Abstract
Background
Atrial fibrillation (AF) is a leading cause of stroke, necessitating effective anticoagulation. While direct oral anticoagulants (DOACs) have improved stroke prevention, bleeding risks remain a concern. Factor XI/XIa inhibitors, targeting the intrinsic coagulation pathway, offer potential for reduced bleeding, although questions remain regarding their efficacy. This systematic review evaluates the efficacy and safety of Factor XI/XIa inhibitors compared to DOACs in AF patients.
Methods
We conducted a systematic review of randomized controlled trials (RCTs) comparing Factor XI/XIa inhibitors with DOACs in AF patients, identified through PubMed and Embase up to January 2025. Data were synthesized narratively due to heterogeneity in study designs and outcomes.
Results
Three RCTs (AZALEA-TIMI 71, OCEANIC-AF, PACIFIC-AF) involving 16 852 patients were included. Factor XI/XIa inhibitors (abelacimab and asundexian) demonstrated significant reductions in bleeding compared to DOACs. In AZALEA-TIMI 71, abelacimab reduced major or clinically relevant non-major bleeding by 62%-69% versus rivaroxaban. In PACIFIC-AF, asundexian reduced bleeding by 50%-84% compared to apixaban. However, OCEANIC-AF showed asundexian was inferior in stroke prevention, with a 3.8-fold higher risk of stroke or systemic embolism compared to apixaban, leading to early trial termination. Abelacimab showed a trend toward higher ischemic stroke rates abelacimab (150 mg: 1.21 vs 0.59 events/100 person-years; and 90 mg: 1.24 vs 0.59 events/100 person-years), though not statistically significant.
Conclusion
Factor XI/XIa inhibitors significantly reduce bleeding risk in AF patients compared to DOACs, but their thrombotic efficacy remains uncertain. While promising, further research is needed to optimize their use.
Keywords
Introduction
Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia, affecting over 33 million people worldwide and posing a significant risk of stroke and systemic embolism due to the formation of atrial thrombi.1,2 The management of AF has evolved significantly with the introduction of direct oral anticoagulants (DOACs),3–5 which have largely replaced vitamin K antagonists (VKAs) due to their improved safety and ease of use. DOACs, including apixaban, rivaroxaban, dabigatran, and edoxaban, have been shown to reduce the risk of stroke and systemic embolism in patients with AF while minimizing the risk of major bleeding compared to VKAs. However, despite these advancements, bleeding remains a significant concern, limiting the use of anticoagulation therapy in many patients, particularly those at high risk of bleeding or with a history of bleeding events.
The persistent challenge of balancing thromboprophylaxis with bleeding risk has driven the exploration of novel anticoagulant targets. Factor XI (FXI) and its activated form-Factor XIa (FXIa)-have emerged as promising targets for anticoagulation therapy.6–9 Unlike traditional anticoagulants that target the extrinsic or common coagulation pathways, FXIa inhibitors aim to disrupt the intrinsic pathway, which plays a critical role in pathological thrombosis but has a limited role in physiological hemostasis. This selective inhibition of FXIa offers the potential for effective thromboprophylaxis with a reduced risk of bleeding, addressing a significant unmet need in the management of AF.
Recent clinical trials10–12 have evaluated the efficacy and safety of Factor XI/XIa inhibitors (AZALEA–TIMI 71[abelacimab: 90 mg or 150 mg monthly], 10 OCEANIC-AF[asundexian: 50 mg daily], 11 and PACIFIC-AF[asundexian: 20 mg or 50 mg daily] 12 ) in comparison to established DOACs in patients with AF. However, their results have been mixed, with some studies demonstrating a lower incidence of bleeding but others raising concerns about the efficacy of Factor XI/XIa inhibitors in preventing thromboembolic events. This systematic review aims to synthesize the available evidence from clinical trials comparing Factor XI/XIa inhibitors with DOACs in patients with AF. We acknowledge that previous systematic reviews7,8 have evaluated Factor XI/XIa inhibitors in various clinical contexts. In contrast, our current review uniquely focuses on AF, synthesizing the latest evidence from three pivotal AF-specific trials (AZALEA-TIMI 71, OCEANIC-AF, and PACIFIC-AF), providing critical data on efficacy-safety trade-offs specific to this patient population.
