Abstract
For individuals who work outdoors in the winter or play winter sports, chemical hand warmers are becoming increasingly more commonplace because of their convenience and effectiveness. A 32-year-old woman with a history of chronic pain and bipolar disorder presented to the emergency department complaining of a “warm sensation” in her mouth and epigastrium after reportedly ingesting the partial contents of a chemical hand warmer packet containing between 5 and 8 g of elemental iron. She had been complaining of abdominal pain for approximately 1 month and was prescribed unknown antibiotics the previous day. The patient denied ingestion of any other product or medication other than what was prescribed. A serum iron level obtained approximately 6 hours after ingestion measured 235 micrograms/dL (reference range 40–180 micrograms/dL). As the patient demonstrated no new abdominal complaints and no evidence of systemic iron toxicity, she was discharged uneventfully after education. However, the potential for significant iron toxicity exists depending on the extent of exposure to this or similar products. Treatment for severe iron toxicity may include fluid resuscitation, whole bowel irrigation, and iron chelation therapy with deferoxamine. Physicians should become aware of the toxicity associated with ingestion of commercially available hand warmers. Consultation with a medical toxicologist is recommended.
Introduction
For individuals who work outdoors in the winter or play winter sports, chemical hand warmers are becoming increasingly more commonplace because of their convenience and effectiveness. An unusual case of a patient who intentionally ingested a piece of a chemical hand warmer that contained between 5 and 8 g of elemental iron is presented.
Case Report
A 32-year-old woman with a history of chronic pain and bipolar disorder who had been prescribed lamotrigine, gabapentin, paroxetine, alprazolam, amphetamine/dextroamphetamine and lorazepam presented to the emergency department (ED). She reported she had a “warm sensation” in her mouth and epigastrium after ingesting the partial contents of a chemical hand warmer the evening prior. The patient recalled eating a solid mass, which she mistook for a granola bar. She also had been complaining of abdominal pain for approximately 1 month and had been prescribed an unknown antibiotic the previous day. The patient denied nausea, vomiting, diarrhea, or ingestion of any product or medication other than what was prescribed.
The patient’s vital signs included: blood pressure, 124/87 mm Hg; heart rate, 67 beats/min; respiratory rate, 19 respirations/min; 98% room air pulse oximetry; and temperature, 37.3°C (99.1°F). Physical examination was remarkable only for a black granular substance in her teeth and mild left lower quadrant tenderness. The material safety data sheet (MSDS) available on the Internet listed the chemical hand warmer’s contents as polymer, iron powder, activated charcoal, water, vermiculite, and salt. After consultation with a medical toxicologist, a single serum iron level was obtained approximately 6 hours after ingestion and measured 235 micrograms/dL (reference range 40–180 micrograms/dL). A complete blood count, metabolic panel, urinalysis, and urine pregnancy test were unremarkable. Serum salicylate, acetaminophen, and ethanol were undetectable.
An abdominal radiograph was performed to try to confirm exposure; it did reveal scattered radiopaque material mixed with stool in the lower right abdomen, consistent with the presence of metal fragments (see Figure 1). Because the patient demonstrated no new abdominal complaints or evidence of systemic iron toxicity, neither a subsequent iron concentration nor treatment was indicated. The patient was educated regarding the potential toxicity of the product and discharged uneventfully.
