Abstract
Introduction
Direct oral anticoagulants (DOACs) demonstrated similar efficacy and lower risk of intracranial hemorrhage than warfarin in patients with atrial fibrillation and venous thromboembolism. Given the lack of data identifying risk factors in patients who bled while on a DOAC, we sought to investigate these characteristics.
Materials and Methods
This retrospective chart review was approved by the Mass General Brigham Institutional Review Board and assessed patients who experienced bleeding events while on DOAC therapy from 6/1/2015 to 7/1/2020. Patient characteristics were evaluated, including age, sex, body mass index (BMI), renal function, concomitant therapies, and baseline comorbidities.
Results
Eighty-seven patients were included for analysis, with a median age of 75.8 years. Most patients were female (51.7%) and 24 (27.6%) had a BMI >30. At time-of-event, 21 patients (24.1%) had acute kidney injury. Thirty-three patients (37.9%) were on concomitant antiplatelet therapy (APT), with 31 (35.6%) on single APT and 2 on dual APT. Pertinent comorbidities included hypertension (74.7%), ischemic cerebrovascular accident (28.7%), thyroid abnormality (23.0%), active cancer (14.9%), and anemia (13.8%). Eleven patients (12.6%) had a prior bleeding event. Most patients were on apixaban (69.0%) for the indication of stroke prevention in nonvalvular atrial fibrillation/flutter (72.4%). FDA-approved dosing was used in most patients (92.0%), and all deviations reflected underdosing. Most bleeding events were defined as major (95.4%), occurred at a critical organ site (72.4%), and developed spontaneously (58.6%).
Conclusions
These data provide insight into characteristics of patients who experience bleeding events while on DOAC therapy. Understanding these potential risk factors may optimize the safe use of these agents.
Introduction
Direct-acting oral anticoagulants (DOACs) confer various advantages over other anticoagulants, including their oral formulation and minimal therapeutic monitoring. In current practice guidelines, DOACs serve as first-line therapy for various disease states, including venous thromboembolism (VTE) treatment and stroke prevention in atrial fibrillation (AF) or flutter.1,2
Various trials have demonstrated similar efficacy and lower rates of intracranial hemorrhage with DOACs as compared to warfarin, with similar or lower risk of overall bleeding.3–10 Although bleeding rates were reported within these landmark trials, subgroup analyses have not been performed to assess the patient characteristics of those who bled while on DOAC therapy. One study captured a cohort of patients with DOAC-related bleeding events and determined that most patients were elderly males with moderate bleeding risk scores. 11
Although risk factors for bleeding have been reported for other anticoagulants, there remains limited data identifying risk factors in patients who experience a bleeding event while on DOAC therapy. Therefore, this descriptive analysis sought to provide insight into the characteristics of patients who experienced a DOAC-related bleed.
Methods
This retrospective, descriptive analysis was approved by the Mass General Brigham Investigational Review Board and was performed at a single, tertiary academic medical center. A system-generated report identified patients with an inpatient or emergency department medication order for one of five reversal agents from June 1, 2015, to July 1, 2020. The reversal agents utilized for initial patient identification included andexanet alfa, anti-inhibitor coagulation complex, recombinant factor VIIa, 4-factor prothrombin complex concentrate (4F-PCC), and idarucizumab. Patients were included for analysis if they received the reversal agent to treat a DOAC-related bleeding event. DOAC-related bleeding events were defined as the occurrence of a bleeding event while actively prescribed one of four DOACs (apixaban, dabigatran, edoxaban, or rivaroxaban). Bleeding event diagnoses were extracted from history and physical exam documentation or other pertinent notes within the electronic medical record. Patients were excluded if they did not receive the ordered reversal agent, received the agent for a reason other than DOAC reversal, or underwent DOAC reversal for the purposes of facilitating a surgical procedure.
