Abstract
The management of device-detected atrial fibrillation in patients with a history of stroke or transient ischemic attack remains a clinical dilemma, balancing the potential benefits of stroke prevention against the risks of anticoagulant-related bleeding. This review synthesizes evidence from the NOAH-AFNET 6 and ARTESIA trials, which evaluated the efficacy and safety of direct oral anticoagulants in this population. Collectively, randomized evidence regarding the use of direct oral anticoagulants in patients with device-detected atrial fibrillation and a history of stroke or transient ischemic attack remains limited and internally discordant. In ARTESIA, apixaban was associated with a reduction in ischemic stroke accompanied by an increase in major bleeding, yielding a relatively coherent benefit–risk signal in this high-risk subgroup. In contrast, NOAH-AFNET 6 demonstrated a modest and statistically non-significant reduction in ischemic stroke alongside a substantial increase in major bleeding. Further research is still needed to refine patient selection and optimize therapeutic strategies.
Keywords
Introduction
Atrial fibrillation (AF) remains a well-established risk factor for ischemic stroke, with oral anticoagulation (OAC) demonstrating proven efficacy in reducing stroke risk among patients with clinically documented AF.1,2 The widespread adoption of cardiac implantable electronic devices (CIEDs) has led to increased detection of brief, asymptomatic atrial tachyarrhythmia episodes, termed atrial high-rate episodes (AHREs)/device-detected atrial fibrillation (DDAF) or subclinical atrial fibrillation (AF) when confirmed on intracardiac electrogram, 3 creating clinical uncertainty regarding anticoagulation initiation, particularly in patients with prior stroke or transient ischemic attack (TIA) who face elevated risks of recurrent thromboembolism.
Prior studies have demonstrated that device-detected atrial tachyarrhythmias confer a two- to five-fold increased risk of stroke or systemic embolism, particularly when episodes exceed 5–6 min or cumulative burden surpasses 24 h.4-6 Early device studies (for example the TRENDS analysis) demonstrated a duration-dependent relationship between atrial arrhythmia burden and stroke, 7 and prolonged electrocardiogram monitoring studies in stroke cohorts confirmed that extended monitoring substantially increases AF detection rates. 8 However, the precise episode-duration thresholds at which the stroke-prevention benefits of anticoagulation outweigh bleeding risks remained uncertain prior to contemporary randomized evaluations.
Recent randomized trials including NOAH-AFNET 6 (Non-Vitamin K Antagonist Oral Anticoagulants in Patients With Atrial High Rate Episodes) and ARTESIA (Apixaban for the Reduction of Thrombo-Embolism in Patients With Device-Detected Sub-Clinical Atrial Fibrillation) have investigated direct oral anticoagulants (DOACs) in DDAF populations.9-12 The most recent pooled analysis of NOAH-AFNET 6 and ARTESIA demonstrated a significant reduction in ischemic stroke and composite cardiovascular events with DOACs, but without clear mortality benefit due to wide confidence intervals and low event rates. 13 These benefits are counterbalanced by an increased risk of major bleeding. 13 Notably, both trials reported lower-than-anticipated stroke rates in DDAF cohorts, raising questions about the risk-benefit ratio of routine anticoagulation across all risk strata.14-17
Management of DDAF in stroke or TIA survivors remains contentious. Recently published subgroup analyses from NOAH-AFNET 6 and ARTESIA offer pivotal new insights.18,19 The objective of this study was to comprehensively identify, map, and summarize randomized or prespecified subgroup evidence evaluating DOACs in patients with DDAF and prior stroke or TIA, and to characterize areas of concordance, discrepancy, and evidence gaps.
Methods
Accordingly, the present work is designed as a systematic scoping review to map and critically synthesize randomized and prespecified subgroup evidence on the use of DOACs in patients with DDAF and a history of stroke or TIA. Given the limited number of eligible randomized data sources and substantial heterogeneity in trial design and comparators, quantitative meta-analysis was not considered methodologically appropriate. This review followed the PRISMA-ScR (Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews) framework to ensure methodological rigor and transparency. 20
Search Strategy
Two investigators independently searched PubMed and Embase from inception to January 2026 using the terms: (‘device-detected atrial fibrillation’ OR ‘atrial high-rate episodes’ OR ‘subclinical atrial fibrillation’) AND (‘non-vitamin K antagonist oral anticoagulants’ OR ‘direct oral anticoagulants’ OR ‘DOAC’ OR ‘edoxaban’ OR ‘apixaban’ OR ‘rivaroxaban’ OR ‘dabigatran’). To complement the electronic search, the references of prior reviews were manually screened to identify any additional relevant studies.
