Abstract
Background
Frostbite injury occurs when exposure to cold results in frozen tissue. We recently reported a novel mouse model for frostbite injury to be used in screening potentially therapeutic drugs and other modalities.
Objective
We used the mouse skin frostbite model to evaluate the effect of poly-
Methods
Sixty mice were studied in a randomized, double-blind method. Standardized 2.9-cm-diameter circles were tattooed on the mouse dorsum. Magnets snap frozen in dry ice (–78.5
Results
Application of PAL before inducing frostbite injury resulted in decreased tissue loss as compared with other treatment conditions.
Conclusions
Applying PAL topically to frostbitten mouse skin caused decreased tissue loss. Poly-
Introduction
We recently invented a novel mouse skin model of induced frostbite injury, with the intention of using the model to allow quantification of the surface area of involved skin, histology of the wound, rate of wound healing, and skin loss in a standardized fashion after frostbite injury.
1
One purpose for this model is to facilitate efforts to establish or refute efficacy of preventive techniques or therapeutic interventions in a screening manner. To this end, we selected a compound containing poly-
Nitric oxide synthase (NOS) is a group of enzymes that helps catalyze production of nitric oxide (NO) from
Poly-
Methods
Frostbite Model
The method of Auerbach et al 1 was used and is briefly recapitulated here. Sixty 8- to 12-week-old C57Bl/6J male mice (Jackson Laboratories, Bar Harbor, ME) were used in a study approved by the Stanford Institutional Review Board and in accordance with Stanford University Institutional Animal Care and Use Committee Guidelines. The animals were housed 5 per cage before and after surgery in a temperature-controlled facility under a 12-hour light/dark cycle.
Skin preparation
Twenty-four hours before day 0, mice were individually anesthetized using isoflurane (Baxter, Deerfield, IL). The dorsal skin surface from the base of the neck to the top of the rear haunches was shaved with an electric clipper, after which a depilatory cream (Nair, Church & Dwight, Princeton, NJ) was applied for 2 minutes to remove any remaining hair. After depilation, the skin was cleansed with a 70% isopropyl alcohol swab. A circle of uniform diameter 2.9 cm was gently traced on the skin with a permanent marker using a silicone sheet template (Invitrogen, Grand Island, NY). The marked circle was then permanently darkened by using an animal tattoo machine (Animal Identification and Marking Systems Inc, Hornell, NY).
Freeze method
Twenty-four hours after skin preparation, ceramic (ferrite) magnets (diameter 0.5 inches, thickness 0.219 inches, weight 3.5 g) were placed in crushed dry ice (–78.5° C) and allowed to freeze for 15 minutes. Using fingers, the back skin of the mouse was lifted into a skin fold and then 2 frozen magnets were placed so that they adhered from opposite sides of the intervening skin fold. Sets of 2 cooled magnets were left in place for 1 minute, then removed to allow new magnets to be immediately placed in the same location against the frozen tissue. The magnet exchange was repeated for a total of 5 applications, resulting in a freeze time slightly longer than 5 minutes. After 5 magnet placements and removals, the skin was allowed to completely thaw. Mice were given subcutaneous injections of buprenorphine (0.05 mg/kg) for analgesia after the thaw. No dressings were applied to the wounds.
Application of poly-l -arginine suspended in AmLactin lotion
One hundred micromolar poly-
Wound Surface Analysis and Tissue Loss Quantification
Digital photographs of the wounds were taken after the thaw on day 1 and every other day thereafter until day 27, at which time the wounds were completely healed by visual inspection by an observer blinded to the treatment conditions. Wound surface area was determined by tracing the wound margin displayed by photograph with a fine-resolution computer mouse. The number of pixels corresponding to a surface area measurement was calculated using ImageJ software (National Institutes of Health, Bethesda, MD). Wound area was reported as percent area of the original maximum frostbite injury ([original wound area – wound area on day “x”] / original wound area). Tissue loss and contraction was reported as area of the original maximum skin area inside the tattooed circle ([original surface area within the tattooed boundary – surface area within the tattooed boundary on day “x”] / original surface area within the tattooed boundary).