Methodology
Literature Search
A thorough search was performed in the PubMed and Embase databases up to January 2025 to identify relevant studies. The search strategy included terms related to: (1) Atrial fibrillation, (2) Factor XI inhibitors or Factor XIa inhibitors, (3) Direct oral anticoagulants or non-vitamin K antagonist oral anticoagulants, including apixaban, rivaroxaban, edoxaban, and dabigatran, and (4) Randomized controlled trials. These terms were combined using the Boolean operator “AND.” A detailed search strategy can be found in Supplemental Table 1. Additionally, reference lists from the selected studies were manually reviewed to identify any additional relevant research that may have been missed during the database search. No language restrictions were applied.
Eligibility Criteria
Inclusion criteria for the studies followed the PICOS framework: (1) Population: Patients diagnosed with AF. (2) Intervention: Administration of Factor XI/XIa inhibitors. (3) Comparison: DOACs (eg, apixaban, rivaroxaban, edoxaban, dabigatran). (4) Outcomes: Effacy and safety outcomes were assessed according to the original study definitions. (5) Study design: Only randomized controlled trials (RCTs) were included.
Exclusion criteria comprised animal studies, case reports, reviews, meta-analyses, editorials, conference abstracts, and observational studies. Additionally, single-arm trials focusing solely on Factor XI/XIa inhibitors without a comparator group were excluded. In the case of duplicate studies, preference was given to the study with the longer follow-up or larger sample size.
Study Selection
Two independent investigators screened titles and abstracts of the identified studies according to the pre-defined exclusion criteria. Full-text review was conducted for studies that met the eligibility criteria. Discrepancies between the two reviewers were resolved through consensus or by involving a third reviewer.
Data Extraction
Data extracted from each study included: (1) Study characteristics: First author, publication year, study design, trial acronym, location, sample size, and follow-up duration; (2) Participant characteristics: Demographic details, CHA2DS2-VASc score, HAS-BLED score, comorbid conditions, and prior treatments; (3) Intervention details: Type and dosage of Factor XI/XIa inhibitors and comparators (DOACs); and (4) Outcomes: Event rates for primary and secondary outcomes.
Risk of Bias Assessment
The quality and potential biases of the included studies were assessed using the Risk of Bias 2 (RoB 2) tool. 13 This tool evaluates five domains: (1) Randomization process, (2) Deviations from the intended interventions, (3) Missing outcome data, (4) Outcome measurement bias, and (5) Bias in the selection of reported results. Each domain was rated as having low risk of bias, some concerns, or high risk of bias.
Data Synthesis
Due to the variability in study designs, patient populations, and outcome measures, along with the limited number of studies, a meta-analysis was not feasible to avoid potentially misleading conclusions.14–16 Therefore, a narrative synthesis was conducted to provide a comprehensive interpretation of the data, highlighting the efficacy and safety profiles of Factor XI/XIa inhibitors compared to DOACs in patients with AF.
Results
Study Selection
The study selection process is summarized in the PRISMA flow diagram (Figure 1). An initial search of the PubMed and Embase databases yielded a total of 23 studies. After removing 5 duplicate publications, the titles and abstracts of the remaining 18 studies were screened. During this phase, 15 studies were excluded due to irrelevance or because they did not meet the inclusion criteria (eg, letter, abstract, or review). Three studies10–12 proceeded to full-text review for detailed evaluation. Following this assessment, all three studies (AZALEA–TIMI 71, 10 OCEANIC-AF, 11 and PACIFIC-AF 12 ) were deemed eligible and included in the narrative synthesis.

The PRISMA Flow Diagram of Study Selection Process.
Baseline Characteristics of Included Studies
Table 1 shows baseline characteristics of three RCTs evaluated Factor XI/XIa inhibitors versus DOACs in patients with AF. The AZALEA-TIMI 71 trial 10 (phase 2b) compared monthly subcutaneous abelacimab (150 mg or 90 mg) against daily oral rivaroxaban in 1287 AF patients. The OCEANIC-AF trial 11 (phase 3) randomized 14 810 patients to oral asundexian (50 mg daily) or apixaban (5 mg or 2.5 mg twice daily). The PACIFIC-AF trial 12 (phase 2) tested asundexian (20 mg or 50 mg daily) against apixaban (5 mg or 2.5 mg twice daily) in 755 patients. Efficacy and safety outcomes between Factor XI/XIa Inhibitors versus DOACs are presented in Table 2.
