Abstract
Introduction
Cold exposure initially induces peripheral vasoconstriction. After 5–10 min, distal blood vessels will transiently and cyclically vasodilate and vasoconstrict in a phenomenon known as cold-induced vasodilation (CIVD). Increased CIVD response is thought to improve dexterity and confer a reduced risk of frostbite injuries. Current guidelines recommend the use of an intravenous synthetic prostacyclin (PGI2) analog for the treatment of some cases of severe frostbite. This double-blind crossover study investigates the effects of the inhaled PGI2 analog epoprostenol on CIVD response through continuous finger temperature measurement during cold water immersion.
Results
Fourteen healthy volunteers completed both sessions of the study and were included in the analysis. Compared to placebo, inhaled epoprostenol sessions showed higher mean finger temperature (9.16 vs 8.34° C; p = .027), mean maxima temperature (10.86 vs 9.88° C; p = .045), and mean minima temperature (7.45 vs 6.80° C; p = .024). No significant difference was detected in the number of cycles (10.0 vs 7.93; p = .104). No hypotension, hypothermia, or hypoxia was observed, and no subject requested discontinuation due to side effects.
Conclusion
In this small study, inhaled epoprostenol induced a statistically significant increase in mean temperature, mean maxima temperature, and mean minima temperature in fingers immersed in a cold-water bath, consistent with an augmented CIVD response. The ability to deliver an inhaled PGI2 analog via nebulizer raises the possibility of early interventions to manage frostbite in austere environments, but further study is needed to draw any conclusions regarding the use of epoprostenol for frostbite care.
Introduction
Cold-induced vasodilation (CIVD) is a physiologic response observed in distal extremities after local cold exposure and has been associated with decreased risk of frostbite. 1 In the distal extremities, the initial response to cold exposure is sympathetically mediated peripheral vasoconstriction, which results in reduced local tissue temperature. However, after 5–10 min of continued cold exposure, distal blood vessels will transiently vasodilate in euthermic subjects. This cycling of vasoconstriction and vasodilation is known as CIVD. The response is thought to confer more dexterity in the cold and reduce susceptibility to frostbite. 1
Frostbite frequently occurs in remote or austere settings, which can limit the use of intravenous or intra-arterial therapies. Current guidelines recommend IV or intra-arterial tissue plasminogen activator (tPA) or IV iloprost for deep or proximal frostbite. Iloprost is a synthetic prostacyclin (prostaglandin I2 or PGI2) analog with vasodilatory and platelet inhibition effects and has shown favorable outcomes in a randomized control trial of the treatment of frostbite. 2 Current guidelines recommend iloprost as first-line therapy for grades 3 and 4 frostbite, < 48 h after thawing, and is suggested for deep frostbite extending to the distal interphalangeal joint or more proximal (grades 2–4), up to 72 h after rewarming, but ideally should be given as soon as possible. 3 Given the inherent risks of thrombolytics, iloprost may represent a safer treatment option in remote or austere settings.
Another synthetic PGI2 analog, epoprostenol, is approved in the United States for treatment of pulmonary hypertension, hypoxemic respiratory failure, and severe acute respiratory distress syndrome (ARDS). 4 Epoprostenol can be administered as a nebulized solution for certain indications and, at the time of this study, is more commonly available in the United States than iloprost. The study of CIVD response to cold water immersion has proven safe in numerous previous studies and offers the prospect of assessing the effects of pharmacologic interventions on the physiologic response to cold in humans without the risks inherent to actual frostbite. Guidelines recommend the use of intravenous iloprost for treatment of frostbite, but at the time of this study, IV iloprost had not yet been approved by the FDA, so we utilized the more widely available epoprostenol. To our knowledge, no studies have established whether inhaled PGI2 analogs affect physiologic responses or the circulation of the distal extremities to cold stimulus. We hypothesize that inhaled epoprostenol will affect CIVD response as measured by continuous finger surface temperature measurement during cold water immersion.
Methods
Subjects
Fifteen volunteers aged 18–65 years provided written consent and were included in this pilot study. Exclusion criteria were a history of frostbite to the upper extremities, vasoconstrictive disease, congenital heart defects, recent surgery (<90 days), allergy to epoprostenol or glycine diluent, current use of vasodilatory antihypertensives, current use of anticoagulants or other bleeding diathesis, and pregnancy. Individuals with recent infection, symptoms of, or high-risk exposures to COVID-19 were also excluded. Subjects were asked to refrain from exercise, smoking, alcohol, and caffeine use for at least 12 h before testing.