Patient characteristics were collected through manual chart review utilizing the electronic medical record. Specific characteristics captured included age, sex, body mass index (BMI), renal function, concomitant therapies, baseline bleeding risk scores, and pertinent baseline comorbidities. DOAC-related data were collected, including the prescribed agent and its indication, dosing, and duration of use. Bleeding events were classified as major or clinically relevant non-major as defined by the International Society on Thrombosis and Haemostasis (ISTH). 12 Bleeding events were further described based on the nature of occurrence (spontaneous vs traumatic), laboratory parameters at the time of event (hemoglobin and hematocrit), and whether or not they occurred at a critical site. Outcomes data included the achievement of hemostasis, mortality within 30 days of reversal, and the occurrence of a thromboembolic event within 30 days following reversal. Thromboembolic events included deep vein thrombosis, pulmonary embolism, ischemic stroke, and intracardiac thrombus. Hemostasis achievement was defined as documentation of bleeding cessation.
Results
Patients and Patient Characteristics
A total of 428 patients were screened, of which 87 were included for analysis. The majority of patients were excluded on the basis of receiving the reversal agent for a reason other than a DOAC reversal (Figure 1).

Patient screening.
Patients had a median age of 76 years, with females accounting for 51.7% of the cohort. The median BMI was 26.8, and most patients were categorized as either overweight (35.6%) or obese (27.6%). At the time of the bleeding event, the majority of patients had a creatinine clearance (CrCl) greater than 30 mL/min (82.8%), with a median CrCl of 51 mL/min. Twenty-one patients (24.1%) were in acute kidney injury at the time of event, as defined per by the Kidney Disease Improving Global Outcomes (KDIGO) criteria (Table 1). Pertinent comorbidities included hypertension (74.7%), history of ischemic cerebrovascular accident (CVA) (28.7%), thyroid disorder (23.0%), active cancer (14.9%), and anemia (13.8%). Eleven patients (12.6%) had a history