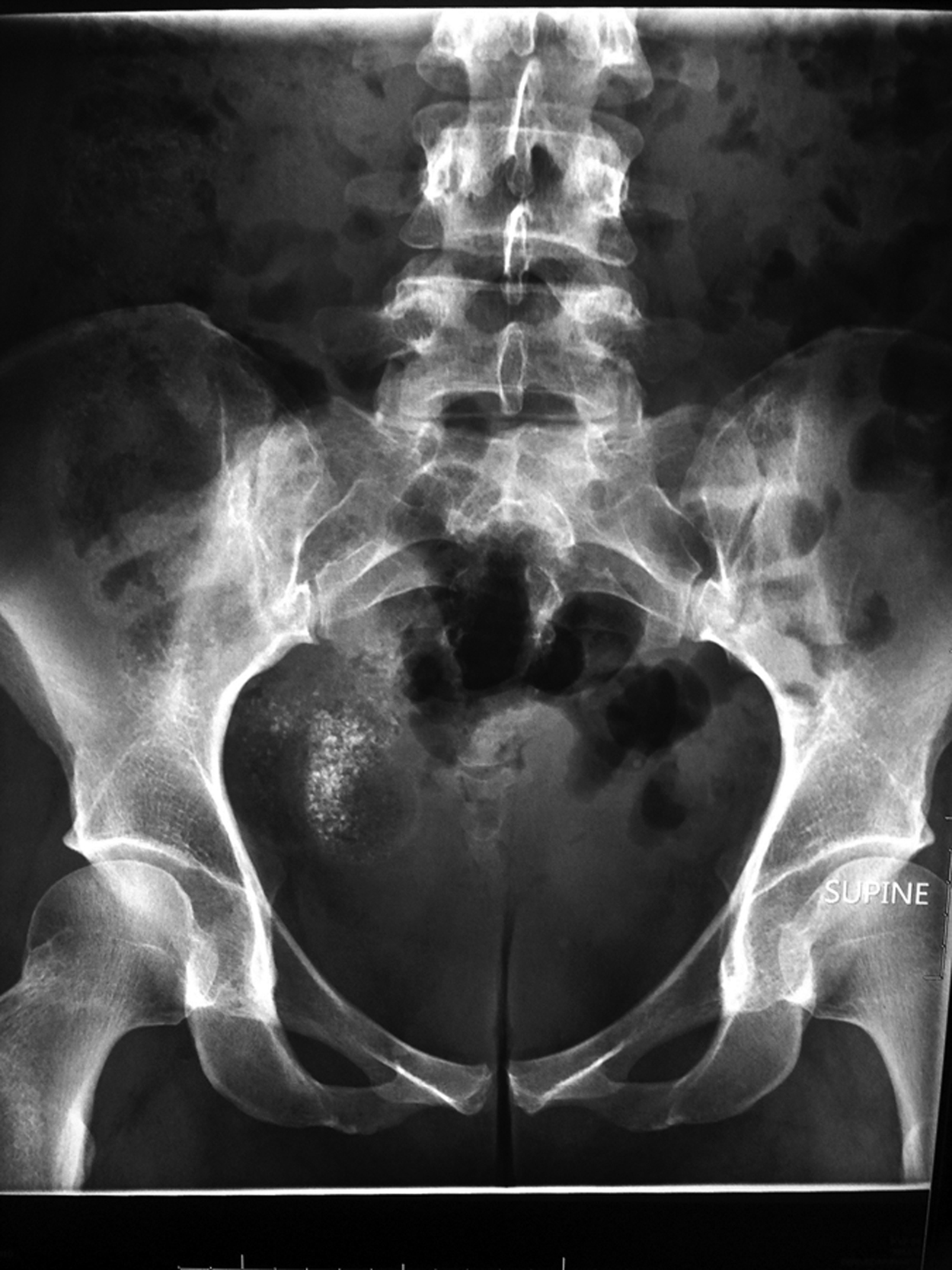
Abdominal X-ray of a patient after reported ingestion of a chemical hand warmer.
Discussion
Ingestion of hand warmer products is uncommon, but has been described in elderly patients suffering from dementia. In these cases most were asymptomatic, despite ingestion of a potentially toxic amount of elemental iron. 1 The patient in this case report suffered from chronic abdominal pain unrelated to the exposure and did not demonstrate any clinical or laboratory evidence of elemental iron toxicity.
Among reviewed case reports, 1 patient exposed to a chemical hand warmer demonstrated severe vomiting and gastritis, as evidenced by endoscopy. 2 However, multiple other case reports describe patients with similar ingestions—some resulting in serum iron concentrations of >200 micrograms/dL, as well as visible radiopacities in abdominal radiographs—who remained entirely asymptomatic. 1
Although the composition of hand warmers differs between manufacturers, the authors noted the following ingredients were common among all products: iron powder, activated charcoal, and salt.1,2 One local distributor’s report includes specific amounts of each ingredient, for example, iron powder, 50%; purified water, 25%; activated carbon, 20%; vermiculite, 3%; and sodium chloride, 2%, with product weights ranging from 95 to 120 g. 1 This suggests a packet of hand warmer may contain as much as 60 g of iron powder.
Hand warmers work by means of an exothermic reaction, the same one that creates rust. With salt and water present, when the warmer is removed from its protective packaging, oxygen immediately interacts with the iron powder through its air-permeable pouch to form iron oxide (Fe2O3) and release heat. 3 Warmers also contain an absorbent material, such as pulverized wood, a polymer such as polyacrylate, or a silicon-based mineral called vermiculite that helps retain moisture in order for the chemical reaction to occur. The activated carbon helps to evenly disperse the heat produced, which can average 57.2°C (135°F). 3 Some manufacturers adjust the amount of iron powder in their formulations to affect the length of time the hand warmer stays hot; the more iron power, the longer the product will stay warm. 3
Unfortunately, the exact amount of product ingested by our patient could not be determined, as she brought in only pieces and crumbs of the original substance. Chemical hand warmers are commonly distributed in tear-open foil packaging, which can be mistaken for food and are easily accessible. Per the manufacturer, this particular warmer is maintained in powder form, becomes solid only after product activation with exposure to air, and contains between 5 and 8 g of elemental iron of unknown type. It is unknown whether ingestion of a potentially air-exposed version would have a different clinical manifestation versus the powdered product.