Study Design
This double-blind crossover design study compared the effects of inhaled nebulized epoprostenol (Flolan, epoprostenol sodium, GlaxoSmithKline, Philadelphia, PA) at 50 ng/kg/min ideal body weight versus inhaled nebulized 0.9% normal saline placebo during 2 cold water immersion sessions. This dose of epoprostenol is the usual starting dose in the treatment of ARDS and the recommended starting dose in the inhaled epoprostenol therapy protocol approved by the University of Utah Health Pharmacy. 5 Subjects were randomly assigned to either the placebo-first or epoprostenol-first groups. Subjects had a washout period of at least 24 h between sessions to minimize any carryover effect. Cold stress procedure followed established protocol previously published in CIVD research, with thermocouples attached to the pads of the 2–5 digits. 6 Subjects wore standardized hospital gowns with ad libitum blankets to ensure euthermia. Body temperature was measured every 5 min. Subjects immersed the 2nd to 5th digits of the dominant hand in a water bath: 5 min in a 35 °C bath to achieve a standardized starting skin temperature, followed by 30 min in a 5 °C water bath during which time subjects received a continuous nebulization of either placebo or epoprostenol. Finger skin temperature was assessed every 0.1 s using wire thermocouples (Omega Engineering, Inc. [Norwalk, CT]). The University of Utah Health Investigational Drug Service Pharmacy provided randomization, blinding, storage, preparation, and disposal of the intervention drug and placebo.
Safety
The study protocol was reviewed and approved by the University of Utah IRB. The research/clinical trial was registered with the University of Utah IRB under the registration number 00131539. Epoprostenol has a half-life of 3–6 min, and subjects were observed for at least 30 min (5–10 half-lives) after the termination of the study drug to ensure adequate clearance. 4 All subjects were placed on hemodynamic monitoring and were under the supervision of a licensed emergency physician in the University of Utah Hospital Emergency Department. Body temperature, blood pressure, heart rate, pulse oximetry, and subjective experience were recorded every 5 min, with side effects graded on a mild/moderate/severe simplified scale.
Data Analysis
The primary outcome variable was mean finger temperature; secondary outcomes included minimum and maximum finger temperature and number of cycles between relative minima and maxima mean finger temps. Raw temperature time series data were smoothed using locally weighted polynomial regression (LOESS). Example plot provided as Figure 1. Local maxima and minima were then identified algorithmically from the smoothed series. Temperature cycles in the smoothed time series were identified by matching maxima with nearest neighbor minima at least 1 °C colder. Where maxima-minima pairs overlapped, the maxima and minima with the highest and lowest temperatures, respectively, were taken. The primary outcome of mean finger temperature was calculated from pooled data across 4 fingers after the initial minima was reached for each finger. Similarly, data for mean maxima and minima temperatures and the number of cycles were pooled across all fingers. Outcomes were examined via a paired t-test and then reassessed using linear mixed-effects models to account for possible data dependence and reduce type I error. The paired nature of the outcome measures was treated as a random effect in the mixed models to account for the within-subject correlation of outcomes. All tests were one-tailed, reflecting the directional hypothesis that inhaled epoprostenol would increase finger temperature compared to placebo, and statistical significance was set at α = 0.05. Data analyses were performed using R version 4.3.1.
