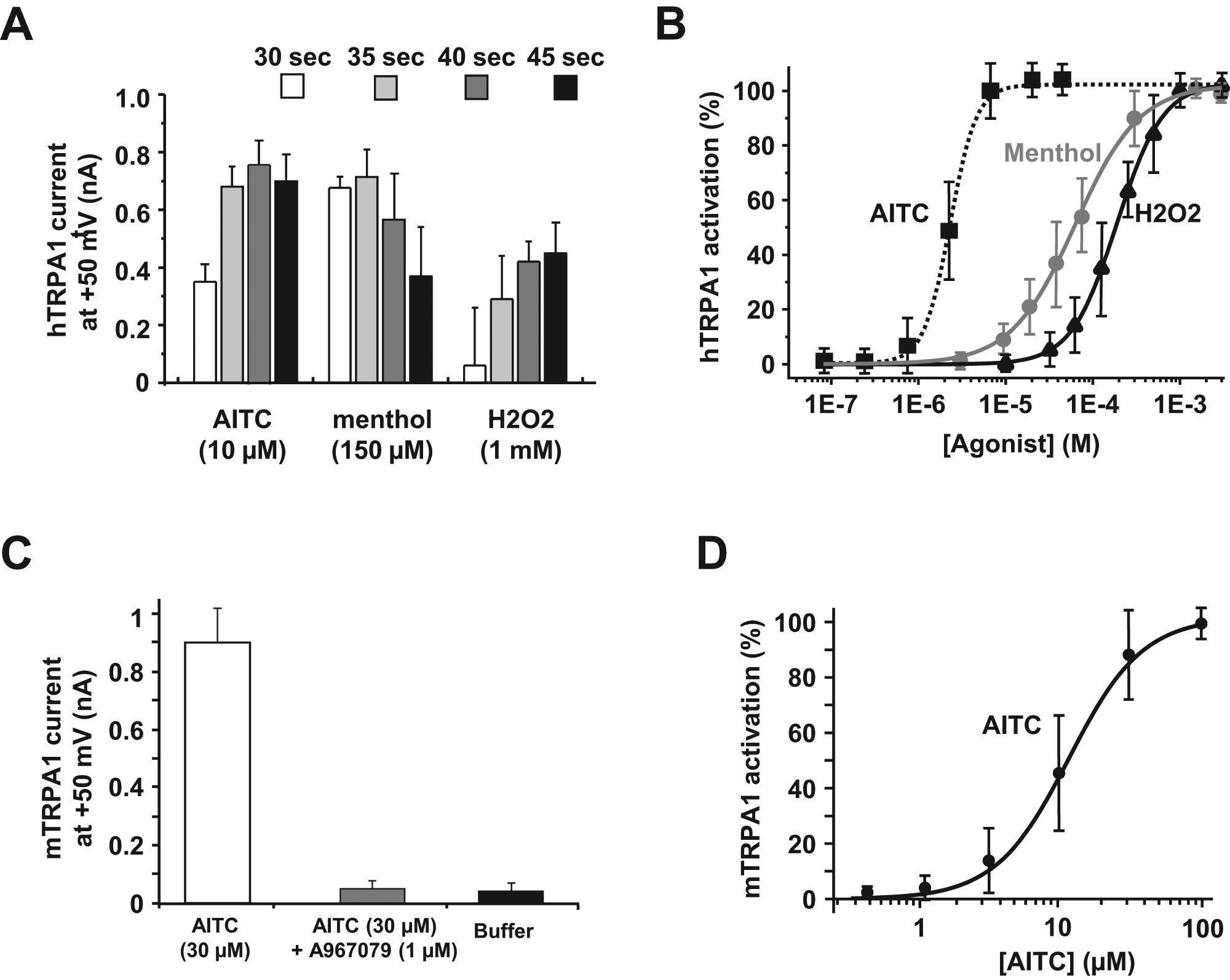
Knowing the effect of several agonists, the next step was the evaluation of such a challenging target in an antagonist mode on an automated patch-clamp platform. To optimize compound effect and match with published data, we noticed that compounds had to be preincubated with the cells prior to opener addition (around 10 min). Following this process, the effect of the described antagonist A-967079 was evaluated and compared with the current amplitude obtained when agonist was added alone. We ran this blocking assay using a single dose of A-967079 (1 µM) versus a panel of described agonists ( Fig. 3A ). For all three tested agonists (AITC, menthol, and H2O2), full block was observed (measured 40 s after application) when 1 µM A-967079 was coapplied. No current was detected when A-967079 was added with buffer (data not shown).
