Abstract
Hand warmer packets are common products used to provide a portable, nonflammable heat source via the exothermic oxidation of iron. We present the first reported case of pediatric hand warmer packet ingestion in a three-year-old male who developed an elevated serum iron concentration (peak 335 ug/dL) and gastrointestinal injury after ingesting the contents of a HOTHANDS hand warmer packet. He was treated with endoscopic gastric foreign body removal and lavage, as well as proton-pump inhibitors and whole bowel irrigation. Hand warmer packs contain reduced elemental iron powder, which has been shown to have a more favorable safety profile when compared to iron salts. The mechanism of toxicity for reduced iron is unknown, though it is thought to be due to conversion to more toxic iron ions in an acidic environment. While the current adult literature suggests that ingestion of a single hand warmer packet is without significant risk, our case demonstrates that even a partial ingestion carries a significant risk of both iron toxicity and direct gastrointestinal caustic injury in a young child. This case demonstrates the need for multidisciplinary care and consideration of urgent endoscopic foreign body removal and gastric lavage followed by whole bowel irrigation to mitigate the potential of severe iron toxicity.
Introduction
Hand warmer packets are common over-the-counter products used to provide a portable, nonflammable heat source. Heat is generated via an exothermic reaction involving the oxidation of iron filings. Ingestion of the contents of hand warmer packets is rarely reported in adult literature and almost universally occurs in geriatric patients mistaking them for food products such as instant coffee or hot cocoa.1–5 To our knowledge, no pediatric cases have been reported and little is known about the potential toxic effects in the pediatric population.
We present the case of a child who developed an elevated serum iron concentration and gastrointestinal injury following ingestion of the contents of a hand warmer packet.
Case Report
A 3-year-old male with a past medical history of nonverbal autism spectrum disorder presented to a major urban pediatric hospital after being found by his parents with the contents of a HOTHANDS hand warmer packet in his mouth and on his hands and clothes. According to the parents, approximately 50% of the packet contents were missing. The parents washed his face and the inside of his mouth with water prior to arrival.