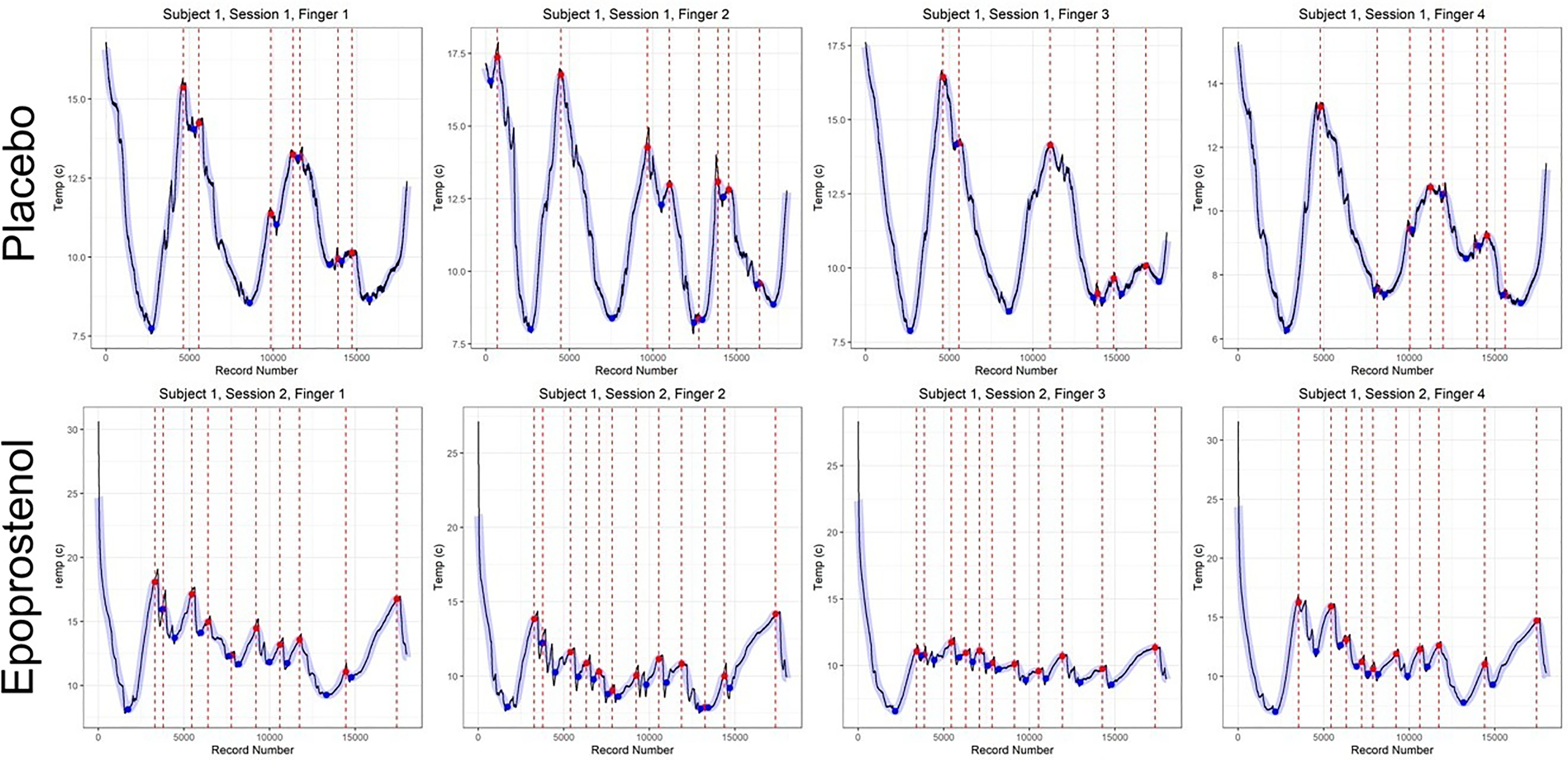
Representative Example of Finger Temperature Plot. Example Finger Temperature Time Series, Demonstrating LOESS Smoothing of Data as Well as Maxima, Minima, and Cycles Identification.
Results
Fifteen subjects completed both sessions of the study; however, one subject had an incomplete recording due to a loose thermocouple wire and was excluded from all data analysis. The mean age of the subjects was 34.2 ± 8.6 years. Nine of 14 subjects (64.3%) were females. No hypotension, hypothermia, notable or sustained tachycardia, or hypoxia was observed, and no subject requested discontinuation due to side effects. The most reported side effect was a mild headache. There was no reported loss of blinding.
Epoprostenol sessions were characterized by higher mean finger temperature (9.16 vs 8.34° C; p = .027) when analyzed with a paired sample t-test. Similarly, secondary outcomes of mean maxima temperature (10.86 vs 9.88° C; p = .045) and mean minima temperature (7.45 vs 6.80° C; p = .024) were similarly higher during epoprostenol sessions than placebo sessions. No significant difference was detected in the number of cycles (10.0 vs 7.93; p = .104) (Table 1).
Comparisons of Primary and Secondary Outcomes Between Epoprostenol and Placebo: One-Tailed Paired Sample t Test (n = 14).

The results from the fitted linear mixed-effects models were similar to those from the paired sample t-test. The beta coefficient (b) for the mean finger temperature was 0.81 (95% CI: 0.01–1.61; p = .025) (Table 2). The b coefficients for mean maximum temperature and mean minimum temperature were also statistically significant at 0.97 (95% CI 0.03–1.93, p = .045) and 0.66 (95% CI 0.01–1.28, p = .022), respectively.