IonWorks Quattro (IWQ) innovative assay for transforming of throughput to identify human and mouse TRPA1 antagonist compounds. (
Four published antagonists—A-967079, ruthenium red, AP-18, and HC-030031—were then screened on the same plate (n = 8 for each tested compound). The percentage of inhibition following application of 10 µM AITC was calculated and data were fitted, giving an IC50 values of 20 ± 2 nM, 257 ± 22 nM, 397 ± 33 nM, and 3.84 ± 1.04 µM, respectively ( Fig. 3B ). These pharmacological data matched with previously published IC50 values even if different technologies (such as calcium mobilization fluorescence assays) were used.8,14,15
Following these positive results obtained with the human TRPA1 antagonist assay, we investigated a similar approach with the rodent ortholog, mTRPA1. Once again, we showed that several TRPA1 agonists (AITC, menthol, and H2O2) elicited mTRPA1 currents, and these currents could be blocked when 1 µM A-967079 was preincubated ( Fig. 3C ).
Finally, four known antagonists (already described in the human TRPA1 assay)—A-967079, ruthenium red, AP-18, and HC-030031—were tested in parallel on the same assay plate on several runs using AITC as the opener. Due to the high reproducibility of current amplitudes from well to well, we were able to extract IC50 values for each tested compound (n = 8). The percentage of inhibition following application of 30 µM AITC was calculated and data were fitted, giving IC50 values of 149 ± 12 nM, 950 ± 121 nM, 1.11 ± 0.12 µM, and 4.95 ± 0.66 µM for A-967079, ruthenium red, AP-18, and HC-030031, respectively ( Fig. 3D ).
TRPV1 and TRPM2, Two Members of the TRP Family Studied Using IonWorks Quattro in PPC Mode
Other targets of the TRP family were also investigated. Cell lines expressing TRPV1 and TRPM2 were available, and automated patch-clamp assay feasibility was assessed.
Using a protocol similar to the one described for the TRPA1 assays, TRPV1 currents could be elicited on the IonWorks Quattro platform when cells expressing the TRPV1 channel were recorded before and after application of 150 nM capsaicin ( Fig. 4A ). At +50 mV from the ramp protocol, an average TRPV1 current of 690 ± 230 pA (n = 8) was obtained ( Fig. 4B ). Application of 1 µM A784168, a published TRPV1 blocker, caused a full block of the TRPV1 capsaicin-elicited conductance. 9 Note that assay success was greater when channel induction was performed only 6 h before assaying (probably due to TRPV1 expression toxicity). Also, a lower doxycycline concentration (10 ng for TRPA1 vs. 1 ng for TRPV1) was used to improve assay quality with Z′ > 0.6. Noninduced cells did not elicit any current following capsaicin application (data not shown).