His initial vital signs were within normal limits for his age. Examinations performed by the emergency department and medical toxicology physicians revealed a well-appearing, nontoxic child without abdominal tenderness. There was no report of abdominal pain or vomiting. The initial serum iron concentration, obtained 4 hours post-ingestion, was 335 ug/dL (normal range 50–120 ug/dL). Serum creatinine was elevated to 0.71 mg/dL (normal range for age 0.20–0.43 mg/dL). The remainder of his laboratory evaluation, including complete blood count and comprehensive metabolic panel, was within normal limits. An abdominal radiograph demonstrated radiopaque material projecting over the body of the stomach (Figure 1).
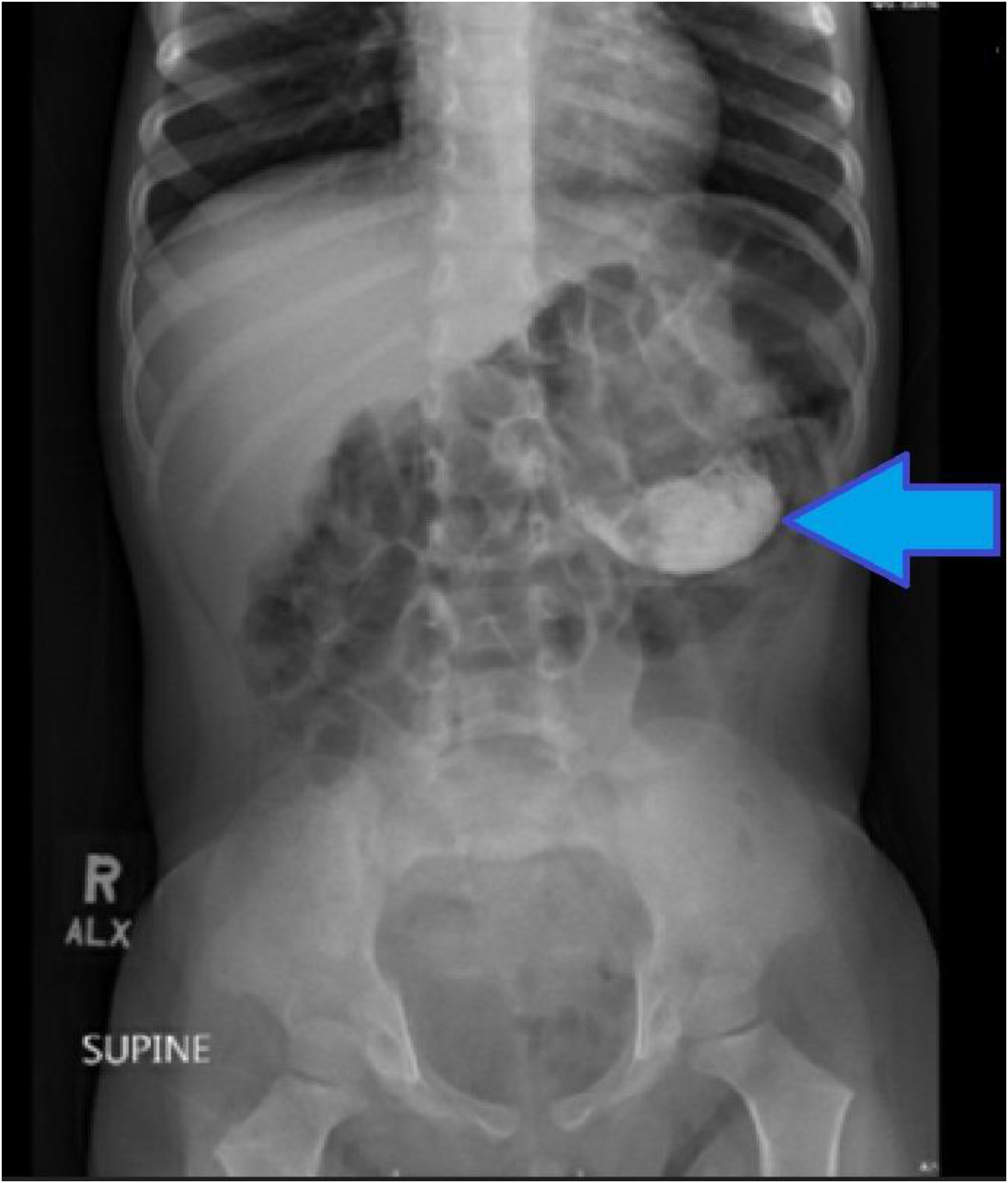
Abdominal radiograph with arrow indicating radiopaque materials projecting over the body of the stomach.
Given the elevated serum iron concentration and radiopaque material in the stomach, pediatric gastroenterology was consulted for urgent endoscopic-assisted gastric foreign body removal and lavage. Esophagogastroduodenoscopy (EGD) performed 5.5 hours postingestion demonstrated a Zargar grade IIA caustic injury in the esophagus and stomach, as well as black adherent powder in the gastric antrum (Table 1). The black powder was noted to be strongly adherent to the mucosa and underlying mucosal ulcerations were noted (Figure 2). Removal of approximately 95% of the visualized material from the stomach and duodenum required approximately 5 liters of high-flow direct irrigation and intraprocedure patient repositioning. Following the procedure, the patient received intravenous pantoprazole, famotidine, and whole bowel irrigation with polyethylene glycol via nasogastric tube.
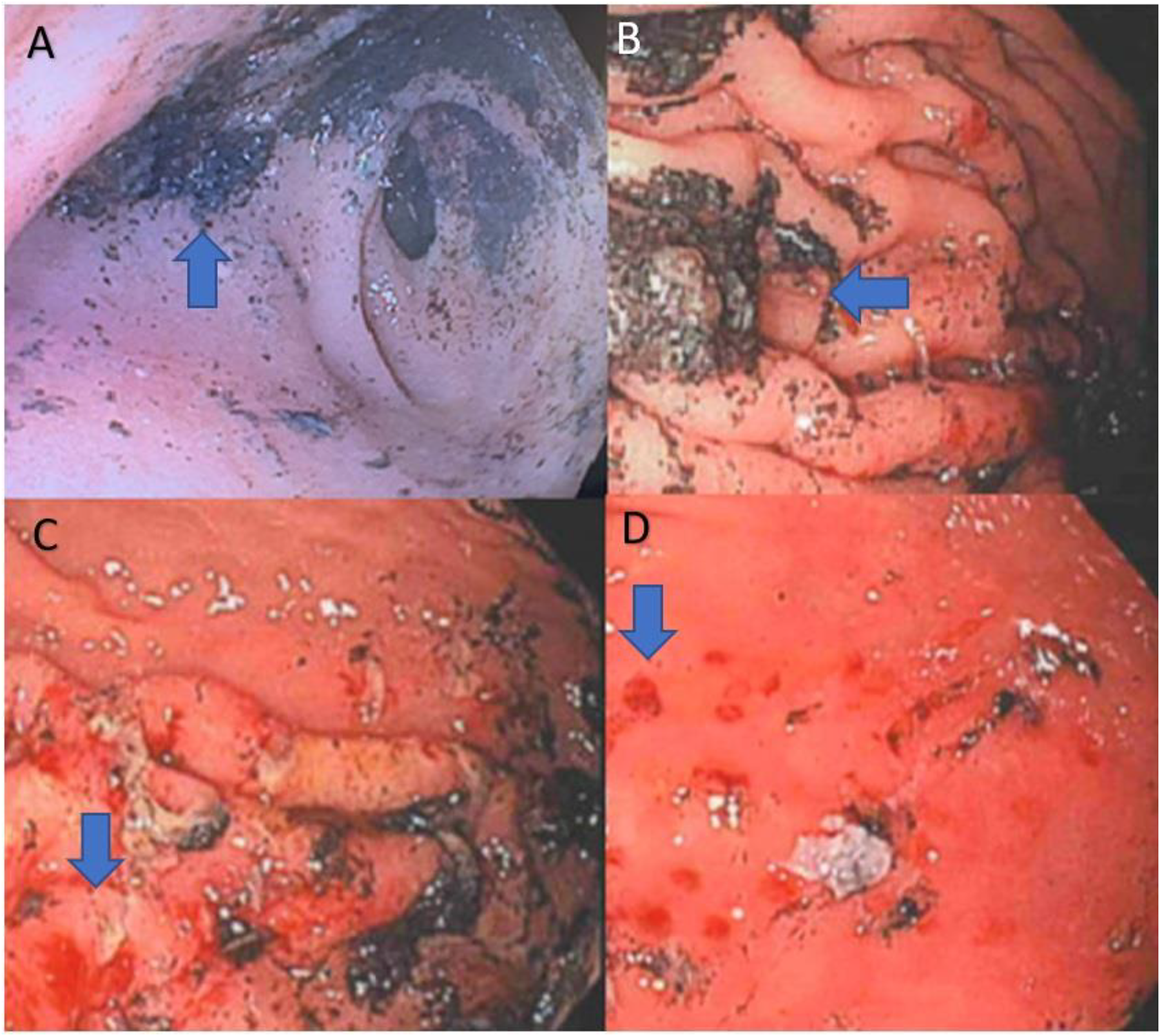
EGD with arrows demonstrating iron powder adherent to the gastric mucosa (A + B) and gastric ulcerations (C + D).
Zargar classification of corrosive gastrointestinal injury.