Eligibility Criteria
Inclusion criteria were as follows: 1) randomized controlled trials (RCTs) or prespecified subgroup analyses evaluating DOACs in adults with DDAF and prior stroke/TIA; 2) comparisons between DOACs and alternative strategies (placebo or antiplatelets); and 3) primary endpoints encompassing stroke, systemic embolism, major bleeding events, or mortality, with secondary outcomes including individual components of the primary outcomes.
Definitions of DDAF
We used the term “DDAF” as an umbrella descriptor for atrial tachyarrhythmia episodes identified by implanted cardiac devices but not previously documented as clinical AF on a standard surface electrocardiogram (Figure 1). ARTESIA required at least one episode of device-detected subclinical AF lasting ≥6 min and <24 h for eligibility (detected by pacemaker, defibrillator, or implantable cardiac monitor). In NOAH-AFNET 6, patients were enrolled on the basis of AHREs lasting ≥6 min, defined according to prespecified device rate thresholds, in the absence of documented clinical AF. Because enrolment thresholds, device types, and the distribution of episode durations differed between trials, reported arrhythmia burdens and event rates are not directly interchangeable between ARTESIA and NOAH-AFNET 6. Throughout this review, DDAF is used as a pragmatic umbrella term to facilitate cross-trial discussion. The term AHRE refers specifically to device-defined AHREs based on programmed rate criteria, whereas subclinical AF denotes DDAF episodes in patients without previously documented clinical AF.

Comparative features of device-detected versus clinically detected Non-valvular atrial fibrillation.
Study Selection
Although this review was initially framed as a systematic scoping review, the structured search process primarily served to confirm the absence of additional RCTs beyond NOAH-AFNET 6 and ARTESIA that met our predefined eligibility criteria. The search identified 186 records; however, only these two prespecified subgroup analyses of NOAH-AFNET 6 and ARTESIA18,19 fulfilled all inclusion criteria (Figure 2 and Supplementary Table S1). Thus, the present review was more accurately characterized as a structured evidence synthesis of a prospectively identified dataset, with the search strategy providing transparency and confirmation of evidentiary completeness rather than expansion of the analytical sample.
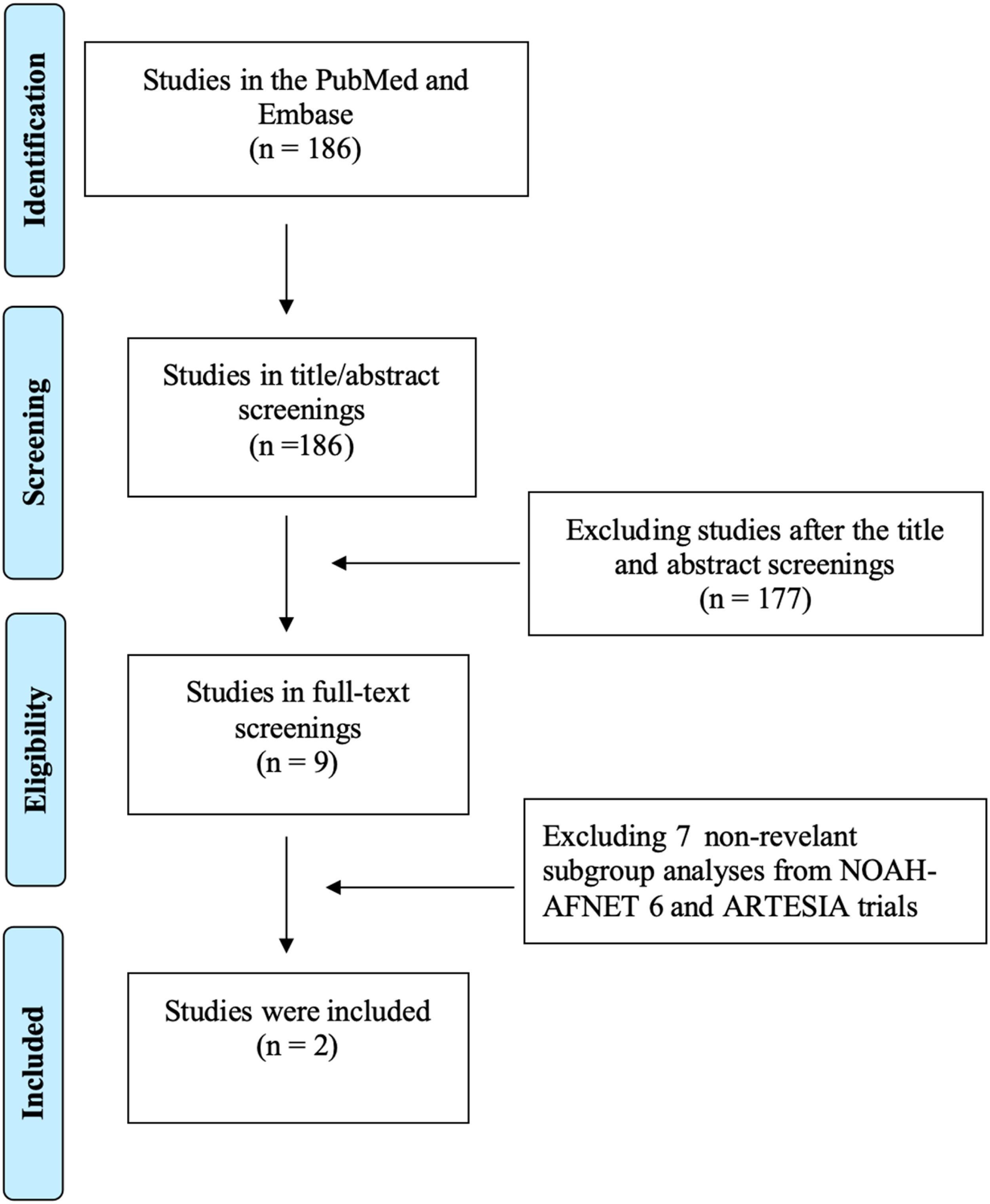
PRISMA flowchart of study selection in this review.
Results