Baseline Characteristics of Included Studies.
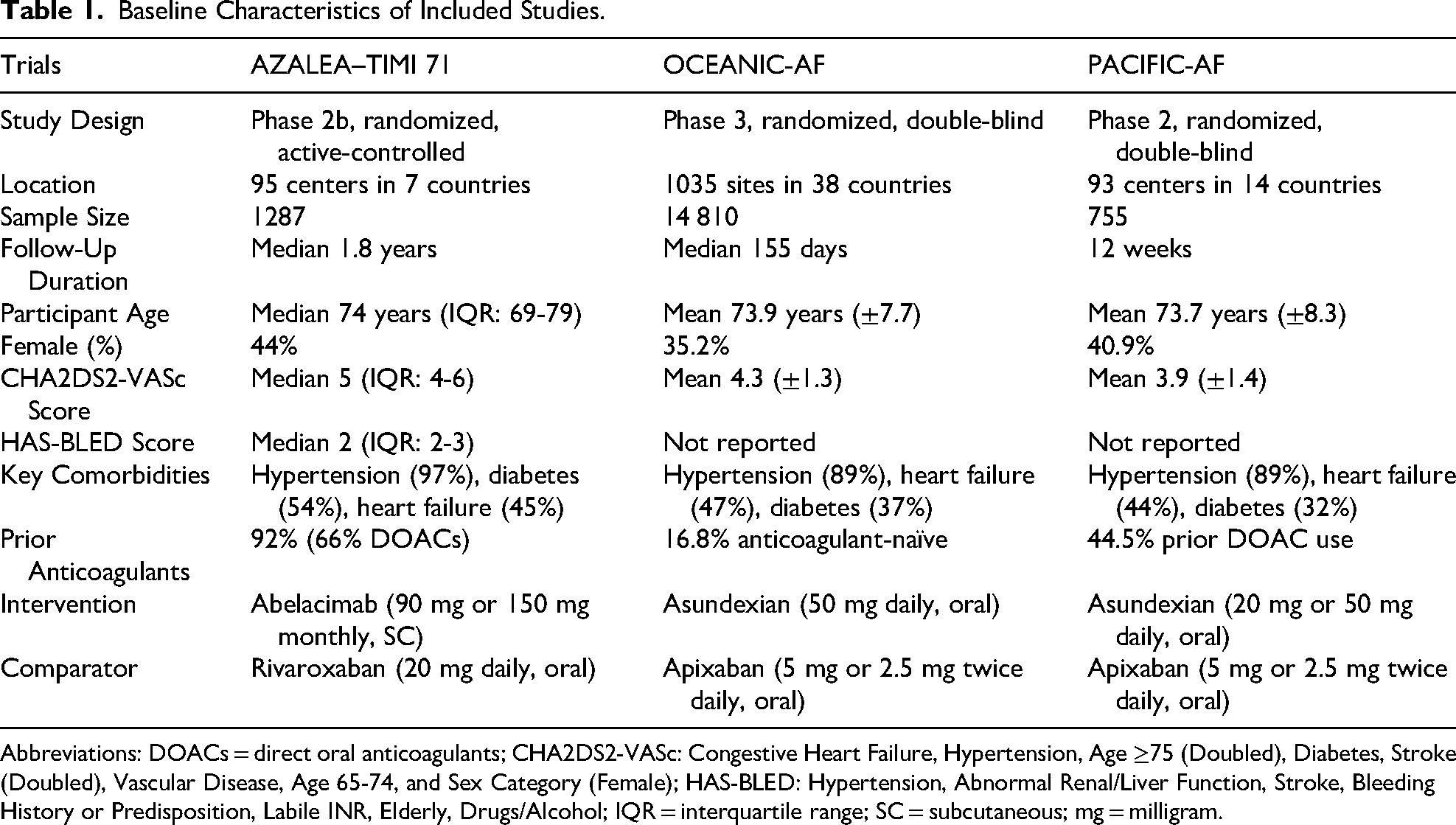
Abbreviations: DOACs = direct oral anticoagulants; CHA2DS2-VASc: Congestive Heart Failure, Hypertension, Age ≥75 (Doubled), Diabetes, Stroke (Doubled), Vascular Disease, Age 65-74, and Sex Category (Female); HAS-BLED: Hypertension, Abnormal Renal/Liver Function, Stroke, Bleeding History or Predisposition, Labile INR, Elderly, Drugs/Alcohol; IQR = interquartile range; SC = subcutaneous; mg = milligram.