Histopathology Examination
The animals were humanely killed, and the entirety of the frostbite wounds were harvested from 2 mice per group on each of days 3, 7, 14, 21, and 27. The tissue samples were fixed overnight in 4% paraformaldehyde, and then embedded in paraffin. Sections (7 µm) were cut and stained with hematoxylin and eosin. Analysis was performed by a board-certified dermatopathologist. Tissue necrosis, reepithelialization, healing rate, acute inflammation, chronic inflammation, fibroblast maturation, collagen deposition, neovascularization, and the overall healing score were analyzed based on a modified version of a scoring system suggested by Abramov et al. 16 All measurements were recorded on a scale of 0 to 3, except for the overall healing score.
Statistical Analysis
Surface area of the wound, tattooed region, and aforementioned silicone stencil were measured in pixels and presented as a mean ± SD. Statistical significance was accepted at P < .05. Statistical analyses were performed using SPSS (Version 17.0; SPSS Inc, Chicago, IL).
Results
Wound Closure and Analysis
The placement of 2 frozen magnets on opposite sides of the dorsal fold resulted in a single frostbite injury spanning both circles and the intervening tissue. The skin between the magnets, as well as a small portion of the surrounding area, was affected by the cold. This single frostbite injury was subsequently monitored (Figure 1).
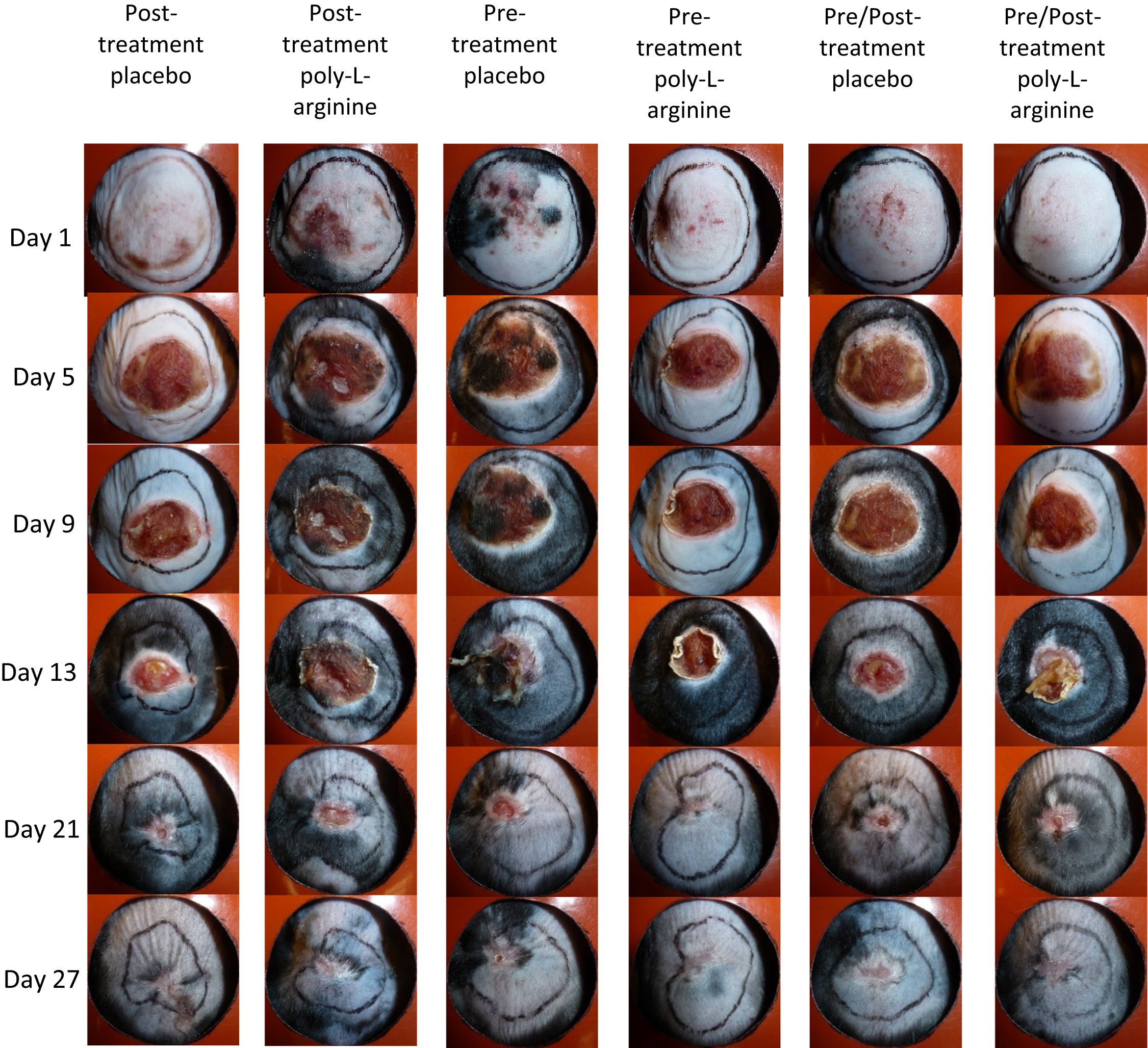
Gross appearance of wound progression at various times after induced frostbite injury under different treatment conditions. Variations among treatments can be seen at intermediate times, although all wounds were completely healed by day 27.