Two prespecified subgroup analyses of NOAH-AFNET 6 and ARTESIA18,19 were included. The small number of included studies reflects the limited availability of randomized or prespecified subgroup analyses specifically evaluating DOACs in patients with DDAF and a history of stroke or TIA. Because only two prespecified subgroup analyses met inclusion criteria and these analyses differed in comparator strategy, arrhythmia burden thresholds, and outcome definitions, statistical pooling was not undertaken. Consistent with PRISMA-ScR recommendations, results are presented as a structured qualitative synthesis emphasizing patterns, discrepancies, and knowledge gaps rather than pooled effect estimates.
Baseline Clinical Characteristics
The baseline clinical characteristics of the NOAH-AFNET 6 and ARTESIA trials reveal notable similarities and differences in their respective study populations, particularly among patients with DDAF and a history of TIA (Table 1). In the NOAH-AFNET 6 trial, the subgroup analysis included 253 patients with prior stroke or TIA, representing 10% of the total randomized population. These patients had a median CHA2DS2-VASc score of 6, reflecting a high thromboembolic risk profile. The mean age was approximately 78 years, with 36.4% being women. Comorbidities such as diabetes (38.5% in the edoxaban group and 33.6% in the placebo group), valvular heart disease (13.9% and 16.8%, respectively), and peripheral arterial disease (11.5% and 14.5%, respectively) were prevalent. Additionally, a significant proportion of these patients had indications for acetylsalicylic acid (82% in the edoxaban group and 84.7% in the placebo group), underscoring the complexity of their comorbid clinical profiles. Cognitive impairment, as assessed by the Montreal Cognitive Assessment (MoCA) score, was present in approximately 60%–67% of patients, further underscoring the frailty of this population.
Baseline Characteristics of Participants with Prior Stroke/TIA in the ARTESiA and NOAH-AFNET6 Trials.

ARTESiA: Apixaban for the Reduction of Thrombo-Embolism in Patients With Device-Detected Subclinical Atrial Fibrillation; NOAH-AFNET 6: Non-Vitamin K Antagonist Oral Anticoagulants in Patients With Atrial High Rate Episodes; BMI: Body Mass Index; CHA2DS2-VASc: Congestins, Hypertension, Age ≥75 (doubled), Diabetes Mellitus, previous Stroke or Transient Ischemic Attack—doubled for age ≥75 and stroke or TIA; Vascular disease, Age 65–74, Sex category (female); eGFR: Estimated Glomerular Filtration Rate; CHF: Congestive Heart Failure; %: Percentage.