Efficacy and Safety Endpoints from the Three Included Studies.

Abbreviations: HR = hazard ratio; CI = confidence interval.
Risk of Bias Assessment
The methodological quality and potential biases of the three included RCTs were assessed using the RoB 2 tool. All three studies demonstrated a low risk of bias across all domains (Supplemental Table 2). The randomization processes were well-described and balanced, interventions were implemented as intended with appropriate blinding, missing outcome data were minimal, outcome measurements were conducted independently and objectively, and there was no evidence of selective reporting. Overall, these studies exhibit high methodological quality, and their results are considered reliable.
Effacy and Safety Outcomes between Factor XI/XIa Inhibitors Versus DOACs
Asundexian Trials
The phase 2 dose-finding study of PACIFIC-AF evaluated asundexian (20 mg or 50 mg daily) against apixaban in 755 AF patients over 12 weeks. Asundexian achieved near-complete factor XIa inhibition (92-94% reduction at 50 mg) and reduced bleeding events. For major or clinically relevant non-major bleeding, the hazard ratio (HR) for asundexian compared to apixaban was 0.33 (95% CI: 0.09-0.97). For any bleeding event, the HR for asundexian versus apixaban was 0.42 (95% CI: 0.26-0.67). No major bleeding events were observed with asundexian. Thrombotic events were rare and balanced across groups (9 total events). Piccini et al concluded that asundexian's bleeding risk was lower than apixaban, supporting further investigation in phase 3 trials. However, the short follow-up and small sample size limited definitive conclusions about thrombotic efficacy.
In the phase 3 trial of OCEANIC-AF comparing asundexian (50 mg daily) with apixaban in 14 810 high-risk AF patients, asundexian demonstrated inferior efficacy for stroke prevention but superior safety. Over a median follow-up of 155 days, the incidence of stroke or systemic embolism was significantly higher with asundexian (1.3% vs 0.4%; HR: 3.79, 95% CI: 2.46-5.83). Conversely, asundexian reduced major bleeding events by 68% (0.2% vs 0.7%; HR: 0.32, 95% CI: 0.18-0.55) and clinically relevant nonmajor bleeding by 52% (0.9% vs 1.9%; HR: 0.48, 95% CI: 0.36-0.64). The trial was terminated early due to the clear efficacy disadvantage of asundexian. Piccini et al concluded that while asundexian exhibited a favorable bleeding profile, its insufficient stroke prevention efficacy at the tested dose precludes clinical use in AF, highlighting the need for dose optimization or alternative mechanisms for factor XIa inhibition.
Abelacimab Trial
In the phase 2b trial of AZALEA–TIMI 71 comparing subcutaneous abelacimab (90 mg or 150 mg monthly) with rivaroxaban in 1287 AF patients, abelacimab significantly reduced bleeding risk. Over a median follow-up of 1.8 years, the incidence of major or clinically relevant nonmajor bleeding was 62%-69% lower with abelacimab (150 mg: 3.22 vs 8.38 events/100 person-years, HR: 0.38; and 90 mg: 2.64 vs 8.38 events/100 person-years, HR: 0.31; both P < .001). Major gastrointestinal bleeding was reduced by 89% (0.5% vs 4.2%). However, ischemic stroke rates were 2-fold higher with abelacimab (150 mg: 1.21 vs 0.59 events/100 person-years; and 90 mg: 1.24 vs 0.59 events/100 person-years), though underpowered for statistical significance. Adverse event rates were similar across groups. The trial was stopped early due to abelacimab's marked bleeding benefit. Ruff et al concluded that abelacimab's favorable safety profile warrants further evaluation in phase 3 trials, particularly in high-bleeding-risk populations, while emphasizing the need for larger studies to confirm its efficacy in stroke prevention.
Discussion
This systematic review synthesizes evidence from three pivotal RCTs (AZALEA–TIMI 71, OCEANIC-AF, and PACIFIC-AF) evaluating the efficacy and safety of factor XI/XIa inhibitors—abelacimab and asundexian—compared to DOACs in patients with AF. The findings illuminate both the promise and challenges of targeting factor XI/XIa, highlighting critical trade-offs between bleeding risk reduction and thrombotic efficacy, while raising mechanistic questions about optimal therapeutic strategies for stroke prevention in AF.