Wound healing
Mice pretreated with placebo AmLactin were completely healed by day 21. Mice pretreated with PAL were completely healed by day 25. Mice posttreated with placebo AmLactin were completely healed by day 17. Mice posttreated with PAL were completely healed by day 26. Mice both pretreated and posttreated with placebo AmLactin were completely healed by day 27. Mice both pretreated and posttreated with PAL were completely healed by day 21. The mice left untreated (control) were completely healed by day 27 (Figure 2).
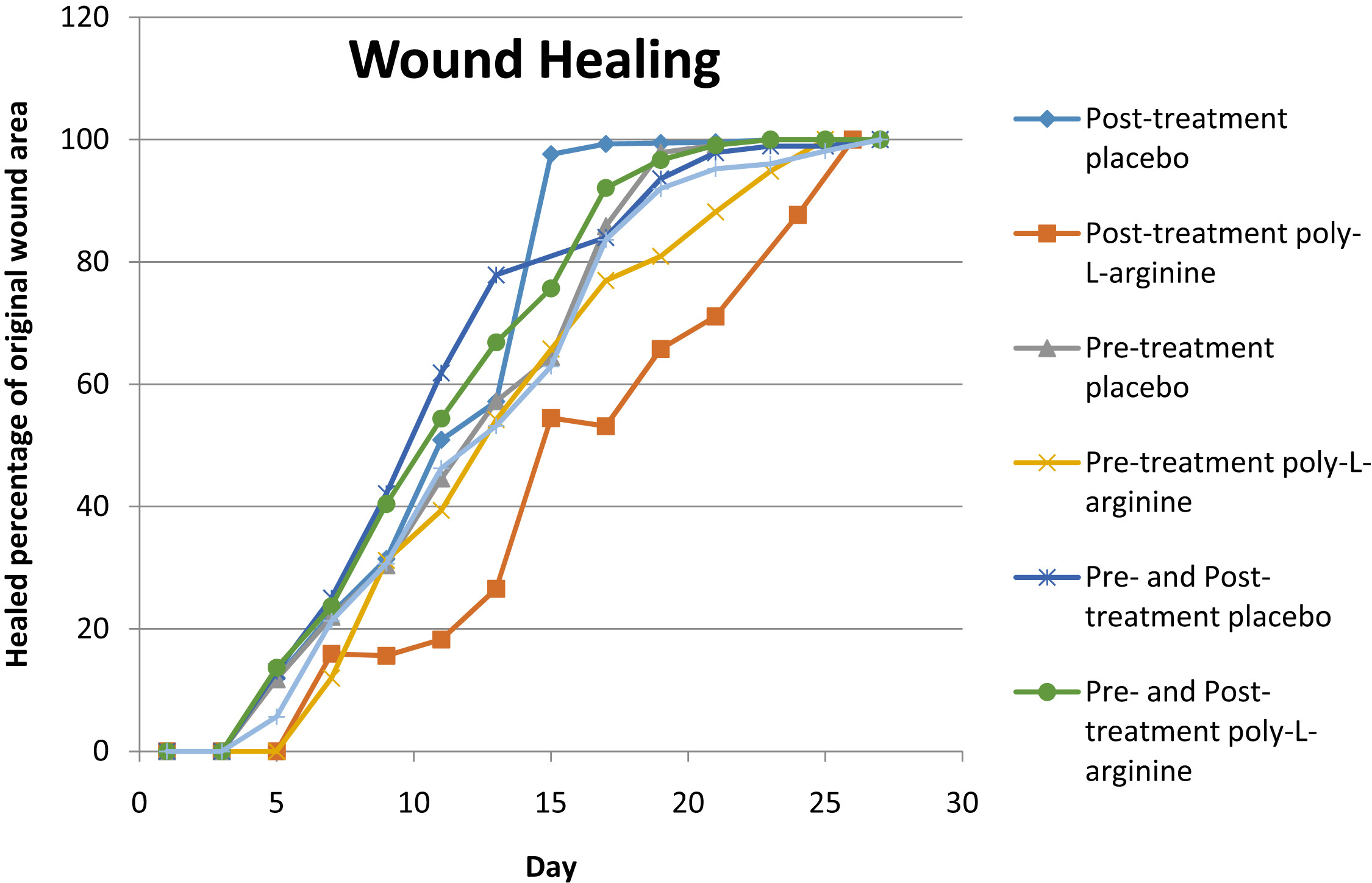
Wound healing showing the percentage of original wound area healed by day. Each data point used to create a curve is the mean healed percentage (confidence interval = 97%) of the original wound area for all mice treated for that condition.
Tissue loss
Tissue loss is the amount of skin area within the bounded area of the tattoo that disappears during the healing process. Mice pretreated with placebo AmLactin lost a mean of 43% of the original skin surface area bounded by the tattoo. Mice pretreated with PAL lost a mean of 39% of the original tattooed skin surface area bounded by the tattoo. Mice posttreated with placebo AmLactin lost a mean of 56% of the original skin surface area bounded by the tattoo. Mice posttreated with PAL lost a mean of 56% of the original tattooed