The ARTESIA trial included 346 participants (8.6% of the total cohort) with a history of stroke or TIA. These patients were slightly younger, with a mean age of 76 years, and had a similar median CHA2DS2-VASc score of 6. The prevalence of documented atherosclerotic disease (50%) was notable. Unlike NOAH-AFNET 6, where most patients had pacemakers or defibrillators, a significant subset in ARTESIA (15%) used implantable cardiac monitors for DDAF detection. The longest duration of subclinical AF episodes prior to randomization varied widely, with median maximum episode durations prior to randomization were 3.1 h in NOAH-AFNET 6 and 4419 min (≈73.7 h) in ARTESIA, reflecting substantial intertrial differences in arrhythmia burden.
Both trials demonstrated that patients with prior stroke or TIA had higher baseline risks compared to those without such history, as evidenced by elevated CHA2DS2-VASc scores and greater comorbidity burdens. However, the ARTESIA population exhibited a higher prevalence of atherosclerotic disease, while the NOAH-AFNET 6 cohort included older patients with a higher frequency of cognitive impairment. Both trials enrolled predominantly male participants (approximately 64% in NOAH-AFNET 6% and 69% in ARTESIA), with comparable gender distributions across treatment arms (p > 0.05 in both studies).
NOAH-AFNET 6: Edoxaban in DDAF with Prior Stroke
The NOAH-AFNET 6 trial investigated the efficacy and safety of oral anticoagulation with edoxaban in patients with DDAF and a history of stroke or TIA. The study included 253 patients with prior stroke or TIA, randomized to either edoxaban or no anticoagulation (placebo or aspirin). The primary outcome, a composite of stroke, systemic embolism, and cardiovascular death, occurred at similar rates in both groups (5.7% per patient-year with edoxaban vs 6.3% without; HR 0.9, 95% CI 0.4-1.8), indicating no significant benefit from anticoagulation in reducing thromboembolic events. Notably, the ischemic stroke rate was lower than expected in this population (edoxaban vs no anticoagulation: 1.6% vs 2.3% per patient-year; HR 0.7, 95% CI 0.2-2.4), suggesting a modest baseline risk. However, anticoagulation was associated with a numerical increase in major bleeding events (3.2% vs 0.8% per patient-year; HR 4.3, 95% CI 0.9-20.1), highlighting a potential safety concern. The study found no interaction between prior stroke/TIA and treatment effects, further underscoring the ambiguous net clinical benefit of anticoagulation in this setting. The findings suggest that the role of edoxaban in patients with DDAF and prior stroke or TIA remains uncertain, particularly due to the low observed stroke risk and the increased bleeding risk.
ARTESIA: Apixaban in DDAF with Prior Stroke
The ARTESIA trial subgroup analysis demonstrated that apixaban significantly reduced the risk of stroke or systemic embolism in patients with DDAF and a history of TIA. Among participants with prior stroke or TIA, apixaban reduced the annual rate of stroke or systemic embolism to 1.20% versus 3.14% per patient-year with aspirin (HR 0.40, 95% CI 0.17-0.95), with consistent benefit across stroke subgroups (stroke: 1.20% vs 3.14%; ischaemic stroke or unknown stroke: 1.20% vs 2.61%). The benefit was further underscored by a marked reduction in disabling or fatal strokes (HR 0.26, 95% CI: 0.07-0.93). While apixaban increased major bleeding (2.26% vs 1.16% per patient-year with aspirin; HR 1.94, 95% CI: 0.77-4.87), the absolute risk difference was outweighed by the stroke prevention benefit, with no significant rise in intracranial hemorrhage or fatal bleeding. The findings support the use of apixaban for secondary stroke prevention in high-risk patients, particularly those with prior stroke/TIA, aligning with the net clinical benefit observed in this subgroup.
Discussion
Divergent Findings from NOAH-AFNET 6 and ARTESIA Trials
The efficacy of DOACs in patients with DDAF and a history of stroke remains a subject of debate, as evidenced by conflicting results from the NOAH-AFNET 6 and ARTESIA trials. In the NOAH-AFNET 6 trial, anticoagulation with edoxaban did not significantly reduce the composite primary outcome of stroke, systemic embolism, or cardiovascular death compared to placebo or aspirin in patients with prior stroke or TIA. Conversely, the ARTESIA trial demonstrated a marked reduction in stroke or systemic embolism with apixaban versus aspirin in a similar population, with an absolute risk reduction of 7% over 3.5 years.