After intervention, serum iron concentrations fell rapidly—to 213 ug/dL 10 h postingestion and 73 ug/dL 14 h postingestion. The patient remained asymptomatic and chelation was not performed as the patient was hemodynamically stable with normal pH and decreasing serum iron concentrations in the setting of source control. Repeat abdominal radiographs performed at 24- and 48-h postingestion demonstrated trace residual radiopaque material in the left upper quadrant and resolution respectively. Whole bowel irrigation was discontinued after 48 h when the patient had clear rectal effluent and resolution of radiopaque material on abdominal radiography. His serum iron concentration continued to decrease and was 8 ug/dL on the morning of discharge on hospital Day 5. The patient's hospital course was complicated by asymptomatic hyponatremia to 123 mEq/L and anemia to a nadir hemoglobin of 7.8 g/dL. The cause of his anemia was likely multifactorial due to systemic and local gastric inflammation, gastrointestinal bleeding, hemodilution, and iatrogenic blood loss. The sodium normalized with the administration of sodium bicarbonate. No blood product transfusions were required.
The patient's gastric lavage fluid, approximately 5 liters in total, was analyzed using the Ferrozine method by the analytical toxicology laboratory at UC San Diego, and 367 mg of soluble iron was recovered.
The patient was seen in the gastroenterology clinic 2 mo after discharge. At that time, it was reported that he had resolution of his dark stools 2 d after discharge and was otherwise asymptomatic without clinical evidence of gastrointestinal sequelae.
Discussion
HOTHANDS hand warmers are composed of 50–60% reduced elemental iron powder, 30–40% water, 5–10% activated charcoal, 5–10% vermiculite, 1–5% water-absorbent resin, and 1–5% sodium chloride according to their material safety data sheet; the exact concentrations are unavailable because they are a trade secret. 6 Once removed from its vacuum seal, the elemental iron oxidizes in air using salt as a catalyst in an exothermic reaction. The charcoal and vermiculite evenly disperse and retain heat, prolonging the duration of action. 5
The hand warmer ingested in this case had a stated weight of 59 g and potentially contained as much as 35 g of elemental iron. As this patient weighed 15 kg and ingested roughly half the packet, he potentially ingested 2333 mg/kg of elemental iron. The noniron ingredients are nontoxic, though they may have contributed to the stronger-than-expected adherence of the iron powder to the gastrointestinal mucosa.