The management of AF has evolved significantly with the introduction of DOACs, which are administered orally, typically once or twice daily (eg, apixaban, rivaroxaban). These agents offer predictable pharmacokinetics without routine monitoring. In contrast, Factor XI/XIa inhibitors represent a novel therapeutic class with diverse administration routes. For example, abelacimab, a monoclonal antibody, is administered subcutaneously at monthly intervals, while asundexian, a small-molecule inhibitor, is taken orally once daily. These differences in dosing frequency and route may influence patient adherence and practical applicability, particularly in high-bleeding-risk populations or those with renal impairment.
The three trials consistently demonstrated that factor XI/XIa inhibitors significantly reduce bleeding risk compared to DOACs, validating the hypothesis that attenuating the intrinsic coagulation pathway spares hemostasis. However, this bleeding benefit came at the expense of thrombotic efficacy. The phase 3 OCEANIC-AF trial demonstrated that asundexian, at a dose of 50 mg once daily, was associated with a 3.8-fold higher risk of stroke or systemic embolism compared to apixaban, leading to early trial termination. This result suggests inadequate thromboprotection with the selected dose of asundexian, highlighting a limitation in its efficacy for stroke prevention in AF. While abelacimab in AZALEA–TIMI 71 did not show statistically significant inferiority in stroke prevention, ischemic stroke rates were 2-fold higher with abelacimab, raising concerns about whether partial factor XI suppression suffices for thromboembolic protection. These divergent outcomes underscore the delicate balance required to uncouple thrombosis from hemostasis and suggest that the degree and mechanism of factor XI/XIa inhibition may critically influence clinical efficacy.
The divergent efficacy outcomes between asundexian and abelacimab may stem from differences in their pharmacodynamic profiles and dosing strategies. While asundexian 50 mg in PACIFIC-AF achieved 92%-94% FXIa inhibition at trough, residual FXIa activity (8-10%) may permit pathological thrombosis in AF, where tissue factor-driven thrombin generation predominates. Conversely, abelacimab's dual inhibition of FXI (zymogen) and FXIa (active enzyme) resulted in sustained >97% FXI suppression, potentially disrupting both intrinsic and extrinsic pathways. The phase 3 OCEANIC-AF results underscore that partial FXIa inhibition at the tested dose inadequately balances thromboprophylaxis and hemostasis. Dose optimization—guided by pharmacokinetic data (eg, targeting trough FXI activity <5%) or alternative mechanisms (eg, combined FXI/XIa inhibition)—warrants investigation. Ongoing trials, such as LIBREXIA-AF evaluating higher-dose milvexian, may clarify whether deeper FXIa suppression or upstream targeting improves efficacy without compromising safety.
The tissue factor-driven extrinsic pathway, central to atrial thrombus formation in AF, 17 may limit the utility of FXIa-selective inhibitors like asundexian. This limitation arises because the extrinsic pathway, initiated by tissue factor (TF)-factor VIIa complexes, directly activates coagulation independently of factor XI by generating thrombin through factor X activation. Since FXIa-selective inhibitors primarily target the intrinsic pathway—where factor XI amplifies thrombin generation—they do not directly interfere with the extrinsic pathway's ability to initiate thrombin production. In AF, persistent endothelial damage and atrial stasis expose TF, driving continuous thrombin generation via the extrinsic pathway. Thus, even with FXIa inhibition, the extrinsic pathway can still produce a significant thrombin “burst” in the left atrium, sustaining thrombus formation. In contrast, abelacimab's dual inhibition of both factor XI (zymogen) and FXIa (active enzyme) may more comprehensively attenuate coagulation by targeting both the initiation phase (reducing TF-FVIIa-driven thrombin) and the amplification phase (blocking FXIa-mediated thrombin amplification). 18 This distinction is critical: selective FXIa inhibitors leave the extrinsic pathway's thrombin-generating capacity intact, whereas dual inhibition disrupts both pathways, addressing the multifactorial pathophysiology of AF-related thrombosis. This mechanistic nuance highlights the need for tailored therapeutic strategies based on the dominant coagulation drivers in specific thrombotic disorders.