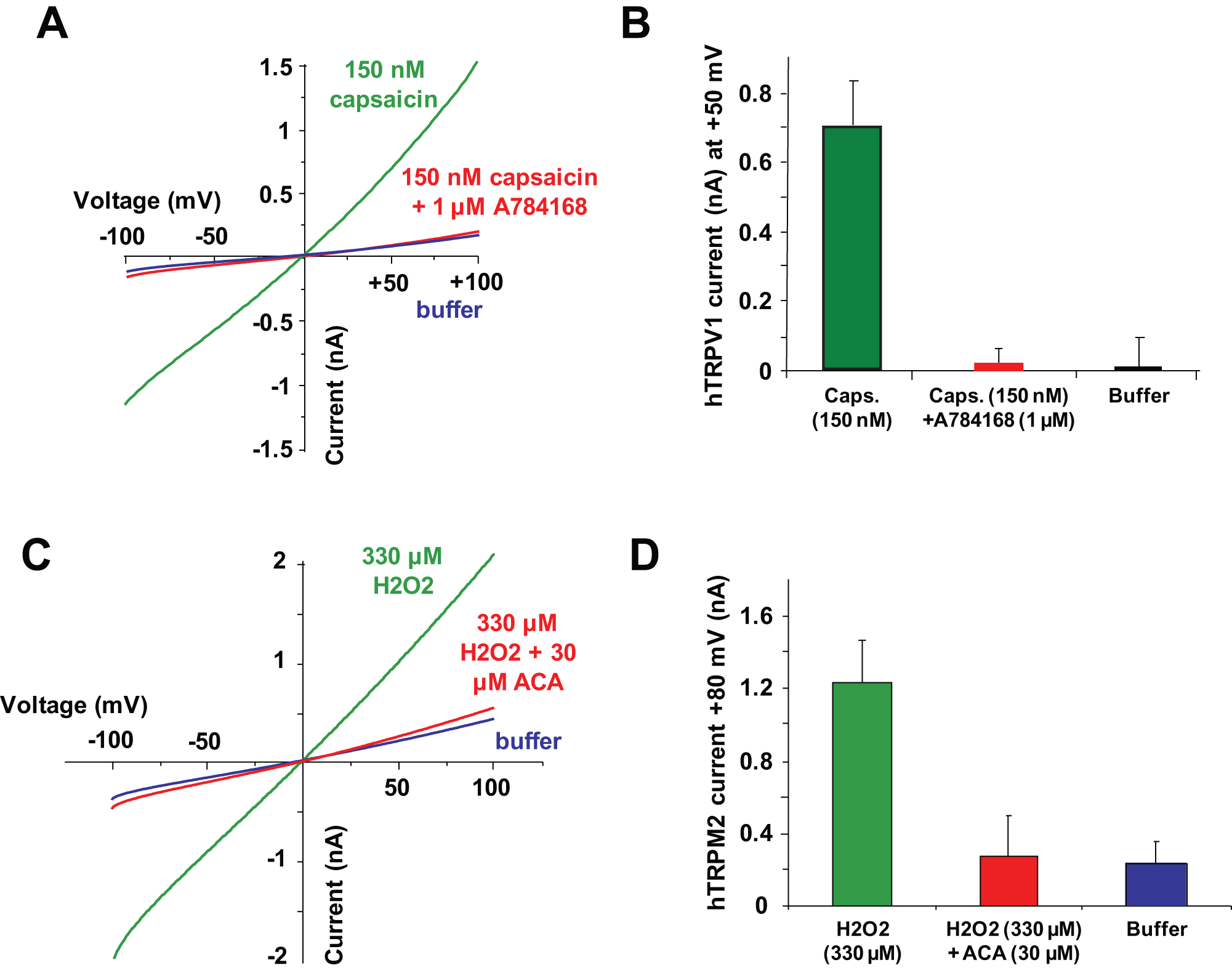
TRPV1 and TRPM2 pharmacology on IonWorks Quattro (IWQ) population patch-clamp (PPC). (
With a high seal success and reliable average resistance (36.2 ± 4.2 MΩ, n = 384), pharmacological experiments were possible. The concentration-response curve for capsaicin was performed and showed an EC50 value of 33 ± 3 nM (n = 16), similar to published data. 16 In addition, an IC50 value for A784168 was determined using 150 nM capsaicin as the opener (data not shown). When the curve-fitting process was applied, an estimated IC50 value of 7.0 ± 0.6 nM (n = 8) was calculated, in the same range as the IC50 value published in the literature. 9
Finally, a recombinant cell line expressing the oxidative stress–sensitive calcium-permeable human TRPM2 channel was used. Hydrogen peroxide was used as an indirect channel opener. Current reading time had to be extended to detect full-channel activation (up to 8 min). Figure 4C shows H2O2-elicited current traces before and after application of 330 µM H2O2. An average current of 1.20 ± 0.28 nA (n = 8) was observed after H2O2 application. The block of this conductance by ACA was examined. A high concentration of 30 µM was applied and resulted in full block of the TRPM2 H2O2-elicited conductance ( Fig. 4D ). No current was detected when buffer was added instead of H2O2 agonist solution. A concentration-response curve was performed using H2O2 at different concentrations, and an EC50 value of 45.9 ± 6.4 µM (n = 16) was calculated, similar to published data. 17 Using the EC80 concentration of 330 µM H2O2, we ran an assay in antagonist mode with ACA preincubation. An IC50 value of 2.81 ± 0.55 µM (n = 8) was extracted from % inhibition obtained at each of the six tested concentrations (data not shown). These data are very well correlated with pharmacological data available from literature. 18
Transforming TRP Channel Drug Discovery
In this article, we describe a new medium-throughput assay on the IonWorks Quattro platform for identifying pharmacological enhancers and blockers of TRP channels. This tool (1) offers an alternative to fluorescence assays with a novel valuable automated electrophysiological method and (2) significantly increases capacities for screening and profiling compounds at TRP channels, which is critical to accelerate drug discovery of such challenging targets.