Reduced iron is created from reduction of iron oxide with hydrogen. 6 Animal studies have compared the toxicity of reduced iron to iron salts and found it to have a more favorable safety profile: the median lethal oral dose in rats is approximately 10% of their body weight, or 100 times the median lethal oral dose of ferrous sulfate. 7 The human median lethal oral dose has not been determined. The mechanism of toxicity for reduced iron is unknown, though it is thought to be due to conversion to free iron ions in an acidic environment. Reaction with hydrochloric acid is hypothesized to form more readily absorbable iron chloride salts.2,7 For example, ferric chloride is comparatively much more dangerous, with one study showing a fatality rate of approximately 6% after ingestion. 8 It has not been established how efficiently reduced iron is absorbed or converted and at what threshold dose reduced iron becomes clinically dangerous. However, in this case, it appears that less than a single packet contained enough iron to potentially cause harm in a young child.
The current adult literature suggests that ingestion of a single hand warmer is without significant risk. 5 However, even partial ingestion in a young child appears to carry a significant risk of both iron toxicity and direct caustic injury to the gastrointestinal mucosa. This is consistent with the only other reported case of pediatric iron oxide ingestion—a child who swallowed a single hematite stone. In that case, the child also had a rapid rise in serum iron concentrations that prompted urgent endoscopic removal. 9 We chose to aggressively manage this patient with whole bowel irrigation and endoscopic gastric lavage due to a paucity of literature on this topic, the high initial serum iron concentration, and the large amount of iron seen on the initial abdominal radiograph. We suspect that that the serum iron concentration would have continued to rise to a potentially life-threatening concentration if not for prompt aggressive intervention and source control. 3
One perplexing element of the patient's presentation is the lack of gastrointestinal symptoms on presentation. Iron poisoning is classically associated with substantial gastrointestinal disturbances, with the “first phase” of iron poisoning characterized by nausea, vomiting, diarrhea, and abdominal pain. 10 Possible explanations include the limitation of gastrointestinal irritation by other substances in the packet or that the mechanism of irritation differs with reduced iron compared with more typical iron salts. Additionally, the patient's age and nonverbal status may have precluded display or recognition of abdominal pain.
Management of ingestion of iron from hand warmer packets may require additional considerations compared to iron salts. First, gastric irrigation with water is a potential treatment, as water terminates the exothermic oxidative reaction and can theoretically decrease thermal injury. 11 Activated charcoal is not effective in these cases, as it does not adsorb iron. Second, the use of acid-reducing medications, such as histamine H2 receptor antagonists and proton pump inhibitors, may decrease the release of free iron ions and formation of iron chloride salts. 3 Third, evaluation for radiopaque material with an abdominal radiograph can assist with confirming and locating the ingested iron. If contained in the proximal bowel, as in this case, consideration of endoscopic-assisted foreign body removal and gastric lavage followed by whole bowel irrigation can be considered, even if the patient is otherwise asymptomatic. Iatrogenic hyponatremia and procedural complications should be considered as risks and weighed against potential benefits. Despite these risks, we favored aggressive treatment with gastrointestinal decontamination via urgent endoscopic gastric lavage and whole bowel irrigation to rapidly reduce serum iron concentrations and avoid potentially dangerous iron toxicity. In cases where gastric lavage is not available, treatment with water by mouth or enteral tube should be performed. Additionally, as with any other iron exposure, chelation should be considered in the appropriate clinical context.
Conclusions
Pediatric ingestion of iron-containing hand warmers is rare but likely more serious than a similar adult ingestion, with the potential for clinically significant iron toxicity and gastrointestinal caustic injury. Aggressive gastrointestinal decontamination may be beneficial for patients with retained gastrointestinal iron. Further research is necessary to determine the best treatment strategies for children.
Footnotes
Author Contribution(s)
Conceptualized the case report, performed the literature review, drafted the initial manuscript, and critically reviewed and revised the manuscript (JH, JS); created the figures, obtained gastric lavage fluid, and reviewed and revised the manuscript (ES); performed laboratory analysis of the gastric lavage fluid including iron quantification, contributed to the drafting of the initial manuscript, and critically reviewed and revised the manuscript (RS); coordinated and supervised patient data collection, revised the manuscript for important intellectual content, critically reviewed the manuscript, and provided general oversight (MS, DL).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