skin surface area bounded by the tattoo. Mice pretreated and posttreated with placebo AmLactin lost a mean of 43% of the original skin surface area bounded by the tattoo. Mice pretreated and posttreated with PAL lost a mean of 37% of the original skin surface area bounded by the tattoo. Mice left untreated (control) lost a mean of 46% of the original skin surface area bounded by the tattoo (Figure 3).
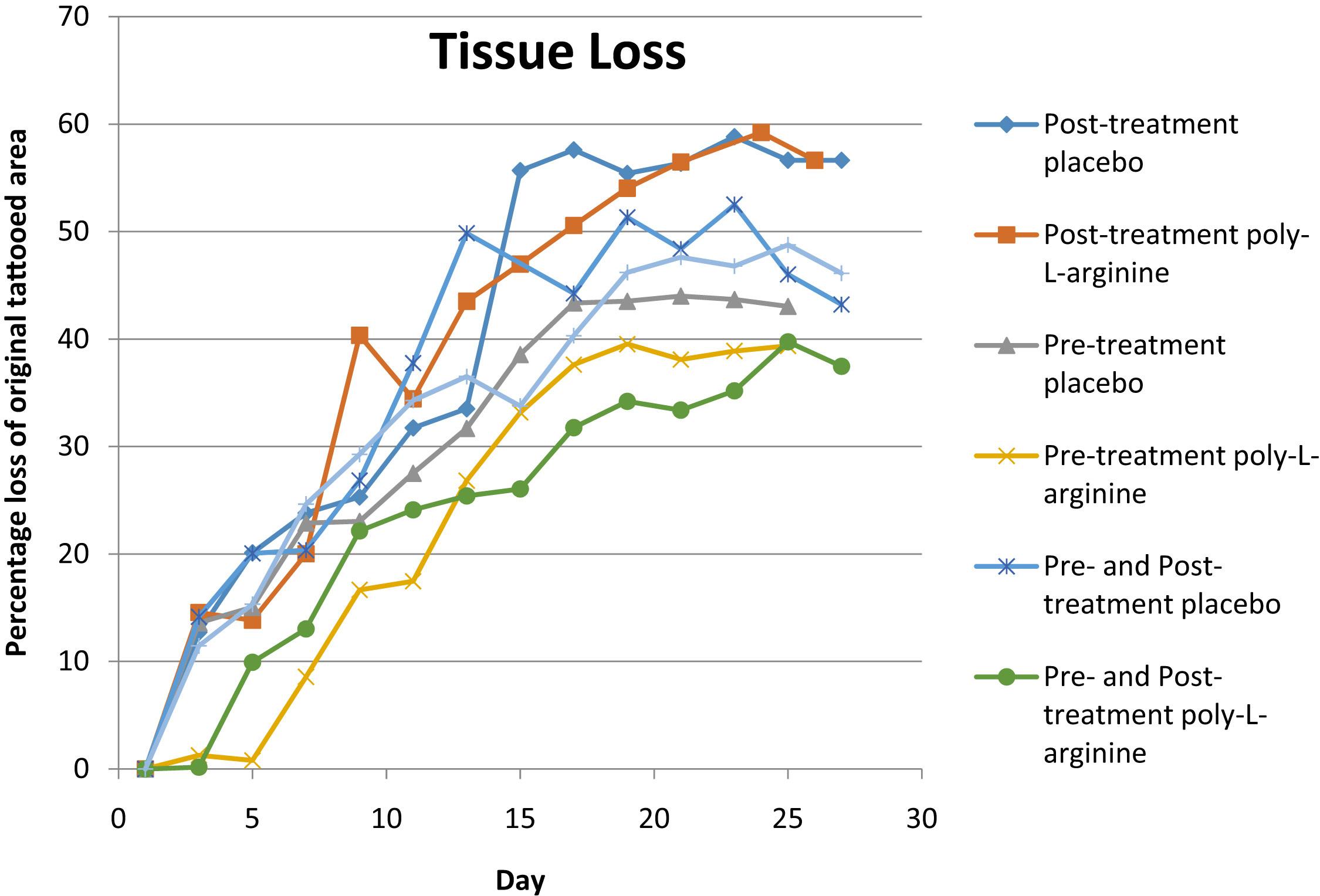
Percentage surface area of tissue lost within the tattooed boundary compared with the original surface area within the boundary. Mice pretreated with poly-
Histology
Histological analysis revealed no significant trend among the treatment conditions (online Supplementary Figure).
Discussion
One goal of establishing our mouse frostbite model was to allow the model to be used for evaluation of pharmacological and other approaches to frostbite prevention and treatment. We used the model to evaluate the effect of topical PAL on a controlled frostbite injury.
Regardless of treatment applied, all wounds were completely healed by day 27. However, certain treatments resulted in shorter healing times than others. The treatments involving poly-
Our most encouraging finding was that mice that received poly-
Histologically, no themes were observed among the treatment conditions. This could be owing to the small sample sizes (n = 2) taken at each time. An increased sample size centered around approximately days 7 to 14 might show changes in the degree or rate of chronic inflammation, amount of granulation tissue, and neovascularization. 1
As previously, we note certain limitations with the mouse frostbite model.
1
Notably, there is not a blistering phase in our model. We did not study a deep tissue or comprehensive limb injury. Mice are not humans, and we do not expect to find absolute comparability in clinical or pathologic presentation. For these and other reasons, it may be necessary to advance PAL to another animal model, such as rabbit limb (which demonstrates deep tissue loss), before undertaking human investigation. However, to provide a counterpoint, given the known human safety profile of poly-
Conclusions
We describe application of a poly-
Footnotes
Acknowledgments
We thank Ms Arnetha Whitmore and Ms Yujin Park for help with experimental procedures. Supported by the National Institute of Diabetes and Digestive and Kidney Diseases (RO1-DK-074095 to GCG), the Howard Hughes Medical Institute Medical Research Fellowship (MGG), and the Division of Emergency Medicine at Stanford.
Supplementary data
Supplementary data associated with this article can be found in the online version at
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