Despite asundexian's failure in OCEANIC-AF, the marked bleeding reductions observed across all trials position factor XI/XIa inhibitors as a compelling option for high-bleeding-risk populations, such as patients with prior gastrointestinal bleeding, advanced chronic kidney disease, or frailty. For example, in AZALEA–TIMI 71, abelacimab reduced major gastrointestinal bleeding by 89%, a finding with profound implications for patients in whom DOAC-related gastrointestinal toxicity limits long-term anticoagulation. Furthermore, the minimal renal elimination of asundexian (<15%) and abelacimab's infrequent dosing (monthly injections) may offer practical advantages in nonadherent populations or those with fluctuating renal function.
However, the current data preclude routine clinical use. The higher stroke risk observed with asundexian and the uncertain thrombotic efficacy of abelacimab highlight the need for caution in interpreting their potential benefits. Importantly, these factor XI/XIa inhibitors are still investigational; they must complete phase 3 trials and undergo regulatory review before any clinical application can be contemplated. Furthermore, while a biomarker-guided strategy—such as titrating doses to maintain FXI activity below a threshold—has been proposed, this recommendation appears redundant given that these agents already achieve 92%-94% suppression of FXI activity. In essence, the focus should be on rigorously establishing their safety and efficacy through appropriately powered trials, rather than on premature clinical implementation or additional dose adjustments.
The ongoing LILAC-TIMI 76 trial (NCT05712200; https://clinicaltrials.gov/study/NCT05712200) evaluating abelacimab against placebo in AF patients unsuitable for DOACs will provide critical insights into thrombotic efficacy and safety in high-bleeding-risk populations. Additionally, the LIBREXIA-AF trial (NCT05757869) 19 is testing the oral FXIa inhibitor milvexian in 15 000 AF patients, with results expected in 2026 .It is important to emphasize that these agents remain investigational and have yet to undergo the necessary phase 3 studies and regulatory review before any definitive clinical role can be assigned. Therefore, categorizing their future use as “adjunctive” is premature; their potential to serve as a safer alternative to DOACs will only be determined once rigorous trials establish both efficacy and safety.
Limitations
This systematic review has several limitations. First, the phase 2 designs of PACIFIC-AF and AZALEA–TIMI 71 lacked statistical power to definitively assess thrombotic outcomes, while the OCEANIC-AF trial's results are rendered unreliable due to its premature termination. Second, the homogeneity of enrolled populations—predominantly White, with high baseline thromboembolic risk (median CHA2DS2-VASc 4-5)—limits generalizability to racially diverse cohorts or patients with lower stroke risk. Third, the trials did not explore combination therapies, such as factor XI/XIa inhibitors paired with antiplatelet agents, which might enhance efficacy in patients with concomitant atherosclerosis.
Conclusion
Factor XI/XIa inhibitors represent a potential paradigm shift in anticoagulation, offering unprecedented reductions in bleeding risk compared to DOACs. However, their inconsistent thrombotic efficacy—particularly the failure of asundexian in OCEANIC-AF—underscores the complexity of targeting the intrinsic pathway in AF-related stroke prevention. While abelacimab's promising safety profile justifies further investigation, clinicians must weigh the bleeding benefits against uncertain thrombotic protection. Until phase 3 data confirm noninferior efficacy, DOACs remain the cornerstone of AF management,1,20,21 with factor XI/XIa inhibitors 22 reserved for clinical trials or highly selected high-bleeding-risk patients.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251335967 - Supplemental material for A Systematic Review of Factor XI/XIa Inhibitors Versus Direct Oral Anticoagulants in Patients with Atrial Fibrillation
Supplemental material, sj-docx-1-cat-10.1177_10760296251335967 for A Systematic Review of Factor XI/XIa Inhibitors Versus Direct Oral Anticoagulants in Patients with Atrial Fibrillation by Zhengbiao Xue, Song Liao, Haiye Fan, Yu Shen and Zhi Nie in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contributions/CRediT
Conceptualization: Zhi Nie and Yu Shen. Methodology: Zhi Nie and Haiye Fan. Formal analysis: Yu Shen and Haiye Fan. Data curation: Yu Shen and Haiye Fan. Writing – original draft preparation: Zhengbiao Xue. Supervision: Zhi Nie and Song Liao. All authors contributed to the writing – review and editing and have read and agreed to the present version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received Ganzhou City Guiding Science and Technology Plan Project (GZ2024ZSF155 and GZ2024YLJ138).
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All relevant data and materials are presented in the paper.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