of a prior bleeding event. When applicable, the median VTE-Bleed score was 3.25, with 9 of 14 patients having a high-risk score of 2 or greater. When applicable, the median HAS-BLED score was 2.67, with 35 of 63 patients having a high-risk score of 3 or greater (Table 2). Thirty-three patients (37.9%) were on concomitant antiplatelet therapy (APT), of which 31 (35.6%) were treated with single APT (SAPT) and 2 (2.3%) with dual APT (DAPT) with aspirin and clopidogrel. Of the patients on SAPT, 25 (28.7%) were on aspirin ≤100 mg, 5 (5.7%) were on clopidogrel, and 1 (1.1%) was on aspirin >100 mg. Seven patients were on concomitant P-glycoprotein (P-gp) inhibitor therapy, with 6 patients (6.9%) on amiodarone and 1 (1.1%) patient on dronedarone. One patient (1.1%) had concomitant use of a strong dual CYP3A4 and P-gp inhibitor. The majority of patients were categorized as medical or were captured through emergency department visits (92.0%), while seven patients were categorized as surgical (8.0%).
Patient Characteristics.
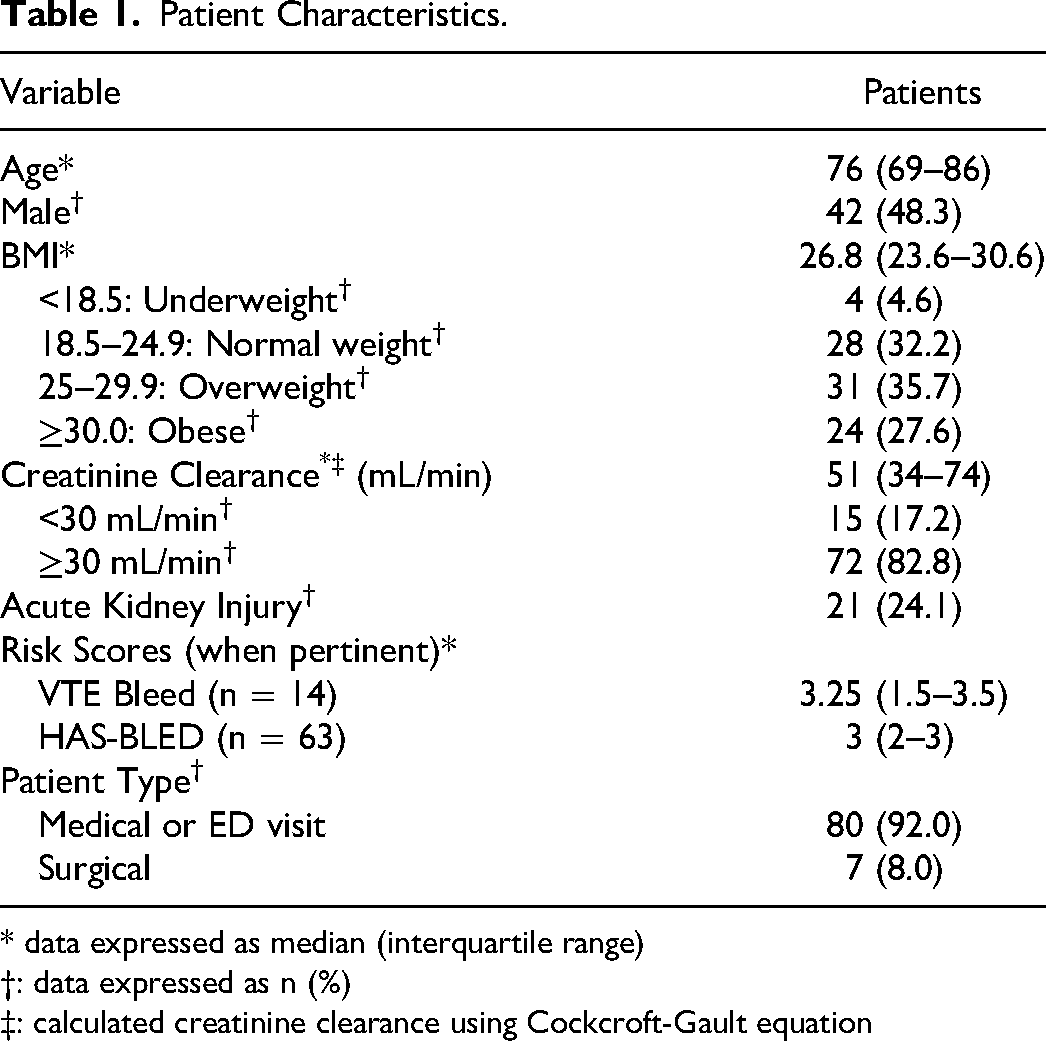
* data expressed as median (interquartile range)
†: data expressed as n (%) ‡: calculated creatinine clearance using Cockcroft-Gault equation
Pertinent Comorbidities.

DOAC Utilization
Apixaban was the most commonly prescribed DOAC (69.0%), followed by rivaroxaban (20.1%), and dabigatran (10.3%). No patients were prescribed edoxaban (Figure 2). Stroke prevention in atrial fibrillation/atrial flutter (72.4%) was the most common primary DOAC indication, followed by VTE treatment (13.8%), and secondary VTE prevention (8.0%). Five patients (5.7%) received a DOAC for an off-label indication, including peripheral vascular disease (2.3%), thromboprophylaxis in an oncology patient (1.1%), intracardiac thrombus (1.1%), and superior vena cava syndrome (1.1%) (Figure 3). The majority of patients (54.0%) had been prescribed a DOAC for more than one year at the time of event (Table 3). Most patients (90.8%) were receiving DOAC dosing that was adherent to Federal Drug Administration (FDA)-approved manufacturer labeling. All deviating doses were lower than that of the FDA-approved manufacturer labeling.