Three members of this new target family were studied: TRPA1 (human and mouse orthologs), TRPV1, and TRPM2. The efficacy of well-known agonists and antagonists for each subunit was confirmed, showing that such technology can support drug discovery from compound library screening until selectivity profiling and lead/candidate selection. In addition, these results strongly suggest that this technology, initially developed for the study of voltage-gated channels, can be used for ion channel subfamilies that display discrete or no voltage dependence. Already described a few years ago for slow deactivating channels, this approach was again supported by recent work showing that calcium-activated potassium channels could also be recorded using the IWQ platform.6,19 Using the conventional patch-clamp method, compound testing on these TRP channels has been highly challenging and time-consuming. Implementation of an automated patch-clamp has been important to release the bottleneck represented by manual electrophysiology.
In addition to the usual clone selection and monitoring (both concentration and time) of the induction system, use of the population patch-clamp approach has been vital to get rid of cell line heterogeneity as well as provide an average current reproducible from well-to-well recordings. Also, the design of the recording system (48-channel E- and F-head) meant that we could reach a minimum interval between agonist application and recording of the ramp current of 25 s when setting a repetitive scan protocol. Using such a protocol, determination of the maximum signal window for each studied target has been one of the challenging steps. The preincubation protocol also has proven to be a crucial improvement to fit with the pharmacological data of all published tool compounds used to validate each of these agonist and antagonist mode assays. Indeed, pharmacological data obtained on the automated patch-clamp platform IonWorks Quattro in both agonist and antagonist modes were in a reasonable range comparable to previous reports for all tested targets, human and mouse TRPA1 as well as human TRPV1 and TRPM2.
Fitting the hardware limit with the biophysical parameters of the TRP channels allowed us to massively increase the throughput for these assays. With a success rate higher than 80%, we were able to test four compounds per hour for dose-response determination. If extrapolated, at 8 plates per day and 5 working days a week, this new approach would transform the TRP channel drug discovery process, allowing approximately 100 IC50 values to be determined per week.
Since electrophysiological data are always mandatory for the most promising compounds, being able to quickly validate compound activity on ortholog channels is a major plus.
We described in this article a new electrophysiological TRP channel assay that could have a real impact on chemical matter identification and timeline reduction, thus transforming drug discovery for these highly relevant therapeutic targets.
Footnotes
Acknowledgements
We thank Dr. Bruno Biton for stimulating discussions and critical reading of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.