Abstract
Trioxane is a stable cyclic trimer of formaldehyde. It is an active ingredient in fuel bars for heating prepackaged foods by military and outdoorspeople. Trioxane depolymerizes to formaldehyde in an acidic environment and is further oxidized to formic acid, which causes neurologic and ocular damage. Because it is solid at room temperature, trioxane is a greater potential hazard to children than aqueous formaldehyde. Little information is available regarding the management of ingestion of solid, compressed fuel bars. We present a case of a 19-mo-old male child who ingested an unknown amount of a trioxane fuel bar, with fortunately limited consequences.
Background
Trioxane (metaformaldehyde; 1,3,5-trioxane; sym-trioxane; 1,3,5-trioxacyclohexane; and trioxymethylene) is a stable cyclic trimer of formaldehyde that exists as a white solid at room temperature (Figure 1). 1 It is the active ingredient in fuel bars used to heat prepackaged food by the military, outdoorspeople, and some relief organizations. Fuel bars are distributed to members of the military but are also available to consumers. They can be purchased at outdoor stores, army surplus stores, and online. The fuel bars typically come packaged in foil wrapping with 3 bars in a box (Figure 2). Trioxane is also used in industry as an intermediate in organic synthesis, specifically in chloromethylation, production of formals, condensations with phenols and amides, and the Mannich synthesis. 2
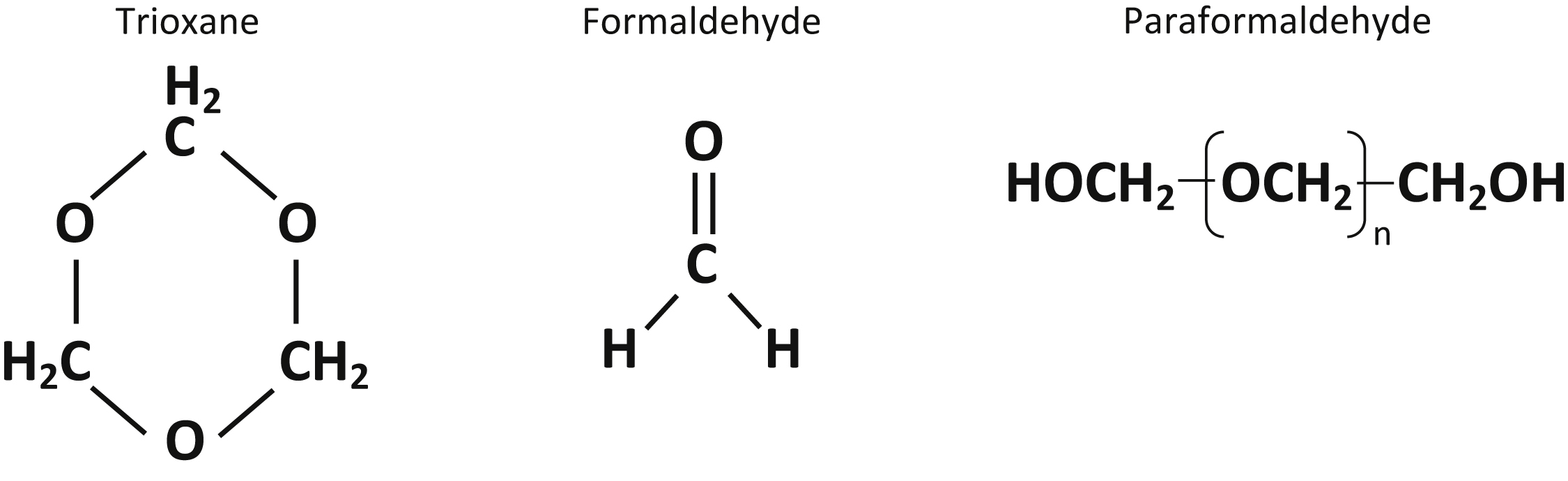
Structures of trioxane, formaldehyde, and paraformaldehyde.

Representative image of a trioxane fuel bar and its packaging.