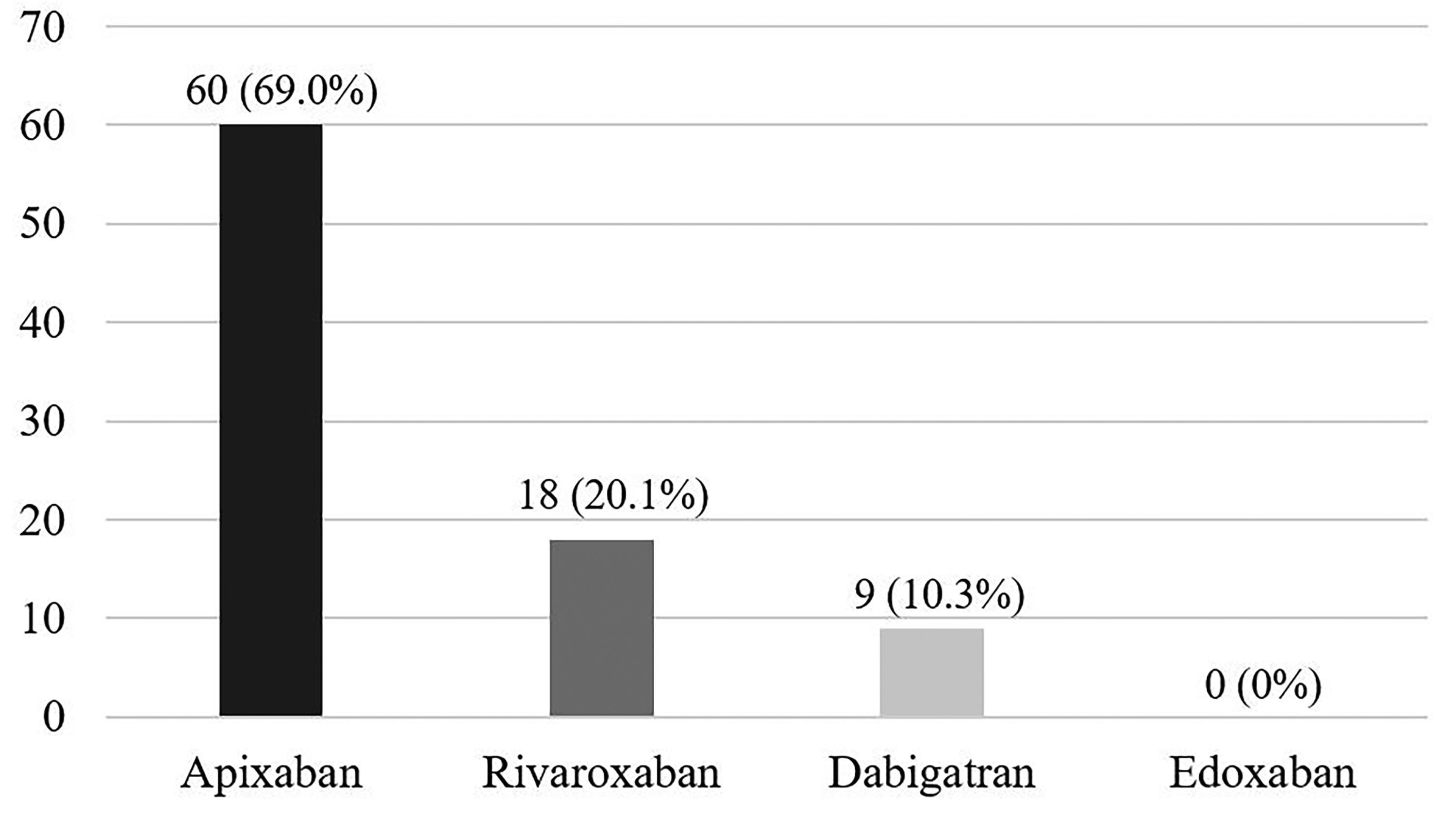
DOAC utilized.
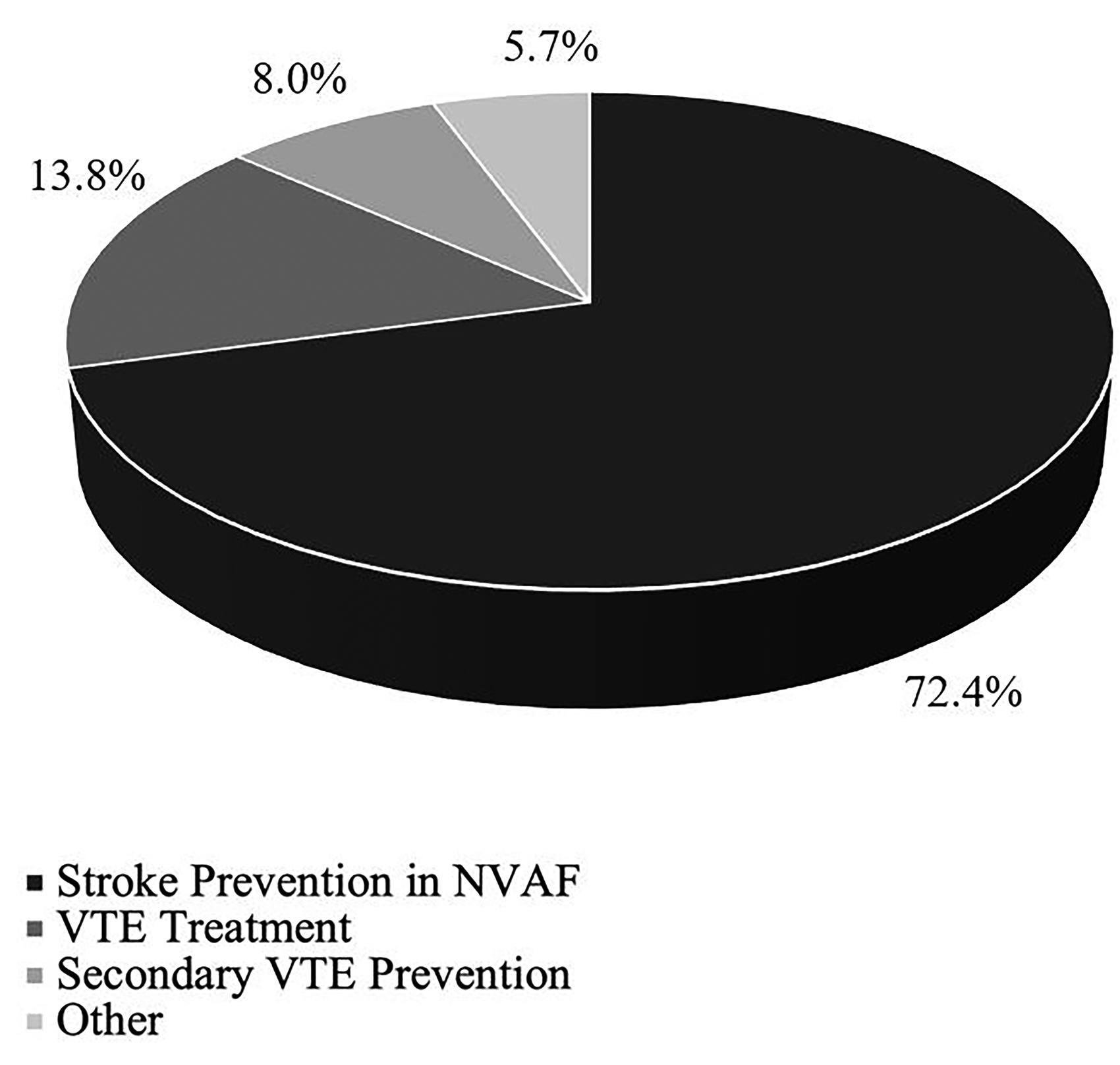
DOAC indication.
DOAC Duration Prior to Event.

Reversal Agents
Sixty-five patients (74.7%) received andexanet alfa for reversal, of which 22 patients also received other products, including 4F-PCC or fresh frozen plasma (FFP). Of the patients who received andexanet alfa, 90.8% received doses consistent with the FDA-approved manufacturer labeling, whereas 2 patients received a higher dose and 4 received only one of two intended administrations. Eight patients (9.2%) received idarucizumab for reversal, of which 1 patient also received FFP. Twenty-one patients (24.1%) received 4F-PCC, accounting for monotherapy in 8 patients (9.2%) and combination therapy in all others. The administered dose of 4F-PCC was 15 units/kg in 2 patients, 25 units/kg in 9 patients, and 50 units/kg in 10 patients. One of the patients who received 4F-PCC also received recombinant factor VIIa as a single dose of 40 mcg/kg. Nineteen patients (21.8%) received FFP. One patient received anti-inhibitor coagulation complex monotherapy at a dose of 100 units/kg (Table 4).
Reversal Agents.

Bleeding Events and Outcomes
Eighty-three (95.4%) of the bleeding events were categorized as major, while 4 patients (4.6%) experienced a clinically relevant non-major bleed. The majority of bleeding events (72.4%) occurred at a critical site organ, of which most were intracranial (63.2%). Spontaneous bleeds occurred in 51 patients (58.6%), whereas 36 patients (41.4%) experienced a traumatic bleed. At the time of event, patients had a median hemoglobin of 11.2 g/dL and a median hematocrit of 33.9%.