Several factors may account for the discordant efficacy results between NOAH-AFNET 6 and ARTESIA. First, the median atrial high-rate episode duration differed substantially (3.1 h vs 73.7 h, respectively), suggesting that the ARTESIA population harbored a higher cumulative atrial arrhythmia burden. Second, NOAH-AFNET 6 used a mixed comparator (placebo or aspirin), which could dilute treatment contrasts compared with the uniform aspirin control in ARTESIA. Third, regional differences in patient characteristics may have influenced outcomes: NOAH-AFNET 6 included an older, frailer European population with more cognitive impairment, whereas ARTESIA had a higher prevalence of atherosclerotic disease and closer follow-up intervals, possibly improving adherence. Finally, pharmacologic differences between edoxaban and apixaban, particularly in renal excretion and anti–Factor Xa potency, may have modestly influenced efficacy in elderly patients with renal dysfunction. Collectively, these disparities underscore that heterogeneous trial design rather than contradictory drug effects likely explains the apparent inconsistency.
Differences in AHRE definition and duration thresholds between trials further complicate cross-study comparisons. ARTESIA enrolled patients with DDAF episodes lasting at least 6 min but typically less than 24 h, whereas NOAH-AFNET 6 required AHREs ≥6 min in duration without clinically documented AF at baseline. Device-based observational data have demonstrated a graded association between AHRE duration and stroke risk, with longer episodes conferring higher thromboembolic risk.5,6 Variations in episode burden and inclusion thresholds likely influenced baseline event rates and may partially explain differences in observed treatment effects. Careful attention to AHRE duration is therefore essential when extrapolating trial findings to broader device-detected populations.
Balancing Efficacy and Safety
The safety profile of DOACs in DDAF patients with prior stroke is equally critical. Both trials reported increased major bleeding with anticoagulation, but the absolute risk differences varied. In NOAH-AFNET 6, major bleeding events were numerically higher with edoxaban, while ARTESIA observed an increase in the risk of major bleeding with apixaban, driven primarily by gastrointestinal bleeding. Importantly, neither trial found a significant rise in intracranial hemorrhage, a reassuring finding for a population already at elevated bleeding risk due to cerebrovascular disease. These outcomes underscore the need for individualized risk assessment, particularly in patients with additional bleeding risk factors (eg, advanced age, renal impairment, or concomitant antiplatelet use). 21 A meta-analysis 13 by McIntyre et al pooling data from both trials suggested a 54% relative risk reduction in stroke or systemic embolism with DOACs, albeit with wider confidence intervals due to limited subgroup data.
Neither NOAH-AFNET 6 nor ARTESIA reported a formally calculated net clinical benefit specifically for the prior stroke/TIA subgroup. Instead, both trials presented separate estimates for ischemic stroke and major bleeding within subgroups.18,19 In NOAH-AFNET 6, recurrent stroke occurred at 1.6% per patient-year with anticoagulation and 2.3% per patient-year without anticoagulation, whereas major bleeding occurred at 3.2% and 0.8% per patient-year, respectively, leading the investigators to describe the overall effect of anticoagulation in this subgroup as ambiguous rather than clearly beneficial. Similarly, ARTESIA presents efficacy and safety outcomes separately in patients with prior stroke or TIA, without integrating them into a composite net-benefit metric. Therefore, current randomized evidence does not demonstrate a proven net clinical benefit of anticoagulation in patients with DDAF and previous stroke or TIA. Clinical decisions should be individualized and based on parallel consideration of stroke risk, bleeding risk, and patient preferences, rather than on an assumption of overall benefit.
Pathophysiological Considerations in DDAF after Stroke
DDAF is increasingly recognized as a manifestation of atrial cardiomyopathy characterized by fibrosis, electrical remodeling, and mechanical dysfunction. These alterations foster endothelial injury and blood-stasis, generating a prothrombotic milieu even in the absence of sustained arrhythmia. In stroke survivors, concurrent inflammation and neurohumoral activation further amplify atrial remodeling, providing a mechanistic rationale for anticoagulation despite brief or subclinical AF episodes.