Inhalation and ingestion are the primary routes of exposure. In an acidic environment, such as the stomach, trioxane slowly depolymerizes to formaldehyde. When heated, trioxane releases formaldehyde vapor. Formaldehyde is quickly oxidized to formic acid in vivo via aldehyde dehydrogenase, as in the final step to metabolism of methanol.3,4 Formic acid inhibits cytochrome oxidase during cellular respiration, causing acidosis and acidemia similar to cyanide and carbon monoxide poisoning. 5 Neurologic and ocular toxicity have been well described from exposure to formaldehyde and formic acid, including blindness and parkinsonian-like movement disorders.6,7
Unlike other chemical species commonly discussed in the context of “toxic alcohols,” formaldehyde is gaseous at standard temperature and pressure. It is most commonly encountered in liquid form as formalin or other aqueous hydrates, solutions, or azeotropes. Most case information available describes ingestion of aqueous formaldehyde resulting in caustic gastrointestinal injury, hemolysis, and death. In some of these cases, however, the ingested formaldehyde was described as formalin. 8 –15 Formalin is an aqueous mixture of formaldehyde, stabilized with methanol. The toxicities associated with formalin are well known, having been described previously. 6
The risk of toxicity from trioxane is mainly extrapolated from animal data and inferred from what is known about formaldehyde or methanol exposures. 16 There is a paucity of previously documented trioxane cases. In 1961, there was report of a case of possible trioxymethylene (trioxane) ingestion in a 22-mo-old, who was treated with induced vomiting and reportedly had no adverse effects. An unspecified amount of the trioxymethylene was sent for testing and was confirmed to be paraformaldehyde, not trioxane, which was reported to be nontoxic at that time. 17 Paraformaldehyde is a polyoxymethylene that can theoretically metabolize to formaldehyde in vivo. 18 Therefore, this raises uncertainty about the actual product ingested in this previously reported case and whether this represents a true trioxane ingestion.
Highly concentrated products containing toxic chemicals have the protentional to result in significant toxicity when ingested. This case report discusses the mechanism of trioxane toxicity and provides a therapeutic approach to an unusual exposure.
Case
A healthy 19-mo-old male child, weighing 12.7 kg, was found by his mother chewing through the packaging of military surplus fuel. The fuel bar was composed of compressed trioxane and contained 95% trioxane by weight, 2 to 5% magnesium stearate, and methylene blue for coloring. The child’s mother found a hole in the package and contacted the regional poison control center. No particulate matter was found in the child’s mouth or on his face or hands, but his mother felt a small amount to be missing from the package. The child was asymptomatic. His mouth was rinsed, and he was brought to a local emergency department.
Blood work done approximately 1.5 h after exposure initially showed an anion gap of 10 mEq·L-1 with bicarbonate of 24 mmol·L-1. It was recommended that laboratory testing be repeated every 2 h and the patient be given intravenous folic acid 2 mg·kg-1 every 4 h. Repeat laboratory testing at 4 h postexposure showed an anion gap of 11 mEq·L-1 and bicarbonate of 19 mmol·L-1. Because of the potential for worsening acidosis, he was transferred to a facility with a pediatric intensive care unit and hemodialysis capability. Repeat laboratory testing showed a stable bicarbonate level of 20 mmol·L-1. Formaldehyde and formic acid levels were not obtained. He was given a total of 3 doses of folic acid and discharged home 20 h postexposure without sequelae.
Discussion