Within 30 days following reversal, 4 patients (4.6%) experienced a thromboembolic event, of which 2 (2.3%) had a deep vein thrombosis and 2 (2.3%) had an ischemic stroke. Thirty-two patients (36.8%) died within 30 days following reversal. At the time of discharge or death, 58 patients (66.7%) had achieved hemostasis, while 29 patients (33.3%) had not (Table 4).
Discussion
Given the benefits of DOACs and their ease of use, prescribing of these agents has increased over the years. DOACs have also demonstrated noninferiority to warfarin with similar or lower risk of bleeding in patients with AF or VTE.3–10 Although bleeding events were captured in the landmark trials, no subgroup analyses were performed to evaluate the characteristics of patients who bled. In the ARISTOTLE, ENGAGE AF TIMI 48, RE-LY, and ROCKET-AF trials, the majority of patients were elderly males, thus limiting the extrapolation of bleeding risk to other populations.3–6 In these studies, as well as the AMPLIFY, EINSTEIN, Hokusai-VTE, and RE-COVER trials, there was also no mention of HAS-BLED or VTE-BLEED risk scores, respectively.3–10 The lack of reporting of baseline bleeding risk, as well as the absence of subgroup analyses creates a gap in understanding the potential factors that increase bleeding risk in patients on DOAC therapy.
Some studies have demonstrated risk factors for bleeding in patients on other anticoagulants, including warfarin and low molecular weight heparin. Observed factors associated with increased bleeding in patients on warfarin include advanced age, elevated international normalized ratio (INR), elevated serum creatinine, concomitant use of acetylsalicylic acid, drug-drug interactions, and prior bleeding.13–16 Other comorbidities associated with increased bleeding on warfarin include hypertension, peripheral vascular disease, congestive heart failure, hepatic and/or renal failure, CVA, alcohol abuse, and cancer. 16 Many of these factors have also demonstrated higher bleeding risk in patients on low molecular weight heparins, with additional characteristics including recent trauma or surgery, female sex, and body surface area.17,18 These observational data set the foundation for the characteristics and factors that were chosen for assessment within this analysis.
Xu, et al published a retrospective cohort study comparing outcomes in nearly 20,000 patients who experienced DOAC-related versus warfarin-related bleeding events. 11 Patients who bled while on DOAC therapy had a median age of 81.2 years, were primarily males (61.1%), and had a mean HAS-BLED score of 2.7. The most commonly utilized DOAC was dabigatran and 44.1% of patients were on concomitant antithrombotic therapies. Although these data provide valuable insight into potential high-risk characteristics, several features of the study limit the application of these results. The cohort only included patients aged 66 years or older with a diagnosis of atrial fibrillation and/or flutter, thus impacting the median age and limiting the extrapolation of observed risk factors to younger patients and those with other indications for anticoagulation. Patients were excluded if they were not bleeding at initial presentation, whereas our study also captures the characteristics of patients who experienced a bleed within an admission.
Our analysis demonstrated that most patients who experienced a bleeding event while on DOAC therapy were elderly, female, and overweight or obese. Interestingly, these three characteristics encompass patients who were not well-captured in major trials. For example, females accounted for less than half of the patients in each of these trials, and BMI or weight stratification data were inconsistent.3–10,19 Most patients were 70–73 years old and 55–58 years old in the landmark trials assessing the use of DOACs in AF and VTE, respectively.3–10 A meta-analysis of 10 randomized control trials assessed the use of DOACs in elderly patients aged ≥75 years old, comparing dabigatran, rivaroxaban and apixaban to warfarin in AF or VTE. This analysis demonstrated that DOACs did not confer a higher bleeding risk than warfarin in this patient population and were noninferior or more effective. 20 Despite the beneficial application of this data, the analysis focused specifically on the comparison of the DOACs to warfarin, as opposed to assessing the bleeding risk comparison between elderly and non-elderly patients on DOAC therapy. Our analysis thus suggests the potential for an association between bleeding risk and age when utilizing these agents. Patients were also more likely to bleed while on longer-term therapy, as most patients within our study experienced a bleeding event after receiving DOAC therapy for more than one year.