Beyond NOAH-AFNET 6 and ARTESIA, observational studies and meta-analyses consistently demonstrate that device-detected AHREs are associated with increased risks of stroke, systemic embolism, and progression to clinical AF, with risk rising in a burden-dependent manner. A systematic review and meta-analysis reported approximately two-fold higher thromboembolic risk among individuals with device-detected AHREs compared with those without such episodes. 4 In parallel, expert consensus remains divided as to whether subclinical AF should be managed similarly to clinical AF, reflecting uncertainty regarding the causal versus marker role of DDAF in stroke pathogenesis. 15
Recent pooled analyses of NOAH-AFNET 6 and ARTESIA further suggest that the relative reduction in stroke or systemic embolism with DOACs emerges earlier than the accumulation of major bleeding events, implying a time-dependent net clinical benefit in selected patients. 16 Although atrial cardiomyopathy provides a plausible mechanistic framework linking atrial disease to stroke in the absence of sustained AF, randomized evidence does not yet support anticoagulation solely on the basis of atrial cardiopathy. Both the ARCADIA and ATTICUS trials failed to demonstrate superiority of apixaban over aspirin in patients with cryptogenic or embolic stroke of undetermined source,22,23 highlighting the need for more precise biological and electrophysiological markers before this concept can be translated into routine anticoagulation strategies.
Mechanistic Insights into Stroke Pathogenesis in DDAF
The available data suggest that anticoagulation with DOACs appears to confer a potential net clinical benefit for selected DDAF patients with prior stroke or TIA, although efficacy signals are driven primarily by the ARTESIA trial, whereas NOAH-AFNET 6 showed neutral results with edoxaban. The pathophysiologic basis for thromboembolic events in DDAF extends beyond transient atrial arrhythmia episodes and likely reflects underlying atrial cardiomyopathy, endothelial dysfunction, and atrial fibrosis that predispose to thrombogenesis even in the absence of sustained AF. Imaging and biomarker studies have identified left atrial enlargement, elevated NT-proBNP, and abnormal P-wave indices as correlates of subclinical atrial dysfunction and stroke risk. Thus, the observed benefit of apixaban in ARTESIA may reflect its ability to mitigate embolic events in patients with such substrate abnormalities rather than solely prevent AF-related embolization. Future research should integrate imaging, electrophysiologic, and biomarker data to refine patient selection criteria for anticoagulation in DDAF.
Limitations
Several unresolved issues merit attention. First, the optimal duration and burden of DDAF required to justify anticoagulation remain undefined. While ARTESIA included episodes as short as 6 min, the stroke risk associated with very brief AHREs may be insufficient to outweigh bleeding risks. Second, the external validity of NOAH-AFNET 6 and ARTESIA is limited. Both trials enrolled patients with implanted or insertable cardiac devices, close follow-up, and relatively stable clinical profiles. Therefore, extrapolation to frailer elderly individuals, patients with very brief AHREs, and those without continuous rhythm monitoring should be undertaken with caution. In addition, the generalizability of these findings to acute stroke populations is limited, as both trials predominantly enrolled patients with remote strokes (>1 year post-event). Studies like FIND-AF 2 (NCT04371055) are investigating whether early anticoagulation post-stroke improves outcomes in DDAF detected via prolonged monitoring.24,25 Third, these subgroup findings of NOAH-AFNET 6 and ARTESIA should be regarded as exploratory and hypothesis-generating, and they do not constitute definitive evidence for routine anticoagulation in all patients with DDAF and prior stroke or TIA.
Conclusion
Current randomized evidence regarding the use of DOACs26,27 in patients with DDAF and a history of stroke or TIA remains limited and internally discordant. ARTESIA suggests a reduction in ischemic stroke at the cost of increased major bleeding, whereas NOAH-AFNET 6 demonstrates increased bleeding without a clearly significant reduction in ischemic stroke. Rather than supporting a uniform favorable risk–benefit profile, these findings highlight genuine uncertainty and underscore the need for individualized risk stratification, particularly with respect to AHRE burden and competing bleeding risk.
Supplemental Material
sj-docx-1-cat-10.1177_10760296261438235 - Supplemental material for Discrepant Outcomes of Direct Oral Anticoagulant Trials in Device-Detected Atrial Fibrillation after Stroke: Insights from NOAH-AFNET 6 and ARTESIA
Supplemental material, sj-docx-1-cat-10.1177_10760296261438235 for Discrepant Outcomes of Direct Oral Anticoagulant Trials in Device-Detected Atrial Fibrillation after Stroke: Insights from NOAH-AFNET 6 and ARTESIA by Heng Liu, Zhangbin Li, Xiaoqing Chen and Junjie Zhou in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Ethical Approval
Not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
All data and materials are presented in the paper.
Supplemental Material
Supplemental material for this article is available online.