Ingestions of 20 mg/kg of elemental iron may produce gastrointestinal symptoms, and ingestion of >60 mg/kg may cause serious toxicity. Total serum iron levels >350 micrograms/dL obtained 2 to 6 hours postingestion correlate with the toxic range, and levels >500 micrograms/dL indicate risk of severe toxicity. Caution must be used in interpreting toxicity values as the serum level may not have reached its peak, or may be misleading due to intracellular iron shifts. 4 Certain clinical findings and laboratory data (including nausea, vomiting, diarrhea, abdominal pain, coma, leukocytosis, hyperglycemia, anion gap metabolic acidosis, and radiopacities on abdominal radiograph) may be helpful in determining toxicity.4,5 While some of these factors have been associated with serum iron concentrations of >200 micrograms/dL, no individual factor was found to be of statistical significance; they also fail to identify cases of toxicity of serum concentrations <500 micrograms/dL.6,7
Iron toxicity is caused by both its local caustic and systemic effects of the free, unbound form. Poisoning may progress through 4 phases, the first of which (0–6 hours) comprises gastrointestinal symptoms as described above. These symptoms may be severe enough to result in shock and death due to fluid loss. A second, latent (or quiescent), phase (6–12 hours) occurs with relative improvement. Severe ingestions may progress immediately to a third stage (12–48 hours), characterized by seizures, hepatic failure, coagulopathy, shock, metabolic acidosis, coma, and death. The fourth phase (4–6 weeks) includes recovery, but with risk of gastric scarring, including pyloric stenosis and intestinal obstructions. 4 ,5,8
Treatment for iron toxicity may include fluid resuscitation, whole bowel irrigation (WBI) and iron chelation therapy with deferoxamine. 4 ,5,9,10 WBI has been recommended in potentially toxic iron ingestions, particularly if large amounts of radiopaque tablets are revealed beyond the pylorus on radiograph, although clinical evidence of this practice’s efficacy is lacking. 4 ,5,9,10 Despite this, current recommendations state that WBI should be considered in substantial iron ingestions due to its significant morbidity, potential mortality, and the lack of other efficacious gastric decontamination methods.9,10 It should be noted that cited studies are referring specifically to ingestion of iron tablets; the results of decontamination may not be generalizable to unsealed iron powder, as was ingested in our case.
Iron chelation with deferoxamine may be indicated for ingestion exceeding 60 mg/kg, a serum iron concentration in the toxic range and clinical evidence of poisoning.4,5 Usually titrated until “vin rose” colored urine disappears, this endpoint of deferoxamine therapy may precede resolution of toxicity and is clinically of little use.4,5 A more appropriate goal for chelation therapy is clinical improvement, resolution of metabolic acidosis, and a serum iron concentration of <100 micrograms/dL.4,5 If a patient survives, the outcome is usually favorable, although significant morbidity may result from intestinal scarring, including pyloric stenosis and intestinal obstructions.4,5
Conclusion
Although rare, physicians should become aware of potential iron toxicity associated with ingestion of commercially-available hand warmers, especially in children, the elderly, or those with significant comorbid conditions. Information regarding the exact amount of iron in these products is neither readily available on the Internet, nor included in the MSDS. Consultation with a medical toxicologist is recommended.
Acknowledgment: The authors thank Bernadette Glenn Porter, BS, for her medical editing assistance with this article.
Author Contributions: Manuscript writing (DAJ); manuscript writing and editing (JLW, KDK); image capture for figures (JLW); data generation (JLW, LKS, DAJ); case description and coordination (KDK).
Financial/Material Support: None.
Disclosures: Ms. Sherrow was a medical student at the New York Institute of Technology College of Osteopathic Medicine, Old Westbury, New York at the time of the case.
Footnotes
☆
Information from this case was presented in abstract form at the North American Congress of Clinical Toxicology/NACCT Annual Meeting, San Francisco, California, October 10-12, 2015, as “Hot Under the Collar.”
Submitted for publication April 2017.
Accepted for publication December 2016.