Comparisons of Primary and Secondary Outcomes Between Epoprostenol and Placebo: Generalized Linear Mixed Models.

Discussion
This study found that inhaled nebulized epoprostenol can induce a statistically significant increase in mean temperature, mean maxima temperature, and mean minima temperature in fingers immersed in a cold water bath, consistent with an augmented CIVD response. Though the absolute difference in mean finger temperature is only 0.82° C, this does surpass the threshold used in other studies to determine a change in CIVD response.6,7 While the paired t-test could produce false positive results due to lack of independence of the data, the estimated coefficients for the fixed effects within the linear mixed effects model closely approximate the treatment differences obtained in the paired t-test. This implies that the mixed model results are consistent with those derived from the paired t-tests, suggesting that variability between subjects was minimal.
Though blood flow was not directly measured, these increased finger temperature measurements suggest that epoprostenol increased peripheral blood flow in the distal finger during cold exposure. In the seminal paper on the use of prostaglandins for frostbite, prostaglandin E1 (PGE1) infusion resulted in a significant treatment effect only if no rapid rewarming was performed. 8 While this could argue that the change in local tissue temperature secondary to vasodilation may have driven the entire observed effect in that animal study, the fact that Cauchy and Cheguillaume still found iloprost effective after all subjects received rapid rewarming would argue that mechanisms other than tissue temperature alone are involved.
This study demonstrates that an inhaled PGI2 analog can affect distal extremity physiologic response to cold stimulus as assessed by CIVD. Current guidelines recommend iloprost for the treatment of frostbite as prolonged IV infusions, but these findings suggest that nebulized PGI2 analogs may be a reasonable alternative treatment modality. Inhaled treatment options could allow earlier in-field treatment, particularly in situations such as high-altitude mountaineering, where oxygen tanks may be readily available. This study's findings should not alter current clinical practice, but they do suggest areas for further research, including the pragmatic use of epoprostenol when iloprost is not available for treating frostbite and a study of the effects of epoprostenol versus iloprost on CIVD. A robust CIVD response has been associated with increased cold tolerance and is thought to reduce the risk of frostbite; however, it remains unclear if a pharmacologic augmentation of CIVD might have clinical utility for the prevention of cold injury, as CIVD only occurs in euthermic subjects. Further study could assess if certain populations, such as people with Raynaud's syndrome, might benefit from prophylactic PGI2 use.
Limitations
This is a small hypothesis-generating pilot study with a short duration of drug exposure and is not powered to detect harm. While the study drug is pharmacologically related to an established frostbite treatment, the size of the study and the use of healthy subjects preclude any conclusions regarding the treatment or prevention of frostbite. In comparison to this 30-min drug exposure, Cauchy et al used 6 h infusions of iloprost in their controlled trial while Yeager et al only saw treatment effect with a 3 h infusion and not with a bolus of prostaglandin. 9 While the temperature plots visibly demonstrate different morphologies of CIVD response between epoprostenol and placebo sessions, it is unclear from this study by what mechanism epoprostenol is creating this effect and what effect epoprostenol might have on finger temperature without cold exposure.
As described in previous studies, CIVD response can be impacted by several variables, including ambient temperatures, time of day, posture, body heat content, recent exercise, high-altitude hypoxia, and food intake. Participants signed consent that they did not meet exclusion criteria or participate in known confounding activities, but these risk factors and activities or other unknown confounders could have altered the observed CIVD effect. Finally, there is significant heterogeneity of chosen outcome measures in the CIVD literature; so, while mean finger temperature has been commonly utilized in previous studies, it is quite plausible that another measure might better assess this physiologic response.
Conclusion
This study found that inhaled nebulized epoprostenol can induce a statistically significant increase in mean temperature, mean maxima temperature, and mean minima temperature in fingers immersed in a cold water bath, consistent with an augmented CIVD response. The ability to deliver epoprostenol via nebulizer and at doses that affect distal extremity circulation raises the possibility of early intervention with inhaled PGI2 analogs for the management of frostbite in remote and austere environments. The exact mechanism by which prostaglandins affect frostbite remains unknown, and further studies are needed to determine if epoprostenol can play a role in the treatment or prevention of frostbite.
Footnotes
Author Contribution(s)
Funding
Research was performed with funding from the WMS Peter Hackett-Paul Auerbach Young Investigator Grant provided by the Wilderness Medical Society and the Academy of Wilderness Medicine.
Declaration of Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