Patients within our study frequently had comorbid hypertension and high bleeding risk scores at baseline. Notably, it is not uncommon for patients to be initiated on anticoagulation therapy with bleeding scores similar to those observed within this analysis. The 2019 American Heart Association/American College of Cardiology/Heart Rhythm Guideline for the Management of Patients with Atrial Fibrillation do not specify a risk threshold at which to forgo anticoagulation, and many patients with AF have associated risk factors for bleeding at baseline. 21 A retrospective analysis of patients initiated on DOAC therapies for AF demonstrated a mean HAS-BLED score of 1.6, which is markedly lower than the median of 2.67 observed in our patient population. 22 Other European analyses have demonstrated average HAS-BLED scores of 2.0 in patients initiated on various anticoagulants for AF.23,24 HAS-BLED scores higher than 2 have been associated with a greater than 85% higher risk of clinically relevant bleeding, a 2.4-fold greater risk of major bleeding, and a 2.9-fold higher risk of death.25,26 Although there is no specific guidance on withdrawing or omitting anticoagulation based on risk scores, it remains important to understand the implications of these scoring tools, especially when considered with other individual risk factors. Overall, given the benefit of anticoagulation in AF patients regardless of bleeding potential, the decision to anticoagulate patients is often a risk versus benefit weigh.
Given that DOACs are primarily renally eliminated, one would expect that patients with renal impairment would experience more bleeding events due to the potential for drug accumulation. The vast majority of the patients within this cohort had a CrCl greater than 30 mL/min; however, this is likely related to prescribing practices as opposed to an implication of risk. Despite this, about one-fourth of patients met the definition for acute kidney injury (AKI) at the time of bleeding event, further supporting the potential association between renal dysfunction and subsequent bleeding. Nearly 40% of patients who bled were receiving concomitant antithrombotic therapy. A prospective registry analysis of patients on DOACs for AF also demonstrated a higher any-bleeding risk in patients on concomitant antiplatelet therapy compared to those without; however, when adjusted for other patient characteristics, the investigators determined that concomitant antiplatelet therapy did not impact bleeding risk independently. 27 Nonetheless, the overall higher bleeding rates render it prudent to evaluate the risks of using multiple agents, and to minimize polypharmacy when appropriate.
Apixaban was the most commonly prescribed DOAC among the patients in our analysis. Although there are no major head-to-head studies comparing the DOACs, landmark trials have demonstrated relatively low bleeding rates with use of apixaban.3–10 Therefore, these data are likely more reflective of prescribing practices and preferential use of apixaban, as opposed to a heightened risk with its use.
Three patients received DOAC doses that deviated from FDA-approved manufacturer labeling and all received lower doses than those which are recommended. Further analysis of these patients can thus provide insight into the characteristics that potentially confer the highest risks. These patients were all elderly females, aged 71, 77, and 95 years. One patient was obese with comorbid hypertension, the second patient was obese with a history of CVA and a prior bleeding event, and the third patient was of normal weight with a history of CVA and a prior bleeding event. Notably, these patient characteristics are consistent with those identified in the aforementioned studies assessing bleeding risk in patients on warfarin and low molecular weight heparin.13–18 All 3 patients were on apixaban therapy, 2 for stroke prevention in AF and 1 for secondary VTE prevention. None of the patients were receiving concomitant antiplatelet therapy.
Although routine monitoring is not required or recommended when using DOACs, the findings of this analysis suggest clinical scenarios in which monitoring serum drug levels may confer clinical benefit. In the absence of established therapeutic ranges, clinicians can utilize available pharmacokinetic studies to compare drug levels in higher-risk patients to previously observed values. While therapeutic monitoring is not established to guide dosage modifications, the availability of this data may allow for cautionary use of DOACs in patients at higher risk of bleeding.
Several limitations of this study exist, including its retrospective nature and the absence of a comparator group. The relatively small patient cohort was extrapolated from a single center, thus potentially limiting generalizability to larger populations. Finally, only patients with bleeds requiring anticoagulation reversal were captured, thus excluding the application of this data to major bleeds not requiring reversal.
Conclusions
Most patients who required reversal for a bleeding event while on DOAC therapy were elderly, overweight or obese, and with comorbid hypertension. Understanding of these potential risk factors may optimize the safe use of these agents. Although DOACs are often considered easier and potentially more effective options to vitamin K antagonists and low molecular weight heparin, these agents still confer risks in certain patient populations that require further investigation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