Significant formaldehyde ingestions are unusual owing to the acrid nature of the substance, which typically limits exposure. Therefore, caustic or systemic metabolic aberrations rarely result from inhalation or ingestion of aqueous formaldehyde. In this case, the potential for a significant exposure was increased by the concentrated nature of the product. It is unknown whether ingestion of trioxane could cause caustic injury in the gastrointestinal tract.
There are no reports of trioxane toxicity; therefore, treatment in this case was based on previous experience with formaldehyde and the metabolic pathway of trioxane. Metabolism of trioxane involves the hydrolysis and oxidation of trioxane to formic acid (Figure 3). Formic acid contributes to the anion gap and causes anion gap metabolic acidosis. Thus, monitoring the bicarbonate and anion gap assesses the presence and degree of trioxane toxicity.
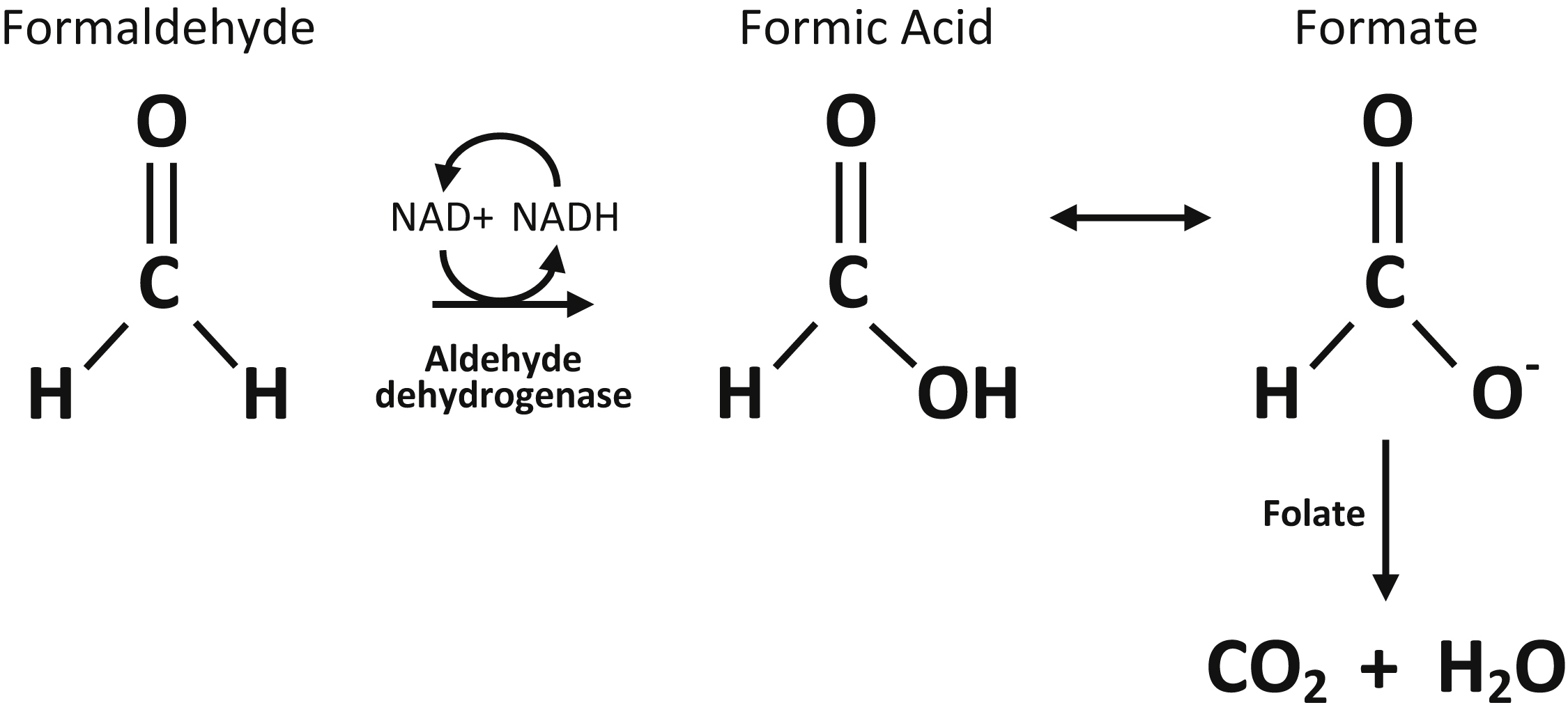
Metabolic pathway of trioxane after conversion to formaldehyde.
Although methanol and formaldehyde both ultimately metabolize to formic acid, fomepizole or ethanol would have been an impractical antidote in this case. Fomepizole inhibits the metabolic enzyme responsible for methanol metabolism, alcohol dehydrogenase. Formaldehyde is not metabolized by this enzyme because it is not an alcohol; thus, blocking it would not prevent metabolism of formaldehyde to formic acid. Theoretical use of an aldehyde dehydrogenase inhibitor, such as disulfiram or diethyldithiocarbamate (not available in the United States), could plausibly retard formation of formic acid and serve as a potential antidote, as described in other toxicities. However, these treatments are not benign, and human experience is not well described. 19 Our patient was given folic acid to enhance conversion of formate to carbon dioxide and water, as has been shown to be effective in animal models, but this is not approved by the Food and Drug Administration. 20 Our patient was transferred to a facility capable of pediatric hemodialysis when his bicarbonate decreased. Had the acidosis worsened, hemodialysis would have been indicated. Hemodialysis enhances elimination of formic acid and corrects underlying acidosis.6,21 Serum alkalization with sodium bicarbonate may also be useful to dissociate formic acid and decrease diffusion into cells. 3
Based on this case, we propose a therapeutic approach for trioxane exposures to consist of decontamination, supportive care, and enhanced elimination. Decontamination should include removing any particulate matter from the oropharynx, typically by rinsing the mouth. Intravenous fluids and sodium bicarbonate are supportive care measures that can maintain intravascular volume and serum pH. Enhanced elimination entails a folic acid infusion to convert formate to carbon dioxide and water and hemodialysis if the patient becomes increasingly acidotic.
Trioxane exposure cannot be confirmed without obtaining a formaldehyde or formic acid level. These levels are not readily available and should not guide treatment in the acute setting.5,12 However, the exposure history in this case was compelling. This report is limited by subjective reporting by family and inability to document the dose ingested.
Conclusions
This case documents a rare pediatric exposure to trioxane. Formaldehyde solid derivatives pose a greater ingestion risk than aqueous solutions, with potential toxicologic consequences. With minimal clinical experience available on ingestion of solid, compressed formaldehyde derivatives, further research is needed to delineate the toxic dose, potential toxicities, and therapeutic management.
Footnotes
Acknowledgements
Author Contributions: All authors were involved in data collection, analysis, and interpretation. JT and MS drafted the article and DG provided critical revision. All authors gave final approval of the article for publication.
Financial/Material Support: None.
Disclosures: None.
This work was previously presented at the 2016 North American Congress of Clinical Toxicology, Boston, MA.
